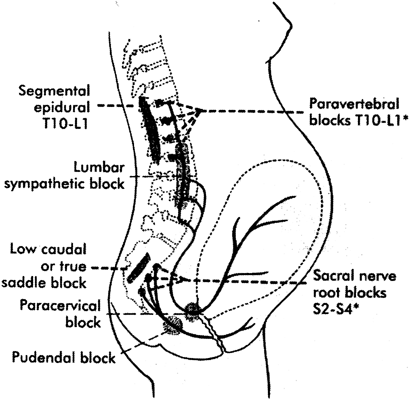
Many physiologic and anatomic changes occur during pregnancy, labor and delivery, and the postpartum period that directly impact the administration of analgesia and anesthesia (Table 1).
TABLE 1. Physiologic and Anatomic Changes During Pregnancy: Anesthetic
Implications
Change | Anesthetic Implications |
Circulation |
|
Hyperdynamic: increased reliance on sympathetic nervous system | Increase in incidence and severity of hypotension after regional analgesia/anesthesia |
Capillary engorgement, increase in airway edema | Increased incidence of difficult airway |
Aortocaval compressio | More profound hypotension with parturient in the supine position |
Metabolism and Respiration |
|
Increase in O2 requirement and CO2 production | Greater risk of desaturation after induction of general anesthesia |
Decrease in FRC | Greater risk of desaturation after induction of general anesthesia |
Gastrointestinal System |
|
Reduced esophageal---stomach barrier pressure | Increased risk of aspiration pneumonitis |
Endocrine |
|
Increased progesterone levels | Increased pain threshold |
Nervous System |
|
Increase in ß-endorphin concentration | Increased tolerance to pain |
Increased susceptibility to local anesthetics | Decrease in local anesthetic dose requirements |
Anatomic changes in the spinal column | Decrease in local anesthetic dose requirements |
Increased susceptibility to CNS depressants | Decrease in dose requirements for general anesthetics and adjuvants |
Pharmacokinetics |
|
Altered volume of distribution | Change in drug pharmacokinetics |
Altered protein binding of drugs | Change in drug pharmacokinetics |
Increased renal blood flow | Change in drug pharmacokinetics |
Altered hepatic microsomal enzyme activity | Change in drug pharmacokinetics |
Coagulation |
|
Hypercoagulable state |
|
FRC, functional residual capacity; CNS, central nervous system. | |
Circulation
In general, the cardiovascular system is hyperdynamic during pregnancy. However, arterial responsiveness to vasopressors is reduced in pregnancy in both in vivo and in vitro animal models.2,3 In contrast, the venous pressor response increases in pregnancy.4 Indeed, hemodynamic stability becomes more dependent on sympathetic nervous system activity as pregnancy progresses,5 probably as a result of dependence on venous return. Sympathetic blockade in the term parturient, a consequence of regional analgesia/anesthesia, results in a marked decrease in blood pressure compared with that in nonpregnant control subjects.5
Capillary engorgement of the oral, nasal, pharyngeal, and tracheal mucosa may contribute to a markedly increased incidence of difficult endotracheal intubations in obstetric patients.6 Nasotracheal intubation should be avoided because of the risk of epistaxis.
AORTOCAVAL COMPRESSION.
Term parturients in the supine position experience a 10% to 20% decrease in cardiac output secondary to aortocaval compression.7 Compression of the vena cava begins as early as 13 to 16 weeks' gestation8 and is nearly complete in a majority of parturients at term. In the lateral decubitus position, there is partial caval compression,9 but collateral circulation through the azygous system maintains venous return.10 There is partial compression of the aorta in the supine position that is completely relieved by the lateral position.11
Objective parameters of fetal well-being, including fetal scalp capillary blood pH, transcutaneous pO2, and fetal heart rate pattern, deteriorate when parturients labor in the supine position.12,13 This is true whether or not regional analgesia/anesthesia-induced sympathetic blockade is present. However, the adverse effects may be more profound in the parturient with sympathetic blockade. In addition, aortocaval compression may occur in the lateral decubitus position when maximum lumbar flexion is assumed during positioning for regional analgesia/anesthesia. Cardiac output is more likely to decrease in this position compared to the sitting position.14
Metabolism and Respiration
Oxygen requirements and carbon dioxide production increase 60% during pregnancy.15 Minute ventilation increases by 45% primarily as a result of an increase in tidal volume.16 In addition to the change in minute ventilation, the most important lung volume/capacity change is a 20% decrease in functional residual capacity (FRC) (i.e., volume of the lung at the end of normal expiration).16
During labor and delivery, minute ventilation may increase up to 300%, oxygen consumption increases by 75%, and the PaCO2 decreases as low as 10 to 15 mmHg.17 Maternal aerobic oxygen requirements exceed oxygen consumption during labor and delivery, resulting in a progressive rise in maternal lactate levels.18
The increased maternal oxygen requirement and decreased FRC (decreased oxygen reserve) have profound implications for the induction of general anesthesia in the parturient. Because of concerns of pulmonary aspiration (see later), parturients are almost never ventilated by mask during the induction of anesthesia. On induction, an intravenous sedative hypnotic (e.g., thiopental) is administered, immediately followed by a neuromuscular blocking agent. Patients remain apneic until the endotracheal tube is placed. Provided there is no difficulty with intubation, the apneic period is approximately 1 minute. During this period, the pO2 in the parturient falls at more than twice the rate of nonpregnant women.19 If oxygen is administered before the induction of anesthesia, resulting in a pO2 close to 500 mmHg, the parturient becomes hypoxemic after 3 minutes of apnea compared to 7 minutes in nonpregnant women.19 Increased minute ventilation and decreased FRC also increase the rate of uptake of inhaled anesthetic agents.
