Surgical Correction of Uterovaginal Anomalies
Authors
INTRODUCTION
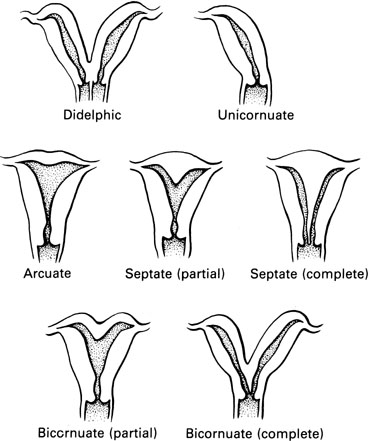
It is useful to classify uterovaginal anomalies into four groups (Table 1). Agenesis of the uterus and the vagina, so-called Mayer-Rokitansky-Küster-Hauser syndrome, represents a dysplasia of the müllerian ducts such that the uterus and the middle and upper thirds of the vagina are absent. Disorders of vertical fusion represent faults in the junction between the down-growing müllerian ducts and the up-growing derivative of the urogenital sinus. Disorders of lateral fusion result from failure of lateral fusion of the two müllerian ducts. Vaginal obstruction that occurs as a result of disorders of lateral fusion is invariably unilateral. Furthermore, unilateral obstruction is accompanied by absence of the ipsilateral kidney. Bilateral obstruction, therefore, would be associated with bilateral kidney agenesis and subsequent nonviability of the developing embryo.
Table 1. Uterovaginal anomalies
Class I | Dysgenesis of the müllerian ducts |
Class II | Disorders of vertical fusion of the müllerian ducts |
Transverse vaginal septum | |
| Obstructed | |
| Unobstructed | |
| Cervical agenesis or dysgenesis | |
Class III | Disorders of lateral fusion of the müllerian ducts |
| Asymmetric-obstructed disorder of uterus or vagina usually associated with ipsilateral renal agenesis | |
| Unicornuate uterus with a noncommunicating rudimentary anlagen or horn | |
| Unilateral obstruction of a cavity of a double uterus | |
| Unilateral vaginal obstruction associated with double uterus | |
| Symmetric-nonobstructed | |
| Didelphic uterus | |
| Complete longitudinal vaginal septum | |
| Partial longitudinal vaginal septum | |
| No longitudinal vaginal septum | |
| Septate uterus | |
| Complete | |
| Complete longitudinal vaginal septum | |
| Partial longitudinal vaginal septum | |
| No longitudinal vaginal septum | |
| Partial | |
| Complete longitudinal vaginal septum | |
| Partial longitudinal vaginal septum | |
| No longitudinal vaginal septum | |
| Bicornuate uterus | |
| Complete | |
| Complete longitudinal vaginal septum | |
| Partial longitudinal vaginal septum | |
| No longitudinal vaginal septum | |
| Partial | |
| Complete longitudinal vaginal septum | |
| Partial longitudinal vaginal septum | |
| No longitudinal vaginal septum | |
T-shaped uterine cavity (DES drug-related) | |
Unicornuate uterus | |
| With a rudimentary horn | |
| With communicating endometrial cavity | |
| Without endometrial cavity | |
| Without rudimentary horn | |
Class IV | Unusual configurations of vertical and lateral fusion defects |
DES, diethylstilbestrol.
Modified from the American Fertility Society classification of müllerian anomalies. Fertil Steril 49(6), 1988; and Rock JA: Surgery for anomalies of the müllerian ducts. In Thompson JD, Rock JA (eds): Telinde’s Operative Gynecology, p 688. Philadelphia, JB Lippincott, 1997
When complete vaginal obstruction occurs, urgent attention is necessary to prevent deterioration of reproductive capacity. This may result from retrograde tubal reflux and subsequent development of endometriosis. Attention to a nonobstructive malformation is seldom an emergency, but a complete evaluation may be necessary to determine whether these müllerian disorders are responsible for a poor reproductive history (Table 2).
Table 2. Obstructive and nonobstructive vaginal anomalies
Obstructive
Unilateral obstructing vaginal septum with ipsilateral renal agenesis
Obstruction of a unilateral rudimentary horn
Complete transverse vaginal septum
Imperforate hymen
Atresia of the uterine cervix and vagina
Nonobstructive
Symmetric double uterus with or without vaginal duplication
Incomplete transverse vaginal septum
This review outlines the management of patients with uterovaginal anomalies and discusses subsequent success as measured by pregnancy outcome or normal sexual function.
CONGENITAL ABSENCE OF THE MÜLLERIAN DUCTS
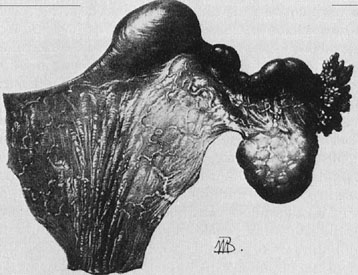
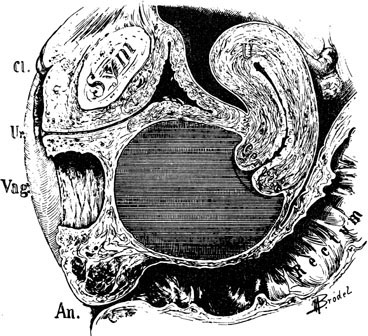
A patient with congenital absence of the müllerian ducts lacks a vagina and uterus. Aplasia or dysplasia of the müllerian ducts is a more accurate term because usually a normal lower vagina has developed, with absence of the middle and upper thirds of the vagina and the uterus. Although the uterus is not present, rudimentary uterine anlagen are often palpable. The tube and ovary are essentially normal (Fig. 1). Although some reported series of congenital absence of the vagina have included patients with the uterus and with absence of various lengths of vagina, these are not generally considered under the heading Rokitansky’s syndrome. There is a homogeneity and specificity of this syndrome (i.e., the absence of a vagina and uterus with a high incidence of urinary tract anomalies), whereas patients with partial agenesis of the vagina or a transverse vaginal septum are considered in the section on disorders of vertical fusion. Patients with disorders of vertical fusion have a low incidence of urinary tract anomalies.
Diagnosis
The growth and development of patients with congenital absence of the müllerian ducts is normal, including secondary sex characteristics. The external genitalia are normal in appearance; however, the vagina is absent or there may be only a shallow vaginal pouch of 3 cm in depth. Prior to menarche, the differential diagnosis between an imperforate hymen and Rokitansky’s syndrome may be difficult. Diagnosis may depend on the ability to determine the presence or absence of a uterus. This often may be accomplished by examining the patient under anesthesia. Laparoscopy is rarely necessary. On occasion, however, these patients may have persistent pelvic pain associated with an active rudimentary uterine anlagen. The patient usually complains of cyclic abdominal pain, and at the time of surgery, retention of a small amount of blood is noted in the rudimentary structure.1 On occasion, functioning endometrium may result in considerable unilateral enlargement of the uterine anlage. In either case, excision of the rudimentary horn results in complete relief of symptoms.
The exact nature of the underlying genetic abnormality responsible for Rokitansky’s syndrome is unknown. However, it is possible to deduce the embryologic abnormality most likely to result in defects of the vagina, kidney, and skeleton. The müllerian duct has two components, a cephalic or anterior portion, which is derived from coelomic epithelium, and a caudal end, which is derived from a wolffian or mesonephric duct. The cephalic end of the müllerian duct is the anlagen of the fallopian tubes, and the caudal portions fuse to form the uterus and attach to the urogenital sinus to induce the development of the vagina. As previously noted, these patients have normal ovaries and fallopian tubes, only solid rudimentary uterine horns, and no vagina. This is a precise lesion that would be predicted if the initial phase of müllerian duct development were normal but subsequent development along the wolffian duct was impaired. Thus, the uterine and vaginal segments of the müllerian duct are dysplastic or absent. The urinary tract abnormalities frequently associated with this disorder (i.e., renal absence or ectopia) and the defects of the müllerian duct could result from a common abnormality of the wolffian duct. Skeleton is also derived from the mesoderm at approximately the same stage of development that has been identified for the defect in the urogenital tract (10–11 mm). The vertebrae develop from adjacent mesodermal cell concentrations at the same somite level as those of the mesonephros. Thus, a defect in the organization of the mesoderm in these somites could cause skeletal abnormalities, as well as defective mesonephric development and subsequent abnormalities in the kidney, uterus, and vagina.
