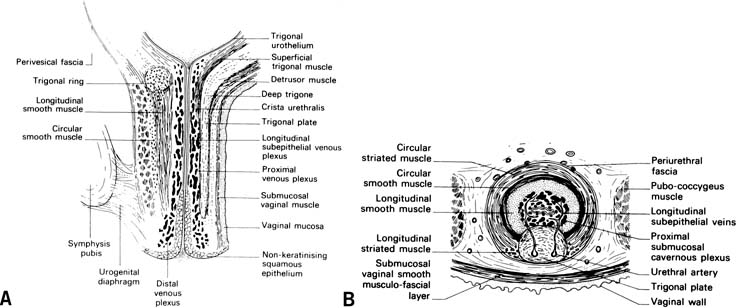
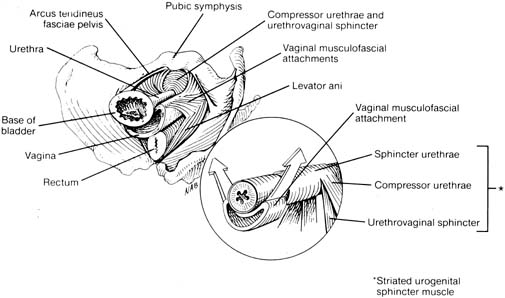
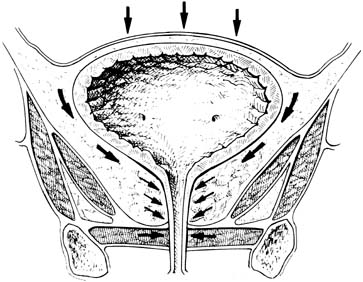
The general principles of any surgical procedure are to relieve suffering, restore
normal anatomy, and maintain function. The function of the
lower urinary tract is to store urine until volitional emptying can occur. Hence, relief
of suffering by resolving untimely and socially embarrassing
urine loss by procedures that compensate for abnormal anatomy
should not overcorrect, causing impairment of storage (diminished
bladder capacity or urge incontinence) or release of urine (retention
or voiding dysfunction). Proper counseling and preoperative
evaluation can be crucial in the avoidance of these problems. The American College of Obstetricians and Gynecologists (ACOG) has (Table 3) recommended that a number of steps be taken before surgical intervention, much
of which has been outlined.32 The presence of stress incontinence and absence of urge incontinence symptoms
should be confirmed in the history. The transient causes of urinary
incontinence, including delirium, infection, hypoestrogenism, pharmaceutical
causes, psychological causes, excessive urine production, restricted
mobility, and stool impaction, should be identified and managed
before other intervention considered. The basic preoperative evaluation
should include a history and physical examination, local neurologic
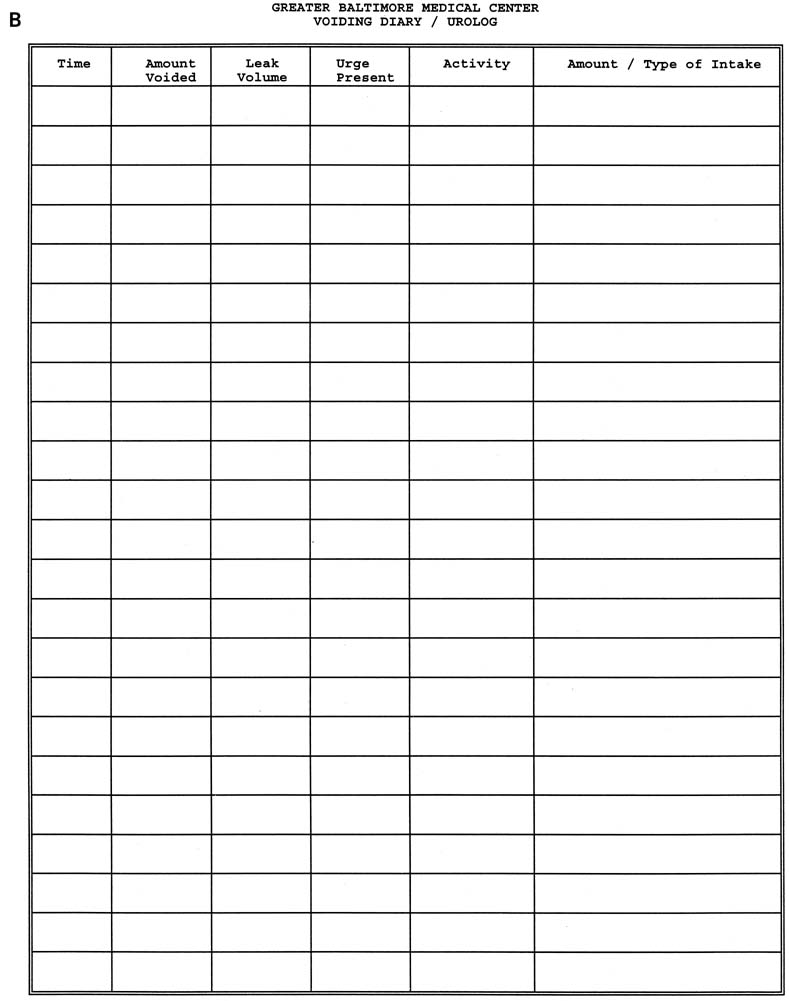
examination, pelvic examination, 24-hour voiding diary, stress
test demonstrating leakage, cotton swab test to confirm urethral
hypermobility, residual urine determination, urinalysis or urine culture, and
a screening cystometrogram. Table 3. ACOG Guidelines for Primary Surgery for SUI
| Confirmation of Indication |
Actions to the Procedure |
| Documentation of stress incontinence |
Document normal voiding habits |
| Identify and manage transient causes of stress incontinence |
Document normal neurological examination |
| Demonstrate stress loss and confirm low-residual urine |
Document absence of previous incontinence or radical
surgery |
| |
Document absence of pregnancy |
| |
Counsel patient regarding alternative therapy |
More than 200 procedures have been described in the literature for the
treatment of stress incontinence. This extraordinary number reflects a
combination of the alteration of techniques and approaches of established
and effective procedures, the introduction of newer technologies
and materials, as well as the discarding of procedures shown to be less
efficacious. In today's surgical practice, it is well-established
that performing an anterior repair or Kelly plication for the
treatment of SUI is substandard compared to more effective procedures.33 Because of significant recurrence rates at even 1 and 2 years of follow-up, long-needle procedures such as the Peyera, Stamey, or
