This chapter should be cited as follows:
Gandhi A, Gandhi A, Glob Libr Women's Med
ISSN: 1756-2228; DOI 10.3843/GLOWM.411353
The Continuous Textbook of Women’s Medicine Series – Obstetrics Module
Volume 13
Obstetric emergencies
Volume Editor: Dr María Fernanda Escobar Vidarte, Fundación Valle del Lili, Cali, Colombia

Chapter
Cardiopulmonary Resuscitation in the Pregnant Woman
First published: February 2021
Study Assessment Option
By answering four multiple-choice questions (randomly selected) after studying this chapter, readers can qualify for Continuing Professional Development points plus a Study Completion Certificate from GLOWM.
See end of chapter for details.
INTRODUCTION
Maternal collapse is a rare but life-threatening event with a wide range of etiology. The outcome for the mother and also for the fetus depends on prompt and effective resuscitation. Maternal collapse is defined as an acute event involving the cardiorespiratory system and/or brain resulting in a reduced or absent conscious level (and potentially death) at any stage in pregnancy and up to 6 weeks after delivery. The incidence of maternal collapse or severe maternal morbidity is unknown as morbidity data are not routinely collected. A recent publication from Dublin showed a severe maternal morbidity rate of 3.2/1000 (320/100,000) births. In the last triennium in the UK, the maternal mortality rate was 14/100,000 births, but again not all maternal deaths are preceded by maternal collapse. The true rate of maternal collapse lies between 0.14 and 6/1000 (14 and 600/100,000) births.1 Cardiac arrest in pregnancy is rarely encountered and considered to occur in 1 : 30,000 births.2 Cardiopulmonary arrest in the pregnant woman triggers highly emotional reactions and can put the parturient at risk. There are four different levels of care to which the obstetric patient can be taken depending on her condition:
Level 0: Normal ward care
Level 1: Patients whose condition is at risk of deteriorating
Level 2: Patients requiring invasive monitoring/intervention or single organ failure support
Level 3: Patients requiring advanced respiratory support with support of more than one organ
Patients requiring care at level 2 and level 3 are likely to develop sudden collapse, but any pregnant woman can develop sudden collapse at any time during the pregnancy.
CLINICAL CAUSES
There are many causes of sudden obstetric collapse, which may be pregnancy related or result from conditions not related to pregnancy and possibly existing before pregnancy. Inclusion of all causes of the obstetric collapse is beyond the scope of the chapter, but the common causes of maternal collapse are discussed below.
Hemorrhage
Hemorrhage is one of the most common causes of maternal collapse and is responsible for most maternal deaths. Causes of major obstetric hemorrhage are postpartum hemorrhage (PPH) and antepartum hemorrhage (APH) from placenta previa/accreta, placental abruption, uterine rupture and ectopic pregnancy. In most cases of massive hemorrhage leading to collapse, the cause is obvious, but the concealed hemorrhage should not be forgotten, including following cesarean section and ruptured ectopic pregnancy. Other rare causes of concealed hemorrhage include splenic rupture and hepatic rupture. Acute inversion of the uterus may also be a cause following improper conduct of the third stage of labor. Resuscitation includes establishment of respiration and restoration of blood volume deficit and arrest of the source of hemorrhage.
Eclampsia
Eclampsia as a cause of maternal collapse is usually obvious in patient settings, often the diagnosis of pre-eclampsia has already been made when the seizure occurs. Epilepsy should also always be considered in cases of maternal collapse associated with seizure activity. Resuscitation includes correction of airway, control of seizures and blood pressure, and delivery.
Amniotic fluid embolism
Amniotic fluid embolism (AFE) presents as collapse during labor, delivery or within 30 minutes of delivery in the form of acute hypotension, respiratory distress and acute hypoxia. Seizures and cardiac arrest may occur. Pulmonary hypertension may develop secondary to vascular occlusion, by both debris or vasoconstriction followed by left ventricular failure and often giving rise to massive PPH. If AFE occurs prior to delivery, profound fetal distress develops acutely. Speedy resuscitation with oxygen administration, positive pressure ventilation, intravenous fluids, vasopressors and, if the fetus is undelivered, immediate delivery should be undertaken. In case of hemorrhage, replacement with red cells, platelets, fresh frozen plasma, cryoprecipitate or fibrinogen is done according to the need.
Pulmonary thromboembolism
Women with a clinical suspicion of deep vein thrombosis may develop release of thrombus. Pulmonary embolism occurs due to a thrombus blocking a pulmonary artery, which causes sudden onset of dyspnea, chest pain and features of collapse. Prompt resuscitation and antithrombotic therapy must be started immediately.
Sepsis
Sepsis has been recognized for centuries as a significant cause of maternal morbidity and mortality. Bacteremia, which may be present even in the absence of pyrexia or a raised white cell count, can progress rapidly to severe septic shock, leading to collapse. Management includes resuscitation, broad-spectrum antibiotic coverage and removal of septic foci.
Cardiac disease
The majority of deaths secondary to cardiac causes occur in women with no previous history. The main causes of death are myocardial infarction, aortic dissection and cardiomyopathy.
Intracranial hemorrhage
Intracranial hemorrhage is a significant complication of uncontrolled systolic hypertension with severe headache followed by maternal collapse.
Anaphylaxis
Anaphylaxis is a severely, life-threatening, generalized or systemic reaction resulting in respiratory, cutaneous and circulatory changes and possibly gastrointestinal disturbances and collapse. Prompt resuscitation with arrest of anaphylaxis is key to management.
Drug toxicity and overdoses
Drug toxicity and overdoses should be considered in all cases of obstetric collapse, and illicit drug overdose should be considered as a potential cause of collapse in the hospital, as should magnesium sulfate in the presence of renal impairment or local anesthetic agents injected intravenously by accident.
