Background
Carcinoma of the cervix is the third most common malignancy of the female reproductive tract and the second most common malignancy in women 15 to 34 years of age, with a peak incidence at 45 to 55 years. Risk factors include low socioeconomic class, black race, early sexual activity, multiple sexual partners, multiparity, and infection with herpes simplex virus type 2. Because of extensive Papanicolaou smear screening, the incidence of invasive cervical cancer continues to decline. In the United States, there are an estimated 15,700 new cases diagnosed every year.1 The vast majority are squamous cell carcinomas. Other histologies include adenocarcinoma, small cell carcinoma, adenoid cystic carcinoma, sarcoma, and lymphoma.
Magnetic Resonance Imaging and Ultrasound Anatomy
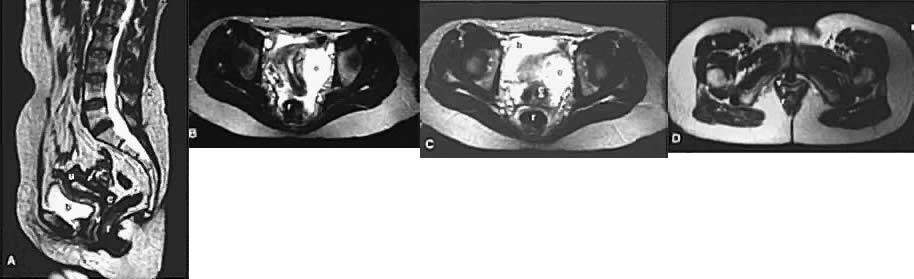
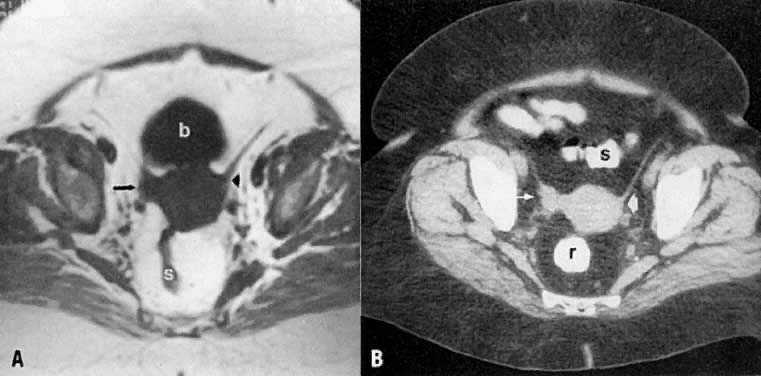
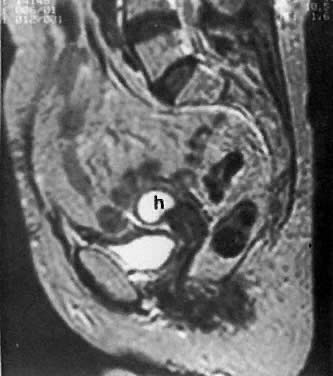
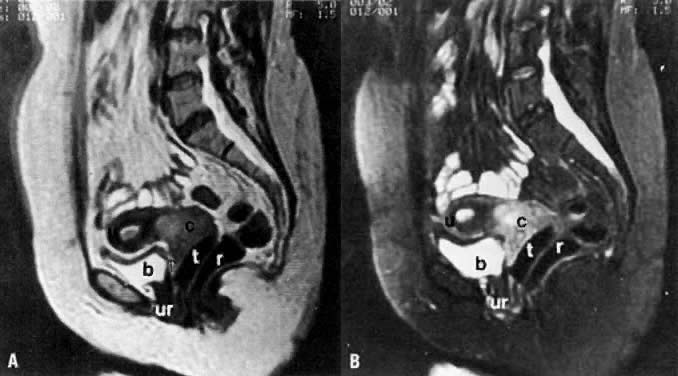
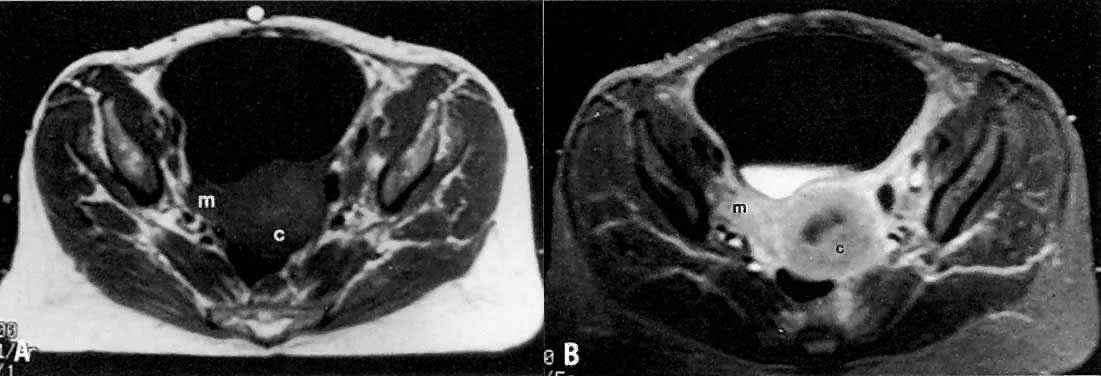
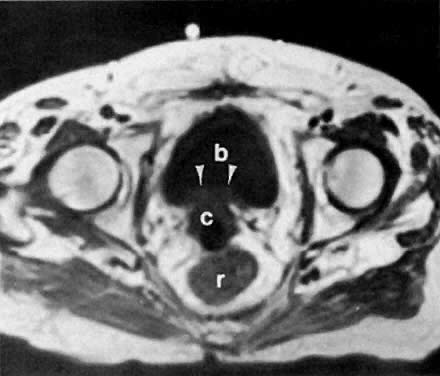
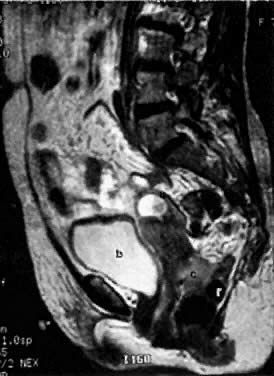
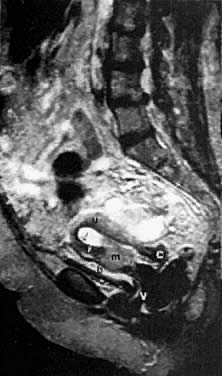
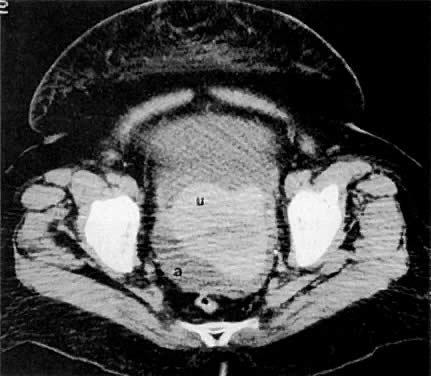
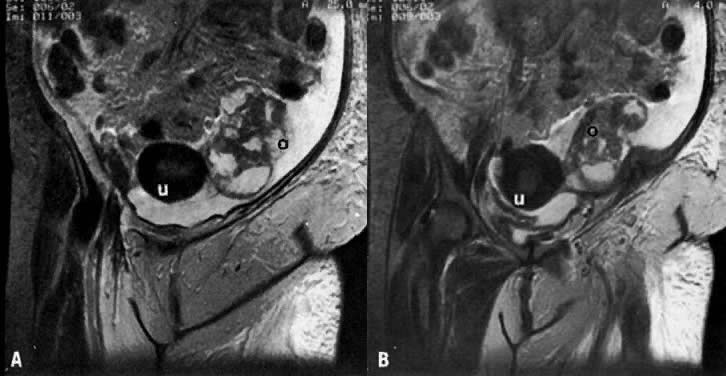
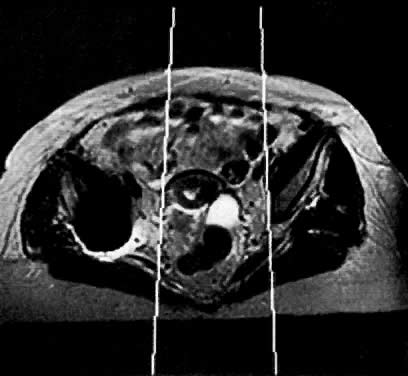
On T2-weighted magnetic resonance imaging (MRI), the normal cervix has two distinct zones (Fig. 1A, Fig. 1B, and Fig. 1C). The central 2- to 3-mm stripe of high signal intensity represents the epithelium and mucous glands in the cervical canal. This stripe is surrounded by a 3- to 8-mm zone of low signal intensity representing the fibromuscular stroma, which is continuous with the junctional zone of the uterus.2,3,4 A third peripheral, 