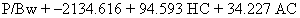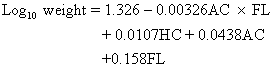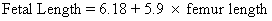A number of formulas for prediction of birth weight are now published in
the literature. The predictive accuracy of these formulas ranges from ± 14.8% to ± 20.2% (2 SD). This accuracy is also related
to the size of the fetus. Thus, the relative error is small (approximately 140 g
to 200 g) in a fetus weighing 1000 g, and large (480 g to 600 g) in
one weighing 4000 g.35,36 Interestingly, the accuracy of BW formulas may appear small if data are
reported in the form of mean % error because the sum of positive and
negative differences is taken into account. By contrast, the absolute
mean % error reflects the variability noted, regardless of sign and as
such is a more accurate predictor of differences from actual BW (Table 9). The format of mean % error can also be misleading because the difference
between actual and predicted weights is expressed in small numbers (1% to 2%) that
do not reflect the true variation in BW. For practical
clinical purposes the variation between predicted and actual BWs is
best expressed in the form of absolute mean % error per 1000 g (see Table 9). TABLE 9. A Comparison Of Mean % Error Versus Mean Absolute % in Reflecting
True Variation Between Actual Birth Weight (A/BW) versus Predicted
Birth Weight (P/BW)*
| A/BW - P/BW | % Error | A/BW - P/BW | % Error |
Fetus 1 | -150 g | -15% | 150 g | 15.0% |
Fetus 2 | + 140 g | + 14% | 140 g | 14.0% |
| -10 g | -1.0% | 290 g | 29.0% |
*Mean % error = -1.0%/2 = -0.5% or -5 g: 1000 g; absolute mean error = 29%/2 = 14.5% or 145 g: 1000 g. It is apparent that the mean % error can
be misleading because it artificially reduces the difference between
actual and predicted BW. Further, the answer is expressed in small numbers (1% to 2%) that
carry large SDs (7% to 10%). Expressing the absolute
mean error per 1000 g will be the most useful way for the practicing
obstestrician.
(Sabbagha RE: Intrauterine growth retardation. In Sabbagha RE [ed]: Diagnostic
Ultrasound Applied to Obstetrics and Gynecology, 2nd
ed, p 121. Philadelphia, JB Lippincott, 1987)Comparison of Birth Weight Formulas A variety of formulas are now used for prediction of fetal weight. Some
of these formulas incorporate BPD and AC whereas others are derived from
HC, and AC measurements, with or without FL data. The formula by Shepard and associates is based only on BPD and AC and yields
a predictive accuracy of ±20% (2DS).35,36 The limiting factors in this formula are the inclusion only of BPD and
AC rather than HC, AC and possibly FL, and the utilization of the same
formula for all fetuses, regardless of gestational age and growth status. These
limiting factors are discussed below: - Changes in head shape such as dolichocephaly or brachycephaly can sufficiently
change the true estimate of cephalic size by BPD.
- The contributions to BW by head size and body size, at different intervals
in pregnancy, are variable. For example, it is known that in the normal
preterm fetus the HC/AC ratio exceeds 1, whereas after 36 weeks' gestation
the ratio is reversed.37 It is also known that the same ratio is dependent on whether a given fetus
is symmetrically or asymmetrically small. Thus, cranial and trunk
proportional contributions to BW are dependent on fetal age as well as
on the specific state of altered growth.
In support of this reasoning Divon and associates found that in pregnancies
complicated by spontaneous rupture of the membranes the estimated
