The hypoestrogenic state seen at menopause is manifested in many women by signs and symptoms of hormonal deficiency in tissues containing estrogen receptors, including the ovary, endometrium, vaginal epithelium, urethra, hypothalamus, and skin. The most common complaints are vasomotor disturbances characterized by hot flushes, genital atrophy, and psychologic symptoms. The decline in estrogen also causes an increased risk of osteoporosis.
Vasomotor Flushes
Vasomotor instability appears to arise not from a lack of estrogen but rather from its withdrawal. Estrogen-deficient patients with gonadal dysgenesis fail to develop hot flushes unless given estrogen replacement therapy (ERT) that is subsequently withdrawn.22 Castrated women with androgen insensitivity experience vasomotor symptoms after the discontinuation of estrogen. Similarly, premenopausal women treated with the antiestrogen clomiphene and postpartum women with very low estrogen levels often complain of hot flushes.
Hot flushes or flashes are experienced by 75% to 85% of all women undergoing natural menopause or who have undergone bilateral oophorectomies.23–26 The sudden drop in estrogen seen in oophorectomized women apparently makes them more prone to vasomotor instability. Hot flushes may begin in the perimenopausal period months or years before menopause, even in the presence of regular menstrual cycles.24 These women can be successfully treated with low-dose estrogen. More than four of five women experience hot flushes for more than 1 year, and 45% of women continue to have hot flushes for 5 to 10 years.25–27 Although one study showed a relationship between circulating estrogen and the occurrence of hot flushes,28 most investigators have not found such a correlation.29,30
The duration of the flush varies from a few seconds to minutes and rarely may last up to an hour. The flushes may be infrequent or may recur as often as every 30 minutes. Flushes appear to be more severe at times of stress and more frequent and severe at night, when they are called night sweats. Simultaneous electroencephalographic recordings show that women awaken before the detection of the physiologic changes seen with each flush.31 Night sweats can lead to significant sleep deprivation. Menopausal women not on replacement hormones have decreased rapid eye movement sleep and increased sleep latency compared with those on estrogen.
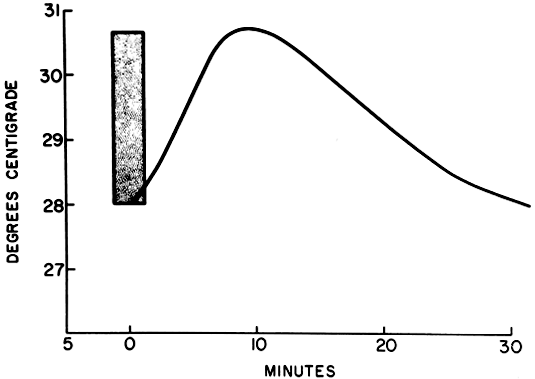
There is a subjective awareness of an impending hot flush, with an aura that may be perceived as a headache accompanied by heart palpitations occurring up to 4 minutes before the actual flush. The subjective sensation of the flush is followed by a change in skin conductance. There is then a rise in finger temperature that reflects cutaneous vasodilation (Fig. 1).32–34 The pulse rate increases an average of nine beats per minute, and a rapid rise is seen in blood flow to the hand before the flush35 (Table 2).
TABLE 2. Documented Physiologic Changes Known to Accompany Hot Flashes
Decrease in skin resistance, increased conductance
Rise in finger temperature
Fall in core body temperature
Increase in pulse rate
Increase in blood flow to hand
Vasodilation along cervical sympathetic pathway
Hormonal changes: increase in LH, ACTH, cortisol, dehydroepiandrosterone, androstenedione
Interruption of REM sleep
ACTH, corticotropin; LH, luteinizing hormone; REM, rapid eye movement.
The actual hot flush is subjectively characterized by a sensation of warmth and heat that starts in the chest and spreads upward over the neck and head. This sensation is accompanied by regional vasodilation, which causes flushing of the neck and face and produces body heat loss. The core temperature falls an average of 0.2°C, resulting in perspiration and chills.36 Other vasomotor symptoms may occur concurrently, including nausea, dizziness, headache, palpitations, and diaphoresis.
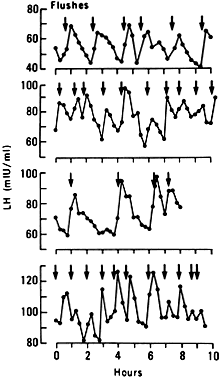
Endocrine studies have shown that hot flushes occur in menopausal women together with pulses of LH (Fig. 2). Although not every LH pulse is accompanied by a hot flush, virtually every hot flush occurs simultaneously with the onset of a pulse of LH.37 Circulating estrone and estradiol levels do not vary before or after the flush; however, the adrenal steroids cortisol, dehydroepiandrosterone, and androstenedione increase significantly at the time of the flush. Corticotropin (ACTH) and cortisone also increase in a pulsatile fashion with the temperature changes.38
The pathophysiology of the hot flush is not fully understood.39 The withdrawal of estrogen seems to alter the hypothalamic thermoregulatory system such that the “set point” lowers, the patient vasodilates to meet this temperature setting, and changes in neural activity alter hormonal secretions. Although LH pulses occur concurrently with flushes,37,38,40 LH does not initiate the hot flush, because patients with pituitary insufficiency and those with hypophysectomies can still have hot flushes.41,42 Injections of gonadotropins do not induce flushes, and administration of a gonadotropin-releasing hormone (GnRH) agonist inhibit pulsatile LH release and precipitate hot flushes.43,44 The hot flush has been associated with GnRH release, because the GnRH neurons are located near the preoptic nucleus of the hypothalamus, the nucleus responsible for temperature regulation. However, GnRH is not the initiating factor, because women with GnRH deficiency can still have hot flushes.
