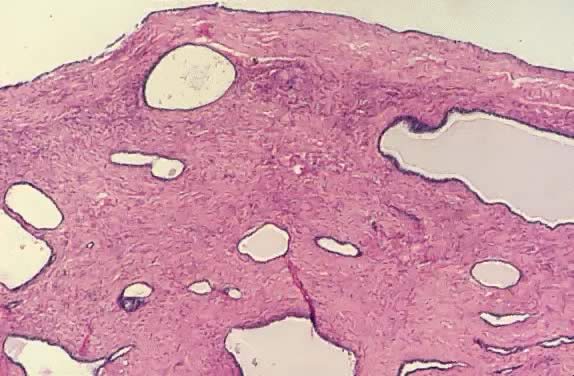
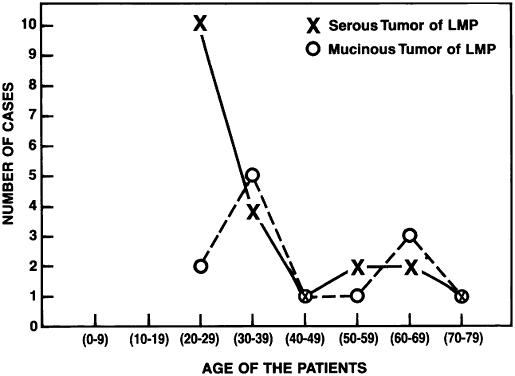
Of all serous tumors, 10% to 15% are LMP tumors.2 In the cases of LMP tumors from Magee-Women's Hospital,8 51% were classified as serous. Most of the patients in the Magee-Women's Hospital series were between 24 and 33 years
of age, which concurs with previous data.7 In general, most of the LMP ovarian tumors occurred in women between 45 and 50 years
of age2 (see Table 2). Jensen and Norris,14 however, reported a few cases occurring in patients between 15 and 19 years
of age. The mean age varies from 40 years in Michigan to 53 years
in Norway.15 The LMP tumors occur at a younger age than do frank carcinomas. The bilaterality
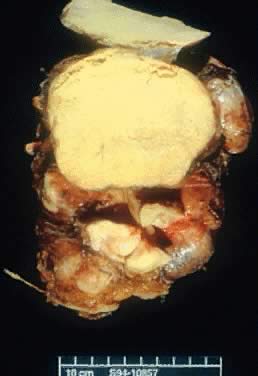
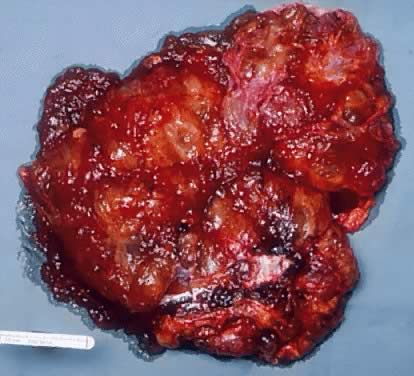
was 25%, which is within the range reported in the literature.16 Gross Examination Serous tumors of LMP are grossly similar to serous cystadenoma. They are
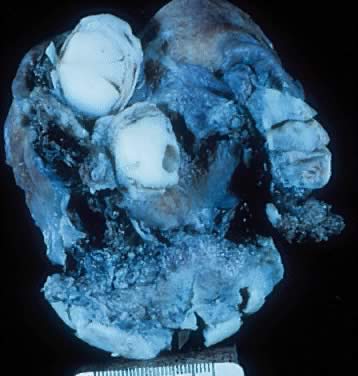
round, ovoid, and multilobulated, and their size varies from a few centimeters
to 28 cm in diameter. On inspection, they are reddish, pink, or
yellowish blue, sometimes with brown discoloration secondary to intramural
or intraluminal hemorrhage. Their surfaces frequently are smooth
and sometimes intermingled with irregular indurated areas representing
thickened fibrous tissue or dystrophic calcification. Veins present
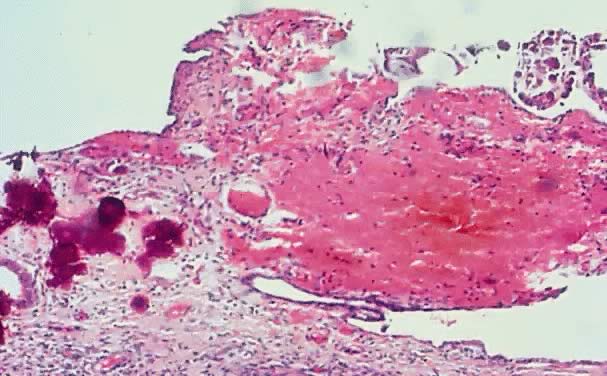