fetal weight by BPD and AC or by AC and FL underestimate BW by 12.4% and 13.2%. respectively.38 They attributed this finding to compression of the fetal head and trunk
observed in some pregnancies with oligohydramnios. Additionally, Weiner
and associates showed that BW in some fetuses with identical BPD and
AC values differed by as much as 1000 g and attributed this difference
to the two factors previously discussed.39 To avoid the errors introduced by changes in head shape and differences
in HC/AC ratios they introduced the concept of targeting BW formulas
to specific populations, for example, preterm or IUGR fetuses. Subsequently, in
a study involving women at high risk for preterm delivery they
derived a formula for prediction of size in low birth weight infants.39 Their formula, shown below, attaches greater significance to HC in these
small fetuses. 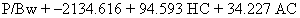
Preliminary data suggest that the prediction of BW in preterm and IUGR
fetuses is enhanced by the use of formulas targeted to such populations.39 In high-risk pregnancies knowledge of BW estimates in small fetuses is
often urgently needed to help in formulating management plans. Thus, time
constraints are placed on deriving these estimates by computer or
calculator, and use of tabulated data is helpful (Table 10). Table 10. Sonographic Prediction of Fetal Weight (200–2200g)*
Head | Abdominal Circumference (cm) |
Circumference (cm) | 13.5 | 14.0 | 14.5 | 15.0 | 15.5 | 16.0 | 16.5 | 17.0 | 17.5 | 18.0 | 18.5 |
13.5 | - | - | - | - | - | - | - | - | - | - | - |
14.0 | - | - | - | - | - | - | - | - | - | - | - |
14.5 | - | - | - | - | - | - | - | - | - | - | - |
15.0 | - | - | - | - | - | - | - | - | - | - | - |
15.5 | - | - | - | - | - | - | - | - | - | - | - |
16.0 | - | - | - | - | - | - | - | - | - | - | - |
16.5 | - | - | - | - | - | - | - | - | - | - | - |
17.0 | - | - | - | - | - | - | - | - | - | - | - |
17.5 | - | - | - | - | - | - | - | - | - | - | - |
18.0 | - | - | - | - | - | - | - | - | | - | 201.24 |
18.5 | - | - | - | - | - | - | - | - | 214.31 | 231.42 | 248.54 |
19.0 | - | - | - | - | - | 210.27 | 227.38 | 244.49 | 261.61 | 278.72 | 295.83 |
19.5 | - | - | 206.22 | 223.34 | 240.45 | 257.56 | 274.68 | 291.79 | 308.90 | 326.02 | 343.13 |
20.0 | 219.30 | 236.41 | 253.52 | 270.63 | 287.75 | 304.86 | 321.97 | 339.09 | 356.20 | 373.31 | 390.43 |
20.5 | 266.59 | 283.70 | 300.82 | 317.93 | 335.04 | 352.16 | 369.27 | 386.38 | 403.40 | 420.61 | 437.72 |
21.0 | 313.89 | 331.00 | 348.11 | 365.23 | 382.34 | 399.45 | 416.57 | 433.68 | 450.79 | 467.91 | 485.02 |
21.5 | 361.18 | 378.30 | 395.41 | 412.52 | 429.64 | 446.75 | 463.86 | 480.98 | 498.09 | 515.20 | 532.31 |
22.0 | 408.48 | 425.59 | 442.71 | 459.82 | 476.93 | 494.05 | 511.16 | 528.27 | 545.39 | 562.50 | 579.61 |
22.5 | 455.78 | 472.89 | 490.00 | 507.12 | 524.23 | 541.34 | 558.46 | 575.57 | 592.68 | 609.79 | 626.91 |
23.0 | 503.07 | 520.19 | 537.30 | 554.41 | 571.53 | 588.64 | 605.75 | 622.87 | 639.98 | 657.09 | 674.20 |
23.5 | 550.37 | 567.48 | 584.60 | 601.71 | 618.81 | 635.94 | 653.05 | 670.16 | 687.27 | 704.39 | 721.50 |
24.0 | 597.67 | 614.78 | 631.89 | 649.01 | 666.12 | 683.23 | 700.35 | 717.46 | 734.57 | 751.68 | 768.80 |
24.5 | 644.96 | 662.08 | 679.19 | 696.30 | 713.42 | 730.53 | 747.64 | 764.75 | 781.87 | 798.98 | 816.09 |
25.0 | 692.26 | 709.37 | 726.49 | 743.60 | 760.71 | 777.83 | 794.94 | 812.05 | 829.16 | 846.28 | 863.39 |
25.5 | 739.56 | 756.67 | 773.78 | 790.90 | 808.01 | 825.12 | 842.23 | 859.35 | 876.46 | 893.57 | 910.69 |
26.0 | 786.85 | 803.97 | 821.08 | 838.19 | 855.42 | 872.42 | 889.53 | 906.64 | 923.76 | 940.87 | 957.98 |
