In the past, irradiation, natural sun light, vitamins, sanatorium care, and
for pelvic tuberculosis in particular, pelvic diathermy and injection
of oxygen intraperitoneally, have all been tried. Before the advent
of effective chemotherapy, surgery was the mainstay of treatment of
genital tract TB, and postoperative complications such as bowel fistula (14%) and
mortality from the primary disease (2.2%) were
high.105 Unfortunately, there are sparse, prospective, controlled clinical studies
regarding treatment of female genital TB, a gap that is found in the
broader context of extrapulmonary TB. The small amount of data, however, suggest
that extrapulmonary TB can be very effectively treated with
some standard short-course regimen.106 Experts suggest that extrapulmonary TB may be even easier to treat than
pulmonary TB owing to the decreased concentration of organisms in these
lesions and the increased accessibility of many of the sites to antituberculous
medication. The advent of antituberculous drugs has revolutionized the management of
this disease. If surgical intervention is needed, chemotherapy makes
this approach safer, easier, and more effective. Three basic principles
have emerged in the years following the advent of chemotherapy for
TB. - Regimen for treatment must contain multiple drugs to which the organism
is susceptible.
- The drugs should be taken regularly.
- The drug therapy should continue for a sufficient period of time.
The treatment of extrapulmonary TB, including genital tract TB, is the
same as the treatment of pulmonary TB (Tables 5 and 6). Thus, the current standards in the treatment of tuberculosis are - A 6-month regimen consisting of isoniazid (INH), rifampin (RIF) and pyrazinamide (PZA) for 2 months, followed by INH and RIF for 4 months, is
the preferred treatment for patients with fully susceptible organism
who adhere to treatment. Ethambutol (EMB) or streptomycin (SM) should
be included in the initial regimen until the results of drug sensitivity
studies are available, unless there is little possibility of drug resistance. This
four-drug, 6-month regimen is effective even when the
infecting organism is resistant to INH. This recommendation applies to
both HIV-infected patients and those who are noninfected with HIV. However, in
the presence of HIV infection, the clinical course should be
closely monitored, and treatment should be prolonged if the course is
determined to be slow or suboptimal.
- A 9-month regimen of INH and RIF is acceptable in patients who cannot tolerate
PZA. EMB or SM should be included until the drug susceptibility
studies are available, unless there is little possibility for drug resistance. Consideration
should be given to treating all patients with
directly observed therapy (DOT)
- The major determinant of the outcome of treatment is patient adherence
to the drug regimen. Virtually all treatment regimens may be given intermittently
if directly observed, thus ensuring adherence.
TABLE 5. Drug Regimens for Genital Tuberculosis
Initial Therapy
INH + RIF + PZA daily for 2 mo
Add EMB or SM where there is high rate of resistance until drug susceptibility
data are known
Continuation Phase
INH + RIF daily for 4−10 mo (total 6−12 mo)
Add pyridoxine 25−50 mg daily to regimens that include INH
EMB, ethambutol; INH, isoniazid; PZA, pyrazinamide; RIF, rifampin; SM, streptomycin.
TABLE 6. Dosage of Antituberculous Drugs
| | Dose (mg/kg)* |
Regimen | INH | RIF | PZA | EMB | SM |
Daily |
5 (300) |
10 (600) |
15–30 (2000) |
15–25 |
15 (1000) |
2x/wk (DOT) |
15 (900) |
10 (600) |
50–70 (4000) |
50 |
25–30 (1500) |
3x/wk (DOT) |
15 (900) |
10 (600) |
50–70 (4000) |
25–30 |
25–30 (1500) |
DOT, directly observed therapy; EMB, ethambutol; INH, isoniazid; PZA, pyrazanimide; RIF, rifampin; SM, stretomycin.
*Maximum daily dose is in parentheses.
Dutt and Stead, based on their study of 478 patients with extrapulmonary
TB including 65 patients with genitourinary TB from 1978 to 1987, concluded
that treatment with short-course antituberculous chemotherapy—consisting
of INH 300 mg orally 4 times a day and RIF 600 mg orally 4 times
a day for 1 month, followed by INH 900 mg and RIF 600 mg
twice a week for another 8 months—was successful in 95% of
their cases.106 Arora and colleagues investigated the efficacy of short-course chemotherapy
in the specific problem related to genital TB in a small prospective
trial.107 Forty-one patients with proven genital TB were given SM/INH/RIF/PZA for 2 months, followed
by INH/RIF for another 7 months. Thirty-two of these
patients completed the full 9 months of therapy, and of these patients, 78% were
considered cured, defined as both symptomatic and
histologic microbiologic resolution.107 A similar outcome was achieved by Jindal and associates in a small prospective
study of 14 female patients with genital TB who were treated with
daily RIF 450 mg and INH 300 mg for 9 months. Follow-up endometrial
biopsies were negative in all patients treated with this regimen. Three
out of 14 patients (21%) conceived after starting therapy.108 Drugs in Current Use ISONIAZID.
