Ectopic Pregnancy
Authors
INTRODUCTION
Lawson Tait,1 the father of gynecologic surgery, reported the first successful operation for ectopic pregnancy in 1883. His main difficulty lay in establishing the diagnosis.
Until a little more than decade ago, little change had occurred in the diagnosis and management of ectopic pregnancy. The clinical use of sensitive pregnancy testing, transvaginal ultrasonography, and diagnostic laparoscopy has had a major impact on the preoperative diagnosis of this condition. The rate of ectopic rupture has declined, and the option of conservative surgical management of an unruptured fallopian tube is now a viable alternative. This chapter briefly reviews the epidemiology of the disease and then focuses on the new diagnostic modalities, options, and therapeutic operations.
INCIDENCE AND RISK FACTORS
The reported incidence of ectopic gestation increased from a total of 17,800 cases in 1970 to 108,800 cases in 1992 (19.7/1000 reported pregnancies).2 Whether the higher reported incidence reflects a true increase in the prevalence of the disease or is secondary to improved diagnostic techniques is unknown. Both factors probably play a role. Factors implicated in the increased incidence of ectopic gestation include widespread use of intrauterine devices (IUDs), higher prevalence of pelvic inflammatory disease (PID), complications of infections, including therapeutic abortions, the wide clinical use of reconstructive tubal surgery, exposure to diethylstilbestrol, and the conservative surgical treatment of ectopic pregnancy itself (Table 1).3, 4, 5, 6 A critical review of the relative contributions of these factors is pertinent. It is widely accepted that when pregnancy occurs in a woman using an IUD, there is an increased likelihood of an ectopic pregnancy. Indeed, the ratio of ectopic pregnancy to intrauterine pregnancy has been reported to have increased sevenfold.7, 8
Table 1. Factors contributing to ectopic pregnancy
Previous pelvic surgery
Contraception
Transperitoneal migration
Diethylstilbestrol exposure
Endometriosis
Infection
Infertility
Maternal age
Maternal race
Hormonal factors
Previous ectopic pregnancy
Previous spontaneous abortion
Alterations in tubal physiology
Assisted reproductive technology
Uterine curettage
The observation that use of an IUD for longer than 2 years increases the risk of an ectopic pregnancy has strengthened the opinion that IUD use increases the overall risks of ectopic pregnancy.9, 10 Lehfeld and colleagues7 reported the findings in 1459 accidental pregnancies occurring in 30,000 women using IUDs. By calculating the incidence of expected intrauterine and ectopic pregnancies, they concluded that IUD use reduced the occurrence of intrauterine pregnancies by 99.5%, tubal pregnancies by 95%, and ovarian implantations not at all.
Ory11 reported the following results of a case-controlled study (the Women's Health Study) regarding the health risks associated with IUD use:
The risk of having an ectopic pregnancy was the same in women with a history of IUD use as in women who had never used this contraceptive device.
The risk was reduced in women who used any form of contraception, including IUDs versus women not using contraception.
The risk was reduced threefold in women who used oral contraceptives compared with users of IUDs and barrier contraception.
The risk triples after 2 years of IUD use but still remains similar to that for women who have never used an IUD.
The risk was independent of the composition of the device (e.g., copper or plastic).
Therefore, it can be concluded that an IUD affords some protection against ectopic gestation for 2 years of use, after which the risk approaches that for women who are not currently using contraception. When accidental pregnancy occurs in a woman using an IUD, there is an increased likelihood that the pregnancy will be an ectopic one.
PID is considered a major etiologic factor in ectopic pregnancy. Because the incidence of PID has risen dramatically over the past two decades, it has been suggested that part of the increased incidence of ectopic gestation can be attributed to this disease. Indeed, women with laparoscopic-proven PID have a sixfold increase of ectopic pregnancy (1:24) over a controlled group undergoing laparoscopy for abdominal pain but without PID (1:146).3 If these figures are applied to the estimate of 500,000 new cases of PID annually in the United States, an expected 13,500 ectopic pregnancies will occur the first year the patients try to conceive, and 25,000 ectopic pregnancies if they try to conceive twice.12 Therefore, PID and its increased incidence are important epidemiologic risk factors for the occurrence of ectopic gestation.
The use of progesterone-only pills has been implicated as a causative factor in ectopic gestation.13 However, when one compares the risk in women using progesterone-only pills (0.6–2.0 per 100 women-years) versus the risk in women using no contraception (0.8–1.2 per 100 women-years) as calculated by Tietze,14 one can conclude that there is no difference in risk for women using progesterone-only pills. There is evidence, however, to suggest a sixfold to eightfold increase in the incidence of ectopic pregnancy in women using progestogen-releasing IUDs compared with that in women using non-progesterone-releasing IUDs, and a threefold increase compared with that in women using no contraception.15, 16
A case-control study conducted in Athens, where abortion is illegal, suggested a 10-fold increased risk of ectopic pregnancy in patients having therapeutic abortions.17 Another study confirmed the increased risk of ectopic pregnancy in patients in whom a therapeutic abortion was associated with a postabortal infection.4
The most important risk factor for ectopic pregnancy is a history of a previous ectopic gestation. This event confers a 10-fold increase in the likelihood of another ectopic pregnancy.18, 19
MORTALITY
Despite the increased frequency of ectopic pregnancy, the case mortality rate has declined from 3.5 deaths per 1000 ectopic pregnancies in 1970 to 0.4 deaths per 1000 ectopic pregnancies in 1985. Although ectopic pregnancies accounted for only 1.5% of the total gestations in 1984 and 1985, they accounted for 14% of maternal deaths in 1984 and 11% in 1985.20
Ectopic pregnancy confers a greater risk of maternal mortality than either childbirth or legal abortion. An extrauterine gestation is 50 times more likely to result in a maternal death than a first-trimester abortion and 10 times more likely than delivery in the third trimester.21
A study of the clinical aspects of ectopic pregnancy mortality in the United States has shown that the most frequent direct causes of death are hemorrhage, infection, and anesthetic complications. Of women dying from hemorrhage, 70% did not undergo surgery. In 50% of women, the condition was misdiagnosed or confused with other pathology such as gastrointestinal disorders, intrauterine pregnancies, spontaneous abortions, PID, sequelae of induced abortion, and psychiatric disorders. In 70% of cases, patients had either called or visited a physician within 1 week of development of symptoms. Diagnostic delay could be ascribed to physician delay in 53% of cases and to combined patient and physician delay in 8% of cases. In 5% of cases, the physician made the diagnosis but did not act promptly enough to prevent maternal death. The site of implantation was also important, as interstitial and nontubal ectopic pregnancies account for only 5–10% of all ectopic pregnancies, but for 20% of all fatalities.22
PATHOPHYSIOLOGY
The mechanisms responsible for ectopic implantation are unknown. The four main possibilities are an anatomic obstruction to the passage of the zygote, an abnormal conceptus, abnormalities in the mechanisms responsible for tubal motility, and transperitoneal migration of the zygote.
Anatomic distortion and obstruction of the fallopian tube are widely believed to be responsible for most ectopic implantations. Obstruction could result from PID, salpingitis isthmica nodosa,5 tubal endometriosis, or postsurgical fibrosis.23 Scarring of the endosalpinx could lead to diverticuli formation, in which the zygote could be trapped, or to simple obstruction of the tubal passage. Support for the contribution of an anatomic cause is the demonstration of histologic and gross evidence of past infection in 30–50% of cases of ectopic gestation.3, 24 Other researchers studying the isthmic portion of the fallopian tube, however, have failed to demonstrate significant pathology (e.g., fibrosis, chronic inflammation of endometriosis) associated with ectopic pregnancy.25 This observation suggests the contribution of nonanatomic factors to the etiology of ectopic pregnancy. Functional causes could include a defective conceptus, abnormalities in the motility of the fallopian tube, or transperitoneal migration.
An abnormal conceptus could theoretically result in defective migration of premature implantation in an ectopic site. This possibility has been investigated by examining the chromosomal constitution of ectopic gestations. Elias and co-workers26 found that the incidence of chromosomal abnormalities in ectopic pregnancies is no different from that in intrauterine pregnancies. The bias with this type of study, however, is that a significant proportion of ectopic pregnancies cannot be adequately karyotyped because of the nonviability of their cells in culture. Similarly, Fedele and colleagues,27 in a case-control study, reported the risk of ectopic pregnancy (after adjustment for maternal age and parity) to be fourfold greater in women with a history of recurrent spontaneous abortion.
