Hygienic Perineal Talc
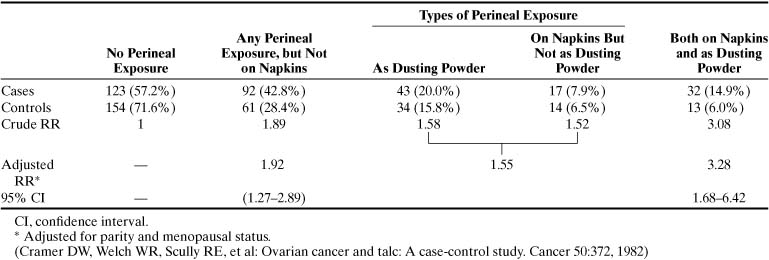
Because it is related to asbestos, talc has been implicated as a cause of ovarian cancer.4 Besides the chemical similarities between talc and asbestos, cosmetic talcum powder manufactured before 1976 contained significant amounts of the three types of asbestos: tremolite, chrysolite, and crocidolite.5,6 Studies by Heller and associates5 and Henderson and colleagues7 showed that talc particles may be found in normal and malignant tissues. Conceptually, talcum powder applied to the perineum can reach the ovaries via retrograde flow. The association between talc and ovarian cancer remains equivocal. Whittemore and associates8 could not show an association between genital talcum powder exposure and ovarian cancer; however, Cramer and coworkers9 reported a relative risk (RR) of 1.9 (p < .03) for ovarian cancer among talc users. The RR was increased further in women who used talcum powder on perineal napkins and dusting powder on the perineum (Table 1). Wong and coworkers10 reported, however, in a case-control study that included 704 controls and 430 cases of ovarian cancer no increased risk for ovarian cancer with the use of perineal talc. There was no increased risk with 20 or more years of use of perineal talc. In a meta-analysis of six studies, Harlow and colleagues11 calculated a modest risk for ovarian cancer development among talc users: RR 1.3 (95% confidence interval [CI], 1.1 to 1.6) (Table 2).11 Although these case-control studies showed a small increased risk for development of ovarian cancer with perineal talc use, in 2000 the scientific advisory panel to the National Toxicology Program, a branch of the National Institutes of Health, voted 7 to 3 against recommending that perineal talc be listed as a human carcinogen.12 The panel next meets in 2002.
Cases | Controls | |||||
Author (Year) |
Total |
Talc Exposure |
Total |
Talc Exposure |
Crude OR |
95% CI |
Cramer et al (1982) |
215 |
92 (42.8%) |
215 |
61 (28.4%) |
1.9 |
1.3–2.0 |
Hartge et al (1983) |
135 |
67 (49.6%) |
171 |
100 (58.5%) |
0.7 |
0.4–2.0 |
Whittemore et al (1988) |
188 |
98 (52.1%) |
539 |
248 (46.0%) |
1.4 |
0.9–2.0 |
Harlow et al (1989) |
116 |
49 (42.2%) |
158 |
64 (40.5%) |
1.1 |
0.7–2.0 |
Booth et al (1989) |
217 |
141 (65.0%) |
434 |
256 (59.0%) |
1.3 |
0.9–1.0 |
Harlow et al (1992) |
235 |
114 (48.5%) |
239 |
94 (39.3%) |
1.5 |
0.9–2.0 |
All studies |
1106 |
561 (50.7%) |
1756 |
823 (46.9%) |
1.3 |
1.1–2.0 |
CI, confidence interval.
(Harlow BL, Cramer DW, Bell DA, et al: Perineal exposure to talc and ovarian cancer risk. Obstet Gynecol 80:19, 1992)
Diet
The major dietary distinction in industrialized nations is the high intake of meat and animal fat, which has been reported to be associated with an increased incidence of ovarian cancer in several studies.13 Shu and associates14 reported a significant (p <.01)dose-response relationship between the intake of fat from animal sources and ovarian cancer risk in a case-control study of 172 epithelial ovarian cancer cases and 172 controls. In a prospective study of 20,305 women, Helzlsouer and colleagues15 showed that women with cholesterol levels greater than 200 mg/dL are at increased risk for ovarian cancer.