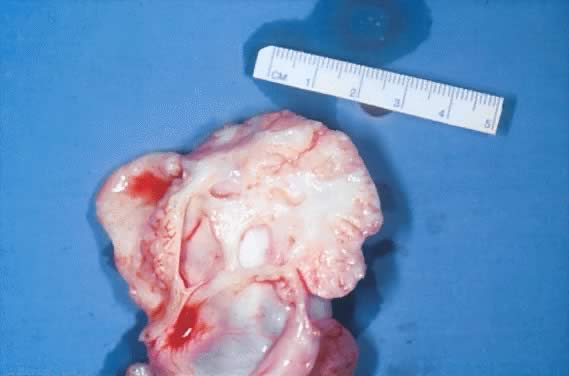
in the cystic wall can be prominent (Fig. 2). Translucency often is observed depending on the cystic contents. 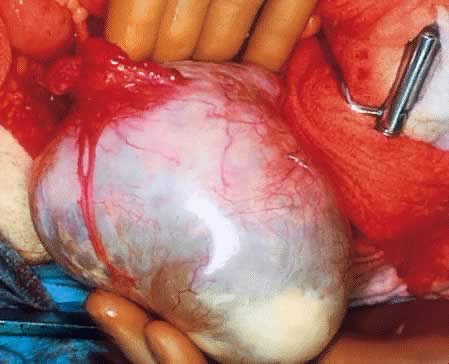 Fig. 2. External surface of a serous cystadenoma of low malignant potential showing
a smooth, tense capsule with prominent veins and thickened fibrous
areas.(Courtesy of Dr. Richard Stock, Naval Hospital, Portsmouth, VA.) Fig. 2. External surface of a serous cystadenoma of low malignant potential showing
a smooth, tense capsule with prominent veins and thickened fibrous
areas.(Courtesy of Dr. Richard Stock, Naval Hospital, Portsmouth, VA.)
|
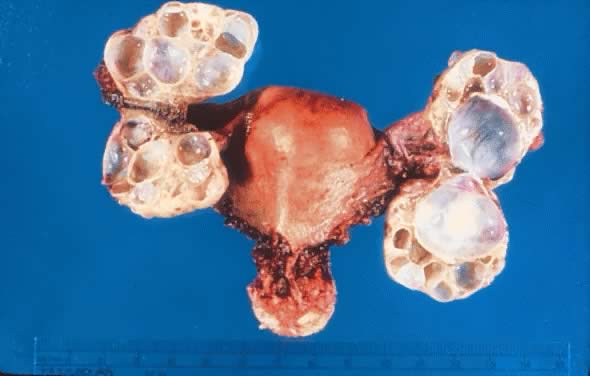
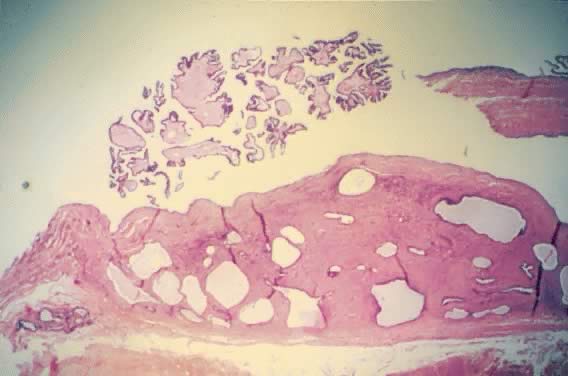
On palpation, the tumors can be fluctuant. Exuberant papillary projections
may be observed on the external surface of the cyst. On section, the
tumors are cystic (unilocular or multilocular), having secondary or “daughter” cysts of varying sizes and shapes,17 with clear, watery contents (and, therefore, serous cysts). Frequently, the
tumor's content is seromucinous, indicating the presence of
serous and mucinous lining epithelium,17 which has been confirmed by electron microscopic study.18 Tumors with this histogenic makeup are designated as mixed LMP tumors (described
later). Mucus can be secreted by the apical portion of serous
tall columnar cells. To designate a tumor as mixed, some investigators
require that there be at least a 10% mixture of epithelial components (serous, mucinous, or