Tubal motility seems to be influenced by the hormonal milieu. The suspicion that some cases of ectopic pregnancy may be due to endocrine abnormalities stems from clinical observations that have suggested an association in patients using a progesterone-only pill, an IUD,13, 15, 16 or human menopausal gonadotropins for ovulation induction.28, 29 It has been suggested that high estrogen levels noted in cases of hyperstimulation with human menopausal gonadotropins interfere with tubal transport. An alternative explanation is that an increased number of eggs are released (superovulation) in hyperstimulated patients, resulting in an increased risk of ectopic implantation. In contrast, subnormal estrogen levels subsequent to vigorous exercise and dietary fads have been hypothesized to contribute to increased ectopic rates in today's more health-conscious society.30
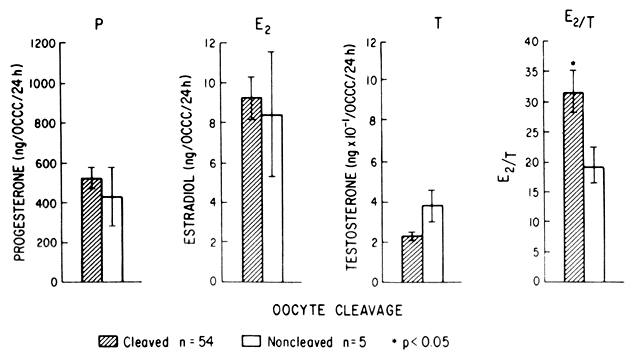
Korenga and Kadota31 have demonstrated the importance of luteal-phase progesterone in circular muscular activity of the fallopian tube. Abnormal progesterone levels in the luteal phase of the cycle could theoretically lead to impaired motility. Laufer and associates32 have shown differences in the secretions of the cumulus cell mass surrounding the ovum in cleaved versus noncleaved eggs (Fig. 1).
Recent SART data from 2001 show that ectopic pregnancy occurs in 1.8% of recipients of embryo transfer during in vitro fertilization and up to 4.3% in patients undergoing zygote intrafallopian transfer (ZIFT).33 Indeed, the first pregnancy reported in humans with this technique was an ectopic pregnancy.34 Postulated mechanisms include the inadvertent injection of embryos into the fallopian tube, uterine irritability stimulating the contractile portion of the tube secondary to cervical and uterine manipulation, and a gravitational effect. It has been suggested that this inadvertent injection or migration of embryos into the fallopian tubes occurs more often than realized. A diseased tube is less likely than a normal tube to propel the embryo back into the uterus.35 In addition, infertile women undergoing gamete and zygote intrafallopian transfer should be counseled regarding at least a theoretic increased risk of tubal gestation.36
PATHOLOGIC AND ANATOMIC CONSIDERATIONS
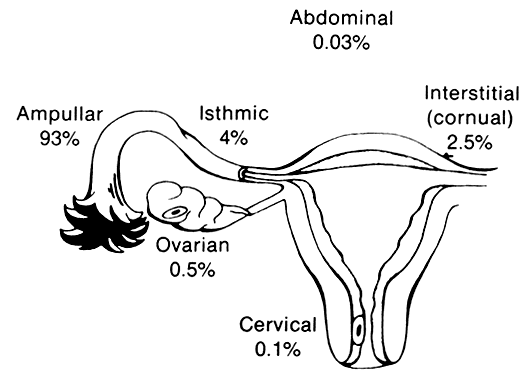
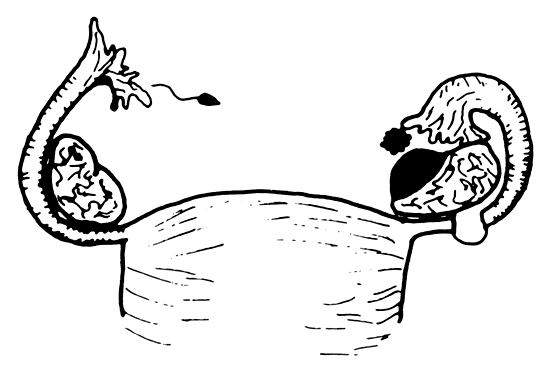
Of all ectopic pregnancies, 97% occur in the fallopian tube (tubal), 2.5% in the uterine cornu, and the remaining 2% in various other locations including the cervix, abdomen, and ovary (Fig. 2). The majority of tubal pregnancies are located in the ampullary portion of the tube. Coexistence of an intrauterine and a tubal pregnancy (heterotopic) was initially reported to occur in 1:30,000 ectopic pregnancies, and the occurrence of bilateral ectopic pregnancy in the same cycle is even rarer.37, 38, 39 With the increased use of ovulation induction, the reported incidence of heterotopic pregnancy has increased to reports of 0.3%.40
|
Cervical pregnancy is a rare cause of mortality today.41 Causes include an endometrial lining that may be unreceptive to implantation (as with infection), uterine fibroids, an atrophic endometrium, a septate uterus, current IUD use, endometrial scarring, and use of oral contraceptives. Cesarean scar pregnancies are also relatively rare occuring at a rate of 1/1800 to 1/2216, but can be a significant cause of morbidity and mortality due to uterine rupture and intractable bleeding.42, 43 These pregnancies are becoming more common due to a rise in the rate of cesarean section. Ovarian pregnancies make up 0.5% of all extrauterine gestations.44, 45, 46 There is a higher proportion of these ectopic pregnancies in patients who use IUDs.46 To be classified as an ovarian pregnancy, a pregnancy must fulfill Spiegelberg's criteria:
The tube on the affected side must be intact.
The fetal sac must occupy a position in the ovary.
The ovary must be connected to the uterus by the ovarian ligament
Decidual ovarian tissue must be found in the sac wall.47
Anatomically, tubal pregnancies can be divided into two distinct types based on the anatomy of the tube in the specific location in which the pregnancy occurs. The first to be considered is an ectopic pregnancy occurring in the ampullary portion. The ampullary portion of the fallopian tube is made up of lining epithelium, loose connective tissue, and an ill-defined muscularis and serosa. This reflects the function of this portion of the tube. It is the incubator where fertilization and early cleavage occur. Therefore, ectopic pregnancies in this area rapidly erode through the tubal epithelium into this loose adventitious area surrounding the lumen; propagation then occurs in the space between the serosa and the tubal lumen, rendering the lining of the tube only partially damaged. This phenomenon was first described by Budowick and co-workers.48 The zygote also usually spends a very brief time in the isthmic portion of the tube. This portion has a very well-defined and functional muscularis, and its primary function is to squeeze the zygote into the waiting endometrial cavity. This compact muscularis causes the embryo to grow within the lumen of the tube, because this is the path of least resistance. The growth of the ectopic pregnancy in this portion of the tube destroys the surrounding luminal epithelium. These differences in the location and growth characteristics of ectopic pregnancies dictate the surgical technique used to correct these specific lesions.
SIGNS AND SYMPTOMS
Schermers49 reviewed the symptoms in 3970 ectopic pregnancies reported in the literature and found pain to be the most common presenting symptom, occurring in 96.3% of the patients; irregular bleeding was second, occurring in 74.1% of the patients. Other symptoms included shoulder pain, gastrointestinal symptoms, and syncope. Also reported as the second most common sign was adnexal tenderness, occurring in 85–95% of patients. The irregular vaginal bleeding might not be a result only of a breakdown of the endometrium, but of blood flowing from the fallopian tube into the uterine cavity and out the cervical os as well.
Other findings also include the Arias–Stella phenomenon and decidual casts (Fig. 3). These are rare occurrences and are more a curiosity than a helpful adjunct in diagnosing an ectopic pregnancy. Physical findings, although helpful, are inconsistent other than in the case of hemorrhagic shock, and a pelvic mass is palpable in only approximately 50% of cases.50 This leads to a need for more objective criteria.
DIAGNOSIS
Patients clinically suspected of having ectopic pregnancy fall into two major categories: those who have an acute abdomen and in whom immediate surgery is indicated, and those who are clinically stable and in whom adjunctive diagnostic procedures can be performed.
Patients with a surgical abdomen are evaluated in the emergency room with a rapid pregnancy test and potentially a culdocentesis. A positive culdocentesis in a patient with a positive pregnancy test result has been reported to correspond with ectopic pregnancy in 99.2% of cases.51
Patients who will benefit most from a culdocentesis are those in whom a clinical suspicion of ectopic pregnancy exists, and who present at a time when expeditious diagnosis is desired and when sophisticated diagnostic modalities, such as ultrasonography and sensitive human chorionic gonadotropin (hCG) assays, cannot be obtained without significant delay. Under these circumstances, culdocentesis is an inexpensive, rapid, and easily performed means of patient evaluation that often provides the impetus for immediate intervention.
