Evaluation of the Male in Infertility
Authors
INTRODUCTION
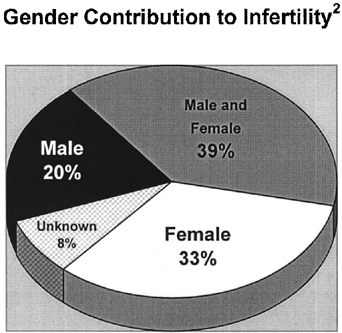
The understanding of the male factor contribution to a couple’s infertility has changed significantly over the past decade such that accurate detection and specific treatment for various etiologies affecting the male factor can effect a pregnancy with strong probability. Bypassing male fertility pathology with utilization of in vitro fertilization (IVF) and intracytoplasmic sperm injection (ICSI) is necessary in a select, well-defined group of patients. The majority of men who present with infertility are found to have medically or surgically correctable causes, and after treatment, natural conception may become possible. A practice survey conducted by the National Regional Advisory Council of the American Society for Reproductive Medicine revealed that the diagnostic and therapeutic services of urologists are significantly underutilized by couples seeking infertility evaluation.1 Approximately 15% of couples attempting to conceive over the period of 1 year are unable to become pregnant.2 Twenty percent of these cases are attributable to a male factor alone, while a male factor is contributory in nearly another 40% of cases (Fig. 1).
A thorough, thoughtful, logical approach to the evaluation of male infertility is imperative because the etiology of compromised fertility is diverse, including abnormalities of sperm production, sperm dysfunction, and obstruction of the excurrent ductal tract anywhere from the efferent ductule to the ejaculatory ducts (Table 1). Most commonly, semen analysis confirms the suspicion that the male partner contributes to the couple’s infertility, and this diagnostic test along with comprehensive history and physical examination are the foundation on which further evaluation may proceed. If necessary, more specific testing to include hormonal evaluation, ultrasound, postejaculatory urinalysis, genetic testing, and specific sperm function tests may be required to establish the diagnosis.
TABLE 1. Etiology of Male Infertility
Abnormalities of sperm production
Primary testicular failure (Hypergonadotropic hypogonadism)
Genetic abnormality (Klinefelter syndrome, Y chromosome microdeletion, other)
Secondary testicular failure
Hypogonadotropic hypogonadism
Cryptorchidism
Atrophy (orchitis)
Exogenous androgen use
Exposure to gonadotoxins/heat
Varicocele
Abnormalities of sperm function
Anti-sperm antibodies
Infection
Varicocele
Acrosome reaction
Biochemical (Reactive oxygen species)
Zona pellucida binding/sperm penetration
Obstruction of excurrent ductal system
Vasectomy
Congenital absence of the vas deferens
Epididymal obstruction (congenital or acquired)
Ejaculatory duct obstruction (congenital or acquired)
Many causes of infertility are treatable, and even patients with an etiology not amenable to surgical reconstruction or medical management, such as congenital bilateral absence of the vas deferens (CBAVD), have the opportunity to produce biologic offspring with sperm retrieval techniques performed in conjunction with IVF. However, CBAVD is associated with cystic fibrosis (CF) gene mutation. Assisted reproduction should be pursued with caution because of the risk of transmission of CF to the offspring if the female partner is a carrier of the CF mutation as is discussed in a subsequent section. The couple must be counseled appropriately. Conversely, a complete absence of sperm production, as is seen in some patients with specific microdeletions of the Y chromosome, necessitates the use of donor sperm or adoption for the couple. Furthermore, at the time of evaluation for male infertility, significant medical pathology requiring further treatment may be discovered by thorough history and complete physical examination.3 For example, patients with unknown pituitary adenomas may present with the complaint of decreased libido secondary to hypersecretion of prolactin, which in turn causes negative feedback inhibition on pituitary secretion of luteinizing hormone (LH) and subsequent reduced testosterone production within the testis. Alternatively, decreased libido may be secondary to an infarct of the anterior pituitary and absence of LH production with diminished testosterone production in the testis, ultimately leading to decreased libido. Further evaluation with determination of serum hormone levels and cranial magnetic resonance imaging (MRI) will ultimately lead to the diagnosis and appropriate treatment. The incidence of testis tumor detection in men who present with infertility is significantly higher than that of the fertile male population, and this risk should be considered for all infertile men.4
Infertility evaluation for both the male and female partner should be pursued simultaneously after 1 year of attempting to achieve a pregnancy. This waiting period is not practical for all couples however. Advanced maternal age (older than 35 years) and a history of cryptorchidism or other factors clearly associated with male infertility should prompt earlier evaluation. Additionally, if either partner has a reason to believe that his or her reproductive function is abnormal, evaluation should be pursued promptly as well. Evaluation of the male partner should be performed by a urologist or other specialist in reproductive medicine and begins with a thorough reproductive and medical history, a physical examination, and two semen analyses obtained at least 4 weeks apart.
BASIC EVALUATION
Reproductive History
Several aspects of a reproductive history are important in assessing a man’s fertility potential. Initially, a distinction between primary infertility (a lifelong inability to effect a pregnancy despite regular, unprotected intercourse) and secondary infertility (initiation of a pregnancy in the past but presently unable to conceive for unknown reason) is made. Prior history of initiating a pregnancy does not ensure lifelong fertility potential, because infertility may be acquired and patients with secondary infertility are evaluated in the same manner as those with primary infertility. The timing and frequency of intercourse are essential components of the reproductive history as well. Intercourse should occur daily, beginning 2 days before and including the day before and day of ovulation, to ensure that spermatozoa are present for fertilization. Many patients are not familiar with the timing of ovulation relative to the onset of the menstrual period and must be counseled appropriately. Finally, the patient should be questioned regarding the use of lubricants with intercourse, as many commonly used lubricants (K-Y Jelly, Surgilube, Lubifax, Astroglide, Replens, Touch, olive oil, saliva) are known to impair the motility of sperm;5,6,7 their use should be avoided.
An assessment of developmental abnormalities and childhood illnesses is imperative as well. Cryptorchidism is associated with decreased spermatogenesis.8 A history of mumps contracted after the onset of puberty may result in mumps orchitis involving one or both testes, causing subsequent atrophy and compromised spermatogenesis as well.9 A history of physical characteristic development during adolescence, such as acquisition of normal hair distribution is essential to establish normal endocrine development. Sexually transmitted diseases, such as gonorrhea and chlamydia are associated with excurrent duct inflammation, which may result in epididymal obstruction.
Several environmental toxins and medications are associated with abnormal sperm production, and the patient should be questioned regarding exposure or use. Agents such as heat, radiation, heavy metals, organic solvents, and pesticides have been shown to affect testicular function as have medications such as sulfasalazine, colchicine, cimetidine, and nitrofurantoin and the drugs caffeine, nicotine, alcohol, marijuana, and cocaine. The effects that these substances have on sperm production are usually reversible with discontinuation of exposure or use. The utilization of androgenic anabolic steroids by athletes depresses sperm production by interfering with the hypothalamic-pituitary-gonadal axis; these effects are not always reversible with discontinuation of use.
