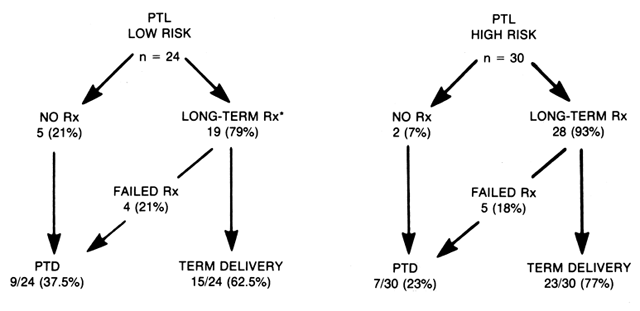
Efforts in the latter half of pregnancy continue to focus on the detection
and prevention of pre-term labor. Detection of IUGR or fetal anomalies
and the determination of the need, if any, of early pregnancy intervention
are new objectives. Foremost of these efforts should be the
early detection and prevention of preterm labor. A considerable effort
should be extended to detect more subtle warning signs. As mentioned
earlier, risk scoring for preterm labor is a tool that offers considerable
promise. Clinically obvious factors to consider are a history of
preterm labor or multiple gestation in the present pregnancy. Major medical/surgical
illness may also result in the delivery of a preterm neonate. Foremost
on this list are severe maternal hypertension, cardiac
disease, and renal disease. Even severe maternal anemia has been implicated.35 Although the above list includes items that are often beyond the control
of the managing clinician, they are nonetheless factors to be considered
in managing the patient. Clearly, any patient who has conceived while
taking ovulation inducing drugs should be evaluated for multiple
gestation. More remotely, multiple gestation should be considered when
conception occurs soon after discontinuance of oral contraceptives.36 Although the above theoretical considerations are interesting and may one
day impact on the prevention of preterm labor, a more pragmatic approach
is required. Should the patient be determined to be at risk for
preterm labor, the patient can be seen at frequent intervals after 20 weeks' gestation. At
these visits, it may be useful to perform routine
cervical examinations and record changes in cervical dilatation, length
in centimeters, and position, station of the presenting pan, and presence
and frequency of uterine contractions. Patient education regarding prophylactic measures, such as bed rest, and
warning signs of premature labor, such as low back pain, cramps, increased
vaginal discharge, or spotting, should be emphasized. It may not
be possible to prevent the onset of preterm labor; however, early detection
and consequent early employment of labor-inhibiting drugs will
undoubtedly have a beneficial effect in a considerable number of patients. Gestational Age Assignment Inaccurate assignment of gestational age is often the limiting variable
in high-risk pregnancy management. The incidence of patients with “suspect” dates
has been estimated to range between 22% and 40%;37, 38 a relatively high number of patients who claim to have a reliable menstrual
history may also fall into that category. It is obvious that failure
to accurately assign gestational age in patients considered for elective
repeat cesarean section or induction at term may result in iatrogenic
prematurity.39, 40, 41 In other circumstances, the impact on gestational age assignment errors
is more subtle and often not apparent unless obstetric versus pediatric
age42 comparisons are made and perinatal statistics by gestational age are carefully
evaluated. For instance, significant differences may exist in
neonatal morbidity and mortality between a fetus inaccurately estimated
to be 29 to 30 weeks' rather than 32 to 33 weeks' gestation. The benefit
of accurate fetal age assignment is particularly important in the
management of premature labor and premature rupture of the membranes
prior to 32 to 34 weeks. In other circumstances, the patient may be spared
the unnecessary apprehension attendant to an unnecessary diagnosis
of postterm pregnancy if a liberal policy of sonographic estimation
of gestational age is employed in early pregnancy. Such a policy may
be cost-effective if one balances the cost of ultra-sound against that
of unnecessary nonstress or stress testing, long inductions, and cesarean
sections performed on the indication, failure to progress in cases
falsely labeled postterm. Early detection of multiple gestation and
congenital anomalies also has obvious benefit. At the other extreme, early
confirmation of dates increases the credibility of routine intervention
at a “true” 42 weeks' gestation, especially if there
is associated oligohydramnios. Clinically, a history of regular menses with minimal variation in flow
duration or quantity (the last normal menstrual period, LMP) is reassuring
regarding the reliability of menstrual data. Close correlation of
fundal height with gestational age prior to 20 weeks, prospective maternal
recognition and documentation of fetal movement (quickening) at 18 to 19 weeks' menstrual
age, and physician detection of fetal heart
sounds at 18 to 20 weeks by fetoscope are useful supporting data. However, the
detection of fetal heart sounds as a measure of gestational age
has limited value unless the patient is seen weekly prior to the anticipated
time of detection; otherwise, the critical observation point
may pass unobserved. Further, total reliance on these clinical estimators
may be hazardous in the management of high-risk pregnancies in which
a difference of 7 to 14 days may be critical. Table 8 projects the interval required to ensure 90% certainly (10% uncertainty) that
an individual pregnancy is at least 38 weeks' gestation.43 It appears that one cannot make decisions that hinge on an accuracy of ± 1 week
even when a menstrual history is designated “reliable.” TABLE 8. Clinical Prediction of Maturity: Interval Necessary to Ensure 90% Confidence
of Pregnancy Being At Least 38 Weeks’ Gestation
Indicator | Interval (Weeks) |
Reliable LMP | 42 |
Unreliable LMP | 45 |
First fetal heart sounds | 21 |
Quickening, nulliparous | 25 |
Quickening, multiparous | 25 | The recent interest and availability of diagnostic ultrasound has dramatically
increased the clinician's ability to assess fetai gestational
age. Table 9 summarizes the predictive range and confidence limits of crown-rump length, biparietal
diameter (BPD), and femur length measurements at various
gestational ages. In normal gestation, the rate of growth of BPD or
femur length is probably a function of genetic predisposition; the variance
about the mean of both measurements increased with advancing gestational
age.44, 45, 46, 47, 48, 49, 50, 51 The increasing biologic variation of the BPD with advancing gestational
age is graphically displayed in Figure 5.46 Fetuses growing in upper percentlie limits have more rapid growth rates
in the third trimester than those growing in the lower percentile ranks. This
phenomenon must be considered in the interpretation of all sonographic
data. TABLE 9. Prediction of Gestational Age by Ultrasonic Determination of Crown-Rump
Length and Biparietal Diameter
| | Predictive | Confidence |
Author | Interval | Range | Limits |
Campbell | Second trimester | ±9 days | 84% |
Varma | Second trimester | ±9 days | 91% |
Sabbagha | 20 to 26 weeks | ±11 days | 90% |
| 27 to 28 weeks | ±14 days | 90% |
|  29 weeks 29 weeks
| ±21 days | 90% |
Sabbagha et al | GASA* | ±1 to 3 days | 95% |
Robinson, | Crown-rump | ±1 to 4 days | 95% |
Fleming | 7 to 14 weeks | | |
* Paired scans: first at 20 to 26 weeks; second at 31 to 33 weeks.
