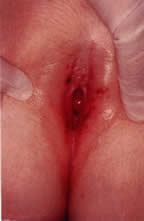
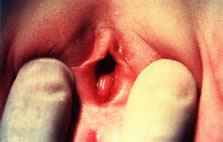
Pathophysiology and Clinical Presentation Vulvitis is defined as inflammation of the labia majora, labia minora, clitoris, and
introitus; vaginitis is inflammation of the vaginal mucosa. Pediatric
vulvovaginitis, involving the vulvar and vaginal tissues, is
a very common diagnosis made by the primary care provider, who often
refers the patient to a specialist when initial treatment is unsuccessful. Thus, it
is important to understand the pathophysiology, know
the various etiologies as they relate to the clinical presentation, and
establish a methodologic approach to the evaluation of vulvovaginitis. Because of anatomic and behavioral factors, the prepubertal girl is at
increased risk for vulvovaginitis. First, with the absence of pubic hair
and labial fat pads, the vaginal vestibule and vulva are less protected
from external irritants, especially when squatting or sitting. Second, the
skin of the vulva and vaginal mucosa is thinner, more sensitive, and
thus more easily irritated by trauma as well as chemical, environmental, and
allergic exposures. Third, the vaginal cavity has a neutral
pH (6.5 to 7.5),20 is warm and moist, and has unestrogenized epithelium that lacks both lactobacilli
and glycogen. All of these factors facilitate bacterial growth. Lastly, prepubertal
children tend to have poor hygiene in terms
of perineal cleansing and hand washing; this can lead to autoinoculation
with fecal bacteria or less commonly from organisms associated with
an infected urinary or respiratory tract.5,21,22 Pediatric vulvovaginitis typically presents as vaginal itching with associated
excoriation, vaginal discharge that may be malodorous, generalized
vulvar, vaginal, or perianal discomfort, or pain or dysuria.23,24 Parents may also report staining, odor, or color on the child's underwear. The
history is very important in narrowing the etiology and
directing treatment. Parents should be asked about the onset, timing, and
duration of symptoms, previous home therapies and medications used (including
prescription and over-the-counter oral and topical therapies), and
prior laboratory tests or evaluative procedures. The possibility
of sexual abuse should be assessed, along with a detailed review of
the developmental, behavioral, and psychosocial history. The child's
past medical or surgical history should be evaluated for other skin
infections, dermatoses, or allergies. Family history of chronic illness, allergies, and
contact sensitivities should also be assessed. A
list of possible acute or chronic irritant exposures such as bubble baths, cleaning
agents and techniques, lotions, powders, fabric softeners, and
hair products should be investigated.25 The child should have a complete physical examination documenting pubertal
stage as well as evidence of chronic disease or other skin abnormalities. With
the patient in the frog-leg or knee-chest position, the perineum
and vulva can be examined for the presence of erythema, discharge, odor, and
edema. An otoscope or colposcope can aid the examiner by
providing focused light and magnification. Vaginal discharge is not
present in most circumstances, but when present it can vary from copious
to minimally dried secretions. It is often helpful to examine the vaginal discharge, but obtaining the
specimen from a child can be challenging. Saline instilled into the vagina
can be “recollected” as it accumulates in the lower
vagina and vestibule. The discharge can also be collected directly with
a dry or saline-moistened bacteriostatic swab, being very careful not
to touch the sensitive hymenal tissue.26,27 Having the child perform a Valsalva maneuver (e.g., cough) can aid in
this procedure. Topical anesthetics should be used with caution because
of initial burning; this may upset the child, prohibiting further examination. Once
the sample is collected, it should be examined with saline
wet mount inspection, potassium hydroxide (KOH), Gram stain, vaginal
pH, and cultures. Based on the history, physical examination, and laboratory evaluation, the