INH is the most widely used antituberculous agent.109
It is bactericidal, relatively nontoxic, easily administered, and inexpensive.
Absorption from the gut is nearly complete, and it is highly active against
M. tuberculosis. The usual dose is 3 to 5 mg/kg body weight. Hepatitis
is the major toxic effect and is increased with increasing age to 65 years.110,111
Alcohol consumption is also identified as a risk cofactor.112,113
Peripheral neuropathy, most likely due to interference with pyridoxine
metabolism, is associated with INH administration but is uncommon at a
dose of 5 mg/kg. In patients at risk for peripheral neuropathy, concomitant
pyridoxine administration is recommended, and it is also recommended in
patients with seizure disorders and in pregnant women.
RIFAMPIN. RIF is bactericidal to M. tuberculosis, relatively nontoxic, and easily administered. It is rapidly absorbed from
the gut. It penetrates well into tissues and cells. The most common
adverse effect to RIF is gastrointestinal upset. Other reactions include
skin eruptions, hepatitis, and rarely, thrombocytopenia and cholestatic
jaundice. RIF induces hepatic microsomal enzymes and may accelerate
clearance of drugs metabolized by the liver.114 These include methadone, warfarin derivatives, glucocorticoids, estrogens, oral
hypoglycemic agents, digoxin, antiarrhythmic agents, theophylline, anticonvulsants, ketoconazole, and cyclosporine. Because RIF interferes
with estrogen metabolism, it can lower the effectiveness of oral
contraceptives. In adults, intermittent administration of RIF in doses
larger than 10 mg/kg may be associated with thrombocytopenia, influenza-like
syndrome, hemolytic anemia, and acute renal failure. RIF is
excreted in urine, tears, sweat, and other fluids, and it colors them
orange. Patients should be advised of this discoloration and also of
the possibility of permanent discoloration of soft contact lenses. PYRAZINAMIDE.
PZA is bactericidal against M. tuberculosis in an acidic environment
and is active against organisms in marcophages. Absorption from the gastrointestinal
tract is nearly complete. PZA penetrates well into most tissues. The most
important side effect of this drug is hepatotoxicity. However, there does
not appear to be an increase in hepatotoxicity when PZA is added to a
regimen of INH and RIF in the initial 2 months of therapy. Hyperuricemia
appears frequently, but acute gout is rare.34,112
Skin rash and gastrointestinal irritation are also seen.
ETHAMBUTOL. EMB in usual doses is generally considered to have a bacteriostatic effect
on M. tuberculosis but may have a bactericidal effect when used in higher doses in intermittent
therapy. The drug is easily administered and has a low frequency
of adverse effects. It accumulates in patients with renal insufficiency. Retrobulbar
neuritis is the most frequent and serious adverse effect
of EMB. Symptoms include blurring of vision, central scotomata, and
red-green colorblindness.112 The complication is dose-related and occurs in less than 1% of
patients receiving a daily dose of 15 mg/kg, increases with a daily dose
of 25 mg/kg, and is increased in patients with renal insufficiency. STREPTOMYCIN. SM is bactericidal in an alkaline environment and is given parenterally. Excretion
of the drug is almost entirely renal. It has good tissue penetration. The
most common serious adverse effect of SM is ototoxicity.115 This usually results in vertigo, but hearing loss may also occur. Nephrotoxicity
may also occasionally occur. A total cumulative dose of more
than 120 g should not be given unless other therapeutic options are
not available. OTHER DRUGS USED AGAINST MYCOBACTERIUM TUBERCULOSIS. Other drugs that have been used against M. tuberculosis and are currently being used, especially in multidrug-resistant disease, are
para-aminosalicylic acid (PAS), cycloserine, capreomycin, kanamycin, thiacetazone, amikacin, ciprofloxacin, and ofloxacin. Ciprofloxacin
and ofloxacin are not licensed for the treatment of TB. SUCCESS RATES AND FOLLOW-UPS. Malkani and Rajani used weekly biopsies to follow a series of 30 patients
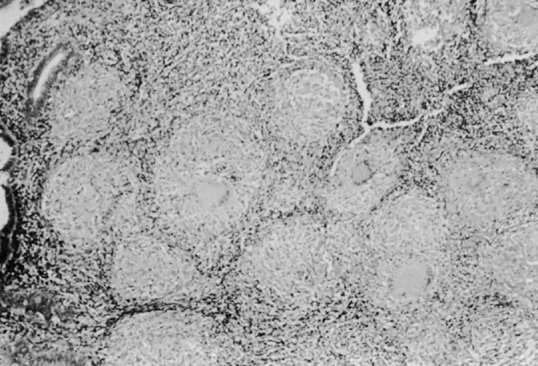
receiving antituberculous drugs for the treatment of tuberculous endometriosis.69 From the 4th week, these patients showed an increasing number of histologically
negative endometria, and after 12 weeks, the endometria in all 30 were
negative by biopsy. According to Kardos, tuberculous endometritis
may be considered “cured” as seen on histologic examination
by using antimicrobial therapy of 3 months-duration, although
he noted the recurrence of endometritis within 4 to 5 years if the
fallopian tubes were not removed.116 Schaefer reviewed 387 cases of tuberculous endometritis treated for periods