Medical History
Additional components of a medical history that should be addressed are medical illnesses that affect infertility secondarily, such as diabetes mellitus, which may cause dysfunction of the bladder neck with retrograde ejaculation. Upper respiratory tract disease may be associated with mutations within the CF transmembrane conductance regulator gene, which in turn are also associated with congenital absence of the vas deferens or congenital epididymal obstruction. A family history is necessary to ascertain whether any male relatives have had a history of infertility as well. A surgical history is also relevant especially with regard to scrotal, inguinal, retroperitoneal, and lower urinary tract procedures. Inguinal herniorrhaphy, renal transplantation, and scrotal surgery are the most common causes of iatrogenic injury to the vas deferens.10 Retroperitoneal procedures may cause ejaculatory dysfunction.
Physical Examination
A complete physical examination should be done in addition to focused examination of the male reproductive tract. The patient’s body habitus is inspected for hair distribution and signs of feminization. Although patients with sex chromosome abnormalities such as Klinefelter syndrome (47,XXY pattern) are classically described as tall and eunichoid with gynecomastia and small testes, the phenotype is variable, and careful observation of body habitus is important. Scrotal examination is best done in a relatively warm room with the scrotum warmed (heating pad) such that the dartos and cremaster muscles are relaxed and the scrotal contents can be more easily palpated. The testicles are assessed for normal size and consistency. Normal testicular volume measures 15 to 25 mL3,11 and the consistency should be firm. Small, soft testes are associated with either primary testicular failure (intrinsic defect in spermatogenesis) or secondary testicular failure, which is caused by endocrine abnormalities or other conditions such as the use of exogenous androgens, or exposure to heat and gonadotoxins. The presence and consistency of the epididymis and the presence of the vas deferens are noted. Epididymal fullness has been found to correlate with epididymal obstruction in men with azoospermia (complete absence of sperm within the ejaculate).12 Diagnosis of congenital absence of the vas deferens can by made by physical examination alone and does not require scrotal sonography.
A varicocele is an abnormal dilation of testicular veins that can be palpated within spermatic cord through the scrotal skin. While 15% of fertile men are found to have varicoceles, 35% of men with primary infertility and 80% of those with secondary infertility have varicoceles.13 Varicoceles are associated with decreased testicular volume, impaired sperm quality, and a decline in Leydig cell function.14 The patient should be examined for the presence of a varicocele in both the supine and the upright position. Grade I varicocele is palpable with Valsalva maneuver only, grade II is palpable in the standing position, and grade III is visually apparent through the scrotal skin as a “bag of worms.”
Digital rectal examination is performed to evaluate the prostate and to assess for the presence of midline cysts, significant prostatic asymmetry, or dilated seminal vesicles. The ejaculatory ducts drain both the vas deferens and the seminal vesicles with ejaculation and course through the central zone of the prostate to enter the prostatic urethra at the level of the utricle. Obstruction of one or both ejaculatory ducts may occur secondary to congenital abnormality of Wolffian duct differentiation, resulting in a midline cyst or dilation of one or both seminal vesicles that may be palpable on digital rectal examination.
Semen Analysis
The principle diagnostic test to determine the extent to which the male partner contributes to a couple’s infertility is semen analysis. This test should be performed according to the guidelines established by the World Health Organization15 by a certified andrology laboratory. The semen specimen is collected by masturbation into a container or by intercourse with a silastic condom that does not contain substances that are toxic to spermatozoa. Collection should be done after an abstinence period of 2 to 3 days. The patient must be instructed carefully with regard to collection of the entirety of the specimen. Incomplete collection resulting in the finding of decreased ejaculate volume is a misleading outcome that may cause unnecessary additional testing. The collection is preferably done in the office, but may be obtained at home and promptly transported to the office for analysis within 1 hour of collection. If collected at home, the patient may transport the specimen to the laboratory in a previously provided sterile container, which should be placed in a shirt or coat pocket during transportation, such that the specimen remains as close to room temperature as possible.
Semen analysis determines the volume of the ejaculate and the sperm concentration, total number, percent motility, and the percentage of spermatozoa with normal morphology according to strict criteria.16 Reference ranges according to the World Health Organization are listed in Table 2.15 These values have been established on the basis of clinical evidence and represent an estimation of the number and quality of spermatozoa adequate to achieve a pregnancy through intercourse and do not reflect the average ranges observed in the semen analyses of fertile men. Repeat semen analysis should be done 4 weeks after the initial evaluation to confirm prior findings.
TABLE 2. Reference Ranges for Semen Analysis15
Ejaculate volume | >2.0 mL |
Sperm concentration | >20 million/mL |
Total sperm count | >40 million/ejaculate |
Motility | >50% |
Normal morphology (Kruger Strict criteria morphology) | >14% |
pH | >7.2 |
EJACULATE VOLUME.
Decreased ejaculate volume may be seen in ejaculatory duct obstruction, primary testicular dysfunction, CBAVD, or retrograde ejaculation. In ejaculatory duct obstruction and in most men with CBAVD, the alkaline seminal vesicle contribution is absent from the ejaculate. The acidic prostatic fluid contribution predominates, and the semen pH reflects the acidic fluid shift (less than 7.2). Because seminal vesicles also secrete fructose, the semen may be tested for the presence of fructose, the level of which correlates with the degree of ejaculatory duct obstruction. In complete, bilateral ejaculatory duct obstruction, the semen analysis shows azoospermia with low volume, acidic pH, and absent fructose.
Bilateral testicular atrophy caused by primary or secondary testicular failure resulting in hypogonadism may also cause low ejaculate volume. Both seminal vesicle and prostate secretions are controlled by androgens, and in the presence of low circulating testosterone, ejaculate volume is reduced. CBAVD is diagnosed by physical examination and is associated with abnormalities of the seminal vesicles (absence, cystic dilation). Retrograde ejaculation is detected by postejaculatory urine analysis and is described in a subsequent section.
SPERM CONCENTRATION AND TOTAL SPERM COUNT.
Azoospermia, the complete absence of sperm on standard microscopic semen analysis, must be confirmed by centrifugation of the semen specimen at 3000g for 15 minutes with meticulous high-powered (400×) microscopic examination of the pellet.
Once the diagnosis of azoospermia is established, it should be classified as obstructive (normal sperm production) or nonobstructive (diminished or absent spermatogenesis). Obstructive azoospermia is caused by obstruction of the excurrent ductal system anywhere from the level of the efferent ductules to the ejaculatory ducts. Nonobstructive azoospermia may be caused by disease that is intrinsic to the testis (primary testicular failure) or by endocrine abnormalities or other conditions such as exogenous androgens, heat, and gonadotoxins that suppress spermatogenesis (secondary testicular failure). Although a low level of sperm production may be present in nonobstructive azoospermia, it is not quantitatively sufficient to allow epididymal transit and ejaculation.17 Extended sperm preparation of a centrifuged semen specimen is necessary, as it may result in detection of sperm for up to 35% of men who are diagnosed with nonobstructive azoospermia prior to proper examination of the centrifuged pellet.18 The finding of sperm in this population may render surgical sperm retrieval unnecessary for assisted reproductive techniques.
