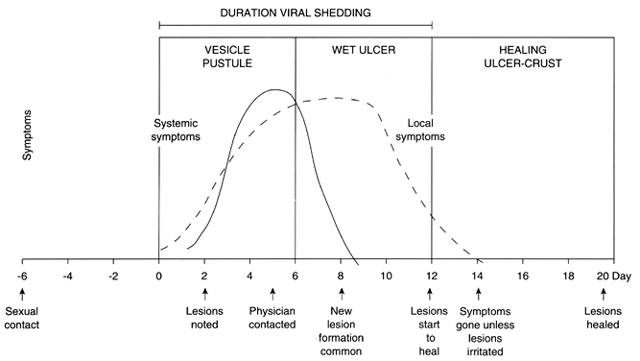
Neonatal HSV infection is the most feared consequence of genital HSV infection
because it carries a high rate of mortality and morbidity. The
increased prevalence of adult HSV between the 1960s and the 1980s was
associated with a parallel increase in the prevalence of neonatal HSV
in the United States.4, 30, 31 Annually, 1500 to 2000 cases of neonatal HSV occur in the United States. Approximately 86% of
neonatal infections are acquired at the time of
birth, 10% are acquired after birth, and 4% are congenital infections.32 HSV-2 causes about two thirds of neonatal herpes and HSV-1 the remaining
third. Genital HSV infection is much more common in pregnancy than is generally
recognized. Only 2% to 5% of pregnant women are aware of a previous
episode of genital herpes; however, characteristic of the asymptomatic
nature of both first-episode and particularly recurrent infection, most
persons carrying the virus do not recognize having had an episode of
HSV infection. Overall, 25% to 35% of pregnant women have serologic
evidence of prior HSV-2 infection.33, 34 The prevalence of antibody to HSV-2 in pregnant women is similar to the
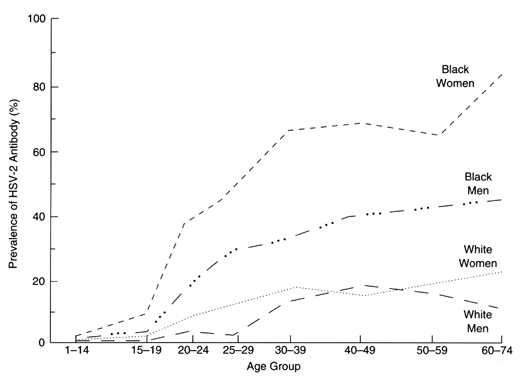
prevalence of HSV-2 antibody in nonpregnant women of reproductive age.11 The prevalence of HSV antibody among women 30 years or older is 25% for
whites and 60% for blacks.11 Therefore, if only 2% to 5% of pregnant women are aware of a prior episode
of HSV whereas 25% to 35% have antibody to HSV, approximately 80% of
pregnant women with latent HSV infection do not report a history of
a prior HSV infection and are unaware that they could possibly expose
their neonate to HSV. Neonatal HSV is most likely to occur as a result of a primary maternal
infection, rather than from the 25% to 35% of women with a recurrent (latent) HSV
infection. Only about 1 in 1000 women have HSV in the genital
tract at delivery from a primary HSV infection. A recent study, however, reported
that neonatal HSV occurred in 15 (41%) of 37 infants born
of mothers with first-episode genital HSV infection at delivery, and
most experts believe that approximately 50% of infants will become
infected when exposed to a primary maternal infection (Table 2).27, 34, 35, 36, 37, 38, 39 The high risk of neonatal HSV from a primary versus a recurrent infection
means that the majority of infected infants are born to mothers with
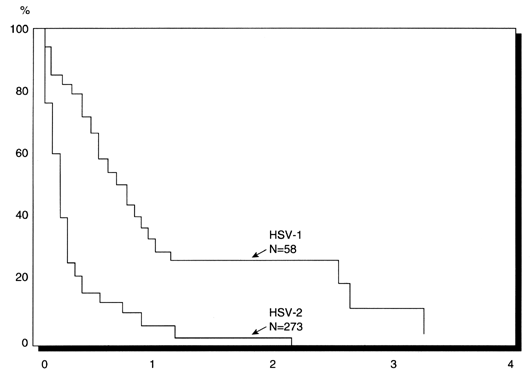
first-episode genital herpes.27 In this study by Brown and colleagues,27 of 52 neonates exposed to HSV in whom the maternal antibody level was
known, 18 (35%) were exposed to a first-episode genital HSV infection
and the remaining 34 (65%) to recurrent HSV. NeonatalHSV infection, however, occurred
in 6 (33%) of the 18 infants exposed to first-episode
genital herpes versus only 1 (3%) of the 34 infants exposed to recurrent
genital herpes (Fig. 4). TABLE 2. Risk Estimates for Neonatal Herpes with Various Exposure Conditions
| Positive Genital | Attack Rate | Neonatal |
Clinical Condition | Culture (%) | (%) | Herpes Odds |
Primary lesion | 100 | 50 | 1 : 2 |
Recurrent genital lesions | 20 | 2* | 1 : 250 |
Recurrent remote lesion † | 2.5 | 5* | 1 : 800 |
History of herpes | 1 | 1* | 1 : 10,000 |
No lesions | 1 | 5* | 1 : 2000 |
*Assumed attack rates.
