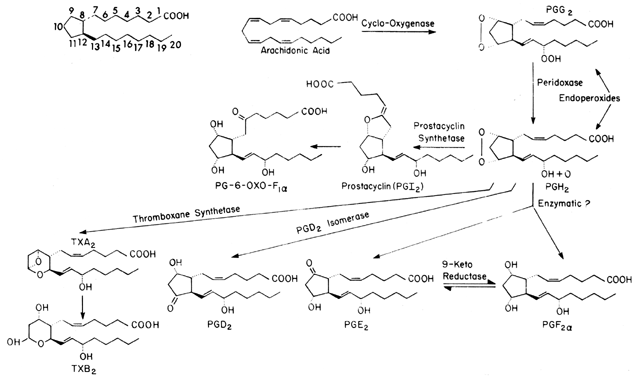
The aforementioned background allows us to discuss the role of prostaglandins
in the reproductive cycle. Although it is clear that prostaglandins
play an important role in a number of reproductive processes, our
understanding of the precise mechanism of action remains very basic. Additionally, the
interrelationships between prostaglandins and other
vasoactive substances such as nitric oxide have added to the complexity
of this area. In many cases, only experimental data from isolated human
tissue and experimental animals are available. However, it is clear
that endogenously occurring compounds are important in clinical medicine. The Menstrual Cycle Peripheral plasma levels of PGF2α and 13, 14dihydro-15-keto-PGF2α have been measured throughout the menstrual cycle.24,25,26,27 However, none of these studies demonstrated a cyclic change. Koullapis
and Collins reported that although extremely variable, levels of 13, 14-dihydro-15-keto-PGF2α consistently showed two peaks: one preovulatory, which was associated
with increasing estrogen, and one premenstrual, which was associated with
rapid decreases in estrogen and progesterone levels.28 The failure to measure cyclic changes in the peripheral plasma in PGF2α most likely reflects the fact that PGF2α is converted quickly to 13, 14-dihydro-15-keto-PGF2α in the lungs and other organs. The failure to routinely measure cyclic
changes in the PGF2α metabolite may reflect a simple dilutional problem. By the time uterine
PGF or its metabolites reach the peripheral circulation, they have been
diluted into a large blood volume. Cyclic changes in PGE and PGF levels do occur in the uterus, as determined
by endometrial and myometrial biopsies.29,30,31,32 In the endometrium, PGE and PGF increased sequentially throughout the
menstrual cycle. The endometrium of the late secretory phase contained
considerably elevated PGF levels and a reversal of the ratio of PGF to
PGE. These increases in PGF levels in the endometrium appear to be caused
by progesterone withdrawal and have been reported for the postovulatory
period.30,32 In the myometrium, the concentration of PGF varied throughout the menstrual
cycle, characterized by two peaks, one occurring during the late
proliferative phase and the other in the late secretory phase. In contrast, PGE
peaked in the late proliferative stage (higher levels than PGF) but
then decreased in the later stages of the cycle.33 The 13, 14-dihydro-15-keto metabolites of PGE and PGE did not show any
cyclic changes in the myometrium, suggesting no cyclic alteration in
PGF and PGE metabolism. The elevated levels of prostaglandins in the periovulatory
period may provide a mechanism to facilitate migration of
ova and spermatozoa, but this has not been proven. The change in the
PGE-to-PGF ratio in both endometrium and myometrium in the late secretory
phase may be important in the onset of menstruation. Menstruation and Dysmenorrhea The postovulatory period is associated with progesterone withdrawal and
increased PGF-to-PGE ratios in the endometrium.29,30,31 PGF2α has been shown to be a vasoconstrictor in the uterus and endometrial blood
vessels, and the local endometrial vasoconstriction that occurs during
menstruation is believed to be due in part to prostaglandins.34,35,36,37,38 In 1957, Pickles proposed that a substance produced by the menstruating
uterus might be responsible for stimulation of uterine contractility
and that this “menstrual stimulant” might be responsible for
primary dysmenorrhea.39 Pickles observed that menstrual fluid of patients with primary dysmenorrhea
had higher PGF2α concentration than that of nondysmenorrheic patients and that these patients
had an increased PGF-to-PGE ratio.29 This study was later confirmed by Lundstrom and Green and substantiated
by the observation that long-term infusions of PGF2α resulted in symptoms similar to dysmenorrhea.40,41 Csapo and associates measured the intrauterine pressures during menstruation
in healthy and dysmenorrheic patients.42 In contrast to the normal resting pressure of 10 mmHg, the dysmenorrheic
patient had levels of 60 mmHg. This increased tone could result in
vascular compression and, thus, reduced uterine blood flow. This would
further compromise a uterine circulation already reduced by the direct
action of PGF2α on blood vessels in the uterus and endometrium. It is this reduced blood
flow and resultant anoxia of uterine tissue that is believed to produce
the pain of dysmenorrhea.29 In the study by Csapo and coworkers,42 treatment of dysmenorrheic patients with the prostaglandin-synthesis inhibitor, naproxen
sodium, reduced the resting pressure as well as the
amplitude and frequency of cyclic intrauterine pressure. Naproxen and
ibuprofen both are associated with a reduction of prostaglandins in menstrual
blood and decreased clinical symptoms of dysmenorrhea.43,44 Additional information is available in a recent review on nonsteroidal
anti-inflammatory drugs and reproduction.45 Treatment of dysmenorrhea with prostaglandin-synthesis inhibitors is an
excellent example of integration of basic research and clinical medicine. This
therapeutic method of treatment appears to be effective in 70% to 80% of
all clinical cases of dysmenorrhea. Ovulation Ovulation has three major phases in which prostaglandins may be involved: (1) follicular
maturation; (2) rupture of the follicle, leading to
oocyte release; and (3) formation of the corpus luteum. The actions of
prostaglandins in hypothalamic and pituitary function, which can influence
