To make cervical cancer prevention programs more accessible to women living
in low-resource settings, the screening test and the screening strategy
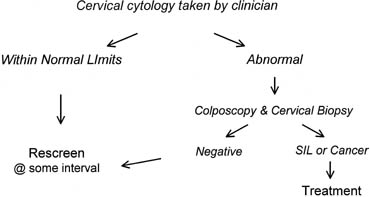
outlined in Figure 6 must be replaced. Two screening methods currently are being considered
as potential alternatives to the Pap test. One is visual screening methods, such
as visual inspection with acetic acid (VIA), which is also
known as direct visual inspection of the cervix. The other is HPV DNA
testing. Neither VIA nor HPV DNA testing has yet been widely accepted
as a replacement for cervical cytology, but large trials that have been
recently completed or are currently under way suggest that both of
these methods have great potential. Screen and Treat Strategies
Simply changing the screening test would not make cervical cancer prevention
programs more accessible to women in low-resource settings because all
of the resources and technologic requirements needed for the systems that
support the test within a prevention program still would be required.
What is needed is a viable alternative that compares favorably in safety
and effectiveness with the screen, diagnose, and treat strategy. It has
been suggested that consolidating screening and diagnosis and eliminating
the requirement for colposcopy and cervical biopsy would reduce greatly
the infrastructure requirements of a cervical cancer prevention program.57,58
Such a “screen and treat” strategy would couple screening
with immediate treatment using simple outpatient ablative methods, such
as cryotherapy or electrofulguration, in all women found to be screen
positive (Fig. 7).
Although the safety and efficacy of screen and treat strategies have not
yet been proved, large-scale clinical trials of this approach are currently
under way, and many cervical cancer screening experts view these strategies
as the only viable option for providing cervical cancer prevention services
in low-resource settings.
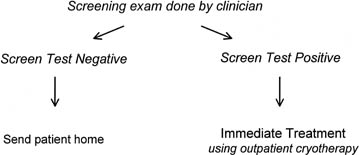 Fig. 7. Screen and treat approach to cervical cancer screening. Human papillomavirus (HPV) testing
or visual inspection with acetic acid (VIA) combined
with immediate ablation. Fig. 7. Screen and treat approach to cervical cancer screening. Human papillomavirus (HPV) testing
or visual inspection with acetic acid (VIA) combined
with immediate ablation.
|
Visual Screening Methods Visual screening methods involve washing the cervix with a chemical contrast
solution, such as a 5% solution of acetic acid or an iodine
solution, and inspecting the cervix either with the naked eye or with
a handheld low-magnification device to determine whether or not a cervical
cancer or precursor lesion is present. This approach is identical
to the Schiller (iodine) test that was introduced in the 1930s.59 The Schiller test consisted of applying an iodine solution to the cervix
and inspecting the cervix with the naked eye to detect nonstaining
areas. The Schiller test initially was well received by clinicians because
it provided the first method that could be used to screen for cervical
cancer precursors. As it became more widely used, however, the low
specificity of the Schiller test was recognized as a significant disadvantage. Only
a small proportion of women who were classified as positive
with the Schiller test had areas of the cervix that did not stain
with iodine because they had a cervical cancer precursor lesion. Most
of these women had areas of cervical ectopy or immature squamous metaplasia
that lacked glycogen and did not stain with iodine.60 Because the only method available for evaluating women with a positive
screening test at that time was a cold knife conization, the test became
unpopular and was largely discontinued after the Pap test, which was
considerably more specific, was introduced in the 1950s.
