Diagnostic Ultrasound in the First Trimester of Pregnancy
Authors
INTRODUCTION
Today's obstetrician gynecologist is required to interpret and in many cases perform ultrasound scans in the first trimester. In fact, certification of residency programs in many countries requires documentation of adequate exposure to and training in the evaluation of first-trimester ultrasound. Failure to understand the limitations of diagnostic ultrasound or inadequate training of physicians in this technique can result in grave complications for the patient and liability for health-care providers.
The benefits of routine transvaginal ultrasound at 6–7 weeks from the LMP include:
- Correct dating of gestational age since 10–15% of women will have inaccurate assignment of gestational age of more than 1 week based on menstrual history.
- Identification of missed abortion, ectopic pregnancy, and C-section scar pregnancies.
- Early identification of multiple gestation and correct determination of chorionicity.
The standard of care for performing routine ultrasound examination at 6–7 weeks varies from country to country. The disadvantages of performing this examination routinely are related to cost, errors in diagnosing ectopic pregnancies that in fact are intrauterine, increased training requirements for providers, and potential biologic hazards to the fetus that are presently unknown.
The potential benefits of a subsequent ultrasound examination at 12–14 weeks from the LMP include:
- Early diagnosis of certain severe and lethal anomalies.
- Early diagnosis of major trisomies as part of nuchal screening.
- Early identification of C-section scar pregnancy.
Similarly the standard of care for performing routine ultrasound at 12–14 weeks' gestation from the LMP varies from country to country. The disadvantages are cost, decreased sensitivity for major anomalies compared to the 20 week exam, and marked increase in training requirements for providers. The role of the early anatomic survey, when free DNA screening for chromosomal anomalies is being utilized, remains to be determined.
Any health-care provider performing diagnostic ultrasound must understand the physics of diagnostic ultrasound and have thorough, supervised training. This includes, but is not limited to, power settings, basic orientation, and proper cleaning of ultrasound probes between uses. For United States readers, the website, aium.org, of the American Institute of Ultrasound in Medicine will be helpful for guidelines. Alternatively for international readers, the website, isuog.org, of The International Society of Ultrasound in Obstetrics and Gynecology should be searched for similar guidelines.
FIRST-TRIMESTER ULTRASOUND: NORMAL LANDMARKS
Intrauterine sacs embryonic structures
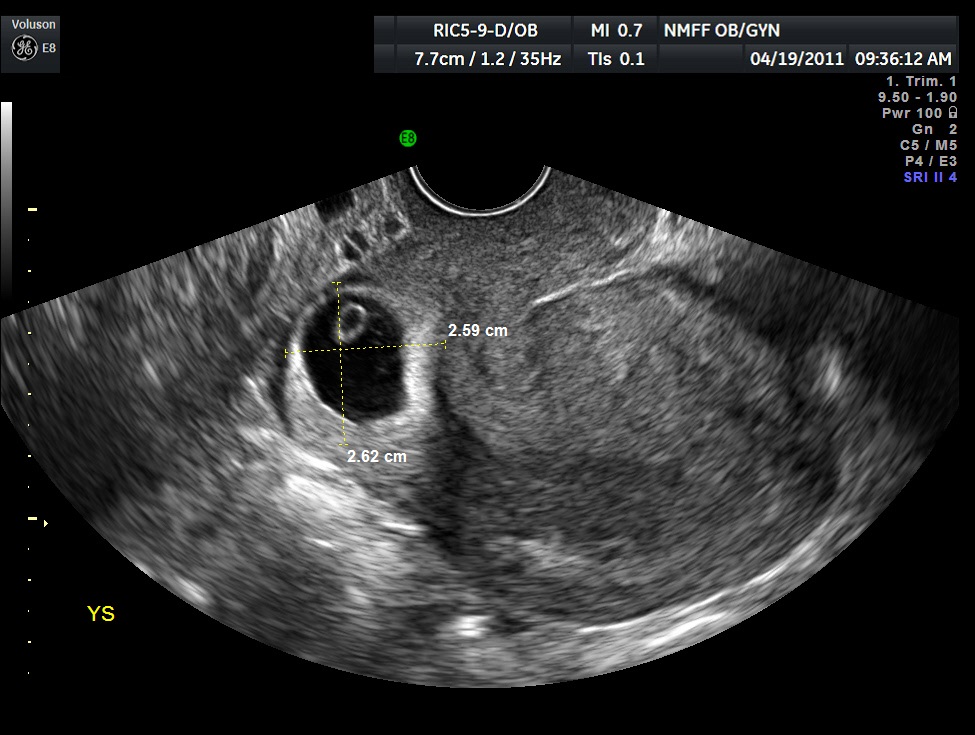
Warren and associates described the orderly appearance of gestational sac, yolk sac, and embryo with heartbeat at a given number of days from the onset of the last menstrual period (Table 1).1 With a transvaginal probe, a 2- to 3-mm gestational sac can usually be seen by 5 weeks from the last menstrual period (Fig 1A). A yolk sac is usually seen by 6 menstrual weeks, or by the time the mean diameter of the sac has reached 10 mm (Fig 1B and Fig 1C). A fetal pole with heart tones is typically seen by the completion of 7 menstrual weeks (Fig 1D). As shown by Fossum and colleagues, the appearance of these structures can be correlated with β-human chorionic gonadotropin (β-hCG) levels (Table 2).2 The literature regarding the correlation between quantitative β-hCG titers and early intrauterine gestational sacs and embryonic structures has been made somewhat confusing by the array of reference standards used to quantify β-hCG. Suffice it to say that the presently used Third International Standard and Fourth International Standard, used by most companies marketing β-hCG kits, corresponds roughly to the First International Reference Preparation. The practitioner must understand that these β-hCG levels are guidelines for the appearance of these structures and are not 100% reliable or accurate. This is discussed subsequently.
Table 1 The appearance of early gestational structures
Days from LMP | 28–35 | 35–42 | 42–49 | 49–56 |
Gestational sac | 100% | |||
Yolk sac | 0% | 91% | 100% | |
Embryo with + FHTs | 0% | 0% | 86% | 100% |
LMP, last menstrual period; +FHTs, positive fetal heart tones
(Warren WB, Timor-Trisch I, Peisner DB et al: Dating the early pregnancy by sequential appearance of embryonic structures. Am J Obstet Gynecol 161:747, 1989)
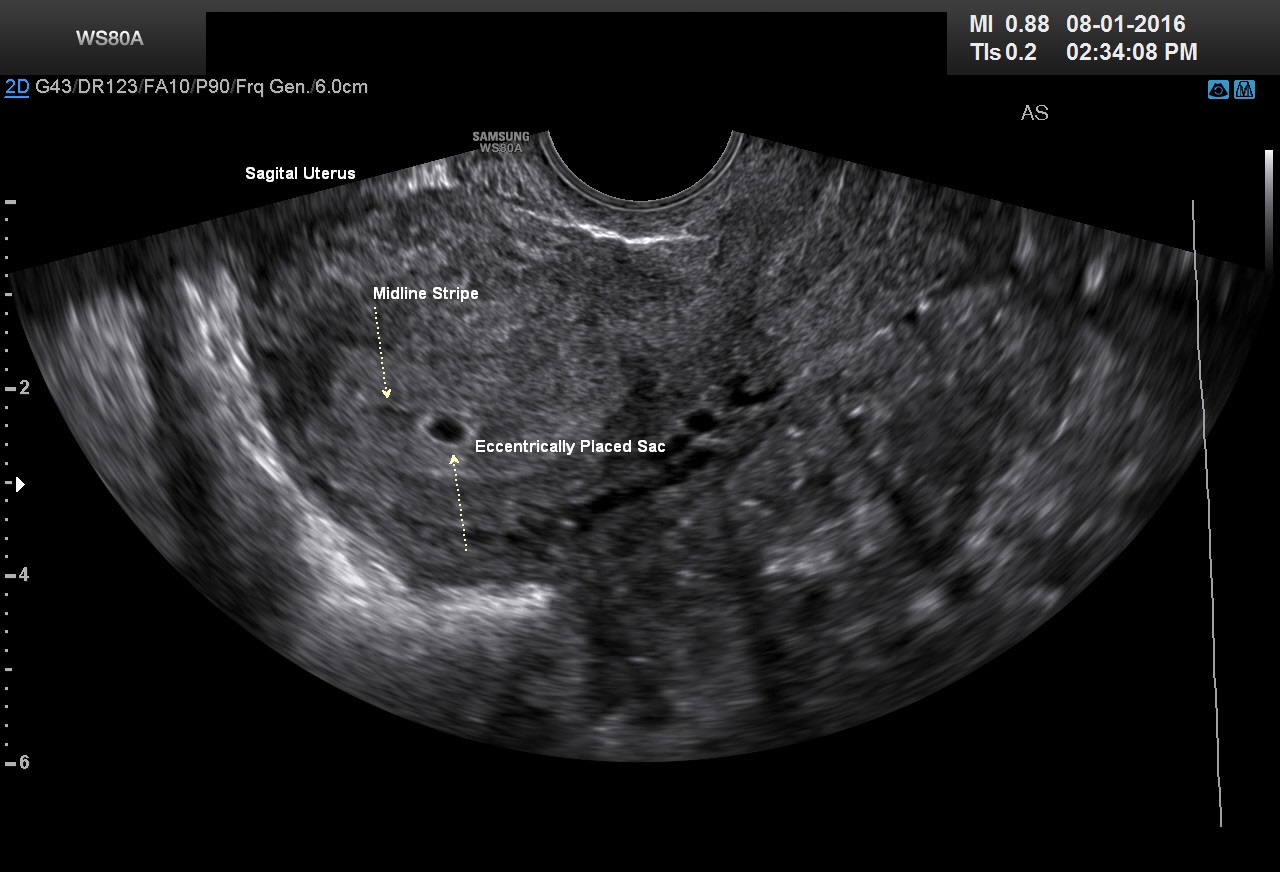 Fig 1A A very early, 3-mm mean diameter intrauterine gestational sac at 5 weeks postmenstruation
Fig 1A A very early, 3-mm mean diameter intrauterine gestational sac at 5 weeks postmenstruation
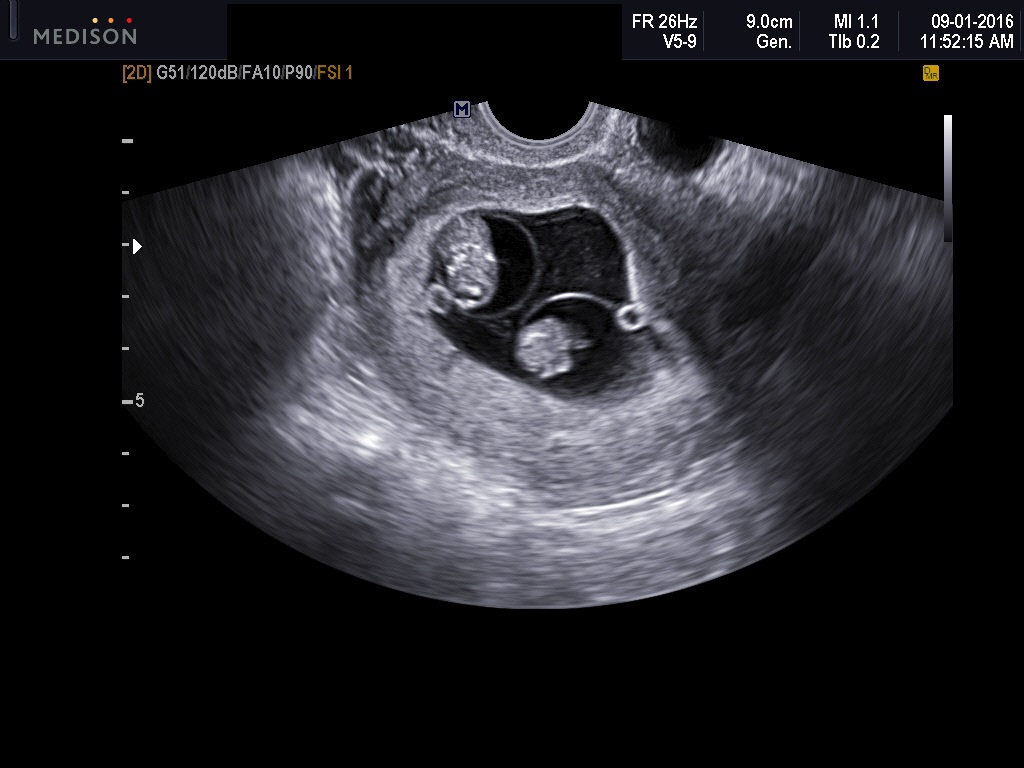
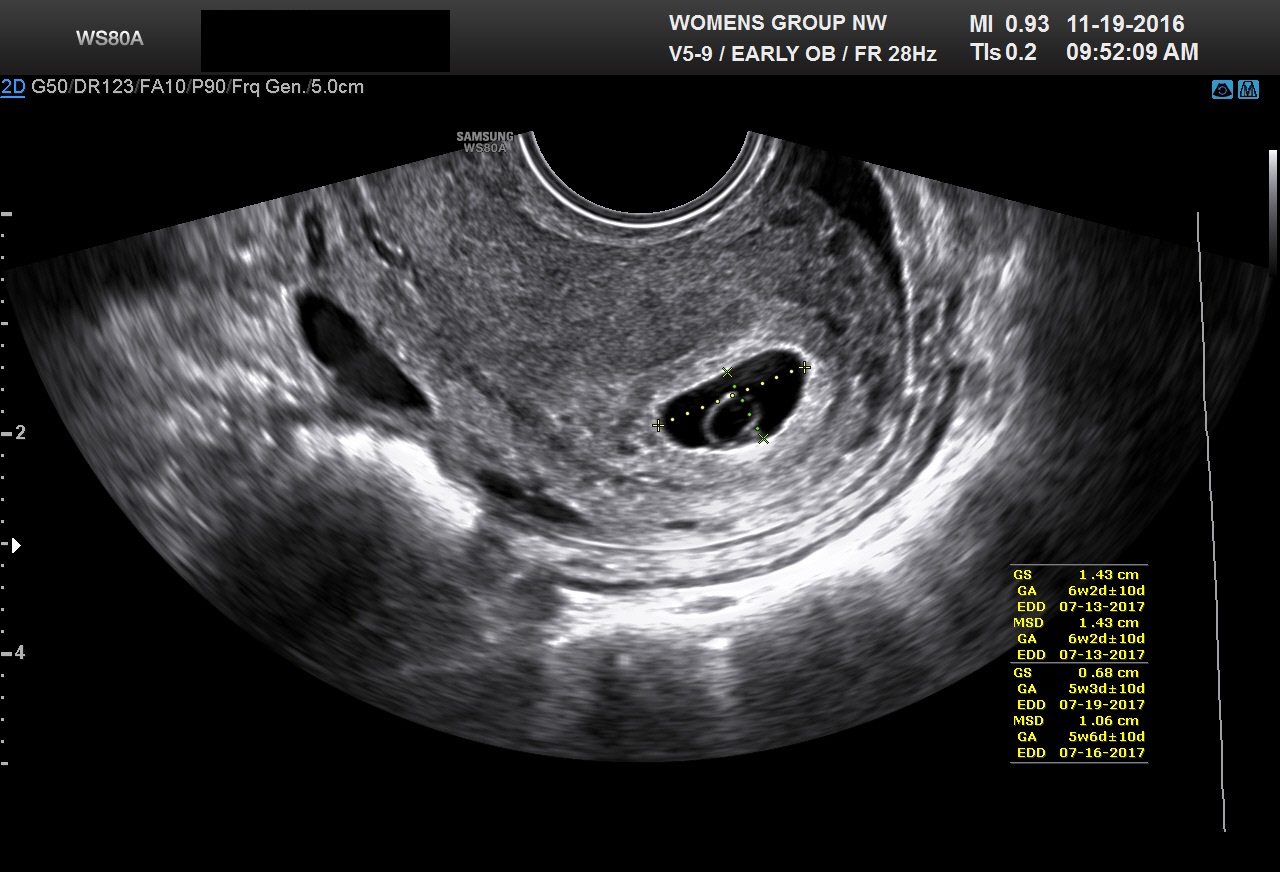 Fig 1B A typical yolk sac. The mean sac diameter of the gestational sac is 10.6 mm. The length and the anterior-posterior dimensions of the gestational sac are measured on this sagittal image of this retroverted uterus
Fig 1B A typical yolk sac. The mean sac diameter of the gestational sac is 10.6 mm. The length and the anterior-posterior dimensions of the gestational sac are measured on this sagittal image of this retroverted uterus
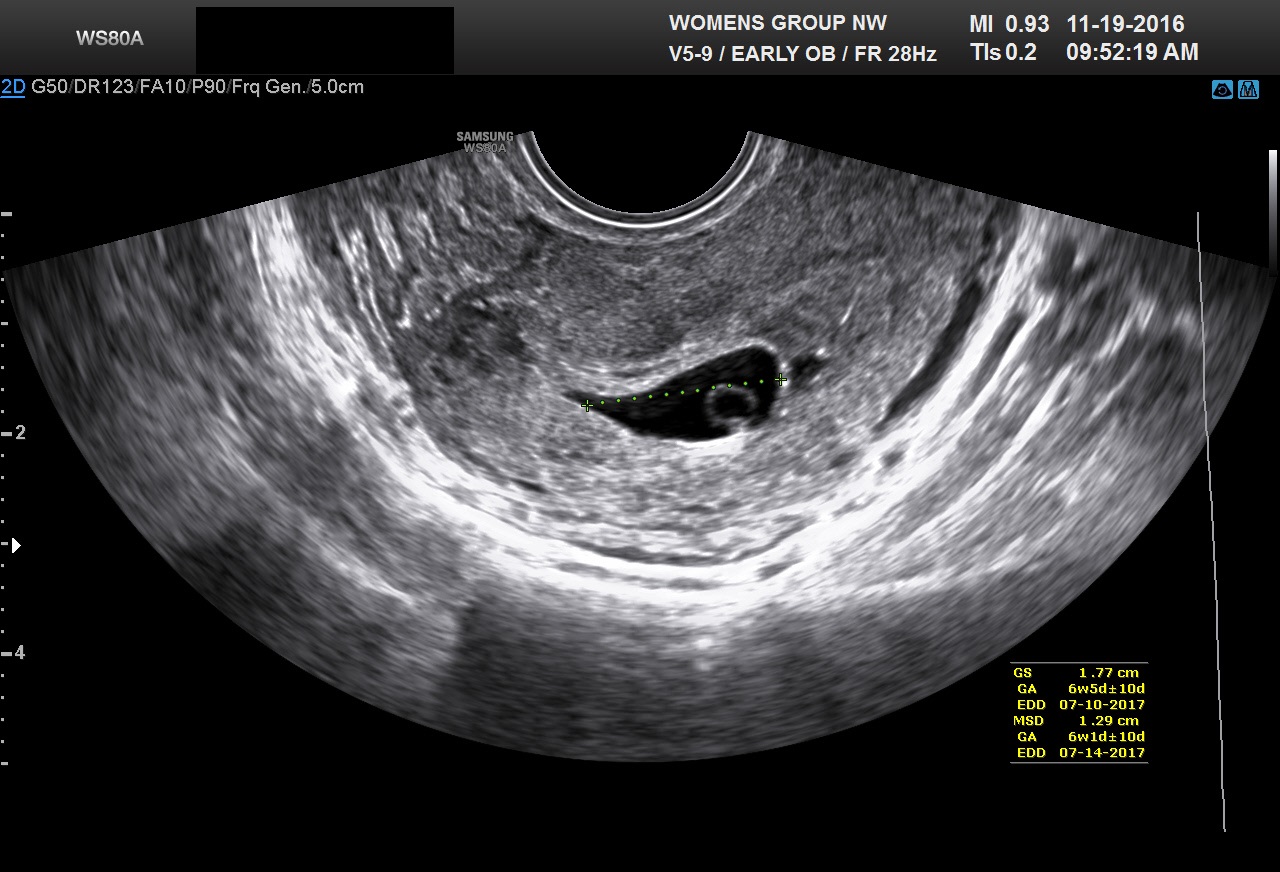 Fig 1C The third diameter of the gestational sac seen in Figure 1B is measured in this image. The mean sac diameter is the average of the three measured diameters (dimensions) of the sac
Fig 1C The third diameter of the gestational sac seen in Figure 1B is measured in this image. The mean sac diameter is the average of the three measured diameters (dimensions) of the sac
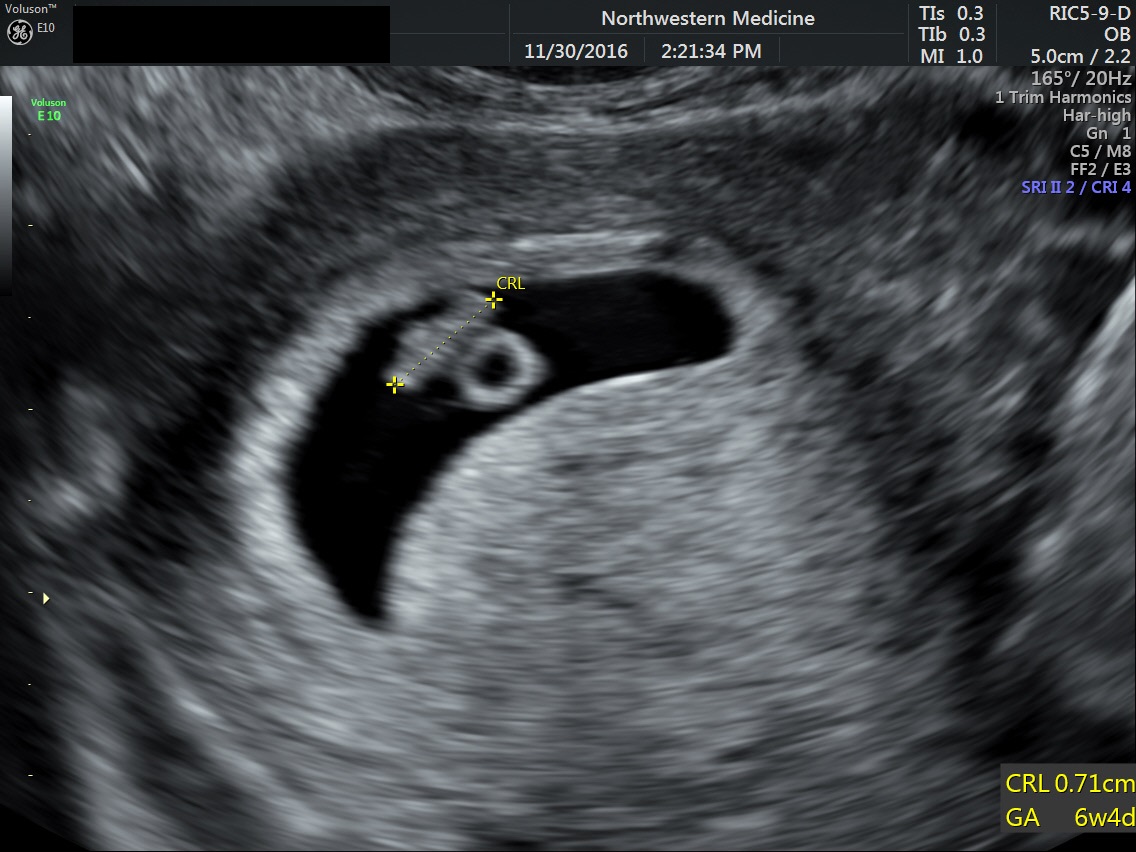 Fig 1D A early 7 mm fetal pole corresponding to 6 weeks 4 days menstrual weeks
Fig 1D A early 7 mm fetal pole corresponding to 6 weeks 4 days menstrual weeks
Table 2 Correlation between β-human chorionic gonadotropin (β-hCG) levels and appearance of early gestational structures
| Structure | Days from LMP | First IRP β-hCG (mIU/ml) | Second IS β-hCG (mIU/ml) |
Sac | 35 | 1400 | 914 |
Fetal pole | 40 | 5100 | 3800 |
Heart motion | 47 | 17,200 | 13,200 |
LMP, last menstrual period; IRP, International Reference Preparation; IS, International Standard
(Fossum GT, Davajan V, Kletzky OA: Early detection of pregnancy with transvaginal ultrasound. Fertil Steril 49:788, 1988)
Discriminatory zones
The generally accepted β-hCG discriminatory zone of 2000mIU/ml for the appearance of an intrauterine gestational sac is not 100% reliable. Recent papers by Doubilet and Benson and Ko and Cheung have provided examples of women with titers greater than 2000 mIU/ml, where transvaginal ultrasound did not identify a early intrauterine sac, and a subsequent ultrasound confirmed a viable pregnancy.3, 4 The β-hCG cut-offs for visualizing a intrauterine pregnancy sac, a yolk sac, and a fetus with heartbeat are guidelines. These cut-offs are accurate in a high percentage of patients with singleton pregnancies, but are subject to biologic as well single test error. These outliers are not limited to multiple gestations. In the series by Doubilet and Benson using transvaginal ultrasound , non-visualization of a early intrauterine pregnancy sac, was seen with a β-hCG level as high as 6567 mIU/ml. These exceptions to the previously accepted cut-offs have important implications in the management of threatened abortion, missed abortion, and ectopic pregnancy.
