Altered Fetal Growth
Authors
INTRODUCTION
Altered fetal growth, either too slow or too rapid, may be caused by several distinctly different fetal and maternal conditions. With the advent of sophisticated antenatal assessment tools and new therapeutic modalities, many of these problems will probably become manageable in the future.
INTRAUTERINE GROWTH RESTRICTION
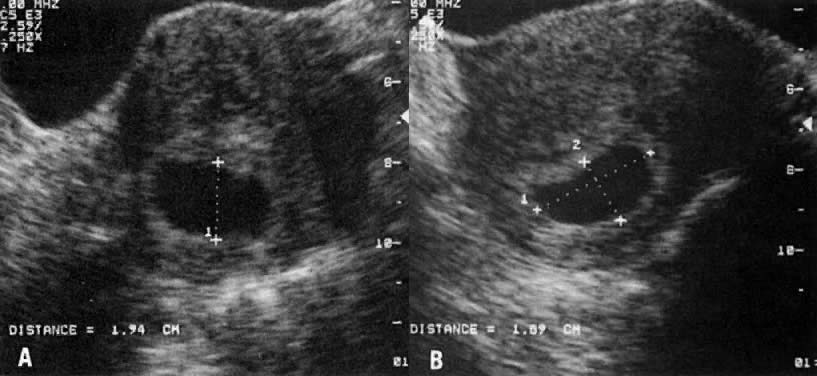
Antenatal diagnosis of intrauterine growth restriction (IUGR) by clinical means is possible in approximately one third of such pregnancies.1 This low yield is attributed to inaccurate assessment of dates and fetal weight relative to fundal height.2, 3, 4, 5 By comparison, a diagnosis of altered fetal growth, using a combination of various sonographic parameters, can either be excluded or verified in over 90% and 80% of cases, respectively. These figures indicate that ultrasound, like other tests used to assess biologic function, is more specific in predicting the unaffected fetus than it is sensitive in the diagnosis of IUGR (Fig. 1).
Yerushalmy reported an eightfold increase in the perinatal mortality of the small-for-gestational-age (SGA) neonate.6 Usher and McLean also found a tenfold increase in perinatal mortality in infants whose birth weight (BW) fell below 2 SD of the mean for dates.7 As to perinatal morbidity, Gruenwald,8 Naeye and Kelly,9 Scott and Usher,10 and Fitzhardinge and Steven11 have all described the nature of immediate and long-term physical and neurologic sequelae of the SGA infant.
Although the etiology of IUGR remains idiopathic in approximately 50% of cases, a number of factors may adversely influence fetal growth, including:
Genetic and chromosomal causes
Chronic fetal infections
Maternal use of drugs (alcohol, narcotics, nicotine, dilantin, beta-adrenergic blockers, corticosteroids)
Maternal disease (cardiovascular, renal, autoimmune, metabolic)
Poor maternal nutrition
Prepregnancy weight <50 kg
High altitude
Multiple pregnancy
Irradiation.
Winick has suggested that early in pregnancy fetal organ growth takes place first by cell hyperplasia or cell division.12 This is followed by cell hypertrophy or increase in cell size. Finally, hyperplasia ceases and growth continues by cellular hypertrophy alone.12
Because hyperplasia normally ceases at some time during fetal life, an early insult is likely to retard cell division and possibly lead to symmetric reduction in organ size.12 By contrast, delayed onset of growth restriction (after organ cell number is complete) is believed to adversely affect only cell size, and result in asymmetric IUGR. In the latter condition cephalic size is relatively spared in comparison to body weight, suggesting that the pathophysiologic process is both recent in onset and potentially remediable.13, 14
The physician involved in antenatal recognition of IUGR should be well versed with current problematic and controversial issues in making such diagnoses. These areas include nonuniformity in:
Definition of IUGR
Lack of due consideration to important factors such as fetal length and growth potential
Use of BW charts inappropriate for pregnant women residing in geographic areas at or near sea level.
In this chapter a discussion of these issues is first presented. Second, a description and critique of the sonographic methodology used in the assessment of fetal growth, BW, and oligohydramnios follows. Finally, the current management modalities of the IUGR fetus are reviewed.
PROBLEMATIC AREAS
Definition
Although most physicians use the 10th centile of BW as the cut-off point for making the diagnosis of IUGR,15, 16, 17, 18, 19 others consider the fifth or even third centile as significant centiles delineating growth restriction.20, 21, 22 On the other hand, some investigators are mainly concerned with the presence of fetal malnutrition (FM) or wasting, clinically apparent by loss of subcutaneous tissue and muscle mass. Fetuses in this latter group usually have a ponderal index (BW × 100/crown–heel length3) below the 10th centile.23, 24
Fetal length
Miller and Merrit emphasized the importance of relating BW to the crown–heel length (CHL) of neonates.24 According to their data the 10th centile of BW for a neonate with a CHL of 51 cm is 2940 g, clearly placing it in an IUGR category (Table 1).24 Nonetheless, many pediatricians find it difficult to make a diagnosis of IUGR in an infant weighing 2940 g.
Table 1. Birthweights (kg) for given centiles in babies of different crown–heel length
Birth weight centiles | Crown–Heel Length (cm) | ||
48.0 | 51.0 | 55.0 | |
95 | 3.16 | 3.79 | 4.76 |
90 | 3.05 | 3.66 | 4.59 |
75 | 2.95 | 3.54 | 4.45 |
50 | 2.81 | 3.37 | 4.21 |
25 | 2.67 | 3.19 | 4.04 |
10 | 2.56 | 3.08 | 3.86 |
5 | 2.45 | 2.94 | 3.69 |
(Adapted from Miller HC, Merritt T: Fetal growth in humans. Chicago, Year Book Medical Publishers, 1979)
Careful attention to neonatal weight versus length (ponderal index) on a universal basis will remedy this situation. Of interest, Hadlock and associates and O'Brien and associates have shown that in the fetus, the length of the femur bone can be used as an indirect estimate of its CHL.25, 26 However, for reasons discussed elsewhere, calculation of fetal ponderal index has not enhanced the diagnosis of the long asymmetrically undergrown fetus. The method of using femur length (FL) in evaluation of BW and IUGR is discussed in other sections.
Growth potential
In specific pregnancies, it is important for the physician to predict fetal growth potential. This can be done in two ways. The first is by serial cephalometry and the second is by comparison of size to sibling weight.
Concerning cephalic growth, Sabbagha and associates showed that 90% of fetuses normally maintain the same biparietal diameter (BPI) centile rank achieved by mid pregnancy.27, 28 To determine cephalic rank, the BPD should be measured serially and classified into one of three categories: large, average, and small. This finding implies that if the BPD growth bracket is defined, for instance by 30 weeks' gestation, fetal cephalic growth potential can be estimated for the remainder of pregnancy. Thus, in specific high-risk pregnancies BPD growth attained at or near term can be compared to that expected, rather than to a mean value derived from a heterogeneous population of fetuses.27
For example, a single BPD value of 9.0 cm is considered normal if the length of pregnancy is 36 weeks (Table 2). However, if the growth potential of the same fetus indicates that the BPD measurement at 36 weeks' gestation should be 9.4 cm rather than 9.0 cm (i.e., at the 80th centile rather than at the 50th centile), the risk for IUGR is increased to 20%.29 Equipped with such information, the physician will continue to carefully monitor fetal status.
Table 2. BPD centile ranges and measurements for both Black and White fetuses
BPD centile values from 16–40 weeks | ||||||||
Fetal age (weeks) | BPD centiles | |||||||
5 | 10 | 25 | 50 | 75 | 80 | 95 | N | |
16 | 3.1 | 3.2 | 3.4 | 3.7 | 4.0 | 4.1 | 4.5 | 12 |
17 | 3.4 | 3.5 | 3.7 | 4.0 | 4.3 | 4.4 | 4.7 | 15 |
18 | 3.7 | 3.8 | 4.0 | 4.3 | 4.5 | 4.6 | 4.9 | 22 |
19 | 3.9 | 4.2 | 4.3 | 4.5 | 4.8 | 4.9 | 5.1 | 33 |
20 | 4.2 | 4.5 | 4.6 | 4.7 | 5.0 | 5.1 | 5.3 | 39 |
21 | 4.5 | 4.8 | 4.9 | 5.0 | 5.3 | 5.4 | 5.5 | 40 |
22 | 4.9 | 5.0 | 5.2 | 5.3 | 5.6 | 5.7 | 5.8 | 48 |
23 | 5.2 | 5.3 | 5.5 | 5.6 | 5.9 | 6.0 | 6.2 | 57 |
24 | 5.5 | 5.6 | 5.8 | 5.9 | 6.2 | 6.3 | 6.6 | 50 |
25 | 5.8 | 5.9 | 6.0 | 6.2 | 6.5 | 6.6 | 7.0 | 47 |
26 | 6.1 | 6.2 | 6.3 | 6.6 | 6.8 | 6.9 | 7.3 | 43 |
27 | 6.4 | 6.5 | 6.7 | 6.9 | 7.1 | 7.2 | 7.6 | 51 |
28 | 6.6 | 6.7 | 7.0 | 7.2 | 7.4 | 7.5 | 7.9 | 51 |
29 | 6.8 | 6.9 | 7.3 | 7.5 | 7.8 | 7.9 | 8.3 | 53 |
30 | 7.1 | 7.2 | 7.6 | 7.8 | 8.0 | 8.2 | 8.6 | 50 |
31 | 7.3 | 7.4 | 7.8 | 8.0 | 8.2 | 8.4 | 8.8 | 48 |
32 | 7.5 | 7.6 | 8.0 | 8.3 | 8.4 | 8.6 | 9.0 | 47 |
33 | 7.7 | 7.8 | 8.3 | 8.5 | 8.6 | 8.8 | 9.1 | 50 |
34 | 7.9 | 8.0 | 8.5 | 8.7 | 8.9 | 9.1 | 9.3 | 50 |
35 | 8.2 | 8.3 | 8.7 | 8.8 | 9.1 | 9.3 | 9.6 | 49 |
36 | 8.3 | 8.5 | 8.9 | 9.0 | 9.3 | 9.4 | 9.7 | 48 |
37 | 8.4 | 8.8 | 9.0 | 9.2 | 9.4 | 9.5 | 9.8 | 43 |
38 | 8.5 | 8.9 | 9.1 | 9.3 | 9.5 | 9.6 | 9.9 | 42 |
39 | 8.7 | 9.0 | 9.2 | 9.4 | 9.6 | 9.7 | 10.0 | 29 |
40 | 8.9 | 9.3 | 9.4 | 9.5 | 9.7 | 9.8 | 10.1 | 15 |
(Sabbagha RE, Barton BA, Barton FB et al: Sonar biparietal diameter II. Am J Obstet Gynecol 126: 485, 1976)
The concept of comparing actual to potential cephalic growth is similar to that proposed by Turner,30 who underscored the importance of comparing fetal size to that of sibling weight. He showed the validity of this concept by showing that 80% of infants affected by congenital rubella were growth restricted in relation to their potential weight, exemplified by that of their normal siblings. However, in comparison to the population as a whole, only 46% of the affected infants fell below normal BW; the remaining 34% were falsely assumed to be normal.
Evaluation of birth weight
In evaluating the normality of BW the physician should be cognizant of several problematic areas. There is presently little unanimity about which BW charts should be used. For example, there is tendency by physicians to universally use the chart by Battaglia and Lubchenco,15 despite the fact that it is only applicable to pregnant women residing in geographic areas, approximately 6000 ft above sea level. Such an approach is likely to result in diagnostic errors. For example, at 36 weeks, a 2100 g neonate born at or near sea level can be approximately placed either in the 15th or fifth centile brackets, depending on which chart is used (Table 3). Second, in identifying long-term sequelae of IUGR infants researchers have focused on very small neonates, those in whom BW falls below the fifth or third centile. Although this approach is justifiable because it is more likely to show measurable ill effects, it is quite restrictive as it excludes the evaluation and follow up of infants with diminished growth, those who are less severely affected and in whom BW falls at the 15th or even 20th centile.
Table 3. Tenth centile of birth weights (g) according to the duration of pregnancy
Week | California | Cleveland | Denver |
27 | 632 | 660 | 770 |
28 | 727 | 770 | 860 |
30 | 966 | 1030 | 1060 |
32 | 1301 | 1310 | 1290 |
34 | 1744 | 1670 | 1600 |
36 | 2229 | 2190 | 2050 |
38 | 2629 | 2510 | 2430 |
40 | 2848 | 2750 | 2630 |
42 | 2965 | 2830 | 2720 |
(Adapted from Williams RL, Creasy RK, Cunningham GC et al: Fetal growth and perinatal viability in California. Obstet Gynecol 59: 624, 1982. Reprinted with permission from The American College of Obstetricians and Gynecologists.)
Furthermore, insufficient attention is directed to the fact that states of normal versus altered growth can be more precisely delineated by evaluation of BW in conjunction with specific growth patterns of the fetal head and body. For example, Tamura and associates reported that in women presenting with premature labor or premature rupture of the membranes fetal cephalic and body size are significantly reduced when compared to normal (i.e., fetuses who delivered at term).31 In their report, BW in the majority of these spontaneously delivered preterm neonates was small but within the low normal range (i.e., between the 10th and 25th centiles). Thus, if the only criterion used in evaluating size were BW <10 centile, the diminution of growth (with all its possible implications in such pregnancies) would have been totally missed.
In conclusion, there is need for uniform applicability of the methods used for diagnosis of IUGR. Each fetus should be evaluated as a separate entity with attention directed to a variety of factors, including:
Comparison of BW to tables appropriate for the fetus, specific to fetal gender
Use of the 5th centile as the cut-off for IUGR and the brackets between the 10th and 20th centiles as the area for identifying diminished growth
Evaluation of the growth potential of each fetus including factors such as maternal ethnicity, height, weight, and age
Measurement of fetal length and emphasis on clinical evidence of “wasting”.