The reconsideration of visual screening as a method for cervical cancer
screening in the 1990s was due to several factors. One was the realization
that cytologic screening is difficult, if not impossible, to implement
and sustain in most low-resource settings. Another reason to reconsider
visual screening was that even when practiced in developed countries,
cytology has a much poorer performance than previously thought. Meta-analyses
of the performance of cervical cytology found that the sensitivity for
the detection of high-grade SIL (CIN 2, 3) is only 0.49 to 0.67 and that
the specificity of cytology is only 0.62 to 0.77.61,62
In addition, the availability of simple outpatient ablative treatment
methods, such as cryotherapy, that were not available at the time the
Schiller test was introduced in the 1930s and 1940s make low specificity
less of a problem than it was in the 1940s, when the only option for screen-positive
women was to undergo a cone biopsy. Ablative outpatient treatment methods
have minimal complications and serious side effects.63,64,65,66
The most widely tested visual inspection method consists of applying a 5% solution
of acetic acid to the cervix and visualizing the cervix
with the naked eye to determine whether an acetowhite “lesion” is
present (Fig. 8). This approach to visual screening has been referred to by a variety
of terms, including VIA, acetic acid washes, acetic acid visualization, cerviscopy, and
direct visual inspection. In most studies, VIA is performed
by specifically trained midlevel nurses, although in one study
from India the examination was performed by cytotechnicians and in one
study from China it was performed by gynecologic oncologists. 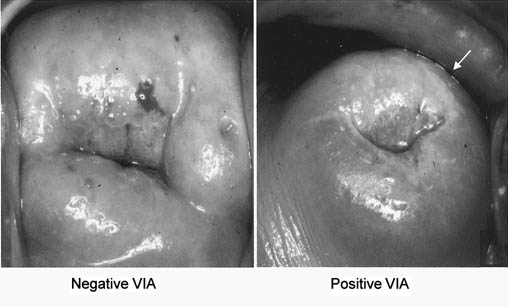 Fig. 8. Visual inspection with acetic acid (VIA). Typical appearance of a negative
screening test (left) and a positive screening test (right). Fig. 8. Visual inspection with acetic acid (VIA). Typical appearance of a negative
screening test (left) and a positive screening test (right).
|
Many large studies have been completed that have evaluated the performance
of VIA when used in the screening setting (Table 10).67,68,69,70,71
VIA has a reported sensitivity of 0.64 to 0.96 for the detection of high-grade
SIL (CIN 2, 3) and invasive cervical cancer and a specificity of 0.65
to 1.0. In studies in which Pap smears72
were obtained and VIA was performed, the sensitivity of the two tests
was comparable, but in most studies the specificity of VIA was significantly
lower. The high sensitivities of VIA that were reported in the studies
by Sankaranarayanan and associates70,71
most likely reflect the fact that relatively few patients who were not
VIA positive underwent colposcopy, and the prevalence of biopsy-confirmed
high-grade SIL (CIN 2, 3) in women who were VIA negative probably was
underestimated to a considerable degree.
TABLE 10. Comparison of Visual Inspection with Acetic Acid and Cervical
Cytology*
| | VIA | Cervical Cytology |
Author | Sensitivity | Specificity | Sensitivity | Specificity |
Megavand et al69 |
0.64 |
0.98 |
0.62 |
0.89 |
|
Sankaranarayanan et al70,71
|
0.90 |
0.92 |
0.92 |
0.90 |
Sankaranarayanan et al109 |
0.96 |
1.00 |
1.00 |
0.63 |
Zimbabwe Project68 |
0.77 |
0.65 |
NA |
NA |
Denny et al67 |
0.67 |
0.83 |
0.75 |
0.90 |
Bellinson et al99 |
0.71 |
0.74 |
0.94 |
0.78 |
VIA, visual inspection with acetic acid; NA, not available.
*For the detection of high-grade squamous intraepithelial lesions (cervical
intraepithelial neoplasia [CIN] 2, 3) and cancer.
