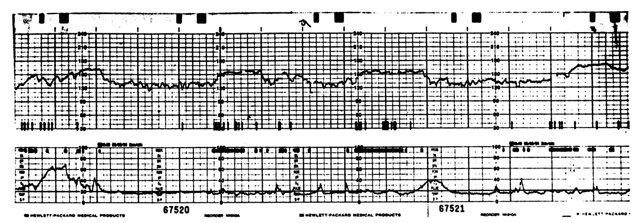
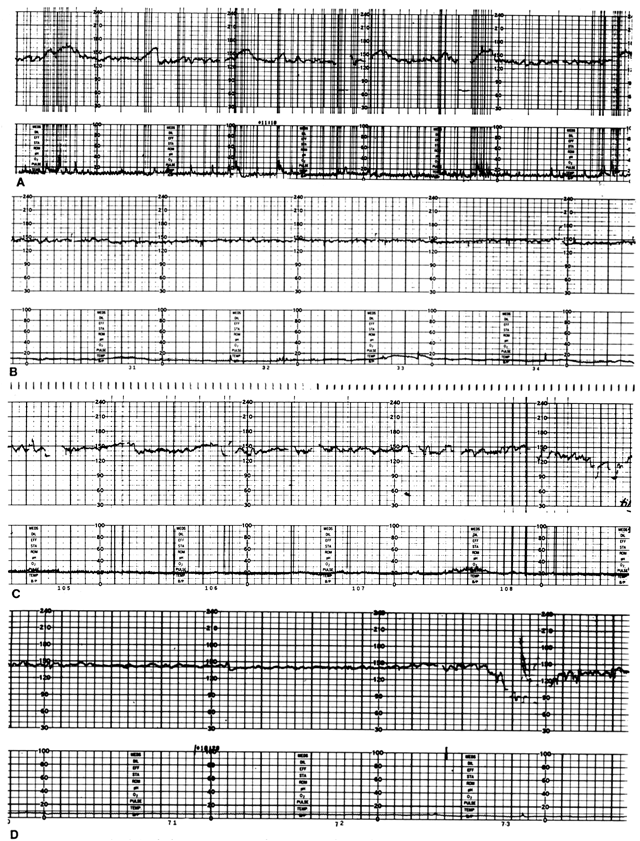
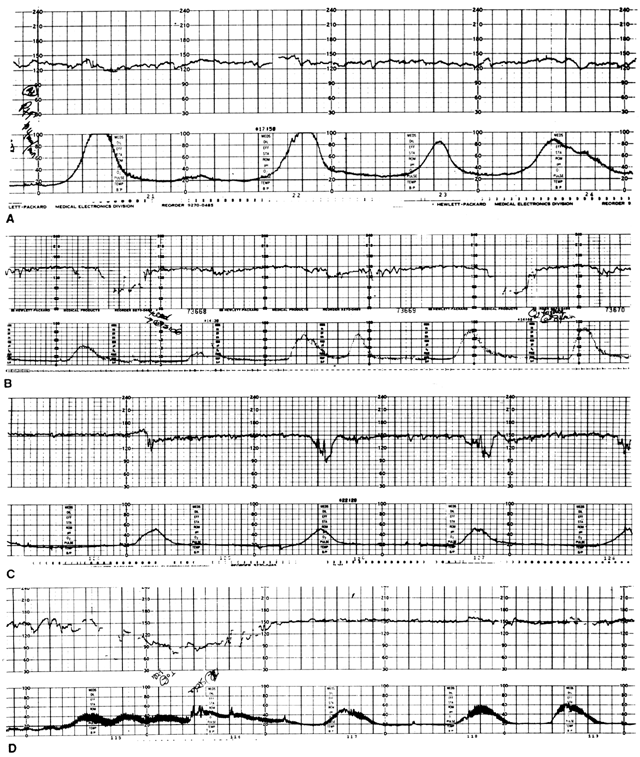
Heart rate patterns of normal fetuses reflect physiological responses to various endogenous and exogenous stimuli.5 The normal baseline record of FHR provides evidence that intrinsic control mechanisms responsible for cardiovascular autoregulation are intact. Control of FHR requires electrical conduction pathways, cellular receptors to circulating neurohormones, reflex arcs, and inherent myocardial contractility.5 The use of specific FHR patterns to evaluate fetoplacental status is based on the association of their components with particular intrauterine events or conditions. These components include baseline rate, rate variation, and episodic rate responses to fetal movements (accelerations) or uterine contractions (decelerations). The characteristics of these FHR components are determined by both cellular and systemic mechanisms.