The results of a culdocentesis can be classified as negative, positive, or nondiagnostic. A negative culdocentesis is indicated by the presence of clear fluid. A positive result refers to the free flow of nonclotting blood. If no fluid is obtained, the test is considered nondiagnostic. When bloody fluid is obtained, a hematocrit of the aspirate is helpful. Ectopic pregnancies are generally associated with hematocrits of more than 15%. Lower hematocrits frequently indicate the presence of cystic fluid. In our series, 86% of all positive culdocenteses performed in the emergency room were associated with an ectopic pregnancy.51 Other causes of positive culdocentesis included ruptured ovarian cysts, retrograde menstruation, endometriosis, torsion of the fallopian tube, and bleeding of unknown etiology. It should be emphasized that a nondiagnostic culdocentesis should not lower the suspicion of an ectopic pregnancy. In our series, 16% of ectopic pregnancies had nondiagnostic culdocentesis, and one-quarter of these were ruptured at the time of operation.51
In clinically stable patients, the approach to the evaluation is based on the combined use of sensitive pregnancy testing, ultrasound examination, and/or laparoscopy. hCG testing is used to screen for pregnancy, and ultrasonography is employed to locate it.52
Evaluation of the patient begins with a sensitive and rapid pregnancy test. Blood and urine pregnancy tests have been used to screen for ectopic pregnancy. Advances in technologies of enzyme-linked immunoassay and monoclonal antibodies to the β subunit have promoted the evolution of rapid, inexpensive, and similar qualitative urinary pregnancy tests. However, we prefer to use the sensitive blood pregnancy testing in the form of a radioimmunoassay against β-hCG. We recommend an assay with a sensitivity of 2.5 mIU/mL because with this system the false-negative rate is 0.5%. The radioreceptor assay, with a sensitivity of 200 mIU/mL, is not suitable for screening because its false-negative rate ranges between 6% and 12%, which is unacceptably high for a life-threatening condition.53, 54
A negative blood pregnancy test virtually rules out pregnancy and thus an ectopic gestation. A positive qualitative hCG result requires further investigation, mainly a determination of the hCG titer and an ultrasonographic examination (either abdominal or vaginal) of the pelvis.
A positive diagnosis of ectopic pregnancy can be made if fetal motion is demonstrated outside the uterus. Unfortunately, this is a rare and late finding, and awaiting its appearance would delay the diagnosis and conceivably increase the risk of tubal rupture. In practice, ultrasonography is used to identify an intrauterine pregnancy, which would render the simultaneous presence of an ectopic pregnancy extremely unlikely (1:30,000). Confirmation of the presence of an intrauterine pregnancy can be made by identifying either a gestational sac or a fetal pole within the endometrial cavity.
Ultrasonographically, ectopic pregnancies can show a single-ring sac due to the presence of blood within the endometrial cavity in association with a significant decidual reaction. This appearance has been shown to occur in 10–20% of all cases (Fig. 4).55, 56, 57, 58, 59, 60, 61, 62, 63
Nyberg and colleagues64 and Bradley and associates65 have proposed morphologic criteria to distinguish the pseudogestational sac of ectopic pregnancy from the gestational sac of a normal intrauterine pregnancy. They have described the normal intrauterine gestational sac as having a double contour produced by the decidua capsularis and the decidua parietalis. The pseudogestational sac of an ectopic pregnancy has only a single ring. The researchers have reported that 98.3% of all patients with a double-ring sac had an intrauterine pregnancy and 64 of 68 patients with a single-ring sac had ectopic pregnancies.
The traditional abdominal ultrasonographic criterion used for the diagnosis of ectopic pregnancy is failure to visualize a gestational sac in the uterus of patients with more than 6 weeks of amenorrhea. The problem with this criterion is that one third of patients with ectopic pregnancy do not know the date of their last menstrual period, and in others irregular bleeding makes interpretation of the menstrual history difficult.
It has been established that the sac of a normal intrauterine pregnancy becomes visible with abdominal ultrasonography when the hCG titer is greater than 6500 mIU/mL. When levels are higher than this, the absence of a sac is associated with an ectopic pregnancy in 86% of cases. This criterion has a sensitivity of 100%, a specificity of 96%, and a negative predictive value of 100%.51 The absence of a sac at levels less than 6000 mIU/mL is a nondiagnostic finding and should not lower or raise the suspicion of ectopic pregnancy.66, 67 Management of these patients depends on the clinical situation, however, if transvaginal ultrasonography is available, it should be employed. If a patient remains clinically stable, serial hCG determinations are useful. Additionally, management can be based upon ultrasonographic findings of the adnexa and clinical suspicion. Romero and colleagues68 prospectively evaluated 220 patients who were suspected of having ectopic pregnancy and who had hCG titers less than 6000 mIU/mL and abdominal ultrasonographic adnexal findings. The demonstration of a noncystic mass, alone or associated with cul-de-sac fluid, was an indication for diagnostic laparoscopy.
The value of transvaginal ultrasonography in the detection of ectopic pregnancy, especially in cases of hCG titers less than 6000 mIU/mL, is now recognized. Proximity of the ultrasound transducer to both the adnexa and the cul-de-sac allows increased resolution and image quality with the detection of an intrauterine pregnancy up to 7 days earlier than with the classic transabdominal approach (Fig. 5).
Vaginal scanning has proved to be more accurate than abdominal scanning in detecting ectopic pregnancies (90% vs 80%) and cul-de-sac fluid (77% vs 46%) and in discerning whether the tubal pregnancy has ruptured (76% vs 50%).69 In the transvaginal ultrasonographic evaluation of pregnancy, Bernaschek and associates,70 using a 5-MHz transducer, proposed a “discriminatory zone” of an hCG titer of 750 mIU/mL (second International Standard) for the detection of an intrauterine gestational sac. Unfortunately, using this proposed criterion and similar ultrasonographic equipment, Fossum and associates71 may have inadvertently surgically investigated several pregnancies that proved to be normal intrauterine gestations (Fig. 6). If an hCG titer exceeds 2000 mIU/mL, we now expect to detect an intrauterine gestational sac using transvaginal ultrasonography.
If ultrasonography is indeterminant, the value of serial hCG determinations cannot be overemphasized. Traditionally, quantitative hCG values were expected to rise by 66% over 2 days. However, this data was based upon a study of 20 patients in 1980 using an 85% confidence interval.72 More recent data indicate the potential for a slower rate of rise in viable intrauterine gestations. Barnhart and colleagues based their data on 281 patients who presented with pain or bleeding in early pregnancy and found that a 53% increase in 2 days represents the minimal rise for a viable pregnancy (confidence interval 99%).73 Another recent study focused on a cohort of patients eventually diagnosed with ectopic pregnancy and found that the hcg profile mimicked a normal viable pregnancy 29% of the time.74
Declining hCG levels may be indicative of a nonviable, nondetectable intrauterine gestation or could still be representative of an ectopic pregnancy. Up to 40% of patients eventually diagnosed with ectopic pregnancy may have initially declining serum hCG values.74 Serum progesterone values may prove a useful adjunct to hCG titers in distinguishing a viable intrauterine pregnancy from that of an ectopic or missed abortion. Accordingly, in two separate reports on 99 women, a progesterone value less than 15 ng/mL was always predictive either of an ectopic pregnancy or a nonviable intrauterine gestation.75, 76
We recommend laparoscopy or dilation and curettage (D&C) for patients with subnormal quantitative increases of hCG. Curettage productive of trophoblastic tissue confirms an abnormal intrauterine pregnancy and completion of therapy. Approximately 30% of patients with abnormally rising quantitative hCG values will have an abnormal intrauterine pregnancy. The remainder will be ectopics. Unfortunately, this test is limited by a false-negative rate of up to 20%.77 Endometrial biopsy with pipelle is even less sensitive than D&C for detection of chorionic villi with a sensitivity somewhere between 30 and 60%.78, 79
Normal increments in hCG are monitored until hCG titers exceed either 2000 or 6500 mIU/mL, at which time either transvaginal or transabdominal ultrasonography, respectively, may be performed. The presence or absence of a gestational sac above these hCG levels should be supportive either of an ectopic or an intrauterine gestation. A good algorithm for the management of ectopic pregnancy is shown below (see Fig. 7).
SURGICAL MANAGEMENT
The ultimate decision regarding surgical management depends on a patient's desire for future fertility. However, it is important to note that whatever procedure is performed, the pregnancy rate after an ectopic pregnancy may be decreased by 40–70%.47 If a patient is not interested in future fertility, the appropriate surgical procedure is salpingectomy. Additional indications for salpingectomy include recurrent ectopic pregnancy in the same tube, a severely damaged tube, prior unsuccessful conservative surgery, and uncontrolled bleeding.80 If a patient does desire future fertility, however, much data from the past few years support performing conservative surgery in a majority of these cases. Conservative options vary from expression of a tubal abortion through the distal end of the tube to linear salpingostomy to segmental resection and secondary anastomosis of an isthmic ectopic pregnancy.
A majority of ectopic pregnancies are associated with contralateral tubal disease, such as salpingitis. The fact that clinical assessment and evaluation of the contralateral tube during the initial surgery correlates poorly with postoperative hysterosalpingographic findings underscores the importance of intraoperative conservative management whenever feasible.81 It is not recommended that a test of tubal patency be done during the operation for an ectopic pregnancy in order to evaluate the status of the contralateral tube. The information provided can be erroneous because decidual reaction around the cornual portion might give a false impression of an occluded tube. The possibility of introducing infection into an already compromised pelvis is also an important consideration.
Laparotomy versus Laparoscopy
A number of early studies documented the appropriateness of laparoscopic treatment of ectopic pregnancies.82, 83, 84 Rates of conception of an intrauterine pregnancy after the procedure were as high as 70% in these cases. Pouly and associates85 reported on 321 women with ectopic pregnancies who underwent conservative laparoscopic treatment. Of the women who did not have a history of infertility or a previous ectopic pregnancy, 86% had a subsequent intrauterine pregnancy.