VIA test results usually are reported as being positive, negative, or suspicious
for invasive cancer, although some investigators also used a “borderline” or “indeterminant” category. Positive
tests frequently are described as well-defined, dense acetowhite
lesions that are adjacent to the squamocolumnar junction. Negative
tests often are defined as including cervices with faint, ill-defined
areas of acetowhitening and small dotlike areas of acetowhite epithelium.73 In one study, 114 cervical photographs taken after the application of
a 5% acetic acid solution were scored by three clinicans experienced
in colposcopy and VIA.73 The degree of intraobserver agreement was only moderate (pairwise unweighted κ statistic, 0.54 to 0.60). Using the cervical photographs, the
performance of VIA varied considerably between the three observers. Sensitivity
for high-grade SIL (CIN 2, 3) and cancer varied between
the three observers from 0.87 to 0.97. Specificity also varied considerably (0.58 to 0.39). As would be expected, the interpreter with the
most colposcopic experience had the highest sensitivity, but also the
lowest specificity. Our group evaluated different definitions of what constitutes a “positive” VIA
result among 2,754 South African women who were
screened using a combination of HPV DNA testing for high-risk types of
HPV, cervical cytology, cervicography, and VIA, with women with a positive
result on any of the screening tests being referred for colposcopy (44% of
all women screened).74 “Definite lesions” were acetowhite lesions with well-circumscribed
borders; “ill-defined lesions” were lesions that
were poorly circumscribed and faintly acetowhite; and “nonconfluent
scattered lesions” were focal, small, punctated areas
of acetowhitening usually involving the transformation zone. When only
definite lesions were classified as a positive result, the sensitivity
of VIA for high-grade SIL (CIN 2, 3) was 0.58. The sensitivity significantly
increased to 0.70 when any lesion was classified as a positive
result. The increase in sensitivity observed with expanding the definition
of what constitutes a positive test result was accompanied by a
significant decrease in test specificity from 0.84 to 0.79. For comparison, the
sensitivity of conventional cervical cytology using a cutoff
of low-grade SIL to define a positive test result produced a sensitivity
of 0.57 and a specificity of 0.96. In the same study, we also evaluated the impact of using a handheld 2.5 × magnification
device while performing VIA and the impact of
age, other demographic factors, and sexually transmitted infections on
the performance of VIA. Magnification resulted in a slight but nonsignificant
increase in sensitivity for high-grade SIL (CIN 2, 3) from 0.70 to 0.74 when
the definition of “positive result” was
the presence of any acetowhite lesion. The nonsignificant increase in
sensitivity was accompanied, however, by a slight but significant decrease
in specificity from 0.79 to 0.77. No significant differences in the
sensitivity and specificity of VIA were associated with the presence
or absence of N. gonorrheae, C. trachomatis, or T. vaginalis or age, parity, contraceptive use, or across the duration of the study. VIA
had a significantly lower specificity among HIV-infected women compared
with uninfected women, and there was a nonsignificant trend toward
greater specificity in women older than age 50 years, which became
significant when women were classified as postmenopausal or not.
The previous discussion highlights several key points regarding VIA.
The first is that currently most experts would place the sensitivity of
VIA for biopsy-confirmed high-grade SIL (CIN 2, 3) between 60%
and 80%, which is slightly greater than that reported for cervical
cytology in most large meta-analyses.61,75
The specificity of VIA appears to be lower, however, than that of cytology
or HPV DNA testing (see later) in many settings. If VIA were used to screen,
considerable numbers of women lacking high-grade SIL (CIN 2, 3) or cancer
would be classified as being screening test positive and would require
either further evaluation or treatment. The second point is that the performance
of VIA depends on the definition of a positive test result. When a more
restrictive definition of a positive test result is used, specificity
is significantly increased, but sensitivity seems to be significantly
decreased. The stringent definition should be used under conditions in
which it is important to limit the number of false-positive results, but
a less stringent definition should be used under conditions in which the
key objective is to identify all women with high-grade SIL (CIN 2, 3),
such as when women would have available only one or two opportunities
to undergo screening in their lifetime. Use of a less stringent definition
and not including magnification also would make the training requirements
for VIA simpler and more appropriate for low-resource settings.