Pseudo-sacs
Considerable caution must be exercised not to mistake collections of fluid within the endometrial cavity for early gestational sacs. These “pseudogestational sacs” can lead to a missed diagnosis of ectopic pregnancy (Figures 2A–2D). Normal early gestational sacs are seen eccentrically placed in the decidua, adjacent to the echogenic central stripe (Figure 1A). This finding is called the intradecidual sign. A review of the literature by Richardson et al., from 2015, found the intradecidual sign was 66% sensitive and 100% specific in identifying an intrauterine pregnancy. The double decidual ring sign was 82% sensitive and 97% specific. Confidence intervals for the reviewed studies were not ideal because of the size of the studies.5 A new study from 2016 by Richardson et al. using high resolution ultrasound found a diagnostic sensitivity of 94% and 100% specificity for the double decidual ring in identifying an intrauterine pregnancy.6 Benson and Doubilet have found that in comparison to early circular intrauterine sacs, pseudo-sacs elongate on the sagittal view.7 Their finding is very useful in differentiating early pregnancy sacs from intracavitary fluid. Our lab at Northwestern relies very heavily on the intradecidual sign and the appearance of the sac on the sagittal view to differentiate very early pregnancy sacs from pseudosacs. We also rescan the patient in 7–10 days to document the appearance of the yolk sac.
Fig 2A An eccentrically placed intrauterine gestational sac 6 weeks postmenstruation (arrow)
Fig 2B In contrast, a pseudosac (arrow) in a patient with ectopic pregnancy representing a collection of blood or fluid collected within the endometrial cavity. This pseudo-sac elongates on the sagittal view
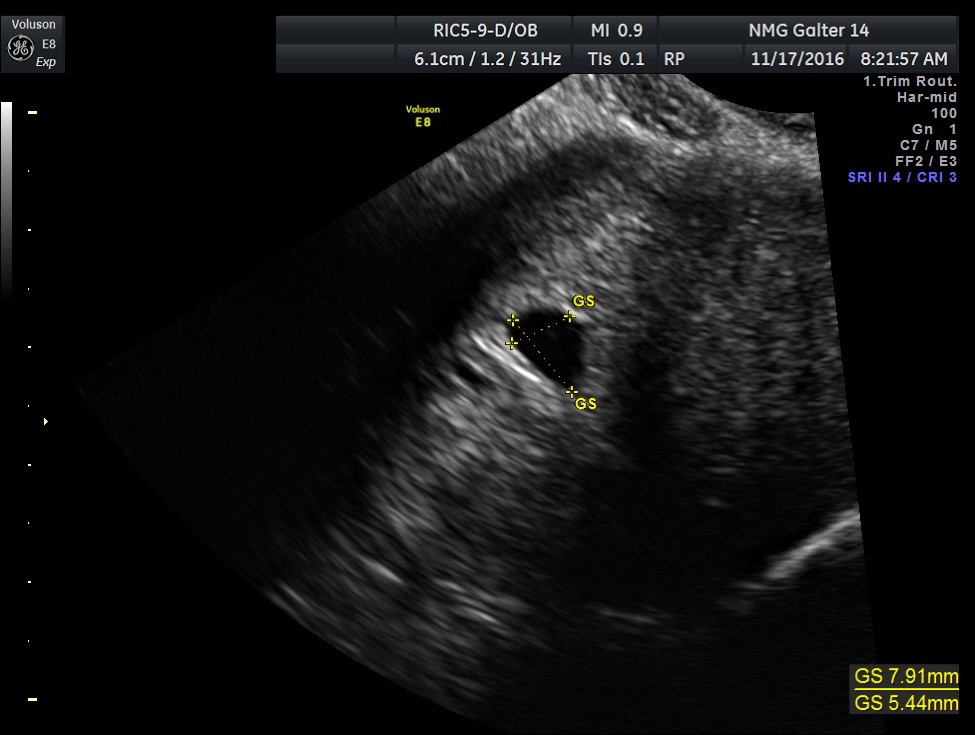 Fig 2C This sac-like structure was misinterpreted as an intrauterine gestational sac
Fig 2C This sac-like structure was misinterpreted as an intrauterine gestational sac
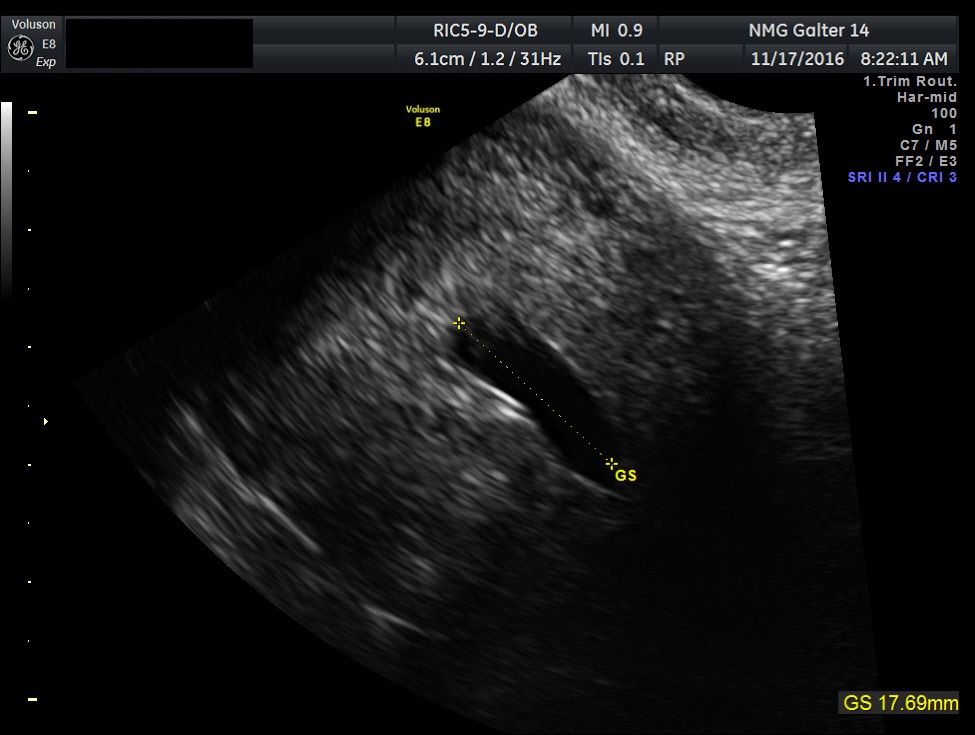 Fig 2D Same patient as Figure 2C. This sac-like structure elongates on the sagittal view. This is another example of a pseudo-sac with an ectopic pregnancy
Fig 2D Same patient as Figure 2C. This sac-like structure elongates on the sagittal view. This is another example of a pseudo-sac with an ectopic pregnancy
Corpus luteum and ovarian masses
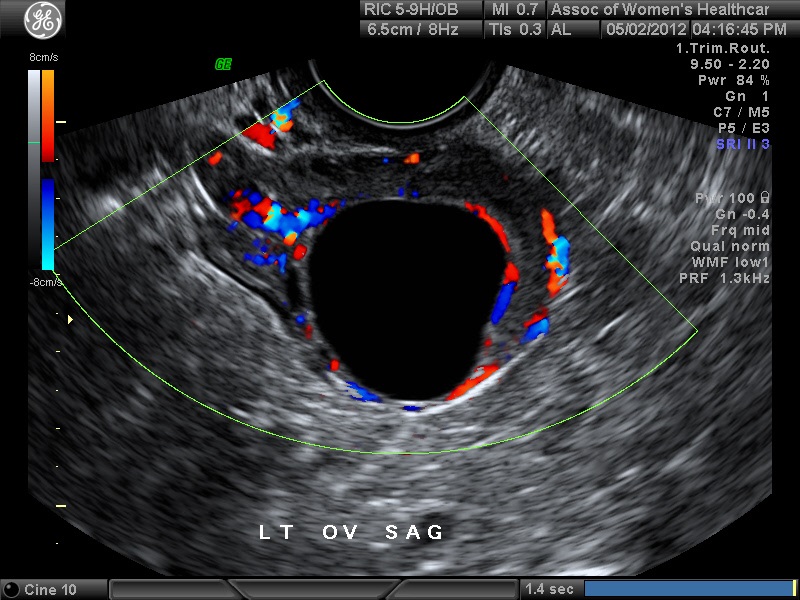
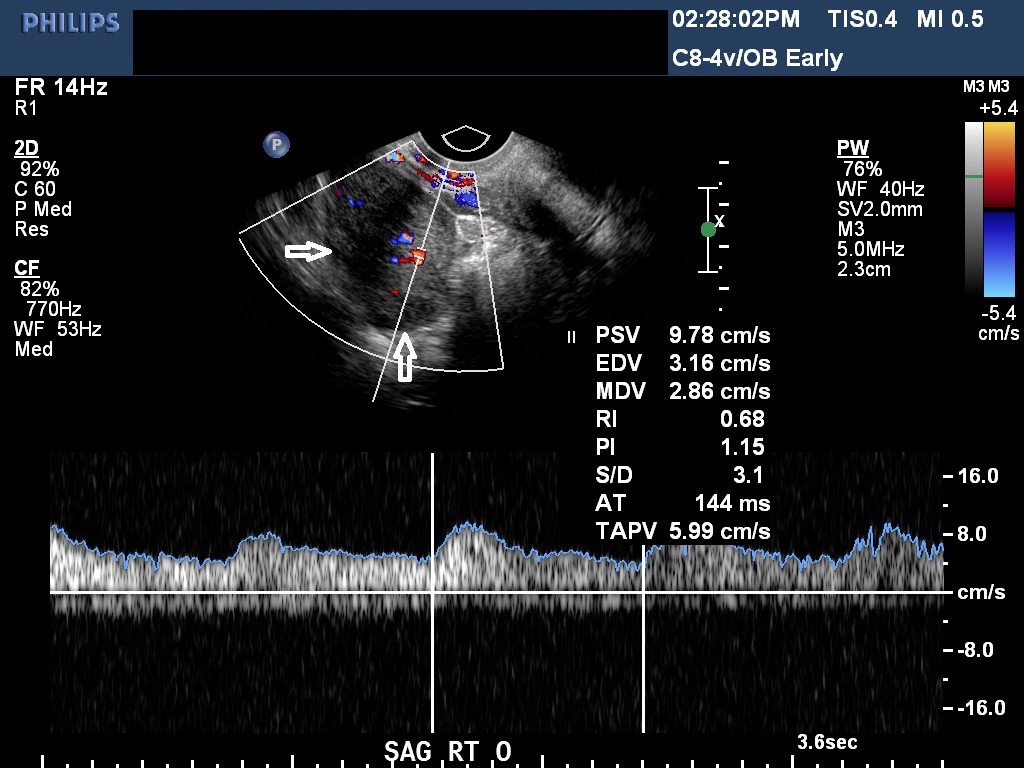
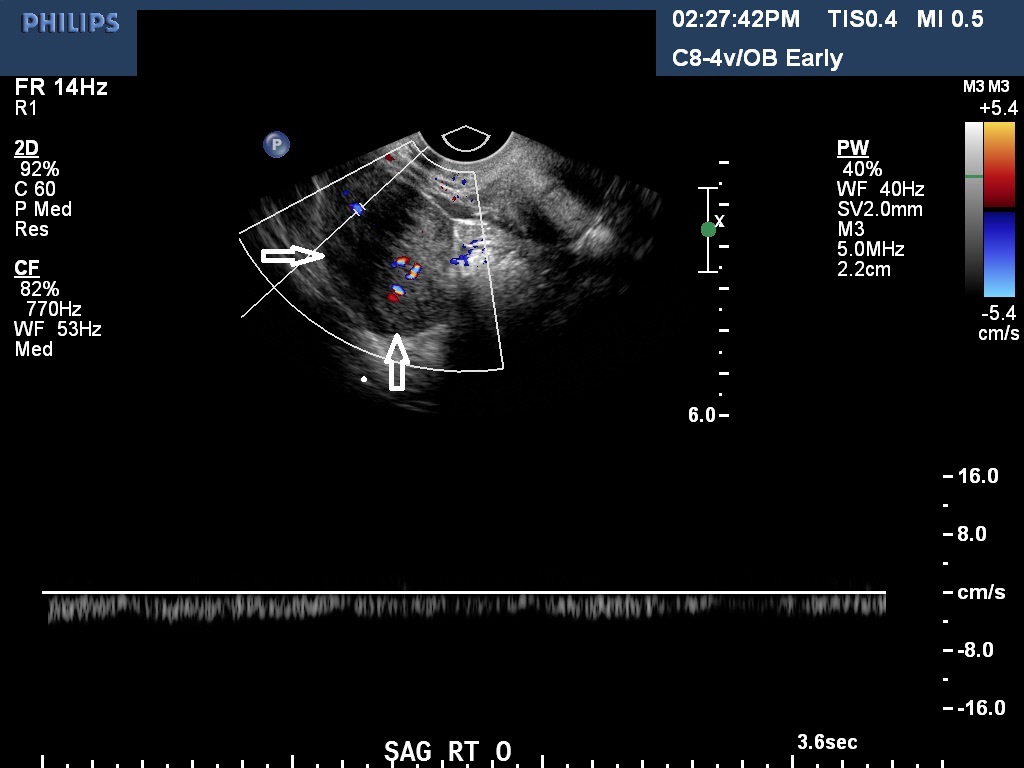
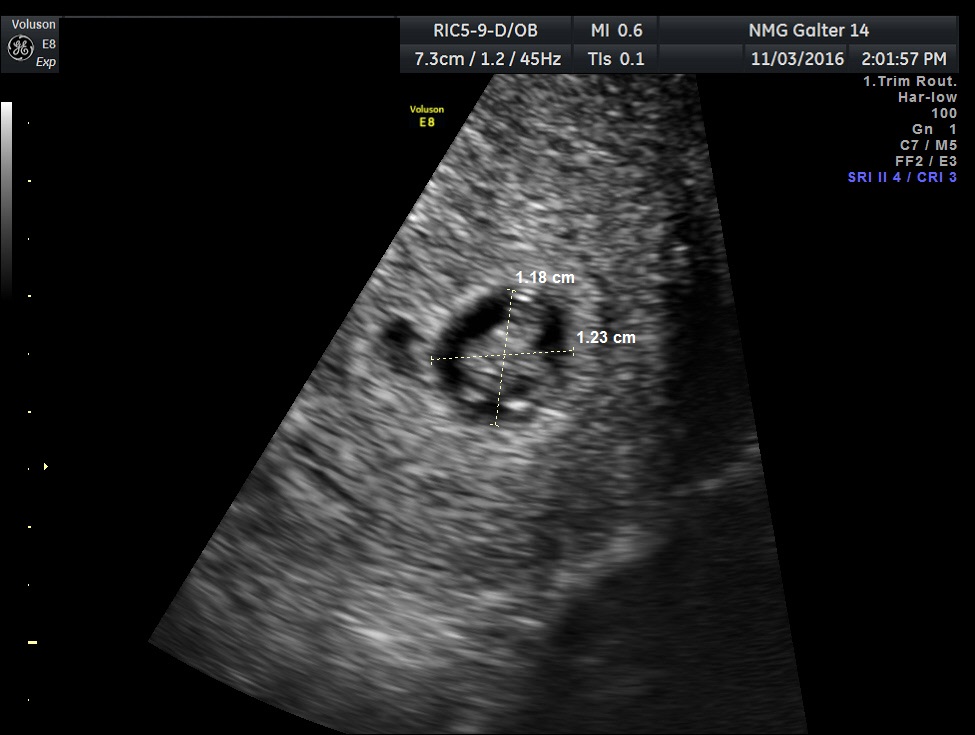
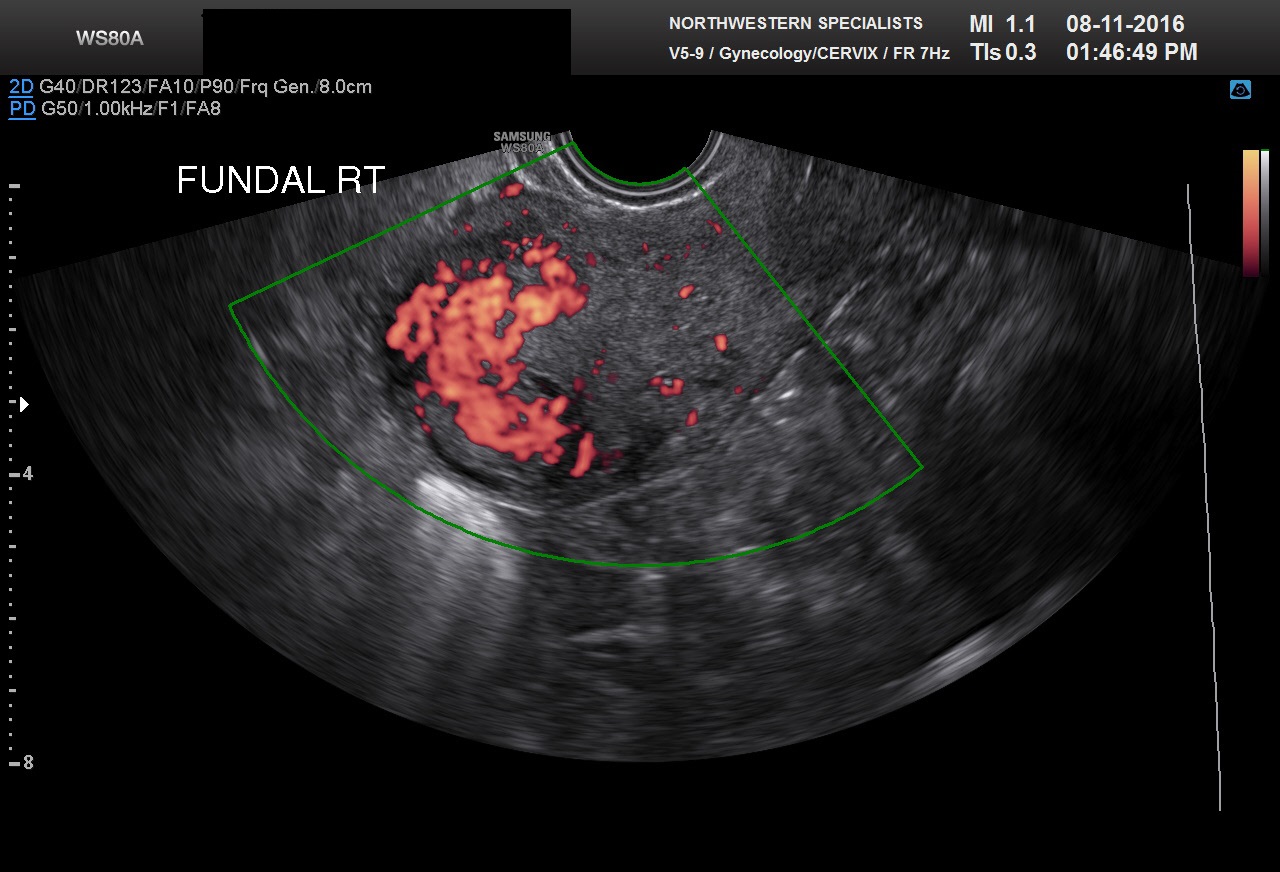
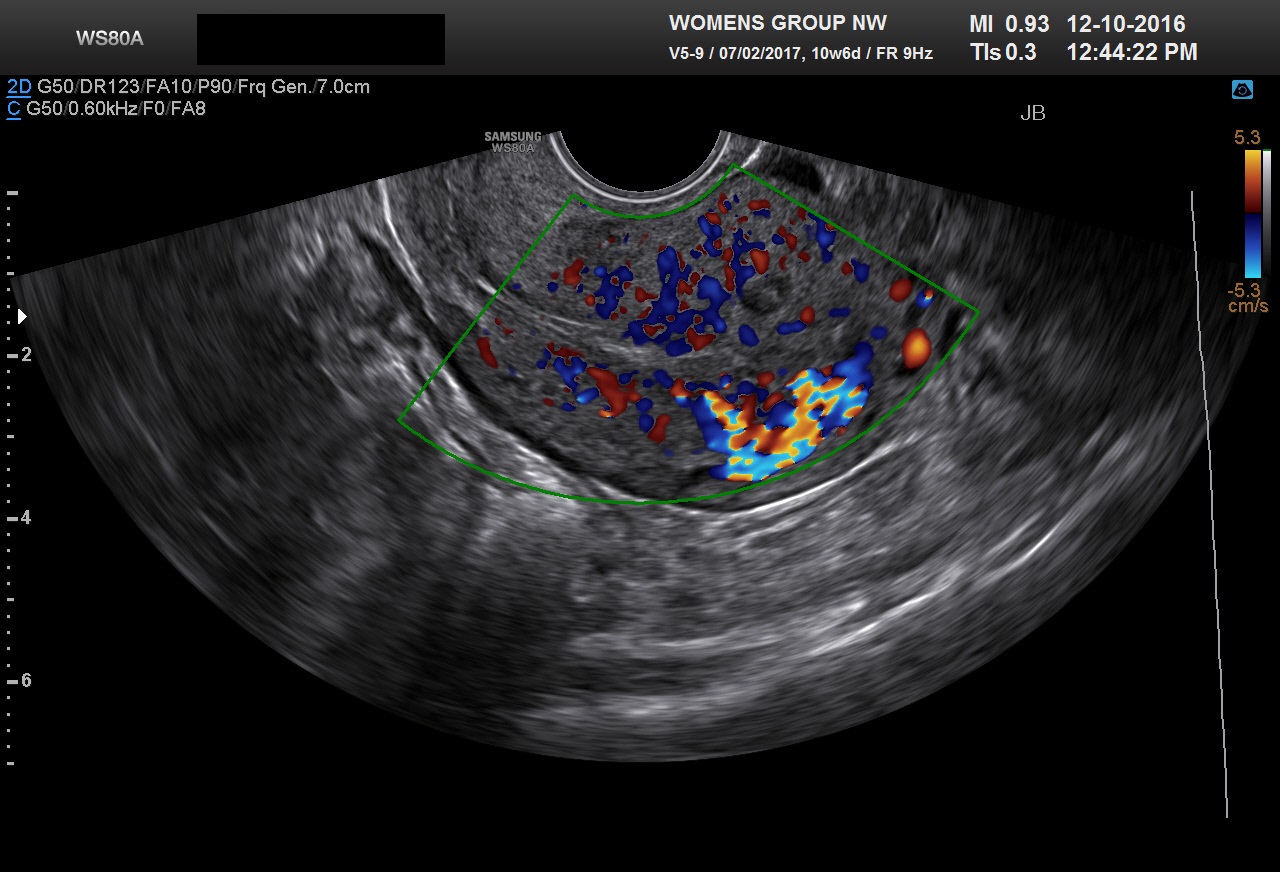
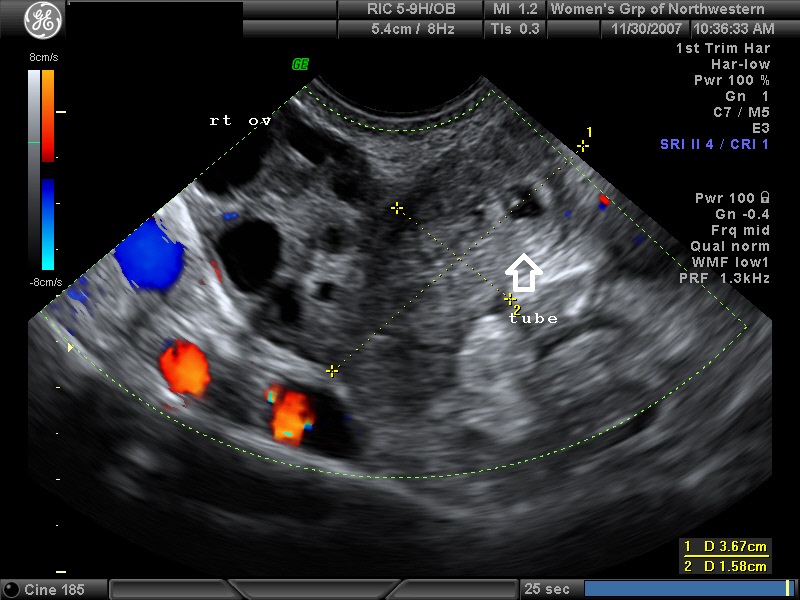
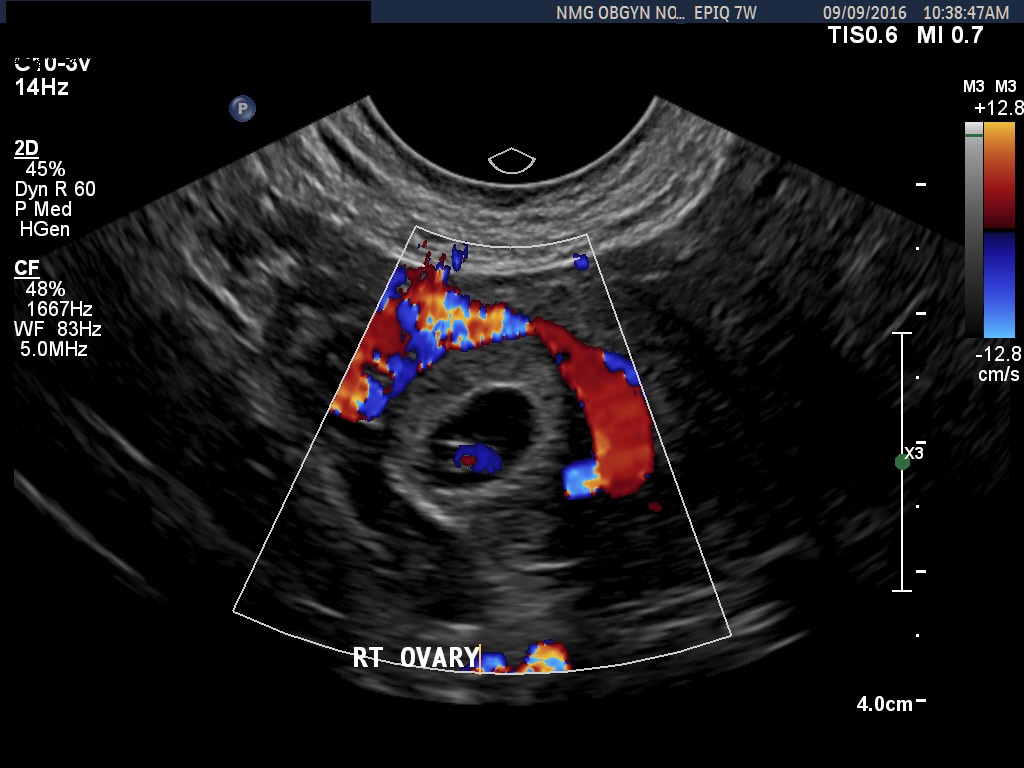
In the first 8 weeks of pregnancy, the corpus luteum is often identified as a cystic mass measuring 1–3 cm in diameter (Fig 3), although they may reach as large as 8 cm.8 The corpus luteum usually resolves spontaneously and is typically not seen by the onset of the second trimester. They can contain areas of complex echogenicity that may masquerade as a neoplasm or an ectopic pregnancy. Consultation with specialists should be obtained if an adnexal mass persists into the second trimester. The two most common persistent ovarian masses that are seen in the second trimester are endometriomas (Fig 4A) and cystic teratomas (Fig 5). Endometriomas and cystic teratomas usually do not require surgical intervention during pregnancy. The increasing accuracy of ultrasound supplemented by MRI in correctly predicting endometriomas, cystic teratomas, and adenofibromas/fibromas has led to a marked decreased in surgical interventions for adnexal masses during pregnancy (unpublished data). Torsion of an adnexal mass is the most common cause for surgery