26.5 | 834.15 | 851.26 | 868.38 | 885.49 | 902.60 | 919.71 | 936.83 | 953.94 | 971.05 | 988.17 | 1005.28 |
27.0 | 881.45 | 898.56 | 915.67 | 932.79 | 949.90 | 967.01 | 984.12 | 1001.24 | 1018.25 | 1035.46 | 1052.58 |
27.5 | 928.74 | 945.86 | 962.97 | 980.08 | 997.19 | 1014.31 | 1031.42 | 1048.53 | 1065.65 | 1082.76 | 1099.87 |
28.0 | 976.04 | 993.15 | 1010.27 | 1027.38 | 1044.49 | 1061.60 | 1078.72 | 1095.83 | 1112.94 | 1130.06 | 1147.17 |
28.5 | 1023.34 | 1040.45 | 1057.56 | 1074.67 | 1091.79 | 1108.90 | 1126.01 | 1143.13 | 1160.24 | 1177.35 | 1194.47 |
29.0 | 1070.63 | 1087.75 | 1104.86 | 1121.97 | 1139.08 | 1156.20 | 1173.31 | 1190.42 | 1207.54 | 1224.65 | 1241.76 |
29.5 | 1117.93 | 1135.04 | 1152.15 | 1169.27 | 1186.38 | 1203.49 | 1120.61 | 1237.72 | 1254.83 | 1271.95 | 1289.06 |
30.0 | 1165.23 | 1182.34 | 1199.45 | 1216.56 | 1233.68 | 1250.79 | 1267.90 | 1285.02 | 1302.13 | 1319.24 | 1336.36 |
30.5 | 1212.52 | 1229.63 | 1246.75 | 1263.86 | 1280.97 | 1298.09 | 1315.20 | 1332.31 | 1349.43 | 1366.54 | 1383.65 |
31.0 | 1259.82 | 1276.93 | 1294.04 | 1311.16 | 1328.27 | 1345.38 | 1362.50 | 1379.61 | 1396.72 | 1413.84 | 1430.95 |
31.5 | 1307.11 | 1324.23 | 1341.34 | 1358.45 | 1375.57 | 1392.68 | 1409.79 | 1426.91 | 1444.02 | 1461.13 | 1478.24 |
32.0 | 1354.41 | 1371.52 | 1388.64 | 1405.75 | 1422.86 | 1439.98 | 1457.09 | 1474.20 | 1491.32 | 1508.43 | 1525.54 |
32.5 | 1401.71 | 1418.82 | 1435.93 | 1453.05 | 1470.16 | 1487.27 | 1504.39 | 1521.50 | 1538.61 | 1555.72 | 1572.84 |
33.0 | 1449.00 | 1466.12 | 1483.23 | 1500.34 | 1517.46 | 1534.57 | 1551.68 | 1568.80 | 1585.91 | 1603.02 | 1620.13 |
33.5 | 1496.30 | 1513.41 | 1530.53 | 1547.64 | 1564.75 | 1581.87 | 1598.98 | 1616.09 | 1633.20 | 1650.32 | 1667.43 |
34.0 | 1543.60 | 1560.71 | 1577.82 | 1594.94 | 1612.05 | 1629.16 | 1646.28 | 1663.39 | 1680.50 | 1697.61 | 1714.73 | Table 10. Sonographic Prediction of Fetal Weight (200–2200g)* (Continued)
Head | Abdominal Circumference (cm) |
Circumference (cm) | 19.0 | 19.5 | 20.0 | 20.5 | 21.0 | 21.5 | 22.0 | 22.5 | 23.0 | 23.5 | 24.0 |
13.5 | - | - | - | - | - | - | - | - | - | - | - |
14.0 | - | - | - | - | - | - | - | - | - | - | - |
14.5 | - | - | - | - | - | - | - | - | - | - | - |
15.0 | - | - | - | - | - | - | - | - | - | - | - |
15.5 | - | - | - | - | - | - | - | - | - | - | - |
16.0 | - | - | - | - | - | - | - | - | - | - | 200.30 |
16.5 | - | - | - | - | - | - | - | - | 213.37 | 230.48 | 247.59 |
17.0 | - | - | - | - | - | 209.32 | 226.44 | 243.55 | 260.66 | 277.78 | 294.89 |
17.5 | - | - | 205.28 | 222.39 | 239.51 | 256.62 | 273.73 | 290.85 | 307.96 | 325.05 | 342.19 |
18.0 | 218.35 | 235.47 | 252.28 | 269.69 | 286.80 | 303.92 | 321.03 | 338.14 | 355.26 | 372.37 | 389.48 |
18.5 | 265.65 | 282.76 | 299.87 | 316.99 | 334.10 | 351.21 | 368.33 | 385.44 | 402.55 | 419.67 | 436.78 |
19.0 | 312.95 | 330.06 | 347.17 | 364.28 | 381.40 | 398.51 | 415.62 | 432.74 | 449.85 | 466.96 | 484.08 |
19.5 | 360.24 | 377.35 | 394.47 | 411.58 | 428.69 | 445.81 | 462.92 | 480.03 | 497.15 | 514.26 | 531.37 |
20.0 | 407.54 | 424.65 | 441.76 | 458.88 | 475.99 | 493.10 | 510.22 | 527.33 | 544.44 | 561.56 | 578.67 |
20.5 | 454.83 | 471.95 | 489.06 | 506.17 | 523.29 | 540.40 | 557.51 | 574.63 | 591.74 | 608.85 | 625.96 |
21.0 | 502.13 | 519.24 | 536.36 | 553.47 | 570.58 | 587.70 | 604.81 | 621.92 | 639.04 | 656.15 | 673.26 |
21.5 | 549.43 | 566.54 | 583.65 | 600.77 | 617.88 | 634.99 | 652.11 | 669.22 | 686.33 | 703.44 | 720.56 |
22.0 | 596.72 | 613.84 | 630.95 | 648.06 | 665.18 | 682.29 | 699.40 | 716.52 | 733.63 | 750.74 | 767.85 |