†Probability for positive culture varies among reports, ranging
from 0% to 22%.
(Hensleigh PA: Genital herpes in pregnancy. In Parer JT (ed): Antepartum
and Intrapartum Management, pp 108–120. Philadelphia, Lea & Febiger, 1989) Fig. 4. Frequency of neonatal herpes infection among women with asymptomatic shedding
of the virus at the time of labor.(Brown ZA, Benedetti J, Ashley R et al: Neonatal herpes simplex virus infection
in relation to asymptomatic maternal infection at the time of
labor. N Engl J Med 324:1247, 1991) Fig. 4. Frequency of neonatal herpes infection among women with asymptomatic shedding
of the virus at the time of labor.(Brown ZA, Benedetti J, Ashley R et al: Neonatal herpes simplex virus infection
in relation to asymptomatic maternal infection at the time of
labor. N Engl J Med 324:1247, 1991)
|
The high rate of neonatal infection during a primary maternal infection
is related to an increase in the concentration of virus, a larger surface
area of lesions, and a high degree of cervical shedding during primary
versus recurrent genital infection, as previously mentioned (see Table 1). A high rate of neonatal infection with exposure to primary HSV also
is related to the lack of a protective effect from HSV serum antibody. HSV
antibodies neutralize the virus, mediate cytotoxicity, and produce
type-specific inhibition. Infected neonates had much lower HSV antibody
titers compared with their mothers or exposed but uninfected neonates.18 Homologous serum HSV antibody also decreases the rate of disseminated
neonatal infection.40 The presence of even heterologous antibody found in cases of nonprimary
first-episode infection influences both pregnancy and neonatal outcome. Adverse
pregnancy outcomes occurred in 6 of 15 neonates born of mothers
with primary firstepisode infections. One of five first-trimester
infections resulted in spontaneous abortion because of herpetic chorioamnionitis, one
of five secondtrimester infections resulted in preterm
delivery, and four of five third-trimester infections were associated
with preterm delivery, growth retardation, and neonatal herpes. No abnormalities
were noted in the 14 neonates born of mothers with nonprimary
first-episode infection.36 The majority of neonatal infections will not only result from primary infection
at or shortly before delivery,34 but the majority of women with a primary infection will be asymptomatic
at delivery. These asymptomatic women with a primary infection at delivery
may have had a recent asymptomatic primary or, occasionally, a
very recent primary infection where symptoms develop only after delivery. In
the study by Brown and associates,27 of 29 women who presented with first-episode genital HSV-2, 15 had a primary
infection and 14 had a nonprimary first-episode infection. All
six infected infants were born of mothers with a primary first-episode
infection in which asymptomatic shedding of HSV-2 at delivery accounted
for nearly all the cases of neonatal infections. In a second study
by Prober and co-workers,38 none of 34 neonates born to mothers with a history of recurrent genital
HSV-2 acquired neonatal herpes. In 31 cases of neonatal herpes, 26% of
mothers had a history of recurrent genital herpes, 29% had primary
infections, 35% had antibody to HSV-2 but no clinical history of genital
herpes, and in 10% of cases the mother was not the source of the infection.18, 34, 36 As many as 80% of women with a history of recurrent infection have a recurrence
of HSV during pregnancy. Pregnancy does not appear to have much
influence on HSV recurrence, although the mean number of recurrences
has been reported to increase from 0.6 in the first trimester to 1.6 in
the third trimester.34, 41 The majority of documented recurrences are associated with external genital
lesions.4 As in nonpregnant women, HSV-2 causes the majority of primary and nonprimary
first-episode genital infections and nearly all recurrent infections
in pregnant women. Most of the recurrences in pregnancy are asymptomatic35, 41; thus, asymptomatic shedding of HSV also is common in pregnancy. The prevalence
of positive cultures, mostly from asymptomatic shedding, at
