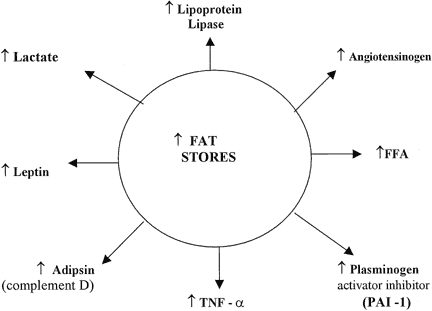
Adiposity is a risk factor for several conditions that contribute to the
increased morbidity and mortality in person who are overweight or obese. Type 2 Diabetes Mellitus Type 2 diabetes mellitus is strongly associated with obesity. The relative
risk for type 2 diabetes is significant even at normal BMI levels. In
the Nurses' Health Study, an increase in BMI from 23 to 25 was associated
with a fourfold risk of developing type 2 diabetes compared with
women whose BMI was less than 22. In subjects with more severe obesity (BMI
more than 35), the risk was 93 times higher than in women whose
BMI was less than 22.19 Moderate weight loss in this population (5 kg) was associated with a 50% reduction
in the risk of developing type 2 diabetes. In a recently
published study of 522 middle-aged subjects with impaired glucose tolerance
investigating the impact of lifestyle changes (weight reduction, increase
in fiber, decrease in total and saturated fat consumption, and
physical activity), the mean reduction of weight in a period of 3.2 years
was 4.2 ± 5.1 kg in the intervention group and 0.8 ± 3.7 in
the control group. The risk of progression to diabetes was
reduced by 58% in the intervention group, showing that type 2 diabetes
can be prevented by lifestyle changes in high-risk individuals.20 Moderate weight reduction (5%–10% of actual weight) has been associated
with marked improvement in insulin resistance and glucose control
and a decreased need for subsequent medical therapy.21,22 Hypertension The mechanism by which obesity leads to hypertension has yet to be elucidated. In
obesity-mediated hypertension there is increased sympathetic
nervous activation and increased renal sodium resorption, possibly mediated
or exaggerated by insulin resistance and/or hyperleptinemia.23 Despite the lack of knowledge of the precise mechanism causing hypertension, the
link between obesity and hypertension is well known: hypertension
is three times more common in obese than in normal-weight individuals.24 Most studies show that a moderate weight loss of as little as 4.5 kg has
a beneficial effect in lowering blood pressure.25,26 In the Dietary Approaches To Stop Hypertension (DASH) trial, a diet rich
in fruits, vegetables, low-fat dairy products, and reduced saturated
and total fat was shown to reduce systolic blood pressure by 5.5 mm
Hg and diastolic pressure by 3.0 mm Hg more than the control diet (typical
American diet).27 Blood pressure was substantially decreased in hypertensive and normotensive
subjects, showing that a healthy diet can have beneficial effects
beyond weight loss.28 Cardiovascular Disease Cardiovascular disease is the number one cause of death in women in the
United States.29 Excess weight is independently associated with an increase in coronary
artery disease in women and men.3 In a Finnish cohort of more than 16000 persons followed for 15 years, the
risk of death from all causes, including cardiovascular disease, increased
with increasing BMI in both men and women.30 In the Nurses' Health Study, during a follow-up of 14 years, the incidence
of coronary heart disease declined by 31%, partially explained by
a 41% decline in smoking, an increase of 175% in hormonal replacement
therapy, and improvement in diet. However, during this period, the proportion
of women who were overweight (BMI more than 24.9) increased by 38%, probably
slowing the decline in the incidence of coronary heart
disease.31 The pattern of fat distribution is an important predictor of the risk of
coronary artery disease, with central obesity being associated with
an increased risk for coronary artery disease.1 In the Nurses' Health Study, a higher waist-to-hip ratio and a larger
waist circumference were independently associated with a greater risk
for coronary artery disease in women between 40 and 65 years of age.32 Gallstones The risk of developing gallstones increases with BMI. The risk of either
gallstones or cholecystectomy is as high as 20 per 1000 women per year
for women with a BMI of more than 40, compared with 3 per 1000 among
women with a BMI less than 24.33 The risk of gallstone formation in the obese is also increased with weight
loss, particularly rapid weight loss,34 and is also associated with weight cycling among women.35 The risk for gallstone formation should be assessed in any weight management
program on the basis of current BMI and rate of weight loss, especially
during the first few weeks of dieting. It has been shown that
ursodeoxycholic acid (600 mg/day), a bile acid salt that reduces cholesterol