follicular development, are not discussed in this chapter. In rabbits, LeMaire
and associates showed that isolated follicles could be
stimulated with human chorionic gonadotropin (HCG) to increase their PGE
and PGF2α levels.46 This increase only occurred in those follicles destined to be ovulated.47 Pretreatment of animals with prostaglandin synthesis inhibitors blocks
ovulation,48,49 although attempts to do this in humans have not been successful. It is
known that rat, bovine, and human follicles predominantly synthesize
PGI2 and lesser amounts of PGE and PGF2α and that follicular fluid concentrations increase as ovulation approaches. Addition
of luteinizing hormone (LH) increases granulosa cell conversion
of arachidonic acid to PGI2 and perfusion of rat ovaries with PGI2 induces rupture of mature follicles, although less efficiently than HCG.50 The majority of ova ovulated by PGI2 were immature and would not undergo germinal vesicle breakdown and maturation, thereby
drawing a distinction between oocyte maturation and follicular
rupture. Prostaglandins may be intermediates in the cyclic adenosine monophosphate (cAMP) signalling
pathway because ovulation induced by forskolin or
dibutyryl cAMP in PMSG-primed rats could be inhibited by indomethacin, an
effect itself reversed by the addition of PGE2.51 There is a progressive increase in dispersion of cells of the mural granulosa
and cumulus oophorus of preovulatory ovine follicles 12 hours
after initiation of the LH surge that can be negated by indomethacin. The
effect of indomethacin itself can be counteracted by PGF2α.52 This suggests that prostaglandins may be involved in reduction of junctional
complexes that adhere granulosa cells to each other and the oocyte
before ovulation. Yoshimura and colleagues50 found that perfusion of ovaries with PGI2 produced vasodilation, extravasation, and filling defects at the apical
region of preovulatory follicles, findings similar to those with HCG
treatment, and suggested that follicular rupture may be secondary to
increased capillary permeability and vasodilation due to PGE2. Although HCG stimulated collagenase in PMSG-primed rats, which may break
down the follicular wall, indomethacin had no effect on collagenase, although
it reduced prostaglandin synthesis.53 More recent biochemical and molecular advances in this field are reviewed
elsewhere.54 Luteinization does not appear to involve prostaglandins because pretreatment
of animals with indomethacin at concentrations that block ovulation
do not alter luteinization.48 Luteolysis Progesterone, which initially is produced in the corpus luteum, is necessary
to prepare the uterus for implantation of a fertilized ova and to
monitor the pregnancy until the placenta can begin progesterone production.55 Interruption of progesterone production by the corpus luteum then leads
to termination of pregnancy. In 1969, it was shown that PGF2α caused luteolysis in rats,56 and in guinea pigs, the uterus was shown to be the source of a luteolysin
identified as PGF2α.57 In sheep and cows, PGF2α of uterine origin appears to be a physiologic luteolysin.58 The PGF2α is transferred to the uterus by a local counter current mechanism59 between uterine vein and ovarian artery or from uterine lymphatic vessels
to the adjacent ovary.60 In nonhuman primates and women, PGF2α also appears to be a luteolysin.61 Only transient decreases in circulating progesterone are seen after systemic
administration of PGF2α62,63 perhaps because of the rapid systemic metabolism of PGF2α. Because hysterectomy in primates or women does not alter ovarian cyclicity, it
was postulated that the luteolysin was generated locally within
the ovary or corpus luteum.64 However, recently, this concept has been questioned.65 The luteolytic action of PGF2α may be directly on luteal cells or may be indirect by directing blood
flow away from the corpus luteum. PGF2α appears to interfere with gonadotropin effects on luteal cells, being
a potent antagonist of LH or HCG action in rat luteal cells and blocking
LH-stimulated cAMP production,66 although not interfering with gonadotropin binding.67 PGF2α inhibits basal and LH-influenced progesterone production by granulosa
cells from preovulatory follicles that spontaneously luteinize in culture.68 Further, PGF2α has an antigonadotropic effect on LH-HCG-stimulated progesterone and cAMP
formation from human luteal tissue in the mid to late luteal phase.69 Pharriss and Wyndarden70 first postulated that the luteolytic action of PGF2α may be due to its vasoconstrictor action, decreasing blood flow to the
corpus-luteum-bearing ovary. Subsequent work has not proven this observation
but does not preclude local effects of PGF2α causing shunting of blood away from the corpus luteum.71 Riley and Carlson72 demonstrated that one of the early events in PGF2α-induced luteolysis is membrane breakdown or decrease in fluidity. This
was associated with a decrease in HCG binding and HCG receptor concentrations. Further, addition of phospholipase A2 gave a similar increased effect on membrane rigidity, suggesting that
a product of phospholipase action (e.g., free fatty acid or lysophosphatidic acid) may be responsible for this
ridigification during luteolysis. Interestingly, addition of prolactin, which
is luteotropic in the rat, was able to attenuate PGF2α-induced increases in membrane rigidity and decreases in plasma progesterone. The rate-limiting step for all steroidogenic tissues is believed to be
the conversion of cholesterol to progesterone. Rajkumer and coworkers73 presented evidence in rats that PGF2 decreases the ability of luteal cells to mobilize endogenous cholesterol