Raz procedures, and their other modifications have also decreased
in popularity over the recent years. Currently, the two primary forms
of surgery for SUI remain the retropubic urethropexy and sling procedures. Injection
of urethral bulking agents also is performed as a less
invasive intervention for SUI in certain patients. Retropubic Urethropexy The gold standard surgical treatment of SUI in patients with a hypermobile
bladder neck but otherwise normally functioning urethra has been accomplished
through a retropubic approach (Fig. 9). The two procedures most studied are the Burch retropubic urethropexy
and the Marshall-Marchetti-Krantz (MMK) procedure. In
a Burch procedure, permanent sutures are placed lateral
to both sides of the mid urethra and UVJ into the fibromuscular layer
of the vagina (avoiding full thickness) and fixed to the ipsilateral
Cooper's ligament for a total of four sutures. Attachment
to the fibrocartilage of the symphysis pubis is performed for the MMK. Placement
of these sutures is aimed at reestablishing the intra-abdominal
location of the proximal urethra and UVJ and to minimize
descent, thus allowing normal pressure transmission to this crucial area
during times of stress. Cure rates for these procedures range from 85% to 90% at 1 to 5 years, and most 10-year data
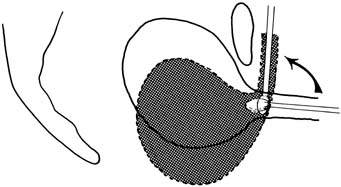
with more than a 70% cure rate.34,35 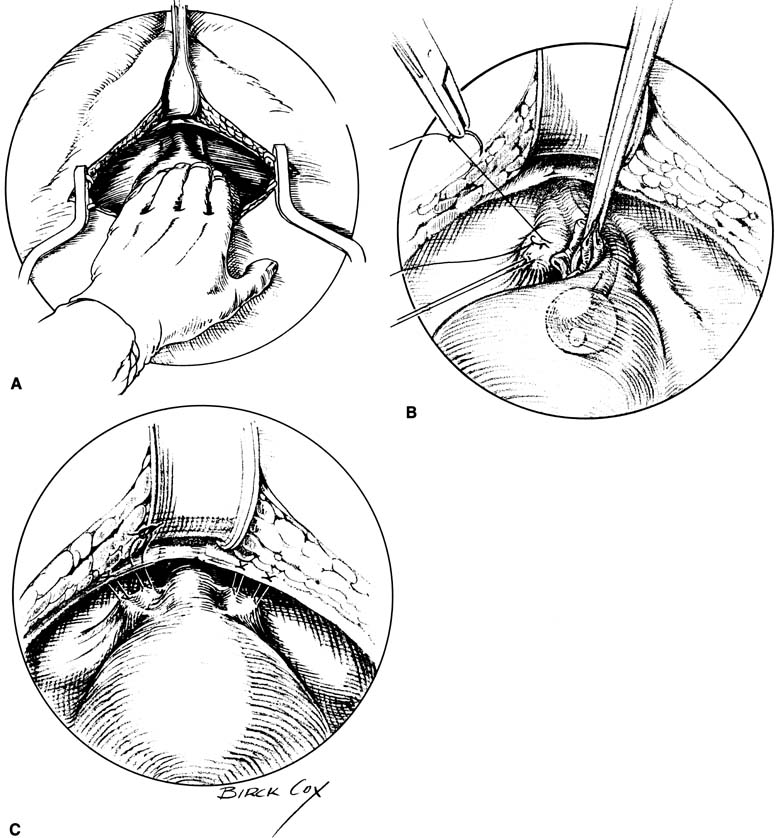 Fig. 9. Burch colposuspension. A. The space of Retzius is entered and a hand placed over the bladder. The
bladder neck is just distal to the fingers, and the vaginal tissues
and pubocervical fascia are located on either side of the urethra. B. The pubocervical fascia is sutured approximately 2 cm lateral to the bladder
neck with a double bite of permanent suture. The sponge stick keeps
the bladder retracted medially and superiorly away from the site
of suture placement in the pubocervical fascia. C. Each arm of each suture is brought up through Cooper's ligament and
tied over the top of the ligament, leaving banjo strings but satisfactorily
elevating the bladder neck. Fig. 9. Burch colposuspension. A. The space of Retzius is entered and a hand placed over the bladder. The
bladder neck is just distal to the fingers, and the vaginal tissues
and pubocervical fascia are located on either side of the urethra. B. The pubocervical fascia is sutured approximately 2 cm lateral to the bladder
neck with a double bite of permanent suture. The sponge stick keeps
the bladder retracted medially and superiorly away from the site
of suture placement in the pubocervical fascia. C. Each arm of each suture is brought up through Cooper's ligament and
tied over the top of the ligament, leaving banjo strings but satisfactorily
elevating the bladder neck.
|
Traditionally, this procedure can be performed in concert with an abdominal
