Four oral diseases have been described as affecting pregnant women to a greater degree than their nonpregnant counterparts: gingivitis, pregnancy granuloma, periodontitis, and dental caries.
Gingivitis
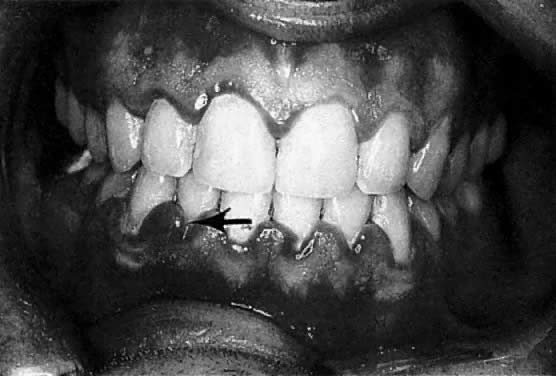
The frequently observed gingival changes that occur during pregnancy were reported as early as 1877.1 For many years, however, there have been questions about the reported prevalence of periodontal disease in pregnancy, the role that local and hormonal factors may have in the pathogenesis, and the implication of certain microorganisms in the etiology of this disease. Based on clinical observation, the reported frequency of so-called pregnancy gingivitis ranges from 35%2 to 100%3 (Fig. 1). This variation may be a reflection of both the populations studied and the clinical parameters used.
|
According to studies using well-defined indices, gingival inflammation is a heightened or exacerbated response to dental plaque during a period of progesterone and estrogen imbalance.4 In addition, the effect of pregnancy on pre-existing gingival inflammation is first noticeable in the second month of gestation and peaks in the eighth month (see Fig. 1). During the last month of gestation, a definite decrease in gingivitis generally occurs, and the gingival status immediately postpartum is found to be similar to that at the second month of pregnancy.3 The greatest relative increase in gingivitis during pregnancy is observed around the anterior teeth, although the molars demonstrate the highest gingivitis scores throughout pregnancy. The papillae (interproximal areas) are the most frequent sites of gingival inflammation both during pregnancy and after parturition.
ETIOLOGY.
The causes of gingivitis in pregnancy can be separated into two general headings: host factors and microbial changes. Relative to host factors, the onset of increased gingival inflammation observed in the second month of gestation coincides with an increase in the circulating levels of estrogen and progesterone. The continuous rise in these two hormone levels up to the eighth month is reflected in the greatest amount of gingival inflammation noted during pregnancy. In addition, a marked reduction in gingivitis after the eighth month correlates with an abrupt decrease of the circulating levels of these hormones. Estrogen and progesterone receptors have been demonstrated in human gingiva,5 indicating that it is a target tissue for hormones.6 Additionally, it has been demonstrated that progesterone is metabolized faster by inflamed human gingiva than by normal gingiva.7 The kinetics of progesterone in the gingiva, coupled with the clinical observations that the abnormal changes in gingiva during pregnancy parallel the circulatory levels of progesterone and estrogen, provide convincing evidence that these two hormones play a role in exacerbating gingivitis.
The mechanisms of action of progesteroneinduced and estrogen-induced gingival changes during pregnancy have become much better understood. Increased circulating levels of progesterone in pregnancy cause dilatation of gingival capillaries, increased capillary permeability, and gingival exudate. Vittek and colleagues8 described the effect of progesterone on the gingival vasculature and the resultant increased exudation. The effects included a direct activation of progesterone on the endothelial cells, possible effects on the synthesis of prostaglandins, and suppression of the cellular immune response.
Progesterone causes dramatic morphologic changes in the gingival microvasculature.9 The morphologic basis of the induced vascular permeability is the formation of gaps in the normally intact endothelial lining, together with channels resulting from coalescence of adjacent vesicles. The changes in both capillaries and venules, as well as the long duration of leakage from these vessels, are unlike the short action of histamine.
The keratinization of the gingiva is known to be decreased during pregnancy, and this, together with an increase in epithelial glycogen, results in a diminution in the effectiveness of the epithelial barrier.10 Estrogen also causes changes in the keratinization of the gingival epithelium and alters the degree of polymerization of ground substance.10 Because of the vascular changes caused by these hormones, there is a more florid response to the irritant effects of dental plaque.11 Increased serum levels of progesterone have been correlated with increased gingival crevicular fluid flow rate, which in periodontal diagnosis has been shown to reflect gingival inflammatory conditions.12
Physiologic levels of estrogen and progesterone in pregnancy have been shown to be stimulatory to prostaglandin synthesis.13,14 Prostaglandins, especially PGE1 and PGE2, act as long-term mediators of inflammation.15 Prostaglandins are synthesized by activated macrophages and, to a lesser degree, by polymorphonuclear neutrophils in response to inflammatory stimuli, both of which increase in number as the gingiva becomes inflamed.16 Prostaglandin concentration within the gingiva and gingival fluid also increases dramatically, with the occurrence of gingival inflammation.17 Along with initiation of vascular changes, stimulation of prostaglandin synthesis illustrates another mechanism that raises progesterone levels in pregnancy, magnifying the clinical features of dental plaque-induced gingivitis.