at relatively short time intervals (4 hours) after PGF2α. At longer time periods (24 hours), there appear to be decreased ability
of these luteal cells to convert cholesterol to progesterone, which
may contribute to the decreased progesterone synthesis. Cholesterol is
imported into luteal cells as a lipoprotein complex by a membrane receptor-mediated
process. However, luteal cells from PGF2α-treated cows74 and PGF2α-treated rats73 do not show decreased utilization of lipoproteins. Thus, although we have a reasonable understanding of how prostaglandins
are involved in luteolysis in some species, we still do not have a clear
indication of how they work in humans.75 Fallopian Tube Motility PGE and PGF have opposite effects on the oviduct; PGE2 causes relaxation, whereas PGF2 induces contractions.76,77 The final response of the oviduct to these prostanoids appears to depend
on the steroid levels associated with the reproductive cycle. In the
rhesus monkey, PGF2α has no effect on the oviduct during the follicular phase but produces
contractions just before ovulation. PGE2 also has no effect during the follicular phase but suppresses activity
during and after ovulation.78,79 It is known that the oviduct can synthesize PGE and PGF2α locally, but endogenous prostaglandins do not appear to be involved in
regulation of spontaneous activity.80,81 The effects of PGI2 and 6-keto-PGF1α have been determined in the fallopian tubes of nonpregnant women. PGI2 is similar to PGE2 and relaxes the fallopian tube, whereas 6-keto-PGF1α causes contractions similar to those seen with PGF2α.82 Although the role of prostaglandins in regulation of fallopian tube motility
is not completely understood, it is thought that prostaglandin-increased
contractile activity is important for ovum transport to the
uterus. Implantation Before the arrival of the blastocyst, the endometrium has undergone profound
changes that are regulated by estrogen and progesterone. In most
mammals, these endometrial changes occur after each ovulation. The information
available about the mechanism of implantation relies heavily
on data acquired from laboratory animals. When the blastocyst arrives
at the uterine wall, changes occur in the underlying endometrium. The
endometrial vessels dilate and exhibit increased vascular permeability. The
trophoblast begin to invade the endometrium, and the blastocyst
undergoes apposition, attachment, adhesion and fusion (known together
as implantation). It currently is unclear whether and how the blastocyst controls the implantation
process. It is clear that changes occur only in the areas of
blastocyst attachment. Prostaglandins have been implicated as the mediator
of the vascular permeability changes that occur at the implantation
site. Prostaglandin levels have been shown to increase significantly
at the site of implantation.83,84,85 The rate-limiting enzyme for prostaglandins synthesis in some situations
may be prostaglandin H synthase (cyclooxygenase). Two isoforms of cyclooxygenase
exist, the constitutive COX-1 and the inducible COX-2 isoforms, which
are regulated differently. Recent studies in the mouse have
shown that the COX-1 gene is expressed in the uterine epithelium during
the peri-implantation period but is down-regulated at the initiation
of attachment.86 This corresponds to the generalized edema in the uterus. In contrast, the
COX-2 gene is expressed in the luminal epithelium and subepithelial
stromal cells surrounding the blastocyst at the time of implantation. Thus, the
COX-1 gene is involved with decidualization or localized endometrial
changes in vascular permeability, whereas the COX-2 gene is
involved with angiogenesis for the establishment of the placenta.86 As these changes occur, expression of prostaglandin receptors EP3 and
FP occur simultaneously in the circular muscle of the myometrium on days 3 to 5 of
pregnancy, which probably is important for embryo transport, spacing, or
accommodation in the uterus. In contrast, the expression
of EP3 in a subpopulation of stromal cells on the mesometrium side
and EP4 receptors in the epithelium and stroma suggest that PGE2 prepares the uterus for implantation.87 The source of the prostaglandins that are generated locally at the site
of implantation still is not clear but is believed to be the endometrium
because the blastocyst has not been shown to produce prostaglandins.83 Indomethacin pretreatment prevents the normal increases in prostaglandins
as well as the changes in vascular permeability in mice,88 rats,83,89 hamsters,84 and rabbits.90 Indomethacin also is effective in blocking implantation in mice,91 rats,92 and rabbits.93 Further evidence for a role of prostaglandins is supported by the fact
that intraluminal administration of PGE2 in rats pretreated with indomethacin results in implantation-like alterations
in vascular permeability,91 but this is not true for PGF2α or PGI2.94,95 Although blockade of implantation would provide a potential method of
contraception, it currently is not known whether similar implantation
mechanisms occur in humans. A recent detailed review on prostaglandins
and implantation appears elsewhere.96 Circulating Levels During Pregnancy Prostaglandins are produced in large quantities in a number of reproductive
tissues during pregnancy, and this production may be affected by
the changing hormonal milieu of pregnancy. A more detailed discussion
of the site of synthesis of these compounds can be found elsewhere.97,98,99 Studies in chronically catheterized animals and measurement of prostaglandins