Lactose, found naturally only in milk, is the only source of dietary galactose. Ovarian tissue has a relative abundance of galactose-metabolizing enzymes, and experimental studies on rats indicated that galactose-rich diets can have an adverse effect on the ovaries.16,17,18,19 Young women with galactosemia, an inborn error of galactose metabolism, experience hypergonadotropic hypogonadism and early ovarian failure.20 Cramer and colleagues21 proposed that increased dietary galactose consumption and low serum levels of galactose-1-phosphate uridyltransferase, an important enzyme in the metabolism of galactose, may be linked to an increased risk of ovarian cancer. In a case-control study, the investigators found that women who consumed significantly more yogurt and cottage cheese, the only foods consumed regularly that distinguished the cases from the controls, had three times the incidence of ovarian cancer than the controls. Women with ovarian cancer were reported to have significantly lower galactose-1-phosphate uridyltransferase levels than controls (Table 3). These authors proposed that in women with an inherited abnormality of galactose-1-phosphate uridyltransferase, the ovaries would be exposed to galactose for a prolonged period, increasing the ovarian cancer risk in these women compared with women who can metabolize galactose efficiently.
TABLE 3. Risks for Ovarian Cancer by Lactose Consumption and Transferase
Activity
No. Subjects (%) | ||||
Lactose Consumption (g/day) | Combination Transferase Activity (mmol/hr per g Hb) | Cases (n = 145) | Controls (n = 127) | RR (95% CI) |
|
≤ 11 |
≥ 22.9 |
29 (20) |
36 (28) |
1.0 |
|
≤ 11 |
< 22.9 |
30 (21) |
35 (28) |
1.1 (0.5–2.2) |
|
> 11 |
≥ 22.9 |
33 (23) |
29 (23) |
1.3 (0.6–2.7) |
|
> 11 |
< 22.9 |
53 (36) |
27 (21) |
2.2 (1.1–4.5) |
Cl, confidence interval; RR, relative risk.
(Cramer DW, Harlow BL, Willett WC, et al: Galactose consumption and metabolism in relation to the risk of ovarian cancer. Lancet 2:66, 1989)
Several other studies found no association between galactose consumption and the incidence of ovarian cancer. In a case-control study of 450 patients, Risch and associates22 showed that galactose consumption did not increase the risk of epithelial ovarian cancer (Table 4). In a 2000 report of the Nurses’ Health Study of 80,326 women who completed a dietary questionnaire, Fairfield and coworkers23 reported, however, a 44% greater risk for the development of invasive epithelial ovarian cancer for women with the highest consumption of lactose compared with women with the lowest consumption (RR, 1.44; 95% CI, 1.01 to 2.07; p for trend 0.07). The largest contributors to dietary lactose were skim and low-fat milk.3 Another, perhaps better relationship between milk consumption and ovarian cancer was proposed by Mettlin and Piver,24 who evaluated this association in terms of animal fat content in milk. The RR for ovarian cancer was 3.1 for women drinking more than one glass of whole milk each day compared with women who never drank whole milk (Table 5). Women who drank only whole milk were at increased risk for ovarian cancer compared with women who drank skim or 2% fat milk. These findings suggest that animal fat, described as the fat content in milk, may be the source of increased risk.
Adjusted Odds Ratio | (95% Confidence Interval) | ||
Nutrient/Item | Unit/d (g) | Model 1 | Model 2 |
Lactose | |||
All subjects |
10 |
1.03 (0.93–1.12) |
0.99 (0.90–1.09) |
Subjects not reporting lactose intolerance |
10 |
1.02 (0.93–1.13) |
0.99 (0.90–1.09) |
Subjects reporting lactose intolerance |
10 |
0.89 (0.64–1.25) |
0.84 (0.60–1.18) |
Subjects who never used OC |
10 |
1.11 (0.97–1.27) |
1.06 (0.93–1.22) |
Subjects who ever used OC |
10 |
0.98 (0.89–1.09) |
0.95 (0.85–1.06) |
Whole milk |
300 |
1.20 (0.80–1.80) |
0.98 (0.64–1.49) |
2% milk |
300 |
0.99 (0.85–1.15) |
0.92 (0.79–1.08) |
Skim milk |
300 |
1.04 (0.84–1.29) |
1.08 (0.87–1.35) |
Yogurt |
30 |
1.02 (0.96–1.09) |
1.03 (0.97–1.10) |
OC, oral contraceptives.