FHR Patterns and Cellular Events
The functional units of the fetal heart are myocardial fibers that act as a syncytium; they are endowed with inherent rhythmicity, apparent from the first trimester onward.6 Cellular events within the myocardium are influenced by oxygen supply, energy substrates, membrane receptors to circulating hormones, and preservation of cell integrity. Cell growth or hypertrophy is a dominant feature of cardiac development in the final trimester of pregnancy, during which most FHR testing is performed. This process is energy consuming and requires adequate transport of oxygen, glucose, and amino acids.
Under normal conditions, the placenta serves as a respiratory and nutritive organ. Nutritive functions, maintained throughout pregnancy, lead to a positive balance of glucose. Eventual glycogen deposition in cardiac and hepatic tissue provides a reservoir for the stresses of parturition and early neonatal life. Under adverse circumstances (e.g., decreased utero-placental perfusion or maternal malnutrition), these stores may be prematurely exhausted; consequently, fetal growth and energy-dependent biophysical activities are curtailed.Placental respiratory failure may alter cellular metabolism. Increased tissue extraction of oxygen from high-affinity fetal hemoglobin may offer short-term protection from this problem. However, the inability of the placenta to exchange oxygen and carbon dioxide results in fetal respiratory acidosis. Excess hydrogen ions accumulate in fetal circulation; progressive cellular hypoxia and diminished aerobic metabolism result in development of a secondary metabolic acidosis. Critical intracellular enzymatic reactions begin to fail, and glucose is broken down to lactate and pyruvate, augmenting metabolic acidosis. Failure to interrupt this sequence of events may lead to cellular death, reduced myocardial contractility, and inability to maintain systemic homeostasis.
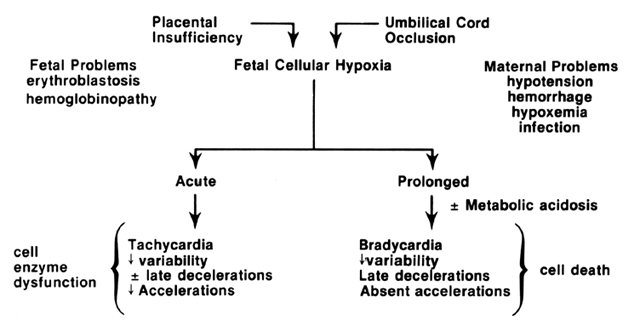
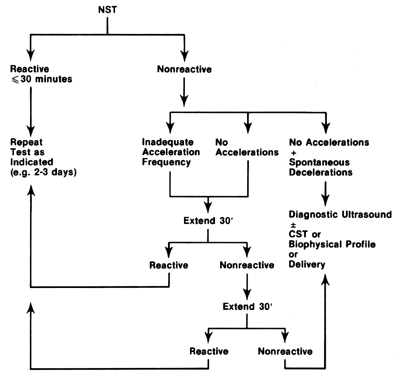
The relationship of cellular events to the pathophysiology of FHR tracings is summarized in Figure 1. Although there is controversy regarding the earliest FHR manifestations of cellular hypoxia and tissue acidosis, their expression will depend on both the chronicity and severity of the actual insults and may not be uniformly appreciated by all compromised fetuses. The ultimate or preterminal patterns associated with cellular hypoxia and systemic asphyxia consist in relatively fixed FHR baselines, reduced or absent FHR variation, absence of FHR accelerations, and the appearance of spontaneous late FHR decelerations.7, 8
FHR and Systemic Influences
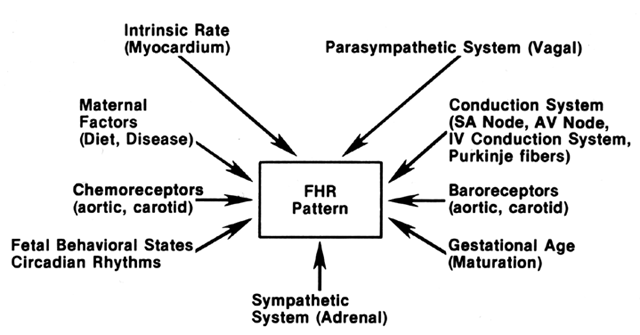
Unstressed or resting FHR tracings are indices of the following: (1) parasympathetic (vagal) tone; (2) sensitivity to sympathetic (adrenergic) discharge; (3) organization of fetal activity or behavioral states; (4) circadian rhythms, (5) the linkage between body movements and accelerations; (6) increasing stroke volume with reduction in resting or intrinsic rate; and (7) integration of reflex responses to momentary fluctuations in arterial blood pressure and gas partial pressures.