in uterine and placental tissue or umbilical cord blood suggest
that peripheral levels of prostaglandins should be elevated significantly
in pregnancy. Several attempts have been made to measure circulating
levels of prostaglandins during pregnancy in humans.100,101 Because the primary prostaglandins PGE and PGF are converted quickly to 13, 14-dihydro-15-keto-metabolites (see previous section on metabolism
and measurement), it was believed that these metabolites would more
accurately reflect gestation changes. Measurements of 13, 14-dihydro-15-keto-PGE2 have not been reliable because this compound is unstable.18 Measurements of the circulating levels of 13, 14-dihydro-15-keto-PGF2α also have failed to show any gestationally related change except during
parturition, when they are increased significantly.101,102,103 The discovery of the potent vasodilator PGI2 and the development of RIAs to measure its metabolite, 6-keto-PGF1α, renewed interest in measuring circulating levels of prostaglandins in
pregnancy. PGI2 is produced in vascular tissue and appears to be important in local vasodilation. During
pregnancy, there is a profound decrease in peripheral
vascular resistance, and the uterine vasculature is almost totally
dilated. It was expected that PGI2 levels (measured as 6-keto-PGF1α) would be increased significantly, reaching a maximum in the third trimester. However, measurements of circulating levels of 6-keto-PGF1α have not shown any gestationally related alterations,99,104,105 probably because of the low circulating concentrations and interference
problems in direct plasma assays. Measurement of the 2,3 dinor 6-keto-PGF1α metabolites in urine have shown an 8- to 10-fold increase in PGI2 synthesis during pregnancy,106 although whether this increase is generalized throughout the whole vascular
tree or confined to certain vascular beds (e.g., uterus and kidney) remains to be established. It was further speculated that low production rates of PGI2 in disease states associated with reduced uterine blood flow (i.e., hypertension, preeclampsia) might be the cause of reduced uteroplacental
perfusion. Again, measurements of peripheral circulating levels of 6-keto-PGF1α were made in patients with a number of diseases associated with pregnancy, and
no differences have been observed.104 In contrast, measurements have shown the ability of umbilical and placental
blood vessels from preeclamptic, chronic hypertensive, and intrauterine
growth retardation (IUGR) pregnancies to produce PGI2 to be reduced considerably.107,108 Again, urinary 2,3 dinor 6-keto-PGF1α measurement confirmed this reduction in PGI2 synthesis in hypertensive or IUGR pregnancies. Thromboxane synthesis also
increases during pregnancy (as measured by urinary metabolites); however, the
ratio of vasodilator PGI2 to vasoconstrictor TxA2 favors PGI2, giving an antithrombotic tendency in normal pregnancy. Whereas, PGI2 synthesis is reduced in hypertensive pregnancy, urinary TxB2 metabolites appear unchanged.106 The net result, however, is an alteration in the PGI2/TxA2 ratio toward TxA2 and a more vasoconstrictive and prothrombotic state. Recently, most of
the interest in the regulation of uterine and systemic vascular resistance
in normal and hypertensive pregnancies has moved away from prostaglandins (with
the exception of prostacyclin) and focused on nitric oxide. Preeclampsia
is believed to be caused by endothelial cell dysfunction, and
this may include reductions in both endothelial cell production
of prostacyclin and nitric oxide synthesis (see discussion elswhere
in these volumes). Uterine Blood Flow Prostaglandins can affect uterine blood flow in three different ways. First, prostaglandins
can have a direct effect on the vascular smooth muscle, acting
through receptors to produce either uterine vasodilation
or vasoconstriction. Second, prostaglandins can increase uterine tone
or contractile activity, resulting in a significant reduction in uterine
blood flow by mechanical compression of the uterine vasculature. The
third potential mechanism is by potentiating or by depressing adrenergic
neurotransmission. This is accomplished by altering the local neurogenic
release of norepinephrine from the presynaptic nerve terminal
or by modifying the responsiveness of the postsynaptic nerve terminal
to norepinephrine. The uterine vasculature effects of prostaglandins have been investigated
mainly in experimental animals. Clark and coworkers have shown that
PGE1 and PGE2 produce vasodilation in the uterine vasculature of the anesthetized nonpregnant
dog, whereas PGF2α does not significantly alter uterine blood flow.109,110 Subsequent studies in unanesthetized chronically catheterized nonpregnant
sheep have shown similar responses for PGE1 and PGE2, whereas PGF2α produced dose-related decreases in uterine blood flow.111,112 Similar studies have shown that PGI2 and PGD2 are potent uterine vasodilators in nonpregnant sheep.37,113 In contrast, the PGI2 breakdown product (6-keto-PGF1α) and TxB2 were very weak vasoconstrictors.37 The ability of prostaglandins to reduce uterine blood flow in nonpregnant
sheep by increasing uterine tone and contractile activity (mechanical
compression) has been documented for PGE2 and PGF2α.37,111,112 In addition, PGE and PGF also have been shown to alter both presynaptic
and postsynaptic sympathetic nerve transmission, thus affecting the
vascular response to uterine sympathetic nerve stimulation and circulating
