Median Episiotomy
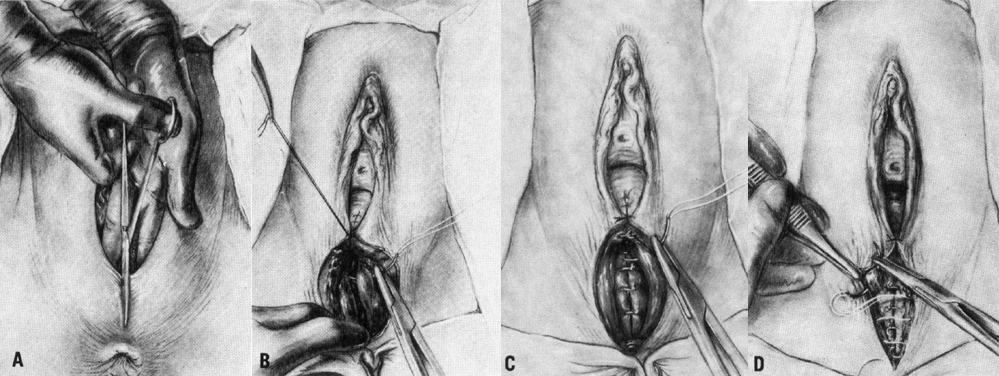
The median episiotomy incision is made in the perineal body from the midline of the hymenal ring through the connective tissue that unites the bulbocavernous muscle, the superficial transverse perineal muscles, and the perineal membrane (urogenital diaphragm).25 The incision is made down to but not including the anal sphincter (Fig. 1A). The vagina should be incised 3 to 4 cm above the hymenal ring, with the incision entering the rectovaginal space. The risk of vaginal lacerations is thus avoided. Scissors or a scalpal is used with care to avoid injury to the fetus.
The depth of the incision is limited to the distance between the vagina and the anal sphincter and thus poses restrictions on the amount of enlargement of the birth canal. If more room is needed, extension into the rectum either spontaneously or intentionally is inevitable. Intentional extension of the incision to involve the anorectal area is referred to as an episioproctotomy. It is not surprising that a median episiotomy is not elected when the perineal body is short or when the infant is thought to be very large. Opinion varies about whether a mediolateral incision is preferable to episioproctotomy. Episioproctotomy carries the risk of fistula and anal incontinence, whereas a mediolateral episiotomy causes greater blood loss and may be more painful. Mediolateral episiotomy, because it can be extended to incise the levator ani (which episioproctotomy does not), provides more room for delivering impacted shoulders or for managing breech delivery.
Repair of the median incision is often deferred until the placenta has delivered and inspection of the cervix and vaginal canal has been performed. Such delay provides adequate exposure for repair of vaginal and cervical lacerations, if present, and manual removal of the placenta, if necessary. Midline episiotomies may bleed briskly at the time of the incision, but after delivery of the fetus there is remarkably less bleeding. This has been believed to be due to a change in the venous congestion of the perineal tissues at the time of delivery.1
Many methods may be used to repair a midline episiotomy, but all require meticulous surgical technique. Richardson and colleagues26 found less perineal soreness after episiotomy repair using careful surgical technique and fine suture material that excites a minimal tissue reaction. They observed the amount of time elapsed before women resumed sexual activity, which was interpreted as a sign of relative perineal comfort. Richardson and associates found the size of the needle and fineness of the suture to be more important than the type of suture material used. However, a 1987 review of suture material used in episiotomy repair found that polyglycolic acid suture was associated with less perineal pain than chromic suture in several of those studies reviewed. Polyglycolic acid suture causes less tissue reaction and provides greater tensile strength than chromic or plain catgut. However, in only three of the studies do the confidence intervals reflect a real effect rather than one of chance.27
The perineum should be reconstructed in layers after adequate anesthesia has been obtained. Whatever the method of repair, the following principles seem evident. Take care to explore the entire extent of the episiotomy to avoid fistulas that can be created by an incomplete repair. Avoid causing ischemia and trauma to the tissues and restore to the midline all of the tissues that have been separated. The vaginal wall is closed first with a continuous suture that starts 1 cm above the apex of the incision, including any retracted blood vessels, which may otherwise result in hematoma formation. The closure continues to the hymenal ring. Each bite should include the rectovaginal fascia so that posterior vaginal support is maintained.25 In some instances, it is preferable to close the vaginal fascia as a separate layer before closing the vaginal mucosa. The connective tissue of the perineal body is then approximated by first using deep interrupted stitches that include the levator fascia (Fig. 1B). Next, the more superficial tissues in the region of the cut edges of the perineal membrane (urogenital diaphragm) and the transverse perineal muscles are approximated (Fig. 1C). This is done either as a continuation of the vaginal suture or as interrupted sutures. The skin is then closed with a continuous subcuticular stitch (Fig. 1D).