2- to 8-mm zone of intermediate signal intensity is only seen in 15% of patients.2,3 The signal intensity of the cervix does not change with the menstrual cycle, but does change with pregnancy. As reported by Holland and associates,5 the predominant signal of the cervix is dark in the nonpregnant woman and in the first trimester, isointense in the second and third trimesters, bright at 72 hours before labor, and isointense postpartum. The parametria are depicted as two thin stripes of moderate signal intensity lateral to the cervix (Fig. 2A).6
|
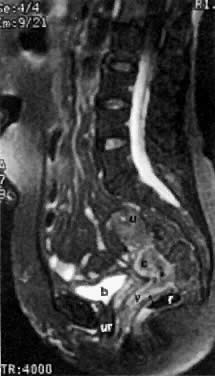
Two distinct layers are seen in the vagina on T2-weighted images: an inner zone of high signal intensity, which represents the vaginal epithelium and mucous glands; and an outer zone of low signal intensity, which represents the vaginal wall. The vaginal wall can be separated easily from the urethra and rectum on T2-weighted images (Fig. 1D).6
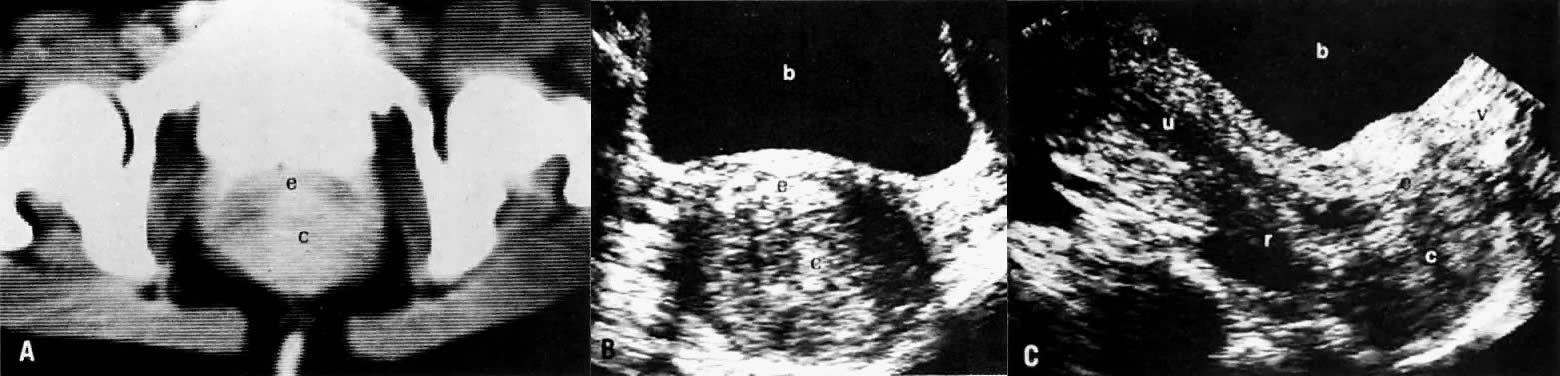
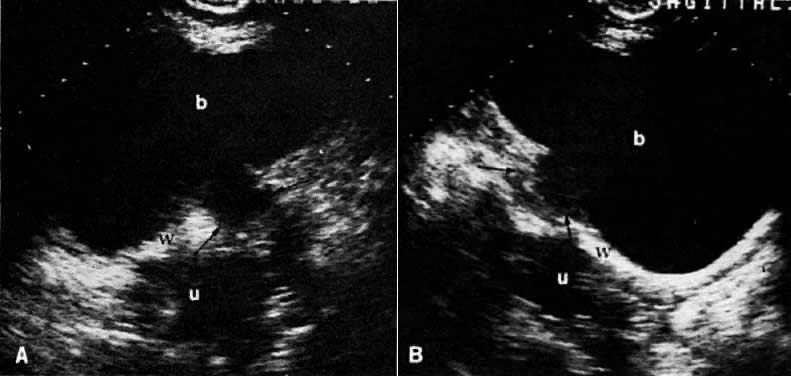
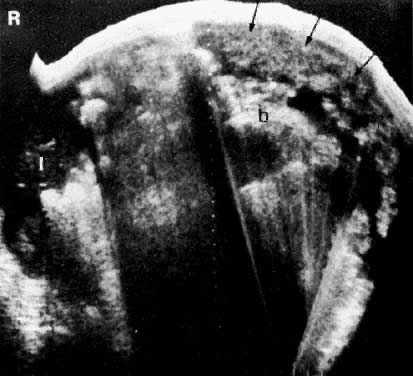
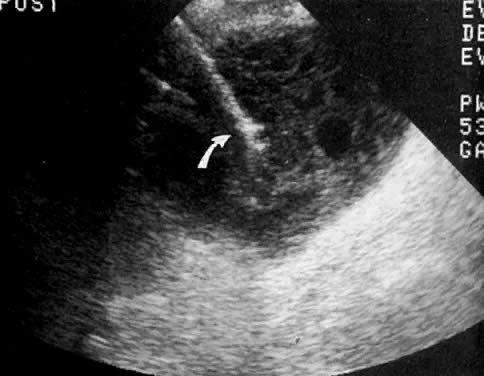
On transvaginal ultrasound (TVUS), the cervix is seen as a medium-echogenicity, 3- to 4-cm long, 3-cm thick cylindrical structure. Two layers can be identified in the cervix: the hyperechoic cervical canal and the medium-echogenicity stroma (Fig. 3A and Fig. 1D). Neither of these layers varies markedly in appearance or thickness during the menstrual cycle. Two distinct layers are seen in the vagina on TVUS: an inner hyperechoic zone, which represents the epithelium and mucous glands; and an outer hypoechoic zone, which represents the muscular wall (Fig. 3B and Fig. 1E).