PHYSIOLOGICAL CHANGES IN PREGNANCY AFFECT RESUSCITATION
There are a number of reasons why the processes of cardiopulmonary resuscitation (CPR) are more difficult to perform and may be less effective in the pregnant compared with the non-pregnant population. Gradually increasing uterus size compromises resuscitative efforts. This may be the case from 20 weeks onwards, but will be more marked as the mother approaches term.
Cardiac output
Cardiac output increases by as much as 50% by 32 weeks’ gestation. At 20 weeks, significant aortocaval compression compromises venous return, and at 30 weeks' gestation, the woman has a significant drop in blood pressure when lying supine.
Vena caval occlusion
After 20 weeks’ gestation, the pregnant woman’s uterus can compress the inferior vena cava and the aorta, impeding venous return, cardiac output and uterine perfusion. The vena cava is completely occluded in 90% of term pregnant women lying supine and the stroke volume may be only 30% of that of a non-pregnant woman. Therefore, in late pregnancy, cardiac output can be increased by as much as 25–30% simply by moving the patient in a left lateral decubitus position.3 Delivery of the fetus during cardiac arrest will reduce the oxygen demands on the mother, empty the uterus, and thus also increase the venous return to the heart, making it more probable that resuscitation will be successful.
Changes in lung function
The pregnant uterus and increased breast size lead to a 20% decrease in functional residual capacity and 45% decrease in chest compliance. With such limited reserve and 20% increased oxygen consumption, there can be a rapid decline in oxygen saturation following hypoventilation.
The presence of mucosal edema and friability, increased secretions and weight gain all contribute to a more difficult airway intubation. There is also a greater risk of aspiration due to the relaxation of the esophageal sphincter. Passive regurgitation of stomach contents is a very real concern as it is greater in volume and more acidic during pregnancy and, therefore, more likely to lead to damaging acid aspiration into the lungs. It is imperative that experienced staff provide a protected airway and adequate ventilation as quickly as possible following cardiac arrest.
Management
Cardiac arrect management has four main steps:
- Immediate resuscitation;
- Supportive management is undertaken immediately once CPR is effective and mother is resuscitated. Move the patient to obstetric ICU;
- Try to undertake a differential diagnosis to determine the cause of the sudden collapse once supportive management is started;
- Continuing management: correct the reversible causes for the sudden collapse.
IMMEDIATE RESUSCITATION
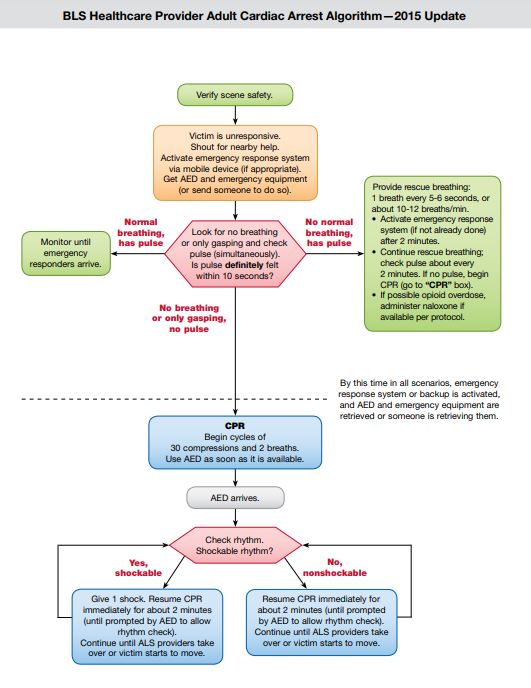
Resuscitation is conducted according to different guidelines. The American Heart Association (AHA) has issued guidelines for basic life support (BLS), adult advanced life support (ALS) and automated external defibrillation (AED) including algorithms and recommendations.4 The 2010 AHA guidelines were updated in 2015, and are now used widely in the resuscitation of the pregnant woman worldwide. It is recognized that the divisions into BLS and ALS are somewhat arbitrary in the hospital setting. In the community setting, BLS should be administered and rapid transfer arranged, unless appropriate personnel and equipments are available.
CPR should be started immediately and should be the same whether the patient is pregnant or not, though with few modifications, if she is pregnant owing to the physiological alterations occurring during pregnancy. The following modifications are necessary for managing cardiac arrest in pregnancy:
- Left lateral displacement of the uterus if gestational age is more than 20 weeks;
- Earlier definitive airway control;
- The removal of the fetal monitor to avoid electrical arcing during defibrillation;
- The defibrillation pads may be positioned in anterior and posterior on the left side of the chest if it is difficult to apply the apical pad in left tilted position of the patient;
- Consideration of an early cesarean delivery.
Basic life support and adult advanced life support for obstetric patient
As per the latest recommendations, emergency management is CAB (circulation, airway and breathing) (not ABC). If defibrillation is available immediately then this may take precedence over BLS.5 Make sure the patient, any bystanders and you are safe before performing CPR. The most important pre-requisite is that anyone performing CPR should be very much alert as their confidence and presence of mind will determine the outcome for the patient.
Adult basic life support for obstetric patient4
- Immediately call for the help. The adult BLS algorithm has been modified to reflect the fact that rescuers can activate an emergency response (i.e., through use of a mobile telephone) without leaving the victim’s side.
- Do not waste precious time before starting resuscitation and do not wait for help to arrive or to know the cause for sudden obstetric collapse. Immediately start resuscitation because management speed makes the difference. A delay of 1 minute may decrease survival chance by 10%.
- Check responsiveness (shake and shout): To confirm whether she is responsive to verbal commands and stimulation. This step is omitted if patient is under anesthesia.
- Turn the patient onto her back.