FETAL GROWTH PARAMETERS
The fetal body parts most frequently used to evaluate growth include biparietal diameter (BPD), head circumference (HC), abdominal circumference (AC), humerus length (HL), and femur length (FL). However, before any of these data are interpreted it is mandatory that physicians become well versed with the definition of predictive value, sensitivity, and specificity of biological tests, and with dependence of the predictive value of tests on prevalence of the disease in specific geographic areas.
The use of these tests is illustrated by relating the sonographic finding such as oligohydramnios to the diagnosis of IUGR (Table 4). Review of this table shows that the sensitivity of oligohydramnios in the diagnosis of growth restriction is 16%. However, the reported sensitivity is correct only to the extent that the incidence of IUGR is 10%. For example, in a high-risk population the prevalence of IUGR fetuses is increased. As a result the predictive value and sensitivity of oligohydramnios will be enhanced.
Table 4. Predictive value, sensitivity, and specificity of oligohydramnios*
| SGA | Not SGA | No. of Patients |
Oligohydramnios | 38 | 58 | 96 |
No oligohydramnios | 207 | 2150 | 2357 |
Total | 245 | 2208 | 2453 |
*Predictive value of oligohydramnios in the diagnosis of IUGR is 38/96 or 40%; sensitivity is 38/245 or 16%; predictive value of normal fluid is 2150/2357 or 91%; and specificity is 2150/2208 or 97%
(Adapted from Philipson EH, Sokol RJ, Williams T: Oligohydramnios: Clinical associations and predictive value for intrauterine growth retardation. Am J Obstet Gynecol 146: 271, 1983)
Biparietal diameter
Growth of the BPD in relation to gestational age is shown in Table 2: this diameter is highly specific, when it remains normal (i.e., greater than 25th centile) (Fig. 2). In such pregnancies IUGR can be excluded in approximately 90% of neonates.29 However, the sensitivity of a small BPD (i.e., <25th centile) is only 52%.29
|
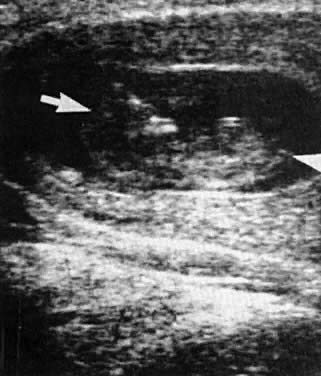
Other limitations in evaluating growth by BPD include imprecision in the estimation of birth weight; poor detection rate of asymmetrically undergrown babies, with normal or near normal cephalic size; and changes in head shape such as side-to-side flattening or dolichocephaly. This latter condition may occur in some pregnancies during the third trimester, rendering the BPD artificially small (Fig. 3). In such situations, unless HC is substituted for BPD, falsely abnormal diagnoses of IUGR can be made.
|
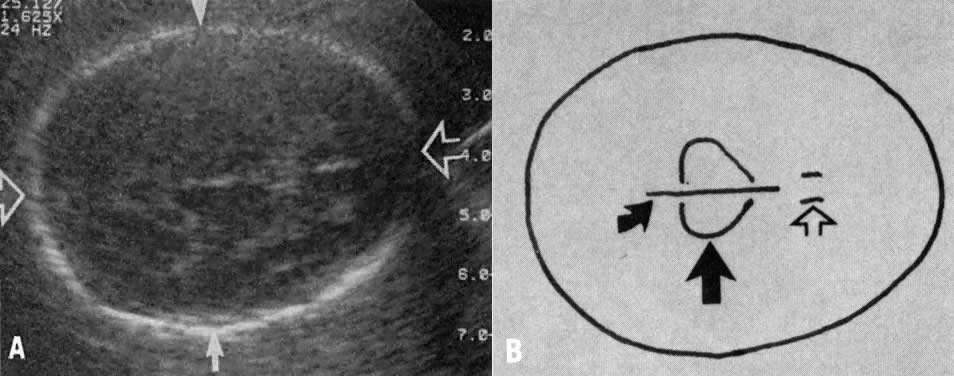
Abdominal circumference
The AC is very useful in assessing nutritional status in normal and altered states of fetal growth (Fig. 4). The reason is that it encompasses the liver and subcutaneous tissue in that area, both of which show reduction in size secondary to chronic hypoxia and decrease in substrate, associated with IUGR.
Biparietal diameter/head circumference and abdominal circumference growth patterns
The methodology of measuring HC and AC is best obtained directly from area tracings directly on the ultrasound machines or alternatively using growth tables (Table 5 and Table 6).32 In the event that this is not possible, mean diameter measurements (derived from outer to outer dimensions of the fetal head and body) should be obtained and then compared to their respective centile ranks (Table 7 and Table 8).32 This is important because transformation of mean diameters to circular or elliptical perimeters often results in erroneous measurements because neither the fetal head nor the fetal trunk conform precisely to these shapes.
Table 5. Centile ranks of fetal head circumference (HC)* relative to gestational age
Week† | Centile Ranks of Head Circumference (cm) | ||||
10 | 25 | 50 | 75 | 90 | |
18 | 14.0 | 15.5 | 16.0 | 17.0 | 17.5 |
19 | 15.0 | 16.0 | 17.0 | 17.5 | 18.0 |
20 | 16.0 | 17.0 | 18.0 | 18.5 | 19.0 |
21 | 17.0 | 18.0 | 19.0 | 19.5 | 20.0 |
22 | 18.0 | 19.0 | 20.0 | 20.5 | 21.0 |
23 | 19.5 | 20.0 | 21.0 | 21.5 | 22.0 |
24 | 21.0 | 21.5 | 22.0 | 22.5 | 23.0 |
25 | 22.0 | 22.5 | 23.0 | 23.5 | 24.0 |
26 | 23.0 | 23.5 | 24.0 | 24.5 | 25.0 |
27 | 24.0 | 25.0 | 26.0 | 26.5 | 27.0 |
28 | 25.5 | 26.0 | 27.0 | 27.5 | 28.0 |
29 | 26.5 | 27.0 | 28.0 | 29.0 | 29.5 |
30 | 27.0 | 27.5 | 28.5 | 29.0 | 30.5 |
31 | 27.0 | 28.0 | 29.0 | 30.0 | 31.0 |
32 | 27.5 | 28.0 | 29.0 | 30.0 | 31.5 |
33 | 28.0 | 28.5 | 29.5 | 30.5 | 32.0 |
34 | 28.5 | 29.0 | 30.5 | 31.5 | 32.5 |
35 | 29.5 | 30.0 | 31.5 | 32.0 | 33.0 |
36 | 30.0 | 31.0 | 32.0 | 33.0 | 34.0 |
37 | 30.5 | 31.5 | 32.5 | 33.5 | 35.0 |
38 | 30.5 | 31.5 | 32.5 | 34.0 | 35.0 |
39 | 31.0 | 32.0 | 33.0 | 34.5 | 35.0 |
40 | 31.5 | 32.5 | 33.5 | 34.5 | 35.5 |
41 | 32.0 | 33.0 | 34.0 | 34.5 | 36.0 |
*Head circumference as measured directly from tracings on the screen of ultrasound machines or alternatively by digitizer from photographs.
†menstrual weeks of pregnancy.
(Sabbagha RE: Intrauterine growth retardation. In Sabbagha RE [ed]: Diagnostic Ultrasound Applied to Obstestrics and Gynecology, 2nd ed, p 116. Philadelphia, JB Lippincott, 1987)
Table 6. Fetal abdominal circumference measurements (cm)*
Weeks of Gestation | Centile | ||||||||
2.5 | 5 | 10 | 25 | 50 | 75 | 80 | 95 | 97.5 | |
18 | 9.8 | 10.3 | 10.9 | 11.9 | 13.1 | 14.2 | 14.5 | 15.9 | 16.4 |
19 | 11.1 | 11.6 | 12.3 | 13.3 | 14.4 | 15.6 | 15.9 | 17.2 | 17.8 |
20 | 12.1 | 12.6 | 13.3 | 14.3 | 15.4 | 16.6 | 16.9 | 18.2 | 18.8 |
21 | 13.7 | 14.2 | 14.8 | 15.9 | 17.0 | 18.1 | 18.4 | 19.8 | 20.3 |
22 | 14.7 | 15.2 | 15.8 | 16.9 | 18.0 | 19.1 | 19.4 | 20.8 | 21.3 |
23 | 16.0 | 16.5 | 17.1 | 18.2 | 19.3 | 20.4 | 20.7 | 22.1 | 22.6 |
24 | 17.2 | 17.7 | 18.3 | 19.4 | 20.5 | 21.6 | 21.9 | 23.3 | 23.8 |
25 | 18.0 | 18.5 | 19.1 | 20.2 | 21.3 | 22.4 | 22.7 | 24.1 | 24.6 |
26 | 18.8 | 19.3 | 19.9 | 21.0 | 22.1 | 23.2 | 23.5 | 24.9 | 25.4 |
27 | 20.4 | 20.9 | 21.5 | 22.6 | 23.7 | 24.8 | 25.1 | 26.5 | 27.0 |
28 | 22.0 | 22.5 | 23.1 | 24.2 | 25.3 | 26.4 | 26.7 | 28.1 | 28.6 |
29 | 23.6 | 24.1 | 24.7 | 25.8 | 26.9 | 28.0 | 28.3 | 29.7 | 30.2 |
30 | 24.1 | 24.6 | 25.2 | 26.3 | 27.4 | 28.5 | 28.8 | 30.2 | 30.7 |
31 | 24.7 | 25.2 | 25.8 | 26.9 | 28.0 | 29.1 | 29.4 | 30.8 | 31.3 |
32 | 25.4 | 25.9 | 26.5 | 27.6 | 28.7 | 29.8 | 30.1 | 31.5 | 32.0 |
33 | 25.7 | 26.2 | 26.8 | 27.9 | 29.0 | 30.1 | 30.4 | 31.8 | 32.3 |
34 | 26.8 | 27.3 | 27.9 | 29.0 | 30.1 | 31.2 | 31.5 | 32.9 | 33.4 |
35 | 28.9 | 29.4 | 30.0 | 31.1 | 32.2 | 33.3 | 33.6 | 35.0 | 35.5 |
36 | 30.0 | 30.5 | 31.1 | 32.2 | 33.3 | 34.4 | 34.7 | 36.1 | 36.6 |
37 | 31.1 | 31.6 | 32.2 | 33.3 | 34.4 | 35.5 | 35.8 | 37.2 | 37.7 |
38 | 32.4 | 32.9 | 33.5 | 34.6 | 35.7 | 36.8 | 37.1 | 38.5 | 39.0 |
39 | 32.6 | 33.1 | 33.7 | 34.8 | 35.9 | 37.0 | 37.3 | 38.7 | 39.2 |
40 | 32.8 | 33.3 | 33.9 | 35.0 | 36.1 | 37.2 | 37.5 | 38.9 | 39.4 |
41 | 33.8 | 34.3 | 34.9 | 36.0 | 37.1 | 38.2 | 38.5 | 39.9 | 40.4 |
*Circumference measurements are obtained from the outer aspect of the fetal abdomen at the area of the liver, which shows the ductus venosus.
(Tamura RK, Sabbagha RE: Percentile ranks of fetal sonar abdominal circumference measurements. Am J Obstet Gynecol 138: 475, 1980)
Table 7. Mean head diameters (biparietal diameter + occipitofrontal diameter/2)
Gestational Age | Sample Size | 10th | 25th | 50th | 75th | 90th |
18 | 10 | 4.19 | 4.57 | 4.73 | 5.16 | 5.29 |
19 | 11 | 4.73 | 4.98 | 5.18 | 5.27 | 5.39 |
20 | 25 | 5.34 | 5.46 | 5.62 | 5.77 | 5.97 |
21 | 22 | 5.61 | 5.70 | 5.90 | 6.11 | 6.27 |
22 | 31 | 5.77 | 6.06 | 6.27 | 6.42 | 6.53 |
23 | 28 | 6.18 | 6.34 | 6.48 | 6.62 | 6.96 |
24 | 21 | 6.68 | 6.77 | 6.86 | 7.02 | 7.17 |
25 | 12 | 6.88 | 6.97 | 7.08 | 7.24 | 7.50 |
26 | 11 | 7.17 | 7.28 | 7.45 | 7.66 | 7.70 |
27 | 10 | 7.51 | 7.54 | 7.76 | 7.90 | 8.29 |
28 | 12 | 7.34 | 7.67 | 7.94 | 8.30 | 8.65 |
29 | 11 | 8.28 | 8.35 | 8.70 | 9.00 | 9.30 |
30 | 24 | 8.50 | 8.65 | 8.92 | 9.30 | 9.81 |
31 | 40 | 8.49 | 8.66 | 8.95 | 9.24 | 9.78 |
32 | 33 | 8.69 | 8.91 | 9.20 | 9.52 | 9.73 |
33 | 35 | 8.56 | 8.82 | 9.29 | 9.52 | 9.77 |
34 | 17 | 8.73 | 9.01 | 9.37 | 9.59 | 9.74 |
35 | 11 | 8.98 | 9.48 | 9.75 | 10.10 | 10.31 |
36 | 11 | 9.24 | 9.48 | 9.65 | 10.04 | 10.35 |
37 | 10 | 9.15 | 9.74 | 10.09 | 10.41 | 10.49 |
38 | 30 | 9.57 | 9.89 | 10.09 | 10.43 | 10.71 |
39 | 29 | 9.58 | 9.75 | 10.00 | 10.42 | 10.62 |
40 | 10 | 9.98 | 10.24 | 10.33 | 10.66 | 11.25 |
41 | 12 | 9.60 | 9.88 | 9.91 | 9.92 | 10.17 |
(Sabbagha RE: Intrauterine growth retardation. In Sabbagha RE [ed]: Diagnostic Ultrasound Applied to Obstetrics and Gynecology, 2nd ed, p 119. Philadelphia, JB Lippincott, 1987)
Table 8. Centile ranks of mean abdominal diameters
Gestational Age | Centile | |||||
N | 10th | 25th | 50th | 75th | 90th | |
18 | 10 | 3.56 | 3.91 | 4.43 | 4.58 | 5.20 |
19 | 11 | 4.36 | 4.45 | 4.65 | 4.85 | 5.22 |
20 | 25 | 4.41 | 4.59 | 4.78 | 4.94 | 5.22 |
21 | 22 | 4.65 | 4.90 | 5.05 | 5.35 | 5.85 |
22 | 32 | 4.98 | 5.05 | 5.45 | 5.90 | 6.19 |
23 | 28 | 5.42 | 5.69 | 5.98 | 6.08 | 6.29 |
24 | 21 | 5.65 | 6.19 | 6.28 | 6.35 | 6.80 |
25 | 12 | 5.36 | 6.04 | 6.29 | 6.66 | 7.05 |
26 | 11 | 6.22 | 6.54 | 6.77 | 6.98 | 7.23 |
27 | 10 | 5.98 | 6.48 | 7.10 | 7.75 | 8.25 |
28 | 12 | 6.48 | 6.62 | 7.65 | 7.85 | 8.62 |
29 | 10 | 7.56 | 7.64 | 8.08 | 8.50 | 9.04 |
30 | 24 | 7.57 | 7.84 | 8.34 | 8.70 | 9.11 |
31 | 40 | 8.06 | 8.45 | 8.69 | 9.00 | 9.40 |
32 | 34 | 8.12 | 8.53 | 9.03 | 9.49 | 9.82 |
33 | 35 | 8.28 | 8.68 | 9.06 | 9.35 | 9.97 |
34 | 18 | 8.60 | 9.08 | 9.50 | 9.92 | 10.47 |
35 | 10 | 9.37 | 9.60 | 9.85 | 11.10 | 11.15 |
36 | 10 | 9.92 | 10.40 | 10.61 | 10.93 | 11.49 |
37 | 10 | 9.95 | 10.33 | 10.90 | 11.28 | 11.84 |
38 | 30 | 10.05 | 10.38 | 10.89 | 11.46 | 12.09 |
39 | 29 | 10.25 | 10.39 | 11.05 | 11.37 | 11.68 |
40 | 12 | 10.32 | 11.15 | 11.43 | 12.15 | 12.30 |
41 | 12 | 10.25 | 11.25 | 12.09 | 12.19 | 12.30 |
(Tamura RK, Sabbagha RE, Pan WH, Vaisrub N: Ultrasonic fetal abdominal circumference: Comparison of direct versus calculated measurement. Obstet Gynecol 67: 833, 1986.Reprinted with permission from The American College of Obstetricians and Gynecologists.)
