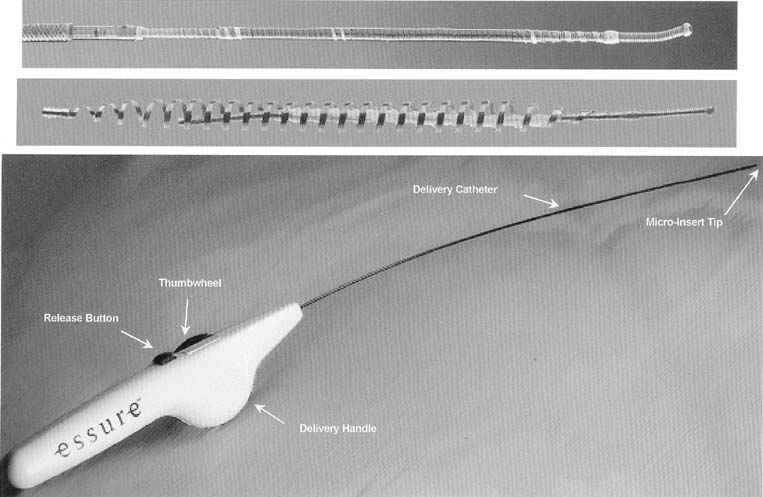
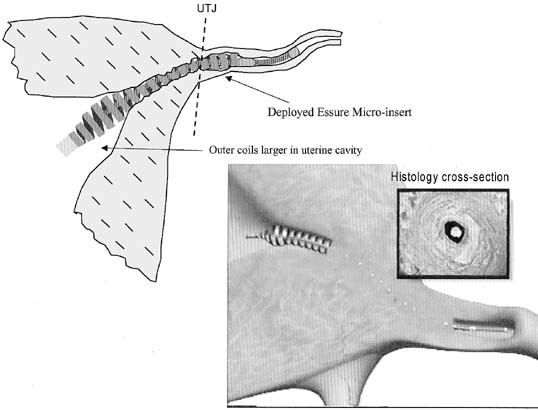
The ESSURE micro-insert is a microcoil that consists of a stainless steel inner coil, an outer coil made from nitinol and polyethylene terephthalate (PET), and Dacron fibers. The inner coil attaches the device to the guidewire used in its placement. The outer coil anchors the device in the fallopian tube on deployment. For the insertion, the device is maintained in a low-profile position through the use of a release catheter. A hydrofilled catheter facilitates the access of the tube and commands the guidewire, release catheter, and the device. The entire system is attached to a handle, aligned for one-hand release of the device. The device is delivered through a 5-French operating channel of a small-caliber hysteroscope (Fig. 1)
The PET fibers have had widespread clinical use for more than 40 years and have been demonstrated to produce an immediate local inflammatory response characterized by macrophages, fibroblasts, foreign body giant cells, and plasma cells. A moderate foreign-body inflammatory reaction is solicited by the PET fibers. This inflammatory response peaks between 2 and 3 weeks, after which the inflammatory response slowly resolves during a 10-week period. Extensive fibrosis results, causing occlusion and chronic anchoring of the PET material and any associated medical device.6
Technique of Insertion and Deployment
The patient is placed in the dorsal lithotomy position and the vaginal area and cervix cleansed with an antiseptic solution. A paracervical block is performed with a local anesthetic. Any local anesthetic can be used to perform the paracervical block, nonetheless, because of its low toxicity; an ester-type of anesthetic is preferable such as chloroprocaine, (Nesacaine) 1% solution, injecting 4 to 6 mL of the solution superficially at the base of each uterosacral ligament. A small amount of anesthetic (.5 mL) can also be applied on the anterior lip of the cervix to place the tenaculum without discomfort to the patient.
A small-caliber, 3.9 to 5.9 mm OD hysteroscope with continuous flow system is used. The tenaculum is optional, placed on the anterior lip of the cervix and hysteroscopy performed without previous cervical dilatation. Low viscosity fluids such as normal saline are preferable when using the hysteroscopic method with a continuous flow system. The normal saline is warmed to room temperature to avoid discomfort to the patient by cooling.
