Intra-amniotic infection is an acute bacterial infection of the amniotic fluid and intrauterine contents during pregnancy. Prospective studies indicate that IAI occurs in 4% to 10% of all deliveries.3–5 Other terms used to describe IAI include amniotic fluid infection, amnionitis, and clinical chorioamnionitis. IAI is clinically diagnosed by maternal fever, uterine tenderness, leukocytosis, and fetal tachycardia and should be distinguished from histologic chorioamnionitis. Histologic chorioamnionitis is a histopathologic diagnosis characterized by inflammatory cell infiltration of the membranes. Histologic chorioamnionitis occurs more frequently than IAI, with a prevalence of 4% to 16% at term to greater than 60% in extremely preterm gestations, and is usually not associated with clinical signs or symptoms.6–8 Unfortunately, many authors have not distinguished between these two distinct entities, making comparisons between studies difficult.
IAI is an important cause of maternal and neonatal morbidity. IAI accounts for 10% to 40% of cases of febrile morbidity in the peripartum period and is associated with 20% to 40% of cases of early neonatal sepsis and pneumonia.2 Maternal bacteremia occurs in 2% to 6% of patients with IAI, and postpartum infectious morbidity increases with bacteremia. An increased risk of dysfunctional labor and cesarean delivery occurs among patients with IAI. A 75% incidence of dysfunctional labor and a 34% incidence of cesarean delivery is reported among patients who developed IAI while in labor.9 IAI also is associated with increased neonatal morbidity and mortality, particularly among preterm neonates. In general, a three- to four-fold increase occurs in perinatal mortality among low-birthweight neonates born to mothers with IAI;10,11 also increased is the incidence of respiratory distress syndrome, intraventricular hemorrhage, and neonatal sepsis.12 Recently, IAI has been implicated in neonatal periventricular leukomalacia and cerebral palsy; the risks of cerebral white matter damage and cerebral palsy are nine-fold greater in the setting of IAI.13,14 Finally, subclinical IAI has been found in at least 10% of women in preterm labor with intact fetal membranes, suggesting that IAI is an important, and potentially preventable, cause of prematurity.15
Pathogenesis
Intra-amniotic infection may result from an ascending infection of cervical or vaginal microbial flora, bacteremia, or iatrogenic causes. Most cases of IAI result from ascending infection after labor or rupture of membranes. Before labor or rupture of membranes, amniotic fluid is nearly always sterile. With the onset of labor or rupture of membranes, bacteria indigenous to the lower genital tract ascend into the amniotic cavity. Increasing duration of labor or rupture of membranes raises the rate of isolation of bacteria from amniotic fluid. In fact, all amniotic fluid samples taken from women with rupture of membranes of a duration of 6 hours or more contained microorganisms,16 and about 90% of those with a “high-virulence” microorganism develop an intrauterine infection.17
The second route of infection is hematogenous spread, across the placenta, from a bacteremic mother. A wide variety of microorganisms are involved, but the most notable infection occurs with Listeria monocytogenes. Transplacental passage of L. monocytogenes may occur in isolated cases or as an epidemic, usually after the ingestion of contaminated dairy products.18 Epidemics of food-borne listeriosis disproportionately affect perinatal cases and are associated with preterm birth, stillbirth, or abortion.
The third mechanism for IAI is the iatrogenic introduction of bacteria during an invasive procedure such as cervical cerclage, amniocentesis, cordocentesis, or intrauterine transfusion. The risk of iatrogenic IAI is small, ranging from less than 1% after amniocentesis, 0.5% to 1% after cordocentesis, and 1% to 2% after routine cervical cerclage.19 Emergency, or rescue, cerclage, performed after dilatation of the cervix has begun, has a much higher rate of IAI: 25% to 40%.20,21 This apparently high risk of IAI after emergency cerclage may represent an underlying subclinical intrauterine infection that precedes cerclage placement. In 33 women at 14 to 24 weeks of gestation with cervical dilatation of 2 cm or more, 7 (21%) had evidence of bacterial invasion of amniotic fluid by Gram stain before cerclage. Further, IAI was subsequently documented even in 50% of cerclage patients with negative amniotic fluid Gram stains.22 Two more recent studies that used a course of broad-spectrum antibiotics with emergency cerclage reduced the risk of IAI to 5% to 16%.23,24
Risk Factors
Many risk factors exist for IAI. The risk falls into three broad categories: prolonged labor or complicated pregnancies, abnormal vaginal flora, and preterm birth.