for an adnexal mass during pregnancy at our institution. The role of color Doppler velocimetry in ruling out torsion is problematic. In some cases both arterial and venous flow may be preserved on pulsed color Doppler (Fig 6). The need for surgical intervention in these cases is based on clinical findings and judgement. The risk of a persistent adnexal mass during pregnancy subsequently diagnosed as borderline or malignant has been estimated to be approximately 1–3% (Fig 7).9, 10 The published studies are retrospective and may be subject to ascertainment bias due to cases referred to major academic medical centers. Decidualized endometriomas can easily be confused with malignant ovarian neoplasms (Fig 4B−D).11 The ultrasound features which can be used to try to distinguish decidualized endometriomas from malignant ovarian cysts have been reviewed by Mascillini et al.12 MRI can be particularly useful in identifying a complex mass as a cystic teratoma, endometrioma, or fibroma. MRI may also be useful in differentiating benign from malignant ovarian/adnexal pathology. This subject has been well reviewed by Yacobozzi.13 The role of MRI with gadolinium contrast during pregnancy is subject to debate. In some countries its use is allowed after the first trimester. In the United States MRI with gadolinium it is considered contraindicated throughout pregnancy.
Fig 3A A 4 cm mean diameter simple corpus luteum cyst at 8 weeks. This cyst resolved by 13 weeks. In some cases the corpus luteum may persist in the second and third trimesters. Typically the circumferential Doppler flow will decrease in this time frame. Complete regression is then seen by 2–3 months postpartum. Failure of complete regression indicates other pathology
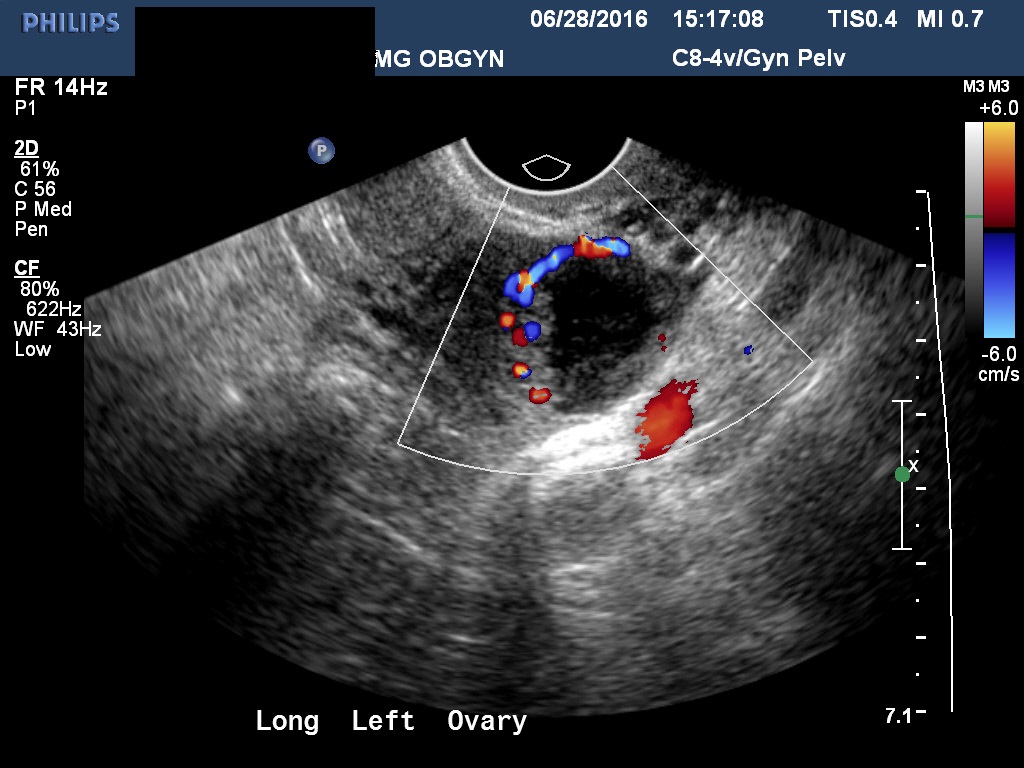 Fig 3B The inner border of this corpus luteum is crenulated. This crenulation can be confused with malignant ovarian pathology
Fig 3B The inner border of this corpus luteum is crenulated. This crenulation can be confused with malignant ovarian pathology
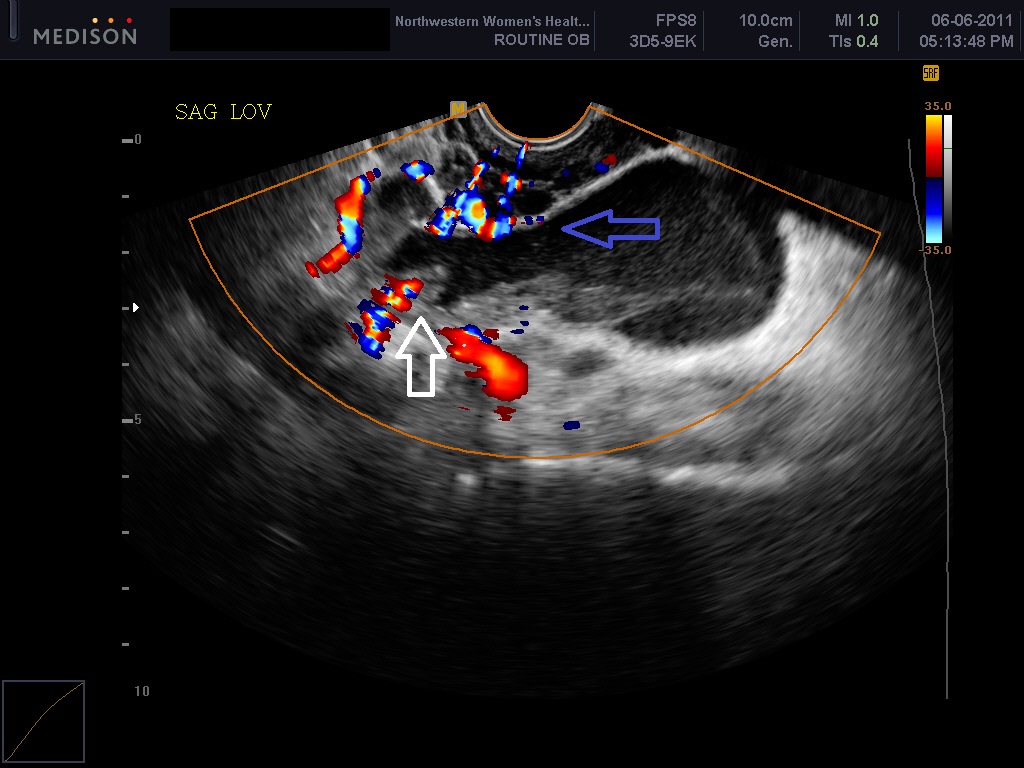 Fig 4A and 4B An endometrioma and decidualized endometrioma at 8 and 13 weeks in the same patient. The decidualized endometrioma’s appearance can be confused with the inner cyst wall. excrescences (white arrow) and septations (blue arrow) seen with malignancy
Fig 4A and 4B An endometrioma and decidualized endometrioma at 8 and 13 weeks in the same patient. The decidualized endometrioma’s appearance can be confused with the inner cyst wall. excrescences (white arrow) and septations (blue arrow) seen with malignancy
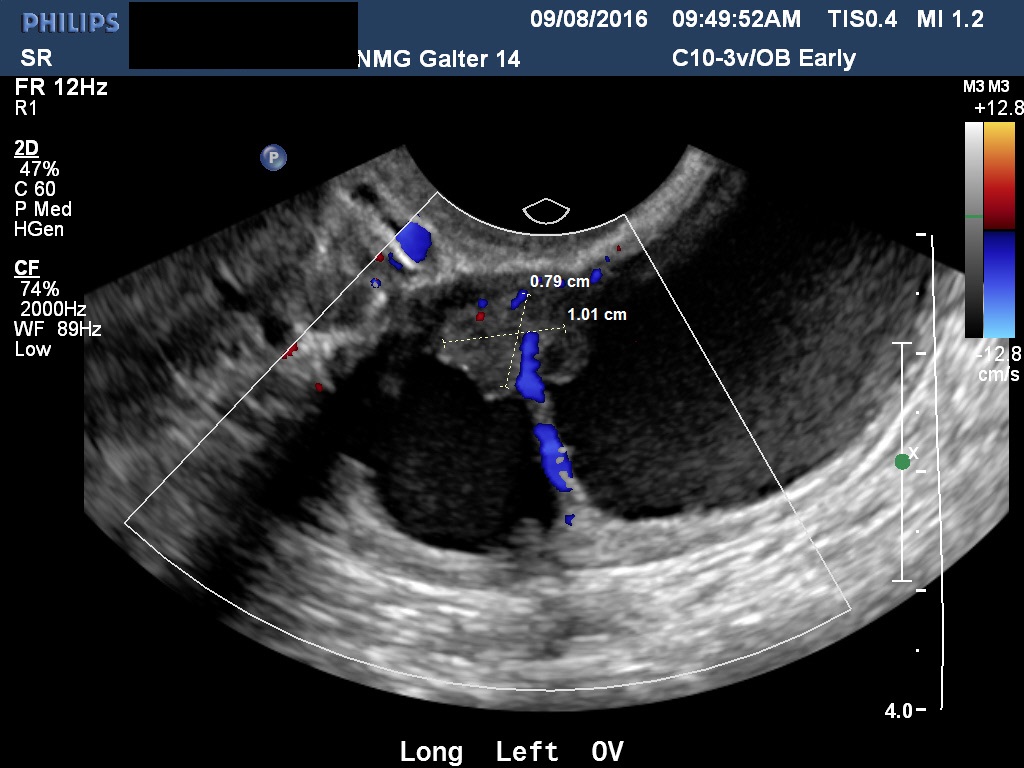 Fig 4C Another example of a decidualized endometrioma. The residual ovarian stroma measures 1.0 x 0.8 cm. The appearance can be easily confused with a malignant excrescence
Fig 4C Another example of a decidualized endometrioma. The residual ovarian stroma measures 1.0 x 0.8 cm. The appearance can be easily confused with a malignant excrescence
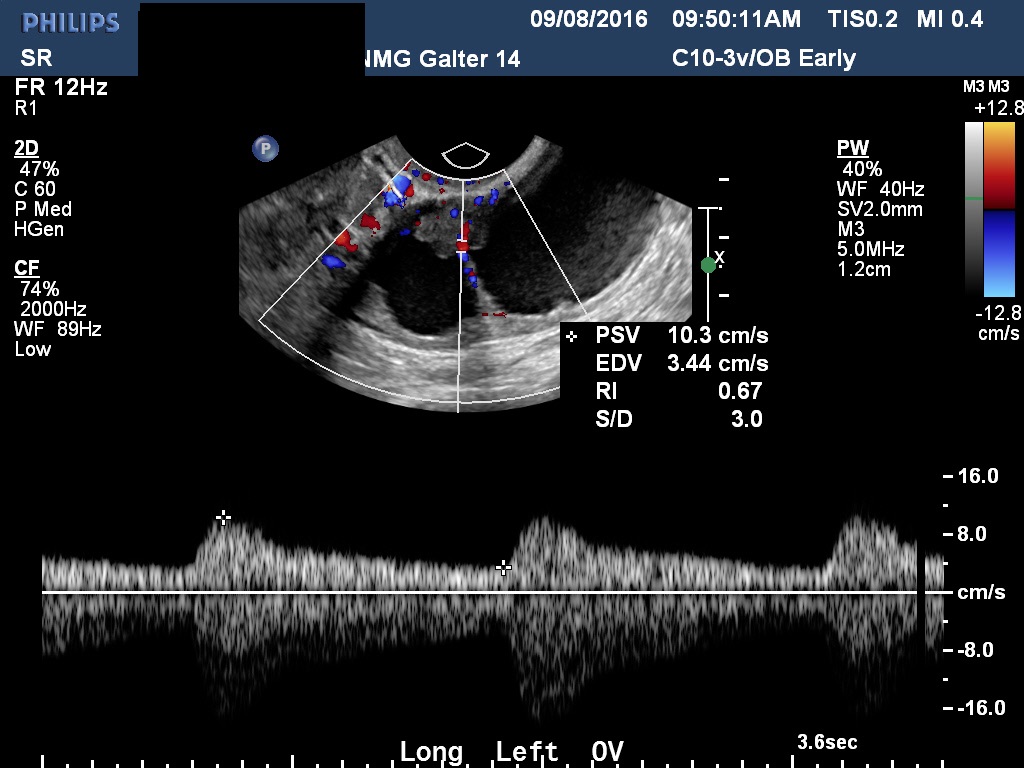 Figure 4D. Doppler velocimetry of the ovarian stroma seen in Figure 4C. The RI value is intermediate. RI values between 0.4 and 0.7 are considered intermediate. This is done mainly for documentation and research purposes. There is a tendency for malignancies to have low RI values. However, an RI cut-off of <0.4 for predicting malignancy is not sufficiently accurate to rely on. Neither are intermediate or high resistance values entirely reassuring. The role of Doppler velocimetry of ovarian masses during pregnancy remains controversial