Normality of fetal development should be assessed in relation to specific cephalic and trunk growth parameters. By placing the fetal BPD or HC and AC in specific centile brackets, nine fetal growth patterns emerge. A fetus classified into growth patterns three or six (normal BPD but small AC) is at high risk for asymmetric IUGR. By contrast, a fetus classified into growth pattern nine is at high risk for symmetric IUGR.32 The centile threshold that best delineates states of normal or altered fetal growth, is still undefined. Nonetheless, preliminary observations suggest that by using the 25th centile as a cut-off point the predictive value of growth patterns three, six, and nine in the diagnosis of IUGR is approximately 80%.
WEIGHT PREDICTING FORMULAS
A number of formulas for prediction of BW are now published in the literature. The predictive accuracy of these formulas ranges from ±14.8% to ±20.2% (2 SD). This accuracy is also related to the size of the fetus. Thus, the relative error is small (approximately 140–200 g) in a fetus weighing 1000 g, and large (480–600 g) in one weighing 4000 g.33, 34
Interestingly, the accuracy of BW formulas may appear small if data are reported in the form of mean % error because the sum of positive and negative differences is taken into account. By contrast, the absolute mean % error reflects the variability noted, regardless of sign and as such is a more accurate predictor of differences from actual BW (Table 9). The format of mean % error can also be misleading because the difference between actual and predicted weights is expressed in small numbers (1–2%) that do not reflect the true variation in BW. For practical clinical purposes the variation between predicted and actual BWs is best expressed in the form of absolute mean % error per 1000 g (see Table 9).
Table 9. A comparison of mean % error versus mean absolute % in reflecting true variation between actual birth weight (A/BW) versus predicted birth weight (P/BW)*
| A/BW − P/BW | % Error | A/BW − P/BW | % Error |
Fetus 1 | −150 g | −15% | 150 g | 15.0% |
Fetus 2 | +140 g | +14% | 140 g | 14.0% |
*Mean % error = −1.0%/2 =−0.5% or −5 g: 1000 g; absolute mean error = 29%/2 = 14.5% or 145 g: 1000 g. It is apparent that the mean % error can be misleading because it artificially reduces the difference between actual and predicted BW. Further, the answer is expressed in small numbers (1–2%) that carry large SDs (7–10%). Expressing the absolute mean error per 1000 g will be the most useful way for the practicing obstestrician.
(Sabbagha RE: Intrauterine growth retardation. In Sabbagha RE [ed]: Diagnostic Ultrasound Applied to Obstetrics and Gynecology, 2nd ed, p 121. Philadelphia, JB Lippincott, 1987)
Comparison of birth weight formulas
A variety of formulas are now used for prediction of fetal weight. Some of these formulas incorporate BPD and AC, whereas others are derived from HC and AC measurements, with or without FL data.
The formula by Shepard and associates is based only on BPD and AC and yields a predictive accuracy of ±20% (2 SD).33, 34 The limiting factors in this formula are the inclusion only of BPD and AC rather than HC, AC, and possibly FL, and the utilization of the same formula for all fetuses, regardless of gestational age and growth status. These limiting factors are discussed below:
- Changes in head shape such as dolichocephaly or brachycephaly can sufficiently change the true estimate of cephalic size by BPD.
- The contributions to BW by head size and body size, at different intervals in pregnancy, are variable. For example, it is known that in the normal preterm fetus the HC:AC ratio exceeds 1, whereas after 36 weeks' gestation the ratio is reversed.35 It is also known that the same ratio is dependent on whether a given fetus is symmetrically or asymmetrically small. Thus, cranial and trunk proportional contributions to BW are dependent on fetal age as well as on the specific state of altered growth.
In support of this reasoning Divon and associates found that in pregnancies complicated by spontaneous rupture of the membranes the estimated fetal weight by BPD and AC or by AC and FL underestimate BW by 12.4% and 13.2%, respectively.36 They attributed this finding to compression of the fetal head and trunk observed in some pregnancies with oligohydramnios. Additionally, Weiner and associates showed that BW in some fetuses with identical BPD and AC values differed by as much as 1000 g and attributed this difference to the two factors previously discussed.37
To avoid the errors introduced by changes in head shape and differences in HC:AC ratios they introduced the concept of targeting BW formulas to specific populations, for example, preterm or IUGR fetuses. Subsequently, in a study involving women at high risk for preterm delivery they derived a formula for prediction of size in low birth weight infants.37 Their formula, shown below, attaches greater significance to HC in these small fetuses.
Predicted BW − 2134.616 + 94.593 HC + 34.227 AC
Data suggest that the prediction of BW in preterm and IUGR fetuses is enhanced by the use of formulas targeted to such populations.37 In high-risk pregnancies knowledge of BW estimates in small fetuses is often urgently needed to help in formulating management plans. (Table 10).
Table 10. Sonographic prediction of fetal weight (200–2200 g)*
Head Circumference (cm) | Abdominal Circumference (cm) | ||||||||||
13.5 | 14.0 | 14.5 | 15.0 | 15.5 | 16.0 | 16.5 | 17.0 | 17.5 | 18.0 | 18.5 | |
13.5 | — | — | — | — | — | — | — | — | — | — | — |
14.0 | — | — | — | — | — | — | — | — | — | — | — |
14.5 | — | — | — | — | — | — | — | — | — | — | — |
15.0 | — | — | — | — | — | — | — | — | — | — | — |
15.5 | — | — | — | — | — | — | — | — | — | — | — |
16.0 | — | — | — | — | — | — | — | — | — | — | — |
16.5 | — | — | — | — | — | — | — | — | — | — | — |
17.0 | — | — | — | — | — | — | — | — | — | — | — |
17.5 | — | — | — | — | — | — | — | — | — | — | — |
18.0 | — | — | — | — | — | — | — | — | — | — | 201.24 |
18.5 | — | — | — | — | — | — | — | — | 214.31 | 231.42 | 248.54 |
19.0 | — | — | — | — | — | 210.27 | 227.38 | 244.49 | 261.61 | 278.72 | 295.83 |
19.5 | — | — | 206.22 | 223.34 | 240.45 | 257.56 | 274.68 | 291.79 | 308.90 | 326.02 | 343.13 |
20.0 | 219.30 | 236.41 | 253.52 | 270.63 | 287.75 | 304.86 | 321.97 | 339.09 | 356.20 | 373.31 | 390.43 |
20.5 | 266.59 | 283.70 | 300.82 | 317.93 | 335.04 | 352.16 | 369.27 | 386.38 | 403.40 | 420.61 | 437.72 |
21.0 | 313.89 | 331.00 | 348.11 | 365.23 | 382.34 | 399.45 | 416.57 | 433.68 | 450.79 | 467.91 | 485.02 |
21.5 | 361.18 | 378.30 | 395.41 | 412.52 | 429.64 | 446.75 | 463.86 | 480.98 | 498.09 | 515.20 | 532.31 |
22.0 | 408.48 | 425.59 | 442.71 | 459.82 | 476.93 | 494.05 | 511.16 | 528.27 | 545.39 | 562.50 | 579.61 |
22.5 | 455.78 | 472.89 | 490.00 | 507.12 | 524.23 | 541.34 | 558.46 | 575.57 | 592.68 | 609.79 | 626.91 |
23.0 | 503.07 | 520.19 | 537.30 | 554.41 | 571.53 | 588.64 | 605.75 | 622.87 | 639.98 | 657.09 | 674.20 |
23.5 | 550.37 | 567.48 | 584.60 | 601.71 | 618.81 | 635.94 | 653.05 | 670.16 | 687.27 | 704.39 | 721.50 |
24.0 | 597.67 | 614.78 | 631.89 | 649.01 | 666.12 | 683.23 | 700.35 | 717.46 | 734.57 | 751.68 | 768.80 |
24.5 | 644.96 | 662.08 | 679.19 | 696.30 | 713.42 | 730.53 | 747.64 | 764.75 | 781.87 | 798.98 | 816.09 |
25.0 | 692.26 | 709.37 | 726.49 | 743.60 | 760.71 | 777.83 | 794.94 | 812.05 | 829.16 | 846.28 | 863.39 |
25.5 | 739.56 | 756.67 | 773.78 | 790.90 | 808.01 | 825.12 | 842.23 | 859.35 | 876.46 | 893.57 | 910.69 |
26.0 | 786.85 | 803.97 | 821.08 | 838.19 | 855.42 | 872.42 | 889.53 | 906.64 | 923.76 | 940.87 | 957.98 |
26.5 | 834.15 | 851.26 | 868.38 | 885.49 | 902.60 | 919.71 | 936.83 | 953.94 | 971.05 | 988.17 | 1005.28 |
27.0 | 881.45 | 898.56 | 915.67 | 932.79 | 949.90 | 967.01 | 984.12 | 1001.24 | 1018.25 | 1035.46 | 1052.58 |
27.5 | 928.74 | 945.86 | 962.97 | 980.08 | 997.19 | 1014.31 | 1031.42 | 1048.53 | 1065.65 | 1082.76 | 1099.87 |
28.0 | 976.04 | 993.15 | 1010.27 | 1027.38 | 1044.49 | 1061.60 | 1078.72 | 1095.83 | 1112.94 | 1130.06 | 1147.17 |
28.5 | 1023.34 | 1040.45 | 1057.56 | 1074.67 | 1091.79 | 1108.90 | 1126.01 | 1143.13 | 1160.24 | 1177.35 | 1194.47 |
29.0 | 1070.63 | 1087.75 | 1104.86 | 1121.97 | 1139.08 | 1156.20 | 1173.31 | 1190.42 | 1207.54 | 1224.65 | 1241.76 |
29.5 | 1117.93 | 1135.04 | 1152.15 | 1169.27 | 1186.38 | 1203.49 | 1120.61 | 1237.72 | 1254.83 | 1271.95 | 1289.06 |
30.0 | 1165.23 | 1182.34 | 1199.45 | 1216.56 | 1233.68 | 1250.79 | 1267.90 | 1285.02 | 1302.13 | 1319.24 | 1336.36 |
30.5 | 1212.52 | 1229.63 | 1246.75 | 1263.86 | 1280.97 | 1298.09 | 1315.20 | 1332.31 | 1349.43 | 1366.54 | 1383.65 |
31.0 | 1259.82 | 1276.93 | 1294.04 | 1311.16 | 1328.27 | 1345.38 | 1362.50 | 1379.61 | 1396.72 | 1413.84 | 1430.95 |
31.5 | 1307.11 | 1324.23 | 1341.34 | 1358.45 | 1375.57 | 1392.68 | 1409.79 | 1426.91 | 1444.02 | 1461.13 | 1478.24 |
32.0 | 1354.41 | 1371.52 | 1388.64 | 1405.75 | 1422.86 | 1439.98 | 1457.09 | 1474.20 | 1491.32 | 1508.43 | 1525.54 |
32.5 | 1401.71 | 1418.82 | 1435.93 | 1453.05 | 1470.16 | 1487.27 | 1504.39 | 1521.50 | 1538.61 | 1555.72 | 1572.84 |
33.0 | 1449.00 | 1466.12 | 1483.23 | 1500.34 | 1517.46 | 1534.57 | 1551.68 | 1568.80 | 1585.91 | 1603.02 | 1620.13 |
33.5 | 1496.30 | 1513.41 | 1530.53 | 1547.64 | 1564.75 | 1581.87 | 1598.98 | 1616.09 | 1633.20 | 1650.32 | 1667.43 |
34.0 | 1543.60 | 1560.71 | 1577.82 | 1594.94 | 1612.05 | 1629.16 | 1646.28 | 1663.39 | 1680.50 | 1697.61 | 1714.73 |
Table 10. Sonographic prediction of fetal weight (200–2200g)* (Continued)
Head Circumference (cm) | Abdominal Circumference (cm) | ||||||||||
19.0 | 19.5 | 20.0 | 20.5 | 21.0 | 21.5 | 22.0 | 22.5 | 23.0 | 23.5 | 24.0 | |
13.5 | - | - | - | - | - | - | - | - | - | - | - |