Sutures must not be placed so tight that they interfere with tissue vascularity, and dead space should be obliterated. Avoid placing sutures in the mucocutaneous portion of the fourchette to help prevent postpartum dyspareunia. The number of knots should be reduced as much as possible on the skin surface and in muscle layers. Careful approximation of the incised tissues layer by layer is required to ensure proper healing. Sutures should be placed perpendicular to the incision line to ensure adequate approximation. Occasionally, when the incision or laceration is curved, approximation can still be achieved by placing the sutures at unequal distances on one side.
When the repair is completed, the vaginal and rectal mucosa is palpated to ensure that repair is adequate, that no suture material extends through the rectal mucosa, and no sponge is left in the vagina. The perineum is rinsed, and an ice pack is applied.
Mediolateral Episiotomy
The type of episiotomy used depends on the judgment of the physician and often the region of practice. Median incisions are believed to be less painful than mediolateral, but when properly repaired there is little difference. Mediolateral incisions are only rarely extended into the rectum and anal sphincter and are often used when more room is required for the delivery process.
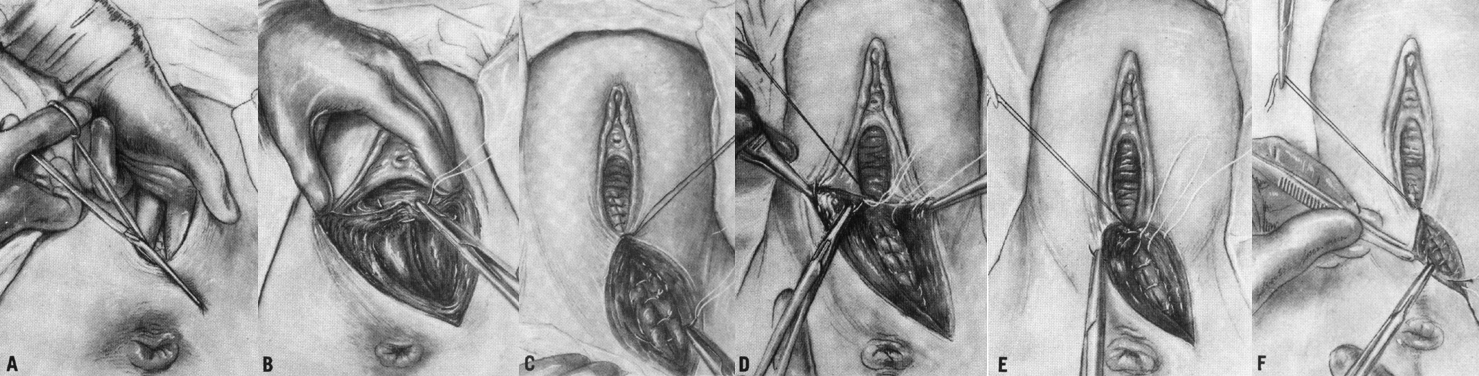
The direction of an episiotomy is dependent on the handedness of the surgeon. A right-handed obstetrician usually incises from the posterior fourchette at the midline toward the patient's left ischial tuberosity (Fig. 2A). The same structures are separated as with the median incision, and the ischiorectal fossa is exposed. In addition, when extra room is needed for a difficult delivery, a mediolateral incision has the advantage that it can be extended through the levator ani muscles, expanding the outlet. This additional room is not available with a median incision, which when extended cannot alleviate resistance from these muscles and their fascia.
It is important to begin the incision at the midline. If the episiotomy is begun in a lateral position on the vaginal outlet, the Bartholin's duct may be incised; at least theoretically, this error could lead to subsequent cyst formations.1 Quilligan and Zuspan24 advise that the mediolateral episiotomy be performed as a two-step procedure. The first incision is made in the soft tissues of the fourchette and the vagina, followed by incision of the perineum, extending in the mediolateral direction. They believe that this allows for an optimal anatomical approximation at the time of repair.
Repair is similar to the midline repair. Blood loss can be greater, however, and for this reason repair often is begun before the placenta has been expressed. Before beginning the repair, the rectal mucosa should be palpated for any defects. If any defects are present, they require initial repair. The vaginal mucosa and underlying supportive tissues are repaired with a running locked suture. The initial stitch is placed 1 cm above the apex of the incision (Fig. 2B). The vaginal closure allows reapproximation of the hymenal ring and subsequent anatomical accuracy. When a deep incision has been made, the levators and deep tissues should be sutured first before the overlying mucosa is closed.
The deep tissues of the perineal body are closed with interrupted fine absorbable suture (Fig. 2C-E). Continuous attention to anatomical approximation is critical. More tissue will appear to be present laterally than medially. Attention made to close the dead space as well as to obtain hemostasis is important, as it is for the median closure. The skin is closed as with the median closure (Fig. 2F). The subcuticular closure is commonly used.1 Vaginal and rectal examination at the conclusion of the procedure is again important to ensure that no suture has been placed in the rectal mucosa and that the closure is adequate.