Diseases of the myocardium may first occur during pregnancy because of myocardial ischemia from previously undiagnosed coronary disease, be a viral cardiomyopathy, result from a systemic illness, or be the strictly defined peripartum cardiomyopathy.9,10 Secondary cardiomyopathy as result of a systemic illness has been observed in cases of malignancy, thyroid dysfunction, hypertension, chronic alcohol abuse, and cocaine use. There are reports of cardiomyopathy related to pheochromocytoma during pregnancy.11,12 Women who have had prior doxorubicin or other anthracyclines for a childhood malignancy might have limited cardiac reserve before pregnancy without clinical evidence of impairment.13 Subclinical disease may manifest as the hemodynamic changes of pregnancy occur—specifically as stroke volume increases, resulting in increased preload, which may cause decompensation in the heart lacking functional reserve.
Cardiomyopathy and Pulmonary Edema
Pulmonary edema may be the initial manifestation of myocardial dysfunction due to systolic or diastolic dysfunction.14–16 The causes of congestive heart failure during pregnancy are summarized in Table 3. Acquired causes of systolic dysfunction during pregnancy may include viral myocarditis,17,18 chronic hypertension,19 preeclampsia19,20 or the use of tocolytic agents.19,21,22 Diastolic dysfunction or impaired ventricular compliance may occur with hypertrophic cardiomyopathy,23 chronic hypertension, or preeclampsia.20 Pulmonary edema may occur during a hypertensive crisis.24 In a study of patients presenting with pulmonary edema during pregnancy, 25% had systolic dysfunction, and 75% had diastolic dysfunction. The pulmonary edema occurred antepartum, and in 50% of patients, dexamethasone was a contributing factor.24 The wedge pressure and the central venous pressure may not correlate. When caused by hypertension or preeclampsia, there may be a low capillary wedge pressure found on hemodynamic monitoring.20 Pulmonary edema of pregnancy associated with chronic hypertension and systolic dysfunction on presentation carries a poor prognosis.25
TABLE 3. Causes of Congestive Heart Failure
Fluid overload
Acute myocarditis
Cardiomyopathy (secondary or idiopathic)
Peripartum cardiomyopathy
Alcohol
Valvular heart disease
Mitral stenosis
Aortic stenosis
Arrhythmia
Atrial fibrillation
Ventricular tachycardia
Preeclampsia or toxemia
The diagnostic criteria for classic peripartum cardiomyopathy require congestive heart failure in the ninth month or within 5 months postpartum; no evidence of preexisting heart disease; no identifiable cause such as fluid overload, tocolytic therapy, eclampsia or toxemia, or prior cardiac dysfunction documented by echocardiography; and an estimated ejection fraction of less than 45% or fractional shortening less than 30%.26 The end-diastolic dimension should be greater than 2.7 cm. Women present with chest pain, cough, orthopnea, paroxysmal nocturnal dyspnea, edema, or fatigue.27 Examination confirms heart failure with rales, sinus tachycardia, and a third heart sound. The cause of classic peripartum cardiomyopathy is unknown.26 Poor prognostic indicators for peripartum cardiomyopathy include a left ventricular end-diastolic dimension greater than 60 mm, fractional shortening less than 21%, no echocardiographic improvement to despite treatment of 6 weeks, or symptoms persisting longer than 2 weeks despite medical therapy. Maternal age greater than 30 may not bode well.19,27
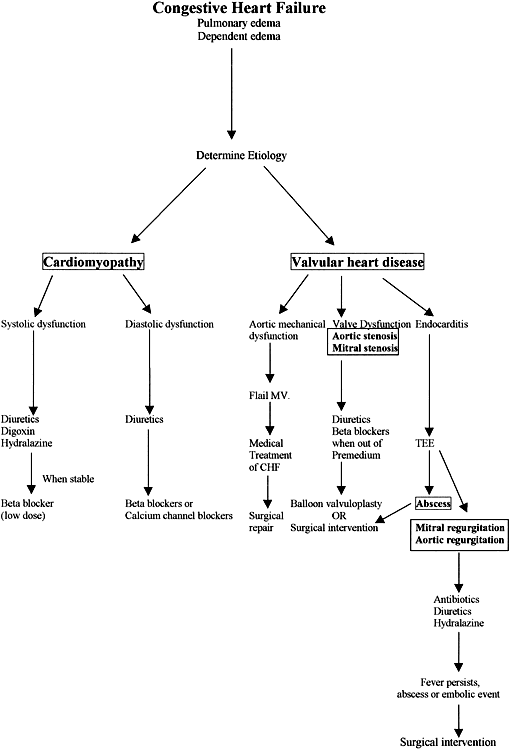
The treatment of pulmonary edema and congestive heart failure depends on the underlying cause (Fig. 1) but generally includes the use of diuretics, digoxin, and afterload reduction. Sodium restriction is effective in the early stages of congestive heart failure. Furosemide or torasemide used during pregnancy has been associated with hypovolemia, hypokalemia, hypomagnesemia, hearing loss, glucose intolerance, and interstitial nephritis. For early signs of mild congestive heart failure, a thiazide diuretic may be effective. Thiazide diuretics cross the placenta and are associated with maternal hypokalemia, volume contraction, pancreatitis, leukopenia, thrombocytopenia, and vasculitis. ACE inhibitors are often used to treat congestive heart failure but are contraindicated in the second and third trimesters of pregnancy.28,29 Hydralazine is preferred for vasodilation and afterload reduction. Adverse effects of hydralazine may include palpitations, flushing, headache, orthostasis, angina, hypotension with reflex tachycardia, and fetal thrombocytopenia.
