Intraperitoneal Fetal Transfusion In 1961, induced early delivery could not be carried out earlier than 31 to 32 weeks' gestation
without encountering prohibitive mortality from
prematurity and severe Rh disease. Eight percent of fetuses become hydropic before 32 weeks' gestation. The
introduction by Liley49 in 1963 of fetal intraperitoneal transfusion (IPT) completely altered
the outlook for these severely affected fetuses. Physiology It has been known since the turn of the century that RBCs placed in the
peritoneal cavity are absorbed and function normally. At one time, IPT
was a favorite method of transfusing children with thalassemia. It was
abandoned in favor of vascular transfusion because of the severe discomfort
that it caused. Absorption is through the subdiaphragmatic lymphatic
lacunae, up the right lymphatic duct, into the venous circulation. Diaphragmatic
movements are necessary for absorption to occur.50 In the absence of hydrops, 10% to 12% of infused RBCs are absorbed daily. The
presence of ascites does not prevent absorption,51 although the rate of absorption is more variable. When the hydropic fetus
is moribund and not breathing, no donor RBCs are absorbed.50 Peritoneal capacity limits the volume of RBCs infused. If the volume is
such that intraperitoneal pressure exceeds umbilical venous pressure, placental
blood flow to the fetus stops, and the fetus dies.52 In nearly every case, RBC volumes infused safely can be calculated by
the following formula: gestation in weeks minus 20 multiplied by 10 mL (i.e., 40 mL at 24 weeks, 80 mL at 28 weeks). Accurate calculation of donor hemoglobin concentration in the fetus at
any time after transfusion allows appropriate spacing of IPT and selection
of the proper gestation (after 33 to 34 weeks) for delivery. After
IPT, 85% of the transfused RBCs are in the fetoplacental circulation. Residual
donor hemoglobin levels in the fetus may be estimated within 1.5 g/dL 95% of
the time using the following parameters: 0.85 times
the fraction of donor RBCs in the fetoplacental circulation; fetal weight
at the gestation donor hemoglobin levels; 125 mL/kg as fetoplacental
blood volume; and 1/120 of the donor hemoglobin infused as the daily
attrition rate of donor RBCs. For example, the donor RBC hemoglobin
concentration at 28 weeks' gestation (fetal weight estimated to be 1150 g) 10 days
after intrauterine transfusion (IUT) of 65 mL of donor RBCs
with a hemoglobin concentration of 30 g/dL can be determined as follows: 
After subsequent IPT, estimations of residual circulating donor hemoglobin
concentrations for each fetal transfusion at any period after the
last transfusion can be made and added together to give the estimated
donor hemoglobin level at that gestation. These calculations can be used to determine the time for the next fetal
transfusion (or for delivery), with the requirement that the donor hemoglobin
level be kept above 10 to 11 g/dL at all times. Because one transfusion
rarely raises donor hemoglobin levels above 10 g/dL, a second
transfusion is carried out 9 to 12 days after the first. Subsequent
transfusion intervals are 3.5- to 4-week intervals, with the last transfusion
rarely being given after 32 weeks' gestation. Delivery is carried
out 3.5 to 4 weeks after the last transfusion, usually between 34 and 36 weeks' gestation. Blood for Intrauterine Transfusion Group O, Rh-negative, Kell-negative RBCs are used for IUT if the alloantibody
is anti-D. If the alloantibody is non-D in the Rh system, group
O, Kell-negative, maternal serum compatible RBCs are used. If the alloantibody
is outside the Rh system, group O, Rh-negative, Kell-negative
RBCs are used that are missing the offending antigen to which the mother
is alloimmunized. The blood should have been drawn from the donor
within 48 to 96 hours of the transfusion. The donor RBCs are carefully
