Borderline Ovarian Tumors - Atypical Proliferative Ovarian Tumors
Authors
INTRODUCTION
The borderline ovarian tumors (BOTs) or atypical proliferative ovarian tumors (APOTs) are of particular importance to the women affected and to gynecologists caring for them as well as to the women's families. They are also important for the pathologists charged with establishing an accurate diagnosis and for the researchers who are trying to explain the tumors' complex pathogenesis.
BOTs or APOTs are of epithelial origin and represent a unique intermediate stage between the benign cystadenomas and the adenocarcinomas. They are separated from cystadenomas by the presence of cellular atypia and from high-grade malignant tumors by the presence of destructive stromal invasion. Some BOTs or APOTs have a minor form of invasion designated as microinvasion; some tumors present small focal areas that exhibit moderate to severe atypia that are designated as intraepithelial carcinomas. BOTs or APOTs share multiple similarities and differences as demonstrated below.1, 2
BOTs or APOTs represent 15–20% of atypical cell proliferations of the ovary.3, 4, 5 In general, these tumors have an excellent disease-free survival after surgical treatment. BOTs or APOTs have been recognized for more than 70 years; nevertheless, several authors going back to the late 1800s reported ovarian tumors with histological and clinical features between benign cystadenoma and high-grade malignant tumors.6, 7, 8 The BOTs or APOTs were separated into a new category of neoplastic processes as a result of the observation made by Taylor and others9, 10, 11 who noted that some tumors displaying papillary features and some with tumor deposits on the peritoneal surface had excellent survival, especially those of serous type. However, other tumors with the same stage and somewhat similar histologic architecture were rapidly fatal. The serous cystadenoma can progress to BOTs or APOTs and finally becomes a serous low-grade carcinoma. These three entities (cystadenoma, BOT or APOT, and low-grade serous carcinoma) are different biologically, in their clinical course and treatment modalities.1, 2, 11, 12, 13, 14
In 1971 the Cancer Committee of the International Federation of Gynecology and Obstetrics15, 16, proposed a classification of common primary epithelial ovarian tumors. They subdivided the tumors into benign cystadenoma, cystadenoma with proliferative activity of the epithelial cells and nuclear abnormalities, but with no infiltrative destructive growth, low malignant potential tumors, and cystadenocarcinoma. Later in 1973, the World Health Organization12 catalogued tumors with histologic characteristics of carcinoma, but with good behavior as “tumors of borderline malignancy”.
In the WHO classification of 2003,15 these neoplasms are simply designated as “borderline tumors”; they have been too widely recognized as ovarian tumors of “low malignant potential,” and as “proliferative ovarian tumors," terminology that was accepted by the WHO in the year 2000.17 At the present time, the use of the designation “low malignant potential” is not recommended.
To further complicate the issue of BOTs or APOTs, the presence of microinvasion has been introduced in the past few decades. Microinvasion may be represented by a single focus or multiple foci of epithelial cells with histologic characteristics identical to those of BOTs or APOTs. The cells forming the focus or foci of microinvasion are seen in the nearby stroma surrounded by an empty space or cleft supposedly filled by serous fluid and without stromal reaction, necrosis or inflammation as illustrated in Figs. 10 and 11. The empty space could also be the result of tissue retraction. The size of the focus or foci of microinvasion is calculated by using three different scales. Bell and Scully18 reported that it should not exceed 3 mm in diameter, later, a 10 mm2 area was suggested by others,19, 20 a 5 mm linear dimension was also suggested and is recommended for all forms of BOTs or APOTs. It is probably the most common dimension used today to calculate microinvasion.21, 22 The 3 mm linear dimension and the 10 mm2 area are arbitrary numbers and have not been scientifically validated.
The atypia in BOTs or APOTs is supposed to be of intermediate degree,23 nevertheless, there are areas of BOTs or APOTs that show changes amounting to small intraepithelial carcinomas (epithelial stratification, loss of polarity, marked nuclear atypia, cribriform architecture, and occasional mitoses).24, 25, 26
Between 20% and 40% of BOTs or APOTs are associated with extraovarian tumors deposits or implants (implants, concomitant tumor or metastasis). The stromal invasion or non-invasive nature of this tumor deposits is a very important histological parameter to determine the tumor behavior.
CLINICAL FINDINGS
In a study by Webb et al.,4 only 16% of the patients with BOTs or APOTs were completely asymptomatic; other investigators have reported no symptoms in up to 30% of cases.27 A sizable number of tumors are discovered during a routine physical, pelvic or abdominal imaging exam, or while performing surgery for other abdominal pathology. The most common symptoms are abdominal pain, abdominal cramps, discomfort or pressure (44%), increase of abdominal girth without abdominal mass (39%), gastrointestinal symptoms including changes in bowel habits (15%), and abdominal mass associated with gynecological symptoms. Patients with ovarian tumors occasionally complain of dyspareunia, infertility, and urinary frequency. The frequency of the symptoms increases with the stage of the disease.4, 10, 27, 28 The symptoms just described are not different from those noted with serous or mucinous cystadenomas or ovarian carcinomas. The symptoms of BOTs or APOTs manifest earlier than those of invasive ovarian carcinomas.
Most patients with BOTs or APOTs are of childbearing age; in a small review at Magee-Womens Hospital29 we found that the age range was 20–73 years (mean 43 years), most of the patients were clustered between 24 and 33 years, and the rest were between 59 and 70 years. In our cases there was a bimodal distribution of age. Other investigators have reported that most BOTs or APOTs occur in the fourth and fifth decades with an average age of 46 years.30, 31 In a review of 247 cases in Singapore,5 the authors found an age range of 16–89 years with a mean of 38 years. The difference between these figures and ours and those of others is probably due to sample size, difference in ethnic populations or geographic variations.
Approximately 70% of BOTs or APOTs are confined to one or both ovaries, 10% have spread to the pelvis, 17% to the abdomen, and less than 0.5% have metastasized beyond the abdomen at the time of diagnosis.32 In the review of Wong et al.5 the authors found different percentages related to stage at time of diagnosis from those mentioned above: stage I (92%), stage II (3.5%), and stage III (2.5%). The overall survival rate of 98% reported in the same review is slightly better than the survival of 94% at 10 years reported by Levi et al.33
In the past decade there have been some concept changes secondary to new knowledge related to mucinous borderline tumors. Earlier, it was believed that the mucinous borderline tumors were the second most common borderline neoplasm, that the mucinous tumors were bilateral, and that they were associated with pseudomyxoma peritonei (PMP). Today, we know that most nonbenign mucinous ovarian tumors are metastases from appendiceal tumors or from tumors in other areas of the GI tract, and that primary mucinous ovarian tumors perhaps are never associated with PMP. Therefore, the primary mucinous tumors are relatively rare; they account for 3% of all ovarian tumors.34, 35
To date, histological characteristics of a well-sampled tumor are the only way to establish an accurate diagnosis of BOTs or APOTs. Consequently, the gross examination, total number of sections examined, and the experience of the pathologist handling these types of lesions are paramount in making the most truthful histologic interpretation.
It should be kept in mind that the classification of a tumor includes a significant component of subjectivity. This is illustrated by an average interobserver reproducibility of 56% among experienced pathologists.36 The marked variability may be due in part to the minimal exposure of most surgical pathologist to these types of lesions, because in general, these kinds of tumors are not common. Russell37 recommends that at least two of the most important histopathologic characteristics of BOTs or APOTs – mild to moderate nuclear atypia (the atypical changes should occupy an area of at least 10% of the tumor epithelial lining), epithelial stratification, presence of rosettes, tufting, lack of destructive invasion (if invasion is present, it should be non-destructive and less than 5 mm in linear dimensions), presence of micropapillae, minimal or no mitotic activity (if mitoses are present they should not be atypical) – should be present before a diagnosis of BOTs or APOTs is rendered. Using this recommendation should lower the chances of misinterpreting an ovarian lesion. Two of the most important steps involved in the process of reaching an accurate diagnosis are extensive tissue sampling of the tumor, followed by meticulous microscopic examination. It is now recommended that two sections should be taken for every centimeter of the tumor diameter or more if necessary.14 The sections should be obtained from solid and papillary areas as well as from areas of nodular thickening, necrosis or focal hemorrhage.
The presence of tumor deposits outside the ovary in association with BOTs or APOTs has been intensely discussed. Are they tumor metastasis, or are they synchronous multifocal tumors?
Clinical findings and symptoms
- Most patients are of childbearing age
- 70% are diagnosed as stage I, <1% as stage 4
- Mucinous tumors are rare, usually unilateral and >12 cm in diameter
- Accurate diagnosis by microscopic examination.
Symptoms
- Most tumors are asymptomatic
- Abdominal pain, abdominal discomfort, fullness and pressure 44%
- Increased abdominal girth without abdominal mass 39%
- Symptomatic pelvic mass
- Gastrointestinal symptoms including abdominal cramps and changes in bowel habits 5%
- Gynecological and urinary symptoms.
Risk and protective factors
SEROUS BORDERLINE OVARIAN TUMORS/ATYPICAL PROLIFERATIVE SEROUS TUMORS/LOW-GRADE SEROUS CARCINOMA
The annual incidence of serous borderline ovarian tumors (SBOTs) or atypical proliferative serous ovarian tumors (APSOTs) in the USA is 2.5/100,000 of which 1.5/100,000 occur in white women.40 Between 1995 and 2004 the incidence rate in Norway and Sweden has been reported as 4.8 per 100.000 women 238,239, almost double that of the USA.41, 42
Serous borderline ovarian tumors or APSOTs account for 10% of the ovarian serous tumors and 56% of the BOTs or APOTs;43 the patients have an average age of 46 years; few are found between 12 and 19 years of age and few in the 9th decade.30, 31, 44, In the series of Longacre et al.44 1.4% of the patients were pregnant at the time of diagnosis. The SBOTs or APSOTs occur in slightly older women than do the cystadenomas, but in younger women than those with ovarian invasive carcinoma. The SBOTs or APSOTs are bilateral in 25–37% of the cases.45, 46, In a more recent series44 bilateralism was reported as 55%.
Gross examination
On gross inspection, SBOTs or APSOTs are similar to serous cystadenomas and to some adenocarcinomas. The tumors are round, ovoid or irregular in shape with the size ranging from 1 to 35 cm in diameter with a mean of 10.4 cm (Fig. 1). They are reddish, pink or yellowish blue; occasionally exhibiting brown discoloration secondary to intraluminal or intramural hemorrhage. Their external surface may be smooth, sometimes with alternating areas with irregular indurations formed by thick fibrous tissue. The indurate areas are occasionally associated with dystrophic calcification. The lining of the cyst wall can show both, serous and mucinous type epithelium with benign features as seeing in Fig. 2.
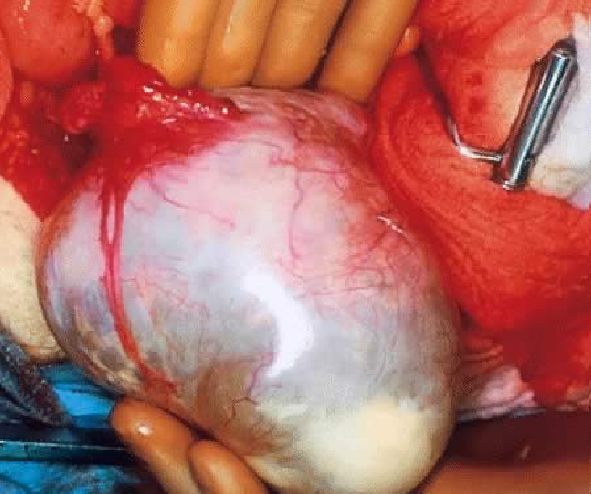 Fig. 1. External surface of a SBOT showing a smooth, tense capsule with prominent vessels and thick fibrous areas. (Photo courtesy of Dr Richard Stock.)
Fig. 1. External surface of a SBOT showing a smooth, tense capsule with prominent vessels and thick fibrous areas. (Photo courtesy of Dr Richard Stock.)
Blood vessels in the cyst wall can be very prominent (Fig. 1) depending on the tumor contents they can be translucent; on palpation it can be fluctuant or somewhat firm. On sectioning the tumor, it can be unilocular or multilocular, and may show secondary or “daughter” cysts of varying size and shape.47 Small or exuberant papillary structures can be seen on the external surface of the cyst wall, identical to those present in the cyst lumen (Figs. 3 and 4). Frequently the fallopian tube is attached to the cyst wall, and could be quite elongated; sometimes it is embedded in the cyst wall (Figs. 23 and 24). Fibrous adhesions are often seen.
The tumor contents can be a clear straw-colored fluid, hemorrhagic or quite frequently thick mucinous. The mucinous content does not necessarily indicate that the tumor is of mucinous type. The cyst inner lining exhibits arborizing or papillary structures; they can fill the entire lumen or alternate with smooth surfaces (Figs. 3 and 4). The papillary structures are generally yellow-brown or tan in color; they are soft and fragile and are covered by serous, brown or mucinous fluid. The ovary can be difficult to identify, especially when the tumor is very large, it may be reduced just to a thickened part of the cyst wall (Fig. 24).
Fig. 2. Section of the wall of a cystadenoma exhibiting two types of epithelia. Low columnar serous type (top) and tall columnar mucinous-type with basally located nuclei (bottom). This mixture of epithelia is found in serous or mucinous ovarian tumors of the ovary.
Fig. 3. SBOT or APSOT demonstrating growth of papillary projections arising from the internal surface of the tumor. Bubbly seromucinous fluid is present. (Courtesy of Dr Richard Stock.)
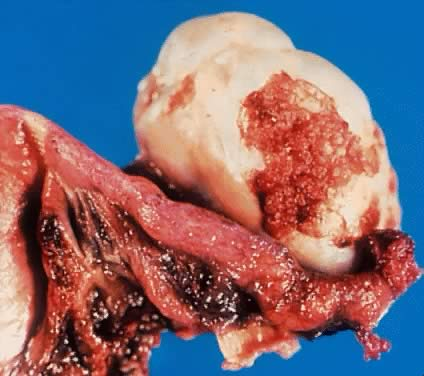 Fig. 4. Adnexal complex exhibiting SBOT or APSOT showing exophytic papillary growth. The comus of the uterus and the Fallopian tubes are seen clearly. (Courtesy of Dr Richard Stock.)
Fig. 4. Adnexal complex exhibiting SBOT or APSOT showing exophytic papillary growth. The comus of the uterus and the Fallopian tubes are seen clearly. (Courtesy of Dr Richard Stock.)
SBOTs or APSOTs are associated with peritoneal implants in approximately 40% of the cases;48 9% of the implants were invasive and 31% noninvasive.
A thorough and meticulous examination of the outer and inner lining of the cyst wall by the pathologist is paramount. This exam may occur when the tumor is opened in the frozen section room, or in the gross room, when an intraoperative consultation did not take place. The pathologist should perform or supervize the gross examination and strongly emphasize to the residents, fellows and pathologist assistants, that a thorough gross examination and specimen sampling is very important for the pathologist to generate an accurate final histopathologic interpretation. The thorough gross examination of the tumor impacts on patient outcome.
Microscopic features
By microscopic examination SBOTs or APSOTs are characterized by the presence of a cyst cavity or complex cystic spaces with infoldings and exuberant hierarchical papillary structures (Figs. 3–7). The papillae can occupy from 10 to 100% of the cyst cavity. In approximately 42% of cases, papillae can be present in both the cyst cavity and on the external surface of the cyst; the presence of papillae on the external surface has been reported in up to 70%.16
If there are papillary structures present only on the external surface of the SBOT or APSOT, which is reported in only 2% of the cases, the lesion would be designated as “serous surface papillary borderline tumor” or atypical proliferating surface serous tumor.
According to Longacre et al.49 69% of the tumors that have an exophytic component are associated with peritoneal implants compared with 16% of those cases where the tumor is only intracystic. Noninvasive and invasive desmoplastic and epithelial peritoneal implants can be present.
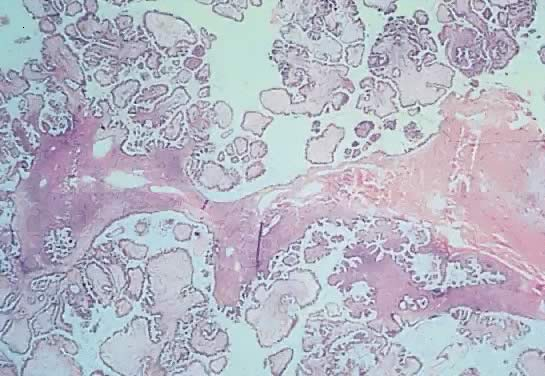
Fig. 5. Photomicrograph of SBOT or APSOT. Observe papillae of different sizes resting on fibrous tissue stroma. Tufting is observed in the interpapillary spaces (hematoxylin and eosin).
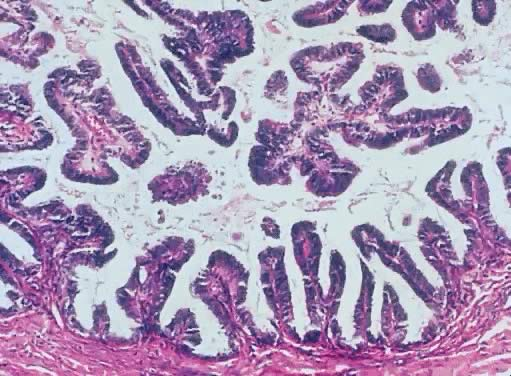 Fig. 6. Inner lining of a serous borderline tumor showing hierarchical papillae with stratification and hyperchromasia. The papillae have fibrous cores. Tufting is seen mixed with fluid in the lumen (hematoxylin and eosin).
Fig. 6. Inner lining of a serous borderline tumor showing hierarchical papillae with stratification and hyperchromasia. The papillae have fibrous cores. Tufting is seen mixed with fluid in the lumen (hematoxylin and eosin).
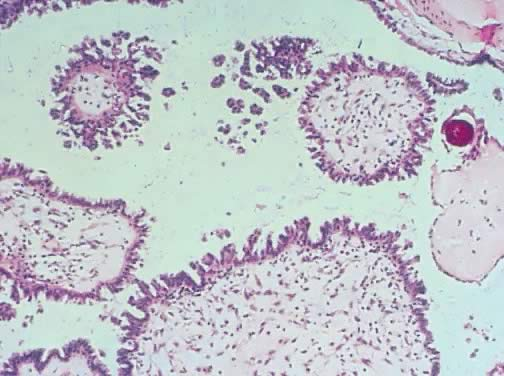 Fig. 7. Photomicrograph of papillae from a serous borderline tumor with edematous stroma, secondary papillary proliferation, and mild inflammatory infiltrate. A small psammoma body is present at the upper right-hand corner
associated with a hyalinized papilla (hematoxylin and eosin).
Fig. 7. Photomicrograph of papillae from a serous borderline tumor with edematous stroma, secondary papillary proliferation, and mild inflammatory infiltrate. A small psammoma body is present at the upper right-hand corner
associated with a hyalinized papilla (hematoxylin and eosin).
The papillae of SBOTs or APSOTs are of multiple lengths, and are branching; they have distinctive edematous, myxoid, or hyalinized connective tissue cores that support the blood vessels. The papillae are lined by different types of epithelia: cuboidal or columnar, tubal (ciliated, clear, and secretory or peg cells), and mucus secreting, either of cervical or enteric types; they usually show an eosinophilic cytoplasm.49, 50, 51 The nuclei are round or oval, are located at the base of the cells and show mild to moderate atypia. Some nuclei may be grooved or creased, nucleoli may be apparent; cellular stratification is seen and is one of the most characteristic histologic features of these tumors (Figs. 5, 6, 30 and 31). Psammoma bodies are seen in about 50% of the cases.
Budding or tufting is very commonly noted as a result of detached groups of epithelial cells from the tips of the papillae, due to lack of connective tissue support. These groups of cells can also form rosette-like structures (Figs. 7 and 9), which are very characteristic of SBOTs or APSOTs.
Budding or tufting, often single cells or clusters of cells float in the tumor fluid. Few areas of the tumor lumen or surface may be covered by micropapillae and/or cribriform structures (the result of the fusion of the tips of the papillae, creating the so called roman bridges) that originate on a thick, fibrotic large papillae core. The micropapillae are filiform, plentiful, and friable, they should not form a confluent area and occupy more that a 5 mm in linear dimension or 10% of the total tumor inner or outer surface, if it does, the lesion is a low-grade papillary serous carcinoma (discussed later). When the tumor is of micropapillary type, the papillae may not show stroma (Fig. 22). The nuclei are located at the base of the cells; they are round to oval and show mild or at most moderate atypia. Some nuclei may be grooved or creased; nucleoli may be apparent. Few SBOTs or APSOTs show severe or high-grade atypia; those tumors would be considered of unclear classification51 and expected to have a more aggressive behavior. In a recent publication McCluggage52 makes the point that the presence of severe nuclear atypia in a serous ovarian lesion, even in the absence of invasion, is sufficient to diagnose the tumor as a high-grade serous carcinoma. Mitotic figures are few and should be typical; probably do not exceed 4 per 10 HPF. Papillary structures present on the external surface of the tumor do not mean that the tumor has penetrated the wall of the cyst; they arise de novo on the ovarian surface epithelium and can be the only component of the tumor.47
The presence of epithelium with atypia should encourage the pathologist to go back to the gross specimen, examine it, and submit more sections in order to exclude a possible area of invasion or a frank high-grade invasive serous carcinoma51 not found in the first inspection. According to some authors, when the severe atypia is only focal (assuming adequate sampling), the pathologist should make a diagnosis of SBOT or APSOT with intraepithelial carcinoma (Fig. 8 and 30).53, 54 Seidman et al.14 classified microinvasive carcinoma as a form of microinvasion, which is typified by the presence of small solid nests of cells associated with micropapillae and distributed in a disorganized fashion. The group of cells, that sometimes show a cribriform pattern, are surrounded by a clear space and accompanied by desmoplasia. This resembles primary ovarian invasive low-grade serous carcinoma, or microinvasive carcinoma. It may be a manifestation of a true invasive serous carcinoma.
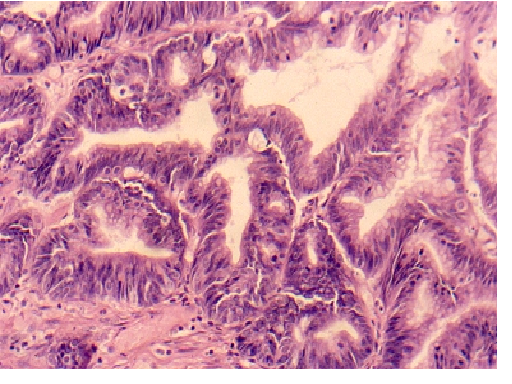 Fig. 8. Photomicrograph illustrating serous borderline tumors with high-grade atypia, stratification, and cribriform architecture representing a focal area of intraepithelial carcinoma (hematoxylin and eosin).
Fig. 8. Photomicrograph illustrating serous borderline tumors with high-grade atypia, stratification, and cribriform architecture representing a focal area of intraepithelial carcinoma (hematoxylin and eosin).
The pathologist should be very careful when examining tumors that exhibit small fine micropapillary structures arising from a thick stromal core and associated with small solid epithelial nests distributed in a disorganized fashion, not to miss a low-grade carcinoma.
The complex papillae of SBOTs or APSOTs produce tufting resulting from detached groups of epithelial cells devoid of connective tissue. Calcifications in the form of psammoma bodies (with laminated structures) are present in up to 50% of cases; they are located between the papillae or in the connective tissue. The histologic changes described as characteristic of SBOTs or APSOTs (cellular atypia, pluristratification, papillary structures, tufting, occasional normal mitoses, and absence of destructive invasion) should be found in at least 10% of the epithelial surface in order to classify the neoplasm as a SBOT or APSOT. If the histologic changes occupy less than 10% of the epithelial surface, the tumor should be classified as a cystadenoma with epithelial proliferation.55 The pathology report should be accompanied by a note saying: epithelial proliferation not sufficient for the diagnosis of SBOT or APSOT.
The atypia in BOTs or APOTs is supposed to be of intermediate degree,23 nevertheless, there are areas of BOTs or APOTs that show changes amounting to an intraepithelial carcinoma (epithelial stratification, loss of polarity, marked nuclear atypia, cribriform architecture, and occasional mitoses),24, 25, 26 this type of lesion is now classified as noninvasive low-grade carcinoma.
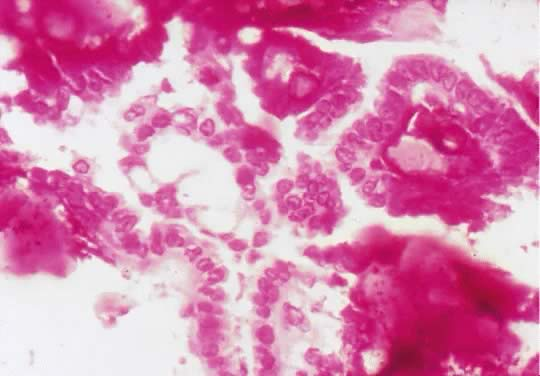 Fig. 9. Photomicrograph of a SBOT or APSOT showing rosette-like structures associated with psammoma bodies (hematoxylin and eosin).
Fig. 9. Photomicrograph of a SBOT or APSOT showing rosette-like structures associated with psammoma bodies (hematoxylin and eosin).
By definition, it is understood that destructive invasion should not be present in any area of the tumor, after making sure that the tumor has been thoroughly examined and abundantly sampled. Invasion can be sometimes very difficult to ascertain because of the presence of epithelial infoldings and tangential sectioning that may simulate epithelial invasion of the stroma, described by some as pseudoinvasion. The areas of destructive invasion should be represented by irregular glands with irregular borders, clusters or single cells with marked cellular atypia with a loose, edematous, and myxoid or desmoplastic stroma. These areas of destruction are at times associated with necrosis or inflammatory reaction and are present in invasive high-grade serous carcinoma.
Histologic characteristic of SBOTs or APSOTs
- Epithelial hyperplasia with hierarchical branching pattern papillae
- Epithelial stratification and tufting
- Nuclear enlargement with mild to moderate atypia
- 1, 2 & 3 present in 10% or more of the tumor total area
- Absence of microinvasion (3 mm linear size or 10 mm2 (WHO) or 5 mm new proposed classification)
- Noninvasive peritoneal implants
- Absence of destructive invasion.
MICROINVASION
SBOTs or APSOTs with microinvasion have been described by several authors.49, 56 It has been reported that approximately 10% of SBOTs or APSOTs have one or more foci of microinvasion.34, 57, 56, 58, 43, 59 Recent studies have quoted microinvasion occurring in 25%.60, 61 Silva et al. reported SBOTs or APSOTs with microinvasive implants, showing microinvasion in the tumor in 56% of the cases.62
The concept of microinvasion in SBOTs or APSOTs has been recognized for close to 50 years.63, 64, 65, 66 Microinvasion is defined as cells, individually or in groups budding off from atypical cells forming part of the SBOTs or APSOTs into the underline stroma, they are usually surrounded by a clear space or cleft. The cells forming the cluster are frequently large and show eosinophilic cytoplasm (Figs. 10 and 11). These cells are usually confused with decidual cells especially if the patient is pregnant when microinvasion is seen.67
The size of the microinvasion should not be more than 3 mm in linear dimension as reported by Bell and Scully63 in 1990 (multiple foci of microinvasion are permitted, their sizes should not be added), or no more than 10 mm2 for each focus, as discussed earlier.3, 56, 63, 64 The values 3 mm and 10 mm2 as expected, are arbitrary and based on a very small number of cases. Five millimeter in linear dimension as the upper limited for microinvasion is what is recommended now for all BOTs or APOTs.16
In practice it has been noted, that microinvasion is found more often during pregnancy; the frequency has been as high as 80%.67, 68
Fig. 10. Photomicrograph of SBOT or APSOT showing a focus of microinvasion, represented by small nests of tumor cells penetrating the tumor stroma near the epithelial–stromal interphase. The cells have eosinophilic cytoplasm and are surrounded by a clear space. There is no stromal necrosis or inflammation around the microinvasive nests
clearly shown in the lower centre of the photomicrograph (hematoxylin and eosin).