causes of pediatric vulvovaginitis are most easily classified into
noninfectious (or nonspecific) and infectious (or specific) groups, with
the latter subclassified into nonsexually and sexually transmitted
infections5,21 (Table 3). Table 3. Prepubertal Vulvovaginitis
Noninfectious Chemical irritants (e.g., bubble baths, perfumes, soaps, and hair products)
Allergic contact
Poor hygiene
Poor perineal aeration
Foreign body
Urologic anatomic abnormalities
Infectious Nonsexually transmitted Bacteria: group A beta-hemolytic streptococcus, Haemophilus influenzae, Staphylococcus aureus, Streptococcus pneumoniae, Streptococcus viridans, Shigella sonnei
Viral: adenovirus, varicella zoster, echovirus, human immunodeficiency
virus
Fungal: Candida albicans
Helminths: Enterobius vermicularis
Sexually transmitted Neisseria gonorrhoeae, Chlamydia trachomatis, Trichomonas, Mycoplasma hominis, Ureaplasma urealyticum, herpesvirus (1 and 2), human papilloma virus, human immunodeficiency
virus, molluscum contagiosum, Treponema pallidum
Noninfectious Vulvovaginitis One of the most common causes of noninfectious vulvovaginitis, usually
referred to as nonspecific vulvovaginitis, is poor perineal hygiene. On
examination, patients usually have mild, nonspecific vulvar inflammation
and may have stool or pieces of toilet paper on the perineal tissue
as well as soiled underwear. Treatment consists mainly of hygiene education
and supportive care. The clinician should recommend that the
child have sufficient opportunities to urinate, uses a front-to-back wiping
technique with soft, white, unscented toilet paper, and washes her
hands regularly, especially after bathroom use. Undergarments should
be 100% cotton, loose fitting, and cleaned or rinsed thoroughly with
mild hypoallergenic unscented detergent without fabric softener. Mild
hypoallergenic perfume-free and dye-free cleansers used to wash the perineal
area gently leave the skin more moisturized than regular soap. Cleaning
agents should never be applied with a washcloth, which can exacerbate
areas of irritation or transfer infectious organisms to that
area. After bathing, the perineal area should be air-dried or patted
dry with a towel, avoiding rubbing. In most patients, acute nonspecific vulvovaginitis of any cause can be
treated symptomatically with frequent sitz baths containing baking soda
or colloidal oatmeal or with wet compresses of Burrow's solution. For
extremely severe cases where other etiologies have been excluded, a 1% hydrocortisone
cream can be used once or twice a day for up to 2 weeks
for itching, or a 1–2-week course of estrogen cream can
be used to facilitate healing. Another common cause of vulvovaginitis is excessive or prolonged exposure
to moisture combined with poor aeration of the perineal tissues. Predisposing
factors to this form of vulvovaginitis include obesity, wearing
tight or synthetic undergarments, and exposure to long periods of
wet undergarments (e.g., enuresis, swimsuits). Physical examination may
show nonspecific inflammation to severe excoriation, which may be associated
with secondary bacterial infections, most commonly due to Staphylococcus aureus. Sitz baths and proper techniques of perineal cleansing and drying should
be reviewed. Patients should wear loose-fitting cotton undergarments
and might find sleeping without undergarments more comfortable. Allergic vulvovaginitis or contact dermatitis may present with pruritus
as the most prominent symptom. Acute or chronic offending agents are
usually topical creams, lotions, perfumed soaps, toilet paper, and poison
ivy. With chronic exposure, the vulva may develop cracks or fissures
and eventually a lichenified appearance. Eosinophils in the vaginal
fluid may be found in cases resulting from allergic reactions.28 Chemical irritants from bubble baths, laundry detergents, soaps, and fabric
softeners produce a similar clinical picture. Treatment consists
of removal of the offending agent, hygiene education, sitz baths, and
a 1% hydrocortisone cream.21 Vaginal foreign bodies can also present as nonspecific vulvovaginitis in