endometrioid); others consider the required
proportion to be at least 40%10 (Fig. 3). Necrosis is rare, although it is more frequent in serous than in mucinous
cysts. It is not always possible to distinguish macroscopically
a benign tumor from an LMP tumor, or the latter from its malignant counterpart. The
presence of papillary projections on the internal or external
surface of the cyst, which frequently are tan, suggest a borderline
tumor or tumor of LMP.2 The papillary projections in the cyst lumen are more complex and luxuriant
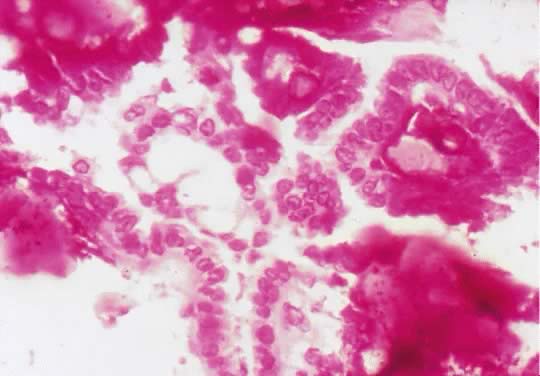
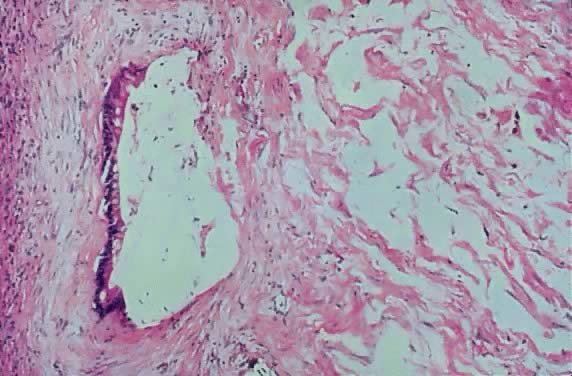
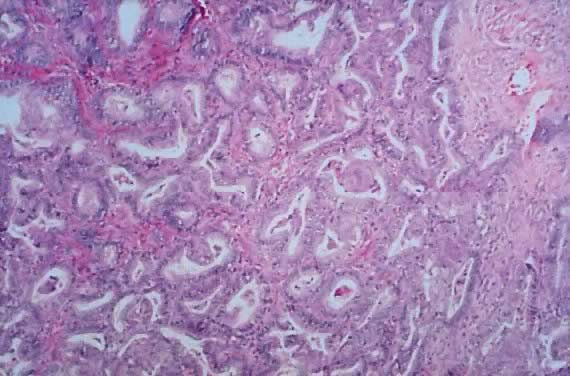
in an LMP tumor than in a benign cystadenoma (Figs. 4 and 5). 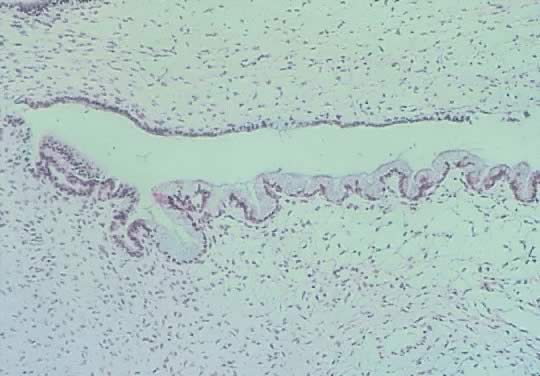 Fig. 3. Cystadenoma exhibiting lining consisting of cuboidal epithelium serous
type ( top) and mucus-producing tall, columnar, endocervical-type epithelium with
basally arranged nuclei ( bottom ). The mixture of epithelia frequently is found in serous or mucinous
cystadenomas of the ovary (hematoxylin and eosin, Fig. 3. Cystadenoma exhibiting lining consisting of cuboidal epithelium serous
type ( top) and mucus-producing tall, columnar, endocervical-type epithelium with
basally arranged nuclei ( bottom ). The mixture of epithelia frequently is found in serous or mucinous
cystadenomas of the ovary (hematoxylin and eosin, 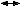 520). 520).