Fig. 11. In this photomicrograph there are cells with abundant eosinophilic cytoplasm that simulate decidual cells. This is a different form of microinvasion. No desmoplasia or inflammation is noted, the nearby cells show some nuclear atypia (hematoxylin and eosin).
Single cells or small clusters of cells with eosinophilic cytoplasm, apparently budding from the atypical epithelium into a space or cleft in the nearby stroma, characterize the most common type of microinvasion.
Bell et al. have designated this type of microinvasion as eosinophilic pattern.19 The nuclei of these cells are slightly enlarge and mildly atypical, sometimes with prominent nucleoli (Figs. 10 and 11). There is no stromal desmoplastic reaction, necrosis or inflammation around the focus of microinvasion. The epithelial cells or glands do not have malignant features. This type of microinvasion can sometimes be confused with lymphatic invasion. Nests of cells with cribriform, papillary or micropapillary architectural growth patterns, haphazardly invading the stroma and frequently surrounded by a clear space or cleft represent the second type of microinvasion. The space or cleft around the cell clusters is probably occupied by fluid supposedly secreted by the serous cells or could be the result of tissue retraction; the stroma in this type of microinvasion may show desmoplastic reaction. McKenney et al.67 described five different types of microinvasion and concluded that the only type that has an aggressive behavior is that composed of micropapillae.
Another type of microinvasion is characterized by epithelial proliferation similar to that present in a well-differentiated carcinoma and also similar to the epithelium present in invasive implants of the peritoneal surface.49 Some authors suggest that tumors with this type of microinvasion be designated as SBOT or APOT with microinvasive carcinoma34, 61 or simple, microinvasive carcinoma.
Microinvasion lacks tissue destructive stromal alterations.16 Patients with tumors with stromal microinvasion have a favorable prognosis and are now classified as low-grade serous carcinomas, if the histological pattern is that of a low-grade serous tumor.
The presence of microinvasion according to some authors63, 64, 65 appears not to adversely affect the patient outcome, especially if it is present in stage I tumors. Many of these patients from whom the data are known were treated conservatively with limited surgery. It is speculated that the pathologist may often miss these microscopic foci of microinvasion;53, 59 this oversight apparently has not changed the patient survival.15
Longacre et al.44 found in a study of 276 cases of SBOTs or APSOTs that stromal microinvasion in the primary tumor, independently of micropapillary features, tumor stage, and types of implants, is associated with adverse outcome. Sporadic cases have been reported, in which recurrence occurred and patients died of disease, when microinvasion was found at the time of original treatment.54
Prat et al. reported that peritoneal implants, exophytic growth, and bilaterality are more commonly present in cases exhibiting microinvasion.54
Lymphovascular space invasion
Sangol et al.69 reported that intratumoral lymphatic space invasion in SBOTs or APSOTs assessed by D2-40 immunostain is significantly associated with the presence of stromal microinvasion (p <0.0001) and is independent of tumor stage, patient’s age, primary tumor histology, and the pattern of microinvasion. The authors found that approximately 65% of the patients diagnosed with stromal microinvasion presented with lymphovascular intratumoral space invasion (Figs. 12 and 13). No lymphovascular intratumoral space invasion was found in tumors without stromal microinvasion.
Fig. 12. Photomicrograph of a SBOT or APSOT with a focus of vascular invasion. Tumor cells are present in what appears to be a vascular space
in the centre of the photomicrograph (hematoxylin and eosin).
Fig. 13. Photomicrograph of a consecutive section of the image above showing endothelial cells stained by positive CD 31 immunostain. Tumor cells are clearly within the vascular space. Myoepithelial cells in the cyst wall are also positive by the CD 31 immunostain (hematoxylin and eosin).
As can be expected, the presence of intratumoral vascular space invasion is associated with an increased risk for lymph node involvement and disseminated disease in several organs.
It has been pointed out70 that SBOTs or APSOTs and serous carcinoma spread more frequently by direct extension than by the lymphatic pathway. Stromal microinvasion was also found in 60% of SBOTs or APSOTs, 31% in low-grade serous carcinoma, and in 35% in high-grade serous carcinoma. Hilar vascular space invasion was more common in serous carcinoma: 15% in low-grade serous carcinoma and 69% in high-grade serous carcinomas. More studies with greater numbers of patients, using the same criteria and long-term follow-up are needed to determine patient outcome.
PERITONEAL IMPLANTS
Implants are ovarian or extraovarian epithelial lesions, in which there is a degree of epithelial or connective tissue proliferation similar to that observed in the ovarian tumor.
Morphologically, implants have been divided into invasive and noninvasive; this division is real and probably the most important prognostic indicator of patient outcome.71, 72 The histologic separation of invasive and noninvasive implants could at times be very difficult. The accurate separation of the two types has profound therapeutic repercussions. The noninvasive implants have been subdivided in desmoplastic implants and epithelial implants. On a given patient, the implants can all be noninvasive, all invasive, or a mixture of the two.72, 73, 74, 75, 76, 77 Noninvasive implants account for 83–96% of all peritoneal implants.
Noninvasive desmoplastic implants
Desmoplastic implants can be found on the ovarian surface (autoimplants), on the surface of the cyst, on the pelvic or abdominal peritoneum, on the omentum and on peritoneal invaginations. They are characterized by dense well-circumscribed tissue plaques that give the impression of being plastered on the surface of the peritoneum. The desmoplastic implants are portrayed by the presence of exuberant fibroblastic proliferation that can simulate granulation tissue. Tumors nests, papillary structures, or single cells with abundant eosinophilic cytoplasm entrapped in the dense fibrous desmoplastic stroma, represent the epithelial elements of the implants (Fig. 14). The fibrous stroma is often associated with edema and mononuclear cell inflammatory infiltrate. There is more stroma than epithelial elements. Psammoma bodies can be seen entrapped in the fibrous tissue. The presence of exuberant fibroblastic proliferation creates a difficult problem when trying to morphologically separate invasive from noninvasive implants.13 The early implants may be associated with hemorrhage and necrosis,72 which increase the challenge of morphologically separating the implants. Noninvasive desmoplastic implants do not invade the underlined tissues.
The autoimplants are multifocal in about 65% of cases, and are from 0.1 to 2.5 cm in diameter; the borders are commonly well demarcated and look very similar to noninvasive desmoplastic implants outside the ovary. What type of mechanism is involved in their development is still not clear. Some suggest that they may be the result of the ovarian surface touching a peritoneal implants elsewhere in the abdominal-pelvic cavities and transplanting some of the neoplastic cells to the ovarian surface.78 Others think that they may be the result of papillae infarction with detachment of viable cells with subsequent adherence to the ovarian surface, or by attachment of papilla exfoliating from the ovarian tumor and reattached to the ovarian surface. None of the possible mechanism of the origin of the autoimplant has been confirmed.
Fig. 14. Photomicrograph of a desmoplastic noninvasive implant with serous type epithelium. Observe the cystic space lined by atypical epithelial cells and small papillae. The stroma surrounding the epithelial components is fibroblastic and somewhat edematous (hematoxylin and eosin).
 Fig. 15. Photomicrograph of a peritoneal epithelial noninvasive implant. The tumor deposits have the histologic characteristics of a SBOT or APSOT. Note epithelial proliferation and papillae formation. The epithelial elements are surrounded by dense desmoplastic stroma. Depending on the epithelial–stromal ratio, the implant can be either of desmoplastic or epithelial type (hematoxylin and eosin).
Fig. 15. Photomicrograph of a peritoneal epithelial noninvasive implant. The tumor deposits have the histologic characteristics of a SBOT or APSOT. Note epithelial proliferation and papillae formation. The epithelial elements are surrounded by dense desmoplastic stroma. Depending on the epithelial–stromal ratio, the implant can be either of desmoplastic or epithelial type (hematoxylin and eosin).
Desmoplastic implants closely resemble reactive mesothelial processes; they can be independent from the ovarian tumor, in other words autochronous. Molecular tests have not yet resolved the issue of invasive versus noninvasive implants.79, 80
Noninvasive epithelial implants
Noninvasive epithelial implants are usually well-circumscribed plaques or small nodules found on the surface of the peritoneum. The implants are characterized by the presence of serous type epithelium forming branching papillary proliferations (Fig. 15) with small epithelial tufts and buds tacked on the peritoneal surface or in subepithelial invaginations without the presence of invasion of the subjacent tissues and without stromal response (desmoplasia).72 The epithelial cell nuclei exhibit minimal to mild atypia and, as in the desmoplastic implants, the mitotic activity is minimal or nonexistent. Occasionally the implants are formed by papillary structures with fibrovascular cores; the epithelial cells lining the papillae are similar to the cells seen in endosalpingiosis. Also, as in the desmoplastic implants, psammoma bodies are frequently present (Fig 15). Salpingitis is observed in 60% of patients with SBOTs or APSOTs or with micropapillary serous carcinoma (MPSC), this inflammatory process does not appear to be the same as that occurring in salpingitis proper.81
Invasive desmoplastic implants
Nine per cent of patients with SBOTs or APSOTs or noninvasive MPSC have invasive implants;57 a higher percentage (12%) is seen in patients with stage III SBOTs or APSOTs.82
The invasive desmoplastic implants show irregular borders. Deformed glandular-like structures are entrapped in the desmoplastic tissue implants; they do not resemble the elements that are typical of BOTs or APOTs and are haphazardly distributed. In general, the desmoplastic process takes over and replaces the adipose tissue. The epithelial elements can be seen infiltrating the adipose tissue lobules, rather than infiltrating in between the lobules.
Fig. 16. Photomicrograph of an invasive desmoplastic implant with serous epithelial elements of SBOT or APSOT histologic characteristics. Single cells and papillary clusters invade the stroma. Inflammatory infiltrate and fibroblastic proliferation is evident. The epithelial cells invade the fat lobules (hematoxylin and eosin).
Often the epithelium present in the invasive implants is more abundant, confluent and may produce complex architectural patterns such as glands, and papillary or cribriform structures. Histologically the epithelium present is similar to that seen on low-grade serous carcinoma (Fig. 16). Sometimes the epithelial elements may be contained in a cystic-like space. Other times the epithelial-like component can be just strapped mesothelial proliferation (cells should be Calretinin positive if they are of mesothelial origin) mimicking serous epithelium, the inflammatory infiltrate is chronic in about 80% and acute in 20% of the cases. Psammoma bodies are present in about 90% of the cases. Occasionally, the desmoplastic implants are completely calcified. The proportion of epithelium to fibrous tissue is larger in invasive implants, contrary to what happens in noninvasive implants. As can be expected, the histological criteria to separate the two types, invasive and noninvasive implants have been the subject of substantial debate.13 Frequently the biopsy material is too small to accurately separate the two types.
Invasive desmoplastic implants
- Irregular borders of epithelial elements
- Epithelial elements deformed and haphazardly distributed
- Epithelial elements simulate low-grade serous carcinoma
- Do not resemble epithelial elements of SBOTs or APSOT
- Intralobular rather than interlobular infiltration
- Abundant epithelium, complex architectural patterns
- Predominantly chronic inflammatory infiltrate
- Frequent psammoma bodies.
Invasive epithelial implants
Invasion can be found in approximately 15% of peritoneal implants as reported by Acs1, and others before him.53, 54, 82, 83 The invasive implants are formed by confluent proliferating epithelial elements that resemble low-grade serous carcinoma, sometimes with a cribriform pattern. The implants may be located in the peritoneum of the abdomen, pelvis and omentum. Grossly, these implants are similar to noninvasive implants. The stroma usually exhibits desmoplastic reaction and the tumor cells have infiltrative margins. Glands, clusters of cells, or single cells can represent invasive implants. The glands or clusters of cells exhibit irregular borders and have a haphazard distribution in the invaded stroma.72 Invasive implants clearly infiltrate the underlying tissues in an irregular fashion; they just do not sit on the peritoneal surface. They infiltrate the adipose tissue rather than migrating in between the fat lobules. The presence of exophytic papillary structures is sufficient to qualify the tumor deposit as invasive implants. Nonbranching micropapillae can be found embedded in the stroma surrounded by an empty space. Sometimes the micropapillae are found within the glands’ lumen. The epithelial cells have mild to moderate atypia in general. Occasionally some cases of invasive implants may show severe atypia and few mitoses. If severe atypia is present, this may represent a high-grade serous carcinoma as mentioned above. Inflammatory infiltrate and psammoma bodies are frequently present. The above-described histologic characteristics encompass the criteria used as described by Bell et al. in 1988,72 extended later by Gershenson in 1998 and 199073, 77 and then again by Bell et al. in 2001.84
Because the tissue samples containing the implants are often small, it is possible that the pathologist cannot see the area of invasion of the subjacent tissue. Therefore, if this is the case, the implant should be reported as noninvasive according with some investigators.53, 84, 85 Nevertheless we should remember that the additional criteria (solid nests of epithelial cells surrounded by clefts, micropapillary architecture, single cell, or clusters invading the stroma) have not been properly validated, although it appears that patients with this type of lesions have a higher mortality rate.54
It has been reported that patients with invasive implants have a less favorable prognosis.33, 46 To further complicate the issue, it has been published that there are groups of tumors with implants that histologically resemble well differentiated (low-grade) serous carcinoma that do not invade the underlined stroma; on the contrary, there are cases with implants that invade deeply the underlined stroma but histologically are similar to typical SBOT or APSOT. Consequently, more studies are needed to clarify these important points. Hopefully molecular techniques will one day help us to separate invasive from noninvasive implants. Nevertheless, the presence of invasive implants is very important and significant for the patient prognosis; there is currently no universally accepted definition for this type of implants. The data available for the relationship between the ovarian tumor and the implants are conflicting.79, 81, 86, 87
The possibility that these tumor deposits are implants and not metastases is suggested by their presence in 66% of patients with exophytic lesions on the surface of the ovary, while only 5% of patients with endophytic lesions show implants.28
Support of the theory that implants are secondary to ovarian tumors and, therefore, metastases, is suggested by a small study by Zanotti87 who found loss of heterozygosity (LOH) on chromosome 17p13 in the implants as well as in the ovarian tumor.
Another theory for the presence of peritoneal implants is that the implants represent autochronous tumors arising de novo in situ, independently of the ovarian neoplasm.88 This theory is supported by the existence of epithelium of the same type lining the surface of the organs in the abdominal cavity and abdominal pelvic region (secondary Müllerian system). Therefore, the implants can be secondary to field effect.89 The existence of extraovarian Müllerian serous carcinomas is a well-established fact; there is no reason to believe that something similar cannot occur on the peritoneal surface giving rise to a serous tumor similar to those arising on the ovary as reported by Bell and Scully in 1990.18 The multiple studies performed in an effort to establish the real origin of the extraovarian implants have shown conflicting results probably because they have been performed in a small number of patients and using different approaches. Patients with borderline ovarian tumors or APSOTs with extraovarian disease do much better than patients with serous carcinoma and peritoneal tumor spread. Therefore, the extraovarian disease in BSOTs or APSOTs should be implants and not metastases.90
The presence of peritoneal implants is associated with higher frequency of bilaterality, micropapillary infarction and microinvasion.54, 89
Invasive epithelial implants
- Glands and irregular solid nests with haphazard distribution in the stroma invading underlying tissues simulating low-grade serous carcinoma
- High ratio of epithelium to stroma
- Small round cell nests of serous or mesothelial type
- Epithelial cells with high N/C ratio and mild to moderate atypia
- Clear space or clefts around tumors nests or cells
- Endophytic or exophytic micropapillary architecture with confluent pattern, loose or dense fibrous stroma
- Psammoma bodies generally infrequent, at times, could be extensive
- Generally mild inflammation.
Tumor deposits in lymph nodes (associated serous lesions in lymph nodes)
Tumor deposits (glandular structures supposed to be of Müllerian origin) or associated serous lesions (ASLs) in examined lymph nodes are found in 5–15% of unselected women and in 42–65% of women with SBOTs or APSOTs and noninvasive micropapillary serous carcinoma.91, 92, 93 There is a significant association of this type of inclusions with the presence of invasive peritoneal implants,94 and micropapillary architecture.93 Unusual cases of distant metastasis from SBOTs or APSOTs and noninvasive micropapillary serous carcinoma to brain, lung, bone, liver and cervical lymph nodes are found in the literature.95, 96
The epithelial deposits or ASLs in the lymph nodes have been divided in four groups. The first is represented by glandular elements with benign characteristics located within lymphatic or sinusoidal spaces in the lymph node capsule or within fibrous septae of the lymph node, but not in the lymph node parenchyma. This glandular structure corresponds to endosalpingiosis (Figs. 17 and 18). The epithelium that forms the glands is ciliated, and shows no atypia. There is no proof that the epithelial cells are clonally related to the ovarian tumor.13
Fig. 17. Photomicrograph of
the capsular region of a lymph node. Note the benign appearance of the epithelial component (hematoxylin and eosin).
Fig. 18. Higher magnification of the above picture (Fig. 17) showing the glands in the capsular region of the lymph node (hematoxylin and eosin).
Fig. 19. Photomicrograph of the periphery of a lymph node with deposits of SBOT or APSOT in the subcapsular region. The epithelium
of the glandular spaces show papillary changes consistent with moderate atypia (hematoxylin and eosin).
Either single cells or clusters of cells with histologic changes that simulate SBOTs or APSOTs represent the second type of tumor deposits.57, 97
The third type of tumor deposits or ASLs is represented by lymph nodes that show clusters of tumor cells that are quite large, and are histologically similar to SBOTs or APSOTs, and are associated with the presence of benign glandular inclusions. These findings suggest that the solid tumor masses probably originated from the benign-looking glands and, therefore, they are not real metastases. The follow-up of patients with epithelial or tumor deposits designated as type 3 or ASLs showed no worse prognosis than that of patients with type 1 or 2 (Fig. 19).13, 90
Single cells or large clusters of cells, glands and papillary structures form the fourth type of epithelial deposits or ASLs in lymph nodes. These deposits or ASLs may represent migrated mesothelial cells lodging in the lymph node sinusoid. If they are mixed with papillary structures, they are not mesothelial. Large cells with eosinophilic cytoplasm are also present on the surface of BOTs or APSOTs. The origin of these mesothelial looking cells can be confirmed or excluded using immunostains such as Calretinin.98 Excluding endosalpingiosis, the remaining three types of ASLs can be reduced to two, merging the third and fourth groups, which are histologically very similar.
On rare occasions there are tumor deposits or ASLs in lymph nodes with changes compatible with high-grade serous carcinoma in the absence of invasive tumor of the ovary or any other site. This suggests that the tumor may have arisen from benign glandular inclusions in the lymph node. The possibility that the ASLs are of mesothelial origin could be confirmed with a positive Calretinin immunostain. Benign mesothelial inclusions have been reported in the absence of any ovarian or peritoneal neoplasm.
Silva et al.99 found that the gland deposits or ASLs are more frequently found in women with stage I SBOTs or APSOTs that recur, than in control women whose tumors do not recur after a follow-up of 15 years.
It is probably safe to say that the presence of epithelial or mesothelial cells, or tumor deposits or ASLs in the lymph nodes does not change the outcome of the patients with SBOTs or APSOTs.13
We should remember that ovarian tumors spread more by direct extension than by the lymphatic or hematogenous route.13, 94
Endosalpingiosis
Endosalpingiosis is found in 30–40% of patients with gynecological malignancies100, 101 and in 10% of patients with lesions other than BOTs or APOTs. Endosalpingiosis is characterized by the presence of glands lined by tubal ciliated-type epithelium without atypia, no mitoses, and absence of endometrial stroma. Occasionally, some of the glandular spaces exhibit non-branching papillae.44 Some investigators102, 103 consider the presence of these glandular inclusions as evidence of metastatic tumors in patients with BOTS or APSOTs. Others,104, 103, 105, 106 together with the general consensus today, consider the presence of those glands as endosalpingiosis, which is a benign process and does not represent metastasis from SBOTs or APSOTs. If there is any doubt regarding the presence of stromal cells around the glands, which may suggest the presence of endometriosis, a CD10 immunostain should be positive on endometrial stroma cells; this immunostain is very helpful to separate endometriosis from endosalpingiosis. Endosalpingiosis should not be confused with peritoneal implants from serous tumors and, therefore, potentially changing the tumor stage.
Endometriosis
Endometrial type glands surrounded by endometrial stroma represent endometriosis; contrary to endosalpingiosis, which as previously discussed, lacks endometrial stroma cells around the glandular structures. Endometriosis primarily can be found on the peritoneal surface, subperitoneal space and in lymphatic channels (Figs. 17 and 18).
Mesothelial cell hyperplasia
Mesothelial cell hyperplasia and mesothelial inclusions can be observed in the lymph node sinusoids, as single cells or forming cell clusters, and glandular structures with abundant eosinophilic cytoplasm with benign-looking nuclei. These cells are transported from hyperplastic foci of mesothelial cells exfoliated from the peritoneal surface.98
MICROPAPILLARY SEROUS BORDERLINE OVARIAN TUMORS/ATYPICAL PROLIFERATIVE MICROPAPILLARY SEROUS OVARIAN TUMORS/LOW-GRADE SEROUS CARCINOMA
Russell66, in 1979 described SBOTs or APSOTs with micropapillary and cribriform histologic characteristics. Later Seidman et al.13 and other investigators14, 107 reported that some SBOTs or APSOTs exhibit nonbranching and hair-like or filliform, micropapillae, some times with a filigree pattern or cribriform architecture. An observation was made that some tumors with lack of destructive invasion at the primary site had histologic characteristic similar to that of a well-differentiated serous carcinoma with papillary features.15, 107, 108 Seidman et al.13 also noted that tumors with invasive and noninvasive implants recur more often, are more frequently bilateral, disseminate like high-grade carcinoma, involve the surface of the ovary more often, afflict younger patients, do not respond to chemotherapy, run a protracted course, and have a different prognosis.
Lesions with micropapillary features in general, represent 6–26% of the SBOTs or APSOTs.11, 54, 82, 109
Base on the above observations, SBOTs or APSOTs with micropapillary component were divided into: serous micropapillary low-grade carcinoma with non-invasive implants, which accounts for about 14% of the micropapillary lesions, and serous micropapillary low-grade carcinoma with invasive implants or invasive low-grade micropapillary carcinoma, which is less frequent and accounts for 4% of the micropapillary tumors.14 The difference between these two forms (noninvasive vs. invasive implants) predicts patient outcome.13, 72 The WHO has accepted micropapillary serous carcinoma terminology since the year 2000.17
The survival rate of patients with stage I SBOTs or APSOTs is practically 100%. Nevertheless, survival of patients with a more advanced stage with noninvasive implants is 95.3%, while the survival rate for tumors with invasive implants is 66%, which is similar to that of high-grade carcinoma. The possibility exists that this extraovarian invasive lesion may represent real metastasis from an ovarian primary high-grade serous carcinoma that was misdiagnosed.
Invasion in serous micropapillary tumors is recognized by the presence of haphazard infiltrative epithelial growth composed of complex glands-like structures showing micropapillae; some times the clusters of glands or disorganized papillae are inside an empty space.
The slender hair-like or filliform micropapillae present in these tumors should originate from the periphery of a dense, fibrous, myxoid, and edematous central core (Fig. 20), although some times they may originate from the surface of the cyst wall. Psammoma bodies can be numerous. Tumor giant cells are not present. The invasive low-grade micropapillary serous carcinoma can display small papillae, micropapillae, macro papillae, small nests and large nests of epithelial proliferation infiltrating the stroma. Immunostains such as Ki67, P53 can help in separating low-grade from high-grade tumors.
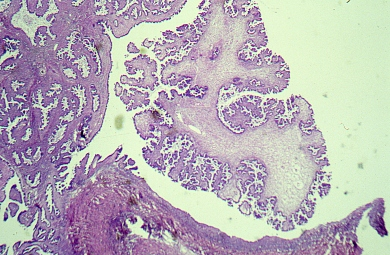 Fig. 20. Photomicrograph showing micropapillae arising on a thick fibrous edematous core. The nuclei are atypical (hematoxylin and eosin).
Fig. 20. Photomicrograph showing micropapillae arising on a thick fibrous edematous core. The nuclei are atypical (hematoxylin and eosin).
Patients with ovarian low-grade micropapillary serous tumors have a median age of 56 years, 10 years younger than patients with high-grade serous carcinoma110 of the micropapillary non-invasive tumors have a medium size of 8 cm in diameter; on average the invasive tumors are 2–3 cm larger that the non-invasive ones; the tumor surface is involved in over half of the cases. On opening the tumor it shows a papillary component with minimal or nonexisting necrosis. Necrosis is very helpful to differentiate, in gross and/or microscopic exam, a low-grade MPSC, where the necrosis is minimal or nonexisting, from high-grade serous carcinoma, where necrosis is plentiful.
In Burks et al.111 series 67% of the micropapillary serous tumors (MPSTs) had omental or peritoneal implants. Tumors with micropapillary component have a more aggressive behavior, which can be manifested by more frequent recurrences and decreased patient survival. Micropapillary serous low-grade carcinomas are associated with non-invasive implants in only 40% of the cases, 40% had only invasive implants, the rest, 20% had invasive and noninvasive implants. Low-grade micropapillary serous carcinomas with invasive implants show higher stage.
There is not clear explanation for the aggressive behavior of the noninvasive tumor as yet. Is the micropapillary architecture a form of invasion? Is the micropapillary component of the tumor a form of carcinoma in situ that exfoliates malignant cells that implant on the peritoneum and become an invasive tumor? Up to this point there is not a definitive answer to these questions.
The volume of the micropapillary component in a tumor varies from case to case, from 10 to 100%.107, 111 The cells covering the hair-like or filliform papillae, which are round, oval or cuboidal, rarely ciliated, are stratified, exhibit high nuclear to cytoplasmic ratio and mild to moderate atypia. Severe nuclear atypia (large and pleomorphic nuclei, irregular nuclear membrane, clumping of the chromatin, and prominent nucleoli) is characteristic of high-grade serous carcinoma and is the best parameter use to separate low-grade from high-grade serous carcinoma. The micropapillae have scanty or no connective tissue vascular cores, the cores can be hyalinized, edematous or myxoid. The papillae should be 5 times as long as they are wide (Fig. 22). The papillary/cribriform pattern should occupy a confluent area of at least 5 mm of linear dimension in order to call the lesion carcinoma, even in the absence of stromal invasion.111 Tufting is present and is due to exfoliation of cells from the papillae due to lack of connective tissue support. The mitotic activity in LGSC is commonly low <12/10 HPF, the mitoses are normal; the number of mitoses is significantly higher in high-grade serous carcinoma.
The presence of micropapillae, as expected, generated a large amount of discussion among pathologist surrounding their significance.112 Micropapillae are present in 6-26% of low-grade papillary serous tumors.54, 82, 109, 113 The microscopic picture created by the micropapillae originating from the large, fibrous, edematous central cores mimics a medusa head (Figs. 21 and 22).1, 53, 114
 Fig. 21. Photomicrograph of a micropapillary borderline ovarian tumor. The papillae are long and thin, they originate on fibrous cores and simulate a Medusa head (hematoxylin and eosin).