any time during pregnancy for women with known recurrent HSV can be as
high as 1% to 2% on any one day.35, 36, 41 At delivery, a mean of approximately 0.2% of all pregnant women will asymptomatically
shed virus,42 and approximately 1% of serologically HSV-2-positive pregnant women have
HSV isolated from the genital tract.34, 43 The site of asymptomatic shedding was the usual site of lesions in 50% to 80%; in 20% to 50% of
women, the site was the cervix.34, 43 Women at risk of asymptomatic shedding are difficult to identify. Asymptomatic
shedding at delivery cannot be predicted on the basis of prior
antenatal culture data. Only 1 of 17 women with asymptomatic shedding
during pregnancy had viral shedding at delivery, and that woman had
a visible lesion.41 Five other women had asymptomatic shedding of HSV at delivery but were
culture-negative during the pregnancy. The duration of asymptomatic shedding
is only 1 to 2 days.20 The attack rate of neonatal HSV infection from a mother with recurrent
HSV infection (with type-specific antibodies)38 is estimated to be 2% (see Table 2; range, 1% to 5%). A word of caution is in order for patients with a recent
primary infection because they are more likely to shed HSV asymptomatically
than are those who had HSV before becoming pregnant. Patients
with a recent primary infection are also more likely to shed HSV asymptomatically
from both the cervix and the vulva than are those with
longstanding infection, who tend to shed from the vulva only.34, 35, 41 A summary of the estimated risk of developing neonatal herpes related to
exposure to a primary or recurrent lesion or asymptomatic shedding is
depicted in Table 2.39 The probability of a positive genital culture is very high with primary
HSV infection, approaching 100%. The attack rate of neonatal HSV is
estimated to be 50% when exposure to virus in a primary HSV lesion occurs. In
contrast to primary HSV, the odds of HSV transmission to the neonate
after exposure to a recurrent lesions is about 100-fold less. Only
approximately 20% of cultures from recurrent lesions contain HSV, and
with the attack rate estimated to be 2%, approximately 1 in 250 of
neonates exposed to a recurrent HSV lesion would develop neonatal HSV. Approximately 2.5% of
patients with HSV lesions remote from the genital
area shed HSV from the genital area. If 5% is used as a high end of
the range of attack rate for recurrent HSV, about 1 in 800 neonates
exposed to a remote HSV lesion would develop HSV. Asymptomatic shedding
of the virus at delivery occurs in approximately 1% of women with recurrent
HSV. The attack rate of neonatal herpes is not known, but the
attack rate of neonatal herpes is probably even lower in the setting of
asymptomatic shedding than with a recurrent HSV lesion. The attack rate
is no more than 5%, but it may be as low as 1%. Thus, the rate of
neonatal herpes from asymptomatic shedding in patients without lesions
would be approximately 1 in 5000 to 1 in 2000 exposures (0.01 × 0.01 to 0.01 × 0.05) This low rate of neonatal infection makes
routine cesarean section unnecessary when HSV lesions are not present
in the patient with a history of recurrent HSV. In some reports, primary HSV infection also has been associated with spontaneous
abortion.36, 37 Congenital infection from the hematogenous spread of virus to the placenta
and fetus is not likely, but if it occurs, it does so during the
viremia of a primary first-episode infection.36 Criteria to establish the diagnosis of congenital infection include identification
of infection within 48 hours of birth, lesions at birth, and
exclusion of other pathogens (e.g., cytomegalovirus, syphilis, toxoplasmosis). Neonates with congenital infection
also may have elevated levels of immunoglobulin M (IgM) in the
cord blood as well as viremia. Congenital infection is unusual, but it
was reported to have occurred in 9 (5%) of 192 neonatal infections.44 Symptomatic first-episode HSV infection also has been associated with preterm
birth, although the number of cases reported has been too small
to permit an accurate assignment of the risk. Preterm delivery occurred
in as many as 33% to 56% of women with symptomatic primary infection
