Umbilical Cord Prolapse and Other Cord Emergencies
Authors
INTRODUCTION
The umbilical cord is a vital intra-amniotic structure that occasionally develops catastrophic complications. Although these events are rare, understanding of risk factors, presentation, and management options can assist in positive outcomes for fetus and mother. The human fetal umbilical cord is derived from embryonic mesodermal layers and yolk sac and can be readily identified as early as the third week after conception. The cord is formed by the union of the body stalk and the omphalomesenteric duct.1 Within the cord are three fetal vessels: normally two arteries and one vein. Initially, there is a second (right) umbilical vein, which undergoes atrophy early in fetal life, at approximately 8 weeks. The umbilical arteries derive from the ventral branches of the paired embryonic dorsal aortas.2 Intraluminal pressures differ between the arteries and vein, producing differing effects during compression. Umbilical vein pressure is between 20 and 35 mmHg, and arterial, approximately 55 mmHg. Arterial compression produces bradycardia, increased arterial pressure, and fetal gasping. When only the umbilical vein is occluded, there is decreased arterial pressure, decreased femoral and carotid blood flow, but no fetal gasping.3 The physiologic herniation of the embryonic midgut into the cord occurs between 6 and 12 weeks, allowing for the rapid growth of the liver. The cord is covered by the amnionic epithelium and, further from the fetus, transforms from a stratified columnar to a simple columnar epithelium.4 The umbilical vessels are sensitive to both physical and chemical stimuli, including cold, touch, oxygen perfusion, serotonin, angiotensin, and oxytocin.5, 6 Wharton's jelly is the connective tissue of the cord that surrounds and supports the vessels. It is composed of a ground substance of open-chain polysaccharides within a fine network of microfibrils and collagen, and contains mast cells and fibroblasts.7
The average length of the umbilical cord is approximately 55 cm (range 32–146 cm).2 The cord exhibits helical twists or coils. Over 80% of cords spiral counterclockwise and coiling can be noted as early as the ninth week of gestation, numbering about 40 turns at term. The coiling of the umbilical cord is thought to enhance the cord's ability to withstand external forces such as tension, compression, stretching, and entanglement and also enhances flexibility. The tensile strength of the cord is directly proportional to the birth weight of the baby by approximately 2.5 times.8 Umbilical cords without coils appear in about 5% of fetuses, and this seems to put the fetus at increased risk for adverse outcomes such as stillbirth, fetal growth retardation, oligohydramnios, meconium-stained amniotic fluid, low Apgar scores, and intrapartum fetal distress.9
UMBILICAL CORD PROLAPSE
Prolapse of the umbilical cord is a true obstetric emergency, and understanding of its predisposing factors can assist in speedy diagnosis and treatment. Although cord compression occurs routinely in normal labors and is commonly seen during fetal heart rate monitoring,identified by variable declerations, severe cord occlusion can result in dangerous alterations in fetal and placental circulation. Historically, the incidence of cord prolapse has been reported as approximately 1 in 300 deliveries. More recently, however, the reported incidence has fallen to about 0.2%, or 1 in 500 of all deliveries, mainly as a result of changing obstetric management, including antenatal ultrasound diagnosis of funic presentation or vasa previa as well as almost universal use of continuous electronic fetal monitoring.10
Morbidity and mortality associated with prolapse of the cord also have declined due to improvements in obstetric management as well as in neonatal care. Mortality from umbilical cord prolapse has fallen from 375 per 1000 to between 36 and 162 per 1000 live births in the last 100 years.11 Interestingly, despite ominous fetal heart rate tracings, abnormal fetal blood gas readings, and low Apgar scores, most of the mortality is attributable to congenital anomalies and prematurity.10
Cord prolapse may be frank or occult. Overt or frank cord prolapse is the most common, and is defined as the cord positioned ahead of the fetal presenting part. This is invariably diagnosed by palpation or visible exam after rupture of the membranes with the cord noted beyond the cervical os and into the vagina or beyond. In contrast, occult cord prolapse is defined as the position of the cord alongside but not past the presenting part. Frequently it is encountered intrapartum, when fetal heart rate abnormalities lead to palpation of loops of cord around or ahead of the presenting part, often with still intact membranes. A large series reported that the cord was found free in the vagina in 45%, at the introitus in 11%, and presenting in 4% of the cases.12
Predisposing factors
Though obstetric intervention has received much of the blame in cord prolapse, the majority of events occur due to nonpreventable risk factors. Umbilical cord prolapse mostly is seen in the clinical setting of a poorly engaged or unengaged presenting part, such as with malpresentation, prematurity, multifetal pregnancy, multiparity, hydramnios, and uterine anomalies (Table 1). However, most cases of cord prolapse are reported with singleton cephalic presentation in 50–63% of the cases.13 Yet, proportionally, breech and transverse presentations have higher risk of prolapse per case. Of breech fetuses, footling breech confers the highest risk due to inadequate filling of the pelvic inlet, compared to frank or complete breech which are usually more fully engaged. The frequency of cord prolapse in vertex presentation was 0.24%; breech 3.5% and transverse 9.6% in a review by Barclay.14 The majority of events occur in cephalic presentations due to the infrequency of nonvertex presentations.