An alteration in the autonomic nervous system has been implicated because neuronal processes from the anterior hypothalamus innervate the superior cervical ganglion, accounting for the distribution of the flush, which spreads along the cervical sympathetic pathway. Norepinephrine, an α-adrenergic agent, releases GnRH and affects thermoregulatory function.45 The role of catecholamines is further supported by the fact that clonidine, an α2-adrenergic agonist, decreases the frequency and intensity of hot flushes, whereas yohimbine, an α2-adrenergic antagonist, provokes hot flushes.46,47 Attempts to measure levels of epinephrine and norepinephrine have produced conflicting results.37,48,49 Central adrenergic activation could cause the release of vasoactive substances such as prostaglandin or histamine. Catecholamines and prostaglandin E have been linked to the release of GnRH50,51 and to temperature regulation.52 These data led to the hypothesis that estrogen deficiency in the central nervous system induces changes in catecholamines and prostaglandins that affect neurons in the locus ceruleus, causing the regional vasodilation associated with the hot flush.22
It has also been suggested that a decrease in gonadal steroids may cause a fall in endogenous opioid activity within the hypothalamus, inducing the symptoms of menopause, which are similar to those of opiate withdrawal.53 This theory is based on several findings. Ovariectomy in the monkey causes a significant decrease in hypothalamic β-endorphin levels.54 Estradiol and progesterone increase β-endorphin secretion.55 Naloxone stimulates the release of LH in certain situations, suggesting inhibition of gonadotropin secretion by opioids.55 When morphine-dependent rats are given clonidine in doses of 10 to 50 μg, there is a modest increase in tail skin temperature, which is blocked if naloxone is also given.45
ERT has been shown clinically to improve vasomotor symptoms and totally eradicates them in most women.56,57 If a patient is unable to take estrogen, progestins have been shown to be effective in reducing hot flushes. Oral medroxyprogesterone acetate (MPA) (20 to 40 mg/day),58 depo-MPA (150 mg given intramuscularly every 3 months),59 or megestrol acetate (40 to 80 mg/day) decreases patient distress. Other agents suggested for the treatment of vasomotor instability include clonidine, naloxone, methyldopa, and a mixture of phenobarbital, ergotamine, and belladonna (Bellergal). The most carefully studied of these other agents is clonidine.46 Large doses of clonidine of up to 400 μg daily reduce the number of objectively recorded hot flushes; however, side effects have caused 40% of women to discontinue using clonidine in controlled studies.60 Moreover, at maximal tolerated dosages, the mean rate of hot flush occurrence decreases by less than one half.
Urogenital Changes
The decline in estrogen in the menopausal woman leads to a number of anatomic changes in the genitourinary tract and elsewhere (Table 3). The uterus becomes smaller and firmer, with a decrease in its total weight from 120 g to less than 50 g. There is also a decrease in myometrial thickness and vascularity. Most commonly, the endometrium becomes atrophic, the so-called senile endometrium; 5% of postmenopausal women develop a weakly proliferative endometrium, and a smaller percentage develop cystic hyperplasia or adenomatous hyperplasia, with even fewer developing neoplasia.
TABLE 3. Anatomic Changes of Menopause
Smaller uterus
Smaller cervix
Adolescent cervix: corpus ratio
Inner migration of squamocolumnar junction of cervix
Contraction of cervical glands
Smaller labia
Decreased Bartholin gland secretion
Smaller clitoris
Thinning of hair on mons pubis and labial folds
Shorter, more narrow vagina
Left shift in maturation index
Flaccid breasts
Shortened urethra
Thinning of skin
Loss of bone density
Increased facial hair
Thinning of scalp hair
Change in fat to muscle ratio
The cervix also decreases in size, although to a lesser degree than the corpus, resulting in a ratio similar to that seen in adolescence. There is an inner migration of the squamocolumnar junction, making the diagnosis of cervical cancer more difficult. The cervical glands contract, resulting in a decrease in cervical mucus.
The postmenopausal vulva loses subcutaneous fat and elastic tissue, which results in a more narrow introitus. The labia majora flatten and wrinkle and shrink more than the labia minora, creating relative proportions similar to those of the prepubertal state. The clitoris decreases in size. The Bartholin glands produce less secretion, causing vaginal dryness and pruritus, and the hair on the mons pubis and labial folds becomes thinner and more coarse. Although the vulva has estrogen receptors, no change is observed after ERT. It is unknown whether estrogen, if instituted at the time of menopause, can prevent these changes.