Major defects in the urinary tract system, such as congenital unilateral absence of a kidney or the presence of a pelvic kidney, occur in approximately 15% of patients with congenital absence of the müllerian ducts. However, if trivial anomalies are included, such as malrotation of the kidney, a partial double collection system on one side, or malposition of the kidney, the percentage of abnormalities rises to approximately 40%. Thus, an intravenous pyelogram should be part of a routine evaluation of all patients with congenital absence of the uterus and vagina.
Anomalies of the bony structures are also common. It is generally believed that these anomalies occur in about 5% of patients. Abnormalities in the lumbar spine, such as sacralization of L5 or the presence of six lumbar vertebrae, are observed. Fusion of the cervical vertebrae can also occur. Of interest is the Klippel-Feil syndrome (congenital fusion of the cervical spine, short neck, low posterior hairline), which has been reported in a few patients with congenital absence of the vagina. A variety of other abnormalities, including abnormalities of the hand, inguinal and femoral hernias, and congenital heart lesions, have also been described.
The karyotype of affected patients is characteristically 46,XX. Jones and Mermut recorded five examples of sisters with this disorder.2 This raised the possibility of a very rare autosomal recessive disorder; however, evidence of this has not been persuasive. Familial aggregates seem best explained on the basis of polygenic or multifactorial inheritance. Single mutant genes are responsible for the McKusick-Kaufman syndrome and the hand-foot-genital syndrome.3 Hand-foot-genital syndrome is a rare, dominantly inherited condition that affects both the distal limbs and the genitourinary tract. A nonsense mutation of the HOXA13 gene has been identified in several families.4, 5 At present, it is not clear whether sporadic cases represent new mutations of a single gene defect that is responsible for the familial disorder or are due instead to multifactorial etiologies.
Clinical management
PSYCHOLOGICAL COUNSELING
Various therapies have been suggested for the treatment of patients with Rokitansky’s syndrome. There is, however, complete agreement that all patients should initially have family counseling instituted by the attending physician or a psychologist. Interaction with other affected young women may also be helpful. Weijenborg and terKuile described the use of a group program for women with Rokitansky’s syndrome. They demonstrated that patients felt less anxious, less depressed, and less sensitive to interpersonal contact after participation in the program.6 Continuity of care should be maintained throughout the hospitalization or dilation program, often continuing several years after initial therapy. Most surgeons agree that the single most important factor in determining the success of vaginoplasty is the psychosocial adjustment of the patient to her anomaly; thus, an intensive-counseling program should definitely be addressed before considering any therapeutic modality.
VAGINOPLASTY
In our institution, three major forms of therapy have been used: the split-thickness graft technique of McIndoe, the Frank technique of vaginal dilation, and the Ingram passive dilation technique. It has been our practice to suggest vaginal dilation as an initial form of therapy. This is quite useful in the motivated patient in whom previous vaginal exploration has not been attempted.7
The Ingram technique for passive dilation has several advantages.8 The patient is not required to press the dilator against the vaginal pouch. A series of graduated lucite dilators (Fig. 2) slowly and evenly dilate the neovaginal space. The patient should be carefully instructed in the use of dilators, as recommended by Ingram, beginning with the smallest dilator. The patient is shown and instructed with the use of a mirror how to place a dilator against the introital dimple. The dilator may be held in place with a supportive undergarment and regular clothing worn over this.
The patient is shown how to sit on a racing-type bicycle seat, which is placed on a stool 24 inches above the floor. She is instructed to sit leaning slightly forward with the dilator in place for at least 2 hours per day at intervals of 15–30 minutes. The patient is followed usually at monthly intervals and can be expected to graduate to the next larger size dilator approximately every month. An attempt at sexual intercourse may be suggested after the use of the largest dilator for 1 or 2 months. Continued dilation is recommended if intercourse is infrequent. In our experience, functional success rates are excellent. Our group recently reported the largest series of vaginal agenesis patients who utilized the Ingram method of dilation to create a neovagina.9 The records of 51 patients with müllerian agenesis were reviewed: 37 patients attempted vaginal dilation and 14 young women underwent a surgical intervention. Functional success was described as satisfactorily achieving intercourse or accepting the largest dilator without discomfort during the clinic visit. All patients were followed for at least 2 years and for an average of 9.25 years. Functional success was achieved in 91.9% of those who attempted dilation (Table 3). Robson and Oliver also reported excellent success with dilation using the Frank method, with only one patient of 25 with müllerian agenesis undergoing surgical management.10 All those women who were sexually active reported satisfactory sexual relations. Thus, passive dilation should be suggested as an initial therapy for vaginal creation. If unsuccessful, operative vaginoplasty is indicated.
Table 3. Dilation success and failure rates
Group | Total (n) | % |
Successful dilation | 34/37 | 91.9 |
Failed dilation | 3/37 | 8.1 |
Adapted from Roberts CP, Haber MJ, Rock JA: Vaginal creation for müllerian agenesis. Am J Obstet Gynecol 185:1349, 2001
Over the past 25 years, the senior author has had experience with the split-thickness graft technique described by McIndoe to line the neovaginal space. Indications for this procedure include failure of vaginal dilation or patient preference.
The Counsellor-Flor modification of the McIndoe technique uses a rolled foam rubber mold covered with a condom in place of a rigid form.11, 12 The initial and important step in this operation is to obtain a satisfactory split-thickness graft. This can be easily obtained with the Padgett electrodermatome, which secures a graft 0.017–0.018 inch thick. The graft is also 10 cm wide and long enough to be twice the vaginal depth. The graft is taken from the buttock to render the donor site invisible, that is, within the lower limits of most bathing suits. Either buttock may be used.
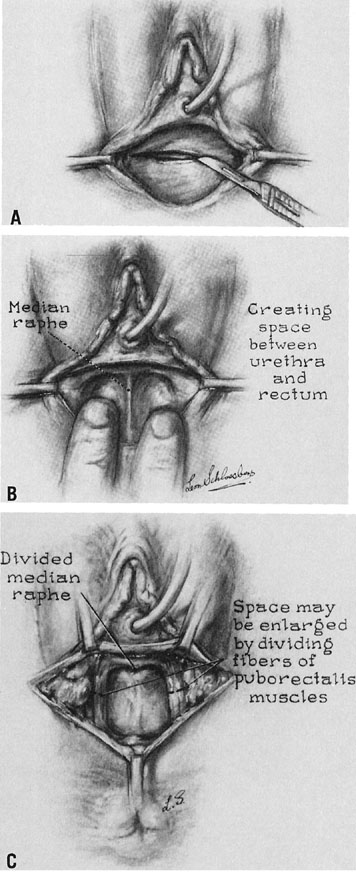
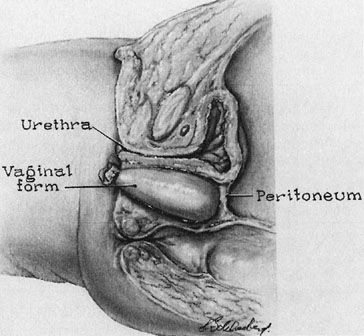
A transverse incision is made at the site of the vaginal orifice. The neovaginal space is developed with digital manipulation. The dissection is easily developed on one or both sides of the median raphe, where the tissue between the bladder and the rectum seems to be condensed almost enough to warrant a designation of a rectourethralis ligament (Fig. 3). The raphe is cut. This dissection should be carried to the peritoneum. However, care must be exercised not to expose too large an area of peritoneum lest an enterocele develop after the procedure.
