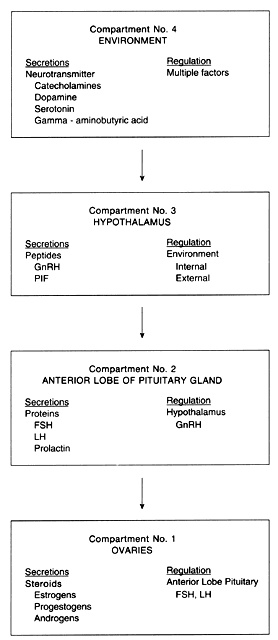
The process of ovulation integrates four compartments that, when functioning normally, eventuate in the cyclic development of ovarian follicles and the release of at least one mature ovum during each cycle. Each of the four compartments is unique with regard to its anatomic characteristics as well as in the nature of its secreted hormone (or hormones). The compartments are as follows: (1) the ovaries, (2) the anterior lobe of the pituitary gland (hypophysis), (3) the hypothalamus, and (4) the environment (Fig. 1).
|
Compartment 1: The Ovaries
The ovary produces steroid hormones (estrogens, progestogens, and androgens). Estrogens are primarily produced by granulosa cells, progestogens by theca and granulosa cells, and androgens by theca cells and stromal cells. Although the premenarcheal ovary possesses the capacity for steroid production, controlling mechanisms at the level of the anterior lobe of the pituitary gland and hypothalamus (discussed above) do not provide the appropriate level of stimulation to effect significant ovarian steroidogenesis until just before menarche. The anterior lobe of the pituitary gland produces protein hormones (FSH, LH, prolactin) that regulate ovarian function. FSH and LH influence ovarian folliculogenesis and steroid hormone secretion via direct action on the ovary. Prolactin influences ovarian function indirectly through modulation of gonadotropin release; there is also compelling evidence that prolactin may have a direct, but incompletely understood, action on ovarian function.19
Compartment 2: The Hypophysis
Histochemical techniques and immunocytochemical methods have demonstrated that LH and FSH are largely individually produced by distinct gonadotropes, but to a lesser extent are secreted by the same cells.20 The observation that both gonadotropins can arise from common cells may explain the midcycle rise of FSH release that accompanies the preovulatory LH surge. Prolactin-secreting cells are also identifiable histologically. These distinct cells, termed lactotropes, are more laterally situated in the hypophysis than the gonadotropes.21 The first few days of the ovulatory cycle are characterized by a predominance of FSH secretion. The midcycle surge of gonadotropins brought about by GnRH is dominated by LH; the concomitant rise in FSH is of lesser magnitude. After ovulation, serum levels of LH and FSH abruptly decline to their preovulatory range.
Compartment 3: The Hypothalamus
LH and FSH secretion as well as that of other hypophysiotropic hormones, specifically the peptide hormones elaborated by the hypothalamus, is regulated mainly by the central nervous system. Ovarian function is regulated by GnRH (a decapeptide), prolactin-inhibiting factor (either a dopamine-like substance or dopamine itself), and thyrotropin-releasing hormone (a tripeptide), which stimulates release of thyroid-stimulating hormone but also serves as a prolactin-stimulating agent. These hypothalamic peptides stimulate the synthesis and secretion of hormones by the anterior lobe of the pituitary gland. GnRH originally was thought to be exclusively related to LH secretion and was thus termed “luteinizing hormone-releasing factor.” It is now apparent that both FSH and LH secretion are dependent on GnRH. The change in nomenclature from LH-releasing factor to the term gonadotropin-releasing hormone reflects this peptide's dual control over both gonadotropins, and also emphasizes that this factor fulfills the criteria necessary to be identified as a hormone. No exclusive FSH-releasing substance has yet been identified.
The following are two important characteristics of hypothalamic control over gonadotropic secretions: (1) varying responsiveness of the pituitary gonadotrope to GnRH throughout the cycle; and (2) pulsatile release of gonadotropins in response to episodic elaboration of GnRH into the hypothalamic-hypophyseal portal vasculature. The pulses of GnRH vary in their frequency and amplitude. The sensitivity of gonadotropes to GnRH depends on circulating levels of estrogens and progestogens, which vary with time in the cycle.
Compartment 4: The Environment
The environmental compartment is not anatomically defined, unlike the other three compartments involved in ovarian functions. It consists of a variety of heterogeneous factors that ultimately influence hypothalamic function. These factors include, among others, diet, climate, medications, light-dark cyclicity, level of stress, and the functional status of other endocrine structures. This spectrum of environmental features—internal and external—is translated into input recognizable by the hypothalamus via biochemical conversion of these factors to neurochemical transmitters. Among these neurochemical transmitters are norepinephrine, dopamine, epinephrine, serotonin, and gamma-aminobutyric acid. Catecholamine and serotonin neuron innervation of the median eminence is particularly important in regulating secretions of the hypothalamic nuclei that control pituitary-ovarian function. In addition to monamines and amino acids, peptides appear to play a modulating role. The vasculature of the hypothalamus and pituitary is tridirectional,22, 23 with flow toward (1) the systemic circulation, (2) the anterior lobe of the pituitary, and (3) the brain. This vascular arrangement facilitates both short and ultrashort feedback relationships acting between the hypothalamus and hypophysis.
A variety of peptide hormones are produced by the neurons in an area of the median eminence that terminates on capillary loops. These neurohormones are released into the hypothalamic-hypophyseal portal system. Opioid peptides such as β-endorphins and enkephalins also play a major role in the regulation of hypothalamic-pituitary function. Opioid receptors have been identified in the hypothalamus as well as in other regions of the central nervous system. Administration of opiates and enkephalin analogs results in inhibition of LH secretion and an increase in prolactin release.24 Opioids suppress LH secretion by inhibiting GnRH neurons in the arcuate nucleus; catecholamines also regulate the elaboration of GnRH. Dopamine inhibits LH secretion, whereas norepinephrine may stimulate LH release.25
The pulsatile release of GnRH by the hypothalamus was first described by Knobil.26 These classic studies, carried out in the Rhesus monkey, revealed that the secretion of LH and FSH is periodic, with pulses at approximately 90-minute intervals in response to the episodic release of GnRH. Continuous infusion of GnRH downregulates the pituitary gland and results in failure of gonadotropin secretion. The use of long-acting analogs of GnRH also inhibits gonadotropin production. Thus the intervals between physiologic episodes of either GnRH elaboration or exogenous GnRH, delivered with an infusion pump, allow the gonadotrope to recover its responsiveness to GnRH, thereby replenishing GnRH receptors. The ultimate elaboration of gonadotropins results from a multitude of factors. The major factor is an appropriate program of GnRH pulses with varying amplitude and frequency delivered via the hypothalamic-hypophyseal portal system. Modulating this mechanism are catecholamines, neuropeptides, opioids, and feedback signals originating at all four compartments.