22.5 | 644.02 | 661.13 | 678.25 | 695.36 | 712.47 | 729.59 | 746.70 | 763.81 | 780.92 | 798.04 | 815.15 |
23.0 | 691.32 | 708.43 | 725.54 | 742.66 | 759.77 | 776.88 | 794.00 | 811.11 | 828.22 | 845.33 | 862.45 |
23.5 | 738.61 | 755.73 | 772.84 | 789.95 | 807.07 | 824.18 | 841.29 | 858.40 | 875.52 | 892.63 | 909.74 |
24.0 | 785.91 | 803.02 | 820.14 | 837.25 | 854.36 | 871.48 | 888.59 | 905.70 | 922.81 | 939.93 | 957.04 |
24.5 | 833.21 | 850.32 | 867.43 | 884.55 | 901.66 | 918.77 | 935.88 | 953.00 | 970.11 | 987.22 | 1004.34 |
25.0 | 880.50 | 897.62 | 914.73 | 931.84 | 948.96 | 966.07 | 983.18 | 1000.29 | 1017.41 | 1034.52 | 1051.63 |
25.5 | 927.80 | 944.91 | 962.03 | 979.14 | 996.25 | 1013.36 | 1030.48 | 1047.59 | 1064.70 | 1081.82 | 1098.93 |
26.0 | 975.10 | 992.21 | 1009.32 | 1026.44 | 1043.55 | 1060.66 | 1077.77 | 1094.89 | 1112.00 | 1129.11 | 1146.23 |
26.5 | 1022.39 | 1039.51 | 1056.62 | 1073.73 | 1090.84 | 1107.96 | 1125.07 | 1142.18 | 1159.30 | 1176.41 | 1193.52 |
27.0 | 1069.69 | 1086.80 | 1103.92 | 1121.03 | 1138.14 | 1155.25 | 1172.37 | 1189.48 | 1206.59 | 1223.71 | 1240.82 |
27.5 | 1116.99 | 1134.10 | 1151.21 | 1168.32 | 1185.44 | 1202.55 | 1219.66 | 1236.78 | 1253.89 | 1271.00 | 1288.12 |
28.0 | 1164.28 | 1181.39 | 1198.51 | 1215.62 | 1232.73 | 1249.85 | 1266.96 | 1284.07 | 1301.19 | 1318.30 | 1335.41 |
28.5 | 1211.58 | 1228.69 | 1245.80 | 1262.92 | 1280.03 | 1297.14 | 1314.26 | 1331.37 | 1348.48 | 1365.60 | 1382.71 |
29.0 | 1258.88 | 1275.99 | 1293.10 | 1310.21 | 1327.33 | 1344.44 | 1361.55 | 1378.67 | 1395.78 | 1412.89 | 1430.01 |
29.5 | 1306.17 | 1323.28 | 1340.40 | 1357.51 | 1374.62 | 1391.74 | 1408.85 | 1425.96 | 1443.08 | 1460.19 | 1477.30 |
30.0 | 1353.47 | 1370.58 | 1387.69 | 1404.81 | 1421.92 | 1439.03 | 1456.15 | 1473.26 | 1490.37 | 1507.49 | 1524.60 |
30.5 | 1400.76 | 1417.88 | 1434.99 | 1452.10 | 1469.22 | 1486.33 | 1503.44 | 1520.56 | 1537.67 | 1554.78 | 1571.89 |
31.0 | 1448.06 | 1465.17 | 1482.29 | 1499.40 | 1516.51 | 1533.63 | 1550.74 | 1567.85 | 1584.97 | 1602.08 | 1619.19 |
31.5 | 1495.36 | 1512.47 | 1529.58 | 1546.70 | 1563.81 | 1580.92 | 1598.04 | 1615.15 | 1632.26 | 1649.37 | 1666.49 |
32.0 | 1542.65 | 1559.77 | 1576.88 | 1593.99 | 1611.11 | 1628.22 | 1645.33 | 1662.45 | 1679.56 | 1696.67 | 1713.78 |
32.5 | 1589.95 | 1607.06 | 1624.18 | 1641.29 | 1658.40 | 1675.52 | 1692.63 | 1709.74 | 1726.85 | 1743.97 | 1761.00 |
33.0 | 1637.25 | 1654.36 | 1671.47 | 1688.59 | 1705.70 | 1722.81 | 1739.93 | 1757.04 | 1774.15 | 1791.26 | 1808.38 |
33.5 | 1684.54 | 1701.66 | 1718.77 | 1735.88 | 1753.00 | 1770.11 | 1787.22 | 1804.33 | 1821.45 | 1838.56 | 1855.67 |
34.0 | 1731.84 | 1748.95 | 1766.07 | 1783.18 | 1800.29 | 1817.41 | 1834.52 | 1851.63 | 1868.74 | 1885.96 | 1902.97 | Table 10. Sonographic Prediction of Fetal Weight (200–2200g)* (Continued)
Head | Abdominal Circumference (cm) |
Circumference (cm) | 24.5 | 25.0 | 25.5 | 26.0 | 26.5 | 27.0 | 27.5 | 28.0 | 28.5 | 29.0 |
13.5 | - | - | - | - | - | - | - | - | - | - |
14.0 | - | - | - | - | - | - | - | - | - | - |
14.5 | - | - | - | - | - | - | - | - | 212.42 | 229.54 |
15.0 | - | - | - | - | - | 208.38 | 225.49 | 242.61 | 259.72 | 276.83 |
15.5 | - | - | 204.34 | 221.45 | 238.56 | 255.68 | 272.79 | 289.90 | 307.02 | 324.13 |
16.0 | 217.41 | 234.52 | 251.64 | 268.75 | 285.86 | 302.97 | 320.09 | 337.20 | 354.31 | 371.43 |
16.5 | 264.71 | 281.82 | 298.93 | 316.04 | 333.16 | 350.27 | 367.38 | 384.50 | 401.61 | 418.71 |
17.0 | 312.00 | 329.12 | 346.23 | 363.34 | 380.45 | 397.57 | 414.68 | 431.79 | 448.91 | 466.02 |
17.5 | 359.30 | 376.41 | 393.52 | 410.64 | 427.75 | 444.86 | 461.98 | 479.09 | 496.20 | 513.32 |
