Steroid Endocrinology of Pregnancy
Authors
INTRODUCTION
To establish and successfully maintain a human pregnancy requires the coordinated secretion of hormones within and between the fetus, mother, and placenta. The placenta synthesizes and secretes steroid and peptide hormones that regulate hormonogenesis by endocrine glands in both the mother and the fetus. Placental hormones also act in a paracrine and/or autocrine manner to regulate growth and differentiation of placental cytotrophoblast and syncytiotrophoblast, growth and maturation of the placental vascular tree, and uterine endovascular invasion by extravillous cytotrophoblast. Moreover, the placenta metabolizes the large quantities of steroid hormones produced by the maternal endocrine glands to protect the fetus and to orchestrate the timing and development of fetal organ systems, the fetal pituitary-adrenocortical axis in particular. Clearly, placental hormonogenesis and metabolism are among the most important determinants of a successful pregnancy; however, despite the numerous advances in our understanding of placental-fetal function, our knowledge of the factors acting and interacting to regulate these processes during human pregnancy remains incomplete.
The purpose of this chapter is to review the classic and more recent concepts regarding: (1) placental trophoblast development, implantation, and uterine endovascular invasion; (2) regulation of estrogen and progesterone production by the placenta; (3) the role of estrogen and placental metabolism of maternal cortisol in maturation of the fetus and the fetal pituitary-adrenocortical axis; (4) development of the fetal gonads; and (5) the mechanisms responsible for the initiation of parturition. Because the study of maternal-fetal-placental function during human pregnancy is necessarily limited by ethical constraints, this chapter draws heavily on relevant information derived from in vivo experiments conducted in nonhuman primate models. Finally, because of its broad scope, this chapter provides a more general review, rather than an in-depth analysis, of the factors important to pregnancy maintenance, parturition, and fetal development. More in-depth reviews of key aspects of placental and fetal adrenocortical-gonadal development have recently been published.1,2,3,4,5,6,7,8,9,10,11,12,13
IMPLANTATION AND EARLY PREGNANCY
Development of the Embryo
The union of individual male and female gametes represents an orderly and highly regulated sequence of events collectively termed fertilization.14 Fertilization involves maturation of spermatozoa15,16,17 by capacitation, and their movement through the cumulus of the ovum,18 although human sperm need not undergo capacitation to traverse the cumulus.16 Sperm binding to zona pellucida proteins (ZP1-4), primarily ZP3,19,20 precedes the acrosome reaction21 that is essential for sperm fusion with the oocyte membrane.22 After fusion of the egg and sperm membranes, release of cortical granules in the oocyte results in conformational changes in ZP2 and ZP3 that render the ovum impenetrable to additional sperm.16
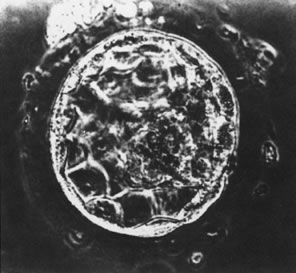
At the 8-cell stage, blastomeres begin to form gap and tight junctions,23,24 a process that initiates segregation of inner cells from outer cells and marks the onset of embryo differentiation.25 After additional cell divisions, the embryo (now termed a morula) enters the uterus, approximately 4 days after ovulation in both humans and nonhuman primates.26 After formation of the blastocoele, cells of the blastocyst differentiate into an inner cell mass destined to form the fetus and an outer mass of cells destined to become the placenta (Fig. 1).27 This process appears to be modulated by cell adhesion molecules, since E-cadherin-null embryos fail to form a trophectoderm.28 The developing blastocyst continues to grow and essentially floats in the uterine cavity for an additional 2 to 3 days during which time the blastocyst (embryo) hatches from the surrounding zona pellucida.29,30,31 Hatching appears to be essential for embryo-uterine contact and for subsequent implantation that occurs between 7 and 9 days after ovulation in humans and most nonhuman primates. Enders32,33 has concluded that prior to attachment to the uterine epithelium, mononucleated cytotrophoblast cells of the trophectoderm fuse to form a syncytiotrophoblast layer. The syncytiotrophoblast appears to initially interact with and adhere to the endometrium. Only after the embryo is totally embedded in the endometrium do cytotrophoblast cells begin to move from the trophoblastic shell to invade the uterus and the uterine vasculature.34,35,36
Preparation of the Uterus for Implantation
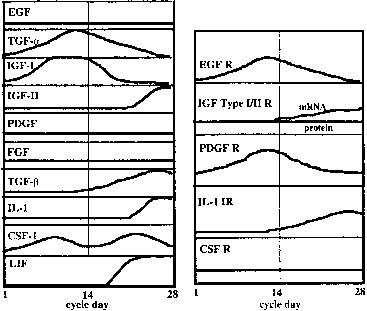
As the embryo (blastocyst) is developing and moving through the fallopian tubes into the uterus, the endometrium undergoes extensive differentiation ultimately to permit embryo attachment (i.e., implantation). In virtually all mammals, preparation of the uterus for implantation is regulated by the coordinated actions of estrogen and progesterone37 produced and secreted by the ovary-corpus luteum and perhaps by the developing embryo itself.38,39 Estrogen and progesterone act on the endometrium and myometrium directly through estrogen-progesterone receptor-mediated events, and indirectly by stimulation of various growth factors (e.g., EGF, TGF-B, insulin-like growth factor I (IGF-I) and IGF-II), proteins (e.g., placental protein 14, 24 kd protein, CA-125), and cytokines (e.g., IL-1, CSF-1) (Fig. 2).40 Uterine receptivity to a putative implantation signal is limited to a discrete period of time during the luteal phase of the menstrual cycle consistent with the concept of a window of implantation that has been advanced in several animal models and in the human.41,42,43 However, despite intense investigation, the molecular basis for the transient nature of uterine receptivity, estimated to span days 19/20 to 24 of the menstrual cycle,35,44,45 remains unknown. During this interval, and presumably under the influence of estrogen and progesterone, the uterine endometrium is thickened and highly secretory in nature and becomes rich in glycogen and lipids.37 Morphologic studies have shown that uterine receptivity is heralded by the formation of pinopods on the apical surface of endometrial epithelial cells, a process that appears to be regulated by progesterone.46 Pinopods may absorb fluid from the uterine cavity, thereby making the endometrium more accessible to the blastocyst. Changes in the composition of the uterine glycocalyx have also been observed during the peri-implantation period. For example, the levels of two transmembrane glycoproteins, mucin (MUC-1) and keratan sulfate, increase on the endometrial glandular surface during the early luteal phase,9,47,48 then decrease as the window for implantation opens.9,48 Because MUC-1 is a relatively large cell surface molecule, its down-regulation may unmask smaller molecules on the uterine surface, such as the cadherins and integrins, thereby mediating specific adhesion of the trophectoderm to the endometrium. Epithelial integrins have been proposed as markers for the window of implantation in the human.49,50 Endometrial integrins are expressed in an “on/off” pattern,51 and both they and their ligands are expressed in trophectoderm.52 Taken together, these observations support the hypothesis that integrins may be involved in early implantation events. The β 3 integrin subunit is expressed on the endometrial surface after day 19 of the human menstrual cycle, just when the endometrial window of implantation opens.53 The fact that this integrin is almost never expressed by epithelial cells, but is expressed by human trophectoderm,54 supports a role for these molecules in implantation. A similar distribution of endometrial integrins and extracellular matrix proteins has been described across the menstrual cycle and early pregnancy in the baboon.55 Human secretory phase endometrium also produces the glycoprotein leukemia-inhibiting factor(LIF).56,57 LIF has the capacity to inhibit embryonic stem cell differentiation in vitro,58 and LIF receptors are detected on human blastocysts.56,57 Moreover, Stewart and colleagues59 have demonstrated that LIF (production of which is regulated by estrogen and progesterone) is essential for implantation in the mouse since implantation of blastocysts did not occur in mutated mice lacking a functional LIF gene. A similar role for LIF in humans and nonhuman primates has not been confirmed.
In addition to expression of integrins, the human blastocyst/embryo secretes a number of factors at various times during its development that may be essential for pregnancy recognition or its normal progression. Almost immediately after fertilization, the embryo secretes platelet-activating factor (PAF), interleukins-1 and -6, and early pregnancy factor. Although inhibition of PAF activity in vivo prevented implantation in rodents,60 a similar role in primate pregnancy has not been demonstrated. By the 8-cell stage, the blastocyst apparently secretes a number of cytokines and growth factors including chorionic gonadotropin(hCG), long recognized as one embryonic factor that is essential for early pregnancy in primate species.31,61,62,63,64
Rescue of the Corpus Luteum by Human Chorionic Gonadotropin
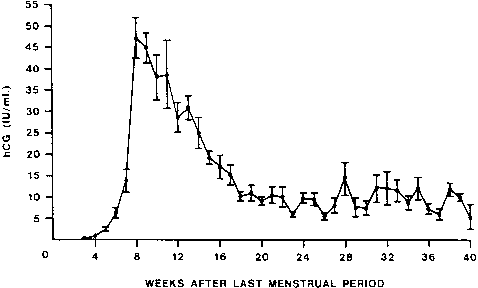
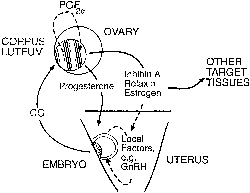
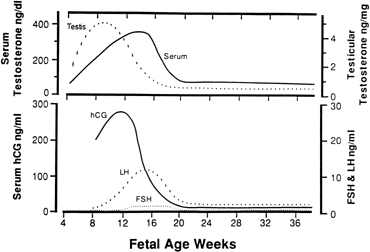
hCG is a glycosylated protein heterodimer composed of noncovalent bound α and β subunits.65 β-hCG is structurally similar to the β subunit of pituitary luteinizing hormone (LH), differing only in the terminal 28 amino acids. Whereas α subunit genes are expressed in the pituitary and placenta,66 the 6 β subunit genes located on chromosome 19q13.3 are expressed only in the placenta.67 The three-dimensional structure of hCG has also been determined68; both the highly glycosylated α and β subunits69 contain several disulfide linkages that form cysteine knot motifs.68,70 Deglycosylation studies indicate that the carbohydrate moieties of the α, but not the β, subunit are critical for activation of the hCG receptor and its associated G-protein coupled signaling systems.70 In human pregnancy, hCG levels in maternal blood are detectable approximately 3 days after implantation71 and rapidly rise thereafter with peak levels being achieved between weeks 8 and 12 of gestation (Fig. 3).72 Although in vitro studies suggest that hCG may be produced by hatched embryos prior to attachment, expression is generally assumed to occur after attachment.26,73 In the absence of hCG, the newly formed corpus luteum will invariably regress; production of estrogen and progesterone declines resulting in endometrial shedding (i.e., menses). The hCG produced by the syncytiotrophoblast binds to the LH/CG receptor in the corpus luteum to stimulate progesterone and estradiol synthesis, presumably by increasing low-density lipoprotein receptor expression and thus facilitating uptake of cholesterol substrate for steroidogenesis (Fig. 4).31 Immunohistochemical studies also suggest that hCG may act to increase the expression of rate limiting steroidogenic enzymes (e.g., 3β-hydroxysteroid [-HSD] and aromatase; Fig. 5).74 In some species, but perhaps not the human, rescue of the corpus luteum by hCG may also involve local inhibition of factors (e.g., prostaglandin) that promote luteal demise (e.g., luteolysis). hCG is also thought to stimulate corpus luteum relaxin production (see Fig. 4) and, as discussed later, appears to enhance testosterone production by the fetal testes that is required for differentiation of the internal duct system and external genitalia in males. The high levels of hCG secreted into the maternal circulation during early gestation apparently are sufficient to bind to the thyroid-stimulating hormone (TSH) receptor and thereby to increase maternal thyroid hormone production.75 Although our current understanding of the regulation of hCG production remains incomplete, it has been proposed that gonadotropin-releasing hormone (GnRH) produced by the placental cytotrophoblast may modulate hCG production by binding to GnRH receptors in the syncytiotrophoblast.5 Interestingly, human placental trophoblast produces a variety of other hypothalamic-/pituitary-like neuropeptides,5 including corticotropin-releasing hormone (CRH), proopiomelanocortin (POMC), neuropeptide Y, oxytocin, somatostatin, and thyrotropin-releasing hormone (TRH). Although the physiologic role of most of these factors remains to be defined, CRH may play a role in parturition,76,77 modulation of uterine blood flow,78 and regulation of maternal pituitary ACTH production.79,80 Finally, the trophoblast also produces several protein hormones (chorionic somatomammotropin [hCS] and growth hormone variant [GH-V]) and growth factors (IGF-I and IGF-II) that are considered essential to the regulation of maternal, and perhaps fetal, intermediary metabolism as well as growth and maturation of the placenta.81
Placental Development, Vascularization, and Intrauterine Endovascular Invasion
DECIDUALIZATION
Upon attachment of the embryo to the uterus, the uterine endometrial glycogen-containing stromal cells are rapidly transformed into large decidual cells that increase in size throughout the course of gestation. The human decidua that represents the maternal component of the placenta is composed primarily of the decidua basalis, underlying the site of implantation, the decidua capsularis that initially overlies the gestational sac but gradually disappears with advancing gestation, and the decidua vera that lines the remainder of the uterine cavity. In humans, as well as the baboon,82 the decidua produces and secretes a variety of factors that include the hormones relaxin and prolactin, the IGF-binding protein (IGF-BP-1), and a variety of other proteins (e.g., placental protein 14, also known as progesterone-associated endometrial protein [PEP], or glycodelin).
In the human, IGF-BP-1 is secreted by the stromal cells that surround the spiral arteries during the late luteal phase, whereas in the baboon, IGF-BP-1 is secreted by the endometrial glands in response to progesterone.83,84 IGF-BP-1 is one of several proteins that bind IGF-I and IGF-II and thereby regulate the ability of these growth factors to interact with their receptors. Ritvos and associates85 have suggested that decidual IGF-BP1 may act to control invasion and/or proliferation of trophoblast cells during implantation/placentation by sequestering the IGFs. IGF-BP-1 levels increase rapidly during early gestation in parallel with decidualization, then transiently decline before increasing again in late pregnancy.86,87 Pregnancy termination with the progesterone receptor antagonist RU-486 is associated with a marked decline in IGF-BP-1 levels, an observation suggesting that decidual IGF-BP-1 production is progesterone dependent.53 It has also been hypothesized that restructuring of the decidual cell cytoskeleton is essential for IGF-B1 gene expression.88
Glycodelin, synthesized by the uterine glandular epithelium, shares homology with the B-lactoglobulins and retinol-binding proteins89 and has been implicated as an immunosuppressive agent.90,91 Close temporal relationships in serum profiles suggest that glycodelin production is regulated by progesterone92,93 and/or relaxin.94 Serum glycodelin levels are elevated in the luteal phase of the menstrual cycle and markedly increase during the first trimester of pregnancy. In the baboon, although the pattern of uterine glycodelin mRNA and protein expression mimics that in the human, glycodelin production is regulated by hCG,95 perhaps by direct action on the endometrium.
Decidual prolactin production begins on day 22 of the idealized human menstrual cycle (approximately 8 days after ovulation)96 and prolactin mRNA and protein expression is observed in the epithelial cells of the deep basal glands of the baboon uterus during the late luteal phase. Decidual prolactin expression increases markedly with advancing gestation and is stimulated by progesterone in both the human97,98 and baboon.99 Decidual prolactin is apparently secreted into the amniotic fluid in humans100,101 and baboons,102 and levels increase with advancing gestation. Although the precise role(s) of decidual prolactin remains unclear, it has been proposed that the hormone may enhance uterine contractility, an action also apparently antagonized by decidual relaxin.103,104
PLACENTAL TROPHOBLAST DEVELOPMENT AND VASCULARIZATION
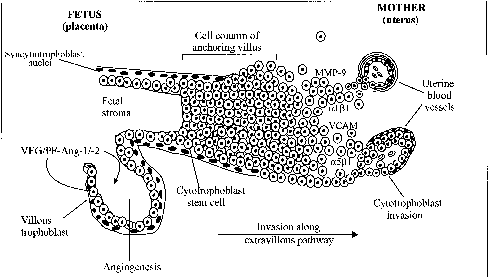
During human and nonhuman primate pregnancy, the placenta simultaneously accesses the maternal blood and develops a vascular network for the transport of nutrients to and waste products from the fetus across the syncytiotrophoblast to ensure fetal growth and development. Both processes depend on the ability of the primordial stem-cell cytotrophoblasts to take either the villous pathway where they remain in the fetal compartment and differentiate morphologically into the syncytiotrophoblast or the extravillous pathway where they proliferate, aggregate into cell columns of the anchoring villi, and invade the endometrial stroma (Fig. 6).105 The syncytiotrophoblast covers the floating chorionic villi that become highly vascularized, whereas the extravillous cytotrophoblasts infiltrate the walls of the spiral arterioles to facilitate the process of placentation.
The vascular network within the placental villous core develops by in situ differentiation of fetal mesenchymal cells into vessels (vasculogenesis) and proliferation of existing vessels (angiogenesis) resulting in secondary and tertiary villi equipped with a functional arterio-capillary-venous system.34 Development of the uteroplacental circulation begins early in pregnancy with neovascularization of both fetal and maternal tissues.106,107,108 Neovascularization results in increased blood flow and increases in the effective exchange area crucial to support fetal development. In humans, placental vasculogenesis begins at approximately 21 days of gestation109 and continues through at least the 26th week of pregnancy.34 Angiogenesis accompanies vasculogenesis and is critical not only early in gestation but also important for early widespread extension of the fetal capillary system and later growth of the fetal vascular compartment during the final third of pregnancy.109,110 Several factors expressed by the placenta, including basic fibroblast growth factor,111,112,113 platelet-derived growth factor,114 placental growth factor,115,116 and vascular endothelial growth/permeability factor (VEG/PF)115 ,117 have been proposed as regulators of placental angiogenesis. Among these, VEG/PF selectively stimulates endothelial cell proliferation and the formation of new blood vessels.118,119 It also induces vascular permeability by actions on endothelial cells resulting in extravasation of plasma proteins that provide an extracellular fibrin matrix for angiogenesis.120 VEG/PF is encoded from a single gene and expressed as 5 isoforms having 121, 145, 165, 189, or 206 amino acids,121,122 the 121 and 165 isoforms exhibiting the greatest angiogenic activity.123,124,125 VEG/PF mRNA and protein are expressed by cytotrophoblasts, syncytiotrophoblast, and Hofbauer macrophage cells within the villous human placenta.114,117,124,126 Inactivation of the VEG/PF gene in transgenic mice results in significant defects in the vasculature of embryonic tissues and organs that are lethal.127
VEG/PF binds to two structurally related transmembrane tyrosine kinase receptors, VEG/PF flt-1 and KDR/flk-1,128,129 which are expressed on placental villous vascular endothelial cells126,129,130 and in villous and extravillous trophoblasts.117,126,131 Both receptors are essential for vascular development. Homozygous KDR/flk-1 defective mice die in utero as a result of an early defect in hematopoietic and endothelial cell development,132 and mouse embryos homozygous for the flt-1 mutation die because of failure to organize normal vascular channels.133
Two other closely related proteins, angiopoietin-1 and -2, work in concert with VEG/PF in signaling vascular morphogenesis by binding to the endothelial cell-specific transmembrane tyrosine kinase receptor Tie-2. In Tie-2 null mice, endothelial cells develop and assemble into tubes, but vessels are immature, lacking branching networks, encapsulation by periendothelial support cells, and proper organization into small and large vessels.134,135 These associations indicate that the Tie-2 receptor may also mediate the capacity of endothelial cells to recruit stromal cells that encase endothelial tubes for vessel stabilization. Transgenic/gene knock-out studies further indicate that angiopoietin-1 signals Tie-2 to recruit vascular support cells and that angiopoietin-2 inhibits this action136,137,138 by competitively inhibiting angiopoietin-1-induced kinase activation of the Tie-2 receptor. During early human pregnancy, angiopoietin-1 is localized to the cytotrophoblast and syncytiotrophoblast, angiopoietin-2 to the cytotrophoblast, and Tie-2 receptor to the endothelium, 139,140 observations that are consistent with the proposed role of the angiopoietin-Tie-2 system in development of the placental circulation.