Finally, high spinal or epidural anesthesia is associated with a further decrease in FRC20; therefore, the parturient is at increased risk, compared to the nonpregnant patient, for having hypoxemia develop during regional anesthesia. For this reason, supplemental oxygen usually is delivered during regional anesthesia.
Gastrointestinal System
Several changes in the gastrointestinal system result in the admonition that a “parturient is always a full stomach.” The gravid uterus displaces the stomach, often resulting in the displacement of the esophagus into the thorax. There is a reduction in lower esophageal sphincter tone secondary to anatomic displacement21 and relaxation of the sphincter by progesterone.22 In addition, intragastric pressure rises. Altogether, these changes result in a reduced esophageal-stomach barrier pressure and the well-known clinical complaint of heartburn.
Gastric emptying, as measured by several techniques, including acetaminophen absorption,23,24 epigastric impedence,25 and applied potential tomography,26 is unchanged in pregnancy. However, labor significantly slows gastric emptying.25,27 Furthermore, opioids administered by any route, including systemic,25 epidural,28,29 or intrathecal,30 slow gastric emptying even more. In the immediate postpartum period (2 to 48 hours), gastric emptying does not appear to be delayed unless the parturient has received opioid analgesia.23–26,31
ANESTHETIC IMPLICATIONS.
Pulmonary aspiration and failed intubation were responsible for 48% of anesthesia-related maternal mortality in the United States between 1979 and 1990.32 Indeed, Mendelson's syndrome, or pulmonary aspiration, was first reported in parturients receiving general anesthesia.33 Ollson34 reported that the incidence of pulmonary aspiration is higher in patients undergoing cesarean delivery (1 in 661) than in the general population undergoing surgery (1 in 2131). In addition, the incidence of aspiration is more than fourfold higher for emergency surgery compared with that of elective surgery.35
Maternal mortality associated with regional anesthesia between 1988 and 1990 decreased compared to 1979 and 1981, whereas the mortality associated with general anesthesia was constant and most often related to difficult intubation or aspiration or both.32 Aspiration often is associated with difficult intubation.36 These data support the premise that regional anesthesia is safer for the parturient.
If general anesthesia is necessary, appropriate precautions are taken. The patient is most at risk for aspiration during induction and emergence from general anesthesia. Aspiration of acidic, compared to neutral, liquid results in more severe pathology.33 Histamine 2 (H2)-receptor antagonists reliably increase gastric pH by decreasing gastric acid production.37 H2-receptor antagonists do not neutralize acid already present in the stomach. Therefore, a nonparticulate antacid (e.g., sodium citrate) is always administered before the induction of general anesthesia. A nonparticulate antacid is used because aspiration of particulate antacid may cause symptoms of aspiration pneumonitis.38
Many anesthesiologists also add metoclopramide to the aspiration prophylaxis cocktail. Metoclopramide increases lower esophageal sphincter tone39 and reduces gastric volume by increasing gastric motility.40
General anesthesia in the parturient is always induced by rapid sequence (see later). Mask ventilation is avoided, as air inadvertently introduced into the stomach increases the risk of aspiration and the airway is not protected with an endotracheal tube. Similarly, deep “conscious” sedation is avoided in the parturient, as this may be associated with obtundation of protective airway reflexes.
As a final precaution, the American Society of Anesthesiologist's (ASA) Guidelines for Obstetrical Anesthesia recommend that before elective cesarean delivery, institutional presurgical fasting guidelines should be followed.41 For most institutions, this means a fasting period for solid food of 6 to 8 hours.