Postmenopausally, the vagina becomes shorter and more narrow because of an increase in submucosal connective tissue. The rugae become less prominent. The fall in estrogen causes the maturation index to shift to the left, with a predominance of parabasal and intermediate cells. Eventually, the smear contains only small parabasal cells. Scant vaginal secretion and thin vaginal epithelium result in dryness and friability. The glycogen content of the vaginal epithelium decreases, as does the acidity of the secretions. Consequently, there is inhibition of lactobacilli with increased growth of other flora, increasing the susceptibility to irritation, trauma, and infection.
The glandular tissue of the breast atrophies, and Cooper's ligaments become less elastic, leading to flaccid breasts consisting primarily of fatty tissue. The nipples become smaller and may lose their erectile properties. The alveoli disappear, and the ducts decrease in size. In 10% of women, the breasts enlarge during the climacteric period, probably because of early unopposed estrogen and increased pituitary stimulation. They may remain enlarged and tender for some years after menopause.
The urethra and the base of the bladder are derived from müllerian tissue and are estrogen sensitive. Epithelial changes in the lining of the urethra and trigone of the bladder after the menopause are similar to those of the vagina and may result in atrophic cystitis, atrophic urethritis, urethral caruncles, urethral shortening, and stress incontinence. The urethral syndrome that results from the loss of bladder sphincter tone is characterized by recurrent bacterial urethritis with dysuria, frequency, urgency, nocturia, and postvoid dribbling. Estrogen replacement is helpful in treating all these symptoms. Between 10% and 15% of postmenopausal women older than 60 experience recurrent urinary tract infections. In a study of 93 women with this condition, a significant reduction in incidence occurred with the use of vaginal estriol cream.61 Pelvic relaxation, including cystocele, rectocele, or uterine prolapse, may develop in part because of the atrophic changes of the vagina and surrounding musculature. Estrogen receptors are not found in the area of the pelvic floor, which is composed of striated muscle; therefore, estrogen replacement is of no benefit for these conditions.
Hypoestrogenism accelerates aging of the skin. Estrogen can reverse some of these changes and has been shown to cause thickening of the epidermis.62 Estrogen receptors are present in the skin, and estrogen may be involved in collagen metabolism. After menopause, the epidermis thins, there is loss of epidermal ridges, the dermal papillae become less prominent, and the collagen and elastic tissue are reduced in the dermis. Other skin changes, including redistribution of fat deposits, loss of muscle tone, and loss of elastic tissue, are not thought to result from estrogen deprivation. There also are fewer hair follicles on the scalp and extremities and decreased production from sebaceous glands. All these changes lead to thin, dry skin with the development of creases and lines, frequent itching, and a tendency toward balding. Because of the state of relative hyperandrogenism occurring with menopause, increased facial hair and loss of scalp hair are common.
Bone Changes and Osteoporosis
Postmenopausal osteoporosis was first described in 1940.63 It is characterized by a progressive loss of bone mass without any change in the chemical composition of bone. The bone loss results in changes in the microscopic architecture of the bone that lead to an increase in fracture rate. Risk factors for osteoporosis include low bone mass at the onset of menopause, slender build, white or Asian race, premature ovarian failure, surgical menopause, a positive family history for osteoporosis, glucocorticoid therapy, and nulliparity (Table 4). Avoidable factors that predispose to osteoporosis include lack of physical exercise, insufficient calcium intake, cigarette smoking, excessive caffeine or alcohol intake, and estrogen deficiency. Premenopausal women with hyperprolactinemia or hypoestrogenic forms of amenorrhea are at increased risk of excessive bone loss because of the associated estrogen deficiency.64 Anorexia nervosa is associated with significant osteoporosis in young women.65 Factors that seem to be protective against osteoporosis include obesity, multiparity, muscular habitus, black race, and possibly the use of oral contraceptive agents for more than 1 year.
TABLE 4. Common Risk and Protective Factors for Osteoporosis
Unavoidable Risk Factors | Avoidable Risk Factors | Protective Factors |
Genetic predisposition | Lack of physical exercise | Obesity |
Low bone mass | Insufficient calcium intake | Multiparity |
Slender build | Smoking | Muscular habitus |
White or Asian race | Excessive alcohol intake | Black race |
Premature ovarian failure | Excessive caffeine intake | Prolonged oral contraceptive use |
Surgical menopause | Estrogen deficiency | Hormone replacement therapy |
Glucocorticoid therapy | Anorexia nervosa |
|
Nulliparity | Excessive exercise |
|
| Excessive thyroid re placement |
|
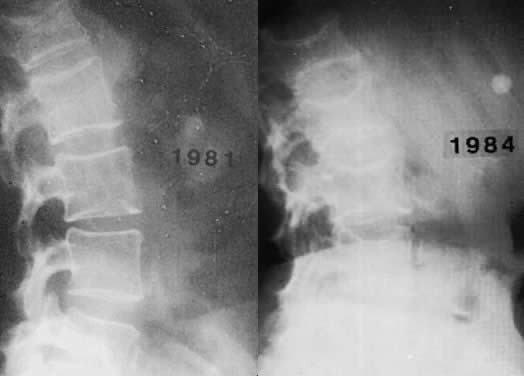
Osteoporosis is an insidious disease and often goes undetected until a painful compression fracture occurs. Only in hypoestrogenic female athletes and dancers may stress fractures be the first sign. Back pain is frequently the first complaint associated with osteoporosis; at first presentation, radiologic changes are usually not evident. A minimum of 25% to 30% of bone mineral content must be lost for osteoporosis to be diagnosed by routine radiographs (Fig. 3). Sudden severe pain may indicate vertebral collapse. The principal complications of osteoporosis include distal forearm fractures, vertebral compression fractures, and hip fractures. Distal forearm fractures (i.e. Colles' fractures) are the first to increase, with a 10-fold rise occurring in Caucasian women as they age from 35 to 60.