Despite the importance of angiogenesis to neovascularization of the developing placenta, very little is known about the regulation of the process and the expression of the VEG/PF-angiopoitin-1/-2 system during human pregnancy. Although hypoxia is a potent stimulus of VEG/PF expression,141 estrogen has also been shown to regulate VEG/PF expression in the rat uterus142,143,144 and in human endometrial cells.145 Moreover, chronic estrogen treatment induced uterine angiogenesis in normal but not estrogen receptor-null transgenic mice.146 In the baboon, cytotrophoblast VEG/PF mRNA levels and vascularization of the villous placenta increase with advancing gestation in parallel with increasing placental estrogen production.147 Thus, the well-established role for estrogen in enhancing uteroplacental blood flow148, 149 may not only reflect changes in vascular reactivity but also enhanced angiogenesis.
At the same time the vascular system is developing within the chorionic villi, a select population of extravillous cytotrophoblasts migrate and invade the spiral arteries of the uterine endometrium at the placental-decidual junction (see Fig. 6). Histologic studies performed during the first half of human, baboon, and macaque pregnancy32, 150,151,152,153,154,155 demonstrate that cytotrophoblasts migrate to and colonize spiral arterioles/arteries by displacing endothelial cells from their basal lamina and partially or completely replacing the smooth muscle component within the tunica media. Consequently, the structure of spiral arteries and the dynamics of blood flow within them are modified by cytotrophoblast invasion, presumably to facilitate implantation and placentation.
As cytotrophoblasts differentiate into cells capable of invading the uterine stroma and blood vessels, their expression of adhesion molecules changes in the human,8,105,156,157,158,159 baboon,160 and rhesus monkey.154,161 As extravillous cytotrophoblasts migrate, expression of the integrin complex α5β1 and α6β4 is lost and that of the α1β1 laminin/collagen receptor is induced.158,162,163 Moreover, interaction of the α1β1 receptor with collagen type IV promotes, whereas interaction of the α5β1 receptor with fibronectin inhibits, invasion of human cytotrophoblasts in vitro.164,165 Zhou and associates105 have suggested that cytotrophoblasts balance invasion-retraining and invasion-promoting adhesion mechanisms as they differentiate. Migratory cytotrophoblast cells also express specific adhesion molecules, specifically vascular cell adhesion molecule (VCAM) and cadherins (e.g., VE-cadherin), that appear to secure cytotrophoblasts to each other and to the endothelium, thereby facilitating their migration against arterial blood flow.161,166,167 Interstitial and spiral arteriole invasion is also associated with the expression of matrix metalloproteinases (MMPs; e.g., MMP-9 collagenase), by intraluminal, extravasating, and intramural cytotrophoblasts,168,169 apparently to disrupt the extracellular matrices in the tunica media to allow cytotrophoblasts to modify the vessel wall. MMP-9 promotes cytotrophoblast invasion; the capacity of human cytotrophoblast to invade is completely inhibited by MMP-9 antibody in vitro.170 Clearly, the process of endovascular spiral artery invasion involves an intricately and temporally ordered expression of integrins, adhesion molecules, and proteinases by the extravillous cytotrophoblasts. This area of research is of intense interest and clinically relevant. Abnormal expression of several of these components has been observed in cytotrophoblasts of women who develop preeclampsia and in whom endovascular invasion is superficial.171 Unfortunately, our current understanding of the factors acting/interacting to regulate the timely expression of ECM and cell adhesion molecules by invading trophoblasts remains incomplete.
PLACENTAL STEROID HORMONE PRODUCTION
Estrogen Production
CONCENTRATIONS IN THE CIRCULATION
The placenta becomes the primary source of estrogens (Fig. 7) after approximately week 9 of human pregnancy (23% of gestational length172). As a result of extensive 16-hydroxylation of c19 steroids within the fetus,173 large quantities of estriol are produced by the placenta during human pregnancy. A fourth estrogen, estetrol, is also produced uniquely but in relatively low levels during human pregnancy.174
Plasma concentrations of estrone, estradiol, and estriol increase as human pregnancy progresses (Fig. 8)174 with daily excretion rates at term approximating 2, 1, and 40 mg, respectively.172,174 In humans, plasma concentrations of estradiol near term range from 6 to 30 ng/mL.175,176 In women with threatened first-trimester abortion, abnormal estradiol concentrations are highly associated with a subsequent pregnancy loss.177 Moreover, a 50% spontaneous abortion rate has been observed among women having a mutation in the amino terminal region of the estrogen receptor involved in transcription.178 In the baboon, reduction of maternal estrogen levels to less than 0.1 ng/mL by daily administration of an inhibitor of placental estrogen synthesis resulted in a 66% incidence of abortion during early gestation that was prevented by treatment with exogenous estradiol.179 Low serum estradiol concentrations during the third trimester are also associated with poor obstetric outcome.180,181 Taken together, these observations suggest that estrogen plays a critically important role in the maintenance of primate pregnancy, but others have refuted this notion182 because pregnancy is maintained in most women having low estrogen levels resulting from deficiencies in various placental enzymes. 183,184 Interestingly, in those cases, although maternal estradiol levels are markedly reduced, concentrations approximate 0.45 ng/mL, or 10-9 mol/L, a concentration that approximates the dissociation constant for estradiol binding to its receptor.185 Differences in the outcome of pregnancy in various women with estrogen deficiency suggest that the important biologic effects of estradiol can be achieved with available receptor and concentrations of estrogen sufficient to interact with it. It would appear that in both human and nonhuman primate pregnancy, estrogen is produced in considerable excess.
FETAL-PLACENTAL UNIT
The primate placenta does not express the P450 17α-hydroxylase/17–20 lyase enzyme (P450c17) and thus cannot convert de novo those c21 progestogens (pregnenolone and progesterone) to the c19 androgens (dehydroepiandrosterone [DHA] and androstenedione).186 As shown by Ryan187 and Siiteri and MacDonald,188 the placenta can convert c19 androgens into estrogen. Because pregnancy with an anencephalic fetus is associated with very low levels of urinary estrogen and fetal adrenal hypoplasia, Frandsen and Stakeman189 suggested that the fetal adrenal may provide the requisite androgen precursors for placental estrogen production. Siiteri and MacDonald188,190 demonstrated that DHAS of maternal and fetal origin contributed about equally to estrone and estradiol synthesis, whereas more than 90% of estriol was synthesized from 16-hydroxy DHAS of fetal origin (Fig. 9).191 They subsequently188 proposed the existence of a fetal-placental unit for estrogen biosynthesis, a concept that was later confirmed by Diczfalusy and colleagues.192,193 A similar fetal-placental unit for the formation of estrogen also exists in several nonhuman primates including the rhesus monkey and the baboon.191
ENZYMES FOR ESTROGEN BIOSYNTHESIS
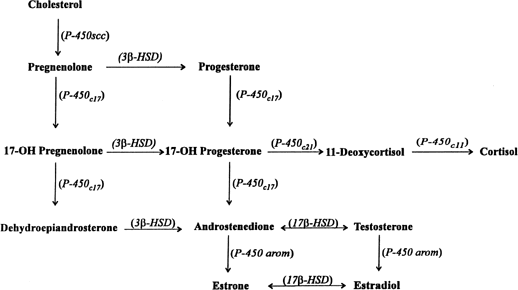
Placental synthesis of estrogens by conversion of c19-steroid precursors requires the enzymes sulfatase, Δ5-3β-hydroxysteroid dehydrogenase/Δ5-4 isomerase (3β-HSD), P450 aromatase(P450arom), and 17β-hydroxysteroid oxidoreductase (17β-HSD) (see Fig. 9). Once secreted by the fetal adrenal gland, DHA is rapidly sulfated in the fetal liver to form DHAS and subsequently hydroxylated at carbon 16 to form 16-hydroxy DHAS, the primary precursor for estriol formation. On arrival in the placenta, sulfurylated c19 steroids precursors are desulfurylated by the enzyme sulfatase to yield unconjugated DHA and 16-hydroxy DHA. Although the regulation of sulfatase is unknown, prolactin and oxytocin stimulate enzyme activity in decidual cells isolated before the onset of human labor.194 Mitchell and colleagues195 have also observed a significant increase in hydrolysis of estrone sulfate by fetal membranes at term that could increase free estrogen concentrations locally within tissues with no associated change in peripheral plasma estrogen concentrations.
Unconjugated Δ5 — c19 steroid precursors in the placenta are subsequently converted to androstenedione/testosterone or 16-hydroxyandrostenedione by the enzyme 3β-HSD. A family of closely related genes encode for 3β-HSD196 and various 3β-HSD isoforms are expressed in a tissue-specific manner. The human type I 3β-HSD is expressed at high levels in placenta, whereas the type II isoform is almost exclusively expressed in the adrenal cortex and gonads. In the placenta, the multinucleated syncytiotrophoblast is the principal site of 3β-HSD expression.
The subsequent conversions of androstenedione and testosterone to estrone and estradiol and 16-hydroxyandrostenedione to estriol (see Fig. 9) are catalyzed by P450arom, an enzyme consisting of an aromatase cytochrome P450 and the flavoprotein, NADPH-cytochrome P450 reductase.197,198P450arom is a member of the cytochrome P450 superfamily of enzymes that includes approximately 220 members.199 The enzyme binds c19-steroid substrates and catalyzes a series of reactions resulting in the formation of the phenolic A ring that is characteristic of estrogens (see Fig. 7). The entire three-step process is catalyzed at one active site by a single cytochrome P450 species.200
Recent studies have shown that the CYP19 gene encoding human cytochrome P450arom is located on chromosome 15 and consists of 9 exons and two polyadenylation sites in the last coding exon downstream from the terminating stop codon that gives rise to the 3.4 and 2.9 kb transcripts that encode human P450arom. Although the entire intron sequences remain to be mapped, the CYP19 gene is at least 70 kb long and is the largest cytochrome P450 gene characterized to date (reviewed elsewhere199).
The interconversions of androstenedione and testosterone and of estrone and estradiol are catalyzed by 17β-HSD. There are at least four isoforms of 17β-HSD, with the placenta expressing 17β-HSD-1 and -2.201,202,203 Placental 17β-HSD-1 has been localized to chromosome 17 and is a cytosolic enzyme that preferentially catalyzes the reduction of estrone to estradiol but does not utilize androgens as substrates.201 In contrast, placental 17β-HSD-2 has been localized to chromosome 16 and catalyzes the oxidation of estradiol to estrone201,204,205 and of testosterone to androstenedione.204,205,206,207 Estrone and estradiol are extensively interconverted within the placenta, but these two estrogens are not secreted in equal amounts into the maternal and fetal compartments in the human,208,209 rhesus monkey,210 or baboon.211 Biologically active estradiol is secreted primarily into the maternal circulation, whereas the weaker estrogen, estrone, is preferentially released into the fetal circulation, perhaps to limit exposure of the developing fetus to estrogen. Although a carrier system specific for estradiol has been suggested to explain this selective secretion,210 differential localization of the 17β-HSD-1 and -2 enzymes within the syncytiotrophoblast may be a more likely explanation, but remains to be confirmed.
REGULATION
The 3β-HSD, sulfatase, and aromatase enzymes are abundantly expressed in the primate placenta,191 and the production of estradiol and estrone per milligram placental protein remains relatively constant throughout pregnancy in women.212 Although cAMP and certain growth factors can modulate estrogen production by transformed trophoblast cell lines in vitro,213,214 placental estrogen production appears most dependent on the amount of substrate provided by the fetal adrenal gland, uteroplacental blood flow, and placental trophoblast mass.
Role of Uteroplacental Blood Flow
The fraction of circulating maternal DHAS converted to estradiol increases significantly from 5% in the first trimester to more than 35% late in gestation,190,215 reflecting increases in both placental mass34 and uteroplacental blood flow. 216 The metabolic clearance rate (MCR) of DHAS increases from approximately 7L/day in the nonpregnant state to more than 60L/day in the third trimester (Fig. 10).215 The influence of uteroplacental blood flow on placental estrogen production has been confirmed by direct experiment. Novy and colleagues217,218 demonstrated that reductions in uteroplacental perfusion caused by graded reductions in maternal distal aortic blood flow in the pregnant baboon resulted in corresponding decreases in the placental clearance of DHA through estradiol formation. Consistent with these observations in the baboon, the MCR of DHAS is 50% lower in women with pregnancy-induced hypertension, presumably reflecting the marked decrease in intervillous perfusion that characterizes this clinical syndrome.219,220,221
Fetal Adrenal c19 Steroids
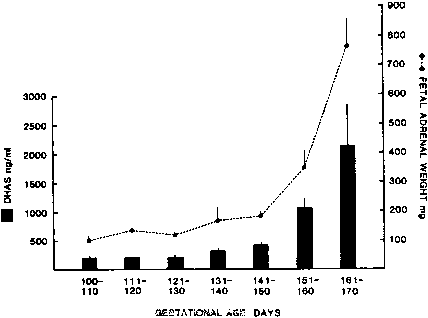
The fetal adrenal is a major source of the c19 steroids required for estrogen production; growth and function of the gland therefore greatly influence the production of estrogen. In fetal baboons, acute stress induced by hypoxemia results in a marked increase in fetal adrenal c19-steroid secretion and results in a corresponding rise in placental estrogen production.222 As pregnancy advances, the primate fetal adrenal exhibits a marked increase in growth, accompanied by a corresponding increase in umbilical serum levels of DHAS (Fig. 11).223,224,225,226 Throughout most of gestation, 75% or more of the human fetal adrenal cortex6 consists of the fetal zone, which expresses the P450c17 and P450scc mRNAs and proteins essential for c19 steroid DHA and DHAS production (see Fig. 5). In contrast, the “transitional zone,” which expresses both the 3β-HSD and P450c17 enzymes for cortisol production, and the “definitive zone,” which expresses 3β-HSD but not P450c17 and makes aldosterone,6,227 do not develop until very late in gestation. A number of studies have endeavored to define the mechanisms controlling growth and differential steroidogenesis in the three zones of the fetal adrenal gland (reviews available elsewhere1,2,6). Suppression of fetal pituitary ACTH secretion by administration of synthetic glucocorticosteroids (e.g., betamethasone) during mid- to late gestation results in fetal adrenal atrophy in humans,228 baboons,229,230 and rhesus monkeys.231 These observations indicate that fetal pituitary ACTH has a critical role in the growth and maintenance of the fetal adrenal in primates. Indeed, fetal anencephaly, accompanied by absence of pituitary ACTH, is associated with fetal adrenal atrophy and a striking decline in mid- to late gestation estrogen production in humans232 and rhesus monkeys.233 Clearly, by stimulating fetal adrenal growth and the overall capacity for c19-steroid formation, fetal ACTH has a major role indirectly in regulating placental estrogen biosynthesis. This tropic action of ACTH appears to be mediated in part by peptide growth factors (e.g., IGF-II, bFGF) in fetal adrenocortical cells.234,235,236,237
ACTH also has an important role in regulating elements of the steroid biosynthetic pathway in the primate fetal adrenal gland. Receptor-mediated uptake of low-density lipoprotein (LDL) cholesterol produced in the fetal liver is a major source of cholesterol substrate for steroidogenesis within the human fetal adrenal.238 Addition of LDL to cultures of human fetal adrenal cells results in increased production of DHAS and cortisol.239,240 ACTH has an important role in stimulating the receptor-mediated uptake and degradation of LDL within the human fetal adrenal to provide cholesterol substrate for steroidogenesis.241,242 In addition, the de novo pathway for the formation of cholesterol may account for up to 30% of the daily secretion of DHAS and cortisol by the human fetal adrenal gland in culture.243,244 Under these conditions, ACTH stimulates HMG CoA reductase, the rate-limiting enzyme for de novo teroidogenesis.244 The ACTH-stimulated LDL pathway appears to be the preferred mechanism for fueling steroidogenesis in the primate fetal adrenal gland, however, because the HMG CoA reductase enzyme is not stimulated in the presence of ACTH and LDL.
In addition to supplying cholesterol substrate to the fetal adrenal cell, ACTH regulates specific enzymes involved in c19-steroid formation. For example, the mRNAs for and activities of the P450 cholesterol side-chain (P450scc) and P450c17 enzymes, and the hydroxysteroid-sulfotransferase (HST) enzyme that catalyzes the sulfurylation of DHA, are stimulated by ACTH in cultures of human fetal adrenal cells. 235,245,246 Moreover, betamethasone suppresses and ACTH restores expression of the mRNAs for the ACTH receptor, P450scc and P450c17, and serum estrogen levels in baboons.229 Thus, by stimulating fetal adrenal c19-steroid formation, fetal ACTH has a pivotal role in regulating placental estrogen biosynthesis during primate pregnancy.
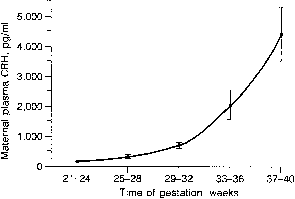
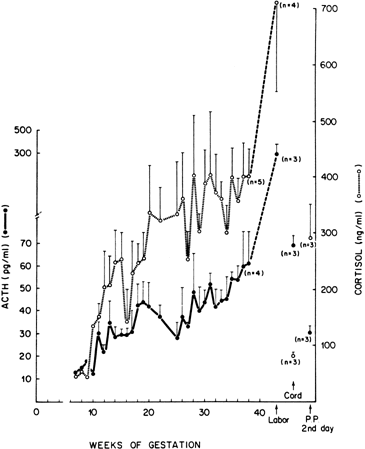
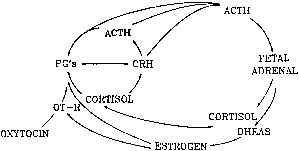
Because estrogen production is only partially reduced after suppression of the fetal hypothalamic pituitary adrenal axis with synthetic cortico-steroids,172,247,248,249 it appears that factors other than ACTH may also be important to the regulation of fetal adrenal hormonogenesis. hCG,250,251 prolactin, growth hormone, and several other peptides, including ACTH, CLIP, and MSH252 stimulate DHAS production by incubates of human fetal adrenal slices. Similar observations have been made in the baboon fetal adrenal both in vitro253 and in vivo.254 Recently, it has also been shown that placental CRH stimulates DHAS production and P450c17 expression by human fetal adrenal cells through activation of a phospholipase C-inositol second messenger system.255 Placental CRH may also regulate the maternal pituitary-adrenocortical axis. Thus, the progressive increase in maternal plasma CRH levels in human pregnancy (Fig. 12)256 is accompanied by a corresponding rise in the concentrations of maternal ACTH and cortisol (Fig. 13)257 as well as levels of DHAS.258 Interestingly, in cultured human trophoblasts, glucocorticoids increase the expression of CRH,80,259 and it has been proposed that a positive feed-forward loop involving the maternal pituitary-adrenocortical axis and placental CRH may operate during primate pregnancy (Fig. 14).260 Whether a comparable feed-forward axis operates in the primate fetus, as originally suggested by Robinson and associates,80 remains to be determined.