Immune mechanisms have also been suggested to have an important role in the initiation and development of gingivitis and periodontitis.18,19 Little is known about the effects of pregnancy on immune response in the oral cavity. Nevertheless, it has been demonstrated that the cell-mediated response is depressed during pregnancy, possibly contributing to the altered responsiveness of the gingival tissue to dental plaque.20
Dental plaque is the principal etiologic factor in gingivitis. In periodontal disease, In periodontal disease, it is well established that the subgingival plaque is characterized by a shift toward a more anaerobic flora. Strong evidence supports the observation that gingival inflammation during pregnancy results from an alteration of the subgingival flora to a more anaerobic state. The anaerobe-to-aerobe ratio increases significantly during the 13th through 60th week of pregnancy and remains high during the third trimester.21 It has been shown that increased proportions of Prevotella intermedia are concomitant with an increase in gingivitis and elevated serum levels of estrogen and progesterone in pregnancy.21 When the proportion of Bacteroides species were monitored in the dental plaque of pregnant women, nonpregnant women, and nonpregnant women taking contraceptives, a 55-fold increase over the control group was noted in the populations of the Bacteroides species in pregnant women and a 16-fold increase in women taking oral contraceptives.22 This concomitant increase in P. intermedia is most pronounced in the second trimester and correlates with increased gingivitis scores.22 Subsequent pure culture studies have shown that the marked increase in the proportion of Bacteroides species during pregnancy seems to be associated with increased serum levels of circulating progesterone and estrogens. Both hormones can substitute for naphthoquinone, which is an essential growth factor for P. intermedia. The studies reported to date indicate that female sex hormones may be capable of altering the gingival vascular system, the immune response, and the normal subgingival flora.
CLINICAL MANIFESTATIONS.
The marginal gingiva and interdental papillae are fiery red and the gingiva is enlarged, mostly affecting the interdental papillae. The gingiva shows an increased tendency to bleed, and in advanced cases, patients sometimes even experience slight pain. During the second and third trimester, the inflammation often becomes more severe.23,24 It should be noted that not all women respond in this fashion: in fact, many do not have a clinically altered gingival condition. When there is no dental plaque-associated gingivitis before pregnancy and attentive oral hygiene is monitored, gingivitis usually does not develop. Preventive measures, such as more frequent dental visits for prophylaxis and meticulous plaque control, are therefore indicated for pregnant women.
Pregnancy Granuloma
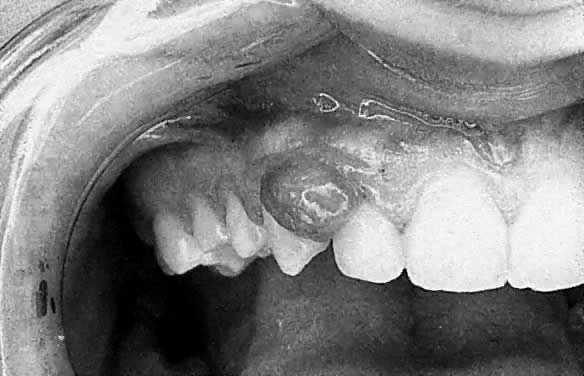
Apart from these generalized gingival changes, pregnancy may also give rise to the formation of tumor-like growths (epulides), along the gingival margin (Fig. 2). A number of terms for this lesion have been suggested, such as pregnancy tumor, epulis gravidarum, and pregnancy granuloma. Of these, pregnancy granuloma is preferred, because the histologic structure is similar to the structure in pyogenic (telangiectatic) granuloma.25 The reported frequency of pregnancy granulomas varies from 0% to 9.6%.26,27 The granuloma occurs more frequently in the maxilla, favoring the vestibular aspect of the anterior region.
|
There appears to be no correlation between the appearance of this lesion and the month of pregnancy, although patients may notice the lesion more often in the second or third trimester, owing to its growth or the complications of its vascular nature.28 The lesion often shows rapid growth, although it seldom becomes larger than approximately 2 cm in diameter. A pregnancy granuloma is generally a pedunculated, soft growth of interdental origin; is fiery red; and often has small, fibrin-covered areas (see Fig. 2). Pregnancy granulomas frequently bleed readily when touched and have a tendency to recur rapidly.