The advantages of laparoscopic removal of an ectopic pregnancy are a shortened operating time, convalescence, and hospital stay. It is imperative, however, that proper case selection be exercised.86 If laparoscopic therapy is to be warranted, the first criterion is the expertise of the operator in performance of a laparoscopic surgical procedure. Patients must be stable without evidence of a significant hematoperitoneum.
One of the complications of conservative surgery via laparoscopy, persistent ectopic pregnancy, appears to be higher with laparoscopy (5–20% vs. 2–11%). This is thought to be associated with the learning curve seen with laparoscopy. Optimally, the ectopic pregnancy should be confined to the ampullary portion of the tube and should at least 2 cm in size.87 The tube must be easily accessible.
Salpingectomy versus Salpingo-oophorectomy
In 1955, Jeffcoate88 suggested that in conjunction with a salpingectomy, which is still the most commonly employed surgical treatment for ectopic pregnancy, an oophorectomy on the ipsilateral side be done as well. The theory behind this is that all ovulations would be into the good tube; this discounts the importance of transmigration. There are no data that justify this conjecture. Several reports have shown that there is no advantage in performing a salpingo-oophorectomy compared with salpingectomy in terms of pregnancy rates and repeat ectopic pregnancies in those patients.89 Considering the successful pregnancy rates with in vitro fertilization and embryo transfer, every effort must be maintained to conserve as much ovarian tissue as possible in any eventuality.
Salpingectomy versus Salpingostomy (Conservative Surgery)
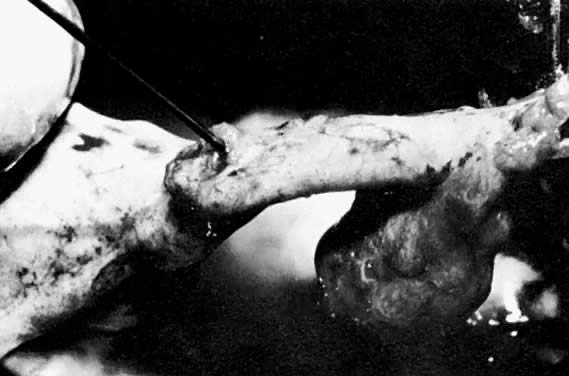
In a stable patient who desires future childbearing, a salpingostomy is the treatment of choice. The incision for a salpingostomy is made on the antimesenteric border of the tube over the area of maximal bulge; as small an incision as possible is made, but a surgeon must make sure that the products of conception are removed entirely. There seems to be no quantitative difference between use of a laser, scalpel, or electrocautery to make the incision. The products of conception can be removed with duckbill forceps or hydrodissection (Fig. 12).84 It is also important not to debride the base or to treat the base too vigorously to achieve hemostasis. These incisions in the tube seem to heal well, as documented in follow-up surgery and hysterosalpingogram. Hemostasis is critical. Preincisional serosal surface injection of vasopressive agents often reduces incisional bleeding, but may distort the area of maximal tubal bulge. If bleeding continues, vessels in the broad ligament can be ligated with fine suture material (4-0), but remember that this ligation devascularizes the tube and may cause some ischemia in the tube that may lead to decreased reproductive function and adhesion formation.90
|
The controversy surrounding salpingectomy versus salpingostomy was based on two assumptions. Those in support of salpingectomy feared (1) that patients undergoing salpingostomy would be more likely to have a repeat ectopic pregnancy, and (2) that salpingostomy is a tedious surgical procedure because tubal hemorrhage during surgery is difficult to control. Neither of these assumptions has proved correct, and in fact, intrauterine pregnancy rates have actually increased or remained stable with the use of conservative surgical techniques (Table 3),91 primarily as a result of the application of modern microsurgical techniques to acute tubal pathology. Early reports showed an increased repeat ectopic pregnancy rate (21% after conservative surgery vs. 12.5% after salpingectomy).92 Since then, results have been contradictory with some demonstrating a lower repeat ectopic pregnancy rate93, 94, 95 and others a higher repeat ectopic pregnancy rate with conservative surgery.96, 97
Table 3. Choice of surgical treatment and subsequent fertility among 151 fatients with ectopic pregnancy
No. of subsequent pregnancies | Incidence of sterility | |||
Surgical treatment | No. of patients | Intrauterine | Repeat ectopic |
|
Conservative | 47 | 39 (83) | 3 (6.4) | 175 |
Radical | 104 | 75 (72.1) | 6 (5.8) | 225 |
(Sherman D, Lunger R, Sadovsky G: Improved fertility following ectopic pregnancy. Fertil Steril 37:497, 1982. Reproduced with permission of the publisher, The American Fertility Society)
There are no substantial data to support the role of cornual resection in conjunction with a salpingectomy. The theoretic rationale is to prevent interstitial implantation. A review of the literature by Kalchman and Meltzer98 demonstrated only 75 cases of interstitial pregnancy following a salpingectomy, leading to the conclusion that this is a rare occurrence and should not dictate surgical technique. Hallatt has suggested that the procedure has some inherent risks secondary to increased bleeding at the time of the original surgery and the possibility of a rupture in the course of a subsequent intrauterine pregnancy.89 Uterotubal fistula formation after this procedure is also a possibility. Therefore, for aesthetic reasons, only a small, shallow cornual resection should be performed.
Salpingostomy versus Salpingotomy
The primary difference between a salpingostomy and a salpingotomy is that, in the latter, the fallopian tube is closed by primary intention. Stromme99 was the first to describe salpingotomy. This procedure involves an antimesenteric incision over the ectopic pregnancy, excising the products of conception and closing the tube in either one or two layers with fine suture material after hemostasis is achieved. The sutures used should be interrupted. Other reports have modified this technique but still espouse primary closure of the tube.91, 94 Anecdotal reports of tuboperitoneal fistulas subsequent to salpingostomy, but not salpingotomy, support performance of the latter procedure.100
A salpingostomy can be performed based on the evidence describing the propagation of ampullary ectopic pregnancies outside of the lumen itself in the adventitious space between the serosa and the tubal epithelium. Salpingostomy may be associated also with an increased incidence (43%) of recurrent ectopic pregnancy, decreased chances of complete trophoblast removal, and delayed postoperative hemorrhage.101, 102, 103, 104
Salpingostomy seems to have no adverse effect on a tube's anatomic and physiologic future. This is a simpler procedure that can be used by all gynecologic surgeons. In 1951, Israel105 performed total linear salpingostomies in eight patients for PID. The procedures were done between 1935 and 1949. Seven of the eight were done for gonorrheal salpingitis and one for tuberculous salpingitis. Five of the eight patients went on to conceive. The procedure was then tried on six dogs. The dogs were operated on at 2-, 4-, and 8-week intervals. At 2 weeks, the raw edges were separated from each other and epithelialized. At 4 weeks, the gaping incision was beginning to be bridged by fibrous tissue, and at 8 weeks the organs appeared totally healed, with “fusion of the tunica propria as well as a conjunction of the circular muscle layer. Its salient feature is the absence of a visible scar.”105
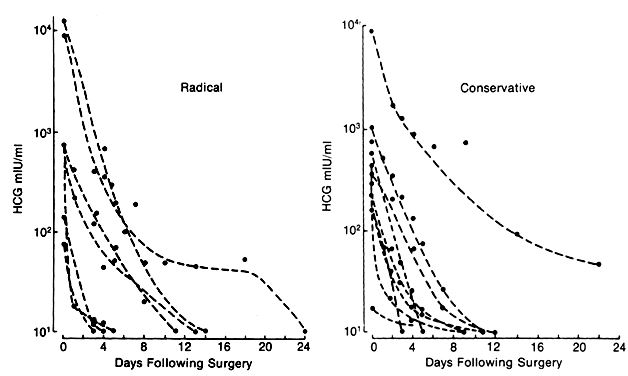
Blood testing demonstrates the disappearance of β-hCG levels over time (Fig. 8.)106
ISTHMIC AND INFUNDIBULAR ECTOPIC PREGNANCY AND TUBAL ABORTIONS
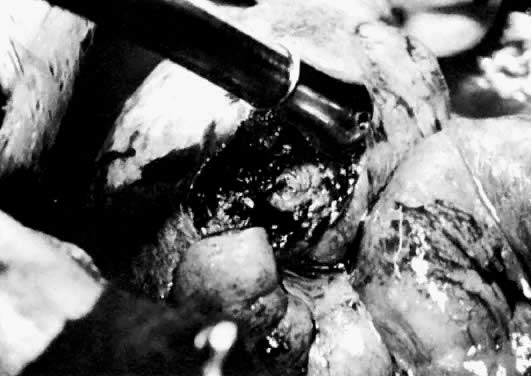
Ectopic pregnancies that occur in the isthmic portion of the tube present a different entity entirely. A recent histopathologic comparison of ampullary and isthmic ectopic pregnancies noted preservation of the ampullary muscularis in 85% of the former cases and only a 43% preservation rate of the muscularis in isthmic pregnancies. The disruption of the tubal wall was also more extensive in isthmic pregnancies.107 This is why patients with isthmic ectopic pregnancies are believed to be the only ectopic pregnancy patients who develop a tuboperitoneal fistula after linear salpingostomy (Fig. 9). Therefore, segmental resection is most commonly recommended.108
The option then becomes to close the tube by primary intention, which has been done successfully by Stangel and colleagues,109 or to perform a second procedure and do an anastomosis at that time. The major advantage of doing an anastomosis at the time of resection of the ectopic pregnancy is that the wide lumen of the tube facilitates the anastomosis. However, edema and the presence of blood may increase the chance of infection. Normal intrauterine pregnancies are achieved regardless of the mode of anastomosis employed. It is recommended that patients waiting for a secondary anastomosis be placed on birth control pills so that they do not form an ectopic pregnancy in the blind distal portion of the tube that has been created (Fig. 10).110 In either event, the anastomosis is performed in two layers using the operating microscope. Four to six 8-0 polyglycolic acid or polydioxanone sutures are placed approximating the muscularis. The serosa is then loosely opposed with running 6-0 Vicryl or PDS sutures (Fig. 11).