Studies also suggest that estrogen itself modulates fetal and perhaps maternal adrenal steroidogenesis. The primate fetal adrenal gland expresses estrogen receptors α and β at mid- and late gestation thereby providing a mechanism for mediating the action of estrogen.261 Estrogen stimulates ACTH-induced DHAS production and inhibits ACTH-induced cortisol synthesis in human fetal adrenal cells in culture.6,262,263,264 It has been proposed,6 therefore, that the availability of DHAS for placental estrogen production is controlled by a positive feedback loop in which estrogen alone and/or in conjunction with other factors enhances production of precursor DHAS by the fetal zone cells. In contrast to the stimulating effect of estrogen in long-term cultures of human fetal adrenal cells, estrogen inhibits ACTH-induced stimulation of DHA both in vivo265 and in short-term incubates of fetal baboon adrenal cells.266 Moreover, in the absence of any changes in MCR, maternal serum levels and production rates of DHA and DHAS are markedly reduced by estrogen treatment of intact or fetectomized baboons.267 Based on these observations, it has been proposed that placental estrogen feeds back to down-regulate the biosynthesis and secretion of DHA by the maternal and fetal adrenal glands to maintain a physiologically normal balance of estrogen production during primate pregnancy.2,3
Progesterone Production
CONCENTRATIONS IN THE CIRCULATION
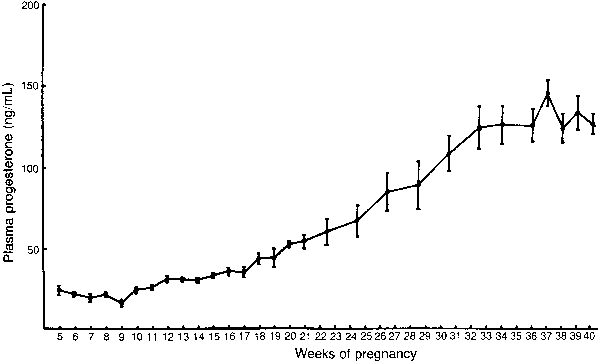
The placenta becomes a significant source of progesterone by approximately weeks 6 to 8 (15% to 20% of gestational length) of human pregnancy.268 In women, serum progesterone concentrations rise in a linear fashion with advancing gestation, ultimately attaining values of 150 to 175 ng/mL at term (Fig. 15).176,269 Fetal progesterone levels are more than twofold greater than in the mother during human,270 baboon,271 and rhesus monkey272 pregnancy, in largest part a reflection of the lower MCR of progesterone in the fetus than in the mother.272 In humans, the placental production rate of progesterone during the third trimester approximates 210 mg/day,273 a value more than 10-fold greater than in the luteal phase of the menstrual cycle.274 Progesterone levels in women with first-trimester threatened abortion are predictive of pregnancy outcome.177 Abortion ultimately results in more than 80% of women with progesterone levels below 10 ng/mL; no viable pregnancy is associated with concentrations below 5 ng/mL.177,275,276 Progesterone concentrations are also typically low in women with ectopic pregnancies.276 In contrast, progesterone levels are elevated in women with hydatidiform mole, particularly between the 10th and 20th weeks of gestation.277 In pregnancies complicated by Rh isoimmunization, progesterone levels are more than twofold higher than normal, presumably reflecting the marked increase in placental mass associated with erythroblastosis.176 Finally, although maternal progesterone concentrations fall dramatically and levels rise in late gestation before the onset of parturition in rats,278 sheep,279 and other laboratory species,191 no such changes occur in primates including humans,176 baboons,280,281,282,283 and rhesus monkeys.284
BIOSYNTHESIS OF PROGESTERONE—LDL PATHWAY
The primate placenta possesses abundant quantities of the P450scc and 3β-HSD enzymes required to convert substrate cholesterol into progesterone. Unlike estrogen formation, progesterone production does not require direct participation of the fetus; however, in contrast to other steroid-secreting organs, the human and nonhuman primate placenta exhibits a very limited capacity for de novo cholesterol and progesterone synthesis from acetate.285,286 The elegant work of Simpson and co-workers285,287 conclusively demonstrated that progesterone biosynthesis in human placental trophoblast cells follows the classical LDL pathway first described by Goldstein and Brown288 in fibroblasts. Earlier studies conducted in pregnant women289,290 and with perfusion of the human fetoplacental unit in vitro291,292 showed that cholesterol in the maternal circulation is taken up and used for progesterone formation by the placenta. Moreover, Winkel and associates287,293 demonstrated that LDL uptake by human trophoblast cells in culture is mediated by high-affinity binding, is saturable and that LDL degradation is a consequence of cellular uptake and internalization. A receptor-mediated pathway, involving coated pits and vesicles, endosomes, and lysosome-like bodies has been identified in human placental trophoblasts for the uptake of colloidal-gold conjugated LDL.294 Henson and colleagues295 have also demonstrated the existence of high-affinity receptor-mediated LDL uptake (i.e., binding and internalization), as well as LDL degradation by trophoblasts isolated from the baboon placenta. High-density lipoprotein also increases progesterone secretion by human trophoblast cultures,287,296 but to a much lesser extent than LDL.
REGULATION
Role of Estrogen
During rat and rabbit pregnancy, estrogen is the major luteotropic stimulus that maintains the corpus luteum and progesterone production.297,298,299 Estrogen stimulates the uptake of high-density lipoprotein cholesterol substrate300 and P450scc expression in the rat301 and rabbit302 corpus luteum, thereby promoting progesterone production. During mid- to late primate pregnancy, when the placenta is the principal source of progesterone, estrogen has a similar regulatory role within trophoblasts. 2,3,191 Placental progesterone formation and serum progesterone concentrations are decreased by administration of the estrogen receptor antagonist ethamoxytriphetol283,303,304 in baboons, an effect that can be reversed by diethylstilbestrol.305 Moreover, placental progesterone production by human trophoblasts in culture is inhibited by treatment with an aromatase inhibitor and restored by estradiol.306 The increase in receptor-mediated LDL uptake307 and LDL receptor308 and P450scc309 mRNA expression in placental trophoblasts observed during the second half of baboon pregnancy when estrogen levels rise, can be suppressed by blocking the action or formation of estrogen.304,310,311,312 In contrast, placental 3β-HSD and adrenodoxin mRNA expression and 3β-HSD activity are not developmentally regulated or altered by antiestrogen treatment in baboons.281,309 Therefore, inhibiting the action or levels of estrogen specifically blocks the developmental increase in placental LDL cholesterol uptake and expression of the P450scc enzyme essential for the metabolism of cholesterol to pregnenolone in baboons.
Effects of cAMP
In cultures of human trophoblast cells, cAMP increases the secretion of progesterone and hCG313,314 and expression of the mRNAs for P450scc.234,315 The observation that cAMP stimulates progesterone formation in cytotrophoblasts that are prevented from transforming into syncytiotrophoblasts suggests that the process is not dependent on syncytia formation. Strauss and co-workers have postulated that cAMP activates a protein kinase that phosphorylates a protein(s) integral to gene transcription of specific components of the progesterone pathway.316,317
Other Factors
Translocation of cholesterol from the outer to the inner mitochondrial membrane accounts for the rapid increase in steroidogenesis within the adrenals and gonads in response to tropic stimulation and is mediated in large part by the steroidogenic acute regulatory protein (StAR; review available elsewhere318). However, because human trophoblast cells do not express StAR,319 other unique mechanisms may be involved in the intracellular trafficking of cholesterol within the placenta. Recent studies317 indicate that the mitochondria of human syncytiotrophoblasts exhibit a morphology that is quite different from cytotrophoblast, adrenal, or gonadal cells. Their unique structure may in some way facilitate cholesterol entry into syncytiotrophoblast mitochondria without the mediation of factors such as StAR.
Various peptide growth factors (e.g., IGF-I and IGF-II) are expressed320,321 and have stimulatory effects on P450scc activity322 in human trophoblasts. EGF also has been observed to stimulate progesterone formation in JEG-3 choriocarcinoma cells in culture.85,323 The relative physiologic significance of peptide growth factors in primate placental progesterone production and their potential interaction with the other factors that have been discussed above remains to be determined.
PLACENTAL STEROID HORMONE METABOLISM
Estrogen Regulates the Developmental Pattern of Cortisol-Cortisone Metabolism by the Primate Placenta
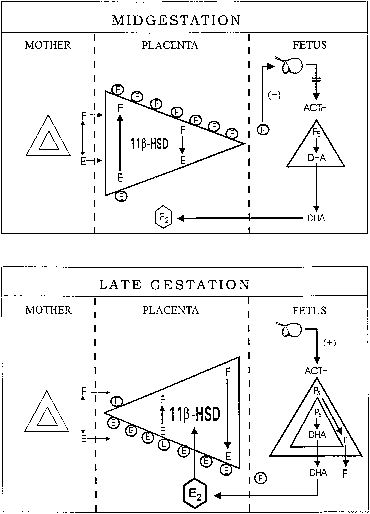
Although steroid hormones and other lipophilic substances of maternal origin rapidly traverse the human and nonhuman primate placenta,2 in most instances these compounds are catabolized to biologically inactive metabolites before entry into the fetal circulation.324,325,326,327 With regard to cortisol, it has generally been considered324 that the major role of the primate placenta was to catabolize biologically active cortisol (i.e., binds to the glucocorticoid receptor) to its inactive (does not bind to receptor) metabolite cortisone. However, in vivo isotope dilution studies in the baboon have shown that in contrast to the relatively constant pattern of cortisol-cortisone metabolism by maternal tissues during both baboon328 and human pregnancy,329 the pattern of placental cortisol-cortisone metabolism changes from preferential reduction(formation of cortisol from cortisone) early in pregnancy to oxidation(conversion of cortisol to cortisone) near term.330 The same may also occur in the human; indirect estimates of placental cortisol metabolism in vivo early in gestation indicate an extensive conversion of cortisone to cortisol across the placenta.331 Moreover, the change in transplacental corticosteroid metabolism is prevented by inhibiting estrogen action in pregnant baboons,332 and placental oxidation of cortisol to cortisone can be induced prematurely at midgestation by estradiol.333 It has therefore been proposed (Fig. 16)334 that estrogen regulates the ontogenetic change in placental glucocorticoid metabolism.2
Estrogen stimulation of placental oxidation of cortisol also appears to occur in fetal tissues. Conversion of cortisol to cortisone (29%) by the baboon fetus at midgestation335 increases twofold (60%) by the time of delivery,336 increases at midgestation (50%) in response to maternal estrogen administration,337 and decreases at term (21%)in neonates delivered to mothers treated with antiestrogen.338 Although the transfer constant measured in peripheral blood represents metabolic contributions of several individual organ systems, one of the sites of estrogen action appears to be the kidney.339
The interconversion of cortisol and cortisone is catalyzed by two 11β-hydroxysteroid dehydrogenase (11β-HSD-1 and -2) enzymes that are the product of two different genes.340,341 In the human and baboon placenta, the mRNA and protein for both 11β-HSD-1 and -2 are primarily expressed in the syncytiotrophoblast.334,342,343,344 Recently, it was demonstrated that the mRNA and protein levels of 11β-HSD-1 and -2,334 and the activity of 11β-HSD oxidase,345 in syncytiotrophoblast increases with advancing gestation and that upregulation may involve a direct estrogen receptor-dependent action of estrogen on the promoters of these two genes.346 Estrogen has also recently been shown to regulate the levels of 11β-HSD-1 and -2 protein in rat uterine endometrium.347 It has been proposed, therefore, that estradiol acts through its receptor to regulate the functional maturation of baboon syncytiotrophoblast that is further manifested by an increase in the genomic expression of the 11β-HSD enzymes controlling cortisol-cortisone interconversion (see Fig. 16).
Although the estrogen-dependent up-regulation of 11β-HSD-2 is consistent with increased transplacental oxidation of cortisol to cortisone at term,330 the concomitant up-regulation of 11β-HSD-1 is surprising since the reduction of cortisone to cortisol across the placenta declined with advancing gestation; however, because the syncytiotrophoblast is a polarized cell, it is possible that once the levels of 11β-HSD-1 and -2 are increased by estrogen, the syncytiotrophoblast may undergo further development resulting in a spatial compartmentalization and functional separation of the two 11β-HSD enzymes.334,348 For example, Burton and associates 347 have shown that 11β-HSD-1 and -2 exhibit marked differences in their expression between basal and labyrinth zones of the rat placenta.
Regulation of the Fetal Pituitary Adrenocortical Axis by Estrogen-Induced Changes in Placental Cortisol-Cortisone Metabolism
Because the transition zone of the primate fetal adrenal develops relatively late in gestation (see1,6 for review), the fetal adrenal gland in human349,350 and nonhuman primates 351,352,353 does not synthesize cortisol de novo for most of intrauterine development. In contrast, it appears that the baboon hypothalamus354,355 and pituitary gland356 develop relatively early in gestation, as previously shown in the human. 357,358 Thus, development of the fetal adrenal appears to be “out-of-phase” with that of the hypothalamus and pituitary, and yet by term, the pituitary adrenocortical axis has become functionally integrated. Studies in the baboon, however, have demonstrated that at midgestation essentially all of the cortisol measured in the fetus is of maternal origin, whereas at term less than 50% derives from the mother.330 Estrogen-dependent regulation of placental metabolism of maternal cortisol and cortisone is an integral step in the sequence of events that control development of the fetal pituitary adrenocortical axis(see Fig. 16). Because placental glucocorticoid metabolism during early to midgestation favors reduction of cortisone to cortisol, the primary maternal corticosteroid reaching the fetal circulation appears to be biologically active cortisol that would inhibit fetal ACTH production and thus limit growth and maturation of the fetal adrenal transitional zone.1,2 With advancing gestation, the estrogen-induced increase in placental 11β-HSD- catalyzed oxidation of cortisol to cortisone would decrease the concentration of maternal cortisol in the fetus causing activation of fetal pituitary ACTH production culminating in maturation of the fetal adrenal gland. Moreover, the concomitant increase in oxidation of cortisol to cortisone within the fetus could act, in concert with increased placental oxidation of maternal stores, to ensure continued synthesis and release of ACTH by the fetal pituitary gland.1,337,338,339 In support of this hypothesis, fetal pituitary ACTH expression,359 the specific activities of rate limiting enzymes,360 and the ontogenesis of de novo fetal baboon adrenal cortisol production361 increase with advancing gestation and can be induced prematurely at midgestation by estrogen induction of placental NAD-dependent 11β-HSD oxidase activity.345
FETAL GONADAL DEVELOPMENT
In humans, bipotential gonads develop as stratifications of the coelomic epithelium at approximately week 4 of gestation. Although most cell types comprising the gonads are derived from mesoderm of the urogenital ridges,10 primordial germ cells originate from ectoderm of the inner cell mass362 and migrate to the coelomic epithelium of the gonadal ridges.363,364,365 By week 5 of gestation, the bipotential gonad consists of germ cells, supporting cells of the coelomic epithelium that give rise to testicular Sertoli cells or ovarian granulosa, and stromal/interstitial cells of mesenchymal origin.10 It appears that two XX chromosomes are required for differentiation of the bipotential gonad into an ovary, whereas a single Y chromosome (e.g., 46XY) encoding the Sry gene and Sry-related transcription factors (e.g., SOX9) are necessary for testicular development.366,367,368 Subsequent development of the gonads continues throughout gestation with histologic differentiation of the fetal testes preceding that of the ovary.369,370
In the male, Leydig cells rapidly proliferate between weeks 12 and 18 of gestation,369,371 accompanied by increasing fetal testicular production and fetal serum testosterone levels (Fig. 17).372 At the same time, Sertoli cells produce androgen-binding protein (ABP)373 and the protein hormone müllerian-inhibiting hormone (MIH) in the developing seminiferous tubules.374,375 Jost demonstrated that castration of rabbit embryos of either sex resulted in the development of the female phenotype,376,377 and thereby established that the female urogenital tract develops spontaneously, whereas secretions of the fetal testes were necessary for male development. Presumably, testosterone binds to ABP to create a locally high concentration of androgen that induces differentiation of the Wolffian duct, whereas MIH causes the demise of the múllerian primordia. Differentiation of the external genitalia/prostate into the male phenotype is induced by testosterone secreted into the fetal circulation that is subsequently converted to 5α-dihydrotestosterone(DHT) by the enzyme 5α-reductase type I378 in the primordia of the external genitalia and by 5α-reductase type II in the prostate.379,380,381,382 Androgen treatment of female embryos induces male development of the internal and external genitalia,383 whereas administration of antiandrogenic agents during embryogenesis impairs male development.384,385 The female fetus is usually protected from testosterone and androstenedione of maternal origin,386,387 presumably because these androgens are either catabolized by the placenta and/or fetal liver,388 and/or are converted to estrogens by placental aromatase.188,389
Testosterone synthesis in the fetal testes appears to be regulated in large part by increasing levels of placental hCG382 and perhaps by fetal pituitary FSH/LH, levels of which peak at approximately weeks 12 to 20 of human pregnancy (see Fig. 17). Observations that testosterone synthesis begins before any significant increase in fetal gonadotropin levels372 and that LH (i.e., hCG) receptors are detected in fetal testicular tissues suggests an important role for hCG.390,391 The observation that male pseudohermaphroditism can result from a defect in testicular gonadotropin receptors provides further evidence.392 LDL cholesterol provides the substrate for fetal testicular steroidogenesis,393 and LDL receptor levels decline after week 20 of gestation in association with the decrease in fetal serum levels of testosterone. Finally, the fetal testes expresses the mRNAs for P450scc, P450c17, and 3β-HSD, all required for formation of testosterone.11,394
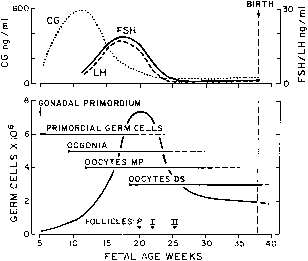
Histologic development of the fetal ovary is strikingly similar in humans and other nonhuman primates, such as rhesus monkeys.369,395,396 In both species, mitotic activity in oogonia is maximal at approximately 45%of gestation, with the number of germ cells falling substantially thereafter. Meiosis is initiated in the largest oogonia (now called oocytes)by the end of the first third of gestation with more germ cells entering meiosis and proceeding through leptotene, zygotene, and pachytene stages as gestation advances (Fig. 18).397 As early as week 10 of gestation, human oocytes become surrounded by a single layer of presumptive granulosa cells, the unit now termed a primordial follicle. Ohno and Smith398 demonstrated that many oocytes regress during the early stages of meiosis unless enveloped by granulosa cells once they have entered the diplotene stage. At approximately 40% of gestation, the process of follicular development intensifies with maturation and subsequent development of primary follicles (i.e., flattened granulosa cells become cuboidal and begin to divide) occurring principally in the medullary (inner)component of the ovary. Preantral follicles containing an enlarged oocyte with a zona pellucida, multiple layers of granulosa cells, and a theca cell layer that originates from the stroma occasionally develop at approximately the 6th to 8th month of gestation in humans11,399 and near term in nonhuman primates.399,400 Finally, it has been shown that the number of degenerating oocytes increases rapidly during folliculogenesis, primarily at the expense of diplotene oocytes. By term, the human and nonhuman primate ovary is thus composed of a relatively wide cortex filled with oocytes and several primordial and growing primary follicles, including preantral follicles primarily located in the inner medullary region. Although the histologic features of fetal ovary development are well defined, our current understanding of the factors that regulate this process remains incomplete.