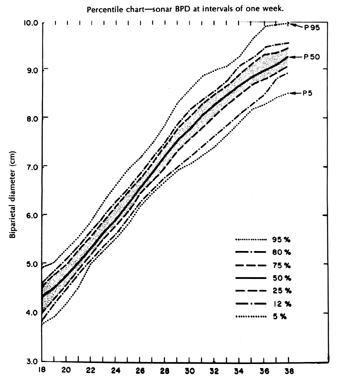 Fig. 5. Gestatonal age as X coordinate of graph depicting normal growth of the
biparietal diameter( BPD ); maximum separation of large versus small BPDs between 30 and 33 weeks’ gestation. (Sabbagha RE, Barton FB, Barton BA: Sonar leiparietal diameter: I. Analysis
of percentile growth differences in two normal populations using
some methodology. Am J Obstet Gynecol 126:497,1976) Fig. 5. Gestatonal age as X coordinate of graph depicting normal growth of the
biparietal diameter( BPD ); maximum separation of large versus small BPDs between 30 and 33 weeks’ gestation. (Sabbagha RE, Barton FB, Barton BA: Sonar leiparietal diameter: I. Analysis
of percentile growth differences in two normal populations using
some methodology. Am J Obstet Gynecol 126:497,1976)
|
Since there appears to be significant advantage to early application of
ultrasound, the clinician must develop a selective plan of management, which
includes ultrasonic estimation of gestational age prior to 26 weeks
in pregnancies at high risk for gestational age inaccuracy (Table 10), IUGR (Table 11), or prematurity (Table 12). Unfortunately, this scheme does not include all patients who will develop
high-risk characteristics in the third trimester. A number of complications
in which age assignment is critical, which may appear for
the first time in the third trimester, are listed in Table 13 Other useful indications for ultrasound are found in Table 14. TABLE 10. Indications for Routine Early Cephalometry: High Risk for Gestational
Age Assignment Inaccuracy
Irregular menses
Recent discontinuation of oral contraceptives
Fundal height versus LMP age discrepancy
Late appearance of fetal movement
Late appearance of fetal heart sounds
Obesity
Maternal age greater than 35 years
TABLE 11. High Risk for Altered Fetal Growth: Paired Scans at 20 to 26 and 31 to 33 Weeks (GASA) Plus Probable Serial Third-Trimester Scans
Prior SGA/intrauterine growth retardation newborn
First-trimester bleeding
First-trimester viral infection
Essential hypertension
Diabetes
Family history of hypertension/diabetes
TABLE 12. High Risk for Preterm Delivery: Indications for Routine Early
Cephalometry and Possible Second Scan at 31 to 33 Weeks
Candidate for elective repeat cesarean section
Candidate for elective induction
Candidate for “indicated induction”
Prior preterm labor
At risk for altered fetal growth
At risk for multiple gestation Family history
Ovulation induction
TABLE 13. Complications First Noted in the Third Trimester in which Gestational
Age or Fetal Growth Assessment Is Important
Premature labor
Premature rupture of membranes
Hydramnios/oligohydramnios
Poor maternal weight gain*
Poor fundal height growth*
Oligohydramnios*
Hypertension/preeclampsia*
* Fetal growth assessment is indicated.
TABLE 14. Ultrasound: Other Useful Indications
Confirm normal pregnancy Gestational sac (5–6 weeks)
Fetal viability (7th week)
Fetal echoes (8th week)
Prior to amniocentesis Locate placenta, umbilical cord, fetal structures
Confirm viability
Follow-up of abnormal α-fetoprotein
Detect fetal anomalies Cephalic
Spina bifida
Genitourinary
Limb reduction abnormalities
Direct fetal surgery
The ability of all single measurement sonographic techniques to predict
gestational age or assess growth is limited by both the inherent biologic
variation of the part (BPD, femur length, crown-rump length) measured
and the precision of the technique. In certain cases at extreme high
risk for uteroplacental insufficiency (UPI), it is thus important
that an attempt be made to determine the actual fetal growth curve and, where
possible, match it to a specific population growth percentile. This
is not possible in most instances, unless gestational age has been
reliably assigned on the basis of a basal body temperature (BBT) or
close correlation of menstrual history and physical findings. Thus, in
most cases, one is forced to assign gestational age by matching a single
BPD or femur length to a point on an established growth curve arbitrarily
at the 50th percentlie. Since biologic variation of the BPD is
most marked (see Table 9) after 26 weeks,46, 47 sonography probably should be used with extreme caution to assign gestational
age if the first scan cannot be accomplished prior to 26 to 28 weeks. Although
it is often acceptable to predict gestational age within
a ± 11-day interval, as may be accomplished by a single scan
prior to 27 weeks, management of many high-risk problems requires a
more accurate estimation. Fortunately, Sabbagha and co-workers, using serial cephalometry, has demonstrated
that approximately 90% of monkey52 and human53 fetuses maintain an apparently predetermined relative cephalic growth
pattern or ranking throughout pregnancy. On the basis of this concept, it
is possible to assign a growth-adjusted sonographic age (GASA), which
predicts gestational age within a ± 5-day (99% confidence limit) range; it
also allows division of cephalic growth curves into three
basic ranks: (1) BPDs greater than the 75th percentile; (2) BPDs between
the 25th and 75th percentile; and (3) BPDs less than the 25th percentile. Variation
in the external environment (UPI) may alter the projected
course somewhat. However, since cephalic growth is spared until
late in the course of most cases of placental insufficiency, this predictable
pattern is disrupted in only a few instances prior to 31 to 33 weeks. Early
onset of severe symmetrical growth retardation may be
an exception to this statement. In such instances, cephalic growth may
diminish prior to 31 weeks; as a consequence, fetal age may be overestimated
by 1 week, even employing GASA. The issue has minimal clinical
relevance, however, as the early failure of cephalic growth will persist
and become a prime issue. To some extent, the failure of cephalic
growth must then be managed independent of age. A similar error may develop
in approximately 5% of cases where there is significant early acceleration
of cephalic growth, which will result in underestimation of
gestational age. To obtain a GASA, the clinician must order two cephalic measurements, the
first at 18 to 26 weeks and the second at 31 to 33 weeks.48, 53 Should the slope of cephalic growth be in excess of the 75th percentile, the
original fetal BPD is assumed to have been large for gestation; in
such a case, the gestational age assigned at the time of the first
scan is reassigned to a gestational age consistent with a BPD at the 75th
percentile (i.e., earlier gestation) at the time of the original scan; to accomplish this, gestational
age is reduced up to 7 to 11 days. In contrast, the fetus
whose head growth slope is less than the 25th percentile is assumed
to have had a BPD at the 25th or lower percentile rather than the 50th
percentile at the time of the initial scan and thus the gestational
age is increased by 7 to l 1 days. The original assignment is maintained
if an average BPD growth slope (25th–75th percentile) is observed. In practice, we first make an assessment of the reliability of menstrual
age. If menstrual age is deemed unreliable, an early scan is ordered (i.e., at 18 weeks). If there is significant discrepancy (greater than 7 days) between
menstrual and sonographic age or the pregnancy is at high risk
for UPI, GASA is planned and early scheduling of a second scan accomplished. In
those cases complicated by preterm labor or preterm premature
rupture of membranes at 27 to 34 weeks, in which no early ultrasound
has been obtained, menstrual age is generally accepted if menstrual
data are felt to be reliable, particularly if the menstrual age falls
within the gestational age range predicted by sonography. In such cases, it
is common to observe a menstrual age that falls at the upper limit
of the gestational age range predicted by the late single scan. It should be obvious from the previous discussion that the system employed
even in perinatal centers with great resources is fraught with many
problems regarding prospective fetal age assignment. In the absence
of a future economic crisis, it is likely that early-pregnancy ultrasound
will become more routine. Certainly, more widespread use of real-time
ultrasound should facilitate a dramatic drop in current ultrasound
charges. This reduction in patient cost, combined with the obvious benefits
to pregnancy management, would include (1) early confirmation of
gestational age, (2) early diagnosis of multiple gestation, and (3) exclusion
of major congenital anomalies. If performed early in the first
trimester, secondary benefits might include early diagnosis of (1) blighted
ova (suspect if no gestational sac is noted after 8 weeks), (2) early
diagnosis of ectopic pregnancy, and (3) early diagnosis of hydatidiform
mole. Crown-rump length is the most accurate technique to assign gestational
age at 8 to 14 weeks.54, 55 There appears to be a transition in the reliability of measurement of
both crown-rump and BPD in the 12- to 14-week interval in that the fetus
is frequently more curled and a BPD may be more difficult to perform
because of the variation in skull calcifications. Femur lengths are