A related controversial issue is maternal oral intake during labor. Although laboring parturients who eat have larger gastric volumes than do parturients who fast,42 an association between eating during labor and an increased risk of aspiration has not been shown. Proponents of eating argue that oral intake improves patient comfort and satisfaction43 and prevents maternal ketoacidosis.42 Pulmonary aspiration is a rare event. Opponents of feeding argue that one cannot predict which parturient will require emergency surgery. Although the risk of aspiration is low, the morbidity and mortality are high. Mild ketosis has not been shown to adversely affect outcome, and a minority of patients find fasting stressful.43 ASA Guidelines state that the “oral intake of modest amounts of clear liquids may be allowed for uncomplicated laboring patients.”41 The American College of Obstetricians and Gynecologists (ACOG) Guidelines of Perinatal Care states that “patients in active labor should avoid oral ingestion of anything except sips of clear liquids.”44
Endocrine
Progesterone concentration rises throughout pregnancy and declines abruptly after delivery of the placenta.45 The increase in plasma progesterone may influence a number of physiologic processes, including response to pain (see later).
Central and Peripheral Nervous System
Anatomic and physiologic changes in the nervous system alter responses to pain and susceptibility to both general and regional anesthesia. Pregnancy-induced neurohumoral changes may alter responses to pain. In a rat model, the pregnancy-induced increased concentration of plasma β-endorphin was associated with an increased tolerance to visceral stimulation, and this effect was reversed by naloxone.46 Pregnancy is associated with lower plasma substance P concentrations47 and higher cerebral spinal fluid (CSF) progesterone levels.48
Nerves from pregnant animals (including humans) appear more susceptible to local anesthetic blockade. In intact rats49,50 and humans,51,52 pregnancy enhanced the effect of central and peripheral local anesthetics. Proposed mechanisms of enhanced neural blockade during pregnancy include hormone-related changes in the actions of spinal cord neurotransmitters, potentiation of the analgesic effect of endogenous analgesic systems, increased permeability of the neural sheath, and other pharmacodynamic or pharmacokinetic differences between pregnant and nonpregnant women.53 The spread of epidural local anesthetic blockade is increased during early pregnancy,51 a phenomenon explained by altered sensitivity to local anesthetics, as opposed to gross changes in spinal column anatomy.
However, anatomic changes in the mother may affect regional anesthesia techniques. The epidural and vertebral foraminal veins are enlarged, resulting in an increased risk of intravascular placement of an epidural catheter and a decreased egress of epidural anesthetic agents from the epidural space. Engorged epidural veins are associated with a decrease cerebral lumbosacral CSF volume.54 An increase in intra-abdominal pressure also is associated with decreased CSF volume.55 In volunteers, decreased lumbosacral CSF volume, as measured by magnetic resonance imaging, was associated with increased sensory blockade after spinal anesthesia.56
Together, these changes result in a 25% reduction in the segmental dose requirement for spinal anesthesia48,57 and a similar segmental dose reduction for epidural anesthesia.51
The central nervous system (CNS) and cardiac toxicity of lidocaine58 and ropivacaine59 are not altered by pregnancy. Although an earlier study concluded that the plasma bupivacaine concentration necessary for cardiac toxicity was markedly decreased in the pregnant sheep l,60 a more recent study does not support this conclusion.61
Just as peripheral nerves are more susceptible to local anesthetic during pregnancy, the parturient also is more susceptible to CNS depressants. The minimum alveolar concentration of volatile halogenated anesthetic agents (the median effective dose) is decreased 30% during pregnancy.62 Similarly, there is increased sensitivity to thiopental.63
Pharmacokinetics
Pregnancy alters disposition or drugs by several mechanisms. Volume of distribution may be altered. For example, the elimination half-life of thiopental is markedly prolonged secondary to a large increase in the volume of distribution.64 Plasma protein concentration decreases, leading to altered drug binding.65 For example, lidocaine is less protein bound during pregnancy, resulting in a higher free fraction in the blood.66 Increased renal blood flow and glomerular filtration and altered hepatic microsomal activity change renal and hepatic drug clearance.67
Coagulation
Pregnancy is a state of enhanced activation of the coagulation system. The levels of most clotting and fibrinolytic factors are elevated.68 However, in otherwise normal parturients, 0.9% have a platelet count of less than 100,000/mm3.69 The platelet count falls after delivery and during the first postpartum day.70
Uteroplacental Unit
UTEROPLACENTAL BLOOD FLOW.