Fig. 21. Photomicrograph of a micropapillary borderline ovarian tumor. The papillae are long and thin, they originate on fibrous cores and simulate a Medusa head (hematoxylin and eosin).
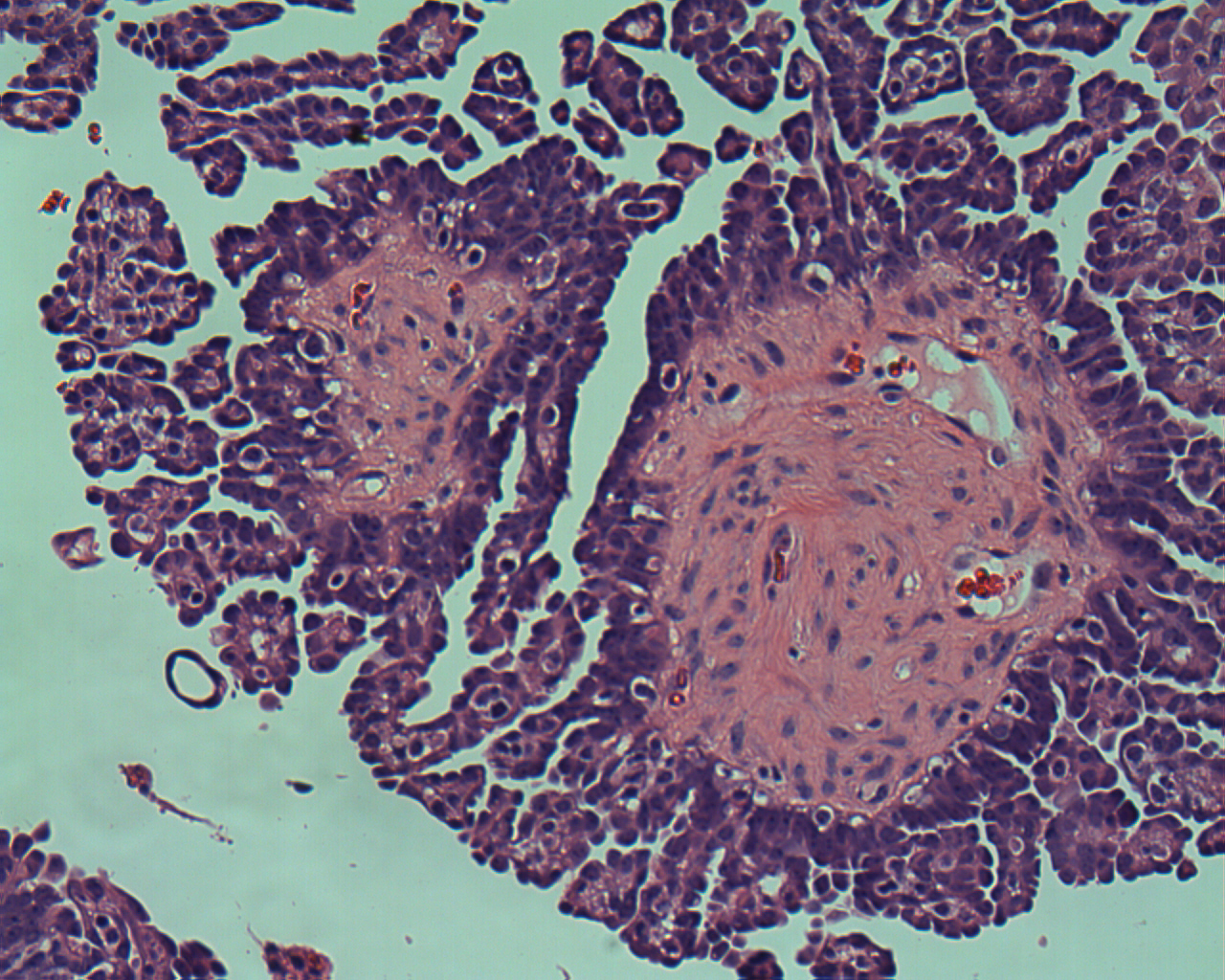 Fig. 22. Higher magnification of Fig. 21 showing the micropapillae arising from a thick fibrous core forming a Medusa head (hematoxylin and eosin).
Fig. 22. Higher magnification of Fig. 21 showing the micropapillae arising from a thick fibrous core forming a Medusa head (hematoxylin and eosin).
Low-grade serous carcinomas occasionally lined by cribriform nests, sometimes accompanied by micropapillae are very often (92%) associated with SBOTs or APSOTs, which suggest that low-grade serous carcinomas develop from a SBOT or APSOT.111
Biologically LGSCs are slothful tumors originating from a completely benign serous cystadenoma passing through SBOTs or APSOTs and reaching the stage of low-grade serous carcinoma. The stepwise transitions from benign serous cystadenoma to invasive low-grade serous carcinoma are substantiated by molecular events such as the presence of KRAS and BRAF mutations, which also separate low-grade from high-grade serous neoplasms.115, 116 On the contrary, TP53 mutations are almost always present in high-grade serous carcinomas but not in low-grade serous carcinomas.117 Low-grade serous carcinomas afflict younger individual than those afflicted by high-grade serous carcinoma, they occur less frequently than high-grade serous carcinoma. These tumors are also substantially different in their prognosis.
Recently Boy118 reported the coexistence in the same ovary of low-grade and high-grade serous carcinoma and undifferentiated carcinoma, the author suggests that probably they can be biologically related, which indicates that in some cases L-GSC and H-GSC may have the same biologic origin.
Some low-grade serous carcinoma may arise de novo, while high-grade serous carcinoma originates from detached distal tubal epithelium implanted on the ovarian.
Serous low-grade ovarian tumors with micropapillae (noninvasive micropapillary low-grade serous carcinoma)
This group of lesions is characterized by changes similar to a SBOT or APSOT with exuberant and friable papillary structures that shows a micropapillary hair-like, filigree or cribriform architectural component, without local invasion and without invasive and non-invasive implants; in other words, a stage I micropapillary serous tumor.
The great majority of the tumor component in MPSBOT or AMPSOTs can be represented by broad hierarchal or branching pattern papillae. The papillae are lined by exuberant and complex epithelial elements composed of stratified columnar or cuboidal cells with eosinophilic cytoplasm and occasionally surface blebs; ciliated cells are often present. The nuclei are uniform and exhibit mild to moderate atypia, associated with cellular stratification, which is characteristic of SBOTs or APSOTs. Budding of epithelial elements is very commonly seeing mixed with clear or mucinous fluid present in the cystic space; nuclear atypia is mild to moderate and mitoses are < 12/10 HPF. Psammoma bodies can be seeing in the interpapillary spaces or in the stroma of the papillae. The focal micropapillae should be long slender, hair-like with scanty connective tissue stroma, which may be hyalinized, edematous or myxoid. The micropapillae should be present in a confluent form occupying an area up to 5 mm in linear dimension (if the micropapillary area is larger than 5 mm and there is increase nuclear atypia, the tumor will be designated low-grade serous carcinoma). For some, the present of micropapillae in an area larger that 5 mm in a background of a SBOT or APSOT represent a low-grade serous carcinoma. The SBOTs or APSOTs with micropapillary component have an adverse behavior. Therefore, it was recommended by some107, 111 that these tumors be designated as low-grade serous carcinomas. SBOTs or APSOTs stage I and without stromal invasion have survival rate of 100%.
Lets emphasize that the presence of severe nuclear atypia (large and pleomorphic nuclei, irregular nuclear membrane, clumping chromatin, and prominent nucleoli) and mitotic index of >12/10 HPF separate high-grade from low-grade serous carcinoma.
There is evidence now that MPSOT/APMPST or MPSC is closer by molecular studies to L-GSC that it is to classical SBOT/APSOT (without micropapillary component).119 Therefore, MPSBOT/APMPST (low-grade serous carcinoma) is precursor of low-grade invasive carcinoma. These findings have staging implications.
By immunohistochemistry, p53 immunostain120 has shown differences among SBOTs or APSOTs, MPSC, and frank invasive high-grade serous carcinoma. The p53 immunostain was shown to be focally positive in SBOTs or APSOTs, moderately diffuse positive in MPSBOTs or APMPSTs, and very strongly positive in most serous high-grade ovarian carcinomas. According to the authors, the SBOTs or APSOTs lack p53 mutation.121
Histologic criteria for micropapillary serous borderline tumors (micropapillary serous carcinoma)
- Long slender non-branching micropapillae with grade I or II nuclei, scanty or stroma
- Papillae should originate in a large fibrotic, edematous myxoid core, or from the cyst wall or both
- Micropapillary or cribriform pattern
MORPHOLOGY, MOLECULAR GENETICS, AND PATHOGENESIS OF SEROUS BORDERLINE OVARIAN TUMORS OR ATYPICAL PROLIFERATIVE SEROUS OVARIAN TUMORS
Continuous effort, especially by the John Hopkins group, has resulted in a proposed new model for ovarian carcinogenesis based on molecular biology and genetics, in association with clinical and morphological evidence.14, 122, 123, 124, 125 According to this model, high-grade serous carcinoma and low-grade serous carcinoma are not related, in other words, high-grade serous carcinoma is not a more advanced form of low-grade serous carcinoma. Present knowledge suggests that ovarian tumors can be divided in two major groups.
The first group, or type I tumors, is composed of neoplasms that in general are limited to the ovary at the time of diagnosis, are genetically stable, develop slowly in a stepwise fashion, and behave in a less aggressive form. Type I tumors usually are found as large neoplasms at stage I. Low-grade serous ovarian carcinoma, a type I tumor, is less responsive to conventional chemotherapy than is high-grade ovarian carcinoma.126
Patients with type I tumors are younger than patients with type II tumors; type II tumors develop relatively fast.
Serous borderline ovarian tumors or APSOTs, low-grade serous carcinoma, mucinous carcinoma, low-grade endometrioid carcinoma, clear cell, and transitional cell carcinoma or Brenner carcinomas are all type I tumors. Type I tumors have benign precursors; they are the result of the progression from a benign cystadenoma passing through BOTs or APOTs, and finally becoming a low-grade carcinoma. Endometrioid neoplasms (endometrioid borderline tumors; EBTs) or atypical proliferating endometrioid tumors (APETs), and low-grade endometrioid adenocarcinoma (LGEAC), as well as clear cell carcinoma (CCC) derive from endometriosis, particularly endometriotic cyst/s.
O’Neill et al.127 reported that p53 mutations are more commonly found in high-grade serous tumors than in low-grade serous tumors; p53 is a tumor suppressor gene located in the short arm of chromosome 17 and is the most frequent genetic abnormality found in cancer.104 The type I tumors often present (60%)128 somatic mutations of KRAS, BRAF, PIK3K and HERBB2 plus other signaling molecules such as PTEN and CTNNB1 (b-catenin).
Type II tumors, contrary to type I tumors, develop rapidly, are very aggressive and are high-grade from the beginning, they do not have a benign BOT or APOTs, and low-grade carcinoma as precursors. Most type II tumors (75%) are high-grade serous neoplasms. This group also includes malignant mixed mesodermal tumors or carcinosarcoma (a rare tumor which is more commonly diagnosed today because better sampling and better microscopic recognition of small areas of sarcomatous component) and undifferentiated carcinomas.129 Researchers used to believe that high-grade neoplasms originated from the surface ovarian epithelium. Recent data suggest that many of those tumors arise from intraepithelial carcinomas located in the fimbriae of the fallopian tubes. Type II neoplasms are characterized by TP53 mutations and they lack mutations of KRAS, BRAF, or ERBB2. High-grade serous carcinomas appear to arise through type II pathway very rarely by pathway I.130
While studying high-grade serous carcinomas, Puls et al.131 found histologically, areas of transition from benign epithelium equal to that of a cystadenoma, atypical epithelial changes similar to BSOTs or APSOTs , and, finally, changes of frank high-grade serous carcinoma in 47% of the cases, suggesting an stepwise evolution from benign to frankly malignant serous carcinoma. Other investigators53 speculated that those benign appearing cells found in malignant tumors are nothing more than maturation of the malignant epithelia and are not benign epithelia. Others132, 133, 134 have shown evidence, although in rare cases, that small foci of high-grade serous tumors, in situ and de novo have been found in ovaries that appear otherwise grossly normal. These histologic observations give support to the theory that malignant tumors develop de novo in the ovary without starting from a benign cystadenoma, passing through SBOTs or APSOTs, to a low-grade serous adenocarcinoma, then to a moderately differentiated carcinoma, and finally, transforming into a poorly differentiated tumor, both of these two theories may be possible.
A recent study suggests the possibility that low-grade serous carcinoma originates from implantation of epithelium exfoliated from the fimbriated end of the fallopian tube onto the ovarian surface, or from cells originating in the fimbriated end of the Fallopian tube end falling into defects created on the surface epithelium by ovulation or other disruptions such as inflammatory processes with subsequent acquisition of KRAS or BRAF, or perhaps other mutations in the transplanted epithelium, resulting in the transformation to serous borderline tumors.135
KRAS and BRAS
Mok et al.136 first reported KRAS mutations in SBOTs or APSOTs. Other investigators137, 115, 138, 139 have confirmed the findings, additionally reporting that mutations of KRAS and BRAF are characteristic or typical of the following neoplastic processes: SBOTs or APSOTs, MPSBOTs or AMPSOTs and MPSBOTs or AMPSOTs with stromal microinvasion, and invasive micropapillary serous carcinoma also designated as low-grade serous carcinomas.
In approximately 86% of cases the type of KRAS and BRAF mutations found in the cystadenoma and the adjacent SBOT or APSOT are identical, suggesting that the mutations are early stages in tumor initiation, similar to what has been observed in melanoma and colorectal carcinoma.140, 141 The results of several studies have suggested that serous cystadenomas are precursors of SBOTs or APSOTs. The fact that the mutations are more frequent in cystadenomas than in SBOTs or APSOTs and that mutations in SBOTs or APSOTs are relatively uncommon, suggests that only a small number of cystadenomas have the potential to progress to SBOTs or APSOTs.142
If KRAS is present, BRAF is absent or vice versa, they are probably mutually exclusive. The KRAS and BRAF mutations occur in approximately 66% in SBOTs or APSOTs and low-grade serous carcinomas.137, 115, 143 The mutations of KRAS and BRAF can occur in either codon 12 or 13 and codon 599, respectively. Mutations of KRAS and BRAF in SBOTs or APSOTs and low-grade serous carcinoma are in contrast to the absence of these types of mutations in high-grade serous carcinomas. It has been suggested that mutations of KRAS and BRAF are involved in early tumor development but the degree of mutation is insufficient for complete malignant transformation to occur. Mutations occur in both genes, which appear to indicate that both have similar effect in tumor development.144
BRCA1 and BRCA2
BRCA1 and BRCA2 are tumor suppressor genes that down regulate the cell cycle; when they mutate, they lose their inhibitory effect on cell proliferation, consequently, contribute to abnormal cell multiplication.
Patients with clinical syndromes characterized by the presence of mutations of BRCA1 and BRCA2 show increased susceptibility to breast and ovarian cancer, mainly due to germline mutation in BRCA1 gene. Additional organs susceptible to the development of cancer include colon, endometrium, cervix, fallopian tube, and peritoneum.
The BRCA2 syndrome has been associated with increased susceptibility to early onset breast cancer, male breast cancer, and to a lesser degree, pancreatic and ovarian cancer. Cancers associated with BRCA1 and BRCA2 mutations represent the majority of what is known as familial related ovarian cancer that accounts for a very small fraction (7–10%) of all ovarian epithelial tumors.144,
Among the tumors associated with BRCA1 and BRCA2 mutations, the most common is the papillary serous carcinoma. The two genes are autosomes; therefore they can be inherited from the mother or the father. Carriers of either one of the two genes can develop cancer at an early age, although, more often with BRCA1. In relation to BRCA1 and BRCA2, mucinous tumors are underrepresented according to several studies.145, 146, 147, 148, 149, 150, 151 The frequency of BRCA1 and BRCA2 occurring in clear cell carcinoma is similar to that of sporadic cases. Some investigators145, 146, 148, 149, 152, 153 have reported that BRCA1 and BRCA2 associated tumors are of higher grade and higher stage than their matched controls.146, 147, 149, 150, 151, 152 The researchers just cited found that all the BRCA1 associated tumors were of advanced stage (stage II–III), but only about 50% were poorly differentiated, as were also the cases without mutations. Johansson et al.148 did not find differences between the cases with BRCA1 mutations carriers and the control population. It has been reported that ovarian cancers occurring in BRCA1 carriers have a somewhat better prognosis154 but it is uncertain whether this is because of the bias in carrier detection in this population or whether they are more sensitive to treatment. If they were more sensitive to treatment, this would refer to platinum therapy, which has been reported prior to the use of taxanes. It is known that high-grade serous carcinoma does not have a precursor component,19 while low-grade serous carcinoma often have a SBOT or APSOT which is frequently a micropapillary type as already mentioned.59, 155, 156
Loss of heterozygosity
SBOTs or APSOTs have loss of heterozygosity (LOH) on chromosome Xq and exhibit microsatellite instability. The high-grade serous carcinoma has p53 mutations and LOH of multiple chromosomes but does not show microsatellite instability.156, 117
It has been published157 that few cases of SBOTs or APSOTs have recurred as high-grade serous carcinomas after surgical treatment. This appears to indicate the possibility that a high-grade serous carcinoma could have been missed at the time of the original diagnosis due to sampling error or, that there is another pathway (separate from the two classical types previously mentioned) for the high-grade serous carcinoma or perhaps that they coexist.
Gilks and associates158 using global mRNA with the hope of obtaining knowledge about the mechanism involved in ovarian carcinogenesis, were able to distinguish SBOTs or APSOTs from high-grade serous carcinoma. This sophisticated molecular biology technique is very promising and it is anticipated that the method will clarify the different pathways involved in tumor development, improve accurate diagnosis, and more importantly, will enhance patient treatment and outcome.
Molecular pathology of serous tumors
- Carriers of BRCA1 and BRCA2 mutations are associated with the familial ovarian cancer (7–10%)
- KRAS and BRAF mutations are present in 66% in cystadenomas, SBOTs or APSOTs and low-grade adenocarcinomas, and are involved in early carcinogenesis
- KRAS and BRAF mutations are mutually exclusive
- KRAS mutation: codon 12 and 13; BRAF mutation codon 599
- LOH occur in chromosome Xq in BOTs or APOTs
- Microsatellite instability is present in BOTs or APOTs
- Global mRNA expression may be useful in separating high-grade serous carcinoma from SBOTs or APSOTs.
PERITONEAL OR EXTRAOVARIAN SEROUS BORDERLINE TUMORS/ATYPICAL PROLIFERATIVE EXTRAOVARIAN SEROUS TUMORS
Peritoneal or serous extraovarian borderline tumors (PEOSBTs) or atypical proliferating extraovarian peritoneal serous tumors (APEOSTs) are usually associated with widespread involvement of peritoneal and omental surfaces and with minimal or no ovarian presence of atypical epithelium, as occurs in the high-grade extraovarian serous carcinoma. The ovaries in patients with PEOSBTs or APEOPSTs are of normal size and may show very small serosal lesions or no lesions at all. This form of presentation could simulate a diffuse malignant mesothelioma or peritoneal carcinomatosis of other origin.
Most of the patients with PEOSBTs or APEOPSTs are women of childbearing age. In the series of 25 cases reported by Bell and Scully,18 the patients were between 19 and 53 years of age with a mean of 31 years, which is similar to the age reported in other series.102, 103 The majority of cases are asymptomatic; they are often diagnosed when surgery has been performed for other pelvic or abdominal pathology. The most important symptoms were infertility and abdominal pain. Rarely these tumors are suspected on a Pap smear, when this occurs, the suspicion comes from the presence of papillary clusters or psammoma bodies on the cytology preparation. On inspection of the abdominal and pelvic cavities, fibrous adhesion, fine granularity or large nodules are seen especially in the pelvic cavity. Most of the lesions are less than 0.6 cm in diameter. The peritoneal surface appears congested and ascitic fluid is present.
Microscopically the tumors are very similar to noninvasive implants, either epithelial or desmoplastic. The epithelial component is usually represented by fine or broad papillary structures on the peritoneal surface, less frequently on invaginations under the mesothelium forming a microscopic cystic space lined by a single layer of cuboidal or flat cells. As observed on the ovarian tumor, cuboidal or polygonal cells with mild atypia cover the papillary structures or the extraovarian tumor nodules. Mitoses are sporadic and are normal. As in the lumen of the ovarian cystic lesions, detached papillary fragments and tufting are seen often nearby, associated with psammoma bodies and single epithelial cells.
Structurally the desmoplastic extraovarian lesions are depicted by marked stromal reaction elicited by the presence of tumor cells. The extraovarian tumors are plastered on the mesothelial surface without underlining tissue invasion; they are rarely associated with endosalpingiosis. The boundaries of the lesions are well demarcated. The epithelial component is characterized by the presence of papillae, glands, and small irregular clusters of cells as occur in peritoneal implants. Psammoma bodies frequently accompany the epithelial component of the tumor. The stroma is formed by dense fibrous tissue that is commonly accompanied by acute inflammatory exudate.
Lesions can be identified on the mesothelium as well as on the fibrous tissue surrounding the adipose tissue lobules of the omentum.
The patient outcome is very similar to that of SBOTs or APSOTs without invasive implants; patient survival is around 95%. This good survival rate indicates that these patients should be treated conservatively, in other words, preserving ovaries, fallopian tubes and uterus to maintain fertility when desired.18 It is intriguing that there is a high percentage (75–85%) of endosalpingiosis associated with EOPSBTs or APEOPSTs.18, 159 Do these tumors originate from endosalpingiosis which sometimes has been found to be atypical, or do they just represent synchronous lesions? This question remains to be answered. Lesions similar to those just described but with deep tissue invasion (invasive peritoneal implants) are found and correspond to low-grade serous extraovarian carcinoma. It should be remembered that some high-grade serous carcinomas might have moderate nuclear atypia, but are associated with destructive stromal reaction or vascular space invasion.
PSAMMOCARCINOMA, PSAMMOMATOUS VARIANT OF PAPILLARY SEROUS CARCINOMA
Psammocarcinoma is a rare variant of low-grade serous carcinoma with an indolent course, and a survival rate of 98–99%. The tumors are unilateral in 95% of the cases.13 The patients mean age has been reported as 40, 50 and 59, this variation is due to the small number of cases published. Most of these tumors are asymptomatic; the most common symptoms are abdominal mass, abdominal and/or pelvic pain, and infertility, many are discovered during abdominal or pelvic surgery for other pathology. These neoplasms might arise either on the ovary or on the peritoneal surface. The neoplasm can involve the wall of the bowel, the myometrium, the omentum, and the fallopian tubes. When the tumor arises on the ovary, the average size is 11 cm in diameter, the surface is usually smooth, and on cut section there are cystic and solid areas. The solid areas are gritty or granular; it gives the sensation of cutting through a bag of sand when examining the specimen. Nodules represent the extraovarian lesions; they range from few millimeters to 2.5 cm in diameter. A single layer of cuboidal cells exhibiting mild to moderate atypia surrounds the psammoma bodies, they psammoma bodies may appear separate or fused and are often laminated (Fig. 9).
In order to make a diagnosis of psammocarcinoma the following histological features most be observed:
1. Destructive invasion of ovarian stroma, vascular invasion, or, in the extraovarian cases, invasion of intraperitoneal viscera;
2. No more than moderate nuclear atypia;
3. No areas of solid epithelial proliferation except for occasional nests with no more than 15 cells in diameter;
4. At least 75% of the papillae or nests should be associated with or completely replaced by psammoma bodies;160
5.
Mitoses are very rare, usually no more than 1/10 HPF. 57,
MUCINOUS BORDERLINE OVARIAN TUMORS/ ATYPICAL PROLIFERATIVE MUCINOUS OVARIAN TUMORS
Several mucinous neoplastic processes can arise in the ovaries; they are mucinous cystadenoma, cystadenoma with focal epithelial proliferation, mucinous borderline ovarian tumors (MBOTs) or atypical proliferative mucinous tumors (APMOTs); MBOTS or APMOTs with intraepithelial carcinoma and/or microinvasion, and mucinous adenocarcinoma. This sequence suggests that these neoplasms develop in a stepwise fashion. They go from mucinous cystadenoma to mucinous cystadenocarcinoma, passing through cystadenoma with atypia, continuing to MBOTs or APMOTs, and few arriving to mucinous carcinoma. Because of the use of more stringent diagnostic criteria, MBOTs or APMOTs and mucinous carcinoma of the ovary are a lot less prevalent now than previously believed. Many of the mucinous tumors found in the ovary are metastases from tumors in other parts of the gastrointestinal tract, particularly the vermiform appendix. Patients with mucinous ovarian carcinomas are around 50 years of age, while patients with MBOTs or APMOTs and cystadenomas are teenagers and young adults. The symptoms of individuals with mucinous tumors are similar to those of other types of ovarian tumors. However, mucinous tumors are the most common of the nonendocrine-cell ovarian tumors to produce manifestation secondary to hormonal secretion, especially of estrogenic and androgenic nature. MBOTs or APMOTs are characterized by the presence of unilocular or multilocular cystic masses; their size fluctuates between few centimeters and 30 cm with a mean of about 20 cm (Fig. 24). Close to 95% of the cases are unilateral with mucus secreting epithelium lining the lumen, infoldings, and glandular structures. They represent almost 5% of all ovarian surface epithelial tumors70 and 40–50% of all nonbenign mucinous ovarian tumors. Mucinous tumors are more common in Japan than in USA and Europe. In 1988, the MBOTs or APMOTs were divided into intestinal or enteric type; this type of tumor is the most common, accounting for 80–85%161 of the total. The endocervical-like mucinous (Müllerian) type tumors represent from 15% to 20%.
INTESTINAL OR ENTERIC MUCINOUS BORDERLINE OVARIAN TUMORS/ATYPICAL PROLIFERATIVE INTESTINAL OR ENTERIC-TYPE MUCINOUS OVARIAN TUMORS
At present, it is known that the survival rate of patients with enteric or intestinal MBOTs or APMOTs is 99–100% when the tumors are confined to the ovary;162, 163, 164 this is in contrast to a survival rate of only 40–50% when the tumors are of an advanced stage. These figures were obtained after examining a large number of patients.163 Riopel and associates165 reviewed the diagnostic criteria and behavior of enteric or intestinal mucinous tumors and concluded that close to 85% of the mucinous tumors reported with advanced stage were associated with pseudomyxoma peritonei (PMP) usually secondary to a ruptured cystadenoma of the appendix in the peritoneal cavity.166 The remaining 15% of PMP are probably from pancreatic, biliary tract and uterine cervix origin. These figures suggest that borderline intestinal mucinous ovarian tumors (IMBOTs) or APIMOTs do not present with peritoneal implants. Mucinous ovarian tumors that have ruptured at the time of surgery are not associated with the development of PMP. It can also be stressed that the IMBOTs or APIMOTs are very benign neoplasms that probably never spread beyond the ovary.
Studies undertaken in the past decade have shown that about 75% of mucinous ovarian tumors are metastatic rather than primary,52, 167 this is because more strict criteria are use for the diagnosis. Consequently, primary IMBOTs or APIMOTs are a lot less frequent than previously thought.