cross-matched against maternal serum; these women are prolific antibody
producers, some having as many as four to six alloantibodies other
than their primary antibody. In such circumstances, finding a compatible
donor may be difficult. Before the transfusion, the unit is tightly
packed, and all plasma and buffy coat are removed. Immediately before
the IUT, 10 to 15 mL of sterile, isotonic saline is added so that the
hemoglobin and hematocrit of the packed RBCs are 27 to 30 g/dL and 85% to 90%, respectively. The donor unit must be human immunodeficiency virus negative, hepatitis
C virus negative, and HBsAg negative and should be irradiated, although
the risk of graft-versus-host disease occurring if nonirradiated blood
is used is small. The Intrauterine Transfusion Team The number of candidates for IUT is decreasing as the number of immunized
Rh-negative women declines because of the success of Rh prophylaxis. IUT
techniques, either intraperitoneal or direct intravascular, appear
simple, but they are not. Complete management of the immunized mother
and baby, before and after birth, requires not only an experienced
obstetrician but also neonatal, ultrasound, and laboratory personnel and
services of the highest order. Procedures should be carried out only
in a tertiary-level perinatal center. A team approach is essential. Each
center in which fetal transfusions are performed should treat at
least five or six fetuses annually, on whom 15 to 20 transfusions are
carried out. To reach this volume of patients, the IUT team must have
all transfusion candidates referred from a population base of 6 million (75,000 to 90,000 deliveries per year). Only under such circumstances
can the team's expertise in IUT and overall management of severely
affected fetuses and infants with hemolytic disease be maintained. Fetal Transfusions of the Hydropic Fetus Although the goals are to predict impending hydrops and to prevent it by
beginning fetal transfusions before hydrops has developed, in 28% to 33% of
cases, hydrops is present at first transfusion because of late
referral, or it develops between the first and second IPTs because one
IPT does not raise donor hemoglobin levels high enough to shut off hepatic
erythropoiesis and interrupt the hepatic chain of events that leads
to the development of ascites and hydrops fetalis. Although direct intravascular transfusion (IVT) is the procedure of choice
when hydrops is encountered, significant salvage of hydropic fetuses
has been produced with IPT in the past. In the ultrasound era in Winnipeg, 60% of
hydropic fetuses (18 of 30) were salvaged with IPT; however, none
of eight moribund, nonbreathing hydropic fetuses subjected
to IPT survived. Hydrops fetalis may be reversed by fetal transfusions. Reversal is more
common when hydrops is found at the second IPT for the first time. Reversal
is common when hydrops is encountered at the first direct IVT. As
donor hemoglobin levels rise after transfusion, erythropoietin levels
fall. If the fetus remains alive, hepatic erythropoiesis diminishes, intrahepatic
circulation improves, portal and umbilical venous pressures
fall, hepatocellular function improves, serum albumin levels rise, and
ascites and fetal anasarca disappear. Direct Intravascular Fetal Transfusion Pioneering attempts at direct IVT into either a fetal or placental vessel
approached through a hysterotomy incision were attempted in the mid-1960s.53,54,55 The results were abysmal because the women almost invariably went into
labor. In 1981, Rodeck and associates56 introduced direct transfusion through a fetoscope. Few others have achieved
his skill with the fetoscope. Blood, meconium, or turbidity in the
amniotic fluid makes fetoscopic visualization of the fetal blood vessels
impossible. With the advent of fetal blood sampling, by the early
to mid-1980s44 it became possible to follow the sampling procedure with direct IVT.57,58,59,60,61 Under ultrasound guidance, the tip of a 22- or 20-gauge spinal needle