the prepubertal child. Possible symptoms include profuse, persistent, foul-smelling
discharge that may be blood stained (Table 4). Table 4. Bloody Genital Discharge
Infectious vulvovaginitis: group A beta-hemolytic streptococcus, Haemophilus influenzae, Shigella sonnei, Shigella flexneri
Foreign body
Sexual abuse
Trauma
Urethral prolapse
Exogenous hormone exposure
Lichen sclerosus
Tumor
Because superinfection is common, antibiotics may provide temporary relief, followed
by the recurrence of symptoms. Thus, recurrent symptoms
requiring repeated trials of medication accompanied by the above history
might warrant vaginoscopy to rule out a foreign body. Although foreign
objects made of firm material may be palpable on rectal examination, a
child may not allow this procedure to be completed. In addition, because
foreign bodies may be multiple or fragmented, irrigation is usually
needed to loosen and remove all debris. Objects can also become
embedded and encased by granulation tissue, which may cause erosion or
perforation into the bladder or bowel tissue. Thus, to evaluate a child
fully for the presence of a vaginal foreign object, an examination
under anesthesia may be necessary. It is important as always with the
diagnosis of vulvovaginitis, but especially with foreign bodies, to keep
a high index of suspicion for sexual abuse. Removal of the offending
agent, most commonly toilet paper, is curative. For developmental or
behavioral reasons, some patients may find that using bulkier items (e.g., disposable
wipes) to clean themselves is helpful in preventing recurrences. Anatomic disorders such as ectopic ureter, urethral prolapse, and fistulas
are rare causes that can present as vulvovaginitis. Instead of opening
into the trigone of the bladder, an ectopic ureter opens elsewhere, usually
along the urethra. It can also open into the vagina or in the
area of the vaginal vestibule, where an additional meatal opening around
the urethral meatus is seen. It is highly associated with other
congenital anomalies such as dilatation and duplicated systems. Patients
typically present with a history of incontinence and a constantly wet
perineum. With the contralateral ureter being normal, these children
can accumulate urine in the bladder and thus have normal voiding habits. Although
the diagnosis can be made prenatally, these patients may
not be diagnosed until adulthood because they can be easily misdiagnosed
as having primary enuresis or stress incontinence. A voiding cystourethrogram
confirms the diagnosis, ultrasound can identify any associated
müllerian structural anomalies, and a renal scan will detect
functioning. Surgical correction is necessary for this condition.21,29 Patients with vesicovaginal fistulas also usually have a history of a constantly
wet perineum and show nonspecific vulvovaginitis and excoriation
on physical examination as a result of the continuous presence of
urine. Presence of feculent vaginal discharge suggests a rectovaginal
fistula. Patients with urethral prolapse usually present with blood staining
on their underwear. Other symptoms include frank vaginal bleeding
with accompanying vulvar pain or dysuria. This problem is often precipitated
by activities that increase intra-abdominal pressure (e.g., coughing, straining
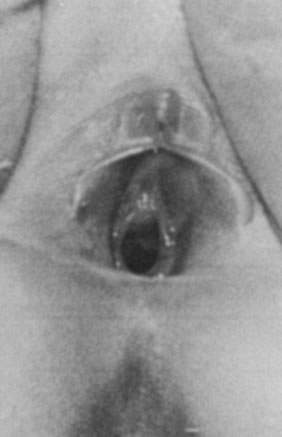
with bowel movements, crying). Examination typically
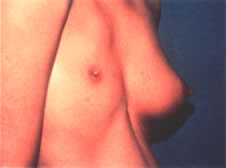
reveals an everted, red, circular mass at the external urethral meatus (Fig. 10). It is most common in African-American children younger than 10 years
of age.5,21,29 Hypothesized factors that place young children at increased risk include
the combination of weak attachments of the different layers of urethral
muscle and episodes of increased intra-abdominal pressure.30 Medical treatment of urethral prolapse includes sitz baths, or estrogen
cream two or three times a day for 2 to 4 weeks. Surgical excision is
considered when it does not resolve or continues to recur. 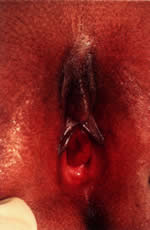 Fig. 10. Urethral prolapse with periurethral adhesions. Fig. 10. Urethral prolapse with periurethral adhesions.