|
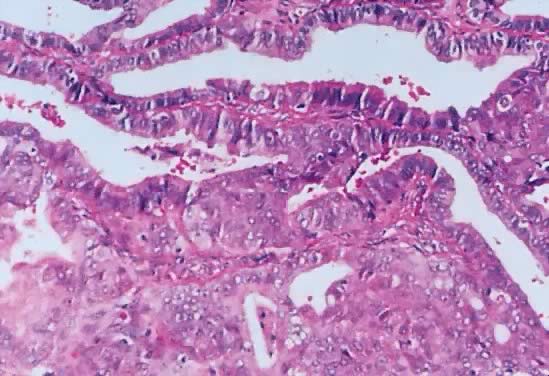
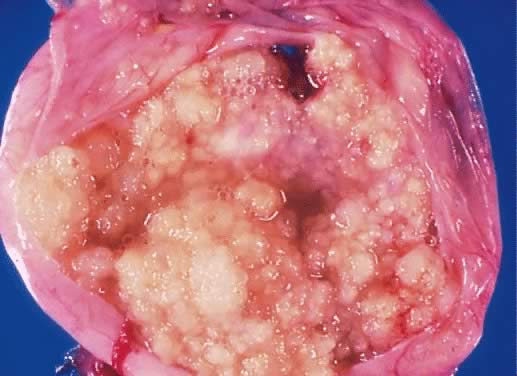 Fig. 4. Serous cystadenoma of low malignant potential demonstrating an exuberant
growth of papillary projections arising from the internal surface of
the tumor. Bubbly serous fluid is present in the lumen.(Courtesy of Dr. Richard Stock.) Fig. 4. Serous cystadenoma of low malignant potential demonstrating an exuberant
growth of papillary projections arising from the internal surface of
the tumor. Bubbly serous fluid is present in the lumen.(Courtesy of Dr. Richard Stock.)
|
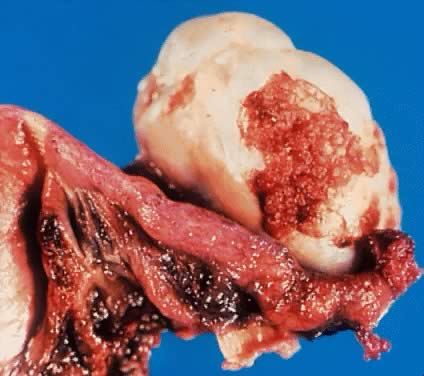 Fig. 5. External surface of an ovary showing a serous cystadenoma of low malignant
potential exhibiting papillary growth. The lateral aspect of the uterus
and the entire fallopian tube are seen clearly. The papillary growth
does not represent invasion.(Courtesy of Dr. Richard Stock.) Fig. 5. External surface of an ovary showing a serous cystadenoma of low malignant
potential exhibiting papillary growth. The lateral aspect of the uterus
and the entire fallopian tube are seen clearly. The papillary growth
does not represent invasion.(Courtesy of Dr. Richard Stock.)
|
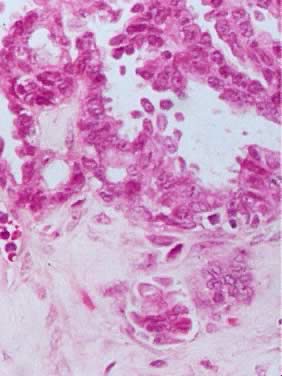
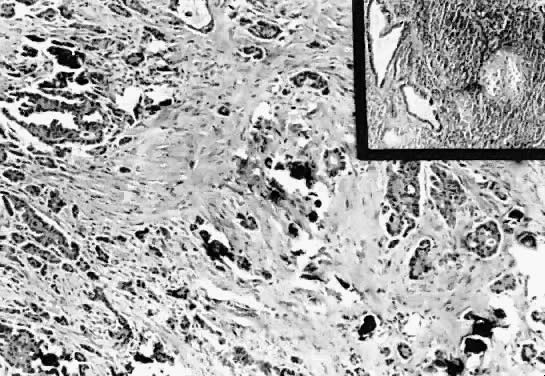
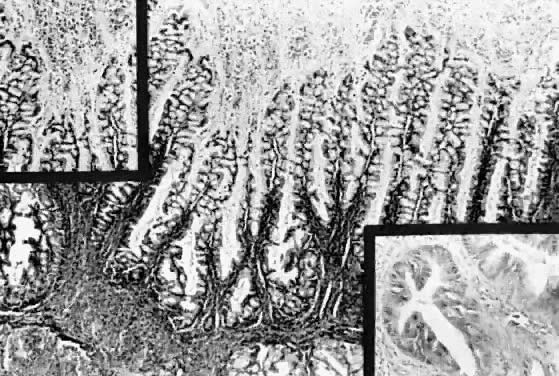
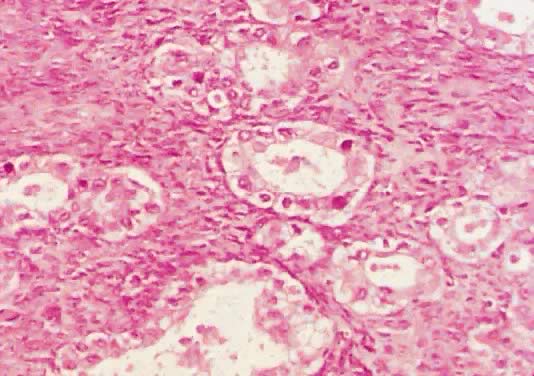
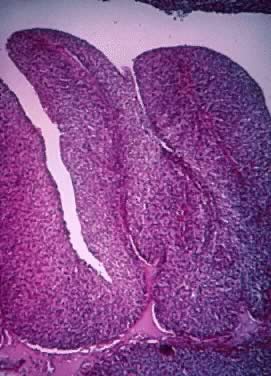
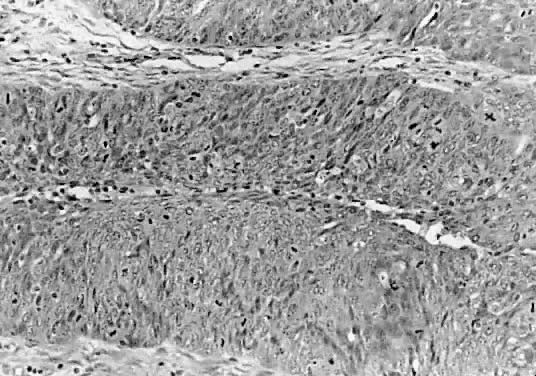
Microscopic Findings On microscopic examination, the LMP serous tumors are characterized by
the presence of simple or complex, broad and fine papillary structures (Fig. 6). They have a distinctive fibrovascular core covered by different types
of columnar epithelia with atypical nuclei and tufting. The papillary
structures may occur on the external surface in approximately 50% of
the stage I tumors (see Fig. 5). Notice that the surface excrescences are the result of tumor growing
from the surface epithelium concomitantly with the excrescences in the
lumen of the cyst. The excrescences on the external surface do not signify
