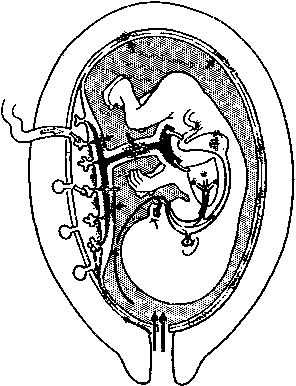
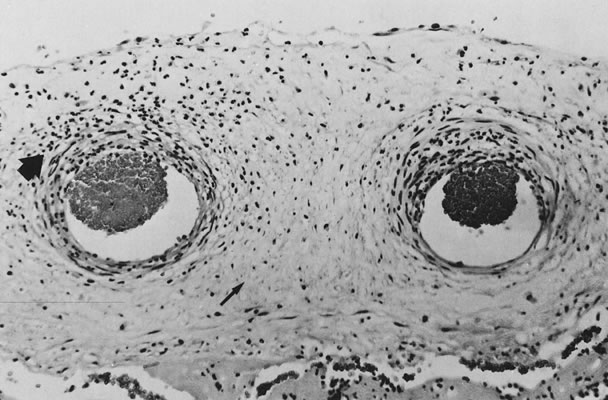
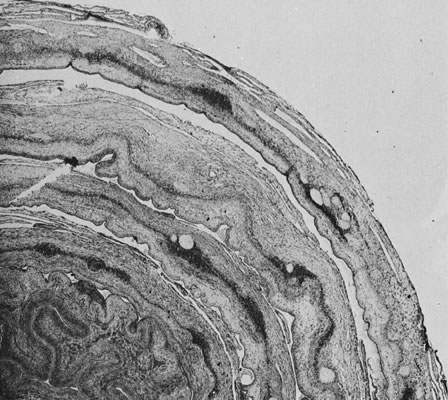
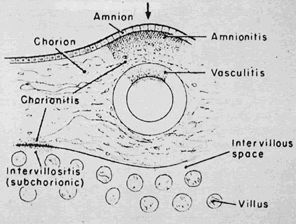
Physiology of Abnormal Uteroplacental Vascular Conversion An outline of the processes of conversion of the uteroplacental vasculature
from a high-resistance/low-capacitance circuit to a high-capacitance
system able to carry large volumes of blood to the intervillous space
is presented in other chapters in this volume. During pregnancy, adaptive
changes associated with endovascular trophoblast invasion, which
occurs in most spiral arteries, lead to dramatic increases in diameter.74 In complicated pregnancies, this vascular adaptation takes place only
in a limited number of placental bed arteries. Failure of uteroplacental
vascular conversion is not specific for preeclampsia or even for hypertensive
disease in pregnancy. Abnormal uteroplacental vascular conversion
and uteroplacental vascular damage also underlie many spontaneous
preterm births.69,75,76 It is reasonable to speculate that abnormal uteroplacental vascular conversion (and
hence uteroplacental perfusion) is the primary process underlying
endothelial cell activation in preeclampsia77 or causing fetal nutritional deprivation in fetal growth retardation and
some cases of intrauterine fetal demise. A causal pathway relating
uteroplacental vascular pathology to preterm labor and premature membrane
rupture has been hypothesized.78 It is critical to keep in mind that the pathologic division between preeclamptic
and nonhypertensive complications of pregnancy may not always
be firm. Likewise, there is a spectrum of uteroplacental vascular changes
that extends from the severely compromised growth-restricted preterm
infant to the term, low ponderal index infant, to the infant electively
delivered due to reduced tolerance of labor. A recent study79 found “pathological changes” in 58% of complicated pregnancies
but also in 40% of normal pregnancies. The authors cited sampling
error as a possible explanation, because a typical biopsy contains only
one or two spiral arteries. This underscores the advantages of generous
placental sampling, which provides a broader view of the uteroplacental
environment. These authors also suggest that additional factors
besides uteroplacental vascular lesions might be required to induce clinical
preeclampsia. This is consistent with the variety of pathways
to preeclampsia recently proposed by Redman and Sargent.80 These pathways include oxidative placental damage and chronic inflammatory
placental lesions, each leading to activation of the maternal endothelium, but
also providing for preeclampsia developing in the context
of a completely normal placenta when the mother has a chronic endothelial
pathology. This last is more common in term preeclampsia, in which
chronic uteroplacental vascular lesions can be uncommon.81 No uteroplacental arterial lesion is distinguished among the different
clinical presentations associated with uteroplacental vascular pathology.69,82 No single uteroplacental histologic feature uniquely characterizes preeclampsia
as opposed to, for example, fetal growth retardation.82,83 Spiral and basal arteries of the placental bed may be more tortuous and/or
densely distributed in preeclamptic than normal term placental bed
arteries.83 Normal placental expansion in the early trimesters stretches the placental
bed arteries.84 The increased tortuosity we observed may be due to reduced placental growth
preeclampsia, with limited expansion of the uterine cavity leaving “redundant” (and therefore tortuous) spiral vessels. This