is introduced into a umbilical blood vessel, preferably the vein but occasionally
the artery, at its insertion into the placenta and rarely
at its insertion into the fetal abdomen. Advantages of Fetal Blood Sampling and Intravascular Fetal Transfusion As stated earlier, in the absence of hydrops, direct measurement of fetal
blood parameters is the most accurate method of determining the severity
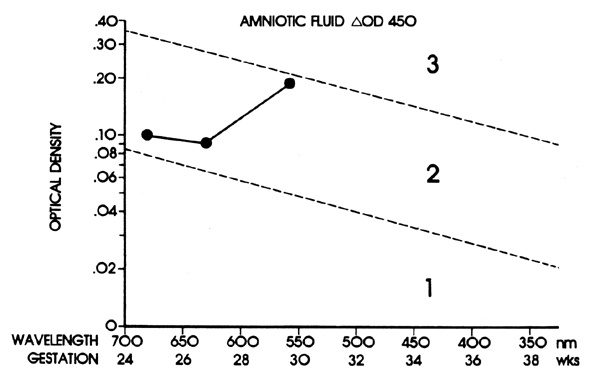
of hemolytic disease, far more accurate than amniotic fluid ΔOD 450 measurements
and ultrasonographic assessments. The latter two
parameters, however, combined with past history and maternal antibody
titers, can identify the fetus at risk who requires a fetal blood sampling
procedure. Direct IVT, as a method of transfusing the severely affected fetus, does
not rely on diaphragmatic movement to increase hemoglobin levels. It
is, therefore, capable of salvaging the moribund nonbreathing fetus, provided
that the fetus still has umbilical blood flow. Also, direct IVT
raises circulating hemoglobin levels in the fetus immediately, rather
than taking the 8 to 10 days required for IPT. Selection of Patients Fetal transfusion is hazardous for the fetus. Only fetuses proven to be
at high risk of hydrops or hydropic before 33 to 34 weeks' gestation
should undergo fetal transfusion. Patients are selected for IPT on the
basis of amniotic fluid findings. Women undergoing amniocentesis are
selected on the basis of history and antibody criteria already outlined. If
the indications are present, amniocentesis should be carried out
initially as early as 18 to 19 weeks' gestation. IPT is indicated when
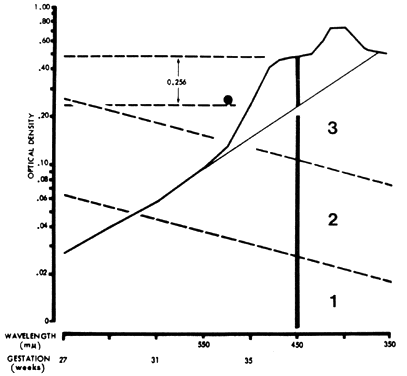
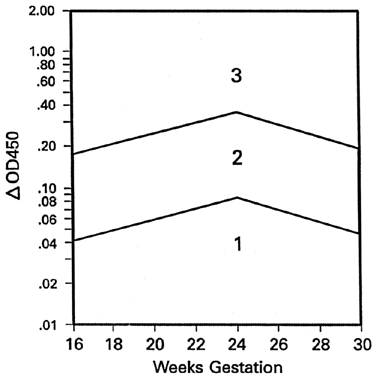
serial ΔOD 450 readings reach the upper 80% to 85% area of zone 2 before 30 weeks' gestation (see Fig. 8) or reach zone 3 between 30 and 33 weeks' gestation. Fetal transfusions
may be indicated at somewhat lower readings if serial ΔOD 450 readings
show a sharp slope rising toward the zone 3 boundary, indicating
rapidly progressive disease. IVT, the preferred transfusion method, is indicated if, at fetal blood
sampling, the circulating hemoglobin concentration in the fetus is less
than 10 to 11 g/dL. Fetal blood sampling is carried out when serial ΔOD 450 readings
reach the 65% to 75% level of zone 2 at any gestation
or when the placenta is anterior and maternal history or antibody
titer place the fetus at risk. Technique of Intraperitoneal Transfusion Real-time ultrasonography is used to determine the site for the needle
insertion on the maternal abdomen and the depth that the needle must be
inserted to enter the fetal peritoneal cavity. After aseptic and local
anesthetic preparation, the operator directs the needle (an 18-cm, 16-gauge
Tuohy needle) through a 5-mm maternal skin incision into the
fetal peritoneal cavity under direct ultrasound guidance, having selected
the site and depth of needle insertion on the basis of real-time ultrasound
assessment. Often, but not invariably, the real-time ultrasound
image shows the needle tip in the fetal abdomen. As the needle enters
the fetal abdominal wall, the operator may note a feeling of resistance
that disappears as the tip enters the peritoneal cavity. The stylet
is removed, and an epidural catheter with its tip and side holes removed
is passed down the needle. If the catheter passes through the end
of the needle, it is lying free in a cavity, which should be the fetal
peritoneal cavity (Fig. 13). Twenty-five to 30 cm of catheter is passed down the needle. The needle
is pulled back over the catheter to lie on the maternal abdomen, and 1 to 2 mL
of radiopaque contrast medium is injected through the catheter. An
anteroposterior radiograph is taken. If the catheter lies free
in the peritoneal cavity, the contrast-filled catheter is seen within