of no value in this interval. Perhaps the best interval to perform routine ultrasound would be in the 14- to 20-week
interval. In this interval, both the BPD44, 49 and the femur length50, 51 are very accurate in the assignment of gestational age. If routine sonography
is performed at 17 to 20 weeks, the clinician is also provided
with the most information regarding structural abnormalities of the cranial
ventricular system, spine, abdominal contents, bladder, limbs, and
abdominal wall. The determination of these abnormalities in this particular
interval is important in increasing the options for future management
of the patient. In rare circumstances, such as observed with
chromosomal or undiagnosed congenital anomalies, the fetus may be symmetrically
small, thus reducing the accuracy of both the fetal femur length
and the BPD. However, in most instances, particularly with unusual
head configuration or significant discrepancies between menstrual and
sonographic age, the femur length is useful in corroborating the date
assigned by BPD. Fetal Evaluation In the presence of stable maternal status, therapeutic decisions regarding
intervention in obstetrics should focus on the issue of benefit (reduced
fetal mortality) versus risk (morbidity and mortality of preterm
delivery). Ideally, intervention should occur at an instant in time
when the risk of intrauterine death (detected by some objective technique) outweighs
that of neonatal death, usually from respiratory distress
syndrome (RDS). Objective clinical evaluation of fetal health status
is a primary goal of obstetric care. Prior to the late 1960s, the management
of pregnancies thought to be associated with increased risk for
fetal mortality was empiric. Pregnancies were frequently terminated
by induction or cesarean section at a gestational age selected by evaluation
of published data comparing the risk of intrauterine death with
the risk of neonatal death at each week of gestation. For example, all
pregnancies complicated by diabetes were interrupted at 37 weeks, the
point in gestation at which the cumulative risk of intrauterine and
neonatal death was the lowest. Large numbers of otherwise normally developing
fetuses were thus delivered prior to full maturation. The benefit
was a reduction in fetal mortality in the subset of high-risk patients
with true UPI; the cost unfortunately was often unnecessary neonatal
morbidity (RDS) or mortality in patients at high risk but not having
true UPI. Management of pregnancy complicated by isoimmune disease
was another example. Inability to specifically assess the status of the
fetus at risk for erythroblastosis fetalis other than by historical
data and antibody titers often resulted in the unnecessary premature
delivery of an Rh-negative fetus in cases in which the father was heterozygous
for the D antigen. The development of amniocentesis to evaluate
the pregnancy at risk for isoimmune disease in the early 1960s revolutionized
obstetric practice and initiated interest in developing other
invasive and noninvasive techniques to selectively evaluate the status
of the individual fetus. Since that time, techniques to evaluate fetal
gestational age and growth, amniotic fluid content, endocrine products
of the fetoplacental unit, instantaneous fetal heart rate antenatally
or in labor, and fetal scalp blood samples have become available
to the practicing clinician. Most techniques if properly applied allow
the physician to delay intervention until maturity is attained. Proper
use of instantaneous fetal heart rate and scalp blood sampling should
promote more selective use of cesarean delivery for fetal distress. As
a result of the availability of new diagnostic tools and superb neonatal
care, the measure of clinical success in a practical sense is no
longer perinatal mortality, which has been reduced dramatically in many
centers, but rather perinatal morbidity. Decisions to intervene or not to intervene in a high-risk pregnancy usually
hinge on evaluation of maternal and fetal health status. In the vast
majority of instances in which intervention is necessary, it is for
fetal purposes; severe preeclampsia is a notable exception: maternal
status deterioration is a more common indication. In all cases, evaluation
of gestational age, lung maturity (also indirectly estimated by
gestational age), and integrity of the fetoplacental unit function are
key variables. Intervention prior to fetal lung maturity should occur
only in the presence of documented evidence of UPI or significant maternal
health deterioration. Ideally, intervention should occur when all
major organ systems, particularly lung and brain, are mature. As a bottom
line, it is emphasized that reassuring test results are more predictive
than nonreassuring ones. Fetal Growth Assessment IUGR may be a function of intrinsic or extrinsic (UPI) factors. Although
there are exceptions, IUGR of the intrinsic type tends to be symmetrically
small, that is, both cephalic and chest-abdominal measurements
are symmetrically two or more standard deviations below the mean for gestational
age. In contrast, the extrinsic IUGR tends to be asymmetrically
small, that is, cephalic head size is proportionately larger. At birth, asymmetrical fetal growth retardation is characterized by subcutaneous
and organ (especially liver) wasting in the presence of cephalic
sparing. Although head growth by population standards may be normal
in many cases, it is not known whether final cephalic growth is normal
compared with the potential established at conception. We do know, however, that
observed fetal growth deviates below that expected on the
basis of the observed tendency of the normal population to maintain
percentile rankings throughout gestation52, 53 In contrast, early and persistent symmetrical reduction in cephalic and
body size is commonly associated with viral (TORCH) infections, multiple
congenital anomalies, or significant chromosomal aberrations; many
are idiopathic. The prenatal diagnosis of IUGR is difficult. A high index of suspicion
is essential; even then the diagnosis may be missed. More frequently, the
prenatal diagnosis is not confirmed until delivery. A history of a
prior SGA newborn, maternal failure to gain 2 lb/month in the third trimester, and
markedly retarded fundal height growth in a patient with
established dates are commonly recognized clinical warning signs. Third-trimester
suspicion of oligohydramnios is a particularly significant
observation. Maternal vascular disease is the most commonly associated
clinical problem, accounting for approximately 20% to 30% of cases. Unfortunately, the
remaining 60% to 80% of cases are idiopathic (40%–60%) or
are associated with congenital infections (20%). RISK FACTORS FOR IUGR. Most risk-scoring systems pertain to risk for general perinatal morbidity
and mortality. Few systems address the specific issue of IUGR. Galbraith
and associates, for instance, have reviewed risk factors in 395 growth-retarded
newborns. In that population, 122 (31%) had no risk factors.56 However, it was noted that patients with “no prenatal risk” had
other prominent medical characteristics. Further, Galbraith's
group found that risk factors for IUGR are addirive. Significant risk
factors according to their time of presentation are shown in Table 15 (obstetrics historical risk factors), Table 16 (medical risk factors), and Table 17 (present pregnancy risk factors) for IUGR. They further found that the
incidence of IUGR in pregnancies complicated by multiple gestation and
preterm delivery was 21% and 11%, respectively. Evaluation of overall
perinatal mortality after excluding: births less than 500 g and those
complicated by congenital anomalies revealed a perinatal mortality rate
of 16.1 (123:7635) per 1000 in the 7635 non-IUGR newborns. In contrast, the
overall perinatal mortality in the IUGR group was 55.7 per 1000 in
the 395 IUGR infants. When IUGR newborns were separated into two
groups according to the presence of risk factors, it was observed that
those with no risk factors had a mortality rate of 32.8 per 1000 (four
deaths). This risk increased to 65.9 deaths per 1000 (18 deaths) in
the IUGR infants at risk. Thus, there is a fourfold increase in perinatal
mortality for the IUGR infant with risk factors as compared with
a normal newborn. In evaluating the effect of multiple risk factors, the
authors observe a progressive increase in incidence of IUGR from 2.3% in
the absence of risk factors to 14.0% with three or more risk factors. Patients
with no prenatal risk factors who subsequently delivered
IUGR infants were significantly different with regard to decreased maternal
weight, decreased maternal weight gain, increased smoking, higher
incidence of first pregnancies, and smaller fundal height at term. TABLE 15. Incidence of IUGR in Pregnancies With Risk Factors in Which There
Had Been a Past Gestational Complication*
| Total | IUGR | IUGR (%) |
IUGR | 384 | 77 | 20 |
Recurrent abortion | 91 | 10 | 11 |
Fetal death (stillbirths) | 106 | 10 | 9.4 |
Neonatal death | 64 | 6 | 9.4 |
Preterm | 260 | 23 | 9 |
Congenital anomaly | 109 | 8 | 8 |
* The factors considered were singly found.