Diagnosis
Diagnosis of cervical carcinoma is usually made by history, physical examination, Pap smear, and in certain cases, dilatation and curettage. Radiology plays a complementary role in the more difficult cases. On MRI7 and other cross-sectional imaging techniques, the cervix is usually normal in appearance in early cervical cancer. Locally confined cancer appears as a focal mass of medium signal intensity, which either distends the endocervical canal or focally disrupts the normal low-signal-intensity cervical stroma on T2-weighted MRI (Fig. 4A).
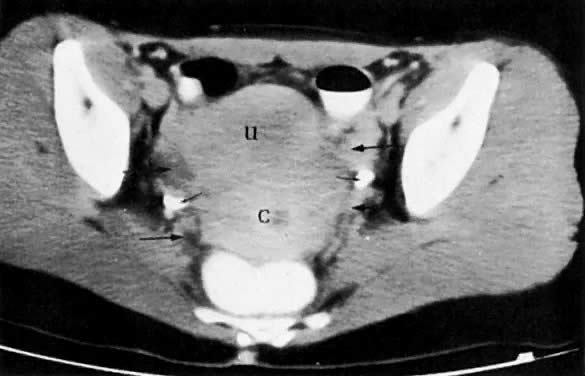
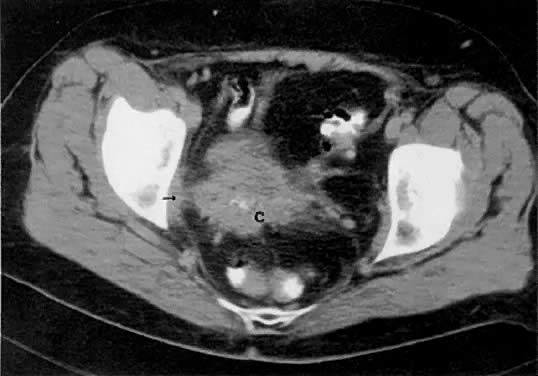
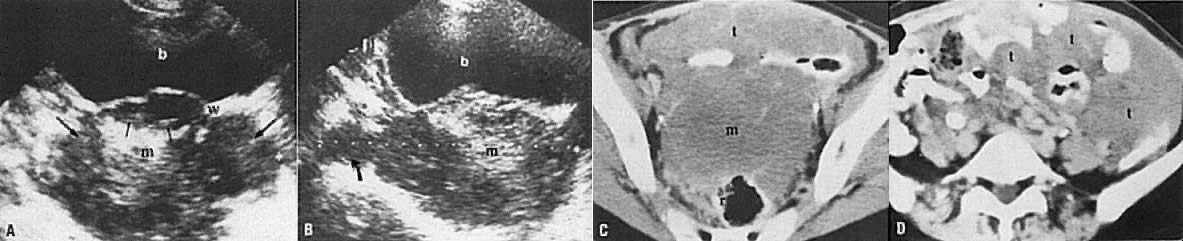
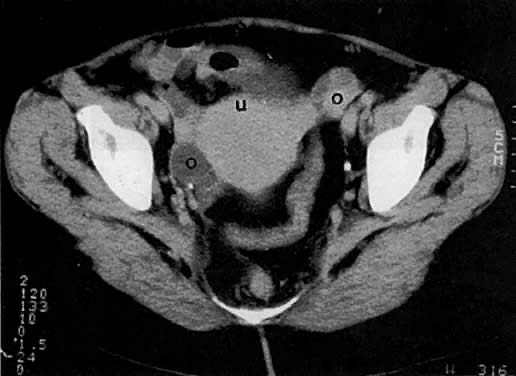
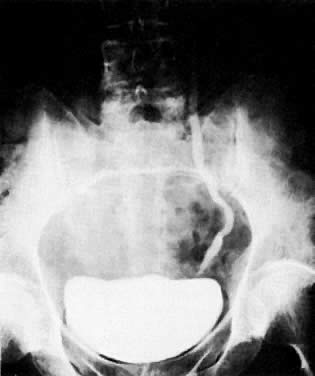