Oligospermia is defined as sperm concentration less than 20 million per milliliter with severe oligospermia defined as less than 5 million per milliliter. Varicocele, the most common etiology among men who present with infertility, may cause oligospermia as well as abnormalities of sperm motility and forward progression. Surgical correction of clinically significant (palpable) varicocele has been found to improve sperm concentration, motility, and morphology with a corresponding increase in pregnancy rate. The most important study to demonstrate the benefit of varicocelectomy showed that 60% of men with abnormal semen analyses secondary to varicocele only, who underwent varicocelectomy experienced pregnancy within 1 year, whereas pregnancy was achieved in only 10% of those couples in which the varicocele went unrepaired (control group).19 The control group then underwent varicocelectomy, and during the second year of the study, 44% initiated a pregnancy. Varicocelectomy improves semen parameters sufficiently such that for most couples, assisted reproductive techniques (ART) are either rendered unnecessary, or the type of ART necessary to bypass the male factor is downstaged.20 Additionally, the benefit of varicocelectomy applies to men with varicocele and nonobstructive azoospermia, because up to 50% of these men will respond to varicocelectomy with return of sperm to the ejaculate; however, most men will still require surgical testicular sperm extraction.21,22
Severe oligospermia is often idiopathic, but may also be caused by significant hormonal deficiencies or by specific microdeletion of the Y chromosome (AZFc region). Hormone evaluation and genetic testing for Y chromosome microdeletion are indicated in this population as is described subsequently.
Total sperm count is a function of sperm concentration and total semen volume. Despite normal concentration, total sperm count may be low if volume is low. Although concentration may be low in the setting of increased volume, the total sperm count may be normal. Fluctuations between sperm concentration and volume are determined by complex factors, including duration of abstinence and stress levels. Critical values to determine fertility potential for an individual man cannot be unequivocally established, and the flux between sperm concentration, total sperm count and volume are assessed as a whole to predict adequacy.
MOTILITY AND FORWARD PROGRESSION.
The percentage of motile sperm with any degree of tail motility is reported as motility, and forward progression is expressed as the percentage of motile sperm that are progressing in one general direction. Deficient sperm movement (asthenospermia) may be caused by several pathologic processes. The presence of anti-sperm antibodies (ASA) within the semen may cause sperm to aggregate and lose the capacity to move effectively. Antisperm antibody testing of the serum and the semen can establish the presence of antibodies. Genital tract infection may impair sperm motion by increasing the number of leukocytes within the semen. White blood cells within the semen cannot be differentiated from immature germ cells on routine examination, both of which are collectively termed round cells. If more than 5 million round cells per milliliter are noted, the sample should be analyzed for the presence of leukocytes, as is described in a subsequent section, and more than 1 million leukocytes per milliliter is indicative of possible infection. If infection is suspected (and many practitioners will routinely evaluate all men for infection), a subsequent semen sample should be cultured for Mycoplasma hominis, Ureaplasma urealyticum, aerobic and anaerobic bacteria, and tested for Neisseria gonorrhoeae, Chlamydia trachomatis, and Trichomonas vaginalis. Prior to providing the sample for culture, the patient should be instructed to prep the penis with betadine to avoid contamination with skin flora.
If no motile sperm are found on semen analysis, a viability stain may be done to differentiate viable, nonmotile sperm from those that are dead. If a large number of sperm are viable, the tails may be unable to flagellate because of a lack of dynein arms within the axoneme of the flagellae of the spermatozoa (immotile cilia syndrome). The cilia of the respiratory tract are likewise afflicted. When associated with situs inversus, immotile cilia syndrome is known as Kartagener’s syndrome. These patients have a history of chronic respiratory tract infection as well as infertility. The diagnosis may be made by electron microscopy of the spermatozoa.
Partial ejaculatory duct obstruction may cause asthenospermia as well, and low semen volume is also present. Partial obstruction of a vasal anastomosis may also cause poor motility and is often associated with detectable levels of ASA. In addition to causing decreased sperm concentration, varicocele may result in diminished sperm motility and forward progression. Significantly prolonged abstinence periods are associated with compromised sperm motility as well. In most cases, impaired motility reflects defective testicular function, and identification of any factors that may adversely affect the testis (e.g. heat, toxins) is needed.
MORPHOLOGY.
The quality of morphologic appearance of spermatozoa within a semen specimen predicts not only the adequacy of spermatogenesis, but also the likelihood of achieving conception through natural intercourse. Of all semen parameters, morphology criteria have undergone most significant revision over the last decade. Previously published reference ranges by the World Health Organization cite normal morphology percentage decreasing sequentially from 50%23 to 30%24 to 14% in the most recent guidelines15 based on the finding that IVF success rates were significantly higher in a population with normal sperm morphology of more than 14% when compared with a population with normal sperm morphology of less than 14%.16 Although this study established the percent normal morphology shown to have high fertilization and pregnancy rates with IVF, the relationship between the Kruger strict criteria morphology and natural fertility (without IVF) is not well established.
Defects in sperm morphology (teratospermia) are categorized by location: head, neck (midpiece), or tail. Cytoplasmic droplets usually located in the midpiece that occupy more than one half of the area of a normal sperm head are noted as a discrete defect as well. For the sperm to be classified as normal, the entire sperm must appear normal. Teratospermia may be observed in patients with varicocele and in men with primary or secondary testicular failure. Directed treatment of the specific disorder can result in improvements in sperm morphology.