14.0 | - | - | - | - | - | - | - | - | - | - | - |
14.5 | - | - | - | - | - | - | - | - | - | - | - |
15.0 | - | - | - | - | - | - | - | - | - | - | - |
15.5 | - | - | - | - | - | - | - | - | - | - | - |
16.0 | - | - | - | - | - | - | - | - | - | - | 200.30 |
16.5 | - | - | - | - | - | - | - | - | 213.37 | 230.48 | 247.59 |
17.0 | - | - | - | - | - | 209.32 | 226.44 | 243.55 | 260.66 | 277.78 | 294.89 |
17.5 | - | - | 205.28 | 222.39 | 239.51 | 256.62 | 273.73 | 290.85 | 307.96 | 325.05 | 342.19 |
18.0 | 218.35 | 235.47 | 252.28 | 269.69 | 286.80 | 303.92 | 321.03 | 338.14 | 355.26 | 372.37 | 389.48 |
18.5 | 265.65 | 282.76 | 299.87 | 316.99 | 334.10 | 351.21 | 368.33 | 385.44 | 402.55 | 419.67 | 436.78 |
19.0 | 312.95 | 330.06 | 347.17 | 364.28 | 381.40 | 398.51 | 415.62 | 432.74 | 449.85 | 466.96 | 484.08 |
19.5 | 360.24 | 377.35 | 394.47 | 411.58 | 428.69 | 445.81 | 462.92 | 480.03 | 497.15 | 514.26 | 531.37 |
20.0 | 407.54 | 424.65 | 441.76 | 458.88 | 475.99 | 493.10 | 510.22 | 527.33 | 544.44 | 561.56 | 578.67 |
20.5 | 454.83 | 471.95 | 489.06 | 506.17 | 523.29 | 540.40 | 557.51 | 574.63 | 591.74 | 608.85 | 625.96 |
21.0 | 502.13 | 519.24 | 536.36 | 553.47 | 570.58 | 587.70 | 604.81 | 621.92 | 639.04 | 656.15 | 673.26 |
21.5 | 549.43 | 566.54 | 583.65 | 600.77 | 617.88 | 634.99 | 652.11 | 669.22 | 686.33 | 703.44 | 720.56 |
22.0 | 596.72 | 613.84 | 630.95 | 648.06 | 665.18 | 682.29 | 699.40 | 716.52 | 733.63 | 750.74 | 767.85 |
22.5 | 644.02 | 661.13 | 678.25 | 695.36 | 712.47 | 729.59 | 746.70 | 763.81 | 780.92 | 798.04 | 815.15 |
23.0 | 691.32 | 708.43 | 725.54 | 742.66 | 759.77 | 776.88 | 794.00 | 811.11 | 828.22 | 845.33 | 862.45 |
23.5 | 738.61 | 755.73 | 772.84 | 789.95 | 807.07 | 824.18 | 841.29 | 858.40 | 875.52 | 892.63 | 909.74 |
24.0 | 785.91 | 803.02 | 820.14 | 837.25 | 854.36 | 871.48 | 888.59 | 905.70 | 922.81 | 939.93 | 957.04 |
24.5 | 833.21 | 850.32 | 867.43 | 884.55 | 901.66 | 918.77 | 935.88 | 953.00 | 970.11 | 987.22 | 1004.34 |
25.0 | 880.50 | 897.62 | 914.73 | 931.84 | 948.96 | 966.07 | 983.18 | 1000.29 | 1017.41 | 1034.52 | 1051.63 |
25.5 | 927.80 | 944.91 | 962.03 | 979.14 | 996.25 | 1013.36 | 1030.48 | 1047.59 | 1064.70 | 1081.82 | 1098.93 |
26.0 | 975.10 | 992.21 | 1009.32 | 1026.44 | 1043.55 | 1060.66 | 1077.77 | 1094.89 | 1112.00 | 1129.11 | 1146.23 |
26.5 | 1022.39 | 1039.51 | 1056.62 | 1073.73 | 1090.84 | 1107.96 | 1125.07 | 1142.18 | 1159.30 | 1176.41 | 1193.52 |
27.0 | 1069.69 | 1086.80 | 1103.92 | 1121.03 | 1138.14 | 1155.25 | 1172.37 | 1189.48 | 1206.59 | 1223.71 | 1240.82 |
27.5 | 1116.99 | 1134.10 | 1151.21 | 1168.32 | 1185.44 | 1202.55 | 1219.66 | 1236.78 | 1253.89 | 1271.00 | 1288.12 |
28.0 | 1164.28 | 1181.39 | 1198.51 | 1215.62 | 1232.73 | 1249.85 | 1266.96 | 1284.07 | 1301.19 | 1318.30 | 1335.41 |
28.5 | 1211.58 | 1228.69 | 1245.80 | 1262.92 | 1280.03 | 1297.14 | 1314.26 | 1331.37 | 1348.48 | 1365.60 | 1382.71 |
29.0 | 1258.88 | 1275.99 | 1293.10 | 1310.21 | 1327.33 | 1344.44 | 1361.55 | 1378.67 | 1395.78 | 1412.89 | 1430.01 |
29.5 | 1306.17 | 1323.28 | 1340.40 | 1357.51 | 1374.62 | 1391.74 | 1408.85 | 1425.96 | 1443.08 | 1460.19 | 1477.30 |
30.0 | 1353.47 | 1370.58 | 1387.69 | 1404.81 | 1421.92 | 1439.03 | 1456.15 | 1473.26 | 1490.37 | 1507.49 | 1524.60 |
30.5 | 1400.76 | 1417.88 | 1434.99 | 1452.10 | 1469.22 | 1486.33 | 1503.44 | 1520.56 | 1537.67 | 1554.78 | 1571.89 |
31.0 | 1448.06 | 1465.17 | 1482.29 | 1499.40 | 1516.51 | 1533.63 | 1550.74 | 1567.85 | 1584.97 | 1602.08 | 1619.19 |
31.5 | 1495.36 | 1512.47 | 1529.58 | 1546.70 | 1563.81 | 1580.92 | 1598.04 | 1615.15 | 1632.26 | 1649.37 | 1666.49 |
32.0 | 1542.65 | 1559.77 | 1576.88 | 1593.99 | 1611.11 | 1628.22 | 1645.33 | 1662.45 | 1679.56 | 1696.67 | 1713.78 |
32.5 | 1589.95 | 1607.06 | 1624.18 | 1641.29 | 1658.40 | 1675.52 | 1692.63 | 1709.74 | 1726.85 | 1743.97 | 1761.00 |
33.0 | 1637.25 | 1654.36 | 1671.47 | 1688.59 | 1705.70 | 1722.81 | 1739.93 | 1757.04 | 1774.15 | 1791.26 | 1808.38 |
33.5 | 1684.54 | 1701.66 | 1718.77 | 1735.88 | 1753.00 | 1770.11 | 1787.22 | 1804.33 | 1821.45 | 1838.56 | 1855.67 |
34.0 | 1731.84 | 1748.95 | 1766.07 | 1783.18 | 1800.29 | 1817.41 | 1834.52 | 1851.63 | 1868.74 | 1885.96 | 1902.97 |
Table 10. Sonographic prediction of fetal weight (200–2200g)* (Continued)
Head Circumference (cm) | Abdominal Circumference (cm) | |||||||||
24.5 | 25.0 | 25.5 | 26.0 | 26.5 | 27.0 | 27.5 | 28.0 | 28.5 | 29.0 | |
13.5 | - | - | - | - | - | - | - | - | - | - |
14.0 | - | - | - | - | - | - | - | - | - | - |
14.5 | - | - | - | - | - | - | - | - | 212.42 | 229.54 |
15.0 | - | - | - | - | - | 208.38 | 225.49 | 242.61 | 259.72 | 276.83 |
15.5 | - | - | 204.34 | 221.45 | 238.56 | 255.68 | 272.79 | 289.90 | 307.02 | 324.13 |
16.0 | 217.41 | 234.52 | 251.64 | 268.75 | 285.86 | 302.97 | 320.09 | 337.20 | 354.31 | 371.43 |
16.5 | 264.71 | 281.82 | 298.93 | 316.04 | 333.16 | 350.27 | 367.38 | 384.50 | 401.61 | 418.71 |
17.0 | 312.00 | 329.12 | 346.23 | 363.34 | 380.45 | 397.57 | 414.68 | 431.79 | 448.91 | 466.02 |
17.5 | 359.30 | 376.41 | 393.52 | 410.64 | 427.75 | 444.86 | 461.98 | 479.09 | 496.20 | 513.32 |
18.0 | 406.60 | 423.71 | 440.82 | 457.93 | 475.05 | 492.16 | 509.27 | 526.39 | 543.50 | 560.61 |
18.5 | 453.89 | 471.00 | 488.12 | 505.23 | 522.34 | 539.46 | 556.57 | 573.68 | 590.80 | 607.91 |
19.0 | 501.19 | 518.30 | 535.41 | 552.53 | 569.64 | 586.75 | 603.87 | 620.98 | 638.09 | 655.21 |
19.5 | 548.48 | 565.60 | 582.71 | 599.82 | 616.94 | 634.05 | 651.16 | 668.28 | 685.39 | 702.50 |
20.0 | 595.78 | 612.89 | 630.01 | 647.12 | 664.23 | 681.35 | 698.46 | 715.57 | 732.69 | 749.80 |
20.5 | 643.08 | 660.19 | 677.30 | 694.42 | 711.53 | 728.64 | 745.76 | 762.87 | 779.98 | 797.09 |
21.0 | 690.37 | 707.49 | 724.60 | 741.71 | 758.83 | 775.94 | 793.05 | 810.17 | 827.28 | 844.39 |
21.5 | 737.67 | 754.78 | 771.90 | 789.01 | 806.12 | 823.24 | 840.35 | 857.46 | 874.57 | 891.69 |
22.0 | 784.97 | 802.08 | 819.19 | 836.31 | 853.42 | 870.53 | 887.65 | 904.76 | 921.87 | 938.98 |
22.5 | 832.26 | 849.38 | 866.49 | 883.60 | 900.72 | 917.83 | 934.94 | 952.05 | 969.17 | 986.28 |
23.0 | 879.56 | 896.67 | 913.79 | 930.90 | 948.01 | 965.13 | 982.24 | 999.35 | 1016.46 | 1033.58 |
23.5 | 926.86 | 943.97 | 961.08 | 978.20 | 995.31 | 1012.42 | 1029.53 | 1046.65 | 1063.76 | 1080.87 |
24.0 | 974.15 | 991.27 | 1008.38 | 1025.49 | 1042.61 | 1059.72 | 1076.83 | 1093.94 | 1111.06 | 1128.17 |
24.5 | 1021.45 | 1038.56 | 1055.68 | 1072.79 | 1089.90 | 1107.01 | 1124.13 | 1141.24 | 1158.35 | 1175.47 |
25.0 | 1068.75 | 1085.86 | 1102.97 | 1120.09 | 1137.20 | 1154.31 | 1171.42 | 1188.54 | 1205.65 | 1222.76 |
25.5 | 1116.04 | 1133.16 | 1150.27 | 1167.38 | 1184.49 | 201.61 | 1218.72 | 1235.83 | 1252.95 | 1270.06 |
26.0 | 1163.34 | 1180.45 | 1197.57 | 1214.68 | 1231.79 | 1248.90 | 1266.02 | 1283.13 | 1300.24 | 1317.36 |
26.5 | 1210.64 | 1227.75 | 1244.86 | 1261.97 | 1279.09 | 1296.20 | 1313.31 | 1330.43 | 1347.54 | 1364.65 |
27.0 | 1257.93 | 1275.05 | 1292.16 | 1309.27 | 1326.38 | 1343.50 | 1360.81 | 1377.72 | 1394.84 | 1411.95 |
27.5 | 1305.23 | 1322.34 | 1339.45 | 1356.57 | 1373.68 | 1390.79 | 1407.91 | 1425.02 | 1442.13 | 1459.25 |
28.0 | 1352.53 | 1369.64 | 1386.75 | 1403.86 | 1420.98 | 1438.09 | 1455.20 | 1472.32 | 1489.43 | 1506.54 |
28.5 | 1399.82 | 1416.93 | 1434.05 | 1451.16 | 1468.27 | 1485.39 | 1502.20 | 1519.61 | 1536.73 | 1553.84 |
29.0 | 1447.12 | 1464.23 | 1481.34 | 1498.46 | 1515.57 | 1532.68 | 1549.80 | 1566.91 | 1584.02 | 1601.13 |
29.5 | 1494.41 | 1511.53 | 1528.64 | 1545.75 | 1562.87 | 1579.98 | 1597.09 | 1614.21 | 1631.32 | 1648.43 |
30.0 | 1541.71 | 1558.82 | 1575.94 | 1593.05 | 1610.16 | 1627.28 | 1644.39 | 1661.50 | 1678.62 | 1695.73 |
30.5 | 1589.01 | 1606.12 | 1623.23 | 1640.35 | 1657.46 | 1674.57 | 1691.69 | 1708.80 | 1725.91 | 1743.02 |
31.0 | 1636.30 | 1653.42 | 1670.53 | 1687.64 | 1704.76 | 1721.87 | 1738.98 | 1756.10 | 1773.21 | 1790.32 |
31.5 | 1683.60 | 1700.71 | 1717.83 | 1734.94 | 1752.05 | 1769.17 | 1786.28 | 1803.39 | 1820.50 | 1837.62 |
32.0 | 1730.90 | 1748.01 | 1765.12 | 1782.24 | 1799.35 | 1816.46 | 1833.58 | 1850.69 | 1867.80 | 1884.91 |
32.5 | 1778.19 | 1795.31 | 1812.42 | 1829.53 | 1846.65 | 1863.76 | 1880.87 | 1897.98 | 1915.10 | 1932.21 |
33.0 | 1825.49 | 1842.60 | 1859.72 | 1876.83 | 1893.94 | 1911.06 | 1928.17 | 1945.28 | 1962.39 | 1979.51 |
33.5 | 1872.79 | 1889.90 | 1907.01 | 1924.13 | 1941.24 | 1958.35 | 1975.46 | 1992.58 | 2009.69 | 2026.80 |
34.0 | 1920.08 | 1937.20 | 1954.31 | 1991.42 | 1988.54 | 2005.76 | 2022.65 | 2039.87 | 2056.99 | 2074.10 |
Table 10. Sonographic prediction of fetal weight (200–2200g)* (Continued)
Head Circumference (cm) | Abdominal Circumference (cm) | |||||||||
29.5 | 30.0 | 30.5 | 31.0 | 31.5 | 32.0 | 32.5 | 33.0 | 33.5 | 34.0 | |
13.5 | - | - | - | 203.40 | 220.51 | 237.62 | 254.73 | 271.85 | 288.96 | 306.07 |
14.0 | - | 216.47 | 233.58 | 250.69 | 267.81 | 284.92 | 302.03 | 319.14 | 336.26 | 353.37 |
14.5 | 246.65 | 263.76 | 280.88 | 297.99 | 315.10 | 332.21 | 349.33 | 366.44 | 383.55 | 400.67 |
15.0 | 293.95 | 311.06 | 328.17 | 345.29 | 362.40 | 379.51 | 396.62 | 413.74 | 430.85 | 447.96 |
15.5 | 341.24 | 358.36 | 375.47 | 392.58 | 409.69 | 426.81 | 443.92 | 461.03 | 478.15 | 495.26 |
16.0 | 388.54 | 405.65 | 422.77 | 439.88 | 456.99 | 474.10 | 491.22 | 508.33 | 525.44 | 542.56 |
16.5 | 435.84 | 452.95 | 470.06 | 487.17 | 504.29 | 521.40 | 538.51 | 555.63 | 572.74 | 589.95 |
17.0 | 483.13 | 500.25 | 517.36 | 534.47 | 551.58 | 568.70 | 585.81 | 602.92 | 620.04 | 637.15 |
17.5 | 530.43 | 547.54 | 564.65 | 581.77 | 598.88 | 615.99 | 633.11 | 650.22 | 667.33 | 684.45 |
18.0 | 577.73 | 594.84 | 611.95 | 629.06 | 646.18 | 663.29 | 680.40 | 697.52 | 714.63 | 731.74 |
18.5 | 625.02 | 642.13 | 659.25 | 676.36 | 693.47 | 710.59 | 727.70 | 744.81 | 761.93 | 779.04 |