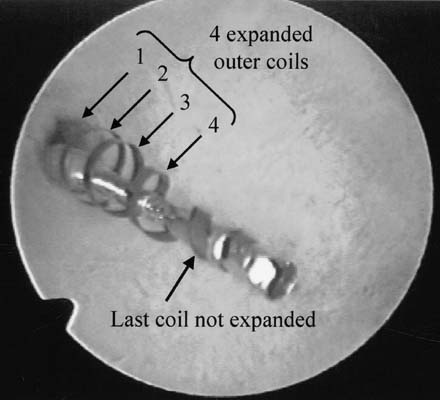
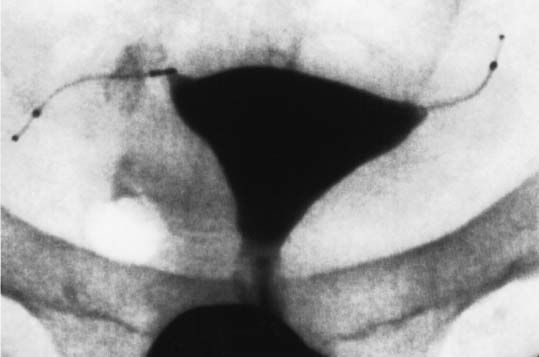
These procedures are best performed with a video camera to share with the patient the steps in performing the sterilization. The uterine cavity is evaluated and both tubal openings are observed. The ESSURE micro-insert is fed through the 5-French operating channel of the hysteroscope and gently guided into the tubal opening until the dark circle of the outer catheter comes into view, signifying time for deployment. The wheel in the handle is activated until a stop occurs, showing the micro-insert in the tubal lumen still not deployed in the catheter. A metal notch marker comes into view, signifying good placement, and by pressing the small button in the handle, the micro-insert is deployed. Further turning of the wheel in the handle will demonstrate the anchoring of the micro-insert by expansion of the spring outer metal. Several counterclockwise turns finally detaches the delivery system and the micro-insert proximal end is evaluated by hysteroscopy to assure that 5 to 10 mm (5 to 10 coils) of trailing micro-insert are in the uterine cavity. The same procedure is performed in the opposite fallopian tube (Figs. 2,3)
|
|
Evaluation of the ESSURE Micro-Inserts in a Prehysterectomy Study
To evaluate the feasibility of insertion, anchoring and complete tubal occlusion produced by the ESSURE micro-inserts, studies were designed to insert these devices in women who required hysterectomy and were willing to have this device tested. The initial study evaluated acute anchoring and feasibility of insertion and the woman underwent immediate hysterectomy. The second group of patients required to wait at least 3 months after the tubal devices were inserted to have a hysterectomy, to evaluate histology and tubal closure. At hysterectomy, the portion of the fallopian tubes containing the device was removed for histologic evaluation. Forty-three patients underwent device placement with the ESSURE micro-inserts. Forty-one patients had device placement in both fallopian tubes and two patients each had device placement in one tube, with a total of 84 tubes treated with device placements. The success of the device placements occurring in at least one tube in 43 patients or 50 patients per 84 (68%) attempts. Because of the uterine pathology present in this patient population, the device placement rate was significantly less than experienced in the intended sterilization population. Failure to place the device was considered to be device-related in only one tube (1%). In this case, the catheter failed to retract from the device. The cause of this failure was identified and resolved with design change. In those patients with uterine myomas or thickened endometrium who permitted visualization of the tubal ostium, placement of the device was achieved. The average procedure time was 15 minutes. No intraoperative adverse effects were noted during placement.6
The patients in this study were followed-up until their hysterectomy, which was performed between 24 hours and 14 weeks after device placement. Most were performed at the completion of almost 3 months. Tubal occlusion was evaluated by hysterosalpingography before the hysterectomy and all tubes (50/50; 100%) were found to be occluded. The histologic evaluation in 27 patients accounting for 47 tubes was also performed with an 80% occlusion rate in those patients with devices that had been in place for more than 4 weeks and consistent with 100% functional occlusion noted by the hysterosalpingogram performed before hysterectomy.