would lead to a hemodynamically vulnerable architecture at greater
risk for shear stress-induced endothelial injury. In the presence of physiologic
vasoconstriction and/or myointimal hypertrophy, the poor hemodynamic
situation would become worse, in a vicious cycle of vascular
injury. Pathologic implantation may also deform the normal complex quadratic
distribution of interstitial trophoblast invasion,85 which may parallel endovascular conversion in normal pregnancy. Another mechanism for vascular injury in failed conversion may be related
to the observation that arterial wall disorganization occurs to some
degree before there is intravascular or even local trophoblast.86 If these early vascular modifications occur but are not followed by trophoblast
invasion and vascular remodeling, the spiral arteries may be
unstable and vulnerable to flow-mediated injury. While purely mechanical
forces may stress the uteroplacental circulation, molecular signals
may also contribute to the genesis or evolution of vascular lesions. Cytotrophoblast
cells isolated from the placenta and placental bed in
cases of fetal growth retardation have been shown to express significantly
higher levels of plasminogen activator inhibitor-1. This may reduce
placental and uteroplacental arterial capacity to lyse fibrin and
has been proposed as a mechanism for restricting endovascular conversion (by
obstruction) and increasing perivillous fibrin deposition.87 Some cytotrophoblasts in preeclampsia have been reported to have increased
xanthine oxidase activity and decreased expression of superoxide
dismutase that would shift the local balance in favor of increased reactive
oxygen species. The associated finding of peroxynitrite deposition
suggests local superoxide/nitric oxide interactions that may reduce
vascular responsiveness to normal modulators.88 Finally, the contributions of maternal systemic pathologies to uteroplacental
vascular disease cannot be ignored. Given the unique circumstances
of spiral arterial remodeling, it might be expected that maternal
circulating atherogenic factors might preferentially initiate uteroplacental
injury by depositing within the fibrinoid, or compromising endothelial
regrowth. Histologic markers of systemic vascular damage such as lipoprotein(a)89 or endothelial activation90 have been widely studied, but primarily in preeclamptic pregnancies. Given
the wide range of pathophysiologies that can lead down the final
common pathway of preeclampsia, it is not surprising that there is little
consensus. Lipoprotein(a) may be a good example of the diverse clinical
manifestations of vascular injury and the diverse uses of hemostatic
molecules in normal pregnancy. Berg and associates91 reported women with very high serum lipoprotein(a) levels delivering consecutive
very-low-birthweight infants with placentas described as “small
and ischemic.” Meekens and coworkers92 found lipoprotein(a) deposition in the placental bed spiral arteries to
be increased in preeclamptic cases compared with normotensive controls. This
deposition appeared to be directly proportional to the severity
of histologic spiral arterial injury. Further work included placental
basal plate arteries in normal term births, term preeclampsia, preterm
preeclampsia, spontaneous preterm birth, and postpartum curettage
and peripartum hysterectomy samples (Table 1).93
Table 1. Distribution of Lipoprotein A Immunoreactivity in Uteroplacental
Vessels of the Placental Bed and Basal Plate
Click here to view Table
1.