(Galbraith RS, Karchmar EJ, Piercy WN et al: The clinical prediction of
intrauterine growth retardation. Am J Obstet Gynecol 133:281, 1979)TABLE 16. Incidence of IUGR in Pregnancies With a Maternal Medical Complication
| Total | IUGR | IUGR (%) |
Hypertension | | | |
Mild to moderate | 93 | 11 | 12.0 |
Severe | 9 | 4 | 44.0 |
Renal | | | |
Nephritis | 24 | 5 | 21.0 |
Other | 79 | 4 | 5.0 |
Urinary tract | | | |
infection | 495 | 39 | 8.0 |
Cardiopulmonary | 73 | 11 | 15.0 | (Galbraith RS, Karchmar EJ, Piercy WN et al: The clinical prediction of
intrauterine growth retardation. Am J Obstet Gynecol 133:281, 1979TABLE 17. Incidence of IUGR in Pregnancies With an Obstetric Complication
| Total | IUGR | IUGR (%) |
Preeclamptic toxemia | | | |
Mild to moderate | 363 | 38 | 10.5 |
Severe | 39 | 12 | 31 |
Antepartum hemorrhage | | | |
First trimester | 640 | 51 | 8 |
First trimester with | | | |
recurrence | 115 | 13 | 11 |
Second trimester | 250 | 19 | 8 |
Second trimester with | | | |
recurrence | 42 | 5 | 12 |
Third trimester | 378 | 41 | 11 | (Galbraith RS, Karchmar EJ, Piercy WN et al: The clinical prediction of
intrauterine growth retardation. Am J Obstet Gynecol 133:281, 1979)It is likely that more widespread application of early and serial ultrasound
as well as gross evaluation of amniotic fluid volume in the third
trimester will increase the likelihood of prenatal diagnosis. Although
ultrasound, particularly serial cephalometry, is helpful, it is by
no means perfect. We continue to more reliably predict a normal outcome
than the reverse. Good judgment and common sense must temper the interpretation
of available data. Factors that may limit the accuracy of
the prenatal diagnosis include limitations of precision between serial
determinations, variation in head shape, and the facts that clinical
problems may not become apparent until after 26 weeks' gestation, slowing
of cephalic growth may be a late manifestation of IUGR, and the pattern
of fetal growth may vary according to IUGR pattern type. If risk
factors for impaired fetal growth do not appear until the third trimester
and an early BPD measurement is not available, the clinician must
accept the ± 14- to 21-day confidence limits for a single scan
at 27 or more weeks’ gestation. Subsequent evaluation of cephalic
growth is thus limited. Since age is uncertain, growth evaluation is
limited to comparison to the mean population growth rate; growth should
average at least 2 mm/ week up to 34 weeks and at least 1 mm/week
measured over a 2- to 3-week interval even in the lowest percentile ranking. Because
of the slow growth rate and limitations in precision (1.0 mm-1.5 mm) of
serial measurements, we require three measurements over
a 3-week interval (days 0, 7 to 10. and 20 to 21) to assign a “no
growth” designation. Serial cephalometry, although helpful, has a false-abnormal diagnosis rate
of at least 28%; these cases with retarded cephalic growth but normal
birth weight are thought to be normal.57 In contrast, approximately 9% of cases have normal serial cephalic measurements
in the presence of birth weights below the normal range for
gestation. These false-normal cases probably represent cephalic sparing. Although
frequently designated normal in the prenatal period, they
are probably at greater risk than their normal counterparts for intrauterine
death secondary to UPI. It is thus obvious that these techniques
must be supplemented to increase the sensitivity and specificity of
the diagnosis. Reevaluation of Campbell's original series (Table 18), in which he was able to identify 68% of 114 SGAs on the basis of serial
cephalometry (but not abdominal circumference), suggests (on the basis
of LBW percentile and retarded cephalic growth) that approximately
three fourths (77: 105) of the SGAs will demonstrate symmetrical growth
retardation.57 Although this seems to be a high ratio of symmetrical IUGR, further evaluation
of assessment employing both cephalic and abdominal measurements
seems worthwhile. TABLE 18. Diagnosis of Small-for-Dates Fetus by Serial Ultrasonic Cephalometry
| Cephalic Growth Patterns | |
Weight | Normal | Borderline* | Retarded† | |
Group | (No./%) | (No./%) | (No./%) | Total |
AGA | 220 (83) | 18 (69) | 21 (18) | 259 |
BDRL SGA | 22 (8) | 4 (15) | 16 (14) | 42 |
SGA | ‡24 (9) | 4 (15) | 77 (68) | 105 |
Total | 266 (100) | 26 (100) | 114 (100) | 406 |
* Distribution of birth weights appears close to that of “normal” growth
rate category.
† Difference between birth weights of “normal” versus “retarded” categories is highly significant (P < 0.001).
‡ 9% false-normal secondary to cephalic sparing in a population
with asymmetrical retardation pattern.
(AGA, appropriate for GA; BDRL, borderline; from Campbell S, Dewhurst CJ: Diagnosis
of the small-for-dates fetus by serial ultrasonic cephalometry. Lancet 2:1002, 1971)Sabbagha has shown that GASA used alone allows the physician to compare
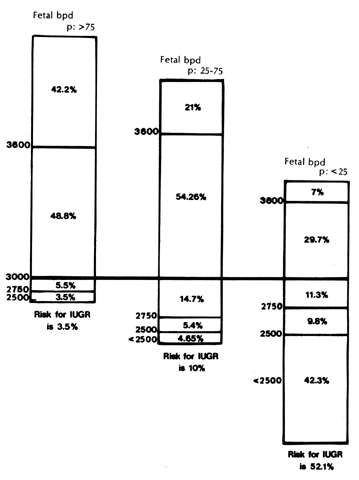
a predicted or potential growth to observed cephalic growth rather than
simply comparing observed growth to a population mean growth rate. In
addition, GASA facilitates prediction of birth weight (Fig. 6) and risk of IUGR.48,53 The likelihood of a newborn's birth weight being less than 2750 g
is 3.5%, 10%, and 52% for the greater than 75th, 25th to 75th, and less
than 25th GASA percentile rankings, respectively. It thus seems reasonable
to more vigorously evaluate the fetus in whom cephalic growth
is less than the 25th percentlie, or in whom there has been significant
deviation in growth subsequent to GASA assignment. Contrarily, it is
reasonable to be more concerned regarding macrosomia if BPD growth is
at greater than the 751h percentile, since 42% of such newborns will
have weights of at least 3600 g. GASA PERCENTILES AND ABDOMINAL CIRCUMFERENCE. Since ultrasound offers the possibility of determining symmetry of growth, it
is reasonable to selectively assess fetal growth pattern in cases
in which cephalic growth has been suboptimal (by GASA or serial third-trimester
scans). Measurements of the abdominal circumference at the
level of the ductus venosus is ideal, since it reflects fetal liver
and subcutaneous tissue volume, both of which are diminished in association
with IUGR.9 Comparison of the abdominal circumference and head circumference, although
time-consuming and costly, can modify future management in the high-risk
group by differentiating the symmetrically growth-retarded fetus
from the asymmetrically growth-retarded fetus. The initial data concerning the ratio of head circumference to abdomen
circumference were reported by Campbell37 Classically, the ratio of the cephaloabdominal circumference is greater
than 1:1 up to 35 to 36 weeks, after which the ratio is less 1. The
transition period may vary about the 35- to 37-week interval, depending
on the time of subcutaneous fat and soft tissue accumulation about the
fetal abdomen. Campbell and Thoms subsequently determined the mean
head-abdominal circumference ratio in 568 normal pregnancies.58 Simply employing the ratio, the authors were able to predict IUGR correctly
in 71% of fetuses; all detected cases were asymmetrically growth
retarded. Recently, Sabbagha has described nine fetal growth patterns that depend
on the relationship of cephalic versus abdominal circumference percentlies (Figure 7).59 The abdominal circumference percentiles are derived from the data of Tamura
and Sabbagha, which provide these percentile measurements (percentiles 2.5 to 97.5) from
the 18th to the 41 st week of pregnancy.59, 60 Growth patterns 3 and 6 reflect a symmetrical growth retardation with
cephalic sparing, whereas persistent slowing of both cephalic and abdominal
growth (pattern 9) two or more standard deviations below the mean
increases the risk for symmetrical retardation. Thus, lets use demonstrating
patterns 3 and 6 should be followed closely for UPI. Cases in
which pattern 9 is observed may require evaluation for viral infection, multiple
anomalies, or chromosomal aberration. In those instances in
which asymmetrical 1UGR is suspected, determination of total intrauterine
volume or relative amniotic fluid volume provides additional useful
information. 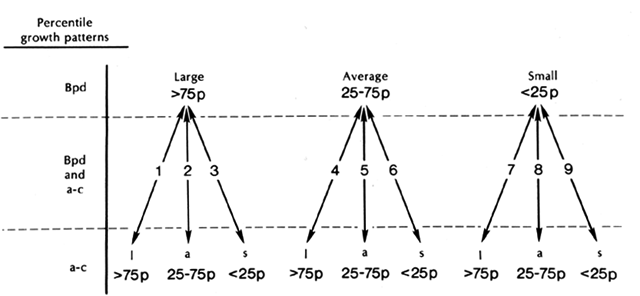 Fig. 7. Nine fetal growth patterns noted by using both biparietal diameter ( Bpd) and abdominal circumference ( a-c) percentiles ( p ). (Tamura RD, Sabbagha RE: Assessment of fetal weight. In Sabbagha RE (ed): Diagnostic