- Left uterine displacement: the gravid uterus at >20 weeks may compress the inferior vana cava and aorta which limits the effectiveness of chest compressions. Shift the uterus a minimum 1.5 inches away from the midline towards the left side. This may be accomplished manually. One person should be dedicated to manually displacing the uterus towards the left to relieve the aortocaval compression known as left uterine displacement (LUD). If rescuer is on patient’s left side, then pull the uterus with two hands. If rescuer is on patient’s right side, push the uterus with the one hand.6 (Do not tilt the woman on left side as that would decrease the performance and effectiveness of chest compressions and vascular access.) This will preserve supine positioning for optimal chest compressions, minimize aortocaval compression, optimize venous return (preload), generate adequate stroke volume during CPR, improve airway and intravenous access, access for defibrillation (Figure 1).
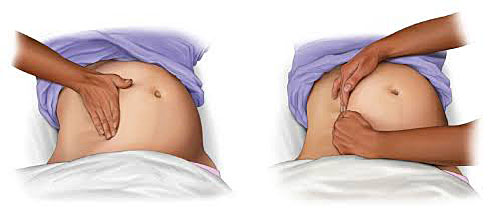
1
Left uterine displacement.6
- Check breathing and simultaneously check for the carotid pulse (within the first 10 seconds).
- If there is no normal breathing and the pateint has a pulse, then provide rescue breathing at the rate of one breath at every 5–6 seconds, or about 10–12 breaths/min. Continue rescue breathing and check pulse at every 2 minutes. If there is no pulse, begin CPR as below.
- If there are no breathing movements or only gasping and no pulse, proceed immediately to high quality compressions. Absence of breathing in the presence of a clear airway is now used as a sign of the absence of circulation. Agonal gasps are present in up to 40% of cardiac arrest victims. Agonal gasps can occur commonly within the first few minutes after sudden cardiac arrest; they are an indication for starting CPR immediately and should not be confused with normal breathing. If the victim is unresponsive with absent or abnormal breathing, the rescuer should assume that the victim is in cardiac arrest. Rescue breathing to be started only after starting compressions.
- Immediately start good quality chest compressions. There should be continued emphasis on the characteristics of high-quality CPR: compressing the chest at an adequate rate and depth, allowing complete chest recoil after each compression, minimizing interruptions in compressions, and avoiding excessive ventilation.
- Put the victim on the flat surface.
- Position yourself on the side of the victim and above the patient’s chest.
- Do not remove her underclothes.
- The gravid uterus at >20 weeks limits the effectiveness of chest compressions. It may be shifted away from the inferior vena cava and aorta manually by Left uterine displacement.
- Perform 100–120 chest compressions/minute. The compression rate has recently beenmodified from 100/min to a range of 100–120/min. The number of chest compressions delivered per minute during CPR is an important determinant of return of spontaneous circulation (ROSC) and survival with good neurologic function. Provision of adequate chest compressions requires an emphasis not only on an adequate compression rate but also on minimizing interruptions to this critical component of CPR. The upper limits of recommended compression rate and compression depth are based on preliminary data suggesting that excessive compression rate and depth adversely affect outcomes as do inadequate compression depth.4
- The compression: breath ratio is 30 : 2 (considered as 1 cycle)
- The person undertaking the CPR should keep their arms straight; elbows should not be bent while performing chest compressions; doing so will deliver weak, ineffective chest compression.
- Compression should be given at the lower border of the sternum between the nipples with heel of the hand.
- Place the heel of one hand there, with the heel of the other hand on the first hand. Interlock the fingers of both the hands and lift the fingers.
- Principle of the chest compression is “Push the chest hard and fast”. Press down on the sternum to depress it at least by 5–6 cm. Compression depth for adults is modified to at least 2 inches (5 cm) but should not exceed 2.4 inches (6 cm). Compressions create blood flow primarily by increasing intrathoracic pressure and directly compressing the heart, which in turn results in critical blood flow and oxygen delivery to the heart and brain. While a compression depth of at least 2 inches (5 cm) is recommended, the 2015 Guidelines Update incorporates new evidence about the potential for an upper threshold of compression depth (greater than 2.4 inches [6 cm]), beyond which complications may occur. Compression depth may be difficult to judge without use of feedback devices, and identification of upper limits of compression depth may be challenging.4
- Give time for chest to recoil in between chest compressions. To allow full chest wall recoil after each compression, rescuers must avoid leaning on the chest between compressions. It will allow full chest wall to recoil in cardiac arrest. Rescuers should allow complete recoil of the chest after each compression, to allow the heart to fill completely before the next compression. Full chest wall recoil occurs when the sternum returns to its natural position during the decompression phase of CPR. Chest wall recoil creates a relative negative intrathoracic pressure that promotes venous return and cardiopulmonary blood flow. Leaning on the chest wall between compressions precludes full chest wall recoil. Incomplete recoil raises intrathoracic pressure and reduces venous return, coronary perfusion pressure, and myocardial blood flow and can influence resuscitation outcomes.4
- One criterion for minimizing interruptions is clarified with a goal of chest compression fraction as high as possible, with a target of at least 60%.4
- Change the rescuer after five such cycles to avoid fatigue of rescuer, otherwise it may lead to ineffective compressions.
- Continue CPR until help arrives or an AED arrives and is ready for use or patient is moved to ICU or patient is revived.4
- Maintaining focus during CPR on the characteristics of compression rate and depth and chest recoil while minimizing interruptions is a complex challenge even for highly trained professionals. It may be reasonable to use audiovisual feedback devices during CPR for real-time optimization of CPR performance and to improve the quality of CPR in actual resuscitations (Figure 2).
