Psychological and Sexual Aspects of Gynecologic Cancer
Authors
INTRODUCTION
Cancer is a major health problem that accounts for 23% of all deaths. Although death rates from heart disease, stroke, and other conditions have been decreasing, deaths resulting from cancer have risen nearly twofold since the 1970s.1 In contrast, the survival picture is more positive for women with gynecologic cancers, which account for 11% of all new cancer cases in women. Since the 1970s, death rates for gynecologic cancers have significantly declined: a reduction of 21% for endometrial cancer, 55% for cervical, and 8% for ovarian.2 Of the approximately 80,000 new gynecologic cases each year, more women are living and necessarily coping with stressors related to the cancer that may become chronic.
Research on the psychological and behavioral aspects of cancer began in the early 1950s; however, significant expansion has occurred since the 1980s. This research has clarified biobehavioral factors in illness, including relations between psychological responses and factors (e.g., personality, mood, coping style) and behavioral variables (e.g., compliance with treatment, diet, exercise), with more recent research incorporating biologic systems (e.g., immune and endocrine) and examining the interaction of these variables and their relationship to disease course.3, 4, 5 This chapter provides a brief overview of the central findings that have emerged regarding the psychological and behavioral aspects of gynecologic cancer. First, the psychological and behavioral outcome literature is reviewed by disease-relevant time points, from prevention to recovery or death. Next, common difficulties for women after treatment regarding sexuality are addressed. Finally, we discuss the role for preventive or rehabilitative interventions to prevent psychological or behavioral morbidity.
PREVENTION
Primary prevention
It has been suggested that much of cancer incidence and premature death can be prevented through changes in behavior. The US Department of Health and Human Services set a goal of a 21% reduction in cancer mortality by 2010 through prevention and control efforts focused, in large part, on lifestyle and genetic factors.6 Primary prevention attempts to reduce the probability of cancer onset by decreasing risk. For women and men, it is estimated that as much as 50% or more of cancer can be prevented through smoking cessation and improved dietary habits, such as reducing fat consumption and increasing fruit and vegetable consumption, physical activity, and weight control7, 8, 9, 10 and these have, therefore been a major focus of prevention efforts.
The isolation of the BRCA1/2 genes has enabled high-risk members of families with hereditary breast/ovarian cancer to learn whether they carry a cancer-predisposing mutation. Women found to carry a mutation in BRCA1 have a 40–65% lifetime risk of ovarian cancer, and an increased risk of colon cancer.11, 12 Those carrying a BRCA2 mutation have a lifetime risk of 11–23% for ovarian cancer13 compared to a risk of 1.7% in the general populaiton.1 Early identification of BRCA1/2 mutation carriers within high-risk families provides the opportunity for targeted surveillance and management strategies. Despite this opportunity, only a subset of high-risk family members are likely to request testing when it is made available, and individuals who do pursue testing tend to be of higher socioeconomic status or have had more relatives affected.14
Although some data suggest that the testing process and subsequent knowledge of carrier status produce significant distress and anxiety,15 other data are not supportive.14, 16 Protocols are available to evaluate the behavioral and psychosocial effects of genetic testing for cancer.17 Closer examination of the literature suggests that upon receipt of genetic results, BRCA1/2 carriers report greater cancer-specific distress than non-carriers, resulting from a reduction in distress in non-carriers versus an increase (or no change) in carriers. Moreover, a subgroup of women within BRCA1/2 carriers appear to be vulnerable to cancer-related distress. In the only study reporting extended follow-up at 5 years after genetic testing18 neither carriers nor non-carriers displayed clinically elevated levels of distress at 5 years. However, 44% of carriers consulted a mental health professional within 5 years after testing, with 50% of those consultations related to their cancer risk. In contrast, 33% of non-carriers consulted with a mental health professional, with only 29% of those consultations related to cancer risk. These results suggest that testing positive for a BRCA1/2 mutation carries long-term psychological significance. Increased gynecologic screening and prophylactic salpingo-oopherectomy are options available to the patient carrying a BCRA1/2 mutation. Overall quality of life in patients electing either option appears comparable to that of the general population, though patients who elect surgery experience a reduction in cancer-related worry and improved outlook related to cancer risk.19 In considering preventive options for patients, this psychological benefit must be balanced against the unfavorable endocrine and sexual sequelae of surgery. Protocols are available to evaluate the behavioral and psychosocial effects of genetic testing for cancer.17
More recently, vaccination has become an option for the primary prevention of cervical cancer. In 2006, the US Food and Drug Administration approved the first human papillomavirus (HPV) vaccine. A second vaccine was also approved for use in Australia and the European Union in 2007. Both vaccines cover the two HPV types (16 and 18) that cause an estimated 70% of cervical cancers worldwide.20 Thus, widespread vaccination has the potential to reduce cervical cancer deaths around the world by as much as two-thirds, as well as to reduce the significant medical costs associated with abnormal Pap tests (e.g., biopsies and invasive procedures).21 The CDC’s Advisory Committee on Immunization Practices recommends routine vaccination for females aged 11–12 years and catch-up vaccination for females aged 13–26 years.22 In one systematic review of 28 studies, most parents reacted positively to the possibility of vaccinating their daughters against HPV, with acceptability higher when people believed the vaccine was effective, a physician recommended it, and HPV infection was likely.23 A number of barriers to vaccination acceptance among parents have also been identified, including lack of knowledge among the US population about HPV transmission and its relation to cervical cancer, parental concerns about vaccines in general, concerns about promoting sexual behavior among adolescents, cost of vaccination programs, and challenges related to adolescents' access to healthcare and insurance coverage.24
Secondary and tertiary prevention
Secondary prevention efforts are those that identify disease at the earliest stages (e.g., when it is preinvasive or localized and asymptomatic) so that effective treatment can be administered sooner and mortality can be reduced. The chances of successful early cancer detection and treatment depend on the clinical characteristics of the disease and the screening strategy, and it is unlikely that secondary prevention can proceed effectively unless certain conditions for both are met. Since its introduction, the Papanicolaou (Pap) test has enabled earlier identification of cervical disease, but there are no comparable early detection methods for endometrial25 or ovarian cancers.26
There have been efforts in the area of tertiary prevention as well, shortening delay to seeking a diagnosis when awareness of symptoms and signs has occurred. In the study of delay in seeking treatment for medical conditions, several definitions of delay have been used, and one is the number of days from the detection of the first symptom to an end point. This variable is less consistently chosen but has included seeing a physician for symptoms, being diagnosed with a medical condition, or beginning treatment for the condition. According to this view, all individuals, including some physicians, would be “delayers” because to some such a delay is considered more “reasonable” than others (e.g., 1 month from symptom appearance to appearance before a physician; see early work in cancer by Pack and Gallo27). Research has shown that the median delays seen among gynecologic cancer patients tend toward longer increments (3 months from symptom onset to treatment for endometrial cancer;25 6 months from symptom onset to presentation for vulvar cancer28). Although correlates of delay can be studied, we have found that delay surrounding symptom interpretation (i.e., the time it takes for a woman to perceive and interpret her signs and symptoms as needing medical evaluation) accounts for most of the delay in seeking a cancer diagnosis.29 For women with gynecologic cancer, the time accounts for approximately 80% of the total delay.
There are likely several reasons for this time of symptom interpretation accounting for the bulk of the delay time. First, the development of malignancy and the appearance of cancer symptoms are often protracted, and a complex and changing symptom picture can be typical, in contrast to the presentation of many other medical problems (e.g., myocardial infarction). Symptoms also can vary with the site and extent of the disease. Ovarian cancer has varied presentations, including pelvic cramping, low back pain, pain or bleeding with intercourse, and urinary frequency irregularities. As cancers progress, they can involve other bodily systems (e.g., gastrointestinal), and the symptom picture tends to change from specific or localized complaints (e.g., vaginal discharge or bleeding) to diffuse ones (e.g., loss of appetite, nausea, cramping). Finally, cancer is a life-threatening, but low-probability disease, and these aspects may foster distress and delay.
RESPONSES TO DIAGNOSIS
An early clinical study suggested that the diagnosis of cancer produces an “existential plight”, meaning that the news brings shock, disbelief, and emotional turmoil.30 For the individual, the manner in which the information is disclosed is important. Physicians who communicate hope have patients who are in turn more hopeful and have a more favorable emotional adjustment; alternatively, communicating “false hope” is unwelcome.31 In a study of gynecologic cancer patients, Roberts and colleagues32 found that although patients expect compassion from their physicians, 89% prefer “straight talk” about their prognosis. This preference points to the need for physicians and other health care professionals to learn more effective and thorough means of communicating information (e.g., diagnosis, treatment, prognosis) to their patients.33, 34
An empirical demonstration of the importance of physician communication was provided by Rutter and associates.35 A time series design was used to test physician communication training as a strategy to reduce patient distress. Patients were treated by physicians before or after the communication training. Training lasting 1 hour and 15 minutes and a handbook on improving the structure and style of patient interactions were provided to the physicians. Based on the work of Ley,36 training included cognitive aids to understanding (e.g., simplification, repetition) and emotional aspects (e.g., conveying warmth, listening, giving feedback). Trained physicians also provided patient information booklets describing adjuvant treatments to encourage patient participation and perceived control. Accrual rate was 80%, and 36 patients participated; most had solid tumors, with 64% having advanced disease. All patients were assessed before and after their physician consultation for adjuvant therapy. A consecutive series of 18 control patients were seen by three physicians before their training. When physician training was completed, and data were gathered from the next 18 patients of the same physicians. A mixed model analysis of variance revealed a significant effect for time, with all patients reporting reductions in anxiety (State-Trait Anxiety Inventory).37 Analyses revealed a significant interaction, with patients seen after physician training reporting fewer depressive symptoms38 and higher levels of satisfaction and personal control. On experimenter measures, the physicians were evaluated as more cognitively skilled after their training than before.
Empirical reports clarify the specific emotions – sadness (depression), fear (anxiety), and anger – that characterize this period. It is not surprising that depression is the most common affective problem. In a review of studies using DSM-III criteria, 23% of gynecologic cancer patients meet the criteria for major depressive disorder, much higher than the rate of 5–6% in the general population.39 When major depression and depressed mood are considered, prevalence rates vary but can be higher (e.g., 21% in Bodurka-Bevers and colleagues,40 16% in Derogatis and colleagues,41 17% in Kornblith and colleagues,42 and 25% in a review by Massie and Holland43). Lower rates have been found when patients are ambulatory with good physical functioning rather than in the midst of or recovering from treatments.42, 43, 44 In a study of Latina cervical cancer patients undergoing radiotherapy,45 depressive symptoms were predicted by lower levels of support from the family, stress, physical symptoms from the treatment, and practical barriers to receiving treatment (e.g., transportation difficulties). In general, depression is more common for patients with advanced rather than early-stage disease, with pain or other disturbing symptoms rather than not,42, 46, 47 in active treatment rather than on follow-up, receiving palliative rather than curative treatment, and with a history of affective disorder or substance abuse.