18.0 | 406.60 | 423.71 | 440.82 | 457.93 | 475.05 | 492.16 | 509.27 | 526.39 | 543.50 | 560.61 |
18.5 | 453.89 | 471.00 | 488.12 | 505.23 | 522.34 | 539.46 | 556.57 | 573.68 | 590.80 | 607.91 |
19.0 | 501.19 | 518.30 | 535.41 | 552.53 | 569.64 | 586.75 | 603.87 | 620.98 | 638.09 | 655.21 |
19.5 | 548.48 | 565.60 | 582.71 | 599.82 | 616.94 | 634.05 | 651.16 | 668.28 | 685.39 | 702.50 |
20.0 | 595.78 | 612.89 | 630.01 | 647.12 | 664.23 | 681.35 | 698.46 | 715.57 | 732.69 | 749.80 |
20.5 | 643.08 | 660.19 | 677.30 | 694.42 | 711.53 | 728.64 | 745.76 | 762.87 | 779.98 | 797.09 |
21.0 | 690.37 | 707.49 | 724.60 | 741.71 | 758.83 | 775.94 | 793.05 | 810.17 | 827.28 | 844.39 |
21.5 | 737.67 | 754.78 | 771.90 | 789.01 | 806.12 | 823.24 | 840.35 | 857.46 | 874.57 | 891.69 |
22.0 | 784.97 | 802.08 | 819.19 | 836.31 | 853.42 | 870.53 | 887.65 | 904.76 | 921.87 | 938.98 |
22.5 | 832.26 | 849.38 | 866.49 | 883.60 | 900.72 | 917.83 | 934.94 | 952.05 | 969.17 | 986.28 |
23.0 | 879.56 | 896.67 | 913.79 | 930.90 | 948.01 | 965.13 | 982.24 | 999.35 | 1016.46 | 1033.58 |
23.5 | 926.86 | 943.97 | 961.08 | 978.20 | 995.31 | 1012.42 | 1029.53 | 1046.65 | 1063.76 | 1080.87 |
24.0 | 974.15 | 991.27 | 1008.38 | 1025.49 | 1042.61 | 1059.72 | 1076.83 | 1093.94 | 1111.06 | 1128.17 |
24.5 | 1021.45 | 1038.56 | 1055.68 | 1072.79 | 1089.90 | 1107.01 | 1124.13 | 1141.24 | 1158.35 | 1175.47 |
25.0 | 1068.75 | 1085.86 | 1102.97 | 1120.09 | 1137.20 | 1154.31 | 1171.42 | 1188.54 | 1205.65 | 1222.76 |
25.5 | 1116.04 | 1133.16 | 1150.27 | 1167.38 | 1184.49 | 201.61 | 1218.72 | 1235.83 | 1252.95 | 1270.06 |
26.0 | 1163.34 | 1180.45 | 1197.57 | 1214.68 | 1231.79 | 1248.90 | 1266.02 | 1283.13 | 1300.24 | 1317.36 |
26.5 | 1210.64 | 1227.75 | 1244.86 | 1261.97 | 1279.09 | 1296.20 | 1313.31 | 1330.43 | 1347.54 | 1364.65 |
27.0 | 1257.93 | 1275.05 | 1292.16 | 1309.27 | 1326.38 | 1343.50 | 1360.81 | 1377.72 | 1394.84 | 1411.95 |
27.5 | 1305.23 | 1322.34 | 1339.45 | 1356.57 | 1373.68 | 1390.79 | 1407.91 | 1425.02 | 1442.13 | 1459.25 |
28.0 | 1352.53 | 1369.64 | 1386.75 | 1403.86 | 1420.98 | 1438.09 | 1455.20 | 1472.32 | 1489.43 | 1506.54 |
28.5 | 1399.82 | 1416.93 | 1434.05 | 1451.16 | 1468.27 | 1485.39 | 1502.20 | 1519.61 | 1536.73 | 1553.84 |
29.0 | 1447.12 | 1464.23 | 1481.34 | 1498.46 | 1515.57 | 1532.68 | 1549.80 | 1566.91 | 1584.02 | 1601.13 |
29.5 | 1494.41 | 1511.53 | 1528.64 | 1545.75 | 1562.87 | 1579.98 | 1597.09 | 1614.21 | 1631.32 | 1648.43 |
30.0 | 1541.71 | 1558.82 | 1575.94 | 1593.05 | 1610.16 | 1627.28 | 1644.39 | 1661.50 | 1678.62 | 1695.73 |
30.5 | 1589.01 | 1606.12 | 1623.23 | 1640.35 | 1657.46 | 1674.57 | 1691.69 | 1708.80 | 1725.91 | 1743.02 |
31.0 | 1636.30 | 1653.42 | 1670.53 | 1687.64 | 1704.76 | 1721.87 | 1738.98 | 1756.10 | 1773.21 | 1790.32 |
31.5 | 1683.60 | 1700.71 | 1717.83 | 1734.94 | 1752.05 | 1769.17 | 1786.28 | 1803.39 | 1820.50 | 1837.62 |
32.0 | 1730.90 | 1748.01 | 1765.12 | 1782.24 | 1799.35 | 1816.46 | 1833.58 | 1850.69 | 1867.80 | 1884.91 |
32.5 | 1778.19 | 1795.31 | 1812.42 | 1829.53 | 1846.65 | 1863.76 | 1880.87 | 1897.98 | 1915.10 | 1932.21 |
33.0 | 1825.49 | 1842.60 | 1859.72 | 1876.83 | 1893.94 | 1911.06 | 1928.17 | 1945.28 | 1962.39 | 1979.51 |
33.5 | 1872.79 | 1889.90 | 1907.01 | 1924.13 | 1941.24 | 1958.35 | 1975.46 | 1992.58 | 2009.69 | 2026.80 |
34.0 | 1920.08 | 1937.20 | 1954.31 | 1991.42 | 1988.54 | 2005.76 | 2022.65 | 2039.87 | 2056.99 | 2074.10 | Table 10. Sonographic Prediction of Fetal Weight (200–2200g)* (Continued)
Head | Abdominal Circumference (cm) |
Circumference (cm) | 29.5 | 30.0 | 30.5 | 31.0 | 31.5 | 32.0 | 32.5 | 33.0 | 33.5 | 34.0 |