Normal uteroplacental vascular involution after placenta delivery is accompanied
by thrombosis, leukocytic infiltration of the vascular wall, and
proliferation/migration of endothelia and vascular smooth muscle.94 Similar processes occur in atherosclerosis, where platelets, lymphocytes, and
mononuclear leukocytes are recruited to the artery wall and change
the vascular microenvironment.95 Andrew and colleagues94 observed that complement and other markers of potential vascular pathology
were present in the normal term placental bed arterial wall. They
described “invariably” the presence of conglutinated erythrocytes
and/or thrombus. In implantation sites obtained remote from delivery, arterial
lipoprotein(a) deposition was a constant feature. We
saw no thrombi in the two cases with immediate, intractable postpartum
hemorrhage, and lipoprotein(a) deposition was entirely absent. Some
preparation of the placental bed, including development of a more thrombogenic
vascular environment, may be critical for normal placental separation
to occur without maternal exsanguination. Lipoprotein(a) has been recently been described as “one of the few
risk factors capable of promoting both early and advanced stages of
atherogenesis,”96 and therefore it may be particularly useful in the transition from free
flow to no flow after placental delivery. How might the proposed “vascular
preparation” develop? The mechanical trauma of labor, compressing
and releasing uteroplacental arteries, may initiate vascular
damage by shear stress effects as well as basic structural deformation. If
lipoprotein(a) deposition occurs within the restricted period
from labor to delivery, the smooth muscle cell deposition characteristic
of chronic vascular damage might not be expected.97 Our cases of uncomplicated term births delivered after labor uniformly
showed dense medial (fibrinoid) deposition. Perhaps the gradual increase
in uterine activity before frank labor98 contributes to the vascular changes common to healthy parturition. Precocious
or more extensive antepartum development of uteroplacental involution
may account for decreased fetal growth velocity in the third trimester. Likewise, the
more extensive lipoprotein(a) in the uteroplacental
vasculature of term preeclampsia (see Table 1) may result in placental ischemia, as has been proposed as the trigger
of the maternal symptoms of preeclampsia.80,99 Lipoprotein(a) deposition in basal plate arteries was also seen in each
of the three types of prematurity studied (preeclampsia, preterm labor, and
premature rupture of membranes; see Table 1). In preterm preeclampsia, placental bed arteries with failed trophoblast
invasion were uniformly lipoprotein(a)-reactive. Lipoprotein(a) deposition
may be facilitated by preexisting vascular damage. All of our
patients with preterm labor and premature membrane rupture were tocolyzed (prolonging
the period of clinical myometrial activity). Therefore, mechanical
trauma to the uteroplacental vasculature would be assumed. Despite
this, placentas from preterm labor and premature membrane rupture
had fewer and less severe uteroplacental lesions and less widespread
placental ischemic damage than placentas from preterm preeclampsia.82 Lipoprotein(a) deposition in preterm labor and premature membrane rupture
may be the result of chronic low-level uteroplacental vascular pathology, the
mechanical trauma of labor, or a combination of the two processes. A significant role of lipoprotein(a) in processes related to uteroplacental
vascular wall damage would explain the recent clinical observation
of homocysteinuria in patients with preeclampsia.100 Homocysteine has been studied as a marker of atherosclerotic vascular
processes. Harpel and Borth101 have shown that sulfhydryl-containing compounds (including homocysteine) increase
the affinity (up to 84-fold) between lipoprotein(a) and fibrin. One
group found that the C677T mutation in the methylene tetrahydrofolate
reductase gene (which slows homocysteine metabolism) carried
a 2.5-fold (confidence interval 1.0 to 6.0) to 3.29-fold (confidence
interval 1.03 to 10.5) odds of placental vasculopathy.102,103 When other procoagulant risk factors (such as protein C deficiency or
resistance) are screened, presence of the C677T mutation and decreased
protein C function bring an additive risk of placental vasculopathy (3.4, confidence