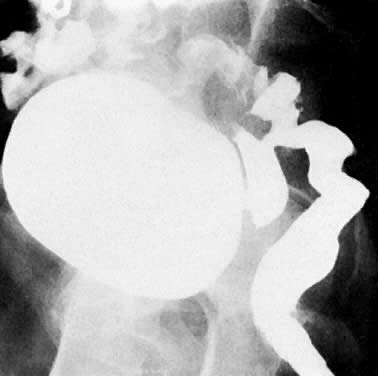
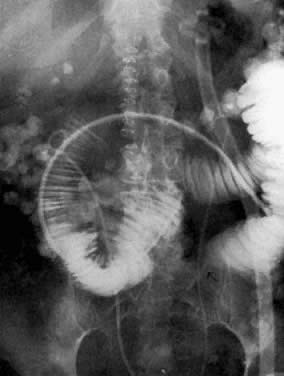
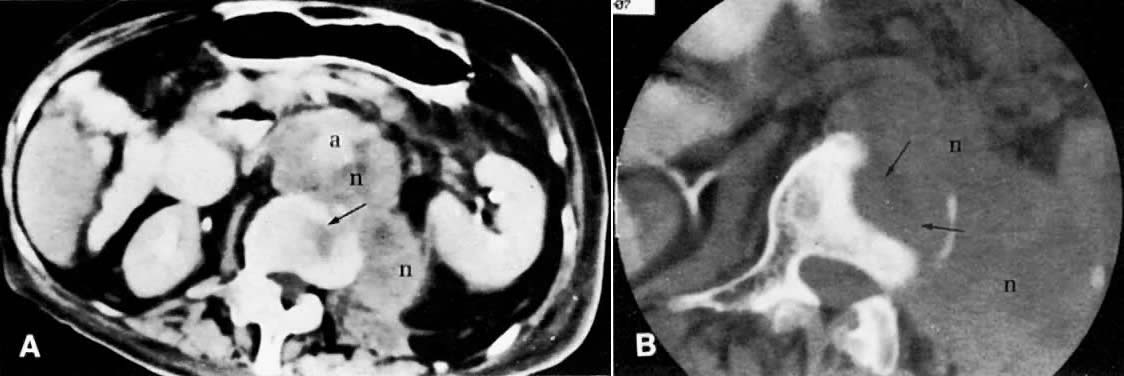
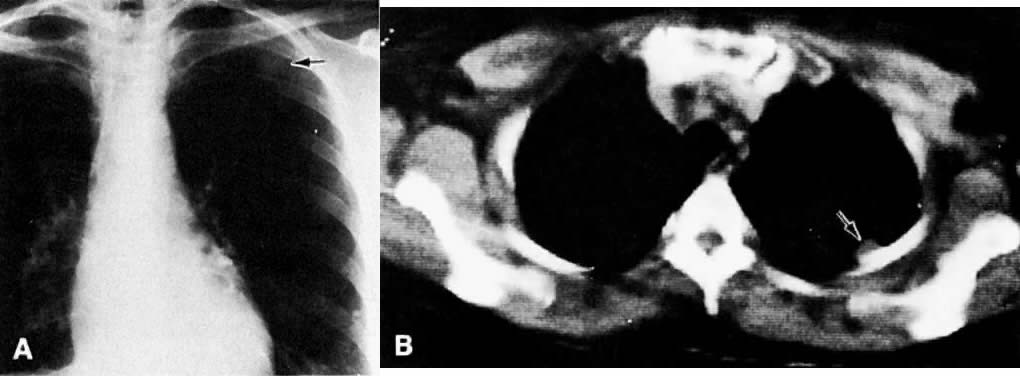
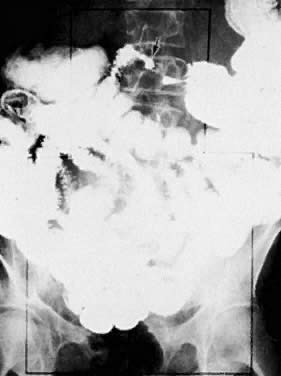
Cervical enlargement (see Fig. 2B; Fig. 5A) is a typical finding of cervical carcinoma on computed tomography (CT). After intravenous contrast material administration, necrotic soft-tissue masses of the cervix can be identified as low attenuation areas.8 Despite technical advances in CT, its accuracy for detecting cervical tumors, which has been reported to be 63% to 80%, remains less than that of the clinical, ultrasound, or MRI examination.9,10
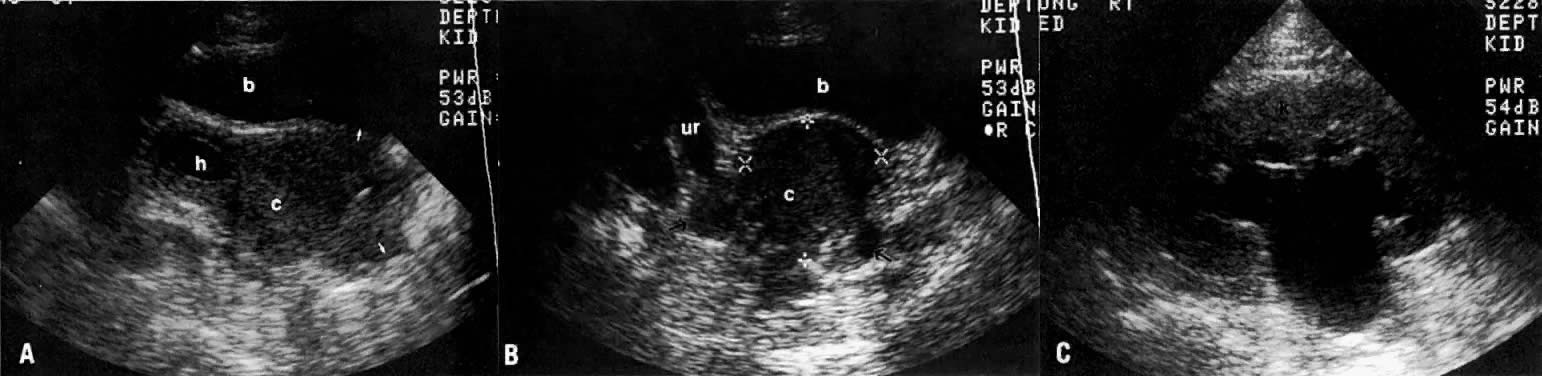
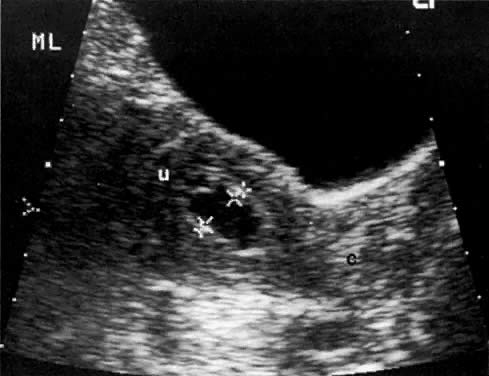
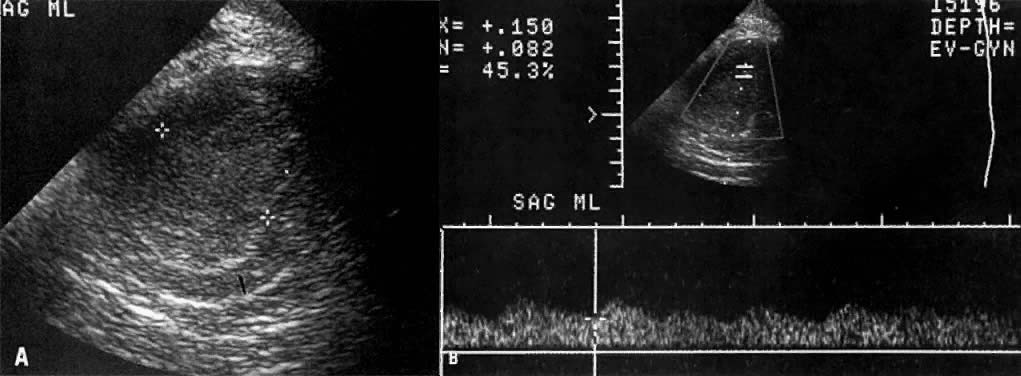