solubility, is highly effective in preventing gallstone formation
in patients experiencing dietary-induced weight reduction.36 Cancer There is epidemiologic evidence of an increased risk of breast cancer in
postmenopausal women with increasing BMI, waist-to-hip ratio, and waist
circumference. Weight loss appears to decrease the risk, especially
when occurring later in life. Possible causative factors linking BMI
with breast cancer include increased levels of endogenous sex hormones, insulin
resistance, and insulin-like growth factors. Interestingly, the
risk for breast cancer in premenopausal women decreases with higher
BMI.37 Obesity also appears to increase the risk of endometrial cancer, gallbladder
cancer, and colon cancer.38 Impact of Obesity on Reproductive Life The level of adiposity has a major impact on a woman's reproductive
life. There is a secular trend toward earlier menarche, correlated with
increasing obesity in all ethnic groups, which is most pronounced
in African-American girls.39 The impact of early menarche on a girl's life extends far beyond
the psychological effect of a shortened childhood. Obese children and
adolescents have lower self-esteem, and as young adults they may experience
an adverse socioeconomic outcome, with a decreased income and lower
educational levels compared with young adults of the same age who
are not obese.40,41 Later in adult life, early menarche and increased body fatness confer
a higher risk of cardiovascular disease and breast cancer.42,43 The polycystic ovarian syndrome (PCOS) occurs in 4% to 7% of women of reproductive
age,44,45 causing infertility and abnormal bleeding and increasing the risk for
endometrial carcinoma and cardiovascular disease.46,47 Hyperandrogenism and insulin resistance are the hallmarks of the syndrome. Forty
to fifty percent of women with PCOS are obese,48 exacerbating the insulin resistance. There is a strong association between
PCOS and glucose intolerance and eventually diabetes,49,50 reinforcing the role of insulin resistance in this syndrome. Diet and
exercise have been shown to be effective in improving insulin sensitivity
and restoring fertility in this group48,51,52 and should always be part of the arsenal of therapy for obese women with
PCOS. Current evidence shows that the use of insulin-sensitizing agents (metformin
and thiazolidinediones) in women with PCOS improves biochemical
manifestations of hyperandrogenism and increases ovulatory rates.53,54 Obese women who become pregnant are at higher risk for maternal and fetal
complications.55 The rates of preeclampsia and eclampsia increase with increasing maternal
weight,56 as does the risk of late fetal death,57 fetal macrosomia, and congenital malformations.58 Gestational diabetes mellitus has been reported in 10% of pregnancies
in obese women, and the prevalence of hypertension is increased tenfold.59 Because of difficulties in labor and delivery, obese pregnant women are
more likely to have a cesarean section than are normal-weight pregnant
women.60 Many women attribute retained weight from pregnancy as a cause for their
subsequent obesity.61 In some women weight gain may indeed be explained by retention of gestational
weight gain as well as negative postpartum lifestyle changes. In
large cohorts of pregnant women, the effect of pregnancy on average
body weight after 18 months postpartum was an increase of less than 0.5 kg; a
subgroup of 15% to 20% of the women gained a more significant
amount of weight after pregnancy.62 In a study of 1300 healthy women aged 18 to 41 years, 6.4% became overweight
after the index pregnancy with high gestational weight gain, young
age at menarche, younger age, and time from menarche to first pregnancy
all being associated with an increased risk of becoming overweight
after pregnancy.63 Weight loss during lactation differs markedly among women, with some women
even gaining weight during lactation.64 An average weight loss of 0.5 kg/week for 10 weeks in lactating overweight
and obese women who were exclusively breastfeeding did not affect
the growth of their infants,65 suggesting that lifestyle modifications and moderate weight loss are safe
in lactating women and their infants. Adding to the beneficial effects of lactation in the neonate, it has been
shown that the risk of becoming overweight during older childhood and
adolescence is lower among children who were breastfed for 7 months
or more compared with those who were breastfed for 3 months or less,66 suggesting a role for breastfeeding as an important tool in the prevention
of obesity during childhood. |  40
40