With the move toward laparoscopic conservative surgery, it is of interest to note Smith and colleagues'111 similar postoperative tubal patency rates when microsurgical anastomosis was compared with linear salpingostomy in the treatment of isthmic ectopic gestation at the time of laparotomy. Pouly and associates85 accordingly treated 22 isthmic ectopic pregnancies by laparoscopic linear salpingostomy, yielding a 54.5% subsequent intrauterine pregnancy rate. DeCherney and Boyers,112 however, showed that treating isthmic pregnancy with linear salpingostomy by laparotomy resulted in subsequent occlusion of the tube in three of four patients.
A rare form of ectopic pregnancy occurs in the infundibular portion of the tube between the fimbria and the ampulla. These are usually treated by “milking or squeezing” the pregnancy out through the distal end. Schermers49 reported a higher incidence of delayed hemorrhage after the milking-out procedure. This experience has been substantiated by Bruhat.82 Success rates are also lowest with this procedure; therefore, these patients also should have a linear salpingostomy rather than “milking out.”
This entity is different from a tubal abortion, in which the products of conception are already partially or completely extruded from the fimbriated end of the fallopian tube. This process can be completed, if necessary, simply by removing the remaining products of conception.
ADJUNCTIVE THERAPY
Consideration should be given to the use of prophylactic antibiotics, such as doxycycline 100 mg twice a day. Additionally, Rh-negative patients should receive Rh0 (D) immune globulin (RhoGAM).113 Postoperatively, patients should be followed with a hysterosalpingogram 4 months after surgery and laparoscopy 1 year after surgery if pregnancy has not occurred.
NONOPERATIVE MANAGEMENT
Nonoperative management can be divided into observation/expectant care and medical therapy. As early as 1955, Lund114 reported on a series of 119 women with ectopic pregnancies treated with observation. In this series, 55% of the cases resolved without intervention; the remainder required laparotomy. More recently, several investigators have reported their experience with expectant management of unruptured ectopic pregnancy. In Maymon's115 review of 8 series of patients (total of 81 patients) managed expectantly, 76% resolved spontaneously, 23% required laparotomy for rupture, and 79% demonstrated ipsilateral tubal patency on follow-up hysterosalpingogram. The subsequent pregnancy rate, however, was only 16%. In all these studies, patients were rigorously selected, with documented unruptured ectopic pregnancies at laparoscopy, with no active bleeding. The only advantage seen in these reports was the avoidance of surgery. Disadvantages included an increased risk of tubal rupture, a possible increase in pelvic adhesions, and a protracted course of illness.
At this time, expectant management should be considered an option only for patients with extreme surgical risk, falling hCG titers, or in a research setting.
MEDICAL THERAPY FOR ECTOPIC PREGNANCY
The use of drug therapy for ectopic pregnancy was first reported in 1982, in a patient with an interstitial pregnancy who refused surgery.116 Since then, many investigators have reported the successful treatment of selected patients using a variety of agents, including parenteral methotrexate, intratubal or intra-amniotic methotrexate, and intratubal osmotic agents.117, 118, 119, 120, 121 To date, there have not been a significant number of reports of successful treatment of ectopic pregnancy using mefipristone (RU-486).
At this time, parenteral (intramuscular) methotrexate is the best studied and most accepted agent for the medical treatment of ectopic pregnancy. Methotrexate is a folic acid antagonist that interferes with the synthesis of DNA. It is most commonly administered intramuscularly, though reports of oral administration are available.122 Patients receiving medical therapy must be hemodynamically stable and desire future fertility. Contraindications include a ruptured ectopic, ectopic mass greater than 3.5 cm, fetal cardiac activity, high level hCG value (10,000 IU), breastfeeding, immunodeficiency, elevated creatinine or liver function tests, alcoholism, and active pulmonary or gastrointestinal disease.
In 1993, Stovall117 reported a prospective study of 120 women treated with a single dose (50 mg/m2) of intramuscular methotrexate. In this series, the author reported a success rate of 94%, with subsequent tubal patency rates similar to those quoted for conservative surgical treatment. Pregnancy rates were nearly 80%, with approximately 88% of these being intrauterine. There were no adverse effects of methotrexate therapy reported in this series; however, there have been recent reports of tubal rupture and hematosalpinx after methotrexate therapy.123, 124 Overall, the outcome of medical treatment of ectopic pregnancy closely approximates results obtained surgically, and there is evidence to support a significant improvement in cost-effectiveness.125 Lipscomb et al. in 1998 reported on 315 patients treated with single-dose methotrexate and reported a success rate of 90.1%.126
There has been some controversy over the use of single-dose versus multiple-dose methotrexate. Direct comparisons of the two therapies have so far found no significant difference.87 A recent study described higher ipsilateral tubal obstruction (as noted by hysterosalpingogram) in patients treated with multiple-dose versus single-dose regimens. This may be attributable to preexisting obstruction or the potential for tubal harm with a multiple dose of methotrexate.127 Additionally, there is some concern for the long-term effects of methotrexate on ovarian function, in that chemotherapy for gestational trophoblastic tumors may hasten menopause by 3 years.128 Although the harm with multiple-dose therapy has not been proven, there does not appear to be any additional benefit over single-dose therapy.
Once a patient is treated with medical therapy for ectopic pregnancy, she must be followed closely to ensure resolution. Day 1 is considered the day of administration of methotrexate. Follow-up hCG levels should be obtained on days 4 and 7, with an expected 15% drop between the two latter values. Thereafter, hCG values should be followed weekly until negative.
Treatment failure is indicated by a less than 15% drop in hCG values between days 4 and 7, worsening abdominal pain concerning for rupture, and increasing or plateauing hCG values after the first week of therapy. If the patient is hemodynamically stable, a second injection of methotrexate can be given with weekly follow-up. A second dose may be required in at least 11% of patients. Tubal rupture may occur in up to 8% of patients requiring subsequent surgery.129
Patients should be counseled regarding the side effects of medical therapy. These can include nausea and vomiting, abdominal pain, diarrhea, stomatitis, dizziness, and rarely neutropenia or reversible alopecia.130
REPRODUCTIVE PERFORMANCE AFTER TREATMENT FOR ECTOPIC PREGNANCY
Conservative surgical treatment can be carried out without lowering the chance of a subsequent intrauterine pregnancy or raising the incidence of an extrauterine pregnancy. A review of the literature summarizing 467 cases operated on for ectopic pregnancy is summarized in Table 4.131 Of these patients, 46% had a subsequent intrauterine pregnancy and 12% had a repeat ectopic pregnancy. These results do not differ greatly from older statistics based on the results after radical surgery. DeCherney and Oelsner131 reviewed 1630 cases treated radically and found a 41% intrauterine pregnancy rate and a 14% repeat extrauterine pregnancy rate. It is hoped that over time, with the use of modern microsurgical techniques, the intrauterine pregnancy rate will increase and the repeat ectopic pregnancy rate will decrease in these patients.