(Adapted from Risch HA, Jain M. Marrett LD, et al: Dietary lactose intake, lactose intolerance, and the risk of epithelial ovarian cancer in Southern Ontario [Canada]. Cancer Causes Control 5:540, 1994)
TABLE 5. Ovarian Cancer and Whole Milk Consumption
Frequency (Total) | Ovarian Cancer (297 Total) | Controls (587 Total) | Relative Risk |
Never |
174 |
410 |
1 |
Less than one glass daily |
62 |
109 |
1.3 |
One glass daily |
25 |
41 |
1.4 |
More than one glass daily |
36 |
27 |
3.1 |
p < 0.001 |
Infertility and Fertility Drugs
Infertility and the use of fertility drugs have been found to increase the risk of ovarian cancer.25,26,27,28 In a case-control study of 3837 women evaluated for infertility, Rossing and colleagues25 observed an increased ovarian cancer risk among infertile women (standardized incidence ratio [SIR], 2.5; 95% CI, 1.3 to 4.5); this study suggested that women with infertility resulting from abnormalities in ovulation were at greater risk for ovarian cancer (SIR, 3.7; 95% CI, 1.4 to 8.1) than women with infertility resulting from tubal abnormalities (SIR, 3.0; 95% CI, 0.4 to 10.8). Similarly, Whittemore and coworkers26 reported that it is not the estimated number of years of ovulation, the number of pregnancies, or the lack of use of oral contraceptives that increases a woman’s risk for ovarian cancer, but rather the inability to conceive among ovulating women who have sexual intercourse for 10 or more years, unprotected by any type of contraception (Table 6).
TABLE 6. Ovarian Cancer Risk of Unprotected Intercourse
Years of Unprotected Intercourse | Ovarian Cancer | Controls | Relative Risk | p Value |
|
< 2 |
91 (50%) |
323(62%) |
1 |
|
|
2–9 |
48 (26%) |
117(22%) |
1.5 |
0.08 |
|
> 10 |
43 (24%) |
82(16%) |
1.8 |
0.01 |
The use of ovulation-inducing agents is increasing in the United States; in the 1980s and 1990s, the number of clomiphene citrate prescriptions dispensed annually nearly doubled.29 In the late 1990s, researchers raised concerns regarding the relationship between ovulation-inducing agents and the development of ovarian cancer. Rossing reported25 an increased risk of ovarian cancer among women who had ever used clomiphene citrate (SIR, 3.1; 95% CI, 1.4 to 5.9) and a further increase in risk of ovarian cancer among women who used clomiphene for 12 or more cycles (SIR, 11.1; 95% CI, 1.5 to 82.3). In a nationwide case-control study from Israel, Shushan and associates28 reported an adjusted odds ratio (OR) of 3.19 (95% CI, 0.86 to 11.82) among women who used human menopausal gonadotropins alone and an adjusted OR of 1.42 (95% CI, 0.65 to 3.12) among women who used human menopausal gonadotropins alone or combined with clomiphene citrate. In an analysis of fertility drug use from eight case-control studies conducted between 1989 and 1999 in the United States, Denmark, Canada, and Australia, Ness and coworkers30 reported, however, no increased risk for the development of invasive epithelial ovarian cancer among infertile women taking clomiphene citrate or human menopausal gonodotropin.
Estrogen Replacement Therapy
Estrogen replacement therapy (ERT) has been used to relieve menopausal symptoms such as vaginal dryness and hot flashes; studies have shown that ERT has significant protective effects benefiting the cardiovascular system, bones, and lipoprotein profile.31,32,33 The association between ERT and ovarian cancer is inconclusive, however.34,35,36,37,38,39 Results from the Collaborative Ovarian Cancer Group showed no risk of ovarian cancer among ERT recipients (OR, 1.1; 95% CI, 0.59 to 2.0), and several studies agreed with these findings.34,39 Cramer and colleagues37 reported an association, although not statistically significant, between ERT and the development of ovarian cancer (RR, 1.56; 95% CI, 0.85 to 2.87). The most controversial report on ERT and ovarian cancer is that of the Department of Epidemiology and Surveillance Research of the American Cancer Society.40 These investigators did not report on the association between the use of ERT and the risk of developing ovarian cancer but rather that among postmenopausal women, the use of ERT in the 1970s and early 1980s for 10 or more years doubled the risk of dying from ovarian cancer (Table 7). The authors assumed that women took estrogen alone (without progesterone) and admitted that they did not know which estrogen product the women took.