Human Papillomavirus DNA Testing TYPES OF HUMAN PAPILLOMAVIRUS. There is now compelling scientific and epidemiologic evidence that specific
high-risk types of HPV cause almost all cases of invasive cervical
cancer. Although a full discussion of the role that HPV plays in the
pathogenesis of cervical cancer is beyond the scope of this chapter, it
is crucial that the nature of the relationship between HPV and cervical
cancer be understood to understand the attractiveness of using HPV
DNA testing as a screening method in low-resource settings. HPV is a
double-stranded DNA virus that is a member of a family of DNA tumor viruses, Papovaviridae. This family also includes polyoma viruses and SV-40 virus. In
humans, 85 types of papillomaviruses have been characterized
and fully sequenced, and more than 120 putative novel types have
been partially characterized.76 Approximately 40 types of HPV can infect the epithelium of the anogenital
tract. Although the different types of HPV are similar structurally, there
are significant differences with respect to the tissue that they
infect and the type of lesions they produce.77
Based on their associations with specific types of lesions, the most
common of the 40 anogenital HPVs have been divided into three oncogenic
risk groups (Table 11). HPV types that usually are
associated with condyloma acuminata of the anogenital tract and occasionally
associated with low-grade SIL but only rarely associated with high-grade
SIL (CIN 2, 3) and almost never associated with invasive squamous cell
carcinomas of the cervix are referred to as low-risk viruses. HPV
types 42, 43, and 44 are included in the low oncogenic risk viruses because
they have a similarly low risk of being associated with cancer. In contrast,
HPV types 16, 18, 45, 56, and 58 are associated most frequently with invasive
carcinomas of the anogenital tract and are referred to as high-risk
viruses.78,79,80,81
Many other HPV types, such as types 31, 33, 35, 39, 51, 52, 59, and 68,
have features of high-risk viruses and can be found in association with
invasive cervical cancers, although less frequently than the typical high
oncogenic risk viruses. These viruses used to be referred to as intermediate-risk
viruses, but because more recent studies indicate that infection with
these intermediate-risk viruses confers similar relative risks for high-grade
SIL (CIN 2, 3) or cancer as does injection with the prototypical high
oncogenic risk viruses, many authorities now classify types 31, 33, 35,
39, 51, 52, 59, and 68 as high-risk viruses.5
TABLE 11. Oncogenic Risk Grouping of Anogenital Human Papillomavirus
Low oncogenic risk | 6, 11, 42, 43, 44, 53 |
High oncogenic risk | 16, 18, 45, 56, 58 |
Other high-risk types | 31, 33, 35, 39, 51, 52, 59, 68 | The only HPV assay that is commercially available (Hybrid Capture II) combines
the 13 most common intermediate-risk and high-risk viruses together
into a single high-risk probe mixture. These are HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, and 68. The commercially available
assay cannot distinguish lesions associated with the prototypical high-risk
types of HPV (e.g., HPV 16, 18, 45, 56, and 58) from those associated
with other high-risk viruses. EVIDENCE LINKING HUMAN PAPILLOMAVIRUS INFECTION WITH CERVICAL CANCER.
There is consistent and compelling epidemiologic evidence that HPV infection
plays a central role in the development of high-grade cervical cancer
precursors (CIN 2, 3) and invasive cervical cancers. Based on this evidence,
it is accepted that infection with a high-risk type of HPV is a major
risk factor for the development of cervical cancer. Epidemiologic studies
have shown that the temporal sequence between infection and subsequent
formation of cancer is correct; associations between infection with high-risk
types of HPV and cervical cancer are relatively specific; and, perhaps
most importantly, the natural history and biologic behavior of HPV infections
and cervical cancer are consistent with a causal association between HPV
and cervical cancer.5 Numerous studies have
tested tissue from invasive cervical cancers using sensitive molecular
methods, and these studies uniformly have identified high-risk types of
HPV DNA in more than 93% of invasive cervical cancers.79,81,82
The same types of HPV are found, regardless of geographic area from which
the cancers are selected (Table 12). When cervical
cancer samples initially found to be HPV DNA negative were retested using
more sensitive HPV DNA detection methods, HPV DNA was identified in almost
all of the HPV DNA–negative samples, with an overall HPV DNA positivity
rate of greater than 99%.83
TABLE 12. Prevalence of Specific Types of Human Papillomavirus in Invasive
Cervical Cancer by Geographic Region
| |
Geographic Region
|
| |
Africa
|
Latin America
|
Southeast Asia
|
Europe
|
North America
|
|
HPV Type
|
No.