The cause of these lesions appears to be an accentuation of the inflammatory reaction of gingivitis in pregnancy, including the endocrine and bacterial changes described earlier. Additionally, the dramatic enlargement in these lesions may be partly related to the hormonal effect on collagen metabolism. The sequential changes in levels of estradiol and progesterone during pregnancy and immediately after parturition suggest that these hormones are important in the regulation of collagenolytic activity. Progesterone and methylhydroxyprogesterone inhibit collagenase production in the culture of postpartum uterine explants.29,30 The inhibition of collagenase production by these two hormones is apparently concentration dependent.
It is well known that endogenous gingival collagenase is the key enzyme involved in physiologic collagen turnover. During pregnancy, the inhibition of collagenase production ultimately results in accumulation of excess collagen within the connective tissue, thereby supplying a possible additional mechanism for the dramatic gingival enlargement of some pregnancy granulomas.
HISTOPATHOLOGY.
A pregnancy granuloma is composed of capillaries, fibrous tissue, and inflammatory cells, with marked vascularity being the most characteristic histologic feature. As such, distinguishing it from a pyogenic granuloma without other clinical data is difficult. The epithelium is generally thin and atrophic, but may be hyperplastic. If the lesion is ulcerated, it shows a fibrous exudate of varying thickness over the surface and a moderately intense infiltration of polymorphonuclear leukocytes, lymphocytes, and plasma cells. The excessive vascularity accounts for the bright red color, and the hyperemia and edema account for the enlargement.31,32
Differential Diagnosis.
The differential diagnosis of a small, pedunculated hemorrhagic lesion of the marginal gingival tissue must include the following:
- Peripheral fibroma
- Pyogenic granuloma
- Peripheral giant granuloma
- Eosinophilia granuloma
- Lymphomas or leukemic infiltrates
- Hemangiomas
TREATMENT.
It is prudent, if possible, to wait until parturition for surgical excision of a pregnancy granuloma, unless the lesion is creating a functional problem or appears to be having a deleterious effect on the adjacent periodontium. These lesions may regress after birth; however, surgical excision is usually warranted. The surgery can be accomplished safely throughout pregnancy with the use of local anesthesia and most effectively with the aid of lasers in place of scalpel blades. Lasers have the tendency to reduce the postsurgical bleeding typically experienced after excision of a pyogenic granuloma.33 Incomplete excision results in recurrence.27 A residual fibrous mass may remain if the lesion is large and is allowed to regress postpartum without surgical intervention.27
Periodontitis
Gingivitis, or inflammation of the gingiva, is considered to be a reversible process. In contrast, periodontitis results in the loss of tooth attachment (periodontal ligament and alveolar bone) and pocket formation. Though gingivitis is often associated with periodontitis, gingivitis does not usually develop into periodontitis because the putative pathogenic bacteria in periodontitis differ from those associated with gingivitis and because periodontitis is believed to be dependent on different immune mechanisms.
A number of investigators have noted sex hormone-mediated alteration of the subgingival flora and the subsequent increase in gingival inflammation.21,22,34 When pregnant and nonpregnant women with periodontitis are compared, however, the differences become less obvious. It has been shown that in contrast to subjects with gingivitis, no significant differences are noted in the total bacterial counts and the proportion of P. intermedia in periodontal pockets of pregnant versus nonpregnant women.34 Although differences exist in the degree of periodontitis between pregnant and nonpregnant female populations, these reported differences are not impressive.35,36,37 Therefore, conventional approaches for the prevention and treatment of periodontitis are indicated for pregnant patients.
Dental Caries
Many laypersons appear to believe that pregnancy is a direct cause of dental caries. The old wives' tale “with each child, a tooth” has been quoted even in dental and medical literature. In 1875 Coles wrote, “We have during pregnancy, an increasing liability to caries, with each generation.”38 He noted that during the first months of pregnancy, patients may have “severe toothache” secondary to caries. He explained this as “a diminution of earthy salts” during pregnancy. This belief has been fostered and has been one of the most stubborn misconceptions to appear in dental and medical literature. There is no scientifically proven evidence to support this belief.39
The hydroxyapatite crystal, of which enamel is made, does not respond to the biochemical and metabolic changes of pregnancy, nor does it respond to changes in calcium metabolism. The belief that morning sickness and vomiting can create an acid pH and therefore increase the decay rate is highly suspect as well. The few seconds that the pH of the oral environment may be lowered is a very brief period of time compared to the months needed for the production of decay.