norepinephrine.109,111,114 The local release of prostaglandins does not appear to be under neurogenic
control because levels of PGE and PGF in uterine venous effluent
were not altered by sympathetic nerve stimulation.114 Prostaglandins act by means of receptors, and, at least in the canine uterus, PGF2α is able to competitively antagonize vasodilation produced by other prostaglandins.115 This is important because the final vascular effects of endogenous prostaglandin
production are dependent on the summation of the individual
prostanoids. Finally, the nonpregnant uterus is composed of two separate
tissue layers, the myometrium and the endometrium. Studies that evaluate
uterine vascular effects of prostaglandins generally combine changes
in both tissue layers. Prostaglandins have been shown to act differently
in the myometrium and the endometrium.27 However, specific tissue changes would not be detected if only total uterine
blood flow were measured. An example of this has been reported
in the dog uterus, in which PGE1 preferentially dilates the myometrium, PGF2α preferentially constricts the endometrium, and PGE2 acts equally on both tissue layers.36 Whether this occurs in other species or during pregnancy has not been
investigated carefully. During the ovarian cycle, estrogen levels increase just before ovulation. This
elevation in estrogen is associated with a significant increase
in uterine blood flow.116,117 Prostaglandins have been implicated in this response because of their
uterine vasoactivity.118,119 Ryan and coworkers have shown that prostaglandin-synthesis inhibitors, meclofenamate
and indomethacin, decrease the uterine responses to estrogen
in rats.120 In contrast, studies in sheep have shown that although estrogen increases
PGI2 levels, inhibition of prostaglandin synthesis does not alter the uterine
vascular response to estrogen.118 These studies strongly suggest that estrogen-induced decreases in uterine
blood flow are not mediated by prostaglandins. More recently, Van
Buren and associates121 showed that estrogen-induced increases in uterine blood flow are mediated
by nitric oxide and are prevented by inhibitors of nitric oxide synthetase. Pregnancy creates a unique cardiovascular challenge for the maternal organism. Blood
volume and cardiac output increase by as much as 40%, yet
arterial blood pressure is not altered significantly. The reason for
this is the development of the uteroplacental vasculature and the fact
that the maternal vasculature dilates. The resistance in the uteroplacental
vasculature is very low. This is due to the development of a new
uteroplacental vascular bed and vasodilating hormonal factors that
appear to play a major role in regulating uteroplacental blood flow during
pregnancy. Prostaglandins are known to be produced in the uterus
and placenta in large quantities and may be important in regulating both
uterine and umbilical blood flow.97,99,122,123 Early studies by Terragno and coworkers124 and Venuto and coworkers125 demonstrated that the pregnant uterus of dogs and rabbits produced large
quantities of PGEs and that after blockade of prostaglandin synthesis, uterine
vascular resistance significantly increased. In contrast, in
animals that recovered from surgery for a period of 5 to 7 days, uterine
blood flow initially decreased but then returned to control levels
within 15 minutes.126,127 In these studies, uterine prostaglandin synthesis was inhibited significantly
by 15 minutes and remained low for the next 4 hours, even though
blood flow returned to control levels.126 These studies suggest that endogenous vasodilator prostaglandins may be
important in maintaining uterine blood flow during stressful situations
but may be less important in regulation of blood flow during normal
pregnancy. When interpreting these results, remember that prostaglandin-synthesis
inhibitors block the formation of all prostaglandins (i.e., vasodilators and vasoconstrictors) and that a complete understanding
of the role of endogenous prostaglandins in regulating uteroplacental
blood flow requires specific inhibition. Also, recent studies discussed
elsewhere strongly suggest that nitric oxide may play a more critical
role in regulating the uterine and systemic vascular resistance. The uterine vascular effects of prostaglandins in pregnancy have been evaluated
mainly in the unanesthetized chronically catheterized pregnant
sheep. PGE2 (a vasodilator in the nonpregnant animal) produced dose-related reductions
in uterine blood flow in pregnant sheep but after administration
to the fetus caused uterine vasodilation.38,128,129 Whether this uterine effect (vasodilation) seen after fetal administration
is the result of PGE2 or PGE2 metabolite is not known. PGE2 did produce a significant increase in myometrial tone after maternal administration, and
thus, decreased blood flow partially may reflect myometrial
compression of the vasculature.38,128,129 PGF2α produced dose-related reductions in uterine blood flow and, at the highest
doses tested, also increased uterine contractile activity and tone.38,129 The initial PGF2α-induced changes in uterine blood flow appear mainly to be the result of
vasoconstriction.38 In contrast, PGI2 produced dose-related increases in uterine blood flow in late term pregnant
sheep, whereas its breakdown product, 6-keto PGF1α, was a weak vasoconstrictor.38 The PGI2-induced increases in uterine blood flow in this species, which were observed
during intra-arterial infusions of PGI2, were largely the result of increases in endometrial blood flow rather