|
(%)
|
No.
|
(%)
|
No.
|
(%)
|
No.
|
(%)
|
No.
|
(%)
|
|
HPV 16 and associated
|
|
HPV 16
|
79
|
(43)
|
255
|
(51)
|
42
|
(43)
|
56
|
(65)
|
33
|
(58)
|
|
HPV 31
|
5
|
(3)
|
35
|
(7)
|
1
|
(1)
|
5
|
(6)
|
3
|
(5)
|
|
HPV 33
|
5
|
(3)
|
18
|
(4)
|
2
|
(2)
|
1
|
(1)
|
0
|
|
|
HPV 35
|
4
|
(2)
|
10
|
(2)
|
1
|
(1)
|
1
|
(1)
|
0
|
|
|
HPV 52
|
4
|
(2)
|
16
|
(3)
|
2
|
(2)
|
3
|
(4)
|
0
|
|
|
HPV 58
|
5
|
(3)
|
11
|
(2)
|
2
|
(2)
|
1
|
(1)
|
0
|
|
|
HPV 18 and associated
|
|
HPV 18
|
33
|
(18)
|
48
|
(10)
|
31
|
(32)
|
7
|
(8)
|
9
|
(16)
|
|
HPV 39
|
0
|
|
13
|
(3)
|
1
|
(1)
|
0
|
|
0
|
|
|
HPV 45
|
23
|
(12)
|
37
|
(7)
|
8
|
(8)
|
2
|
(2)
|
8
|
(14)
|
|
HPV 59
|
0
|
|
14
|
(2.8)
|
1
|
(1)
|
0
|
|
0
|
|
|
HPV 68
|
|
Other
|
|
HPV 56
|
6
|
(3)
|
3
|
(1)
|
3
|
(3)
|
2
|
(2)
|
2
|
(4)
|
|
Miscellaneous
|
5
|
(3)
|
16
|
(3)
|
4
|
(4)
|
1
|
(1)
|
0
|
|
|
Undetermined
|
2
|
(1)
|
8
|
(2)
|
0
|
|
1
|
(1)
|
1
|
(2)
|
|
HPV negative
|
19
|
(10)
|
36
|
(7)
|
7
|
(3)
|
4
|
(5)
|
4
|
(7)
|
|
Total samples
|
186
|
|
505
|
|
98
|
|
86
|
|
57
|
|
HPV, human papillomavirus.
Modified from Bosch FX, Manos MM, Munoz N, et al: Prevalence of human papillomavirus
in cervical cancer: A worldwide perspective. International
biological study on cervical cancer (IBSCC) Study Group. J Natl Cancer
Inst 87:779, 1995.