the peritoneal cavity, and contrast appears within the cavity, outlining
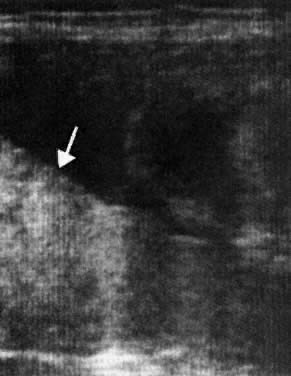
the negative shadow of liver and semilunes of small bowel (Fig. 14). 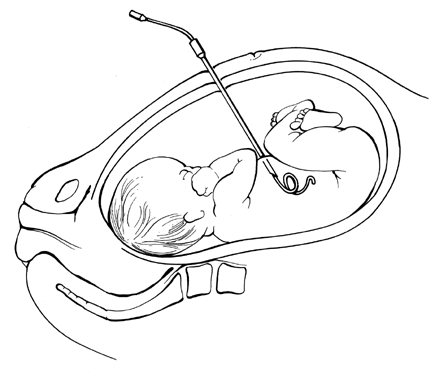 Fig. 13. Intrauterine transfusion. The Tuohy needle has been inserted across the
maternal abdominal wall and uterine wall into the fetal peritoneal cavity, and
the epidural catheter has been threaded into the peritoneal
cavity of the fetus. The safest position for the fetus at intrauterine
transfusion is not with its abdomen anterior (as shown in this diagram) because
the umbilical vessels will lie in the center of the target
area.(Bowman JM: Blood-group incompatibilities. In Iffy L, Kaminetzky HA s]: Principles
and Practice of Obstetrics and Perinatology, p 1193. New
York, John Wiley & Sons, 1981) Fig. 13. Intrauterine transfusion. The Tuohy needle has been inserted across the
maternal abdominal wall and uterine wall into the fetal peritoneal cavity, and
the epidural catheter has been threaded into the peritoneal
cavity of the fetus. The safest position for the fetus at intrauterine
transfusion is not with its abdomen anterior (as shown in this diagram) because
the umbilical vessels will lie in the center of the target
area.(Bowman JM: Blood-group incompatibilities. In Iffy L, Kaminetzky HA s]: Principles
and Practice of Obstetrics and Perinatology, p 1193. New
York, John Wiley & Sons, 1981)
|
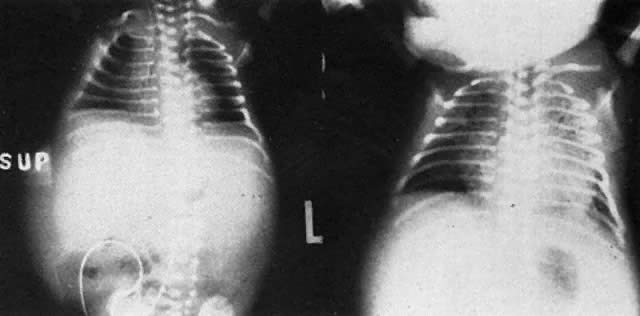
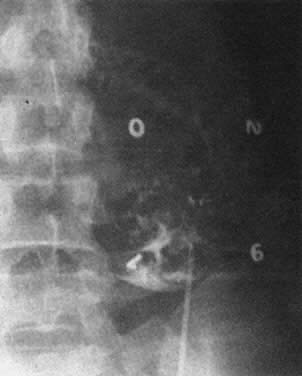 Fig. 14. Successful catheterization of the fetal peritoneal cavity as shown by radiopaque
contrast medium in the fetal peritoneal cavity, outlining negative
shadows of small bowel.(Bowman JM: Maternal blood group immunization. In Creasy RK, Resnik R s]: Maternal-Fetal
Medicine: Principles and Practice, pp 561–602. Philadelphia, WB
Saunders, 1984) Fig. 14. Successful catheterization of the fetal peritoneal cavity as shown by radiopaque
contrast medium in the fetal peritoneal cavity, outlining negative
shadows of small bowel.(Bowman JM: Maternal blood group immunization. In Creasy RK, Resnik R s]: Maternal-Fetal
Medicine: Principles and Practice, pp 561–602. Philadelphia, WB
Saunders, 1984)
|
Diagnosis of fetal ascites should be made by ultrasound before the fetal
transfusion. Ascitic fluid obtained at first transfusion is characteristically
bright yellow, clear, and slightly viscous because of its protein
content. It looks different from the dull yellow, slightly turbid, less
viscous amniotic fluid. If ascites is present at second and subsequent
fetal transfusions, it is mixed with residual donor blood to
various degrees. When ascites is present, a radiograph taken after injection
of contrast medium reveals characteristic diffusion of dye into
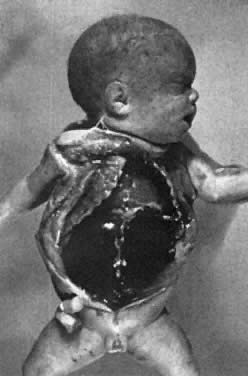
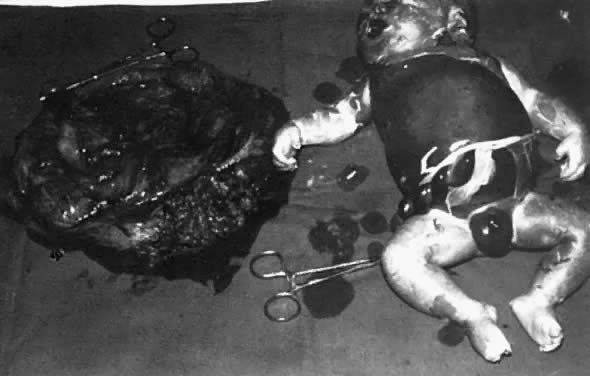
the ascitic fluid, with no visible small bowel semilunes or other landmarks (Fig. 15). 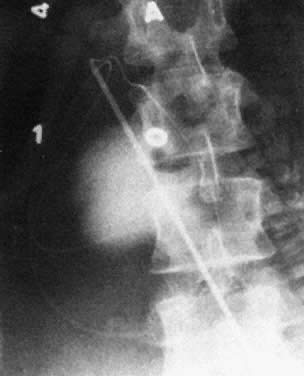 Fig. 15. Hydrops fetalis at intrauterine transfusion. Note gross ascites at first
transfusion (31.5 weeks' gestation), which was more severe at second
transfusion 4 days later (not shown). Severe residual hydrops was seen
at emergency cesarean section (33.5 weeks' gestation). Cord hemoglobin, 6.2 g/100 mL (99% donor in origin); cord bilirubin, 5.4 mg/100 mL. Five
exchange transfusions, respirator care for 4 days, and 3 weeks of
intensive nursery care were required.(Bowman JM: Maternal blood group immunization. In Creasy RK, Resnik R s]: Maternal-Fetal
Medicine: Principles and Practice, pp 561–602. Philadelphia, WB
Saunders, 1984) Fig. 15. Hydrops fetalis at intrauterine transfusion. Note gross ascites at first
transfusion (31.5 weeks' gestation), which was more severe at second
transfusion 4 days later (not shown). Severe residual hydrops was seen