19.0 | 672.32 | 689.43 | 706.54 | 723.66 | 740.77 | 757.88 | 775.00 | 792.11 | 809.22 | 826.34 |
19.5 | 719.61 | 736.73 | 753.84 | 770.95 | 788.07 | 805.18 | 822.29 | 839.41 | 856.52 | 873.63 |
20.0 | 766.91 | 784.02 | 801.14 | 818.25 | 835.36 | 852.48 | 869.59 | 886.70 | 903.82 | 920.93 |
20.5 | 814.21 | 831.32 | 848.43 | 865.55 | 882.66 | 899.77 | 916.89 | 934.00 | 951.11 | 968.22 |
21.0 | 861.50 | 878.62 | 895.73 | 912.84 | 929.96 | 947.07 | 964.18 | 981.30 | 998.41 | 1015.52 |
21.5 | 908.80 | 925.91 | 943.03 | 960.14 | 977.25 | 994.37 | 1011.48 | 1028.59 | 1045.70 | 1062.82 |
22.0 | 956.10 | 973.21 | 990.32 | 1007.44 | 1024.55 | 1041.66 | 1058.78 | 1075.89 | 1093.00 | 1110.11 |
22.5 | 1003.39 | 1020.51 | 1037.62 | 1054.73 | 1071.85 | 1088.96 | 1106.07 | 1123.18 | 1140.30 | 1157.41 |
23.0 | 1050.69 | 1067.80 | 1084.92 | 1102.03 | 1119.14 | 1136.26 | 1153.37 | 1170.48 | 1187.59 | 1204.71 |
23.5 | 1097.99 | 1115.10 | 1132.21 | 1149.33 | 1166.44 | 1183.55 | 1200.66 | 1217.78 | 1234.89 | 1252.00 |
24.0 | 1145.28 | 1162.40 | 1179.51 | 1196.62 | 1213.74 | 1230.85 | 1247.96 | 1265.07 | 1282.19 | 1299.30 |
24.5 | 1192.58 | 1209.69 | 1226.81 | 1243.92 | 1261.03 | 1278.14 | 1295.26 | 1312.37 | 1329.48 | 1346.60 |
25.0 | 1239.88 | 1256.99 | 1274.10 | 1291.22 | 1308.33 | 1325.44 | 1342.55 | 1359.67 | 1376.78 | 1393.89 |
25.5 | 1287.17 | 1304.29 | 1321.40 | 1338.51 | 1355.62 | 1372.74 | 1389.85 | 1406.96 | 1424.08 | 1441.19 |
26.0 | 1334.47 | 1351.58 | 1368.70 | 1385.81 | 1402.92 | 1420.03 | 1437.15 | 1454.26 | 1471.37 | 1488.49 |
26.5 | 1381.77 | 1398.88 | 1415.99 | 1433.10 | 1450.22 | 1467.33 | 1484.44 | 1501.56 | 1518.67 | 1535.78 |
27.0 | 1429.06 | 1446.18 | 1463.29 | 1480.40 | 1497.51 | 1514.63 | 1531.74 | 1548.85 | 1565.97 | 1583.08 |
27.5 | 1476.36 | 1493.47 | 1510.58 | 1527.70 | 1544.81 | 1561.92 | 1579.04 | 1596.15 | 1613.26 | 1630.38 |
28.0 | 1523.66 | 1540.77 | 1557.88 | 1574.99 | 1592.11 | 1609.22 | 1626.33 | 1643.45 | 1660.56 | 1677.67 |
28.5 | 1570.95 | 1588.06 | 1605.18 | 1622.29 | 1639.40 | 1656.52 | 1673.63 | 1690.74 | 1707.86 | 1724.97 |
29.0 | 1618.25 | 1635.36 | 1652.47 | 1669.59 | 1686.70 | 1703.81 | 1720.93 | 1738.04 | 1755.15 | 1772.27 |
29.5 | 1665.54 | 1682.66 | 1699.77 | 1716.88 | 1734.00 | 1751.11 | 1768.22 | 1785.34 | 1802.45 | 1819.56 |
30.0 | 1712.84 | 1729.95 | 1747.07 | 1764.18 | 1781.29 | 1798.41 | 1815.52 | 1832.63 | 1849.75 | 1866.86 |
30.5 | 1760.14 | 1777.25 | 1794.36 | 1811.48 | 1828.59 | 1845.70 | 1862.82 | 1879.93 | 1897.04 | 1914.15 |
31.0 | 1807.43 | 1824.55 | 1841.66 | 1858.77 | 1875.89 | 1893.00 | 1910.11 | 1927.23 | 1944.34 | 1961.45 |
31.5 | 1854.73 | 1871.84 | 1888.96 | 1906.07 | 1923.18 | 1940.30 | 1957.41 | 1974.52 | 1991.63 | 2008.75 |
32.0 | 1902.03 | 1919.14 | 1936.25 | 1953.37 | 1970.48 | 1987.59 | 2004.71 | 2021.82 | 2038.93 | 2056.04 |
32.5 | 1949.32 | 1966.44 | 1983.85 | 2000.66 | 2017.78 | 2034.89 | 2052.00 | 2069.11 | 2086.23 | 2103.34 |
33.0 | 1996.62 | 2013.73 | 2030.85 | 2047.96 | 2065.07 | 2082.18 | 2099.30 | 2116.41 | 2133.52 | 2150.64 |
33.5 | 2043.92 | 2061.03 | 2078.14 | 2095.26 | 2112.37 | 2129.48 | 2146.59 | 2163.71 | 2180.82 | 2197.93 |
34.0 | 2091.21 | 2108.33 | 2125.44 | 2142.55 | 2159.66 | 2176.78 | 2193.89 | 2211.00 | 2228.12 | 2245.23 |
*Based on P wt = 94.593 (HC) + 34.226 (AC) − 2134.616
Fetus's with femur length greater than or less than 2 SDs from norm should be corrected by (FFL/MFL) (P wt)
(Sabbagha RE: Intrauterine growth retardation. In Sabbagha RE [ed]: Diagnostic Ultrasound Applied to Obstetrics and Gynecology, 2nd ed, p 122. Philadelphia, JB Lippincott, 1987)
Hadlock and associates derived from the following formula for prediction of BW from HC, AC, and FL:34
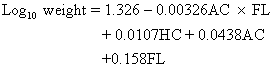
They recommended utilization of this formula for all birth weight categories because the range in accuracy is relatively small, namely ± 14.8% (2 SD). Their proposal seems reasonable if fetuses falling in the BW category <1500 g are excluded. In the latter group, the accuracy of BW prediction reported in their study deteriorates to + 19.4% (2 SD)34 and use of a “targeted” approach is more appropriate.37
Of importance, by using the same formula in 361 normal fetuses, Hadlock and associates showed that the shape of the ultrasonically derived weight curve was identical to that derived by Williams and associates, based on approximately 2 million newborns.34, 38, 36, 37, 39 Previously, Ott and Doyle suggested that the two curves (ultrasonic vs actual BWs) were different.40 However, their conclusion was based on BWs derived by the less accurate formula of Shepard and associates.
The improvement in the prediction of BW by Hadlock's formula may be mainly related to the use of the HC rather than HC and FL. The reason for this is that other investigators, notably Woo and associates and Weiner and associates, found no significant benefit to routine inclusion of the femur length.37, 41 This observation seems contrary to the previously presented rationale of incorporating fetal length in the estimation of fetal or neonatal size. However, closer examination shows that routine inclusion of FL dilutes any change or effect resulting from long or short fetuses because the majority are of average length. In fact, Weiner and associates showed that the variation in BW can be reduced by incorporating the FL selectively, that is, only in long or short fetuses, those differing from the mean by 2 SD.37
Routine use of birth weight prediction
Routine prediction of BW by formula is not necessary and can be misleading. The difference between actual and predicted BWs (2 SD = 15%) is large enough to produce false-positive and false-negative diagnoses of IUGR. For example, a fetus weighing 3000 g may be falsely assumed to be growth retarded, whereas a small 2500 g fetus may be assumed to be normal.
Antenatal prediction of BW has proven to be especially useful in the management of women at high risk of premature delivery. The reasons include
1. Imprecision in the BW estimates by the history and clinical examination. For example, Paul and associates showed that 35% of preterm infants clinically judged to be less than 1000 g, in fact exceeded that weight.42
2. Improvement in the perinatal outcome of very low birth weight babies; this is attributed to the advent of technologically sophisticated neonatal intensive care facilities and the management of high-risk pregnancies at tertiary care centers. The most dramatic improvement is witnessed in infants weighing less than 1000 g but not necessarily those under 750 g.43, 44
Thus, accurate estimation of BW in the range of 750–1000 g is important because when equipped with this information, the physician managing labor will pursue sound obstetric principles directed at the prevention of asphyxia, a step essential for effective intensive neonatal care.
Oligohydramnios
Manning and associates and Chamberlain and associates found that the presence of oligohydramnios was predictive of IUGR with an accuracy approaching 89.6% and 78.9%, respectively.45, 46 However, the pregnancies studied were close to term and were at high risk for IUGR, by previous ultrasound or clinical data or both. In their studies oligohydramnios was diagnosed when only a single pocket of amniotic fluid, approximately 1– 2 cm in size, was noted in a plane vertical to the uterine surface.
Using the “1-cm rule” Hoddick and associates, in a retrospective study, showed that oligohydramnios accurately predicted only 4/52 or 8% of IUGR fetuses.47 In other words the sensitivity of the test was poor.
In a large unselected population of pregnant women Philipson and associates reported that the incidence of oligohydramnios was 3.9% and of those only 40% were growth retarded.48 However, when projected to the total population the sensitivity of the finding of oligohydramnios was only 16% (see Table 4);48 this level of sensitivity was also confirmed by Chamberlain and associates.46 In conducting their study Philipson and associates diagnosed oligohydramnios by careful subjective assessment of overall amniotic fluid volume, rather than by strict adherence to the 1-cm or 2-cm “rule”.
The difference observed in the three studies can be explained by the number of high-risk pregnancies (prevalence of IUGR) in each group. It is clear that oligohydramnios is more predictive of poor outcome in pregnancies at high risk for IUGR. This association between oligohydramnios and high-risk pregnancies is further illustrated by reference to the entity of post-date pregnancies. A retrospective study of such pregnancies by Phelan and associates showed that amniotic fluid was marginal (>1 cm, but subjectively decreased) and decreased (<1 cm) in 38 and seven patients, respectively.49 In these two groups, antenatal fetal heart tests showed decelerations and bradycardia in 44.7% and 71%, respectively. By comparison, fetal heart rate tests were abnormal in 21% of patients with normal amniotic fluid volume. In the presence of oligohydramnios the overall incidence of late decelerations requiring cesarean section was high (5.25% and 16.7%) when compared to the group with normal fluid (1.05%).
The authors also found that only 14% of post-date pregnancies with oligohydramnios were delivered of an infant with the stigmata of post-maturity, including meconium-stained fluid. Nonetheless, the authors recommended that post-date pregnancies with oligohydramnios should be considered for trial of labor with continuous electronic fetal monitoring.
OTHER ADJUNCTIVE TESTS
A number of indices, ratios, and volume calculations have been derived from various fetal structures and subsequently tested with regards to accuracy in the diagnosis of IUGR. These include ponderal index (PI), HC:AC ratio, total intrauterine volume (TIUV) measurements, and placental grade.