13.5 | - | - | - | 203.40 | 220.51 | 237.62 | 254.73 | 271.85 | 288.96 | 306.07 |
14.0 | - | 216.47 | 233.58 | 250.69 | 267.81 | 284.92 | 302.03 | 319.14 | 336.26 | 353.37 |
14.5 | 246.65 | 263.76 | 280.88 | 297.99 | 315.10 | 332.21 | 349.33 | 366.44 | 383.55 | 400.67 |
15.0 | 293.95 | 311.06 | 328.17 | 345.29 | 362.40 | 379.51 | 396.62 | 413.74 | 430.85 | 447.96 |
15.5 | 341.24 | 358.36 | 375.47 | 392.58 | 409.69 | 426.81 | 443.92 | 461.03 | 478.15 | 495.26 |
16.0 | 388.54 | 405.65 | 422.77 | 439.88 | 456.99 | 474.10 | 491.22 | 508.33 | 525.44 | 542.56 |
16.5 | 435.84 | 452.95 | 470.06 | 487.17 | 504.29 | 521.40 | 538.51 | 555.63 | 572.74 | 589.95 |
17.0 | 483.13 | 500.25 | 517.36 | 534.47 | 551.58 | 568.70 | 585.81 | 602.92 | 620.04 | 637.15 |
17.5 | 530.43 | 547.54 | 564.65 | 581.77 | 598.88 | 615.99 | 633.11 | 650.22 | 667.33 | 684.45 |
18.0 | 577.73 | 594.84 | 611.95 | 629.06 | 646.18 | 663.29 | 680.40 | 697.52 | 714.63 | 731.74 |
18.5 | 625.02 | 642.13 | 659.25 | 676.36 | 693.47 | 710.59 | 727.70 | 744.81 | 761.93 | 779.04 |
19.0 | 672.32 | 689.43 | 706.54 | 723.66 | 740.77 | 757.88 | 775.00 | 792.11 | 809.22 | 826.34 |
19.5 | 719.61 | 736.73 | 753.84 | 770.95 | 788.07 | 805.18 | 822.29 | 839.41 | 856.52 | 873.63 |
20.0 | 766.91 | 784.02 | 801.14 | 818.25 | 835.36 | 852.48 | 869.59 | 886.70 | 903.82 | 920.93 |
20.5 | 814.21 | 831.32 | 848.43 | 865.55 | 882.66 | 899.77 | 916.89 | 934.00 | 951.11 | 968.22 |
21.0 | 861.50 | 878.62 | 895.73 | 912.84 | 929.96 | 947.07 | 964.18 | 981.30 | 998.41 | 1015.52 |
21.5 | 908.80 | 925.91 | 943.03 | 960.14 | 977.25 | 994.37 | 1011.48 | 1028.59 | 1045.70 | 1062.82 |
22.0 | 956.10 | 973.21 | 990.32 | 1007.44 | 1024.55 | 1041.66 | 1058.78 | 1075.89 | 1093.00 | 1110.11 |
22.5 | 1003.39 | 1020.51 | 1037.62 | 1054.73 | 1071.85 | 1088.96 | 1106.07 | 1123.18 | 1140.30 | 1157.41 |
23.0 | 1050.69 | 1067.80 | 1084.92 | 1102.03 | 1119.14 | 1136.26 | 1153.37 | 1170.48 | 1187.59 | 1204.71 |
23.5 | 1097.99 | 1115.10 | 1132.21 | 1149.33 | 1166.44 | 1183.55 | 1200.66 | 1217.78 | 1234.89 | 1252.00 |
24.0 | 1145.28 | 1162.40 | 1179.51 | 1196.62 | 1213.74 | 1230.85 | 1247.96 | 1265.07 | 1282.19 | 1299.30 |
24.5 | 1192.58 | 1209.69 | 1226.81 | 1243.92 | 1261.03 | 1278.14 | 1295.26 | 1312.37 | 1329.48 | 1346.60 |
25.0 | 1239.88 | 1256.99 | 1274.10 | 1291.22 | 1308.33 | 1325.44 | 1342.55 | 1359.67 | 1376.78 | 1393.89 |
25.5 | 1287.17 | 1304.29 | 1321.40 | 1338.51 | 1355.62 | 1372.74 | 1389.85 | 1406.96 | 1424.08 | 1441.19 |
26.0 | 1334.47 | 1351.58 | 1368.70 | 1385.81 | 1402.92 | 1420.03 | 1437.15 | 1454.26 | 1471.37 | 1488.49 |
26.5 | 1381.77 | 1398.88 | 1415.99 | 1433.10 | 1450.22 | 1467.33 | 1484.44 | 1501.56 | 1518.67 | 1535.78 |
27.0 | 1429.06 | 1446.18 | 1463.29 | 1480.40 | 1497.51 | 1514.63 | 1531.74 | 1548.85 | 1565.97 | 1583.08 |
27.5 | 1476.36 | 1493.47 | 1510.58 | 1527.70 | 1544.81 | 1561.92 | 1579.04 | 1596.15 | 1613.26 | 1630.38 |
28.0 | 1523.66 | 1540.77 | 1557.88 | 1574.99 | 1592.11 | 1609.22 | 1626.33 | 1643.45 | 1660.56 | 1677.67 |
28.5 | 1570.95 | 1588.06 | 1605.18 | 1622.29 | 1639.40 | 1656.52 | 1673.63 | 1690.74 | 1707.86 | 1724.97 |
29.0 | 1618.25 | 1635.36 | 1652.47 | 1669.59 | 1686.70 | 1703.81 | 1720.93 | 1738.04 | 1755.15 | 1772.27 |
29.5 | 1665.54 | 1682.66 | 1699.77 | 1716.88 | 1734.00 | 1751.11 | 1768.22 | 1785.34 | 1802.45 | 1819.56 |
30.0 | 1712.84 | 1729.95 | 1747.07 | 1764.18 | 1781.29 | 1798.41 | 1815.52 | 1832.63 | 1849.75 | 1866.86 |
30.5 | 1760.14 | 1777.25 | 1794.36 | 1811.48 | 1828.59 | 1845.70 | 1862.82 | 1879.93 | 1897.04 | 1914.15 |