PROLONGED LABOR
Nulliparity, prolonged duration of ruptured membranes, prolonged duration of labor, multiple vaginal examinations, and internal fetal monitoring are consistently linked to IAI.3,4,25 One or more of these risk factors is present in most patients, and the strength of association is usually strong. Prelabor rupture of membranes with more than eight vaginal examinations increased the risk of IAI five-fold, and duration of labor of 12 hours or longer increased the risk of IAI four-fold compared with patients without these risk factors.25 In a stepwise logistic regression, duration of rupture of membranes and duration of internal fetal monitoring were the major risk factors for IAI, and patients with these two risks had the relative risk of IAI increase 19.7-fold.3 About 70% of patients who ultimately developed IAI met risk criteria several hours before the diagnosis was clinically evident.3 This is consistent with the view that clinical indicators of infection develop late in the time course of the infection, as discussed below. In addition, meconium-stained amniotic fluid, maternal colonization with group B streptococcus, prior history of an elective or spontaneous abortion, and prolonged second stage of labor are identified as risk factors for IAI.25–27
ABNORMAL VAGINAL FLORA
An association between bacterial vaginosis (BV) and IAI exists.28–32 The association between BV and IAI is based on: 1) epidemiologic studies showing an increased risk of IAI, with odds ratios of 1.5 to 6.8, for women with BV;28–32 2) the association between the presence of specific BV-associated microorganisms, or the absence of Lactobacillus sp. in the vagina and IAI;31,33 3) the recovery of BV-associated microorganisms from chorioamnion among women with histologic chorioamnionitis and preterm delivery;6 and 4) the recovery of BV-associated microorganisms from amniotic fluid of women with clinical IAI.30
The association of BV and IAI in the setting of preterm birth is particularly relevant, considering the prevalence of BV. BV is the most prevalent vaginal infection in sexually active women, occurring in approximately 20% of pregnant women.34 Anaerobes, frequently those associated with BV, were recovered from amniotic fluid significantly more often in women with IAI giving birth to low-birthweight neonates than in women with IAI at term.35 BV-associated microorganisms, including Mycoplasma hominis, Gardnerella vaginalis, and anaerobes, were recovered from the chorioamnion of 42% of women delivering at less than 37 weeks of gestation and from only 7% of women delivering at term (odds ratio 9.5).6 Thus, BV is associated both with IAI and with preterm birth.
PRETERM BIRTH
The incidences of IAI, microbial invasion of chorioamnion, and histologic chorioamnionitis are all strongly inversely correlated with gestational age. Histologic chorioamnionitis occurs in up to 60% to 90% of gestations ending between 20 and 24 weeks,7,8 and microbial infection of the chorioamnion occurs in up to 60% of patients with preterm delivery.6 The incidence of clinically apparent IAI also varies inversely with gestational age. In a literature review, the incidence of clinical IAI was 41% at gestational ages less than 27 weeks, 15% at gestational ages 27 to 37 weeks, and 2% at gestations of 38 weeks or greater.2 Bacteria indigenous to the lower genital tract also are recovered from the amniotic fluid of 10% to 20% of all women in preterm labor with intact fetal membranes and no clinical signs of IAI15 and of up to 67% of women in preterm labor with pregnancies ending at 23 to 24 weeks.36 Most patients with IAI deliver rapidly, and clinically apparent IAI develops in a few. These observations support the hypothesis that ascending, initially subclinical IAI precedes preterm labor and may be an important cause of extreme preterm deliveries.
Coitus has been suggested in epidemiologic studies to be a risk factor for chorioamnionitis.37 However, there is no direct evidence relating coitus with IAI, and a more recent study of 10,000 Israeli women found no increased risk of premature rupture of membranes, low birthweight, or perinatal death associated with coitus in the third trimester.38
Diagnosis
Ideally, an early diagnosis of IAI is important to allow timely treatment and intervention. Unfortunately, the early diagnosis is difficult because the clinical signs and symptoms of IAI occur late and are neither sensitive nor specific. In a nonhuman primate model, where experimental IAI was induced with group B streptococcus, fever and leukocytosis were present in only 50% of the primates at the onset of infection-induced preterm labor, which occurs 28 to 40 hours after experimental infection.39 Thus, to avoid a delay in diagnosis, a high index of suspicion is warranted, together with appropriate adjunctive laboratory tests. The clinical criteria commonly used to diagnose IAI include maternal fever (more than 37.8°C), and two or more of the following: maternal leukocytosis (15,000/mm3 or more), maternal or fetal tachycardia, uterine tenderness, or foul-smelling amniotic fluid (Table 1).10 Because low-grade maternal fever may arise from other sources, other nonobstetric causes of fever should be excluded.
Table 1. Criteria for the Clinical Diagnosis of Intra-amniotic Infection
Maternal fever  37.8°C
37.8°C
Two or more of the following:
Maternal leukocytosis  15,000/mm3
15,000/mm3
Uterine tenderness
Maternal tachycardia >100 beats per minute
Fetal tachycardia >160 beats per minute
Malodorous amniotic fluid
No other apparent source of infection.