Spinal compression fractures, the most frequently occurring of the osteoporotic fractures, may cause pain, loss of height, and postural deformities. Such fractures are 10 times more common in white women than in men, occurring in 25% of Caucasian women over the age of 60. The average postmenopausal woman shrinks approximately 6.35 cm. Some loss of height is secondary to narrowing of the disc space. Small vertebral fractures are responsible for anterior collapse of the vertebral bodies, leading to dorsal kyphosis (i.e. dowager's hump).
Osteoporotic fractures of the femur occur later in life, usually at age 70 to 75. The incidence of hip fractures increases from 0.3 per 1000 to 20 per 1000 from age 45 to 85, with 20% to 25% of all women sustaining such a fracture by age 90. In 80% of the cases, osteoporosis is the cause. Fifteen percent of these women die within a 3-month period because of complications of the fracture, including pulmonary edema, myocardial infarction, and pulmonary embolism.66 Another 25% of these women suffer significant morbidity. Taking into account the significant morbidity, the direct and indirect costs of osteoporosis were estimated for 1995 to have been 13.8 billion dollars in the United States.67
Cortical bone predominates in the shaft of long bones, whereas trabecular bone is found primarily in the vertebrae, pelvis, and other flat bones, as well as the ends of long bones. Trabecular bone is more sensitive to factors that affect remodeling because it is metabolically more active than cortical bone. Vertebral fractures, Colles' fractures of the distal arm, and increased loss of teeth are seen with postmenopausal osteoporosis because the vertebral body, ultradistal radius, and mandible contain large amounts of trabecular bone. Trabecular bone loss begins at age 30 to 35, with a linear decrease of 1.2% per year. Although one series reported a similar rate of loss in postmenopausal and premenopausal women,68 most studies have shown a significant increase in bone loss after menopause. One study reported a 12% loss of density in the lumbar vertebrae 2 years after surgical oophorectomy.69 This accelerated phase of bone loss appeared to result from an increased rate of bone resorption relative to bone formation.70,71 This phase is more easily discernible in oophorectomized women, who have an abrupt loss of estrogen, than in women undergoing physiologic menopause, who have a gradual decline in estrogen levels.
The loss of bone mass correlates with the duration of estrogen deficiency. When women of a mean age of 50 who had been castrated 20 years before were compared with women of a mean age of 70 who had undergone menopause 20 years earlier, the bone mass was similar in both groups, with a 15% to 28% loss of radial bone.72 The actual estrogen levels, the metabolic clearance rates, and the rates of conversion of androgen to estrogen appear to be of no value in predicting which women will have a more rapid rate of bone loss.
Cortical bone loss begins at approximately age 40, at which time there is a slow rate of loss of 0.3% to 0.5% per year until menopause, when the rate increases to 2% to 3% per year. This rate continues for 8 to 10 years, at which point the slow loss rate is resumed.73–75 Consequently, there is a much greater loss of trabecular than cortical bone.
The belief that estrogen deficiency plays a major role in bone loss is strongly supported by the high incidence of osteoporosis seen in untreated oophorectomized women and women with gonadal dysgenesis. The rapid bone loss seen in untreated women is prevented with the use of estrogen replacement.76 Increased urinary excretion of calcium and hydroxyproline is associated with estrogen deficiency, presumably because of an increase in bone resorption (Table 5).77 ERT reduces the rate of excretion of these substances to the range observed in premenopausal women.
TABLE 5. Effects of Estrogen Decline on Bone Metabolism
Acceleration of bone loss
Decreased secretion of parathyroid hormone
Increased secretion of calcitonin
Decreased production of 1,25-dihydroxycholecalciferol
Decreased calcium absorption
Increased urinary excretion of calcium and hydroxyproline
Parathyroid hormone (PTH) causes an increase in serum calcium by increasing bone resorption, increasing tubal reabsorption of calcium in the kidney, and producing the enzyme 1α-hydroxylase, which is needed to make the active form of vitamin D necessary for calcium absorption from the gut. Premenopausally, estrogen is thought to block the action of PTH on bone resorption, possibly by increasing calcitonin levels. After menopause, estrogen levels fall. It is believed that the actions of PTH are then less inhibited, leading to an acceleration of bone loss. There is decreased secretion of PTH, increased secretion of calcitonin, and impaired 1α-hydroxylase activity, causing a decrease in the production of 1,25(OH)2 vitamin D and diminished calcium absorption from the gastrointestinal tract.