A prosthesis is then developed using foam rubber such as may be obtained from an upholstery shop. This is readily sterilized in blocks about 10 × 10 × 20 cm. The foam rubber may be cut to a suitable size using scissors. The form should be cut about twice the desired size and further compressed by covering with two rubber sheaths, which are ordinary condoms. Once compressed, the form is placed into the neovagina and allowed to assume the shape of the space. The end of the condom is then tied so that the proper amount of air is trapped within the foam rubber to conform to the vaginal space. The skin graft is sewn to the form with interrupted, vertical mattress sutures of fine, nonreactive suture so that the exteriorized undersurface of the graft is approximated to exteriorize the undersurface of the sutured edges (Fig. 4).
After the graft has been inserted into the cavity, the edges of the graft may be sutured to the cut edges of the vaginal epithelium (Fig. 5). Only a few sutures should be placed, however, to allow drainage of any serum that may collect under the graft. Rather, large braided sutures may be placed through the labia to prevent the extrusion of the form. A suprapubic catheter is recommended to prevent urethral pressure. Postoperatively, the patient should be kept on a low-residue diet, given antibiotics, and kept at bed rest. The labial sutures may be removed in approximately 4 days, while the patient is at bed rest. Seven days after the procedure, the patient is again returned to the operating room; the suprapubic catheter is removed, and the vaginal cavity is irrigated. A second vaginal form may be molded so that the patient has two forms to use at home while the healing process is completed. Twenty-four hours after removal of the original mold, the patient is instructed in self-removal and the use of a low-pressure, clear, warm water douche. After approximately 6 weeks, the soft form is replaced with a Silastic form, which is inserted after it is moistened with tap water.
|
In our series, there have been no serious complications following vaginal creation with the split-thickness graft technique. There have been no fistulas. Abandonment of the balsa wood form, the use of a suprapubic catheter, and careful operative technique are believed to be responsible for the anatomic success, which approaches 90%, of this operative vaginoplasty. The artificial vagina is of sufficient depth and width to allow comfortable sexual relations. Minor complications have been noted in 21 of 79 (26%) patients in our series. These include a small vaginal hematoma with secondary infection (n = 2) and the presence of granulation tissue resulting from variable graft loss (n = 19). The graft take was 100% in 60 patients, 80–90% in 14 patients, 60–70% in three patients, and 50% in two patients.7
Several investigators have reported successful modifications of alternative methods of vaginoplasty for patients with vaginal agenesis. With the ease of laparoscopy, modifications of procedures previously performed by laparotomy have gained interest. The Vecchietti approach, using a pulley system to pull the vaginal apex superiorly, has been adapted and performed laparoscopically.13 In addition, the Davydov procedure, dissection of the rectovesical space and mobilization of the peritoneum to the introitus after a vaginal space has been created, has been adapted for use with the laparoscope.14 These modified techniques certainly offer some advantages owing to the acknowledged benefits of minimally invasive surgery, such as shorter hospital stays and less postoperative discomfort. The authors report excellent success over the initial follow-up period; however, it will be important to continue to gather appropriate long-term data regarding functional success.
Vaginal vault prolapse, after both nonsurgical and surgical treatment of müllerian agenesis, has been described. It is proposed that, owing to the lack of müllerian development and appropriate suspension to the bony pelvis, these women are at risk for vault prolapse. The incidence of prolapse in young women with this disorder is still unknown, regardless of the method of creation of the neovaginal space.15, 16, 17
In summary, the use of the Ingram method of dilation, coupled with careful instruction and counseling, may meet with good success. In fact, in our experience, the most common reason for failure with vaginal dilation has been an attempt at hymenectomy prior to referral. Thus, a high success rate may be expected in women without previous vaginal surgery. Additionally, dilation obviates the need for hospitalization and the inherent risks of surgery. However, vaginal creation using a split-thickness graft is highly successful in skilled hands and should be offered to those patients unwilling or unable to obtain an adequate neovagina with dilation (Table 4). Preoperative counseling is essential to the success of either procedure.
Table 4. Success following vaginal creation for müllerian agenesis
Procedure | Patients (n) | Functional Success | Anatomic Success |
Split-thickness graft | 80 | 80 (100%) | 73 (91%) |
Rock JA, Reeves LA, Retto H, et al: Success following vagi-nal creation of müllerian agenesis. Fertil Steril 39:809, 1983
DISORDERS OF VERTICAL FUSION
Obstructive
TRANSVERSE VAGINAL SEPTUM
A transverse vaginal septum may result from a failure of complete canalization of the primordial vaginal plate or from lack of union, which represents the junction of the urogenital sinus and the paramesonephric duct contribution. A high transverse vaginal septum probably occurs at about the junction of the upper fourth and the lower three-fourths of the vagina. The etiology may lie in a local abnormality of the vaginal mesoderm or in the lack of canalization of the epithelial vaginal plate. As judged by a pedigree study of an inbred Amish community, there is evidence that a complete transverse vaginal septum may be the result of a rare female sex–limited autosomal recessive transmission.2
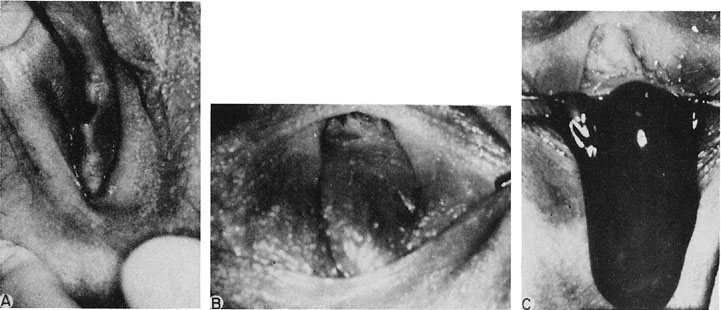
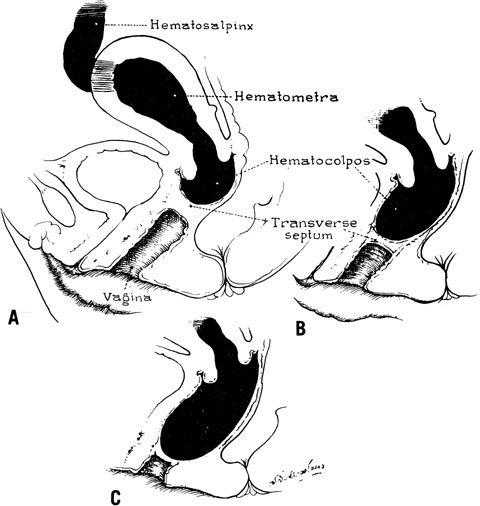
The site of obstruction may occur at any point along the vaginal canal, but it is most frequently at the junction between the middle and the upper thirds of the vagina (Fig. 6). The diagnosis of this condition in infancy is not easy unless a hydromucocolpos occurs. Often, an abdominal mass is noted and exploration performed. It is not unusual to observe serious urinary tract obstruction associated with the large mucocolpos. The diagnosis is difficult because there is no bulging of the outlet.
The onset of symptoms usually occurs after puberty, with the onset of menstruation. Pelvic pain and pressure are primarily due to the large hematocolpos (Fig. 7). The delay in diagnosis may be considerable, approaching 2–3 years.18 Symptoms are associated with obstruction to the outflow of menstrual blood.