The premature fetus has a higher incidence of cord prolapse events due to its smaller volume filling the pelvis, as well as the relative increase in the amount of fluid surrounding it.
There has been concern over the role of obstetric maneuvers in promoting prolapse of the cord. Intrapartum interventions and procedures such as amniotomy, amnioinfusion, cervical ripening, and labor induction have been thought to increase risks of umbilical cord prolapse. Amniotomy especially has received blame for cord prolapse. In a study by Roberts and colleagues, no increased risk was identified between artificial and spontaneous rupture of the membranes in their series, nor a difference in cervical dilation at the time of diagnosis of prolapse.15
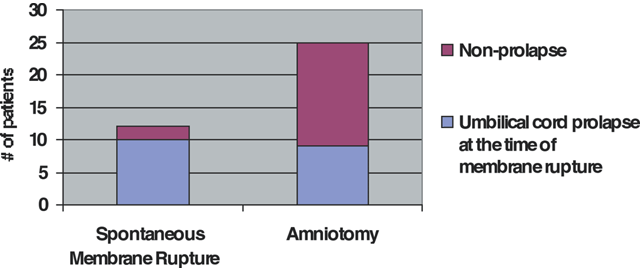
Figure 1 illustrates the proximity of ruptured membranes to cord prolapse, with 83% of diagnoses made after spontaneous rupture versus 36% of the artificially ruptured group. In the group with artificial rupture, two-thirds of the patients with cord prolapse had abnormal fetal heart tracings, which prompted intervention for applying an internal fetal electrode or performing amnioinfusion to treat cord compression. In artificial rupture of membranes, the mechanism of action thought to be responsible for cord prolapse is disengagement of the fetal presenting part followed by the sudden release of fluid that propels the cord past the presenting part. However, study of this clinical scenario has not confirmed this theory. In addition, those diagnosed with prolapse had a significantly higher station and little descent of the presenting part during labor. Often, amniotomy was performed for evaluation of existing intrapartum fetal heart rate abnormalities, implying a preexisting occult cord prolapse. Other investigators have not been able to confirm risk associated with other maneuvers, including application of fetal scalp electrode, insertion of intrauterine pressure catheters, and external cephalic version as well as application of forceps vacuum or manual rotation. Labor induction, amnioinfusion and cervical ripening treatments also have not been shown to increase the incidence of cord prolapse.
Table 1. Factors associated with cord prolapse in 79 patients
Factor | No. | % |
Abnormal presentation | 40 | 50.6 |
Prematurity <2500 g or <37 weeks | 30 | 38.0 |
Obstetric manipulation | 15 | 19.0 |
Multiparity (<5 pregnancies) | 10 | 12.7 |
Multiple gestation | 8 | 10.1 |
Placental problems (abruption, abnormal implantation, bleeding) | 6 | 7.6 |
(Levy H, Meier PR, Makowski EL: Umbilical cord prolapse. Obstet Gynecol 64:499, 1984.)