ADVANCED EVALUATION
Hormone Assessment
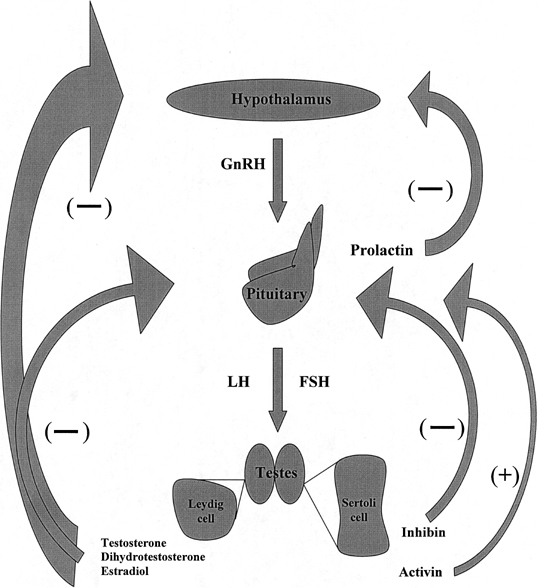
Derangements of the hypothalamic-pituitary-gonadal axis are associated with abnormalities in spermatogenesis and in sexual function. Men with abnormalities on semen analysis (especially sperm concentration less than 10 million per milliliter), decreased libido, or other clinical manifestations of endocrinopathy should undergo hormone evaluation.25 Initial testing includes determination of serum testosterone and FSH. If abnormalities are detected, serum LH and prolactin should also be determined. Figure 2 depicts normal hormone production and negative feedback inhibition within the hypothalamic-pituitary-gonadal axis. The relation between serum hormone values in an individual patient is characteristic of a specific diagnosis (Table 3). Men who are azoospermic secondary to testicular failure often present with small, soft testes measuring less than 10 cm3 with a small, flat epididymis. In the setting of primary testicular failure, decreased testosterone production produces diminished negative feedback inhibition on the pituitary, which in turn is stimulated to increase FSH secretion (hypergonadotropic hypogonadism). These men will often have excess conversion of testosterone to estradiol (aromatase activity) and respond to aromatase inhibitor therapy with normalization of testosterone levels and improved sperm production. Men with azoospermia secondary to obstruction have normal FSH levels, as testosterone production is normal, and the entire homone profile reflects the normal state. Although a significantly elevated FSH is consistent with spermatogenic failure, not all men with abnormal spermatogenesis have an elevated FSH. The combination of physical examination and hormone evaluation is extremely useful for differentiating whether azoospermia is caused by obstruction or by testicular failure. Of men with obstructive azoospermia, 96% have been found to have FSH levels of 7.6mIU/mL or less or testicular long axis greater than 4.6 cm, whereas 89% of men with nonobstructive azoospermia had FSH levels greater than 7.6 mIU/mL or testicular long axis of 4.6 cm or more.26 Men with primary testicular failure, small, soft testes, elevated FSH, and typically low ejaculate volume secondary to low androgen levels should be advised to undergo genetic evaluation to rule out chromosomal abnormalities such as Klinefelter syndrome and microdeletion of the Y chromosome.
|
TABLE 3. Correlation Between Serum Hormone Levels and Diagnosis
Diagnosis | T | FSH | LH | Prolactin |
Normal | Normal | Normal | Normal | Normal |
Primary Testicular | Low | High | Normal→High | Normal |
Failure (Hypergonadotropic hypogonadism) | ||||
Hypogonadotropic hypogonadism | Low | Low | Low | Normal |
Functional pituitary adenoma | Low | Low→Normal | Low | High |
FSH, follicle-stimulating hormone; LH, luteinizing hormone; T, testosterone.
Low testosterone, low FSH, and low LH are typical findings in hypogonadotropic hypogonadism wherein the pituitary does not produce adequate FSH resulting in low testosterone production. Hopogonadotropic hypogonadism may be caused by Kallman syndrome, a disorder of the hypothalamus in which sufficient GnRH is not produced, or by a pituitary defect, including pituitary adenoma. Kallman syndrome is also associated with midline abnormalities such as anosmia27 and less commonly synkinesia, unilateral renal agenesis and high, arched palate. Men with this hormone profile should undergo cranial MRI to evaluate the sella turcica for the presence of a pituitary adenoma.
Functional adenomas of the pituitary may produce high levels of prolactin, which in turn causes negative feedback inhibition on the gonadotropes of the pituitary gland with subsequent decrease in FSH, LH, and testosterone. Decreased libido may precede the subsequent establishment of a hypoandrogenic state that results in inadequate virilization. Cranial MRI is indicated to assess the pituitary gland. Although medical treatment for microadenomas and macroadenomas is usually the same, sizeable macroadenomas that may cause significant morbidity from mass effect should be detected for follow-up and possible neurosurgical consultation if the mass enlarges or causes other neurologic signs.
Recent investigation has shown that many men with nonobstructive azoospermia have an elevated testosterone:estradiol (T:E2) ratio when compared with the fertile population.28 Aromatase inhibitors decrease the conversion of testosterone and androstenedione to estradiol and estrone, respectively, thereby increasing serum testosterone levels. Administration of aromatase inhibitors has been found to not only restore the T:E2 toward normal, but also to significantly improve semen parameters including sperm concentration and motility in oligospermic men.28,29 An E2 level may be obtained in infertile, oligospermic men to determine the T:E2 ratio, although these data were not available for review and inclusion in the current recommendations of the Male Infertility Best Practice Policy Committee.25
Postejaculatory Urinalysis
Low-volume or absent ejaculate suggests bladder neck dysfunction and retrograde flow of ejaculate into the bladder. This may be seen in patients with diabetes mellitus or in men who have undergone surgical procedures of the urogenital tract such as bladder neck reconstruction or transurethral resection of the prostate. Men with ejaculate volume less than 1.0 mL who are not found to have hypogonadotropic hypogonadism or CBAVD should undergo postejaculatory urinalysis.25 Fluid production within the seminal vesicles and the prostate is dependent on androgen stimulation, and the hypoandrogenic state of hypogonadotropic hypogonadism does not support normal contribution of these glands to the semen. CBAVD is associated with hypoplasia of the seminal vesicles, also resulting in low-volume ejaculate that is acidic and often, but not always, lacks fructose.
Postejaculatory urinalysis determines the number of sperm that are present within the bladder after centrifugation of the specimen and careful examination of the pellet with light microscopy under 400× magnification. If any sperm are noted within the urine of a patient with azoospermia, then retrograde ejaculation is diagnosed. Patients with oligospermia are difficult to assess for retrograde ejaculation, as the sperm found in the urine may reflect sperm that are washed out of the urethra with urination, and the results must be interpreted cautiously.
Ultrasound
Transrectal ultrasonography (TRUS) is indicated for those patients with palpable vasa who have an acidic, low-volume ejaculate that contains absent or minimal fructose to assess for ejaculatory duct obstruction. In partial or unilateral ductal obstruction, the semen may contain fructose and the pH may be only subtly acidic. On ultrasound, the seminal vesicles can appear dilated (more than 1.5 cm in anteroposterior diameter) and the ejaculatory ducts may be visualized as well with obstruction.30 In an azoospermic patient thought to have ejaculatory duct obstruction, the seminal vesicles may be aspirated under ultrasound guidance,31 and intraoperative microscopic analysis of the aspirate is done. If present, motile sperm should be cryopreserved for use in IVF with ICSI. Definitive treatment is provided with transurethral resection of the ejaculatory ducts after which sperm may be seen to return to the ejaculate in 50% to 75% of cases with pregnancy occurring in 25% of couples.32 If viable, but poor-quality sperm return to the ejaculate, IVF with ICSI is recommended.
Unilateral vasal agenesis may be associated with contralateral segmental atresia of the vas deferens or seminal vesicle.33 TRUS can be done for evaluation of the ampullary portion of the contralateral vas and seminal vesicle. Furthermore, approximately 25% of men with congenital unilateral absence of the vas deferens and 10% of men with CBAVD have unilateral renal agenesis; abdominal ultrasound is indicated for this population for assessment of the kidneys.34
Transscrotal ultrasound is rarely necessary because the majority of scrotal pathology can be palpated on physical examination. Clinically significant varicoceles, congenital absence of the vas deferens, and testis tumors are most commonly diagnosed with palpation alone. Scrotal ultrasound is not indicated unless physical examination cannot be adequately accomplished or findings on physical examination are equivocal.