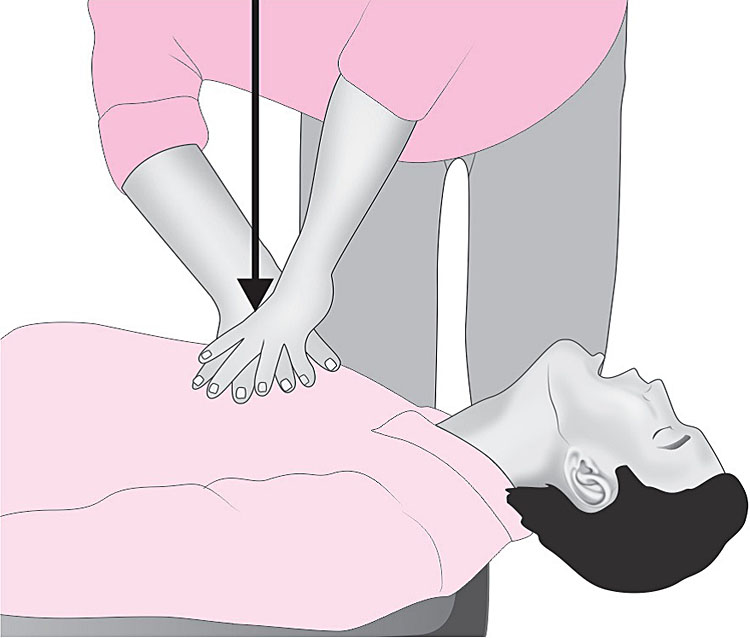
2
Chest compressions.
- Breathing:
- Mouth to mouth breathing: Ensuring head tilt and chin lift. Close the soft part of the patient’s nose with your thumb and index finger. Open her mouth a little but maintain chin lift. Take a breath and place your lips around her mouth, making sure that you have good seal. Blow steadily into her mouth over 1 second and watching for her chest to rise. Maintaining head tilt and chin lift, take your mouth away from the patient and watch for her chest to fall as the air comes out. Take another breath and repeat the sequence to give another effective breath. Give two effective breaths with use of barrier devices if available to provide ventilation like pocket mask, face shield, bag valve mask. Return to chest compressions quickly.
There have been few incidents of rescuers suffering adverse effects from undertaking CPR, with only isolated reports of infections such as tuberculosis (TB) and severe acute respiratory distress syndrome (SARS). Transmission of HIV during CPR has never been reported. - Ventilation breaths: Keep the airway open and provide ventilation with appropriate adjuncts. This might be a pocket mask or self-inflating bag with mask (AMBU bag). Bag and mask ventilation should be undertaken until intubation can be achieved. Each ventilatory breath should last about 1 second and should make the chest rise as if a normal breath. High flow oxygen should be added as soon as possible.
- Intubated ventilation: Tracheal intubation is the most effective way of providing adequate ventilation and should be performed as soon as a trained member of staff is available. Apply continuous cricoids pressure during ventilation and intubation due to the risk of regurgitation. Early intubation is mandatory, with attention to the use of an endotracheal tube (ETT) 0.5–1 mm smaller in internal diameter than that which would be used for a non-pregnant woman because the airway may be narrowed from edema. Avoid nasal intubation because of the increased mucosal friability during pregnancy. A laryngoscope with a shorter handle is useful as the presence of large breasts may interfere with access. Once the patient is intubated, ventilation should continue at ten breaths per minute but does not need to be synchronized with chest compressions. It should then be uninterrupted. For patients with ongoing CPR and an advanced airway in place, a simplified ventilation rate of 1 breath every 6 seconds (10 breaths per minute) is recommended. During pregnancy, there is increased oxygen requirements and rapid onset of hypoxia, and therefore it is important to ensure optimal oxygen delivery by high-flow 100% oxygen to whatever method of ventilation is being employed.
If circulation is present but no breathing (respiratory arrest), continue rescue breathing and bag and mask ventilation at a rate of 10 breaths/minute and a tidal volume large enough to raise the chest.
It must be noted that hyperventilation is harmful and should be avoided.7 If the patient starts to breathe on her own but remains unconscious, turn her into the recovery position and apply oxygen at the rate of 15 L/min. Check her condition and be ready to turn her back to start rescue breathing if she stops breathing.
- Mouth to mouth breathing: Ensuring head tilt and chin lift. Close the soft part of the patient’s nose with your thumb and index finger. Open her mouth a little but maintain chin lift. Take a breath and place your lips around her mouth, making sure that you have good seal. Blow steadily into her mouth over 1 second and watching for her chest to rise. Maintaining head tilt and chin lift, take your mouth away from the patient and watch for her chest to fall as the air comes out. Take another breath and repeat the sequence to give another effective breath. Give two effective breaths with use of barrier devices if available to provide ventilation like pocket mask, face shield, bag valve mask. Return to chest compressions quickly.
- Defibrillation: If automated external defibrillator (AED) is available, attach it, analyze the rhythm and defibrillate the patient. In majority of these types of cases, ventricular fibrillation or flutter is the dying rhythm of heart. In this case defibrillation with defibrillator is the definitive treatment. Defibrillate the victim using standard ACLS defibrillation doses. The most frequent initial rhythm in the context of sudden collapse is ventricular fibrillation (VF). The AED allows for early defibrillation by lesser-trained personnel as it performs rhythm analysis and gives information by voice or visual display, and the delivery of the shock is then delivered automatically. The 2010 Guidelines recommended the establishment of AED programs in public locations where there is a relatively high likelihood of witnessed cardiac arrest (eg, airports, casinos, sports facilities, etc.).There is clear and consistent evidence of improved survival from cardiac arrest when a bystander performs CPR and rapidly uses an AED. For witnessed adult cardiac arrest when an AED is immediately available, it is reasonable that the defibrillator be used as soon as possible. For adults with unmonitored cardiac arrest or for whom an AED is not immediately available, it is reasonable that CPR be initiated while the defibrillator equipment is being retrieved and applied and that defibrillation, if indicated, be attempted as soon as the device is ready for use. With in-hospital sudden cardiac arrest, there is insufficient evidence to support or refute CPR before defibrillation. However, in monitored patients, the time from ventricular fibrillation (VF) to shock delivery should be under 3 minutes, and CPR should be performed while the defibrillator is readied. CPR should be provided while the AED pads are applied and until the AED is ready to analyze the rhythm. If an AED is not available, manage with immediate ECG, if possible to differentiate the shockable and non-shockable rhythm and plan defibrillation as follows:
Shockable rhythms
There is no evidence that shocks from a direct current defibrillator have adverse effects on the heart of the fetus. If fetal or uterine monitors are in place, remove them before delivering shocks.