Ultrasound, Chap 9, p 99. Hagerstown, Harper & Row, 1980) Fig. 7. Nine fetal growth patterns noted by using both biparietal diameter ( Bpd) and abdominal circumference ( a-c) percentiles ( p ). (Tamura RD, Sabbagha RE: Assessment of fetal weight. In Sabbagha RE (ed): Diagnostic
Ultrasound, Chap 9, p 99. Hagerstown, Harper & Row, 1980)
|
In practice, we perform serial BPD measurements at (1) 18 to 24 weeks, (2) 31 to 33 weeks, and (3) 35 to 38 weeks. GASA is determined on the
basis of the first two scans, which also provide relative risk of IUGR. The
long interval between the first and second scan increases the likelihood
of detection of the small difference in growth of fetal head
size of fetuses with large versus small cephalic growth patterns.61 Performance of the second scan at 31 to 33 weeks has only minor limitations
in the detection of IUGR, since the majority of IUGR pregnancies
do not develop subnormal growth until after 32 weeks' gestation.61 The final reading at 35 to 38 weeks is used to evaluate the trend of growth
subsequent to attaining the abdominal circumference at the time
of the second scan. TOTAL INTRAUTERINE VOLUME (TIUV). Oligohydramnios accompanies and increases the risk of fetal death in many
cases of significant IUGR. As a result, uterine volume (fetal and
placental mass plus amniotic fluid) assessment, obtained by measuring
transverse and longitudinal diameters of the uterus, has been advocated.62 A value more than 1.5 standard deviations below the mean for gestational
age is highly suggestive of IUGR; a value of 1.0 to 1.5 standard deviations
below the mean for gestation represents a gray zone where the
diagnosis is less secure. Recently, clinicians have begun to assess amniotic
fluid volume more simply with real-time ultrasound. A pocket of
fluid greater than 1 cm in diameter is helpful in reducing the risk
of growth retardation. This more simplified approach reduces the necessity
to know the precise gestational age or to perform the calculations
required for TIUV measurements. Fetoplacental Function Evaluation Fetoplacental status may be assessed in the third trimester by biochemical (estriol
or placental lactose) or biophysical nonstress test, oxytocin
challenge test means. These subjects are eloquently discussed elsewhere
in these volumes and are mentioned in overview to reflect some
personal bias of the author. In most instances, the anticipated result
is patient and physician reassurance that pregnancy can be safely continued. The
overwhelming majority of reassuring results are highly specific
and are associated with favorable outcome (low false-normal rate); the
incidence of false-positive tests (no evidence of fetal or placental
disease during labor or at delivery following an abnormal test result) in
most instances ranges from 40% to 60%. Consideration to intervene
on the basis of a single abnormal result, particularly in the presence
of uncertain fetal pulmonary status, should be rare unless maternal
status deteriorates. Fetal assessment tests should be ordered in conditions at risk for UPI. By
design, they offer little or no useful information in the evaluation
of pregnancies with subsequent morbidity and mortality that are the
consequence of trauma, congenital anomalies, or cord accidents. Since
most assays/ procedures are expensive, the clinician should consider
if the test to be ordered actually provides useful information not detectable
by other means. Historically, biochemical assay, particularly
urinary estriols, has been commonly used. However, in recent years, biophysical
assessment of fetal placental respiratory function-nonstress
test (NST) or contraction stress test--tCST) or, more recently, of fetal
activity has largely replaced biochemical assays. In general, the
NST/CST is considered more reliable, can be performed at less frequent
intervals, and is less expensive than currently available biochemical
measures of fetoplacental function. OXYTOCIN CHALLENGE TEST. Since it is well known that uterine contractions are associated with a
reduction in uteroplacental blood flow, it is reasonable to employ spontaneous
or oxytocin-induced contractions with a frequency of three in 10 minutes
as a standard test of fetoplacental respiratory function.63, 64, 65, 66, 67, 68, 69 Stress of that magnitude has been proved clinically to be used in separating
the occasional fetus with suboptimal oxygen reserve from the vast
majority of cases with normal reserve.65, 66, 67 This stress does not significantly compromise the normal fetus with adequate
reserves. In contrast, a fetus can be presumed to have diminished “fetal
reserve” should repetitive late decelerations associated
with most contractions of any frequency be noted; the absence
of accelerations and baseline variability increases the specificity of
the diagnosis. In ordinary circumstances, an oxytocin challenge test (OCT) or
CST is initiated in pregnancies at high risk for UPI once the
diagnosis is suspected. Circumstances in which the CST or OCT is indicated
are as follows: Nonreactive NST
Diabetes mellitus
Preeclampsia
Chronic hypertension
Intrauterine growth retardation
Post-term pregnancy (42+ weeks)
History of previous stillbirth
Narcotic addiction
Sickle cell hemoglobinopathy
Chronic pulmonary disease
Organic heart disease
Rh isoimmune disease
Meconium-stained amniotic fluid
Testing may begin as early as 28 to 30 weeks in conditions such as early-onset
maternal pre-eclampsia (Figure 8). We routinely begin NST/CST testing at 30 weeks' gestation in class D
or greater diabetics. In contrast, we delay initiation of routine testing
until 32 weeks in class C, 34 weeks in class B, and 36 to 37 weeks
in uncomplicated class A diabetics.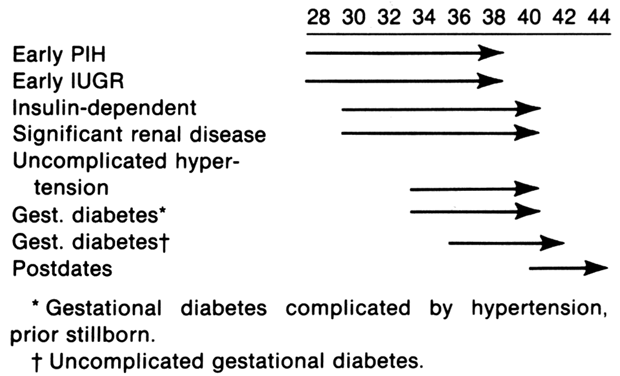 Fig. 8. Scheme for initiation of NST/CST testing according to weeks of gestation. ( PIH, pregnancy-induced hypertension) Fig. 8. Scheme for initiation of NST/CST testing according to weeks of gestation. ( PIH, pregnancy-induced hypertension)
|
In some instances, oxytocin is not required (spontaneous CST); in most
cases, however, it is necessary to induce uterine activity by administering
intravenous oxytocin using an oxytocin infusion pump. The fetal
heart rate is recorded for a baseline (nonstress) period of 15 to 20 minutes. If
three contractions per 10 minutes are observed within this
time interval, oxytocin need not be administered. If three contractions
are not observed, oxytocin is administered beginning at a 0.5 mu/min
interval. In most instances, it is not necessary to exceed 20 mu/min. The
CST (Figure 9) is negative (normal) if there are no late decelerations associated with
a contraction frequency of three in 10 minutes. Such a result is very
reassuring; it is associated with a 1 to 2:1000 false-normal rate. In
most instances, the procedure is repeated on a weekly basis. However, more
frequent testing may be indicated for insulin-dependent diabetics. patients
with moderate to severe preeclampsia, or postdate pregnancies, particularly
when associated with oligohydramnios. 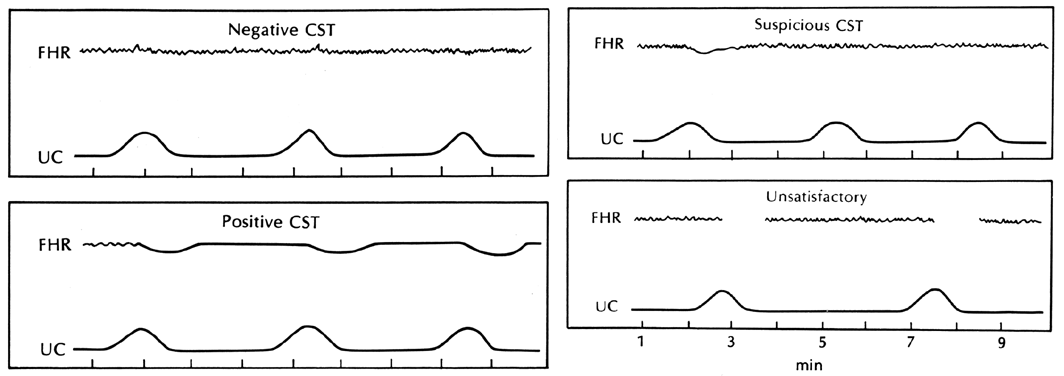 Fig. 9. Representation of possible contraction stress test ( CST) outcomes. The upper channel of each strip is the fetal heart rate ( FHR ); the lower channel reflects uterine contractions ( UC) in a 10-minute time interval. (Depp R: Assessment of fetal weight. In Sabbagha RE (ed): Diagnostic Ultrasound
Applied to Obstetrics and Gynecology. Hagerstown, Harper & Row, 1980) Fig. 9. Representation of possible contraction stress test ( CST) outcomes. The upper channel of each strip is the fetal heart rate ( FHR ); the lower channel reflects uterine contractions ( UC) in a 10-minute time interval. (Depp R: Assessment of fetal weight. In Sabbagha RE (ed): Diagnostic Ultrasound
Applied to Obstetrics and Gynecology. Hagerstown, Harper & Row, 1980)
|
The presence of consistent and persistent late decelerations with most
uterine contractions regardless of their frequency is diagnostic of a
positive CST (Figure 10). In many instances, this is associated with decreased variability and
absence of fetal heart rate accelerations with fetal movement. Since
a positive CST is an indication of UPI, delivery should be accomplished