Genetic Analysis
Chromosome abnormalities are clearly associated with male infertility. The best characterized genetic anomalies are mutations within the CF transmembrane conductance regulator (CFTR) gene, the sex chromosome abnormality Klinefelter syndrome (47,XXY), and microdeletions of the Y chromosome.
MUTATIONS WITHIN THE CYSTIC FIBROSIS TRANSMEMBRANE CONDUCTANCE REGULATOR GENE.
CBAVD, congenital unilateral absence of the vas deferens, congenital bilateral partial absence of the vas or epididymides, and congenital epididymal obstruction comprise the spectrum of vasal aplasia. At least 80% of men with CBAVD are found to carry mutations within at least one allele of the cystic fibrosis transmembrane conductance regulator (CFTR) gene,35 located on the short arm of chromosome 7. The gene encodes a protein that acts as an ion channel but also affects formation of the distal two-thirds of the epididymis, the vas deferens, seminal vesicle, and ejaculatory duct. As greater numbers of mutations within the CFTR gene are discovered, the percentage of men with CBAVD who are found to harbor CFTR mutations increases. Possibly, all men with CBAVD carry mutations within the CFTR gene, and failure to detect these mutations represents both limitations within current testing methodologies and also practical issues with regard to the number of mutations that should be tested in a given patient, because some mutations carry a low frequency. Approximately 4% of Caucasians are carriers of CFTR gene mutations. Because men with evidence of vasal agenesis can be assumed to be carriers of CF mutations, the female partner must be evaluated for CF gene mutations prior to attempting ART to determine the risk for transmitting CF or CBAVD to the offspring. The penetrance of the CF carrier state in causing CBAVD appears to be low.36
KLINEFELTER SYNDROME.
Up to 10% of men with nonobstructive azoospermia have karyotypic abnormalities. The most common chromosome abnormality is Klinefelter syndrome (47,XXY or 46,XY;47,XXY mosaicism). The clinical spectrum of Klinefelter syndrome ranges from varied degrees of impaired spermatogenesis with complete absence of spermatogenesis on one end of the spectrum to severe oligospermia in rare cases. Testicular spermatozoa may be recovered in most men with Klinefelter syndrome using microsurgical testicular sperm extraction (TESE). Thus far, all children from our center born after ICSI with use of retrieved sperm have had normal karyotype. Other karyotypic abnormalities in infertile men include autosomal translocations.37 Preimplantation diagnosis (evaluation of fertilized embryos by biopsy during IVF) using fluorescence in situ hybridization for identification of normal chromosome composition may be considered when using sperm from men with known chromosomal abnormalities.38
MICRODELETIONS OF THE Y CHROMOSOME.
Y chromosome microdeletions are identified in up to 7% of infertile men.39 Relevant microdeletions may be detected on three nonoverlapping regions of the long arm of the Y chromosome, designated as AZF (AZoospermic Factor) a, b, and c.40 Because they are too small to be identified by karyotype analysis alone, deletions must be identified by polymerase chain reaction-based technique utilizing multiple sequence-tagged–sites. The presence of a deletion cannot be predicted by phenotype. Deletions within the different regions are associated with different sperm retrieval rates. Men with deletions of the AZFc region may have sufficient spermatogenesis to produce spermatozoa within the ejaculate,41 but if not, sperm may be retrieved by microdissection TESE for most of these men. Men with deletions of the AZFa or AZFb regions, however, have a poor prognosis for successful sperm retrieval.42,43,44 Deletions of the recently elucidated AZFd region are associated with normal spermatogenesis, and the clinical significance of deletions within this region remains to be determined.45
Given that the Y chromosome will be passed on to all male children, concern for transmission of impaired spermatogenesis to all male offspring exists, and vertical transmission of deletions has been demonstrated.46,47,48 Because of the prognostic value and implications for offspring, all azoospermic men considered for ICSI should be screened for both chromosomal abnormalities with karyotype analysis and Y chromosome microdeletions.25 The couples should be counseled not only regarding the possibility of passing infertility to the offspring, but also that genetic abnormalities may exist that have not yet been elucidated and are not detectable by current genetic testing methodologies.
Analysis of Sperm Function
Although sufficient sperm with adequate motility and normal morphology are found on semen analysis, the sperm may be unable to fertilize an oocyte. For this reason, additional analysis of sperm function may be indicated to identify specific deficiencies in some of the multitudinous components involved in normal sperm action, although utility in this effort is realized only if the findings will affect treatment options.
LEUKOCYTOSPERMIA.
Excess leukocytes within the semen are detrimental to sperm function and motility. Infertile men have been found to contain a greater number of white blood cells in their ejaculates when compared with fertile men.49 On routine semen analysis, several cell types appear similar and cannot be differentiated from one another, including epithelial cells, prostate cells, immature germ cells (round spermatids, spermatocytes, spermatogonia), and leukocytes. These cells are collectively referred to as round cells. If the number of round cells in a semen analysis exceeds 5 million per milliliter, then the percentage of these cells represented by leukocytes should be determined to assess the likelihood of genital tract infection. The histochemical peroxidase stain using ortho-toluidine or the immunocytochemical pan-leukocyte monoclonal antibody test, which has the advantage of detecting activated polymorphonuclear granulocyes as well as leukocytes that do not contain peroxidase, can be performed to determine the concentration of white blood cells within the semen. A concentration of white blood cells greater than 1 million per milliliter warrants semen culture for Mycoplasma hominis, Ureaplasma urealyticum, aerobic and anaerobic bacteria, and testing for Neisseria gonorrhoeae, Chlamydia trachomatis, and Trichomonas vaginalis. Prior to submitting a semen specimen for culture, the patient should carefully wash the penis with Betadine to diminish the likelihood of specimen contamination with skin flora.
ANTI-SPERM ANTIBODIES.
The blood-testis barrier created by tight junctions between Sertoli cells normally isolates sperm from immune recognition. When the barrier is disrupted and sperm are exposed to blood, however, an antigenic response is elicited. ASA may be found within the serum, semen, or bound to sperm. Those that are bound to sperm are most clinically significant, because they may disable sperm either by impeding transport through the cervical mucous50 or by altering interaction between the sperm and oocyte such that fertilization cannot occur.51 Antibodies found in semen are predominantly IgA and IgG, both of which may diffuse into the genital tract, and IgA is also secreted in the male reproductive tract. ASA are found in 80% of men who have undergone vasectomy52 and are also associated with vasoepididymostomy or vasovasostomy, testicular biopsy, infection, varicocele, cryptorchidism, and testicular torsion or trauma.53
Pregnancy rates are clearly lower in men with ASA than in men without, and among those men with ASA, significantly higher pregnancy rates are achieved if fewer than 50% of the sperm are bound by antibodies.54 Men with semen analyses that show clumping or agglutination of sperm or asthenospermia should be considered for ASA assessment. Either the immunobead test or the mixed antiglobulin reaction test can be used to screen for ASA. A fresh semen sample is used, and at least 200 sperm must be available for evaluation.15
SPERM VIABILITY.