Shockable rhythms are treated with a shock. It is followed by immediate continuation of CPR without stopping for a rhythm or pulse check. Every 2 min the rhythm is assessed and if necessary a further shock is delivered. Injection adrenaline (epinephrine) 1 mg IV is given immediately before the third and every subsequent alternate shock, i.e. approximately every 4 min. Amiodarone 300 mg IV is given before the fourth shock.
Defibrillator safety management
The patient should not be near inflammable fluids, fumes or chemicals that could ignite.
If paddles are used rather than the pads: Keep paddles pressed firmly on the patient’s chest – failure to do so may result in a flash arc. The defibrillation paddles should be placed back in the appropriate containers as soon as the shocks have ended. Do not discharge the paddles in the air.8 The operator’s hands should be dry prior to applying a shock; the operator must check that nobody has direct or indirect contact with the patient. Advise all bystanders and personnel to “stand clear” (Figure 3).Non-shockable rhythms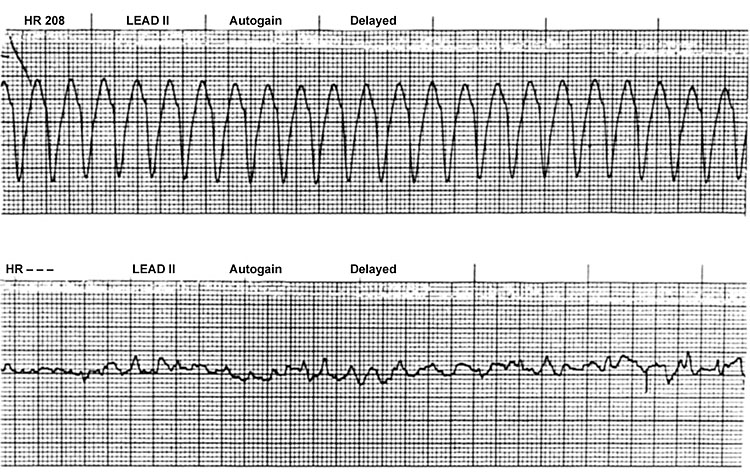
3
Cardiac rhythms: ventricular flutter and fibrillation.
In case of the non-shockable rhythm, i.e. pulseless electrical activity or asystole, adrenaline (epinephrine) 1 mg should be given intravenously immediately. Atropine 3 mg IV may be given once for asystole or slow rate, i.e. <60 bpm. This will minimize any vagal tone if present. The combined use of vasopressin and epinephrine offers no advantage to using standard-dose epinephrine in cardiac arrest. Also, vasopressin does not offer an advantage over the use of epinephrine alone. Therefore, to simplify the algorithm, vasopressin has been removed from the Adult Cardiac Arrest Algorithm – 2015 Update. It may be reasonable to administer epinephrine as soon as feasible after the onset of cardiac arrest due to an initial nonshockable rhythm.
Steroids may provide some benefit when bundled with vasopressin and epinephrine in treating an in hospital cardiac arrest (IHCA). While routine use is not recommended pending follow-up studies, it would be reasonable for a provider to administer the bundle for IHCA. When rapidly implemented, extracorporeal cardiopulmonary resuscitation (ECPR) can prolong viability, as it may provide time to treat potentially reversible conditions or arrange for cardiac transplantation for patients who are not resuscitated by conventional CPR (Figure 4).4
BLS Healthcare Provider Adult Cardiac Arrest Algorithm – 2015. From Amercian Heart Association,4 with permission.
- Resuscitative cesarean section delivery (Also called perimortem cesarean delivery) (PMCD): It is important to remember that both, mother and infant may die if the provider cannot restore blood flow to the mother’s heart. At gestational age >22 weeks and if CPR is not effective, consider the need for an emergency cesarean delivery which requires delivery to begin about 3–4 min after cardiac arrest. It will also facilitate effectiveness of CPR. Perimortem cesarean section packs are available on resuscitation trolleys in all areas where maternal collapse may occur.5
- PMCD needs to be initiated within 3–4 minutes of cardiac arrest and is essentially done to save the mother irrespective of whether fetus is alive or dead.
- Perimortem cesarean section should be performed where resuscitation is taking place. The principle of successful cesarean delivery is rapid incision, rapid delivery and rapid closure.
- It is best obtained with large vertical abdominal incision and closure of the uterus and then the abdomen with large running sutures in a single layer.
- There is no need to scrub the abdomen.
- No need to administer anesthesia, as patient is not responsive.
- Cesarean deliver will be relatively bloodless, since there is no circulation and cardiac output.
- Immediately deliver the baby and handover to neonatologist.
- The placenta should be delivered manually.
- The best survival rate for infants >28 weeks occurs when delivery of infant occurs in <5 minutes after the mother’s heart stops beating.
- Chest compressions and ventilation should be continued during PMCD.
- Emptying of the uterus will also facilitate effectiveness of CPR.