within a relatively short interval if fetal pulmonary maturity is present. It
should be remembered, however, that approximately 24% to 60% of
such cases are actually false-positive (false-abnormal) when not preceded
by a nonreactive NST; if allowed to labor subsequently, such patients
will demonstrate no further late decelerations. For this reason, it
may be safe to attempt a vaginal delivery if it is possible to rupture
the membranes, apply a direct electrode, and allow the patient
to labor in a lateral decubitus position. In the absence of pulmonary
maturity, evaluation of fetal status can be supplemented by daily plasma
or urinary estriols; the pregnancy may be allowed to continue as long
as the estriol levels are stable or rising. Although a positive CST
result is less predictive (24%–60% false-positive) than a negative
one (99% true-negative), the certainty of diagnosis may be increased
by evaluating the fetal heart rate baseline for the presence or absence
of fetal heart rate accelerations with fetal movement.65 Most cases in which the positive OCT is accompanied by accelerations with
a magnitude of at least 15 beats per minute in response to fetal movement
will tolerate labor or can tolerate some additional time interval
to delivery. If accelerations are absent and baseline variability
is poor or absent, it is unlikely that the supplemental use of serial
estriols will be helpful in allowing the pregnancy to continue until maturity
is obtained and unlikely that labor will be tolerated without
further evidence of late decelerations. In those cases in which cesarean
is planned after a positive CST, it may be desirable to stop the oxytocin, begin
administration of oxygen, and allow time for intrauterine
recovery prior to delivery. 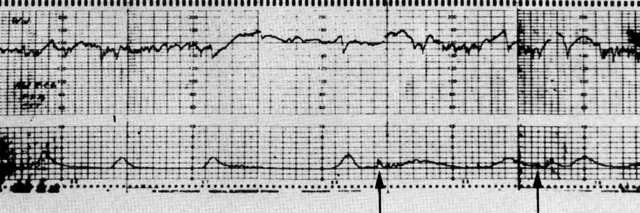 Fig. 10. Reactive (negative) result of contraction stress test using an abdominal
electrocardiogram as source. The lower channel reflects uterine contractions
of 40- to 60-second duration. Small superimposed peaks represent
fetal movement. Fetal heart rate (FHR) baseline (upper channel) is 145 to 150. Note
accelerations of FHR in response to fetal movement ( arrows) on lower channel. The estimated magnitude of accelerations is 25 to 30 beats/min. (Depp R: Dynamics of fetal growth. In Sabbagha RE (ed): Diagnostic Ultrasound
Applied to Obstetrics and Gynecology. Hagerstown, Harper & Row, 1980) Fig. 10. Reactive (negative) result of contraction stress test using an abdominal
electrocardiogram as source. The lower channel reflects uterine contractions
of 40- to 60-second duration. Small superimposed peaks represent
fetal movement. Fetal heart rate (FHR) baseline (upper channel) is 145 to 150. Note
accelerations of FHR in response to fetal movement ( arrows) on lower channel. The estimated magnitude of accelerations is 25 to 30 beats/min. (Depp R: Dynamics of fetal growth. In Sabbagha RE (ed): Diagnostic Ultrasound
Applied to Obstetrics and Gynecology. Hagerstown, Harper & Row, 1980)
|
NONSTRESS TEST. Although the CST/OCT is highly predictive with a very low false-negative
rate, the test is time-consuming, requires intravenous fluids and oxytocin, and
in most instances must be performed in a labor unit. As a
consequence, the procedure is costly as well as invasive. Early work
by Kubli and Rutgers70 and Hammacher71 established the characteristics of the fetal heart rate baseline in normal
and abnormal fetuses. At this point in the evolution of antepartum
fetal heart rate monitoring, it became obvious that it was rare to observe
repetitive late decelerations consistent with a positive CST/OCT
following an initial baseline observation period (prior to initiation
of oxytocin) characterized by significant accelerations of the fetal
heart rate in association with fetal movements. The observation of the
importance of the reactive nonstress portion of the CST thus paved the
way for development of the nonstress test (NST) as proposed in this
country by Lee72, 73, 74 and subsequently employed by Schifrin75 and Evertson76 and their colleagues and numerous other investigators (Figure 9). It is generally accepted that a reactive (negative or reassuring) NST is
characterized by the presence of two or more accelerations of the fetal
heart rate observed in 20 or less minutes; the accelerations must
be at least 15 beats per minute above the baseline. Apparently it does
not matter if the accelerations observed are associated with spontaneous
movement or in response to manual stimulation. If the fetus is not
reactive within the first 20 minutes, the fetus should be stimulated
artificially and observed for an additional 20 minutes before making the
designation “nonreactive.” The latter step minimizes the
possibility of lack of activity associated with fetal sleep cycles. A
non-reactive NST is characterized by accelerations of less than 15 beats
per minute or less than the normal CST. Like the normal CST, a reactive
NST is very reassuring. The false-negative (false-normal) rate
approximates that of a CST; most series report a false-negative rate of 1 to 3:1000. It
is currently thought that the unsuspected death is a
consequence of unforeseen cord accidents, such as tight nuthal cords, cord
knots, or “cord vulnerability” associated with oligohydramnios. Such
deaths are currently not preventable, as is probably
the case with sublethal insults resulting in cerebral palsy. There has been some concern that late intervention may occur if the clinician
delays until a nonreactive NST followed by a positive CST evolves. This
concern does not seem realistic for several reasons: (1) The
NST appears to be a very conservative indicator of fetal status in that
the nonreactive rate may be as high as 20% to 35% for the NST versus
only a 3% positive rate for the CST. This significant increase in the
population judged to be at special risk identifies a subset population
that may be better served by the CST with its associated lower false-abnormal
rates. (2) There appears to be a gradation of non-reactive results (decreasing
acceleration magnitude, more obvious loss of variability); clearly
the more fiat the base-line, the less favorable the outcome. Since
these changes are generally observed over time, a CST can
be ordered to evaluate fetal status at a very early time in development
of the nonreactive state. (3) The majority of false-positive CSTs are
characterized by a baseline that is reactive. (4) There is little if
any increase in perinatal mortality when comparing the patient with
a reactive NST and one with a nonreactive NST followed by a negative CST. (5) From
a practical standpoint, the diminished cost and the patient
convenience of the NST allow the clinician to assess certain high-risk
pregnancies more frequently and less expensively than is realistically
possible if the CST is used as the primary test procedure. In most cases, an NST is used as a screening test; reactive tests may safely
be repeated at weekly intervals; however, some investigators repeat
them more frequently in insulindependent diabetics, preeclamptics, and
patients at high risk for IUGR or postmaturity. Twenty percent or
more of tests are nonreactive; such cases require CST. Ninety percent
of subsequent CSTs will be negative. Most of the few who subsequently
have a positive CST have good outcome; deaths are associated with prematurity
or, occasionally, congenital anomalies. If there is a deficiency
in the NST, when compared to the CST, there may be a possible diminished
ability to detect variable decelerations, which may be detected
in up to 10% of CSTs but in a considerably smaller number of NSTs. The
etiology of these variable decelerations has been specifically determined. Primary
concerns would be the presence of a nuchal cord (currently
not detectable in the prenatal period) or vulnerable cord as a consequence
of oligohydramnios. Possible solutions to this potential deficiency
of the NST are (1) real-time scanning for adequacy of amniotic
fluid volume for patients with nonreactive NSTs or clinical suspicion
of diminished amniotic fluid volume on the basis of physical examination
or variable deceitrations noted on NST and (2) nipple stimulation to
induce uterine activity in all patients, to facilitate physiologically
induced uterine activity. The latter approach may or may not be effective, in
that uterine hypertonus and hyperactivity may be induced in