Sperm viability is critical to ICSI success, and nonmotile sperm may not be viable. Clinical scenarios in which this is relevant include the use of testicular sperm, asthenospermia, or cryptozoospermia (sperm found only after centrifugation of the semen specimen within the pellet). Men with nonobstructive azoospermia who undergo microdissection testicular sperm extraction typically have low numbers of sperm that are found, and few may be motile. In this case, it is impossible to ascertain which sperm are viable for use in ICSI without conducting a sperm viability test. Viability testing is also important for cryopreserved specimens after the thaw process. Cryopreservation has been shown to decrease the number of viable sperm by 50%,55 and for specimens that contain few sperm prior to cryopreservation, rare sperm may be available following thaw. Viability testing may be necessary to select nonmotile, viable sperm for ICSI. In patients who are not azoospermic, viability testing will differentiate viable, nonmotile sperm from dead sperm, and if a large number of sperm on routine semen analysis are nonmotile, but viable, then defects of microtubule action (immotile cilia syndrome) must be considered.
Viability testing is indicated if less than 50% of sperm are motile.15 Two different types of testing modality are available: dye exclusion with eosin Y or trypan blue and hypo-osmotic swelling (HOS). With dye exclusion, the dead sperm with damaged plasma membranes allow dye to fill the cell, whereas viable cells exclude the dye. After the dye exclusion test, sperm do not remain viable and cannot be used for ICSI.
In hypo-osmotic fluid, water follows the solute concentration gradient and diffuses into viable sperm, causing the plasma membrane to bulge and the tail to curl,56 which can be easily observed with phase-contrast microscopy. A nonviable cell cannot maintain the osmotic gradient and does not swell. Sperm that react in the HOS test may be selected for use in ICSI. It is not clear that routine use of the HOS test improves ICSI results.
SPERM-CERVICAL MUCUS INTERACTION.
To achieve fertilization, the sperm must pass through the cervical mucus to enter the uterus and finally the fallopian tube. Just prior to ovulation, the cervical mucus changes so that it is receptive to spermatozoa and effectively filters out abnormal sperm, allowing those that are fit to pass into the uterus. The mucus also serves as a nutritive, protective reservoir to supply sperm to the uterus following intercourse. The postcoital test (PCT) is a microscopic evaluation of the cervical mucus after intercourse as close to ovulation as possible.57 The presence of motile sperm with adequate forward progression is a good indicator that the interaction between cervical mucus and sperm is not hostile.58 The routine application of the PCT has not been shown to confer benefit to infertile couples,59 and an absence of consensus exists as to the clinical value of this test. Although the significance of the test relative to fertilization potential is vague, the test may be useful to ascertain the efficacy of intercourse and proper deposition of semen within the vaginal vault.
SPERM PENETRATION ASSAY (ZONA-FREE HAMSTER OOCYTE TEST).
Removal of the species-specific zona pellucida from hamster oocytes allows sperm from a different species to fertilize one oocyte. This process requires sperm capacitation including the acrosome reaction, fusion with the oolemma and incorporation of sperm into the oocyte. Because multiple interactions are required, failure of an adequate number of sperm to penetrate one oocyte, or failure of sperm to penetrate an adequate percentage of oocytes, cannot be attributed to one specific defect. For example, this artificial test relies on the occurrence of spontaneous in vitro or unnaturally induced acrosome reactions.60 An absence of penetration is somewhat difficult to interpret, as the defect may lie within the acrosome reaction or within an indeterminate mechanism of penetration itself. Couples for whom the sperm penetration assay (SPA) test was negative have achieved success with IVF, and therefore extrapolation of SPA results is controversial.61
Variability also exists in the scoring of test results. Some laboratories assess the percentage of oocytes that are penetrated, while others assess the number of penetrations per oocyte. The sperm capacitation index, or mean number of sperm to penetrate an oocyte, was proposed to increase sensitivity of the test.62 Despite this change in result analysis, correlation of SPA results with pregnancy remains unclear, and the test should be considered only for those patients for whom the outcome of the test will influence treatment decisions.
COMPUTER-ASSISTED SEMEN ANALYSIS.
Computer-assisted semen analysis (CASA) was introduced as a technology intended to provide a precise, automated, reproducible, objective assessment of spermatozoon characteristics, specifically movement pattern and concentration. Computer analysis of digitized images procured with a video camera to identify specific characteristics of sperm motion may predict fertility potential. For production of meaningful, reproducible results, CASA requires standardization of sample preparation, frame rate and sperm concentration63 in addition to strict guideline adherence for use of the instruments. Widespread use and application of CASA is a complex endeavor, and unequivocal predictability of kinematic pattern effect on conception has not been demonstrated. Whether this technology offers additional information of clinical benefit to the infertile couple over routine manual semen analysis remains to be established.
ACROSOME REACTION.
The acrosome is a membrane-bound structure derived from the Golgi apparatus located most anteriorly in the head of the sperm, appearing as a fluid-filled nuclear cap, the outer acrosomal membrane located subjacent to the plasma membrane and the inner acrosomal membrane located anterior to the nuclear membrane. The sperm binds to the zona pellucida, the acrosome reaction occurs when the outer acrosomal membrane fuses with the plasma membrane, and acrosomal enzymes responsible for penetration of the zona pellucida are released. A high level of spontaneous acrosome loss and a lack of response to calcium ionophore A23187, which causes artificial induction of the acrosome reaction, are observed in men with infertility.64 Both fluorescent-labeled lectins and monoclonal antibodies can be used to assess the outer and the inner acrosomal membrane as well as acrosomal contents after artificial induction of the acrosome reaction. In vitro acrosome reaction dysfunction can be detected by this assay, but the clinical relevance of this observation is elusive.
HUMAN ZONA PELLUCIDA BINDING TESTS.
The hemizona assay is one of the tests that evaluates the ability of sperm to bind to the zona pellucida. The zona pellucida from a nonviable human oocyte (obtained from autopsy, surgical specimens, or most commonly from an IVF center) is divided in half. One half is incubated with patient sperm and the other with fertile control sperm at the same concentration. The number of patient sperm bound is assessed relative to the number of control sperm bound. Other tests differentially tag patient and control sperm allowing assessment of the general ability of the patient sperm to accomplish binding to the zona pellucida. Absent or low-frequency binding of sperm suggests a defect in this discrete step in fertilization. The test may have specific utility in assessing sperm that fail IVF with normal results on SPA, wherein the oocytes lack the zona pellucida. Widespread use of this testing is not possible due to extensive experience required with micromanipulation techniques and limited availability of human oocytes.
BIOCHEMICAL TESTING.
Reactive Oxygen Species.