Gibbs RS, Castillo MA, Rodgers PJ: Management of acute chorioamnionitis. Am J Obstet Gynecol 136:709, 1980.
Because of the inconsistency of clinical features (Table 2), other adjunctive laboratory tests are used to aid in the diagnosis of IAI. These include measurement of maternal C-reactive protein, direct examination of amniotic fluid for leukocytes or bacteria by Gram stain, amniotic fluid culture, and measurement of amniotic fluid glucose concentrations, amniotic fluid leukocyte esterase, bacterial organic acids by gas-liquid chromatography, various amniotic fluid or vaginal cytokines (e.g, interleukins 2, 4, 6, granulocyte colony-stimulating factor, and tumor necrosis factor-alpha), matrix metalloproteinase-9, lactoferrin, and fetal activity (biophysical profile) by ultrasonography. Cytokine or other biochemical factor measurement is expensive and generally not clinically available. Further, these tests are no better than more readily available traditional tests such as amniotic fluid Gram stain and culture, amniotic fluid glucose concentrations, and detection of amniotic fluid leukocyte esterase. The efficiency of these tests is summarized in Table 3.40 Although all have reasonable sensitivity, specificity, and predictive value, none is sufficiently sensitive or specific to be used independently of clinical features in the diagnosis of IAI. Unfortunately, only a few studies of amniotic fluid markers for IAI have directly compared Gram stain, glucose, or leukocyte esterase in the same patients,41–47 and only one study has directly compared all three (Table 4).48 The range of the predictive values observed in these studies may depend, in part, on both the variable prevalence of IAI in the study population and on the cross-sectional nature of the studies, because amniotic fluid is obtained only at a single time during a temporally progressive infection. Animal models that allow longitudinal sampling of amniotic fluid after infection are helpful to delineate the natural progression of laboratory markers in infection.
Clinical Sign | Frequency (%) |
Intrapartum fever | 100* |
Maternal leukocytosis >15,000/mm3 | 70–90 |
Maternal tachycardia >100 bpm | 20–80 |
Fetal tachycardia >160 bpm | 40–70 |
Malodorous amniotic fluid | 5–22 |
Uterine tenderness | 2–25 |
*Intrapartum fever required by study for diagnosis of intra-amniotic infection.
Data are based on a summary of reported studies.
|
| Reported Range (%) | |||
| Standard Used |
|
| Positive | Negative |
Test | for Diagnosis | Sensitivity | Specificity | Predictive Value | Predictive Value |
Gram stain | AF culture | 36–80 | 83–98 | 78–87 | 56–92 |
Glucose | AF culture | 63–87 | 92–99 | 78–87 | 56–92 |
Leukocyte esterase | AF culture | 19–83 | 85–100 | 42–100 | 67–78 |
Biophysical profile | Clinical intra-amniotic | 67–90 | 83–86 | 31–60 | 96–98 |
| infection |
|
|
|
|
* Adapted from data in Ohlsson A, Wang E: An analysis of antenatal tests to detect infection at preterm rupture of the membranes. Am J Obstet Gynecol 162:809, 1990.
|
|
|
|
|
|
|
| Positive Predictive | ||
|
| Sensitivity (%) | Specificity (%) | Value (%) | ||||||
Author | Patients | GS | Glu | LE | GS | Glu | LE | GS | Glu | LE |
Romero41 | 171 | 36 | - | 19 | 95 | - | 87 | 78 | - | 42 |
Hoskins42 | 42 | 62 | - | 91 | 81 | - | 95 |
|
|
|
Egley43 | 44 | 54 | - | 81 | 83 | - | 100 | 82 | - | 100 |
Romero44 | 168 | 65 | 87 | - | 99 | 92 | - | 61 | 63 | - |
Kiltz45 | 86 | 50 | 63 | - | 100 | 99 | - | 100 | 99 | - |
Gauthier46 | 204 | 30 | 79 | - | 97 | 94 | - | 87 | 87 | - |
Odibo47 | 88 | 26 | 28 | - | 95 | 95 | - | 88 | 88 | - |
Gauthier48 | 33 | 49 | 87 | 80 | 97 | 90 | 84 | 92 | 87 | 78 |
Gs, Gram Stain; Glu, glucose; LE, leukocyte esterase.
With the exception of maternal C-reactive protein and ultrasonography, these tests require amniotic fluid for analysis. Amniotic fluid may be obtained by aspiration through a transcervical intrauterine pressure catheter among women in labor with ruptured membranes. However, for women in preterm labor with intact membranes or for women with premature rupture of the membranes not in labor, an amniocentesis is required to obtain fluid, and the risks of amniocentesis must be weighed against the possibility of infection. With premature rupture of the membranes, amniocentesis is successful in only about half of patients. Thus, for many patients with clinically apparent IAI, amniotic fluid may not be available, or necessary. Amniocentesis may be clinically more useful among those in preterm labor with intact fetal membranes in whom infection is suspected.