|
Infectious Vulvovaginitis, Nonsexually Transmitted Vaginal discharge is a more prominent finding associated with infectious
causes of vulvovaginitis than with noninfectious causes. It is best
to make the diagnosis of infection based on culture rather than treating
empirically so that appropriate antibiotics can be prescribed. Also, the
recovery of certain organisms may prompt a sexual abuse evaluation
that may not have been performed otherwise. There are limited data
about the normal vaginal flora in the prepubertal child. Organisms cultured
from prepubertal asymptomatic “control” subjects have
included Bacteroides species, lactobacilli, shape Staphylococcus epidermidis, and other enteric organisms.31,32 Although not well studied, antibiotic treatment may be warranted when
these organisms are found in cultures of patients with symptoms that do
not resolve with supportive care. The poor hygiene habits of the young child commonly promote autoinoculation
of respiratory, gastrointestinal, or urinary pathogens, while the
unprotected, unestrogenized prepubertal vaginal tissues support their
growth. Bacterial respiratory pathogens, such as group A beta-hemolytic
streptococci (GAS), Streptococcus pneumoniae, and Hae-mophilus influenzae can cause a purulent vaginal discharge with an associated vulvovaginitis
with or without other symptoms.33 GAS in particular can have a dramatic appearance, with severe vulvovaginal
erythema, edema, and discharge; it may be or not be associated with
a concurrent scarlet fever and a positive throat culture. It is also
associated with desquamation that can take place a few weeks later. Bacterial
cultures of the perineum can confirm the diagnosis.26,33,34 GAS vulvovaginitis can be treated with amoxicillin 40 mg/kg divided three
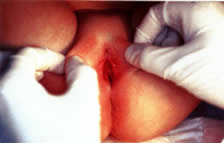
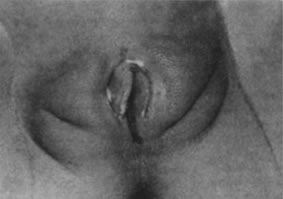
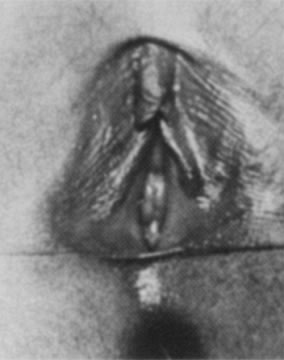
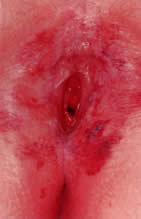
times a day for 10 days. Viral pathogens usually present as ulcerative lesions in the vaginal area (Fig. 11). Offending viral pathogens include adenovirus, varicella, echovirus, Epstein-Barr
virus, and herpesvirus 1 and 2. A special viral culture medium
is necessary for a definitive diagnosis. Depending on the severity, presumptive
treatment with acyclovir may be indicated while awaiting
cultures. Gastrointestinal pathogens such as Shigella can produce an
acute or chronic vaginal discharge that is bloody, purulent, and foul-smelling, with
associated vulvovaginal erythema. It is sometimes associated
with diarrhea in the patient or family members. Culture of the
vaginal discharge is diagnostic, and sensitivities should be obtained
so that appropriate systemic antibiotics can be administered.35,36 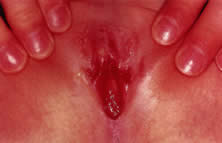 Fig. 11. Ulcerative lesions on vaginal mucosa. Fig. 11. Ulcerative lesions on vaginal mucosa.