Digoxin is recommended for treatment of congestive heart failure due to systolic dysfunction and has been used safely in pregnancy. Digoxin may slow the heart rate and cause conduction system disease. Anticoagulation with heparin may be indicated for the treatment of thromboembolic events, unstable angina, cardiomyopathy, or atrial fibrillation. It can be administered twice or three times daily.
The low-molecular-weight heparins may obviate the need for continuous blood testing, but their use for cardiac disorders has not been extensively reported.30,31 With standard heparin therapy, there is an associated risk of bleeding, osteoporosis, and thrombocytopenia. Coumadin use has been contraindicated during pregnancy because of teratogenicity.
The prognosis for postpartum cardiomyopathy varies widely.32,33 Strictly defined peripartum cardiomyopathy may not improve, and the patient may eventually require cardiac transplantation or, although clinically improved, may experience recurrence with subsequent pregnancies. Other causes may be associated with a better prognosis. Decisions regarding subsequent pregnancies should be made only after full recovery.34
Coronary Artery Disease
The older gravida at risk for coronary artery disease may first develop angina with pregnancy, although this is fortunately rare.34 Risk factors for coronary artery disease most often identified in this population are smoking and hypertension, but homocystinuria and diabetes may also be the cause.35,36 Theoretically, women with anticardiolipin antibodies may be at risk for thrombotic events.37 Complications of anomalous coronary arteries may first arise during pregnancy.38
The evaluation and treatment of chest pain during pregnancy may proceed as during the nonpregnant state. The differential diagnosis includes aortic dissection, esophageal spasm, and a pulmonary process.39 Early in pregnancy, stress testing to a predetermined heart rate (140 to 150 bpm) may confirm coronary artery disease, especially when combined with echocardiography. Echocardiography to detect wall motion abnormalities induced by ischemia is preferred over nuclear imaging during exercise, because the radioisotope is excreted by the kidneys and may concentrate in the bladder. If indicated, cardiac catheterization may be done with abdominal shielding. Beta blockers and aspirin are the initial treatment choice. Additional antianginal medical therapy may include nitrates and calcium channel blockers. These drugs have had limited use during pregnancy for angina, but calcium channel blockers such as diltiazem and verapamil have been used more often for treatment of arrhythmia. Although aspirin (81 mg) has been associated with bleeding and premature ductal closure, it has been used for other indications during pregnancy.
Acute myocardial infarction occurs rarely during pregnancy and has a reported incidence of 1 case in 10,000.40–44 In a review of women who had myocardial infarction during pregnancy, the average maternal age was 32 years.45 Infarction most often occurred in the third trimester. On review of the clinical factors, the women who had infarcts were most often multiparous. The anterior walls of the heart were most often involved, and when the women underwent cardiac catheterization, nearly one third had normal coronary arteries. Maternal mortality was approximately 21%, with a 13% fetal mortality. It was hypothesized that the infarcts resulted from vasospasm or localized dissection.45,46 Dissecting aneurysm of a coronary artery has been reported.47
The treatment of a myocardial infarction during pregnancy should follow guidelines for the nonpregnant patient with the use of nitrates, beta blockers, and aspirin. Nitrates may cause hypotension. Aspirin should be used in low doses, because there is a risk of bleeding. There are case reports in the literature on the use of thrombolytic therapy for the treatment of myocardial infarction during pregnancy.48,49 Reports have included the use of streptokinase, urokinase, or tissue plasminogen activator.48,49 The agent has been administered at any time from 9 to 38 weeks. There has been no evidence of teratogenic effects. However, maternal complications have included hemorrhage, preterm delivery, fetal loss, spontaneous abortion, and uterine hemorrhage. Primary angioplasty for acute myocardial infarction has been reported and may be preferred over aggressive thrombolytic therapy.50–54 Stent implantation has been used for treatment of acute myocardial infarction. This procedure required periprocedure and subsequent use of the antiplatelet agents abciximab and ticlodipine in addition to aspirin.54 Ticlodipine may cause neutropenia and thrombocytopenia. Cardiac surgery during pregnancy has been reported.55,56 Most reports are of surgery for valvular heart disease. Cardiac surgery requiring generalized hypothermia may precipitate uterine contractions resulting in preterm labor. Normothermic conditions are recommended with continuous fetal monitoring.55,56