HISTOLOGIC EVALUATION
There were 47 patients enrolled and who underwent implantation with devices in the prehysterectomy trial. Table 1 provides the length of the device-wearing for each of the devices interaction in this study. There were no reports of pain during device-wearing. No pain was reported during pelvic examinations conducted just before hysterectomy in any of the patients. There was no evidence of inflammation, ulceration, or hemorrhage on gross examination of the uterus, except one patient with adenomyosis who was noted to have ulceration and hemorrhage in the uterine cavity; both fallopian tubes in this patient were unremarkable.
Table 1. Length of Device Wearing by Device Iteration
| Length of Device Wearing | No. Patients |
| <2 wk | 02 |
| 2–5 wk | 05 |
| 5–7 wk | 01 |
| 7–9 wk | 00 |
| 9–11 wk | 02 |
| 11–13 wk | 06 |
| 13–15 wk | 16 |
| >15 wk | 01 |
| Pending hysterectomy | 00 |
| Total | 33 |
Tubal occlusion was evaluated just before the hysterectomy by HSG. In tubes in which device movement or perforation did not occur occlusion was noted in 50 of 50 (100%) tubes evaluated.6
Methodology for Histologic Evaluation
Fallopian tubes of all patients enrolled in the study underwent both gross and microscopic examination. Care was taken at the time of hysterectomy to remove the uterus and tubes in block, without cutting into the device or using electrosurgery on the tubes. Once the tubes were removed, the uterus and tubes were x-rayed to determine the position of the devices in the tubes. Markers were placed at the uterotubal junction (UTJ) as a landmark. The uteri were then bivalved and the uterine end of the device was evaluated for position as well as to examine the local response to the device in the uterus. The uterine cornua and tubes were then excised and placed in formalin. An independent histopathologist then