than increases in placental blood flow.130 Studies evaluating PGD2 also have shown it to be a potent vasodilator of the uterine vasculature
in late-term pregnant sheep, but whether it is able to dilate the placental
vasculature is unknown. In pregnant rabbits, PGD2 had a biphasic response, producing vasodilation in the myometrium and
vasoconstriction in the placenta.38,131 Similar studies have not been conducted in the sheep or other species. To
date, very little data exist that suggest that prostaglandins can
dilate the maternal placental vasculature. This most likely is because
the maternal placental vasculature is dilated maximally. If prostaglandins
have a role in regulation of uteroplacental blood flow, PGI2 would be the most likely candidate, but it would share this role with
nitric oxide. Umbilical Blood Flow Until recently, the effect of prostaglandins on the umbilical vasculature
in the intact animal has been limited to PGE2 and PGF2α, both of which were shown to be vasoconstrictors. PGE2 was by far the more potent vasoconstrictor.129,132 It is widely accepted in the clinical literature that prostacyclin is
a potent vasodilator of the uterine, umbilical, and placental vasculature
and that reduction of PGI2 production is associated with preeclampsia. The decrease in PGI2 is thought to be associated with reduced uterine and umbilical blood flow. Recently, several
studies have evaluated the umbilical vascular response
to PGI2.133,134 In both of these studies, PGI2 produced dose-related decreases in fetal arterial pressure, increases
in fetal heart rate, and decreases in umbilical blood flow that were associated with increases in umbilical
vascular resistance. These data are interpreted to suggest that systemic
prostacyclin administration leads to peripheral vasodilation and
shunting of blood away from the placenta, thus leading to decreases in
umbilical blood flow. Additionally, it is believed that because the
placental vasculature is dilated maximally and has no vascular tone, prostacyclin
could not produce any further relaxation. This hypothesis
however was tested in two separate studies134,135 by increasing umbilical vascular tone by simultaneous infusion of the
thromboxane A2-like compound U46619. Even under the condition of increased tone, prostacyclin (PGI2) could not dilate the umbilical circulation. Thus, to date, there is no
evidence in favor of an active prostaglandin dilator system in the umbilical
circulation, although several members of the prostaglandins are
effective vasoconstrictors in the umbilical circulation. Thus, the
proof that prostaglandins modulate umbilical blood flow does not currently
exist, and further investigation is required. Prostaglandins and Preeclampsia Although the aforementioned studies suggest that PGI2 is not a dilator in the umbilical circulation, no studies have ruled out
the possibility that PGI2 produced in the vessel walls is not important in regulating vascular tone. It
is clear that umbilical blood vessels from patients with preeclampsia, hypertension, and
IUGR all show significantly depressed PGI2 synthetic capabilities.107 These disease states also are associated with reduced uterine or umbilical
blood flow. Because of this, many investigators have made an association
between the two phenomena. The hypothesis, which is not proven, is that decreased PGI2 production in the uterine and umbilical vasculature results in reduced
blood flow in these vascular beds. Preeclampsia is thought to be caused
by endothelial dysfunction, which results in altered production of
several vasoactive substances such as PGI2 and nitric oxide. A second reasonable interpretation that could be made, based
on the current state of our knowledge, is that reduced uterine
and umbilical synthesis of PGI2 and nitric oxide leads to increased platelet aggregation, which in turn
leads to increased release of thromboxane A2 and serotonin. Thus, the increasing levels of thromboxane A2 or serotonin could be responsible for the significant decreases in uterine
and umbilical blood flow. Thromboxane A2 (evaluated as the thromboxane mimetic U46619) is a potent vasoconstrictor
of the uterine (unpublished data) and umbilical vasculature.134,135 Serotonin is also a potent vasoconstrictor in both the uterine and umbilical
vasculature.136,137 Based on the accepted belief that preeclampsia and reduced uteroplacental
blood flow are caused by inadequate production of vasoactive substances
such as prostacyclin, Fidler and colleagues138 attempted to treat preeclamptic patients with intravenous PGI2. In this study, the patient's diastolic blood pressure was decreased
from 140 to 80 mmHg. PGI2 was used only when other therapeutic agents failed to control the blood
pressure. It should be stressed that the safety of infusing this endogenous
prostanoid into pregnant women currently is not known. Uteroplacental
blood flow is extremely dependent on perfusion pressure, and PGI2 infusions that decrease mean arterial pressure significantly may lead
to significant reductions in uteroplacental blood flow, which could lead
to fetal hypoxia.139 Pregnancy also is associated with a reduced vascular responsiveness to
angiotensin II, both in humans and in sheep.140,141 Before the development of pregnancy-induced hypertension (PIH), angiotensin
II refractoriness is lost.140 This increase in vascular responsiveness probably is not specific for
angiotensin II but rather reflects increased vascular reactivity to all
vasoconstrictors.142 It originally was believed that angiotensin II increased uterine blood