at emergency cesarean section (33.5 weeks' gestation). Cord hemoglobin, 6.2 g/100 mL (99% donor in origin); cord bilirubin, 5.4 mg/100 mL. Five
exchange transfusions, respirator care for 4 days, and 3 weeks of
intensive nursery care were required.(Bowman JM: Maternal blood group immunization. In Creasy RK, Resnik R s]: Maternal-Fetal
Medicine: Principles and Practice, pp 561–602. Philadelphia, WB
Saunders, 1984)
|
When the contrast injection radiograph demonstrates the catheter lying
in the fetal peritoneal cavity (see Fig. 14), the transfusion is started. The infusion is carried out in 10-mL aliquots, with
each 10-mL injection taking 3 to 6 minutes. The syringe is
rinsed after each injection. The fetal heart rate is monitored by Doppler
ultrasound at the end of each 10-mL injection and continuously for
the last 10 to 15 mL of the infusion. The volume infused is calculated
according to the following formula: gestation (in weeks) minus 20 multiplied
by 10 mL. If the fetus is in good condition, fetal heart rate
increases to 160 to 190 beats/min during the procedure. Fetal bradycardia
early in the transfusion, a rare event, is ominous, indicating the
probability of transfusion trauma and fetal death. Fetal bradycardia
toward the end of the transfusion is uncommon and is an indication for
prompt termination of the procedure because intraperitoneal pressure
may be approaching umbilical venous pressure. After completion of the procedure, the catheter is removed slowly with
continuous heart rate monitoring. Marked bradycardia of vagal origin may
occur. Further catheter removal should be delayed until the fetal heart
rate improves. The needle site is swabbed with iodine, and a small
dressing is applied. Fetal heart rate is monitored periodically after
the transfusion. Fetal movement may be markedly reduced for the first 24 hours
after the procedure. The mother should be reassured that this
is not an ominous sign. She usually can be discharged 24 to 36 hours
after the procedure. If gross ascites is noted (see Fig. 15), every effort should be made to aspirate a significant quantity of fluid
through the needle before the catheter is passed. After passage of
the catheter and injection of contrast, if the postinjection radiograph
reveals a considerable amount of residual ascitic fluid, a further
volume of fluid (as much as possible) should be aspirated through the
catheter. If a greater volume of ascitic fluid is aspirated than the planned RBC
volume to be injected, the total volume to be transfused may be increased
but should remain 15 to 20 mL less than the volume of ascitic fluid
removed. IPT is no longer recommended for treatment of the hydropic
fetus; direct IVT is the preferred method of transfusion. Indeed, direct
IVT is now the preferred route for all fetal transfusions. Problems With Intraperitoneal Transfusion Although IPT represented a major advance in the management of severe erythroblastosis
fetalis, it was associated with some serious problems. The
procedure is of no value for the nonbreathing moribund hydropic fetus. In
this situation, RBCs are not absorbed, and the fetus dies. Also, when
the placenta is implanted on the anterior uterine wall and must
be transfixed by the Tuohy needle to enter the fetal peritoneal cavity, the
traumatic death rate per procedure is 7%. Moreover, after IPT, there
is a 30% spontaneous labor rate per patient, with delivery earlier
than otherwise planned; however, most of such deliveries occur after 30 weeks' gestation. Seventy percent of fetuses born after spontaneous
labor survive. Finally, although serial amniotic fluid ΔOD 450 measurements
have increased the accuracy of prediction of severity
of hemolytic disease, inaccuracies do occur—the occasional only
moderately affected fetus despite a zone 3 reading and, less commonly, hydrops
despite only a moderate (60%) zone 2 reading. Technique of Direct Intravascular Fetal Transfusion Because of the previously outlined problems with IPT, direct IVT has become