Polyhydramnios or excessive amniotic fluid (index above 25, or vertical pocket of 8 cm) has been shown to increase the risk of umbilical cord prolapse. Many patients with excessive fluid have high station at labor onset. In a retrospective study of more than 60,000 singleton deliveries at term, Maymon and colleagues noted an almost 10-fold increase in the risk of cord prolapse (2.2% vs. 0.3%) in patients with hydramnios.16
Multifetal pregnancies have an increased risk of cord prolapse. When attempting vaginal delivery of twin pregnancy, even in vertex–vertex presentation, there is a higher proportion of cord prolapse with the second twin after successful vaginal delivery of the first twin, often requiring cesarean delivery. In a study of 106 sets of vertex–vertex twins, Sullivan and associates noted a 35% incidence of cord prolapse in the second twin among those who required cesarean delivery of the second twin.17 Abnormal cord insertion, placenta previa, and low-lying placenta also are risk factors for prolapse of the cord.
Antepartum diagnosis and management
Funic presentation before rupture of membranes is a predisposing factor to umbilical code prolapse. This is visualized on ultrasound in approximately 1 in 167 (0.6%) live births.18 The increased use of antepartum obstetric ultrasound, especially with color flow Doppler, has assisted in making the diagnosis of a presenting cord and preparing for cesarean delivery before overt or occult prolapse. Figure 2 illustrates the usefulness of color flow Doppler in this diagnosis. Every patient with fetal malpresentation, multiple gestation, prematurity or known abnormal placentation should undergo ultrasound examination before labor to exclude the possibility of funic presentation. Ultrasound also can be helpful for the preterm fetus in assessing the contents of hour-glassing or prolapsing membranes.
During labor, the diagnosis of cord prolapse must be in the differential diagnosis whenever fetal heart rate abnormalities are noted, such as an episode of prolonged bradycardia or recurrent variable decelerations. If risk factors for cord prolapse are present, gentle vaginal examination should be performed, with the assistance of confirming bedside ultrasound. Once the diagnosis has been confirmed, the cord should be manipulated as little as possible because the vessels of the cord may spasm and further compromise blood flow. Cervical dilation, effacement, and station of the presenting part should be ascertained. Because of the possibility of spasm, controversy exists as to whether an attempt at replacement of the cord should be done.
Management of cord prolapse requires prompt delivery of the fetus. If the cervix is completely dilated and rapid vaginal delivery is feasible, then an effort at replacement is not harmful, during which simultaneous preparations for abdominal delivery are made. The head is gently disengaged and elevated while replacing the cord back into the pelvis. With continuous fetal monitoring, the success or failure of this maneuver will rapidly be evident. In a small series, the maneuver was successful in 5 of 8 patients, with no morbidity.19 In a setting where immediate cesarean delivery is not readily available, attempt at cord replacement is appropriate.
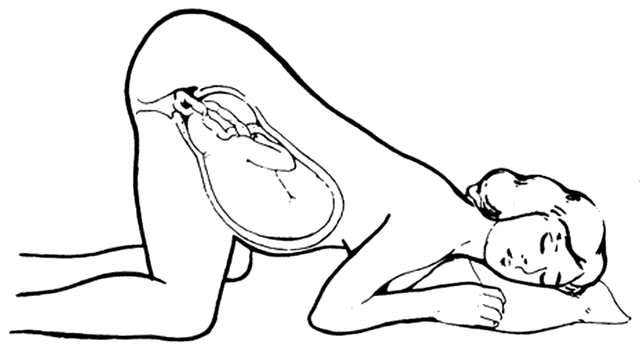
Several maneuvers are available to attempt to relieve compression on the cord. The patient should be immediately repositioned in knee-chest position (Fig. 3), which reduces the pressure of the presenting part on the cord. If the patient is unable to assume the knee-chest position because of epidural or body habitus, or for patient comfort during prolonged repositioning while awaiting cesarean delivery, steep Trendelenburg position also may be used. Oxygen should be administered to the mother. In addition, the examiner's hand should remain in the vagina to manually elevate the presenting part, if possible. Another technique described by numerous authors is that of filling the mother's urinary bladder with 500–1000 mL of fluid to assist in elevating the presenting part while preparations for delivery are made. This may be combined with the maneuvers previously described. If the cord has prolapsed beyond the introitus, it should be wrapped in sponges or a sterile towel soaked with warm saline or replaced back into the vagina and held there. Another useful therapy is to decrease force and frequency of contractions of labor using subcutaneous terbutaline. Immediate delivery is the treatment of choice, providing the fetus still is viable.