some patients, while in others the contractile activity generated may
not be equivalent to the traditional CST. Further research must be done
in this area. FETAL ACTIVITY. Assessment of presence or absence of fetal activity has been used on a
subjective basis by clinicians for many years.77 Recently, it has been reconfirmed that active fetal movements are an expression
of fetal well-being. A sudden increase (cord compression or
abruption) or a decrease (chronic UPI) in activity may precede fetal death. Some
have suggested that antepartum surveillance of patients with
decreased fetal movement would be more beneficial in reducing perinatal
morbidity than simply monitoring all classically high-risk patients. Total
reliance on such a scheme is unlikely, in that patients tend
to observe a decrease in fetal movement as gestation progresses; patient
reliability also varies considerably. ESTRIOLS. Third-trimester biochemical assessment of the fetoplacental unit is most
commonly done using either urinary or plasma estriols.78, 79, 80, 81 Since estriol is the product of fetal and placental compartments, its
assay had great theoretical potential.78 Prior to the mid 1970s, serial urinary estriol assays were the only commonly
used technique of evaluating fetal health. Since the advent of
NST/CST and biophysical profile programs, biochemical surveillance has
been largely replaced by biomechanical surveillance. This is a function
of potential limitations of the technique where normal values are not
strictly related to suboptimal fetal health, as well as the gestational
age dependency of normal values. The clinician who employs estriol determinations in his management must
understand the estriol synthesis pathway and the potential for its disruption. For
instance, chronically low for gestational age estriol values
may be seen in association with anencephaly (presumed failure of
central stimulation or absence or aplasia of the adrenal). Further, liver
or biliary disease or disturbance of gut flora by oral antibiotics
may reduce estriol levels detected in the maternal urine, since approximately
half of estriol conjugates are excreted by way of bile into the
enterohepatic circulation where they are hydrolyzed by intestinal bacteria
to free estriol, which is then reabsorbed. In addition, the clinician
must be aware of the complexities of the analysis of plasma or
urinary estriols. The first step in analysis is acid or enzyme hydrolysis. There
may be significant inconsistent losses of estriol in the assay. Significant
reduction of recovery also occurs in the presence of
urinary antiseptic methenamine. Many laboratories correct for these recovery
losses by using an internal standard of radioactive estriol-16-glucuronide; however, many do not. The clinician should be aware of the
laboratory policies in his institution. Although serial estriols are
used with considerably less frequency, when used they are probably best
employed in the management of pregnancy complicated by diabetes, hypertension, suspected
IUGR, or postmaturity. In most instances, serial
values should be compared to both a standard curve and to the average
of the preceding two or three values. Collection of specimens is usually
on a daily basis in diabetes or two to three times per week in hypertension, suspected
IUGR, or post-maturity. A single value should virtually
never be used for assessment of management. Abnormal patterns
may be manifest in one of three patterns: (1) progressive downward slope, (2) rapid
fall (35%–40% of baseline established by average of
values for preceding 3 days), (3) persistent low values. However, the
clinician must be hesitant in acting immediately on the basis of low
values. Persistent low values may be seen with failure to collect a true 24-hour
specimen, renal function impairment, or overestimation of
gestational age. High urinary glucose or methenamine therapy for cystitis
may also be associated with false-abnormal values. Assay of 24-hour
urinary creatinine excretion in parallel with estriol provides a reasonable
internal standard to determine if 24-hour collection is complete. Sudden
drops in estriol must be interpreted with caution; adequacy
of the collection can be verified by noting 24-hour urine creatinine
values; the average patient excretes 12 mg to 15 mg of creatinine per
kilogram (approximately 1 g) of body weight per 24 hours in her urine. Intelligent
assessment of gestational age and renal function is also
helpful. Some investigators have favored collection of serial short-term urine collections
of which an estriol/creatinine ratio is calculated. However, in
those institutions still using estriols, the recent trend is to favor
plasma over urinary estriol assay. Plasma estriols may be assayed
as (1) unconjugated (free) estriol, which comprises 8% to 10% of total
estriol;80 (2) total estriol;80 and (3) immunoreactive estriol.81 Patient inconvenience and inability to reliably collect a true 24-hour
urinary specimen have provided the impetus to assess plasma estriols. Plasma
estriol collection is rapid (obviating delays in obtaining results), simple, and
complete. Rising or stable values within normal limits
are very reassuring. False-positive results unfortunately are common. Plasma unconjugated estriol levels are primarily dependent on fetoplacental
production and secretion rates; they are therefore less affected
by disorders of maternal liver or kidney. Distler and co-workers feel
that plasma free urinary estriols are the most useful of the plasma tests
in the management of the pregnant diabetic.80 Total plasma estriol includes both the unconjugated and the conjugated (glucuronides
and sulfates) fractions. The assay includes solvent extraction
or enzyme hydrolysis prior to radioimmunoassay. As a consequence, assay
time is 6 to 8 hours in a routine laboratory. Some advocate the use of immunoreactive estriol, which reflects approximately 40% of
total estrio.81 This assay has the advantage of eliminating the extraction process and
thus the assay time is considerably less than that of either total or
unconjugated estriol. At the present time, clinical utilization of plasma
estriols is not widespread. HUMAN PLACENTAL LACTOGEN. Human placental lactogen (HPL) is a single-chain polypeptide hormone immunologically
similar to human growth hormone, with a molecular weight
of approximately 21,000 and half-life of approximately 25 minutes.82, 83 It is produced by the syncytiotrophoblast. Although the precise function
of this hormone has not been determined, it does have inherent somatotrophic, lactogenic, mammo-trophic, and luteotrophic activities. As
a result, it has also been called human chorionic somatomammotropin. The clinical significance of HPL is controversial. It was originally thought
that HPL levels might be useful in predicting the outcome of patients
with threatened abortion. However, many patients with low HPL values
do not abort; clinical over assessment of gestational age may be
a factor. First-trimester real-time ultrasound evaluation of uterine contents
is probably more accurate in predicting eventual outcome than
is HPL. At one time, HPL levels were thought to be of value in fetal placental
function in hypertensive diseases of pregnancy, diabetes, and
postmaturity or in assessing or predicting the presence of IUGR; subsequent
studies have not supported this conclusion. Unfortunately, there
is a high false-positive and false-negative rate when HPL is used to
assess these conditions. Furthermore, HPL levels do not necessarily fall
prior to or immediately after intrauterine fetal death. HPL levels
correlate best with placental weights; however, this correlation is not
consistent. As a consequence, the value of HPL in assessing placental
integrity is limited. If there is a clinical application, it may be
for high-risk screening.84 Fetal Maturity Assessment Spontaneous premature labor, premature rupture of the membranes, or high-risk
pregnancies requiring premature intervention for maternal-fetal
indications account for most newborns delivered prior to functional pulmonary
maturity. However, as recently as 1976, 8% to 15% of newborns
developing RDS followed failure to use currently available techniques
to assess fetal pulmonary maturity or to correctly assess gestational
age.39, 40 Although most cases of RDS are not preventable, a significant fraction