In the adult, a transverse vaginal septum adjacent to the cervix is quite thick. In fact, a considerable segment of vagina may be underdeveloped, and if the segment is quite prolonged, so that it involves the lower portion of the vagina, the situation arises that is sometimes described in the literature as congenital absence of the vagina with a uterus present. This situation is considered to be an extreme form of the transverse vaginal septum.
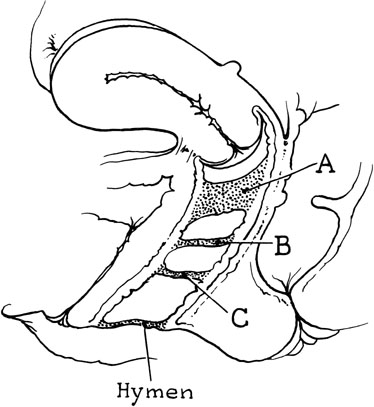
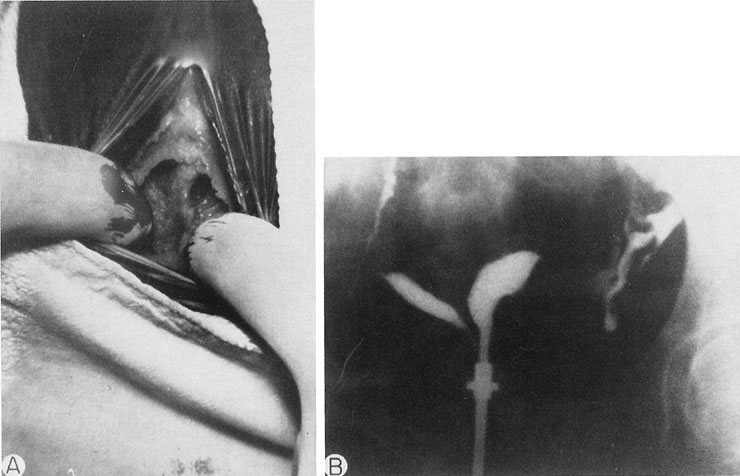
The persistent hymen should be mentioned, although it is not included as a disorder of vertical fusion. The hymen consists of a membrane separating the vaginated caudal end of the fused paramesonephric ducts from the evaginated urogenital sinus. The hymen may persist if centrally placed epithelial cells do not degenerate and produce the expected hymenal remnants. With accumulated menstrual blood, a bulge in the hymen is often recognized (Fig. 8). The hymen may be excised with a cruciate incision.
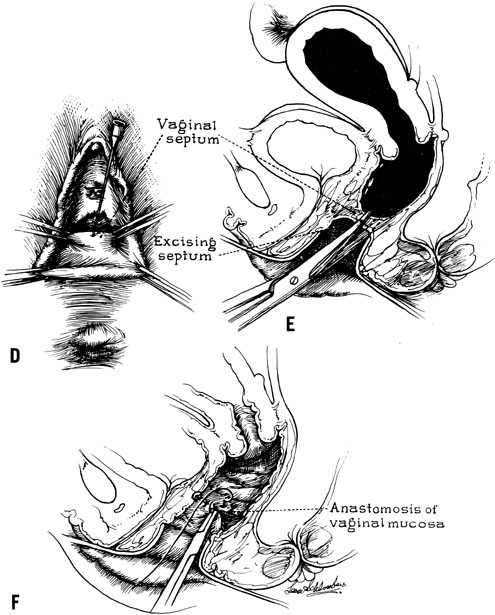
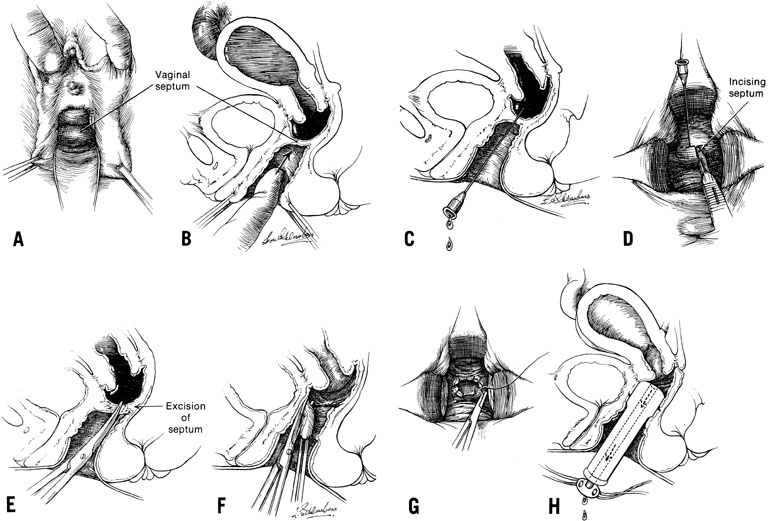
When symptoms dominated by severe cyclic pain require therapy for a complete obstructing transverse vaginal septum in the lower and middle thirds of the vagina, therapy should consist of removing the obstructing membrane and reanastomosing the upper and lower vaginal segments (Fig. 9).
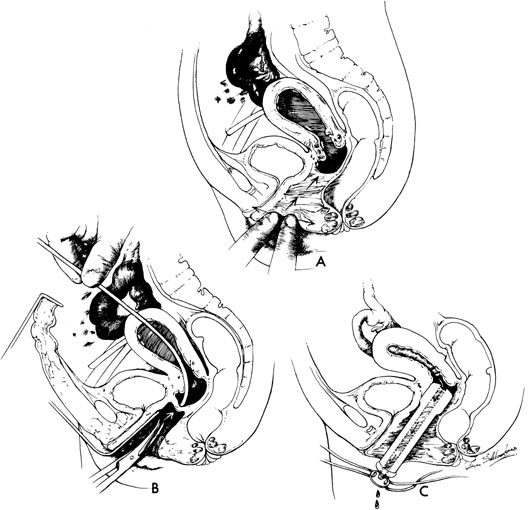
In some instances, however, the length of the obstructing transverse vaginal septum may be such that reanastomosing the upper and lower vagina is not possible. This is particularly true when one encounters a high transverse vaginal septum (Fig. 10). In this situation, a space is created between the rectum and the bladder so as to identify the obstructed upper vagina. It is important to distinguish the mass from the bladder anteriorly and the rectum posteriorly. This identification may be facilitated by the mass, which has resulted from accumulated menstrual blood. If one cannot be certain, however, exploratory laparotomy may be performed and a sound passed through the fundus of the uterus to tent the vaginal septum and allow the operator from below to incise and resect it safely (Fig. 11). In most instances, it is possible to identify the obstructing membrane, and the operator may then probe the mass with an aspirating needle to identify old menstrual blood. The upper vagina may then be opened and the septum excised. Because reanastomosis of the vagina cannot be performed, an indwelling lucite form is placed into the vagina. This form consists of a bulbous end and a channel through which menstruation may occur. This form may be held in place with a retaining harness. In most instances, the bulbous end of the form is retained in the upper vagina. This form should be left in place for 4–6 months. It is anticipated that epithelialization from the lower and upper vagina will have joined by proliferation of the epithelium. Following removal of the lucite form, vaginal dilation on a daily basis may be necessary for 2–4 months to prevent contracture.
Repair of a vaginal obstruction due to an imperforate hymen as compared with a complete transverse vaginal septum is associated with a higher subsequent pregnancy success rate (Table 5). Furthermore, pregnancy success is increased in patients with a complete transverse vaginal septum of the lower third of the vagina as compared with those with a high septum (Table 6). However, when making such a comparison between an imperforate hymen and a complete vaginal septum, one should keep in mind that the interval between the onset of symptoms and diagnosis and treatment is usually longer in patients with a complete transverse vaginal septum. It is attractive to assume that greater vaginal distention is permitted to accommodate trapped menstrual blood in patients with imperforate hymen than in patients with a high transverse vaginal septum. In fact, in patients with a high vaginal septum, only a small portion of vagina is present, and retrograde menstruation occurs early after the onset of menstruation and may predispose these patients to the development of endometriosis. Endometriosis could well explain the reduced fertility in these patients. Thus, prompt diagnosis and surgical correction to drain accumulated blood may preserve reproductive capacity by preventing this sequela.