during pregnancy,36, 37 including four of five cases involving a primary third-trimester infection.36 Women with an asymptomatic first-episode HSV infection during pregnancy
seem to have no increased rate of adverse outcome.45 Pregnant women possibly have an increased risk of disseminated HSV infection. Dissemination
of HSV can cause serious hepatic and central nervous
system disease. Signs of disseminated HSV in the mother should be
treated with intravenous acyclovir to reduce the severity of maternal
disease. Management of Recurrent Herpes Simplex Virus in Pregnancy Pregnant women with recurrent HSV often have periodic outbreaks of genital
HSV, and their outbreaks may even become slightly closer together
in late pregnancy, but there is a limited chance of an HSV outbreak at
the time of labor. Cesarean section for delivery is indicated only for
an active HSV lesion, but only about 1 in 500 women in labor have HSV
in the genital tract from a recurrent HSV lesion.28 Cesarean section is unnecessary for the patient with prior recurrent HSV
who has no prodromal symptoms and no lesions at the onset of labor
or with rupture of membranes because the rate of neonatal infection is
so small (see Table 2). Women with a history of recurrent HSV should be reassured that both
cesarean and neonatal infection are uncommon in this setting. The routine practice of weekly viral cultures in the last month of pregnancy
does not predict asymptomatic viral shedding among patients known
to have had genital HSV.35, 41 The previous practice of weekly viral cultures for women with known recurrent
HSV is no longer recommended.46 None of the rapid tests to identify HSV in the asymptomatic woman without
lesions is sensitive enough to exclude reliably the presence of HSV, and
rapid tests are not indicated for women in labor. It is possible
that oral acyclovir suppresses recurrent HSV or asymptomatic shedding, but
the use of acyclovir is not recommended for this purpose because
of the lack of data on efficacy and safety.42 Symptomatic infection is reduced with the use of acyclovir, but asymptomatic
shedding still can occur.47 Patients with prior recurrent HSV should be advised to report to the hospital
early in labor and shortly after membranes rupture for a detailed
physical examination to exclude the presence of herpetic lesions. All
women with a known history of prior HSV infection require a detailed
physical evaluation during labor or upon rupture of membranes to establish
whether or not HSV lesions are present. It has been recommended
that HSV cultures be obtained from women with a history of prior HSV, even
in the absence of lesions.42 The quality of these screening cultures for HSV in the absence of lesions
has not been established and do not represent the standard of care. A
single Dacron applicator can be used to swab the cervix, the vulva, and
the genital area usually involved in recurrent lesions. The applicator
is placed in transport media and processed as a single culture. A
negative culture is reassuring. A positive culture identifies neonates
potentially exposed to HSV, but there is a 48-hour waiting period
for culture results. It is not known whether prophylactic treatment of
asymptomatic infants exposed to HSV is of benefit. These infants do benefit, however, from
very close evaluation for early symptoms of neonatal
HSV. Some physicians also perform surface and oropharynx cultures
of the neonate.42 Serial cultures of high-risk infants after birth, including infants born
of mothers with an HSV-positive culture taken during labor, also are
recommended by some pediatricians. An HSV-positive culture from the
neonate 24 hours after delivery is considered evidence of neonatal infection, rather
than contamination from secretions from the mother. These
infants should be considered candidates for acyclovir treatment. Approximately 1 in 250 neonates exposed to an active recurrent HSV lesion
will acquire HSV (see Table 2), and cesarean section is recommended for the few patients with recurrent
HSV who have active genital lesions at the time of labor or rupture
of membranes. Cesarean might also be considered if typical prodromal
symptoms are present in patients with no genital lesions. Cesarean section