could probably be eliminated by more specific diagnostic techniques to
assess fetal health status or willingness to rely on reassuring results, thus
avoiding unnecessarily early intervention. Fetal lung maturation is marked by biochemical changes of surfactant with
increasing gestational age. RDS/hyaline membrane disease (HMD) is a
clinical state of respiratory distress/pathologic findings at autopsy
of hyaline membrane associated with a deficiency of surfactant. Surfactant
is a generic term for a complex mixture of substances that act as
a detergent to lower alveolar surface tension (reduce the tendency to
collapse). It is possible to assess surfactant in both a quantitative
and qualitative sense by performing a number of biochemical assays on
amniotic fluid. Eighty to 90% (by weight) of surfactant is lipid; 80% to 90% of
the lipid is phospholipid. Lecithin is the major phospholipid (70%–80%)85, 86, 87 Protein (10%–20%) and carbohydrate 11%–2%) compose the remaining 10%–20%. Saturated (palmitic acid at C-1 and C-2) dipalmitoyl
lecithin (DPL) accounts for 50% of total lecithin88 Phosphatidylcholine (PC) and phosphatidylglycerol (PG) are two acidic
phospholipids that act to stabilize lecithin in the surfactant layer.89, 90 PG seems to be most active in this regard. Sphingomyelin, a sphingolipid, also
has surface-active properties. LECITHIN-SPHINGOMYELIN (L/S) RATIO. Under ordinary circumstances, surface-active lecithin begins to appear
in the amniotic fluid at approximately 24 to 26 weeks. Sphingomyelin
appears earlier and in higher concentrations early in the third trimester. Since
sphingomyelin concentrations change very little with gestation, it
is useful as an “internal standard” for lecithin in
performing the L/S ratio.91, 92, 93 In uncomplicated pregnancies, prior to 30 to 32 weeks, the ratio is generally
less than 1.5. At 34 to 35 weeks, there is a fourfold rise in
lecithin versus sphingomyelin, which remains relatively stable or actually
decreases at the time of the iecithin surge. An L/S ratio of 2, ordinarily
achieved by 35 to 36 weeks' gestation, is clinically accepted
as a reliable indicator of functional pulmonary maturity. The timing of lung maturation may vary considerably in high-risk pregnancies.91 In many instances, conditions known to modify the timing of suffactant
appearance are known only in retrospect; a diagnosis of class A diabetes
or IUGR is unfortunately made only after delivery. It is thus not
surprising in unselected patient series that the L/S ratio is independent
of gestational age and birth weight. As a result, the clinician should
not assume the presence of pulmonary maturity unless there is early
sonographic confirmation of dates and the patient is at term.94 Consequently, consideration of surfactant activity assessment is important
whenever intervention for maternal or fetal indications or inhibition
of preterm labor is considered. This consideration may be important
very early in the third trimester, since approximately 19% of cases
at risk for premature delivery at 28 to 32 weeks and 35% at risk to deliver
between 33 and 36 weeks demonstrate an L/S ratio in excess of 2.95 An L/S ratio in excess of 2 correctly predicts maturity in approximately 98% of
cases; only 2% of patients develop RDS (false-mature/false normal). Most
false-mature results are associated with perinatal asphyxia, maternal
diabetes, or Rh isoimmune disease. In contrast, an L/S ratio
less than 2 is not as predictive; approximately 35% of newborns delivering
within 72 hours of an L/S ratio less than 2 actually develop RDS (true-abnormal).96, 97 LUNG PROFILE. Recently, Gluck's group has developed a more specific measure of
surfactant activity, the lung profile.97, 98 In addition to the L/S ratio, the profile includes assay of the percentage
of DPL, PI, and PG (Figure 11). The profile seems to have particular merit in patients with an L/S ratio
less than 2 and perhaps in diabetics. Ordinarily, PI increases in
parallel with a rise in the L/S ratio up to a value of 2; thereafter, it
falls to early third-trimester values. In contrast, PG first appear
in normal pregnancies at 35 weeks; its presence in excess of 3% signals
the presence of stable mature function of the neonatal lung. In a
recent series of 57 cases with an L/S ratio less than 2, the profile correctly
predicted maturity in 14 of the 18 cases (78% reduction of the
false-immature) in which the L/S ratio was less than 2. Prediction of
true outcome thus increased from 68% to 93% in that series. 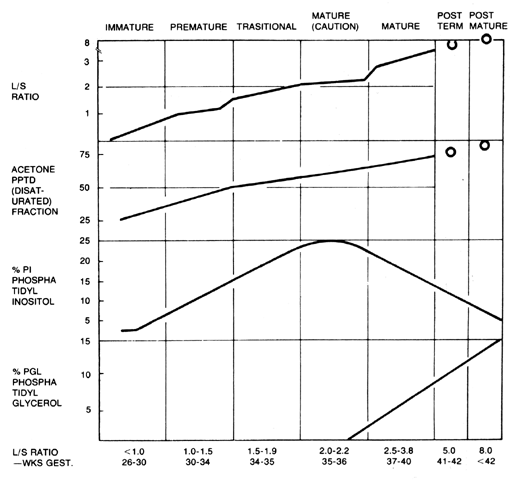 Fig. 11. Form used to report lung profile. L/S ratio, acetone-precipitated desaturated
lecithin, phosphatidyl inositol, and phosphatidyl glycerol are
recorded at points on curves. Values usually fall within a given grid, and
lung maturity may be read off at top of form. (Kulovich MV, Hallman MB, Gluck L: The lung profile: I. Normal pregnancy. Am
J Obstet Gynecol 135:57, 1979. Copyright © by the Regents of
the University of California, 1977) Fig. 11. Form used to report lung profile. L/S ratio, acetone-precipitated desaturated
lecithin, phosphatidyl inositol, and phosphatidyl glycerol are
recorded at points on curves. Values usually fall within a given grid, and
lung maturity may be read off at top of form. (Kulovich MV, Hallman MB, Gluck L: The lung profile: I. Normal pregnancy. Am
J Obstet Gynecol 135:57, 1979. Copyright © by the Regents of
the University of California, 1977)
|
SURFACTANT ASSESSMENT IN HIGH-RISK PREGNANCY. The timing of functional maturation (L/S ratio greater than 2) of the
fetal lung may vary according to maternal or fetal placental disease states. It
may occur as early as 28 weeks in association with class F and
R diabetics, in prolonged rupture of the membranes, and in cases complicated
by severe hypertension-proteinuria syndromes. In contrast, maturation
is often delayed in class A diabetics. The issue of surfactant assessment in pregnancies complicated by maternal
diabetes is both controversial and worrisome. The infant of the diabetic
mother has a six fold increase in risk for developing RDS.99 It is generally accepted that traditional assessment of suffactant using
the L/S ratio in diabetics has a false-mature rate of 3% to 5.5%100, 101 versus an expected rate of 2%97 in the normal population. A higher incidence of cesarean section, asphyxia, and
premature delivery, all of which increase the risk of RDS, may
influence the difference in predictability.102 The diabetic is also prone to gestational age assignment errors. Fortunately, it
appears that the presence of PG in excess of 3% ensures greater
likelihood of pulmonary maturity.98 Pregnancy complicated by hypertensive disease, particularly when severe
and associated with significant proteinuria or placental infarction, may
be associated with accelerated pulmonary maturity manifest by early
elevation of the L/S ratio.91, 93 In some cases. although the L/S ratio is less than 2, PG may be elevated
to a mature 3% level as early as 29 to 30 weeks.98 Advancement of maturation is generally not the case in milder cases or
more acute terminal cases. Although there is some controversy, most investigators feel that prolonged
rupture of the membranes is associated with early appearance of pulmonary
maturity.103, 104, 105 Most series to date are retrospective and do not control for other factors
that may impinge on suffactant induction. However, in cases in which
membranes are ruptured longer than 24 hours, the lung profile when
corrected for gestational age demonstrates an earlier appearance of PG (by 1.5 weeks), higher
mean L/S ratios, and an early appearance of a
mature L/S ratio.98 Preparation for Delivery The obstetrician managing the high-risk mother should make an attempt when
possible to plan delivery timing in cooperation with the neonatologist. Except
in the case of fetal distress, there are few instances when
the obstetrician cannot afford to vary the timing of the delivery according
to availability of the neonatologist. Such planning not only
is important for increasing the chance of survival of the high-risk newborn
but also is very useful in fostering professional interrelationships
and a positive relationship between the mother and the neonatologist, which
is so vital to perinalal care. |
 10 = high risk.
10 = high risk.

 25th p).
25th p). 