An interesting clinical report provided detail on the specific emotions and concerns of patients. Steginga and Dunn48 interviewed 82 Australian women, at least 60% of whom were less than 18 months’ postdiagnosis. Diagnoses included cervical (45%), endometrial (30%), ovarian (20%), and vulvar (4%) cancers. At the time of diagnosis and treatment, greater than 80% of the women described depression (49%), anxiety (37%), and fear of dying (35%) being significant and problematic. The problematic symptoms during treatment included fatigue (14%), pain (11%), and bladder dysfunction (9%). All of these problems persisted for some women for 2 years after diagnosis, with rates of 13%, 15%, and 23% for depression, anxiety, and fear of dying and rates of 10–15% for pain, fatigue, and bladder and vaginal infections. The data on the chronic worries of recurrence are consistent with other clinical reports suggesting these are the same reasons women voice for wanting to continue their medical follow-ups.49
Psychological variables seem to play a role in the magnitude of emotional distress. Individuals who perceive their illness as severe50 or who have a sense of pessimism about life51 tend to have greater mood disturbance. Carver and associates51 found that general pessimism (versus optimism) at diagnosis predicted poorer well-being (mood and life satisfaction) at 1 day before surgery and at 3-, 6-, and 12-month follow-ups (see Miller and coworkers52 for similar results at 2- and 4-month follow-ups). Alternatively, when patients use positive ways to cope, such as seeking social support and assistance from friends and family, they have less anxiety and depression; when they use avoidance, their moods worsen.53
The clinical problem of diagnosis and treatment-related distress can be alleviated through psychological interventions.54, 55 McQuellon and colleagues56 developed a brief oncology clinic orientation program in the hopes of reducing anxiety, distress, and uncertainty in newly diagnosed cancer patients. The standardized program was designed to minimize the appraisal of threat in the new and unfamiliar setting. It lasted approximately 15–20 minutes and comprised a tour of the oncology clinic, a verbal description of clinic administrative procedures, a handout including important phone numbers and maps, and a brief question-and-answer session (patients were encouraged to address all medical questions to the medical staff). A total of 150 subjects who attended the clinic for their initial consultation were recruited. They were randomized into the treatment and control groups (control patients completed the two assessment batteries but did not receive the orientation). At the initial assessment, there were no significant differences between the two groups on any of the sociodemographic or psychological variables assessed. When followed-up 1 week after their initial visit, anxiety and mood disturbance decreased significantly for subjects who received the intervention, whereas mood disturbance increased for subjects in the control condition. More recently, Powell et al. 57 randomized new patients in a gynecologic cancer clinic to either a single-visit individual psychosocial intervention or an assessment-only control. Those who received the intervention reported greater decreases in anxiety, depression, and overall distress over the three month follow-up period; anxiety and overall distress decreased for controls as well, but depression increased. Improvements such as these in mood and coping are more impressive because they often are achieved with brief, cost-effective interventions (e.g., 10 therapy hours with delivery in a group format).54
RESPONSES TO TREATMENT
A component of the emotional distress occurring at diagnosis is due to the anticipation of treatment. Current therapies include surgery, radiotherapy and radioactive substances, chemotherapy, hormonal therapy, immunotherapy, and combination regimens and procedures (e.g., intraoperative radiotherapy). Some patients also undergo difficult diagnostic or treatment monitoring procedures, and all treatments are preceded or followed by physical examinations, tumor surveys, and laboratory studies. The diagnostic process of selecting the appropriate therapy and the subsequent treatment events can represent multiple occasions of medical stressors. As discussed subsequently, the data are consistent in their portrayal of more distress (particularly fear and anxiety), slower rates of emotional recovery, and perhaps higher rates of other behavioral difficulties (e.g., food aversions, continued fatigue, and malaise) than are found with individuals undergoing medical treatment for illnesses other than cancer. As cancer treatments vary considerably in their intent, morbidity, and mortality, we review each of the major modalities separately and discuss clinical problems that are common across therapies.
Surgery
There have been few investigations of cancer surgery, but there are numerous descriptive and intervention studies of the reactions of healthy individuals undergoing surgery for benign conditions. The latter studies are consistent in their portrayal of (1) high levels of self-reported preoperative anxiety predictive of lowered postoperative anxiety and (2) postoperative anxiety predictive of recovery (e.g., time out of bed, pain reports). Cancer surgery patients may be distinguished by higher overall levels of distress and slower rates of emotional recovery. Gottesman and Lewis58 found greater and more lasting feelings of crisis and helplessness among cancer patients compared with benign surgery patients for 2 months after discharge.
As noted earlier, considerable research has been done on the psychological and behavioral aspects of response to surgery, and many effective interventions have been tested. Components of these interventions include procedural information (i.e., how the surgery is to be performed and preoperative and postoperative events from the perspective of the patient), sensory information on the actual physical sensations of the surgery or preparatory events, behavioral coping instructions, cognitive coping interventions, relaxation, hypnosis, and emotion-focused interventions. In a meta-analysis of the literature, Johnston and Vogele59 reported that procedural information and behavioral instructions show consistent and strong positive effects on postoperative recovery. Effects are significant for a broad band of measures, including ratings of negative affect and pain, amount of pain medication, length of stay, behavioral recovery, and physiologic indices.
Radiotherapy
Radiotherapy is an important treatment for gynecologic cancer patients, particularly patients with cervical or endometrial disease. Noncompliance with radiotherapy includes treatment refusal, premature terminations, or receipt of fewer radiation sessions than prescribed. These circumstances occur for all cancer patients,60 although patients with low income or low educational levels may be at greater risk. The regimen of multiple weeks of daily treatments can pose a significant burden for patients employed, with children, or with transportation difficulties.61
To understand radiation fears, the surgical anxiety studies described previously have been a paradigm. Anxiety (and sleeplessness) often can cause more overall distress than physical symptoms62 and are predictors of treatment response.63 If interventions to reduce distress (especially anticipatory anxiety) are not conducted, heightened post-treatment anxiety is also found64, 65 and might be maintained for 3 months after therapy, particularly when treatment symptoms linger (e.g., decreased appetite; cramping; vaginal dryness, discharge, and itching;66 bladder or bowel dysfunction; fatigue67). When acute side effects resolve (usually by 12 months post-treatment), there seems to be no higher incidence of emotional difficulties for radiotherapy patients than for cancer surgery patients.68 Distress remains, however, if local symptoms, such as frequent voiding and diarrhea and vaginal changes (e.g., shortening, dryness), become chronic.67 The changes to the vagina occur early (e.g., shortening after a single intravaginal treatment69) and only increase with time. Leon-Pizarro and colleagues 70 have demonstrated that brief training in relaxation and guided imagery techniques can significantly reduce anxiety, depression, and overall discomfort in patients undergoing brachytherapy in Spain. All participants received one 45-50 minute introductory session prior to hospitalization, intervention participants also received just 10 minutes of relaxation training and a cassette tape to practice at home prior to receipt of radiotherapy.
Chemotherapy
The routine use of antiemetic drugs and the development of new chemotherapy agents have resulted in a lower incidence and severity of nausea and vomiting as a clinical problem. This change in clinical practice may have been offset, however, by the use of more toxic regimens and the adjuvant treatment for disease types or stages that previously were not treated with chemotherapy.71 In general, antiemetics need to be used from the beginning not only to control nausea and vomiting, but also to reduce the likelihood of the development of anticipatory reactions. When anticipatory nausea and vomiting develops in response to treatment, antiemetics are less effective,72 and this points to the need for the use of behavioral strategies to aid patients.71 Research has focused on eliminating or reducing these problems through a variety of techniques, including hypnosis, progressive muscle relaxation with guided imagery, systematic desensitization, cognitive distraction, and biofeedback.73
A common problem reported by most patients receiving radiotherapy or chemotherapy is fatigue.74, 75 Fatigue, described by patients as tiredness, lack of energy, sleepiness, confusion, and poor concentration, has been related to cancer morbidity and poor treatment compliance. Until more recently, little research had investigated variables associated with fatigue; however, a few studies conducted in the mid-1990s showed a consistent relationship between fatigue and the availability of physical, emotional, and social resources;76 severity of anxious and depressive symptoms;77 emotional distress; muscle weakness; numbness; sleep disturbances; and difficulty concentrating.78 Psychological and behavioral interventions have focused on alleviating or increasing tolerance to fatigue through preparatory information on side effects and activity and rest cycle recommendations (e.g., naps in the afternoon)74, 75 and other activity-planning coping efforts (e.g., planning and scheduling activities, decreasing nonessential activities, and relying on others for assistance as needed).79
Psychological interventions have focused on a variety of methods to improve patient adjustment. The novel physician communication training intervention of Rutter and coworkers35 discussed earlier alone and with efforts to provide additional information (e.g., orientation to a chemotherapy clinic56) are cost-effective. Both intervention efforts produced positive, immediate outcomes on measures of anxiety and depression. These interventions and relaxation therapy alone80 are the type of efforts that could be implemented widely to all cancer patients. With adequate training and supervision, peers or volunteers could be used successfully as the “therapists”. With a triage model, intensive (and expensive) efforts, such as group or individual interventions, could be provided selectively to moderate-risk to high-risk/high-distress patients. Most psychological interventions delivered to individual patients or to patient groups are multimodal and include components consisting of stress reduction (progressive muscle relaxation training), disease and treatment information, cognitive–behavioral coping strategies, and social support.54
PSYCHOLOGICAL OUTCOMES
Recovery from treatment and short-term adjustment
As patients recover and resume life patterns, there may be residual emotional distress and difficulties requiring continued coping. We discuss each of these circumstances and provide examples of emotional and physical challenges that may confront women during the year after their diagnosis. Lingering emotional distress from the trauma of diagnosis, treatment, and more generally life threat may occur for a small subset (perhaps 5–7%) of cancer patients.81 When pronounced, such distress has been likened to post-traumatic stress disorder. It is unlikely, however, that such extreme distress would occur for the “average” cancer patient; it is more likely for patients who have undergone the most difficult treatment regimens (e.g., bone marrow transplant); lengthy toxic chemotherapies, as might be given to stage III ovarian cancer patients; or life-altering or disfiguring cancer treatments (e.g., pelvic exenteration). Longitudinal studies provide a glimpse of the mood, social adjustment, employment, and marital adjustment outcomes. Andersen and colleagues82 followed women for 1 year after their diagnosis. Regarding mood, there were significant elevations of depression, anxiety, and confusion at the time of diagnosis; however, these effects stabilized within normal ranges during the post-treatment year. Similarly, there was no significant disruption of social relationships or activities during recovery, and women gradually resumed pretreatment levels. There was a high incidence of sexual and fertility disruption (see discussion later). This incidence in part has prompted concern regarding the possibility of marital disruption among gynecologic cancer patients. An early clinical study of women receiving radical mastectomy noted the realistic feelings of body disfigurement that the women and their spouses felt – prompting sexual retreat, emotional estrangement, and marital disruption.83 Despite the emotional distress and, for some, accompanying sexual disruption that couples experience, data from retrospective studies with comparison groups84 and from the controlled longitudinal studies indicate that marriages remain intact and satisfactory and occasionally stronger, as found in data from single assessment.85, 86, 87 More recent data indicate that when marital breakdown occurs, it is most likely among couples with pre-existing (i.e., precancer) difficulties.88
In addition to the cancer patient, stress may become significant for individuals closest to her, such as spouse or children.89 Problems facing the family members of a cancer patient include loneliness, isolation, and role overload. Family strain seems to be affected by illness variables (e.g., prognosis, stage or duration of illness, caregiving demands, patient’s distress), family variables (e.g., age and gender of family members, socioeconomic status, other family stressors), and relational variables (e.g., quality of marriage, marital communication, family stage, and social support).90 Young families, in which the wife/mother has cancer and young children are in the home, may be at heightened risk for relational difficulties.91 Ell and coworkers92 found that kin who were functioning poorly (e.g., lower perceived personal control, less adequate emotional support from close others, and greater stress unrelated to cancer) when the patient was diagnosed or who lost personal and social resources during the patient’s treatment and recovery tended to function poorly at follow-up. A subset of partners and family members seem to be at psychosocial risk.
Long-term adjustment
Models have been proposed to predict which patients are at risk for psychosocial morbidity.5 In a longitudinal study comparing cervical and endometrial cancer patients, predictors of risk at 6 and 12 months post-treatment included well-being before treatment (as would be expected), site of disease (with a cervix diagnosis conferring a greater risk), and the presence of children in the home.93 Studies of this sort are important in identifying the types of variables before treatment that could be used to identify women at risk. These data are consistent with those of Greimel and Friedl,94 who reported lower quality of life at 3 months post-treatment for cervical and ovarian cancer patients compared with endometrial patients. Chan and colleagues95 reported that lower education levels predict poorer outcomes for Asian women. Kornblith and colleagues42 reported that one third of ovarian cancer patients report significant psychological distress, and impaired physical functioning is an important correlate. Although global adjustment problems do not occur for most gynecologic cancer survivors, worry about the disease persists 96 as does more general distress for a subset of patients 97, 98, 99. An emerging literature has documented psychosocial factors that protect patients from adverse psychological outcomes; such factors include self-efficacy,100 social support,100 engaging in preventive health behaviors,101 and deriving meaning from the cancer experience.97
There are few data on the aspects of physical health that might influence quality of life for gynecologic cancer survivors, but the available data suggest three trends. First, side effects of treatment are common and persistent.102, 103 In fact, some patients rate the physical sequelae of treatment as the most significant challenge of survivorship.104 Second, comparisons between normative samples and gynecologic cancer survivors indicate few group differences in mood and quality of life. Still, a portion of patients, ranging from 20% to 63% across studies, have quality of life and persistent psychological maladjustment.103 Third, there appears to be a relationship between physical impairment and psychological maladjustment, particularly for adjuvant therapy patients.102, 105 Ovarian patients might be at greater risk for difficulty, due in part to aggressive treatment regimens.106 Thus, the primary difficulties in gynecologic cancer survivorship include physical impairments and psychological outcomes; however, physical health might pose a greater challenge. In addition, other data suggest that gynecologic cancer patients are at higher risk for psychological maladjustment than other cancer survivors. Parker et al.107 interviewed breast, gastrointestinal, gynecologic, and urologic cancer survivors (n = 351) and assessed depressive symptoms, anxiety symptoms, and psychological well-being. Depressive and anxiety symptoms were elevated for all survivors, but gynecologic survivors reported significantly higher anxiety scores than all other patients, and higher depression scores and lower well-being scores than gastrointestinal and urologic survivors.