PREGNANCY MAINTENANCE AND PARTURITION
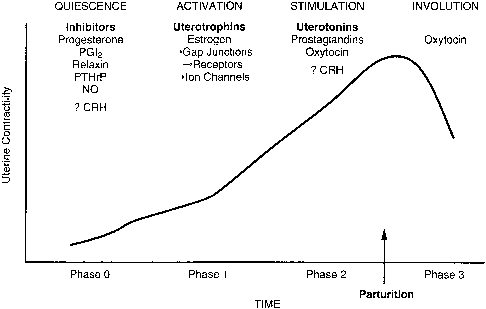
Throughout most of gestation, the uterus remains relatively quiescent. Although uterine contractions occur, they are of low amplitude, not synchronized, and very little uterine pressure is developed. This pattern of uterine activity generally classified as Braxton Hicks contractions in humans, and contractures in baboons, rhesus monkeys, and sheep401,402 has been termed phase 0 (Fig. 19)403 of the parturition process.12,404,405 At term, the uterus demonstrates well-coordinated contractions. The myometrium becomes highly excitable and generates contractions that increase in both frequency and amplitude. Transition from a quiescent to an active myometrium is termed activation or phase 1. During phase 2, an activated myometrium becomes increasingly responsive to various stimulatory factors.405,406,407 In this sequence, the initiation of parturition corresponds to the transition from phase 0 to phase 1 or from uterine quiescence to uterine activation (see Fig. 19).
Progesterone is generally considered to be a gene suppressor and indeed does down-regulate a number of genes that are considered essential for parturition, including the gap junction protein connexin 43.408,409 Moreover, progesterone alone and/or in conjunction with estradiol has been shown to restrain uterine contractility during pregnancy by (1) controlling calcium uptake/availability from external stores through both calcium channels and fast sodium channels; (2) mobilization of calcium from internal stores by regulation of α-1-adrenergic function; (3) removal of calcium from the uterine cytosol; and (4) coupling of β2-receptor to adenylate cyclase and the activation of protein kinase A to reduce calcium-calmodulin binding to myosin-light chain kinase (MLCK) and thus phosphorylation and activity of MLCK.2,12 In species in which progesterone withdrawal normally precedes labor (e.g., rats, rabbits, sheep), administration of progesterone prevents delivery,410 whereas treatment with antiprogestins late in gestation results in premature parturition similar to spontaneous labor induction. In contrast, in species in which progesterone production does not decline prior to labor (e.g., nonhuman primates and humans), exogenous progesterone does not prevent parturition at term411; however, treatment with the progesterone receptor antagonist RU-486 does lead to increased uterine activity and induction of premature labor.412 Moreover, suppression of progesterone production in late rhesus monkey gestation causes premature vaginal delivery that can be prevented by treatment with progesterone.411 Treatment with antiprogestins also increases myometrial responsiveness to contractile agents but does not necessarily result in preterm birth.413 For example, in monkeys, although antiprogestin treatment alone during late gestation does not induce labor, concomitant administration of oxytocin facilitates parturition.414,415 As discussed earlier, estrogen is also essential for maintenance of pregnancy179 and presumably, therefore, phase 0 of uterine function. In baboons, suppression of placental estradiol production by treatment with an aromatase inhibitor between early and midgestation was associated with a high rate of miscarriage, effects prevented by concomitant treatment with estradiol.
Nitric oxide (NO) is a potent endogenous muscle relaxant that elicits its actions through cyclic guanosine monophosphate and calcium channels.409 The mRNA levels of inducible nitric oxide synthase(iNOS) are highest in myometrium of preterm human patients not in labor;mRNA and protein levels decrease in term myometrium.416 Moreover, iNOS activity in myometrium of a variety of species prior to parturition appears to decrease progressively.416 Therefore, several investigators have proposed that NO acts in a paracrine manner alone and/or in conjunction with progesterone to maintain uterine quiescence during pregnancy.409
Relaxin may have a dual role in pregnancy by inhibiting myometrial contractility417 and regulating changes in cervical connective tissue.418 Relaxin can suppress spontaneous uterine contractility in the rat,417 and expression of relaxin is up-regulated in patients with premature rupture of membranes.419 Relaxin receptors are localized to decidua and chorionic trophoblast cells, and relaxin acts through these receptors to up-regulate various metalloproteinases.419 In the cervix, relaxin also enhances expression of metalloproteinses-1 and -3 and inhibits the activity of tissue inhibitor of metalloproteinases.418
As discussed by Challis,12,403 activation of the myometrium in phase 1 is associated with up-regulation of several contraction-associated proteins (CAPs) including connexin-43, oxytocin receptor, prostaglandin F receptor, and various ion channel proteins. It appears that uterine stretch as modified by the hormonal milieu is critical. Connexin-43 and oxytocin receptor mRNA expression are increased in rat myometrium after initiation of a quick stretch response.407,420 Significantly, this response to stretch is diminished in rats treated with progesterone and in unilateral pregnant rats in which serum progesterone levels are still relatively high. Although these observations implicate the increase in fetal growth to induction of CAPs and may explain the gradual increase in connexin-43 mRNA levels in the human uterus with advancing gestation,12 they further highlight the important role of progesterone in maintaining uterine quiescence. Recent observations indicate that the biochemical changes occurring in the uterus during phase 1 are associated with and are perhaps regulated by increased activity of the fetal pituitary-adrenocortical axis (i.e., increased production of cortisol and DHA/DHAS by the developing fetal adrenal gland).2,12 Increased production of estrogen421,422 and withdrawal of the production and/or action of progesterone appear to be important mediators of phase 1.12,403
In phase 2, increased production of the uterotonins oxytocin and prostaglandins stimulates the uterus that was activated in phase 1. In humans, the likely source of prostaglandins that stimulate myometrial contractility is the decidua and/or the myometrium because the level of the prostaglandin-catabolizing enzyme prostaglandin dehydrogenase (PGDH) in trophoblast chorion is sufficiently high to prevent movement of bioactive prostaglandins produced in the amnion to the myometrium.403
In animals such as the sheep and goat, parturition is initiated by activation of the fetal hypothalamic-pituitary-adrenocortical axis423 that results in increased production of cortisol. Thus, lesions in the paraventricular nucleus, hypophysectomy, or adrenalectomy of the sheep fetus prolong gestation, whereas infusion of ACTH or glucocorticoid into the fetus induces premature parturition. Fetal-derived cortisol apparently acts on the sheep placenta to induce P450c17 expression that results in decreased output of progesterone, a rise in estrogen, and subsequently, a marked increase in prostaglandin production by intrauterine tissues.424,425
The role of the human fetus in initiating parturition is not as well defined. In anencephalics and infants with other abnormalities that prevent cortisol synthesis, parturition is not significantly delayed426 ;however, in the rhesus monkey fetus, there is an increase in the distribution of both premature and postmature births after adrenalectomy427 or experimental anencephaly.233 Treatment of the monkey fetus with dexamethasone does not induce premature labor231 ; however, chronic infusion of androstenedione to pregnant rhesus monkeys increases estrogen production and results in premature birth,422 effects that are blocked by co-administration of an aromatase inhibitor.428 Interference with the conversion of androgen to estrogen locally within the placenta also blocks the patterns of myometrial contractility induced by administration of precursor androstenedione alone.428 In primates, androgens produced by the fetal adrenal as a source of substrate for placental estrogen synthesis may represent a link between the fetus and mother in the initiation of parturition.12 The role of cortisol appears more tenuous. In the human, glucocorticoids have the capacity to increase PGDH activity in the chorion429 and thereby to increase prostaglandin production. As discussed earlier, it has been suggested that placental CRH, production of which is upregulated by cortisol,430 may play a role in the onset of labor in humans.403 Indeed, maternal CRH levels are increased in women with preterm labor,431,432 and at 26 to 28 weeks of gestation, elevated maternal CRH levels may discriminate patients with apparent premature labor who go on to deliver within 24 to 48 hours from those who do not.433 CRH receptors are present in the myometrium and fetal membranes.434 CRH also stimulates the release of prostaglandins from human decidua and amnion in vitro435 and can potentiate the action of oxytocin and prostaglandins both in vitro436,437 and in vivo.438 Thus, by regulating production of placental CRH and trophoblast chorion PGDH activity, fetal-derived cortisol may play a significant role in phase 1 and/or 2 of uterine contractility integral to the onset and progression of labor.
REFERENCES
Pepe GJ, Albrecht ED: Regulation of the primate fetal adrenal cortex. Endocr Rev 11: 151, 1990 |
|
Pepe GJ, Albrecht ED: Actions of placental and fetal adrenal steroid hormones in primate pregnancy. Endocr Rev 16: 608, 1995 |
|
Albrecht ED, Pepe GJ: Central integrative role of oestrogen in modulating the communication between the placenta and fetus that results in primate fetal-placental development. Placenta 20: 129, 1999 |
|
Enders AC, Lantz KC, Peterson PE et al: From blastocyst to placenta: The morphology of implantation in the baboon. Human Reprod Update 3: 561, 1997 |
|
Petraglia F, Florio P, Nappi C et al: Peptide signaling in human placenta and membranes: Autocrine, paracrine, and endocrine mechanisms. Endocr Rev 17: 156, 1996 |
|
Mesiano S, Jaffe RB: Developmental and functional biology of the primate fetal adrenal cortex. Endocr Rev 18: 378, 1997 |
|
Hild-Petito S, Donnelly KM, Miller JB et al: A baboon ( Papio anubis) simulated-pregnant model: Cell specific expression of insulin-like growth factor binding protein-1 (IGFBP-1) type I IGF receptor (IGF-1R) and retinol binding protein (RBP) in the uterus. Endocrine J 3: 639, 1995 |
|
Aplin JD: The cell biology of human implantation. Placenta 17: 269, 1996 |
|
Aplin JD, Seif MW, Graham RA et al: The endometrial cell surface and implantation: Expression of the polymorphic mucin MUC-1 and adhesion molecules during the endometrial cycle. Ann NY Acad Sci 734: 103, 1994 |
|
George FW, Wilson JD: Sex determination and differentiation. In Knobil E, Neill JD (eds): The Physiology of Reproduction, 2nd ed, pp 3–28. New York, Raven Press, 1994 |
|
Rabinovici J, Jaffe RB: Development and regulation of growth and differentiated function in human and subhuman primate fetal gonads. Endocr Rev 11: 532, 1990 |
|
Challis JRG, Matthews SG, Gibb W et al: Endocrine and paracrine regulation of birth at term and preterm. Endocr Rev 21: 514, 2000 |
|
Nathanielsz PW: Comparative studies on the initiation of labor. Eur J Obstet Gynecol Reprod Biol 78: 127, 1998 |
|
Carr B: Fertilization, implantation, and endocrinology of pregnancy. In Griffin JE, Ojeda SR (eds): Textbook of Endocrine Physiology, p 186. Oxford, Oxford University Press, 1988 |
|
Austin CR: Capacitation and the release of hyaluronidase from spermatozoa. J Reprod Fertil 3: 31, 1960 |
|
Yanagimachi R: Mammalian fertilization. In Knobil E, Neill JD (eds): The Physiology of Reproduction, 2nd ed, p 189. New York, Raven Press, 1994 |
|
Bedford JM: Significance of the need for sperm capacitation before fertilization in eutherian mammals. Biol Reprod 28: 108, 1983 |
|
Saling PM: Fertilization: Mammalian gamete interactions. In Adashi EY, Rock JA, Rosenwaks Z (eds): Reproductive Endocrinology, Surgery and Technology, p 404. Philadelphia, Lippincott-Raven, 1996 |
|
Liang LF, Dean J: Oocyte development: Molecular biology of the zona pellucida. Vitam Horm 158: 35, 1993 |
|
Wassarman PM: Gamete interactions during mammalian fertilization. Theriogenology 41: 31, 1994 |
|
Foltz KR: Sperm-binding proteins. Int Rev Cytol 163: 249, 1995 |
|
Yanagimachi R: Sperm-egg fusion. In Duzgunes N, Bronner F (eds): Current Topics in Membranes and Transport, pp 3–43. San Diego, Academic Press, 1988 |
|
Dale B, Gualtieri R, Talevi R et al: Intercellular communication in the early human embryo. Mol Reprod Dev 29: 22, 1991 |
|
Lo CW: The role of gap junction membrane channels in development. J Bioenerg Biomembr 28: 379, 1996 |
|
Wong KHH, Adashi EY: Gametogenesis, fertilization, and implantation. In Reece EA, Hobbins JC (eds): Medicine of the Fetus and Mother, 2nd ed. Philadelphia, Lippincott-Raven, 1999 |
|
Webley GE, Hearn JP: Embryo-maternal interactions during the establishment of pregnancy in primates. In Charlton HM (eds): Oxford Reviews of Reproduction Biology. Oxford, Oxford University Press, 1994 |
|
Buster JE, Bustillo M, Rodi IA et al Biological and morphologic development of donated human ova recovered by nonsurgical uterine lavage. Am J Obstet Gynecol 153:211, 1985 |
|
Larue L, Mami O, Hirchenhain J et al: E-cadherin null mutant embryos fail to form a trophectoderm epithelium. Proc Natl Acad Sci USA 91: 188, 1994 |
|
Perona RM, Wassarman PM: Mouse blastocysts hatch in vitro by using a trypsin-like proteinase associated with cells of mural trophectoderm. Dev Biol 114: 42, 1986 |
|
Sathananthan H: Ultrastructure of preimplantation human embryos co-cultured with human ampullary cells. Hum Reprod 5: 309, 1990 |
|
Stouffer RL, Hearn JP: Endocrinology of the transition from menstrual cyclicity to establishment of pregnancy in primates. In Bazer FW (ed): The Endocrinology of Pregnancy, p 35. Totowa, NJ, Humana Press, 1998 |
|
Enders AC: Trophoblast differentiation during the transition from trophoblastic plate to lacunar stage of implantation in the rhesus monkey and human. American Journal of Anatomy 186: 85, 1989 |
|
Enders AC: Current topic: Structural responses of the primate endometrium to implantation. Placenta 23: 309, 1991 |
|
Benirschke K, Kaufmann P: Pathology of the Human Placenta. New York, Springer-Verlag, 1995 |
|
Hertig AJ, Rock J, Adams EC: A description of 34 human ova within the first 17 days of development. American Journal of Anatomy 98: 435, 1956 |
|
Hamilton WJ, Boyd JD: Development of the human placenta. In Philip EE, Barnes J, Newton M (eds): Scientific Foundations of Obstetrics and Gynecology, p 185. London, William Heinemann, 1970 |
|
Weitlauf HM: Biology of implantation In Knobil E, Neill JD (eds): The Physiology of Reproduction, p 391. New York, Raven Press, 1994 |
|
Shutt DA, Lopata A: The secretion of hormones during the culture of human preimplantation embryos with corona cells. Fertil Steril 35: 413, 1981 |
|
Laufer N, Decherney AH, Haseltine FP et al: Steroid secretion by the human egg-corona cumulus complex in culture. J Clin Endocrinol Metab 58: 1153, 1984 |
|
Giudice LC, Saleh W: Growth factors in reproduction. Trends Endocrinol Metab 6: 60, 1995 |
|
Finn CA: The implantation reaction. In Wynn RM (ed): Biology of the Uterus, p 245. New York, Plenum, 1977 |
|
Psychoyos A: Uterine receptivity for nidation. Ann N Y Acad Sci 476: 36, 1986 |
|
Rogers PAW, Murphy CR: Uterine receptivity for implantation: Human studies in blastocysts implantation. In Yoshinaga K (ed): Blastocyst Implantation, p 231. Boston, Adams Publishing, 1989 |
|
Formigli L, Formigli G, Roccio C: Donation of fertilized uterine ova to infertile women. Fertil Steril 47: 162, 1988 |
|
Bergh PA, Navot D: The impact of embryonic development and endometrial maturity on the timing of implantation. Fertil Steril 58: 537, 1992 |
|
Martel D, Frydman R, Glissant M et al: Scanning electron microscopy of postovulatory human endometrium in spontaneous cycles and cycles stimulated by hormone treatment. J Endocrinol 114: 319, 1987 |
|
Aoki D, Kawakami H, Nozawa S et al: Differences in lectin binding patterns of normal human endometrium between proliferative and secretory phases. Histochemistry 92: 177, 1989 |
|
Hey NA, Graham RA, Seif MW et al: The polymorphic epithelial mucin MUC1 in human endometrium is regulated with maximal expression in the implantation phase. J Clin Endocrinol Metab 78: 337, 1994 |
|
Lessey BA, Damjanovich L, Coutifaris C et al: Integrin adhesion molecules in the human endometrium: Correlation with the normal and abnormal menstrual cycle. J Clin Invest 90: 188, 1992 |
|
Lessey BA, Castelbaum AJ, Buck CA et al: Further characterization of endometrial integrins during the menstrual cycle and in pregnancy. Fertil Steril 62: 497, 1994 |
|