These complex interrelationships (Fig. 2) are also affected by maturational effects, maternal state factors, and pathologic conditions. FHR accelerative responses are regulated through accelerator nerve fibers arising in the upper thoracic roots and are fine-tuned in the hypothalamic and medullary regions of the brain, which are sensitive to momentary changes in oxygen tension, acid-base balance, circulating catecholamines, and endorphins. These pathways are evident as early as the midtrimester.6 FHR decelerative responses are partly generated through parasympathetic pathways, resulting from direct vagal stimulation and baroreceptor and chemoreceptor arcs (aortic arch, carotid sinus).9
Fetal movements become increasingly frequent in the midtrimester and act as triggers for transient baseline alterations with stronger linkages as term is approached.10, 11 Natale and co-workers10 have shown that from 24 to 32 weeks, despite a gradual decrease in the incidence of fetal movements, the association of accelerations with movements increases and the amplitude and duration of accelerations become greater. Others11, 12 have reported that the frequency of movement-associated decelerations decreases with gestational age, especially after 29 to 32 weeks.
Behavioral organization becomes more important in the late third trimester, since clustering of movements and accelerations become more apparent during this general time frame.13, 14 Animal studies suggest that the increasing tendency to generate accelerations with body movements may also result from increased myocardial sensitivity to endogenous catecholamines, presumably a reflection of increasing numbers of myocardial receptor sites.15
The element of time plays a greater role in the occurrence of FHR patterns as fetal cardiovascular control systems mature. Nonrandom, periodic cycles of FHR are generated, lasting from 60 to 500 minutes,16 with a mean duration of approximately 90 minutes at term. As state organization becomes better defined, epochs of low variability and decreased movement incidence recur, lasting as long as 90 to 120 minutes, with a mean of approximately 20 minutes.17 These sequences alternate, through transitions, with periods of increased fetal breathing and rapid eye movements associated with occasional body movements. These states of active sleep occupy 40% to 60% of the average 24-hour day18 and are also reflected by increased baseline FHR variability. Brief episodes of “wakeful” activity, approximately 10% to 15% of the total day, account for the majority of epochs during which reactive FHR accelerations are observed. Diurnal fluctuations in body movements have been reported, with peaks occurring between 0100 and 0700 hours.19 Throughout the day, mean frequency of FHR accelerations exceeding 15 beats per minute amplitude is 15 to 20 per hour.17
Maternal factors may influence the patterns present in resting FHR tracings. Maternal fed state, responsible for maternal glycemia, has had an inconsistent effect on reported FHR responses.20, 21 These reports failed to demonstrate an increase in either fetal movement or acceleration frequency following maternal glucose loading. Maternal activity levels22 are also associated with a variety of FHR patterns. Normal ambulation23 appears to have little appreciable influence on either subsequent baseline rate or acceleration incidence; graded vigorous exercise may cause transient but unpredictable tachycardias and bradycardias.22 Numerous drugs administered to pregnant women near term have been studied.24 β-blocking agents such as propranolol tend to lower mean baseline FHR and reduce acceleration frequency.25, 26 Long-acting central nervous system depressants may theoretically increase the time required for eliciting reactive accelerations,27 whereas nicotine may transiently elevate baseline rate, reduce uteroplacental perfusion, and delay the onset of a normal reactive pattern.28