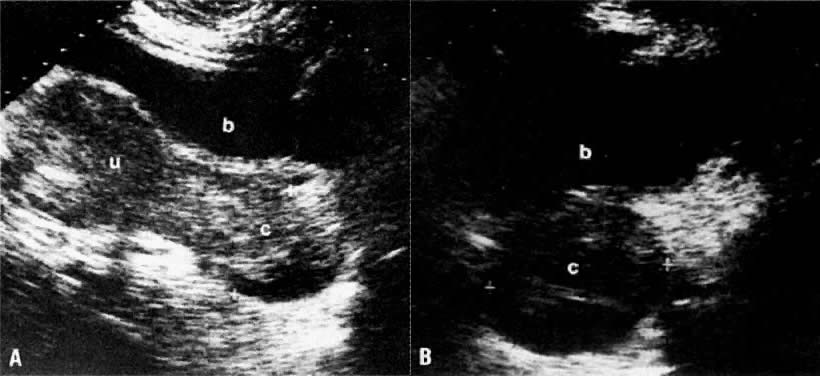
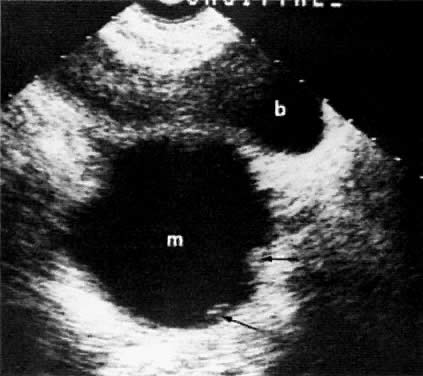
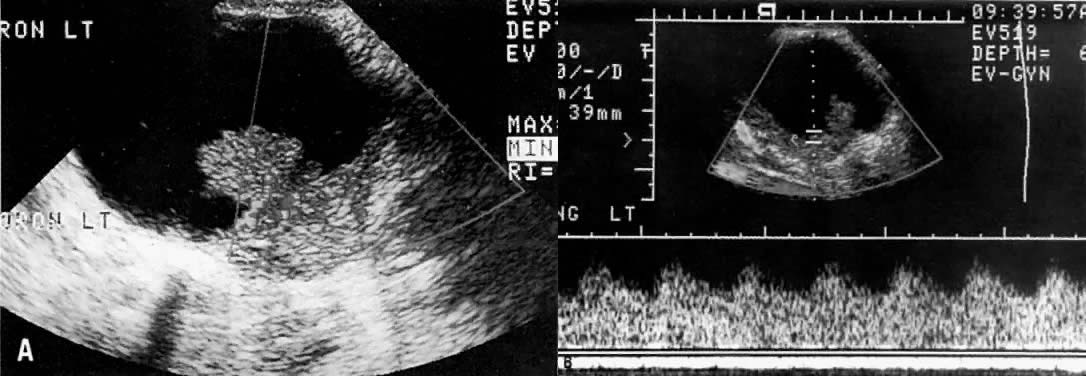
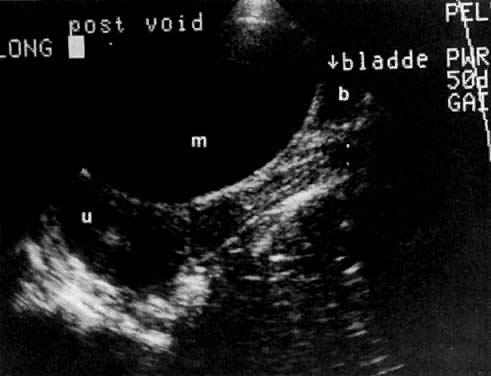
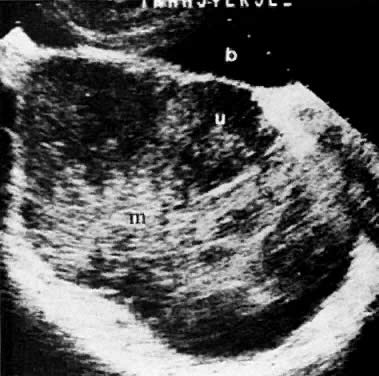
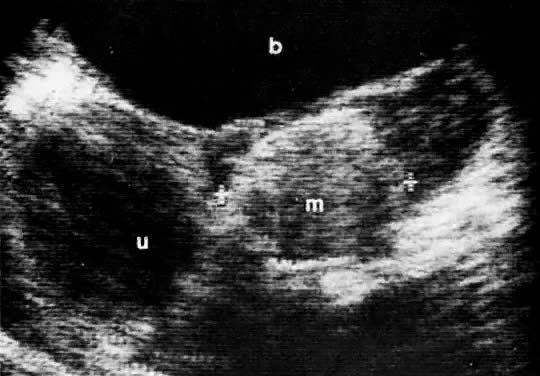
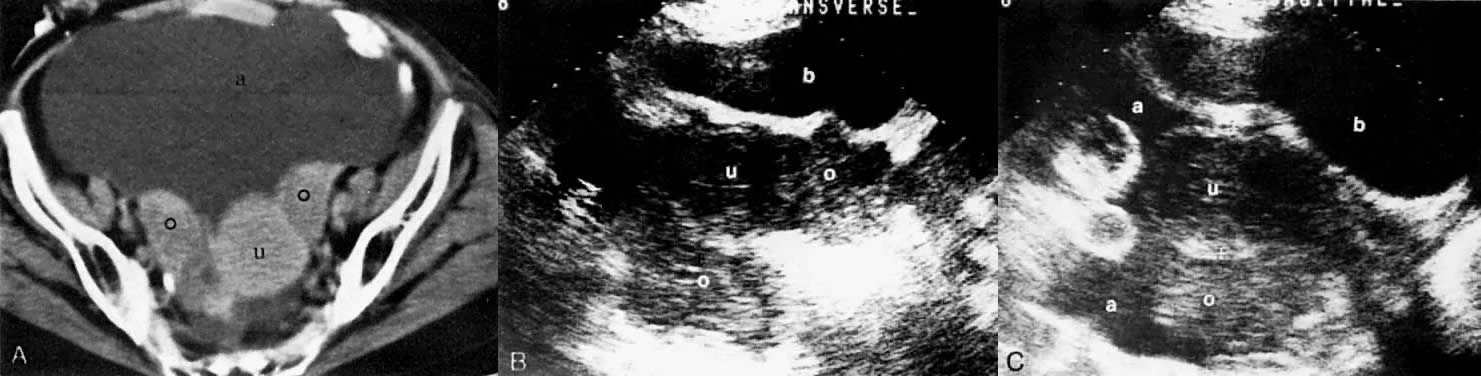
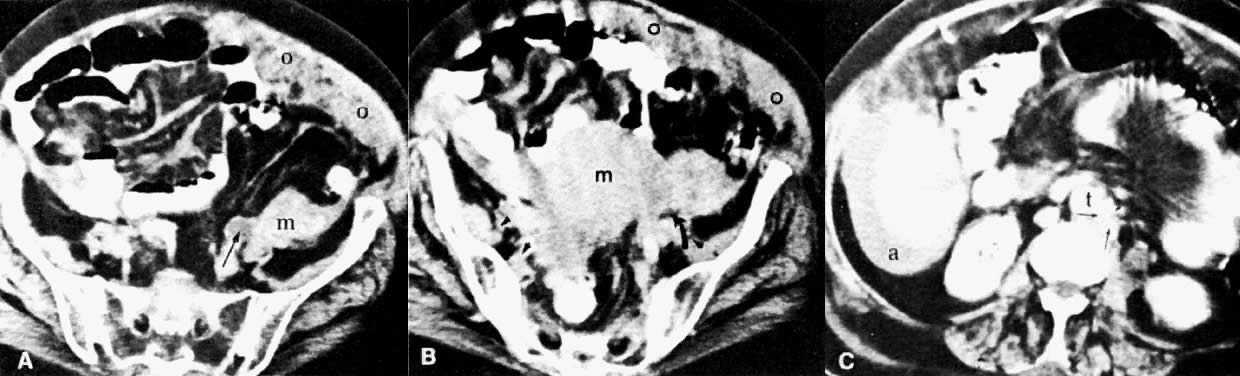
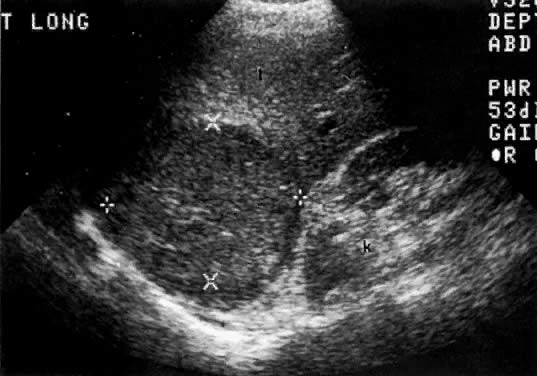
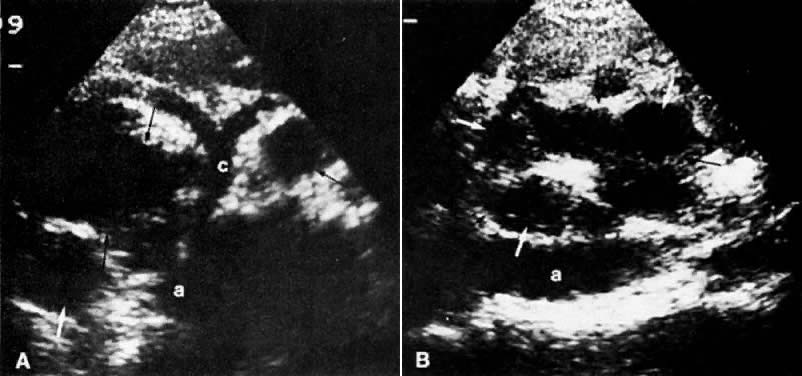
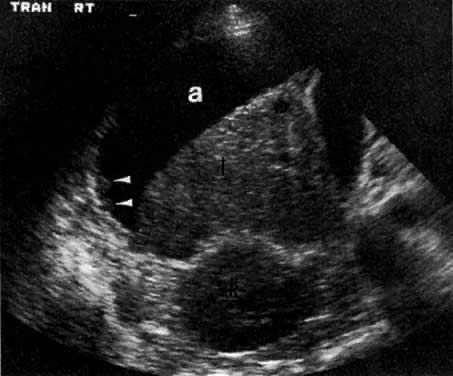
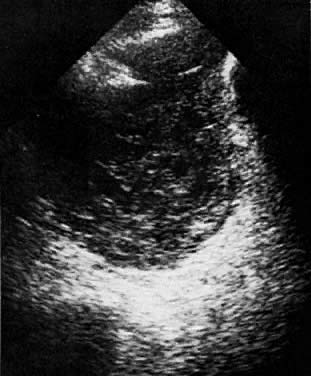
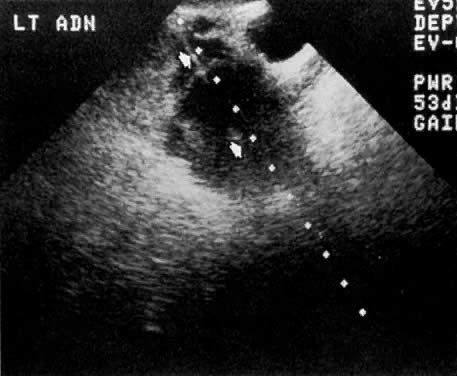
In locally confined disease, a cervical mass may be seen by ultrasound (Fig. 5B and C). In locally advanced disease, the cervix may become diffusely enlarged, inhomogeneous, and irregularly marginated (Fig. 6A and B).