flow and released PGEs from the uterus, which modulated uterine and systemic
pressor response to angiotensin II in pregnancy.124,125,143 This possibility has been addressed in two studies, one that showed release
of prostaglandins (PGE2 and PGI2 measured as 6-keto PGF1a) from the uteroplacental vasculature at high doses of angiotensin II144 and one that did not show any change.145 Neither study showed sufficient uterine release of prostaglandins to alter
systemic responses to angiotensin II or systemic vascular resistance. However, angiotensin
II did produce uterine vasoconstriction.141,144,145 In the study that found no uterine release of prostaglandins, bradykinin
released large quantities of both PGE and 6-keto-PGF1α, serving as a positive control.145 Because prostaglandins are metabolized quickly or undergo nonenzymatic
breakdown, the likelihood of a circulating vasodilator prostaglandin
released from the pregnant uterus being responsible for reduced systemic
vascular resistance or responsiveness does not seem likely. If prostaglandins
are responsible for regulating vascular responsiveness of blood
vessels, this would have to occur by local vessel synthesis. Parturition The role of prostaglandins in parturition has been investigated extensively
both in humans and in sheep. The similarities and differences in
endocrine changes that occur at the time of parturition in these two species
have been reviewed.146,147,148 Although peripheral levels of most prostaglandins do not change throughout
gestation, the levels of 13, 14-dihydro-15-keto-PGF2α, the PGF2α metabolite, are elevated during the last days of pregnancy and during
labor in both species.99,101,102,103 In contrast, the levels of PGE2, PGF2α, and 6-keto-PGF1α all are elevated in amniotic fluid during labor,102,149 and a recent report suggests that increased amniotic fluid prostaglandins
precede the onset of labor.150 Increasing amounts of data strongly suggest that prostaglandins play a
major role in the mechanism of parturition in both species. The observation
that PGE2 and PGF2α, as well as their synthetic analogues (15-methyl and 16, 16-dimethyl), are
effective in induction of abortions and labor and the fact that prostaglandin-synthesis
inhibitors can block premature labor and delay
parturition add further support to this concept. All the intrauterine tissues produce prostaglandins; however, the tissues
that produce these specific oxytocic prostaglandins that cause uterine
contractility at parturition and the signals that initiate this increased
production are not firmly identified. In sheep, the placenta appears
to be the tissue that produces prostaglandins at parturition as
a result of a signal from the fetal hypothalamic-pituitary-adrenal-placental
axis.147 No similar signal route occurs in human pregnancy at term, so current
attention is focused on paracrine mechanisms that operate in the fetal
membranes to initiate prostaglandin synthesis and stimulate labor.151 The amnion has been recognized as a major site of PGE2 synthesis at the time of labor in humans.152,153 The amnion is an extremely rich source of arachidonic acid, the precursor
of prostaglandins esterified in membrane phosphatidylethanolamine
or phosphatidylinositol. Whereas before labor, amnion synthesizes mainly
lipoxygenase products of arachidonic acid,154 during labor, PGE2 synthesis predominates with release of substrate arachidonic acid from
membrane phospholipids under the control of phospholipase A2 being seen.155 To reach the myometrium and thus cause contraction, PGE2 first has to cross the chorion, a rich tissue source of prostaglandin
dehydrogenase. However, in vitro transfer experiments have shown that
PGE2 can traverse amnion-chorion-decidua path intact, apparently by means of
an extracellular route,156 although the relative amount transferred is hotly debated.157,158,159 Therefore, PGE2 produced by the amnion could mediate uterine contractility. However, measurements
of peripheral plasma prostaglandin metabolites in labor showed
that PGF2α metabolites are increased,160 with little or no data available for increase in the corresponding PGE2 metabolites. This PGF2α does not appear to arise by reduction of PGE2 by fetal tissues; rather, it may represent true PGF2α synthesis. Wilson and associates161 recently noted that the decidua produces increased concentrations of PGF2α during labor and that this PGF2α is oxytocic. In the ovine model, changes in placental estrogen/progesterone synthesis
in favor of estrogen, under the influence of increased fetal adrenal
cortisol, appear to regulate prostaglandin synthesis. Such changes in
peripheral plasma estrogen or progesterone have not been demonstrated
in humans, giving rise to the concept of local changes in estrogen/progesterone
synthesis in the fetal membranes, which then locally regulate
prostaglandin synthesis.162 In addition, a variety of peptide hormones (cytokines,163 epidermal growth factor,164 transforming growth factor alpha,165 relaxin166) have been shown to stimulate fetal membrane prostaglandin synthesis in
vitro and have been suggested variously as signals for parturition. To
increase prostaglandin production, arachidonic acid must be liberated
from membrane phospholipids by phospholipases and the activity of PGH
synthase must increase (perhaps under the influence of peptide hormones) to
convert that arachidonic acid to prostaglandin. Activity of phospholipase
may be regulated directly or indirectly. Gravidin, a 58,000 molecular
weight protein produced by the chorion and found in amniotic
fluid, recently has been described as an inhibitor of phospholipase