ERT Status | No. of Deaths | Adjusted Rate Ratio | 95% Confidence Interval |
Never used |
689 |
1.00 |
|
Ever used |
255 |
1.23 |
1.06–1.43 |
Years of use baseline users |
|||
<10 |
31 |
1.14 |
0.79–1.65 |
≥10 |
31 |
2.20 |
1.53–3.17 |
Years of use former users |
|||
<10 |
158 |
1.10 |
0.92–1.31 |
≥10 |
35 |
1.59 |
1.13–2.25 |
Genetic Factors
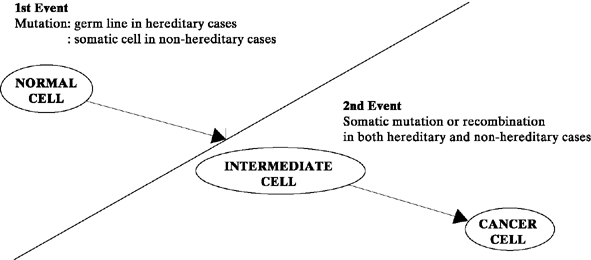
It is commonly accepted that cancer results from a series of genetic alterations.41,42 In 1972, Knudson and Strong43 proposed the two-hit mutation model for the development of cancer (Fig. 1). According to this concept, in inherited cancers, the first mutation (single-gene alteration) is prezygotic, making the fetus predisposed to the development of cancer from conception onward. Cancer develops in the somatic cells (in this case, the ovary), however, only when a postzygotic second mutation occurs after birth. In contrast, nonhereditary cancers occur when both mutations occur postzygotically in the somatic cell line (the ovary). Knudson and Strong’s hypothesis helps explain why hereditary cancers frequently occur 10 to 20 years earlier than nonhereditary cancers (i.e., because the first mutation has occurred prezygotically).
|
Genetic changes in two categories of normal cellular genes—proto-oncogenes and tumor-suppressor genes—have been proposed to cause cancer.41,42,44 Genetic changes (e.g., point mutations, translocations, amplifications) in proto-oncogenes facilitate the transformation of a normal cell to a malignant cell by producing an altered or overexpressed gene product. Products of proto-oncogenes include cellular growth factors, growth factor receptors, enzymes, and DNA-binding proteins.45,46 Tumor-suppressor genes are proposed to prevent cancer.42,44,45,46,47,48 Inactivation or loss of these genes contributes to the development of cancer because of the lack of a functional gene product. Dysfunction of tumor-suppressor genes could result in abnormal cell division, abnormal gene expression, or the increased ability of cells in tissue to detach.
Activation of several proto-oncogenes (particularly K-ras, h-ras, c-myc, and HER-2/neu) has been assayed by various techniques in ovarian tumors and in established ovarian tumor cell lines.49,50,51 These techniques have measured the ability of isolated tumor DNA to transform the murine fibroblast cell line (NIH/3T3) and the amplification of proto-oncogene messenger RNA and protein by tumors; data from these investigations showed that activation of proto-oncogenes occurs in ovarian tumors. The significance of proto-oncogene activation and the role, if any, it plays in the development of ovarian cancer remain to be determined.
Until more recently, there was little evidence in the literature that the origin of some ovarian cancers was either genetic or familial. In 1979, we reported a family in which epithelial ovarian cancer developed in five members, spanning three generations: a grandmother, three daughters, and a granddaughter.52 Shortly thereafter, five members of a second family, spanning two generations (three sisters, a first cousin, and her daughter), came under our care for epithelial ovarian cancer.53 Our review of the literature revealed that before 1970, familial ovarian cancer had been reported in only five families. In contrast, during the 1980s, 26 more families were documented as having familial ovarian cancer. In response to this seemingly real increase in the incidence of familial ovarian cancer, and because of the need for appropriate genetic counseling, a Familial Ovarian Cancer Registry was established at Roswell Park Cancer Institute in 1981 to document the number of cases occurring in the United States and to study the mode of inheritance. It was postulated that if an autosomal dominant mode of transmission was established, genetic counseling for prophylactic oophorectomy at an appropriate age might lead to a decrease in the death rate from ovarian cancer in such families.
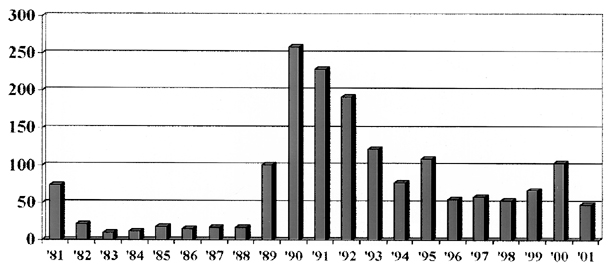
The Registry’s first report in 1984 documented 201 cases of ovarian cancer in 94 families.54 After the initial collection of 76 cases in 1981, the average number of cases enrolled between 1982 and 1996 was 189 per year (range, 32 to 635). Because of an increased awareness of a possible genetic link generated by the media, the Registry now receives daily information on new families with familial ovarian cancer. In 1990, the Registry was renamed the Gilda Radner Familial Ovarian Cancer Registry (GRFOCR) in honor of the comedic actress, Gilda Radner, who died of ovarian cancer. Through December 31, 2001, the Registry has enrolled 1652 families with two or more close relatives with ovarian cancer (Fig. 2). The Registry has made it clear that familial ovarian cancer is not a rare occurrence, as originally conceived in the four decades from 1930 to 1970.