Table 5. Pregnancy success following surgical correction of an intact hymen versus correction of a complete transverse vaginal septum
Hymen | Transverse Septum | Total | |
Patients (n) | 22 | 26 | 48 |
Adequate follow-up | 18 | 22 | 40 |
Patients attempting pregnancy | 15 | 19 | 34 |
Patients pregnant* | 13 (86%) | 9 (47%) | 22 (65%) |
Patients with a living child | 10 (66%) | 7 (36%) | 17 (50%) |
*p <0.05 (Χ analysis).
Adapted from Rock JA, Zacur HA, Dlugi AM et al: Pregnancy success following surgical correction of imperforate hymen and complete transverse vaginal septum. Obstet Gynecol 59:448, 1982
Table 6. Pregnancy success following surgical correction of a complete transverse vaginal septum (Johns Hopkins Hospital)
Site of Septum | ||||
Lower Third | Middle Third | Upper Third | Total | |
Patients (n) | 5 | 9 | 12 | 26 |
Adequate follow-up | 5 | 8 | 9 | 22 (84%) |
Patients attempting pregnancy | 4 | 7 | 8 | 19 (73%) |
Patients pregnant | 4 (100%) | 3 (43%) | 2 (25%) | 9 (47%) |
Adapted from Rock JA, Zacur HA, Dlugi AM, et al: Pregnancy success following surgical correction of imperforate hymen and complete transverse vaginal septum. Obstet Gynecol 59:448, 1982
CONGENITAL ABSENCE OF THE CERVIX
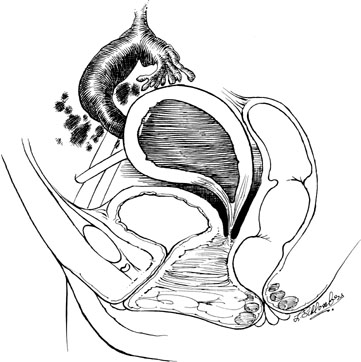
Rarely, a defect in development includes the cervix, which results in a functioning, but obstructed, uterine corpus and a defect in cervical and vaginal development. A large portion of the vagina may be absent (Fig. 12). Cervical agenesis involves a complete lack of any substantial cervical tissue, where the lower uterine segment narrows to terminate in a peritoneal sleeve at a point well above the normal communication with the vaginal apex. Cervical dysgenesis, in which varying portions of the cervical body develop, may also occur.
|
Until recently, preservation of reproductive potential in this situation has seldom achieved success. Although some have suggested that a fistulous opening be maintained between the endometrial cavity and the vagina using a splinting system, our experience (and that of others) has shown that attempts to create a fistulous tract between the vagina and the uterus have been unsuccessful.19, 20, 21, 22, 23 Repeated dilations have been necessary, and in every instance, both patient and surgeon have chosen to solve the problem by removal of the uterus. Therefore, hysterectomy has been the recommended primary therapy, especially in cases of cervical agenesis. Chakravarty and colleagues reported on 18 adolescents with complete or partial cervicovaginal atresia managed with a surgical cannulation technique.24 Patients were treated with amniotic membrane vaginoplasty and bisection of the partial cervical cord. The incised cervical edges served as an ectocervix and were affixed to the neovagina. A mold that allowed drainage was left in place for 6 months to 1 year, and a cervical stent was removed after 1 year. Complications included stenosis treated with repeated cervical dilation, cervical incision, and laparotomy. The authors noted that all patients have regular menses and two thirds have satisfactory vaginal length and distensibility. Two spontaneous pregnancies, with delivery of viable neonates, were also reported.
There have been several reports of pregnancy success after varied attempts at vaginoplasty and cervical stenting for partial cervical atresia.25, 26 Pregnancy success is generally associated with cervical reconstruction with a well-formed cervical body. Additionally, successful pregnancy and delivery by cesarean section have been reported after zygote intrafallopian transfer (ZIFT) in a patient with congenital cervical atresia.27 Spontaneous missed abortions have also been reported.28 It is essential to counsel patients about the potential necessity for removal of an abnormally developing pregnancy by laparoscopic surgery or laparotomy.
Nonobstructive
Occasionally, a transverse vaginal septum is not complete, so the accumulation of mucus or menstrual blood is not a factor. However, the incomplete transverse vaginal septum may cause dyspareunia or result in a complication that compromises reproduction. There may be a small opening in the transverse vaginal septum allowing the egress of blood. Other incomplete septa may cause no dyspareunia, and pregnancies have occurred so it is clear that sperm have traversed the opening from the partial transverse septum to the cervix.
DISORDERS OF LATERAL FUSION
Obstructive
OBSTRUCTED UTERINE HORN
Disorders of lateral fusion result from a failure of lateral fusion of two müllerian ducts. In addition, on one side, there is failure of the lumen to communicate with the outside. At times, an obstruction may involve an isolated horn of the uterus with minimal connection to the unobstructed side (Fig. 13). These patients usually present with signs and symptoms suggestive of an ectopic pregnancy. Early removal is desirable so that retrograde menstruation will not cause endometriosis and compromise subsequent reproduction. Fortunately, in some instances of a rudimentary horn with obstruction, there is also a failure of communication of the cavity of the uterus with the fallopian tube so there is no opportunity for spill of menstrual blood. The excision of these horns gives complete relief of symptoms.
DOUBLE UTERUS ASSOCIATED WITH AN OBSTRUCTED HEMIVAGINA AND IPSILATERAL RENAL AGENESIS
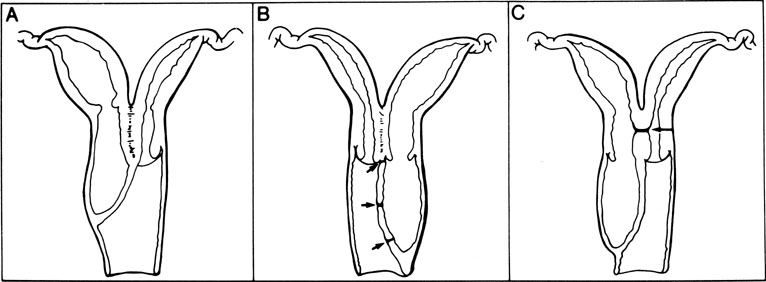
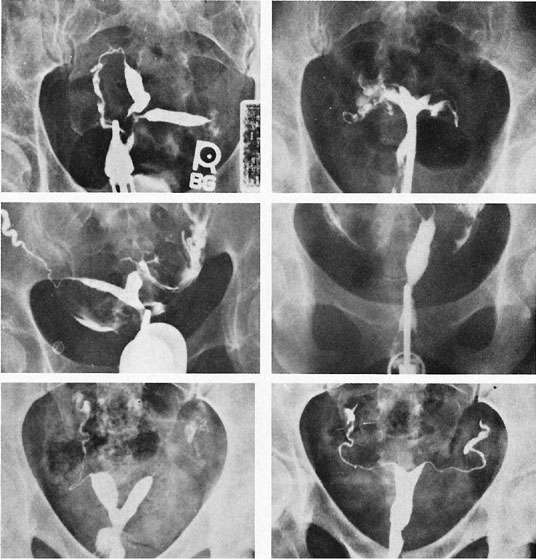
The unique clinical syndrome of double uterus, unilateral partial or complete obstruction of the vagina, and ipsilateral renal aplasia is rare (Fig. 14; see Fig. 13). The clinical presentation varies depending on the uterovaginal relationships, which may obscure the true diagnosis. Three groups of patients have emerged from a review of our own experience (Table 7).29 Group 1 consists of patients with a complete unilateral vaginal obstruction without uterine communication, which results in a perivaginal mass with severe dysmenorrhea, lower abdominal pain, and regular menses. Group 2 consists of patients with incomplete unilateral vaginal obstruction without uterine communication. These patients have lower abdominal pain, severe dysmenorrhea, excessive foul mucopurulent discharge, and intermenstrual bleeding. Group 3 patients have complete vaginal obstruction with a laterally communicating double uterus. They have a perivaginal mass, lower abdominal pain, dysmenorrhea, and regular menses. Irregular menstrual bleeding usually confuses the physician such that unless she or he thinks of the possibility of the diagnosis, the diagnosis may be overlooked. A hysterogram, intravenous pyelogram, and careful pelvic examination often allow the diagnosis to be made. When there is complete unilateral obstruction, a large amount of blood may accumulate, and this condition may go unrecognized for a number of years after the onset of menstruation. This is due primarily to a distensible vagina that can accommodate to the increments of blood resulting from each menstrual cycle. Absorption of enough menstrual blood occurs between periods so that succeeding flow may add to the accumulated blood without pain. Nevertheless, once retrograde menstruation occurs, endometriosis invariably results. Thus, if the septum may be removed before the tubes and ovaries are compromised by infection or endometriosis, reproduction may be consistent with uterine duplication without obstruction.