Sueoka K, Shiokawa S, Miyazaki T et al: Integrins and reproductive physiology: Expression and modulation in fertilization, embryogenesis, and implantation. Fertil Steril 67: 799, 1997 |
|
Coutifaris C, Dardik R, Omigbodun A: Cell adhesion molecules and embryo implantation. In Rock JA, Faro S, Gant NF et al (eds): Advances in Obstetrics and Gynecology, Vol 4, p 163. St. Louis, CV Mosby, 1997 |
|
Strauss J, Coutifaris C: The endometrium and myometrium: Regulation and dysfunction. In Yen SSC, Jaffe RB, Barbieri RL (eds): Reproduction Endocrinology, Physiology, Pathophysiology, and Clinical Management, 4th ed, p 218. Philadelphia, WB Saunders, 1999 |
|
Campbell S, Swann HR, Seif MW et al: Cell adhesion molecules on the oocyte and preimplantation human embryo. Hum Reprod 10: 1571, 1995 |
|
Fazleabas AT, Bell SC, Fleming S et al: Distribution of integrins and the extracellular matrix proteins in the baboon endometrium during the menstrual cycle and early pregnancy. Biol Reprod 56: 348, 1997 |
|
Charnock-Jones D, Sharkey A, Fenwick P et al: Leukaemia inhibitory factor mRNA concentration peaks in human endometrium at the time of implantation and the blastocyst contains mRNA for the receptors at this time. J Reprod Fertil 101: 421, 1994 |
|
Cullinan EB, Abbondanzo SJ, Anderson PS et al: Leukemia inhibitory factors (LIF) and LIF receptor expression in human endometrium suggests a potential autocrine/paracrine function in regulating embryo implantation. Proc Natl Acad Sci USA 93: 3115, 1996 |
|
Rathjen PD, Nichols J, Toth S et al: Developmentally programmed induction of differentiation inhibiting activity and the control of stem cell populations. Genes Dev 4: 2308, 1990 |
|
Stewart CL, Kaspar P, Brunet LJ et al: Blastocyst implantation depends on maternal expression of leukemia inhibitory factor. Nature 359: 76, 1992 |
|
Spinks NR, O'Neill C: Embryo-derived platelet-activating factor is essential for establishment of pregnancy in the mouse. Lancet 1: 106, 1987 |
|
Prahalada S, Venkatramaiah M, Rao AJ et al: Termination of pregnancy in macaques ( Macaca radiata) using monkey antiserum to ovine LH. Contraception 12: 137, 1975 |
|
Stevens VC: Immunization of female baboons with hapten-coupled gonadotropins. J Obstet Gynecol 42: 496, 1973 |
|
Talwar GP, Sharma NC, Dubey SK: Isoimmunization against hCG with conjugates of processed β -subunit of the hormone and tetanus toxoid. Proc Natl Acad Sci USA 73: 218, 1976 |
|
Thau RB, Sundaram K: The mechanisms of action of an anti-fertility vaccine in the rhesus monkey: Reversal of the effects of antisera to β -oLH by medroxyprogesterone acetate. Fertil Steril 33: 317, 1980 |
|
Ryan RJ, Keutmann HT, Charlesworth MC et al: Structure-function relationships of gonadotropins. Recent Prog Horm Res 43: 383, 1987 |
|
Parsons TF, Pierce JG: Free α-like material from bovine pituitaries: Removal of its 0-linked oligosaccharide permits combination with lutropin- β. J Biol Chem 259: 2662, 1984 |
|
Jameson JL, Hollenberg AN: Regulation of chorionic gonadotropin gene expression. Endocr Rev 14: 203, 1993 |
|
Lapthorn AJ, Harris DC, Littlejohn A et al: Crystal structure of human chorionic gonadotropin. Nature 369: 455, 1994 |
|
Ren S-G, Braunstein GD: Human chorionic gonadotropin. Semin Reprod Endocrinol 10: 95, 1992 |
|
Halvorson LM, Chin WW: Gonadotropic hormones: Biosynthesis, secretion, receptors and action. In Yen SSC, Jaffe RB, Barbieri RL (eds): Reproduction Endocrinology, Physiology, Pathophysiology, and Clinical Management, 4th ed, p 81. Philadelphia, WB Saunders, 1999 |
|
Lenton EA: Gonadotropins of the menstrual cycle and implantation. Establishing a Successful Human Pregnancy 66: 33, 1990 |
|
Braunstein GD, Rasor J, Hanser H et al: Serum human chorionic gonadotropin levels throughout normal pregnancy. Am J Obstet Gynecol 126: 678, 1976 |
|
Seshagiri PB, Terasawa EI, Hearn JP: Secretion of chorionic gonadotropin by the peri-implantation embryo of rhesus and marmoset monkeys, with its possible relationship to embryo-derived GnRH. In Puri CP, Van Look PFA(eds): Current Concepts in Fertility Regulation and Reproduction, p 395. New Delhi, Wiley Eastern, 1996 |
|
Albrecht ED, Pepe GJ: Placental steroidogenesis in primate pregnancy. In Knobil E, Neill J (eds): Encyclopedia of Reproduction, Vol 3, pp 889–898, Boston, Academic Press, 1998 |
|
Glinoer D: The regulation of thyroid function in pregnancy: Pathways of endocrine adaptation from physiology to pathology. Endocr Rev 18: 404, 1997 |
|
Challis JRG: CRH, a placental clock and preterm labour. Nat Med 1: 416, 1995 |
|
Reis FM, Fadalti M, Florio P et al: Putative role of placental corticotropin-releasing factor in the mechanisms of human parturition. J Soc Gynecol Investig 6: 109, 1999 |
|
Gu W, Jones CT, Parer JT: Production of CRF-like immunoreactivity by placenta in response to alteration of uterine blood flow in pregnant sheep. Int Symposium on Fetal and Neonatal Development. Oxford UK, 6 (abstr), 1987 |
|
Riley SC, Walton JC, Herlick JM et al: The localization and distribution of corticotropin-releasing hormone in the human placenta and fetal membranes throughout gestation. J Clin Endocrinol Metab 72: 1001, 1991 |
|
Robinson BG, Emanuel RL, Frim DM et al: Glucocorticoid stimulates expression of corticotropin-releasing hormone in human placenta. Proc Natl Acad Sci USA 85: 5244, 1988 |
|
Han VKM: Growth factors in placental growth and development. In Rice GE, Brennecke SP (eds): Molecular Aspects of Placental and Fetal Membrane Autocoids, p 395. Boca Raton, FL, CRC Press, 1993 |
|
Fazleabas AT, Kim JJ, Srinivasan S et al: Implantation in the baboon:Endometrial responses. Semin Reprod Endocrinol 17: 257, 1999 |
|
Waites GT, James RFL, Bell SC: Immunohistological localization of the human endometrial secretory protein pregnancy-associated endometrial 1-globulin (1-PEG) an insulin-like growth factor binding protein, during the menstrual cycle. J Clin Endocrinol Metab 67: 1100, 1988 |
|
Fazleabas AT, Verhage HG, Waites G, et al: Characterization of an insulin-like growth factor binding protein, analogous to human pregnancy-associated secreted endometrial a1 globulin, in decidua of the baboon ( Papio anubis) placenta. Biol Reprod 40: 873, 1989 |
|
Ritvos O, Ranta T, Jalkanen J et al: Insulin-like growth factor (IGF) binding protein from human decidua inhibits binding and biological action of IGF-I in cultured choriocarcinoma cells. Endocrinology 122: 2150, 1988 |
|
Bell SC: Secretory endometrial and decidual proteins: Studies and clinical significance of a maternally derived group of pregnancy-associated serum proteins. Hum Reprod 1: 29, 1986 |
|
Giudice LC, Farrell EM, Pham H et al: Insulin-like growth factor binding proteins in maternal serum throughout gestation and in the puerperium: Effects of a pregnancy associated serum protease activity. J Clin Endocrinol Metab 71: 806, 1990 |
|
Kim JJ, Jaffe RC, Fazleabas AT: Insulin-like growth factor binding protein-1 expression in baboon endometrial stromal cells: Regulation by filamentous actin and requirement for de novo protein synthesis. Endocrinology 140: 997, 1999 |
|
Garde J, Bell SC, Eperon IC: Multiple forms of mRNA encoding human pregnancy-associated endometrial α-globulin, a β -lactoglobulin homologue. Proc Natl Acad Sci USA 88: 2456, 1991 |
|
Seppala M, Angervo M, Riittinen L et al: Peptides and proteins in the human endometrium. Reprod Med Rev 1: 37, 1992 |
|
Seppala M, Julkunen M, Riittinen L et al: Endometrial proteins: A reappraisal. Hum Reprod 7: 31, 1992 |
|
Joshi SG: A progestagen-associated protein of the human endometrium: Basic studies and potential clinical applications. J Steroid Biochem Mol Biol 19: 751, 1983 |
|
Seppala M, Koistinen H, Koistinen R et al: Glycodelins as regulators of early events of reproduction. Clin Endocrinol (Oxf) 46: 381, 1997 |
|
Stewart DR, Erickson MS, Erickson ME et al: The role of relaxin in glycodelin secretion. J Clin Endocrinol Metab 82: 839, 1997 |
|
Hausermann HM, Donnelly KM, Bell SC et al: Regulation of the glycosylated β -lactoglobulin homolog, glycodelin (placental protein 14[PP14 ]) in the baboon ( papio anubis) uterus. J Clin Endocrinol Metab 83: 1226, 1998 |
|
Ben-Jonathan N, Mershon JL, Allen DL et al: Extrapituitary prolactin: Distribution, regulation, functions, and clinical aspects. Endocr Rev 17: 639, 1996 |
|
Maurer RA, Kim KE, Day RN et al: Regulation of prolactin gene expression by estradiol. In Sato G, Stevens JL (eds): Molecular Endocrinology and Steroid Hormone Action, p 159. New York, Liss, 1990 |
|
Frasor J, Gaspar CA, Donnelly KM et al: Expression of prolactin and its receptor in the baboon uterus during the menstrual cycle and pregnancy. J Clin Endocrinol Metab 84: 3344, 1999 |
|
Huang JF, Tseng L, Bischof P et al: Regulation of prolactin production by progestin, estrogen, and relaxin in human endometrial stromal cells. Endocrinology 121: 2011, 1987 |
|
Riddick DH, Kusnik WF: Decidua: A possible source of amniotic fluid PRL. Am J Obstet Gynecol 127: 187, 1977 |
|
McCoshen JA, Tomita K, Fernandez C et al: Specific cells of human amnion selectively localize prolactin. J Clin Endocrinol Metab 55: 166, 1982 |
|
Nguyen T, Diveky L, Fedirko B et al: Daily changes in plasma and amniotic fluid prolactin during the last third of pregnancy in the baboon. Biol Reprod 56: 597, 1997 |
|
Bryant-Greenwood GD: Relaxin as a new hormone. Endocr Rev 3: 62, 1982 |
|
Bigazzi M, Nardi E: Prolactin and relaxin: Antagonist on the spontaneous motility of the uterus. J Clin Endocrinol Metab 53: 665, 1981 |
|
Zhou Y, Fisher SJ, Janatpour M et al: Human cytotrophoblasts adopt a vascular phenotype as they differentiate: A strategy for successful endovascular invasion? J Clin Invest 99: 2139, 1997 |
|
Assali NS, Clark KE, Zugaib M et al: Effects of estrogenic hormones on uteroplacental hemodynamics and progesterone production in the sheep. Int J Fertil 23: 219, 1978 |
|
Kaufmann P, Luckhardt M, Leiser R: Three dimensional representation of the fetal vessel system in the human placenta. Trophoblast Research, Vol 3, p 113. New York, Plenum, 1988 |
|
Rosenfeld CR, Morriss FH, Makowski EL et al: Circulatory changes in the reproductive tissues of ewes during pregnancy. Gynecol Invest 5: 252, 1974 |
|
Demir R, Kaufmann P, Castellucci M et al: Fetal vasculogenesis and angiogenesis in human placental villi. Acta Anat 136: 190, 1989 |
|
Stoz F, Schuhman RA, Schebesta B: The development of the placental villus during normal pregnancy: Morphometric database. Acta Gynecol Obstet 244: 23, 1988 |
|
Arany E, Hill DJ: FGF-2 and FGF receptor-1 mRNA expression and peptide localization in placentae from normal and diabetic pregnancies. Placenta 19: 133, 1998 |
|
Ferriani RA, Ahmed A, Sharkey A et al: Colocalization of acidic and basic FGF in human placenta and the cellular effects of bFGF in trophoblast cell line JEG-3. Growth Factors 10: 259, 1994 |
|
Hamai Y, Fujii T, Yamashita T et al: Evidence for bFGF as a crucial angiogenic growth factor, released from human trophoblasts during early gestation. Placenta 19: 149, 1998 |
|
Jackson MR, Carney EW, Lye SJ et al: Localization of two angiogenic growth factors (PDECGF and VEGF) in human placentae throughout gestation. Placenta 15: 341, 1994 |
|
Shore VH, Wang TH, Wang CL et al: Vascular endothelial growth factor, placental growth factor and their receptors in isolated human trophoblasts. Placenta 18: 657, 1997 |
|
Vuorela P, Hatva E, Lymboussaki A et al: Expression of vascular endothelial growth factor and placenta growth factor in human placenta. Biol Reprod 56: 489, 1997 |
|
Cooper JC, Sharkey AM, McLaren J et al: Localization of VEG/PF and its receptor, flt-1, in human placenta and decidua by immunohistochemistry. J Reprod Fertil 105: 205, 1995 |
|
Leung DW, Cachianes G, Kuang W-J et al: Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 246: 1306, 1989 |
|
Ferrara N, Davis-Smyth T: The biology of vascular endothelial growth factor. Endocr Rev 18: 4, 1997 |
|
Brown LF, Detmar M, Claffey K et al: Vascular permeability factor/vascular endothelial growth factor: A multifunctional angiogenic cytokine. EXS 79: 233, 1997 |
|
Klagsbrun M, D'Amore PA: Regulators of angiogenesis. Annu Rev Physiol 53: 217, 1991 |
|
Ferrara N, Houck K, Jakeman L et al: Molecular and biological properties of vascular endothelial growth factor family of proteins. Endocr Rev 13: 18, 1992 |
|
Sharkey DS, Sharkey AM, Rajput-Williams J et al: Identification and localization of alternative spliced mRNAs for VEG/PF in human uterus and estrogen regulation in endometrial carcinoma cell lines. Biol Reprod 48: 1120, 1993 |
|
Sharkey AM, Charnock-Jones DS, Boocock CA et al: Expression of mRNA for vascular endothelial growth factor in human placenta. J Reprod Fertil 99: 609, 1993 |
|
Cheung CY: VEGF: Possible role in fetal development and placental function. J Soc Gynecol Investig 4: 169, 1997 |
|
Clark DE, Smith SK, Sharkey AM et al: Localization of VEGF and expression of its receptors flt and KDR in human placenta throughout pregnancy. Hum Reprod 11: 1090, 1996 |
|
Carmellet P, Ferreira V, Breler G et al: Abnormal blood vessel development and lethality in embryos lacking a single VEGF gene. Nature 380: 435, 1996 |
|
Kaipainen A, Korhonen J, Pajusola K et al: The related FLT4, FLT1, and KDR/flk-1 receptor tyrosine kinases show distinct expression patterns in human fetal endothelial cells. J Exp Med 178: 2077, 1993 |
|
Barleon B, Hauser S, Schollmann C et al: Differential expression of the two VEGF receptors flt-1 and KDR/flk-1 in placenta and vascular endothelial cells. J Cell Biochem 54: 56, 1994 |
|
Sunderkotter C, Steinbrink K, Goebeler M et al: Macrophages and angiogenesis. J Leukoc Biol 55: 410, 1994 |
|
Clark DE, Smith SK, He Y et al: A vascular endothelial growth factor inhibitor is produced by the human placenta and released into the maternal circulation. Biol Reprod 59: 1540, 1998 |
|
Shalaby F, Rossant J, Yamaguchi T et al: Failure of blood-island formation and vasculogenesis in flt-1-deficient mice. Nature 376: 62, 1995 |
|
Fong GH, Rassant J, Gertenstein M et al: Role of flt-1 receptor tyrosine kinase in regulating the assembly of vascular endothelium. Nature 376: 66, 1995 |
|
Dumont DJ, Gradwohl GJ, Fong GH et al: Dominant-negative and targeted null mutations in the endothelial receptor tyrosine kinase, tek, reveal a critical role in vasculogenesis of the embryo. Genes Dev 8: 1897, 1994 |
|
Sato TN, Tozawa Y, Deutsch U et al: Distinct roles of the receptor tyrosine kinases Tie-1 and Tie-2 in blood vessel formation. Nature 376: 70, 1995 |
|
Hanahan D: Signaling vascular morphogenesis and maintenance. Science 277: 48, 1997 |
|
Suri C, Jones PF, Patan S et al: Requisite role of angiopoietin-1, a ligand for the TIE-2 receptor, during embryonic angiogenesis. Cell 87: 1171, 1996 |
|
Maisonpierre PC, Suir C, Jones PF et al: Angiopoietin-2 a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science 277: 55, 1997 |
|
Zhang EG, Smith SK, Charnock-Jones DS: Placental expression of human angiopoietin-2 and tie-2 but not angiopoietin-1 or tie-1, are regulated during gestation. J Soc Gynecol Investig 6: 158, 1999 |
|
Goldman-Wohl DS, Ariel I, Greenfield C et al: Tie-2 and angiopoietin-2 expression at the fetal-maternal interface: A receptor ligand model for vascular remodeling. Mol Hum Reprod 6: 81, 2000 |
|
Shweiki D, Itin A, Sofer D et al: Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature 359: 843, 1992 |
|
Friedrici HH: The early response of uterine capillaries to estrogen stimulation. Lab Invest 17: 322, 1967 |
|
Cullinan-Bove K, Koos RD: Vascular endothelial growth factor/vascular permeability factor expression in the rat uterus: Rapid stimulation by estrogen correlates with estrogen-induced increases in uterine capillary permeability and growth. Endocrinology 133: 829, 1993 |
|
Hyder SM, Stancel GM, Ciappetta C et al: Uterine expression of VEGF is increased by estradiol and tamoxifen. Cancer Res 56: 3954, 1996 |
|
Charnock-Jones DS, Sharkey AM, Rajput-Williams J et al: Identification and localization of alternatively spliced mRNAs for vascular endothelial growth factor in human uterus and estrogen regulation in endometrial carcinoma cell lines. Biol Reprod 48: 1120, 1993 |
|
Johns A, Freay A, Fraser W et al: Disruption of estrogen receptor gene prevents 17 β estradiol-induced angiogenesis in transgenic mice. Endocrinology 137: 4511, 1996 |
|
Hildebrandt VA, Babischkin JS, Koos RD et al: Developmental regulation of vascular endothelial growth/permeability factor messenger ribonucleic acid levels in and vascularization of the villous placenta during baboon pregnancy. Endocrinology 142: 2050, 2001 |
|
Magness RR, Rosenfeld CR: Local and systemic estradiol-17α effects on uterine and systemic vasodilation. Am J Physiol 256: E536, 1989 |
|