As in SBOTs or APSOTs, and any other borderline ovarian tumor, IMBOTs or APIMOTs should have at least 10% of the tumor area involved by atypical proliferating intestinal or enteric type epithelium. Patients’ age varies from 9 to 70 years, with a mean age of 35 years.168, 169 Most of the patients are in the fifth or sixth decade. The tumors bilateralism is about 6%; 80–90% are stage I at the time of diagnosis,165, 170 Primary mucinous tumors of the ovary are in general large, with a mean diameter of 20 cm (Figs. 23 and 24). This is in contrast to metastatic mucinous tumors, which are on average about 10 cm (Fig. 26) in diameter with the exception of those of pancreatic, and small and large bowel origin that produce large and cystic tumors. The tumors of pancreatic and cervical origin are difficult to differentiate morphologically from primary mucinous ovarian tumors, as all these tumors are very similar morphologically.165, 167, 170
Fig. 23.
Gross photograph of an intestinal type MBOT. Observe multiple nodules, a collapsed hemorrhagic cyst on the lower left hand corner representing endometriosis. On the upper left-hand corner the dilated Fallopian tube. Prominent blood vessels are evident.
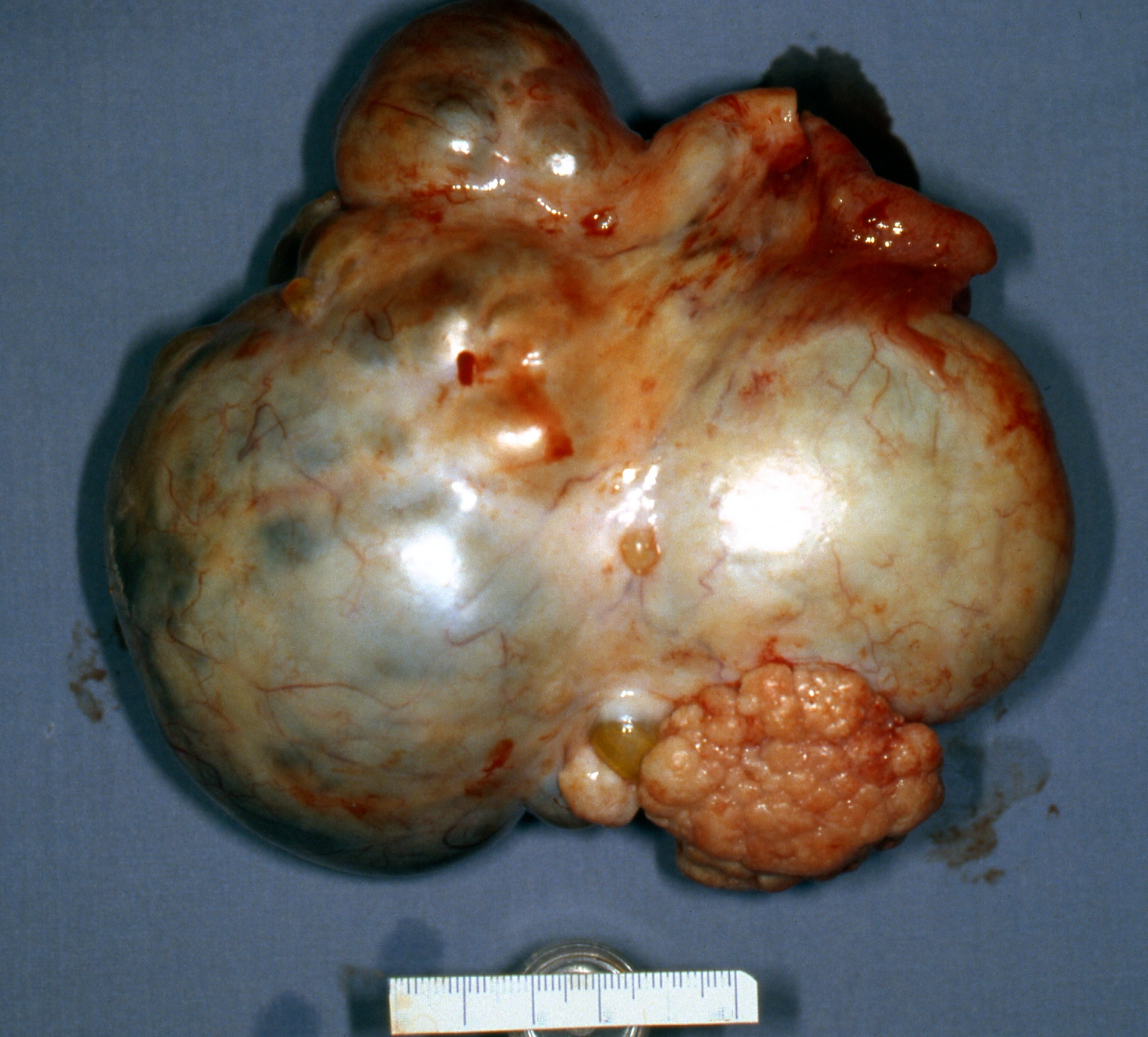 Fig. 24. Gross photograph of an MBOT or APMOT with exophytic papillary growth visible at the upper right-hand corner. In the upper part of the tumor between the two large lobules a thick yellow area is noted and corresponds to ovarian tissue. A segment of the Fallopian tube is present at the upper right-hand corner.
Fig. 24. Gross photograph of an MBOT or APMOT with exophytic papillary growth visible at the upper right-hand corner. In the upper part of the tumor between the two large lobules a thick yellow area is noted and corresponds to ovarian tissue. A segment of the Fallopian tube is present at the upper right-hand corner.
 Fig. 25. Photograph of an open seromucinous BOT. The lumen shows a central yellow-brown papillary area and small clear papillae nearby; the other areas exhibit hemorrhage within the wall that could be associated with endometriosis.
Fig. 25. Photograph of an open seromucinous BOT. The lumen shows a central yellow-brown papillary area and small clear papillae nearby; the other areas exhibit hemorrhage within the wall that could be associated with endometriosis.
 Fig. 26. Photograph of bilateral borderline mucinous cystic adenofibroma-like tumors. The tumors are less than 10 cm in diameter. The inner lining is smooth and shows focal areas of dense fibrosis separating small cysts. The cysts contents were clear mucinous material. (Courtesy of Dr Richard Stock.)
Fig. 26. Photograph of bilateral borderline mucinous cystic adenofibroma-like tumors. The tumors are less than 10 cm in diameter. The inner lining is smooth and shows focal areas of dense fibrosis separating small cysts. The cysts contents were clear mucinous material. (Courtesy of Dr Richard Stock.)
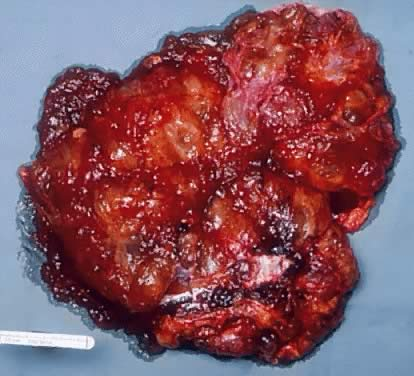 Fig. 27. Photograph of a mucinous BOT enteric or intestinal type showing nodules and lobules, a collapsed hemorrhagic cyst on the left hand corner representing endometriosis. A dilated Fallopian tube is present at the upper left hand corner; prominent blood vessels are evident.
Fig. 27. Photograph of a mucinous BOT enteric or intestinal type showing nodules and lobules, a collapsed hemorrhagic cyst on the left hand corner representing endometriosis. A dilated Fallopian tube is present at the upper left hand corner; prominent blood vessels are evident.
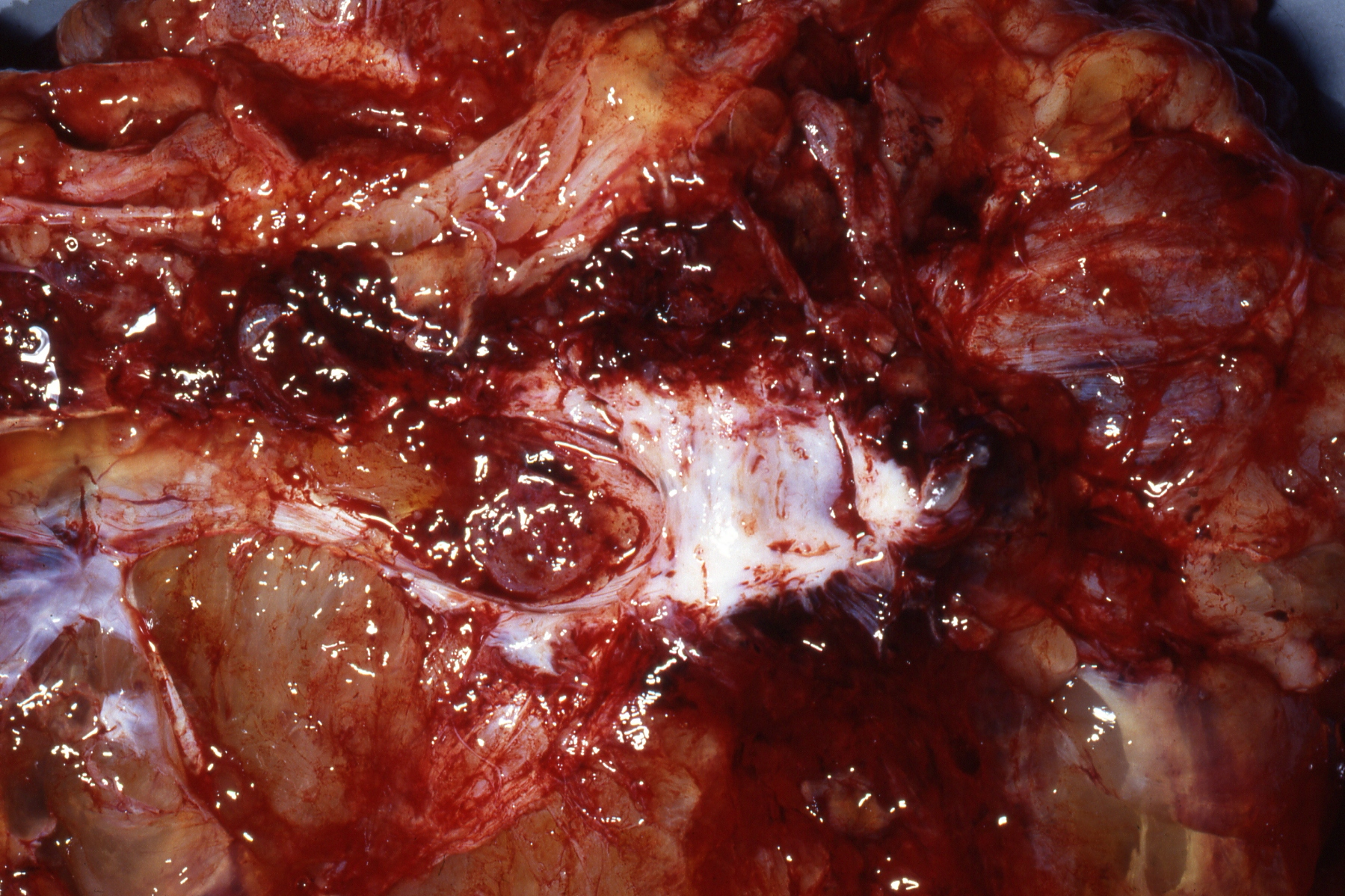 Fig. 28. Gross photograph of the tumor depicted above (Fig. 27) exhibiting cystic cavities of different sizes filled with hemorrhagic mucinous material. A dense fibrous area is noted in the center.
Fig. 28. Gross photograph of the tumor depicted above (Fig. 27) exhibiting cystic cavities of different sizes filled with hemorrhagic mucinous material. A dense fibrous area is noted in the center.
Some mucinous enteric or intestinal-type tumors have a germ cell origin. They develop from the intestinal or enteric-type epithelium found in a cystic teratoma. The rest of the tumors are supposed to have an origin in the mucinous neometaplasia of the ovarian epithelium.171 Mucinous tumors found in the ovary are often confused with metastatic mucinous tumors to the ovary, which commonly mimic a primary mucinous ovarian tumor.53, 172, 173
Grossly the IMBOTs or APIMOTs are characterized by masses that are most often unilateral. Their size varies but is normally more than 12 cm in diameter and can reach huge sizes and weigh many kilograms. Mucinous tumors are as a rule, larger than the metastatic tumors that frequently mimic a primary mucinous ovarian tumor. They are similar to cystadenomas and to some mucinous cystadenocarcinomas.172, 174 In general the mucinous tumors are multilocular, the external surface could be smooth or show fibrous adhesions (Figs. 23–25 and 27). The blood vessels on the cyst wall may be prominent. The cyst wall could be thin, thick and fibrous, exhibiting yellowish pink or blue color. The lumen contains serous, mucinous or seromucinous straw-colored and sometimes hemorrhagic fluid. The inner surface of the tumor could be smooth or show arborescent structures that are soft and delicate, although, sometimes they can be solid or firm when associated with a cystic adenofibroma component, or formed by closely packed small cysts (Fig. 28). The solid areas should be meticulously examined to exclude the possibility of invasion or the presence of a mural nodule of anaplastic carcinoma.
Microscopically, the epithelium lining the cyst cavities or the papillary structures is often a mixture of different mucinous cell types, including intestinal, gastric, endocervical, argyrophilic and rarely Paneth cells (Figs. 28, 29 and 33). This mixture of mucinous secreting-type cells, as expected, could result in a difficult task in determining accurately the tumor type. Cellular atypia of various degrees is seen. When severe atypia is present, the focus with severe atypia should be considered as an intraepithelial carcinoma. We emphasize that because of the presence of multiple degrees of atypia including carcinoma in mucinous tumors, a meticulous gross examination and generous sampling are strongly recommended.
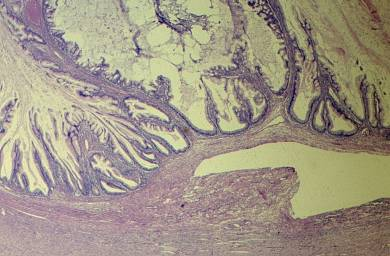 Fig. 29. Photomicrograph of a cervical-like mucinous BOT. The atypical epithelium is represented by tall columnar mucus secreting cells forming papillae and infoldings. Thick clear mucinous material fills the lumen (hematoxylin and eosin).
Fig. 29. Photomicrograph of a cervical-like mucinous BOT. The atypical epithelium is represented by tall columnar mucus secreting cells forming papillae and infoldings. Thick clear mucinous material fills the lumen (hematoxylin and eosin).
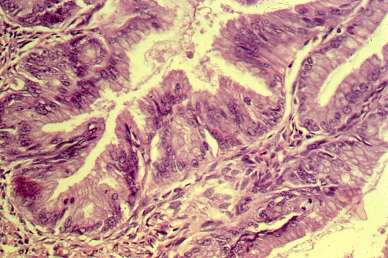 Fig. 30. Higher magnification of the above image (Fig. 29) showing hyperchromatic nuclei, cribriform architecture, and frequent mitotic figures (hematoxylin and eosin).
Fig. 30. Higher magnification of the above image (Fig. 29) showing hyperchromatic nuclei, cribriform architecture, and frequent mitotic figures (hematoxylin and eosin).
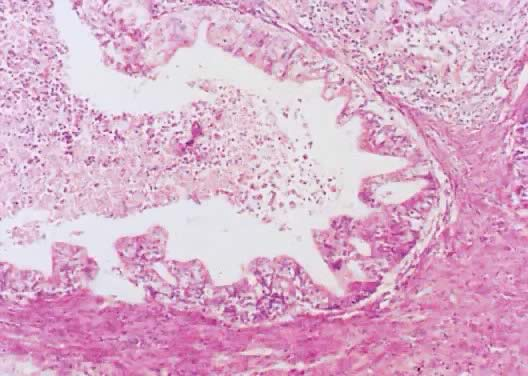 Fig. 31. Photomicrograph of a cervical-like mucinous BOT showing atypia, mitoses, and focal cribriform architecture. The lumen contains cellular debris and mucus material (hematoxylin and eosin).
Fig. 31. Photomicrograph of a cervical-like mucinous BOT showing atypia, mitoses, and focal cribriform architecture. The lumen contains cellular debris and mucus material (hematoxylin and eosin).
 Fig. 32. Higher magnification of the above cervical-like mucinous BOT (Fig. 31) showing epithelial stratification, nuclear atypia, and mitoses. Cellular debris is seen in the cyst lumen (hematoxylin and eosin).
Fig. 32. Higher magnification of the above cervical-like mucinous BOT (Fig. 31) showing epithelial stratification, nuclear atypia, and mitoses. Cellular debris is seen in the cyst lumen (hematoxylin and eosin).
The atypical epithelial lining cells are stratified in two or three layers (Figs. 30, 33 and 36). Papillary tufts are rare, the mitotic activity can be mild to moderate, and if present the mitosis should be of normal type. Often the atypical lining epithelium is mixed with benign-looking mucus secreting cells of gastric or cervical type.174 For some investigators174 the presence of stratification should be considered an indication of poor prognosis. This point has been substantiated. Furthermore, determining stratification is often complicated because histologic sections are not properly oriented. Glandular formation is seen as well as multiple epithelial infoldings, which may mimic stromal invasion. Cribriform structures, or glands within glands are often present (Figs. 30, 33, 35 and 36); if they are observed in the tumor stroma they should be interpreted as evidence of invasion. If the cribriform or glandular structures are less than 5 mm in linear dimension, by definition, they should be diagnosed as microinvasion, but if they are more than 5 mm, the tumor would be diagnosed as mucinous carcinoma.
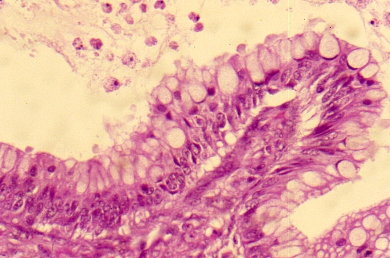 Fig. 33. Photomicrograph of an area of an enteric or intestinal mucinous tumor showing a short papilla with numerous goblet cells with stratification and nuclear atypia. The lumen shows cellular debris (hematoxylin and eosin).
Fig. 33. Photomicrograph of an area of an enteric or intestinal mucinous tumor showing a short papilla with numerous goblet cells with stratification and nuclear atypia. The lumen shows cellular debris (hematoxylin and eosin).
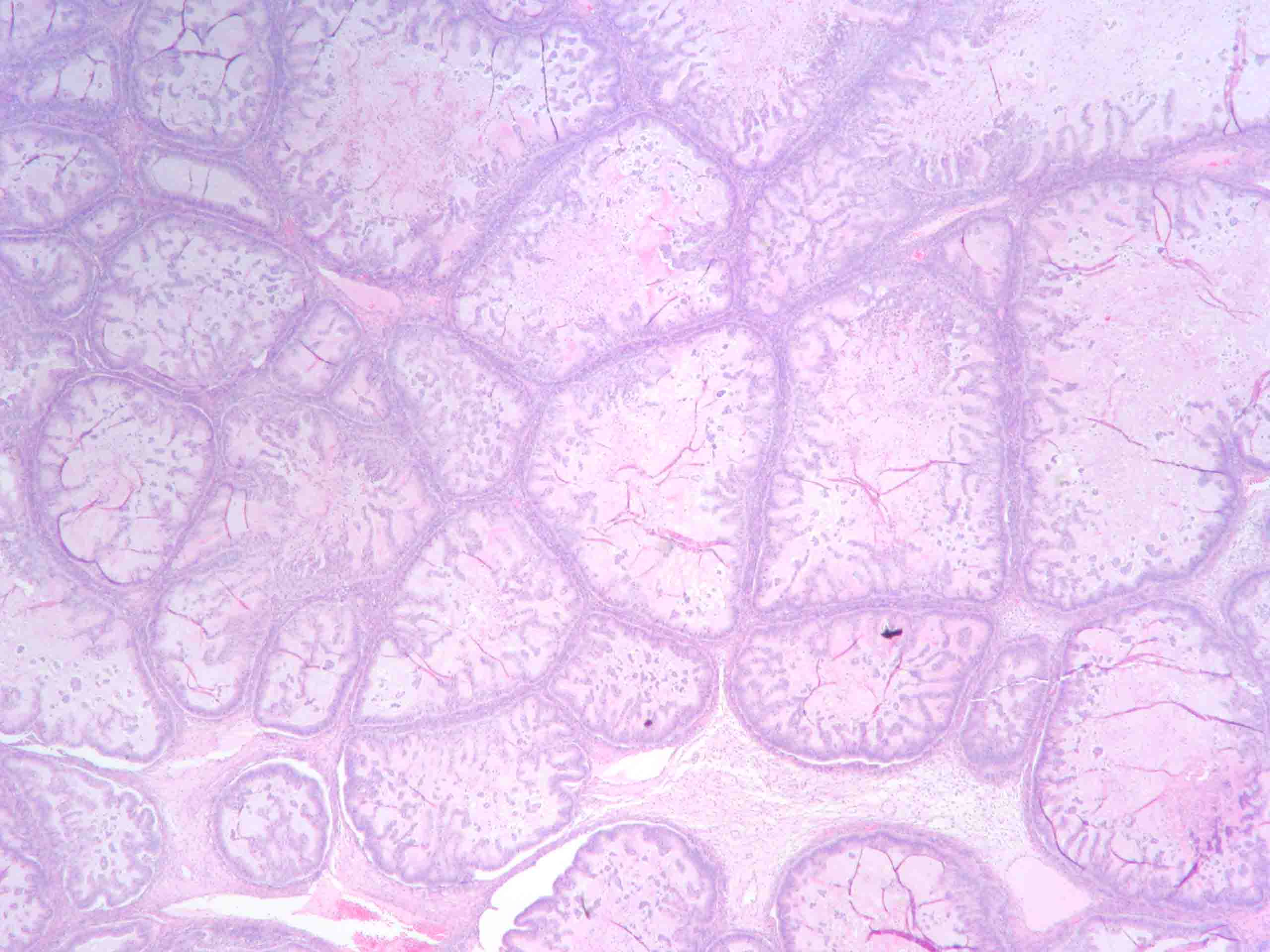 Fig. 34. Photomicrograph of mucinous BOT with multiple small cysts with mucus material in their lumens (hematoxylin and eosin).
Fig. 34. Photomicrograph of mucinous BOT with multiple small cysts with mucus material in their lumens (hematoxylin and eosin).
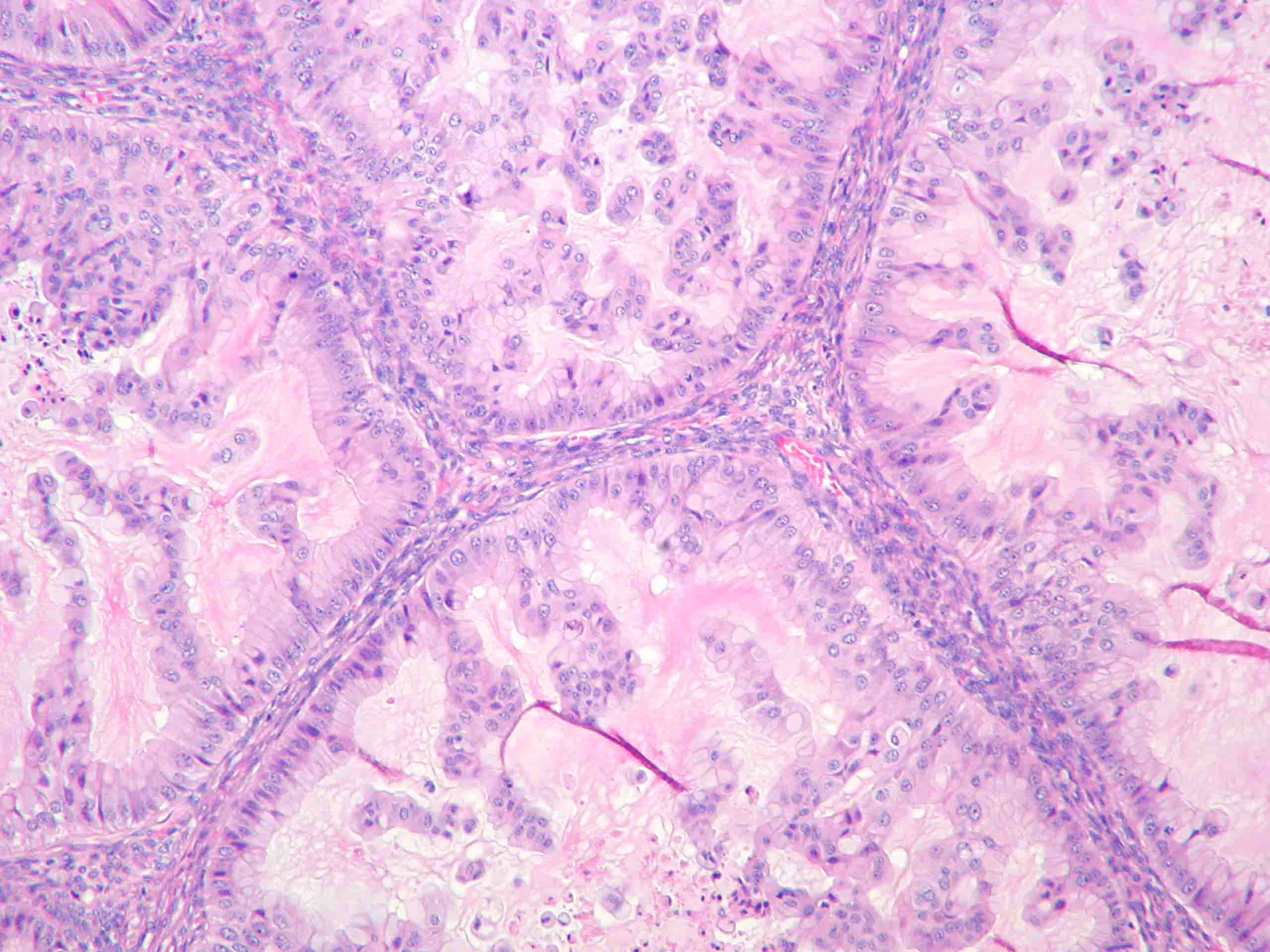 Fig. 35. Higher magnification of the above photomicrograph (Fig. 34) showing micropapillae and mucus material in the lumens (hematoxylin and eosin).
Fig. 35. Higher magnification of the above photomicrograph (Fig. 34) showing micropapillae and mucus material in the lumens (hematoxylin and eosin).