Figure 4D. Doppler velocimetry of the ovarian stroma seen in Figure 4C. The RI value is intermediate. RI values between 0.4 and 0.7 are considered intermediate. This is done mainly for documentation and research purposes. There is a tendency for malignancies to have low RI values. However, an RI cut-off of <0.4 for predicting malignancy is not sufficiently accurate to rely on. Neither are intermediate or high resistance values entirely reassuring. The role of Doppler velocimetry of ovarian masses during pregnancy remains controversial
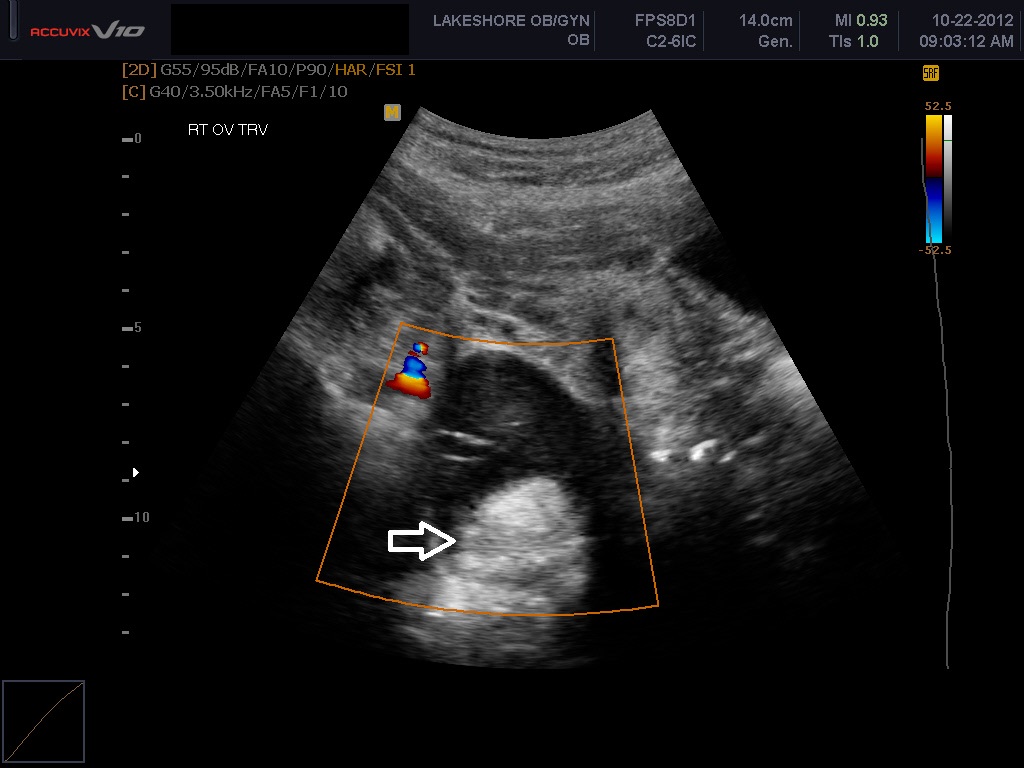
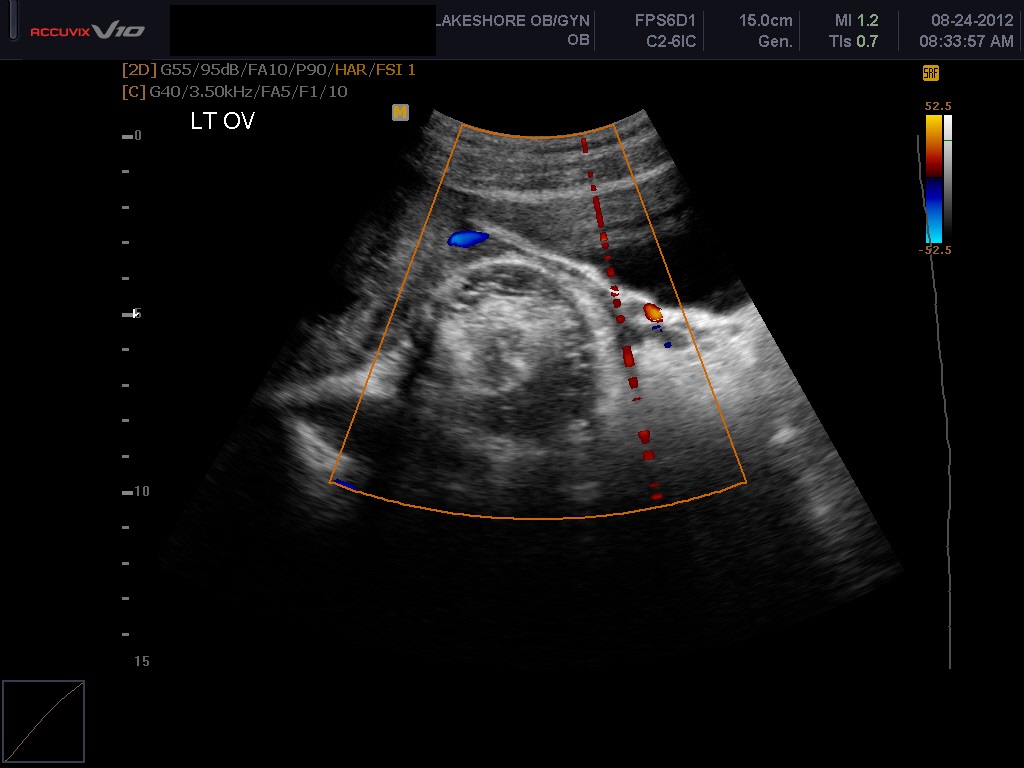 Figure 5A and 5B Two cystic teratomas. The first is in an “iceberg pattern”. The second has scattered areas of dense echogenicity throughout the mass. Cystic teratomas typically display posterior shadowing and exhibit minimal or absent flow on color Doppler centrally with the central region of the mass
Figure 5A and 5B Two cystic teratomas. The first is in an “iceberg pattern”. The second has scattered areas of dense echogenicity throughout the mass. Cystic teratomas typically display posterior shadowing and exhibit minimal or absent flow on color Doppler centrally with the central region of the mass
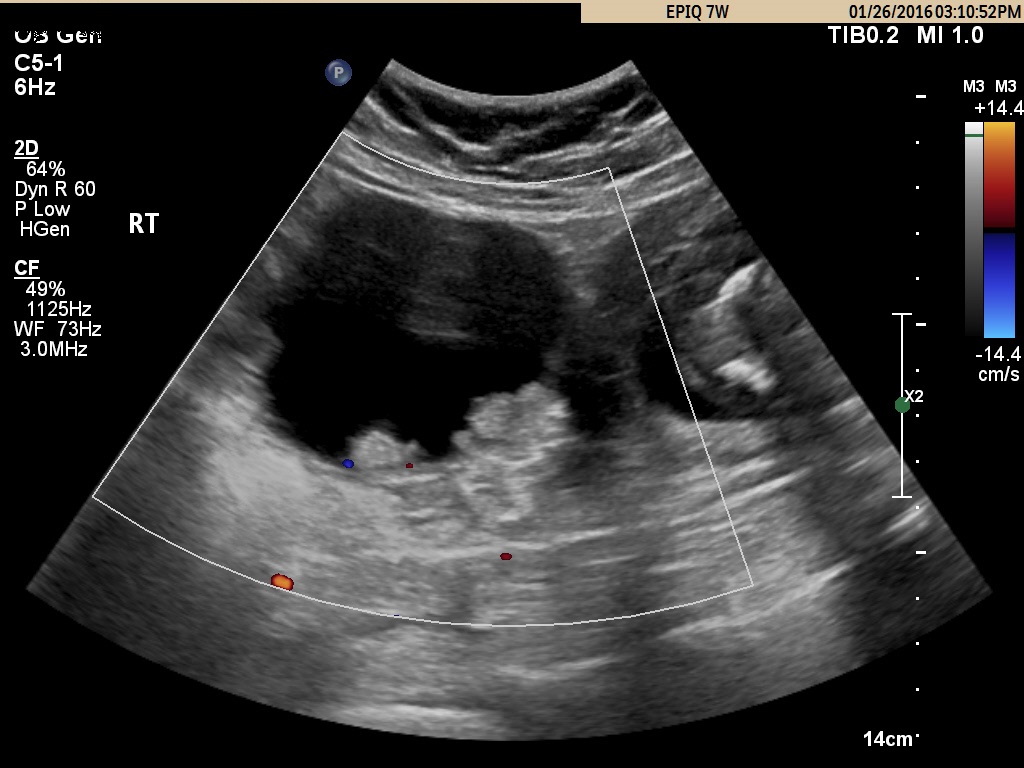
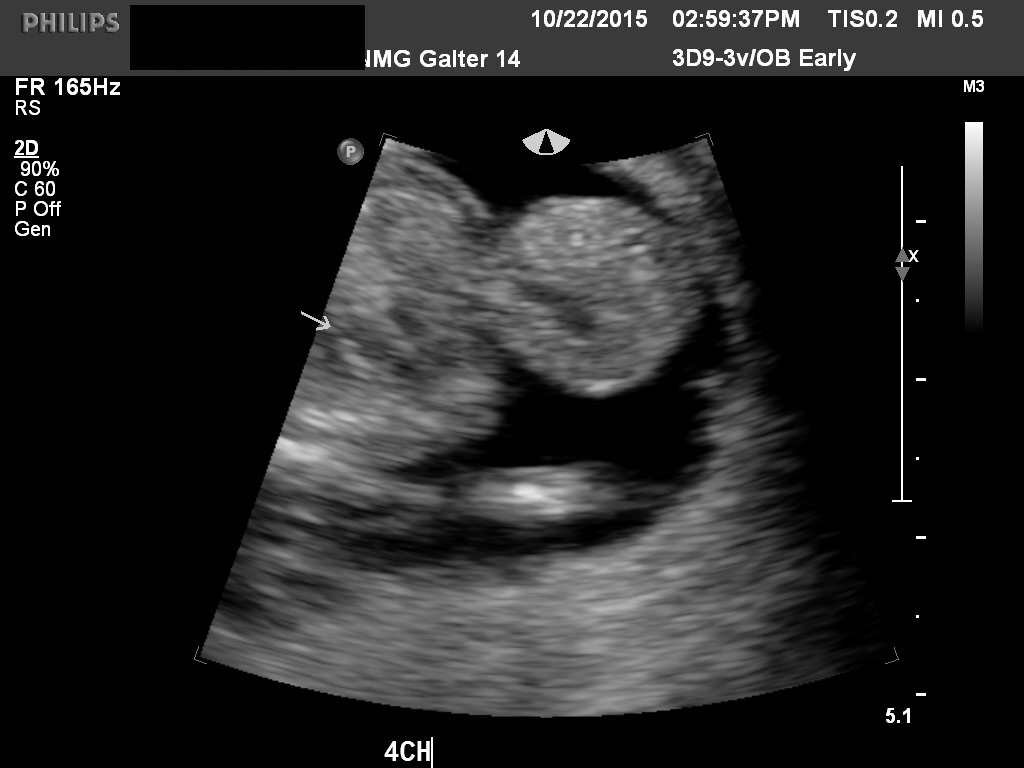
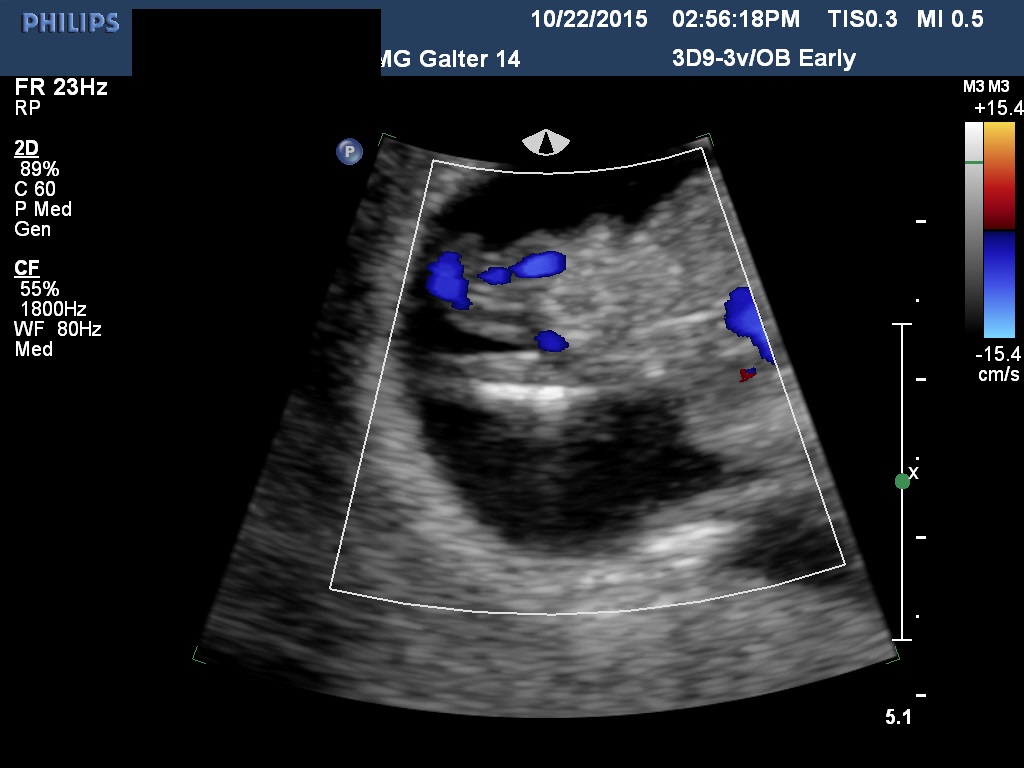
Fig 6A and 6B A torsion of an ovary with preserved arterial and venous flow on Doppler velocimetry
Fig 7A A typical borderline tumor of the ovary at 14 weeks
Fig 7B 3D imaging of multiple internal excrescences seen in many borderline tumors. In the United States these are typically surgically treated in the early second trimester. In other countries surgery is performed 2–3 months postpartum or at the time of cesarean section
THREATENED ABORTION AND COMPLETED ABORTION
Approximately 15% of women have a spontaneous, clinically recognized pregnancy loss in the first trimester. Ball and co-workers found that a subchorionic bleed is associated with an increased risk of miscarriage, stillbirth, abruptio placentae, and preterm labor (Fig 8). Their data suggested that the risk of spontaneous abortion increased in proportion to an increase in the size of the subchorionic bleeds; however, a larger sample size was needed to determine statistical significance.14 A recent meta-analysis by Tuuli et al. found that subchorionic hematoma is associated with an increased risk of early and late pregnancy loss, abruption, and premature rupture of membranes.15 Bradycardic fetal heart rates, small sac size (Fig 9), enlarged yolk sacs (Fig 10) have also been associated with an increased risk of first-trimester pregnancy loss. A recent paper by Tan et al confirmed that a higher risk of spontaneous miscarriage when yolk sacs were larger than 7 mm at 6−7 weeks.16
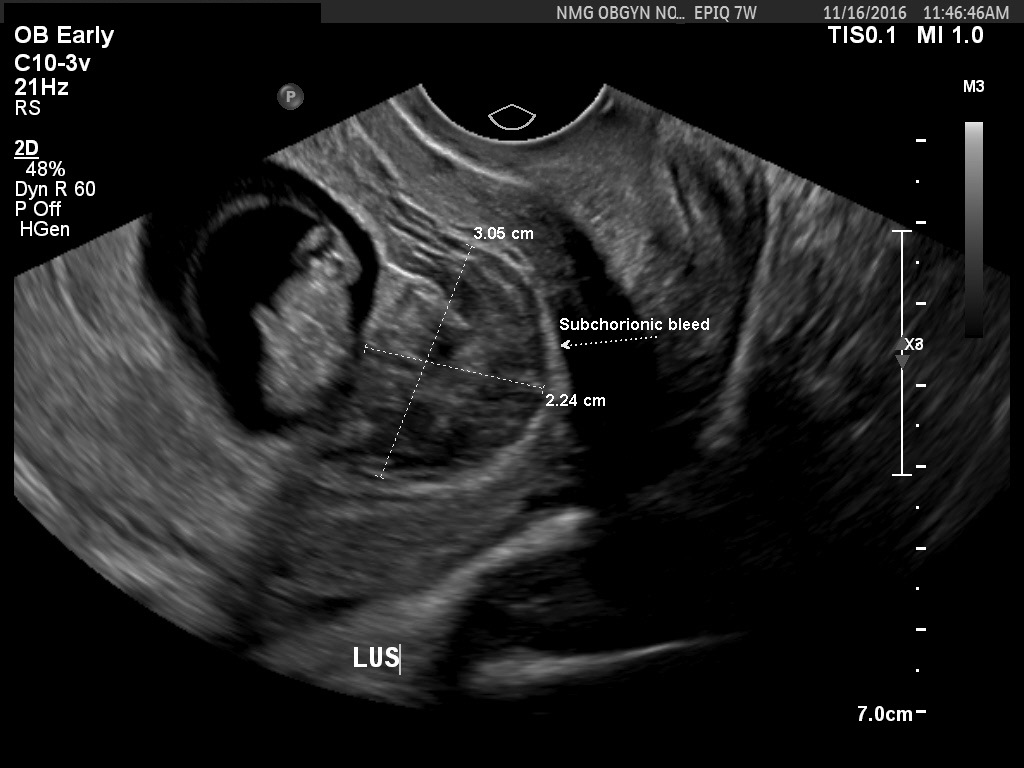
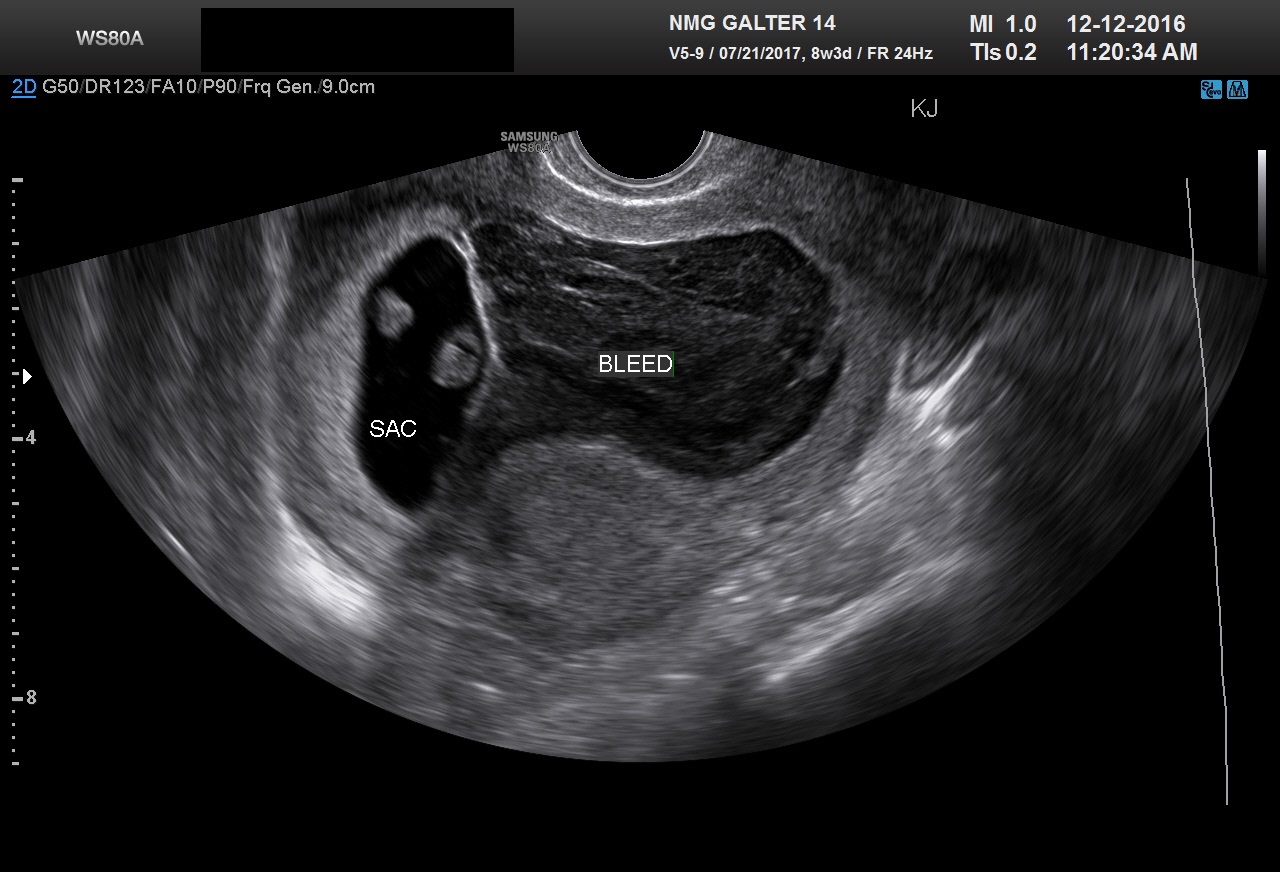 Fig 8A and 8B Two examples of large subchorionic bleeds
Fig 8A and 8B Two examples of large subchorionic bleeds
Fig 9 An example of a fetus crowded in a sac. Note that only two dimensions of the sac are measured on this image. Early gestational sacs should be measured in three dimensions
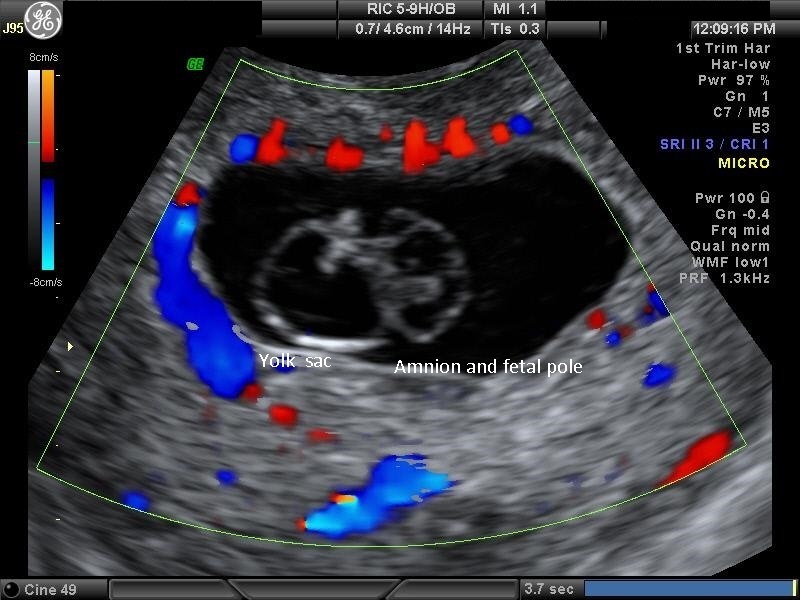
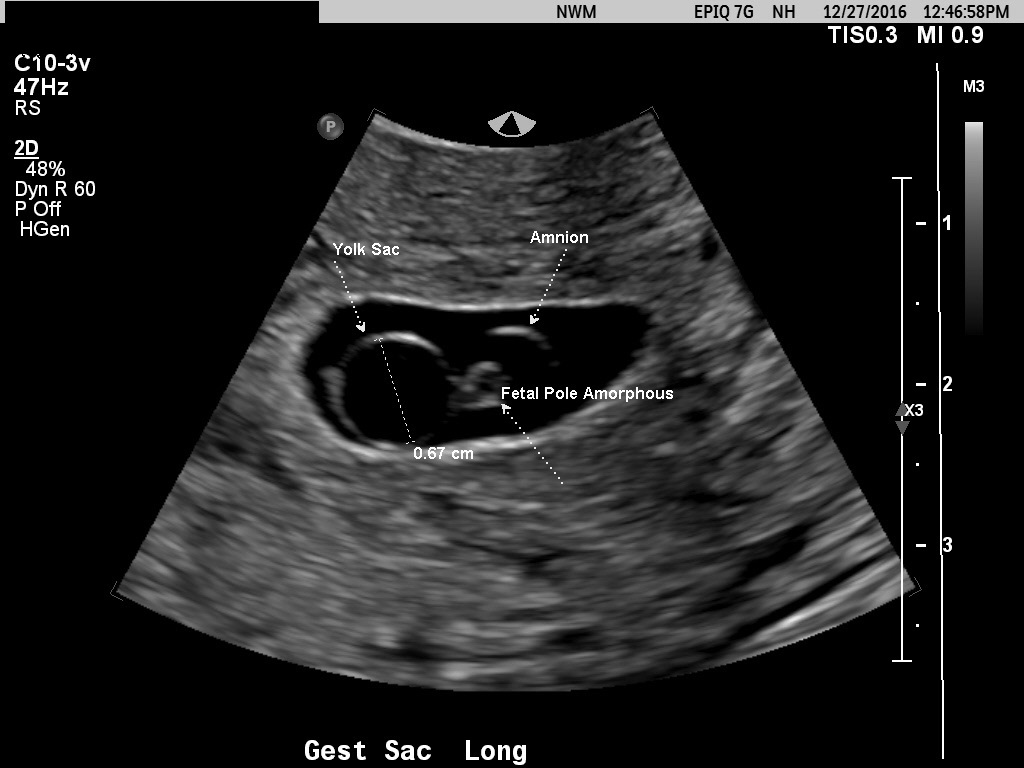 Fig 10A and B Two examples of patients with eventual early pregnancy loss. Both yolk sacs are enlarged to approximately 7 mm
Fig 10A and B Two examples of patients with eventual early pregnancy loss. Both yolk sacs are enlarged to approximately 7 mm
Missed abortion
Two papers by Doubilet and Benson in 2010 and Abdallah et al. both in 2011 recommended that using previous guidelines for diagnosing missed abortion could result in inadvertent termination of pregnancy in a small percentage of women with suspect missed abortion.17, 18, The previous guidelines from the earlier version of this chapter are given below.