that the tumor has penetrated the wall of the cyst. In few cases, the
papillary excrescences represent the only tumor present in the
ovary.17,18 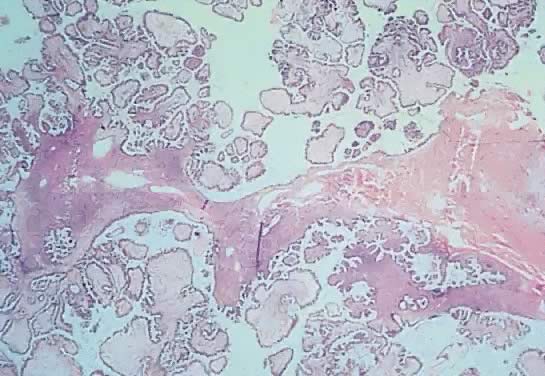 Fig. 6. Photomicrograph of a serous cystadenoma of low malignant potential. The
tumor is composed mainly of papillary structures of different sizes resting
on stroma made up of fibrous tissue. Tufting is observed in the
interpapillary space ( Fig. 6. Photomicrograph of a serous cystadenoma of low malignant potential. The
tumor is composed mainly of papillary structures of different sizes resting
on stroma made up of fibrous tissue. Tufting is observed in the
interpapillary space (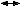 100). 100).
|
The LMP serous tumor may display all of the types of epithelial cells normally
seen in the fallopian tube: ciliated, clear, intermediate, and
mucus secreting.19,20 The cytoplasm of the serous cells is clear or eosinophilic. The basally
located nucleus is somewhat irregular, hyperchromatic, and slightly
pleomorphic. The epithelium, when stratified, should not have more than
five layers of cells, which display loss of polarity and atypia. The
mitoses are not atypical, although, their activity usually is increased. The
frequency of mitosis varies among cases. In our series,8 there were cases in which the mitotic count varied from 0 per 10 high-power
fields (HPF) to 9 per 10 HPF (mode, 0 to 2 per 10 HPF). Psammoma bodies are observed frequently. The border between the abnormal
epithelium and the subjacent or surrounding mesenchymal tissue is sharp, regular, and
smoothly contoured. The invagination of the epithelium
may create diagnostic difficulties; in these cases, observation of
their margins reveal smooth rather than jagged edges and the absence of
stromal edema, inflammation, or neovascularization. The changes of the
epithelium are of the same type throughout. It is unusual to find areas
of the tumor exhibiting totally benign epithelium alternating with