Systemic influences on resting FHR tracings due to ongoing maternal or fetoplacental pathology have a common pathway in which oxygenation and energy substrates are reduced. In the absence of acidosis, acute disturbances of placental respiratory or nutritive function may result in sudden and profound decrease in fetal movement incidence. The corollary to this situation would be marked decreases in acceleration frequency. More commonly, diminished placental functions are more subtle, tend to be chronic, and lead to gradual declines in fetal movement incidence and acceleration frequencies as compensatory visceral shunting of the fetal circulation occurs.29, 30 Consequently, days or weeks may elapse before the impact of chronic placental failure can be appreciated from alterations in resting FHR patterns.
Uterine Activity and FHR Patterns
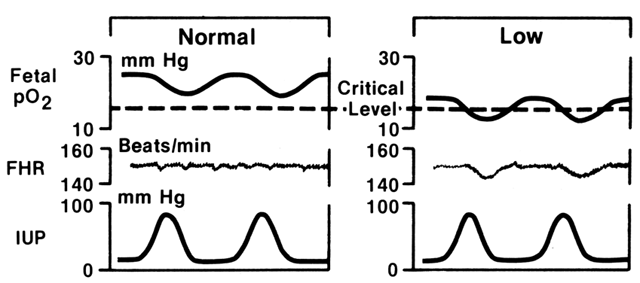
As term pregnancy approaches, the frequency of spontaneous uterine activity increases and individual contractions tend to become longer and more intense. Early data, using direct measurements of intrauterine pressure, suggest that a contraction intensity of more than 35 mmHg is needed before the effects of transient hypoxia are consistently appreciated by borderline or compromised fetuses.1 The concept of gauging the impact of such uterine activity on basal fetal oxygenation and subsequent alteration of FHR is illustrated in Figure 3. During any given contraction of moderate to strong intensity, intervillous space blood flow is greatly reduced or abolished, restricting oxygenation of the fetus. The hypoxemic fetus tolerates such stress poorly, and myocardial homeostasis becomes insufficient to maintain effective cardiac output during this period. The resulting late FHR decelerations have both a reflex component (i.e., vagotonic in origin) and a component directly related to myocardial depression. In general, the depth and duration of this response are reflections of the intensity and duration of the uterine contraction itself, whereas the “lag” between the peak of the contraction and nadir of the deceleration is a reflection of the pre-existing level of myocardial depression. It should be carefully noted that other mechanisms may be responsible for late FHR decelerations: (1) intrinsic maternal hypoxemia (respiratory disease, anemia); (2) maternal hypotension (aortocaval compression, drugs); or (3) compromise of umbilical venous blood flow (partial cord occlusion).
Uterine activity may also be associated with fetal movements and FHR accelerations.31 These FHR alterations occur commonly during labor and are typically associated with a healthy, well-oxygenated fetus.32 Unless the fetus is being directly visualized during periods of uterine activity, it may not be possible to distinguish accelerations associated with fetal movements from those associated with mild cord compression. Therefore, this clinical correlation should be interpreted cautiously during antepartum monitoring.