Despite numerous clinical and epidemiologic studies, the etiology of placental abruption is yet to be precisely determined, but it is thought to be a disease of the decidua and uterine blood vessels. Several conditions continue to be associated with abruption. However in more than 40% of cases, no cause can be identified.19 Chronic factors include maternal vascular disease, chronic and pregnancy-induced hypertension (PIH), cigarette smoking, drug ingestion, nutritional deficiency, uterine anomalies and tumors, supine hypotension syndrome, antiphospholipid syndrome, congenital thrombophilias (including activated protein C resistance, deficiencies of protein C, protein S, and antithrombin III), hyperhomocystinemia, and, rarely, congenital hypofibrinogenemia.
Acute factors include maternal trauma, decompression of the overdistended uterus, and perhaps the acute vascular changes secondary to cocaine abuse. Amniocentesis has been a rare cause of abruption. Other factors such as a short umbilical cord and chorioamnionitis have been associated with abruptio placentae. Placental abruption also has been reported after the insertion of catheter tip intrauterine pressure transducers.20 Evidence and strength of associations linking some of these conditions with abruption are summarized later and in Table 1.
| Evidence | ||
| Risk Factors | Strength | RR or OR |
| Maternal age and parity | + | 1.1–3.7 |
| Cigarette smoking | ++ | 1.4–2.5 |
| Cocaine and drug use | +++ | 5.0–10.0 |
| Multiple gestations | ++ | 1.5–3.0 |
| Chronic hypertension | ++ | 1.8–5.1 |
| Mild and severe preeclampsia | ++ | 0.4–4.5 |
| Chronic hypertension + preeclampsia | +++ | 7.8 |
| Premature rupture of membranes | ++ | 1.8–5.1 |
| Oligohydramnios | + | 2.5–10.0 |
| Chorioamnionitis | ++ | 2.0–2.5 |
| Dietary/nutritional deficiency | +/− | 0.9–2.0 |
| Male fetus | +/− | 0.9–1.3 |
RR, relative risk; OR, odds ratio.
*These estimates are the ranges of RR or OR found in independent studies.
Maternal Age and Parity
Advanced maternal age and multiparity have been associated with an increased risk for placental abruption. However, they often are interrelated, and studies have produced inconsistent results. Although some studies have found women of advanced maternal age (but not parity) to be at an increased risk for abruption,21,22,23,24 others have demonstrated that parity plays an important role in the etiology of placental abruption.25,26 On the contrary, the U.S. Perinatal Collaborative Project performed from 1959 to 1966 and a population-based study failed to show a relationship between placental abruption and either maternal age or parity.27
Recently, Ananth and colleagues demonstrated by using population-based cohort data from Nova Scotia, Canada that young, multiparous women (aged 20–24 years and parity of 3 or higher) were at a 3.2-fold increased risk for abruptio placentae compared with nulliparous women aged 25 to 29 years.28 However, neither maternal age nor parity were independent risk factors for abruption. A larger, population-based retrospective cohort study in the United States found that the risk for abruption among women with singleton gestations was 1.85-fold higher among multigravid (3 or higher) women aged younger than 20 years, relative to primigravid women aged 25 to 29 years.16 In this same study, the risk for placental abruption among twin births was the highest among multigravid (3 or higher) women aged 35 to 49 years (relative risk 1.52).16 The implications of an age or parity effect on the increased incidence of abruption are not clear. For instance, whether the increased risk for abruption seen among young, multiparous women reflects a true biologic risk or is a reflection of other confounding factors is not clearly evident.
Maternal Vascular Disease
It has long been recognized that many patients with placental abruption also exhibit chronic hypertension or PIH. Although this is true for abruption overall, it appears that the more severe or higher grades have the strongest association with hypertension.23,29 Attention has focused on the arterial or afferent segment of the placental circulation to determine if pathologic changes could be attributed to hypertension. Williams previously had described degenerative changes in the intima of the small uterine arteries and postulated a relationship of preexisting toxemia.30 McKelvey's histologic examination of normal placentas at term showed degenerative lesions in the intima of the arterioles of the upper decidua basalis.31 Hertig demonstrated an acute degenerative arteriolitis with subsequent atheroma formation and fibroplastic deposition in the intima of the same vessels.32 He believed that these changes were pathognomonic of toxic separation; they were later deemed to be related to preexisting vascular disease. Subsequent investigation has shown that intimal changes of the spiral arterioles are more severe with hypertensive complications of pregnancy.