Uterine blood flow (UBF) increases markedly during pregnancy, from 50 to 100 mL/min before pregnancy to 700 to 900 mL/min at term. There is a generalized decrease in vascular sensitivity to endogenous and exogenous vasopressors; however, the diminution in response may vary between uterine and systemic vessels. The altered vascular reactivity in pregnancy may be secondary to (1) changes in receptor number or function, (2) changes in metabolism or clearance of drugs, (3) changes in release of endogenous vasoactive substances, or (4) changes in sensitivity to vasoactive substances.71 In general, however, UBF is related to uterine perfusion pressure and vascular resistance:

UBF is not autoregulated. Any decrease in uterine arterial pressure (systemic hypotension), increase in venous pressure (caval compression, uterine contraction, Valsalva maneuver), or increase in uterine vascular resistance (increase in uterine vasoconstriction relative to systemic vasoconstriction) decrease UBF. The net effect on UBF of any therapeutic intervention depends on relative changes in systemic and uterine vessels as they affect the determinants of UBF.
EFFECT OF REGIONAL ANALGESIA/ANESTHESIA ON UTEROPLACENTAL BLOOD FLOW.
Regional analgesia/anesthesia may effect UBF, in both directions, by a number of mechanisms.71 Pain causes activation of the sympathetic nervous system. In pregnant ewes, acute stress increases plasma norepinephrine levels by 25% and decreases UBF by 50%.72 Labor pain also is associated with a marked increase in epinephrine concentration,73,74 which has been associated with an increased incidence of abnormal fetal heart rate patterns.75 Labor analgesia is associated with a marked reduction in circulating catecholamines.73,74 Pain also causes maternal hyperventilation. Hyperventilation may cause a decrease in UBF.76
Regional anesthesia-induced sympathetic blockade does not appear to alter UBF in normal parturients in the absence of hypotension.77 In the setting of severe pre-eclampsia, epidural analgesia with local anesthetic actually may increase intervillous blood flow.78
Regional analgesia/anesthesia may adversely effect UBF primarily as a result of sympathetic blockade-induced maternal hypotension. Maternal hypotension decreases uterine arterial pressure and increases uterine vascular resistance secondary to reflex release of vasoconstrictors.71 Regional analgesia has been associated with uterine hypertonus resulting in a decrease in UBF (see later).
Unintentional intravenous injection of local anesthetic may decrease UBF by several mechanisms. A test dose containing epinephrine often is administered through the epidural catheter to rule out inadvertent intravenous placement of the catheter. If the catheter is in a vein, the intravenous epinephrine will cause transient maternal tachycardia and hypertension. In gravid ewes, intravenous epinephrine also caused a transient decrease in UBF.79 The epidural administration of local anesthetic solutions containing epinephrine does not appear to effect UBF in the healthy parturient.80 However, there is some concern that the epidural injection of epinephrine containing local anesthetics may adversely effect UBF in patients with pre-eclampsia.81
The amount of local anesthetic contained in a normal test dose does not effect uterine perfusion if injected into a vein,82 nor does the amount of local anesthetic absorbed from the epidural space after epidural anesthesia. However, higher concentrations of local anesthetics, achieved after inadvertent intravenous injection or after a paracervical block (see later), may cause uterine hypertonus, a decrease in UBF, and subsequent fetal bradycardia.71
EFFECT OF GENERAL ANESTHESIA ON UTEROPLACENTAL BLOOD FLOW.
Anesthetic agents and adjuvants, in general, have no direct effect on UBF. Hypotension associated with general anesthesia may adversely affect UBF. The typical rapid sequence induction of general anesthesia for cesarean delivery (induction agent, followed by neuromuscular blocking agent, followed by laryngoscopy) often is accompanied by a catecholamine surge that may cause a decrease in UBF.83 Mechanical hyperventilation decreases UBF.84,85 It is unclear whether this is a result of hypocarbia or a direct result of mechanical hyperventilation (resulting in decreased venous return and decreased cardiac output).84
The Placenta and Fetus
The anatomy and physiology of the placenta are discussed in detail elsewhere in this text. Whether anesthetic agents cross the placenta depends on a number of factors, including drug concentration and electrochemical gradients across the placenta, molecular weight, lipid solubility, degree of ionization, membrane surface area and thickness, maternal and fetal blood flow, placental binding and metabolism, and degree of maternal and fetal protein binding.86 Most drugs that parturients receive during the administration of analgesia/anesthesia cross the placenta. This includes volatile anesthetic gases, nitrous oxide, sedative/hypnotics, opioids, local anesthetics, antihypertensive agents, and vasopressors.86 Neuromuscular blocking agents and most anticholinesterases are charged molecules and have limited placental transfer. The effects of individual anesthetic agents on the fetus/neonate are discussed later.