Table 7. Patients with a double uterus associated with unilateral vaginal obstruction and ipsilateral renal aplasia, treated with excision of the obstructing vaginal septum (Johns Hopkins Hospital, 1950–1980)
Anatomic Findings | Patient | Age | Signs and Symptoms | Subsequent Pregnancy |
Group 1 | BS | 16 | Perivaginal mass, severe dysmenorrhea, lower abdominal pain, regular menses | Term pregnancy |
CC | 21 | Not desired | ||
MS | 13 | Undetermined | ||
WW | 20 | Infertile | ||
JF | 17 | Term pregnancy | ||
ML | 21 | Term pregnancy | ||
Group 2 | PM | 17 | Lower abdominal pain, severe dysmenorrhea, excessive foul mucopurulent discharge, intermenstrual bleeding | Not desired |
CM | 19 | Term pregnancy | ||
LH | 14 | Not desired | ||
RP | 21 | Term pregnancy | ||
Group 3 | AC | 24 | Perivaginal mass, lower abdominal pain, dysmenorrhea, regular menses | Term pregnancy |
EC | 14 | Not desired |
Adapted from Rock JA, Jones HW Jr: The double uterus associated with an obstructed hemivagina and ipsilateral renal agenesis. Am J Obstet Gynecol 138:339, 1980
Rarely, a situation exists in which a low vaginal obstruction is associated with a lateral communication between the two horns of the uterus (see Fig. 14C). Patients may complain of a disappearing mass at the vaginal outlet. A careful hysterogram under fluoroscopy is often useful in identifying the communication.
The treatment for a unilateral vaginal obstruction is the careful excision of this vaginal septum. In most instances, there is no indication for abdominal exploration. No treatment is indicated for lateral communication of the uterine horns. Reproductive performance is consistent with a double uterus unless delay in diagnosis and treatment has destroyed tubal function or resulted in significant endometriosis. Haddad and coworkers retrospectively reviewed the management of 42 patients with a blind hemivagina.30 All patients underwent laparoscopy to clarify the uterine malformation. Surgical treatment consisted of excision of the vaginal septum in 37 (88%) patients. Only one procedure was necessary in 92% of cases. Examination at 3-month assessment was satisfactory in 93% of patients. Patients also completed reproductive questionnaires an average of 6.5 years after the procedure. Nine women who had undergone vaginal septum excision experienced 20 pregnancies after the surgical procedure.
The epithelium of an obstructed vagina is usually composed of cuboid cells of müllerian origin. If the obstruction is incomplete, the cuboidal epithelium is replaced with the squamous epithelium of the vagina. Thus, when the vaginal septum is removed in the obstructed circumstances, the newly opened vagina is lined with adenomatous epithelium, that is, the patient has vaginal adenosis. The adenosis epithelium is slowly replaced by a metaplastic process, which may require 2–3 years. In most instances, adenomatous epithelium results in an increased mucous discharge.
Nonobstructive
DIDELPHIC AND UNICORNUATE UTERUS
Unobstructed failure of lateral fusion involving both the uterus (didelphic) and the vagina is usually without symptoms; nevertheless, the narrowness of the vagina may result in significant dyspareunia when coitus is attempted (Fig. 15). This may require the removal of the vaginal septum. The vaginal septum is often thick. Complete resection of the septum is not necessary because the tissue often retracts and is not visible as a ridge. Generous pedicles should be taken such that hemostasis is ensured, avoiding troublesome bleeding. Reproduction is modestly compromised in patients with didelphic uteri. Unfortunately, information is anecdotal and consists of case reports or small series recording examples of primary infertility, pregnancy wastage, and premature labor. There is no indication for surgical intervention in a didelphic condition except for removal of a vaginal septum that might cause dyspareunia.
|
Reproduction in a unicornuate uterus is not different from that of the didelphic uterus. The didelphic uterus is but a symmetrically inversed duplication of a unicornuate uterus. Small series have noted a compromised reproductive history, including infertility, pregnancy wastage, and premature labor. However, most pregnancies appear to result in a live-born infant. Cerclage has been suggested in some instances of repeated miscarriage and premature labor. Clarification of pregnancy success associated with the unicornuate and didelphic uteri awaits a large comprehensive study of carefully documented histories of patients with these uterine anomalies.
BICORNUATE AND SEPTATE UTERUS
A symmetric, nonobstructed double uterus may be associated with reproductive failure (Fig. 16). Generally, there is no problem in achieving pregnancy, but difficulty arises from repeated pregnancy loss. This may be first trimester or second trimester loss. In the event the patient carries the pregnancy to term, obstetric malpresentation or difficulties in delivery are not unusual. Primary infertility in a patient with a symmetric double uterus is sometimes observed, but the etiologic relationship between infertility and the anomaly is an unresolved problem.
|
It is necessary to distinguish between the bicornuate and the septate uterus because the bicornuate uterus is infrequently associated with reproductive difficulty, whereas the septate uterus is frequently associated with reproductive failure. The distinction between these two types of uteri cannot be made by examination of a hysterogram: the image of the cavities may be exactly the same. With the bicornuate uterus, two distinct horns are observed; with a septate uterus, the exterior configuration of the uterus is normal.
Although the diagnosis of müllerian anomalies traditionally has been made with the use of hysterosalpingogram, sonogram, diagnostic laparoscopy, and occasionally, laparotomy, magnetic resonance imaging (MRI) has been shown to be useful in characterizing and delineating more clearly the exact nature of the müllerian defect.31
There have been no studies attempting to clarify the reasons why some double uteri cause reproductive loss whereas others provide a series of normal pregnancies. It has been suggested that increasing intrauterine pressure with relative cervical incompetence may be responsible for premature delivery, whereas habitual abortion may be the result of a poor blood supply to the endometrium through the septum.32 It has also been suggested that the luteal defect associated with a double uterus may be a result of the local vascular insufficiency and not a hormonal deficiency. The etiology of reproductive failure in patients with a double uterus awaits further study by histologic techniques of various parts of the endometrium with hormonal correlation and the use of blood-flow studies. Until this is done, identification of the double uterus that causes difficulty may be made only after the fact by clinical trial.
A small percentage of patients with a symmetric double uterus have abnormalities of the urinary tract; 9% of patients with a double uterus have been noted to have congenital absence of a single kidney.33 As a rule, the more severe the uterine anomaly, the more likely an anomaly of the urinary tract. Urologic evaluation is recommended for all patients with a double uterus.