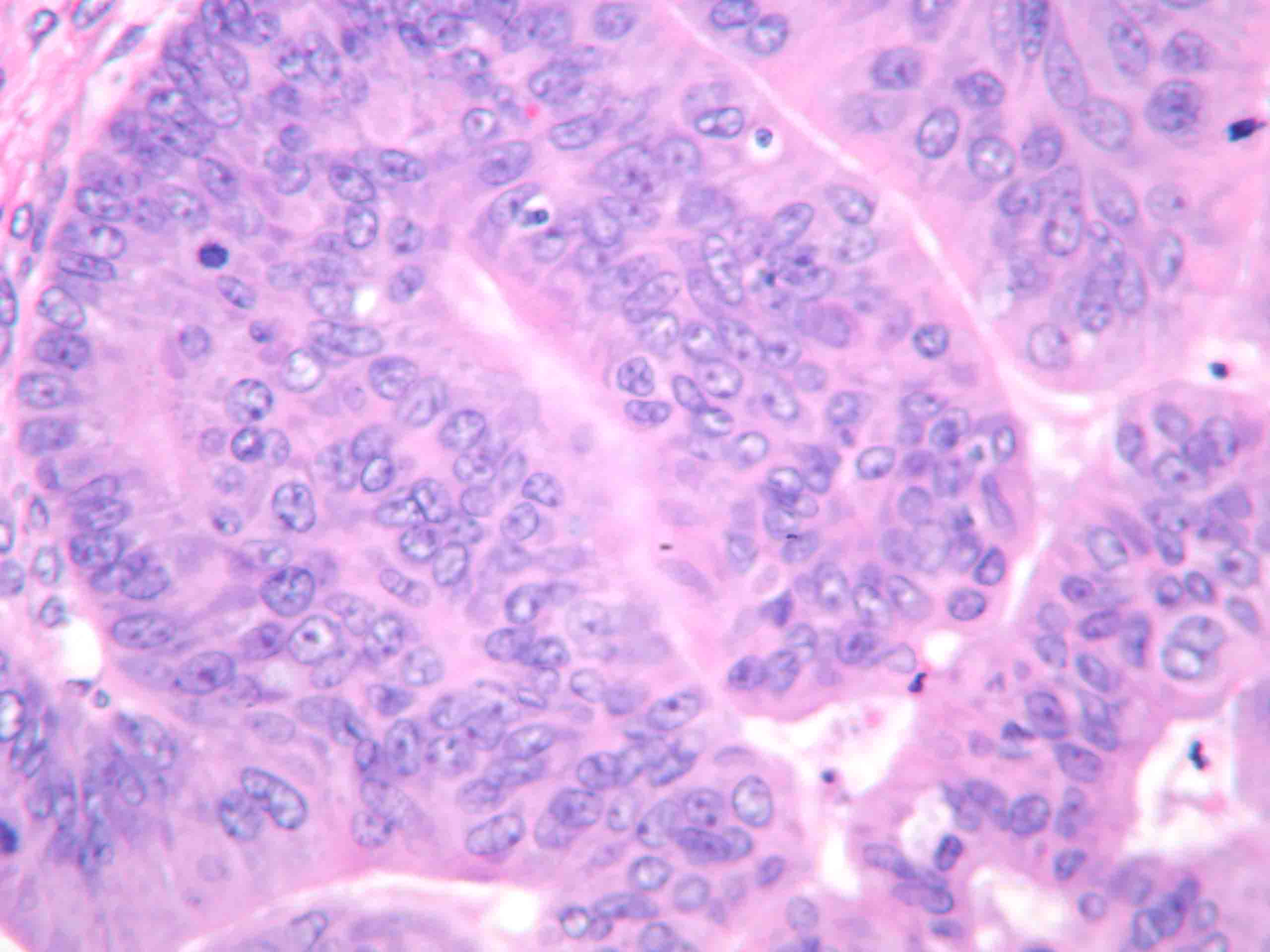 Fig. 36. Intestinal type mucinous tumor; higher magnification of Fig. 35 showing stratification, papillary and cribriform architecture with cellular atypia and mitoses (hematoxylin and eosin).
Fig. 36. Intestinal type mucinous tumor; higher magnification of Fig. 35 showing stratification, papillary and cribriform architecture with cellular atypia and mitoses (hematoxylin and eosin).
As with other BOTs or APOTs, destructive invasion is the most important criterion to separate BOTs or APOTs from frank invasive carcinoma. The evaluation of destructive invasion is particularly more difficult in mucinous than in serous tumors. This potential problem is due to the presence of epithelial infoldings and tumor stroma that simulates ovarian stroma. Hart and Norris175 suggested that when the stratification is more than four layers, the tumor should be diagnosed as carcinoma in situ. Later, high-grade cytologic atypia, the presence of cribriform architecture or of solid atypical epithelial nests or papillary structures devoid of supporting stroma, were added to the four layers of stratification as part of the criteria for the diagnosis of carcinoma in situ.24 After evaluation of multiple series using the different criteria for diagnosis, it was found that there were no changes in patient outcome and that tumors with the characteristics just described should be designated as IMBOTs or APIMOTs with intraepithelial carcinoma.
Intestinal or enteric mucinous borderline tumors (IMBOTs or APIMOTs)
- Patient mean age 35 years, range 9–70 years
- Few tumors are of germ-cell origin
- 75% are supposed to be metastatic
- 95% unilateral, 20 cm mean size
- 80–95% are MBOTs or APMOTs
- 98–99% patient survival when stage I
- 40–50% survival if advanced stage
- No PMP if mucinous ovarian tumors are ruptured at surgery
- Primary ovarian tumors probably never spread beyond the ovary.
ENDOCERVICAL-LIKE (Müllerian) MUCINOUS BORDERLINE OVARIAN TUMORS/ATYPICAL PROLIFERATIVE ENDOCERVICAL-LIKE MUCINOUS TUMORS
Endocervical-like mucinous borderline ovarian tumors (E-LMBOTs) or atypical proliferative endocervical-like mucinous tumors (APE-LMOTs) represent approximately 5–14% of all MBOTs or APMOTs and are the second most common MBOTs or APMOTs.162 The endocervical subtype of E-LMBOTs or APE-LMTs is clinically and pathologically closely related to SBOTs or APSTs with which are often mixed (seromucinous) E-LMBOTs or APE-LMOTs. Endocervical type mutinous tumors are distinct from the enteric type, they are less common, frequently bilateral, smaller in diameter than the enteric or intestinal type tumors, unilocular, and found in younger women, and show acute inflammatory infiltrate in the stroma as reported by Rodriguez et al. and others.63, 176, 177 These tumors were first described by Rutgers and Scully178 and are infrequently unilateral tumors, with an average diameter of 13 cm.177 The patients in the Rodriguez series176 had an age range of 22.5–75.2 years with a mean of 40 years. The symptoms were similar to those of all BOTs or APOTs. Thirty five per cent of them were associated with endometriosis. They possibly could be associated with peritoneal implants.
On gross exam the E-LMBOTs or APE-LMOTs show one or few locules (Figs. 23 and 24) filled with clear or viscous material. Papillae are present in their lumen/s and they are lined by mucinous endocervical-type epithelium with more epithelial proliferation than is seen in a cystadenoma (Figs. 28 and 32). Approximately 80% of the patients are stage I at the time of diagnosis. The remaining are usually stage II and III with peritoneal implants and lymph nodes (deposits) metastasis.178 In more than half of the E-LMBOTs or APE-LMOTs, a transition between benign epithelium and E-LMBOT or APE-LMOTs was observed.
The outcome of the patients with this type of tumor appears to be excellent even when they have peritoneal implants, microinvasion, and lymph nodes deposits.58, 177, 179
Histologically, the epithelium is usually tall columnar or endocervical-like, sometimes alternating with polygonal eosinophilic and ciliated cells (seromucinous). The epithelial lining of the tumors is often stratified and the cells exhibit mild to moderate histological atypia, represented by basally located pleomorphic, hyperchromatic nuclei, and eosinophilic cytoplasm (Figs. 29, 30 and 32). The nuclei are usually grade I, or II, rarely grade III; nucleoli may be seen. Sometimes the epithelial proliferations are associated with focal necrosis but without destructive stromal invasion. The arborizing epithelium forms clusters or rosettes, some attached to the papillae and some detached resulting in tufting. Prominent fibrovascular cords that could be associated with marked edema represent the supporting stroma. Single cells are often found floating in the cyst fluid. The stroma of the papillae is frequently infiltrated by acute inflammatory exudates, which is also observed within the extracellular mucoid material. Occasionally, squamous or endometrial-type cells or components are found creating the so-called mixed cell type endocervical-like mucinous borderline tumors.
Endocervical-like mucinous borderline tumors or APE-LMTs
- Second most common MBOT or APMOT
- 10–15% of all MBOTs or APMOTs
- Often associated with endometriosis (35%)
- Small size and frequent bilateralism
- Often mixed with serous component (seromucinous)
- Bulbous papillae often edematous and infiltrated by PMNs also present in the mucus material
- Occasional squamous or endometrial cell component
- Excellent prognosis even in those cases with microinvasion or intraepithelial carcinoma.
ENDOCERVICAL-LIKE MUCINOUS BORDERLINE OVARIAN TUMORS WITH INTRAEPITHELIAL CARCINOMA/ATYPICAL PROLIFERATIVE ENDOCERVICAL-LIKE MUCINOUS TUMORS WITH INTRAEPITHELIAL CARCINOMA
The endocervical-like mucinous borderline ovarian tumors (E-LMBOTs) or atypical proliferating endocervical-like tumors (APE-LMOTs) with intraepithelial carcinoma are of two types. The first type could be identified as a tumor with a focus or foci of confluent epithelial growth with cribriform architecture in the cystic wall or in cellular papillae devoid of stroma.
The second type is characterized by epithelial proliferation with diffuse high-grade epithelial atypia (nuclear grade 3 or nuclear size greater than threefold), in other words, by cells with nuclear enlargement, pleomorphism, irregular nuclear membrane, coarse chromatin, and prominent nucleoli representing an intraepithelial carcinoma. Intraepithelial carcinoma was found in 13% of cases of E-LMBOT or APE-LMOTs. As mentioned in Rodriguez et al. report,176 cellular stratification is not necessary, since the lack of it is accepted as characteristic of E-LMBOTS or APE-LMOTs. In general, there is no size limit required of the type of alteration present to make the diagnosis of intraepithelial carcinoma;178 if we think that atypical changes are extensive, the lesion should be designated as endocervical type mucinous carcinoma. ( The author refers to "if changes are extensive,” but he does not define “extensive” nor does he define stratification, how large should be the endocervical-like epithelial component, or how many how many layers are sufficient for the diagnosis of carcinoma.)
ENDOCERVICAL-LIKE MUCINOUS BORDERLINE OVARIAN TUMORS WITH MICROINVASION/ATYPICAL PROLIFERATIVE ENDOCERVICAL-LIKE MUCINOUS TUMORS WITH MICROINVASION
Endocervical-like mucinous borderline ovarian tumors (E-LMBOTs) or APE-LMOTs with microinvasion are defined as tumors with one or more foci of single cells, irregular glands, or small nests of confluent mucinous cells infiltrating the underlying stroma. The dimensions of the microinvasive focus or foci should not exceed 10 mm2 in area (≤3 mm in each of the two linear dimensions).176 The authors pointed out that up to the present time, there is no established histological criterion for the diagnosis of microinvasion for this particular tumor; they use the criteria for the intestinal type tumor. According to the authors, the most frequent form of microinvasion was that of single cells found in the stroma of the papillae or under the epithelium. The focus of microinvasion is frequently multiple and measures 1 and 2 mm in linear dimension in most of cases. As mention earlier in this chapter, 5 mm has been the recommended size to diagnose microinvasion in all BOTs or APOTs. Up to this point, there is no unanimous agreement concerning the size for microinvasion.
Because these tumors are very rare, there are insufficient data concerning survival or recurrence. The survival, as described above, for intraepithelial carcinoma is around 95%. It is also consider that the metastatic potential is lowDIFFERENTIAL DIAGNOSIS OF MUCINOUS TUMORS
Tumors with mucus secreting epithelium developing in the ovary can be benign, MBOTs or APMOTs of borderline malignancy or atypical proliferating type, with microinvasion, with intraepithelial carcinoma, and high-grade or frankly malignant mucinous carcinoma.
In the presence of a mucinous tumor of the ovary, the first thing to consider is whether it is a primary or metastatic. Grossly, if the tumor is bilateral, is less than 10 cm in diameter, shows surface involvement, and is associated with pseudomyxoma peritonei or pseudomyxoma ovarii, there is a strong possibility that the tumor is metastatic.
It should be remembered that other ovarian tumors might be associated with mucinous epithelium, including Brenner tumors, Sertoli-Leydig cell tumors, and ovarian teratomas. Immunostains can be very helpful in establishing the origin of the neoplasm.
Grossly the mucinous tumors are in general very similar. Microscopically, the cystadenomas exhibit mucus-secreting epithelium that is most often of intestinal or enteric type, and of endocervical type. The mucus secreting cells in the mucinous cystadenomas have benign characteristics. Lesions designated as borderline or atypical proliferating, are characterized by the presence of papillary structures and proliferating atypical cells; mitoses are rare and appear normal. The mucinous adenocarcinoma is characterized by high-grade atypia and stromal invasion. Microscopically the separation of benign and frankly malignant tumors is not a problem in the majority of cases. The diagnosis of borderline or atypical proliferating tumors can be difficult at times, as it can be difficult to separate MBOTs or APMOTs from benign proliferative lesion or from carcinoma. Occasionally, the separation of serous and mucinous tumors can be challenging. It can also be challenging to determine whether the mucinous tumor is primary or metastatic to the ovary. This point is very important, and the real origin should be determined, during surgery, so the primary tumor can be localized and excised. Mucinous ovarian tumors have a gastrointestinal origin most of the time. Seidman et al.167 reported a simple rule that classified all bilateral mucinous tumors as metastatic, unilateral mucinous carcinoma ≤10 cm as metastatic, and unilateral carcinomas ≥10 cm as primary. Using this simple practical rule, the authors were able to correctly determine the primary or metastatic origin in 84% of the cases overall; including 100% of the primary ovarian tumors, and 77% of the metastatic mucinous tumors. The accurate separation of the tumors based on the origin, primary or metastatic, will assist in the appropriate surgical management of the patient. This algorithm has been modified by Yamelyanova and associates180 improving the accuracy by increasing the diameter of the tumors from 10 to 13 cm. The invasive and noninvasive nature of the tumors can be determined by measuring the size of invasion. If it is more than 5 mm the tumor should be classified as a frank invasive carcinoma. If the area of invasion is less than 5 mm, the tumor will be designated as MBOT or APMOT with microinvasion. Similar results can be achieved by determining whether the neoplasm in question has an atypical area that represents 10% or more of the total tumor neoplastic area. If it is <10% the tumor is a cystadenoma, perhaps with focal atypia, insufficient for the diagnosis of MBOT or APMT. Metastatic tumors to the ovary exhibit a nodular surface involvement, and the infiltration has a haphazard infiltrative pattern of the stromal. Mucinous tumors should be also differentiated from endometrioid adenocarcinoma with mucinous differentiation, Sertoli-Leydig cell tumors with heterologous elements such as gastrointestinal epithelium; these tumors are usually solid or soft. The neuroendocrine markers will be positive in the neuroendocrine cell.
The immunohistochemical markers CK7, CK20, and Dpc4 can be useful in the differential diagnosis of primary and metastatic mucinous tumors. Dpc4 is inactivated in approximately 55% of ductal pancreatic adenocarcinomas, while it is expressed in almost all mucinous primary ovarian carcinomas. Tumors of gastrointestinal origin may also express CEA and CA19.9 markers the levels of which may be very elevated in the patient serum too. The mucinous tumors of endocervical type express CA125, ER and PR, which are negative in intestinal-type neoplasms.52
MOLECULAR PATHOLOGY OF MUCINOUS TUMORS
Epidemiological studies181 indicate that there is a sequence in the development of mucinous ovarian cancer, starting with mucinous cystadenoma, continuing through MBOT or APMOT, and ending with mucinous invasive adenocarcinoma.182, 183, 184
It has been reported that the KRAS gene shows mutations on mucinous tumors similar to those described on serous tumors. The MBOTs or APSOTs have mutations more frequently than mucinous cystadenomas but less frequently than mucinous carcinomas. KRAS mutations increase from 14–56% in mucinous cystadenomas to 29–73% in MBOTs or APMOTs to 40–85% in mucinous carcinomas.136, 139, 162, 185, 186, 187, 188, 189 These findings support the view that invasive mucinous adenocarcinoma develops through a stepwise process from the benign cystadenoma to MBOT or APMOT, and ending in invasive carcinoma. The KRAS mutations in mucinous tumors have been found in areas with different histologic characteristics within the same tumor, in other words, in areas of cystadenoma, MBOT or APMOT, and adenocarcinoma.136, 185, 186
Several researchers have found an increase in the frequency of KRAS mutations in codons 12 and 13 in cystadenomas, MBOTs or APMOTs and mucinous carcinoma from 33% to 86%.136, 139 187, 188, 189 These findings indicate that KRAS is involved in early carcinogenesis and that the mucinous carcinoma has an origin in cystadenomas passing through MBOT or APMOT.
The pattern of gene expression of mucinous carcinoma can easily be distinguishable from serous, endometrioid and clear cell carcinoma. Ovarian mucinous tumors express the genes UC2, MUC3 and MUC17 that are typical of tumors of mucinous linage, primary or metastatic.190 Among other genes expressed by mucinous tumors is the CDx2, which is a very useful immunohistochemical marker to differentiate intestinal from non-intestinal mucinous tumors.
Microsatellite instability
Microsatellite instability (MSI) has rarely been reported in MBOTs or APMOTs except in the X chromosome.191 Microsatellite instability is defined as an alteration of the DNA reparatory genes. Consequently, cells with mutation in these genes cannot effectively repair the defects that build up during normal DNA replication. Sanz Casla et al.192 reported that MSI was not found in six microsatellites examined in benign ovarian tumors. The authors also found that MSI was rarely seen in sporadic ovarian tumors and that MSI lacks a significant role in the origin and progression of ovarian tumors.
Loss of heterozygosity
Loss of heterozygosity (LOH) at chromosomes 5q14-21 and 17q11.2 was found more frequently in MBOTs or APMOTs and mucinous carcinoma, suggesting that MBOTs or APMOTs with LOH at the same loci, progresses into mucinous carcinoma. It appears that MBOTs or APMOTs with LOH at chromosomes 7q35 tended to remain as MBOTs or APMTs; they do not have the tendency to progress to carcinoma. When an MBOT or APMOT and mucinous carcinoma coexist in the same cystic neoplastic lesion, the carcinoma portion harbors LOH in addition to the loci where the morphologically MBOT or APMOT portion allowed LOH. This finding suggests that when a MBOT or APMOT and mucinous carcinoma coexist in the same cystic lesion, the MBOT or APMOT is the precursor of the invasive carcinoma.193 In summary, it appears that the MBOTs or APMOTs with LOH at chromosome 7q35 remain as such, while MBOTs or APMOTs with LOH at chromosomes 5q14-21 and 17q11.2 will progress to carcinoma, therefore, MBOTs or APMOTs with LOH at 5q14-21 and 17q11.2 are the precursor of invasive mucinous carcinoma in the adenoma–carcinoma sequence.
Using customized oligonucleotide microarray, it has been shown that mucinous ovarian carcinomas express genetic profiles that are different to but also overlap with other subtypes of epithelial ovarian cancers.
Molecular pathology of mucinous tumors
KRAS mutations
- Cystadenoma 14–56%
- MBOTs or APMOTs 29–73%
- Mucinous carcinoma 40–85%
- Lacks significant role
Loss of heterozygosity
- On chromosomes 5q14-21 and 17q11.2; most frequent in MBOTs and in mucinous carcinoma
- MBOTs or APMOTs with LOH at chromosome 7q35 tends to remains as such
- MBOTs or APMOTs with LOH on chromosomes 5q14-21 and 17q11.2 are the precursors of adenocarcinoma.
PSEUDOMYXOMA PERITONEI (MUCINOUS ASCITES, DISSEMINATED PERITONEAL ADENOMUCINOSIS, AND PERITONEAL MUCINOUS CARCINOMATOSIS)
Pseudomyxoma peritonei (PMP) has been defined as the intraoperative clinical finding of mucus or gelatinous fluid in a localized or generalized form, occupying the pelvic or abdominal cavity with involvement of the omentum. Occasionally the gelatinous material forms masses that adhere to the organs of the cavities. The mucus material sometimes infiltrates the stroma of intra-abdominal organs. In the case of the ovary, the resulting condition is known as pseudomyxoma ovarii (Fig. 37). PMP is a clinical term, and, therefore, should not be used as a pathological diagnosis,162, 192 and should not be included in the pathology report. If the pathologist wants to mention it, he or she should do so in a comment.
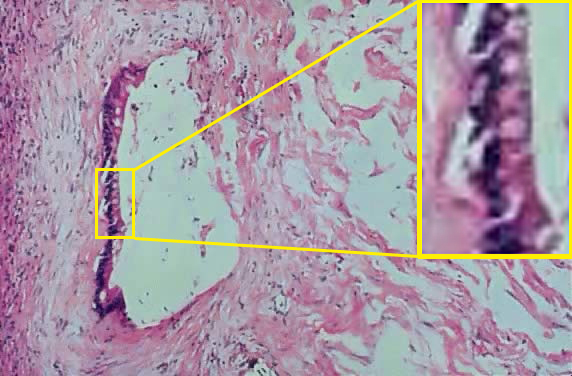 Fig. 37. Photomicrograph of ovarian stroma with a space partially lined by tall columnar mucinous secreting epithelial cells that could be of gastrointestinal origin as suggested by the presence of goblet-type-cells seen in the inset, at the right-hand side. Nearby mucus diffusely infiltrates the ovarian stroma (hematoxylin and eosin).
Fig. 37. Photomicrograph of ovarian stroma with a space partially lined by tall columnar mucinous secreting epithelial cells that could be of gastrointestinal origin as suggested by the presence of goblet-type-cells seen in the inset, at the right-hand side. Nearby mucus diffusely infiltrates the ovarian stroma (hematoxylin and eosin).
In the past, it was believed that PMP was the method of spread of IMBOTs or APIMOTs, particularly if the tumor was ruptured.66, 178 Now it is believed that this event probably never occurs; this idea is strongly supported by a large study of ruptured ovarian tumors by Hart and Norris175 in which there was no association with PMP. Therefore, they are most probably the result of an appendiceal or other mucus-secreting tumor in another area of the GI tract. In these cases the tumors of the ovary and appendix have similar or identical histological features.166, 194, 195
In a study by Prayson et al.194 the authors concluded that cases with acellular PMP have a very good outcome and that the appendix is the primary site of origin of PMP in the vast majority of cases; in both men and women. The authors also concluded that the associated ovarian tumors are the result of incorporation or implantation of mucus and epithelium on the ovarian surface (Fig. 38). In many cases the appendiceal tumor is not easily recognized grossly, and therefore, the entire appendix has to be serially sectioned and entirely submitted for microscopic examination to be able to find the primary site. There have been a few reported cases, in which the gastrointestinal origin cannot be found even when an extra effort has been made to locate it. Consequently, the possibility exists that in very few cases could the PMP be of ovarian origin. This possibility has been supported by reports of ovarian mucinous tumors that had originated in the gastrointestinal component of a mature cystic teratoma.162, 196 We have personally seen a couple of cases in which the origin of the mucinous tumor has been located morphologically and by immunohistochemistry profiles in a gastrointestinal epithelium of a cystic teratoma.
If the ovarian tumors associated with PMP are compared with IMBOTs or APIMOTs not associated with PMP, they differ morphologically, immunohistochemically, and by molecular genetic evidence, supporting the clonality and the appendiceal origin of PMP in women. In 100% of the cases of PMP analyzed by Szych et al.,197 the author demonstrated identical KRAS mutations in the appendiceal adenoma and the synchronous ovarian tumors. The authors identified KRAS mutations in 69% of appendiceal adenomas not associated with PMP, and in 75% of MBOTs or APMOTs. Of the cases in question, two PMP showed identical allelic losses in the matched ovarian and appendiceal tumors.
When the mucus material is acellular (it is said that when the mucus material is extensively sampled, epithelial cells should always be found166, 198, 199 or have cells with mild atypia) the lesion is designated by some as peritoneal ascites or disseminated peritoneal pseudomucinosis.166, 200, 201 In this case the appendiceal lesion is generally an adenoma or a mucocele; these cases are associated with a good patient outcome. It is known that the presence of mucus on the peritoneal surface produces irritation and the development of adhesion, which can eventually end up with the patient death secondary to intestinal obstructions. However, when the mucus material shows an epithelial component with high-nuclear or atypical grade, it should be designated as peritoneal mucinous carcinomatosis.202 In this case the disseminated tumor corresponds to metastasis from a carcinoma of the appendix or colon in which case the patient outcome is poor. The epithelial component is present in the form of glands, epithelial nest, or single cells with high-grade nuclear atypia and frequent mitoses; essentially malignant characteristics. Not infrequently in these cases there are associated parenchymal and lymph node metastases.
It has been suggested that the presence of epithelial component mixed with the mucus material should be included in the pathology report together with the degree of atypia.162 This piece of information is important for treatment and to determine the patient outcome.
Often the PMP occurs with ovarian synchronous neoplasm showing histologic features of adenoma or IMBOT or APIMT that are discovered during the operative procedure, before the leaking appendiceal tumor is found.194 The ovarian metastases result from the neoplastic mucinous cells being deposited on the ovarian surface or having penetrated in the ovarian stroma.194, 195
Pseudomyxoma peritonei (mucinous ascites or disseminated peritoneal adenomucinosis)
- Mucinous or gelatinous material in abdominal or pelvic cavity sometimes adherent to abdominal organs
- Mild, moderate, severe cellular atypia probably always secondary to appendiceal tumor or tumor elsewhere in the gastrointestinal tract
- Associated with bilateral ovarian tumors most frequently present in the right ovary
- Often coexistent with appendiceal tumor
- Similar histology of appendiceal lesion and the ovarian tumor.
MURAL NODULES
Mural nodules (MNs) are neoplastic processes that are found occasionally associated with mucinous cystadenomas, MBOTs, APMTs or mucinous carcinomas. They may exhibit a wide variety of histological features. MNs have been classified as sarcoma-like mural nodules (benign), nodules of anaplastic carcinoma, and sarcoma, or a mixture of the two. The sarcoma-like MNs occur in younger women203, 204, with a mean age of 39 years; the tumors are red brown ranging from 0.6 to 6 cm in diameter. Half of the time the nodules are associated with mucinous carcinomas and in the remaining cases MNs are associated with either benign or MBOTs or APMTs (Figs. 38, 39 and 40). Very often the MNs are multiple and show a heterogeneous cell population. Multinucleated giant cells of the epulis type, atypical spindle cells, and inflammatory cells characterize the MNs. The cell proportion varies from nodule to nodule. In some, spindle cells predominate, in others, the giant cells predominate. Giant cells are pleomorphic, mononucleated or binucleated and exhibit increased mitotic activity, 5–10/10 HPF including atypical mitoses.205
Fig. 38. Photograph of a 44 cm and 39.5 pound mucinous carcinoma with a mural nodule in its lumen. The surface shows focal blue discoloration, and fibrous capsule with prominent vascularity.
Fig. 39. Lumen of the above tumor showing multiple papillary and nodular structures bathed by clear mucus. Two well-circumscribed red nodules are seen on the inferior aspect of the tumor corresponding to a mural nodule.
Fig. 40. Photomicrograph of an area of the mucinous carcinoma showing tall columnar mucinous secreting cells forming cystic spaces of different sizes and focal areas of dystrophic calcifications (hematoxylin and eosin).
Fig. 41. Photomicrograph of a malignant mural nodule (carcinosarcoma) with marked nuclear hyperchromasia and pleomorphism. The spaces between tumor cells are filled with fresh blood. Giant cells and single cells with abundant cytoplasm are present as well abundant atypical mitoses (hematoxylin and eosin x 400).
The benign MNs (sarcoma-like mural nodules) are found in the cyst inner lining. These nodules can be differentiated from the malignant MNs by the presence of gross and microscopic sharply circumscribed borders, absence of vascular invasion, and relatively small size (1–6 cm). The cells of the nodules are positive for histiocytic markers and vimentin and focally positive for cytokeratins. This immunoprofile suggests a mesenchymal or mesothelial cell origin205 probably secondary to the irritation produced by the intramural hemorrhage or to the mucinous cyst contents that stimulate the cells to react. They behave in a benign fashion, and do not recur.
The anaplastic carcinoma MNs are single or multiple and measure from <1 cm to 10 cm in diameter.53, 203, 204, 206, 207, 208 Microscopically these MNs nodules are formed by several types of cells: one type is represented by sheets of large rhabdoid cells with abundant eosinophilic cytoplasm showing eccentric nuclei and multiple nucleoli; another type has a spindle sarcomatoid appearance exhibiting vesicular nuclei forming a herringbone pattern; and the final type is represented by pleomorphic cells. The cells of these nodules are in general strongly cytokeratin positive and are very different histologically from the cells of the mucinous carcinoma (Figs. 41 and 42).