conducted gross examination of the tubes. Subsequent to the gross examination, the tubes were sent for processing at the Core Laboratory and then returned to the independent histopathologist for microscopic evaluation.6 The tubes were processed at the Core Laboratory as follows: the tubes were x-rayed to determine placement of the device within the tube. The specimens were then dehydrated in alcohol and xylene and embedded in methacrylate (MMA) until polymerized.
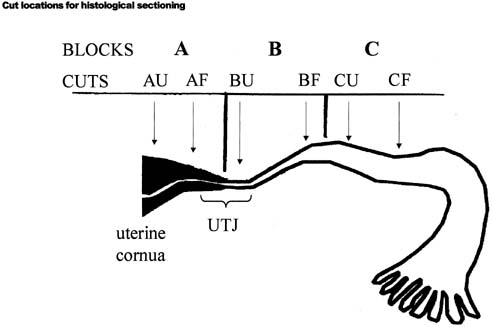
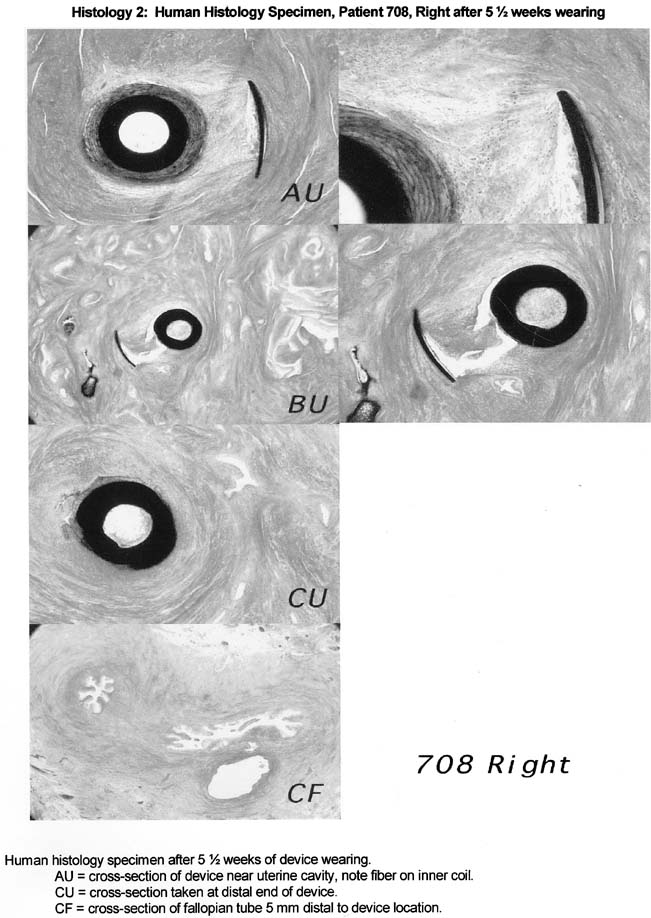
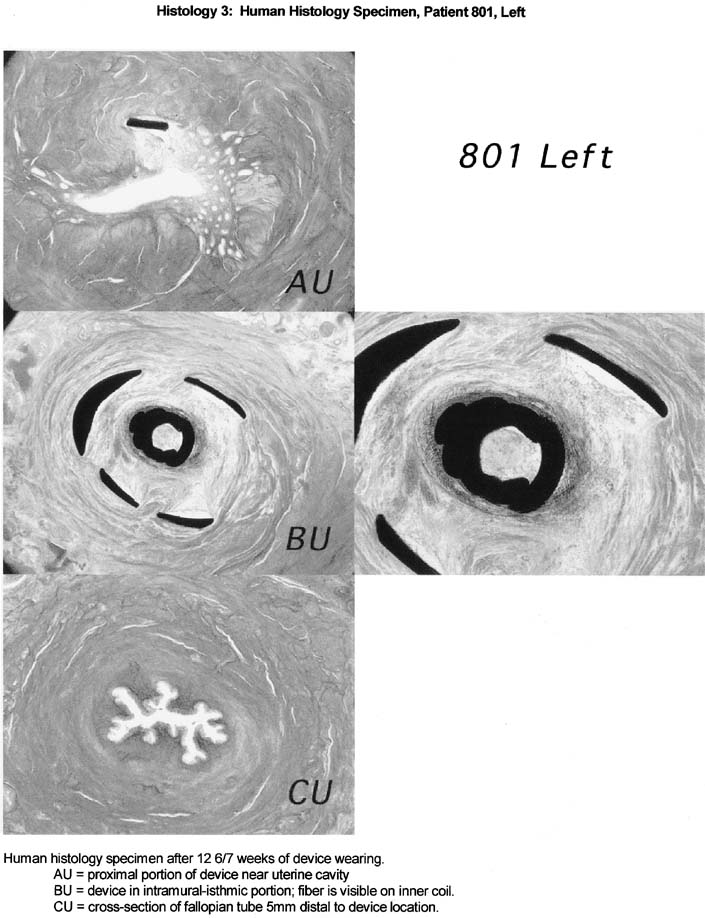
The resulting block was then compared with the x-ray and the imbedded fallopian tube was divided into three blocks according to the device position, using a diamond blade saw. (Figs. 4,5)
|
Block A included the uterine cornua up to the (UTJ). Block B included the UTJ to the proximal isthmic portion of the tubes, and block C included the proximal isthmic portion to a point within 5 mm distal to the end of the device. Two cross-sectional sections were then taken from the uterine end of blocks A, B, and C, and the fimbrial end of block C, using a diamond saw.
These sections were ground down to a thickness of approximately 50 to 100 microns for microscopic evaluation. Slides were then prepared from each section: One was stained in H and E and the other one with Goldner's trichrome. The MMA embedding process and the diamond saw cutting were undertaken to observe the integrity of the relationship with the device and the tissue.
Results of Histologic Evaluation
Patients wore the device ranging from 8 days to 14 weeks. The distribution of wearing time is noted in Table 1. Table 2 provides some quantitative information derived from the histology slides. The information is presented by wearing time to more clearly view the increasing tissue response seen over time.