31.0 | 1807.43 | 1824.55 | 1841.66 | 1858.77 | 1875.89 | 1893.00 | 1910.11 | 1927.23 | 1944.34 | 1961.45 |
31.5 | 1854.73 | 1871.84 | 1888.96 | 1906.07 | 1923.18 | 1940.30 | 1957.41 | 1974.52 | 1991.63 | 2008.75 |
32.0 | 1902.03 | 1919.14 | 1936.25 | 1953.37 | 1970.48 | 1987.59 | 2004.71 | 2021.82 | 2038.93 | 2056.04 |
32.5 | 1949.32 | 1966.44 | 1983.85 | 2000.66 | 2017.78 | 2034.89 | 2052.00 | 2069.11 | 2086.23 | 2103.34 |
33.0 | 1996.62 | 2013.73 | 2030.85 | 2047.96 | 2065.07 | 2082.18 | 2099.30 | 2116.41 | 2133.52 | 2150.64 |
33.5 | 2043.92 | 2061.03 | 2078.14 | 2095.26 | 2112.37 | 2129.48 | 2146.59 | 2163.71 | 2180.82 | 2197.93 |
34.0 | 2091.21 | 2108.33 | 2125.44 | 2142.55 | 2159.66 | 2176.78 | 2193.89 | 2211.00 | 2228.12 | 2245.23 |
*Based on P wt = 94.593 (HC) + 34.226 (AC) - 2134.616
Fetus's with femur length greater than or less than 2 SDs from norm
should be corrected by (FFL/MFL) (P wt)
(Sabbagha RE: Intrauterine growth retardation. In Sabbagha RE [ed]: Diagnostic
Ultrasound Applied to Obstetrics and Gynecology, 2nd
ed, p 122. Philadelphia, JB Lippincott, 1987)Role of Femur Length Hadlock and associates derived from the following formula for prediction
of BW from HC, AC, and FL:36 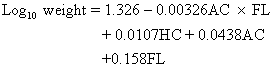
They recommended utilization of this formula for all birth weight categories
because the range in accuracy is relatively small, namely ± 14.8% (2 SD). Presently, their proposal seems reasonable if fetuses
falling in the BW category <1500 g are excluded. In the latter group, the
accuracy of BW prediction reported in their study deteriorates
to + 19.4% (2 DS)36 and use of a “targeted” approach is more appropriate.39 Of importance, by using the same formula in 361 normal fetuses, Hadlock
and associates showed that the shape of the ultrasonically derived weight
curve was identical to that derived by Williams and associates, based
on approximately 2 million newborns.36,37,38,39,40 Previously, Ott and Doyle suggested that the two curves (ultrasonic vs
actual BWs) were different.41 However, their conclusion was based on BWs derived by the less accurate
formula of Shepard and associates. The improvement in the prediction of BW by Hadlock's formula may be
mainly related to the use of the HC rather than HC and FL. The reason
is that other investigators, notably Woo and associates and Weiner and
associates, found no significant benefit to routine inclusion of the femur length.39,42 This observation seems contrary to the previously presented rationale
of incorporating fetal length in the estimation of fetal or neonatal size. However, closer
examination shows that routine inclusion of FL dilutes
any change or effect resulting from long or short fetuses because
the majority are of average length. In fact, Weiner and associates showed
that the variation in BW can be reduced by incorporating the FL selectively, that is, only in long or short fetuses, those differing from the mean
by 2 SD.39 To correct for femur length they multiplied the BW estimate by the ratio
of actual FL/FL at 50 P for dates. Routine Use of Birth Weight Prediction Routine prediction of BW by formula is not necessary and can be misleading. The
difference between actual and predicted BWs (2 SD = 15%) is large
enough to produce false-positive and false-negative diagnoses of
IUGR. For example, a fetus weighing 3000 g may be falsely assumed to be
growth retarded, whereas a small 2500 g fetus may be assumed to be normal. Antenatal prediction of BW has proven to be especially useful in the management
of women at high risk of premature delivery. The reasons include - Imprecision in the BW estimates by the history and clinical examination. For