Recurrence and death
Cancer recurrence is devastating and fear of recurrence represents a significant concern for survivors of gyencologic cancer (24% of survivors in one study99 reported clinically significant anxiety related to fears of recurrence). The magnitude of distress is greater than that found with the initial diagnosis108 and studies contrasting cancer patients showing no evidence of disease with patients receiving palliative treatment109 reported the greatest distress for patients with disseminated or persistent disease.110, 111 Acute fear also can be a problem. In a sample of gynecologic cancer patients faced with the prospect of end-of-life decisions, Roberts and colleagues32 found that 39% exhibited acute fears, the most common being fear of abandonment (32%) and fear of social isolation (17%). Difficult decisions (e.g., beginning a regimen that offers little chance for cure and has side effects versus no treatment) are made in a context of extreme emotional distress and physical debilitation. The few studies of psychological interventions for adult patients indicated that important emotional gains can be achieved during terminal stages112 and that children, adolescents, and adults can make independent decisions about the continuation of therapy when death is imminent.113
At this time of significant emotional turmoil and physical difficulty, psychological interventions seem to enhance the quality of life. One example is an investigation of a group support intervention for women with breast cancer conducted by Speigel and colleagues.114, 115 Women were randomized to no treatment or a group treatment intervention that included discussion of death and dying, family problems, communication problems with physicians, and living fully in the context of a terminal illness. The intervention subjects were randomized a second time to two conditions: no additional treatment or self-hypnosis for pain problems,115 which was incorporated into the support group format. All intervention groups met for weekly meetings for 1 year, for a total of 75 therapy hours. At the end of the first year, the groups formally ended, but members could continue to meet as they wished or were able; some groups lasted for an additional 2 years. A total of 86 women, 50 intervention and 36 no treatment control, with metastatic breast cancer and referred to the intervention from the oncologist, participated. After random assignment, there was subject loss (e.g., refusal, too weak, death) with the study beginning with 34 intervention and 24 control participants; however, the survival data are reported for the original sample of 86. Analyses indicated that the intervention group reported significantly fewer phobic responses and lower anxiety, fatigue, and confusion and higher vigor than the controls. These differences were evident at all assessments, but the magnitude increased from 4 to 12 months. There was also a significant decrease in the use of maladaptive coping responses by the intervention group. Regarding the findings from the hypnosis substudy, women receiving hypnosis within the group support intervention reported no change in their pain sensations during the year, whereas pain sensations significantly increased for the women in group support who did not receive hypnosis. Similar findings were reported for pain suffering – a slight decrease for the women who also received hypnosis and a significant increase in suffering for the remaining intervention women. Pain sensation scores for both groups were, however, significantly lower than those for the no intervention controls, suggesting that the hypnosis component provided an additive analgesic effect to other group treatment components. The most startling data from this project were reported in a survival analysis. A variety of follow-up analyses, controlling for initial disease stage, days of radiotherapy, or use of androgen or steroid treatments were conducted, and all indicated the same survival differences favoring the intervention participants. In contrast to other studies, interventions for terminal patients tend to be intensive and lengthy, such as “several sessions” or “until death”.
Finally, a frequent complication of disseminated disease is pain. Although it might also be one of the first symptoms of cancer or might be present when disease is only localized or spread regionally, pain is more common and less controllable for patients with metastatic disease.116 The major cause of cancer pain, accounting for roughly 70% of the cases, is due to direct tumor involvement (e.g., metastatic bone disease, nerve compression). Another 20–30% of cases are due to medical therapy (e.g., postoperative pain, radiation-induced pain). The remaining cases are individuals with pain problems unrelated to the cancer. The most difficult case is chronic pain associated with disease progression (e.g., patients with carcinoma of the pancreas, in which the pain escalates), in which combinations of antitumor therapy, anesthetic blocks, and behavioral approaches to pain control are considered.
The quality-of-life consequences of chronic pain are important. Rummans and colleagues117 reported that in a sample of recurrent breast and gynecologic cancer patients, 62% of patients (n = 114) stated that their pain interfered with their ability to function in some way. In the same study, the investigators reported that the frequency and amount of pain, in addition to its level of interference, correlated moderately with a variety of measures of overall physical and social functioning, with Spearman r ranging from 0.430 to 0.676. In a similar report, other researchers showed that outpatients with cancer-related pain (n = 200) reported significantly more mood disturbance than cancer patients who were pain-free (n = 169). They showed that, in addition to correlations with overall mood disturbance, the intensity and duration of pain modestly but significantly correlated with higher levels of depression, fatigue, confusion, and anger in cancer patients.118
SEXUAL FUNCTIONING OUTCOMES
When gynecologic cancer survivors are studied longitudinally, sexuality is the life area that undergoes major, or even permanent, change.119 There are several reliable and valid strategies to assess quality of life (e.g., SF-36, FACT)120 and sexuality,121 and research has documented the acceptability of such assessments in clinical trials.122 This discussion is divided into three areas: (1) review of sexual difficulties for gynecologic cancer survivors, (2) identification of medical and psychological variables related to morbidity risk, and (3) discussion of the merits of psychological interventions for cancer survivors.
In the literature on sexual functioning outcomes, reports often examined outcome as a function of treatment modality. Specifically, many studies provide data on surgery versus radiotherapy, usually in the treatment of cervical cancer. With one exception,123 randomized trials testing for sexual outcomes have not been conducted, even though it is likely that there are systematic differences between treatment modalities. Occasionally, ovary or endometrial cancer patients also may be included in the analysis. Although this inclusion adds to the variability in outcomes, the variations in the same modality seem to be less important in predicting sexual outcomes than the effects of different treatment modalities (e.g., surgery versus radiotherapy). For this reason, we review the retrospective data within treatment regimens, specifically surgery (e.g., radical hysterectomy), radiotherapy (external beam, with or without intracavitary treatment), and combination surgery and radiotherapy.119
Review of the literature: retrospective studies
RADICAL HYSTERECTOMY AND RELATED SURGERIES
Historically there have been conflicting reports on the sexually disruptive effects of hysterectomy per se. The vaginal shortening with radical hysterectomy may contribute to subjective feelings for the woman that the vagina is “too short” for intercourse. Nerve and vascular disruption to the pelvis (as may occur with lymphadenectomy) may result in loss of sensitivity and orgasmic disruption. The latter issues and other types of morbidity (e.g., bladder or bowel dysfunction) have led to the consideration of nerve-sparing techniques for radical hysterectomy.124
With few exceptions,125, 126 the data on radical hysterectomy and related surgeries come from small sample reports, which are more subject to threats of internal validity, including subject selection (i.e., biased sample), history (i.e., the effects of events before and after cancer and cancer treatment), and maturation (i.e., the effects of normal developmental events, such as menopause). Although large sample sizes do not solve these research design flaws (i.e., the data have the same problems with validity), they may provide more reliable (stable) estimates of sexual outcomes. Review suggests that approximately 20% of women report being sexually inactive at follow-up, and of the women remaining active, approximately 30% reported significant declines in the frequency of intercourse. The rates of dysfunctions ranged from 25% to 40% for the sexually active women, with dyspareunia in particular a problem for 30% of women.
RADIATION THERAPY
The effects of radiation on the vagina have been detailed.127 The rapid cell renewal system of the vagina’s epithelium provides for the natural exfoliation of the outermost layer of nondividing cells, which in turn makes the epithelium sensitive to the effects of radiation. The damage causes depletion of the cell supply, which is compensated by mucosal shrinkage. Significant vaginal shortening occurs.128 The slow occlusion of blood vessels and the gradual laying down of fibrosis in the submucosal tissues leads to tissue hypoxia and progressive narrowing of the vaginal canal with a significant decrease in elasticity and a decrease in sensation. The patients may have genital scarring and vaginal fibrosis. The thinning of the vaginal epithelium and lubrication loss increase susceptibility to trauma and infection. Pelvic stenosis and fibrosis cause decreased blood flow and vaginal secretions, which are coupled with decreases in vaginal length and diameter. It is estimated that radiation-induced tissue changes continue for 36 months after the completion of therapy.
Radiation to the pelvis can destroy ovarian functioning for the premenopausal woman, inducing menopausal symptoms and further adding to vaginal atrophy and lubrication difficulties. The exact dose required to cease ovulation permanently is unknown; however, the nearer to menopausal age is the woman, the lower is the dose required. Because current standard doses for gynecologic tumors consist of 4000–6000 cGy to the whole pelvis,129 excluding the dosage received with intracavitary treatments (see Andersen et al.130 for a discussion of the psychological aspects of this difficult treatment), cessation of ovulation is virtually certain for women of any age. Estrogen therapy after treatment can control menopausal symptoms such as hot flushes and aid in the healing of the vaginal epithelium,131 but dyspareunia still may occur. Estrogen replacement therapy has not been offered to many patients.
As with the surgery studies, several data are available. The percentages of women discontinuing sexual activity or reporting decreases in frequency are comparable to the surgery studies, with approximately 20% reporting no activity and 40% reporting significant decreases. The percentages of women reporting response cycle disruption are higher, however, in the range of 40–50%. Supporting descriptive data on pelvic examinations suggest the contribution of radiation effects on the vagina coupled with estrogen deficiency to the occurrence of sexual disruption.
COMBINED SURGERY AND RADIOTHERAPY
There are two common scenarios in receiving combination therapy. Radiotherapy can follow surgery if the pelvic lymph nodes are positive for malignancy. When radiotherapy precedes surgery, the apex of the vagina, which is exposed to the largest dose of radiation when intracavitary treatment is included, may be removed during surgery. If this is done, the remaining portion of the vagina is less affected and, perhaps, less vulnerable to problems of dyspareunia. In the retrospective studies, researchers do not always mention the sequence of the combined treatment, making interpretation difficult and comparison of sequences impossible.
It is believed that these women would have greater risk for sexual morbidity because of the combination therapy. With the exception of higher rates of dyspareunia (in the range of 35%), however, the outcomes appear similar to those for the single-modality groups. Summary analyses suggest that approximately 15% reported no sexual activity, whereas 40% reported declines in frequency, and 25–50% of women reported difficulties with desire, excitement, or orgasm.
RADICAL SURGERIES
Pelvic exenteration
This surgery is disfiguring and produces many functional problems with the obvious sexual sequelae.132 Clinical articles commonly have reported the cessation of sexual activity for most women (i.e., 80–90% of those surveyed). Available reports are from limited to Canada,133 the United Kingdom,134 the Netherlands,135 and the United States;136, 137, 138, 139, 140 all reveal a negative scenario for sexual outcomes and the majority suggest significant psychological distress, including depression and anxiety-related problems. For most women and couples, the prospect of ending their sexual life (as most couples cease all sexual activity when intercourse becomes impossible) is distressing and may be a source of continuing marital discord.136
Vaginal reconstruction is possible for some and enables a woman to maintain sexual activity that includes intercourse; however, many sexual difficulties often remain, and some physicians have described the outcomes as disappointing.141 Some women have difficulties with the physical characteristics of the new vagina (e.g., the cavity is too large or too narrow); others have general problems with arousal or orgasm or specific impediments such as dyspareunia or bleeding with intercourse. Regardless of whether or not women with pelvic exenteration undergo vaginal reconstruction, these women face the greatest disruption to their body and functioning of any female cancer group. It is remarkable that little systematic descriptive or intervention work has been done with these women in view of the curative intent of this surgery.