Some studies demonstrate an elevated risk of abruption among those with hypertensive diseases, with odds ratios ranging from 1.8 to 4.5,3,33 whereas others show inconclusive results.26,34,35 This discrepancy may be at least partly caused by inconsistent definitions of preeclampsia. In addition, with the exception of two recent studies,3,34 none of the earlier studies separated chronic hypertension and preeclampsia and other types of hypertension to determine the risk for abruption.
In summary, vascular disease plays a predisposing role in the disruption of the placental bed in some abruptions. Because hypertension has been shown to cause arteriolar degeneration, the compromised afferent blood supply to the placenta has been emphasized. In addition, superimposed arteriolar spasm would tend to aggravate the ischemia. Perhaps the entire entity may be a progressive stress or accelerated degeneration of an already defective vasculature of the placental bed.
Trauma
Maternal trauma, whether sustained during an automobile accident, from falls, or from physical abuse, is a significant cause of abruptio placentae. Most reports in the literature stress that abruptio placentae is the major pregnancy complication after blunt abdominal trauma and the leading cause of fetal death after automobile accidents associated with maternal survival.36 Maternal trauma accounts for 1% to 2% of severe or grade III abruptions. Likewise, abruptio placentae complicates 1% to 5% of minor injuries and 40% to 50% of major ones (life-threatening).36 Physicians should continue to emphasize the importance of lap and shoulder restraints for their pregnant patients traveling by automobile. Careful consideration of abruptio placentae should be undertaken when the history of an automobile accident or other maternal trauma is noted. The occurrence of abruption may not be able to be predicted based on the severity of abdominal trauma. Abruptions have occurred after seemingly minor maternal injuries. Therefore, all patients sustaining abdominal trauma, especially after a motor vehicle accident, should be monitored closely for signs of clinical abruption and fetomaternal hemorrhage, even if the trauma severity appears mild. A minimal amount of trauma can produce a mild abruption that can progress to severe over the course of 24 hours.37 Certainly, pregnant women involved in high-speed accidents should also be deemed at high risk for abruption regardless of presentation and/or restraint use and must require evaluation. A recent case-control study found that pregnant women involved in severe automobile accidents, i.e., higher speed (>30 mph) or injury severity score, were more likely to have abruptio placentae.36 Because the placenta does not contain elastic tissue, it cannot expand and contract. By contrast, because the uterus contains elastic tissue, it can react to acceleration–deceleration forces by changing its shape. Very high pressures can be generated in the uterus during abrupt deceleration, and high-pressure and marked distortion create a shearing effect on the placental attachment, which results in abruption.38
A complete workup should be performed in suspected cases, including a sufficient period of maternal and fetal observation, along with fetal heart rate monitoring. Recommendations include continuous monitoring for at least 24 hours, and even up to 48 hours, because of possible delayed abruption (seen up to 5 days after trauma).39 Those who should receive at least 24 hours of continuous fetal heart rate monitoring include patients with six or more contractions per hour, abdominal or uterine tenderness, vaginal bleeding, evidence of hypovolemia, or those with nonreassuring fetal heart rate patterns.40 In the absence of uterine activity along with a reassuring fetal heart rate tracing, the duration of monitoring may be less. In a pregnant trauma patient, the admission white blood cell count (WBC) may also be useful in assessment for placental abruption.41 The white count (WBC × 103/mm3) has been found to be significantly elevated in patients with abruption versus those without (27 ± 4.6 vs. 17 ± 7.8; p =.005), along with the band count (10 ± 6.6% vs. 4 ± 6.1%; p = .03). Therefore, a WBC more than 20,000/mm3 on admission should raise one's suspicion of the possibility of abruptio placentae. Conversely, a WBC of 20,000/mm3 or less may be associated with a very small, if any, risk for abruption, because the negative predictive value is 100%.41
Other traumatic etiologies of abruption include separation of the placenta in a twin gestation after delivery of the first twin with subsequent decompression, attempted external cephalic version, and drainage of amniotic fluid associated with polyhydramnios (in rare cases after amniocentesis for maturity studies). In the case of polyhydramnios, the abruption can occur both from the needle insertion and from the loss of fluid and uterine decompression. Rapid decompression should be avoided. A short umbilical cord (<20 cm) may be a factor in some abruptions. Excessive fetal traction on the placenta either before or during labor also can result in premature separation.