example, Paul and associates showed that 35% of preterm infants
clinically judged to be less than 1000 g, in fact exceeded that weight.43
- Improvement in the perinatal outcome of very low birth weight babies; this
is attributed to the advent of technologically sophisticated neonatal
intensive care facilities and the management of high-risk pregnancies
at tertiary care centers. The most dramatic improvement is witnessed
in infants weighing less than 1000 g but not necessarily those under 750 g.44,45
Thus, accurate estimation of BW in the range of 750 g to 1000 g is important
because when equipped with this information, the physician managing
labor will pursue sound obstetric principles directed at the prevention
of asphyxia, a step essential for effective intensive neonatal care. Oligohydramnios Manning and associates and Chamberlain and associates found that the presence
of oligohydramnios was predictive of IUGR with an accuracy approaching 89.6% and 78.9%, respectively.46,47 However, the pregnancies studied were close to term and were at high risk
for IUGR, by previous ultrasound or clinical data or both. In their
studies oligohydramnios was diagnosed when only a single pocket of amniotic
fluid, approximately 1 cm to 2 cm in size, was noted in a plane
vertical to the uterine surface. Using the “1-cm rule” Hoddick and associates, in a retrospective
study, showed that oligohydramnios accurately predicted only 8/52 or 8% of
IUGR fetuses.48 In other words the sensitivity of the test was poor. In a large unselected population of pregnant women Philipson and associates
reported that the incidence of oligohydramnios was 3.9% and of those
only 40% were growth retarded.49 However, when projected to the total population the sensitivity of the
finding of oligohydramnios was only 16% (see Table 4);49 this level of sensitivity was also confirmed by Chamberlain and associates.47 In conducting their study Philipson and associates diagnosed oligohydramnios
by careful subjective assessment of overall amniotic fluid volume, rather
than by strict adherence to the 1-cm or 2-cm “rule.” The difference observed in the three studies can be explained by the number
of high-risk pregnancies (prevalence of IUGR) in each group. It is
clear that oligohydramnios is more predictive of poor outcome in pregnancies
at high risk for IUGR. This association between oligohydramnios
and high-risk pregnancies is further illustrated by reference to the
entity of post-date pregnancies. A retrospective study of such pregnancies
by Phelan and associates showed that amniotic fluid was marginal (> 1 cm, but
subjectively decreased) and decreased (<1 cm) in 38 and 7 patients, respectively.50 In these two groups, antenatal fetal heart tests showed decelerations
and bradycardia in 44.7% and 71%, respectively. By comparison, fetal heart
rate tests were abnormal in 21% of patients with normal amniotic
fluid volume. In the presence of oligohydramnios the overall incidence
of late decelerations requiring cesarean section was high (5.25% and 16.7%) when
compared to the group with normal fluid (1.05%). The authors also found that only 14% of post-date pregnancies with oligohydramnios
were delivered of an infant with the stigmata of post-maturity, including
meconium-stained fluid. Nonetheless, the authors recommended
that post-date pregnancies with oligohydramnios should be considered
for trial of labor with continuous electronic fetal monitoring. | 
 75th p, 25th–57th p, and
75th p, 25th–57th p, and  25th p).
25th p).