Surgeries for vulva cancer
Despite the sexual morbidity of vulva surgery, attention to the sexual or psychological outcomes for women is relatively recent, with the first substantive reports not appearing until 1983. As with pelvic exenteration, vulvar treatments can have a dramatic impact on sexuality and other life areas. After early observations on the benefits of conservative therapy142 and the advocacy of individualized treatment approaches,143 a paradigm shift in treatment occurred. In addition to less radical resection of the primary lesion, this shift has included selective omission of groin dissection and routine pelvic lymphadenectomy, the possibility of sentinel node biopsy rather than lymphadenectomy,144 preoperative radiation to obviate the need for exenteration in patients with advanced disease, and postoperative radiation to decrease the incidence of groin recurrence.145, 146 The data for in situ and invasive disease are reviewed separately because of the considerable difference in the treatment regimens. Also, on average, in situ patients are younger than patients with invasive disease.
Andersen and colleagues147 provided extensive data on the sexual outcomes for women treated with wide local excision and related treatments for in situ disease. In situ patients are more likely to be sexually inactive at follow-up, whether or not they have available sexual partners, than age-matched healthy counterparts. If the woman has a sexual relationship, however, the rates of sexual dysfunction are only slightly higher than those for healthy women.
The outcomes for women with in situ disease contrast markedly with the outcomes for women with invasive disease, many of whom are treated with radical vulvectomy, with or without groin dissection. The early retrospective studies, conducted in the 1980s, were limited by their small sample sizes (range 9–52) and retrospective evaluations, but the trends were consistent.136, 148, 149, 150 At least 30–50% of patients became sexually inactive, and of the women remaining active, 60–70% had multiple sexual dysfunctions. Research efforts with larger samples confirmed these findings and documented the high frequency of sexual dysfunctions, including ones that are difficult to treat (e.g., sexual aversion and hypoactive sexual disorder). Size of excision,151 advanced age,152, 151 depression 152and lower performance status 152 are correlates of risk for sexual dysfunction. Reasons women and their partners end intercourse include the physical changes to the body and severe dyspareunia, such as may occur with a narrowed introitus. Rather than being resigned to this circumstance, most women would have preferred to remain sexually active.136
Review of the literature: prospective studies
In the prospective designs, women have been recruited for study participation shortly after diagnosis and assessed longitudinally. These studies usually have detailed assessments of sexual functioning. Some studies also have included women treated for benign disease to estimate the magnitude of sexual problems with gynecologic disease and treatment, and healthy women to estimate the base rate of sexual problems. The latter designs are most powerful when longitudinal assessment is included for all groups.
SEXUAL DISRUPTION WITH THE INITIAL APPEARANCE OF SYMPTOMS AND SIGNS OF CANCER
The sexually disruptive effects of early signs and symptoms were described by Andersen and coworkers.153 In the United States, 41 women with early-stage cervical or endometrial cancer were studied before treatment, and their responses were compared with a matched group (i.e., age, menopausal status, and sexually active status) of healthy women in no gynecologic distress. Because 75% of the women with cancer experienced a substantial change in sexual functioning, it is likely that such obvious and disruptive sexual problems influenced the women to interpret their gynecologic disease signs and symptoms negatively and to seek medical consultation. These controlled data replicate an extension of the findings of Harris and colleagues,154 also in the United States, who interviewed 63 recently diagnosed women (stage of disease not specified). Although 85% of the women were satisfied with their sexual life before the appearance of symptoms, only 48% felt similarly afterward. Of the women, 50% stopped having intercourse, and another 30% reduced the frequency. Of women remaining sexually active, the percentage of women able to experience orgasm dropped from 58% to 15%. Finally, women reported that they initiated the changes in intercourse frequency because of bleeding, pain, and anxiety.
SINGLE-GROUP LONGITUDINAL DESIGNS
The only experiment ever to have been conducted to test for differential sexual outcomes was reported by Vincent and associates123 in 1975 in the United States. A total of 50 women with early-stage cervical disease were randomly assigned to receive either radical hysterectomy or radiotherapy. The groups were matched for age, education, socioeconomic status, marital status, parity, race, and disease stage. In contrast to the results from the retrospective studies, the changes in sexual desire and activity from pretreatment to 6 months post-treatment were comparable. Estimates of diminished desire were obtained from 24% of the radiotherapy patients and 20% of the surgical patients. Decreased frequency of intercourse was reported by 29% of the radiotherapy patients and 33% of the surgical patients. This experiment provides convincing evidence that, in general, the rates of sexual behavior disruption and dysfunction were comparable for the particular treatment options offered in 1975.
Schover and coworkers155 reported sexual outcomes for 61 women treated for stage Ia (8%), Ib (85%), or IIa (7%) cervical cancer. Of women, 26 received radical hysterectomy and 35 received radiotherapy; however, 19 of the latter also received surgery. There was a 20% refusal rate for study participation. Although data were gathered at 6 and 12 months after therapy, data were available only for 48% and 42% of subjects, respectively. The report does not indicate significant sexual disruption. Rates of loss of sexual desire are reported as 24% for the initial assessment and 25% at 6 months and 25% at 12 months. Similarly the rate of excitement problems at 12 months was 20% and 27% for orgasmic dysfunction, respectively. Despite these findings, the rates of sexual complaints were high. At 12 months, 21% noted that vaginal penetration was painful, 45% indicated deep thrusting was painful, and 24% reported postcoital vaginal pain. Also, 29% reported continued vaginal bleeding at 12 months. Sexual problems of this type have been reported to be significantly distressing and meriting diagnoses of sexual dysfunction in other studies.
Flay and Matthews156 studied 16 New Zealand women treated with surgery or radiotherapy for stages I through III cervical cancer. By 14 weeks after the end of radiotherapy, approximately two thirds of the women were moderately to extremely dissatisfied with the low frequency of sexual activity and their low levels of desire, arousal, and satisfaction. Women indicated the following problems as contributing to their difficulties: vaginal shortening (64%), dryness (43%), narrowing (43%), and bleeding (36%). Dyspareunia was reported by 43% of the sample.
Hansen157 in Denmark provided sexual outcome data for women treated with surgery, radiotherapy, or combined treatment for stages Ib or IIa cervical cancer. The outcome data are combined, and results are reported only for 78 women who had no sexual difficulties before therapy. Of this subset, 73% resumed intercourse and had no sexual complaints. Of the 27% who stopped intercourse, it seemed that 38% of them reported loss of desire, 17% had orgasmic dysfunction, 24% had dyspareunia, and 19% no longer had a sexual partner. The length of the time to follow-up was not reported.
The only report on psychological or sexual adjustment of African women comes from Adelusi,158 who prospectively studied 101 women treated for stage I through III cervical cancer at the University Hospital in Ibadan, Nigeria. The report is remarkable for its large sample; longitudinal assessment (pretreatment and 12–18 months’ post-treatment); and specific data on intercourse frequency, causes of difficulty, and marital and relationship status. Despite the modest statistical analyses, important findings indicated significant negative effects: 27% of the patients became separated or divorced after treatment, and 75% became sexually inactive. Reasons noted for sexual difficulty included dyspareunia, vaginal bleeding (36%), and fear of cancer or recurrence (18%). This magnitude of problems may be due to racial, cultural, or other related factors; however, this is difficult to discern because of the absence of other studies from Africa.
LONGITUDINAL STUDIES WITH COMPARISON GROUPS AND DATA
Andersen and colleagues82, 159 examined the nature and timing of sexual difficulties for women with early-stage disease and other quality-of-life outcomes. A total of 47 women with stage I or II cervical or endometrial disease were assessed before treatment and at 4, 8, and 12 months after treatment. Two matched comparison groups, women diagnosed and treated for benign disease (e.g., uterine fibroid treated with simple hysterectomy) and gynecologically healthy women, also were followed longitudinally. The former provided an estimate of sexual disruption resulting from disease in and treatment to the pelvis, and the latter estimated the base rate of sexual difficulties resulting from normal life circumstances.
Analyses indicated that the primary sexual behavior disrupted by the disease and treatment process for women with malignant or benign disease was the frequency of intercourse, declining from an average of 9.5 occasions per month to 6–7 occasions per month during the post-treatment period. The absence of change in other sexual behavior variables (e.g., range of current sexual activities) indicated that when couples engaged in intercourse, albeit less often, the women reported the same sexual activities (e.g., body caressing, oral–genital stimulation) as having occurred. There were no significant differences between groups in the percentage of women becoming sexually inactive, with the estimates ranging from 5% to 15% across the assessments. Half of the sexually inactive cases in the cancer group (i.e., two of four women) were due to disease-related causes, however. Considering the data on the sexual response cycle, difficulty with sexual excitement for both disease groups was substantial. A likely reason for the arousal deficits was the co-occurrence of significant disrupters (e.g., dyspareunia, resulting in part from radiation effects or induced menopause). By 12 months’ post-treatment, 30–40% of the sample had sexual dysfunctions, including inhibited desire, excitement, or orgasm and dyspareunia.
Weimar Schultz and associates160 studied women with stage Ia through IIIb cervical cancer. Comparison groups included ten women treated with simple hysterectomy for benign disease, 21 gynecologically healthy women, and 12 women treated 1 year previously for cervical cancer. The benign group was followed longitudinally; however, there was only one assessment for the healthy and previously treated cervical patients. Follow-up data were available only for 48% of the sample, resulting in 26 women. Analyses of 12-month post-treatment data indicated that there were no differences between the women with cancer and healthy women in the areas of current sexual activity or motivation for sexual activity; however, there were significant decrements in sexual arousal, disrupting genital sensations during intercourse, and increases in sexual dissatisfaction. However, this pattern of difficulties was similar to sexual disruptions reported by the women treated for benign disease. These outcomes for the women treated for disease are consistent with the data of Andersen and coworkers.159
A report by Kylstra and colleagues161 assessed a predominantly cervical cancer sample (73%; n = 58) with stage I or II disease before treatment and at 6 and 12 months after treatment. Most of the sample (76%) was treated with surgery alone. Neither sexual activity nor sexual satisfaction changed significantly from pretreatment to post-treatment. These positive outcomes may be accounted for by the circumstance of availability of sexuality information from the physician for virtually all (92%) of the women.
Predictors of sexual morbidity risk
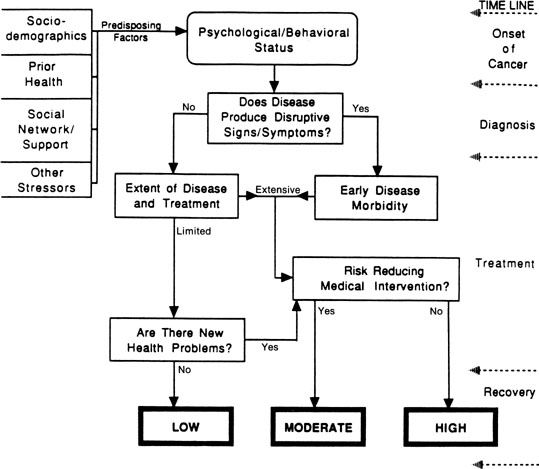
Across studies, we have been reminded of the significant role that medical factors, such as the extent of disease and magnitude of treatment, seem to play in determining sexual outcomes for women with gynecologic cancer. We have formulated a model of disease and treatment pathways that can yield differential levels of risk for sexual morbidity (Fig. 1). Variables are introduced according to a disease time line, from cancer appearance to the immediate post-treatment recovery period, as disease-relevant events provide a meaningful framework to understand psychological adjustment to cancer.162 Turning to the model, at the onset of cancer, we consider the occurrence of disruptive signs and symptoms. When the disease is detected and produces sexual disruption, this is the first point of psychological and behavioral morbidity. This early change is included because of its role in increasing a woman’s emotional distress and alerting her to the potential for subsequent life changes.