|
Depending on the setting and the availability of anesthesia and surgical facilities, vaginal delivery may be the most rapid route of delivery using forceps or vacuum, given the appropriate cervical dilation and station. Breech extraction of a second twin is still an option, despite a prolapsing cord. However, other presentations, such as breech, face, transverse, shoulder, and other unstable presentations, should be delivered abdominally. In a large British series of cord prolapses, 71% of 132 patients were delivered by cesarean section, with a perinatal mortality of 91 of 1000.10 Low cord pH (less than 7.10) and low Apgar scores were not predictive of intact outcome. Only one baby did not survive as a direct result of cord prolapse. In this study, time to delivery was not a critical factor; however, Praboulous and Phillipson, in their study of 65 cases of cord prolapse of 26,000 deliveries, found that infants in the group having a shorter time to delivery (mean 11 minutes) had a worse outcome, including all cases of asphyxia, suggesting a pre-existing problem such as anomaly or prematurity and the severity of the cord compression.20
In addition, time spent prior to delivery on in utero fetal resuscitation measures may help improve cord pH and Apgar scores. The presence of a limp, pulseless cord does not necessarily indicate fetal death. Auscultation and ultrasound assessment of fetal cardiac activity also should be performed. Driscoll and colleagues report patients having pulseless cord but also the presence of cardiac activity by ultrasound, with subsequent delivery of liveborn infants who did well.21 After the delivery, whether vaginal or abdominal, analysis of cord blood gases should be performed, as well as pathologic evaluation of the placenta.
Included in the differential diagnosis of fetal heart tracing abnormalities is abruptio placentae and umbilical cord prolapse. Often, emergent abdominal delivery is done without knowing which entity is present. According to Johnson and Richards, umbilical cord acid-base values at delivery can be highly predictive.22 The data suggest that careful analysis of umbilical arterial and venous blood gases may elucidate the cause of fetal acidosis. The study noted acidosis in arterial pH for both abruptio and cord prolapse of 6.87 and 7.05, respectively. There was, however, a marked difference in umbilical venous pH values, which also were acidotic, with a mean of 6.98 in the abruptio group and a near-normal value at 7.28 in the cord prolapse group.22
Acidosis in the newborn usually results from either of two different clinical situations: reduced maternal oxygen delivery to the placenta, or reduced fetal placental blood flow. In abruptio placentae—as well as maternal hypotension, hypoxemia, or increased arterial resistance, as in uterine hyperstimulation—there is poor oxygen delivery to the placenta and fetus, giving rise to low pH in both umbilical arterial and venous gases. In contrast, umbilical cord prolapse, as well as in other conditions of reduced fetal cardiac output, leads to reduced fetal perfusion of the placenta, large differences in arterial and venous oxygen saturation and pH, and a totally different mechanism for fetal acidosis.