Ponderal index
Neonate pediatricians use the ponderal index (BW/CHL3) to determine whether some infants with BWs >10th centile are fetally malnourished or undergrown, relative to their length.23 In the fetus, Hadlock and associates and Vintzileos and associates derived a fetal PI from in utero mean estimates of FL and AC.50, 51 They based their premise on the fact that fetal AC is related to BW and the femur length to the CHL in the neonate.25, 26 This formula for PI is the reverse of the neonatal ponderal index, and calculates the FL:AC ratio rather than the AC:FL. The latter PI may be more appropriate because it may be compared to that of the neonate (Table 11). In any event the mean PI for FL:AC ratio was reported to be 22.3 ± 2.4 (mean ± 2 SD) and did not vary with advancing gestational age.50
Table 11. Fetal ponderal index calculations based on abdominal circumference (AC)* and femur length (FL) 50th centile values at different weeks (WK) of pregnancy
WK | AC | FL | FL3 | AC:FL | AC × 100 FL3 |
26 | 22.1 | 4.75 | 107.17 | 4.65 | 20.6 |
27 | 23.7 | 5.00 | 125.0 | 4.74 | 18.9 |
28 | 25.3 | 5.25 | 144.7 | 4.82 | 17.5 |
29 | 26.9 | 5.45 | 161.8 | 4.82 | 16.6 |
30 | 27.4 | 5.75 | 190.1 | 4.76 | 14.4 |
31 | 28.0 | 5.95 | 210.6 | 4.70 | 13.3 |
32 | 28.7 | 6.2 | 238.0 | 4.62 | 12.0 |
33 | 29.0 | 6.30 | 325.0 | 4.60 | 11.6 |
34 | 30.01 | 6.50 | 274.6 | 4.61 | 10.9 |
35 | 32.2 | 6.7 | 300.7 | 4.80 | 10.7 |
36 | 33.3 | 6.8 | 314.4 | 4.89 | 10.6 |
37 | 34.4 | 7.0 | 343.0 | 4.91 | 10.0 |
38 | 35.7 | 7.1 | 357.9 | 5.02 | 10.0 |
39 | 35.9 | 7.3 | 389.0 | 4.91 | 9.2 |
40 | 36.1 | 7.4 | 405.2 | 4.87 | 8.9 |
*The mean AC:FL ratio is 4.78 and varies only slightly (4.60–5.02) with advancing gestation. By contrast the AC × 100/FL3 decreases with advancing gestation and varies considerably from 8.9 to 20.6.
(Sabbagha RE: Intrauterine growth retardation. In Sabbagha RE [ed]: Diagnostic Ultrasound Applied to Obstetrics and Gynecology, 2nd ed, p 127. Philadelphia, JB Lippincott, 1987)
Conceptually, a low PI in the fetus (AC:FL <10th centile) may be diagnostically useful only in a small proportion of asymmetric IUGR fetuses, those in whom the AC is small but the FL is long. In other words, most of the asymmetrically undergrown infants will have normal PIs. Similarly, in symmetric IUGR, PIs are normal.
To test the procedure accuracy of in utero PIs, Vintzileos and associates compared fetal and neonatal PI data in 113 pregnancies.52 In their study, fetal weight was estimated by BPD and AC, using the model by Shepard and associates, and length was derived by the following formula,53 namely
Fetal length = 6.18 + 5.9 x femur length
Ponderal indices in the fetus and neonate were calculated by the standard formula
Ponderal index = [Birth weight/(crown–heel length)3] x 100
The results showed a weak correlation (r = 0.364) between fetal and neonatal PIs. The predictive value of the fetal PI in the diagnosis of asymmetric IUGR neonates with PIs <10th centile was also low (Table 12). The sensitivity of the test was better (76.9%). However, the prevalence of IUGR neonates with low PIs in the study was high (see Table 12). As a result, the sensitivity of the test in the general population will be lower.
Table 12. Predictive value of fetal ponderal index (PI) in the diagnosis of infants with asymmetric growth restriction and ponderal indexes below the 10th centile
Fetal PI | Neonatal Ponderal Index | ||
<10th Centile | >10th Centile | Total | |
<10th centile | 10 | 18 | 28 |
>10th centile | 3 | 82 | 85 |
Total | 13 | 100 | 113 |
The predictive value of fetal PI in the diagnosis of infants with a neonatal PI <10th centile is 10/28 or 35%.The sensitivity is 10/13 or 76.9%. The predictive value of normal PI is high: 82/85 or 96.4%.
(Adapted from Vintzileos AM, Lodeiro JG, Feinstein SJ et al: Value of fetal ponderal index in predicting growth retardation. Obstet Gynecol 67: 584, 1986. Reprinted with permission from The American College of Obstetricians and Gynecologists.)
The fetal PI was also not useful in the prediction of macrosomia. Although Benson and associates showed that the mean values of FL:AC ratios were significantly different from those of normal fetuses, the predictive value of the PI “test” in the diagnosis of macrosomia was poor (36–42%), and only slightly greater than the prevalence of the condition (26%) in their study.54
The low positive predictive value of fetal PI is related to the use of imprecise BW formulas (± 2 SD error of 15–22%) and fetal CHL estimates, an approach that will only compound the variation inherent in AC and FL measurements. To avoid false-positive results the normality of AC and FL should be assessed by comparing these measurements directly to their respective growth centiles, as discussed elsewhere.
Total intrauterine volume
Gohari and associates proposed sonar measurement of total intrauterine volume (TIUV) for predicting IUGR.55 This is an indirect method of estimating fetal size from sagittal and transverse echograms of the pregnant uterus, views that can only be obtained using static image scanners. Chinn and associates showed that the positive predictive value of TIUV was poor.56 Grossman and associates questioned the reproducibility of the measurement on the basis that appropriate landmarks were difficult to identify and varied relative to bladder filling.57
Head circumference:abdominal circumference ratio
Campbell and Thoms showed that in approximately 70% of asymmetrically undergrown fetuses the head circumference:abdominal circumference (HC:AC) ratio was >2 SD above the mean (Table 13).35 Thus, the ratio can be used as an adjunctive test to further substantiate asymmetric IUGR. The probability of an accurate diagnosis (70%) may be enhanced if it is used in conjunction with abnormal growth patterns of the head and trunk.
Table 13. Mean head circumference:abdomen circumference ratio versus gestational age (GA)
GA (Weeks) | Mean HC:AC Ratio |
+2 SD |
28 | 1.13 | 1.21 |
32 | 1.075 | 1.17 |
34 | 1.04 | 1.13 |
36 | 1.02 | 1.12 |
38 | 0.99 | 1.06 |
40 | 0.97 | 1.05 |
(Campbell S, Thoms A: Ultrasound measurement of fetal head to abdomen circumference ratio in the assessment of growth retardation. Br J Obstet Gynaecol 84: 165, 1977.)
It should be remembered that the HC:AC ratio is poor in detecting symmetric IUGR because all fetal dimensions are reduced. Further, its use is dependent on clear definition of gestational age because the HC:AC ratio is normally high in preterm fetuses.35
ANTENATAL MANAGEMENT OF INTRAUTERINE GROWTH RETARDATION
As stated above, the physician using ultrasound antenatally can identify a population of fetuses at high risk for symmetric or asymmetric IUGR. Once this is accomplished, it becomes feasible, cost-effective, and mandatory to monitor this high-risk group more intensively. In this way, the most appropriate time for delivering the compromised fetus can be determined, prior to the onset of hypoxia, asphyxia or intrauterine fetal death. The bioelectric tests used in fetal heart rate monitoring include the nonstress test (NST), and the contraction stress test (CST).
Nonstress test
In the NST fetal heart rate (FHR) accelerations occurring in response to fetal movement are recorded on a monitor strip. The NST is referred to as an external FHR monitoring strip. This is because the transducer is placed on the maternal abdomen rather than on the fetus.
A normal or reactive test depends on detection of at least two FHR accelerations, 15 beats per minute (bpm) above baseline, in a 20-minute test period.58 Other indicators of well-being include variability in the FHR, a FHR baseline between 120 bpm and 160 bpm and the absence of repetitive FHR decelerations.58
A nonreactive NST is one in which FHR accelerations, above the baseline, are less than 15 bpm. The presence of repetitive spontaneous decelerations with or without contractions may also be associated with adverse outcome.58 In addition, the loss of variability is significant. The reason is that a flat baseline (absence of variability) cannot be artificially produced by ultrasound and may be associated with the fetal hypoxic state.
A reactive NST is predictive of good outcome (absence of asphyxia) for a period of 1 week in 62–89% of cases. However, nonreactive tests are predictive of fetal death (within 1 week of testing) in approximately 3–29% of cases.56 Thus, an abnormal NST should be followed by other tests to assess for fetal well-being.
Contraction stress test
The CST is performed by administering intravenous (IV) Pitocin, in a controlled fashion to the pregnant woman. The IV drip is gradually increased until three uterine contractions are registered within a 10-minute interval. Uterine contractions are known to reduce intervillous blood flow but not to the extent of altering normal FHR patterns.59 On the other hand, a compromised fetus will not tolerate the “stress” of uterine contractions. As a result fetal myocardial hypoxia develops. This pathophysiologic process manifests in the form of repetitive uniform late decelerations the hallmark of a positive CST.
Overall, CSTs will be normal in 83–93% of pregnancies examined. In the remaining group, the test results are either unsatisfactory (poor recording or less than three contractions over 10 minutes) or equivocal/suspicious (only occasional late decelerations noted). Although the incidence of false-positive CSTs is high (30–60%), the false-normal rate is low.
Biophysical profile
The reactivity observed in the NST as well as in biophysical “functions”, such as fetal breathing movements (FBM), muscle tone, and gross body movements, are based on an intact fetal central nervous system (CNS). However, absence of some of these dynamic states may reflect either normal periodicity or CNS depression. In compromised fetuses the likelihood that more than one biophysical “function” can be altered is increased.
Because of these observations, Manning and associates postulated that evaluation of several fetal biophysical variables or a biophysical profile may better predict fetal health status. Subsequently, they conducted a number of studies to test the predictive ability of five biophysical variables including NST, FBM, fetal tone (FT), fetal movements (FM), and oligohydramnios. Their evaluation system was simple, a score of 2 was assigned for each normal variable and 0 when the biophysical parameter was absent or abnormal.
Recently, however, Manning and associates showed that the addition of the NST did not improve discriminative accuracy, when all the other four variables were normal.60 In that study, testing was started at a mean gestational age of 34.3 weeks and the examination time was extended for 30 minutes, before any variable was scored as abnormal. The outcome used for measuring true or false normal test results was fetal death within 1 week of a normal biophysical profile. Their observed false-negative rate was 0.634:1000, a result worse than that recorded for CST (0.4:1000) but superior to NST accuracy (3.2:1000).61
Overall, the results of the biophysical profile were normal, equivocal, or abnormal in 97.5%, 1.72%, and 0.76% of tests, respectively. For comparison, the rates for normal CST and NST results, reported in other studies, were in the range of 85% and 89%, respectively.58, 59 Interestingly, the predictive value of an abnormal biophysical score was not reported and comparison with CST or NST is, therefore, not possible.
The accuracy of the biophysical profile may be enhanced if guidelines to define prerequisites for the test are established. For example, to properly evaluate FBM (a function governed by circadian rhythm and maternal glucose level), during the day. Additionally, prior to testing it should be determined that the mother is not on medication known to inhibit the fetal CNS.
Umbilical artery Doppler study
The umbilical arteries (UA) and therefore Doppler interrogation of these vessels allows for the assessment of placental circulation. In the normal placenta, the resistance to passive flow decreases with increasing gestational age. However, in the growth restricted fetus, often the resistance to flow increases leading to an overall decease in flow. It should be noted that it is the pathological conditions leading to IUGR and not fetal growth restriction itself that lead to abnormal umbilical artery Doppler values.
A number of authors have evaluated and tested the efficacy of UA Dopplers in the monitoring of the growth restricted fetus. In addition, there is continued debate regarding the implications of abnormal UA Doppler results. Unfortunately, the Doppler studies in isolation do not predict the timing of perinatal insults, and they are not able to predict which neonates will be compromised. Therefore, this type of monitoring should be used as an adjunct to other perinatal testing. It may be used to help guide the frequency of NST or biophysical profile testing in the at risk pregnancy. Progressive decline in the flow, absent and finally reversed flow in the UA Doppler studies should prompt intensive fetal surveillance and may guide the decision to deliver when gestational age and other fetal surveillance are taken into account.
Management protocols
The sequence of bioelectric and biophysical testing, used to predict IUGR and/or fetal asphyxia, varies in different institutions. In most centers management protocols list use of the NST as the initial evaluation of the fetus with altered growth. In the event the test is nonreactive, a biophysical score is then obtained. UA Dopplers are used to determine the frequency of fetal testing, as abnormal Dopplers will prompt more frequent NSTs.
Most obstetricians agree that in the face of IUGR and abnormal biophysical and bioelectric tests, intervention, regardless of pulmonary maturity, is necessary to prevent fetal death or asphyxia possibly leading to long-term CNS deficits.
By comparison the question of when to deliver an IUGR fetus, who has attained pulmonary maturity but in whom biophysical and bioelectric tests remain normal, has not been scientifically answered yet. Some physicians feel that, under these circumstances, delivery should be effected by 36–38 weeks' gestation, in the hope of preventing long-term CNS abnormalities.1 Others are not yet convinced of the benefits of early delivery.