The model indicates that the extent of disease and treatment are major determinants of risk. As indicated, the disease and treatment status are summarized into routes of limited versus extensive; these alternatives should have supporting medical end point data and stage and magnitude of treatment information. The limited/extensive distinction is based in part on the classification of disease stage – localized versus regional or metastatic – and its direct relationship to survival in gynecologic cancer (i.e., across sites, 5-year survival rates are 89%, 54%, and 20% for localized, regional, and distant gynecologic disease).163 The model goes one step further and suggests that the extent of disease be considered in the context of the magnitude of treatment that is received. Women with localized or regional vulvar cancer often receive the same disfiguring surgical treatment – radical vulvectomy – and might differ only in the provision of radiotherapy for the groin nodes in the case of regional disease.
For women whose therapy cannot be modified, the model next considers the availability of risk-reducing medical interventions. For women with extensive disease and treatment, the availability of such interventions might reduce the level of risk from high to moderate. Examples of rehabilitative medical efforts include vaginal reconstruction for pelvic exenteration patients or labia preservation for vulvar cancer patients. For patients who have had such interventions, psychological, behavioral, and sexual outcomes are significantly better, although the interventions are not panaceas136 (see Berek and Andersen for a review164). For patients with limited disease and treatment, the availability of medical interventions could reduce their risk from low to approximately that of healthy individuals undergoing related experiences (i.e., the base rate). Some women experiencing a surgical menopause after treatment for localized cervical cancer can be treated with hormonal replacement therapy, similar to healthy women who experience a natural menopause with its attendant difficulties.165
The final contributor to risk is new health problems (e.g., hormonal changes or continuing stressors from the disease and treatment). Consideration of hormonal changes includes two issues: induced menopause and infertility. Most women who were premenopausal before gynecologic cancer become postmenopausal afterward either because of ovary removal or because of the inability to be treated with estrogens. Menopausal changes produce significant sexual effects.166 Also, ovary removal or sterilization ends childbearing – a potential stressor for a young woman with cancer. Continuing stressors can be heterogeneous. Examples include chronic fatigue or nausea and vomiting from toxic or lengthy chemotherapy regimens or late morbidities from treatments (e.g., a bowel fistula after pelvic irradiation). Should such problems arise, the model considers the availability of effective treatments. As can be seen, multiple pathways can lead to high or moderate risk for morbidity, but only patients who have limited disease and treatment and who have no new or continuing problems are hypothesized to have the lowest risk.
In addition to medical contributors, we have focused on identifying sexually relevant psychological factors in predicting risk for sexual morbidity. Before turning to this discussion, we note that the Figure 1 model begins with “baseline” sexual status. In addition to the individual difference variable described subsequently, straightforward predictors include such factors as age, sexual status (active or inactive), and prior frequency of important sexual activities (e.g., intercourse). These factors have emerged as important predictors of sexual activity in the earliest research on sexuality.167 These variables are important predictors in studies of healthy individuals, and their predictive utility is found in studies of individuals with chronic conditions and illnesses.168 We note these variables in preface to the subsequent discussion.
In the effort to identify a sexually relevant individual difference variable, we explored the concept of body image because it has been hypothesized as relevant to sexuality for healthy women169 and women with cancer.170 Our studies found, however, weak theoretical notions of the construct and poor operationalizations (i.e., measures of poor reliability and validity).171 In empirical tests, we found that measures could not predict outcome either among breast cancer patients172 or in multiple samples of women with gynecologic cancer.171 This finding led us away from considering a view of the body per se to a more central perspective – a woman’s view of herself as a sexual person.
We have proposed that sexual self-schema (self-concept) is a cognitive view about sexual aspects of the self; it is derived from past experience, it is manifest in current experience, and it guides the processing of domain-relevant social information.173 The concept includes two positive aspects – an inclination to experience romantic or passionate emotions and a behavioral openness to sexual experiences and relationships – and a negative aspect – embarrassment or conservatism, which seems to be a deterrent to sexual expression. Having developed a valid and reliable measure of sexual schema and tested it in the prediction of sexual behavior, attitudes, and responsiveness in healthy women, we wished to examine its predictive power in the context of risk for sexual morbidity after gynecologic cancer. Consistent with our definition of the construct, we that found that women “low or negative” in sexual self-concept, in contrast to women with a “high or positive” sexual self-concept, are at greatest risk for sexual difficulty. We anticipated that women with more negative sexual schemata would have more difficulties because they are, in general, less romantic or passionate in their emotions, less open to sexual experiences, and more likely to have negative feelings about their sexuality. In the context of cancer, with disease or treatment factors causing direct changes to the sexual body or sexual responses, we anticipated that women with low sexual self-schemata would be at greater risk. Women with negative self views of their sexuality might find that their sexual arousability would worsen; they might be less apt to try new sexual activities as a way to cope with their sexual difficulties; or they may have more negative cognitions or feelings, such as embarrassment, about any body changes.
We tested these hypotheses with gynecologic174, 175 and breast cancer patients.176 For the women with gynecologic cancer, 62 women who were currently disease-free but who had received treatment 6 months to 5 years previously for stage I or II disease were assessed. Comparison subjects included 68 women seeking routine gynecologic care. Analysis of the quality-of-life data replicated earlier prospective longitudinal findings82, 159 – specifically, sexuality is the major life area of disruption for the survivor. There were no differences between the groups in the areas of mental health (emotional distress, depression) or social functioning. The survivors did report, however, slightly more negative evaluations of their physical functioning and their perceptions of their health, yet there were no differences in reports of physical symptoms. In contrast, a comparison between the samples in terms of current sexual functioning found significant differences, with the cancer sample reporting lower levels of sexual behavior, sexual responsiveness, and global evaluations. We tested the risk for morbidity model162 and the utility of the schema construct with the cancer sample in the prediction of sexual responsiveness (i.e., desire, excitement, orgasm, and resolution) and sexual behavior (frequency of intercourse) with regression analyses. Sexual self-schema accounted for a significant and large portion of the variance (26%) in the prediction of current sexual responsiveness. In a subsequent study, a sample of survivors (n = 175) 2–10 years posttreatment were assessed during routine follow-up. Not only did sexual self schema account for significant variance in the prediction of current sexual behavior, responsiveness, and satisfaction, it also moderated the relationship between sexual satisfaction and psychological outcomes, suggesting that a positive sexual self schema might “buffer” patients from depressive symptoms when their sexual satisfaction is low. Furthermore, the combination of a negative sexual self schema and low sexual satisfaction appeared to heighten survivors’ risk for psychological distress, including depressive symptomatology.175 These general effects replicated in predicting sexuality and body change stress for women with breast cancer176 provide important, basic information on the processes of female sexuality and the specific relevance of the schema construct in understanding sexuality after gynecologic cancer.
The role for psychological interventions in improving sexual outcomes
Interventions can reduce distress, hasten resumption of routine activities, and improve social outcomes for groups at high risk for quality-of-life morbidity, such as patients with disseminated or recurrent disease and patients at low or moderate risk.3, 177 When controlled studies have been reviewed, effective therapy components have included an emotionally supportive context to address fears and anxieties about the disease, information about the disease and treatment, behavioral coping strategies (e.g., role playing difficult discussions with family or the medical staff), cognitive coping strategies, and relaxation training to lower “arousal” and enhance one’s sense of control. There are insufficient data to choose among components, but the literature suggests an emphasis on relaxation, coping, social support, and disease-specific components. The need for interventions to prevent or remediate sexual difficulties for these patients is apparent, but there is a gap between clinical knowledge and practice. Stead et al.178 interviewed 43 physicians and nurses regularly treating women with ovarian cancer. Ninety-eight per cent reported that they felt sexual issues should be discussed with patients, but only 21% reported doing so. When discussed, only 58% of healthcare professionals mentioned the potential for inhibited desire, 48% mentioned the possibility of fears about sexual activity, 42% noted dyspareunia, 30% altered arousal/vaginal dryness, and 7% altered pleasure or frequency of sexual activity.
Few intervention studies have been conducted, and of them only four have included sexuality as a treatment target or outcome.179, 180, 181, 182 In the earliest of these trials, Robinson and colleagues181 evaluated the effectiveness of a psychoeducational group in improving sexual outcomes for women with cervical and endometrial cancer (stage I or II) receiving intracavitary and external-beam radiotherapy. These women are at high risk for sexual morbidity because of the vaginal changes associated with radiation treatment (e.g., decrease in tissue elasticity; vaginal fibrosis, shortening, or dryness), all of which contribute to pain with intercourse. The use of vaginal dilators, which helps break fibrous adhesions as they develop, is often recommended to women who desire sexual activity. A total of 32 women completed the study (14 intervention and 18 control). The control condition included a brief counseling session in which subjects received a booklet on sexuality and cancer and were directed to the appropriate sections to answer any questions. The intervention comprised two 90-minute group sessions that included three components: information (extensive verbal and written instruction about sexuality and cancer), motivation (“sex can be pleasurable despite cancer treatment”), and behavioral skills (instruction on use of dilators, lubricants, and Kegel exercises). Subjects were reassessed every 3 months for 1 year. Analyses revealed that women receiving the intervention had significantly less fear about sex after treatment and were significantly more likely to follow recommendations for vaginal dilation than women in the control arm. This was particularly true for younger women; 44% of women aged 41 years and younger in the intervention complied with dilation recommendations compared with 6% of controls.
Caldwell and colleagues180 piloted a 12-week group therapy intervention in a sample of women who had been treated for any gynecologic cancer. Participants were randomly assigned to groups of three to five members that met weekly; session lasted 90 minutes. The intervention focused on issues related to body image and sexuality, with general themes outlined for each session, including communication with the sexual partner, coping with infertility, bodily changes relevant to sexuality, and changes in sexual desire. In this pretest–posttest design, assessments occurred pretreatment, posttreatment, and at 3 months posttreatment. Nineteen women completed the intervention and at least one follow-up assessment. While there was no control condition, repeated measures analyses indicated that sexual functioning improved posttreatment (p <0.05), with a trend toward sustained benefits in sexual arousal, sexual pleasure, and overall sexual functioning at 3 months (p <0.10). Brotto and colleagues developed an individual-focused pilot intervention for early-stage cervical and endometrial cancer patients meeting DSM-IV-TR criteria for Female Sexual Arousal Disorder.177 The intervention consisted of three 1-hour sessions (timed four weeks apart) combining elements of cognitive-behavioral therapy for sexual dysfunction and mindfulness training. Participants (n = 22) were assessed pre- and posttreatment. Results indicated that, posttreatment, participants demonstrated significant improvements in levels of sexual desire, arousal, orgasm, and sexual distress. Participants also reported significantly greater sexual satisfaction, fewer depresive symptoms, and better mental health quality of life following treatment.
Because cancer is known to affect both patients and partners, Scott et al.179 employed a couples therapy approach to improving sexual and psychological outcomes. Ninety-four married women with early-stage breast or gynecologic cancer and their partners were randomly assigned to a couples-based coping training (Can-COPE) intervention, individual coping training for the woman, or a medical education control. Outcomes included couples’ observed support communication and self-reported cancer-related distress, coping, and sexual adjustment. Those in the control condition received booklets containing information about their cancer and five brief phone calls focusing on patients’ understanding of their diagnosis and medical treatments. Patients in the individual therapy condition received the same educational materials as controls and also four 2-hour in-person therapy sessions (conducted in their homes) and two 30-minute telephone sessions focusing on common psychological reactions to cancer diagnosis, coping training, and supportive counseling. The Can-COPE intervention consisted of five 2-hour, in-person couple therapy sessions and two 30-minute telephone sessions. Sessions were held in the couples’ homes. In addition to the educational materials, coping skills training, and counseling received by those in the individual therapy condition, Can-COPE participants received training in supportive communication, partner support, and counseling about their sexuality. Participants were assessed at diagnosis, after cancer surgery, and at 6- and 12-month follow-ups. Prior to intervention, cancer-related cessation of sexual activity was reported in 72% of patients with gynecologic cancer, compared to 17% of patients with breast cancer. Results indicated that participants in the Can-COPE intervention experienced less decline in sexual intimacy than those in the other conditions; Can-COPE also produced significant improvements in couples’ supportive communication, reduced psychological distress, and coping effort.