Fig. 42. Photomicrograph from the malignant mural nodule showing clearly the glandular epithelial and the stromal components of the tumor (carcinosarcoma). The tumor cell nuclei of both components are highly pleomorphic, hyperchromatic and atypical (hematoxylin and eosin).
The malignant nodules are usually associated with MBOTs or APMTs or mucinous carcinomas. It is suggested that these nodules can be considered as a variant of mucinous carcinomas or carcinosarcomas. Invasion of the tissues around the nodule is seen frequently and occasionally there is vascular invasion.207 Recent studies suggest that when the malignant nodules are found in unruptured stage I mucinous cystic tumors, the patient outcome is not as bad as previously thought.209 Histologically, they look like anaplastic epithelial or spindle cells with eosinophilic cytoplasm. The tumor cells exhibit high-grade nuclear atypia. The carcinosarcoma MNs207, 210 are formed by large pleomorphic epithelioid cells with eosinophilic cytoplasm, large nuclei, and prominent nucleoli. Spindle cells are noted in the background and can be dominant in the tumor; they are also positive for cytokeratins, as are other malignant cells.
The sarcomatous nodules occur more often in older patients and can be uniform, variegated, and yellow colored or poorly circumscribed, and exhibit hemorrhage or necrosis on gross inspection. They are larger than the benign nodules. These malignant nodules are associated with poor patient outcome.203, 210 The nodules can be classified as fibrosarcoma, rhabdomyosarcoma, and undifferentiated sarcoma. The malignant nodules can be mixed forming carcinosarcomas. Sometimes when there are multiple nodules, some can be of one histologic type while others are of another type.204 Malignant mural nodules are fatal in 50% of the cases. The sarcoma-like mural nodules are thought as benign, although, they should be observed with caution because their morphology is worrisome.
ENDOMETRIOID BORDERLINE OVARIAN TUMORS/ATYPICAL PROLIFERATIVE ENDOMETRIOID TUMORS
This tumor represent only 0.2% of all the ovarian epithelial tumors.43, 211, The designation of endometrioid borderline ovarian tumors (EBOTs) or atypical proliferative endometrioid tumors (APEOTs) is taken from the histologic similarities of these neoplastic processes to the atypical complex, atypical simple hyperplasia of the endometrium, or to a well differentiated endometrial adenocarcinoma. In most cases the tumor simulates an adenofibroma with complex endometrial architecture of crowded glands. If the glands are only mildly crowded and if there is not cellular atypia, the tumor should be designated as endometrioid adenofibroma. In other cases the epithelial component has villoglandular features similar to those of villoglandular adenocarcinoma of the endometrium.
A relatively small number of cases of EBOTs or APEOTs have been published. The patients’ age range is 28–86 years with a mean of 54.9 years. Only 3% of patients had more than stage I disease. In the most recent series by Roth et al.211, 47% of the tumors were of adenofibromatous pattern and 43% showed squamous differentiation; of those, 7% presented with intraepithelial carcinoma and 7% with stromal microinvasion. The bilaterality ranges from 2% to about 5% at the time of diagnosis. In one series, only one patient showed a colonic implant.212 The tumor diameter fluctuates between 1 and 22 cm with a mean of 10.7 cm. The tumors show a mixture of solid and cystic masses, and their contents are usually hemorrhagic brownish green in color. Often a gross neoplasm is not identified and the lesion is found incidentally as a focus of endometriosis with atypical or malignant histologic characteristics
Microscopically, the tumors with squamous differentiation exhibit squamous morules in 33% of cases and keratin pearls in 13%. Villoglandular pattern was present in 37%; some tumors show a mixture of villoglandular, papillary and squamous features, especially in the intracystic tumors. Cribriform epithelial component was present in 33% with one of those showing intraepithelial carcinoma. In this series211 the authors found endometriosis in 63% of cases, a percentage that is higher than previously reported.212 Endometrial hyperplasia was present in 11% and endometrial adenocarcinoma in 28%.
Some tumors show histologic architecture similar to that of a cystic adenofibroma composed of endometrial-like glands in a dense fibrous stroma, although this pattern is relatively rare.
The epithelial glandular component of EBOTs or APEOTs is associated with glandular crowding without supporting stroma, mild to moderate atypia and epithelial stratification. These changes are consistent with atypical complex hyperplasia or with a well-differentiated endometrial adenocarcinoma (Figs. 43–45). EBOTs or APEOTs have been identified or designated with several different terms. The plurality of names is the result of different criteria use by different authors who have reported on this lesion. Moreover, to date, there is no universal agreement on the criteria for diagnosis. The World Health Organization criteria defined EBOTs or APETs as tumors formed by glands or cysts lined by atypical or cytologically malignant endometrioid type cells without obvious stromal invasion.57, 213, 214 When the epithelial component of the tumors shows histological features of carcinoma (grade III nuclear atypia) and is ≤3 mm in diameter the tumor is designated as intraepithelial carcinoma.53 Fig. 43. Ovary with dense stroma, focal edema, and multiple glandular elements. Cystic spaces are noted at the top probably representing dilated lymphatic channels or dilated glands. The glands are of different sizes and shapes and even at this low magnification the epithelium appears hyperchromatic (hematoxylin and eosin).
Fig. 43. Ovary with dense stroma, focal edema, and multiple glandular elements. Cystic spaces are noted at the top probably representing dilated lymphatic channels or dilated glands. The glands are of different sizes and shapes and even at this low magnification the epithelium appears hyperchromatic (hematoxylin and eosin).
 Fig. 44. Higher magnification of a different area of Fig. 43 showing back to back glands with rounding of nuclei and atypical epithelial changes. The glands are still separated by connective tissue. This image is similar to atypical complex endometrial hyperplasia (hematoxylin and eosin).
Fig. 44. Higher magnification of a different area of Fig. 43 showing back to back glands with rounding of nuclei and atypical epithelial changes. The glands are still separated by connective tissue. This image is similar to atypical complex endometrial hyperplasia (hematoxylin and eosin).
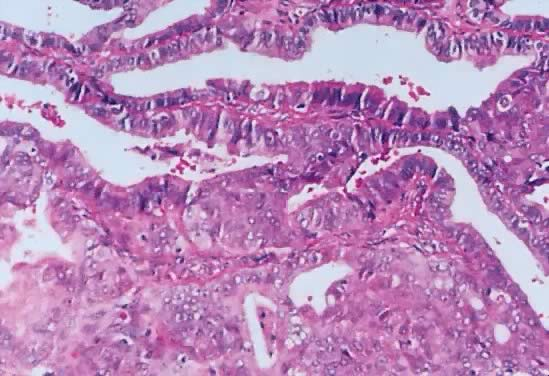 Fig. 45. Higher magnification of an area of Fig. 44 showing squamous differentiation. Observe the presence of connective tissue between the glands and nuclear atypia involving the glands lumen (hematoxylin and eosin).
Fig. 45. Higher magnification of an area of Fig. 44 showing squamous differentiation. Observe the presence of connective tissue between the glands and nuclear atypia involving the glands lumen (hematoxylin and eosin).
As in other BOTs or APOTs, microinvasion is arbitrarily designated as the presence of one focus or several foci of epithelial changes characteristic of carcinoma that measures less than 10 mm2 in area or ≤3 mm in diameter of confluent growth. The epithelium should have mild to moderate atypia and exhibit stratification, absence of stromal desmoplasia, and destructive invasion. Some investigators have used 5 mm214 as the limit to separate EBOTs or APOTs from invasive adenocarcinoma. It seems that 5 mm is now more commonly accepted as the upper limit for microinvasion. These tumors behave as benign neoplasms suggesting that they perhaps should not be called EBOTs or APOTs. These tumors were designated earlier213 as atypical proliferating endometrioid tumor that is probably realistic given the excellent prognosis but confusing because of the previous use of this term, as a synonym of EBOTs. In those cases the glandular epithelial atypia is mild to moderate. Snyder et al.212 called tumors with severe glandular atypia, invasion of <5 mm, and mitotic activity up to 10/10 HPF as tumors of low malignant potential. Only few cases of microinvasion or intraepithelial carcinoma have been reported and they all appeared to have a biologically benign behavior.
Intraepithelial carcinoma is represented by a very similar pattern of confluent growth of ≤3 mm or 5 mm in diameter but with severe glandular and nuclear atypia.215
Peritoneal implants of EBOTs or APOTs are extremely rare. In a case reported earlier,213 it is speculated that the implant may represent an independent focus of tumor arising in endometriosis. Because implants occur very rarely, there is no description of such event.
Borderline endometrioid tumor
- Similar to endometrial atypical simple or complex hyperplasia or well-differentiated adenocarcinoma
- Very rare tumor
- Only 3% are more than stage I
- 7% have adenofibromatous pattern
- 43% associated with squamous metaplasia
- 7% associated with intraepithelial carcinoma
- 7% associated with microinvasion
- Bilaterality 2–5%
- Up to 63% associated with endometriosis
- 11% associated with endometrial hyperplasia
- 28% associated with endometrial adenocarcinoma
- Biologically benign behavior.
CLEAR CELL BORDERLINE OVARIAN TUMORS/ATYPICAL PROLIFERATIVE CLEAR CELL TUMORS
Clear cells borderline ovarian tumors (CCBOTs) or atypical clear cell ovarian tumors (APCCOTs) are relatively rare, they represent only 0.2% of ovarian epithelial tumors,14 and the majority of clear cell tumors are clear cell adenocarcinomas.215, 216, 217, 218, 219, 220, 221, 222 The benign tumors are very rare and the CCBOTs or APCCOTs are rare. Only about a dozen benign tumors and around 30 CCBOTs or APCCOTs have been described in the literature.66, 213, 215, 223, 224, 225, 226 Endometriosis is quite often (60–65%) associated with clear cell tumors.218, 221 About 25% of clear cell tumors arise from the epithelial lining of an endometriotic cyst. Bell and Scully133 have reported the origin of the tumor as being from the surface ovarian epithelium. Roth et al.211 consider the CCBOTs or APCCOTs as originating from endometriosis and not from the surface epithelium. Shevchuk et al.227 reported in 1981 that clear cell carcinomas often coexist with endometrioid carcinoma, both on the ovary and in the endometrium, and therefore, they should be considered as just a histological variant of endometrioid carcinoma. These tumors are accepted to be of Müllerian origin rather than of mesonephric origin.215, 223, 228 The Müllerian origin of these tumors is supported by their association with endometriosis and endometrioid carcinoma, the coexistence of endometriosis, and clear cell tumor in the same ovary. Furthermore, the fact that this type of neoplasia occurs in the endometrium and that it can be found in vaginal adenosis suggests that it is of Müllerian origin.53
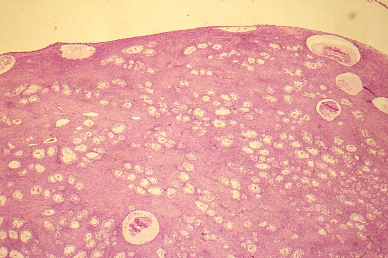 Fig. 46. Low power photomicrograph of a borderline clear cell tumor showing tubulo-cystic pattern. The presence of multiple tubules and cysts give the tumor the spongy appearance and consistency on palpation (hematoxylin and eosin).
Fig. 46. Low power photomicrograph of a borderline clear cell tumor showing tubulo-cystic pattern. The presence of multiple tubules and cysts give the tumor the spongy appearance and consistency on palpation (hematoxylin and eosin).
 Fig. 47. Higher magnification of the above image showing the tubulo-cystic pattern. There is no destructive invasion and tubules are lined by epithelial cells with loss of nuclear polarity and presence of nuclear atypia (hematoxylin and eosin).
Fig. 47. Higher magnification of the above image showing the tubulo-cystic pattern. There is no destructive invasion and tubules are lined by epithelial cells with loss of nuclear polarity and presence of nuclear atypia (hematoxylin and eosin).
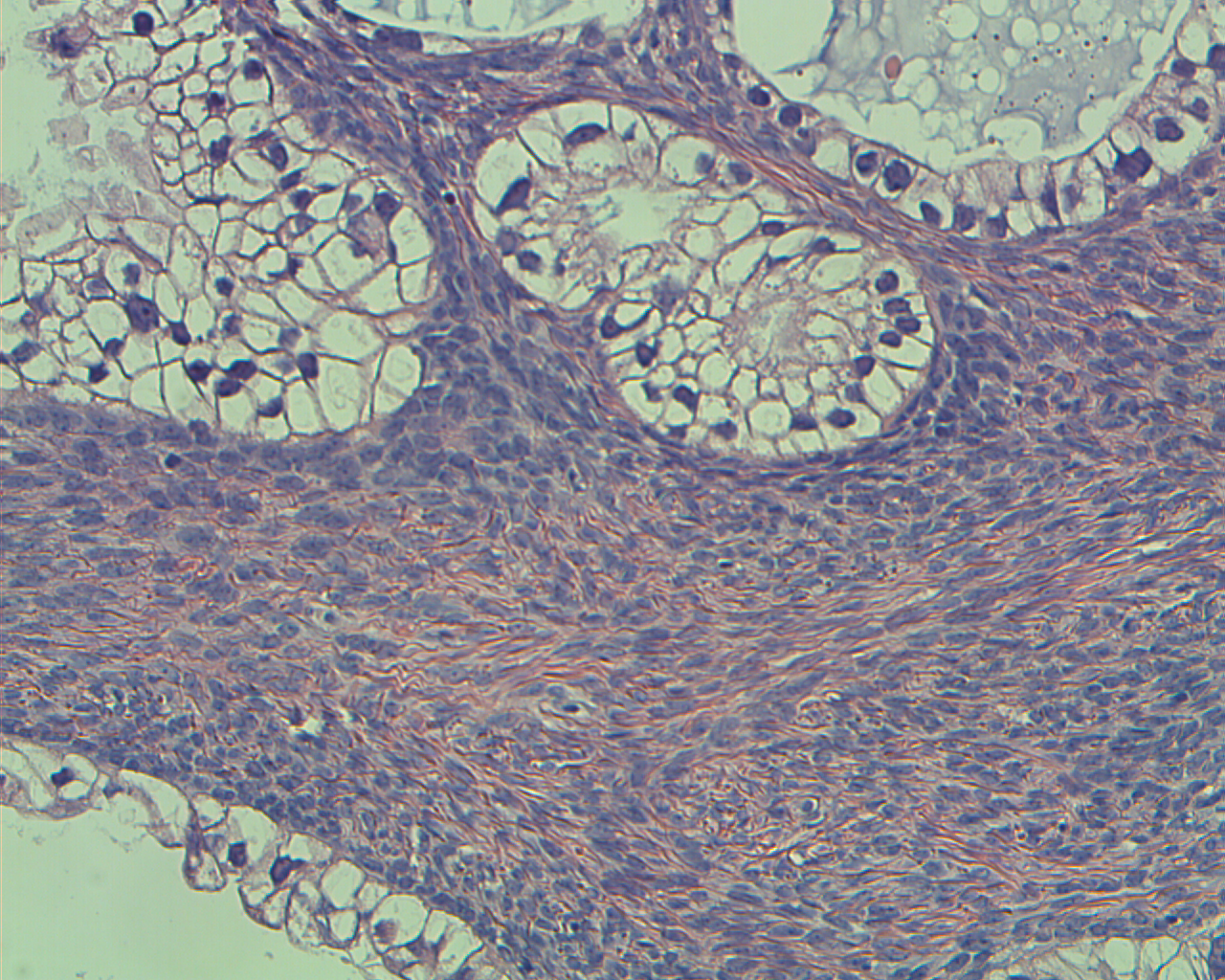 Fig. 48. Photomicrograph
of a clear cell tumor showing tubulo-cystic spaces lined by epithelial cells with abundant clear cytoplasm.
The nuclei are hyperchromatic, pleomorphic and atypical. The lumens of the tubules display mucus-like secretions (hematoxylin and eosin).
Fig. 48. Photomicrograph
of a clear cell tumor showing tubulo-cystic spaces lined by epithelial cells with abundant clear cytoplasm.
The nuclei are hyperchromatic, pleomorphic and atypical. The lumens of the tubules display mucus-like secretions (hematoxylin and eosin).
The age of the patients with benign clear cell tumors is around 45 years. According to Rusell and Farnsworth229 the age of patients with CCBOTs or APCCOTs is in their experience, similar to that of those with benign clear cell tumors. Although the cases that were reported by Roth et al.223 and Bell and Scully224, were in patients in the seventh decade, which is significantly older than patients with clear cell carcinoma.
Czernobilsky230 reported, almost at the same time, that clear cell tumors represent a subset of endometrioid adenofibromatous tumors of the ovary. There are a few examples of clear cell carcinoma arising at extraovarian locations such as the broad ligament,231 the mesocolon of the sigmoid,232 and in pelvic or abdominal foci of endometriosis.233 No cases of extraovarian implants of clear cell borderline ovarian tumor have been reported. Bell and Scully224, published the only case of a possible metastasis of clear cell borderline tumor to the lung.
The CCBOTs or APCCOTs have a nonspecific gross appearance.224 They are about 15 cm in average diameter. The external surface is sometimes shaggy due to the presence of fibrous adhesions. Because of the presence of cystic glands as a tumor component, the mass has a spongy or honeycomb features. The tumors are predominantly solid or predominantly cystic, unilocular or multilocular. When the tumors are cystic, a yellow, brown or white mass is found protruding into the cyst lumen.218, 219, 221, 222 The cyst contents are chocolate-colored if associated with endometriosis, and can be serous or mucinous, similar to those of cystic adenofibromas of the ovary.66, 215
Microscopically the tumor is composed of dense stromas in which, there are glands and tubules arranged in a disorganized fashion, contrary to what can be seen in the adenofibromas. The stroma is more abundant than the glandular component. Atypical clear cells line the infoldings, glandular and tubular structures; they are arranged in single or multiple rows (Figs. 46–48). Atypical clear cells occasionally line a cyst, which is diagnosed as atypical endometriosis. The nuclear atypia is of varying degrees, sometimes with prominent nucleoli and epithelial tufting, mitotic figures are up to 3 per 10 HPF. The epithelial cells can have scanty cytoplasm with the nuclei located at one edge, giving the cell the appearance of a hobnail, quite similar to the cells observed in the Arias-Stella phenomenon, which are characterized by a large hyperchromatic nucleus protruding in the glandular or cyst lumen. The cytoplasm exhibits a clear cell pattern shows abundant granular, and eosinophilic material (Figs. 47 and 48). Frequently, squamous, ciliated, and other types of metaplasia are present; sometimes endometriosis is seen. The glandular or tubal lumens may show mucinous content and the cytoplasm may display diastase-digestible periodic acid-Schiff-positive material which confirms the presence of glycogen.223 It is not unusual that a focus of cells with malignant features is found within the tumor but without evidence of invasion. Assuming that the tumor has been generously sampled, this focus should be diagnosed as CCBOT or APCCOT with intraepithelial carcinoma.
If areas consistent with invasion measuring less than 5 mm are present, the interpretation of the lesion should be that of a CCBOT or APCCOT with microinvasion.
Differential diagnosis of clear cell ovarian tumors
As described above, CCBOTs or APCCOTs are rare, the following neoplasms should be considered in the differential diagnosis:
Dysgerminoma (associated with lymphocytes in the stroma, usually younger patients, round cells rather than polygonal, one to four prominent nucleoli)
Yolk sac tumor (presence of other types of germ cell neoplasia alpha-fetoprotein positive, rarely in clear cell tumor)
Juvenile granulosa cell tumors (young patients, associated estrogenic symptoms, and alpha inhibin positive)
Krukenberg tumor (predominantly composed of signet-ring cell containing mucus, history of mucinous primary tumor [GI tract or breast], about 80% are bilateral)
Metastatic renal cell carcinoma, (rarely metastasis to ovary)
Steroid cell tumor (centrally located nuclei, less cytologic atypia difficult to separate from the CCBOT or APCCOT also with mild cytologic atypia. Steroid cell tumors are usually immunoreactive for Calretinin, inhibin, and melan-A).
Clear cell borderline ovarian tumors
- Tumor about 12 cm in diameter, spongy features
- Dense fibrous stroma
- Frequently or perhaps always associated with endometriosis
- Glandular or tubular epithelial elements
- Clear cells and hobnail cells lining glands and tubules
- Periodic acid-Schiff positive material in cell cytoplasm
- No extraovarian implants reported to date.
TRANSITIONAL CELL (BRENNER) BORDERLINE OVARIAN TUMORS/ATYPICAL PROLIFERATING BRENNER TUMOR
Fritz Brenner described this neoplasm in 1907. The designation of the lesions as Brenner tumors is still commonly used in the medical literature. Transitional cell ovarian neoplasms or Brenner tumors represent 10% of all ovarian tumors, and the great majority of tumors are benign.
Only 3–5% of Brenner tumors are transitional cells borderline ovarian tumors (TCBOTs) or atypical proliferating transitional cell Brenner tumors or (APTCBTs).51, 234, 235 They are almost always unilateral and are confined to the ovary. The patients have an age range of 30–70 years with a mean age of 59 years; most of the patients are over 50 years of age. The patients complain of abdominal mass and sometimes of vaginal bleeding. The tumor size ranges between 10 and 28 cm in diameter with a mean size of 18 cm. On cut section the tumors are generally cystic with friable papillary or polypoid masses protruding into the lumen (Fig. 49 and 50). The papillary masses are similar microscopically to noninvasive transitional cell carcinoma of the urinary bladder. Areas of benign transitional type epithelium with scant stroma are noted forming solid masses. Commonly, there is a firm area of the tumor that is composed of histologically benign Brenner tumor,236 which sometimes is calcified (Figs. 45 and 46). Of five series reported,234, 235, 236, 232, 237 none of the tumors had recurred or metastasized.
Transitional cell tumors as defined by World Health Organization are those composed of epithelial cells that resemble histologically the cells of the urothelium. The cells of transitional cells Brenner tumors are characterized by oval shaped nuclei that exhibit frequent longitudinal grooves. Roth and Sternberg described Brenner borderline ovarian tumors or APBOTs in 1971.238 The Brenner tumor has some transitional differentiation; but the transitional cell carcinoma without a benign Brenner component is a variant of high-grade serous carcinoma. The transitional-type cells of the Brenner tumors are histologically similar to the cells of the Walthard rests. The prognosis for patients with transitional cell borderline Brenner tumor or APBOT is excellent.
The Brenner tumor appears to be derived from the ovarian surface epithelium through metaplasia, giving origin to the so-called urothelial-like epithelium from which, the TCBOTs or APTCOTs and the Walthard rests develop. The Walthard rest and the urothelial-like epithelium are histologically and ultrastructurally similar. However, the Walthard rests and the transitional cells are different in their reaction to cytokeratins. The transitional tumors are negative to CK20 as are the tissues derived from the Müllerian system, rather than being CK20 positive, as is the urothelium.239, 240 EGFR and Ras are generally weakly positive or negative; Müllerian derived tissue and urothelium can both express CK7, CEA, EMA, and CA19-9. The CEA-125 is positive in ovarian tumors.
Because very few transitional cell tumors have been found in association with mature cystic teratomas, struma ovarii, and carcinoid tumors, a germ cell origin is possible in at least few cases.23, 53 Transitional cell tumors are characterized by epithelial proliferation with atypical or malignant features, beyond that present in the benign transitional cell or Brenner tumor. Transitional cell tumors should be devoid of destructive stromal invasion. Microinvasive carcinoma, borderline carcinoma, carcinoma in situ, and extraovarian implants have not been clearly defined as yet. Transitional cell tumors not associated with benign Brenner tumors probably represent a separate group of carcinomas.234
 Fig. 49. Gross photograph of a previously formalin fixed borderline Brenner tumor. On the upper left-hand corner, two gray solid masses are seen (one mass split) on the smaller fragment a cystic space is noted. The rest of the tumor has an irregular shaggy and hemorrhagic cystic cavity.
Fig. 49. Gross photograph of a previously formalin fixed borderline Brenner tumor. On the upper left-hand corner, two gray solid masses are seen (one mass split) on the smaller fragment a cystic space is noted. The rest of the tumor has an irregular shaggy and hemorrhagic cystic cavity.
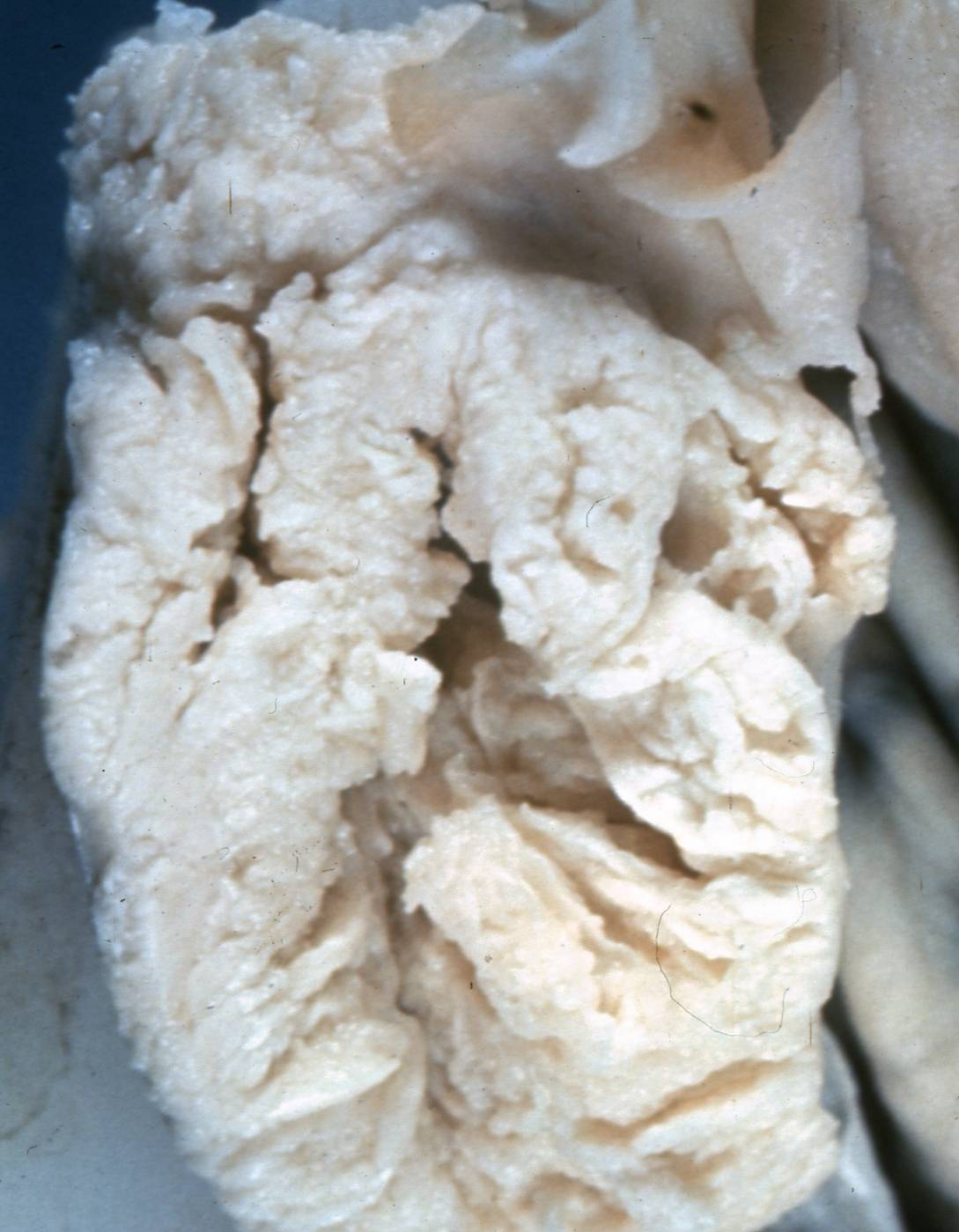 Fig. 50. Gross photograph of a borderline Brenner tumor (borderline transitional cell tumor). The tumor is cystic. The cystic cavity is filled with soft fragile papillary structures. (Courtesy of Dr Lawrence M. Roth, Indianapolis, Indiana.)