areas of LMP tumor.21 In some cases, there is penetration of the stroma by glandular-like elements
and microcysts with papillae without exhibiting stromal reaction; therefore, it
should not be interpreted as an stromal invasion. In
some cases, sharply delineated desmoplastic plaques, most frequently on
the external surface, are noted. Sometimes these thickened areas are
observed on the inner aspect of the wall. These have been designated
as autoimplants.22 Micropapillary, cribriform, or solid histologic patterns also can be seen. These
histologic patterns have been described only sporadically. Therefore, their
behavioral significance are not known. The micropapillary
and cribriform types can be associated with poor prognosis, but this
has not been corroborated.23 The LMP serous tumor and benign serous cystadenoma usually have cilia. These
cilia are absent in the cyst adenocarcinoma.20 Unfortunately, this histologic factor is not of any practical value in
the differential diagnosis. Epithelial cells devoid of connective tissue detach, fall, and form rosette-like
structures that float in the cystic fluid; this phenomenon is
designated as tufting (Figs. 7 and 8). Hyalinization of the connective tissue of the papillary structures is
seen more often in the LMP serous tumors than in serous cystadenomas.19 Papillae undergoing ischemic changes show edema and frequently slight
acute inflammation of the stroma. In more advanced cases, frank necrosis
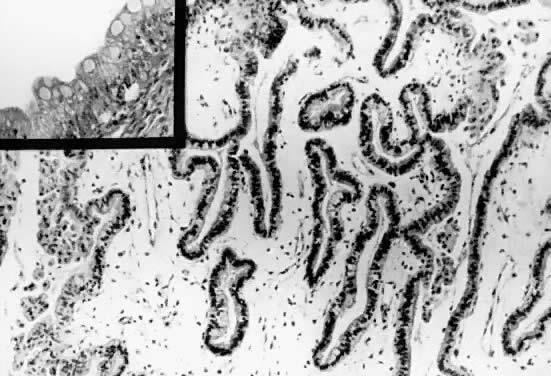
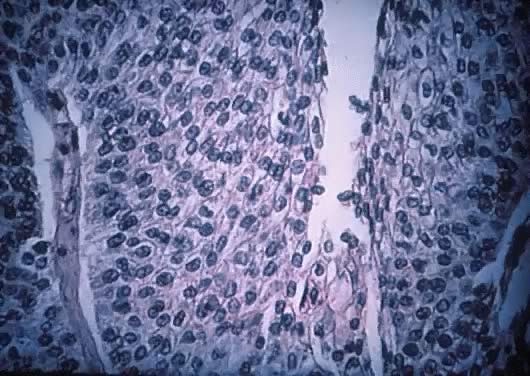
may be evident. 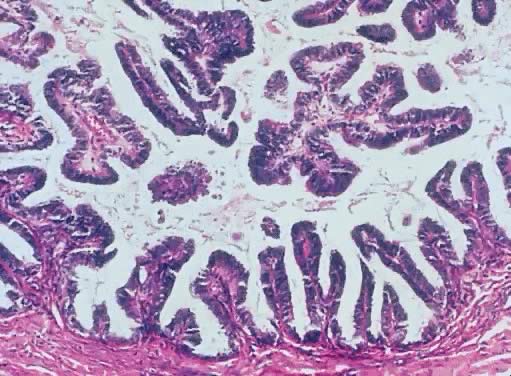 Fig. 7. Inner lining of a serous cystadenoma of low malignant potential showing
papillations with epithelial pluristratification, hyperchromasia, and
tufting; there is some degree of nuclear atypia ( Fig. 7. Inner lining of a serous cystadenoma of low malignant potential showing
papillations with epithelial pluristratification, hyperchromasia, and
tufting; there is some degree of nuclear atypia ( 420). 420).