the fetal transfusion procedure of choice. When it has been decided
from amniotic fluid and ultrasound assessment that fetal blood sampling
and direct IVT are required, the mother is heavily premedicated and
transferred to the fetal assessment and therapy unit. A highly skilled, experienced
perinatal ultrasonographer is an essential component
of the IVT group. Great care and considerable time are spent in identifying
the target blood vessel (Fig. 16). Packed RBCs used for IVT are of the same hemoglobin and maternal cross-match
compatibility as those used for IPT. Immediately before the planned
procedure, with great care, 8 to 10 mL of sterile saline is added
to the packed RBC unit. A blood transfusion set is inserted into one
of the outlet ports of the packed RBC unit. The side arm of a metal
stopcock is inserted into the other end of the transfusion tubing. A length
of sterile connector tubing is attached to the male end of the stopcock, and
a 10-mL glass syringe with finger-ring assembly is attached
to the other end of the stopcock. Packed RBCs are run down the transfusion
tubing through the stopcock into the extension tubing. The stopcock
lever is then set at an angle so that blood will not run down the
transfusion tubing in any direction. With a 45-degree turn of the stopcock
in one direction, the syringe may be filled with the packed RBC
unit; changing the lever 90 degrees then allows blood to be transfused
down the extension tubing. Packed RBCs treated in this manner can be
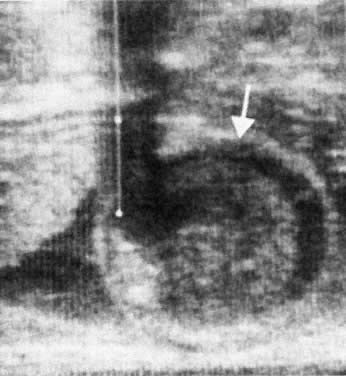
readily infused through a 22-gauge spinal needle without hemolysis. 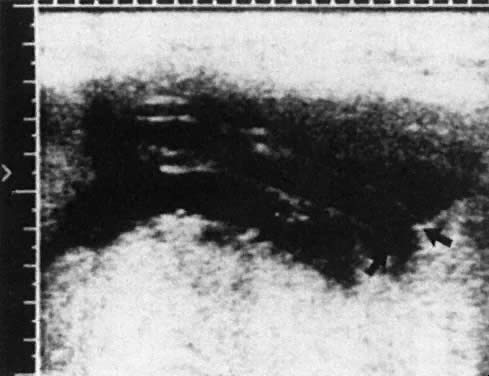 Fig. 16. Ultrasonogram of the insertion of the umbilical vein into the placenta ( arrows ), the target area for insertion of the needle at direct intravascular
transfusion.(Bowman JM: Maternal Blood Group Immunization. In Creasy RK, Resnik R (eds): Maternal-Fetal
Medicine: Principles and Practice, 2nd ed, pp 613–655. Philadelphia, WB Saunders, 1989) Fig. 16. Ultrasonogram of the insertion of the umbilical vein into the placenta ( arrows ), the target area for insertion of the needle at direct intravascular
transfusion.(Bowman JM: Maternal Blood Group Immunization. In Creasy RK, Resnik R (eds): Maternal-Fetal
Medicine: Principles and Practice, 2nd ed, pp 613–655. Philadelphia, WB Saunders, 1989)
|
After careful aseptic preparation of the maternal abdomen, the obstetric
surgeon drapes the maternal abdomen, infiltrates the skin with local
anesthetic, and inserts a 20- or 22-gauge spinal needle tip through the
maternal skin immediately over the target site selected previously
by the obstetric ultrasonographer. The ultrasound transducer, enclosed
in sterile plastic, is applied to the maternal abdomen immediately adjacent
to the needle insertion site. The ultrasonographer and venipuncturist
select a plane and position the transducer to allow simultaneous
ultrasonographic identification of the blood vessel target site and
the needle tip. The needle is guided toward the blood vessel, with appropriate
ultrasound directed corrections to keep the tip on target. In most instances, the needle tip can be identified as it penetrates the
target blood vessel. Because the needle tip is being advanced in a three-dimensional
plane using ultrasound, which is in two dimensions, the
operator cannot always be certain that the needle tip has been correctly
inserted into the lumen of the vein. When the tip is at the proper
depth, the needle stylet is withdrawn, and aspiration is attempted
using a lightly heparinized 1-mL tuberculin syringe. If nothing is obtained
or if amniotic fluid is aspirated, withdrawal, but not removal, of
the needle tip and reinsertion under ultrasound guidance are carried
out. If a free-flowing blood sample is aspirated, the tip of the needle