interval 1.8 to 6.42).103 The C677T gene mutation appears to affect the uteroplacental vasculature
via elevations of maternal serum homocysteine.104 Plasma levels of homocysteine may be higher in women with preeclampsia,105 possibly reducing the maternal systemic endothelial threshold independent
of placental vascular disease, as proposed by Redman and Sargent.80 Uteroplacental vascular disease may be more common in this condition; 31% (26/84) of
women with placental infarct or abruption were found to
have hyperhomocysteinemia compared with 9% (4/46) of controls.106 Many other heritable risk factors for cardiovascular disease, including
factor V Leiden, methylenetetrahydrofolate reductase, and prothrombin
gene, and for deficiencies of protein C, protein S, and antithrombin
III have also been identified as more prevalent among women with severe
preeclampsia. Among women with obstetric vascular complications, a
threefold greater prevalence of at least one of these heritable factors
or acquired anticardiolipin antibodies has been described, and a more
than tenfold increase in “double hits” (presence of two
thrombophilic factors).107 The same group studied obstetric vascular complications more broadly, including
severe preeclampsia, abruptio placentae, fetal growth restriction, and
stillbirth. Fifty-seven patients had a thrombophilic mutation, as
compared with 19 women with normal pregnancies (52% and 17%, respectively; p< .001). Deficiency of protein S, protein C, or antithrombin III or
anticardiolipin antibodies was found in 14 additional patients compared
with 1 control (13% and 1%, respectively; p< .001).108 However, not all reports are so conclusive.109,110,111 There are several likely explanations: first, that these factors are not
associated with vascular complications in obstetrics, no more than
they are in adult vascular disease;112 that there is population heterogeneity for the known gene polymorphisms,113 so that prevalence of specific polymorphisms will naturally vary widely, and
population-specific polymorphisms may not be identified; and that
thrombophilia is a multigenic disease.114 It is unlikely that current laboratory testing can label every biologic
variable that can predispose to clotting. It is also unlikely that every
functional mutation of those genes known to alter hemostasis has been
identified in every racial or ethnic group. As of 1997, 650 functional
mutations of the cystic fibrosis gene had been identified.115 Only a handful of polymorphisms have been identified in the candidate
thrombophilia genes (see above). Finally, the maternal environment likely
contributes to vascular fragility, including older maternal age and
stress. A “thrombophilia workup” is fast becoming common
in the at-risk pregnancy evaluation. In the absence of a positive family
history for early-onset cardiovascular disease and placental evidence
of uteroplacental vascular pathology, this may not be cost-effective. Given
the prevalence of many of these polymorphisms, broad application
of thrombophilia screening may result in unnecessary therapy and
anticoagulation of asymptomatic carriers whose pregnancy pathology may
lie elsewhere. Placental pathology can be a useful screen to select those
who might most benefit from a thrombophilia screen. It can also identify
patients with vascular pathology that cannot currently be labeled
with our laboratory methods who may yet benefit from vascular-directed
therapy and surveillance. Pathology of Abnormal Uteroplacental Vascular Conversion Placental bed biopsy specimens are not easy to obtain, and their interpretation
may be even more complicated than placenta histopathologic examination. In
the clinical setting, some questions that are critical to
the obstetrician are: Is there histologic evidence of uteroplacental
vascular pathology? Is the pathology (in the most general of terms) “acute” rather
than “chronic”? Is there associated
placental damage that may have impaired placental function (and therefore
fetal well-being)? These questions can all be answered by careful
examination of the delivered placenta, without a placental bed biopsy. The
delivered placenta has several advantages over the placental
bed biopsy; failing a hysterectomy, biopsy of the placental bed of necessity
provides only a small and focal sample of the placental bed, within
which there is much regional variation.85 Four or five en face slices of the basal plate (which can all fit into a single cassette) may