should be done as soon as possible after membrane rupture. Cesarean
delivery within 4 to 6 hours of membrane rupture reduces the rate
of HSV transmission to neonates of mothers with an active recurrent HSV
lesion.37 Cesarean section does not prevent all cases of neonatal HSV, even when
it is performed within 4 to 6 hours of membrane rupture or even before
membrane rupture. In a recent survey, 15 (8%) of 184 infants with neonatal
HSV were delivered by cesarean before membrane rupture.13 However, patients with active lesions generally are delivered by cesarean
section, even after a prolonged period of membrane rupture, because
cesarean delivery may slightly reduce HSV transmission to the neonate. If
the cesarean section is performed 6 hours or more after membrane
rupture, the mother and neonate should be cultured for HSV and counseled
that neonatal infection could still occur. Management becomes problematic for the patient in labor with an active
extragenital HSV lesion present some distance from the genital area. Viral
shedding from the cervix occurs in approximately 2.5% of women with
an extragenital lesion, which is similar to the 3% rate of cervical
HSV shedding in patients with recurrent vulvar lesions.34, 39, 41 Extragenital HSV lesions can be covered, and because the risk of cervical
shedding is estimated to be only 2.5%, vaginal delivery can be considered; however, because
the estimated risk of neonatal herpes is still 1:800 (see Table 2), there is no unanimous opinion on the management of delivery in these
cases. Many experts recommend that a cesarean section be performed when
extragenital lesions are present, and this recommendation appears prudent.42 The management of women with active recurrent HSV and membrane rupture
before term is particularly difficult. The physician must weigh the chance
of a preterm infant developing an ascending HSV infection versus
the chance of serious morbidity or death from respiratory distress syndrome. If
the fetus has a low chance of respiratory distress syndrome (i.e., the fetus is more than 34-weeks' gestation) an immediate cesarean section
would be appropriate. If the fetus is very immature and has a high
chance of either respiratory distress syndrome or death, or both, all
of the following can be considered: - Delaying delivery until betamethasone has been given (even when there has
been a membrane rupture)
- Performing a cesarean section, relying on the administration of topical
surfactants to the neonate after birth
- Administering acyclovir therapy to reduce the chance of neonatal HSV exposure.
None of these three management regimens have been used extensively, and
each case requires individual consideration. A summary of the management
of patients with recurrent genital HSV is provided in Table 3.42 TABLE 3. Considerations for the Management of Pregnant Women with Recurrent
Genital Herpes
- Sequential cultures during late gestation are not indicated unless they
are done after clinically evident recurrent infection to document cessation
of virus excretion.
- Cesarean delivery should not be suggested as a “prophylactic” measure
to women who do not have active genital lesions at delivery.
- Symptomatic recurrences of genital herpes during the third trimester will
be brief; vaginal delivery is appropriate if no active lesions are
present at delivery.
- Obtaining specimens for culture at delivery from women with a history
of HSV infection may help in identifying exposed infants, but the clinical
benefit of this procedure has not been established.
- Options for the management of women with active genital herpes at onset
of labor:
- If the membranes have ruptured, the mother is afebrile, and the fetal lungs
are immature, the following steps may be taken:
- Delay delivery until betamethasone can be given.
- Proceed with cesarean delivery and give topical surfactant to the infant
or
- Manage the patient expectantly with or without acyclovir therapy.
- If term gestation or fetal lung maturity has been established and the membranes
have ruptured, cesarean delivery is indicated.
*Because the value of acyclovir therapy is not known, its routine use is
not indicated.