- If the mother is resuscitated and pulse returns then immediately move her to ICU.
- Start broad-spectrum antibiotics.
- If the patient has sudden collapse during cesarean delivery, chest compressions and ventilation should be started immediately. The baby should be delivered immediately and the uterus emptied. The principle again here for successful resuscitation is “Rapid incision, Rapid delivery and Rapid closure.” The placenta should be removed manually, and the abdomen closed with large running sutures in a single layer.
- Document the procedure in the patient’s record.
- All patients who progress to brain death or circulatory death after initial cardiac arrest should be considered potential organ donors.
A study was conducted Katz et al.9 of cases from 1985 to 2004 identified 38 cases of PMCD; 34 infants survived (three sets of twins, one set of triplets). Of the 34 infants (25–42 weeks’ gestation), time of delivery after maternal cardiac arrest was available for 25. Eleven infants were delivered within 5 minutes, four in from 6 to 10 minutes, two in 11 to 15 minutes, and seven in more than 15 minutes. Of 20 PMCD deliveries with potentially resuscitatable causes, 13 mothers were resuscitated and discharged from the hospital in good condition. One other mother was successfully resuscitated after the delivery, but died within 24 hours from complications related to AFE. In 12 of 18 reports that documented hemodynamic status, cesarean delivery preceded return of maternal pulse and blood pressure, often in a dramatic fashion. Eight other cases noted improvement in maternal status. Importantly, in no case was there deterioration of the maternal condition with the cesarean delivery.9 Cesarean delivery might be necessary to accomplish a successful resuscitation even if the fetus has died.7
Laparoscopic patient
Bradycardia may occur during laparoscopic surgeries because of insufflating gas which may compress the inferior vana cava and reduce the venous return. If bradycardia is not managed quickly, it may lead to asystole. Immediately releasing the insufflating gas usually restores venous return and improves the heart rate. Injection atropine or glycopyrrolate may be given to improve the heart rate.
SUPPORTIVE MANAGEMENT
Studies of patients after cardiac arrest have found that a systolic blood pressure less than 90 mmHg or a mean arterial pressure of less than 65 mmHg is associated with higher mortality and diminished functional recovery, while systolic arterial pressures of greater than 100 mmHg are associated with better recovery. The 2015 AHA Guidelines Update recommendations for postcardiac arrest care recommended identification and correction of hypotension in the immediate postcardiac arrest period.4 After return of spontaneous circulation (ROSC), fluids and vasoactive infusions should be used to maintain a systolic blood pressure above the fifth centile for age. Early, rapid IV administration of isotonic fluids is widely accepted as a cornerstone of therapy. After ROSC, normoxemia should be targeted. When the necessary equipment is available, oxygen administration should be weaned to target an oxyhemoglobin saturation of 94–99%. Hypoxemia should be strictly avoided. Ideally, oxygen should be titrated to a value appropriate to the specific patient condition.
Supportive management is started immediately after CPR is effective and mother resuscitated:
- Two wide-bore cannulas should be inserted as early as possible.
- There should be an aggressive approach to volume. Fluids – isotonic normal saline, Ringer's lactate or plasmalyte A are given rapidly in bolus as per an individual case. Caution must be exercised in the case of severe pre-eclampsia and eclampsia to avoid pulmonary edema owing to fluid overload.
- Oxygen administration is continued to prevent hypoxia.
- Catheterization should be done.
- Broad spectrum antibiotics should be started.
- If required, should be managed for blood transfusion.
- The patient should be examined for resuscitation related injuries, e.g. rib fractures.
- Immediately the pateint should be moved to obstetric ICU. The patient should be accompanied by qualified personnel during transportation to obstetric ICU.
- Senior experienced staff should be involved at an early stage.
- The patient should be supervized by an adequately skilled team.
- Blood samples should be collected and sent for basic initial laboratory investigations during initial resuscitation mainly CBC, blood sugar, LFT, renal function test, bleeding and coagulation profile, pH, bicarbonate, etc.
- Abdominal ultrasound by a skilled operator can assist in the diaganosis of concealed hemorrhage and to know the condition of a fetus. The interpretation of fetal well being by external electronic monitoring may be of utmost importance.
- Multidisciplinary teamwork is involved as per the requirement of an individual case.
- Along with CPR, correct the reversible causes of cardiac arrest – 4 Hs and 4 Ts:
Four Hs
Hypoxia
Hypovolemia (hemorrhage or sepsis)
Hyperkalemia and other metabolic disorders
Hypothermia
Hypoxia
Effective ventilation and supplementation is ensured with 100% oxygen delivery as soon as possible.
Regular checks should be done to ensure adequate airway and ventilation.
Hypovolemia
Fluid replacement should be given if hypovolemia is suspected. Large-bore cannula is inserted to allow rapid infusion of fluids.10
Hyper/hypokalemia, hypocalcemia and metabolic disorders
Electrolyte imbalance may lead to cardiac arrest – it should be confirmed or ruled out with arterial blood gases and serum electrolytes analysis. ECG monitoring and baseline blood tests should be done for urea and electrolytes at regular interval.
Hypothermia
Hypothermia is defined when the core temperature is below 35°C. Record the patient’s temperature as soon as possible after a cardiac arrest. In case of hypothermia, consider active warming with warm blankets.
Four Ts
Thromboembolism
Toxicity (drugs associated)
Tension pneumothorax
Cardiac tamponade
IDENTIFY THE RESPONSIBLE CAUSE FOR THE SUDDEN COLLAPSE
The spot diagnosis of probable differential diagnosis for the sudden collapse should be undertaken as further management depends on the responsible cause for the situation. It is imperative to identify reversible causes of cardiac arrest. The gestational age should be quickly established in order to determine fetal viability. Abdominal ultrasound examination can be performed for this purpose, but it should not delay the resuscitation procedures. The etiology can also be classified into anesthesia-related causes and/or non-anesthesia-related causes.