Supine Hypotensive Syndrome
Approximately 10% of pregnant women at term display a significant decrease in blood pressure in the supine position.1 This decreased peripheral pressure is presumed to be secondary to compression of the aorta and vena cava by the gravid uterus and to blockage of the uterine veins at the pelvic sidewalls. Supine hypotension generally is relieved by placing the patient in the lateral decubitus position. Isolated case reports suggest a relationship between placental abruption and the supine hypotensive syndrome. However, the absence of a higher incidence of abruption in patients with large infants or multiple gestations makes it unlikely that supine hypotension is of major clinical significance.
Cigarette Smoking, Dietary Deficiency, and Drug Use
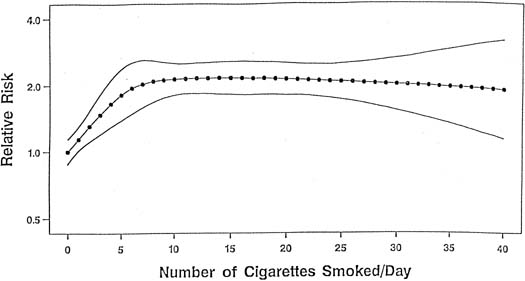
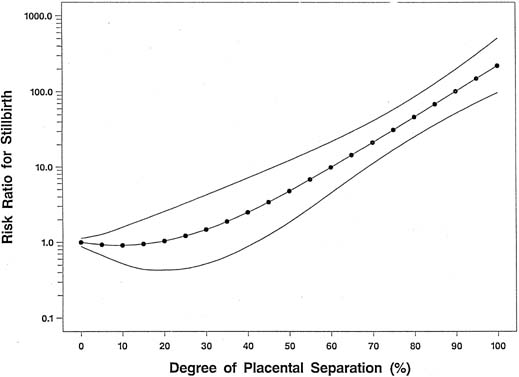
Although the mechanism linking tobacco smoke to the development of placental abruption still is unclear, several studies have found smoking to be a risk factor for abruption, whereas other studies show that the strength and magnitude of association between smoking and placental abruption were inconsistent. A recent meta-analysis, however, on the association between smoking during pregnancy and abruption, based on 1,385,352 pregnancies, indicates that smoking was associated with a 90% increase in the odds of developing abruption.42 This yielded a population attributable risk of 20%, indicating that 20% of abruption cases are potentially preventable if women quit smoking. These authors found a dose–response relationship between the average number of cigarettes smoked per day and the abruption risk. The relative risk increased in proportion to the increasing number of cigarettes smoked per day (Fig. 1). At 10 or more cigarettes per day, the relative risk remained fairly constant at 2.1, which suggests a possible “saturation” effect at 10 cigarettes.
Several potential mechanisms relating smoking to the development of placental abruption have been proposed. Based on pathologic examination, Spira and colleagues suggest that smoking leads to chronic hypoxia, which in turn produces placental enlargement, increasing the risk for placental abruption.43 Christianson also found placentas of smokers to be enlarged and to have lesions characteristic of underperfusion of the uterus.44 Others speculate that smoking may cause endothelial changes, which subsequently lead to vasoconstriction and rigidity of the arteriolar walls, resulting in placental underperfusion. This, in turn, may lead to ischemia of the decidua basalis, with eventual decidual necrosis and hemorrhage.45
A recent study also examines the relationship between smoking and preeclampsia and between smoking and preeclampsia superimposed on chronic hypertension.3 When compared with nonsmoking normotensive women, smokers with severe preeclampsia were at 5.9-times greater risk for abruption. Among smokers with preeclampsia superimposed on chronic hypertension, the relative risk for abruption was 7.8.
Other authors attribute an increased frequency of placental abruption to anemia or deficiencies of folic acid, vitamin A, or calcium.25 These deficiencies probably reflect the overall poor nutrition in women of a lower socioeconomic group, who are more prone to this complication. In addition, folate deficiency is related to hyperhomocystinemia, which itself has been found in women with placental abruption (see section on hyperhomocystinemia later).