probably is positioned properly in the fetal blood vessel. At initial
fetal blood sampling, this may be confirmed by determining that the
blood is fetal by alkaline denaturation screening. After the first IVT, the
amount of donor blood present in the fetal circulation obscures
any alkaline denaturation test to confirm its fetal origin. Confirmation that the needle tip is placed properly is done easily and
accurately by injecting 0.5 mL of sterile saline. If the needle tip is
in the vein, streaming ultrasound turbulence is seen as the saline passes
down the vein. Conversely, if the needle tip is in the artery, the
turbulence of the saline may be seen moving in the opposite direction
onto the surface of the placenta. If turbulence can be seen in the amniotic
fluid, the needle tip is in the amniotic cavity. If no turbulence
can be seen, the needle tip may be dislodged and embedded in the umbilical
cord. In this situation, injection of blood should not be carried
out because life-threatening umbilical vein compression may occur. Only
if streaming of saline in the vessel can be seen should blood infusion
be begun. An immediate hemoglobin estimation is carried out on the fetal blood sample. Subsequently, complete investigation of the fetal blood sample is
carried out in the Rh Laboratory. If the immediate hemoglobin reading is below 10 to 11 g/dL, the IVT proceeds. If
fetal movements are likely to disturb the needle insertion (posterior
cord insertion), the fetus is paralyzed by the intravenous injection
of pancuronium. The obstetric venipuncturist inserts the filled
blood transfusion tubing connector into the needle hub and holds it
firmly. The obstetric ultrasonographer continuously watches the blood
flow turbulence in the fetal blood vessel. The transfusionist (the third
member of the transfusion team) transfuses the packed RBCs in 10-mL
aliquots. Because there is only 2 or 3 mm of needle tip within the fetal
blood vessel, which may be as small as 2 to 3 mm in diameter, the
tip is in danger of early dislodgment. For this reason, infusion of packed
RBCs is carried out as rapidly as possible. Each aliquot is transfused
in 1 to 2 minutes until the desired blood transfusion volume is
attained. The usual transfusion volume is 50 mL/kg of estimated nonhydropic
fetal weight. Because of the expansile placental vascular bed, large
RBC volumes transfused rapidly are tolerated. The ultrasonographer
monitors fetal heart rate and cardiac ventricular size. If there is
evidence of significant bradycardia or marked ventricular dilation (a
rare event), the transfusion is discontinued before the full volume is
administered. Because the aim is to shut off erythropoiesis so that
hepatosplenomegaly diminishes and hydrops is prevented or reversed, a
second procedure is carried out as soon as it is estimated that the total
circulating hemoglobin concentration has dropped into the range of 9 to 10 g/dL. The
average donor hemoglobin attrition rate is 0.4 g/dL/d. Subsequent
IVTs usually are required every 3 to 4 weeks after the
estimated residual circulating hemoglobin concentration has dropped into
the range of 9 to 10 g/dL. Intravascular Transfusion of a Hydropic Fetus In the presence of hydrops fetalis, IVT is preferred over IPT. IVT has
the potential for salvaging all hydropic fetuses, even those who are moribund
and not breathing, with the exception of those that are dying
and have no blood flow in their umbilical vessels. The procedure is not
different from IVT of a nonhydropic fetus, other than that the volume
administered in the first transfusion frequently is confined to 30 to 40 mL/kg
of estimated nonhydropic fetal body weight. When the transfusion has been completed, the needle is cleared with 0.5 mL
of normal saline. A 1-mL posttransfusion blood sample is withdrawn, again
into a heparinized tuberculin syringe. All blood samples, before