Table 4. Results of radical and conservative surgery for extrauterine pregnancy
| Women desiring pregnancy | Women with | Women with |
Radical sugery | 1630 | 667 (41%) | 231 (14%) |
Conservative surgery | 467 | 215 (46%) | 54 (12%) |
(Adapted from DeCherney AH, Oelsner G: Ectopic pregnancy. In Garcia C-R, Mikuta JJ, Rosenblum NG (eds): Current Therapy in Surgical Gynecology. Toronto, BC Decker, 1986)
Based on the similar findings in radically and conservatively treated patients, an important question is whether salpingostomy works at all. This begs the question that perhaps those patients who conceive are using the tube that was not operated on. This possibility has been reviewed, and the intrauterine pregnancy rate and the repeat ectopic pregnancy rate seem to be the same in patients who had salpingostomies performed on only one fallopian tube with an ectopic pregnancy for both the group receiving radical treatment and the group receiving conservative treatment.47
These statistics do not hold, however, for patients who have had two or more ectopic pregnancies. In these patients, the incidence of repeat ectopic pregnancy approximates that of a subsequent intrauterine gestation.132, 133 An unanswered question at this point remains: How many conservative procedures for an ectopic pregnancy can a patient undergo before her reproductive future is compromised to the point where in vitro fertilization is the only viable alternative (Table 5)?132, 133
Table 5. Reproductive Outcome of 47 Patients After Two Ectopic Pregnancies
| Number of Patients (% in parentheses) | |
Outcome | DeCherney et al. N = 23 | Tulandi N = 24 |
Sterilized (unable to conceive) | 6 (26.1) | 4 (16.7) |
At least one tube present | 17 (73.9) | 20 (83.3) |
Using contraception | 4 (23.5) | 4 (20) |
Trying to conceive | 13 (76.5) | 16 (80) |
Intrauterine pregnancy | 4 (30.8) | 8 (50.0) |
Term | 4 (30.8) | 5 (31.3) |
Spontaneous abortion | 0 (0.0) | 3 (18.8) |
Third ectopic gestation | 1 (20.0) | 3 (27.3) |
(% of those trying to conceive) | (7.7) | (18.8) |
Not able to conceive | 8 (61.5) | 5 (31.3) |
(Adapted from DeCherney AT, Silidker JS, Mezer HC et al: Reproductive outcome following two ectopic pregnancies. Fertil Steril 43:82, 1985; Tulandi T: Reproductive performance of women after two ectopic pregnancies. Fertil Steril 50:164, 1988, Reproduced with permission of the publisher, The American Fertility Society) With the advent of effective medical therapy for ectopic pregnancy, there exist a wide variety of available treatment choices. A recent review compared laparoscopic salpingostomy, multiple-dose methotrexate, single-dose methotrexate, and expectant management. The authors concluded that there was no significant clinical difference between the four groups with subsequent intrauterine pregnancy rates varying from 52-61% and subsequent ectopic pregnancy rates ranging from 8–13%. However, successful resolution with expectant management appeared lower at 68% (compared with 87–93% for other therapies).134 Therefore it seems reasonable to conclude that treatment choice should be based upon physician comfort and patient characteristics.
CONCLUSION
During the past decade, the incidence of ectopic pregnancy has noticeably increased. Although 50% of ectopic pregnancies are attributable to infection, the remainder remain unexplained. Clinical findings such as adnexal tenderness, irregular bleeding, and abdominal pain still represent reliable but imperfect clues that a patient has an ectopic pregnancy. It is the advent of newer diagnostic techniques, including laparoscopy, serial β-hCG testing, and transvaginal ultrasonography, that has allowed for the earlier diagnosis of ectopic pregnancy. Previously, 85% of ectopic pregnancies were diagnosed as ruptured and 15% as unruptured.135 Today, this ratio is reversed. The diagnosis of many cases of small, unruptured ectopic pregnancies has led to a reversal from surgical management to medical management with methotrexate especially in patients desirous of future fertility. The hallmark of improved diagnostic management remains the β-hCG test, with failure to achieve a normal slope of increase during the early stages of gestation heralding an ectopic pregnancy or an inevitable abortion.136
REFERENCES
Tait RL: Five cases of extrauterine pregnancy operated upon at the time of pregnancy. Br Med J 1: 1250, 1884 |
|
Centers for Disease Control and Prevention (CDC). Ectopic pregnancy--United States, 1990-1992. MMWR Morb Mortal Wkly Rep. 1995;44(3):46-8 |
|
Westrom L, Bengtsson LPH, Mardh PA: Incidence trends, and risks of ectopic pregnancy in a population of women. Br Med J (Clin Res) 282: 15, 1981 |
|
Chung CS, Smith RG, Steinhoff PG et al: Induced abortion and ectopic pregnancy in subsequent pregnancies. Am J Epidemiol 115: 879, 1982 |
|
Majumdar BH, Henderson PH, Semple L: Salpingitis isthmica nodosa: A high-risk factor for tubal pregnancy. Obstet Gynecol 62: 73, 1983 |
|
Wolf GC, Thompson NJ: Female sterilization and subsequent ectopic pregnancy. Obstet Gynecol 55: 17, 1980 |
|
Lehfeld H, Tietze C, Gorstein F: Ovarian pregnancy and the intrauterine device. Am J Obstet Gynecol 108: 1005, 1970 |
|
Vessey MP, Johnson B, Doll R et al: Outcome of pregnancy in women using an intrauterine device. Lancet 1: 495, 1974 |
|
Tatum JH, Schmidt FM, Jain AK: Management and outcome of pregnancies associated with the copper T intrauterine contraceptive device. Am J Obstet Gynecol 126: 869, 1976 |
|
Sivin I: Copper 1 IUD use and ectopic pregnancy rates in the United States. Contraception 19: 151, 1979 |
|
Ory HW: Ectopic pregnancy and intrauterine contraceptive devices: New perspectives. Obstet Gynecol 57: 137, 1981 |
|
Urquhart J: Effect of the venereal disease epidemic on the incidence of ectopic pregnancy: Implications for the evaluation of contraceptives. Contraception 19: 455, 1979 |
|
Bonnar J: Progesterone-only contraception and tubal pregnancies. Br Med J 1: 287, 1974 |
|
Tietze C: Extrauterine pregnancy and intrauterine devices. Br Med J 2: 302, 1966 |
|
Snowden RF: The Progestasert and ectopic pregnancy. Br Med J 4: 1600, 1977 |
|
Diaz S, Croxatto HB, Pavez M et al: Ectopic pregnancy associated with low dose progesterone-releasing IUD's. Contraception 22: 259, 1980 |
|
Panayotou PP, Kaskarelis DB, Mietinnen OS et al: Induced abortion and ectopic pregnancy. Am J Obstet Gynecol 114: 507, 1972 |
|
DeCherney A, Minkin MJ, Spangler S: Contemporary management of ectopic pregnancy. J Reprod Med 26: 519, 1981 |
|
Douglas ES, Singleton HM, Crist T: Surgical management of tubal pregnancy: Effect on subsequent fertility. South Med J 62: 954, 1969 |
|
Lawson HW, Atrash HK, Saftlas AF et al: Ectopic pregnancy surveillance, United States 1970 - 1985. MMWR 37: 9, 1988 |
|
Dorfman SF: Deaths from ectopic pregnancy. United States, 1979 to 1980. Obstet Gynecol 64: 386, 1984 |
|
Dorfman SF: Ectopic pregnancy mortality, United States, 1979 to 1980: Clinical aspects. Obstet Gynecol 64: 386, 1984 |
|
Siegler AM: Salpingotomy and salpingectomy: indications and techniques for tubal pregnancy. In Sciarra JJ (ed): Gynecology and Obstetrics. Philadelphia, JB Lippincott, 1991 |
|
Hallatt JG: Ectopic pregnancy associated with the intrauterine device: A study of seventy cases. Am J Obstet Gynecol 106: 227, 1976 |
|
Fortier KJ, Haney AF: The pathologic spectrum of uterotubal junction obstruction. Obstet Gynecol 65: 93, 1985 |
|
Elias S, LeBeau M, Simpson JL et al: Chromosome analysis of ectopic human conceptuses. Am J Obstet Gynecol 141: 698, 1981 |
|
Fedele L, Acaia B, Parazzini F et al: Ectopic pregnancy and recurrent spontaneous abortion: Two associated reproductive failures. Obstet Gynecol 73: 206, 1989 |
|