MACROSOMIA
Optimal diabetic antenatal care entails the early detection and appropriate management of fetal growth acceleration. The large-for-gestational age (LGA) neonate is at greater risk for perinatal mortality and morbidity when compared with the normal sized infant.62 Sack reported a threefold increase in perinatal mortality when infants weighed more than 4500 g.63 A 5-year follow-up study of 200 LGA neonates revealed severe neurologic disability in 11.4%. In another study of 312 infants weighing more than 4500 g, neonatal morbidity, neonatal mortality, and maternal morbidity were 13.2%, 3.2%, and 21.2%, respectively.64 Fetuses weighing more than 4000 g are reported to be at increased risk for prolonged second-stage labor, midforceps delivery, shoulder dystocia, and immediate neonatal injury.65 Furthermore, macrosomia in the infant of a diabetic mother (IDM) is associated with an increased rate of perinatal loss.66, 67
Definition
For clinical purposes, macrosomia is generally defined as a fetal weight in excess of 4000 g.65, 68 Fetuses weighing more than 4000 g and those weighing in excess of 4500 g account for 10% and 1% of all deliveries, respectively.69 Among diabetic pregnancies, however, the incidence of macrosomia has ranged from 16% to 45%.70, 71, 72 Unfortunately, absolute weight criteria may be too restrictive for the determination of macrosomia because birth weight represents merely an endpoint of accelerated growth. To effect improvement in perinatal care, the evolution as well as the extent or severity of macrosomia should be determined and quantified earlier in pregnancy and prior to term or attainment of maximal size and weight. Toward this end, we concur with others who believe that fetal weight above the 90th centile for gestational age is a more meaningful clinical definition of macrosomia than is absolute birth weight.72, 73
Our experience parallels that of Hadlock and associates who contend that LGA fetuses are a nonhomogeneous population with two main forms of macrosomia.74 The first is symmetric macrosomia whereby accelerated growth of all fetal parameters (i.e., fetal weight, length, head circumference, and abdominal circumference all exceed the 90th centile for gestational age). Generally, this form of “constitutional macrosomia” is more commonly noted in patients with large stature and tends to recur in subsequent pregnancies. On the other hand, asymmetric macrosomia occurs in diabetic patients falling into White's A–C classification. In these asymmetric LGA fetuses the head and femur measurements vary in size and length but fall below the 90th centile rank. Fetal abdominal circumference and thigh diameter, however, both reflect soft tissue mass and may be significantly larger than normal.74, 75
Antenatal prediction
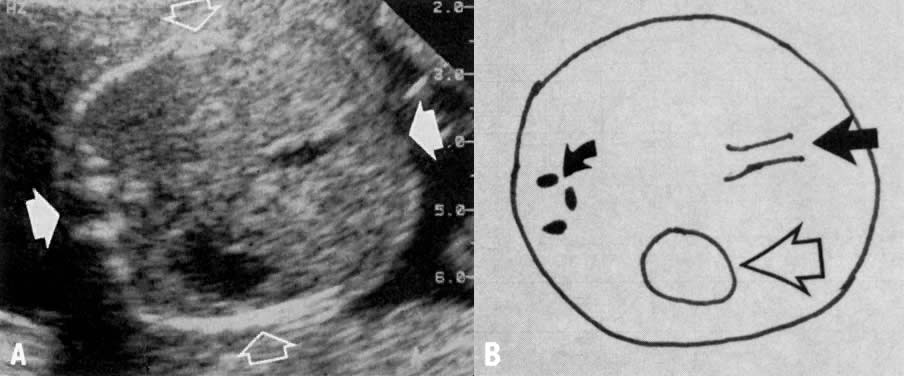
Fetal macrosomia is difficult to predict in the antenatal period.68 The detection of evolving diabetic macrosomia, however, was reported by serial ultrasound measurements of the fetal BPD and AC (Fig. 5).76 The fetal BPD was followed as an index of brain growth, which is relatively “insensitive” to insulin action.77, 78, 79 Additionally, the fetal AC was used to evaluate growth of the liver and subcutaneous abdominal fat, tissues which are relatively insulin “sensitive”.77, 80, 81 Specifically, in 23 insulin-dependent diabetes mothers (classes A–C), the BPD values of all fetuses fell within the normal range (mean +2 SD). However, AC values for only 13 of the 23 fetuses fell within the normal range (mean +2 SD) of measurements for fetuses of nondiabetic pregnancies. In the other ten fetuses AC values exceeded the upper limits of normal (2 SD above the mean) for nondiabetic pregnancies between 28 and 32 weeks' gestation; delivered at term, the reliability of the macrosomia index for detection of evolving macrosomia (prior to term) has not been determined. Platt and associates reported that a transverse fetal thigh diameter greater than 5.6 cm was associated with macrosomia in all their study population.75 Unfortunately, false-negative and false-positive rates were not reported. In 147 fetuses of diabetic mothers, Tamura and associates compared BPD, HC, and AC centile ranks to BW centiles in the latter part of pregnancy.71 Estimates of fetal weight by Shepard's method were also calculated from these data and compared to actual BW centile values.33 They noted AC values greater than the 90th centile correctly predicted macrosomia (defined as a BW greater than the 90th centile for age) in 78% of cases, and that the BPD and HC centiles were significantly less predictive of macrosomia. By comparison, estimated fetal weights greater than the 90th centile correctly predicted macrosomia in 74% of cases.
|
When both the AC and the estimated fetal weight exceeded the 90th centile, however, macrosomia was correctly diagnosed in 88% of cases. Of clinical importance, vaginal delivery was attempted in 109 of the 147 diabetic gravidas. The probability of cesarean delivery for disproportion in fetuses predicted to be macrosomic was 28.3%. On the other hand, the probability of cesarean section in fetuses predicted not to be macrosomic was <0.7%, a statistically significant difference (X2p <0.05).
Although several formulas are now available for sonographic estimation of fetal weight, to date, only two studies have evaluated the accuracy of fetal weight estimation for the detection of macrosomia. This is presumably because of the significant errors associated with such estimates which range from ± 16% to 20% (±2 SD). Because of this variation, a fetus weighing 4000 g may be estimated to weigh as little as 3200 g or as much as 4800 g. Clearly, such a wide range of estimates would result in a significant number of false-negative and false-positive results. Korndorffer and associates, in a preliminary study, compared three methods for predicting macrosomia and were able to correctly diagnose only five of ten LGA fetuses by the best method.82 Tamura and associates tested the applicability of fetal weight estimation formulas in 64 fetuses of diabetic mothers.83 Using the BPD and the AC, the best fitting equation for estimating fetal weight was log(EFW) = 0.02597 AC + 0.2161 BPD − 0.1999(AC × BPD2)/1000 + 1.2659. The standard deviation of differences was 322.26 g and the multiple r was 0.781. These authors caution that the large standard deviations of mean differences behoves the obstetrician to use clinical judgment and centile ranks of fetal sonar parameters in addition to weight estimates when planning delivery of fetuses suspected of being macrosomic.
ASSESSMENT OF FETAL WELL-BEING
Available methods for fetal antenatal assessment in diabetic pregnancies include biochemical (urinary estriols), bioelectric (antepartum fetal heart rate testing), and biophysical (ultrasound) modalities. Biochemical tests, specifically serial urinary estriol determinations, have served as the standard of fetal well-being against which the value of fetal heart rate testing has been compared. Recent reports, however, have seriously questioned the value of estriol determinations as the standard. Dooley and associates observed drops in the estriol:creatinine ratio of 40% or greater in only 0.6% of tests (19 of 3085) and found that only two of these abnormal drops correctly predicted fetal distress.84 Thus, the high numbers of false-positive estriol drops precludes clinical usefulness of the test in the management of pregnant diabetics. Further, the cost of serial estriols is high and there is a delay in obtaining laboratory results. Golde and associates used bioelectric and biophysical testing to manage insulin-dependent diabetic pregnant women.85
Antepartum fetal heart rate testing (AFHRT) is, at present, the predominant approach used to assess fetal status in the insulin-dependent diabetic mother. As a result of the low false-negative rate, the contraction stress test (CST) has had the greatest clinical usefulness in diabetic gestations.86, 87 Although Gabbe and associates have noted a false-positive rate of 42% when either the CST or urinary estriol was abnormal, attention to the presence or absence of fetal heart rate accelerations associated with fetal movements may improve the accuracy of the CST in IDMs.87 Freeman's group reported that nonreactive positive CSTs were significantly more ominous than reactive positive ones.88, 89 Thus, when obstetric facilities are adequate and direct fetal electronic heart rate monitoring as well as blood gas sampling are available, most clinicians will attempt a trial of labor in those diabetic patients with reactive positive CSTs.86, 87, 90
The nonstress test (NST), although widely used, has not been tested in large numbers of diabetic pregnancies. Whereas both types of AFHRT are reflective of fetal status, nonreactive NSTs are associated with more false-negatives than are positive CSTs. Whittle and associates and Dooley and associates have demonstrated that NSTs are superior to estriol determinations.84, 91 However, because of the finding of fetal distress within 1 week of normal AFHRT, most investigators recommend testing at least twice weekly to detect imminent fetal demise in IDMs.91, 92
Dynamic ultrasound imaging provides another screening modality by allowing in utero observation of fetal activity. Although several aspects of fetal behavior have been studied, at present, fetal breathing movements (FBM) and body movements (FM) appear to be the most promising. Under normal conditions, FBM may be observed from 30% to 90% of the time; normal frequency is 30–70 per minute.93, 94 In near-term fetuses, the “inspiratory” effort results in an inward anterior fetal chest wall movement of about 2–5 mm and an outward anterior abdominal wall excursion of 3–8 mm.95 Although continuous breathing activity is rarely seen for more than 10 minutes,96 apneic periods of up to 108 minutes have been observed in normal fetuses.94 Diurnal variation in the incidence of FBM has been reported and Patrick and associates have correlated significant increases in FBM following meals that appear to be associated with elevations in maternal plasma glucose concentrations.94 Evidence fails to indicate whether the increase in maternal plasma glucose concentration directly affects FBM or indirectly mediates FBM as a result of its effect on other substances, that is free fatty acids, hormones, and pCO2. Natale and associates hypothesize that local excesses of carbon dioxide produced by increased glucose oxidation might stimulate fetal medullary chemosensitive areas and produce increased FBM.97 Nevertheless, rare observations of rapid FBM activity have been recorded in fetuses of poorly controlled diabetics.93, 98, 99 However, until these findings can be correlated to clinical situations, their value in the management of diabetic pregnancies is largely experimental.
Improved neonatal care has sharply reduced the incidence of neonatal deaths in IDMs. As a result attention is presently being focused on antepartum and intrapartum methods that allow for the early detection and treatment of fetal disease. Because asphyxia and major anomalies account for about 85% of fetal mortality in unscreened and untested populations, Manning and associates have devised a method for reliably detecting both of these causes of fetal mortality.100 Using dynamic real-time ultrasound, they studied five fetal biophysical variables and devised a risk management protocol for asphyxia. In a prospective, blinded, clinical study they found that perinatal mortality consistently rose when the last test score prior to delivery fell.6 Although a specific diabetic population was not longitudinally studied, 9.2% of all the referred patients had pregnancies complicated by diabetes mellitus. Whereas 97.52% of the tests were normal, 17.2% were equivocal, and 0.76% were abnormal. Of the 93 perinatal deaths, 66.7% were the result of major anomalies, 7.5% were attributed to isoimmunization, and 25.8% were due to asphyxia. Of prognostic significance, completely normal test scores were associated with a perinatal mortality rate of 0.652:1000 whereas with completely abnormal scores the mortality rate was 187:1000 tests. Clearly, large numbers of diabetic pregnancies must be serially assessed by biophysical profile testing before its accuracy can be precisely determined. Nonetheless, the principle on which the biophysical profile scoring system rests is sound, namely, “that the more complete the examination of the fetus, its activities and its environment, the more accurate may be the differentiation of fetal health from disease states.”6
REFERENCES
Tejani N, Mann LI, Weiss RR: Antenatal diagnosis and management of the small-for-gestational-age fetus. Obstet Gynecol 47: 31, 1976 |
|
Dewhurst DJ, Beazley JM, Campbell S: Assessment of fetal maturity and dysmaturity. Am J Obstet Gynecol 113: 141, 1972 |
|
Beazley JM, Underhill RA: Fallacy of the fundal height. Br Med J 4: 404, 1970 |
|
Lind T: The estimation of fetal growth and development. Br J Hosp Med 3: 501, 1970 |
|
Ong HC, Sen DK: Clinical estimation of fetal weight. Am J Obstet Gynecol 112: 877, 1972 |
|
Yerushalmy J: Relation of birth weight, gestational age, and rate of intrauterine growth to perinatal mortality. Clin Obstet Gynecol 13: 107, 1970 |
|
Usher R, McLean F: Normal fetal growth and the significance of fetal growth retardation. In Davis JA, Dobbing J (eds): Scientific Foundations of Paediatrics, p 69. London, Heinemann, 1974 |