There is little information on long-term outcomes of deliveries complicated by umbilical cord prolapse. In a series of 132 case followed by Murphy, only 1 of 120 survivors suffered any developmental deficit.23
VELAMENTOUS CORD INSERTION AND VASA PREVIA
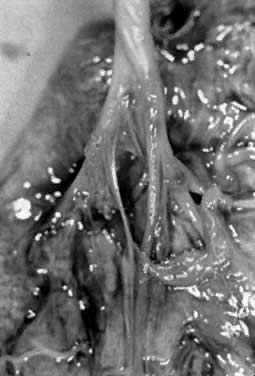
Other abnormalities of the umbilical cord can complicate labor and delivery. The umbilical cord normally inserts near the center of the placenta. In approximately 1% of placentas, the cord insertion is on the membranes, known as velamentous insertion.1 The umbilical vessels course over the membranes separately, without the protection of Wharton's jelly and thus are more vulnerable to trauma, including rupture of membranous vessels and vasa previa (Fig. 4). Vasa previa, occurring in 1 of 200024 to 5000 deliveries, is a variation of a funic presentation, although it carries possibly more serious risks, since the unprotected umbilical vessels precede the presenting part. Spontaneous rupture of the vessels can occur, or iatrogenic laceration can occur during artificial rupture of membranes or other obstetric manipulation, including cervical ripening, insertion of monitoring devices, or scalp gas sampling. If the vessels are lacerated, rapid fetal exsanguination can occur if delivery is not accomplished immediately. Mortality rate of 70–100% is expected with ruptured vasa previa.25
Vasa previa is suspected if severe fetal heart decelerations occur immediately after a vaginal examination, since the vessels spasm in response. Sinusoidal fetal heart rate patterns also are associated with compression of the umbilical vessels in vasa previa. When vaginal bleeding is associated with rupture of the membranes, vasa previa must be suspected, especially if fetal heart rate decelerations occur out of proportion to the amount of bleeding. If brisk vaginal bleeding ensues with fetal bradycardia, emergent delivery is indicated. Theoretically, Kleihauer–Betke or the Apt test can be used to determine if the bleeding is from fetal or maternal source, although in reality, if laceration of fetal umbilical vessels has occurred, there is little time to waste, since total fetal blood volume is only 300–500 mL.
Ultrasound has been used to assess umbilical cord insertion, especially in pregnancies that have a high risk of abnormal insertions. Velomentously inserted cords are associated with twins and two-vessel cord as well as marginal, bilobed, and low-lying placentas.24 Ultrasound evaluation of the umbilical cord throughout its length is best done between 24 and 27 weeks. During this period, it is possible to characterize the cord in over 90% of patients, including the insertion.26 Vasa previa is thought to evolve from an early placenta previa, or low-lying placenta. As the uterus grows, the placenta “recedes” from the cervical os while the membranes, with the vessels attached, remain at the os until delivery.27 Ultrasound assessment before labor should be considered in any patient with a history of early placenta previa or low-lying or marginal placenta to exclude vasa previa. Three-dimensional ultrasound also has been shown to clearly identify velamentous insertion and vasa previa. Patients in whom vasa previa is known or suspected can be monitored closely, educated, and delivered electively by cesarean section before the onset of labor. There is no current consensus on the timing of delivery for patients with a diagnosis of vasa previa. Most experts agree that a course of betamethasone between 32 and 34 weeks is practical and elective delivery between 34 and 37 weeks without confirmation of fetal lung maturity should be performed.28, 29
REFERENCES
Benirschke K, Kaufman P: The umbilical cord and major fetal vessels. In: Pathology of the Human Placenta, 2nd edn. New York, Springer-Verlag, 1990: 182 |