Fig. 50. Gross photograph of a borderline Brenner tumor (borderline transitional cell tumor). The tumor is cystic. The cystic cavity is filled with soft fragile papillary structures. (Courtesy of Dr Lawrence M. Roth, Indianapolis, Indiana.)
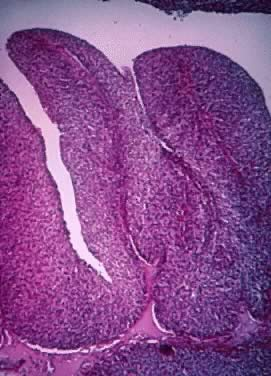 Fig. 51. Papillary growth showing thin central fibrovascular cores. The epithelium has 10–20 cell layers of transitional cell-type epithelium. The architectural and histologic characteristics are similar to those of a transitional cell carcinoma grade I or II of the urinary tract (hematoxylin and eosin).
Fig. 51. Papillary growth showing thin central fibrovascular cores. The epithelium has 10–20 cell layers of transitional cell-type epithelium. The architectural and histologic characteristics are similar to those of a transitional cell carcinoma grade I or II of the urinary tract (hematoxylin and eosin).
Histologically the TCBOTs or APTCOTs show a greater degree of epithelial complexity than that observed in the benign tumors. The epithelial complexity is created by fibrovascular papillae with a branching pattern (Figs. 50 and 51) that project into the cyst lumen. The papillary fibrovascular cores are covered by transitional type epithelium that may exhibit the same histological changes observed in the transitional cell tumors of the urinary bladder. As mentioned in the gross description, a focus of benign TCBOT or APTCOT or benign Brenner tumor is almost always present in borderline or malignant Brenner tumors; this focus could be easily missed because of its small size. Mitotic activity is increased and focal necrosis is rather common. Mucinous metaplasia is observed sometimes and could be quite prominent. The tumors have a large fibroma or thecoma component, which may be responsible for the hormonal effect (usually estrogenic) seen in some patients with Brenner tumors. There has been a subdivision of this tumor into intermediate grades. The subdivision criteria are still a matter of controversy.236 One division will encompass the proliferative Brenner tumors characterized by histological changes with grade I or II atypia, similar to the changes present in transitional cell carcinoma grade I or II of the urinary bladder (Fig. 51). The second subdivision corresponds to the borderline Brenner tumor or atypical proliferative Brenner tumor, which is compared histologically with the transitional cell carcinoma of the urinary bladder with grade III cellular atypia. Unfortunately, the term proliferating tumors corresponding to the first subdivision is confusing because of the use of “atypical proliferative” as a synonym for borderline tumors. Others,53 designate lesions with grade I nuclei as borderline Brenner tumors and those with grade II and III as borderline Brenner tumor with intraepithelial carcinoma. In all cases stromal invasion is absent. Microinvasion has not been reported.
It should be remembered that almost always, malignant invasive Brenner tumors are associated in some areas with histological characteristics of TCBOTs or APTCTs. Meticulous gross examination and generous sampling should be performed to avoid missing an invasive lesion.
Liao et al.241 reported on the use of p63 protein expression as a marker for Brenner tumors. The authors found p63 nuclear high immunoreactivity in 17 benign Brenner tumors, less intensive (80%) in five borderline Brenner tumors, less intensive (75%) in only one of six malignant Brenner tumors, and in none of the ovarian transitional cell carcinomas. Immunoreactivity to p63 was found in the Walthard cells rests of the Fallopian tubes, in epidermal basal cells of immature cystic teratomas, in transitional cell metaplasia of the cervix, and extensively, in transitional cell tumors of the urinary bladder.
It has been reported earlier that Brenner tumors have similar immunoreactivity to normal urothelium.241, 242, 243 It has also been reported that ovarian transitional cell tumors are quite similar histologically to transitional cell tumors of the urinary bladder, but that in spite of the morphological similarities they are immunophenotipically240 and genetically different. These tumors probably represent poorly differentiated ovarian tumors of surface epithelial origin (Müllerian) with transitional cell features.240, 242 This type of tumors has an immunophenotype similar to that expressed by surface epithelial tumors.
Transitional cell borderline (Brenner) tumor/atypical proliferating Brenner tumor
- Epithelium resembling urothelium
- 3–5% of all Brenner tumors
- Unilateral more than 90%, some say always unilateral
- 16–20 cm unilocular or multilocular
- Generally cystic with friable papillary masses
- 25–30% associated with endometriosis
- Some may be of germ cell origin
- None has recurred or metastasized
- Approximately 25% associated with benign mucinous tumor
- Papillary structures with grade I –III cellular atypia
- Brenner tumors express p63 nuclear immunoreactivity.
REFERENCES
Acs, G., Serous and mucinous borderline (low malignant potential) tumors of the ovary. Am J Clin Pathol, 2005. 123 Suppl: p. S13-57. |
|
Karseladze, A.I., Serous borderline ovarian tumors: where are we now? Eur J Gynaecol Oncol, 2005. 26(4): p. 355-61. |
|
Scully RE, Clement PB., Tumors of the Ovary and Maldeveloped gonades, fallopian tube and broad dligament.. In Atlas of Tumor Pathology, 3th Series, fascicle 23, Washingto DC,: Armed Froces Institue of Pathology. 1998. |
|
Webb, P.M., et al., Symptoms and diagnosis of borderline, early and advanced epithelial ovarian cancer. Gynecol Oncol, 2004. 92(1): p. 232-9. |
|
Wong, H.F., et al., Ovarian tumors of borderline malignancy: a review of 247 patients from 1991 to 2004. Int J Gynecol Cancer, 2007. 17(2): p. 342-9. |
|
Abel C, S.W., Blander. Gynecological Pathology. A Manual of Microscopic Techniques and Diagnosis. In Gynecological Practices for Students and Physicians. William Woods & company, New York, 1901. |
|
Pickel, H. and O. Reich, History of gynecological pathology XIX. Walther Schauenstein: an early austrian pioneer of cervical pathology with comments on his successors. Int J Gynecol Pathol, 2006. 25(2): p. 195-8. |
|
Pickel, H. and K. Tamussino, History of Gynecological Pathology: XIV. Hermann Johannes Pfannenstiel. Int J Gynecol Pathol, 2003. 22(3): p. 310-4. |
|
Taylor, H.C., Jr., Malignant and semimalignant tumors tumors of the ovary. . Surg Gynecol Obstet,, 1929. 48: p. 204-230. |
|
Munnell, E.W. and H.C. Taylor, Jr., Ovarian carcinoma; a review of 200 primary and 51 secondary cases. Am J Obstet Gynecol, 1949. 58(5): p. 943-59, illust. |
|
Serov Sf, S.R., Sobin LH.,, Histologic typing of ovarian tumors.Geneva, Switzerland: World Helath rganization. 1973. |
|
Classificatin and staging of malignant tumors in the female pelvis. Acta Obstet Gynecol Scand, 1971. 50: p. 1-7. |
|
Seidman, J.D.K., R. J., Ovarian serous borderline tumors: a critical review of the literature with emphasis on prognostic indicators. Hum Pathol, 2000. 31(5): p. 539-57. |
|
Seidaman JD, Cho KR, Ronnett BM, Kraus RJ. Surface Epithelial Tumors of the Ovary, In Blaustein’s Pathology of the female genital tract, Chapter 14, Six Edition, Springer, New York, 2011 |
|
Lee KR, T.F., PratJ, et al., Surface Epithelial-Stromal tumors. In Tavassoli FA, Devilee P, eds. Pathology and Genetics of Tumors of the Breast and Female Genital Organs. Lyon, France: IARC Press; 2003, 117-145. World Health Organization Classification of Tumors. 2003. |
|
Kempson, R.L. and M.R. Hendrickson, Ovarian serous borderline tumors: the citadel defended. Hum Pathol, 2000. 31(5): p. 525-6. |
|
Fritz A, Pervey C, Jack A et al.,(2000)) International classifcationof the diseases for oncology, 3thedn, WHO, Geneva. |
|
Bell, D.A. and R.E. Scully, Serous borderline tumors of the peritoneum. Am J Surg Pathol, 1990. 14(3): p. 230-9. |
|
Bell, D.A., et al., Serous borderline (low malignant potential, atypical proliferative) ovarian tumors: workshop perspectives. Hum Pathol, 2004. 35(8): p. 934-48. |
|
Santesso L, Kofumeier HL, General classification of ovarian tumors, in Gentil F, Junqueira AC, (eds): Ovarian Cancer, New York, Springer-Verlag, 1968, pp1-8); |
|
Smith Sehdev AE, Sehdev PS, Kurman RJ: Noninvasive and invasive micropapillary (low-grade) serous carcinoma of theovary: a clinicopathologic analysis of 135 cases. Am J Surg Pathol. 2003 Jun;27(6):725-36. |
|
Silva EG, Kurman RJ, Russell P et al: Symposium: ovarian tumors of borderline malignancy. Int J Gynecol Pathol. 1996 Oct;15(4):281-302. |
|
Scully RE, R.L., Histologic typing of ovarian tumors, 2nd edn. In: World Health Organaization International Histological Classification of Tumors. Springer-Verlag. Berlin, Heidelberg, 1. 1999. |
|
Hart WR, Ovarian epithelial tumors of borderline malignancy (carcinomas of low malignant potential). Hum Pathol, 1977. 8(5): p. 541-9. |
|
Riman, T., et al., Risk factors for epithelial borderline ovarian tumors: results of a Swedish case-control study. Gynecol Oncol, 2001. 83(3): p. 575-85. |
|
Modugno, F., R.B. Ness, and J.E. Wheeler, Reproductive risk factors for epithelial ovarian cancer according to histologic type and invasiveness. Ann Epidemiol, 2001. 11(8): p. 568-74. |
|
Chambers, J.T., Borderline ovarian tumors: a review of treatment. Yale J Biol Med, 1989. 62(4): p. 351-65. |
|
Bostwick, D.G., et al., Ovarian epithelial tumors of borderline malignancy. A clinical and pathologic study of 109 cases. Cancer, 1986. 58(9): p. 2052-65. |
|
Amortegui AJ, T.G., Comercy J,, Ovarian Tumors of Low Maligant Potenhtial (Borerline Ovarian tumors) In Gynecology and Obstetrics. Sciarra JJ (edd) Volume 4, Chapter 29. 2001. |
|
Koonings, P.P., et al., Relative frequency of primary ovarian neoplasms: a 10-year review. Obstet Gynecol, 1989. 74(6): p. 921-6. |
|
Scully RE, Ovary.In Henderson DE, Albores-Saavedra J, eds.Ther pathology of incipient neoplasia Philadelphia, PA Saunders; 1986:p279-293 1986. |
|
Petterson F, Annual report of the result of treatment in gynecological cancer . Stockholm Sweden: Internbational Federationof Gynecology and Obstetrics; 1991. |
|
Levi, F., et al., Borderline ovarian tumours in Vaud, Switzerland: incidence, survival and second neoplasms. Br J Cancer, 1999. 79(1): p. 4-6. |
|
Seidman, J.D., et al., The histologic type and stage distribution of ovarian carcinomas of surface epithelial origin. Int J Gynecol Pathol, 2004. 23(1): p. 41-4. |
|
Remenyi A, Tomilin A, Scholer HR et al: Differential activity by DNA-induced quarternary structures of POU transcriptionfactors. Biochem Pharmacol. 2002 Sep;64(5-6):979-84. |
|
Stalsberg, H., et al., Observer variation in histologic classification of malignant and borderline ovarian tumors. Hum Pathol, 1988. 19(9): p. 1030-5. |
|
Russell P, Surface Epithelial Tumors of the Ovary. In Kurman RJ (ed) Blaustein's Pathology of the Female Genital Tract, p 712. New York, Springer-Verlag, 1994. |
|
Beral V, Doll R, Hermon C et al: Ovarian cancer and oral contraceptives: collaborative reanalysis of data from 45epidemiological studies including 23,257 women with ovarian cancer and 87,303controls. Lancet. 2008 Jan 26;371(9609):303-14. |
|
Zhou B, Sun Q, Cong R et al: Hormone replacement therapy and ovarian cancer risk: a meta-analysis. Gynecol Oncol. 2008 Mar;108(3):641-51. Epub 2008 Jan 24. |
|
Mace J, Sybil Biermann J, Sondak V et al: Response of extraabdominal desmoid tumors to therapy with imatinib mesylate. Cancer. 2002 Dec 1;95(11):2373-9. |
|
Tropé CG, Kaern J, Davidson B. Borderline ovarian tumours.Best Pract Res Clin Obstet Gynaecol. 2012 Jun;26(3):325-36. doi: 10.1016/j.bpobgyn.2011.12.006. Epub 2012 Feb 7. |
|
Skírnisdóttir I, Garmo H, Wilander E, Holmberg L. Borderline ovarian tumors in Sweden 1960-2005: trends in incidence and age at diagnosis compared to ovarian cancer. Int J Cancer. 2008 Oct 15;123(8):1897-901. |
|
Seidman JD, Surface epithelial tumors of the ovary. In Kurman nRJ, (ed) Blaustein'sd pathology of the Female Genital Tract, New York, Springer-Verlag, 2002: p.791. |
|
Longacre, T.A., et al., Ovarian serous tumors of low malignant potential (borderline tumors): outcome-based study of 276 patients with long-term (> or =5-year) follow-up. Am J Surg Pathol, 2005. 29(6): p. 707-23. |
|
Segal, G.H. and W.R. Hart, Ovarian serous tumors of low malignant potential (serous borderline tumors). The relationship of exophytic surface tumor to peritoneal implants. Am J Surg Pathol, 1992. 16(6): p. 577-83. |
|
Michael, H., L.M. Roth, and P.K. Kotylo, Recent developments in the pathology of ovarian epithelial tumors of low malignant potential and related neoplasms. Pathol Annu, 1993. 28 Pt 2: p. 1-22. |
|
Colgan, T.J. and H.J. Norris, Ovarian epithelial tumors of low malignant potential: a review. Int J Gynecol Pathol, 1983. 1(4): p. 367-82. |
|
Kurman, R.J. and J.D. Seidman, Ovarian serous borderline tumors: the citadel defended. Hum Pathol, 2000. 31(11): p. 1439-42. |
|
Longacre, T.A., R.L. Kempson, and M.R. Hendrickson, Well-differentiated serous neoplasms of the ovary. Pathology (Phila), 1993. 1(2): p. 255-306. |
|
Klemi, P.J. and T.J. Nevalainen, Ultrastructural and histochemical observations on serous ovarian cystadenomas. Acta Pathol Microbiol Scand [A], 1978. 86(4): p. 303-12. |
|
Dietel, M. and S. Hauptmann, Serous tumors of low malignant potential of the ovary. 1. Diagnostic pathology. Virchows Arch, 2000. 436(5): p. 403-12. |
|
McCluggage WG: The pathology of and controversial aspects of ovarian borderline tumours. Curr Opin Oncol. 2010 Sep;22(5):462-72. |
|
Scully RE, Y.R., Clement PB,, Tumors of the overy and maldeveloped gonads, fallopian tube and broad ligament. In Atlas of tumor pathology 3th Series, fascicle 23, Washington DC,: Arm Forces Institute of Pathology. 1998. |
|
Prat, J. and M. De Nictolis, Serous borderline tumors of the ovary: a long-term follow-up study of 137 cases, including 18 with a micropapillary pattern and 20 with microinvasion. Am J Surg Pathol, 2002. 26(9): p. 1111-28. |
|
Seidman JD, S.R., Vang R, et a.. Ovarian borderline tumors: A co nsensus approach: Definitions and imagens. Hum Pathol, 2004. 35: p. 918-933. |
|
Leitao, M.M., Jr., et al., Clinicopathologic analysis of early-stage sporadic ovarian carcinoma. Am J Surg Pathol, 2004. 28(2): p. 147-59. |
|
Seidman JD, Surface epithelial tumors of the ovary. In Kurman RJ (ed) Blaustein's pathology of the female genital tract, New York, Springer-Verlar 2002. 2002: p. 791. |
|
Nayar, R., et al., Microinvasion in low malignant potential tumors of the ovary. Hum Pathol, 1996. 27(6): p. 521-7. |
|
Silva, E.G., et al., Symposium: ovarian tumors of borderline malignancy. Int J Gynecol Pathol, 1996. 15(4): p. 281-302. |
|
Hogg R, Scurry J, Kim SN et al: Microinvasion links ovarian serous borderline tumor and grade 1 invasivecarcinoma. Gynecol Oncol. 2007 Jul;106(1):44-51. Epub 2007 Apr 27. |
|
Morice P, Camatte S, Rey A et al: Prognostic factors for patients with advanced stage serous borderline tumours ofthe ovary. Ann Oncol. 2003 Apr;14(4):592-8. |
|
Silva EG, Gershenson DM, Malpica A et al: The recurrence and the overall survival rates of ovarian serous borderlineneoplasms with noninvasive implants is time dependent. Am J Surg Pathol. 2006 Nov;30(11):1367-71. |
|
Bell, D.A. and R.E. Scully, Ovarian serous borderline tumors with stromal microinvasion: a report of 21 cases. Hum Pathol, 1990. 21(4): p. 397-403. |
|
Tavassoli, F.A., Serous tumor of low malignant potential with early stromal invasion (serous LMP with microinvasion). Mod Pathol, 1988. 1(6): p. 407-14. |
|
Katzenstein, A.L., et al., Proliferative serous tumors of the ovary. Histologic features and prognosis. Am J Surg Pathol, 1978. 2(4): p. 339-55. |
|
Russell P, The pathological assessment of ovarian neoplasms. II: The proliferating 'epithelial' tumours. Pathology, 1979. 11(2): p. 251-82. |
|
McKenney JK, Balzer BL, Longacre TA: Patterns of stromal invasion in ovarian serous tumors of low malignant potential(borderline tumors): a reevaluation of the concept of stromal microinvasion. Am J Surg Pathol. 2006 Oct;30(10):1209-21. |
|
Mooney, J., et al., Unusual features of serous neoplasms of low malignant potential during pregnancy. Gynecol Oncol, 1997. 65(1): p. 30-5. |
|
Sangoi, A., McKenney JK, Dadras SS, Longacre TA., Lymphatic Vascyular Invaqsion in Ovarian Serous Tumors of Low Malignant Potential with Stromal Microinvasion. A case Study. Am J Surg Pathol, 2008. 32(2): p. 261-268. |
|
Seidman, J.D., B.M. Ronnett, and R.J. Kurman, Pathology of borderline (low malignant potential) ovarian tumours. Best Pract Res Clin Obstet Gynaecol, 2002. 16(4): p. 499-512. |
|
Hart W. R, Borderline epithelial tumors of the ovary. Mod Pathol, 2005. 18 Suppl 2: p. S33-50. |
|
Bell, D.A., M.A. Weinstock, and R.E. Scully, Peritoneal implants of ovarian serous borderline tumors. Histologic features and prognosis. Cancer, 1988. 62(10): p. 2212-22. |
|
Gershenson, D.M., et al., Serous borderline tumors of the ovary with noninvasive peritoneal implants. Cancer, 1998. 83(10): p. 2157-63. |
|
Michael, H. and L.M. Roth, Invasive and noninvasive implants in ovarian serous tumors of low malignant potential. Cancer, 1986. 57(6): p. 1240-7. |
|
Russell, P., Borderline epithelial tumours of the ovary: a conceptual dilemma. Clin Obstet Gynaecol, 1984. 11(1): p. 259-77. |
|
McCaughey, W.T., et al., Peritoneal epithelial lesions associated with proliferative serous tumours of ovary. Histopathology, 1984. 8(2): p. 195-208. |
|
Gershenson, D.M., et al., Ovarian serous borderline tumors with invasive peritoneal implants. Cancer, 1998. 82(6): p. 1096-103. |
|
Rollins SE, Young RH, Bell DA: Autoimplants in serous borderline tumors of the ovary: a clinicopathologic studyof 30 cases of a process to be distinguished from serous adenocarcinoma. Am J Surg Pathol. 2006 Apr;30(4):457-62. |
|
Gu, J., et al., Molecular evidence for the independent origin of extra-ovarian papillary serous tumors of low malignant potential. J Natl Cancer Inst, 2001. 93(15): p. 1147-52. |
|
Sherman ME, Berman J, Birrer MJ et al: Current challenges and opportunities for research on borderline ovarian tumors. Hum Pathol. 2004 Aug;35(8):961-70. |
|
Seidman JD, Sherman ME, Bell KA et al: Salpingitis, salpingoliths, and serous tumors of the ovaries: is there aconnection? Int J Gynecol Pathol. 2002 Apr;21(2):101-7. |
|
Gilks CB, Alkushi A, Yue JJ et al: Advanced-stage serous borderline tumors of the ovary: a clinicopathological studyof 49 cases. Int J Gynecol Pathol. 2003 Jan;22(1):29-36. |
|
Kennedy, A.W. and W.R. Hart, Ovarian papillary serous tumors of low malignant potential (serous borderline tumors). A long-term follow-up study, including patients with microinvasion, lymph node metastasis, and transformation to invasive serous carcinoma. Cancer, 1996. 78(2): p. 278-86. |
|
Bell, K.A., A.E. Smith Sehdev, and R.J. Kurman, Refined diagnostic criteria for implants associated with ovarian atypical proliferative serous tumors (borderline) and micropapillary serous carcinomas. Am J Surg Pathol, 2001. 25(4): p. 419-32. |
|
Rubin, S.C., et al., Clinical and pathological features of ovarian cancer in women with germ-line mutations of BRCA1. N Engl J Med, 1996. 335(19): p. 1413-6. |
|
Sieben NL, Kolkman-Uljee SM, Flanagan AM et al: Molecular genetic evidence for monoclonal origin of bilateral ovarian serousborderline tumors. Am J Pathol. 2003 Apr;162(4):1095-101. |
|
Zanotti, K.M., et al., Allelic imbalance on chromosome 17p13 in borderline (low malignant potential) epithelial ovarian tumors. Int J Gynecol Pathol, 1999. 18(3): p. 247-53. |
|
Link, C.J., Jr., E. Kohn, and E. Reed, The relationship between borderline ovarian tumors and epithelial ovarian carcinoma: epidemiologic, pathologic, and molecular aspects. Gynecol Oncol, 1996. 60(3): p. 347-54. |
|
Kraus JA, Seidman JD: The relationship between papillary infarction and microinvasion in ovarianatypical proliferative ("borderline") serous and seromucinous tumors. Int J Gynecol Pathol. 2010 Jul;29(4):303-9. |
|
Morice P., et al., Borderline ovarian tumors:pathological diagnostic dilemma and risk factors for invasive or lethal recurrence The lancet Oncol. 2012; 13; p e103- e115 |
|
Claus, E.B., et al., The genetic attributable risk of breast and ovarian cancer. Cancer, 1996. 77(11): p. 2318-24. |
|
Moore, W.F., et al., Some mullerian inclusion cysts in lymph nodes may sometimes be metastases from serous borderline tumors of the ovary. Am J Surg Pathol, 2000. 24(5): p. 710-8. |
|
Chang SJ, Ryu HS, Chang KH et al: Prognostic significance of the micropapillary pattern in patients with serousborderline ovarian tumors. Acta Obstet Gynecol Scand. 2008;87(4):476-81. |
|
McKenney JK, Balzer BL, Longacre TA: Lymph node involvement in ovarian serous tumors of low malignant potential(borderline tumors): pathology, prognosis, and proposed classification. Am J Surg Pathol. 2006 May;30(5):614-24. |
|
Bergman, F., Carcinoma of the ovary. A clinicopathological study of 86 autopsied cases with special reference to mode of spread. Acta Obstet Gynecol Scand, 1966. 45(2): p. 211-31. |
|
Verbruggen, M.B., et al., Serous borderline tumor of the ovary presenting with cervical lymph node involvement: a report of 3 cases. Am J Surg Pathol, 2006. 30(6): p. 739-43. |
|
Tan, L.K., S.D. Flynn, and M.L. Carcangiu, Ovarian serous borderline tumors with lymph node involvement. Clinicopathologic and DNA content study of seven cases and review of the literature. Am J Surg Pathol, 1994. 18(9): p. 904-12. |
|
Clement, P.B., et al., Hyperplastic mesothelial cells within abdominal lymph nodes: mimic of metastatic ovarian carcinoma and serous borderline tumor--a report of two cases associated with ovarian neoplasms. Mod Pathol, 1996. 9(9): p. 879-86. |
|
Silva, E.G., et al., Tumor recurrence in stage I ovarian serous neoplasms of low malignant potential. Int J Gynecol Pathol, 1998. 17(1): p. 1-6. |
|
Karp, L.A. and B. Czernobilsky, Glandular inclusions in pelvic and abdominal para-aortic lymph nodes. A study of autopsy and surgical material in males and females. Am J Clin Pathol, 1969. 52(2): p. 212-8. |
|
Reich, O., et al., Benign mullerian inclusions in pelvic and paraaortic lymph nodes. Gynecol Oncol, 2000. 78(2): p. 242-4. |
|
McCaughey, W.T., et al., Peritoneal epithelial lesions associated with proliferative serous tumours of ovary. Histopathology, 1984. 8(2): p. 195-208. |
|
Burmeister, R.E., R.E. Fechner, and R.R. Franklin, Endosalpingiosis of the peritoneum. Obstet Gynecol, 1969. 34(3): p. 310-8. |
|
Parker, R.L., et al., Early recurrence of ovarian serous borderline tumor as high-grade carcinoma: a report of two cases. Int J Gynecol Pathol, 2004. 23(3): p. 265-72. |
|
Zinsser, K.R. and J.E. Wheeler, Endosalpingiosis in the omentum: a study of autopsy and surgical material. Am J Surg Pathol, 1982. 6(2): p. 109-17. |
|
Ehrmann, R.L., J.M. Federschneider, and R.C. Knapp, Distinguishing lymph node metastases from benign glandular inclusions in low-grade ovarian carcinoma. Am J Obstet Gynecol, 1980. 136(6): p. 737-46. |
|
Seidman, J.D. and R.J. Kurman, Subclassification of serous borderline tumors of the ovary into benign and malignant types. A clinicopathologic study of 65 advanced stage cases. Am J Surg Pathol, 1996. 20(11): p. 1331-45. |
|
Serov Sf, S.R., Sobin LH., Histologic typing of ovarian tumors.Geneva, Switzerland: World Helath rganization. 1973. |
|
Pettersson F, Annual report on gynecological cancer, FIGO, Stockholm, Stockhol Panorama Press. 1988. 20. |
|
Diaz-Padilla et al., Ovarian low-grade serous carcinoma: a comprehensive update. Gynecol Oncol. 2012. 126: p 279-285 |
|