Differential diagnosis of sudden collapse in pregnancy
Is it APH or PPH or inversion of uterus? If not, then consider for non-hemorrhagic causes.
History of and probable diagnosis
Obstetric or medical causes
History of hypertension in pregnancy and convulsions – eclampsia
History of grand multipara or previous uterine scar or instrumental delivery – rupture of the uterus
History of mismanaged third stage of labor, short cord or manual removal of placenta (MRP) – inversion of uterus
History of previous cardiac problems, complaints of acute left-sided chest pain, gabhraman, hypotension –maternal cardiac problems, mainly myocardial infarction
History of vehicular accidents or domiciliary violence – trauma
History of collapse after administration of drugs, signs and symptoms of allergic reactions – drug reaction or overdose
History of painful stimuli, injections, etc. – anaphylactic reaction
History of collapse immediately after delivery, mainly in multipara or in precipitate labor and no obvious cause or in any case always consider the possibility of amniotic fluid embolism – AFE
History of sudden onset of unexplained dyspnea and tachypnea, especially in western countries because of venous stasis and hypercoagulability of blood – pulmonary thromboembolism.
Anesthesia-related causes
History of spinal anesthesia (SA) in higher position, difficult SA during surgery, complaints of heaviness in the chest, gabhraman, breathlessness within few minutes of SA – high spinal anesthesia
History of vomiting under anesthesia and problem starts within few hours – Mendelson’s syndrome
History of fall in the blood pressure within few minutes after SA – supine spinal shock
Mortality related to airway problems during extubation of the trachea has increased with spinal anesthesia-related mortality.11,12 Of all maternal deaths due to anesthesia 27% occurred among obese women, whereas 24% occurred among overweight women.
CONTINUING THERAPY: CORRECT THE REVERSIBLE CAUSES FOR THE SUDDEN COLLAPSE
Continuing therapy can be directed towards the specific cause of the caused sudden collapse in a pregnant woman to optimize the outcome and continued in an obstetric ICU. Management of each individual cause is not within the scope of this chapter. Decisions should not be delayed if surgical intervention is required.
PREARM TO PERFORM

5
Basic life support algorithm.4
Drugs used in adult cardiac arrest
Adrenaline
Adrenaline causes vasoconstriction and increases perfusion to myocardium and cerebrum.13 The usually prescribed dosage of adrenaline is 1 mg IV (1 mL of 1 : 1000 or 10 mL of 1 : 10,000). Ventricular fibrillation/pulseless ventricular tachycardia (VF/pulseless VT) after initial counter shocks have failed (after second shock and then after every second cycle).
Asystole and pulseless electrical activity in initial cycle (and then every second cycle).
Do not interrupt CPR to administer medications.
Adverse effects include tachyarrhythmias and hypertension after the person is resuscitated.
Amiodarone
Amiodarone is an antiarrhythmic drug. It is given for VF/pulseless VT (between the third and fourth shock when refractory to defibrillator shocks and a vasopressor). If there have been three unsuccessful defibrillation shocks for VF/pulseless VT, the adult bolus dose of 300 mg (5 mg/kg) of amiodarone IV should be given. An additional bolus of 150 mg could be considered. This may be followed by an infusion of 15 mg/kg over 24 h. Major side-effects include bradycardia, heart block and hypotension.
Lignocaine and amiodarone should not be given together.14,15
Vasopressor
Epinephrine may reasonably be administered as soon as feasible after the onset of cardiac arrest due to an initial nonshockable rhythm.4
Vasopressin in combination with epinephrine offers no advantage as a substitute for standard-dose epinephrine in cardiac arrest.4
Intravenous lipid emulsion
It may be reasonable to administer intravenous lipid emulsion, concomitant with standard resuscitative care, to patients who have premonitory neurotoxicity or cardiac arrest due to local anesthetic toxicity.
Sodium bicarbonate
Sodium bicarbonate is given in the dose of 50 mmol IV to patients when arrest is associated with hyperkalemia. It is also given in response to severe acidosis when pH is less than 7.1, usually in cases of septicemia and diabetic ketoacidosis. It should be used with caution, because rapid correction of maternal acidosis can reduce the compensatory hyperventilation.16
Calcium chloride
A dose of 10 mL 10% calcium chloride (6.8 mmol Ca2+) IV can be used if it is thought that pulseless electrical activity (PEA) is caused by hyperkalemia, hypocalcemia, overdose of calcium channel blocking drugs or overdose of magnesium sulfate (for treatment of pre-eclampsia).
Inotropic agent
Dopamine: Initial intravenous infusion of 2–5 μg/kg/min and increase up to 5–10 μg/kg/min as needed
Dobutamine: Intravenous infusion of 2–40 μg/kg/min
Noradrenaline: 2–4 μg/kg/min dose depending upon blood pressure
Intra-arrest and postarrest prognostic factors
Low end-tidal carbon dioxide (ETCO2) in intubated patients after 20 minutes of CPR is associated with a very low likelihood of resuscitation. While this parameter should not be used in isolation for decision making, providers may consider low ETCO2 after 20 minutes of CPR in combination with other factors to help determine when to terminate resuscitation.
Multiple factors should be considered when trying to predict outcomes of cardiac arrest. Multiple factors play a role in the decision to continue or to terminate resuscitative efforts during cardiac arrest and in the estimation of potential for recovery after cardiac arrest. No single intra-arrest or postcardiac arrest variable has been found that reliably predicts favorable or poor outcomes.