activity.167 The activity of gravidin was reported to decrease during labor, thereby
paradoxically promoting an increase in phospholipase activity. Gravidin
may be a member of the annexin family of calcium-phospholipid-binding
proteins that inhibit phospholipase activity indirectly by binding
to the substrate phospholipid. Activity of annexin I in human fetal membranes
has been found to decrease at the time of parturition168 consistent with an increase in phospholipase activity. Two isoforms of
the PGH synthase (cyclooxygenase) enzyme have been described, the constitutively
expressed PGHS-1 (COX-1) isoform and the PGHS-2 (COX-2) isoform, which
is inducible by a variety of cytokines and growth factors. The
cytokine interleukin-1 (IL-1) and growth factors such as epidermal
growth factor (EGF), transforming growth factor (TGF), and tumor necrosis
factor (TNF) all can rapidly induce PGHS-2 and stimulate prostaglandin
synthesis in human amnion cells.169 Whereas the specific activity of PGHS enzyme in the human amnion increases
sharply before the onset of labor,170 the abundance of PGHS-1 mRNA in the amnion, the choriodecidua, and the
placenta does not change with labor and delivery.171 However, expression of PGHS-2 is increased significantly in both the amnion
and the chorion at the time of labor,172 suggesting that upregulation of PGHS-2 mediates the increased prostaglandin
synthesis. The use of indomethacin—a nonselective PGHS inhibitor—as
a tocolytic is limited because it causes oliguria, suggesting
that PGHS-2-specific inhibitors may be more useful tocolytics. Arachidonic acid for prostaglandin synthesis by PGHS enzyme is liberated
from cell membrane phospholipids by phospholipase enzymes. Recently, several
isoforms of the phospholipase A2 enzyme have been described, including the 85-kDa cytosolic phospholipase
A2 (cPLA2), which has a preference for substrate phospholipid-containing arachidonic
acid in the SN-2 position.173 Cytokines and growth factors also rapidly induce cPLA2 expression and activity in human fetal membranes, and IL-1β coordinately
induces both cPLA2 and PGHS-2 in amnion-derived epithelial cells for sustained PGE2 synthesis.169 Activity of cPLA2 in the human amnion increases throughout gestation and is highest before
the onset of labor,174 suggesting that it plays a role in prostaglandin production at labor. Since the first studies of Karim and coworkers and Bygdeman and coworkers
in 1968, it has been clear that PGE2 and PGF2α can induce labor both in term pregnancies and in the first and second
trimesters.175,176 The problem with these agents was not their effectiveness but rather the
persistent side-effects of vomiting and diarrhea. Attempts were made
to produce analogues of these primary prostaglandins that would reduce
side-effects. In the original studies, large quantities of PGE2 or PGF2α had to be used because of the efficient metabolism of primary prostaglandins
after intravenous infusion. Thus, one of the major objectives in
producing synthetic analogues was to generate compounds that were not
metabolized quickly, allowing a lowering of the therapeutic dose. These
synthetic attempts gave rise to the 15-methyl and 16, 16-dimethyl
analogues, which have greatly improved effectiveness but still can produce
some nonuterine side-effects. Hundreds of articles have appeared that discuss the benefits and disadvantages
of different routes of administration (oral, intravenous, vaginal, intra-amniotic), dosages to be used, and types of prostaglandins
to be used (PGE2, PGF2α, or an analogue) for the induction both of labor and abortions. This area
of research is still quite active because pharmaceutical corporations
continue to seek the most effective agent with the fewest side-effects. Several
reviews of the use of prostaglandins for induction of labor
and abortion have appeared; the reader is referred to them for additional
information.177,178 In addition to normal labor, prostaglandins also have proved to be very
effective in inducing labor in cases of intrauterine fetal death, anencephaly, and
hydatidiform mole.179,180,181 Reports also exist in which 15-methyl-PGF2α has been used effectively to treat postpartum or postabortion bleeding
due to uterine atony.182,183 The final clinical use of prostaglandins to be discussed is that of cervical
ripening. In early studies in which prostaglandins were used to
induce abortion and labor, it was noted that the cervix seemed to dilate, soften, and
efface even before strong uterine contractions. Tissue
samples taken from the human cervix in the first trimester have been
shown to produce PGE, PGF, the PGF2α metabolite 13, 14-dihydro-15-keto-PGF2α, and 6-keto-PGF1α. This production is enhanced at term.99 Using an animal model, Stys and coworkers have shown that intracervical
administration of PGE2 results in significant changes in compliance of the cervix of the pregnant
sheep at a time when no uterine contractile activity is observed.184 This study clearly demonstrates that PGE2 can effectively change cervical compliance. In recent years, PGE2-impregnated gels have been used effectively to ripen the cervix before
induction of labor in all three trimesters. These gels are applied directly
to the cervix by means of a transcervical catheter (12 hours before
induction) and produce ripening of the cervix in a large percentage
of primigravidas and multigravidas. Once the cervix is ripe, the outcome
of labor is improved significantly. A further discussion of this
clinical use can be found elsewhere.185,186 |