Gemzell G, Guillome J, Wang CF: Ectopic pregnancy following treatment with human gonadotropins. Am J Obstet Gynecol 143: 761, 1982 |
|
McBain JC, Evans JH, Pepperell RJ et al: An unexpectedly high rate of ectopic pregnancy following the induction of ovulation with human pituitary and chorionic gonadotrophin. Br J Obstet Gynaecol 87: 5, 1980 |
|
James WH: A hypothesis on the increasing rates of ectopic pregnancy. Paediatr Perinat Epidemiol 3: 189, 1989 |
|
Korenga M, Kadota T: Changes in mechanical properties of the circular muscle of the isthmus of the human fallopian tube in relation to hormonal domination and post-ovulatory time. Fertil Steril 36: 343, 1981 |
|
Laufer N, Tarlatzis BC, DeCherney AH et al: Steroid secretion by human oocyte-corona-cumulus complexes associated with conceptions following in vitro fertilization. J In Vitro Fert Embryo Transfer 2: 156, 1985 |
|
American Society for Reproductive Medicine/Society for Assisted Reproductive Technology. Assisted reproductive technology in the United States: 2001 results generated from the American Society for Reproductive Medicine/Society for AssistedReproductive Technology registry. Fertil Steril 2007;87(6):1253-66. Epub 2007 Feb 2. |
|
Steptoe PC, Edwards RG: Reimplantation of the human embryo with subsequent tubal pregnancy. Lancet 1: 880, 1976 |
|
Correy JF, Watkins RA, Bradfield GF et al: Spontaneous pregnancies and pregnancies as a result of treatment on an in vitro fertilization program terminating in ectopic pregnancies or spontaneous abortions. Fertil Steril 50: 85, 1988 |
|
Olive DL, Taylor N, Cothran GE et al: Gamete intrafallopian transfer (GIFT) complicated by bilateral ectopic pregnancy. Fertil Steril 49: 719, 1988 |
|
Maheux R, DeCherney AH: Surgical management of tubal pregnancy. In DeCherney AH, Polan ML (eds): Seminars in Reproductive Endocrinology, Vol 2, pp 175 - 185. New York, Thieme-Stratton, 1984 |
|
Winer AE, Bergman WD, Fields C: Combined intra- and extrauterine pregnancy. Am J Obstet Gynecol 74: 1701, 1957 |
|
Shah Y, Zevallos H, Moody L: Combined intra- and extrauterine pregnancy. J Reprod Med 25: 290, 1980 |
|
Barrenetxea G, Barinaga-Rementeria L, Lopez de Larruzea A et al: Heterotopic pregnancy: two cases and a comparative review. Fertil Steril 2007;87(2):417.e9-15. Epub 2006 Oct 30 |
|
Parenta JT, Ou CS, Levy J et al: Cervical pregnancy analysis: A review and report of five cases. Obstet Gynecol 62: 79, 1983 |
|
Reyftmann L, Vernhet H, Boulot P: Management of massive uterine bleeding in a cesarean scar pregnancy. Int J Gynaecol Obstet. 2005 May;89(2):154-5. |
|
Jurkovic D, Hillaby K, Woelfer B et al: Cesarean scar pregnancy. Ultrasound Obstet Gynecol 2003;21(3):310 |
|
Bobrow ML, Winelstein LB: Intrafollicular ovarian pregnancy. Am J Surg 91: 991, 1956 |
|
Hallatt JG: Primary ovarian pregnancy: A report of twenty-five cases. Am J Obstet Gynecol 143: 55, 1982 |
|
Groycol Ruffolo EH: Ovarian pregnancy associated with intrauterine contraceptive devices. Am J Obstet Gynecol 132: 134, 1978 |
|
DeChemey AH, Maheux R: Modern management of tubal pregnancy. Am Prob Obstet Gynecol 6: 1, 1983 |
|
Budowick M, Johnson TR Jr, Genadry R et al: The histopathology of the developing tubal ectopic pregnancy. Fertil Steril 34: 169, 1980 |
|
Schermers JA: Ectopic Pregnancy: A Morphological and Endocrine Study. Amsterdam, Boek-en Offsetdrukkerij Los-Naarden, 1984 |
|
Kallenberger DA, Ronk DA, Jimerson GK: Ectopic pregnancy: A 15-year review of 160 cases. South Med J 71: 758, 1978 |
|
Romero R, Copel JA, Kadar N et al: Value of culdocentesis in the diagnosis of ectopic pregnancy. Obstet Gynecol 65: 519, 1985 |
|
Romero R, Taylor KW Jr, Kadar N et al: The diagnosis of ectopic pregnancy. In Steril WB, Cochrine WJ (eds): Clinics in Diagnostic Ultrasound: Gynecologic Ultrasound, p 123. New York, Churchill-Livingstone, 1984 |
|
Berry CM, Thompson JD, Hatcher R: The radioreceptor assay for hCG in ectopic pregnancy. Obstet Gynecol 54: 43, 1973 |
|
Romero R, Kadar N, Copel JA et al: The value of serial human chorionic gonadotropin testing as a diagnostic tool in ectopic pregnancy. Am J Obstet Gynecol 155: 392, 1986 |
|
Kadar N, Taylor KW Jr, Rosenfield A et al: The sonographic appearance of the uterus in ectopic pregnancy. Am J Roentgenol 141: 609, 1980 |
|
Mueller CE: Intrauterine pseudogestational sac in ectopic pregnancy. J Clin Ultrasound 7: 133, 1979 |
|
Spirt BA, O'Hare KR, Gordon L: Pseudogestational sac in ectopic pregnancy: Sonographic and pathologic correlation. J Clin Ultrasound 9: 338, 1981 |
|
Weiner CP: The pseudogestational sac in ectopic pregnancy. Am J Obstet Gynecol 1139: 959, 1981 |
|
Mantoni M, Pedersen JF: Massive pseudogestational sac in ectopic pregnancy. J Clin Ultrasound 11: 29, 1983 |
|
Abramovici H, Auslender R, Lewin A et al: Gestational pseudogestational sac: A new ultrasonic criterion for differential diagnosis. Am J Obstet Gynecol 145: 377, 1983 |
|
Marks WM, Filly RA, Callen PW et al: The decidual case of ectopic pregnancy: A confusing ultrasonographic appearance. Radiology 133: 451, 1979 |
|
O'Neill AGB, Hammond JG, Reid SE: Problems in the diagnosis of ectopic pregnancy: The pseudogestational sac. Aust NZ J Obstet Gynaecol 22: 94, 1982 |
|
Dunne MG: Shed decidual cast simulating an intrauterine fetus: Case reports. Am J Roentgenol 139: 591, 1982 |
|
Nyberg DA, Laing FC, Filly et al: Ultrasonographic differentiation of the gestation sac of early intrauterine pregnancy from the pseudogestational sac of early intrauterine pregnancy from the pseudogestational sac of ectopic pregnancy. Radiology 146: 755, 1983 |
|
Bradley EG, Fiske CE, Filly RA: The double ring sac sign of early intrauterine pregnancy: Use in exclusion of ectopic pregnancy. Radiology 143: 223, 1982 |
|
Kadar N, DeVore G, Romero R: The discriminatory hCG zone: Its use in sonographic evaluation for ectopic pregnancy. Obstet Gynecol 50: 156, 1980 |
|
Romero R, Kadar N, Jeaty P et al: The value of the discriminatory hCG zone in the diagnosis of ectopic pregnancy. Obstet Gynecol 66: 357, 1985 |
|
Romero R, Kadar N, Castro D et al: The value of adnexal sonographic findings in the diagnosis of ectopic pregnancy. Am J Obstet Gynecol 158: 52, 1988 |
|
Cacciatore B, Stenman UH, Ylostalo P: Comparison of abdominal and vaginal sonography in suspected ectopic pregnancy. Obstet Gynecol 73: 770, 1989 |
|
Bernaschek G, Rudelstorfer R, Csaicsich P: Vaginal sonography versus serum human chorionic gonadotrophin in early detection of pregnancy. Am J Obstet Gynecol 158: 608, 1988 |
|
Fossum GT, Davajan V, Kletzky OA: Early detection of pregnancy with transvaginal ultrasound. Fertil Steril 49: 788, 1988 |
|
Kadar N, Caldwell B, Romero R: A method of screening for ectopic pregnancy and its indications. Obstet Gynecol 58: 162, 1980 |
|
Barnhart KT, Sammel MD, Rinaudo PF et al: Symptomatic patients with an early viable intrauterine pregnancy: HCG curves redefined. Obstet Gynecol. 2004;104(1):50-5 |
|
Silva C, Sammel MD, Zhou L et al: Human chorionic gonadotropin profile for women with ectopic pregnancy. Obstet Gynecol. 2006;107(3):605-10 |
|
Yeko TR, Gorrill MJ, Hughes LH et al: Timely diagnosis of early ectopic pregnancy using a single blood progesterone measurement. Fertil Steril 48: 1048, 1987 |
|
Matthew CP, Coulson PB, Wild RA: Serum progesterone levels as an aid in the diagnosis of ectopic pregnancy. Obstet Gynecol 68: 390, 1986 |
|
Lindahl B, Ahlgren M: Identification of chorion villi in abortion specimens. Obstet Gynecol. 1986;67(1):79-81 |
|
Ries A, Singson P, Bidus M et al: Use of the endometrial pipelle in the diagnosis of early abnormal gestations. Fertil Steril. 2000;74(3):593-5 |
|
Barnhart KT, Gracia CR, Reindl B et al: Usefulness of pipelle endometrial biopsy in the diagnosis of women at risk forectopic pregnancy. Am J Obstet Gynecol. 2003;188(4):906-9. |