|
Although intestinal infestation with Enterobius vermicularis (pinworms) usually causes perianal itching, vulvovaginitis has also been
reported. Because of close proximity, the pinworms may crawl into the
vagina (or be transferred by scratching), bringing eggs and attached
enteric organisms. Physical examination may show vaginal discharge, nonspecific
inflammation, and excoriation from scratching. The diagnosis
is confirmed by observation of pinworm ova and/or adults with a saline
wet mount or with Scotch tape testing, which is best done when the
patient is asleep during the night, when the worms emerge to feed. Patients
are treated with mebendazole; empiric treatment of the entire family
is given to avoid reinfection. Candidal vulvovaginitis, although very common after puberty, is extremely
rare in healthy prepubertal children and is often overdiagnosed and
overtreated.24 Predisposing factors include recent antibiotic use, poor perineal aeration, inflammatory
skin conditions such as seborrheic dermatitis, and
chronic diseases such as diabetes mellitus and immunodeficiency syndromes. Pruritus
and dysuria are the most common complaints, along with the
presence of diffuse vulvar erythema, thick cheesy vaginal discharge, excoriation
from scratching, and the presence of white plaques on the
vaginal mucosa. “Satellite lesions” and erythematous prominence
in the creases are characteristic of candidal rashes, especially
in those who wear diapers. A patient with a candidal infection will
have a low vaginal pH, and budding yeast and pseudohyphae can be seen
with a saline wet mount and KOH preparation. Treatment consists of topical
or oral antifungal agents such as fluconazole; the area should be
kept as dry as possible. Persistent candidal infections, especially
in the prepubertal child, should prompt investigation for the presence
of diabetes or HIV or other immunodeficiency syndromes.5,21 Infectious Vulvovaginitis, Sexually Transmitted Organisms associated with sexual transmission that are found in prepubertal
girls require an evaluation for sexual abuse. Practitioners should
always have a high index of suspicion and incorporate questions about
sexual abuse into the routine history. They should also know their local
mandated reporting laws and should perform a thorough physical examination
as well as an extensive psychosocial history. When sexual abuse
is discovered, referral to a child abuse specialist in the community
should be made to ensure proper and complete care, especially because
the collection of evidence may vary from the office guidelines. Acute
injury and infection require immediate attention and possible referral
to an emergency department. Cultures should always be obtained for
gonorrhea and chlamydia before treatment. Antibiotic choices and doses
vary with age, weight, and pregnancy and are outlined in the “American
Academy of Pediatrics Red Book.”37 The lack of estrogenized epithelium in this age group inhibits sexually
transmitted organisms from extending upward into the pelvis. Thus, symptoms
typically include “lower tract” symptoms of pruritus, dysuria, vaginal
discharge, and odor. Physical findings consist of
vulvovaginitis, external lesions, and vaginal discharge. Genital infection
with Neisseria gonorrhoeae is essentially pathognomonic for sexual abuse. It is associated with a
purulent thick yellow discharge along with vulvar erythema, edema, and
excoriation and inguinal lymphadenopathy. Diagnosis is confirmed with
a culture and the patient is treated with cefixime, ceftriaxone, or
ciprofloxacin. Chlamydia trachomatis can infect the atrophic vaginal squamous cells of a prepubertal child, causing
vulvovaginitis with pruritus and a vaginal discharge. The presence
of C. trachomatis is also indicative of sexual abuse, but it can also be acquired perinatally. Infants
born to mothers with chlamydia have been shown to have
asymptomatic carriage for up to 18 months.38 Culture of the vaginal discharge confirms the diagnosis, and the patient
should be treated with azithromycin or doxycycline. Trichomonas is diagnosed by observing the organisms with a saline wet mount
and can be recognized by the presence of a fishy odor (positive “whiff”) when
potassium hydroxide is added to the sample of
vaginal discharge. It is treated with metronidazole. Bacterial vaginosis is predominantly associated with sexual abuse but has
been reported in “control” subjects.39 It is diagnosed by the presence of a positive “whiff,” a higher-than-normal
pH, and the presence of “clue cells” with
a saline wet mount. Treatment consists of metronidazole. Herpes simplex virus (HSV) produces vesicular lesions with a resultant
ulcerative vulvovaginitis and usually includes inguinal lymphadenopathy
and systemic symptoms. HSV can be treated with acyclovir. Human papilloma virus (HPV) causes condyloma acuminata, or painless, soft, moist, granular, and
friable lesions that predominate in the vaginal
vestibule and perianal area. The lesions can become secondarily infected
and produce pruritus, pain, and discharge. Therapy consists of cryotherapy
or serial applications of TCA. Molluscum contagiosum are waxy, centrally umbilicated lesions 2 to 5 mm
in diameter. Treatment can include imiquimod cream (Aldara; 3M Pharmaceuticals, St. Paul, MN) or curettage. Vulvovaginitis from syphilis is usually due to the manifestation of secondary
syphilis, which includes a rash over the perineum and inner thighs
and development of condyloma lata on the vulva and anus. Serologic
and cerebrospinal fluid testing confirms the diagnosis and establishes
the therapy.21,40 |