As only one in five patients with a double uterus will have reproductive loss as a result of the uterine anomaly, a complete evaluation for habitual abortion should be performed.33 This should include an endometrial biopsy and karyotypic analysis. Further studies, as indicated by history, should include thyroid-stimulating hormone, thyroid profile, mycoplasma culture, and antibody titers for toxoplasmosis, herpes, and cytomegalic inclusion disease. Uterine unification as treatment for repeated pregnancy loss should be attempted only after a complete and thorough evaluation. The history of the miscarriage is particularly significant. A characteristic story is that of an early midtrimester loss associated with a minilabor starting with cramps and followed by bleeding. In miscarriages that occur in the first trimester, when there is a history of lack of recognition of an embryo, it is necessary to suspect that the cause is other than the anatomic defect of the uterus. Nevertheless, histories that differ from the typical one described previously are sometimes encountered.
When a correctable endocrine or metabolic cause of reproductive loss is identified, it should be corrected before any surgical procedure is considered. When the double uterus is the only factor associated with repeated pregnancy loss, surgical correction may be performed.
Several authors have presented concerns with reference to the type of metroplasty for a particular uterine anomaly. Technical simplicity, vascularity of the incision, trauma to the remaining myometrium, soundness of the uterine scar, and interference of subsequent uterine activity have been stressed. The suggestion that little or no myometrial tissue be removed with the repair has also been emphasized.
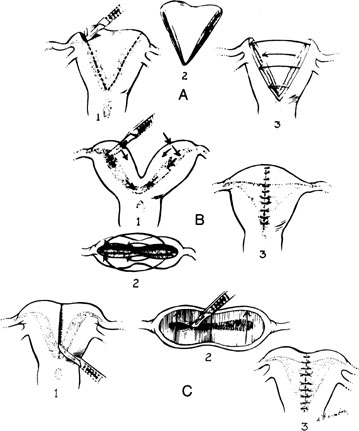
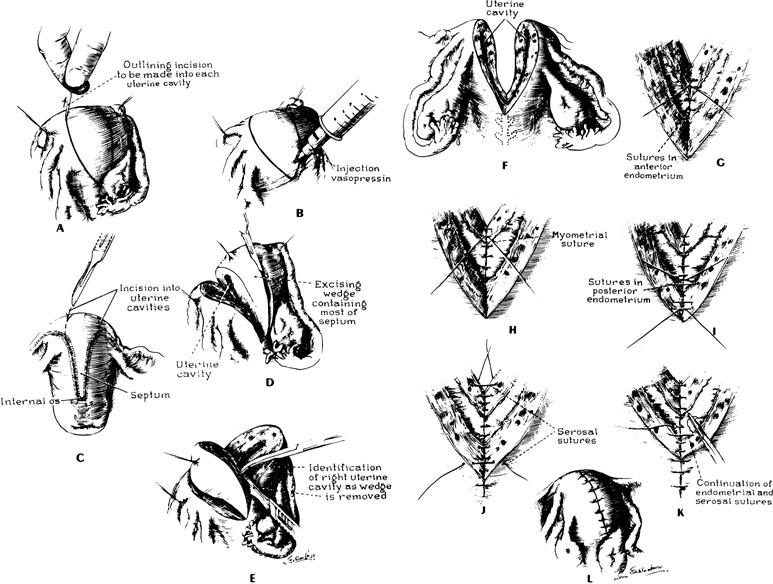
There are three principal types of abdominal procedures (Fig. 17).34, 35, 36 Modifications of these procedures are numerous, each claiming some advantage. Our own experience has been exclusively with excision of the septum by wedge. Excessive blood loss should not present a problem with any unification procedure with the injection of a dilute solution (20 units in 20 mL saline solution) of vasopressin (Pitressin) into the myometrium. The wedge technique invariably results in a small reduction in the size of the uterine cavity. However, this does not appear to interfere with subsequent pregnancy success.
Several points should be emphasized in performing a wedge metroplasty (Fig. 18):
- When the incisions in the fundus are approaching the endometrial cavity, care must be taken that the cavity is not transected.
- After the removal of the wedge, the uterus should be closed in three layers of interrupted sutures; the first layer of sutures should be placed such that the knots are tied within the uterine cavity.
- The uterine serosa should be approximated with fine suture to minimize the incidence of adhesions to the fundus of the uterus.
At the conclusion, the uterus appears rather normal in its configuration, but the striking feature will be the proximity of the insertion of the fallopian tubes; in placing the final sutures, it is important that the sutures not obstruct the interstitial portions of the tube. The size of the uterine cavity after metroplasty seems unimportant (Fig. 19). At times, the reconstructed cavity may be quite small compared with a normal uterus; however, of more importance is the symmetry of the uterus. The postoperative hysterogram usually reveals small “dog-ears,” which are leftover tags from the original bifid condition of the uterus.
Duplication of the cervix occurs only rarely. It is important not to attempt to unify the cervix. This may cause an incompetent cervix. Surgical reconstruction needs to be applied only to the corpus.
The healing of the nonpregnant uterus after abdominal metroplasty does not compare with healing after cesarean section because there is a lack of involution. In our series, all patients after abdominal metroplasty were delivered by cesarean section. This may be recommended as a matter of precaution because most patients have experienced a long, disappointing obstetric history and are unwilling to accept the possibility of uterine rupture during labor. To allow the uterine incision to heal properly, a 4–6-month delay in becoming pregnant after surgical reconstruction should be advised.
Pregnancy success following abdominal wedge metroplasty has now approached 81% (Table 8). Success rates following transcervical septal incision are comparable. Individualization of surgical repair of various uterine malformations may have some merit. Although abdominal metroplasty is usually reserved for the very broad uterine septum, hysteroscopic metroplasty has largely replaced abdominal metroplasty.
Table 8. Reproductive performance of patients with double uterus: before and after wedge metroplasty (Johns Hopkins Hospital, 1936–1983)
Before | After | |
With adequate follow-up (n) | 63 | 59 |
Pregnant (n) | 63 (100%) | 57 (96%) |
With living children (n) | 4 (6%) | 48 (81%) |
Total pregnancies | 167 | 78 |
| Term | 0 | 60 (77%) |
| Premature | 9 (5%) | 4 (5%) |
| Abortion | 158 (95%) | 14 (18%) |
| Living children | 4 (2%) | 58 (75%) |
An alternative to the Jones or Tompkins approach to abdominal metroplasty for surgical correction of the septate uterus is the transcervical hysteroscopic procedure for lysis of the uterine septum. Many reports, including those of Israel and March, have shown that the septate uterus may be successfully corrected using a hysteroscopic approach with postoperative term pregnancy results equal to, or slightly higher than, the 80% achieved with traditional transabdominal approaches to metroplasty.37 This technique avoids the need for abdominal and myometrial incisions and thus reduces the risks of postoperative morbidity and pain. Moreover, the necessity of a cesarean section is also avoided.
Although abdominal approaches to metroplasty might be advised for extremely wide uterine septums, transcervical lysis can be accomplished in most cases; however, a second attempt might be necessary in certain instances to completely incise the septum transcervically.
Surgery is performed in the follicular phase of the cycle, or patients are preoperatively treated with a gonadotropin-releasing hormone analog, to eliminate the possibility of endometrium diminishing clarity of view during surgery. Preoperative antibiotics are often empirically given, despite the lack of good evidence to support it. Hyskon is used as the uterine distending medium, and a simultaneous laparoscopy is helpful to exclude the diagnosis of a bicornuate uterus and to help prevent a uterine perforation. The laparoscopist can benefit from observation with his or her light turned off. The uterine septum may be incised with scissors or with the elecrocautery loupe of the resectoscope (Fig. 20).