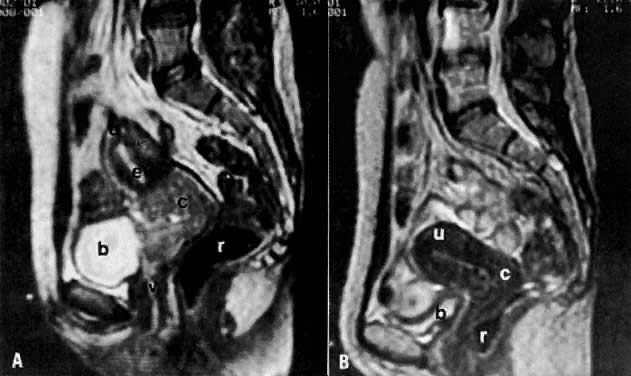
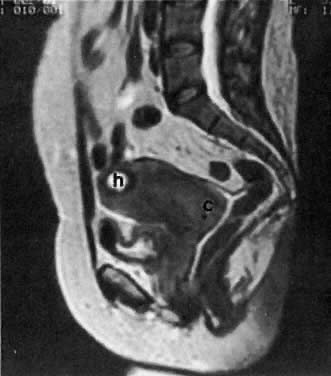
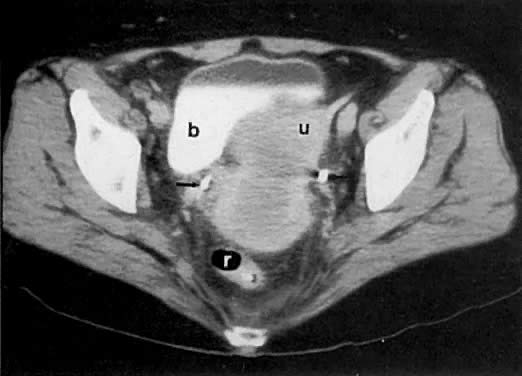
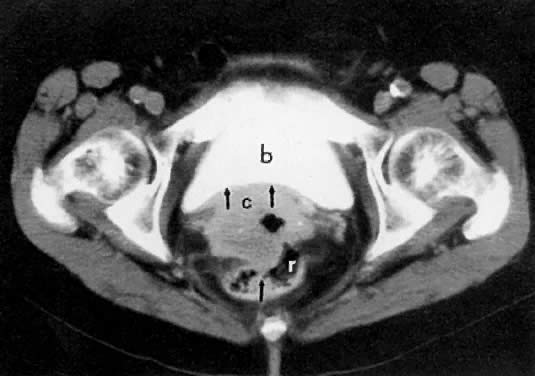
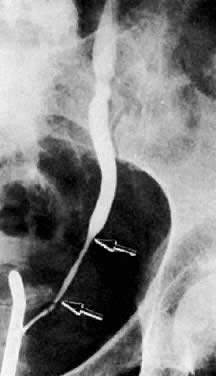
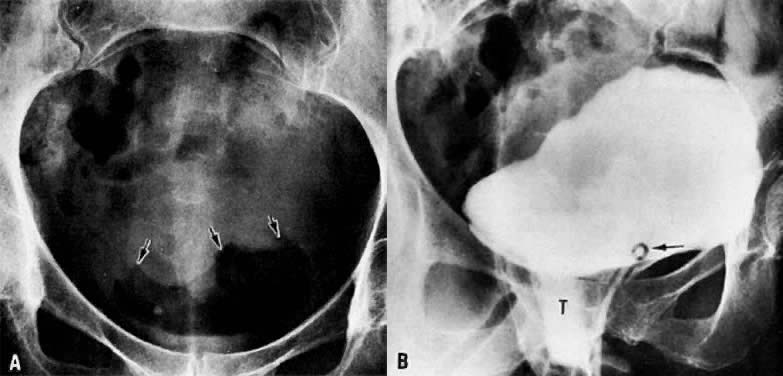
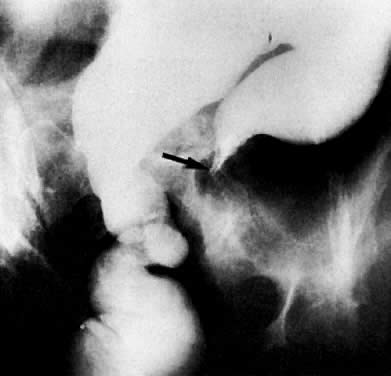
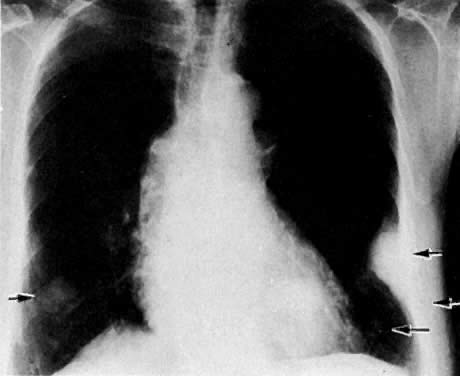
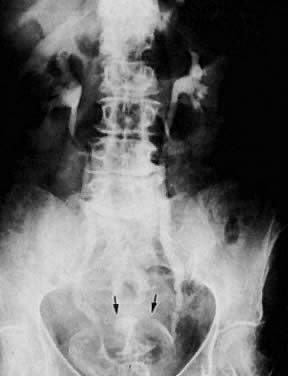
The presence of hydrometras should raise the suspicion of cervical or endometrial carcinoma. It is best demonstrated by ultrasound (see Fig. 6A), but can be seen by both MRI (Fig. 7) and CT (Fig. 8).11,12 This finding, however, is nonspecific because it can also be seen in benign cervical strictures caused by either previous cervical inflammation (Fig. 9) or pelvic irradiation (Fig. 10).