|
Gruenwald P: Infants of low birth weight among 5000 deliveries. Pediatrics 34: 157, 1964 |
|
Naeye RL, Kelly JA: Judgment of fetal age. III. The pathologist's evaluation. Pediatr Clin North Am 13: 835, 1966 |
|
Scott KE, Usher R: Fetal malnutrition: Its incidence, causes and effects. Am J Obstet Gynecol 94: 951, 1966 |
|
Fitzhardinge PM, Steven EM: The small-for-dates infant. II. Neurological and intellectual sequelae. Pediatrics 50: 50, 1972 |
|
Winick M: Fetal malnutrition. Clin Obstet Gynecol 13: 526, 1970 |
|
Winick M, Brasel JA, Velasco EG: Effects of prenatal nutrition upon pregnancy risk. Clin Obstet Gynecol 16: 185, 1973 |
|
Villar J, Belizan JM: The timing factor in the pathophysiology of the intrauterine growth retardation syndrome. Obstet Gynecol 37: 499, 1982 |
|
Battaglia FC, Lubchenco LO: A practical classification of newborn infants by weight and gestational age. J Pediatr 71: 159, 1967 |
|
Rosenberg K, Grant J, Hepburn M: Antenatal detection of growth retardation-actual practice in a large maternity hospital. Br J Obstet Gynaecol 89: 12, 1982 |
|
Verma UL, Tajani NA, Chatterjee S et al: Screening for small-for-gestational-age by the roll-over test. Obstet Gynecol 56: 591, 1980 |
|
Scott A, Moar V, Ounsted M: The relative contributions of different maternal factors in small-for-gestational-age pregnancies. Eur J Obstet Gynecol Reprod Biol 12: 157, 1981 |
|
Tejani NA: Recurrence of intrauterine growth retardation. Obstet Gynecol 59: 329, 1982 |
|
Belizan J, Villar J, Nardin JC et al: Diagnosis of intrauterine growth retardation by a simple clinical method-measurement of uterine height. Am J Obstet Gynecol 131: 643, 1978 |
|
Sparks JW, Girard JR, Battaglia FC: An estimate of the caloric requirements of the human fetus. Biol Neonate 38: 113, 1980 |
|
Rosenberg K, Grant J, Tweedie I et al: Measurement of fundal height as a screening test for fetal growth retardation. Br J Obstet Gynaecol 89: 447, 1982 |
|
Lubchenco LO, Hansman C, Boyd E: Intrauterine growth in length and head circumference as estimated from live births at gestational ages from 26–42 weeks. Pediatrics 37: 403, 1966 |
|
Miller HC, Merritt T: Fetal growth in humans. Chicago, Year Book Medical Publishers, 1979 |
|
Hadlock FP, Deter RL, Foecher E et al: Relation of fetal femur length to neonatal crown-heel length. J Ultrasound Med 3: 1, 1984 |
|
O'Brien GD, Queenan JT: Ultrasound fetal femur length in relation to intrauterine growth retardation. Am J Obstet Gynecol 144: 35, 1982 |
|
Sabbagha RE, Barton BA, Barton FB et al: Sonar biparietal diameter. II. Predictive of three fetal growth patterns leading to a closer assessment of gestational age and neonatal weight. Am J Obstet Gynecol 126: 485, 1976 |
|
Sabbagha RE, Turner JH, Chez RA: Sonar biparietal diameter growth standards in the rhesus monkey. Am J Obstet Gynecol 121: 371, 1975 |
|
Sabbagha RE: Intrauterine growth retardation: Antenatal diagnosis by ultrasound. Obstet Gynecol 52: 252, 1978 |
|
Turner G: Recognition of intrauterine growth retardation by considering comparative birth weights. Lancet 2: 1123, 1971 |
|
Tamura RK, Sabbagha RE, Depp R et al: Diminished growth in fetuses born preterm after spontaneous labor or premature rupture of membranes. Am J Obstet Gynecol 148: 1105, 1984 |
|
Tamura RK, Sabbagha RE: Percentile ranks of sonar fetal abdominal circumference measurements. Am J Obstet Gynecol 138: 475, 1980 |
|
Shepard MJ, Richards VA, Berkowitz RL et al: An evaluation of two equations of predicting fetal weight by ultrasound. Am J Obstet Gynecol 142: 47, 1982 |
|
Hadlock FP, Hardst RB, Sharman RS et al: Estimation of fetal weight with the use of head, body, and femur measurements-a prospective study. Am J Obstet Gynecol 151: 333, 1985 |
|
Campbell S, Thoms A: Ultrasound measurement of the fetal head to abdomen circumference ratio in the assessment of growth retardation. Br J Obstet Gynaecol 84: 165, 1977 |
|
Divon MY, Chamberlain PF, Sipos L, Platt LD: Underestimation of fetal weight in premature rupture of membranes. J Ultrasound Med 3: 529, 1984 |
|
Weiner CP, Sabbagha RE, Vaisrub N, Socol ML: Ultrasonic fetal weight prediction: The role of head circumference and femur length. Obstet Gynecol 65: 812, 1985 |
|
Campbell S, Thoms A: Ultrasound measurement of the fetal head to abdomen circumference ratio in the assessment of growth retardation. Br J Obstet Gynaecol 84: 165, 1977 |
|
Williams RL, Creasy RK, Cunningham GC et al: Fetal growth and perinatal viability in California. Am J Obstet Gynecol 59: 624, 1982 |
|
Ott WJ, Doyle S: Normal ultrasonic fetal weight curve. Obstet Gynecol 59: 603, 1982 |
|
Woo JSK, Wan CW, Cho KM: Computer assisted evaluation of ultrasonic fetal weight prediction using multiple regression equations with and without the femur length. J Ultrasound Med 4: 65, 1985 |
|
Paul RH, Koh KS, Monfared AH: Obstetric factors influencing outcome in infants weighing from 1001 to 1500 grams. Am J Obstet Gynecol 133: 503, 1979 |
|
Eden RD, Jelovsek FR, Kodack LD et al: Accuracy of ultrasonic fetal weight prediction in preterm infants. Am J Obstet Gynecol 147: 43, 1983 |
|
Killam AP, Barrett JM, Cotton RB: The impact of a tertiary perinatal center on survival of the very low birth weight infant. J Tenn Med Assoc 74: 870, 1981 |
|
Manning FA, Hill LM, Platt LD: Qualitative amniotic fluid volume determination by ultrasound-antepartum detection of intrauterine growth retardation. Am J Obstet Gynecol 139: 254, 1981 |
|
Chamberlain PF, Manning FA, Morrison I et al: Ultrasound evaluation of amniotic fluid volume: The relationship of marginal and decreased amniotic fluid volumes to perinatal outcome. Am J Obstet Gynecol 150: 245, 1984 |
|
Hoddick WH, Callen PW, Filly RA, Creasy RK: Ultrasonographic determination of qualitative fluid volume in intrauterine growth retardation: Reassessment of the 1 cm rule. Am J Obstet Gynecol 149: 758, 1984 |
|
Philipson EH, Sokol RJ, Williams T: Oligohydramnios: Clinical associations and predictive value for intrauterine growth retardation. Am J. Obstet Gynecol 146: 271, 1983 |
|
Phelan JP, Platt LD, Yeh SY et al: The role of ultrasound assessment of amniotic fluid volume in the management of the postdate pregnancy. Am J Gynecol 151: 304, 1985 |
|
Hadlock FP, Deter RL, Harrist RB et al: A date independent predictor of intrauterine growth retardation: Femur length/abdominal circumference ratio. Am J Roentgenol 141: 979, 1982 |
|
Vintzileos AM, Neckles S, Campbell W et al: Three fetal ponderal indexes in normal pregnancy. Obstet Gynecol 65: 807, 1985 |
|
Vintzileos AM, Lodeiro JG, Feinstein SJ et al: Value of fetal ponderal index in predicting growth retardation. Obstet Gynecol 67: 584, 1986 |
|
Vintzileos AM, Campbell WA, Neckles S et al: The ultrasound femur length as a predictor of femur length. Obstet Gynecol 64: 779, 1984 |
|
Benson CB, Doubilet PM, Saltzman DH et al: Femur length/abdominal circumference ratio: Poor predictor of macrosomic fetuses in diabetic mothers. J Ultrasound Med 5: 141, 1986 |
|
Gohari P, Berkowitz RL, Hobbins JC: Prediction of intrauterine growth retardation by determination of total intrauterine volume. Am J Obstet Gynecol 127: 255, 1977 |
|
Chinn DH, Filly RA, Callen PW: Prediction of intrauterine growth retardation by sonographic estimation of intrauterine volume. J Clin Ultrasound 9: 175, 1981 |
|
Grossman M, Flynn JJ, Aufrichtif D et al: Pitfalls in ultrasonic determination of intrauterine volume. J Clin Ultrasound 10: 17, 1982 |
|
Lavery PJ: Nonstress fetal heart rate testing. Clin Obstet Gynecol 25: 689, 1982 |
|
Collea JV, Holls WM: The contraction stress test. Clin Obstet Gynecol 4: 707, 1982 |
|
Manning FA, Morrison I, Lange MB et al: Fetal assessment based on fetal biophysical profile scoring: Experience in 12,620 referred high-risk pregnancies: Perinatal mortality by frequency and etiology. Am J Obstet Gynecol 151: 343, 1985 |
|
Freeman RE, Anderson G, Dorcester W: A prospective multi-institutional study of antepartum fetal heart rate monitoring. I. Risk of perinatal mortality and morbidity according to antepartum fetal heart rate test results. Am J Obstet Gynecol 143: 771, 1982 |
|
Chase HC: Perinatal mortality overview and current trends. Clin Perinatol 1: 3, 1974 |
|
Sack RA: The large infant: A study of maternal, obstetric and newborn characteristics, including a long term pediatric followup. Am J Obstet Gynecol 104: 195, 1969 |
|
Nelson JH, Rover IW, Barter RH: The large baby. South Med J 51: 23, 1958 |
|
Benedetti TJ, Gabbe SG: Shoulder dystocia: A complication of fetal macrosomia and prolonged second stage of labor with midpelvic delivery. Obstet Gynecol 52: 526, 1978 |
|
Gellis SS, Hsia DY: The infant of the diabetic mother. J Dis Child 97: 1, 1974 |
|
Freeman RK: Obstetric management of the diabetic patient. Contrib Gynecol Obstet 1: 58, 1973 |
|
Kitzmiller J, Cloherty J, Younger MD et al: Diabetic pregnancy and perinatal morbidity. Am J Obstet Gynecol 131: 560, 1978 |
|
Houchang D, Modanlou HD, Dorchester WL et al: Macrosomia-maternal, fetal, and neonatal implications. Obstet Gynecol 55: 420, 1980 |
|
Coustan DR, Lewis SB: Insulin therapy for gestational diabetes. Obstet Gynecol 51: 306, 1978 |
|
Tamura RK, Sabbagha RE, Depp R et al: Diabetic macrosomia: Accuracy of third trimester ultrasound. Obstet Gynecol 67: 828, 1986 |
|
Coustan DR, Imarah J: Prophylactic insulin treatment of gestational diabetes reduces the incidence of macrosomian, operative delivery, and birth trauma. Am J Obstet Gynecol 150: 836, 1985 |
|
Golditch IM, Kirkman K: The large fetus. Management and outcome. Obstet Gynecol 52: 26, 1978 |
|
Hadlock FP, Deter RL, Harrist RB: Sonographic detection of abnormal fetal growth patterns. Clin Obstet Gynecol 27: 342, 1984 |
|
Platt LD, Golde SH, DeVore GR et al: The use of femoral soft tissue diameters in the prediction of macrosomic babies, p 97. Official Proceedings, AIUM Annual Meeting, Denver, 1982 |
|
Ogata ES, Sabbagha RE, Metzger BE et al: Serial ultrasonography to assess evolving fetal macrosomia: Studies in 23 pregnant diabetic women. JAMA 243: 2405, 1980 |
|
Naeya RL: Infants of diabetic mothers: A quantitative morphologic study. Pediatrics 35: 980, 1965 |
|
Hill DE: Effect of insulin on fetal growth. Semin Perinatol 2: 319, 1978 |
|
Susa JB, McCormick KL, Widness JA et al: Chronic hyperinsulinemia in the fetal rhesus monkey: Effects on fetal growth and composition. Diabetes 28: 1058, 1979 |
|
Olser M, Pedersen J: The body composition of newborn infants of diabetic mothers. Pediatrics 26: 985, 1960 |
|
Fee B, Well WB: Body composition of infants of diabetic mothers by direct analysis. Ann NY Acad Sci 110: 869, 1963 |
|
Korndorffer FA, Miller JM, Gabert HA: A comparison of three methods of predicting fetal overgrowth, p 160. Official Proceedings of the Society of Perinatal Obstetricians, Annual Meeting, San Antonio, 1984 |
|
Tamura RK, Sabbagha RE, Doolely SL et al: Real-time ultrasound estimations of weight in fetuses of diabetic gravid women. Am J Obstet Gynecol 153: 57, 1985 |
|
Dooley SL, Depp R, Socol ML et al: Urinary estriols in diabetic pregnancy: A reappraisal. Obstet Gynecol 64: 469, 1984 |
|
Golde SH, Montoro M, Good-Anderson B et al: The role of nonstress tests, fetal biophysical profile, and contraction stress tests in the outpatient management of insulin-requiring diabetic pregnancies. Am J Obstet Gynecol 148: 269, 1984 |
|
Schneider JM, Curet LB, Olson RW et al: Ambulatory care of the pregnant diabetic. Obstet Gynecol 56: 144, 1980 |
|
Gabbe SG, Mestman JH, Freeman RK et al: Management and outcome of pregnancy in diabetes mellitus, classes B to R. Am J Obstet Gynecol 129: 723, 1977 |
|
Evertson LR, Gauthier RJ, Collea JV: Fetal demise following negative contraction stress tests. Obstet Gynecol 51: 671, 1978 |
|
Braly P, Freeman RK: The significance of fetal heart rate reactivity with a positive oxytocin challenge test. Obstet Gynecol 50: 689, 1977 |
|
Gauthier RJ, Evertson LRL, Paul RH: Antepartum fetal heart rate testing. Am J Obstet Gynecol 133: 34, 1979 |
|
Whittle MJ, Anderson D, Lowensohn RI et al: Estriol in pregnancy. VI. Experience with unconjugated plasma estriol assays and antepartum fetal heart rate testing in diabetic pregnancies. Am J Obstet Gynecol 135: 764, 1979 |
|
Gabbe SG, Quilligan EJ: General obstetric management of the diabetic pregnancy. Clin Obstet Gynecol 24: 91, 1981 |
|
Boddy F, Dawes GS: Fetal breathing. Br Med Bull 31: 3, 1975 |
|
Patrick J, Natale R, Richardson B: Patterns of human fetal breathing activity at 34 to 35 weeks' gestational age. Am J Obstet Gynecol 132: 507, 1978 |
|
Patrick J, Fetherston W, Vick H et al: Human fetal breathing movements and gross fetal body movements at weeks 34 to 35 of gestation. Am J Obstet Gynecol 130: 693, 1978 |
|
Manning FA: Fetal breathing as a reflection of fetal status. Postgrad Med 61: 116, 1976 |
|
Natale R, Patrick J, Richardson B: Effects of human maternal venous plasma glucose concentrations on fetal breathing movements. Am J Obstet Gynecol 132: 36, 1978 |
|
Manning FA, Heaman M, Boyce D et al: Intrauterine fetal tachypnea. Obstet Gynecol 58: 399, 1981 |
|
Romero R, Chervenak FA, Berkowitz RL et al: Intrauterine fetal tachypnea. Am J Obstet Gynecol 144: 356, 1982 |
|
Manning FA, Platt LD, Sipos L: Antepartum fetal evaluation: Development of a fetal biophysical profile. Am J Obstet Gynecol 136: 787, 1980 |