Communication
Wherever possible, senior personnel from the obstetric, anesthesia and midwifery should communicate properly. Ensure that the family is looked after and kept informed about patient’s conditions and management undertaken. All interventions should be documented accurately with timings.
To improve outcomes for maternal collapse in future:
- Always be set up ready to tackle such cases. All obstetric units must be ready to tackle the unpredictable.
- Form a permanent team which should include the cardiac arrest team, a senior obstetrician, an obstetric anesthetist a senior midwife.
- Develop systems that assemble more staff.
- Plan regular training of the staff.
- Plan and perform regular practice drills for medical and paramedical staff.
- Review, revise and audit each such event and update the protocol as and when required.
- Identify and treat the patient in response to early signs of demise may prevent deterioration of situation to cardiac arrest. An early warning sign score (EWS) can help to identify deteriorating patients and can be given attention to prevent cardiac arrest.
CONCLUSION
CPR in the pregnant patient with cardiac arrest should be performed with consideration of the physiological changes associated with pregnancy. The standard algorithm should be followed according to basic and advanced life support protocols, with few exceptions. Attention should be given to lateral displacement of the uterus, to secure early airway, aggressive airway management and early consideration of emergency cesarean delivery which are major modifications in the management of maternal sudden cardiac arrest. Immediate cesarean delivery not only improves survival of infant but also facilitates maternal resuscitation. CPR drill should be performed regularly for medical and paramedical staff.
PRACTICE RECOMMENDATIONS
- Maternal collapse is defined as an acute event involving the cardiorespiratory system and/or brain resulting in a reduced or absent conscious level (and potentially death) at any stage in pregnancy and up to 6 weeks after delivery.
- There are many causes of sudden obstetric collapse, which may be pregnancy related or result from conditions not related to pregnancy and possibly existing before pregnancy.
- Hemorrhage is one of the most common causes of maternal collapse and is responsible for most maternal deaths.
- Cardiopulmonary resuscitation (CPR) is more difficult to perform and may be less effective in the pregnant compared with the non-pregnant population.
- Emergency management is CAB (circulation, airway and breathing) (not ABC).
- The 2015 AHA Guidelines Update recommendations for postcardiac arrest care recommended identification and correction of hypotension in the immediate postcardiac arrest period.
- Along with CPR, the reversible causes of cardiac arrest the 4 Hs and 4 Ts should be corrected.
- Continuing therapy can be directed towards the specific cause of the caused sudden collapse in a pregnant woman to optimize the outcome and continued in an obstetric ICU.
- Decisions should not be delayed if surgical intervention is required.
- CPR drill should be performed regularly for medical and paramedical staff.
CONFLICTS OF INTEREST
Author(s) statement awaited.
Feedback
Publishers’ note: We are constantly trying to update and enhance chapters in this Series. So if you have any constructive comments about this chapter please provide them to us by selecting the "Your Feedback" link in the left-hand column.
REFERENCES
Maternal collapse in pregnancy and the puerperium .Green-top Guideline No. 56. RCOG 2011;1:24. | |
Morris S, Stacey M. Resuscitation in pregnancy. BMJ 2003;327:1277–9. | |
Lapinsky SE, Kruczynski K, Slutsky AS. Critical care in the pregnant patient. Am J Respir Crit Care Med 1995;152:427–55. | |
Women and Newborn Health Services, King Edward Memorial Hospital. Clinical Guidelines-Obstetrics & Gynaecology, Advanced Life Support. Government of Western Australia Department of Health. | |
Sinz E, Lavonas EJ, Jeejeebhoy FM. American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Part 12: cardiac arrest in special situations. Circulation 2010;122:S829–61. | |
Ezri T, Lurie S, Weiniger C, Golan A, et al. Cardiopulmonary resuscitation in the pregnant woman – an update. IMAJ 2011;13:306–10. | |
Defibrillation theory and practice. In: Moule P, Albarran J (eds.) Practical resuscitation recognition and response. Melbourne: Blackwell Publishing, 2005;151–76. | |
Katz V, Balderston K, DeFreest M. Perimortem caesarean delivery: were our assumptions correct? Am J Obstet Gynecol 2005;192:1916–20; discussion. | |
Australian Resuscitation Council. Guideline 11.2 protocols for adult advanced life support Dec 2010: ARC/NZRC. 2010. Available from: http://www.resus.org.au/policy/guidelines/section_11/guideline-11-7dec10.pdf. | |
Arendt KW. Present and emerging strategies for reducing anesthesia-related maternal morbidity and mortality. Curr Opin Anaesthesiol 2009;22:330–5. | |
Mhyre JM, Riesner MN, Polley LS, et al. A series of anesthesia-related maternal deaths in Michigan, 1985–2003. Anesthesiology 2007;106:1096–104. | |
Gillimore D. Understanding the drugs used during cardiac arrest response. Nurs Times 2006; 102:24. | |
Thomas J (ed.) APP guide. Australian prescription products guide. Melbourne: Australian Pharmaceutical Publishing Company Ltd.; 2006. | |
Atta E, Gardner M. Cardiopulmonary resuscitation in pregnancy. Obstet Gynecol Clin North Am 2007;34:585–97. | |
Australian Resuscitation Council, New Zealand Resuscitation Council. Guideline 11.7: post-resuscitation therapy in adult advanced life support: Australian Resuscitation Council. Dec 2010. Available from: http://www.resus.org.au/policy/guidelines/section_ 11/guideline-11-7dec10.pdf. |
Online Study Assessment Option
All readers who are qualified doctors or allied medical professionals can automatically receive 2 Continuing Professional Development points plus a Study Completion Certificate from GLOWM for successfully answering four multiple-choice questions (randomly selected) based on the study of this chapter. Medical students can receive the Study Completion Certificate only.
(To find out more about the Continuing Professional Development awards program CLICK HERE)