Metabolism of oxygen can result in the formation of reactive oxygen species (ROS) that, when present in abundance, are toxic to aerobic cells. The detection of ROS formation in the semen of 40% of infertile men, but not in azoospermic and fertile men,65 suggested that ROS was a major potential cause or mediator of idiopathic infertility. ROS are normally produced by sperm and are necessary for normal sperm function. Low superoxide anion scavenging capacity in seminal plasma may be responsible for the accumulation of high levels of ROS.66 In fertile and infertile men, leukocytes are the major producers of ROS, but in oligospermic men, sperm may produce up to 167 times more ROS than in a corresponding fertile group.67 Motility, midpiece abnormalities and sperm-oocyte fusion are affected by ROS through peroxidation of sperm lipid membranes.68 Chemiluminescent probes can be used to detect the amount of ROS produced by sperm cells.
Creatine Phosphokinase.
Creatine phosphokinase is a key enzyme in the generation, transport, and use of energy within spermatozoa. Clinical evidence has suggested that elevated total creatine phosphokinase and perturbation in isozyme forms is associated with infertility,69 but others have demonstrated that total creatine kinase activity and isozyme distribution are not predictive of male fertility.70 Investigation is ongoing in biochemical tests of sperm function.
CONCLUSION
Evaluation of the male in infertility has evolved into an organized, logical process beginning with thorough reproductive and medical history, physical examination, and semen analysis. The results of this initial protocol determine either the course of treatment or the necessity for further evaluation with additional studies. When followed, the assessment described herein results in the effective accumulation of a large amount of information contributing to a diagnosis. Directed treatment that is appropriate for the individual patient may be instituted, such that the male factor contribution to infertility is most effectively surmounted.
REFERENCES
Corson SL, Maislin G: The National Registry Advisory Council Practice Survey for 2000. Fertil Steril 77:448, 2002 |
|
Thonneau P, Marchand S, Tallec A, et al: Incidence and main causes of infertility Corson in a resident population (1,850,000) of three French regions (1988–1989). Hum Reprod 6:811, 1991 |
|
Honig SC, Lipshultz LI, Jarow J: Significant medical pathology uncovered by a comprehensive male infertility evaluation. Fertil Steril 62:1028, 1994 |
|
Norbert C, Goldstein M: Increased incidence of testicular cancer in men with infertility and abnormal semen analysis [abstract O-132]. Fertil Steril 76:S50, 2001 |
|
Anderson L, Lewis SEM, McClure N: The effects of coital lubricants on sperm motility in vitro. Hum Reprod 13:3351, 1998 |
|
Kutteh WH, Chao CH, Ritter JO, et al: Vaginal lubricants for the infertile couple: effect on sperm activity. Int J Fertil Menopausal Stud 41:400, 1996 |
|
Goldenberg RL, White R: The effect of vaginal lubricants on sperm motility in vitro. Fertil Steril 26:872, 1975 |
|
Lee PA: Fertility in cryptorchidism: Does treatment make a difference? Endocrinol Metab Clin North Am 22:479, 1993 |
|
Beard CM, Benson RC Jr, Kelalis PP, et al: The incidence and outcome of mumps orchitis in Rochester, Minnesota, 1934–1974. Mayo Clin Proc 52:3, 1977 |
|
Sheynkin JR, Hendin BN, Schlegel PN, et al: Microsurgical repair of iatrogenic injury to the vas deferens. J Urol 159:139, 1998 |
|
Prader A: Testicular size: Assessment and clinical importance. Triangle 7:240, 1966 |
|
Kolettis PN: Is physical examination useful in predicting epididymal obstruction? Urology 57:1138, 2001 |
|
Gorelick JI, Goldstein M: Loss of fertility in men with varicocele. Fertil Steril 59:613, 1993 |
|
World Health Organization: The influence of varicocele on parameters of fertility in a large group of men presenting to infertility clinics. Fertil Steril 57:1289, 1992 |
|
World Health Organization: WHO Laboratory Manual for the Examination of Human Semen and Semen-Cervical Mucus Interaction. New York, Cambridge University Press, 1999 |
|
Kruger TF, Menkveld R, Stander FS, et al: Sperm morphologic features as a prognostic factor in in vitro fertilization. Fertil Steril 46:1118, 1986 |
|
Silber SJ, Nagy Z, Devroey P, et al: Distribution of spermatogenesis in the testicles of azoospermic men: The presence or absence of spermatids in the testes of men with germinal failure. Hum Reprod 12:2422, 1997 |
|
Ron-El R, Strassburger D, Friedler S, et al: Extended sperm preparation: an alternative to testicular sperm extraction in non-obstructive azoospermia. Hum Reprod 12:1222, 1997 |
|
Madgar I, Weissenberg R, Lunenfeld B, et al: Controlled trial of high spermatic vein ligation for varicocele in infertile men. Fertil Steril 63:120, 1995 |
|
Çayan S, Erdemir F, özbey İ, et al: Can varicocelectomy significantly change the way couples use assisted reproductive technologies? J Urol 167:1749, 2002 |
|
Kim ED, Leibman BB, Grinblat DM, et al: Varicocele repair improves semen parameters in azoospermic men with spermatogenic failure. J Urol 162:737, 1999 |
|
Matthews GJ, Matthews ED, Goldstein M: Induction and spermatogenesis and achievement of pregnancy after microsurgical varicocelectomy in men with azoospermia and severe oligoasthenospermia. Fertil Steril 70:71, 1998 |
|
World Health Organization Laboratory Manual for the Examination of Human Semen and Sperm-Cervical Mucus Interaction. 2nd ed. New York, Cambridge University Press, 1987 |
|
World Health Organization Laboratory Manual for the Examination of Human Semen and Sperm-Cervical Mucus Interaction. 3rd ed. New York, Cambridge University Press, 1992 |
|
Jarow JP, Sharlip ID, Belker AM, et al: Best practice policies for male infertility. J Urol 167:2138, 2002 |
|
Schoor RA, Elhanbly S, Niederberger CS, et al: The role of testicular biopsy in the modern management of male infertility. J Urol 167:197, 2002 |
|
Seminara SB, Hayes FJ, Crowley WF Jr: Gonadotropin-releasing hormone deficiency in the human (idiopathic hypogonadotropic hypogonadism and Kallmann’s syndrome): Pathophysiological and genetic considerations. Endocr Rev 19:521, 1998 |
|
Pavlovich CP, King P, Goldstein M, et al: Evidence of a treatable endocrinopathy in infertile men. J Urol 165:837, 2001 |
|
Raman JD, Schlegel PN: Aromatase inhibitors for male infertility. J Urol 167:624, 2002 |
|
Carter SS, Shinohara K, Lipshultz LI: Transrectal ultrasonography in disorders of the seminal vesicles and ejaculatory ducts. Urol Clin North Am 16:773, 1989 |