as long as 4 months.117 Twenty-two percent showed disease recurrence within 3 years. To determine
the effect of antituberculous drug therapy on the fallopian tubes, which
may be the cause of disease recurrence, and to arrive at the optimal
duration of therapy, Schaefer treated a series of patients with
genital TB for periods varying from 6 weeks to 3 years before removing
the fallopian tubes.117 In patients with minimal genital TB, no evidence of active TB was found
in the tubes after 10 months of therapy. Schaefer subsequently advocated a regimen of treatment using INH 300 mg
daily, and EMB 20 mg/kg or approximately 1200 mg/day.72 At 6 months and 12 months, endometrial curettings or biopsies are examined
bacteriologically and microscopically. If, during the course of treatment, the
endometrial curettings become positive or tuboovarian masses
appear, RIF in a dose of 600 mg daily is added for 3 months, and
the patient is advised to have surgery. Schaefer advocated treatment using
INH and EMB for at least 2 years if there were no complications.117 Subsequently, his patients continued to receive INH for an indefinite
period. For advanced disease, Schaefer advocated INH and EMB for a period of 3 to 4 months, and
if the tuboovarian mass still persisted, he advised surgery
followed by antimicrobial therapy.117 In general, over 95% of patients treated for TB for the first time, using
a combined drug regimen, undergo successful treatment if they
complete the prescribed drug course. Most patients become noninfectious
very rapidly, usually within 2 weeks.115 In patients who have had previous chemotherapy, who have had contact with
a INH-resistant case, or who acquired their infection in an area with
a high prevalence of resistant organisms, such as Asia, Latin America, or
Africa, drug resistance should be suspected and drug sensitivity
tests should be performed. Careful compliance is the most important
factor in preventing the development of drug resistance.34 Corticosteroids have been proposed by some as an adjuvant therapy in the
management of TB and may ameliorate certain aspects of the inflammatory
response involved in the disease. Elsbach and Edsall stated that
there was no evidence of an adverse effect on the course of the disease
when chemotherapy was adequate.118
Patient response to treatment should be monitored by doing endometrial
curettage 6 and 12 months after the initiation of treatment. Patients
need to be followed closely for an indefinite period. Recurrence or dissemination
to other organs is rare but occasionally occurs. Chest radiographs, urine
cultures for M. tuberculosis, and uterine curettage should be repeated
at the end of the treatment course, with a repeat curettage or endometrial
biopsy every 6 to 12 months. Sutherland suggested that patients needed
follow-up assessment for several years.40,52
Surgical Treatment The role of surgery in the treatment of genital TB in modern times is clearly
as adjunctive therapy. Given the excellent results that can be
obtained by treating extrapulmonary TB with antituberculous chemotherapy, surgery
has become the back-up plan in most cases. Surgery is advocated
only if there is no other choice of treatment.
Indications for surgical intervention in the management of pelvic TB
include (1) persistent and recurrent disease despite adequate treatment;
(2) persistent or recurrent pelvic masses after 6 months of adequate therapy;
(3) persistent or recurrent symptoms such as pelvic pain and abnormal
bleeding; (4) a persistent nonhealing fistula; (5) multidrug-resistant
disease; and (6) concomitant genital tract neoplasia or other pathology.5,9,36,37,40,119
When surgery is advocated, the patient should be given chemotherapy for
at least 1 to 2 weeks preoperatively. Surgery should be performed at
midcycle in premenopausal patients, and chemotherapy should be continued
for 6 to 12 months postoperatively. Under antituberculous treatment, surgery
is technically much less difficult, and morbidity and mortality
are significantly reduced. Sutherland reported on 77 cases in which
surgery was undertaken during antituberculous drug treatment; no patient
developed a fistula, and there was no mortality.119 Late complications were infrequent. When TB is first diagnosed postoperatively after histologic examination, antituberculous
treatment is given immediately and continued for 6 to 12 months. The operation of choice is total abdominal hysterectomy with bilateral
salpingo-oopherectomy followed by hormone replacement therapy, especially
in a premenopausal woman. If the patient is premenopausal and the
ovaries look normal, they may be conserved. |