- Pennell and associates, using transvaginal scanning (TVS), found that a 12-mm mean diameter sac is seen at approximately 6+ menstrual weeks. Failure to see a yolk sac or small fetal pole when the sac size reaches this diameter should heighten concern of a loss. If a TVS repeated 7–10 days later fails to reveal embryonic structures, the diagnosis of missed abortion can be made unequivocally.19
- By TVS, fetal heart motion should be seen 100% of the time when the fetal pole reaches 5 mm; absence of fetal heart motion at this stage is a strong indication of missed abortion. By transabdominal scan (TAS), fetal heart motion should be seen when the fetal pole reaches 12 mm. The reliability of TAS can be compromised by maternal obesity, obscuring leiomyomas, and retroversion.20
- Goldstein and colleagues, using TVS, found that fetal heart motion should be seen when the mean sac diameter reaches 20 mm. Absence of fetal heart motion at this stage is consistent with a missed abortion. By TAS, fetal heart motion is usually seen at a diameter of 25 mm. Again, diagnosis of missed abortion via TAS may be unreliable in the presence of maternal obesity, leiomyomas, or retroversion.21
- For patients who appear not to believe the diagnosis of pregnancy loss, a repeat scan at an appropriate interval may be indicated.
- Many patients expect that TVS will be performed. Both they and their physicians may be uncomfortable if the diagnosis of early pregnancy loss is not confirmed by this technique. In addition a full bladder is not required for TVS. The majority of patients are more comfortable being scanned transvaginally with an empty bladder. Furthermore, anesthesiologists prefer that patients do not have full stomachs from drinking large volumes of fluid.
A new standard in the United States was reached at a multispecialty conference sponsored by the Society of Radiologists in Ultrasound. It was summarized and published in the New England Journal in 2013 by Doubilet et al.22, The guidelines listed above have been changed as follows.
1a. Absence of embryo with heartbeat >2 weeks after gestational sac visualized without a yolk sac.
1b. Absence of a embryo with a heartbeat >11 days after a gestational sac visualized with a yolk sac.
2. The CRL is >7 mm and fetal heart tones are absent.
3. The mean sac diameter is >25 mm and no embryo is seen.
The older guidelines #1, #2, and #3 above are now considered suspicious but not diagnostic for a missed abortion. Statistical analysis by Jeve et al. in 2011 of the older guidelines revealed a confidence interval of 90–100% for the specificity.23 Most ultrasound labs using the older guidelines had already modified their practice by rescanning 7–10 days after a missed abortion was diagnosed by old guidelines #2 and #3. The new guidelines are meant to be 100% specific with a positive predictive value as close as possible to 100% for predicting missed abortion.
Completed abortion
TVS can be used to evaluate women thought to have completed abortions. In a study by Rulin and co-workers, 48 of 49 women determined as having an empty uterus using TVS were spared dilatation and curettage.24
Expectant management or pharmacologic methods can be used to treat early missed abortions in 80–90% of patients without dilation and curettage (D and C) being required. Cowett et al. have reported that unless the patient has excessive bleeding or other compelling symptoms, the absence of a gestational sac after medical abortion, demonstrates that a dilation and curettage will not be required.25 Small amounts of clot, fragments of tissue, and small regions of color Doppler flow at the implantation site can be usually be managed expectantly. Suspected endometritis or subinvolution of the placental site may require antibiotic or cytotec. Rarely patients will have persistent benign trophoblast. On Doppler these patients will have extension deep into the myometrium. Two examples are given in Figures 11A and 11B.
Fig 11A and B Persistent benign trophoblast is likely in both of these cases. Both patients had presented with bleeding at 7–8 weeks with intrauterine pregnancy sacs and missed abortion was confirmed. Both were treated with cytotec. We recommend transvaginal scan be performed 1 week after cytotec to document completed abortion or sooner if there is excessive bleeding. The gestational sacs are no longer seen in these two examples. However, areas of markedly increased color Doppler are noted in the myometrium fundally. Both patients had persistent bleeding and β-hCG levels that were not falling properly. Management of these cases varies between practitioners and institutions. Careful D and C under ultrasound guidance gives a histologic diagnosis. However, methotrexate may still be required if β-hCG levels fail to become negative. This can be seen even when benign trophoblast is found on pathology. Alternatively, the patient may be treated initially with methotrexate. This requires that the pretreatment ultrasound shows no evidence of molar tissue or marked hydropic change.
The practitioner must not forget that it is quite common for a patient to pass a decidual cast and falsely think they have a spontaneous abortion of an intrauterine pregnancy, when they actually have an ectopic pregnancy. For the practitioner to be satisfied with an ultrasound diagnosis of completed abortion, one of three conditions must be met:
- A previous ultrasound documented an intrauterine pregnancy, and the endometrial cavity is now empty.
- Products of conception have been identified pathologically, and the uterine cavity appears empty.
- Quantitative titers are heading toward zero at an appropriate rate, and the uterine cavity appears empty. This may also be consistent with a nonviable ectopic or tubal abortion.
Both sonologist and practitioner must also entertain the possibility of a heterotopic pregnancy, which is a simultaneous intrauterine and extrauterine twin pregnancy. Particularly with the rise of patients undergoing assisted reproduction, this entity is being encountered more frequently.26
ECTOPIC PREGNANCY
The incidence of ectopic pregnancy has now reached nearly 2%. Any patient with a history of ectopic pregnancy, tubal ligation or tubal surgery, or pelvic inflammatory disease should undergo TVS by 6 weeks from the last menstrual period (LMP). For patients who are not at high risk for ectopic pregnancy, the two most common presenting symptoms are bleeding and pelvic pain. The pain is typically lateralized over the adnexa.
In a 1981 study, Kadar and associates found that a “discriminatory” β-hCG value of 6000 mIU/ml could be used as a cut-off for when an intrauterine gestational sac should be seen via TAS.27 With the availability of TVS in the late 1980s, many publications found that an early gestational sac was typically seen at a β-hCG level of 1500–2000 mIU/ml (Third International Standard). The normal doubling time for β-hCG levels in early gestation was felt to be every 48–72 hours. In the presence of slowly rising or plateauing β-hCG titers and nonvisualization of an intrauterine gestational sac, the patient can be diagnosed with an early intrauterine pregnancy loss or ectopic pregnancy.
Recent papers by Condous et al. and Doublilet and Benson have questioned the safety of using a 2000 mIU/ml cut-off for identifying a intrauterine pregnancy.28, 29, In a hemodynamically stable patient with no evidence of an extrauterine pregnancy on scan, a single β-hCG value greater than this cut-off should not be considered diagnostic for a ectopic pregnancy. A paper by Barnhart found that β-hCG levels may double more slowly than expected in some viable pregnancies.30
The sensitivity of TVS in detecting actual ectopic adnexal masses is probably dependent on both β-hCG levels and the skill of the sonographers. Stovall and co-workers visualized an adnexal mass suspicious for ectopic pregnancy in 94% of patients. The mean pretreatment β-hCG level was 3950 mIU/ml. However, they noted a fetal heartbeat in only 12% of these ectopic pregnancies.31 This compared to 23% in the study by Timor-Tritsch and colleagues.32, The corpus luteum itself and hemoperitoneum secondary to it can lead to a false-positive diagnosis of ectopic pregnancy. In addition, the clinician should be aware that very small ectopic pregnancies identified on ultrasound may be difficult to identify laparoscopically. The American Society of Reproductive Medicine guidelines for the medical treatment of ectopic pregnancy were most recently modified in 2013.33 An excellent French overview and guidelines are available from 2016.34
Examples of various locations for ectopic pregnancy are given Figures 12–17.
Fig 12 Ampullary tubal ectopic
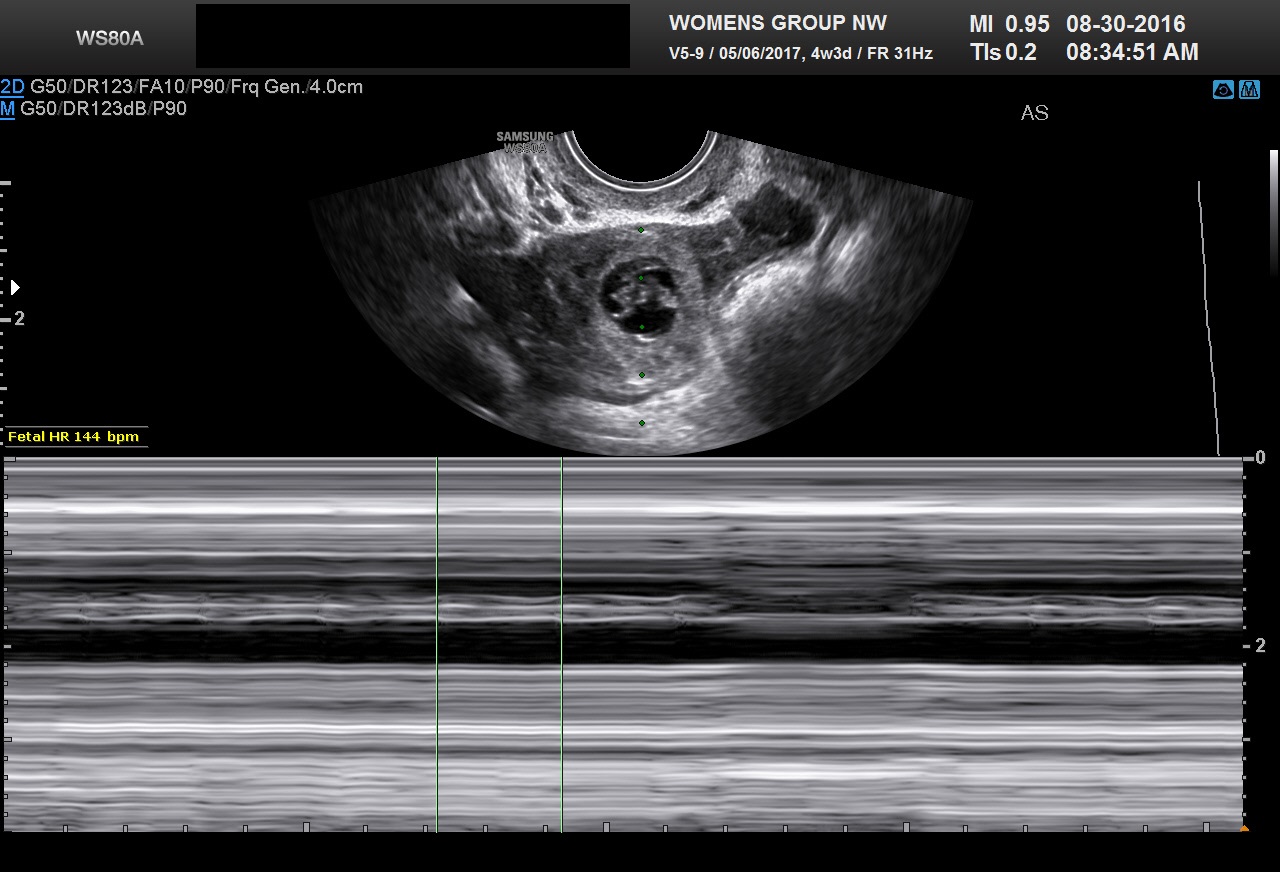 Fig 13 Ampullary tubal ectopic
Fig 13 Ampullary tubal ectopic
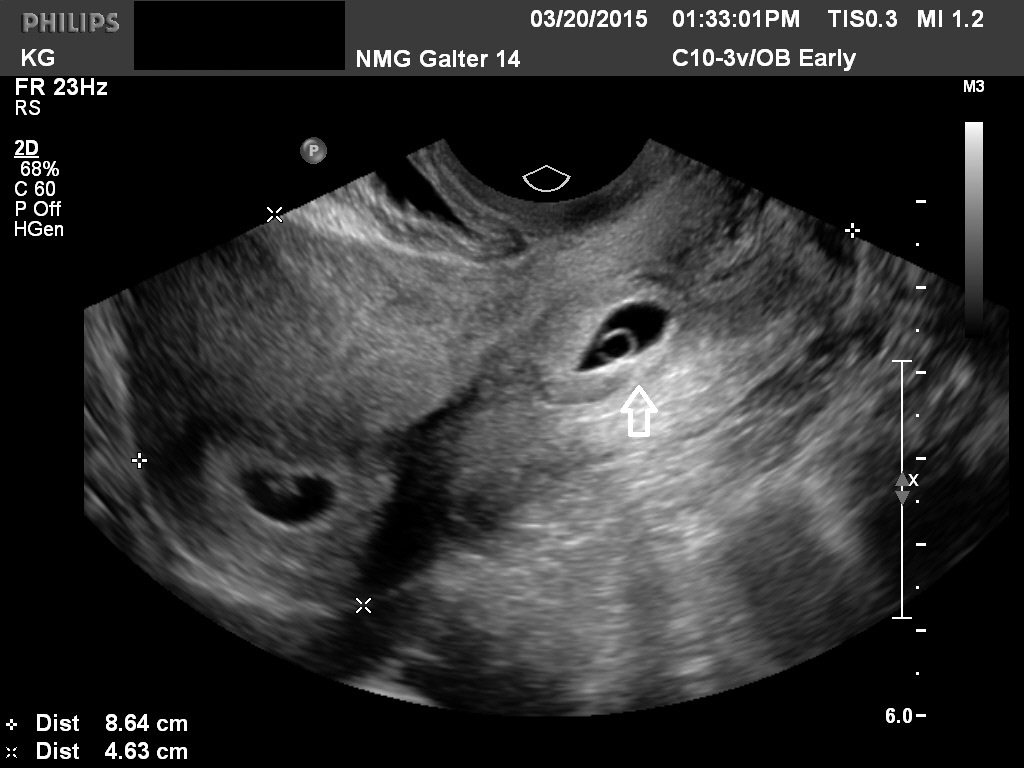 Fig 15A Concurrent cervical ectopic and intrauterine pregnancy
Fig 15A Concurrent cervical ectopic and intrauterine pregnancy
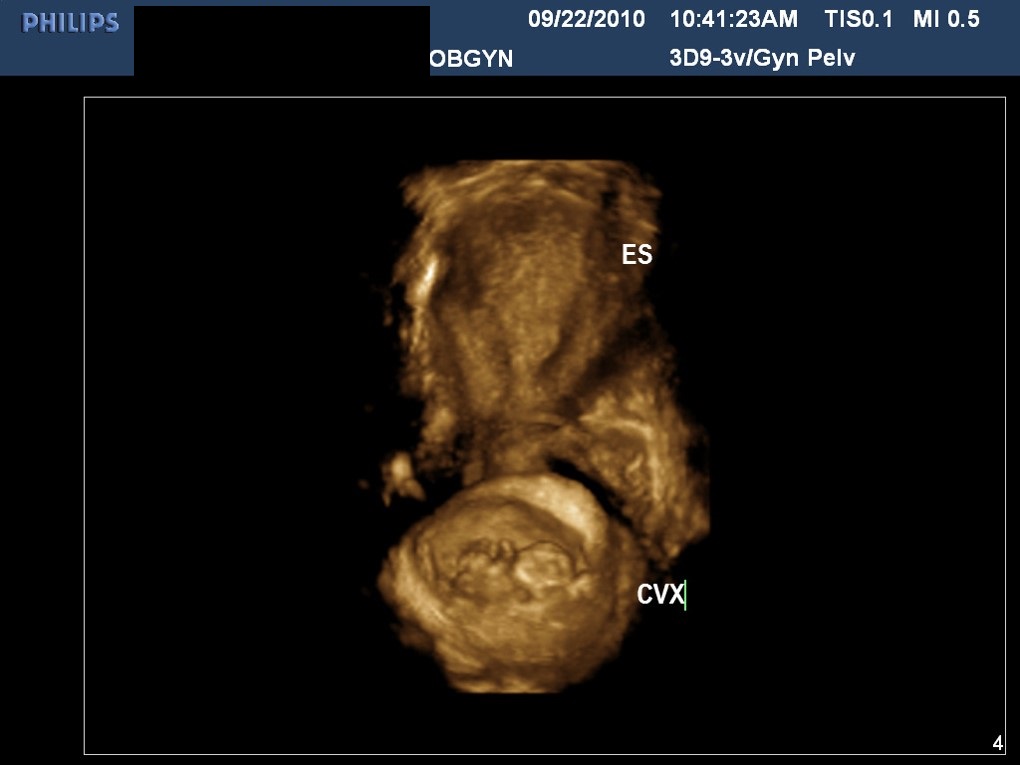 Fig 15B Three-dimensional image of a 10 week cervical pregnancy
Fig 15B Three-dimensional image of a 10 week cervical pregnancy
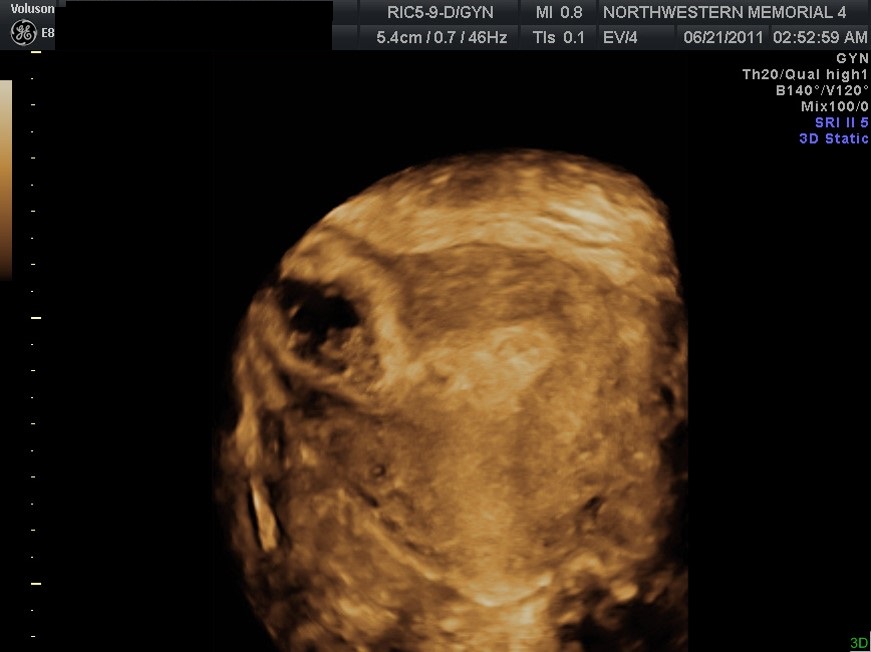 Fig 16A Interstitial ectopic. Three-dimensional is very useful in distinguishing angular pregnancies from interstitial ectopic pregnancies. Repeat studies are sometimes required at 7–10 day intervals to differentiate the two. The gestational sac will start filling the endometrial cavity for angular implantations
Fig 16A Interstitial ectopic. Three-dimensional is very useful in distinguishing angular pregnancies from interstitial ectopic pregnancies. Repeat studies are sometimes required at 7–10 day intervals to differentiate the two. The gestational sac will start filling the endometrial cavity for angular implantations
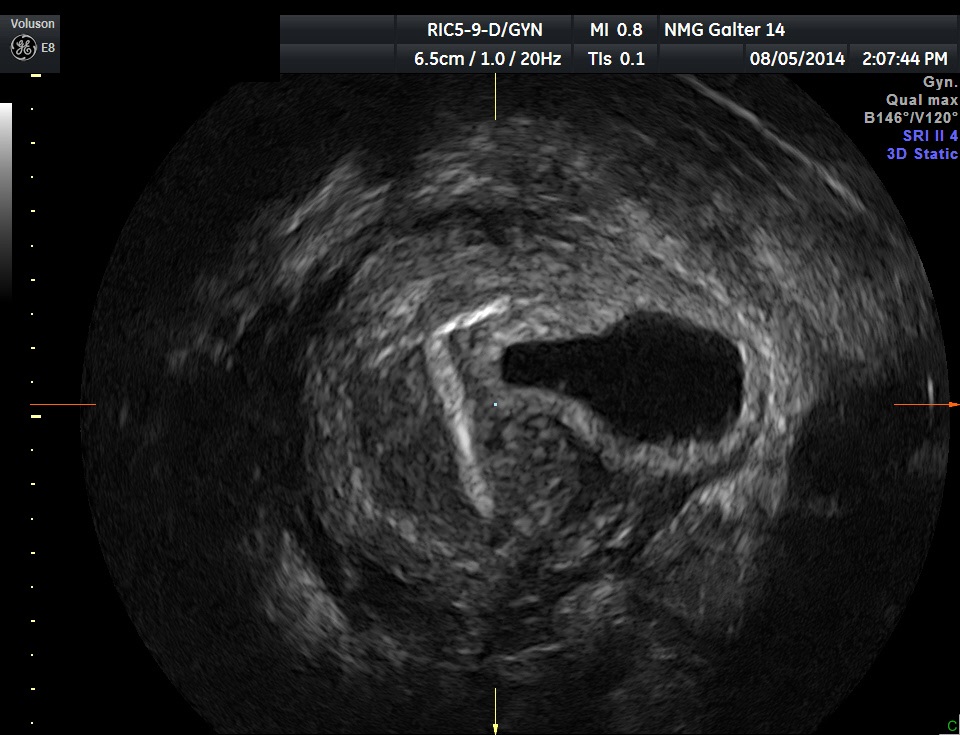 Fig 16B A interstitial ectopic pregnancy with an IUD in place
Fig 16B A interstitial ectopic pregnancy with an IUD in place
Fig 16C A normal implantation with an IUD in place. The IUD was removed. Patient had a normal term pregnancy
Fig 17 A cesarean section scar pregnancy. These cases are typically managed at our institution with intra sac methotrexate followed by standard IM dose of methotrexate
Although the pros and cons of the medical management of ectopic pregnancy with methotrexate are beyond the scope of this chapter, two points are worth making. First, LMP dating is off by at least 1 week 15% of the time. Failure to check serial titers can result in improper administration of methotrexate to patients with healthy pregnancies. Litigation has occurred in cases where methotrexate was inadvertently given to patients subsequently found to have an early intrauterine pregnancy. Second, methotrexate therapy for proven ectopic pregnancies appears to work best if the ectopic adnexal mass is less than 4 cm and the β-hCG titer less than 5000 mIU/ml. The reader is referred to the excellent paper by Lipscomb in the New England Journal of Medicine.35 Pregnancies that do not meet these criteria are more likely either to require multiple doses of methotrexate or to be unresponsive to treatment.