|
Jarow JP: Seminal vesicle aspiration in the management of patients with ejaculatory duct obstruction. J Urol 152:899, 1994 |
|
Jarow JP: Diagnosis and management of ejaculatory duct obstruction. Tech Urol 2:79, 1996 |
|
Hall S, Oates RD: Unilateral absence of the scrotal vas deferens associated with contralateral mesonephric duct anomalies resulting in infertility: Laboratory, physical and radiographic finding, and therapeutic alternatives. J Urol 150:1161, 1993 |
|
Schlegel PN, Shin D, Goldstein M: Urogenital anomalies in men with congenital absence of the vas deferens. J Urol 155:1644, 1996 |
|
Oates RD, Amos JA: The genetic basis of congenital bilateral absence of the vas deferens and cystic fibrosis. J Androl 15:1, 1994 |
|
Shin D, Gilbert F, Goldstein M, et al: Congenital absence of the vas deferens: incomplete penetrance of cystic fibrosis gene mutations. J Urol 158:1794, 1997 |
|
Rucker B, Mielnik A, King P, et al: Preoperative screening for genetic anomalies in men with non-obstructive azoospermia prior to sperm extraction. J Urol 160:2068, 1998 |
|
Tournaye H, Staessen C, Liebaers I, et al: Testicular sperm recovery in nine 47XXY Klinefelter patients. Hum Reprod 11:1644, 1996 |
|
Pryor JL, Kent-First M, Muallem A, et al: Microdeletions in the Y chromosome of infertile men. N Engl J Med 336:534, 1997 |
|
Vogt PH, Edelmann A, Kirsch, S, et al: Human Y-chromosome azoospermia factors (AZF) mapped to different subregions in Yq11. Hum Mol Genet 5:933, 1996 |
|
Mulhall JP, Reijo R, Alagappan R, et al: Azoospermic men with deletion of the DAZ gene cluster are capable of completing spermatogenesis: Fertilization, normal embryonic development and pregnancy occur when retrieved testicular spermatozoa are used for intracytoplasmic sperm injection. Hum Reprod 12:503, 1997 |
|
Kamp C, Huellen K, Fernandes S, et al: High deletion frequency of the complete AZFa sequence in men with Sertoli-cell-only syndrome. Mol Hum Reprod 7:987, 2001 |
|
Krausz C, Quintana-Murci L, McElreavey K: Prognostic value of Y deletion analysis: What is the clinical prognostic value of Y chromosome microdeletion analysis? Hum Reprod 15:1431, 2000 |
|
Brandell RA, Mielnik A, Liotta D, et al: AZFb deletions predict the absence of spermatozoa with testicular sperm extraction: Preliminary report of a prognostic genetic test. Hum Reprod 13:2812, 1998 |
|
Kent-First M, Muallem A, Shultz J: Defining regions of the Y-chromosome responsible for male infertility and identification of a fourth AZF region (AZFd) by Y-chromosome microdeletion detection. Mol Reprod Dev 53:27, 1999 |
|
Silber SJ, Repping S: Transmission of male infertility to future generations: Lessons from the Y chromosome. Hum Reprod Update 8:217, 2002 |
|
Cram DS, Ma K, Bhasin S, et al: Y chromosome analysis of infertile men and their sons conceived through intracytoplasmic sperm injection: Vertical transmission of deletions and rarity of de novo deletions. Fertil Steril 74:909, 2000 |
|
Chang PL, Sauer MV, Brown S: Y chromosome microdeletion in a father and his four infertile sons. Hum Reprod 14:2689, 1999 |
|
Wolff H, Politch JA, Martinez A, et al: Leukocytospermia is associated with poor semen quality. Fertil Steril 53:528, 1990 |
|
Haas GG Jr: The inhibitory effect of sperm-associated immunoglobulins on cervical mucus penetration. Fertil Steril 46:334, 1986 |
|
Mahoney MC, Blackmore PF, Bronson RA, et al: Inhibition of human sperm-zona pellucida tight binding in the presence of antisperm antibody positive polyclonal patient sera. J Reprod Immunol 19:287, 1991 |
|
Alexander NJ, Anderson DJ: Vasectomy: Consequences of autoimmunity to sperm antigens. Fertil Steril 32:253, 1979 |
|
Turek PJ: Infections, immunology, and male infertility. Infertil Reprod Med Clin North Am 10:435, 1999 |
|
Ayvaliotis B, Bronson R, Rosenfeld D, et al: Conception rates in couples where autoimmunity to sperm is detected. Fertil Steril 43:739, 1985 |
|
Prins GS, Dolgina R, Studney P, et al: Quality of cryopreserved testicular sperm in patients with obstructive and nonobstructive azoospermia. J Urol 161:1504, 1999 |
|
Jeyendran RS, Van der Ven HH, Perez-Pelaez M, et al: Development of an assay to assess the functional integrity of the human sperm membrane and its relationship to other semen characteristics. J Reprod Fertil 70:219, 1984 |
|
Moghissi KS: Post-coital test: Physiologic basis, technique, and interpretation. Fertil Steril 27:117, 1976 |
|
Oei SG, Helmerhorst FM, Keirse MJ: When is the post-coital test normal? A critical appraisal Hum Reprod 10:1711, 1995 |
|
Oei SG, Helmerhorst FM, Keirse MJ: Routine postcoital testing is unnecessary. Hum Reprod 16:1051, 2001 |
|
Aitken RJ, Buckingham DW, Huang GF: Analysis of the response of human spermatozoa to A23187 employing a novel technique for assessing the acrosome reaction. J Androl 14:132, 1993 |
|
Aitken RJ: Diagnostic value of the zone-free hamster oocyte penetration test and sperm movement characteristics in oligozoospermia. Int J Androl 8:348, 1985 |
|
Smith RG, Johnson A, Lamb DJ, et al: Functional tests of spermatozoa: Sperm penetration assay. Urol Clin North Am 14:451, 1987 |
|
Davis RO, Katz DF: Standardization and comparability of CASA instruments. J Androl 13:81, 1992 |
|
Fenichel P, Donzeau M, Farahifar D, et al: Dynamics of human sperm acrosome reaction: Relation with in vitro fertilization. Fertil Steril 55:994, 1991 |
|
Iwasaki A, Gagnon C: Formation of reactive oxygen species in spermatozoa of infertile patients. Fertil Steril 57:409, 1992 |
|
de Lamirande E, Gagnon C: Human sperm hyperactivation in whole semen and its association with low superoxide scavenging capacity in seminal plasma. Fertil Steril 59:1291, 1993 |
|
Aitken RJ, Buckingham D, West K, et al: Differential contribution of leucocytes and spermatozoa to the generation of reactive oxygen species in the ejaculates of oligozoospermic patients and fertile donors. J Reprod Fertil 92:451, 1992 |
|
Kim JG, Parthasarathy S: Oxidation and the spermatozoa. Semin Reprod Endocrinol 16:235, 1998 |
|
Huszar G, Vigue L, Oehninger S: Creatine kinase immunocytochemistry of human sperm-hemizona complexes: Selective binding of sperm with mature creatine kinase-staining pattern. Fertil Steril 61:136, 1994 |
|
Rolf C, Gehre HM, Cooper TG, et al: Creatine kinase activity in human spermatozoa and seminal plasma lacks predictive value for male fertility in in vitro fertilization. Fertil Steril 69:727, 1998 |