Three additional studies included minor sexuality components. Two studies included women at low morbidity risk. Capone and coworkers183 provided a brief, crisis-oriented intervention. For sexually active women, a sexual therapy component included sexual information and methods to cope and reduce anxiety when resuming intercourse. There were 56 newly diagnosed women (88% were stage I through III). A nonequivalent control group was obtained by recruiting previously treated women. Analyses indicated no differences between groups or within the intervention group on the measures of emotional distress. A trend in the percentages of women returning to work favored the intervention participants (e.g., 50% versus 25% at 3 months). Substantial differences were found between the groups in the resumption and frequency of intercourse across all post-treatment assessments (e.g., 16% of the intervention versus 57% of the control women reported less or no sexual activity at 12 months’ post-treatment). The second quasiexperimental investigation was reported by Houts and associates184 and used a peer-counseling model. Interventions were delivered in telephone contacts and booklet descriptions of the coping strategies. A total of 32 women (14 intervention and 18 control) participated. Analyses indicated no differences between groups at any point in time. The only study with moderate-risk gynecologic cancer patients is that of Cain and associates185 comparing individual and group therapy formats. The intervention included discussion of the causes of cancer at diagnosis, impact of the treatments on body image and sexuality, relaxation training, and setting goals for the future to cope with uncertainty and fears of recurrence, delivered in eight sessions. A total of 72 women (21 individual intervention, 22 group intervention, and 29 no treatment control) completed the study. Analyses indicated that there were no differences between the intervention formats, but both groups reported less depression and anxiety, better psychosocial adjustment (including health perspectives and use of leisure time), and better sexual functioning than the no-treatment control group. These quasi-experimental designs suggest that broad-based interventions produce modest gains in psychological outcomes, but, in contrast, significant improvement can be found in sexual functioning.
CONCLUSIONS
For most patients, gynecologic cancer is a survivable disease. Future research will likely test the generalizability of these descriptive data and formulate general principles of adjustment to illness. While providing estimates of the magnitude of quality-of-life problems, these data can be used for models that predict which patients might be at greatest risk for adjustment difficulties.5 The latter is an important step toward designing interventions tailored to the difficulties and circumstances of cancer patients.
A growing literature on the use of psychological interventions to improve cancer patients’ quality of life exists.3, 119 The effectiveness of these interventions is robust, as they have reduced distress and enhanced the quality of life of many cancer patients differing in disease stage and disease site. Despite the challenges of studying these patients, well-controlled investigations have been conducted. Improvements in emotional distress are found at the end of the interventions with continued gains at follow-up. In addition, changes in other areas – self-esteem and self-concept, death perceptions, life satisfaction, and locus of control – have been found. Important for quality of life, psychological interventions also could lower or stabilize pain reports. The positive outcomes for terminal patients are notable considering their worsening pain or increasing debilitation.
The descriptive data also highlight the need for focused interventions for sexual functioning. Available data suggest cautious optimism that psychological and sexual outcomes can be improved through general psychological interventions, but it is likely that greater gains could be achieved if sexuality-specific interventions were a central part of a therapeutic effort. A foundation of clinical research in gynecologic cancer and basic research in sexuality has led to the targeting of important individual difference variables. As shown in Figure 1, we offer a strategy for considering the magnitude of disease and treatment. This is a working model, although it is empirically supported. Longitudinal data have highlighted the early trajectory of sexual problem development. Issues of quality of life are important, and positive results have been achieved by behavioral sciences. As with most issues, further commitment and action are needed.
REFERENCES
Jemal A, Siegel R, Ward E et al: Cancer statistics, 2008. CA Cancer J Clin 58(2): 71–96, 2008 |
|
Reis LAG, Eisner MP, Kosary CL et al: SEER cancer statistics review, 1975-2002. Bethesda, MD: National Cancer Institute, 2004 |
|
Andersen BL: Psychological interventions for cancer patients to enhance the quality of life. J Consult Clin Psychol 60: 552, 1992 |
|
Andersen BL, Kiecolt-Glaser JK, Glaser R: A biobehavioral model of cancer stress and disease course. Am Psych 49: 389, 1994 |
|
Andersen BL: Surviving cancer. Cancer 74: 1484, 1994 |
|
Department of Health and Human Services: Healthy People 2010: Understanding and Improving Health. 2nd ed. Washington, DC, U.S. Government Printing Office, 2000 |
|
U.S. Department of Health and Human Services (HHS). The Health Benefits of Smoking Cessation. DHHS Publication No. CDC 90-8416. Atlanta, GA: Public Health Service, Centers for Disease Control, Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, 1990 |
|
Willet, W. Diet and nutrition. In: Schottenfeld, D., and Fraumeni, Jr., J.F.; eds. Cancer Epidemiology and Prevention. 2nd ed. New York, NY: Oxford University Press, 1996, 438–461 |
|
Greenwald P, Kramer B, Weed DL. eds. Cancer Prevention and Control. New York, NY: Marcel Dekker, 1995, 303–327 |
|
HHS. Physical Activity and Health: A Report of the Surgeon General. Atlanta, GA: Centers for Disease Control and Prevention (CDC), 1996 |
|
Easton DF, Bishop DT, Ford D, Crockford DP: Genetic linkage analysis in familial breast and ovarian cancer: Results from 214 families. Am J Hum Genet 52: 678, 1993 |
|
Ford D, Easton DF, Bishop DT et al: Risks of cancer in BRCA-1 mutation carriers. Breast Cancer Linkage Consortium Lancet 343: 692, 1994 |
|
Antoniou A, Pharoah PD, Narod S et al: Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2mutations detected in case Series unselected for family history: a combinedanalysis of 22 studies. Am J Hum Genet. 2003 May;72(5):1117-30. Epub 2003 Apr 3. |
|
Lerman C, Narod S, Schulman K et al: BRCA1 testing in families with hereditary breast-ovarian cancer : A prospective study of patient decision making and outcomes. JAMA 275: 1885, 1996 |
|
Smith PM, Schwartz PE: New fears in gynecologic cancer. Cancer 76: 2133, 1995 |
|
Coyne JC, Benazon NR, Gaba CG et al: Distress and psychiatric morbidity among women from high-risk breast and ovarian cancer families. J Consult Clin Psychol 68: 864, 2000 |
|
Botkin JR, Croyle RT, Smith KR et al: A model protocol for evaluating the behavioral and psychosocial effects of BRCA1 testing. J Natl Cancer Inst 88: 872, 1996 |
|
van Oostrom I, Meijers-Heijboer H, Lodder LN et al: Long-term psychological impact of carrying a BRCA1/2 mutation and prophylactic J Clin Oncol. 2003 Oct 15;21(20):3867-74. |
|
Madalinska JB, Hollenstein J, Bleiker E et al: Quality-of-life effects of prophylactic salpingo-oophorectomy versus gynecologicscreening among women at increased risk of hereditary ovarian cancer. J Clin Oncol. 2005 Oct 1;23(28):6890-8. Epub 2005 Aug 29. |
|
Munoz N, Bosch FX, Castellsague X et al: Against which human papillomavirus types shall we vaccinate and screen? Theinternational perspective. Int J Cancer. 2004 Aug 20;111(2):278-85. |
|
Steinbrook R: The potential of human papillomavirus vaccines. N Engl J Med. 2006 Mar 16;354(11):1109-12. |
|
Centers for Disease Control and Prevention. Quadravalent Humanpapillomavirus Vaccine: Recommendations of the Advisory Committee on Immunization Practices. MMWR 2007;56(No. RR-2). |
|
Brewer NT, Fazekas KI: Predictors of HPV vaccine acceptability: a theory-informed, systematic review. Prev Med. 2007 Aug-Sep;45(2-3):107-14. Epub 2007 Jun 2. |
|
Dempsey AF, Davis MM: Overcoming barriers to adherence to HPV vaccination recommendations. Am J Manag Care. 2006 Dec;12(17 Suppl):S484-91. |
|
Pirog EC, Heller DS, Westhoff C: Endometrial adenocarcinoma: Lack of correlation between treatment delay and tumor stage. Gynecol Oncol 67: 303, 1997 |
|
Franco K, Belincon J, Casey G et al: Adjusted to perceived ovarian cancer risk. Psycho-oncology 9: 411, 2000 |
|
Pack GT, Gallo JS: Culpability for delay in treatment of cancer. Am J Cancer 33: 443, 1938 |
|
Menczer J: Diagnosis and treatment delay in gynecological malignancies: Does it affect outcome? Int J Gynecol Cancer 10: 89, 2000 |
|
Andersen BL, Cacioppo JT, Roberts DC: Delay in seeking a cancer diagnosis: Delay stages and psychophysiological comparison processes. Br J Soc Psychol 34: 33, 1995 |
|
Weisman AD, Worden JW: The existential plight in cancer: Significance of the first 100 days. Int J Psychiatry Med 7: 1, 1976 |
|
Sardell AN, Trierweiler SJ: Disclosing the cancer diagnosis. Cancer 72: 3355, 1993 |
|
Roberts JA, Brown D, Elkins T, Larson DB: Factors influencing views of patients with gynecologic cancer about end-of-life decisions. Am J Obstet Gynecol 176: 166, 1997 |
|
Sell L, Devlin B, Bourke SJ et al: Communicating the diagnosis of lung cancer. Respir Med 87: 61, 1993 |
|
Woodard LJ, Pamies RJ: The disclosure of the diagnosis of cancer. Prim Care 19: 657, 1992 |
|
Rutter DR, Iconomou G, Quine L: Doctor-patient communication and outcome in cancer patients: An intervention. Psychol Health 12: 57, 1996 |
|
Ley P: Communicating with Patients. London, Chapman & Hall, 1988 |
|
Spielberger CD, Gorsuch RL, Lushene RD: Manual for the State-Trait Anxiety Inventory (STAI). Palo Alto, CA, Consulting Psychologists Press, 1970 |
|
Beck AT, Beck RW: Screening depressed patients in family practice: A rapid technique. Postgrad Med 52: 81, 1972 |
|
Locke BZ, Regier DA: Prevalence of selected mental disorders, p 1. In Taube CA, Barrett SA (eds): Mental Health United States. Rockville, MD, National Institute of Mental Health, 1985 |
|
Bodurka-Bevers D, Basen-Engquist K, Carmack CL et al: Depression, anxiety, and quality of life in patients with epithelial ovarian cancer. Gynecol Oncol 78: 302, 2000 |
|
Derogatis LR, Morrow GR, Fetting J et al: The prevalence of psychiatric disorders among cancer patients. JAMA 249: 751, 1983 |
|
Kornblith AB, Thaler HT, Wong G et al: Quality of life of women with ovarian cancer. Gynecol Oncol 59: 231, 1995 |
|
Massie MJ, Holland JC: Depression and the cancer patient. J Clin Psychiatry 51: 12, 1990 |
|