Table 2. Histology Results by Tube
| Wearing Time (wks) | ||||||
| 1–4 n = 9 | 4–8 n = 5 | 8–12 n = 3 | 12–16 n = 8 | <16 n = 1 | Total N = 23 | |
| Tissue Reaction | Tubes | Tubes | Tubes | Tubes | Tubes | Tubes |
| Moderate/extensive acute inflammation | 7/9 | 2/5 | 0/3 | 17/29 | 0/1 | 26/47 |
| Moderate extensive chronic inflammation | 8/9 | 4/5 | 3/3 | 26/29 | 1/1 | 42/47 |
| Moderate/extensive loose fibrosis | 7/9 | 3/5 | 3/3 | 26/29 | 1/1 | 40/47 |
| Moderate/extensive dense fibrosis | 7/9 | 2/5 | 3/3 | 25/29 | 1/1 | 38/47 |
| Moderate/extensive disruption of epithelium | 7/9 | 4/5 | 3/3 | 29/29 | 1/1 | 44/47 |
| Moderate/extensive disruption of lamina propria | 7/9 | 4/5 | 3/3 | 28/29 | 1/1 | 43/47 |
| 80–100% obliteration of tubal lumen | 4/9 | 5/5 | 3/3 | 26/29 | 1/1 | 39/47 |
| Overall reaction to device = extensive | 3/9 | 1/5 | 3/3 | 26/29 | 1/1 | 34/47 |
| Overall reaction to device = moderate | 2/9 | 3/5 | 0/3 | 01.29 | 0/1 | 06/47 |
| Overall/reaction to device = mild | 4/9 | 1/5 | 0/3 | 02/29 | 0/1 | 07/47 |
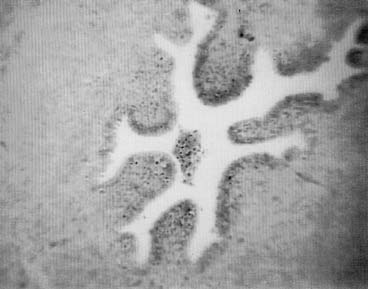
The histological response to the ESSURE micro-insert is characteristic of the histological response observed with the use of PET fibers in other anatomical sites. Specifically, the PET fibers appear to elicit strong fibrous and inflammatory tissue response that extends into the space viewed in the inner and outer coils of the ESSURE micro-inserts. The tissue response consists predominantly of macrophages and mononuclear cells, with some foreign body-type giant cells and acute inflammatory cells response. The fibrous response consists of both loose and dense fibrosis. In the studied cases with 3 months of wearing time, smooth muscle cells are also observed migrating from fallopian tube wall into the space viewed with the inner and outer coil. This response clearly demonstrated with comparable cross-sections of tubal segments of patients with and without the devices.
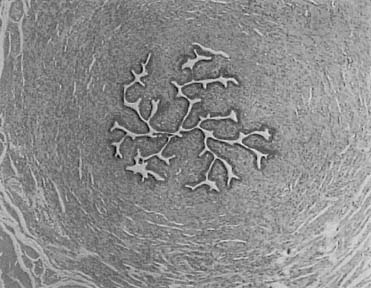
In addition, the fibrosis and tissue reaction were noted to be localized to the inner portions of the fallopian tube wall. There was no evidence that the fibrosis induced by the device will extend beyond the wall of the fallopian tube or cause peritubal adhesions or serositis (Figs. 6, 7, 8).
Normal tubal architecture was present within 5 mm distal to the end of the device. The histological analysis revealed normal tubal segment that were absent of inflammatory cells. This can be observed in specimens collected that were distal to the device (Fig. 9)
An independent histopathologist for the study concluded that the initial response to the ESSURE micro-insert is occlusive in nature and should provide for long-term anchoring of the device and pregnancy prevention. The study demonstrated that the tissue reaction is predictable, occurred in all specimens that contained fiber, and is localized to the device.