|
Moore KL: The Developing Human: Clinically Oriented Embryology, 5th edn. Philadelphia, WB Saunders, 1995: 124 |
|
Veth AFL, van Bemmel JH: The role of the placental vascular bed in the fetal response to cord occlusion. In Longo LD, Reneau DD (eds): Fetal and Newborn Cardiovascular Physiology. New York, Garland Publishing, 1978: 579–614 |
|
Sinha AA: Ultrastructure of human amnio and amniotic plaques of normal pregnancy. Zellforsch 122: 1, 1971 |
|
Winters RH: Unique vascular relationship in human umbilical cord. Nature 226: 656, 1970 |
|
Le Donne AT, Mc Gowan L: Effect of an oxytocic on umbilical cord venous pressure. Obstet Gynecol 30: 103, 1967 |
|
Yamada K, Shimizu S: Concanavalin A-peroxidase-diaminobenzidine (Con A-PO-DAB)-alcian blue (AB): A reliable method for dual staining of complex carbohydrates. Histochemistry 47: 159, 1976 |
|
Ghosh KG, Ghosh SN, Gupta AB: Tensile properties of human umbilical cord. Indian J Med Res 79: 538, 1984 |
|
Strong TH, Elliot JP, Radin TG: Noncoiled umbilical blood vessels: A new marker for the fetus at risk. Obstet Gynecol 81: 409, 1993 |
|
Murphy DJ, MacKenzie IZ: The mortality and morbidity associated with umbilical cord prolapse. Br J Obstet Gynaecol 102: 826, 1995 |
|
Panter, KR, Hannah, ME. Umbilical cord prolapse: so far so good? Lancet 347: 74, 1996 |
|
Yla-Outinen A, Heinonen PK, Tuimala R: Predisposing and risk factors of umbilical cord prolapse. Acta Obstet Gynecol Scand 64: 567, 1985 |
|
Clark DO, Copeland W, Ullery JC: Prolapse of the umbilical cord: A study of 117 cases. Am J Obstet Gynecol 101: 84, 1968 |
|
Barclay, M. Umbilical cord prolapse and other cord accidents. In: Sciarra JJ (ed). Gynecology and Obstetrics. JB Lippincott, Philadelphia, PA 1989: 1. |
|
Roberts WE, Martin RW, Roach HH: Are obstetric interventions such as cervical ripening, induction of labor, amnioinfusion, or amniotomy associated with umbilical cord prolapse? Am J Obstet Gynecol 176:1181; discussion 1183, 1997 |
|
Maymon E, Ghezzi F, Shotham-Vard I et al: Isolated hydramnios at term gestation and the occurrence of peripartum complications. Eur J Obstet Gynecol Reprod Biol 77: 157, 1998 |
|
Sullivan CA, Hawkins D, Seago DP et al: Cesarean delivery of the second twin in vertex vertex presentation: Operative indications and predictability. South Med J 91: 155, 1998 |
|
Lange IR: Determine cord position in malpresentation. Contemp Obstet Gynecol 29: 112, 1987 |
|
Barrat, JM. Funic reduction for the management of umbilical cord prolapse. Am J Obstet Gynecol, 1991; 165-654. |
|
Praboulos AM, Philipson EH: Umbilical cord prolapse: Is time to delivery critical? J Reprod Med 43: 129, 1998 |
|
Driscoll JA, Sadan O, Van Gelderan CJ et al: Cord prolapse: Can we save more babies? Case reports. Br J Obstet Gynaecol 94: 594, 1987 |
|
Johnson JWC, Richards DS: The etiology of fetal acidosis as determined by umbilical cord acid base studies. Am J Obstet Gynecol 177: 274, 1997 |
|
Murphy, DJ, McKenzie, IZ. The mortality and morbidity associated with umbilical cord prolapse. Br J Obstet Gynaecol 1995; 102:826 |
|
Quek SP, Tan KL: Vasa praevia. Aust NZ J Obstet Gynaecol 12: 206, 1972 |
|
Pent D: Vasa previa. Am J Obstet Gynecol 134: 151, 1979 |
|
Hata T, Aoki S, Hata K et al: Three dimensional ultrasonographic assessment of the umbilical cord during the 2nd and 3rd trimesters of pregnancy. Gynecol Obstet Invest 45: 159, 1998 |
|
Lee W, Lee VL, Kirk JS: Vasa previa: Prenatal diagnosis, natural evolution, and clinical outcome. Obstet Gynecol 95: 572, 2000 |
|
Society of Maternal-Fetal (SMFM) Publications Committee, Sinkey RG, Odibo AO, Dashe JS. #37: Diagnosis and management of vasa previa. Am J Obstet Gynecol 2015; 213:615. |
|
Swank ML, Garite TJ, Maurel K, et al. Vasa previa: diagnosis and management. Am J Obstet Gynecol 2016; 215:223.e1. |