Magness RR: Maternal cardiovascular and other physiologic responses to the endocrinology of pregnancy. In Bazer FW (ed): The Endocrinology of Pregnancy, p 507. Totowa, NJ, Humana Press, 1999 |
|
Ramsey EM, Houston ML, Harris JWS: Interactions of the trophoblast and maternal tissues in three closely related primate species. Am J Obstet Gynecol 124: 647, 1976 |
|
Boyd JD, Hamilton WJ: The Human Placenta. Cambridge, Heffer & Sons, 1970 |
|
Pijnenborg R: Trophoblast invasion. Reprod Med Rev 3: 53, 1994 |
|
Enders AC: Cytodifferentiation of trophoblast in the anchoring villi and trophoblastic shell in the first half of gestation in the macaque. Microsc Res Tech 38: 3, 1997 |
|
Enders AC, Blankenship TN: Modification of endometrial arteries during invasion by cytotrophoblast cells in the pregnant macaque. Acta Anat 159: 169, 1997 |
|
Enders AC, King BF: Early stages of trophoblastic invasion of the maternal vascular system during implantation in the macaque and baboon. Am J Anat 192: 329, 1991 |
|
Huppertz B, Kertschanska S, Frank HG et al: Extracellular matrix components of the placental extravillous trophoblast: Immunocytochemistry and ultrastructural distribution. Histochem Cell Biol 106: 291, 1996 |
|
Aplin JD, Charlton AK: The role of matrix macromolecules in the invasion of decidua by trophoblast. Trophoblast Res 4: 139, 1990 |
|
Aplin JD: Expression of integrin α6 β 4 in human trophoblast and its loss from extravillous cells. Placenta 14: 203, 1993 |
|
Burrows TD, King A, Loke YW: Expression of adhesion molecules by endovascular trophoblast and decidual endothelial cells: Implications for vascular invasion during implantation. Placenta 15: 21, 1994 |
|
Pijnenborg R, D'Hooghe T, Vercruysse L et al: Evaluation of trophoblast invasion in placental bed biopsies of the baboon with immunohistochemical localization of cytokeratin, fibronectin, and laminin. J Med Primatol 25: 272, 1996 |
|
Blankenship TN, Enders AC, King BF: Trophoblastic invasion and the development of uteroplacental arteries in the macaque: Immunohistochemical localization of cytokeratins, desmin, type IV collagen, laminin, and fibronectin. Cell Tissue Res 272: 227, 1993 |
|
Damsky CH, Fitzgerald ML, Fisher SJ: Distribution pattern of extracellular matrix components and adhesion receptors are intricately modulated during first trimester cytotrophoblast differentiation along the invasive pathway, in vivo. J Clin Invest 89: 210, 1992 |
|
Vicovac L, Jones CJ, Aplin JD: Trophoblast differentiation during formation of anchoring villi in a model of the early human placenta in vitro. Placenta 16: 41, 1995 |
|
Damsky CH, Librach C, Lim KH et al: Integrin switching regulates normal trophoblast invasion. Development 120: 3657, 1994 |
|
Aplin JD, Haigh T, Jones CJP et al: Development of cytotrophoblast columns from explanted first-trimester human placental villi: Role of fibronectin and integrin α5 β 1. Biol Reprod 60: 828, 1999 |
|
Blankenship TN, King BF: Macaque intra-arterial trophoblast and extravillous trophoblast of the cell columns and cytotrophoblastic shell express neural cell adhesion molecule (NCAM). Anat Rec 245: 525, 1996 |
|
Blankenship T, Enders AC: Expression of platelet-endothelial cell adhesion molecule-1 (PECAM) by macaque trophoblast cells during invasion of the spiral arteries. Anat Rec 247: 413, 1997 |
|
Blankenship TN, King BF: Identification of 72-kilodalton type IV collagenase at sites of trophoblastic invasion of macaque spiral arteries. Placenta 15: 177, 1994 |
|
Blankenship T, Enders AC: Trophoblast cell-mediated modifications to uterine spiral arteries during early gestation in the macaque. Acta Anat 158: 227, 1997 |
|
Librach CL, Werb Z, Fitzgerald ML et al: 92-KD type IV collagenase mediates invasion of human cytotrophoblasts. J Cell Biol 113: 437, 1991 |
|
Zhou Y, Damsky CH, Fisher SJ: Preeclampsia is associated with failure of cytotrophoblasts to mimic a vascular adhesion phenotype: One cause of defective endovascular invasion in this syndrome? J Clin Invest 99: 2152, 1997 |
|
Oakey RE: The progressive increase in oestrogen production in human pregnancy: An appraisal of the factors responsible. Vitam Horm 28: 1, 1970 |
|
Milewich L, MacDonald PC, Carr BR: Estrogen 16α-hydroxylase activity in human fetal tissue. J Clin Endocrinol Metab 63: 404, 1986 |
|
Levitz M, Young BK: Estrogens in pregnancy. Vitam Horm 35: 109, 1977 |
|
Lindberg BS, Johansson EDB, Nilsson BA: Plasma levels of nonconjugated oestrone, oestradiol-17 β and oestriol during uncomplicated pregnancy. Acta Obstet Gynecol Scand 32: 21, 1974 |
|
Tulchinsky D, Hobel CJ, Yeager E et al: Plasma estrone, estradiol, estriol, progesterone and 17-hydroxyprogesterone in human pregnancy. Am J Obstet Gynecol 112: 1095, 1972 |
|
Nygren KG, Johansson EDB, Wide L: Evaluation of the prognosis of threatened abortion from the peripheral plasma levels of progesterone, estradiol and human chorionic gonadotropin. Am J Obstet Gynecol 116: 916, 1973 |
|
Lehrer S, Sanchez M, Song HK et al: Oestrogen receptor B-region polymorphism and spontaneous abortion in women with breast cancer. Lancet 335: 622, 1990 |
|
Albrecht ED, Aberdeen GW, Pepe GJ: The role of estrogen in the maintenance of primate pregnancy. Am J Obstet Gynecol 182: 432, 2000 |
|
Tulchinsky D, Korenman SG: The plasma estradiol as an index of fetoplacental function. J Clin Invest 50: 1490, 1971 |
|
Lindberg BS, Johansson EDB, Nilsson BA: Plasma levels of nonconjugated oestradiol-17 β and oestriol in high-risk pregnancies. Acta Obstet Gynecol Scand 32: 37, 1974 |
|
Morishima A, Grumbach MM, Simpson ER et al: Aromatase deficiency in male and female siblings caused by a novel mutation and the physiological role of estrogens. J Clin Endocrinol Metab 80: 3689, 1995 |
|
France JT: Steroid sulfatase deficiency. J Steroid Biochem 11: 647, 1979 |
|
Shozu M, Akasofu K, Harada T et al: A new cause of female pseudohermaphroditism: Placental aromatase deficiency. J Clin Endocrinol Metab 72: 560, 1991 |
|
Younes MA, Besch NF, Besch PK: Estradiol and progesterone binding in human term placental cytosol. Am J Obstet Gynecol 141: 170, 1981 |
|
Levitz M, Emerman S, Dancis J: Sterol synthesis in perfused human placenta. Excerpta Med Internat Congr Series (AMST) 51: 266, 1962 |
|
Ryan KJ: Biological aromatization of steroids. J Biol Chem 234: 268, 1959 |
|
Siiteri PK, MacDonald PC: The utilization of circulating dehydroisoandrosterone sulfate for estrogen synthesis during human pregnancy. Steroids 2: 713, 1963 |
|
Frandsen VA, Stakeman G: The site of production of oestrogenic hormones in human pregnancy. Acta Endocrinol 38: 383, 1961 |
|
Siiteri PK, MacDonald PC: Placental estrogen biosynthesis during human pregnancy. J Clin Endocrinol Metab 26: 751, 1966 |
|
Albrecht ED, Pepe GJ: Placental steroid hormone biosynthesis in primate pregnancy. Endocr Rev 11: 124, 1990 |
|
Diczfalusy E: Endocrine function of the human fetal placental unit. Fed Proc 23: 791, 1964 |
|
Bolte E, Mancuso S, Eriksson G et al: Studies on the aromatisation of neutral steroids in pregnant women: III. Overall aromatisation of dehydroepiandrosterone sulphate circulating in the foetal and maternal compartments. Acta Endocrinol (Copenh) 45: 576, 1964 |
|
Braverman MB, Gurpide E: In vitro effects of human prolactin and oxytocin on sulfatase activity in isolated human decidual cells. J Clin Endocrinol Metab 63: 725, 1986 |
|
Mitchell BF, Cross J, Hobkirk R et al: Formation of unconjugated estrogens from estrone sulfate by dispersed cells from human fetal membranes and decidua. J Clin Endocrinol Metab 58: 845, 1984 |
|
Mason JI, Keeney DS, Bird IM et al: The regulation of 3 β -hydroxysteroid dehydrogenase expression. Steroids 62: 164, 1997 |
|
Thompson EA Jr, Siiteri PK: Utilization of oxygen and reduced nicotinamide adenine dinucleotide phosphate by human placental microsomes during aromatization of androstenedione. J Biol Chem 249: 5364, 1974 |
|
Thompson EA Jr, Siiteri PK: Partial resolution of the placental microsomal aromatase complex. J Steroid Biochem 7: 635, 1976 |
|
Simpson ER, Zhao Y, Agarwal VR et al: Aromatase expression in health and disease: Recent Prog Horm Res 52:185, 1997 |
|
Bellino FL, Osawa Y: Evidence of the direct aromatization of testosterone and different aromatization sites for testosterone and androstenedione in human placental microsomes. Biochemistry 13: 1925, 1974 |
|
Blomquist CH: Kinetic analysis of enzymic activities: Prediction of multiple forms of 17 β -hydroxysteroid dehydrogenase. J Steroid Biochem Mol Biol 55: 515, 1995 |
|
Poutanen M, Isomaa V, Peltoketo H et al: Role of 17 β -hydroxysteroid dehydrogenase type 1 in endocrine and intracrine estradiol biosynthesis. J Steroid Biochem Mol Biol 55: 525, 1995 |
|
Andersson S, Moghrabi N: Physiology and molecular genetics of 17 β -hydroxysteroid dehydrogenases. Steroids 62: 143, 1997 |
|
Wu L, Einstein M, Geissler WM et al: Expression cloning and characterization of human 17β-hydroxysteroid dehydrogenase type 2, a microsomal enzyme processing 20α-hydroxysteroid dehydrogenase activity. J Biol Chem 12:12,964, 1993 |
|
Andersson S: Molecular genetics of androgenic 17 β -hydroxysteroid dehydrogenases. J Steroid Biochem Mol Biol 55: 533, 1995 |
|
Strickler RC, Tobias B: Estradiol 17 β -dehydrogenase and 20α-hydroxysteroid dehydrogenase from human placental cytosol: One enzyme with two activities? Steroids 36: 243, 1980 |
|
Blomquist CH, Lindemann NJ, Hakanson EE: 17 β -hydroxysteroid and 20α-hydroxysteroid dehydrogenase activities of human placental microsomes: Kinetic evidence for two enzymes differing in substrate specificity. Arch Biochem Biophys 239: 375, 1985 |
|
Gurpide E, Schwers J, Welch MT et al: Fetal and maternal metabolism of estradiol during pregnancy. J Clin Endocrinol Metab 26: 1355, 1966 |
|
Gurpide E, Marks C, DeZiegler D et al: Asymmetric release of estrone and estradiol derived from labeled precursors in perfused human placentas. Am J Obstet Gynecol 144: 551, 1982 |
|
Walsh SW, McCarthy MS: Selective placental secretion of estrogens into fetal and maternal circulations. Endocrinology 109: 2152, 1981 |
|
Waddell BJ, Albrecht ED, Pepe GJ: Utilization of maternal and fetal androstenedione for placental estrogen production at mid and late baboon pregnancy. J Steroid Biochem Mol Biol 41: 171, 1992 |
|
Ogino M: Productivity of estrogen by human placental organ culture at different stages of gestation. Endocrinol Jpn 32: 607, 1985 |
|
Mendelson CR, Cleland WH, Smith ME et al: Regulation of aromatase activity of stromal cells derived from human adipose tissue. Endocrinology 111: 1077, 1982 |
|
Mendelson CR, Corbin CJ, Smith ME et al: Growth factors suppress and phorbol esters potentiate the action of dibutyryl adenosine 3',5'-monophosphate to stimulate aromatase activity of human adipose stromal cells. Endocrinology 118: 968, 1986 |
|
Gant NF, Hutchinson HT, Siiteri PK et al: Study of the metabolic clearance rate of dehydroisoandrosterone sulfate in pregnancy. Am J Obstet Gynecol 111: 555, 1971 |
|
Stock MK, Metcalfe JM: Maternal physiology during gestation. In Knobil E, Neill JD (eds): The Physiology of Reproduction, p 947. New York, Raven Press, 1994 |
|
Fritz MA, Stanczyk FZ, Novy MJ: Relationship of uteroplacental blood flow to the placental clearance of maternal dehydroepiandrosterone through estradiol formation in the pregnant baboon. J Clin Endocrinol Metab 61: 1023, 1985 |
|
Fritz MA, Stanczyk FZ, Novy MJ: Maternal estradiol response to alterations in uteroplacental blood flow. Am J Obstet Gynecol 155: 1317, 1986 |
|
Worley RJ, Everett RB, Madden JD et al: Fetal considerations: metabolic clearance rate of maternal plasma dehydroisoandrosterone sulfate. Semin Perinatol 2: 15, 1978 |
|
Worley RJ: Steroid endocrinology of pregnancy. In Sciarra JJ, Speroff L(eds) Gynecology and Obstetrics, Vol 5. New York, Harper and Row, 1980 |
|
Everett RB, Porter JC, MacDonald PC et al: Relationship of maternal placental blood flow to the placental clearance of maternal plasma dehydroisoandrosterone sulfate through placental estradiol formation. Am J Obstet Gynecol 136: 435, 1980 |
|
Shepherd RW, Stanczyk FZ, Bethea CL et al: Fetal and maternal endocrine responses to reduced uteroplacental blood flow. J Clin Endocrinol Metab 75: 301, 1992 |
|
Seron-Ferre M, Taylor NF, Rotten D et al: Changes in fetal monkey plasma dehydroepiandrosterone sulfate: Relationship to gestational age, adrenal weight and preterm delivery. J Clin Endocrinol Metab 57: 1173, 1983 |
|
Jaffe RB, Seron-Ferre M, Parer JT et al: The primate fetal pituitary-adrenal axis in the perinatal period. Am J Obstet Gynecol 131: 164, 1978 |
|
Townsley JD, Pepe GJ: Serum dehydroepiandrosterone and dehydroepiandrosterone sulfate in baboon ( Papio papio) pregnancy. Acta Endocrinologica 85: 415, 1977 |
|
Walsh SW, Norman RL, Novy MJ: In utero regulation of rhesus monkey fetal adrenals: Effects of dexamethasone, adrenocorticotropin, thyrotropin-releasing hormone, prolactin, human chorionic gonadotropin and α-melanocyte stimulating hormone on fetal and maternal plasma steroids. Endocrinology 104: 1805, 1979 |
|
Mesiano S, Coulter CL, Jaffe RB: Localization of cytochrome P450 cholesterol side-chain cleavage, cytochrome P450 17α-hydroxylase/17,20-lyase, and 3 β -hydroxysteroid dehydrogenase isomerase steroidogenic enzymes in human and rhesus monkey fetal adrenal glands: Reappraisal of functional zonation. J Clin Endocrinol Metab 77: 1184, 1993 |
|
Easterling WE, Simmer HH, Dignam WJ et al: Neutral c19-steroids and steroid sulphates in human pregnancy. II. Dehydroepiandrosterone sulfate, 16α-hydroxydehydroepiandrosterone sulfate in maternal and fetal blood of pregnancies with anencephalic and normal fetuses. Steroids 8: 157, 1966 |
|
Aberdeen GW, Leavitt MG, Pepe GJ et al: Effect of maternal betamethasone administration at moderation on baboon fetal adrenal gland development and adrenocortico-tropin receptor messenger ribonucleic acid expression. J Clin Endocrinol Metab 83: 976, 1998 |
|
Townsley JD: Utilization of dehydroepiandrosterone and its sulphate for oestrogen production by pregnant baboons ( Papio papio). Acta Endocrinol (Copenh) 81:830, 197 |
|
Novy MJ, Walsh SW: Dexamethasone and estradiol treatment in pregnant rhesus macaques: Effects on gestational length, maternal plasma hormones, and fetal growth. Am J Obstet Gynecol 145: 920, 1983 |
|
Johannisson E: Aspects of the ultrastructure and function of the human fetal adrenal cortex. Contrib Gynecol Obstet 4: 109, 1979 |
|
Novy MJ, Walsh SW, Kittinger GW: Experimental fetal anencephaly in the rhesus monkey: Effect on gestational length and fetal and maternal plasma steroids. J Clin Endocrinol Metab 45: 1031, 1977 |
|
Voutilainen R, Miller WL: Coordinate tropic hormone regulation of mRNAs for insulin-like growth factor II and the cholesterol side-chain-cleavage enzyme, P450scc, in human steroidogenic tissues. Proc Natl Acad Sci USA 84: 1590, 1987 |
|
Voutilainen R, Picardo-Leonard J, Di Blasio AM et al: Hormonal and developmental regulation of adrenodoxin messenger ribonucleic acid in steroidogenic tissues. J Clin Endocrinol Metab 66: 383, 1988 |
|
Mesiano S, Jaffe RB: Interaction of insulin-like growth factor-II and estradiol directs steroidogenesis in the human fetal adrenal toward dehydroepiandrosterone sulfate production. J Clin Endocrinol Metab 77: 754, 1993 |
|
Aberdeen GW, Pepe GJ, Albrecht ED: Developmental expression of and effect of betamethasone on the messenger ribonucleic acid levels for peptide growth factors in the baboon fetal adrenal gland. J Endocrinol 163: 123, 1999 |
|
Carr BR, Simpson ER: Cholesterol synthesis in human fetal tissues. J Clin Endocrinol Metab 55: 447, 1982 |
|
Simpson ER, Carr BR, Parker CR Jr et al: The role of serum lipoproteins in steroidogenesis by the human fetal adrenal cortex. J Clin Endocrinol Metab 49: 146, 1979 |
|
Carr BR, Parker AR, Milewich L et al: The role of low density, high density, and very low density lipoproteins in steroidogenesis by the human fetal adrenal gland. Endocrinology 106: 1854, 1980 |
|
Carr BR, Porter JC, MacDonald PC et al: Metabolism of low density lipoprotein by human fetal adrenal tissue. Endocrinology 107: 1034, 1980 |
|
Ohashi M, Carr BR, Simpson ER: Effects of adrenocorticotropic hormone on low density lipoprotein receptors of human fetal adrenal tissue. Endocrinology 108: 1237, 1981 |
|
Carr BR, Simpson ER: De novo synthesis of cholesterol by the human fetal adrenal gland. Endocrinology 108: 2154, 1981 |
|
Mason JI, Rainey WE: Steroidogenesis in the human fetal adrenal: A role for cholesterol synthesized de novo. J Clin Endocrinol Metab 64: 140, 1987 |
|
Hornsby PJ, Aldren KA: Steroidogenic enzyme activities in cultured human definitive zone adrenocortical cells: Comparison with bovine adrenocortical cells and resultant differences in adrenal androgen synthesis. J Clin Endocrinol Metab 58: 121, 1984 |
|