|
|
|
|
Methods of Spread
Cervical cancer spreads in a stepwise fashion by either local infiltration into parametria, pelvic sidewall, and adjacent organs (vagina, ureters, bladder, and rectum); or by lymphatic spread to the pelvic (obturator and external, internal, and common iliac) and para-aortic lymph nodes.13 In advanced cervical cancer and small cell cervical cancer, hematogenous metastases to the lungs, liver, brain, and bones can occur.14
Clinical Staging
Standard clinical workup for cervical cancer staging includes a history and physical examination with pelvic and rectal examinations. Ideally, the pelvic examination is performed under anesthesia along with proctoscopy and cystoscopy in advanced cases, with or without dilatation and curettage. Laboratory studies include a complete blood count and blood chemistries. The staging system (Table 1) was last revised in 1984 by FIGO.15
TABLE 1. Staging System for Carcinoma of the Uterine Cervix (FIGO 1995)
Stage | Characteristics |
IA | Invasive cancer identified only microscopically. (Depth of invasion less than 5 mm, width less than 7 mm) |
IB | Clinical lesion confined to the cervix or preclinical lesion greater than stage IA. |
IIA | Extension to the vagina (not the lower third), with no parametrial extension |
IIB | Parametrial invasion (but not to pelvic sidewall) |
IIIA | Tumor involves lower third of the vagina, with no extension to pelvic sidewall |
IIIB | Tumor extends to the pelvic sidewall or causes hydronephrosis or nonfunctioning kidney |
IVA | Tumor invades mucosa of bladder or rectum and/or extends beyond the true pelvis |
IVB | Distant metastases |
The tumor prognosis, as well as the choice and success of therapy, depends on accurate clinical staging and volume assessment of the tumor.16,17 For example, the differentiation between stage I and stage IIB disease is of special significance because patients with parametrial spread are treated with primary radiation therapy, whereas those with lesser stages may be treated with either a radical hysterectomy or radiation therapy. Also, the presence of nodal involvement would change patient management by adding radiation therapy to the para-aortic area and/or chemotherapy. Clinical examination may not always provide optimal evaluation of the tumor extent and volume.18,19,20,21 It differs from surgical staging in approximately 30% to 40% of cases,18,19 and its accuracy decreases as the disease advances in stage.
Local Radiologic Staging
Radiologic studies that have been used to evaluate the locoregional extent of cervical cancer include excretory urography (EU), lymphangiography, barium enema, CT, MRI, and ultrasound. Abdominopelvic CT has replaced EU and lymphangiography, especially in locally advanced disease, because it provides accurate information about ureteral encasement, parametrial and pelvic sidewall extension, and pelvic and para-aortic nodal involvement. Barium enema should be considered for the evaluation of patients with stage III and stage IV disease or when symptoms suggest bowel involvement. MRI and ultrasound are complementary studies that may provide useful information about local extension to the parametria, bladder, ureters, and rectosigmoid.
LOCAL CT STAGING.
Poor soft-tissue contrast and restriction to the axial plane limit the ability of CT to determine the depth of stromal invasion or to assess tumor volume. Tumor confined to the cervix may be seen as an inhomogeneous, hypodense area in an enlarged cervix; however, the size of the cervix cannot be used in staging cervical carcinoma. The criteria used to stage confined disease include smooth, well-defined cervical margins; no prominent parametrial soft-tissue strands; no parametrial soft-tissue mass; and preservation of the periureteral fat planes (see Fig. 5A).22 CT has a limited role, however, in the detection of tumor extension into the vagina and body of the uterus.
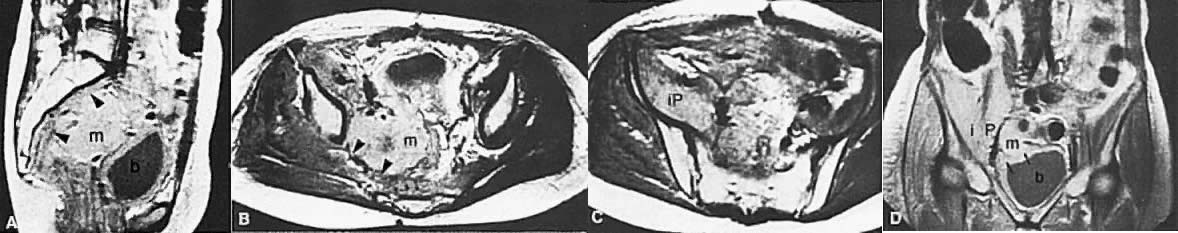
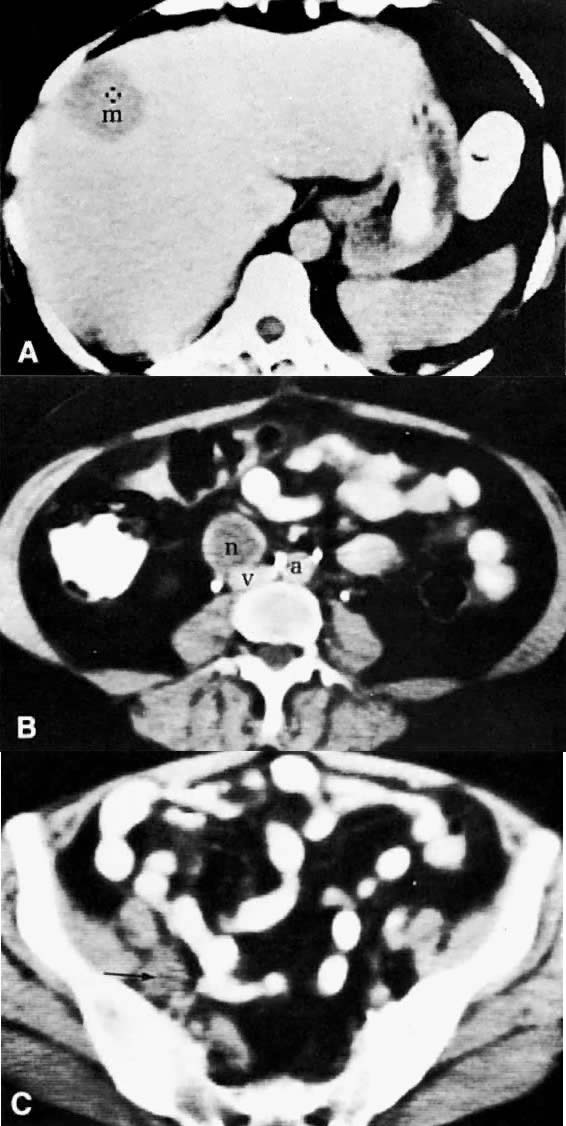
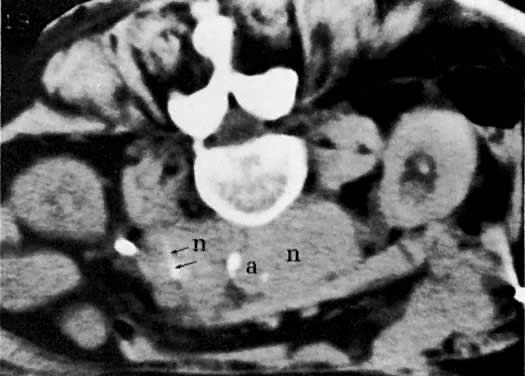
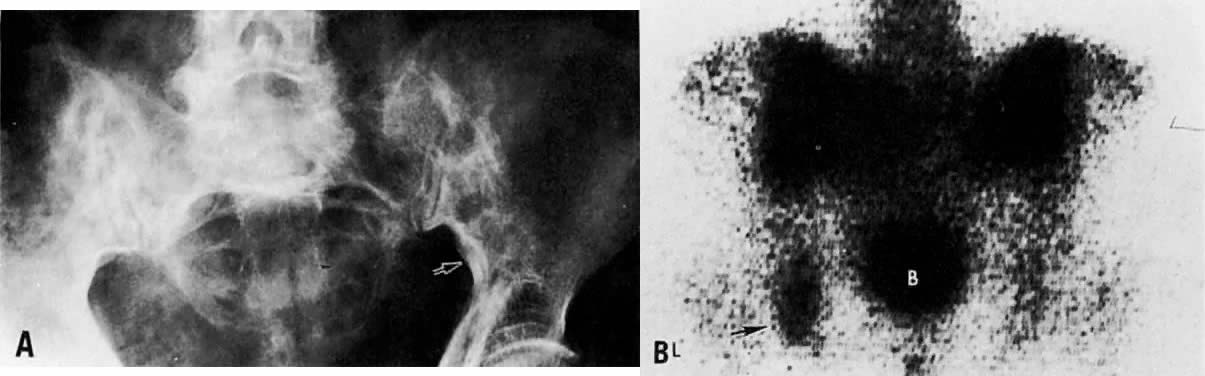
CT can be helpful in the diagnosis of parametrial invasion and can be used in conjunction with the physical examination in differentiating stage I cervical carcinoma from stage IIB or more advanced disease. The criteria used to diagnose parametrial invasion include irregularity or poor definition of the lateral cervical margins, prominent parametrial soft-tissue strands, eccentric soft-tissue mass (see Fig. 2B), and obliteration of the periureteral soft-tissue planes without hydroureter (Fig. 11). Prominent strands can be seen with inflammation, but are more commonly seen with tumor invasion. One should be careful in diagnosing parametrial invasion in patients with asymmetry of the cervix because tampon insertion occasionally causes angulation of the uterus. The reported accuracy of parametrial involvement by CT is 70%, positive predictive value 33%, and negative predictive value 67%.9
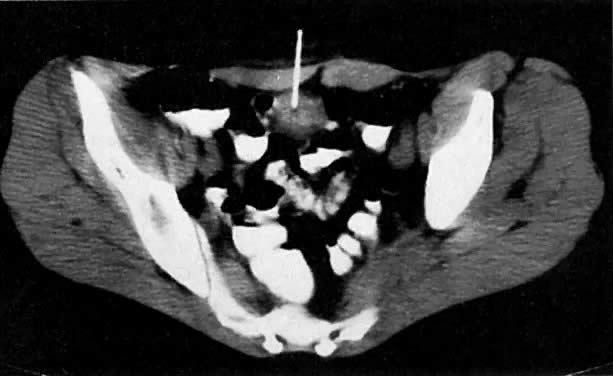