and after transfusion, are tested for hemoglobin, hematocrit, bilirubin, Kleihauer
fetal/adult RBC ratios, platelet counts, plasma protein, and
blood gas estimations. The initial pretransfusion blood sample
is blood grouped and direct Coombs' tested. Intrauterine Transfusion Survival Rates The Winnipeg overall salvage rate with IPT from January 1964 to October 1986 (the
last date that a fetus was given IPT alone) was 63% (222 of 353 fetuses
transfused 862 times). Survival rates with IPT and IVT in the modern ultrasound era are outlined
in Table 7, which shows IVT survival rates that are superior to IPT survival rates
in every category. More IVTs than IPTs are required per patient (4.3 versus 2.7), for
three reasons. First, IVTs are being carried out earlier
in gestation. Second, perinatal mortality per procedure is less, allowing
multiple procedures in more patients. Third, because of somewhat
smaller IVT volumes, the intervals between IVTs are modestly shorter
and the gestation of delivery moderately longer (37 to 38 weeks). TABLE 7. Intrauterine Transfusions (IUT): Winnipeg Ultrasound Era
| 204 IPT | 696 IVT |
| (July 1980-October 1986) | (May 1986-May 1997) |
Fetuses | 75 | 161* |
Survivors | 57 (76%) | 140 (88%) |
Nonhydrops | 45 | 111* |
Survivors | 39 (86.7%) | 104 (95%) |
Hydrops | 30 | 50 |
Incidence | 40% | 31% |
Survivors | 18 (60%) | 36 (72%) |
Nonmoribund hydrops | 22 | 31 |
Survivors | 18 (82%) | 24 (77%) |
Moribund hydrops | 8 | 19 |
Survivors | 0 (0%) | 12 (63%) |
IUT per fetus | 2.7 | 4.3 |
Risk per IUT | 3.5% | 0.8% |
*Two in utero are expected to survive.
IPT, intraperitoneal transfusion; IVT, intravenous transfusion.
Pros and Cons of Intravascular Transfusion and Intraperitoneal Transfusion If feasible, IVT is the procedure of choice. Only through IVT can the moribund, nonbreathing, hydropic fetus be salvaged; 12 of 19 (63%) such
fetuses in the Winnipeg series survived. What is gratifying is the much
lower overall risk with IVT than with IPT (0.8% versus 3.5% per procedure). Although the risk of IVT is materially less, this low risk can only be
achieved when the obstetric ultrasonographer and the obstetric venipuncturist
have great skill and experience with the procedure. Otherwise, there
are hazards that increase the risk of IVT: overtransfusion, with
cardiac failure (not observed in the Winnipeg series); exsanguination (a
problem, particularly if the fetus is hydropic, thrombocytopenic, and
the cord insertion posterior62); and inadvertent injection of blood into the cord around the vein, producing
a cord hematoma, compressing and interfering with umbilical blood
flow. This third hazard has caused fetal death and can be prevented
only by an alert, experienced obstetric ultrasonographer. Despite the great advantages of IVT, there are two situations in which
IPT may still be required; therefore, the skill in carrying out IPT must
be maintained. One is the rare situation, before 20 to 21 weeks' gestation, when
the cord vessels may be too small for successful venipuncture. The
second is the more common situation late in pregnancy (after 30 weeks' gestation) when, after several successful IVTs, increasing
fetal size totally obscures a posterior cord vessel insertion, making
venipuncture impossible. Development in Fetal Transfusion Survivors Physical and intellectual development in most fetal transfusion survivors
is normal. Because prematurity may be marked (as early as 27 weeks' gestation) and
residual severity of disease great, some problems may
be anticipated. In the Winnipeg Rh Laboratory, of 101 children tested
at 18 months or later, 87 appeared completely normal, 10 had minor developmental
delay (probably transient and due to prematurity), and 4 were
definitely abnormal: 1 had a normal IQ but spastic hemiparesis; 2 had
IQs of 75 and 85; and the 4th, in whom fetal hydrops had been reversed, had
cerebral agenesis that was probably unrelated to the hydrops. |