Three conditions are associated with familial ovarian cancer: (1) site-specific ovarian cancer, in which the familial risk is restricted to ovarian cancer; (2) breast/ovarian cancer syndrome, with clustering of ovarian and breast cancer cases in extended pedigrees; and (3) cancer syndrome family, also known as Lynch syndrome II, which consists of hereditary nonpolyposis colorectal cancer (with most of the cancers occurring in the proximal colon) in association with ovarian or endometrial carcinoma or both.55 In all of these syndromes, inheritance seems to follow an autosomal dominant pattern with variable penetrance. In addition, several genetic factors are associated with increased gonadal dysgenesis (dysgerminoma),56 Peutz-Jeghers syndrome (mucocutaneous pigmentation or gastrointestinal polyposis, granulosa cell tumor, and cystadenoma),57 and basal cell nevus syndrome (fibroma).58
Hall and associates59 in 1990 mapped by genetic linkage a locus for familial ovarian cancer to the long arm of chromosome 17 in the interval 17q12-21. Before the cloning of BRCA1 by Miki and coworkers60 in 1994, the GRFOCR participated in the Breast Cancer Linkage Consortium Study of 145 breast/ovarian cancer families. 61 These families consisted of three or more cases of early onset (<60 years old) breast or ovarian cancer. It was estimated that 92% of the families in which there was no male breast cancer and with two or more cases of ovarian cancer were linked to BRCA1. The GRFOCR participated in another international study of nine families with site-specific ovarian cancer defined as three or more cases of epithelial ovarian cancer and no case of breast cancer diagnosed at less than 50 years of age. This study documented that 89% of such site-specific ovarian cancer families are linked to BRCA1.62,63
In 1995, Wooster et al64 cloned BRCA2, which previously had been mapped by genetic linkage to chromosome 13q12. It is now estimated that BRCA1 and BRCA2 are responsible for 5% to 10% of all epithelial ovarian cancers with 52% BRCA1, 32% BRCA2, and 16% other yet undefined genes.65 In the United States, lifetime risk of developing epithelial ovarian cancer is 1 in 55 or 1.8% compared with 28% to 44% for BRCA1 carriers and 27% for BRCA2 carriers.
Because familial ovarian cancer (site-specific) is thought to be inherited in an autosomal dominant pattern with variable penetrance, sisters and daughters in families with a history of familial ovarian cancer (two or more first-degree relatives, mother/sister/daughter, who share one half of one’s genes) may have a 50% chance of disease development. This figure compares with a 1.8% (1 in 55) in women without a family history. It has been recommended that such women undergo a prophylactic oophorectomy by age 35 if they have completed childbearing. A word of caution: There have been reports of women with a family history of ovarian cancer who have been diagnosed with intra-abdominal carcinomatosis histologically similar in appearance to ovarian adenocarcinoma (also known as extraovarian papillary carcinoma of the peritoneum) after prophylactic oophorectomy.66 In some cases, the prophylactically removed ovaries were cancerous on retrospective review67; however, in other cases of extraovarian papillary carcinoma, the prophylactically removed ovaries were cancer-free. It is postulated that whatever causes ovarian cancer also affects the peritoneum to form an extraovarian papillary carcinoma. From 1981 to July 1982, the GRFOCR had enrolled 931 families totaling 1222 cases of familial ovarian cancer. Of these, 324 had undergone prophylactic oophorectomy.68 Of these 324 women, 6 or 1.8% developed primary peritoneal carcinoma indistinguishable from epithelial ovarian cancer 1, 2, 5, 13, 15, and 27 years after prophylactic oophorectomy. Although these data suggest that development of primary peritoneal carcinoma after prophylactic oophorectomy for a family history of familial ovarian cancer is not a common event, they do point out that all women who undergo such preventive surgery need to be under lifetime surveillance for the possible development of primary peritoneal carcinoma.