Signs of pelvic sidewall invasion (stage IIIB) on CT include irregular soft-tissue strands extending to pelvic muscles (Fig. 12), a fat plane of less than 3 mm between these muscles and the tumor, muscle encasement by the tumor, and ureteral obstruction.23 CT is much more accurate than clinical examination in detecting pelvic sidewall invasion. Its reported accuracy ranges from 66% to 80%. Although CT can detect invasion of the bladder and colon by tumor, it may be difficult to differentiate invasion from mere contiguity to these structures based on the loss of fat planes.24,25 The presence of bladder or rectal wall thickening and intraluminal mass in association with focal fatty plane obliteration may help in differentiation (Fig. 13).23
|
|
LOCAL MRI STAGING.
With its improved soft-tissue contrast resolution and multiplanar capability, MRI is very useful in local staging of cervical carcinoma. Low signal intensity surrounding the tumor in the cervix and preservation of fat planes are consistent with tumor confined to the cervix (Fig. 14A). Stage IIA is indicated by the loss of the normal low signal intensity of the vaginal wall (Fig. 15). Stage IIB is indicated by the presence of abnormal thickening of the parametrium on T1-weighted MRI (see Fig. 2A; Fig. 16A). On T2-weighted images, stage IIB is depicted as increased signal intensity in the parametrium with the loss of the normal low signal intensity of the cervical stroma.9 Stage IIIA is indicated by the loss of low signal intensity of the wall of the lower third of the vagina. In stage IIIB, the lateral pelvic wall muscles lose their normal low signal intensity, with loss of the fat planes separating these muscles from the tumor on T1-weighted images (Fig. 17A and B). Stage IVA is indicated by loss of the normal low signal intensity of the bladder wall (see Fig. 17D; Fig. 18) or rectal wall (Fig. 19), associated with wall thickening and/or intraluminal mass.26,27
|
Several studies have shown that MRI improves the accuracy of parametrial assessment over that of the clinical examination.28,29,30,31 In one study with surgical correlation, the accuracy of detecting parametrial involvement by MRI was 92% compared with 78% by clinical examination, and the positive predictive value was 89%, and negative predictive value was 92%.9 Most errors occur because of overstaging. In a study of 57 patients by Hricak and colleagues,26 MRI overstaged the tumor in 11 patients and understaged it in 3 patients, yielding an overall accuracy of 81%. In this study, the accuracy of MRI was 88% for detecting parametrial extension, 95% for vaginal extension, 93% for pelvic sidewall extension, and 96% for bladder wall invasion. In another study of 67 patients, Togashi and co-workers27 overstaged the tumor in 6 patients and understaged it in 10 patients. Accuracy was 89% for parametrial tumor extension and 83% for vaginal involvement. In patients with large tumors or tumors located in an area difficult to evaluate, MRI can accurately assess tumor volume and extent.2,6,28,29,30,31,32 As tumor diameter approaches 3 cm, approximately 50% of these patients will have lymph node metastasis.
Peritumoral inflammatory reaction can mimic parametrial involvement, causing tumor overstaging on T2-weighted imaging.33 In addition to edema, factors including volume averaging, bowel peristalsis artifact, close proximity of structures, obliquity of planes, and anatomic variations may obliterate fat planes between the tumor and the surrounding organs, thus increasing the false-positive rate, especially in advanced disease.34 Gadolinium-enhanced T1-weighted MRI techniques do not seem to improve visualization of stromal invasion of cervical carcinoma, but do improve staging in advanced cases (see Fig. 16A and Fig. 16B).35 Recent studies have shown that gadolinium-enhanced dynamic MRI may be superior to T2-weighted MRI in the evaluation of stromal invasion.12 Fat suppression techniques may also better delineate the tumor margins (Fig. 14B) and detect bladder or rectal wall invasion.36 MRI using endorectal surface coils may also prove to be a promising technique because of improved resolution over conventional MRI techniques.37
LOCAL ULTRASOUND STAGING.
Ultrasound may play a role in the evaluation of tumor extension to the parametrium and the pelvic sidewall in patients with an equivocal pelvic examination.38,39 In stage IB, the tumor is confined to the cervix on ultrasound, without parametrial extension (see Fig. 5B and C). In stage IIB, a tongue of the hypoechoic soft-tissue mass extends laterally from the cervix (see Fig. 6B). More recently, high-resolution transrectal ultrasound was found to be more sensitive in assessing parametrial tumor spread compared to clinical evaluation (78% vs 52%, respectively).40 Ultrasound is also capable of detecting tumor extension into the bladder (Fig. 20),39,41 hydronephrosis (Fig. 6C),38 and/or ureteral obstruction (see Fig. 6B). Unlike CT, however, it cannot always reliably evaluate the level and cause of the obstruction.