Numerous studies have shown a clear, consistent association between infection
with specific high-risk types of HPV and invasive cervical cancer
and its precursor lesions. They also have shown that exposure to HPV
precedes the development of cervical disease. This evidence, combined
with evidence identifying the molecular pathways by which HPV could
cause cervical cancer, supports a causal relationship between HPV infection, acquired
through sexual contact, and the development of SIL and
invasive cervical cancer. Based on these data, the IARC has classified
HPV 16 and 18 as carcinogens in humans.4 New data on the role of other types of HPV in the pathogenesis of cervical
cancer were reviewed by Bosch and coworkers,5 and they concluded that HPV types 31, 33, 35, 39, 51, 52, 59, and 68 also
should be classified as carcinogens in humans. NATURAL HISTORY OF HUMAN PAPILLOMAVIRUS INFECTIONS. Much has been learned in recent years regarding the natural history of
HPV infections. Anogenital HPV infections are rare in virgins but become
common when young women initiate sexual intercourse. A prospective
study of female college students in the United States who were followed
at regular intervals over a 3-year period found that 26% of the
women were HPV DNA positive at entry into the study.84 After only 3 years of follow-up, however, cervicovaginal HPV infections
were identified in an additional 43% of the women. Two thirds
of these young, sexually active college women were found to have anogenital
HPV infection at some point in the study. Other studies have confirmed
a high prevalence of anogenital HPV infections in sexually active
young women. Of female college students from the University of California, 46% were
found to have anogenital HPV infections using
sensitive polymerase chain reaction assays.85 Even these relatively high prevalence figures may underestimate the number
of HPV-infected women because shedding of sufficient quantities of
HPV DNA to be detected with the currently available molecular methods
occurs only transiently in many young women. When female college students
were tested weekly for HPV DNA using polymerase chain reaction, only 26% of
the women were HPV DNA positive at the first examination, but
after 10 visits, the cumulative prevalence of HPV DNA positivity
was 44%.86 In general, anogenital HPV infections tend to be transient and of relatively
short duration in both young and older women. In the study by Ho
and colleagues84 of college women, the median duration of HPV infections was only 8 months, and
by 24 months of follow-up, 91% of HPV-infected women had
become HPV DNA negative. The duration of infection was longer for women
infected with high-risk types of HPV than for women infected with
low-risk types of HPV. Similarly, in a study of young women from San
Francisco, Moscicki and coworkers87 reported that approximately 70% of young women positive for HPV
had regression of their infections by 24 months. Women with low-risk
types of HPV infections also were more likely to show HPV regression than
were women with high-risk types of HPV infection in this study. In
another study of older women from New York City, it was found that persistent
HPV infections (defined as the same type of HPV detected twice
over a 12-month period) occurred in only 16% of the women who
were HPV DNA positive at enrollment. In the New York City study, persistence
was more common among women infected with high-risk types of HPV
compared with low-risk types of HPV.88 Because most HPV-infected women have spontaneous resolution of their infections, the
prevalence of HPV infections decreases with increasing
age.
It seems that most sexually active young women are exposed to HPV at
some point after initiating sexual activity.84,85,87,89
Most women develop transient HPV infections that are of relatively short
duration, and eventually most become HPV DNA negative. Only a small proportion
of women exposed to HPV become persistently infected and continue to have
detectable levels of HPV DNA in the genital epithelium. These persistently
infected women are at risk for having persistent high-grade SIL and of
developing invasive cervical cancer.90,91,92
Although the exact factors that regulate the clearance of an anogenital
HPV infection are unknown, immunologic factors and viral type are important.
The role of immunologic factors is shown by the finding that persistence
of HPV infections is more common in HIV-infected compared with HIV-uninfected
women and that rates of persistence in this population increase with increasing
levels of immunosuppression.88 A possible
role for humoral immunity in the loss of infection is suggested by the
finding that 67% of women enrolled in a prospective follow-up study
who developed an incident HPV 16 infection subsequently developed serum
antibodies against HPV 16.93 The median
time to seroconversion was 8.3 months, which is similar to the median
duration of incident HPV infections in most studies.93
Evidence supporting a central role for persistence of infection with
high-risk types of HPV in the pathogenesis of high-grade SIL (CIN 2, 3)
and invasive cervical cancer comes from two types of studies. One is case-control
studies of women developing high-grade SIL (CIN 2, 3) or invasive cervical
cancer in which archived conventional Pap tests were tested for the presence
of HPV using sensitive polymerase chain reaction–based methods.
These case-control studies showed that women who are persistently infected
with high-risk types of HPV are at least 30 times more likely to develop
high-grade SIL (CIN 2, 3) than women who are high-risk HPV DNA negative.