An intrauterine device is not placed after the procedure. After the procedure, uterine bleeding may be controlled using a Foley catheter to tamponade the cavity for 4–6 hours, if necessary. The patient can be discharged home by 4 hours after the procedure and continued on antibiotics for 5 days. Hormonal therapy is often prescribed after the procedure to promote rapid epithelialization and decrease the risk of intrauterine adhesions. Attempts at pregnancy should be postponed for 2 months after surgery because postoperative hysteroscopy with biopsy has shown the uterine cavity to be normal at 8 weeks after surgery.38
In patients in whom the uterine septum is complete from the fundus to the cervix, this procedure is also safe and efficacious. It may be completed after a Foley catheter is placed into one cavity while the other cavity is distended with Hyskon through the hysteroscope. Incision of the septum should begin above the level of the internal cervical os and then be continued superiorly until completely incised, except for the lower portion of the septum inferior to the internal cervical os.39 The cervical portion of the septum is preserved. In 1999, Rock and associates published one of the largest series of patients managed with this approach.40 The hysteroscopic approach to lysis of the uterine septum allows the surgeon to cut the septum just as in the Tompkins procedure without the need for myometrial and abdominal incisions.
REFERENCES
Rock JA, Baramki TA, Parmley TH, et al: A unilateral functioning uterine anlage with müllerian duct agenesis. Int J Gynaecol Obstet 18:99, 1980 |
|
Jones HW Jr, Mermut S: Familial occurrence of congenital absence of the vagina. Am J Obstet Gynecol 114:1100, 1972 |
|
McKusick VA, Weiboecher RG, Gragg GW: Recessive inheritance of a congenital malformation syndrome. JAMA 204:113, 1968 |
|
Simpson JL: Genetics of the female reproductive ducts. Am J Med Genet (Semin Med Genet) 89:224, 1999 |
|
Goodman FR, Bacchelli C, Brady AF, et al: Novel HOXA13 mutations and the phenotypic spectrum of hand-foot-genital syndrome. Am J Med Genet 67:197, 2000 |
|
Weijenborg PTM, terKuile MM: The effect of a group programme on women with the Mayer-Rokitansky-Küster-Hauser syndrome. Br J Obstet Gynaecol 107:365, 2000 |
|
Rock JA, Reeves LA, Retto H, et al: Success following vaginal creation for müllerian agenesis. Fertil Steril 39:809, 1983 |
|
Ingram JM: The bicycle seat stool in the treatment of vaginal agenesis and stenosis: A preliminary report. Am J Obstet Gynecol 140:867, 1981 |
|
Roberts CP, Haber MJ, Rock JA: Vaginal creation for müllerian agenesis. Am J Obstet Gynecol 185:1349, 2001 |
|
Robson S, Oliver GD: Management of vaginal agenesis: Review of 10 years’ practice at a tertiary referral centre. Aust N Z Obstet Gynaecol 40:430, 2000 |
|
Counsellor VS, Flor FS: Congenital absence of the vagina. Surg Clin North Am 37:1107, 1957 |
|
McIndoe A: Treatment of congenital absence and obliterative conditions of the vagina. Br J Plast Surg 2:254, 1950 |
|
Veronikis DK, McClure GB, Nichols DH: The Vecchietti operation for constructing a neovagina: Indications, instrumentation, and techniques. Obstet Gynecol 90:301, 1997 |
|
Templeman CL, Hertweck SP, Levine RL, et al: Uses of laparoscopically mobilized peritoneum in the creation of a neovagina. Fertil Steril 74:589, 2000 |
|
Ramaswamy S: Müllerian agenesis with vaginal prolapse. Case report Br J Obstet Gynaecol 93:640, 1986 |
|
Peters WA, Uhlir JK: Prolapse of a neovagina created through self-dilatation. Obstet Gynecol 76:904, 1990 |
|
Schaffer J, Fabricant C, Carr BR: Vaginal vault prolapse after nonsurgical and surgical treatment of müllerian agenesis. Obstet Gynecol 99:947, 2002 |
|
Rock JA, Zacur HA, Dlugi AM, et al: Pregnancy success following surgical correction of imperforate hymen and complete transverse vaginal septum. Obstet Gynecol 59:448, 1982 |
|
Farber M, Marchant DJ: Reconstructive surgery for congenital atresia of the uterine cervix. Fertil Steril 27:1277, 1976 |
|
Baker ER, Horger EO, Williamson HO: Congenital atresia of the uterine cervix. J Reprod Med 27:39, 1982 |
|
Geary LW, Weed JC: Congenital atresia of the uterine cervix. Obstet Gynecol 42:213, 1973 |
|
Dillon WP, Mudaliar NA, Wingate MB: Congenital atresia of the cervix. Obstet Gynecol 54:126, 1979 |
|
Niver DH, Barrette G, Jewelewicz R: Congenital atresia of the uterine cervix and vagina: Three cases. Fertil Steril 33:25, 1980 |
|
Chakravarty B, Hiralal K, Chowdhury NNR: Pregnancies after reconstructive surgery for congenital cervicovaginal atresia. Am J Obstet Gynecol 183:421, 2000 |
|
Hampton HL, Meeks GR, Bates GW, et al: Pregnancy after successful vaginoplasty and cervical stenting for partial atresia of the cervix. Obstet Gynecol 76:900, 1990 |
|
Anttila L, Penttla TA, Suikkari AM: Successful pregnancy after in-vitro fertilization and transmyometrial embryo transfer in a patient with congenital atresia of the cervix. Hum Reprod 14:1647, 1999 |
|
Thijssen RFA, Hollanders JMG, Willemsen WNP, et al: Successful pregnancy after ZIFT in a patient with congenital cervical atresia. Obstet Gynecol 76:902, 1990 |
|
Suganuma N, Furuhashi M, Moriwaki T, et al: Management of missed abortion in a patient with congenital cervical atresia. Fertil Steril 77:1071, 2002 |
|
Rock JA, Jones HW Jr: The double uterus associated with an obstructed hemivagina and ipsilateral renal agenesis. Am J Obstet Gynecol 138:339, 1980 |
|
Haddad B, Barranger E, Paniel BJ: Blind hemivagina: Long-term follow-up and reproductive performance in 42 cases. Hum Reprod 14:1962, 1999 |
|
Carrington BM, Hricak H, Nuruddin RN, et al: Müllerian duct anomalies: Magnetic resonance imaging evaluation. Radiology 176:715, 1990 |
|
White MM: Uteroplasty in infertility. Proc R Soc Med 53:1006, 1960 |
|
Rock JA, Jones HW Jr: The clinical management of the double uterus. Fertil Steril 28:798, 1977 |
|
Jones HW Jr, Jones GES: Double uterus as an etiological factor of repeated abortion: Indication for surgical repair. Am J Obstet Gynecol 65:325, 1953 |
|
Strassman EO: Operations for double uterus and endometrial atresia. Clin Obstet Gynecol 4:240, 1961 |
|
Tompkins P: Comments on the bicornuate uterus and twinning. Surg Clin North Am 42:1049, 1962 |
|
Israel R, March CM: Hysteroscopic incision of the septate uterus. Am J Obstet Gynecol 149:66, 1984 |
|
Candiani GB, Vercellini P, Fedele L, et al: Repair of the uterine cavity after hysteroscopic septal incision. Fertil Steril 54:991, 1990 |
|
Rock JA: Surgery for anomalies of the müllerian ducts. In Thompson JD, Rock JA (eds): TeLinde’s Operative Gynecology. p 603, Philadelphia, JB Lippincott, 1992 |
|
Rock JA, Roberts CP, Hesla JS: Hysteroscopic metroplasty of the class Va uterus with preservation of the cervical septum. Fertil Steril 72:942, 1999 |