Levin SH, Jones LD, Sack DA: Evaluation and treatment of depression, anxiety, and insomnia in patients with cancer. Oncology 7: 119, 1993 |
|
Meyerowitz BE, Formenti SC, Ell KO, Leedham B: Depression among Latina cervical cancer patients. J Soc Clin Psychol 19: 352, 2000 |
|
Bukberg J, Penman D, Holland JC: Depression in hospitalized cancer patients. Psychosom Med 46: 199, 1984 |
|
Cella DF, Orofiamma B, Holland JC et al: The relationship of psychological distress, extent of disease, and performance status in patients with lung cancer. Cancer 60: 1661, 1987 |
|
Steginga SK, Dunn J: Women’s experiences following treatment for gynecologic cancer. Oncol Nurs Forum 24: 1403, 1997 |
|
Bradley EJ, Pitts MK, Redman CWE, Calvert E: The experience of long-term hospital follow-up for women who have suffered early stage gynecological cancer: A qualitative interview study. Int J Gynecol Cancer 9: 491, 1999 |
|
Marks G, Richardson JL, Graham JW, Levine A: Role of health locus of control beliefs and expectations of treatment efficacy in adjustment to cancer. J Person Soc Psychol 51: 443, 1986 |
|
Carver CS, Pozo-Kaderman C, Harris SD et al: Optimism versus pessimism predicts the quality of women’s adjustment to early stage breast cancer. Cancer 73: 1213, 1994 |
|
Miller DL, Manne SL, Talyor K et al: Psychological distress and well-being in advanced cancer: The effects of optimism and coping. J Clin Psychol Medical Settings 3: 115, 1996 |
|
Lutgendorf SK, Anderson B, Rothrock N et al: Quality of life and mood in women receiving extensive chemotherapy for gynecologic cancer. Cancer 89: 1402, 2000 |
|
Andersen BL: Sexuality and quality of life of women with vulvar cancer, p 202. In Luesley DM (ed): Cancer and Pre-Cancer of the Vulva. London, Arnold, 2000 |
|
Hamilton AB: Psychological aspects of ovarian cancer. Cancer Invest 17: 335, 1999 |
|
McQuellon RP, Wells M, Hoffman S et al: Reducing distress in cancer patents with an orientation program. Psycho-oncology 7: 207, 1998 |
|
Powell CB, Kneier A, Chen LM et al: A randomized study of the effectiveness of a brief psychosocial intervention forwomen attending a gynecologic cancer clinic. Gynecol Oncol. 2008 Oct;111(1):137-43. Epub 2008 Aug 13. |
|
Gottesman D, Lewis M: Differences in crisis reactions among cancer and surgery patients. J Consult Clin Psychol 50: 381, 1982 |
|
Johnston M, Vogele C: Benefits of psychological preparation for surgery: A meta-analysis. Ann Behav Med 15: 245, 1993 |
|
Vokes EE, Haraf DJ, Stenson K et al: Concomitant chemoradiotherapy as primary therapy for loco-regionally advanced head and neck cancer. J Clin Oncol 18: 1652, 2000 |
|
Formenti SC, Meyerowitz BE, Ell K et al: Inadequate adherence to radiotherapy in Latina immigrants with carcinoma of the cervix. Cancer 75: 1135, 1995 |
|
Formenti SC, Meyerowitz BE, Ell K et al: Inadequate adherence to radiotherapy in Latina immigrants with carcinoma of the cervix. Cancer 75: 1135, 1995 |
|
Wallace LM, Priestman SG, Dunn JA, Priestman TJ: The quality of life of early breast cancer patients treated by two different radiotherapy regimens. Clin Oncol 5: 228, 1993 |
|
Andersen BL, Karlsson JA, Anderson B, Tewfik HH: Anxiety and cancer treatment: Response to stressful radiotherapy. Health Psychol 3: 535, 1984 |
|
Andersen BL, Tewfik HH: Psychological reactions to radiation therapy: Reconsideration of the adaptive aspects of anxiety. J Person Soc Psychol 48: 1024, 1985 |
|
Fieler VK: Side effects and quality of life in patients receiving high-dose rate brachytherapy. Oncol Nurs Forum 24: 545, 1997 |
|
Klee M, Thranov I, Machin D: The patients’ perspective on physical symptoms after radiotherapy for cervical cancer. Gynecol Oncol 76: 14, 2000 |
|
Hughson AV, Cooper AF, McArdle CS, Smith DC: Psychosocial effects of radiotherapy after mastectomy. BMJ 294: 1515, 1987 |
|
Katz A, Njuguna E, Rakowski E et al: Early development of vaginal shortening during radiation therapy for endometrial or cervical cancer. Int J Gynecol Cancer 11: 234, 2001 |
|
Leon-Pizarro C, Gich I, Barthe E et al: A randomized trial of the effect of training in relaxation and guided imageryand breast brachytherapy patients. Psychooncology. 2007 Nov;16(11):971-9. |
|
Andrykowski MA: The Morrow/Hickok article reviewed: Behavioral treatment of chemotherapy-induced nausea and vomiting. Oncology 7: 93, 1993 |
|
Morrow GR, Hickok JT: Behavioral treatment of chemotherapy-induced nausea and vomiting. Oncology 7: 83, 1993 |
|
Redd WH, Montgomery GH, DuHamel KN: Behavioral intervention for cancer-treatment side-effects. J Natl Cancer Inst 93: 810, 2001 |
|
Nail LM, King KB: Fatigue. Semin Oncol Nurs 3: 257, 1987 |
|
Smets EM, Garssen B, Schuster-Uitterhoeve AL, deHaes JC: Fatigue in cancer patients. Br J Cancer 68: 220, 1993 |
|
Smets EM, Visser MRM, Garssen B, et al: Understanding level of fatigue in cancer patients undergoing radiotherapy. J Psychosom Res 45: 277, 1998 |
|
Hann DM, Garovoy N, Finkelstein B et al: Fatigue and quality of life in breast cancer patients undergoing autologous stem cell transplantation: A longitudinal comparative study. J Pain Symptom Manage 17: 311, 1999 |
|
Jacobsen PB, Hann DM, Azzarello LM et al: Fatigue in women receiving adjuvant chemotherapy for breast cancer: Characteristics, course, and correlates. J Pain Symptom Manage 18: 233, 1999 |
|
Rhodes VA, Watson PM, Hanson BM: Patients’ descriptions of the influence of tiredness and weakness on self-care abilities. Cancer Nursing 1988 11: 186, 194 |
|
Larsson G, Starrin B: Relaxation training as an integral part of caring activities for cancer patients: Effects on well-being. Scand J Caring Sci 6: 179, 1992 |
|
Smith MY, Redd WH, Peyser C, Vogl D: Post-traumatic stress disorder in cancer: A review. Psycho-oncology 8: 521, 1999 |
|
Andersen BL, Anderson B, deProsse C: Controlled prospective longitudinal study of women with cancer: II. Psychological outcomes J Consult Clin Psychol 57: 692, 1989 |
|
Bard M, Sutherland AM: Adaptation to radical mastectomy. Cancer 8: 656, 1952 |
|
Cella DF, Tross S: Psychological adjustment to survival from Hodgkin’s disease. J Consult Clin Psychol 54: 616, 1986 |
|
Baider L, Sarell M: Couples in crisis: Patient-spouse differences in perception of interaction patterns and the illness situation. Fam Ther 11: 115, 1984 |
|
Lichtman RR, Taylor SE: Close relationships and the female cancer patient, p 233. In Andersen BL (ed): Women with Cancer: Psychological Perspectives. New York, Springer-Verlag, 1986 |
|
Meyerowitz BE, Watkins IK, Sparks FC: Psychosocial implications of adjuvant chemotherapy: A two-year follow-up. Cancer 52: 1541, 1983 |
|
Taylor-Brown J, Kilpatrick M, Maunsell E, Dorval M: Partner abandonment of women with breast cancer: Myth or reality? Cancer Pract 8: 160, 2000 |
|
Baider L, Cooper CL, De-Nour AK (eds): Cancer and the Family. New York, Wiley, 2000 |
|
Sales E, Schulz R, Biegel D: Predictors of strain in families of cancer patients: A review of the literature. J Psychosoc Oncol 10: 1, 1992 |
|
Vess JD, Moreland JR, Schwebel AI: A follow-up study of role functioning and the psychosocial environment of families of cancer patients. J Psychosoc Oncol 3: 1, 1985 |
|
Ell K, Nishimoto R, Mantell J, Hamovitch M: Longitudinal analysis of psychological adaptation among family members of patients with cancer. J Psychosom Res 32: 429, 1988 |
|
Eisemann M, Lalos A: Psychosocial determinants of well-being in gynecologic cancer patients. Cancer Nurs 22: 303, 1999 |
|
Greimel ER, Friedl W: Functioning in daily living and psychological well-being of female cancer patients. J Psychosom Obstet Gynecol 21: 25, 2000 |
|
Chan YM, Hnan HYS, Yip PSF et al: Psychosocial adjustment in gynecologic cancer survivors: A longitudinal study on risk factors for maladjustment. Gynecol Oncol 80: 387, 2001 |
|
Klee M, Thranov I, Machin D: The patients’ perspective on physical symptoms after radiotherapy for cervical cancer. Gynecol Oncol 76: 14, 2000 |
|
Simonelli LE, Fowler J, Maxwell GL et al: Physical sequelae and depressive symptoms in gynecologic cancer survivors:meaning in life as a mediator. Ann Behav Med. 2008 Jun;35(3):275-84. Epub 2008 Apr 3. |
|
Matulonis UA, Kornblith A, Lee H et al: Long-term adjustment of early-stage ovarian cancer survivors. Int J Gynecol Cancer. 2008 Jan 22. |
|
Hodgkinson K, Butow P, Fuchs A et al: Long-term survival from gynecologic cancer: psychosocial outcomes, supportivecare needs and positive outcomes. Gynecol Oncol. 2007 Feb;104(2):381-9. Epub 2006 Oct 5. |
|
Champion V, Williams SD, Miller A et al: Quality of life in long-term survivors of ovarian germ cell tumors: a GynecologicOncology Group study. Gynecol Oncol. 2007 Jun;105(3):687-94. Epub 2007 Mar 13. |
|
Costanzo ES, Lutgendorf SK, Bradley SL et al: Cancer attributions, distress, and health practices among gynecologic cancersurvivors. Psychosom Med. 2005 Nov-Dec;67(6):972-80. |
|
Carlsson M, Strang P, Bjurstrom C: Treatment modality affects long-term quality of life in gynaecological cancer. Anticancer Res. 2000 Jan-Feb;20(1B):563-8. |
|
Matthews AK, Aikens JE, Helmrich SP, Anderson DD, Herbst AL, Waggoner SE. Sexual functioning and mood among long-term survivors of clear-cell adenocarcinoma of the vagina or cervix. Journal of Psychosocial Oncology. 1999;17(3-4):27-45 |
|
Wenzel LB, Donnelly JP, Fowler JM et al: Resilience, reflection, and residual stress in ovarian cancer survivorship: agynecologic oncology group study. Psychooncology. 2002 Mar-Apr;11(2):142-53. |
|
Manne S, Rini C, Rubin S et al: Long-term trajectories of psychological adaptation among women diagnosed withgynecological cancers. Psychosom Med. 2008 Jul;70(6):677-87. |
|
Miller BE, Pittman B, Case D et al: Quality of life after treatment for gynecologic malignancies: a pilot study in anoutpatient clinic. Gynecol Oncol. 2002 Nov;87(2):178-84. |
|
Parker PA, Baile WF, De Moor C, Cohen L. Psychosocial and demographic predictors of quality of life in a large sample of cancer patients. Psycho-Oncology. 2003;12(2):183-193 |
|
Thompson L, Andersen BL, DePetrillo D: The psychological processes of recovery from gynecologic cancer. In Coppleson M, Morrow P, Tattersall M (eds): Gynecologic Oncology. p. 1499, 2nd ed. Edinburgh, Churchill Livingstone, 1992 |
|
Cassileth BR, Lunk EJ, Strouse TB et al: A psychological analysis of cancer patients and their next-of-kin. Cancer 55: 72, 1985 |
|
Bloom JR: Psychological response to mastectomy. Cancer 59: 189, 1987 |
|
Le T, Hopkins L, Fung Kee Fung M: Quality of life assessments in epithelial ovarian cancer patients during andafter chemotherapy. Int J Gynecol Cancer. 2005 Sep-Oct;15(5):811-6. |
|
Linn MW, Linn BS, Harris R: Effects of counseling for late stage cancer patients. Cancer 49: 1048, 1982 |
|
Nitschke R, Humphrey GB, Sexauer CL et al: Therapeutic choices made by patients with end-stage cancer. J Pediatr 101: 471, 1982 |
|
Speigel D, Bloom JR, Yalom I: Group support for patients with metastatic cancer: A randomized outcome study. Arch Gen Psychiatry 38: 527, 1981 |
|
Speigel D, Bloom JR: Group therapy and hypnosis reduce metastatic breast carcinoma pain. Psychosom Med 45: 333, 1983 |