Also, women who are persistently infected with HPV DNA are 213 times more
likely to develop invasive cervical cancer than are women who are not
infected with HPV.90,94,95
Long-term prospective follow-up studies also have documented a central
role for HPV persistence in the pathogenesis of SIL. Hopman and associates92
followed 68 women who were cytologically negative but HPV DNA positive,
using cytology, colposcopy, and HPV DNA testing for 34 months. During
follow-up, 17 (25%) of these women subsequently developed an abnormal
Pap test, and 8 (12%) developed biopsy-confirmed high-grade SIL.
Of the women developing a cervical abnormality, 94% had a persistent
high-risk HPV DNA infection.92 Similar findings
were reported by Ellerbrock and coworkers,91
who found that 12% of cytologically and colposcopically normal
women who had high-risk HPV infections subsequently developed biopsy-confirmed
SIL during 36 months of follow-up. Women who were persistently infected
with high-risk types of HPV were significantly more likely to develop
incident SIL on follow-up than were women who were transiently infected
with high-risk types of HPV.
HUMAN PAPILLOMAVIRUS DNA TESTING FOR CERVICAL CANCER SCREENING. Performance in Clinical Studies.
Although none of the commercially available HPV DNA testing methods is
approved by the U.S. Food and Drug Administration for primary screening,
many studies have evaluated HPV DNA testing as a cervical cancer screening
method. We conducted a study of previously unscreened South African women
older than age 35 years that compared the sensitivity and specificity
of conventional cytology and Hybrid Capture II for the detection of high-grade
SIL (CIN 2, 3) and cervical cancer (Table 13).96
HPV DNA testing had a significantly higher sensitivity but a lower specificity
than cytology. A large National Cancer Institute–sponsored study
conducted in Costa Rica also found that HPV DNA testing was more sensitive
but less specific than cytology (see Table 13).97
In the Costa Rica study, the specificity of HPV DNA testing was better
than that observed in South Africa. Other studies conducted in the United
Kingdom, Canada, Germany, France, and China also found that the sensitivity
of HPV DNA testing is superior to that of cytology (see Table
13).98,99,100,101
TABLE 13. Screening for High-Grade Cervical Neoplasia with Human Papillomavirus
DNA Testing
| | | Pap Smear | Hybrid Capture II |
Author | Country |
Sensitivity*
| Specificity |
Sensitivity*
| Specificity |
Kuhn96 | South Africa |
0.78 |
0.97 |
0.88 |
0.82 |
Schiffman97 | Costa Rica |
0.78 |
0.94 |
0.88 |
0.89 |
Cuzick100 | United Kingdom |
0.86 |
|
0.95 | |
Belinson99 | China† |
0.94 |
0.78 |
0.98 |
0.85 |
Ratnam98 | Newfoundland |
0.40 |
0.92 |
0.68 |
0.91 |
Clavel101 | France† |
0.88 |
0.93 |
1.00 |
0.86 |
*For biopsy-confirmed high-grade squamous intraepithelial lesions
(cervical intraepithelial neoplasia [CIN] 2, 3) and cervical cancer.
†Liquid-based cytology.
One of the most significant drawbacks to using HPV DNA testing as a screening
test is the high rate of HPV DNA positivity in women lacking cervical
disease (i.e., its apparent low specificity). Although these cases
frequently are described as a being “false positives,” these
women do not have false-positive results in the standard sense
of the term. These women are truly infected with high-risk types of
HPV and have a significantly elevated risk for subsequently developing
SIL (CIN), even though they may not have SIL (CIN) at the time the test
is obtained. There are many explanations for the relatively high rate
of HPV DNA positivity in some of the published screening studies. In
the South African screening study, almost 8% of the women were
HIV-infected, 9% were infected with either gonorrhea or chlamydia, and 25% had
culture-proven trichomonas. The studies from
Costa Rica and Canada included many young women, and this may account
for the high rates of HPV DNA positivity in women lacking cervical disease.