During the 1980s, cocaine abuse reached epidemic proportions among Americans and still is widespread. Many case reports of pregnant women using cocaine, having transient episodes of hypertension, and subsequent placental abruptions with its associated complications, have been reported in the literature.46,47 Fetal deaths secondary to placental abruption have been reported in women abusing cocaine during pregnancy.48 A recent meta-analysis also confirms that cocaine use during pregnancy causes abruption,49 and another study involving more than 17,000 women found that the association existed even after adjustment for several confounding factors.50
Cocaine is an alkaloid prepared from the leaves of the coca plant.7,8 It can be administered intranasally;51 injected subcutaneously, intramuscularly, or intravenously; or administered orally, vaginally, sublingually, or rectally.52,53 In particular, a form of cocaine freebase called “rock” or “crack,” because of the popping sound made when the user heats it,54,55 has become extremely popular. Cocaine acts by blocking the presynaptic reuptake of the neurotransmitters norepinephrine and dopamine. Therefore, there is an excess of transmitter at the postsynaptic receptor sites,56 resulting in activation of the sympathic nervous system and producing vasoconstriction, tachycardia, an abrupt increase in blood pressure, and a predisposition to ventricular arrhythmia and seizures.56 Placental vasoconstriction also occurs,57 decreasing blood flow to the uteroplacental–fetal unit.58 The hypertension and vasoconstriction probably are responsible for the placental abruptions that are encountered in women soon after the intake of cocaine.58 Notice that cocaine abuse may be an etiologic factor in large and unusually early abruptions.47
Congenital Hypofibrinogenemia and Dysfibrinogenemia
Several case reports linking congenital hypofibrinogenemia and recurrent placental abruption appear in the obstetric literature. Ness and coworkers describe a patient who had a history of recurrent abruptio placentae and subsequently was found to have congenital hypofibrinogenemia.59 The patient had no bleeding problems in the nonpregnant state. Other members of the patient's family also displayed decreased fibrinogen levels. Pritchard has theorized that the hypercoagulable, hyperfibrinogenemic state of the normal pregnancy plays a role in maintaining placental attachment and controlling small placental separations and hemorrhage that often occur.29
A recent study has also described a patient having placental abruption in whom dysfibrinogenemia was subsequently diagnosed.60 It was discovered that her father and brother also had dysfibrinogenemia found through coagulation studies. This is an autosomal-dominant disorder that is caused by genetic mutations that create dysfunctional fibrinogen resistant to thrombin cleaving. These patients have greater risks for obstetric complications, such as spontaneous abortion and postpartum thrombosis, when compared with the general population.60
Hyperhomocystinemia and Other Congenital Thrombophilias
Homocysteine remethylation to the essential amino acid methionine depends on folate and vitamin B12. Enzyme deficiencies involved in homocysteine remethylation and folate and vitamin B12 metabolism, as well as malnutrition and malabsorption of these vitamins, may result in hyperhomocysteinemia.61 One enzyme involved in the remethylation of homocysteine to methionine is methylenetetrahydrofolate reductase (MTHFR), of which there are several documented mutations in this gene (C677T and A1298C). Hyperhomocystinemia induces endothelial cell injury and dysfunction and leads to premature arteriosclerosis and thromboembolism.62 Elevated homocysteine levels have also been shown to damage the vascular endothelium and result in placental vasculopathy.63 The relation between mild hyperhomocystinemia and complications in pregnancy leading to placental abruption has been demonstrated by several studies.64,65,66 One study found that of 84 women with a history of placental abruption or infarction, hyperhomocystinemia was found in 31%,64 whereas another study showed a 26% incidence.65 Owen and associates found hyperhomocystinemia in 29% of women with unexplained abruptio placentae.67 Interestingly, a recent study found that combined heterozygosity for MTHFR mutations C677T and A1298C was detected in 22.2% of their population of abruption cases.63 They suggest that combined heterozygosity for these two MTHFR mutations may represent a genetic marker for abruption. These authors also found that the A1298C mutation occurred significantly more frequently in mothers with abruptio placentae as compared with the control cohort. In addition, the combination of hyperhomocystinemia and thrombotic factors like activated protein C resistance, protein C, protein S, antithrombin, and factor V Leiden increases the risk for placental abruption three to seven times.68
Hyperhomocystinemia can be treated with a combination of folic acid and vitamin B6 (pyridoxine). Folic acid enhances remethylation of homocysteine to methionine and vitamin B6 is a cofactor for cystathionine-beta synthase activity, which converts homocysteine to cystathionine.65 It is not known whether normalization of hyperhomocystinemia by periconceptional folate administration favors pregnancy outcome in women with placental abruption or infarction.64 There seems to be no evidence of adverse effects of high folic acid levels or vitamin B6 levels on mothers during pregnancy that are caused by the supplementation dosages.