Estrogen receptors have been identified in osteoblastic cells in tissue culture.78,79 Although added estrogen increases secretion of alkaline phosphatase and α1-procollagen by such cells in vitro, it is not clear whether this is the mechanism by which estrogen affects bone loss. This is especially true in view of the diverse effects of estrogens on metabolic factors that can affect bone. Progesterone receptors are present in bone as well.80
Only 20% of castrated women and 30% of women undergoing natural menopause lose sufficient bone mass to result in pathologic fractures. It is difficult to predict which women are at risk for osteoporosis. A variety of radiologic methods have been used to assess bone mineralization, including radiogrammetry, linear photon absorptiometry, dual photon absorptiometry, dual energy x-ray absorptiometry, computed tomography, quantitative computed tomography, neutron activation analysis, and ultrasound. These tests are costly and sometimes inaccurate in the presence of arthritis, scoliosis, and compression fractures. The dual energy x-ray absorptiometer claims accuracy of 3% to 6% and precision of 0.5% to 1.2% while having the advantage of very low radiation exposure (1 to 3 mrem). The instrument can evaluate lumbar spine, hip, and wrist, as well as total body fat and calcium content. The World Health Organization has proposed guidelines for the diagnosis of osteoporosis when using bone mineral density testing (Table 6). Less expensive screening devices are being investigated.
TABLE 6. World Health Organization Criteria for Diagnosis of Osteoporosis
Category | T Score* |
Normal | Above -1.0 STD |
Osteopenia | -1.0 to -2.5 STD |
Osteoporosis | Below -2.5 STD |
*The T score represents the number of standard deviations (STD) from the peak, young, healthy bone mineral density of the reference group.
The diagnosis of osteoporosis is one of exclusion. The radiologic changes are late manifestations. There are usually only minimal alterations in laboratory values. Serum calcium levels are typically normal; phosphorus concentrations may be slightly low, normal, or slightly elevated; serum PTH and alkaline phosphatase levels are normal; and urinary excretion of calcium and hydroxyproline is usually increased. The ratio of urinary calcium to creatinine is elevated.81
Drugs for osteoporosis can be divided into two categories: prevention and treatment (Table 7). Estrogen, saloxifene, and alendronate have been approved by the U.S. Food and Drug Administration (FDA) for prevention and treatment. The relation between lack of estrogen and osteoporosis was described 50 years ago by Albright.63 ERT has been found to increase calcium absorption and decrease bone resorption.82
TABLE 7. FDA Approved Therapy for Osteoporosis
Indication | Therapy |
Prevention and treatment | Raloxifene |
Treatment only | Calcitonin |
Prevention and treatment | Hormone replacement therapy |
Prevention and treatment | Alendronate |
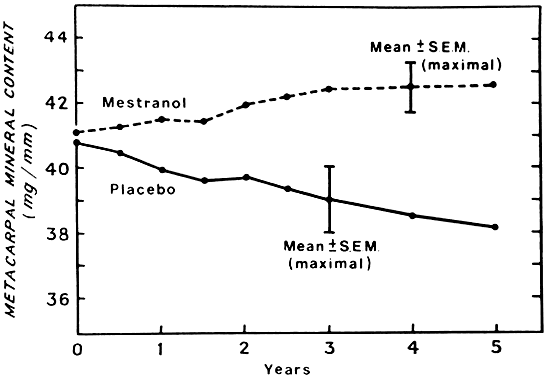
Estrogen does not appear to stimulate osteoblastic formation of new bone directly but may have a positive effect through insulin-like growth factor, transforming growth factor, and prostaglandins.83–85 Estrogen prevents bone mineral loss if begun within 2 months of surgical castration of premenopausal women or before physiologic menopause.76,86 If therapy is delayed 3 to 6 years after castration, there is still a response to estrogen therapy with an increase in bone density, which seems to be maintained for at least a 5-year period (Fig. 4).86 If estrogen therapy is stopped, bone loss resumes, with bone mineral content rapidly declining to the low levels of nontreated women. It may be necessary to continue ERT indefinitely to maintain the benefit. However, women who do not start ERT until age 60 may fare almost as well as the women who initiate ERT at menopause, provided they continue it long term.87,88 Estrogen replacement also decreases the incidence of fractures of the radius and hip.89–91 In one study, women taking exogenous estrogen had less than one half of the fractures of the placebo group, with the most apparent decline seen after 5 or more years of therapy.90 Estrogen use was found to protect against hip fractures in the subsequent 2 years in women younger than 65 and in those 65 to 74 years old.92 Various doses of the different estrogen preparations have been shown to be protective of bone mineral density (Table 8). Many of the bone density studies have been controlled, randomized trials. Fracture data, however, have been drawn primarily from observational studies.