DATING
Crown–rump measurements at 6–10 weeks are accurate in assigning gestational age with a 95% confidence interval [CI] ± 3–5 days. This compares to biparietal diameter assessment at 16–24 weeks, which has a 95% [CI] of + 7 to 10 days. In contrast, as noted by Gardosi, LMP dating is less accurate, with a 95% CI of –9 to +27 days. The inaccuracy of LMP dating can lead to errors in assessing both preterm and post-term pregnancy rates, as well as false-positive PANAFP screens. A recent, well-referenced editorial by Gardosi discusses the inaccuracy of LMP dating and advocates routine ultrasound confirmation of dates.36
Robinson and Fleming published the first crown–rump length tables. More recent studies with timed ovulation have shown that their table underestimated gestational age by about 1 week (Table 3). Crown–rump lengths at gestational ages greater than 10 weeks are less accurate.37, 38, 39
A review article by Napalitano from 2014 summarizes and reviews the multiple articles published on crown–rump dating.40
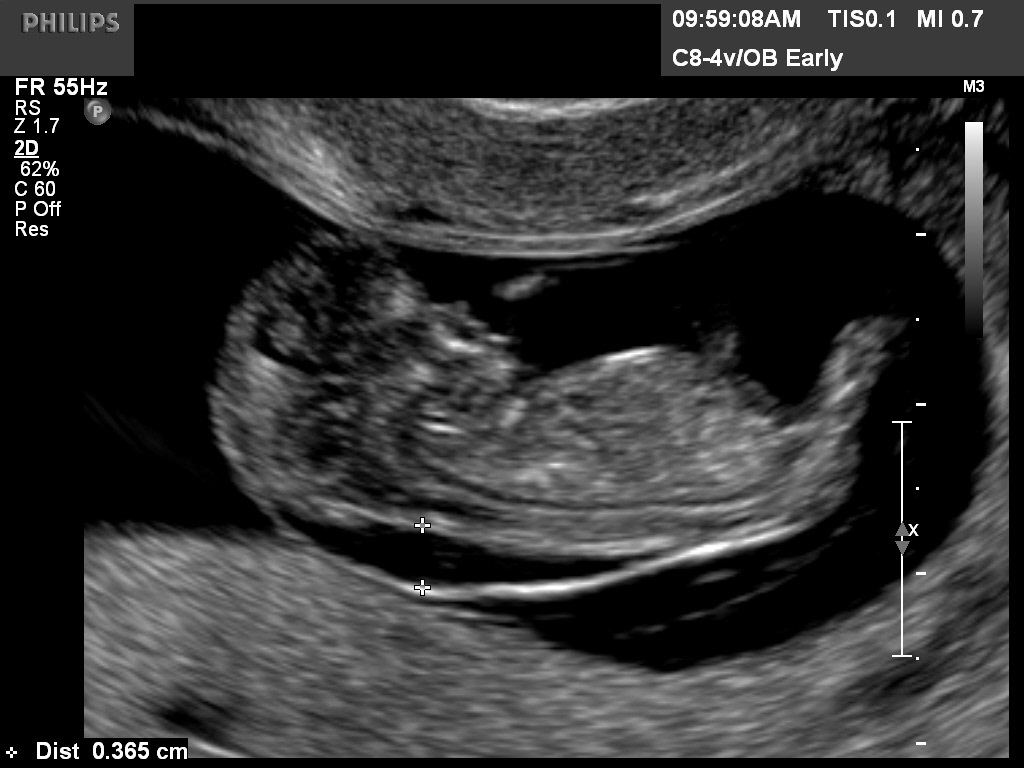
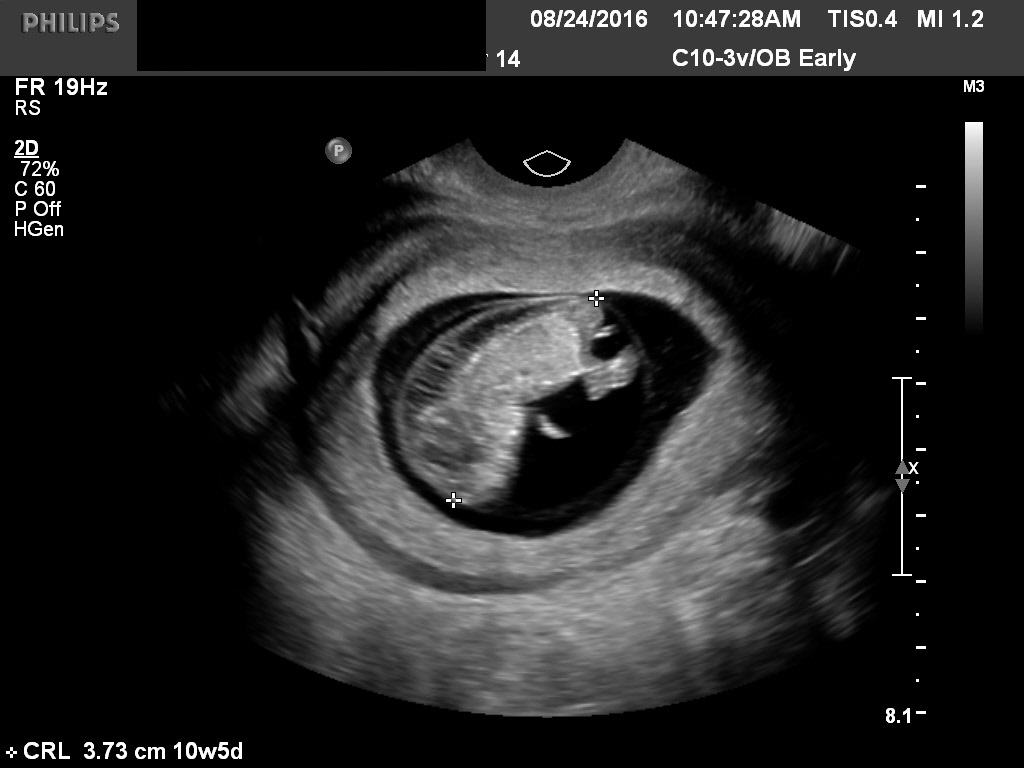
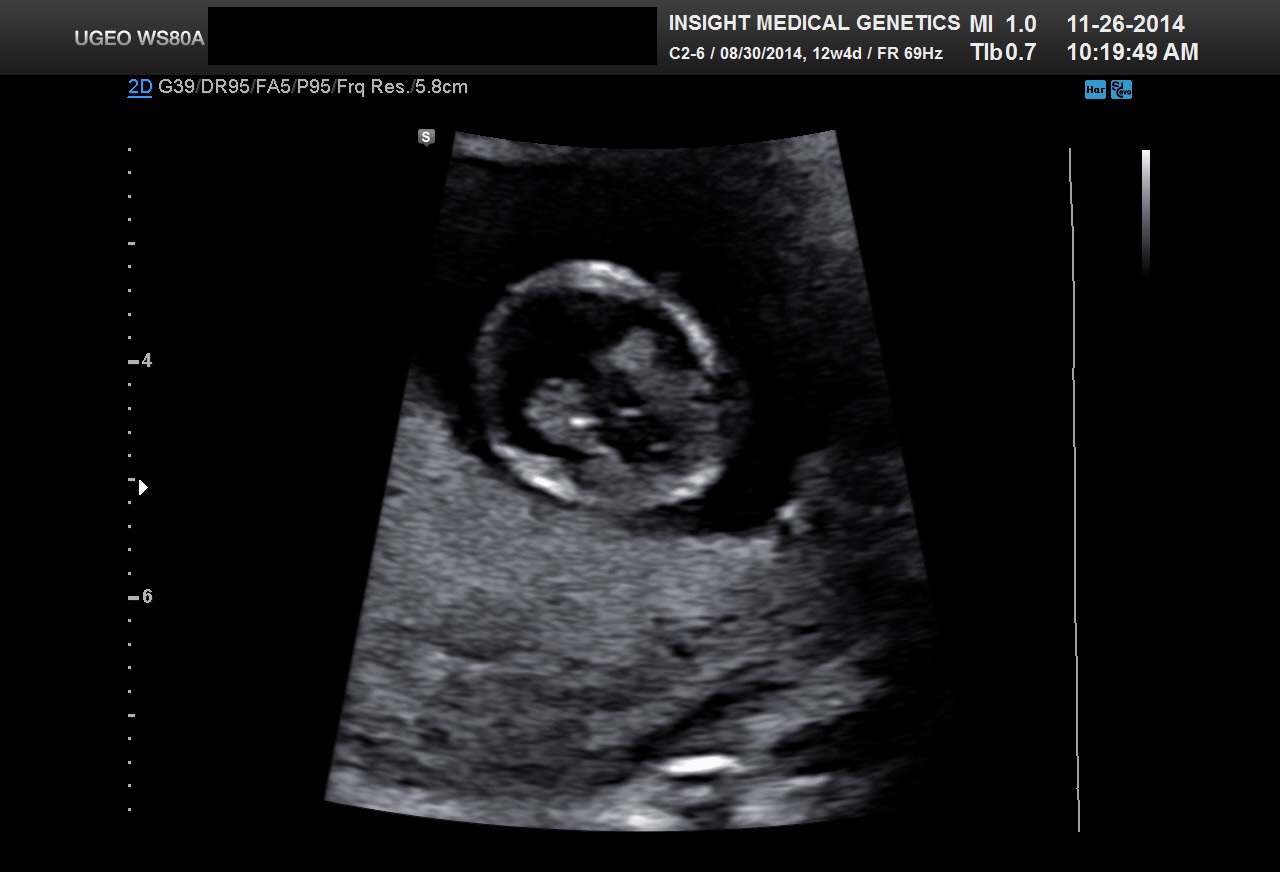
A typical 9 week CRL is shown in Figure 18. A typical 7 week CRL is shown in Figure 1D.
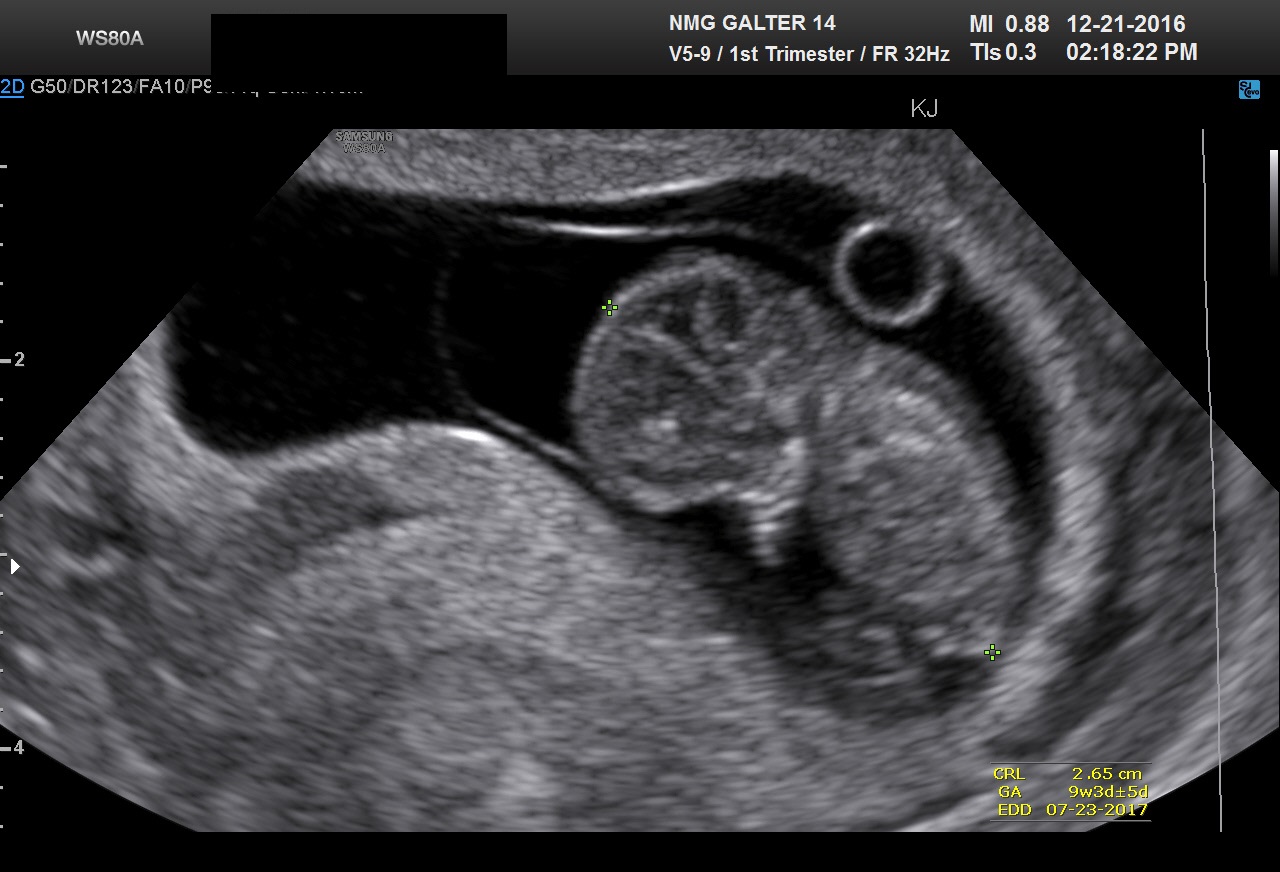 Fig 18 A 9 week CRL. Please note the fetus may start to curl and may be undermeasured. The amnion is now well seen.
Fig 18 A 9 week CRL. Please note the fetus may start to curl and may be undermeasured. The amnion is now well seen.
Table 3 Gestational age (menstrual age) estimates relative to crown–rump length
Crown–rump length (cm) | Gestational age (weeks + days) | Crown–rump length (cm) | Gestational age (weeks + days) | Crown–rump length (cm) | Gestational age (weeks + days) |
1 | 7 + 5 | 3 | 9 + 6 | 5 | 11 + 6 |
1.1 | 7 + 6 | 3.1 | 10 + 0 | 5.1 | 12 + 0 |
1.2 | 8 + 0 | 3.2 | 10 + 1 | 5.2 | 12 + 0 |
1.3 | 8 + 1 | 3.3 | 10 + 2 | 5.3 | 12 + 1 |
1.4 | 8 + 1 | 3.4 | 10 + 2 | 5.4 | 12 + 1 |
1.5 | 8 + 2 | 3.5 | 10 + 3 | 5.5 | 12 + 2 |
1.6 | 8 + 3 | 3.6 | 10 + 4 | 5.6 | 12 + 3 |
1.7 | 8 + 4 | 3.7 | 10 + 4 | 5.7 | 12 + 3 |
1.8 | 8 + 5 | 3.8 | 10 + 5 | 5.8 | 12 + 4 |
1.9 | 8 + 5 | 3.9 | 10 + 6 | 5.9 | 12 + 4 |
2 | 8 + 6 | 4 | 10 + 6 | 6 | 12 + 5 |
2.1 | 9 + 0 | 4.1 | 11 + 0 | 6.1 | 12 + 6 |
2.2 | 9 + 1 | 4.2 | 11 + 1 | 6.2 | 12 + 6 |
2.3 | 9 + 1 | 4.3 | 11 + 1 | 6.3 | 13 + 0 |
2.4 | 9 + 2 | 4.4 | 11 + 2 | 6.4 | 13 + 0 |
2.5 | 9 + 3 | 4.5 | 11 + 3 | 6.5 | 13 + 1 |
2.6 | 9 + 4 | 4.6 | 11 + 3 | 6.6 | 13 + 2 |
2.7 | 9 + 4 | 4.7 | 11 + 4 | ||
2.8 | 9 + 5 | 4.8 | 11 + 5 | ||
2.9 | 9 + 6 | 4.9 | 11 + 5 |
(Adapted from MacGregor SN, Tamura RK, Sabbagha RE et al: Underestimation of gestational age by conventional crown-rump length dating curves. Obstet Gynecol 70:344, 1987)
MULTIPLE GESTATION
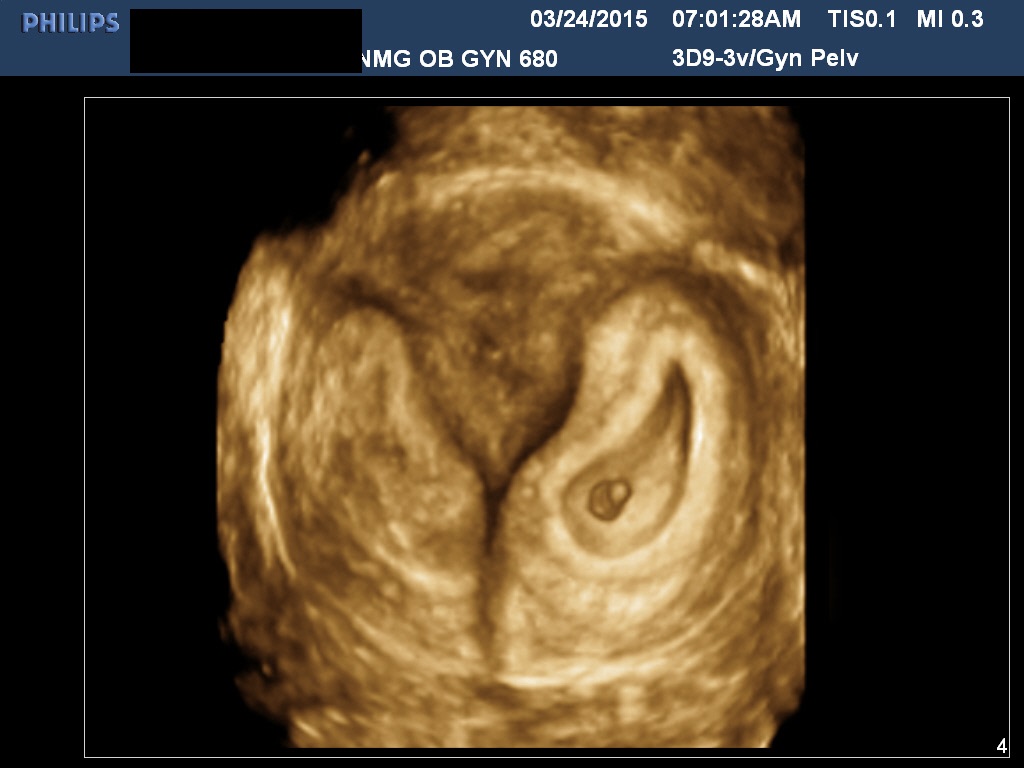
The determination of chorionicity of multiple gestations is of obvious interest to the obstetrician because of the greatly increased morbidity and mortality in monochorionic pregnancies and in particular monoamniotic–monochorionic twin pregnancies. In a well-illustrated study, Monteagudo and co-workers demonstrated the extreme reliability of first-trimester ultrasound in predicting chorionic and amniotic type (Figs. 19 and 20).41 Sepulveda and associates, in a series of 288 twins, correctly identified all 63 monochorionic twins at 10–14 weeks using the lambda sign, which is a triangular projection of placenta where dichorionic placentas meet (Fig 11).42 Sepulveda and colleagues also described the ipsilon zone, where the chorionic membranes converge centrally, which is useful in identifying the chorionicity of most triplet pregnancies (Fig 12).43
Fig 19A and B Two diamniotic monochorionic pregnancies. The amnions may not be well seen until 8–9 weeks
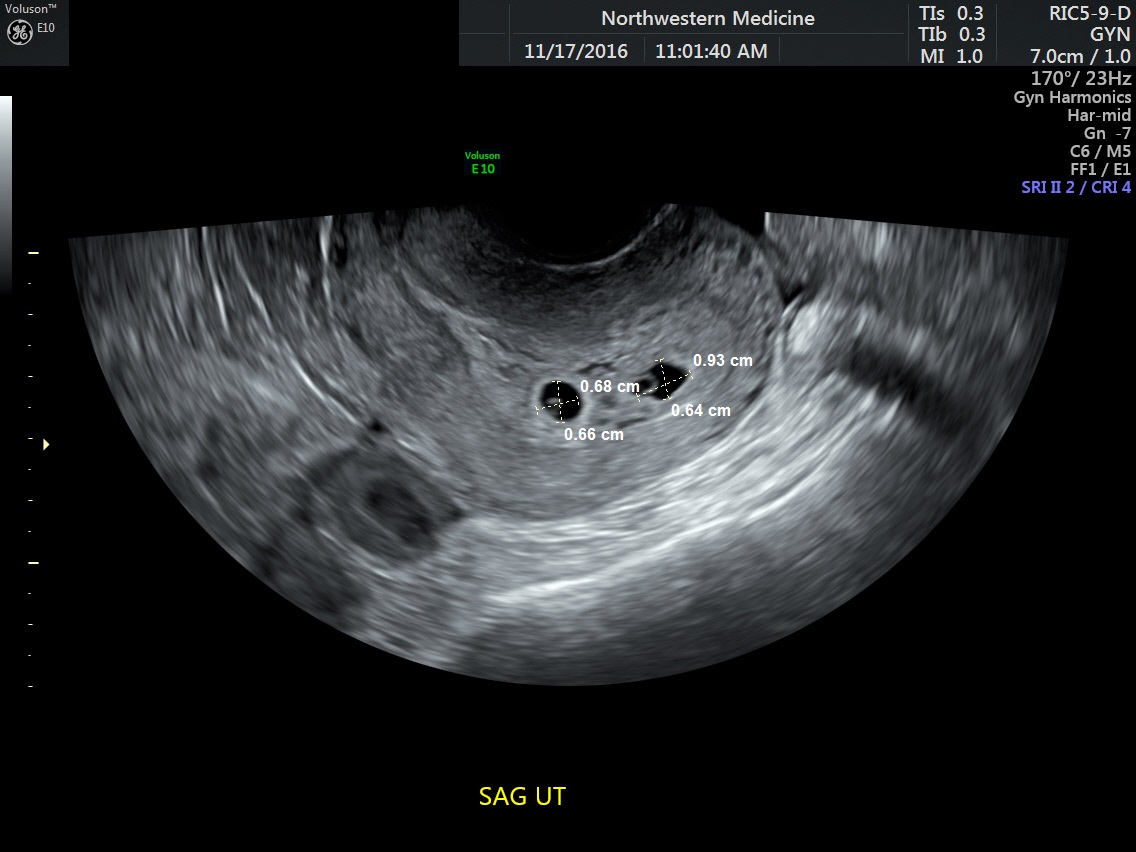 Fig 20A 6-week dichorionic pregnancy. Two separate gestational sacs clearly seen
Fig 20A 6-week dichorionic pregnancy. Two separate gestational sacs clearly seen
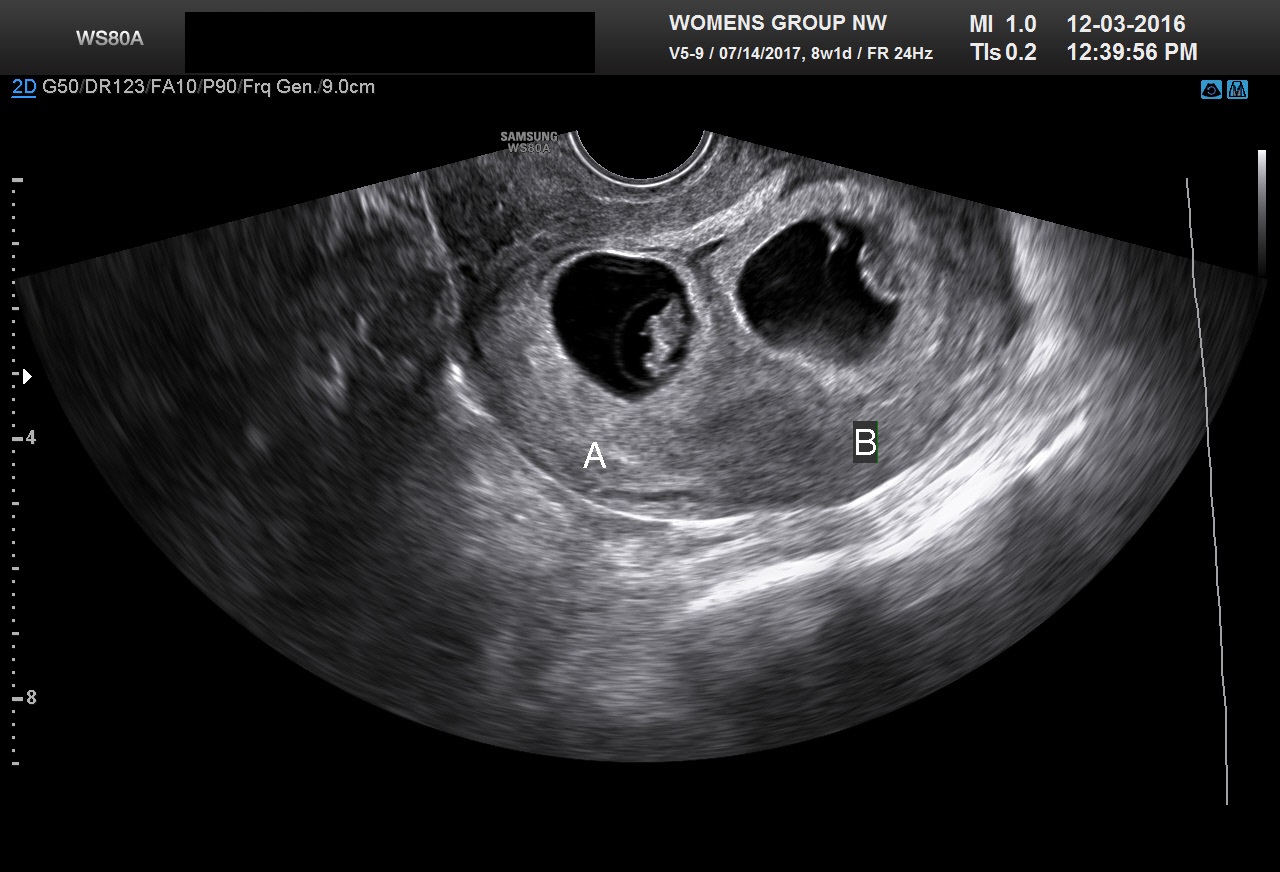 Fig 20B 7-week dichorionic pregnancy. Two separate gestational sac sacs clearly seen
Fig 20B 7-week dichorionic pregnancy. Two separate gestational sac sacs clearly seen
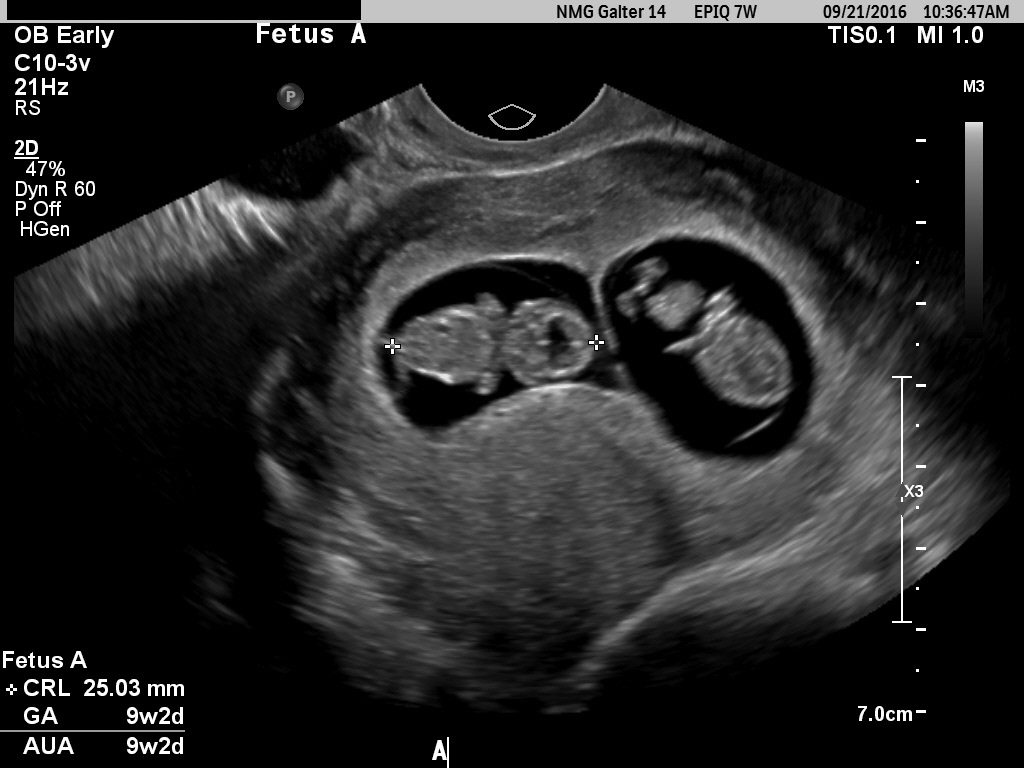 Fig 20C 9-week dichorionic pregnancy. The delta sign clearly identifies a dichorionic pregnancy
Fig 20C 9-week dichorionic pregnancy. The delta sign clearly identifies a dichorionic pregnancy
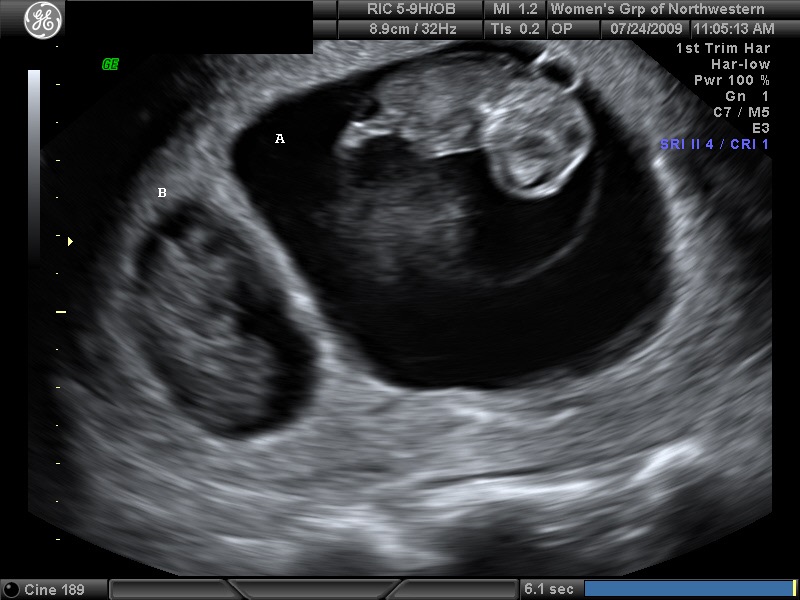 Fig 20D A demised second twin is displayed
Fig 20D A demised second twin is displayed
