The first step in management is anticipation of shoulder dystocia, including recognition of the previously described antepartum and intrapartum risk factors. Every effort should be made to prepare a rapid, ingrained, and well-coordinated stepwise plan. When fetal macrosomia is suspected, the patient should be thoroughly counseled regarding the risks and benefits of a trial of vaginal delivery.37
At the time of delivery, if shoulder dystocia is a concern, some clinicians have empirically advocated immediately proceeding to delivery of the fetal shoulders to maintain the forward momentum of the fetus. Others support a short delay in delivery of the shoulders, arguing that the endogenous rotational mechanics of the second stage may spontaneously alleviate the obstruction.
Shoulder dystocia usually is heralded by the classic “turtle sign.” After the fetal head delivers, it retracts back onto the maternal perineum. As maternal efforts to expel the fetal body continue, the fetal shoulder becomes further impacted behind the symphysis pubis. If the physician is alone, immediate assistance should be recruited. Additional support from anesthesia, nursing, and pediatrics also should be recruited. The mother should be instructed to stop pushing while attempts are made to dislodge the impacted shoulder. Most cases of shoulder dystocia resolve within a few minutes, although it may seem longer.
McRoberts Maneuver
The McRoberts maneuver is recommended as the initial technique for disimpaction of the anterior shoulder. In a retrospective review of 236 cases of shoulder dystocia occurring between 1991 and 1994 at Los Angeles County-University of Southern California Medical Center, this maneuver alone alleviated 42% of cases.38 When combined with suprapubic pressure, the McRoberts maneuver results in resolution of 58% of cases.39 Trends toward lower rates of maternal and neonatal morbidity have been associated with the McRoberts maneuver.38 Objective testing also has shown that the McRoberts maneuver may reduce fetal shoulder extraction forces and brachial plexus stretching.40
The McRoberts maneuver involves flexion of the mother's legs onto her abdomen. An x-ray study of the McRoberts maneuver confirms that it is associated with an increase in the mean angle of inclination between the symphysis pubis and the sacral promontory. There also was a 24% decrease in the angle created by drawing a line bisecting the symphysis pubis relative to the horizontal. With the McRoberts maneuver, the angle created by a line bisecting the longitudinal axis of the fifth lumbar vertebra and the longitudinal axis of the upper sacrum also increased.41
The McRoberts maneuver does not change the actual dimensions of the maternal pelvis; instead, it presumably works by straightening the maternal sacrum relative to the lumbar spine, with consequent cephalic rotation of the symphysis pubis. This position is thought to also enhance passage of the posterior fetal shoulder over the sacrum and through the pelvic inlet, positioning the plane of the pelvic inlet to its maximal dimension perpendicular to the maximum maternal expulsive force. Limitations of the technique include the need for two assistants and the extra time required to elevate and flex the patient's legs. Potential difficulty also can be encountered in moving the very obese patient or the patient with a dense epidural motor blockade.
The need for additional maneuvers after the McRoberts maneuver has been associated with larger fetal birth weights, longer active phases, and longer second stages of labor.38 Simultaneous suprapubic pressure, applied either posteriorly or laterally, can displace the impacted shoulder into the oblique diameter and effect delivery. Because shoulder dystocia is considered to be a “bony dystocia,” cutting a proctoepisiotomy may not be necessary. If the McRoberts maneuver fails, however, enlarging the episiotomy may allow more room for the next proposed maneuver.
Fetal Manipulation
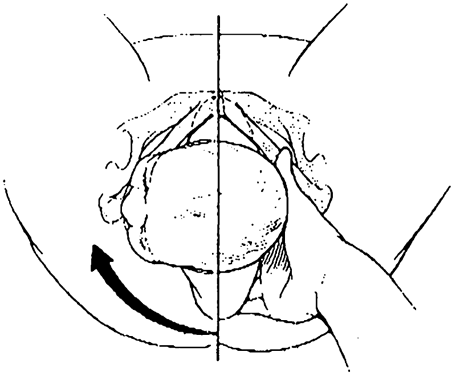
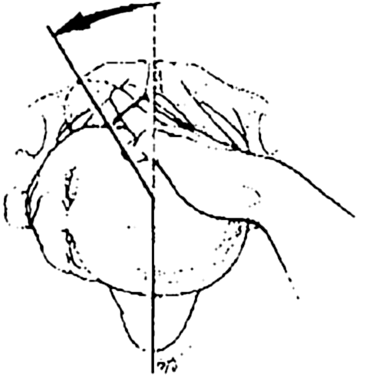
In the Woods (corkscrew) maneuver (Fig. 1), the practitioner pushes the posterior shoulder through a clockwise 180-degree arc by applying pressure on the anterior surface of the posterior shoulder. In the reverse Woods maneuver, or Rubin maneuver (Fig. 2), pressure is applied to the posterior surface of the anterior shoulder to effect counterclockwise rotation of the posterior shoulder.