|
Ahles TA, Ruckdeschel JC, Blanchard EB: Cancer related pain: I. Prevalence in an outpatient setting as a function of stage of disease and type of cancer J Psychosom Res 28: 115, 1984 |
|
Rummans TA, Frost M, Suman VJ et al: Quality of life and pain in patients with recurrent breast and gynecologic cancer. Psychosomatics 39: 437, 1998 |
|
Glover J, Dibble SL, Dodd MJ, Miaskowski C: Mood states of oncology outpatients: Does pain make a difference? J Pain Sympt Manage 10: 120, 1995 |
|
Andersen BL: Biobehavioral outcomes following psychological interventions for cancer patients. J Consult Clin Psychol 70: 590, 2002 |
|
Carter J, Saltzman A, Chen D et al: Quality of life assessment in gynaecological cancer patients. Aust N Z Obstet Gynaecol 35: 293, 1995 |
|
Syrjala KL, Schroeder TC, Abrams JR et al: Sexual function measurement and outcomes in cancer survivors and matched controls. J Sex Res 37: 213, 2000 |
|
Stead ML, Crocombe WD, Fallowfield LJ et al: Sexual activity questionnaires in clinical trials: Acceptability to patients with gynaecological disorders. Br J Obstet Gynaecol 106: 50, 1999 |
|
Vincent CE, Vincent B, Greiss FC, Linton EB: Some marital-sexual concomitants of carcinoma of the cervix. South Med J 68: 552, 1975 |
|
Trimbos JB, Maas CP, Deruiter MC et al: A nerve-sparing radical hysterectomy: Guidelines and feasibility in Western patients. Int J Gynecol Cancer 11: 180, 2001 |
|
Froewis J, Picha E: Der Einfluss der Radikaloperationen bei Carcinoma colli uteri auf das Sexualleben der Frau mit besonderer Berucksichtigung der Wertheimschen Operation [The influence of radical operations upon the sexual activity of women with special regards to Wertheim surgery]. Geburtshilfe Frauenhilkd 15: 806, 1955 |
|
Lau HU: Rehabilitation bei kollumkarzinomen [Rehabilitation for cervical carcinoma]. Zentralbl Gynakol 89: 1649, 1967 |
|
Cartwright-Alcarese F: Addressing sexual dysfunction following radiation therapy for a gynecologic malignancy. Oncol Nurs Forum 22: 1227, 1995 |
|
Katz A, Njuguna E, Rakowski E et al: Early development of vaginal shortening during radiation therapy for endometrial or cervical cancer. Int J Gynecol Cancer 11: 234, 2001 |
|
Eifel PF: Radiation therapy, p 117. In Berek JS, Hacker NF (eds): Practical Gynecologic Oncology. 3rd ed. Philadelphia, Lippincott Williams & Wilkins, 2000 |
|
Andersen BL, Karlsson JA, Anderson B, Tewfik HH: Anxiety and cancer treatment: Response to stressful radiotherapy. Health Psychol 3: 535, 1984 |
|
Pitkin RM, van Voorhis LW: Post irradiation vaginitis: An evaluation of prophylaxis with topical estrogen. Therap Radiol 99: 417, 1971 |
|
Turns D: Psychosocial issues: Pelvic exenteration. J Surg Oncol 76: 224, 2001 |
|
Lamont JA, dePetrillo AD, Sargeant EJ: Psychosexual rehabilitation and exenterative surgery. Gynecol Oncol 6: 236, 1978 |
|
Corney RH, Crowther ME, Everett H et al: Psychosexual dysfunction in women with gynaecological cancer following radical pelvic surgery. Br J Obstet Gynaecol 100: 73, 1993 |
|
Roos EJ, de Graeff A, van Eijkeren MA et al: Quality of life after pelvic exenteration. Gynecol Oncol. 2004 Jun;93(3):610-4. |
|
Andersen BL, Hacker NF: Psychosexual adjustment after vulvar surgery. Obstet Gynecol 62: 457, 1983 |
|
Brown RS, Haddox V, Posada A, Rubio A: Social and psychological adjustment following pelvic exenteration. Am J Obstet Gynecol 114: 162, 1972 |
|
Dempsey GM, Buchsbaum HJ, Morrison J: Psychosocial adjustment to pelvic exenteration. Gynecol Oncol 3: 325, 1975 |
|
Knorr NJ: A depressive syndrome following pelvic exenteration and ileostomy. Arch Surg 94: 258, 1967 |
|
Vera MI: Quality of life following pelvic exenteration. Gynecol Oncol 12: 355, 1981 |
|
Gleeson N, Baile W, Roberts WS et al: Surgical and psychosexual outcome following vaginal reconstruction with pelvic exenteration. Eur J Gynaecol Oncol 15: 89, 1994 |
|
DiSaia PJ, Creasman WT, Rich WM: An alternate approach to early cancer of the vulva. Am J Obstet Gynecol 33: 825, 1979 |
|
Hacker NF, Berek JS, Nieberg RK et al: Individualization of treatment for stage I squamous cell vulvar carcinoma. Obstet Gynecol 63: 155, 1984 |
|
Makar APH, Scheistroen M, Van den Weyngaert D, Trope CG: Surgical management of stage I and II vulvar cancer: The role of the sentinel node biopsy: Review of the literature. Int J Gynecol Cancer 11: 255, 2001 |
|
Hacker NF: Radical resection of vulvar malignancies: A paradigm shift in surgical approaches. Curr Opin Obstet Gynecol 11: 61, 1999 |
|
Rodrigues M, Sevin B-U, Averette HE, et al: Conservative trends in the surgical management of vulvar cancer: A University of Miami Patient Care Evaluation study. Int J Gynecol Cancer 7: 151, 1997 |
|
Andersen BL, Turnquist D, LaPolla J, Turner D: Sexual functioning after treatment of in-situ vulvar cancer: Preliminary report. Obstet Gynecol 71: 15, 1988 |
|
Andresson B, Moth I, Jensen SB, Bock JE: Sexual function and somatopsychic reactions in vulvectomy-operated women and their partners. Acta Obstet Gynecol Scand 65: 7, 1986 |
|
Weijmar Schultz WCM, Wijma K, Van de Wiel HBM et al: Sexual rehabilitation of radical vulvectomy patients: A pilot study. J Psychosom Obstet Gynecol 5: 119, 1986 |
|
Tamburini M, Filiberti A, Ventafridda V, DePalo G: Quality of life and psychological state after radical vulvectomy. J Psychosom Obstet Gynecol 5: 263, 1986 |
|
Likes WM, Stegbauer C, Tillmanns T et al: Correlates of sexual function following vulvar excision. Gynecol Oncol. 2007 Jun;105(3):600-3. Epub 2007 Feb 15. |
|
Green MS, Naumann RW, Elliot M et al: Sexual dysfunction following vulvectomy. Gynecol Oncol 77: 73, 2000 |
|
Andersen BL, Lachenbruch PA, Anderson B, deProsse C: Sexual dysfunction and signs of gynecologic cancer. Cancer 57: 1880, 1986 |
|
Harris R, Good RS, Pollack L: Sexual behavior of gynecologic cancer patients. Arch Sex Behav 11: 503, 1982 |
|
Schover LR, Fife M, Gershenson DM: Sexual dysfunction and treatment for early stage cervical cancer. Cancer 63: 204, 1989 |
|
Flay LD, Matthews JHL: The effects of radiotherapy and surgery on the sexual function of women treated for cervical cancer. Int J Radiat Oncol Biol Phys 31: 399, 1995 |
|
Hansen MK: Surgical and combination therapy of cancer of the cervix uteri stages Ib and IIa. Gynecol Oncol 11: 275, 1981 |
|
Adelusi B: Coital function after radiotherapy for carcinoma of the cervix uteri. Br J Obstet Gynaecol 84: 821, 1980 |
|
Andersen BL, Anderson B, deProsse C: Controlled prospective longitudinal study of women with cancer: I. Sexual functioning outcomes J Consult Clin Psychol 57: 683, 1989 |
|
Weijmar Schultz WCM, Van de Wiel HBM, Bouma J et al: Psychosexual functioning after treatment for cancer of the cervix: A comparative and longitudinal study. Int J Gynecol Cancer 1: 37, 1991 |
|
Kylstra WA, Leenhouts GHMW, Everaerd W et al: Sexual outcomes following treatment for early stage gynecological cancer: A prospective multicenter study. Int J Gynecol Cancer 9: 387, 1999 |
|
Andersen BL: Health psychology’s contribution to addressing the cancer problem: Update on accomplishments. Health Psychol 8: 683, 1989 |
|
American Cancer Society: Cancer Facts and Figures—1994. New York, ACS, 1994 |
|
Berek J, Andersen BL: Sexual rehabilitation: Surgical and psychological approaches, p 401. In Hoskins WJ, Perez CA, Young RC (eds): Principles and Practice of Gynecologic Oncology. Philadelphia, JB Lippincott, 1992 |
|
Matthews KA, Wing RR, Kuller LH et al: Influences of natural menopause on psychological characteristics and symptoms of middle-aged healthy women. J Consult Clin Psychol 58: 345, 1990 |
|
Walling MK, Andersen BL, Johnson SR: Hormonal replacement therapy for postmenopausal women: Sexual outcomes and related gynecologic effects. Arch Sex Behav 19: 119, 1990 |
|
Kinsey A, Pomeroy W, Martin C, Gebhard P: Sexual Behavior in the Human Female. Philadelphia, WB Saunders, 1953 |
|
Curry SL, Levine SB, Jones PK, Kurit DM: Medical and psychosocial predictors of sexual outcome among women with systemic lupus erythematosus. Arthritis Care Res 6: 23, 1993 |
|
Cash TF: Body Image Therapy: A Program for Self-Directed Change. New York, Guilford, 1991 |
|
Derogatis LR: Breast and gynecologic cancers: Their unique impact on body image and sexual identity in women, p 1. In Vaeth JM (ed): Frontiers of Radiation Therapy and Oncology. Basel S, Karger AG, 1980 |
|
Andersen BL, LeGrand J: Body image for women: Conceptualization, assessment, and a test of its importance to sexual dysfunction and medical illness. J Sex Res 28: 457, 1991 |
|
Andersen BL, Jochimsen PR: Sexual adjustment among breast cancer, gynecologic cancer, and healthy women. J Consult Clin Psychol 53: 25, 1985 |
|
Andersen BL, Cyranowski JC: Women’s sexual self-schema. J Person Soc Psychol 67: 1079, 1994 |
|
Andersen BL, Woods X, Cyranowski JC: Sexual self schema predicts sexual morbidity following cancer. Can J Hum Sexuality 3: 165, 1994 |
|
Carpenter KM, Andersen BL, Fowler JM et al: Sexual Self Schema as a Moderator of Sexual and Psychological Outcomes for Gynecologic Cancer Survivors. Arch Sex Behav. 2008 Apr 17. |
|
Yurek D, Farrar W, Andersen BL: Breast cancer surgery: Comparing surgical groups and determining individual differences in post operative sexuality and body change stress. J Consult Clin Psychol 68: 697, 2000 |
|
Brotto, L. A., & Heiman, J. R: Mindfulness in sex therapy: Applications for women with sexual difficulties following gynecologic cancer. Sex Relationship Ther 22, 1468-1994, 2007 |
|
Stead, M. L., Brown, J. M., Fallowfield, L., & Selby, P: Lack of communication between healthcare professionals and women with ovarian cancer about sexual issues. Br J Cancer, 88, 666-671, 2003 |
|
Scott, J. L., Halford, W. K., & Ward, B. G: United we stand? The effects of a couple-coping intervention on adjustment to early stage breast or gynecological cancer. J Consult Clin Psychol 72, 1122-1135, 2004 |
|
Caldwell, R., Classen, C., Lagana, L., McGarvey, E., Baum, L., Duenke, S. D., et al:Changes in sexual functioning and mood among women treated for gynecological cancer who receive group therapy: A pilot study. J Clin Psychol Med Settings 10, 149-156, 2003 |
|
Robinson JW, Faris PD, Scott CB: Psychoeducational group increases vaginal dilation for younger women and reduces sexual fears for women of all ages with gynecologic carcinoma treated with radiotherapy. Int J Radiat Oncol Biol Phys 44: 497, 1999 |
|
Capone, M. A., Good, R. S., Westie, K. S., & Jacobson, A. F: Psychosocial rehabilitation of gynaecologic oncology patients. Arch Phys Med Rehabil 61, 128-132, 1980 |
|
Capone MA, Good RS, Westie KS, Jacobson AF: Psychosocial rehabilitation of gynecologic oncology patients. Arch Phys Med Rehabil 61: 128, 1980 |
|
Houts PS, Whitney CW, Mortel R, Bartholomew MJ: Former cancer patients as counselors of newly diagnosed cancer patients. J Natl Cancer Inst 76: 793, 1986 |
|
Cain EN, Kohorn EI, Quinlan DM et al: Psychosocial benefits of a cancer support group. Cancer 57: 183, 1986 |