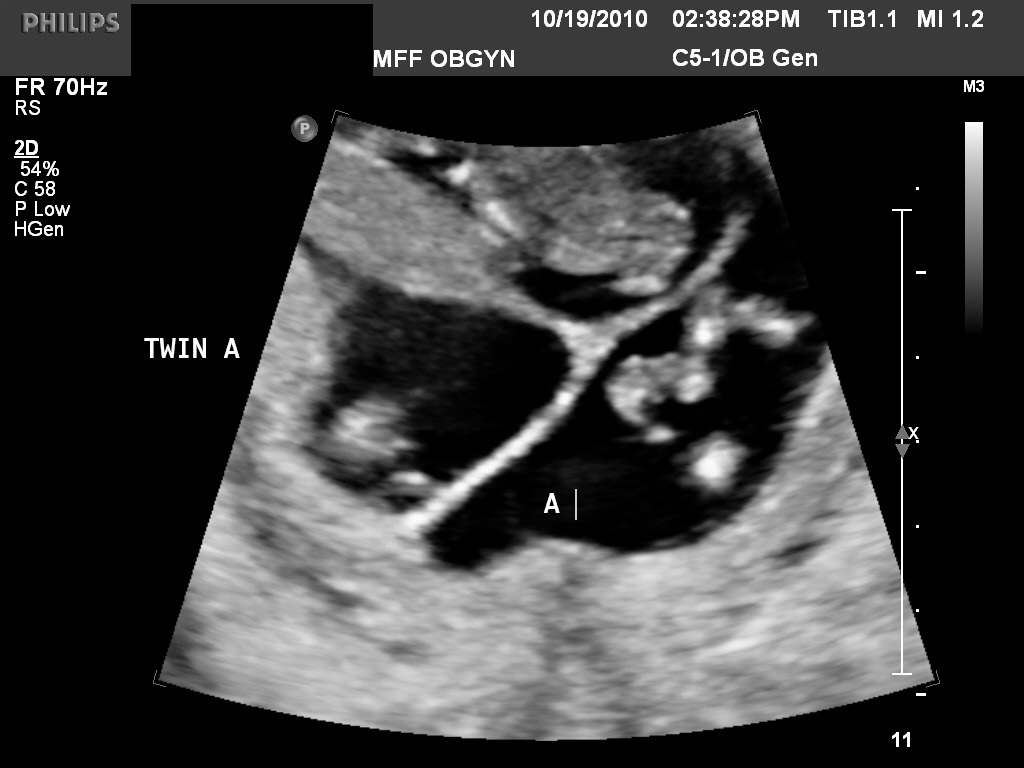 Fig 20E A ipsilon sign in a triplet pregnancy
Fig 20E A ipsilon sign in a triplet pregnancy
TRISOMY SCREENING
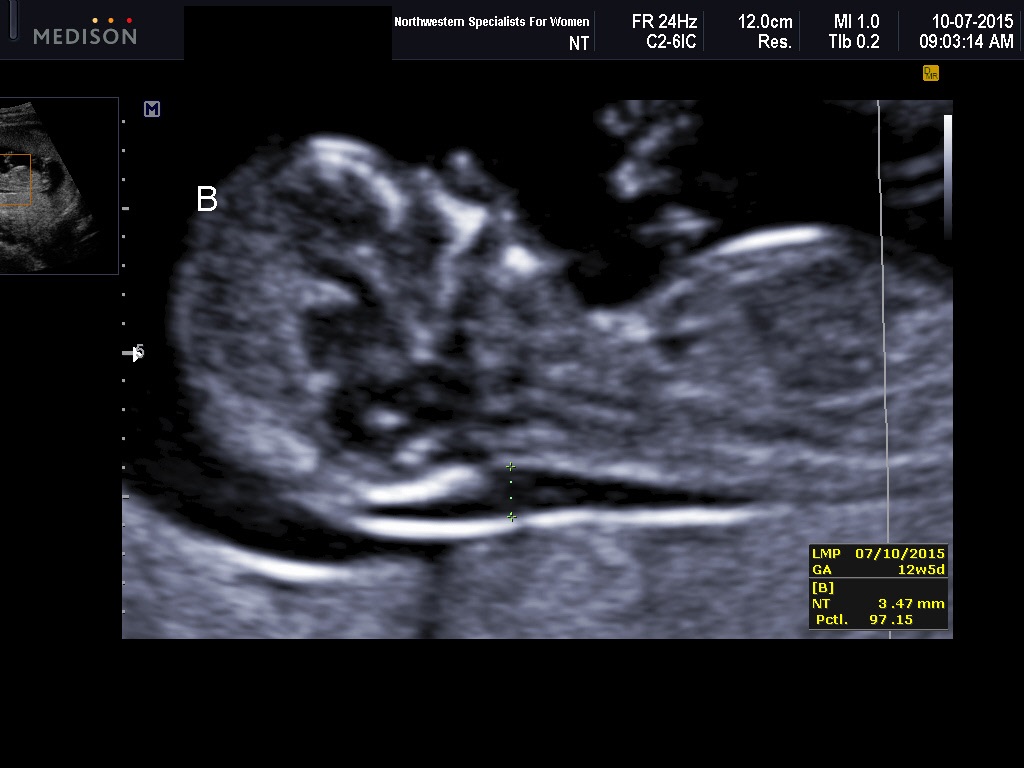
From the mid 1990s until the present nuchal screening has been combined in various paradigms with biochemical markers to screen for fetal trisomy. Pandya and co-workers, in a study of 20,804 English women scanned at 10–14 weeks' gestation, achieved an 80% detection for trisomy with 5% of the population being identified at risk.44 In another study of 1303 Italian women less than 35 years of age, Orlandi and associates found combination of first-trimester nuchal translucency thickness measurement at 10–13 weeks and biochemical markers (free β-hCG and PAPP-A) yielded an 87% sensitivity for trisomy 21, with a false-positive rate of 5%. In the same group, the detection rate for trisomy 18 was 76%, with a 1% false-positive rate.45, The issue of training sonographers and sonologists in obtaining nuchal translucency measurements was reviewed by Braithwaite and colleagues.46, The publication of the BUN study and FASTER study in the United States have demonstrated the role of both first trimester screening and sequential first and second trimester screening for fetal trisomy.47, 48 With the availability of free DNA testing many women over 35 are choosing free DNA screening.49 Recent data suggest that the false positive rate may be low enough to include younger women.49 The exact role of nuchal screening in women having free DNA testing remains to be determined.50
Examples of abnormal nuchals are given in Figures 21, 22 and 23.
Fig 21A A fetus with trisomy 21 confirmed by CVS. The nuchal measurement is enlarged. No nasal bone was identified
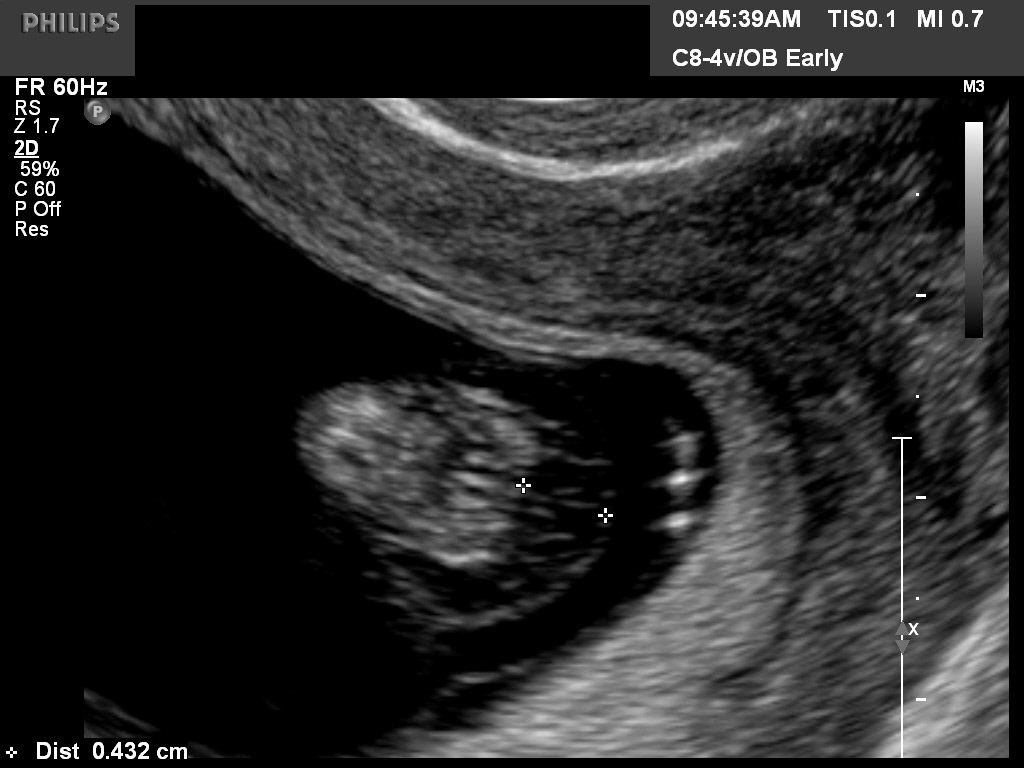 Fig 21B A septated hygroma is noted on an axial view of the neck. CVS revealed trisomy 21. In our lab nuchal measurements greater than 3.0 are counselled for CVS. If the genetics is normal, the patient is referred for early anatomic survey including pediatric fetal echo
Fig 21B A septated hygroma is noted on an axial view of the neck. CVS revealed trisomy 21. In our lab nuchal measurements greater than 3.0 are counselled for CVS. If the genetics is normal, the patient is referred for early anatomic survey including pediatric fetal echo
Fig 22 A fetus confirmed trisomy 18. Nuchal screening revealed a nuchal measurement of 3.5 mm. Micrognathia noted
Fig 23 A fetus with a large septated hygroma extending to the lower back
DIAGNOSIS OF FETAL ABNORMALITIES
The early anatomic survey at 11–14 weeks well reviewed in 1997 by Souka and Nicolaides.51 They found early survey holds some promise for the early detection of major anomalies. However, a review by Rossi and Prefumo in 2013 of 19 studies found that the sensitivity for the detection of major anomalies was less than ideal compared to the later anatomic surveys.52 It is important to note that a multicenter paper by Syngelaki et al., found that the 11–13 week exam performed abdominally at the time of nuchal screening was 100% sensitive for certain major anomalies. This included acrania, alobar holoprosencephaly, gastroschisis, megacystis, and body stalk anomalies. However only 34% of major congenital heart defects were detected.53 A recent paper by Iliescu et al. suggest that if both transabdominal and transvaginal techniques are used and if Doppler of the fetal heart is utilized, that 76% of all major defects and 90% of major congenital heart defects can be detected.54 It should also be remembered that chromosomally normal fetuses with increased nuchal translucency are at increased risk for cardiac abnormalities. About 20% of major congenital heart defects can be found as a result of this screen.55, 56
Screening for congenital anomalies at this gestational age requires additional training. ISUOG Practice Guidelines for performing the first-trimester exam are available at their website.57 The reader is referred to the review by Yagel and co-workers, who outlined the limitations of early pregnancy scanning for fetal anomalies.58 The training of sonographers to perform these transvaginal studies has been reviewed by Timor-Tritsch et al.59
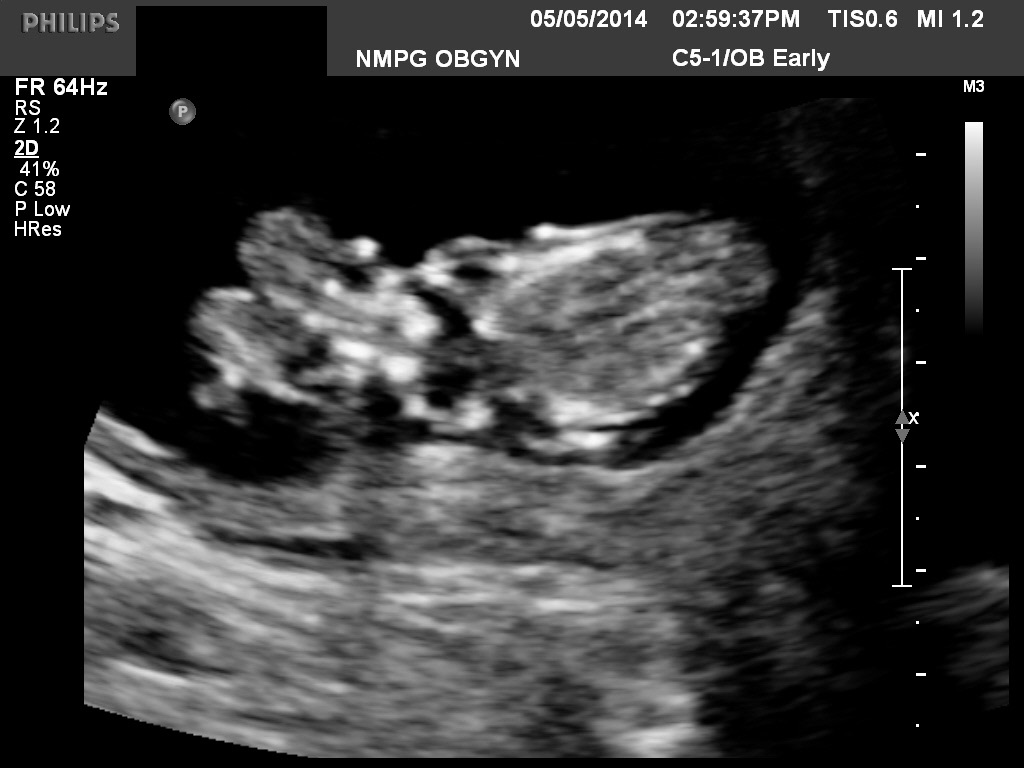 Fig 24 A fetus with acrania at 13 weeks
Fig 24 A fetus with acrania at 13 weeks
Fig 25A and B Two fetuses with omphalocoeles at 13 weeks. Invasive testing is recommended in these fetuses to exclude trisomy 18
Fig 26 A fetus at 12 weeks with holoproscencephaly. Invasive testing revealed trisomy 13
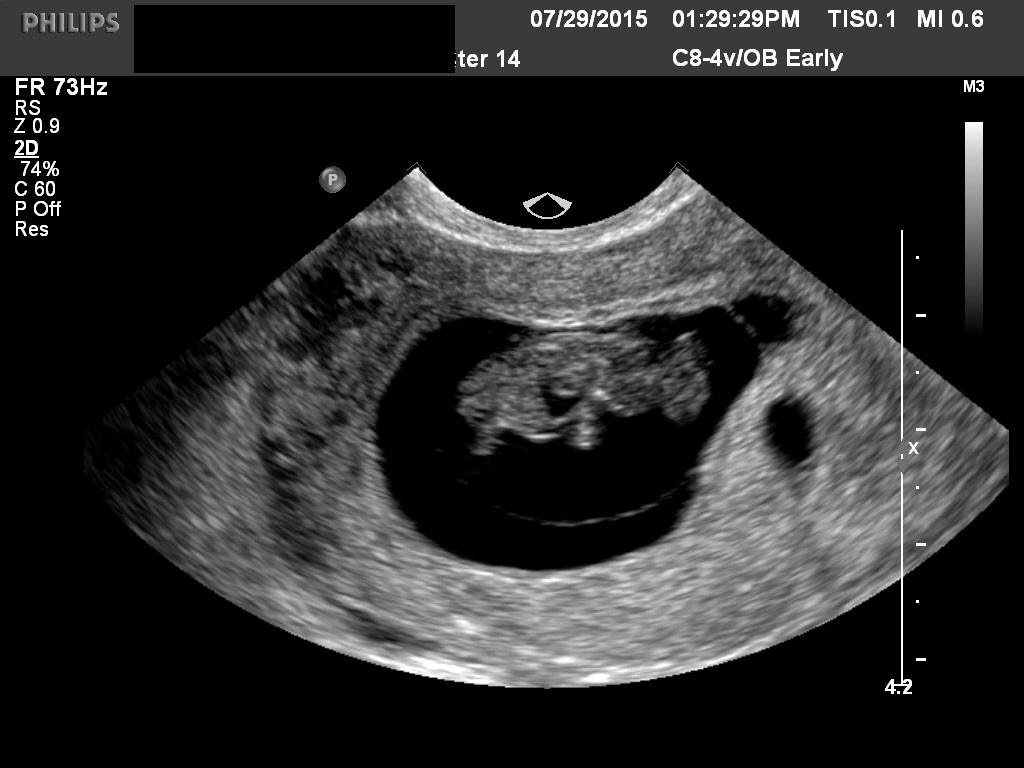 Fig 27 A fetus with acrania and pericardial effusion.