Burks, R.T., M.E. Sherman, and R.J. Kurman, Micropapillary serous carcinoma of the ovary. A distinctive low-grade carcinoma related to serous borderline tumors. Am J Surg Pathol, 1996. 20(11): p. 1319-30. |
|
Gershenson, D.M. and E.G. Silva, Serous ovarian tumors of low malignant potential with peritoneal implants. Cancer, 1990. 65(3): p. 578-85. |
|
Deavers, M.T., et al., Micropapillary and cribriform patterns in ovarian serous tumors of low malignant potential: a study of 99 advanced stage cases. Am J Surg Pathol, 2002. 26(9): p. 1129-41. |
|
Seidman, J.D., et al., Borderline ovarian tumors: diverse contemporary viewpoints on terminology and diagnostic criteria with illustrative images. Hum Pathol, 2004. 35(8): p. 918-33. |
|
Singer, G., et al., Mutations in BRAF and KRAS characterize the development of low-grade ovarian serous carcinoma. J Natl Cancer Inst, 2003. 95(6): p. 484-6. |
|
Kurman RJ, et al,. The origin and pathogenesis of epithelial ovarian cancer- a proposed unifying theory. Am J Surg Pathol.2010, 34(3): p 433-343 |
|
Singer, G., et al., Patterns of p53 mutations separate ovarian serous borderline tumors and low- and high-grade carcinomas and provide support for a new model of ovarian carcinogenesis: a mutational analysis with immunohistochemical correlation. Am J Surg Pathol, 2005. 29(2): p. 218-24. |
|
Boy C.,et al., Low-grade ovarian serous neoplasms (Low-grade serous carcinoma and serous borderline tumor) associated with high grade serous or undifferentiated carcinoma: Report of a series of cases of an unusual phenomenon. Am J Surg Pathol, 2012. 36(3);p 368-375 |
|
May T., et al,.Low malignant potential tumors with micropapillary features are molecularly similar to low-grade serous carcinoma of the ovary. Gynecol Oncol, 2010. 117(1) p 9-17 |
|
Katabuchi, H., et al., Micropapillary serous carcinoma of the ovary: an immunohistochemical and mutational analysis of p53. Int J Gynecol Pathol, 1998. 17(1): p. 54-60. |
|
O'Neill CJ, Deavers MT, Malpica A et al: An immunohistochemical comparison between low-grade and high-grade ovarian serous Am J Surg Pathol. 2005 Aug;29(8):1034-41. |
|
Kurman RJ, Shih IeM: The origin and pathogenesis of epithelial ovarian cancer: a proposed unifyingtheory. Am J Surg Pathol. 2010 Mar;34(3):433-43. |
|
Cho KR, Shih IeM: Ovarian cancer. Annu Rev Pathol. 2009;4:287-313. |
|
Kurman RJ, Shih IeM: Pathogenesis of ovarian cancer: lessons from morphology and molecular biology andtheir clinical implications. Int J Gynecol Pathol. 2008 Apr;27(2):151-60. |
|
Kurman RJ, Visvanathan K, Roden R et al: Early detection and treatment of ovarian cancer: shifting from early stage tominimal volume of disease based on a new model of carcinogenesis. Am J Obstet Gynecol. 2008 Apr;198(4):351-6. |
|
Schmeler KM, Gershenson DM: Low-grade serous ovarian cancer: a unique disease. Curr Oncol Rep. 2008 Nov;10(6):519-23. |
|
O'Neill CJ, Deavers MT, Malpica A et al: An immunohistochemical comparison between low-grade and high-grade ovarian serous Am J Surg Pathol. 2005 Aug;29(8):1034-41. |
|
Singer, G., et al., Patterns of p53 mutations separate ovarian serous borderline tumors and low- and high-grade carcinomas and provide support for a new model of ovarian carcinogenesis: a mutational analysis with immunohistochemical correlation. Am J Surg Pathol, 2005. 29(2): p. 218-24. |
|
Dehari R, Kurman RJ, Logani S et al: The development of high-grade serous carcinoma from atypical proliferativemorphologic and molecular genetic analysis. Am J Surg Pathol. 2007 Jul;31(7):1007-12. |
|
Vang R, Shih IeM, Kurman RJ: Ovarian low-grade and high-grade serous carcinoma: pathogenesis,clinicopathologic and molecular biologic features, and diagnostic problems. Adv Anat Pathol. 2009 Sep;16(5):267-82. |
|
Puls, L.E., et al., Transition from benign to malignant epithelium in mucinous and serous ovarian cystadenocarcinoma. Gynecol Oncol, 1992. 47(1): p. 53-7. |
|
Agoff, S.N., et al., Unexpected gynecologic neoplasms in patients with proven or suspected BRCA-1 or -2 mutations: implications for gross examination, cytology, and clinical follow-up. Am J Surg Pathol, 2002. 26(2): p. 171-8. |
|
Bell, D.A. and R.E. Scully, Early de novo ovarian carcinoma. A study of fourteen cases. Cancer, 1994. 73(7): p. 1859-64. |
|
Shaw, P.A., et al., Histopathologic features of genetically determined ovarian cancer. Int J Gynecol Pathol, 2002. 21(4): p. 407-11. |
|
Li J, Abushahin N, Pang S et al: Tubal origin of 'ovarian' low-grade serous carcinoma. Mod Pathol. 2011 Nov;24(11):1488-99. doi: 10.1038/modpathol.2011.106. Epub 2011 |
|
Mok, S.C., et al., Mutation of K-ras protooncogene in human ovarian epithelial tumors of borderline malignancy. Cancer Res, 1993. 53(7): p. 1489-92. |
|
Singer, G., et al., Diverse tumorigenic pathways in ovarian serous carcinoma. Am J Pathol, 2002. 160(4): p. 1223-8. |
|
Sieben, N.L., et al., In ovarian neoplasms, BRAF, but not KRAS, mutations are restricted to low-grade serous tumours. J Pathol, 2004. 202(3): p. 336-40. |
|
Caduff, R.F., et al., Comparison of mutations of Ki-RAS and p53 immunoreactivity in borderline and malignant epithelial ovarian tumors. Am J Surg Pathol, 1999. 23(3): p. 323-8. |
|
Pollock, P.M., et al., High frequency of BRAF mutations in nevi. Nat Genet, 2003. 33(1): p. 19-20. |
|
Chan, T.L., et al., BRAF and KRAS mutations in colorectal hyperplastic polyps and serrated adenomas. Cancer Res, 2003. 63(16): p. 4878-81. |
|
Shih le-Ming et al,. Molecular pathogenesis f ovarian borderline tumors: New insights and old challenges. Clin Cancer Res. 2005; 11(20) p7273-7279 |
|
Ho CL, Kurman RJ, Dehari R et al: Mutations of BRAF and KRAS precede the development of ovarian serous borderlinetumors. Cancer Res. 2004 Oct 1;64(19):6915-8. |
|
Gayther, S.A., et al., The contribution of germline BRCA1 and BRCA2 mutations to familial ovarian cancer: no evidence for other ovarian cancer-susceptibility genes. Am J Hum Genet, 1999. 65(4): p. 1021-9. |
|
Aida, H., et al., Clinical features of ovarian cancer in Japanese women with germ-line mutations of BRCA1. Clin Cancer Res, 1998. 4(1): p. 235-40. |
|
Berchuck, A., et al., Frequency of germline and somatic BRCA1 mutations in ovarian cancer. Clin Cancer Res, 1998. 4(10): p. 2433-7. |
|
Bodian, C.A., et al., Prognostic significance of benign proliferative breast disease. Cancer, 1993. 71(12): p. 3896-907. |
|
Johannsson, O.T., et al., Survival of BRCA1 breast and ovarian cancer patients: a population-based study from southern Sweden. J Clin Oncol, 1998. 16(2): p. 397-404. |
|
Pharoah, P.D., et al., Survival in familial, BRCA1-associated, and BRCA2-associated epithelial ovarian cancer. United Kingdom Coordinating Committee for Cancer Research (UKCCCR) Familial Ovarian Cancer Study Group. Cancer Res, 1999. 59(4): p. 868-71. |
|
Werness, B.A., et al., Histopathology of familial ovarian tumors in women from families with and without germline BRCA1 mutations. Hum Pathol, 2000. 31(11): p. 1420-4. |
|
Zweemer, R.P., et al., Clinical and genetic evaluation of thirty ovarian cancer families. Am J Obstet Gynecol, 1998. 178(1 Pt 1): p. 85-90. |
|
Rubin, S.C., et al., Clinical and pathological features of ovarian cancer in women with germ-line mutations of BRCA1. N Engl J Med, 1996. 335(19): p. 1413-6. |
|
Stratton, J.F., et al., Contribution of BRCA1 mutations to ovarian cancer. N Engl J Med, 1997. 336(16): p. 1125-30. |
|
Ben David, Y., et al., Effect of BRCA mutations on the length of survival in epithelial ovarian tumors. J Clin Oncol, 2002. 20(2): p. 463-6. |
|
Malpica, A., et al., Grading ovarian serous carcinoma using a two-tier system. Am J Surg Pathol, 2004. 28(4): p. 496-504. |
|
Feeley, K.M. and M. Wells, Precursor lesions of ovarian epithelial malignancy. Histopathology, 2001. 38(2): p. 87-95. |
|
Parker, R.L., et al., Early recurrence of ovarian serous borderline tumor as high-grade carcinoma: a report of two cases. Int J Gynecol Pathol, 2004. 23(3): p. 265-72. |
|
Gilks, C.B., et al., Distinction between serous tumors of low malignant potential and serous carcinomas based on global mRNA expression profiling. Gynecol Oncol, 2005. 96(3): p. 684-94. |
|
Clement PB, Diseases of the Peritoneun. In Robert J Kuman, Blaustein's Pathology of the Female Genital Trac, Chapert 17 pp 771. 2002. |
|
Gilks CB, Bell DA, Scully RE: Serous psammocarcinoma of the ovary and peritoneum. Int J Gynecol Pathol. 1990;9(2):110-21. |
|
Fukumoto, M. and K. Nakayama, Ovarian epithelial tumors of low malignant potential: are they precursors of ovarian carcinoma? Pathol Int, 2006. 56(5): p. 233-9. |
|
Lee, K.R. and R.E. Scully, Mucinous tumors of the ovary: a clinicopathologic study of 196 borderline tumors (of intestinal type) and carcinomas, including an evaluation of 11 cases with 'pseudomyxoma peritonei'. Am J Surg Pathol, 2000. 24(11): p. 1447-64. |
|
Siriaunkgul, S., et al., Ovarian mucinous tumors of low malignant potential: a clinicopathologic study of 54 tumors of intestinal and mullerian type. Int J Gynecol Pathol, 1995. 14(3): p. 198-208. |
|
Chen YC, Lin KH: Chemotherapy-related side effects in children with acute lymphocytic leukemia inTaiwan: a nursing perspective. Zhonghua Min Guo Xiao Er Ke Yi Xue Hui Za Zhi. 1992 Mar-Apr;33(2):119-29. |
|
Riopel, M.A., B.M. Ronnett, and R.J. Kurman, Evaluation of diagnostic criteria and behavior of ovarian intestinal-type mucinous tumors: atypical proliferative (borderline) tumors and intraepithelial, microinvasive, invasive, and metastatic carcinomas. Am J Surg Pathol, 1999. 23(6): p. 617-35. |
|
Ronnett, B.M., et al., Pseudomyxoma peritonei in women: a clinicopathologic analysis of 30 cases with emphasis on site of origin, prognosis, and relationship to ovarian mucinous tumors of low malignant potential. Hum Pathol, 1995. 26(5): p. 509-24. |
|
Seidman, J.D., R.J. Kurman, and B.M. Ronnett, Primary and metastatic mucinous adenocarcinomas in the ovaries: incidence in routine practice with a new approach to improve intraoperative diagnosis. Am J Surg Pathol, 2003. 27(7): p. 985-93. |
|
Fukunaga, M., et al., Ovarian atypical endometriosis: its close association with malignant epithelial tumours. Histopathology, 1997. 30(3): p. 249-55. |
|
Guerrieri, C., et al., Mucinous borderline and malignant tumors of the ovary. A clinicopathologic and DNA ploidy study of 92 cases. Cancer, 1994. 74(8): p. 2329-40. |
|
Young, R.H. and R.E. Scully, Metastatic tumors in the ovary: a problem-oriented approach and review of the recent literature. Semin Diagn Pathol, 1991. 8(4): p. 250-76. |
|
Prat, J., Ovarian tumors of borderline malignancy (tumors of low malignant potential): a critical appraisal. Adv Anat Pathol, 1999. 6(5): p. 247-74. |
|
Lee, K.R. and R.H. Young, The distinction between primary and metastatic mucinous carcinomas of the ovary: gross and histologic findings in 50 cases. Am J Surg Pathol, 2003. 27(3): p. 281-92. |
|
Ronnett, B.M., et al., The morphologic spectrum of ovarian metastases of appendiceal adenocarcinomas: a clinicopathologic and immunohistochemical analysis of tumors often misinterpreted as primary ovarian tumors or metastatic tumors from other gastrointestinal sites. Am J Surg Pathol, 1997. 21(10): p. 1144-55. |
|
Young, R.H. and R.E. Scully, Mucinous ovarian tumors associated with mucinous adenocarcinomas of the cervix. A clinicopathological analysis of 16 cases. Int J Gynecol Pathol, 1988. 7(2): p. 99-111. |
|
Hart WR, N., H. J., Borderline and malignant mucinous tumors of the ovary. Histologic criteria and clinical behavior. Cancer, 1973. 31(5): p. 1031-45. |
|
Rodriguez, I.M., J.A. Irving, and J. Prat, Endocervical-like mucinous borderline tumors of the ovary: a clinicopathologic analysis of 31 cases. Am J Surg Pathol, 2004. 28(10): p. 1311-8. |
|
Nomura, K. and S. Aizawa, Clinicopathologic and mucin histochemical analyses of 90 cases of ovarian mucinous borderline tumors of intestinal and mullerian types. Pathol Int, 1996. 46(8): p. 575-80. |
|
Rutgers, J.L. and R.E. Scully, Ovarian mixed-epithelial papillary cystadenomas of borderline malignancy of mullerian type. A clinicopathologic analysis. Cancer, 1988. 61(3): p. 546-54. |
|
Khunamornpong, S., P. Russell, and J.C. Dalrymple, Proliferating (LMP) mucinous tumors of the ovaries with microinvasion: morphologic assessment of 13 cases. Int J Gynecol Pathol, 1999. 18(3): p. 238-46. |
|
Yemelyanova AV, Vang R, Judson K et al: Distinction of primary and metastatic mucinous tumors involving the ovary:analysis of size and laterality data by primary site with reevaluation of analgorithm for tumor classification. Am J Surg Pathol. 2008 Jan;32(1):128-38. |
|
Jordan, S.J., et al., Risk factors for benign, borderline and invasive mucinous ovarian tumors: epidemiological evidence of a neoplastic continuum? Gynecol Oncol, 2007. 107(2): p. 223-30. |
|
Bell, D.A., Origins and molecular pathology of ovarian cancer. Mod Pathol, 2005. 18 Suppl 2: p. S19-32. |
|
Scott, M. and W.G. McCluggage, Current concepts in ovarian epithelial tumorigenesis: correlation between morphological and molecular data. Histol Histopathol, 2006. 21(1): p. 81-92. |
|
Jordan, S., A. Green, and P. Webb, Benign epithelial ovarian tumours-cancer precursors or markers for ovarian cancer risk? Cancer Causes Control, 2006. 17(5): p. 623-32. |
|
Cuatrecasas, M., et al., K-ras mutations in mucinous ovarian tumors: a clinicopathologic and molecular study of 95 cases. Cancer, 1997. 79(8): p. 1581-6. |
|
Mandai, M., et al., Heterogeneous distribution of K-ras-mutated epithelia in mucinous ovarian tumors with special reference to histopathology. Hum Pathol, 1998. 29(1): p. 34-40. |
|
Ichikawa, Y., et al., Mutation of K-ras protooncogene is associated with histological subtypes in human mucinous ovarian tumors. Cancer Res, 1994. 54(1): p. 33-5. |
|
Enomoto, T., et al., K-ras activation occurs frequently in mucinous adenocarcinomas and rarely in other common epithelial tumors of the human ovary. Am J Pathol, 1991. 139(4): p. 777-85. |
|
Gemignani, M.L., et al., Role of KRAS and BRAF gene mutations in mucinous ovarian carcinoma. Gynecol Oncol, 2003. 90(2): p. 378-81. |
|
Heinzelmann-Schwarz VA, Gardiner-Garden M, Henshall SM et al: A distinct molecular profile associated with mucinous epithelial ovarian cancer. Br J Cancer. 2006 Mar 27;94(6):904-13. |
|
Shih, Y.C., et al., No evidence for microsatellite instability from allelotype analysis of benign and low malignant potential ovarian neoplasms. Gynecol Oncol, 1998. 69(3): p. 210-3. |
|
Sanz Casla, M.T., et al., Role of microsatellite instability in borderline ovarian tumors. Anticancer Res, 2003. 23(6D): p. 5139-41. |
|
Nakayama, K., et al., Comprehensive allelotype study of ovarian tumors of low malignant potential: potential differences in pathways between tumors with and without genetic predisposition to invasive carcinoma. Int J Cancer, 2001. 94(4): p. 605-9. |
|
Prayson, R.A., W.R. Hart, and R.E. Petras, Pseudomyxoma peritonei. A clinicopathologic study of 19 cases with emphasis on site of origin and nature of associated ovarian tumors. Am J Surg Pathol, 1994. 18(6): p. 591-603. |
|
Young, R.H., C.B. Gilks, and R.E. Scully, Mucinous tumors of the appendix associated with mucinous tumors of the ovary and pseudomyxoma peritonei. A clinicopathological analysis of 22 cases supporting an origin in the appendix. Am J Surg Pathol, 1991. 15(5): p. 415-29. |
|
Ronnett, B.M. and J.D. Seidman, Mucinous tumors arising in ovarian mature cystic teratomas: relationship to the clinical syndrome of pseudomyxoma peritonei. Am J Surg Pathol, 2003. 27(5): p. 650-7. |
|
Szych, C., et al., Molecular genetic evidence supporting the clonality and appendiceal origin of Pseudomyxoma peritonei in women. Am J Pathol, 1999. 154(6): p. 1849-55. |
|
Hoerl, H.D. and W.R. Hart, Primary ovarian mucinous cystadenocarcinomas: a clinicopathologic study of 49 cases with long-term follow-up. Am J Surg Pathol, 1998. 22(12): p. 1449-62. |
|
Ronnett, B.M., et al., Patients with pseudomyxoma peritonei associated with disseminated peritoneal adenomucinosis have a significantly more favorable prognosis than patients with peritoneal mucinous carcinomatosis. Cancer, 2001. 92(1): p. 85-91. |
|
Ronnett, B.M., et al., Pseudomyxoma peritonei: new concepts in diagnosis, origin, nomenclature, and relationship to mucinous borderline (low malignant potential) tumors of the ovary. Anat Pathol, 1997. 2: p. 197-226. |
|
Cuatrecasas, M., X. Matias-Guiu, and J. Prat, Synchronous mucinous tumors of the appendix and the ovary associated with pseudomyxoma peritonei. A clinicopathologic study of six cases with comparative analysis of c-Ki-ras mutations. Am J Surg Pathol, 1996. 20(6): p. 739-46. |
|
Ronnett, B.M., et al., Disseminated peritoneal adenomucinosis and peritoneal mucinous carcinomatosis. A clinicopathologic analysis of 109 cases with emphasis on distinguishing pathologic features, site of origin, prognosis, and relationship to pseudomyxoma peritonei. Am J Surg Pathol, 1995. 19(12): p. 1390-408. |
|
Prat, J. and R.E. Scully, Ovarian mucinous tumors with sarcoma-like mural nodules: a report of seven cases. Cancer, 1979. 44(4): p. 1332-44. |
|
Bague, S., I.M. Rodriguez, and J. Prat, Sarcoma-like mural nodules in mucinous cystic tumors of the ovary revisited: a clinicopathologic analysis of 10 additional cases. Am J Surg Pathol, 2002. 26(11): p. 1467-76. |
|
Prat, J., Ovarian carcinomas, including secondary tumors: diagnostically challenging areas. Mod Pathol, 2005. 18 Suppl 2: p. S99-111. |
|
Nichols, G.E., et al., Spindle cell mural nodules in cystic ovarian mucinous tumors. A clinicopathologic and immunohistochemical study of five cases. Am J Surg Pathol, 1991. 15(11): p. 1055-62. |
|
Prat, J., R.H. Young, and R.E. Scully, Ovarian mucinous tumors with foci of anaplastic carcinoma. Cancer, 1982. 50(2): p. 300-4. |
|
Chan, Y.F., et al., Ovarian mucinous tumor with mural nodules of anaplastic carcinoma. Gynecol Oncol, 1989. 35(1): p. 112-9. |
|
Rodriguez, I.M. and J. Prat, Mucinous tumors of the ovary: a clinicopathologic analysis of 75 borderline tumors (of intestinal type) and carcinomas. Am J Surg Pathol, 2002. 26(2): p. 139-52. |
|
Baergen, R.N. and J.L. Rutgers, Mural nodules in common epithelial tumors of the ovary. Int J Gynecol Pathol, 1994. 13(1): p. 62-72. |
|
Roth, L.M., R.E. Emerson, and T.M. Ulbright, Ovarian endometrioid tumors of low malignant potential: a clinicopathologic study of 30 cases with comparison to well-differentiated endometrioid adenocarcinoma. Am J Surg Pathol, 2003. 27(9): p. 1253-9. |
|
Snyder, R.R., H.J. Norris, and F. Tavassoli, Endometrioid proliferative and low malignant potential tumors of the ovary. A clinicopathologic study of 46 cases. Am J Surg Pathol, 1988. 12(9): p. 661-71. |
|
Bell, D.A. and R.E. Scully, Atypical and borderline endometrioid adenofibromas of the ovary. A report of 27 cases. Am J Surg Pathol, 1985. 9(3): p. 205-14. |
|
Bell, K.A. and R.J. Kurman, A clinicopathologic analysis of atypical proliferative (borderline) tumors and well-differentiated endometrioid adenocarcinomas of the ovary. Am J Surg Pathol, 2000. 24(11): p. 1465-79. |
|
Lee KR, The pathology of surface epithelial-stromal tumors of the ovar, Chapter 27, pp 846. In In Christopher PC, Knneth R. Lee (eds) Diagnostic Gynecologic and Obstetric Pathology Elsevier Saunders. 2006: p. 846. |
|
Scully RE, Tumors of the ovaries and maldevelpoped gonades. In Atlas of tumors pathology, series 2, fascicle 16, pp 1672. Washington DC, Armed Froces Institute of Pathology, 1978. 1978. |
|
Scully, R.E., Ovarian tumors. A review. Am J Pathol, 1977. 87(3): p. 686-720. |
|
Brescia, R.J., N. Dubin, and R.I. Demopoulos, Endometrioid and clear cell carcinoma of the ovary. Factors affecting survival. Int J Gynecol Pathol, 1989. 8(2): p. 132-8. |
|
Crozier, M.A., et al., Clear cell carcinoma of the ovary: a study of 59 cases. Gynecol Oncol, 1989. 35(2): p. 199-203. |
|
Jenison, E.L., et al., Clear cell adenocarcinoma of the ovary: a clinical analysis and comparison with serous carcinoma. Gynecol Oncol, 1989. 32(1): p. 65-71. |
|
Montag, A.G., et al., Ovarian clear cell carcinoma. A clinicopathologic analysis of 44 cases. Int J Gynecol Pathol, 1989. 8(2): p. 85-96. |
|
Kennedy, A.W., et al., Ovarian clear cell adenocarcinoma. Gynecol Oncol, 1989. 32(3): p. 342-9. |
|
Roth, L.M., et al., Ovarian clear cell adenofibromatous tumors. Benign, of low malignant potential, and associated with invasive clear cell carcinoma. Cancer, 1984. 53(5): p. 1156-63. |
|
Bell, D.A. and R.E. Scully, Benign and borderline clear cell adenofibromas of the ovary. Cancer, 1985. 56(12): p. 2922-31. |
|
Tsurunaga, T., et al., Ovarian clear cell adenofibromatous tumor: report of 2 cases]. Nippon Sanka Fujinka Gakkai Zasshi, 1992. 44(1): p. 105-8. |
|
Katsube, Y., et al., Ovarian clear cell adenofibroma of borderline malignancy. A case report. Hiroshima J Med Sci, 1989. 38(2): p. 87-90. |
|
Shevchuk, M.M., et al., Clear cell carcinoma of the ovary: a clinicopathologic study with review of the literature. Cancer, 1981. 47(6): p. 1344-51. |
|
Scully, R.E. and J.F. Barlow, Mesonephroma of ovary. Tumor of Mullerian nature related to the endometrioid carcinoma. Cancer, 1967. 20(9): p. 1405-17. |
|
Rusell P, a.F.A., Clear cell tumors In Surgical Pathology of the Ovaries, Chapter 27pp 335, Peter Rusell and Annabelle Farnsworth, eds. Second Edition, Chrchill Livingstone, New Yorki, 1997. 1997. |
|
Czernobilsky, B., Endometrioid neoplasia of the ovary: a reappraisal. Int J Gynecol Pathol, 1982. 1(2): p. 203-10. |
|
Evans, H., et al., Clear cell carcinoma of the sigmoid mesocolon: a tumor of the secondary mullerian system. Am J Obstet Gynecol, 1990. 162(1): p. 161-3. |
|
Woodruff, J.D., et al., Proliferative and malignant Brenner tumors. Review of 47 cases. Am J Obstet Gynecol, 1981. 141(2): p. 118-25. |
|
Hitti, I.F., S.S. Glasberg, and S. Lubicz, Clear cell carcinoma arising in extraovarian endometriosis: report of three cases and review of the literature. Gynecol Oncol, 1990. 39(3): p. 314-20. |
|
Hallgrimsson, J. and R.E. Scully, Borderline and malignant Brenner tumours of the ovary. A report of 15 cases. Acta Pathol Microbiol Scand Suppl, 1972. 233: p. 56-66. |
|
Miles, P.A. and H.J. Norris, Proliferative and malignant brenner tumors of the ovary. Cancer, 1972. 30(1): p. 174-86. |
|
Roth, L.M., G. Dallenbach-Hellweg, and B. Czernobilsky, Ovarian Brenner tumors. I. Metaplastic, proliferating, and of low malignant potential. Cancer, 1985. 56(3): p. 582-91. |
|
Trebeck, C.E., et al., Brenner tumours of the ovary: a study of the histology, immunohistochemistry and cellular DNA content in benign, borderline and malignant ovarian tumours. Pathology, 1987. 19(3): p. 241-6. |
|
Roth, L.M. and W.H. Sternberg, Proliferating Brenner tumors. Cancer, 1971. 27(3): p. 687-93. |
|
Soslow, R.A., et al., Transitional cell neoplasms of the ovary and urinary bladder: a comparative immunohistochemical analysis. Int J Gynecol Pathol, 1996. 15(3): p. 257-65. |
|
Ordonez, N.G., Transitional cell carcinomas of the ovary and bladder are immunophenotypically different. Histopathology, 2000. 36(5): p. 433-8. |
|
Liao, X.Y., et al., p63 expression in ovarian tumours: a marker for Brenner tumours but not transitional cell carcinomas. Histopathology, 2007. 51(4): p. 477-83. |
|
Ogawa, K., S.L. Johansson, and S.M. Cohen, Immunohistochemical analysis of uroplakins, urothelial specific proteins, in ovarian Brenner tumors, normal tissues, and benign and neoplastic lesions of the female genital tract. Am J Pathol, 1999. 155(4): p. 1047-50. |
|
Riedel, I., et al., Brenner tumors but not transitional cell carcinomas of the ovary show urothelial differentiation: immunohistochemical staining of urothelial markers, including cytokeratins and uroplakins. Virchows Arch, 2001. 438(2): p. 181-91. |