TABLE 8. Doses of Estrogen Preparations that Prevent Bone Loss
Estrogen | Dose |
Conjugated estrogens | 0.3 mg |
Oral estradiol-17β | 0.5 mg |
Transdermal estradiol-17β | 0.025 mg |
0.3 mg |
The bisphosphonate alendronate has been approved by the FDA at a dose of 5 mg/day for prevention and 10 mg/day for treatment. An increase of approximately 9% at the spine and 7% at the hip was reported in the treatment group over a period of 3 years.93 Fracture data indicate an approximately 50% reduction in fractures of the spine and hip.94 Very few side effects are reported with this medication; however, an uncommon but serious risk of esophageal or gastric ulceration with potential hemorrhage has been reported. It is thought that strict adherence to dosing guidelines can reduce the incidence of this side effect. Patients must be willing and able to take the medication first thing in the morning on an empty stomach with a full glass of water. They need to remain upright sitting or standing and not consume any other food or beverage for a full 30 minutes. Patients with preexisting esophageal problems or significant hiatal hernias may not be good candidates for this medication. Other bisphosphonates are under investigation. As a class, they have very low absorption rates—hence the specific dosing instructions. In general, they have a very short serum half-life and a very long half-life (years) of adsorption to hydroxyapatite in the bone. Long-term effects on bone metabolism remain to be determined.
Nasal spray calcitonin has been approved by the FDA for the treatment of osteoporosis. It is recommended as a treatment option for the woman with osteoporosis who is more than 5 years past menopause and not using ERT. Estrogen has been shown to stimulate the release of calcitonin,95 and women with postmenopausal osteoporosis have been found to have diminished calcitonin levels.96 With use of a calcium clamp, calcitonin release was found to be blunted in women with osteoporosis compared with age-matched women without osteoporosis.97 Other techniques, however, have shown that calcium sometimes stimulates calcitonin release in patients with osteoporosis.98 Osteoporosis may or may not be associated with an alteration in calcitonin. Nevertheless, calcitonin has been shown to be effective in a dose-related fashion in suppressing bone loss.99,100 When calcitonin was given with calcium, a slight gain in bone mass was observed over 2 years, and the fracture rate was reduced by two thirds.101 Exercise may release calcitonin, explaining the positive effect of exercise on bone. Calcitonin acts to inhibit bone resorption by reducing the activity and the number of osteoclasts.102 Calcitonin results in increased levels of circulating β-endorphins, which may explain the analgesic effect reported by patients with osteoporosis treated with calcitonin. Calcitonin may decrease the bone pain associated with osteoporosis. Dosing for the nasal spray calcitonin is 200 units or one spray per nostril per day. The patient should alternate nostrils each day. An earlier version of calcitonin can be administered subcutaneously in doses ranging from 50 units every other day to 100 units daily.
Antibodies to calcitonin may develop and impede its usefulness. Several studies suggest that calcitonin may lose effectiveness after a period of time. Investigation is under way to determine if intermittent administration of the drug eliminates the development of resistance. Human calcitonin is less immunogenic than the salmon calcitonin. Anti-salmon calcitonin antibodies do not bind to human calcitonin in vitro. Patients with Paget's disease who have developed a resistance to salmon calcitonin respond favorably to treatment with human calcitonin,103 and this may prove to be a promising therapy for osteoporosis.
Raloxifene was approved by the FDA for the prevention and treatment of osteoporosis. Raloxifene is a benzothiophene and belongs to a class of drugs designated as selective estrogen receptor modulators (SERMs). Two other drugs in this category are clomiphene and tamoxifen, both triphenylethelenes. These drugs have estrogen agonist effects at some end-organ sites and estrogen antagonist effects at other sites and were previously known as mixed estrogen agonist-antagonists. Tamoxifen, for example, has antiestrogenic effects at the breast and reduces breast cancer recurrence for at least 5 years of treatment. It has mildly estrogenic effects on bone, lipids, and the uterus. It is not recommended for use for longer than 5 years because any protective effect on the recurrence rate for breast therapy appears lost after this time.104 Decreased bone loss has been observed in postmenopausal women using tamoxifen as treatment for breast cancer.105 There have also been reports of an increased incidence of cancer of the uterus in women using this drug.106,107
Raloxifene has a slightly different profile. It has mildly estrogenic effects on the bone and lipid profile with no observable effect on the breast or uterus. Unlike oral estrogens, raloxifene does not increase triglycerides. It also has no apparent benefit on the symptoms of hot flushes and vaginal dryness and may increase these symptoms in some women. In a 2-year trial, raloxifene produced a 1% to 2% increase in bone mineral density at the spine and hip compared with baseline and a 2.0% to 2.4% increase compared with the placebo group.108 Side effects include a 6% incidence of leg cramps and a threefold increase in venous thromboembolic events (VTEs), an increase similar to that reported with ERT. A history of VTEs is a contraindication to using raloxifene.
Sodium fluoride has been used for many years in the treatment of osteoporosis. The drug has many proponents and opponents. It appears to increase bone density substantially;109 however, it has lost favor since the publication of data indicating marked improvement in bone density with no improvement in fracture rate110 in women being treated with fluoride. A high frequency of side effects can be encountered with use of fluoride, including gastrointestinal problems, anemia, and joint and tendon inflammation. A slow-release fluoride formulation under investigation may provide better fracture protection with fewer side effects, but at present it is advisable to limit the use of fluoride to research settings until better data are available.