Fig 27 A fetus with acrania and pericardial effusion.
MAJOR UTERINE ANOMALIES
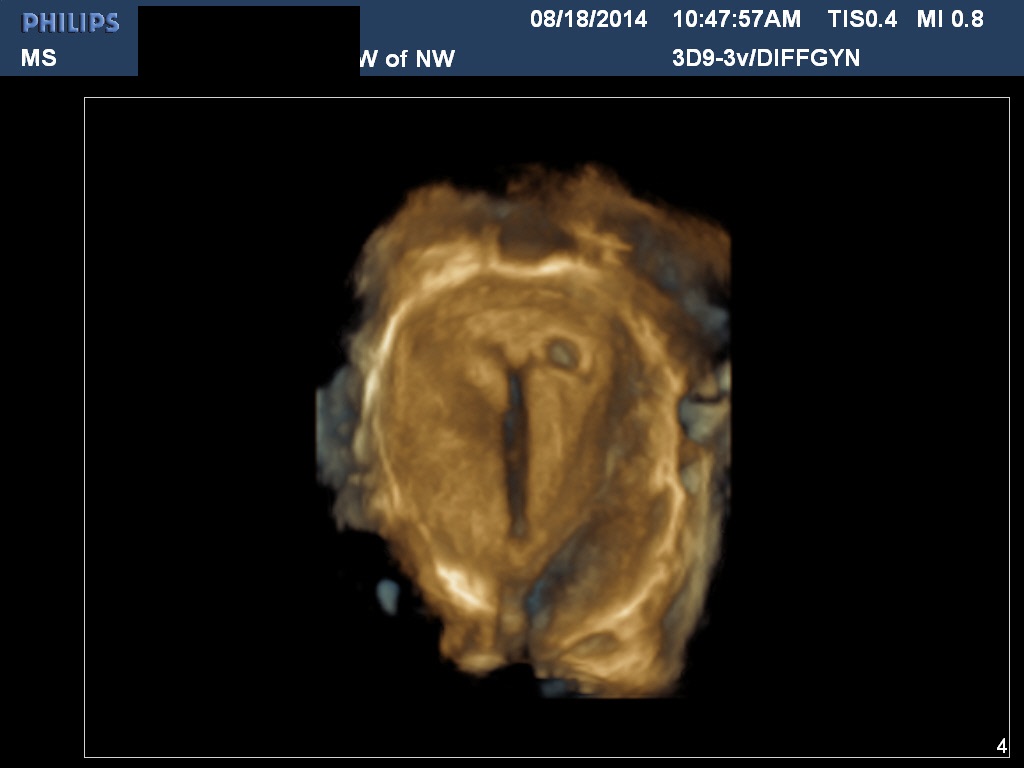
Major uterine anomalies are not infrequently diagnosed during the first-trimester ultrasound examination. The bicornuate uterus is characterized by its widened transverse diameter and a notched fundus. The septated uterus has a normal uterine contour but a septated endometrial cavity. Three-dimensional imaging has markedly simplified the detection and classification of suspect uterine anomalies.
Fig 28 Three-dimensional image. Septated uterus. Pregnancy is to left of septum
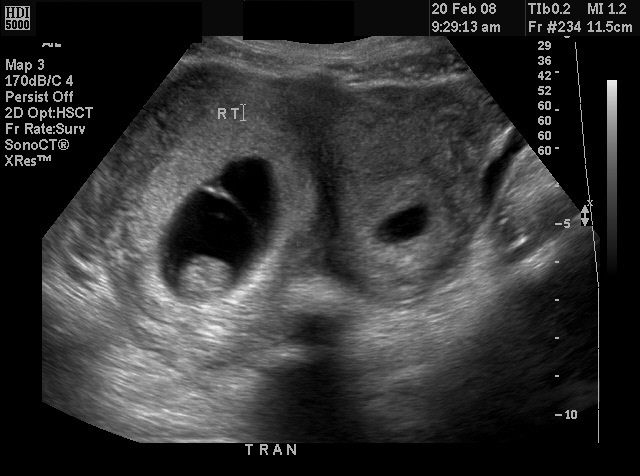 Fig 29A Three-dimensional image. Didelphus uterus. Pregnancy is in right horn
Fig 29A Three-dimensional image. Didelphus uterus. Pregnancy is in right horn
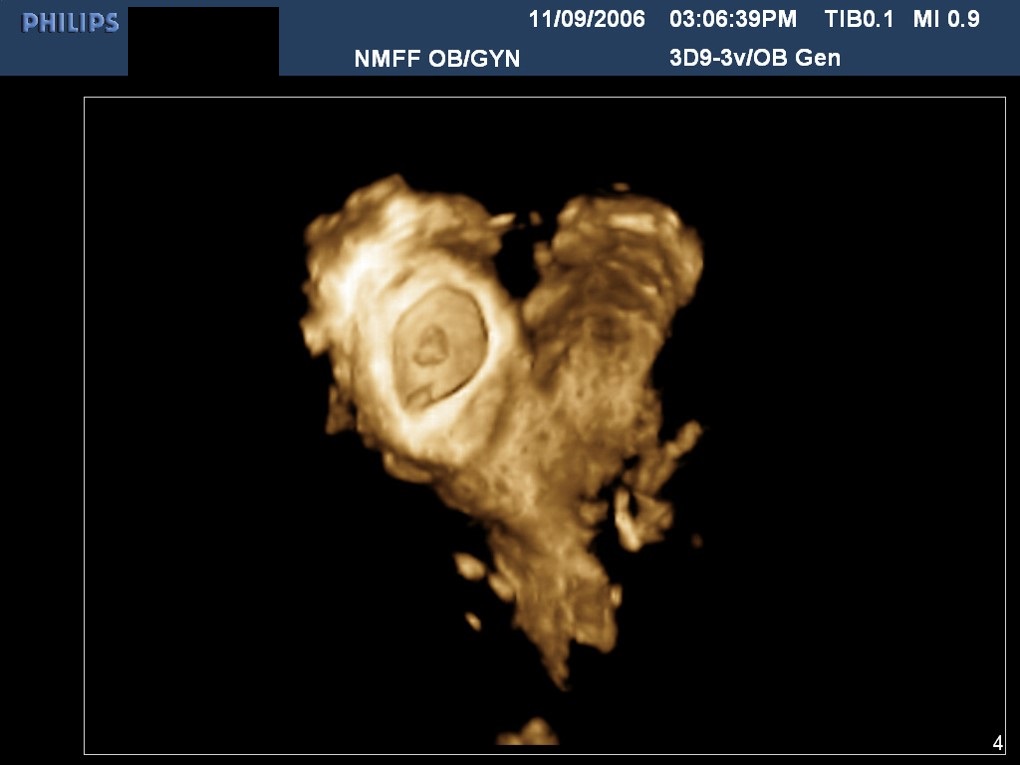 Fig 29B Three-dimensional image. Bicornuate uterus. Pregnancy is in right horn
Fig 29B Three-dimensional image. Bicornuate uterus. Pregnancy is in right horn
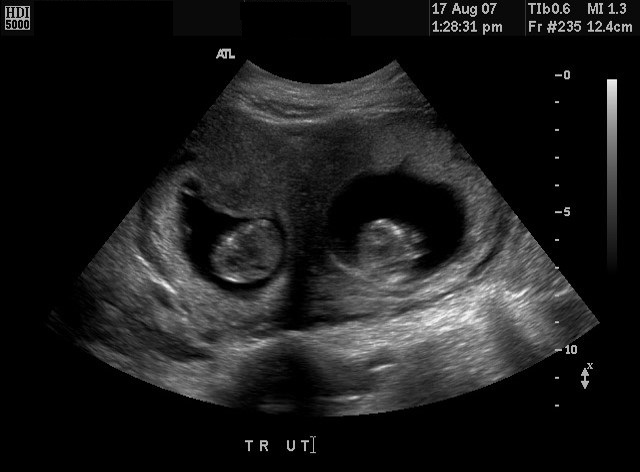 Fig 29C 2D image. Bicornuate. Twin pregnancy. One fetus is in each horn
Fig 29C 2D image. Bicornuate. Twin pregnancy. One fetus is in each horn
MOLAR PREGNANCY
The characteristic “grape-like clusters” or vesicular pattern seen in molar pregnancy is easily identified on TVS (Fig 30). In rare cases, a molar pregnancy will be noted concomitant with a normal twin pregnancy. For a discussion of the management of these cases, the reader is referred to the article by Fishman and associates (Fig 31).60
Fig 30 Typical ultrasound appearance of a complete molar pregnancy
Fig 31 A normal twin (placenta not in view) shown adjacent to a molar pregnancy
REFERENCES
Warren WB, Timor-Tritsch I, Peisner DB et al: Dating the early pregnancy by sequential appearance of embryonic structures. Am J Obstet Gynecol 161: 747, 1989 |
|
Fossum GT, Davajan V, Kletzy OA: Early detection of pregnancy with transvaginal ultrasound. Fertil Steril 49: 788, 1988 |
|
Doubilet PM and Benson CB: Further evidence against the reliability of human chorionic gonadotropin discriminatory level. J Ultrasound Med 2011; 30:1637-1642. |
|
Ko J, Cheung V. Time to revisit the human chorionic gonadotropin discriminatory level in the management of pregnancy of unknown location. J Ultrasound Med 2014;33:465-471. |
|
Richardson A, Gallos I, Dobson S et al. Accuracy of first trimester ultrasound in diagnosis of intrauterine pregnancy prior to visualization of the yolk sac: a systematic review and meta-analysis. Ultrasound Obstet Gynecol 2015; 46:142-149.. |
|
Richardson A, Hopkisson J, Campbell B, Raine-Fenning N. Use of double decidual sac sign to confirm intrauterine pregnancy location prior to ultrasonographic visualization of embryonic contents: a diagnostic accuracy study. Ultrasound Obstet Gynecol 2016 May 19. doi 10:1002/uog.15966. [Epub ahead of print] |
|
Benson CB , Doubilet PM, Peters HE, and Frates MC:Intrauterine fluid with ectopic pregnancy. J Ultrasound Med 2013; 32:389-393. |
|
Perkins KY, Johnson JL, Kay Helen HK: Simple ovarian cysts: Clinical features on first trimester ultrasound. J Reprod Med 1997; 42: 440-444. |
|
Bromley B, Benacerraf B: Adnexal masses during pregnancy: Accuracy of sonographic diagnosis. J Ultrasound Med 1997; 16: 447-452.. |
|
Chiang G and Levine D. Imaging of adnexal masses in pregnancy. J Ultrasound Med 2004; 23:805-819. |
|
Barbieri M, Somigliana E, Oneida S et al. Decidualized ovarian endometriosis in pregnancy : a challenging diagnostic entity. Human Reproduction 2009; 24:1818-1824. |
|
Mascilini F, Savelli L, Scifo MC et al .Ovarian masses with papillary projections diagnosed and removed during pregnancy:ultrasound features and histological diagnoses. Ultrasound Obstet Gynecol 2016 Aug 3. doi 10.1002/uog.17216. [Epub ahead of print] |
|
Yacobozzi M, Nguyen D, Rakita D. Adnexal Masses in Pregnancy. Sem Ultrasound CT MR 2012; 33:55-64. |
|
Ball RH, Ade CM, Schoenborn JA, Crane J: The clinical significance of ultrasonographically detected subchorionic hemorrhages. Am J Obstet Gynecol 1966;174: 996-1002. |
|
Tuuli MG, Norman SM, Odibo AO, Macones GA, Cahill AG.Perinatal outcomes in women with subchorionic hematoma: a systematic review and meta-analysis. Obstet Gynecol 2011;117:1205-1212. |
|
Tan S, Tangal NG,Kanat-Pektas M et al. Abnormal sonographic appearances of the yolk sac: which can be associated with adverse perinatal outcome? Medical Ultrasound 16:15-20,2014. |
|
Doubilet PM, Benson CB. First do no harm… too early pregnancies. J Ultrasound Med 2010; 29:685-689. |
|
Abdallah Y, Daemen A, Kirk et al. Limitations of the current definitions of miscarriage using mean gestational sac diameter and crown-rump length measurements: a multicenter observational study. Obstet Gynecol 2011; 38:497-452. |
|
Pennell RG, Needleman L, Pajak T et al: Prospective comparison of vaginal and abdominal ultrasound in normal early pregnancy. J Ultrasound Med 1991; 10: 63-67. |
|
Falco P, Milano V, Pilu G et al: Sonography of pregnancies in with first trimester bleeding and a viable embryo: A study of prognostic indicators by logistic regression. Ultrasound Obstet Gynecol 1996; 7: 165-169. |
|
Goldstein I, Zimmer EA, Tamir A et al: Evaluation of normal gestational sac growth: Appearance of embryonic heartbeat and embryo movements using transvaginal technique. Obstet Gynecol 1991; 77: 885-888. |
|
Doubilet PM, Benson CB, Bourne T, Blaivas M. Diagnostic Criteria for nonviable pregnancy early in the first trimester. New England Journal 2013; 369:1443-1446. |
|
Jeve Y, Rana R, Bhide A, Thangaratinam S. Accuracy of first trimester ultrasound in the diagnosis of early embryonic demise: a systematic review. Ultrasound Obstet Gynecol 2011;38:489-496,2011. |
|
Rulin MC, Bornstein SG, Campbell JD: The reliability of ultrasonography in the management of spontaneous abortion thought to be complete: A prospective study. Am J Obstet Gynecol 1993;168: 12-15. |
|
Cowett AA, CohenLS, Lichtenberg ES, Stika CS. Ultrasound evaluation of the endometrium after medical termination of pregnancy. Obstet Gynecol 2004; 103:871-875. |
|
Tal J, Haddad S, Gordon N, Timor-Tritsch I: Heterotopic pregnancy after ovulation induction and assisted reproduction and assisted reproductive technology: A literature review from 1971–1993. Fertil Steril 1996: 66: 1-12. |
|
Kadar N, Devore G, Romero R: Discriminatory beta-hCG zone: Its use in the sonographic evaluation of ectopic pregnancy. Obstet Gynecol 1981; 58: 156-161. |
|
Perkins KY, Johnson JL, Kay Helen HK: Simple ovarian cysts: Clinical features on first trimester ultrasound. J Reprod Med 42: 440, 1997 |
|
Condous G, Van Calster B, Kirk E et al. Prediction of ectopic pregnancy in women with a pregnancy of unknown location. Ultrasound Obstet Gynecol 2007; 29: 680-687. |
|
Barnhart KT, Samuel MD, Rinaudo PF et al. Symptomatic patients with early viable intrauterine pregnancy:HCG curves redefined. Obstet Gynecol 2004; 104:50-55. |
|
Stovall TG, Ling FW, Gray LA: Single dose methotrexate for the treatment of ectopic pregnancy. Obstet Gynecol 1991;77: 754-777. |
|
Timor-Tritsch IE, Yeh MN, Peisner DB et al: The use of transvaginal ultrasound in the diagnosis of ectopic pregnancy. Am J Obstet Gynecol 1989; 161: 157-161. |
|
Practice Committee of Society of Reproductive Medicine .Medical Treatment of Ectopic Pregnancy: A Committee Opinion. Fertility Sterility 2013;100:638-644. |
|
Marret H, Fauconnier A, Dubemard G, et al. Overview of guidelines of off-label use of methotrexate in ectopic pregnancy: report of CNGOF. Eur J Obstet Gynecol Reprod Bio 2016;205:105-109. |
|
Lipscomb GH, McCord ML, Stovall TG et al. Predictors of methotrexate treatment in women with tubal ectopic pregnancy. N Engl J Med 1999; 341:1974-1978. |
|
Gardosi J: Dating of pregnancy: Time to forget the last menstrual period. Ultrasound Obstet Gynecol 1997; 9: 367-368. |
|
Robinson HP, Fleming JEE: A critical evaluation of sonar “crown-rump length” measurements. Br J Obstet Gynaecol 1975; 82: 702-710. |
|
MacGregor SN, Tamura SK, Sabbagha RE et al: Underestimation of gestational age crown-rump length dating curves. Obstet Gynecol 1987; 70: 344-348. |
|
Rossavik IK, Torjusen GO, Gibbons WE: Conceptual age and ultrasound measurements of gestational sac and crown-rump length in vitro fertilization pregnancies. Fertil Steril 1988; 49: 1012. |
|
Naplitano R,Dhami J, Ohuma EO et al. Pregnancy dating by fetal crown-rump length: a systematic review of charts. BJOG 2014; 121:556-665. DOI:10.1111/1471-0528.12478. |
|
Monteagudo A, Timor-Tritsch IE, Sharma S: Early and simple determination of chorionic and amniotic type in multifetal pregnancy in the first fourteen weeks of pregnancy by high frequency transvaginal ultrasound. Am J Obstet Gynecol 1994; 170: 824-829. |
|
Sepulveda W, Sebire NJ, Hughes K et al: The lambda sign at 10–14 weeks gestation as a predictor of chorionicity in twin pregnancies. Ultrasound Obstet Gynecol 1996; 7: 421-423. |
|
Sepulveda W, Sebire NJ, Psarra A et al: Prenatal detection of chorionicity of triplet pregnancy by ultrasonographic examination of the ipsilon zone. Obstet Gynecol 1996; 88: 855-858. |
|
Pandya PP, Snidjers RJM, Johnson SJ et al: Screening for fetal trisomies by maternal age and fetal nuchal translucency at 10 to 14 weeks gestation. Br J Obstet Gynaecol 1995;102: 957-962. |
|
Orlandi F, Damiani G, Hallahan TW et al: First trimester screening for fetal aneuploidy: Biochemistry and nuchal translucency. Ultrasound Obstet Gynecol 1997;10: 381-386. |
|
Braithwaite JM, Kadir RA, Pepera TA et al: Nuchal translucency measurements: Training of potential examiners. Ultrasound Obstet Gynecol 1996 ;8: 192-195. |
|
Wapner R, Thom E, Simpson JL et al. First-trimester screening for trisomies 21 and 18. N Engl J Med 2003;349:1405-13 |
|
Malone FD, Canick JA, Ball RH et al: First- and second-trimester screening or both, for Down's syndrome. N Engl J Med 2004; 353: 2001-11 |
|
Norton M.E., Jacobsson B., Swamy G.K. et al. Cell-free DNA analysis for non-invasive examination of trisomy. NEJM 2015; 372: 1589-1597. |
|
Reiff ES, Little SE, Dobson L, et al. What is the role of the 11 – 14 week ultrasound in women with negative cell-free DNA screening for aneuploidy? Prenat Diagn; 2016:36:260-265. |
|
Souka AP, Nicolaides: Diagnosis of fetal abnormalities at the 10–14 week scan. Ultrasound Obstet Gynecol 1997;10: 429-442. |
|
Rossi AC and Prefumo F.: Accuracy of ultrasonography at 11-14 weeks of gestation for detection of fetal structural anomalies: a systematic review. Obstet Gynecol 2013 ;122:1160-1167. |
|
Syngelaki A, Chelemen T, Daklis et al. Challenges in the diagnosis of fetal non-chromosomal abnormalities at 11-13 weeks. Prenatal Diagnosis 2011;;31:90-102. |
|
Iliescu D, Tudorache S, Comanescu A et al. Improved detection rate of structural abnormalities in the first trimester using an extended examination protocol. Ultrasound Obstet Gyneco 2013l;42:300-309. |
|
Hyett J, Moscoso G, Papapangiotou G et al: Abnormalities of the heart and great vessels in chromosomally normal fetuses with increased nuchal translucency thickness at 11–13 weeks of gestation. Ultrasound Obstet Gynecol 1996; 7: 245-250. |
|
Simpson L, Malone F, Bianchi D et al: Nuchal translucency and the risk of congenital heart disease. Obstet Gynecol 2007; 109: 376-83. |
|
ISUOG Practice Guidelines: performance of first-trimester fetal ultrasound scan. Ultrasound Obstet Gynecol 2013;41:102-113. |
|
Yagel S, Achiron R, Ron M et al: Transvaginal ultrasonography at early pregnancy cannot be used alone for targeted organ ultrasonographic examination in a high risk population. Am J Obstet Gynecol 1995; 172: 971-975. |
|
Timor-Tritsch IE, Bashiri A, Montageudo A, Arslan A. Qualified and trained sonographers in the US can perform early fetal anatomy scans between 11 and 14 weeks. Am J Obstet Gynecol 2004; 191: 1247-52. |
|
Fishman DA, Padilla L, Keh P et al: Management of twin pregnancies consisting of a complete hydatidiform mole and normal fetus. Obstet Gynecol 1998; 91: 546-550. |