|
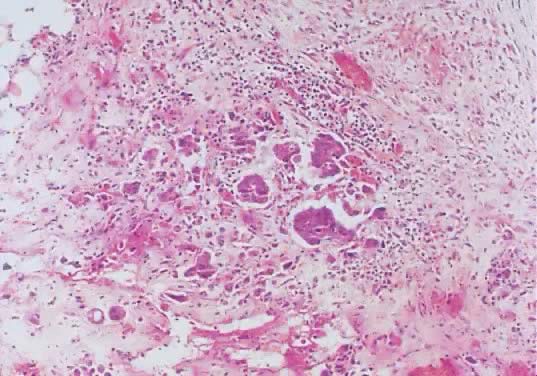
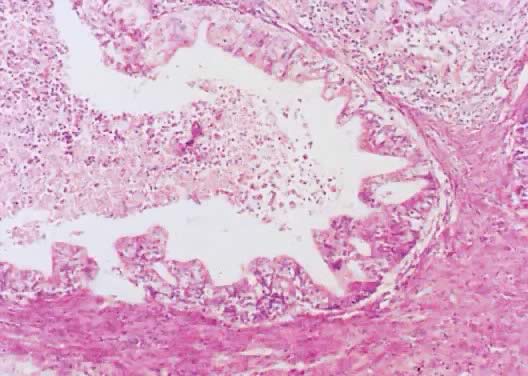
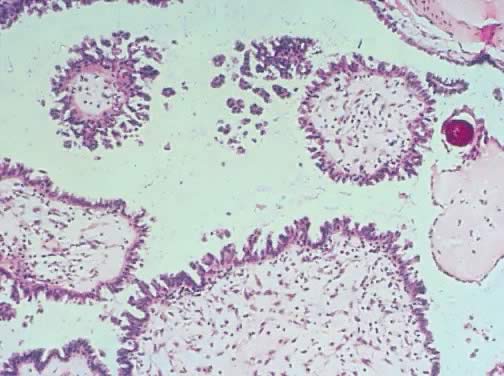 Fig. 8. Serous cystadenoma of low malignant potential demonstrating papillae, stratification
of epithelial lining, tufting, stromal edema with hyalinization, and
focal acute inflammatory cell infiltrate. On the upper right-hand
corner, a psammoma body is clearly seen (hematoxylin and eosin, Fig. 8. Serous cystadenoma of low malignant potential demonstrating papillae, stratification
of epithelial lining, tufting, stromal edema with hyalinization, and
focal acute inflammatory cell infiltrate. On the upper right-hand
corner, a psammoma body is clearly seen (hematoxylin and eosin,  420). 420).
|
Of all the histologic characteristics discussed, the most helpful and important, which
can be used to separate the tumor of LMP from a malignant
neoplasm, is the stromal invasion by the epithelium. Unfortunately, the
determination of stromal invasion can be difficult in some LMP tumors. Another
problem is the lack of uniformity among pathologists in
determining the existence of invasion. In general, invasive serous carcinoma
is typified by the presence of poorly defined nests of cells with
marked atypia and irregular borders that infiltrate the stroma. This
stroma exhibits a loose, edematous, myxoid, or desmoplastic pattern, sometimes
associated with necrosis and inflammatory infiltrate. Histochemically, the epithelial cells of serous LMP tumors contain material
that is periodic acid-Schiff-positive but diastase resistant. The
same material also reacts positively with mucicarmine and Alcian blue
on its luminal border.17 When viewed with the electron microscope, the cell nuclei are convoluted
and the cytoplasm reveals the presence of cilia, which, as mentioned
earlier, are absent in serous cystadenocarcinoma.24 Studies of epithelial nuclear deoxyribonucleic acid (DNA) in LMP tumor
produce conflicting results. One study demonstrates that these neoplasms
exhibit diploid patterns more frequently in early stages (I and II) than
in stage III.25 Friedlander and coworkers26 demonstrated that the DNA content of cells derived from LMP serous tumors
are diploid in 95% (38 patients) and aneuploid in 5% (2 patients). Of
these two, one patient died of progressive disease. Retrospectively, it
has been shown than some of the aneuploid cases were carcinomas
that were erroneously diagnosed as LMP tumors. In a study27 of 44 primary ovarian serous tumors of LMP and 16 implants, about half
of the primary tumors had a diploid pattern, and the other half, an aneuploid
pattern. In 30% of the cases, the ploidy of the implants differed
from that of the primary neoplasms. Of the 44 patients, 4 died: of
these 4 patients, 1 had a diploid DNA pattern and the other 3, an aneuploid
pattern. The implants in the four patients were aneuploid. Therefore, if
the implants are invasive and aneuploid, the prognosis is unfavorable. Overall, however, there is little utility to flow cytometric
examination in LMP tumors. In a series investigated by Kabawat and associates,28 most of the serous malignant tumors, the LMP serous tumors, and 11% of
the benign serous cystadenomas expressed the OC-125 monoclonal murine
antigen on their surface. Its presence does not correlate with the morphologic
pattern and, therefore, probably is not significant for the
determination of tumor differentiation. Another frequent finding in the LMP serous tumors is the presence of calcium
deposits, some of which have a concentric laminated structure and
are known as psammoma bodies (see the section of the serous psammocarcinoma
of the ovary later). They are more abundant in the peritoneal
implants within the fibrous tissue than in the ovarian tumor itself. In
our review of LMP tumors, psammoma bodies were present in 60% of the
cases. The psammoma bodies are discussed in more detail later. Eichhorn and coworkers29 report 40 cases of serous tumors diagnosed as borderline with cribriform
pattern, micropapillary pattern, or both. Of the 40 cases, 26 tumors (65%) were
of the micropapillary type, 11 (27.5%) were cribriform, and 3 (7.5%) had
both patterns. The micropapillary pattern was found in
patients 21 to 76 years of age (mean, 38 years). The patients with cribriform
pattern were 34 to 79 years (mean, 60 years). Those with both
patterns also had a mean age of 38 years. The criteria30,31 for the diagnosis of these two subsets of serous tumors of LMP are as
follows: - Tumors with micropapillary pattern characterized by small, uniform papillae