|
|
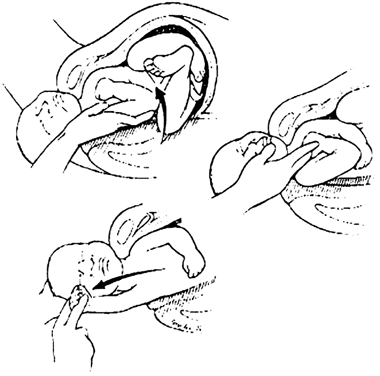
Should these maneuvers fail, the physician's hand can be passed into the vagina following the posterior arm to the elbow. After pressure is applied at the antecubital fossa to flex the fetal forearm, the arm is swept out over the infant's chest and delivered over the perineum (Fig. 3). Rotation of the fetal trunk to bring the posterior arm anteriorly may be required.
|
Zavanelli Maneuver, Symphysiotomy, and Hysterotomy
Before attempting the Zavanelli maneuver or a symphysiotomy, all of the previously described maneuvers it would be reasonable to reattempt with the patient under anesthesia. In the Zavanelli maneuver, the fetal head is rotated back to a direct occiput anterior position and then flexed. Constant firm pressure is used to push the head back into the vagina; a cesarean section subsequently is performed. Tocolytic agents or uterine-relaxing general anesthesia may be administered in preparation for and during the maneuver. Among 92 reported cases of partially born fetuses in vertex presentation, the Zavanelli maneuver successfully returned 78 fetuses into the vagina.42 Severe maternal complications of the Zavanelli maneuver include uterine infection requiring hysterectomy, vaginal “rupture,” laceration of the lower uterine segment, and uterine rupture.42
To perform a symphysiotomy, the patient should be placed in an exaggerated lithotomy position and have a Foley catheter placed to identify the urethra. With the physician's index and middle finger displacing the urethra laterally, the cephalad portion of the symphysis is incised with a scalpel blade. Because of the significant maternal morbidity associated with this procedure, it represents a last attempt to preserve fetal life and should be initiated within 5 to 6 minutes of delivery of the fetal head. Among three recently described cases, two patients had significant lower urinary tract complications and also required blood transfusion.43
For catastrophic cases unresponsive to the traditional maneuvers, performance of hysterotomy may be used to either primarily resolve the shoulder dystocia or assist with vaginal techniques.44 O'Shaughnessy describes delivery of the posterior fetal arm through a transverse uterine incision with subsequent passage of the hand through the vagina to an assistant. Posterior arm delivery then was completed vaginally while the abdominal surgeon applied pressure on the anterior fetal shoulder to allow rotation to the oblique pelvic diameter.45
Other Maneuvers
Initially described in 1976 by Gaskin, the “all-fours” maneuver consists of placing the gravid patient onto her hands and knees.46 In their analysis of 82 consecutive cases of shoulder dystocia, Bruner and coworkers noted that 68 patients (83%) had successful delivery of the fetus with this maneuver alone.47 Rates of maternal and neonatal morbidity were 1.2% and 4.9%, respectively, with a single case of maternal postpartum hemorrhage, one infant with a fractured humerus, and three neonates with low Apgar scores. The average time needed to assume the all-fours position and complete delivery was 2 to 3 minutes. The downward force of gravity or a favorable change in pelvic diameter produced by this maneuver may be the mechanisms that allow for disimpaction of the fetal shoulder.47
Fracture of the clavicle may be attempted by applying direct pressure away from the fetal lung. In reality, however, this is difficult to accomplish in the setting of shoulder dystocia.