Other maternal congenital thrombophilias have been found in patients with abruptio placentae, such as protein S and protein C deficiencies,65 along with antithrombin III deficiency. Also, resistance to activated protein C from factor V Leiden mutation has been found frequently (25%–30%) in patients with placental abruption,69,70 and the authors suggest that the occurrence of abruption should lead clinicians to seek this diagnosis.69 Pregnant patients with antiphospholipid antibody syndrome, which involves two antibodies (lupus anticoagulant and anticardiolipin antibody) that are believed to cause thrombosis, may experience not only recurrent pregnancy loss, preeclampsia, and intrauterine growth restriction but also placental abruption.71 In patients with placental abruption, deVries and coworkers report a 13% prevalence of anticardiolipin antibodies (versus 4% in the general population).65 A recent study has found that compared with a control group, the prothrombin gene mutation was significantly more prevalent (18.5%) in women with abruption (odds ratio of 5.8).72
Association With Prolonged Preterm Premature Rupture of the Membranes
Premature rupture of the membranes (PROM) is a common obstetric complication, occurring in approximately 10% to 15% of pregnancies.73,74 Previous studies suggest that women with prolonged preterm PROM are at an increased risk for having placental abruption.75,76,77,78 Nelson found a three-fold to eight-fold increased risk for abruption in patients who were followed-up expectantly after prolonged preterm PROM. In their case reports, patients had significantly decreased amniotic fluid.79 Vintzileos and associates retrospectively studied 298 patients with prolonged PROM and noted those with severe oligohydramnios (<1 cm) to be at particular risk for abruptio placentae.78
Results of a recent meta-analysis based on pooled data from four studies indicate that PROM is associated with a three-fold increased risk for abruption.80 The meta-analysis, however, revealed widespread disagreement among study results. Methodologic inconsistencies among these studies in the definition of both preterm PROM and placental abruption may have contributed to this disagreement. For instance, two studies defined preterm PROM as ruptured membranes not followed by the initiation of labor or delivery within 24 hours and that resulted in preterm delivery (<37 weeks of gestation).76,78 In other studies, however, the definition of preterm PROM was not reported. The clear lack of consensus with preterm PROM on the definition of minimum latency time (between membrane rupture and the onset of labor) makes comparison of study results difficult.
There appears to be several plausible biologic mechanisms that may be responsible for the increased risk for placental abruption in relation to preterm PROM. Vintzileos and colleagues conclude that infections do not seem to be implicated in the mechanism by which preterm PROM predisposes to the development of abruptio placentae.74 Nelson and associates speculated that an acute reduction in the uterine volume and intrauterine surface area as a consequence of preterm PROM could ultimately lead to a disruption in the site of placental attachment, thereby resulting in abruption.79
Association With Elevated MSAFP (Maternal Serum Alpha-Fetoprotein) Levels
A relationship has been found between unexplained elevated MSAFP levels in the second trimester and pregnancy complications such as placental abruption.81 One group found that in such women presenting with preterm labor, there was an increased relative risk for abruption.82 Bartha and associates measured MSAFP at the time of admission in women presenting with preterm labor and intact membranes.83 They found elevated levels of MSAFP in two thirds of patients subsequently found to have placental abruption.
Other Etiologic Factors
Other possible associations that have been noted in the literature include abruptio placentae after snake bite,84 nipple stimulation,85 and prostaglandin E2 use.86 Uterine fibroids, especially when located behind the implantation site of the placenta, also may predispose to abruption.