Microbiology
Intra-amniotic infection is frequently polymicrobial, involving both facultative and anaerobic bacteria. In a case-control study of 52 patients with IAI and 52 matched controls, an average of 2.2 microbes were isolated from amniotic fluid of women with IAI.49 Among those with IAI, 48% had both facultative and anaerobic microorganisms isolated, 38% had only facultative organisms, and 8% had only anaerobes. Further, patients with IAI were more likely than controls to have higher concentrations of high-virulence microorganisms (e.g, anaerobes, group B streptococci, Escherichia coli) recovered from amniotic fluid. In the largest series, the most frequent microorganisms recovered from amniotic fluid of IAI included G. vaginalis, Bacteroides bivius, other gram-negative anaerobes, and the genital mycoplasmas M. hominis and Ureaplasma urealyticum (Table 5).35 In an important observation, although group B streptococci and E. coli were recovered from amniotic fluid in only about 20% of patients with IAI, these two microorganisms accounted for approximately two thirds of maternal or neonatal bacteremia.35 Bacteremia of the mother or neonate occurs in 25% of patients with group B streptococci recovered from amniotic fluid. Bacteremia develops in 33% of mother or neonate pairs when E. coli is recovered from amniotic fluid. Although it is frequently recovered from amniotic fluid, anaerobic maternal or neonatal bacteremia is rare. This may be interpreted to mean that anaerobes are locally active but not particularly invasive.50
Table 5. Microorganisms Recovered From Amniotic Fluid in 404 Women With
Intra-amniotic Infection
|
| % Patients With |
|
| Total Isolates | Organisms | % Total Isolates |
Facultative |
|
|
|
Gardnerella vaginalis | 99 | 24.5 | 11.3 |
Group B streptococcus | 59 | 14.6 | 3.8 |
Escherichia coli | 33 | 8.2 | 3.8 |
Enterococci | 22 | 5.4 | 2.5 |
Anaerobic |
|
|
|
Bacteroides bivius | 119* | 29.5 | 13.6 |
Peptostreptococcus sp. | 38 | 9.4 | 4.3 |
Fusobacterium sp. | 22* | 5.4 | 2.5 |
Bacteroides fragilis | 14 | 3.5 | 1.6 |
Gram-negative anaerobe | 155* | 38.4 | 17.7 |
Genital Mycoplasma |
|
|
|
Ureaplasma urealyticum | 190 | 47.0 | 21.7 |
Mycoplasma hominis | 123 | 30.4 | 14.3 |
*Isolated significantly more frequently from amniotic fluid of women giving birth to low-birthweight neonates (<2500 g).
Adapted from Sperling RS, Newton E, Gibbs RS: Intraamniotic infection in low-birthweight infants. J Infect Dis 157:113, 1988.
In the series of 404 patients with IAI, pregnancies resulting in low-birthweight neonates (less than 2500 g) were more likely than pregnancies with normal-birthweight infants to have anaerobes(B. bivius, Fusobacterium sp, and gram-negative rods) in the amniotic fluid.35 In contrast, facultative microorganisms were recovered with equal frequency among preterm and term gestations.
M. hominis and U. urealyticum are recovered from amniotic fluid in 30% to 50% of women with IAI, making them the two most frequent isolates. However, they are frequently recovered in association with other virulent microorganisms, and their pathogenic potential is uncertain. M. hominis was recovered from the amniotic fluid of 35% of patients with IAI and only 8% of control patients.51 Bacteremia with M. hominis occurs in 2% of patients with both IAI and M. hominis in amniotic fluid.52 Thus, a pathologic potential of M. hominis clearly exists. In contrast, U. urealyticum was recovered from 50% of both patients with IAI and in noninfected control patients; neither U. urealyticum bacteremia nor a maternal serologic response occurred that would suggest infection.51,52 However, U. urealyticum is the most common microorganism recovered from amniotic fluid of women in preterm labor with intact fetal membranes,53,54 and the recovery of U. urealyticum from chorioamnion membranes is strongly associated with histologic chorioamnionitis.6,55 In an experimental nonhuman primate model of chorioamnionitis, intra-amniotic inoculation of U. urealyticum, in the absence of other pathogens, caused preterm delivery, IAI, histologic chorioamnionitis, and fetal pneumonitis and meningitis.56 Thus, U. urealyticum is a potential pathogen. The role of M. hominis and U. urealyticum in human intra-amniotic or intrauterine infection remains uncertain because most patients recover from infection despite antibiotic therapy that is not effective against either microorganism.