DiBlasio AM, Voutilainen R, Jaffe RB et al: Hormonal regulation of messenger ribonucleic acids for P450scc (cholesterol side-chain cleavage enzyme) and P450c17 (17α-hydroxylase/17,20-lyase) in cultured human fetal adrenal cells. J Clin Endocrinol Metab 65: 170, 1987 |
|
Brown JB, Beischer NA, Smith MA: Excretion of urinary oestrogens in pregnant patients treated with cortisone and its analogues. J Obstet Gynecol Br Comm 75: 819, 1968 |
|
Oakey RE, Heys RF: Regulation of the production of oestrogen precursors in the foetus. Acta Endocrinol 65: 502, 1970 |
|
Charnvises S, Fencl MD, Osathanondh R et al: Adrenal steroids in maternal and cord blood after dexamethasone administration at midterm. J Clin Endocrinol Metab 61: 1220, 1985 |
|
Seron-Ferre M, Lawrence CC, Jaffe RB: Role of hCG in regulation of the human fetal adrenal gland. J Clin Endocrinol Metab 46: 834, 1978 |
|
Seron-Ferre M, Lawrence CC, Siiteri PK et al: Steroid production by the definitive and fetal zones of the human fetal adrenal gland. J Clin Endocrinol Metab 47: 603, 1978 |
|
Brown JB, Ginz B, Milne CM et al: Stimulation by polypeptides of dehydroepiandrosterone sulfate synthesis in human fetal adrenal slices. J Endocrinol 41: 111, 1981 |
|
Walker ML, Pepe GJ, Albrecht ED: Regulation of baboon fetal adrenal androgen formation by pituitary peptides at mid and late gestation. Endocrinology 122: 546, 1988 |
|
Pepe GJ, Waddell BJ, Albrecht ED: The effects of adrenocorticotropin and prolactin on adrenal dehydroepiandrosterone secretion in the baboon fetus. Endocrinology 122: 646, 1988 |
|
Chakravorty A, Mesiano S, Jaffe RB: Corticotropin-releasing hormone stimulates P450 17α-hydroxylase/17,20-lyase in human fetal adrenal cells via protein kinase C. J Clin Endocrinol Metab 34: 3732, 1999 |
|
Goland RS, Wardlaw SL, Fortman JD et al: Plasma corticotropin-releasing factor concentrations in the baboon during pregnancy. Endocrinology 131: 1782, 1992 |
|
Carr BR, Parker Cr, Madden JD et al: Maternal plasma adrenocorticotropin and cortisol relationships throughout human pregnancy. Am J Obstet Gynecol 139: 416, 1981 |
|
Buster JE, Chang RJ, Preston DL et al: Interrelationships of circulating maternal steroid concentrations in third trimester pregnancies. I: c21steroids: 16α-hydroxyprogesterone, 17α-hydroxyprogesterone, 20α-dihydroxyprogesterone, Δ5 -pregnenolone, Δ5 -pregnenolone sulfate, dehydroepiandrosterone sulfate, and 17-hydroxy Δ5 -pregnenolone. J Clin Endocrinol Metab 48: 133, 1979 |
|
Jones SA, Brooks AN, Challis JRG: Steroids modulate corticotropin-releasing hormone production in human fetal membranes and placenta. J Clin Endocrinol Metab 68: 825, 1989 |
|
Challis JRG, Brooks AN: Maturation and activation of hypothalamic-pituitary-adrenal function in fetal sheep. Endocr Rev 10: 182, 1989 |
|
Albrecht ED, Babischkin JS, Davies WA et al: Identification and developmental expression of the estrogen receptor α and β in the baboon fetal adrenal gland. Endocrinology 140: 5953, 1999 |
|
Voutilainen R, Kahri AI, Salmenpera M: The effects of progesterone, pregnenolone, estriol, ACTH and hCG on steroid secretion of cultured human fetal adrenals. J Steroid Biochem 10: 695, 1979 |
|
Voutilainen R, Kahri AI: Placental origin of the suppression of 3 β -hydroxysteroid dehydrogenase in the fetal zone cells of human fetal adrenals. J Steroid Biochem 13: 39, 1980 |
|
Fujieda K, Faiman C, Reyes FI et al: The control of steroidogenesis by human fetal adrenal cells in tissue culture. IV. The effects of exposure to placental steroids. J Clin Endocrinol Metab 54: 89, 1982 |
|
Pepe GJ, Waddell BJ, Albrecht ED: Effect of estrogen on pituitary peptide-induced dehydroepiandrosterone secretion in the baboon fetus at midgestation. Endocrinology 125: 1519, 1989 |
|
Albrecht ED, Pepe GJ: Effect of estrogen on dehydroepiandrosterone formation by baboon fetal adrenal cells in vitro. Am J Obstet Gynecol 156: 1275, 1987 |
|
Albrecht ED, Pepe GJ: Suppression of maternal adrenal dehydroepiandrosterone and dehydroepiandrosterone sulfate production by estrogen during baboon pregnancy. J Clin Endocrinol Metab 80: 3201, 1995 |
|
Csapo AI, Pulkkinen MO, Ruttner B et al: The significance of the human corpus luteum in pregnancy maintenance. I. Preliminary studies. Am J Obstet Gynecol 112: 1061, 1972 |
|
Johansson EDB: Plasma levels of progesterone in pregnancy measured by a rapid competitive protein binding technique. Acta Endocrinol 61: 607, 1969 |
|
Tulchinsky D, Okada DM: Hormones in human pregnancy. IV. Plasma progesterone. Am J Obstet Gynecol 121: 293, 1975 |
|
Waddell BJ, Pepe GJ, Albrecht ED: Progesterone and 20α-hydroyxpregn-4-en-3-one (20α-OHP) in the pregnant baboon: Selective placental secretion of 20α-OHP into the fetal compartment. Biol Reprod 55: 854, 1996 |
|
Ducsay CA, Stanczyk FZ, Novy MJ: Maternal and fetal production rates of progesterone in rhesus macaques: Placental transfer and conversion to cortisol. Endocrinology 117: 1253, 1985 |
|
Lin TJ, Lin SC, Erlenmeyer F et al: Progesterone production rates during the third trimester of pregnancy in normal women, diabetic women, and women with abnormal glucose tolerance. J Clin Endocrinol Metab 34: 287, 1972 |
|
Lin TJ, Billiar RB, Little B: Metabolic clearance rate of progesterone in the menstrual cycle. J Clin Endocrinol Metab 35: 879, 1972 |
|
Stovall TG, Ling FW, Gray LA et al: Single-dose methotrexate for treatment of ectopic pregnancy. Obstet Gynecol 77: 754, 1991 |
|
Stovall TG, Ling FW, Gray LA et al: Methotrexate treatment of unruptured ectopic pregnancy: A report of 100 cases. Obstet Gynecol 77: 749, 1991 |
|
Teoh ES, Das NP, Dawood MY et al: Serum progesterone and serum chorionic gonadotropin in hydatidiform mole and choriocarcinoma. Acta Endocrinol 70: 791, 1972 |
|
Waddell BJ, Bruce NW: Changes in the blood concentrations of progesterone and 20α hydroxypreg-4-en-3-one during late pregnancy in the conscious rat: A specific role for metabolic clearance rate. Biol Reprod 40: 1231, 1989 |
|
Challis JRG, Olson DM: Parturition. In Knobil E, Neill JD (eds): The Physiology of Reproduction, p 2177. New York, Raven Press, 1988 |
|
Albrecht ED, Pepe GJ: Endocrinology of pregnancy. In Brans YW, Kuehl TJ (eds): Non-Human Primates in Perinatal Research, p 13. New York, John Wiley and Sons, 1988 |
|
Henson MC, Pepe GJ, Albrecht ED: Transuterofetoplacental conversion of pregnenolone to progesterone in antiestrogen-treated baboons. Endocrinology 121: 1265, 1987 |
|
Albrecht ED, Townsley JD: Serum progesterone in the pregnant baboon ( Papio papio ). Biol Reprod 14: 610, 1976 |
|
Albrecht ED, Pepe GJ: Effect of the antiestrogen ethamoxytriphetol (MER-25) and lutectomy on serum progesterone concentrations in pregnant baboons. Endocrinology 115: 1717, 1984 |
|
Walsh SW, Kittinger GW, Novy MJ: Maternal peripheral concentrations of estradiol, estrone, cortisol and progesterone during late pregnancy in rhesus monkeys ( Macaca mulatta) and after experimental fetal anencephaly and fetal death. Am J Obstet Gynecol 135: 37, 1979 |
|
Simpson ER, Porter JC, Milewich L et al: Regulation by plasma lipoproteins of progesterone biosynthesis and 3-hydroxy-3-methyl glutaryl coenzyme A reductase activity in cultured human choriocarcinoma cells. J Clin Endocrinol Metab 47: 1099, 1978 |
|
Simpson ER, Burkhart MF: Acyl CoA: Cholesterol acyl transferase activity in human placental microsomes: Inhibition by progesterone. Arch Biochem Biophys 200: 79, 1980 |
|
Winkel CA, Gilmore J, MacDonald PC et al: Uptake and degradation of lipoproteins by human trophoblastic cells in primary culture. Endocrinology 107: 1892, 1980 |
|
Goldstein JL, Brown MS: Binding and degradation of low-density lipoproteins by cultured human fibroblasts. J Biol Chem 249: 5153, 1974 |
|
Bloch K: The biological conversion of cholesterol to pregnanediol. J Biol Chem 157: 661, 1945 |
|
Hellig H, Gattereau D, Lefebvre Y et al: Steroid production from plasma cholesterol. I. Conversion of plasma cholesterol to placental progesterone in humans. J Clin Endocrinol Metab 30: 624, 1970 |
|
Telegdy G, Weeks JW, Lerner U et al: Acetate and cholesterol metabolism in the human foetoplacental unit at midgestation. 1. Synthesis of cholesterol. Acta Endocrinol (Copenh) 63: 91, 1970 |
|
Baird DT, Cockburn F, Galbraith A et al: Formation of progesterone and pregnenolone from [4-14 C] cholesterol by the intact midterm human foeto-placental unit perfused in vitro. J Endocrinol 56: 187, 1973 |
|
Winkel CA, Snyder JM, MacDonald PC et al: Regulation of cholesterol and progesterone synthesis in human placental cells in culture by serum lipoproteins. Endocrinology 106: 1054, 1980 |
|
Malassine A, Besse C, Roche A et al: Ultrastructural visualization of the internalization of low density lipoprotein by human placental cells. Histochemistry 87: 457, 1987 |
|
Henson MC, Babischkin JS, Pepe GJ et al: Effect of the antiestrogen ethamoxytriphetol (MER-25) on placental low density lipoprotein uptake and degradation in baboons. Endocrinology 122: 2019, 1988 |
|
Rabe T, Kalbfleisch H, Haun A et al: Influence of human lipoproteins on the progesterone synthesis of human term placenta in organ culture. Biol Res Pregnancy Perinatol 4: 75, 1983 |
|
Rothchild I: The regulation of the mammalian corpus luteum. Recent Prog Horm Res 37: 183, 1981 |
|
Holt JA: Regulation of progesterone production in the rabbit corpus luteum. Biol Reprod 40: 201, 1989 |
|
Gibori G, Khan I, Warshaw ML et al: Placental-derived regulators and the complex control of luteal cell function. Recent Prog Horm Res 44: 377, 1988 |
|
Gibori G, Chen YD, Khan I et al: Regulation of luteal cell lipoprotein receptors, sterol contents, and steroidogenesis by estradiol in the pregnant rat. Endocrinology 114: 609, 1984 |
|
Goldring NB, Durica JM, Lifka J et al: Cholesterol side-chain cleavage P450 messenger ribonucleic acid: Evidence for hormonal regulation in rat ovarian follicles and constitutive expression in corpora lutea. Endocrinology 120: 1942, 1987 |
|
Keyes PL, Kostyo JL, Hales DB et al: The biosynthesis of cholesterol side-chain cleavage cytochrome P450 in the rabbit corpus luteum depends upon estrogen. Endocrinology 127: 1186, 1990 |
|
Albrecht ED: A role for estrogen in progesterone production during baboon pregnancy. Am J Obstet Gynecol 136: 569, 1980 |
|
Henson MC, Pepe GJ, Albrecht ED: Regulation of placental low-density lipoprotein uptake in baboons by estrogen: Dose-dependent effects of the antiestrogen ethamoxytriphetol (MER-25). Biol Reprod 45: 43, 1991 |
|
Castracane VD, Goldzieher JW: The relationship of estrogen to placental steroidogenesis in the baboon. J Clin Endocrinol Metab 62: 1163, 1986 |
|
Wunsch DM, Anderson LD, Pepe GJ et al: Regulation of progesterone formation by human placental cells in culture. Endocrinology 119: 998, 1986 |
|
Henson MC, Pepe GJ, Albrecht ED: Developmental increase in placental low density lipoprotein uptake during baboon pregnancy. Endocrinology 130: 1698, 1992 |
|
Albrecht ED, Babischkin JB, Koos RD et al: Developmental increase in expression of messenger ribonucleic acid levels for the low density lipoprotein receptor in syncytiotrophoblasts during baboon pregnancy. Endocrinology 136: 5540, 1995 |
|
Babischkin JS, Pepe GJ, Albrecht ED: Developmental expression of placental trophoblast P450 cholesterol side-chain cleavage, adrenodoxin and Δ5 -3 β -hydroxysteroid dehydrogenase/isomerase messenger ribonucleic acids during baboon pregnancy. Placenta 17: 595, 1996 |
|
Albrecht ED, Henson MC, Pepe GJ: Regulation of placental low density lipoprotein uptake in baboons by estrogen. Endocrinology 128: 450, 1991 |
|
Babischkin JS, Pepe GJ, Albrecht ED: Regulation of progesterone biosynthesis by estrogen during baboon pregnancy: Placental mitochondrial cholesterol side-chain cleavage activity in antiestrogen (ethamoxytriphetol, MER-25)-treated baboons. Endocrinology 124: 1638, 1989 |
|
Babischkin JS, Pepe GJ, Albrecht ED: Estrogen regulation of placental P450 cholesterol side-chain cleavage enzyme messenger ribonucleic acid levels and activity during baboon pregnancy. Endocrinology 138: 452, 1997 |
|
Caritis SN, Hirsch RP, Zeleznik AJ: Adrenergic stimulation of placental progesterone production. J Clin Endocrinol Metab 56: 969, 1983 |
|
Feinman MA, Kliman HJ, Caltabiano S et al: 8-Bromo-3',5'-adenosine monophosphate stimulates the endocrine activity of human cytotrophoblasts in culture. J Clin Endocrinol Metab 63: 1211, 1986 |
|
Ringler GE, Kao LC, Miller WL et al: Effects of 8-bromo-cAMP on expression of endocrine functions by cultured human trophoblast cells: Regulation of specific mRNAs. Mol Cell Endocrinol 61: 13, 1989 |
|
Strauss JF, Kido S, Sayegh R et al: The cAMP signalling system and human trophoblast function. Placenta 13: 389, 1992 |
|
Strauss JF, Martinez F, Kiriakidou M: Placental steroid hormone synthesis: unique features and unanswered questions. Biol Reprod 54: 303, 1996 |
|
Stocco DM, Clark BJ: Regulation of the acute production of steroids in steroidogenic cells. Endocr Rev 17: 221, 1996 |
|
Sugawara T, Holt JA, Driscoll D et al: Human steroidogenic acute regulatory protein: Functional activity on COS-1 cells, tissue specific expression, and mapping of the structural gene to 8p11.2 and a pseudogene to chromosome 13. Proc Natl Acad Sci USA 92: 4778, 1995 |
|
Fant M, Munro H, Moses AC: An autocrine/paracrine role for insulin-like growth factors in the regulation of human placental growth. J Clin Endocrinol Metab 63: 499, 1986 |
|
Han VKM, Lund PK, Lee DC et al: Expression of somatomedin/insulin-like growth factor messenger ribonucleic acids in the human fetus: Identification, characterization, and tissue distribution. J Clin Endocrinol Metab 66: 422, 1988 |
|
Nestler JE, Williams T: Modulation of aromatase and P450 cholesterol side-chain cleavage enzyme activities of human placental cytotrophoblasts by insulin and insulin-like growth factor I. Endocrinology 121: 1845, 1987 |
|
Bahn RS, Speeg KV Jr, Ascoli M et al: Epidermal growth factor stimulates production of progesterone in cultured human choriocarcinoma cells. Endocrinology 107: 2121, 1980 |
|
Migeon CJ, Bertrand J, Wall PE: Physiologic disposition of 4 14C cortisol during late pregnancy. J Clin Invest 36: 1350, 1957 |
|
Juchau MR, Lee QH, Louviaux GL et al: Oxidation and reduction of foreign compounds in tissues of the human placenta and fetus. In Boreus LO (ed): Fetal Pharmacology, p 3231. New York, Raven Press, 1971 |
|
Juchau MR, Pedersen MG, Fantel AG et al: Drug metabolism by placenta. Clin Pharm Ther 14: 673, 1973 |
|
Juchau MR, Symms KG, Zachariah PK: Drug-metabolizing enzymes in the placenta. In Dancis J, Hwant JC (ed): Perinatal Pharmacology: Problems and Priorities, p 89. New York, Raven Press, 1974 |
|
Pepe GJ, Ehrenkranz RA, Townsley JD: The metabolic clearance rates and interconversion of cortisol and cortisone in pregnant and nonpregnant baboons. Endocrinology 99: 597, 1976 |
|
Beitins IZ, Bayard F, Ances IG et al: The metabolic clearance rate, blood production, interconversion, and transplacental passage of cortisol and cortisone in pregnancy near term. Pediatr Res 7: 509, 1973 |
|
Pepe GJ, Albrecht ED: Transuteroplacental metabolism of cortisol and cortisone during mid- and late gestation in the baboon. Endocrinology 115: 1946, 1984 |
|
Pasqualini JR, Nguyen BL, Uhrich F et al: Cortisol and cortisone metabolism in the human foeto-placental unit at midgestation. J Steroid Biochem 1: 209, 1970 |
|
Pepe GJ, Albrecht ED: Fetal regulation of transplacental cortisol-cortisone metabolism in the baboon. Endocrinology 120: 2529, 1987 |
|
Pepe GJ, Waddell BJ, Stahl SJ et al: The regulation of transplacental cortisol-cortisone metabolism by estrogen in pregnant baboons. Endocrinology 122: 78, 1988 |
|
Pepe GJ, Babischkin JS, Burch MG et al: Developmental increase in expression of the messenger ribonucleic acid and protein levels of 11 β -hydroxysteroid dehydrogenase types 1 and 2 in the baboon placenta. Endocrinology 137: 5678, 1996 |
|
Waddell BJ, Albrecht ED, Pepe GJ: Metabolism of cortisol and cortisone in the baboon fetus at midgestation. Endocrinology 122: 84, 1988 |
|
Pepe GJ: The production and secretion of cortisol in neonatal baboons ( Papio papio ). Steroids 33: 251, 1979 |
|
Waddell BJ, Albrecht ED, Pepe GJ: Effect of estrogen on the metabolism of cortisol and cortisone in the baboon fetus at midgestation. Biol Reprod 38: 1006, 1988 |
|
Albrecht ED, Johnson DK, Pepe GJ: Effect of maternal administration of the antiestrogen ethamoxytriphetol throughout the last third of baboon gestation upon neonatal corticoid production. Steroids 37: 45, 1981 |
|
Pepe GJ, Townsley JD: Catabolic regulation of blood cortisol in premature and term baboon neonates. J Steroid Biochem 8: 187, 1977 |
|
Tannin GM, Agarwal AK, Monder C et al: The human gene for 11 β -hydroxysteroid dehydrogenase: Structure, tissue distribution, and chromosomal localization. J Biol Chem 266: 16653, 1991 |
|