hysterectomy or alone, through a retropubic approach without opening
the peritoneal cavity. With the increased trend toward minimally invasive
surgery, methods for laparoscopic urethropexy have been developed. Besides
either an intraabdominal or an extraperitoneal approach, numerous
modifications have been suggested including using only two sutures, substituting
mesh, or using tacks, anchors, and other tools to
elevate the bladder neck. These variations significantly lower cure rates
as compared with traditional open urethropexy. Placement of four permanent
sutures identical to an open procedure, though, has yielded similar 1- and 2-year cure rates as an open Burch (93% and 89%, respectively),36 although no prospective trial between abdominal and laparoscopic Burch
procedures has been published. Currently, it is our practice to place
four sutures identical to the open procedure when using a laparoscopic
approach for optimal results. Transvaginal placement of urethropexy
sutures has also been reported in small numbers, but no RCTs demonstrating
comparable efficacy have been performed.37 Suburethral Slings Placement of a sling under the bladder neck and proximal urethra had traditionally
been used for patients with severe incontinence and poorly
functioning urethras, and for those with recurrent stress incontinence. More
often, though, suburethral slings (SUS) are being used
as treatment for primary stress incontinence, an approach advocated
by some urologists.38,39 The debated increase in morbidity, specifically urinary retention and
urge incontinence, as compared with urethropexy is the point of contention
regarding primary use of this procedure for treatment of SUI.40 Intermediate (1 to 2 years) and long-term (2 to 10 years) cure
rates of 85% to 90% and 70% to 85%, respectively, are cited in the literature for most slings.39,41 In general, SUS can be considered full-length slings extending from
rectus fascia all the way around the bladder neck and back up to the
contralateral rectus fascia, or patch slings that use suture bridges
that are tied over or sutured to the fascia. Full-length autologous
slings can be obtained by harvesting fascia either from the rectus
sheath or from the fascia lata of the leg. The sling is usually sutured
into place with delayed absorbable sutures under the bladder neck. The
tails of the full-length slings are sutured to the rectus
fascia directly with good care to keep the sling loose. Similarly, the
permanent suture bridge of patch slings is tied over the fascia or
to the fascia loosely to protect against overcorrection and outlet occlusion. The two slings described are the most common types of autologous slings
cited in the literature. Other less-proven autologous materials
include vaginal wall slings and rectus abdominus slings. Cadaveric fascia
lata, dermal allograft, porcine dermis, and porcine small intestinal