Both Chlamydia trachomatis and Neisseria gonorrhoeae have been associated with preterm labor and premature rupture of the membranes.28,57 However, neither is usually recovered from amniotic fluid among women with IAI, although C. trachomatis can grow in amniotic cell monolayers in vivo. In a prospective study of perinatal mortality complicated by maternal chlamydial infection, two of six fetal deaths in the chlamydial infection group were associated with histologic chorioamnionitis, in contrast to only one of eight in controls without chlamydial infection.58 Similarly, a higher rate of intrapartum fever was present among patients with than without antepartum cervical chlamydial infections.59 Thus, a role of C. trachomatis in acute IAI is suggested but not confirmed by intrapartum cultures, perhaps because C. trachomatis would be expected to grow poorly in the relatively cell-free environment of amniotic fluid.
Treatment
Three compelling principles guide the treatment of IAI. First, IAI is frequently a polymicrobial infection, with both facultative and anaerobic microorganisms, and broad-spectrum parenteral antibiotics are indicated. Second, although group B streptococci and E. coli account for only about 20% of isolates from IAI, these two pathogens are responsible for 60% to 70% of cases of neonatal sepsis and meningitis. Thus, antibiotics selected to treat IAI must have activity against these two pathogens. Finally, antibiotic therapy should be begun in the intrapartum period, as soon as the diagnosis is confirmed, because intrapartum antibiotics improve neonatal outcome.
A combination of parenteral ampicillin, 2 g intravenously every 6 hours, and an aminoglycoside such as gentamicin, 1.5 mg/kg intravenously every 8 hours, are most often used because of excellent activity against both group B streptococci and E. coli. However, these agents provide limited anaerobic coverage. Thus, clindamycin, 900 mg intravenously every 8 hours, should be added to this regimen for patients with IAI who undergo cesarean delivery to prevent postpartum endomyometritis. Because dysfunctional labor leads to cesarean delivery in 40% of patients with IAI, a high proportion of patients with IAI will ultimately require triple antibiotic therapy, with its associated expense and potential toxicity. For most infections, single-agent therapy with a broad-spectrum antibiotic is equally efficacious and more cost-effective. Recommended intravenous regimens include cefotetan (2 g every 12 hours), piperacillin or mezlocillin (4 g every 6 hours), or ampicillin/sulbactam (3 g every 6 hours) (Table 6). Because of the greater risk of postpartum endomyometritis after cesarean delivery, the chosen antibiotic regimen should be continued until the patient has been afebrile and symptoms have resolved for at least 24 hours. For patients delivering vaginally, the duration of antibiotic therapy is more arbitrary; most patients defervesce promptly, and more than one dose of antibiotics postpartum may be unnecessary.60
Table 6. Antibiotic Treatment of Intra-amniotic Infection
2 g IV every 6 hours | |
+ |
|
Gentamicin | 1.5 mg/kg IV every 8 hours, |
| or 4.5 mg/kg IV every 24 hours |
If delivered by cesarean, add clindamycin 900 mg IV | |
every 8 hours |
|
Ampicillin/sulbactam | 3.0 g IV every 6 hours |
Piperacillin | 4.0 g IV every 6 hours |
Mezlocillin | 4.0 g IV every 6 hours |
Cefotetan | 2.0 g IV every 12 hours |
Antibiotic therapy should begin in the intrapartum period, as soon as the diagnosis of IAI is confirmed. Therapy should not be deferred until after delivery unless delivery is imminent. The benefits of intrapartum treatment clearly outweigh the potential disadvantage to the pediatrician to perform neonatal cultures without the presence of antibiotics. These antibiotics all rapidly penetrate into the fetal compartment and achieve therapeutic concentrations in cord blood after maternal intravenous administration.61 Recent studies comparing intrapartum with postpartum treatment consistently report a lower incidence of neonatal sepsis with intrapartum therapy.11,62,63 In the only randomized, prospective trial of intrapartum versus postpartum treatment, significant reductions in the rate of neonatal sepsis or pneumonia occurred in those receiving intrapartum treatment (0%) versus those with postpartum treatment (32%; p < 0.05).63 Intrapartum treatment led to a 2-day reduction in the average neonatal hospital length of stay.63 Further, maternal postpartum febrile morbidity also was reduced by intrapartum treatment.
Delivery is essential for adequate treatment, and prompt action to effect delivery should be begun at the time of diagnosis, but cesarean section should be reserved for usual obstetric indications. No advantage was attributed to cesarean delivery per se, and it increased maternal postpartum febrile morbidity. When cesarean delivery is necessary, extraperitoneal cesarean offers no benefit to reduce the risk of postpartum endomyometritis.64 Few data exist about the effect on neonatal infection of the interval between the diagnosis of infection and delivery. A critical time has not been identified beyond which maternal and neonatal complications increase. Most patients with IAI deliver within 5 to 7 hours of diagnosis. Several studies found no increase in neonatal infections for an interval of at least 12 hours among patients receiving intrapartum antibiotics.10,65,66 Whether intervals longer than 12 hours increase the risk of neonatal infections has not been determined. Continuous electronic fetal monitoring should be strongly considered. The combination of villous edema, hyperthermia, and fetal infection may contribute to fetal acidosis.2 Fetal tachycardia may be associated with maternal pyrexia but also is an independent predictor of fetal sepsis and pneumonia. Persistent fetal tachycardia may therefore portend a hemodynamically unstable neonate, and personnel skilled in neonatal resuscitation should be present at such deliveries.