Agarwal AK, Rogerson FM, Mune T et al: Gene structure and chromosomal localization of the human HSD11K gene encoding the kidney (type 2) isozyme of 11 β -hydroxysteroid dehydrogenase. Genomics 29: 195, 1995 |
|
Pepe GJ, Burch MG, Albrecht ED: Expression of the 11 β -hydroxysteroid dehydrogenase types 1 and 2 proteins in human and baboon placental syncytiotrophoblast. Placenta 20: 575, 1999 |
|
Stewart PM, Rogerson FM, Mason JI: Type 2 11 β -hydroxysteroid dehydrogenase messenger ribonucleic acid and activity in human placenta and fetal membranes: Its relationship to birth weight and putative role in fetal adrenal steroidogenesis. J Clin Endocrinol Metab 80: 885, 1995 |
|
Sun K, Yang K, Challis JRG: Differential expression of 11 β -hydroxysteroid dehydrogenase types 1 and 2 in human placenta and fetal membranes. J Clin Endocrinol Metab 82: 300, 1997 |
|
Baggia S, Albrecht ED, Pepe GJ: Regulation of 11 β -hydroxysteroid dehydrogenase activity in the baboon placenta by estrogen. Endocrinology 126: 2742, 1990 |
|
Pepe GJ, Davies WA, Dong K et al: Cloning of the 11 β -hydroxysteroid dehydrogenase (11 β -HSD) -2 gene in the baboon: Effects of estradiol on promoter activity of 11 β -HSD-1 and -2 in placental JEG-3 cells. Biochim Biophys Acta 1444: 101, 1999 |
|
Burton PJ, Krozowski ZS, Waddell BJ: Immunolocalization of 11 β -hydroxysteroid dehydrogenase type 1 and 2 in rat uterus: Variation across the estrous cycle and regulation by estrogen and progesterone. Endocrinology 139: 376, 1998 |
|
Pepe GJ, Burch MG, Albrecht ED: Localization and developmental regulation of 11 β -hydroxysteroid dehydrogenase-1 and -2 in the baboon syncytiotrophoblast. Endocrinology 142: 68, 2001 |
|
Lanman JT: The adrenal fetal zone: Its occurrence in primates and a possible relationship to chorionic gonadotropin. Endocrinology 61: 684, 1957 |
|
Neville AM, O'Hare MH: Origin and development of the adrenal gland. In The Human Adrenal Cortex, p 12. Berlin, Springer-Verlag, 1982 |
|
Pepe GJ, Titus JA, Townsley JD: Increasing fetal adrenal formation of cortisol from pregnenolone during baboon ( Papio papio) gestation. Biol Reprod 17: 701, 1977 |
|
Kerr GR, Kennan AL, Waisman HA et al: Growth and development of the fetal rhesus monkey. I. Physical growth. Growth 33: 201, 1969 |
|
McNulty WP, Novy MJ, Walsh SW: Fetal and postnatal development of the adrenal gland in Macaca mulatta. Biol Reprod 25: 1079, 1981 |
|
Scott DE, Pepe GJ: The fetal baboon median eminence as a circumventricular organ: I. Transmission electron microscopy. Brain Res Bull 19: 87, 1987 |
|
Davies WA, Albrecht ED, Pepe GJ: Hypothalamic corticotropin-releasing hormone expression in the baboon fetus at mid and late gestation. Biol Reprod 55: 559, 1996 |
|
Berghorn KA, Albrecht ED, Pepe GJ: Responsivity of the baboon fetal pituitary to corticotropin-releasing hormone in utero at midgestation. Endocrinology 129: 1424, 1991 |
|
Blumenfeld Z, Jaffe RB: Hypophysiotropic and neuromodulatory regulation of adrenocorticotropin in the human fetal pituitary gland. J Clin Invest 78: 288, 1986 |
|
Ackland JF, Ratter SJ, Bourne GL et al: Corticotropin-releasing factor-like immunoreactivity and bioactivity of human fetal and adult hypothalami. J Endocrinol 108: 171, 1986 |
|
Pepe GJ, Davies WA, Albrecht ED: Activation of the baboon fetal pituitary-adrenocortical axis at midgestation by estrogen: Enhancement of fetal pituitary proopiomelanocortin messenger ribonucleic acid expression. Endocrinology 135: 2581, 1994 |
|
Pepe GJ, Albrecht ED: Activation of the baboon fetal pituitary-adrenocortical axis at midgestation by estrogen:Adrenal Δ5 -3 β -hydroxysteroid dehydrogenase and 17α-hydroxylase-17,20-lyase activity. Endocrinology 128: 2395, 1991 |
|
Pepe GJ, Waddell BJ, Albrecht ED: Activation of the baboon fetal hypothalamic-pituitary-adrenocortical axis at midgestation by estrogen-induced changes in placental corticosteroid metabolism. Endocrinology 127: 3117, 1990 |
|
Gardner RL, Lyon MF, Evans EP et al: Clonal analysis of X-chromosome inactivation and the origin of the germ line in the mouse. J Embryol Exp Morphol 88: 349, 1985 |
|
Blandau RJ, White BJ, Rumery RE: Observations on the movements of the living primordial germ cells in the mouse. Fertil Steril 14: 482, 1963 |
|
Fujimoto T, Miyayama Y, Fuyuta M: The origin, migration and fine morphology of human primordial germ cells. Anat Rec 188: 315, 1977 |
|
Witschi E: Migration of the germ cells of human embryos from the yolk sac to the primitive gonadal folds. Contributions to Embryology, Carneige Institution of Washington, DC, 32: 67, 1948 |
|
Davis RM: Localisation of male determining factors in man: A thorough review of structural abnormalities of the Y chromosome. J Med Genet 18: 161, 1981 |
|
Page DC, Mosher R, Simpson EM et al: The sex-determining region of the human Y chromosome encodes a finger protein. Cell 51: 1091, 1987 |
|
Koopman P, Gubbay J, Vivian N et al: Male development of chromosomally female mice transgenic for Sry. Nature 351: 117, 1991 |
|
van Wagenen G, Simpson ME: Embryology of the ovary and testis. In Homo Sapiens and Macaca Mulatta, New Haven, CT, Yale University Press, 1965 |
|
Kurilo LF: Oogenesis in antenatal development in man. Hum Genet 57: 86, 1981 |
|
Jirasek JE: Development of the Genital System and Male Pseudohermaphroditism. Baltimore, MD, Johns Hopkins University Press, 1971 |
|
Wartenberg H: Differentiation and development of the testes. In Burger H, de Kretzer D (eds): The Testis, p 67. New York, Raven Press, 1989 |
|
Ritzen EMS, Neyfeh SN, French FS et al: Demonstration of androgen-binding components in rat epididymis cytosol and comparison with binding components in prostate and other tissues. Endocrinology 89: 143, 1971 |
|
Blanchard M, Josso N: Source of the anti müllerian-inhibiting hormone synthesized by the fetal testis: müllerian-inhibiting activity of fetal bovine Sertoli cells in tissue culture. Pediatr Res 8: 968, 1974 |
|
Josso N: Permeability of membranes to the müllerian-inhibiting substance synthesized by the human fetal testis in vitro: A clue to its biochemical nature. J Clin Endocrinol Metab 34: 265, 1972 |
|
Jost A: Problems of fetal endocrinology: the gonadal and hypophyseal hormones. Recent Prog Horm Res 8: 379, 1953 |
|
Jost A: A new look at the mechanism controlling sexual differentiation in mammals. Johns Hopkins Med J 130: 38, 1972 |
|
Ellsworth K, Harris G: Expression of the type 1 and 2 steroid 5α-reductases in human fetal tissues. Biochem Biophys Res Comm 215: 774, 1995 |
|
Wilson JD: Recent studies on the mechanisms of action of testosterone. N Engl J Med 287: 1284, 1972 |
|
Wilson JD: Testosterone uptake by the urogenital tract of the rabbit embryo. Endocrinology 92: 1192, 1973 |
|
Wilson JD Gloyna RE: The intranuclear metabolism of testosterone in the accessory organs of reproduction. Recent Prog Horm Res 26: 309, 1980 |
|
Siiteri PK, Wilson JD: Testosterone formation and metabolism during male sexual differentiation in the human embryo. J Clin Endocrinol Metab 38: 113, 1974 |
|
Schultz FM. Wilson JD: Virilization of the Wolffian duct in the rat fetus by various androgens. Endocrinology 94: 979, 1974 |
|
Neumann F, von Berswordt-Wallrabe R, Elger W et al: Aspects of androgen-dependent events as studied by antiandrogens. Recent Prog Horm Res 26: 337, 1970 |
|
Goldman AS: Production of hypospadias in the rat by selective inhibition of fetal testicular 17α-hydroxylase and C-lyase. Endocrinology 88: 257, 1971 |
|
Resko JA, Ellinwood WE, Pasztor LM et al: Sex steroids in the umbilical circulation of fetal rhesus monkeys from the time of gonadal differentiation. J Clin Endocrinol Metab 50: 900, 1980 |
|
Resko JA, Buhl AE, Phoenix CH: Treatment of pregnant rhesus macaques with testosterone propionate: Observations on its fate in the fetus. Biol Reprod 37: 1185, 1987 |
|
Resko JA, Abdelgadir SE, Connolly PB: Androgen metabolism by hepatic and renal tissues of the fetal rhesus monkey. J Steroid Biochem Mol Biol 38: 513, 1991 |
|
Walsh SW: Regulation of progesterone and estrogen production during rhesus monkey pregnancy. In Albrecht ED, Pepe GJ (eds): Research in Perinatal Medicine (IV) Perinatal Endocrinology, p 219. Ithaca, NY, Perinatology Press, 1985 |
|
Huhtaniemi IT, Korenbrot CC, Jaffe RB: hCG binding and stimulation of testosterone biosynthesis in the human fetal testis. J Clin Endocrinol Metab 44: 963, 1977 |
|
Huhtaniemi IT, Korenbrot CC, Seron-Ferre M et al: Stimulation of testosterone production in vivo and in vitro in the male rhesus monkey fetus in late gestation. Endocrinology 100: 839, 1977 |
|
Schwartz M, Imperato-McGinley J et al: Male pseudohermaphroditism secondary to an abnormality in Leydig cell differentiation. J Clin Endocrinol Metab 53: 123, 1981 |
|
Carr BR, Parker CRJ, Ohashi M et al: Regulation of human fetal testicular secretion of testosterone: Low-density lipoprotein-cholesterol and cholesterol synthesized de novo as steroid precursor. Am J Obstet Gynecol 146: 241, 1983 |
|
Voutilainen R, Miller WL: Developmental expression of genes for the steroidogenic enzymes P450scc (20,22-desmolase), P450c17 (17α-hydroxylase/17,20-lyase), and P450c21 (21-hydroxylase) in the human fetus. J Clin Endocrinol Metab 63: 1145, 1986 |
|
Baker TG: A quantitative and cytological study of germ cells in human ovaries. Proc R Soc Biol Med 158: 417, 1963 |
|
Baker TG: A quantitative and cytological study of oogenesis in the rhesus monkey. J Anat 100: 761, 1966 |
|
Reyes FI, Faiman C, Winter JSD: Development of the regulatory mechanisms of the hypothalamic-pituitary-gonadal system in the human fetus: The chorionic-hypothalamic-pituitary-gonadal axis. In Novy MJ, Resko JA (eds): Fetal Endocrinology, p 285. New York, Academic Press, 1981 |
|
Ohno S, Smith JB: Role of fetal follicular cells in meiosis of mammalian oocytes. Cytogenesis 3: 324, 1964 |
|
Peters H, Byskov AG, Grinsted J: Follicular growth in fetal and prepubertal ovaries of humans and other primates. J Clin Endocrinol Metab 7: 469, 1978 |
|
Gulyas BJ, Hodgen GD, Tullner WW et al: Effects of fetal or maternal hypophysectomy on endocrine organs and body weight in infant rhesus monkeys ( Macaca mulatta ): With particular emphasis on oogenesis. Biol Reprod 16: 216, 1977 |
|
Harding R, Poore ER, Bailey A et al: Electromyographic activity on the nonpregnant and pregnant sheep uterus. Am J Obstet Gynecol 142: 448, 1982 |
|
Nathanielsz PW, Binlenda Z, Wimsalt J et al: Patterns of myometrial activity and their regulation in the pregnant monkey. In McNellis D, Challis J, MacDonald P et al (eds): The Onset of Labor: Cellular and Integrative Mechanisms, p 359. Ithaca, NY, Perinatology Press, 1988 |
|
Challis JRG: Characteristics of parturition. In Creasy RK, Resnik R (eds): Maternal-Fetal Medicine, 4th ed, p 484. Philadelphia, WB Saunders, 1999 |
|
Casey ML, MacDonald PC: Human parturition: Distinction between the initiation of parturition and the onset of labor. Semin Reprod Endocrinol 11: 272, 1993 |
|
Casey ML, MacDonald PC: The endocrinology of human parturition. Ann N Y Acad Sci 828: 273, 1997 |
|
Lye SJ: Initiation of parturition. Anim Reprod Sci 42: 495, 1997 |
|
Lye SJ, Challis JRG: Paracrine and endocrine control of myometrial activity. In Gluckman PD, Nathanielsz PW (eds): Liggins Symposium, p 361. Ithaca, NY, Perinatology Press, 1989 |
|
Garfield RE, Blennerhassett MG, Miller SM: Control of myometrial contractility: Role and regulation of gap junctions. Oxford Rev Reprod Biol 10: 436, 1988 |
|
Chwalisz K, Garfield RD: Regulation of the uterus and cervix during pregnancy and labor: Role of progesterone and nitric oxide. Ann N Y Acad Sci 828: 273, 1997 |
|
Csapo AI: Force of labor. In Iffy L, Kaminetzky HA (eds): Principles and Practice of Obstetrics and Perinatology, Vol 2, p 761. New York, John Wiley, 1981 |
|
Haluska GJ, Cook MJ, Novy MJ: Inhibition and augmentation of progesterone production during pregnancy: Effects on parturition in rhesus monkeys. Am J Obstet Gynecol 176: 682, 1997 |
|
Avrech OM, Golan A, Weintraub Z et al: Mifepristone (RU-486) alone or in combination with a prostaglandin analogue for termination of early pregnancy: A review. Fertil Steril 56: 385, 1991 |
|
Chwalisz K: The use of progesterone antagonists for cervical ripening and as an adjunct to labour and delivery. Hum Reprod 9 (suppl 1): 131, 1994 |
|
Wolf JP, Simon J, Itskovitz J et al: Progesterone antagonist (RU-486) accommodates, but does not induce labour and delivery in primates. Hum Reprod 8: 759, 1989 |
|
Wolf JP, Sinosich MJ, Anderson T et al: Progesterone antagonist (RU-486) for cervical dilatation, labor induction, and delivery in monkeys: Effectiveness in combination with oxytocin. Am J Obstet Gynecol 160: 45, 1989 |
|
Bansal RK, Goldsmith PC, He Y et al: A decline in myometrial nitric oxide synthase expression is associated with labor and delivery. J Clin Invest 99: 2502, 1997 |
|
Porter DG: The myometrium and the relaxin enigma. Anim Reprod Sci 2: 77, 1979 |
|
Goldsmith LT, Palejwala S, Weiss G et al: The role of relaxin in preterm labor (abstr). In Preterm Birth, Etiology, Mechanisms and Prevention. Charleston, SC, UAB Press, 1997 |
|
Bryant-Greenwood GD, Yamomoto S, Bogic L et al: Relaxin: A role in the premature rupture of the fetal membranes independent of infection (abstr). In Preterm Birth, Etiology, Mechanisms and Prevention. Charleston, SC, UAB Press, 1997 |
|
Ou CW, Orsini A, Lye SJ: Expression of connexin-43 and connexin-26 in the rat myometrium during pregnancy and labor is differentially regulated by mechanical and hormonal signals. Endocrinology 138: 3976, 1998 |
|
Figueroa JP, Honnebier MBOM, Binienda Z et al: Effect of a 48-hour intravenous Δ4 A androstenedione infusion on the pregnant rhesus monkey during the last third of gestation: Changes in maternal plasma estradiol concentrations and myometrial contractility. Am J Obstet Gynecol 161: 481, 1989 |
|
Mecenas CHA, Giussani DA, Owing JR et al: Production of premature delivery in pregnant rhesus monkeys by androstenedione infusion. Nat Med 2: 443, 1996 |
|
Liggins GC, Fairclough RJ, Grieves SA et al: The mechanism of initiation of parturition in the ewe. Recent Prog Horm Res 29: 111, 1973 |
|
Liggins GC, Thorburn GD: Initiation of parturition. In Lamming GE (ed): Marshall's Physiology of Reproduction, 4th ed, Vol 3, p 863. Pregnancy and Lactation, part 2. London, Chapman and Hall, 1994 |
|
Thorburn GD, Liggins GC: Role of the fetal pituitary adrenal axis and placenta in the initiation of parturition. In Lamming GE (ed): Marshall's Physiology of Reproduction, 4th ed, Vol 3, p 1003. Pregnancy and Lactation, part 2. London, Chapman and Hall, 1994 |
|
Honnebier WJ, Jobsis AC, Swaab DF: The effect of hypophysial hormones and human chorionic gonadotropin (hCG) on the anencephalic fetal adrenal cortex and on parturition in the human. J Obstet Gynaecol Br Comm 81: 423, 1974 |
|
Mueller-Heubach E, Myers RE, Adamsons K: Effects of adrenalectomy on pregnancy length in the rhesus monkey. Am J Obstet Gynecol 112: 221, 1972 |
|
Nathanielsz PW, Jenkins SL, Tame JD et al: Local paracrine effects of estradiol are central to parturition in the rhesus monkey. Nat Med 4: 456, 1998 |
|
Patel FA, Clifton V, Challis JRG: Regulation of prostaglandin dehydrogenase activity by cortisol in human term placenta and fetal membranes (abstr 125). 44th Annual Meeting of Society for Gynecologic Investigation, San Diego, CA, 1997 |
|
Karalis K, Goodwin G, Majzoub JA: Cortisol blockage of progesterone: A possible molecular mechanism involved in the initiation of human labor. Nat Med 2: 556, 1996 |
|
Campbell EA, Linton EA, Wolfe CDA et al: Plasma corticotropin-releasing hormone concentrations during pregnancy and parturition. J Clin Endocrinol Metab 64: 1054, 1987 |
|
McLean M, Bisits A, Davies J et al: A placental clock controlling the length of human pregnancy. Nat Med 1: 460, 1995 |
|
Korebrits C, Yu DH, Ramirez MM et al: Antenatal glucocorticoid administration increases corticotropin-releasing hormone in maternal plasma. Br J Obstet Gynaecol 105: 556, 1998 |
|
Hillhouse EW, Grammatopoulos D, Milton NGN et al: The identification of a human myometrial corticotropin-releasing hormone receptor that increases in affinity during pregnancy. J Clin Endocrinol Metab 76: 736, 1993 |
|
Jones SA, Challis JRG: Local stimulation of prostaglandin production by corticotropin-releasing hormone in human fetal membranes and placenta. Biochem Biophys Res Commun 159: 192, 1989 |
|
Benedetto C, Petraglia F, Marozio L et al: Corticotropin-releasing hormone increases prostaglandin F activity on human myometrium in vitro. Am J Obstet Gynecol 171: 126, 1994 |
|
Quartero HWP, Fry CH: Placental corticotropin-releasing factor may modulate human parturition. Placenta 10: 439, 1989 |
|
McLean M, Thompson D, Zhang HP et al: Corticotropin-releasing hormone and β -endorphin in labour. Eur J Endocrinol 131: 167, 1994 |