Many studies have documented the prevalence of HPV DNA positivity in
women of different ages (Table 14).78,98,100,102,103,104
In the studies from the United Kingdom, the Netherlands, Costa Rica, and
Newfoundland, the prevalence of high-risk types of HPV in women age 35
years or older was 5% or less. This is not any higher than the
rate of cytologic abnormalities in women undergoing cytologic screening
in the United States.105 Much higher prevalences
of HPV DNA positivity were identified in older women from Zimbabwe. More
than 50% of the women from Zimbabwe were infected with HIV, however,
and the strong associations between HIV and HPV infection explain the
high rates of HPV DNA positivity observed in the older women in that population.104
TABLE 14. Impact of Age on Rates of Human Papillomavirus DNA Positivity
| | | % HPV DNA Positive by Age Group |
Author | Country | <25 years | 25–34 years | 35–44 years | >45 years |
Jacobs et al102 | Netherlands*† |
13 |
10 |
2 |
2 |
Herrero et al78 | Costa Rica*† |
10 |
6 |
3 |
3 |
Ratnam et al98 | Newfoundlandठ|
17 |
12 |
5 |
4 |
Cuzick et al100 | United Kingdom†§ |
|
3 |
3 |
5 |
Womack et al110 | Zimbabweठ|
|
32 |
22 |
24 |
HPV, human papillomavirus.
*Women without squamous intraepithelial lesions.
†By polymerase chain reaction.
‡By Hybrid Capture II for high-risk types of HPV.
§All women.
POTENTIAL STRATEGIES FOR USING HUMAN PAPILLOMAVIRUS DNA TESTING FOR SCREENING. There are several ways in which HPV DNA testing could be incorporated into
cervical cancer screening programs. One potential strategy would be
to use HPV DNA testing as a “stand-alone” screening method, much
as cytology is used today. When used as a stand-alone screening
method, it is likely that HPV testing would need to be restricted
to women older than age 35 years because of its low specificity in younger
women. This restriction should not be a problem in low-resource
settings, however, where the target age for screening typically begins
at 35 to 40 years. Another strategy would be to perform HPV DNA testing
and cervical cytology simultaneously. This strategy might be useful
for women of all ages, especially if HPV DNA status were used to determine
screening frequency, rather than to refer women to colposcopy. Another
strategy would be to combine HPV DNA testing and cytology sequentially
into a two-stage screening program (Fig. 9). In a two-stage strategy, all women would be screened using the first
test, and only women who are positive on the first test would be screened
using the second test.58 This strategy would increase specificity greatly, but also would reduce
sensitivity. 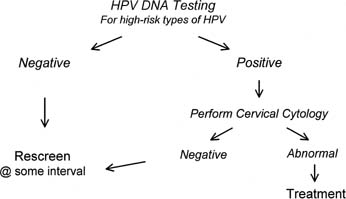 Fig. 9. Two-stage approach to cervical cancer screening.(Data from Denny L, Kuhn L, Risi L, et al: Two-stage cervical cancer screening: An
alternative for resource-poor settings. Am J Obstet Gynecol 183:383, 2000.) Fig. 9. Two-stage approach to cervical cancer screening.(Data from Denny L, Kuhn L, Risi L, et al: Two-stage cervical cancer screening: An
alternative for resource-poor settings. Am J Obstet Gynecol 183:383, 2000.)
|
ADVANTAGES OF HUMAN PAPILLOMAVIRUS DNA TESTING. HPV DNA testing has many advantages as a screening test compared with cervical
cytology. Because HPV DNA testing is more sensitive than cytology, it
is less likely that a high-grade SIL (CIN 2,3) would be missed
when HPV DNA testing is used. The performance of screening is improved, especially
when screening is done at extended intervals, such as every 5 years. Another
advantage is that HPV DNA testing can be performed
on self-collected vaginal samples; this allows screening to be performed
without a speculum examination.106 This may be advantageous for low-resource settings because it eliminates
the need to train providers in how to collect samples for screening. Another
important advantage is that in contrast to cytology, which simply
determines whether or not a patient has a lesion at the time screening
takes place, HPV DNA testing identifies which women are at greatest
risk for developing cervical disease in the future. Screening programs
might be able to be tailored to an individual’s specific
risk for developing cervical disease in the future. |