Outcome
Significant maternal and perinatal morbidity may occur from IAI. However, with prompt diagnosis and antibiotic treatment, maternal and neonatal outcome may vastly improve.
MATERNAL OUTCOME
Dysfunctional labor and cesarean delivery are the most common adverse maternal outcomes associated with IAI; up to 75% of women with IAI have dysfunctional labor. The rate of cesarean delivery is increased approximately three-fold, to 35% to 40% with IAI, mainly as a result of dystocia.9,10,67 Women with IAI frequently have longer labors and slower rates of cervical dilatation, even at a high concentration of intravenous oxytocin.68 Complications associated with cesarean delivery, including blood loss, wound infection, and endomyometritis, are also increased among women with IAI.
NEONATAL OUTCOME
IAI increases the incidence of complications among term neonates despite the use of intrapartum antibiotics. Term neonates have increased risks of a low Apgar score, pneumonia, and sepsis, although most ultimately do well, and long-term neurologic deficits are rare.66,69
By contrast, preterm or low-birthweight neonates have a much higher frequency of morbidity and mortality from IAI. In addition to sepsis and pneumonia, preterm neonates also are at increased risk for death, respiratory distress syndrome, intraventricular hemorrhage, and neurologic deficits.12,35,70 A case-control study of 92 preterm neonates with IAI (cases) and 602 preterm controls found that a significant increase occurred among cases in the incidence of death (25% vs. 6%), respiratory distress syndrome (62% vs. 35%), intraventricular hemorrhage (56% vs. 22%), and sepsis (28% vs. 11%).12 More recently, a cohort analysis of 95 low-birthweight neonates born after IAI revealed an increased adjusted odds ratios for intraventricular hemorrhage (2.8, 95% confidence interval, 1.6 to 4.8), periventricular leukomalacia (3.4, 1.6 to 7.3), and early neonatal seizures (2.9, 1.2 to 6.8) when adjusted for preterm rupture of membranes, pregnancy-associated hypertension, cesarean birth, gestational age, and birth weight.70 These complications occurred more frequently in the low-birthweight population than among a cohort of term neonates born in a demographically similar population with IAI reported from the same institution (Table 7).69 Taken together, these data clearly show significant deleterious effects of IAI on the preterm or low-birthweight neonate.
| Preterm IAI (n = 95) | Term IAI (n = 5144) |
Outcome | Odds Ratio (95% CI) | Odds Ratio (95% CI) |
Apgar, | 2.1 (1.3–3.3) | 1.3 (0.8–2.0) |
Umbilical artery pH, < 7.0 | 1.9 (0.5–6.9) | 1.2 (0.8–2.0) |
Sepsis | 2.7 (1.03–7.1) | 2.9 (2.1–4.1) |
Respiratory distress syndrome | 2.9 (1.5–5.5) |
|
Intraventricular hemorrhage, | 2.8 (1.6–4.8) |
|
grade 3 and 4 |
|
|
Periventricular leukomalacia | 3.4 (1.6–7.3) |
|
Early seizures | 2.9 (1.2–6.8) | 1.1 (0.6–2.0) |
Neonatal death | 1.7 (0.9–3.3) |
|
CI, confidence interval.
Adapted from Alexander JM, Mclntire DM, Leveno KJ et al: Chorioamnionitis and the prognosis for term infants. Obstet Gynecol 94:274, 1999, and Alexander JM, Gilstrap LC, Cox SM et al: Clinical Chorioamnionitis and the prognosis for very low birth weight infants. Obstet Gynecol 91:725, 1998.