submucosa are other sources of nonsynthetic sling material. Synthetic
slings, used as either full-length or patches, include Mersilene, Marlex, Prolene, Gore-tex, and silastic bands. Some of
these novel materials have used bone anchors or other minimally invasive
means to place a sling under the bladder neck, although long-term
data regarding efficacy of these modifications, as compared with
traditional slings, remain lacking. A variation of the classic sling was developed in Europe by Ulmsten and
colleagues using a Prolene mesh placed under the mid urethra (Fig. 10).42 The increasingly popular tension-free vaginal tape (TVT) and
its subsequent competitors (Sparc, Uretex) were developed
as a minimally invasive outpatient procedure that can be performed
under intravenous sedation and local anesthesia. Placement of the mesh
is accomplished through a small 1- to 2- cm incision
under the mid urethra with blind passage of delivery needles up behind
the pubic bone and through two small 5-mm supra-pubic
skin incisions. The mesh is positioned loosely under the mid urethra, the
protective plastic sheath removed, and the tails trimmed, which retract
under the skin. These slings are not sutured in place, but rather
the friction of the mesh pores keeps it in position initially until
ingrowth of fibroblasts scars the sling in place. Most studies report
an 85% cure rate with an additional 5% to 10% significantly
improved.42,43,44 Because the needles are passed blindly, common complications of bladder
perforation (4%–9%) and, less commonly, hematoma
formation (1%) can occur.42,43,44,45 The low incidence of postoperative urge incontinence and obstructive voiding
as well as the quick return to normal voiding has been attributed
to the minimal peri-urethral dissection and loose application
of the sling as directed by the developers. Although a lower incidence
of erosion (< 1%) has been seen with this synthetic
mesh compared with historical numbers for synthetic slings, the blind
passage of the needles has caused some notable trauma to bowel, iliac
vessels, and epigastric vessels, resulting in life-threatening
situations.  Fig. 10. The tension-free vaginal tape (TVT) device. Top left: polypropylene
mesh tape in plastic sheath . Top right: introducer handle. Bottom
left: mesh tape attached to introducer needles. Bottom right: rigid
catheter guide used to deflect urethra and bladder away from
side of tape placement (Reproduced with permission from Gynecare, Somerville, NJ). Fig. 10. The tension-free vaginal tape (TVT) device. Top left: polypropylene
mesh tape in plastic sheath . Top right: introducer handle. Bottom
left: mesh tape attached to introducer needles. Bottom right: rigid
catheter guide used to deflect urethra and bladder away from
side of tape placement (Reproduced with permission from Gynecare, Somerville, NJ).