Most postmenopausal and castrated women are in negative calcium balance because of insufficient calcium intake. Those not on estrogen require approximately 1.5 g per day to remain in balance, whereas those on estrogen require 1 g per day.111 However, calcium supplementation alone does not seem effective in preventing osteoporosis. When high-dose calcium was given to menopausal women, it was no more effective in preventing trabecular bone loss than the average daily intake of 500 to 600 mg of calcium. Calcium does, however, seem to suppress cortical bone loss112–114 and reduce the risk of developing osteoporosis-related fractures.115 Although calcium supplementation does not appear to arrest trabecular bone loss in the early postmenopausal phase, it may have a more positive effect in the elderly. Women with a mean age of 84 who were given calcium and vitamin D supplements were found to have 43% fewer hip fractures.116 Other studies have reported a beneficial effect of calcium on bone loss in the older postmenopausal woman.117,118 Reports have indicated that there may be more vitamin D deficiency among the elderly than was suspected. Replacing vitamin D and calcium in the elderly can have significant benefits.116–118
Because there is no cure for osteoporosis, prevention of accelerated bone loss is of the greatest value. Adequate dietary intake of calcium and sufficient exercise should be encouraged throughout life. Avoidable risk factors for osteoporosis should be addressed (Table 4), and ERT therapy should be considered for those women who are eligible and deemed to be at risk for osteoporosis.
Cardiovascular and Lipid Changes
The hypoestrogenic state that occurs after menopause may be a significant factor in the development of ischemic heart disease. Premenopausal females are relatively protected against atherosclerosis and coronary heart disease compared with males the same age. Women rarely suffer heart attacks until after the menopause, when the risk of cardiovascular disease approximates that of men by age 65.119 This sex difference seems to be apparent only in affluent populations. Some investigators feel the decrease in the male to female ratio of death rates results solely from a decrease in male mortality with age. This hypothesis is supported by a study that showed a linear increase in the incidence of coronary heart disease in women between the ages of 30 and 90, with no apparent change in rate at menopause.120 However, an increase in the incidence of heart disease has been reported after castration in young women,121–123 with autopsy studies showing the presence of more coronary disease than in age-matched controls.124 A large, prospective study found bilateral oophorectomy but not natural menopause to increase the risk of coronary heart disease.125 This increase seemed to be prevented by ERT. In contrast, other studies have found no difference in the incidence of coronary disease in women with and without oophorectomy.126 Women with premature ovarian failure have also been shown to have an increased incidence of coronary heart disease 10 to 20 years after the cessation of menstruation, with significantly higher cholesterol and triglyceride levels than age-matched women who underwent physiologic menopause.127
Smokers have an earlier natural menopause by 1 to 2 years. When the risk factors of smoking and age were considered, the increased risk seemed to affect only women castrated and those not on ERT.128 Although hypertension is a significant factor in premature development of coronary disease, it presumably plays no role in normal women because no change in diastolic or systolic blood pressure is found with menopause.129
Cholesterol levels increase with age in men and women. Values greater than 250 ng/ml correlate with an increased risk of heart disease. However, after age 50, this association is not as strong. Serum cholesterol levels rise after natural or surgical menopause to values seen in men. It is unknown whether this change results from estrogen depletion. When castrated women were treated with 50 μg of ethinyl estradiol or 1.25 mg of conjugated estrogens daily, no change was seen in their cholesterol levels,130 but when 2.5 mg of conjugated estrogen was administered, cholesterol levels fell.131
Individuals with high low-density lipoprotein (LDL) cholesterol and low high-density lipoprotein (HDL) cholesterol are at increased risk for coronary heart disease. Elevated levels of HDL are found in people who exercise and those at low risk for atherosclerotic heart disease. At the time of menopause, there is a shift in the lipid pattern to one more similar to the pattern found in men, with increases in phospholipids, α-lipoproteins, and triglycerides.132 Menopause seems to have only a slight effect on HDL cholesterol levels. Although estrogen replacement cannot totally reverse this pattern, it does decrease LDL cholesterol concentrations, and raise HDL cholesterol concentrations.133 Natural estrogen increases the HDL2 and HDL3 fractions.134
The beneficial effect of oral estrogen administration on the lipid profile has been attributed to its first passage through the liver, and it is thought to be more beneficial in this regard than percutaneous application. However, percutaneous administration has been shown to produce similar changes of a lower magnitude, but a longer time is required to see this beneficial effect.135
Estrogen use in postmenopausal women has been reported to protect against coronary heart disease.91,136 When mortality rates from ischemic heart disease in postmenopausal women on estrogen were compared with those in living and deceased women not on estrogen, estrogen and excessive alcohol use were shown to decrease the chance of dying, whereas diabetes, hypertension, and smoking increased the risk of dying. Even women who smoked and took estrogen had decreased mortality.137 When mortality rates from all causes were evaluated in age-adjusted women, those on ERT had significantly lower rates than nonusers.138
There are studies that refute the reported beneficial effects of estrogen on coronary heart disease.139–141 In one study of incident acute myocardial infarctions occurring in a large managed care program, there was no statistically significant reduction in relative risk of myocardial infarction among current users of ERT.141 No definitive conclusions can be made regarding the beneficial or detrimental effects of estrogen on coronary disease. The preponderance of data, however, suggests that estrogen administration to postmenopausal women may reduce the incidence of death from cardiovascular disease by as much as one half; however, most of these reports were observational studies and not randomized or double-blind trials. The best data probably will come from the very large Women's Health Initiative Trial sponsored by the National Institutes of Health. Results may not be available until 2005 or later.