INTRA-AMNIOTIC INFECTION, PRETERM BIRTH, AND CEREBRAL PALSY
Considerable evidence exists that IAI is an important cause of preterm birth and cerebral palsy.14,71 The prevalence of histologic chorioamnionitis is inversely related to gestational age and positively related to cerebral palsy.8,14 Bacteria indigenous to the lower genital tract are recovered from amniotic fluid of 10% to 20% of women in preterm labor with intact fetal membranes.15 The prevalence of amniotic fluid infection also is inversely related to gestational age, occurring in up to 67% of pregnancies ending at 23 to 24 weeks of gestation.36 Proinflammatory cytokines play a central role in the pathogenesis of infection-associated preterm delivery. The proinflammatory mediators (interleukin [IL]-1, IL-6, IL-8, and tumor necrosis factor [TNF]) are produced by maternal and fetal macrophages and decidual cells in response to a wide variety of bacteria or bacterial products. A role for selected cytokines in the onset of preterm labor is based on the following observations:15 1) elevated concentrations of IL-1, IL-6, and TNF and prostaglandins are found in the amniotic fluid of patients with IAI and preterm labor; 2) bacterial products stimulate the productions of IL-1, IL-6, and TNF by decidua; 3) these cytokines stimulate production of prostaglandins by amnion and decidua; and 4) intra-amniotic infusion of IL-1 induces preterm contractions and delivery in nonhuman primates, contractions that are inhibited by immunomodulators, including IL-10 (an anti-inflammatory cytokine) or dexamethasone.72,73
Recently, the role of intrauterine infections in the pathogenesis of periventricular leukomalacia and cerebral palsy has become a major focus of research, both in term and preterm infants.13,14 Periventricular leukomalacia, a cerebral lesion characterized by necrosis of the white matter near the lateral ventricles, is found in 7% to 26% of neonates with a birth weight of less than 1500 g, and 60% to 80% of these infants develop cerebral palsy.74,75 The role of infection, with release of proinflammatory cytokines, in the pathogenesis of periventricular leukomalacia and cerebral palsy was first hypothesized by Leviton in 1993.76 In this hypothesis, infection results in increased fetal concentrations of proinflammatory cytokines, which leads to not only synthesis of prostaglandins and preterm labor but also to damage to the cerebral white matter by one or more of the following mechanisms: hypotension and ischemia; intravascular coagulopathies; activation of platelet activating factor; or direct destruction of oligodendrocytes and toxic effects on myelin.
This hypothesis is supported by human observational studies and animal experimental studies. The risk of cerebral white matter damage was 9.4-fold greater among preterm neonates with purulent amniotic fluid than those with nonpurulent fluid.13 Similarly, maternal intrapartum fever or chorioamnionitis was associated with a 9.3-fold increased risk of cerebral palsy among infants of normal birthweight born mostly at term.14 Studies subsequently reported elevated concentrations of amniotic fluid proinflammatory cytokines, including IL-6, IL-1β, and TNF-α, and elevated umbilical cord plasma concentrations of IL-6 among neonates with periventricular leukomalacia or cerebral palsy.77,78 Higher neonatal blood concentrations of interleukins 1, 6, 8, and TNF-α have also been reported among neonates with cerebral palsy.79 Finally, animal models using pregnant rabbits with experimental intrauterine infection showed brain white matter lesions characterized by increased karyorrhexis, rarefaction, disorganization of white matter, and increased apoptosis in the cerebral cortex.80
The findings of increased umbilical cord IL-6 and neonatal proinflammatory cytokine concentrations suggest a fetal origin to the destructive inflammatory response, consistent with a “fetal inflammatory response syndrome.” Increased expression of IL-6 and TNF-α within the brain lesions of infants dying with periventricular leukomalacia offers further support.81 A fetal inflammatory response to IAI suggests that intrapartum antibiotic treatment alone may be insufficient to prevent these neurologic sequelae, and that immunomodulator therapy may play a role in preventing periventricular leukomalacia.
Prevention
Premature rupture of the membranes (PROM), defined as rupture of the membranes before the onset of labor, occurs in 8% to 10% of all pregnancies; preterm PROM is responsible for one third of all preterm births. Because most IAIs result from ascending infections after rupture of membranes, strategies to prevent IAI are directed at either induction of labor to shorten the duration of PROM, or antibiotic treatment after PROM to prevent ascending infection.