(Prober CG, Corey L, Brown ZA et al: The management of pregnancies complicated
by genital infections with herpes simplex virus. Clin Infect Dis 15:1031, 1992)Management of Primary Herpes Simplex Virus Infections in Pregnancy Neonatal exposure to a primary maternal HSV infection results in a 50% rate
of neonatal HSV. First-episode infection is associated with a large
number of viral lesions, often comprising a large surface area, and
a 90% chance of cervical HSV infection. Thus, the neonate must be protected
from exposure to primary HSV. Cesarean section should be offered
to all women with active first-episode HSV, even if it was a nonprimary
first-episode infection. Transplacentally acquired heterologous antibodies
to HSV-1 does little to influence the acquisition of HSV-2 by
the neonate.28 Cesarean section should be done immediately in the patient has a membrane
rupture. Although most infections of the neonate have occurred after
the membranes have been ruptured for more than 4 to 6 hours, neonatal
infection after exposure to primary infection has occurred when cesarean
section was performed within 4 to 6 hours of membrane rupture.36, 41, 42 When the fetus is mature, most physicians will still perform cesarean
section beyond 6 hours of membrane rupture for primary HSV because the
neonatal infection rate is so high that any incremental reduction of
this attack rate would be of benefit.39 The guidelines for women with rupture of membranes in the preterm infant
are similar between those with primary and recurrent infections. The
much-increased chance of neonatal infection in pregnancies complicated
by primary versus recurrent infection, however, needs to be considered; in
cases of primary infection, there is an increased pressure toward
early delivery.42 Women with a first-episode HSV infection earlier in pregnancy should have
a viral culture to document that HSV is the cause of the lesions and
to type the strain as either HSV-2 or HSV-1. Accurate serology testing
is available in only a few areas of the United States. Commercial serologic
testing is discouraged because of inaccuracies in distinguishing
between HSV-1 antibody and HSV-2 antibody.48, 49 Accurate differentiation of these antibodies has a potential advantage
because many first-episode infections will be nonprimary ones, which
are associated with less maternal morbidity and fewer complications of
pregnancy than are primary infections. The patient with a primary infection
should be advised of the limited data on spontaneous abortion, congenital
infection, growth retardation, and preterm labor. It is not
known whether acyclovir reduces this morbidity.42 Amniocentesis for viral culture is not indicated because of the poor predictive
value of the results.42 To date, the data are too limited to use culture evidence as an indication
for the presence of congenital infection: Congenital infection has
occurred despite a negative amniotic fluid culture, and it has not occurred
in some persons with a positive amniotic fluid culture. A tenfold increased risk of asymptomatic shedding occurs for several months
after a primary infection, compared with infections that have occurred 6 to 12 months
or more earlier. 36, 41, 50 This high risk of viral shedding immediately after a primary infection
means that HSV may still be present in the cervix or genital area if
the patient goes into labor within 2 months of a primary HSV infection. Patients
with primary genital HSV late in pregnancy should be followed
with weekly cervical or vaginal cultures, or both. If viral shedding
stops by the time of delivery and no lesions are present, vaginal delivery
can be undertaken with the precautions in place for recurrent HSV, including
careful examination for lesions and cultures of patients
without lesions. A cesarean section should be performed, however, for
the patient in labor with a recent primary HSV infection if viral shedding
continues until delivery or if lesions are present. It may be wise
to favor a cesarean section for women with a primary HSV infection
within 4 to 6 weeks of delivery, given the difficulties in predicting
HSV shedding immediately after a primary episode.42 These guidelines are not intended to represent a standard of care but
are offered based on our experience. Prevention During Pregnancy Prevention of a primary episode of HSV during pregnancy would reduce the
stage of HSV infection to which the infant is most susceptible. Sexual
contact obviously should not occur when the sexual partner has an active
HSV lesion or prodromal symptoms where the risk of infection increases
further. Sexual contact with condom protection could resume once
the lesions have healed. Condom use also should be considered for any
sexual contact with a male partner with a known history of genital herpes
to protect the pregnant woman from acquiring HSV during asymptomatic
shedding by the male partner. There is a small but definite rate
of primary HSV during pregnancy transmitted even by stable asymptomatic
partners. In a study of private obstetric patients, 32% of women and 25% of
their husbands had antibody to HSV-2. Two thirds of the women
with HSV antibody had no history of genital herpes. Of the 190 couples, 73% were
serologically concordant; of these couples, 57% were both antibody
negative and 16% were both antibody positive. The discordant couples
had been sexually intimate for a mean of 6 years.12 Annually, 7% to 10% of sexual partners are expected to acquire genital
herpes from a partner with a history of HSV, and two in three infections
will be acquired from an asymptomatic sexual partner.12, 51 Unfortunately, the majority of sexual partners with latent HSV have always
been asymptomatic and are unaware that they have ever had HSV in
the first place. Rate of condom use by the asymptomatic male with no known
history of prior HSV would be very low. Thus, prevention of much
of HSV infection becomes problematic short of abstinence. Postnatal infection appears to account for approximately 10% of neonatal
HSV. The higher rate of HSV-1 isolation from neonatal infection (30%) than
from genital infection (10% to 25%), is suspected to be related
to postnatal transmission from adults with active oral HSV-1. Parents, other
relatives, friends, and hospital personnel with active oral labial
HSV should avoid direct contact with newborns when they have active
lesions, and they should wash their hands with soap to reduce the chance
of HSV transmission to the newborn from free virus contaminating
the hands. Because postnatal HSV infection is preventable, all attempts
should be made to reduce exposure by advising unsuspecting adults about
the contagious nature of active oral lesions. Neonatal Morbidity and Mortality Neonatal HSV is one of the most life-threatening of all infections in newborns. Disseminated illness is associated with the greatest risk of death, the death rate ranging
from 57% to 82%.32, 33 Antiviral therapy does little to modify this risk.32 The morbidity and mortality of neonatal HSV are influenced by several
factors, classified into four groups: - Infection localized to the skin, eyes, or mouth (15%)
- Infection primarily localized to the central nervous system (20% to 30%)
- Disseminated infection with or without central nervous system involvement (70%)
- Asymptomatic infection (rare).