|
Injectable Treatments for Stress Incontinence Injectable agents designed to bulk the proximal urethra and bladder neck
have been developed as a minimally invasive, office-based procedure
for the treatment of SUI. The numerous agents now in use and in
development are primarily aimed for the treatment of hypomobile stress
incontinence in patients with poorly functioning urethral sphincters. Cystoscopically
guided placement of these agents, either transurethrally
or periurethrally, under the urethral mucosa aids in proximal urethral
and bladder neck closure during times of increased stress (Fig. 11). Although primarily aimed to treat patients with immobile bladder
necks, patients with severe comorbidities precluding more extensive
procedures for their stress incontinence may benefit from injections despite
a hypermobile UVJ.46 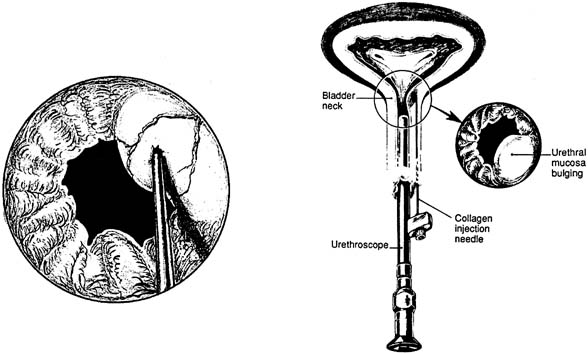 Fig. 11. Collagen injection therapy. On the right is the periurethral injection
technique. The collagen material is deposited underneath the urethral
mucosal lining while the surgeon observes with a 0-degree lens
and scope. On the left is the transurethral technique. The needle is in
the urethral mucosa at the 2 o'clock position, ready to inject (Bent AE: Periurethral collagen injections. Oper Tech Gynecol Surg
2: 51 1997.) Fig. 11. Collagen injection therapy. On the right is the periurethral injection
technique. The collagen material is deposited underneath the urethral
mucosal lining while the surgeon observes with a 0-degree lens
and scope. On the left is the transurethral technique. The needle is in
the urethral mucosa at the 2 o'clock position, ready to inject (Bent AE: Periurethral collagen injections. Oper Tech Gynecol Surg
2: 51 1997.)
|
Similar to the variations on slings and colposuspension, many agents have
been used or are in trials as a urethral bulking substance (Table 4). The many substances reflects the desire for an agent that is easily
injectable, is permanent and will not be degraded by the host, and
does not migrate or cause a damaging host reaction. The most commonly
used agent is sterile bovine dermal collagen that is cross-linked
with glutaraldehyde (Contigen; Bard Collagen Implant). A
skin test to detect an allergic response is required before injection. Cure
rates generally range from 20% to 40% with up to 70% to 80% improved and satisfied at 1 year.47 Degradation of the foreign collagen, which may occur between 3 months
and 3 years after placement, is the most common cause necessitating reinjection. Similar
results have been seen with trials of carbon beads (Durasphere), the next most commonly used bulking agent, when
compared with collagen. Table 4. Biomaterials and Synthetic Injectable Agents in Use and Development
as Urethral Bulking Materials
| Human and Xenograft Injectable Agents |
Synthetic Injectable Agents |
| Bovine cross-linked collagen |
Carbon beads |
| Autologous fat |
Teflon |
| Human collagen |
Ethylene vinyl alcohol copolymer-DMSO |
| Autologous cartilage |
Silicone |
| Protein/elastin admixture |
Injectable suture material |
| Bovine elastin/collagen |
Implantable microballoons |
| Autologous muscle derived cells |
Hyaluronic acid and dextranomener microspheres |
| |
Adjustable volume balloons |
| |
Cross-linked hyaluronic acid |
| |
Calcium hydroxyapatite |
Other Treatments Placement of artificial urethral sphincters tends to be performed by urologists
in patients who have not had symptoms cured by other aforementioned
interventions. Most of these devices involve an occlusive cuff
placed around the proximal urethra that is fed by a reservoir situated
in the labia majora for patient control. Consensus regarding suitable
patients and efficacy is still in question because of small cohort studies
and poor standardization. A success rate of between 80% and 90% is
most quoted, but a significant rate of revision, explantation, or
erosion of 5.9%48 to 17%49 has been seen. A different, minimally invasive, treatment modality being developed involves
radiofrequency thermal energy to cause tissue shrinkage at the bladder
neck (SURx; Thermatrx). This procedure, performed as
an outpatient procedure by a laparoscopic or vaginal approach, is designed
to cause scarring at the bladder neck to restrict mobility. In a
single small study of 94 women who underwent the laparoscopic procedure,50 subjective improvement, and cure rates, as well as objective cure (negative
stress test) were seen in approximately 80% of
patients. Further studies and longer follow-up are obviously needed. |