IAI occurs in 10% to 50% and postpartum infection in 2% to 30% of women with preterm PROM. Neonatal sepsis occurs in 1% to 25% after preterm PROM. Approximately 75% of patients with preterm PROM deliver within 1 week of rupture.82 Despite differences in study design, patient inclusion criteria, adjunctive use of tocolysis and corticosteroids, and antibiotics used, many studies in the past decade have documented that antibiotic therapy after preterm PROM reduces the incidence of delivery within 7 days, intra-amniotic and postpartum infections, and neonatal sepsis and pneumonia. One recent meta-analysis of antibiotic treatment after preterm PROM included 13 trials, among them 6 placebo-controlled treatment trials.82 Women receiving antibiotics, compared with untreated women, had significant reductions in deliveries within 1 week (62% vs. 76%; odds ratio 0.5, 95% confidence interval 0.4 to 0.7), IAI (12% vs. 23%; odds ratio 0.5, confidence interval 0.3 to 0.6), and postpartum infection (8% vs. 12%; odds ratio 0.6, confidence interval 0.4 to 0.9).82 Fetal morbidity, including sepsis (5% vs. 9%; odds ratio 0.6, confidence interval 0.4 to 0.9), pneumonia (1% vs. 3%; odds ratio 0.3, confidence interval 0.1 to 0.9), and intraventricular hemorrhage (9% vs. 14%; odds ratio 0.7, confidence interval 0.5 to 0.9) also occurred less frequently after maternal antibiotic therapy. No significant differences were observed with treatment in overall neonatal survival, respiratory distress syndrome, or necrotizing enterocolitis (Table 8). Three recent randomized, placebo-controlled trials confirmed a significant prolongation of the latent period after rupture of membranes and before delivery, and significant reductions in neonatal sepsis, respiratory distress, neonatal oxygen dependency, or ventilator requirements with maternal antibiotic treatment.83–85 The incidence of maternal IAI also was reduced (10.5% with treatment vs. 32.4% without treatment, p < 0.05) in the one study that specifically addressed this outcome.85 A variety of antibiotic regimens were used, but most included broad-spectrum antibiotics such as erythromycin base and ampicillin or amoxicillin/clavulanate, initially administered intravenously, followed by oral therapy for a total of 7 to 10 days. The largest study of 4826 women with preterm PROM compared four groups of patients who received erythromycin, amoxicillin/clavulanate, or erythromycin plus amoxicillin/clavulanate, or placebo.84 Reductions in neonatal morbidity were observed only when erythromycin was administered; by contrast, amoxicillin/clavulanate alone was associated with an increase in the incidence of neonatal necrotizing enterocolitis (3.8% with treatment vs. 2.4% without, p < 0.005). The reasons for this small reported increase in the incidence of necrotizing enterocolitis remain speculative but point out the need for caution in interpreting data before widespread administration of antibiotics.
| With Antibiotic |
|
| Odds Ratio | |
Outcome | Treatment | Control | (95% CI) | ||
Maternal |
|
|
|
|
|
Delivery | 281/457 | (62%) | 349/460 | (76%) | 0.5 |
|
|
|
|
| (0.4–0.7) |
Chorioamnionitis | 74/639 | (12%) | 147/649 | (23%) | 0.5 |
|
|
|
|
| (0.3–0.6) |
Postpartum infection | 36/478 | (8%) | 55/479 | (12%) | 0.6 |
|
|
|
|
| (0.4–0.9) |
Neonatal |
|
|
|
|
|
Confirmed sepsis | 33/649 | (5%) | 56/647 | (9%) | 0.6 |
|
|
|
|
| (0.4–0.9) |
Pneumonia | 4/373 | (1%) | 12/369 | (3%) | 0.3 |
|
|
|
|
| (0.1–0.9) |
Respiratory distress | 217/577 | (38%) | 232/580 | (40%) | 0.9 |
|
|
|
|
| (0.7–1.2) |
Necrotizing enterocolitis | 34/619 | (6%) | 35/616 | (6%) | 0.9 |
|
|
|
|
| (0.6–1.6) |
Intraventricular hemorrhage | 58/619 | (9%) | 85/616 | (14%) | 0.7 |
|
|
|
|
| (0.5–0.9) |
Survival | 626/673 | (93%) | 626/684 | (92%) | 1.2 |
|
|
|
|
| (0.8–1.8) |
CI, confidence interval. Confidence intervals that include 1.0 reflect no significant difference between groups.
Adapted from Mercer BM, Arheart KL: Antimicrobial therapy in expectant management of preterm premature rupture of the membranes. Lancet 346:1271, 1995.
Approximately two thirds of cases of PROM occur in women at term. The most significant maternal or neonatal risk after term PROM is IAI, a risk that increases with the duration of rupture of membranes (discussed previously). Historically, to reduce this risk, term PROM was treated by immediate induction of labor. However, two studies showed that immediate induction of labor among nulliparous patients with unfavorable cervixes resulted in increased cesarean rates and/or increased intrapartum febrile morbidity compared with no induction.86,87 Further, most patients with term PROM deliver within 28 hours of rupture of membranes.88 Expectant management may, therefore, be an option for nulliparous patients with an unfavorable cervix. However, a recent large study of more than 5000 patients with term PROM found greater patient acceptance of immediate induction versus expectant management and a 50% reduction of clinical chorioamnionitis, from 8.6% among those with expectant management to 4.0% among those with immediate induction.88 A 50% reduction in the incidence of postpartum fever, from 3.6% to 1.9%, was also shown.88 Immediate induction of labor after term PROM would therefore seem prudent. Appropriate intrapartum antibiotics to prevent neonatal group B streptococcal disease should be administered in accordance with Centers for Disease Control guidelines if the patient selects expectant management.89
Because BV is a risk factor for IAI, antepartum identification and treatment may reduce IAI. However, no study to date has shown a reduction in the risk of IAI by antenatal screening and treatment of BV.
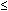 10–14 mg/dL
10–14 mg/dL