with cores containing little or no stroma emanating in a nonhierarchical
fashion from large fibrotic, edematous, or myxoid stalk or directly
from the cyst wall; or a cribriform pattern of the cells lining
the stalk or cyst walls; or both
- Involvement by either or both patterns of at least one confluent area (5 mm) in
greatest dimension on at least one slide
- No more than grade 2 nuclear atypia (with grade 3 atypia being defined
as the presence of “markedly enlarge pleomorphic and vesicular” nuclei
with “prominent eosinophilic nucleoli”)
MICROPAPILLARY PATTERN. The criteria just outlined for micropapillary LMP serous tumors was modified
by Eichhorn and coworkers,29 who require that the papillae are at least “five times as long as
wide.” Microinvasion was defined as the presence in the stroma
of single cells or small nests of cells with little or no stromal response
occupying one or more foci with an area of less than 10 mm2. To establish the presence of invasive implants, invasion of the underlining
tissue with tumor replacing or destroying it should be demonstrated. The micropapillary tumors were between 2.5 and 19 cm in diameter; 69% were
bilateral, and 50%, intracystic. Only 15% were exophytic, and 35% were
both intracystic and exophytic. Four percent of the tumors were microinvasive. The
micropapillary component of the epithelial area was 10% to 25% in
seven cases. In another seven cases, the micropapillary
area was between 25% and 50%. In 12 cases, the micropapillary area was
over 50%. In all cases, the mitotic rate was not more than 1 per 10 HPF. CRIBRIFORM PATTERN. The size of the tumors with cribriform pattern was similar to that of
the micropapillary tumors, and the bilaterality was only 36%. These tumors
were intracystic in 55% of the cases—more frequent than the
micropapillary tumors. Of 11 tumors, 1 (9%) was exophytic, 4 (36%) were
both intracystic and exophytic, and 1 showed microinvasion. In 3 of 11 cases, the
cribriform pattern represented 10% to 25% of the epithelial
surface. In two cases, the cribriform pattern occupied between 25% and 50% of
the epithelial area. In two cases, the cribriform architecture
occupied more than 50% of the epithelial surface. Mitoses were no
more than 1 of 10 HPF. The micropapillary and cribriform subtypes of serous tumors of LMP were
more frequently associated with invasive implants. In the series by Eichhorn
and coworkers,29 12% of serous LMP tumors were micropapillary and cribriform; of those, 65% were
pure micropapillary, 27.5% were cribriform, and 7.5% were mixed. Both
micropapillary and cribriform types of serous LMP tumors were
treated almost 10 years earlier than those with pure serous LMP tumors. The
patients with micropapillary-type tumors presented for treatment
more frequently in a more advanced stage, the ovarian surface was involved
more frequently, and they were bilateral more often. In the cases
reported by Burks and colleagues,30 67% were stage II or III, whereas the typical serous LMP tumors were stage
II or III in 32% of the cases. The micropapillary tumors were bilateral
twice as frequently as were the typical serous LMP tumors. They
also exhibited surface components more often than the typical LMP tumors. As
mentioned earlier, micropapillary tumors had invasive implants
more frequently than the typical serous LMP tumors. Patients with invasive
implants died twice as frequently as those without invasive implants. Siedman
and Kurman31 state that “a micropapillary pattern even in the absence of destructive
infiltrative growth is a manifestation of an invasive tumor.” Therefore, it
has been suggested that this group of tumors be designated
as micropapillary carcinomas.24,30,32 In the cases reported by Eichhorn and coworkers,29 none of the noninvasive implants were micropapillary. These authors also
state that some implants of overt ovarian serous carcinoma are not
invasive, but cells with obviously malignant features present in a noninvasive
implant are sufficient to make the diagnosis of carcinoma. These
controversial findings should be resolved by studies of larger series
of the subtypes in question. It appears that patients with micropapillary
or cribriform types of LMP serous tumors had 5- and 10-year survival
rates similar to those of typical LMP serous tumors and different
from those observed with serous carcinomas. The survival rates vary
considerably among studies. In the annual report of FIGO,33 the 5-year patient survival rate for LMP ovarian serous tumors for stages
I to III was between 90% and 95%. Similar figures have been reported
in subsequent series. |


 520).
520).