|
Mukul LV, Teal SB: Current management of ectopic pregnancy. Obstet Gynecol Clin North Am. 2007;34(3):403-19, x |
|
Mitchell DE, McSwain HF, McCarthy JA et al: Hysterosalpingographic evaluation of tubal patency after ectopic pregnancy. Am J Obstet Gynecol 157: 618, 1987 |
|
Bruhat MA, Manhes H, Mage G et al: Treatment of ectopic pregnancy by means of laparoscopy. Fertil Steril 33: 411, 1980 |
|
Shapiro HI, Adler DLH: Excision of an ectopic pregnancy through the laparoscope. Am J Obstet Gynecol 117: 290, 1973 |
|
DeCherney AH, Romero R, Naftolin F: Surgical management of unruptured ectopic pregnancy. Fertil Steril 35: 21, 1981 |
|
Pouly JL, Mahnes H, Mage G et al: Conservative laparoscopic treatment of 321 ectopic pregnancies. Fertil Steril 46: 1093, 1986 |
|
Brumsted J, Kessler C, Cison C et al: A comparison of laparoscopy and laparotomy for the treatment of ectopic pregnancy. Obstet Gynecol 71: 889, 1988 |
|
Lipscomb GH, Givens VM, Meyer NL et al: Comparison of multidose and single-dose methotrexate protocols for the treatmentof ectopic pregnancy. Am J Obstet Gynecol 2005;192(6):1844-7; discussion 1847-8. |
|
Jeffcoate TNA: Salpingectomy or salpingo-oophorectomy. Br J Obstet Gynaecol 135: 74, 1955 |
|
Hallatt JG: Repeat ectopic pregnancy: A study of 123 consecutive cases. Am J Obstet Gynecol 122: 520, 1975 |
|
Schinfeld JS, Reedy G: Mesosalpingeal vessel ligation for conservative treatment of ectopic pregnancy. J Reprod Med 28: 823, 1983 |
|
Sherman D, Langer R, Sadovsky G: Improved fertility following ectopic pregnancy. Fertil Steril 37: 497, 1982 |
|
Timonen S, Nieminen U: Tubal pregnancy, choice of operative method of treatment. Acta Obstet Gynecol Scand 46: 327, 1967 |
|
DeCherney AH, Kase N: The conservative surgical management of unruptured ectopic pregnancy. Obstet Gynecol 54: 451, 1979 |
|
Langer R, Bukovsky I, Herman A et al: Conservative surgery for tubal pregnancy. Fertil Steril 38: 427, 1982 |
|
DeCherney AH, Polan ML, Kort H et al: Microsurgical technique in the management of tubal ectopic pregnancy. Fertil Steril 34: 324, 1980 |
|
Yao M, Tulandi T: Current status of surgical and nonsurgical management of ectopic pregnancy. Fertil Steril. 1997;67(3):421-33 |
|
Hajenius PJ, Mol F, Mol BW et al: Interventions for tubal ectopic pregnancy. Cochrane Database Syst Rev 2007 Jan 24;(1):CD000324. |
|
Kalchman CG, Meltzer RM: Interstitial pregnancy following homolateral salpingectomy. Am J Obstet Gynecol 96: 1139, 1966 |
|
Stromme WB: Salpingotomy for tubal pregnancy. Obstet Gynecol 1: 472, 1953 |
|
Semm K: Operative Manual for Endoscopic Abdominal Surgery, p 192. Friedrich ER (trans). Chicago, Year Book, 1982 |
|
Oelsner G, Rabinovitch O, Movad J et al: Reproductive outcome after microsurgical treatment of tubal pregnancy in women with a single fallopian tube. J Reprod Med 31: 483, 1986 |
|
Richards BC: Persistent trophoblast following conservative operations for ectopic pregnancy. Am J Obstet Gynecol 150: 100, 1984 |
|
Kelly RW, Martin SA, Strickler RC: Delayed hemorrhage in conservative surgery for ectopic pregnancy. Am J Obstet Gynecol 133: 225, 1979 |
|
Johnson TRB, Sanborn JR, Wagner KS et al: Gonadotrophin surveillance following conservative surgery for ectopic pregnancy. Fertil Steril 33: 207, 1980 |
|
Israel SL: Total linear salpingostomy: Clinical and experimental observations. Fertil Steril 2: 505, 1951 |
|
Kamrava MM, Taymor ML, Berger MJ et al: Disappearance of human chorionic gonadotrophin following removal of ectopic pregnancy. Obstet Gynecol 62: 484, 1983 |
|
Senterman M, Jibodh R, Tulandi T: Histopathologic study of ampullary and isthmic tubal ectopic pregnancy. Am J Obstet Gynecol 159: 939, 1988 |
|
DeCherney AH, Naftolin F, Graebe R: Isthmic ectopic pregnancy: Segmental resection and anastomosis to conserve fertility. Fertil Steril 41: 458, 1984 |
|
Stangel JJ, Reyniak JV, Stone ML: Conservative surgical management of tubal pregnancy. Obstet Gynecol 48: 241, 1976 |
|
Cartwright PS, Entman SS: Repeat ipsilateral tubal pregnancy following partial salpingectomy: A case report. Fertil Steril 42: 647, 1984 |
|
Smith HO, Toledo AA, Thompson JD: Conservative surgical management of isthmic ectopic pregnancies. Am J Obstet Gynecol 157: 604, 1987 |
|
DeCherney AH, Boyers SP: Isthmic ectopic pregnancy: Segmental resection as the treatment of choice. Fertil Steril 44: 307, 1985 |
|
Grimes DA: Rh immunoglobulin utilization after ectopic pregnancy. Am J Obstet Gynecol 140: 246, 1981 |
|
Lund J: Early ectopic pregnancy. Obstet Gynaecol J Br Empire 62: 70, 1955 |
|
Maymon R: The new gynecological epidemic disease: Review of the modern work up of ectopic pregnancy and the non surgical treatment option. Int J Fertil 37 (3): 146, 1992 |
|
Tanaka T, Hayashi H, Kutsuzawa T et al: Treatment of interstitial ectopic pregnancy with methotrexate: Report of a successful case. Fertil Steril 37: 851, 1982 |
|
Stovall T: Single dose methotrexate: An expanded clinical trial. Am J Obstet Gynecol 168: 6, 1993 |
|
Yeko T: A prospective series of unruptured ectopic pregnancies treated by tubal injection of hyperosmolar glucose. Obstet Gynecol 85: 2, 1995 |
|
Sauer MV, Gorrill MJ, Rodi IA et al: Nonsurgical management of unruptured ectopic pregnancy: An extended clinical trial. Fertil Steril 48: 752, 1987 |
|
Rodi IA, Sauer MV, Gorrill MJ et al: The medical treatment of unruptured ectopic pregnancy with methotrexate and citrovorum rescue: Preliminary experience. Fertil Steril 46: 811, 1986 |
|
Ichinoe K, Wake N, Shinkai N: Nonsurgical therapy to preserve oviduct function in patients with tubal pregnancies. Am J Obstet Gynecol 156: 484, 1987 |
|
Lipscomb GH, Meyer NL, Flynn DE et al: Oral methotrexate for treatment of ectopic pregnancy. Am J Obstet Gynecol. 2002;186(6):1192-5 |
|
Frishman G, Seifer D: Hematosalpinx after methotrexate treatment of unruptured ectopic pregnancy. Fertil Steril 60: 3, 1993 |
|
Abbot J: Ruptured ectopic pregnancy after medical management: Current management strategies. Am J Emerg Med 11: 5, 1993 |
|
Creinin M, Washington A: Cost of ectopic treatment: Surgery vs. methotrexate. Fertil Steril 60: 6, 1993 |
|
Lipscomb GH, Bran D, McCord ML et al: Analysis of three hundred fifteen ectopic pregnancies treated with single-dosemethotrexate. Am J Obstet Gynecol. 1998;178(6):1354-8. |
|
Guven ES, Dilbaz S, Dilbaz B et al: Comparison of the effect of single-dose and multiple-dose methotrexate therapy ontubal patency. Fertil Steril. 2007;88(5):1288-92. Epub 2007 Apr 6 |
|
Bower M, Rustin GJ, Newlands ES et al: Chemotherapy for gestational trophoblastic tumours hastens menopause by 3 years. Eur J Cancer. 1998;34(8):1204-7 |
|
Agostini A, Blanc K, Ronda I et al: Prognostic value of human chorionic gonadotropin changes after methotrexateinjection for ectopic pregnancy. Fertil Steril. 2007;88(2):504-6. Epub 2007 Apr 6 |
|
: ACOG practice bulletin. Medical management of tubal pregnancy. Number 3, DecemberCollege of Obstetricians and Gynecologists. Int J Gynaecol Obstet. 1999;65(1):97-103 |
|
DeCherney AH, Oelsner G: Ectopic pregnancy. In Garcia C-R, Mikuta JJ, Rosenblum NG (eds): Current Therapy in Surgical Gynecology. Toronto, BC Decker, 1986 |
|
DeCherney AH, Silidker JS, Mezer HC et al: Reproductive outcome following two ectopic pregnancies. Fertil Steril 43: 82, 1985 |
|
Tulandi T: Reproductive performance of women after two tubal ectopic pregnancies. Fertil Steril 50: 164, 1988 |
|
Buster JE, Krotz S: Reproductive performance after ectopic pregnancy. Semin Reprod Med 2007;25(2):131-3 |
|
Breen JL: A 21 year survey of 654 ectopic pregnancies. Am J Obstet Gynecol 106: 1004, 1970 |
|
DeCherney AH, Romero R, Polan ML: Ultrasound in reproductive endocrinology. Fertil Steril 37: 323, 1982 |