Neonatal death depends most on the location of infection (Fig. 5). High death rates (60% to 80%) occur with disseminated infection, moderate
rates (20% to 30%) with local central nervous system infection, and
low rates (15%) with HSV localized to the skin or eye. Death rates
are increased with concomitant prematurity, reduced level of consciousness, and
disseminated intravascular coagulopathy.32 Permanent neurologic sequelae are common among survivors of neonatal HSV, but
sequelae vary with the invasiveness of infection. The neurologic
impairment in survivors is 63% after disseminated disease, 41% after
localized central nervous system infection, and 6% after localized skin
infections.32 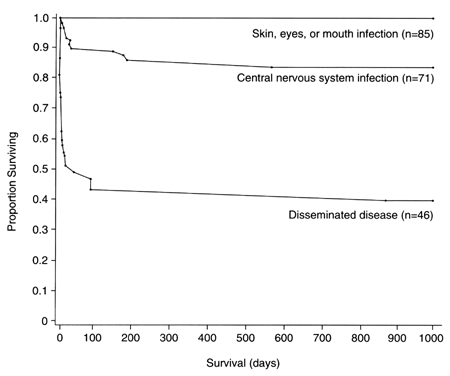 Fig. 5. Survival of infants with neonatal herpes infection, according to the extent
of disease (p < 0.001 for all comparisons) Fig. 5. Survival of infants with neonatal herpes infection, according to the extent
of disease (p < 0.001 for all comparisons)
|
Inadvertent exposure of the neonate to HSV at delivery should lead to prompt
notification of the primary caregiver and the parents. If exposure
to a recurrent infection occurred, the rate of neonatal infection is
low. The parents should be advised of symptoms and signs of lethargy, poor
feeding, or lesions (Table 4). If exposure occurred to a primary maternal infection, where the risk
of neonatal infection is high, it is advised that viral cultures be taken
of the eyes, throat, urine, stool, and cerebrospinal fluid.42 Some physicians culture the infant's eyes, nose, mouth, and skin
weekly thereafter for 4 to 6 weeks (see Table 4). Therapy using acyclovir is recommended if any HSV culture is positive 48 hours
after delivery or the cerebrospinal fluid is abnormal.42 Acyclovir can be given prophylactically to an inadvertently exposed infant, but
acyclovir appears to have limited ability to prevent all of
the manifestations of neonatal infection. Acyclovir needs to be studied
in this situation before it can be routinely recommended. The infrequency
of transplacental passage of HSV and the problems of diagnosis of
congenital HSV make identification and management of possible congenital
HSV experimental. TABLE 4. The Management of Infants Exposed to Herpes at Delivery
- If the infant has been exposed to a first episode of maternal genital
herpes, the following steps are recommended:
- Obtain specimens of urine, stool, and CSF as well as specimens from the
eyes and throat for culture.
- Instruct parents to report signs of lethargy, poor feeding, fever, or lesions.
- If the result of a culture performed for an infant >48 hours of age
is positive or if findings on examination of CSF are abnormal, initiate
therapy with IV acyclovir.
- If the infant has been exposed to recurrent maternal genital herpes, the
following steps are recommended:
- Instruct parents to report signs of lethargy, poor feeding, fever, or lesions.
- Perform weekly surveillance cultures of specimens from the eyes, nose, mouth, and
skin for 4–6 weeks after delivery on an optional basis.
CSF = cerebrospinal fluid.
(Prober CG, Corey L, Brown ZA et al: The management of pregnancies complicated
by genital infections with herpes simplex virus. Clin Infect Dis 15:1031, 1992) |