Data on the use of estrogen for secondary prevention of coronary heart disease (CHD) in women were published in 1998.142 This prospective randomized trial of 2,763 women with documented CHD followed the subjects an average of 4.1 years. The women were randomly assigned to a continuous regimen of 0.625 mg conjugated equine estrogens and 2.5 mg medroxyprogesterone acetate administered daily or placebo. At the end of four years there were no significant differences between the two groups with regard to the occurrence of angioplasty, coronary bypass procedures, myocardial infarction, or CHD death. The group on HRT had more surgical gallbladder disease and thromboembolic events. Prior observational studies had reported a significant benefit with HRT in the setting of secondary prevention of CHD. The results of this study underscore the importance of well-designed clinical trials in confirming or disputing the results of observational studies. The Women's Health Initiative will provide more data on this subject.
Estrogen also has an effect on liver function. The effects are influenced by the dose, the route of administration, and the type of estrogen used. The usual estrogen replacement dose, 0.625 mg of conjugated equine estrogen daily, is about one fifth as potent as one dose commonly used in low-dose oral contraceptives, 30 μg of ethinyl estradiol. The synthetic estrogens used in oral contraceptives, unlike the natural estrogens used for estrogen replacement, significantly increase globulins produced in the liver, including renin substrate (angiotensinogen), causing elevated blood pressure in some women.143 Natural estrogens also stimulate hepatic synthesis of renin substrate but to a much lesser extent. Blood pressure is unaffected by postmenopausal ERT,144,145 except for the 5% to 8% of such women who develop an idiosyncratic elevation.146 Several observational and prospective reports have indicated an increased risk of VTEs with use of ERT.147–152 The increase in relative risk was 2.0 to 3.6. The absolute risk is small because of the overall low incidence of these events (less than 1 in 10,000). However, because of the potential serious and life-threatening sequelae of VTEs caution should be exercised in assessing the risk to benefit ratio in individuals deemed to be at increased risk of VTEs.
Several small studies of different progestins suggest that the synthetic progestins commonly used together with estrogen for replacement therapy have an adverse effect on lipoprotein metabolism, causing a substantial increase in LDL cholesterol and a significant decrease in HDL cholesterol.153 In contrast, data from the prospective Postmenopausal Estrogen-Progestin Intervention Trial showed that micronized progesterone did not adversely affect the beneficial effects on lipids of conjugated equine estrogens.151 The effects of progestins on lipoproteins are greater with the 19-nortestosterone derivatives and appear to be dose dependent.154 During the phase of the treatment cycle when estrogen and progestin are given together, it appears that the beneficial effects of estrogen on lipoprotein cholesterol levels are negated at least in part.155–157 These findings have led to the common recommendation that progestogens, if prescribed, should be given in the lowest possible dose needed to achieve the desired histologic changes in the endometrium and should not be given to women who have undergone hysterectomy. The clinical significance of the effect of progestins on the lipid profile is not known. Early research on cardiovascular health and HRT focused on the beneficial effect of HRT on the lipid profile.
Estrogen interacts with the cardiovascular system in many ways. Documented effects of estrogen include a decrease in fibrinogen, lipoprotein a, plasminogen activator inhibitor, fasting glucose, collagen formation, and neointimal proliferation.158–160,151 Estrogen appears to enhance vasodilation and collateral vessel formation161 The effects of estrogens on lipids may be of little added benefit to their apparently beneficial cardiovascular effects. Several groups have attempted to estimate the effects of estrogen alone and estrogen given with a progestin to postmenopausal women for HRT.162,163 More studies are needed to define the benefits of estrogens on cardiovascular disease and to determine the optimal way in which estrogens and progestins should be administered.
Psychologic and Other Symptoms
Complaints of fatigue, nervousness, headache, insomnia, depression, irritability, joint and muscle pain, forgetfulness, inability to concentrate, dizziness, palpitations, and formication are also indicative of the menopausal syndrome. Many of these symptoms can be attributed to the sleep deprivation that results from frequent night sweats and may be avoided by taking estrogen. Postmenopausal women treated with estrogen replacement in one study had an improvement in their hot flush frequency and severity, insomnia, urinary frequency, vaginal dryness, frequency of headaches, mood, and memory. Treatment in postmenopausal women without flushes and night sweats improves only their vaginal atrophy, memory, and mood.164 In this same study, estrogen was not found to improve arthralgia, backache, or vaginal discomfort and had no effect on skin condition, coital satisfaction, or frequency of orgasm. It is unclear whether fatigue, irritability, anxiety, nervousness, and depression are caused by estrogen deficiency or another alteration at the central nervous system level. It has been suggested that estrogen is an inhibitor of monoamine oxidase, an enzyme whose levels are elevated in some depressed women.165
A decrease in the intensity and duration of the sexual response and a decline in libido is seen in some women after menopause. Most menopausal women remain sexually active, and estrogen replacement with the use of a vaginal lubricant has been helpful in relieving the dyspareunia associated with vaginal atrophy. One major reason for decreased sexual activity among older women is lack of an able partner.