include the distal (basal plate) segments of several different uteroplacental
arteries, located at different sites in the placental bed.116 Variation in uteroplacental vascular anatomy and pathology implies also
that one section of the placental villi may not be representative of
placental function. Thorough examination of the delivered placenta can
provide a better picture of the intrauterine environment of the fetoplacental
unit than a placental bed biopsy. Lesions of the end-vascular
distribution of the uteroplacental circulation (in the basal plate) may
not perfectly mirror myometrial pathology. The ability to routinely
identify failure of uteroplacental vascular adaptation, fibrinoid necrosis/atherosis, persistence of endovascular trophoblasts, thrombosis, and
chronic vasculitis in the basal plate can clarify the nature and
mechanisms involved in pregnancy compromise. For example, in a recent
study of women at risk for pregnancy loss and spontaneous preterm birth, both
acute ascending infection and decidual vascular lesions were
identified. These two patterns were found to segregate with the clinical
change of the cervix; those with progressive cervical shortening had
significantly more acute inflammation, whereas those without cervical
shortening had primarily decidual vascular lesions.117 It can be useful to remember that acute inflammatory and decidual vascular
lesions often tend to be mutually exclusive. Arias and colleagues118 proposed two “distinct subgroups among patients with preterm labor
and preterm ruptured membranes.” Factor72 and cluster analyses119 have confirmed the tendency for these lesions to segregate in separate
subgroups of preterm infants. This distinction can be routinely made
by reviewing basal plate uteroplacental vessels whenever a placenta is
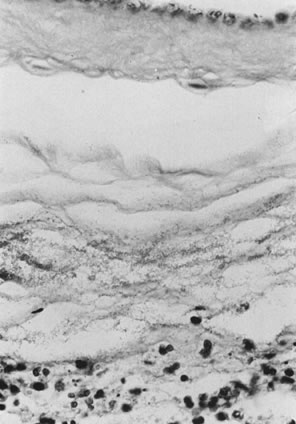
referred for pathology examination (Fig. 12).  Fig. 12. Maternal surface of the term placenta showing the entry point of a maternal
spiral artery into the central area of a cotyledon. Fig. 12. Maternal surface of the term placenta showing the entry point of a maternal
spiral artery into the central area of a cotyledon.
|
In the basal plate or the placental bed, uteroplacental arteries with absent, incomplete, or
failed adaptation have variable persistence of vascular
muscle and elastic lamina (Fig. 13). This leads to increased uterine vascular resistance, decreased capacitance, and
decreased total blood flow to the placenta. Doppler and isotope
studies in the human suggest that uteroplacental flow is decreased
to 50% to 70% of normal, which may explain the often associated fetal
growth retardation.120,121 Increased uterine artery resistance parallels histologic evidence of impaired
trophoblast migration.122 It is been often held that trophoblast invasion of the uteroplacental
vasculature is completed at the myometrial level by the middle of the
second trimester. Trophoblast vascular conversion at those deeper levels
commonly continues even into the early third trimester. Endovascular
trophoblast in the superficial (basal plate) uteroplacental arteries
later than the early midtrimester may reflect abnormal (delayed) uteroplacental
vascular conversion. Fibrinoid necrosis of the vessel wall
with mural foamy cells (“atherosis,” see Fig. 13) is accompanied by dense lipoprotein(a) deposition within the vascular
wall, reflecting a vascular pathology similar to atherosclerosis. Other
common uteroplacental arterial lesions are thrombosis and chronic vasculitis. For
cases of preeclampsia in which uteroplacental vascular
pathology is common, Redline and Patterson123 explored the hypothesis that there is a generalized maturation defect
in the extravillous trophoblast that leads to increased accumulation of
trophoblast in the superficial layers of the implantation site. Increased
thickness of basal cytotrophoblast and increased cytotrophoblast
proliferation (marked by immunostaining with proliferating cell nuclear
antigen) were seen in preeclamptic placentas 24 to 40 weeks of age
compared to term. Whether trophoblast “pile-up” at the placental/decidual
interface reflects an intrinsic invasive defect, or whether
their migration is inhibited by maternal factors is not clear. This
pile-up is not, in our view, specific to preeclampsia. It is, however, yet
another marker of abnormal uteroplacental interaction assessable
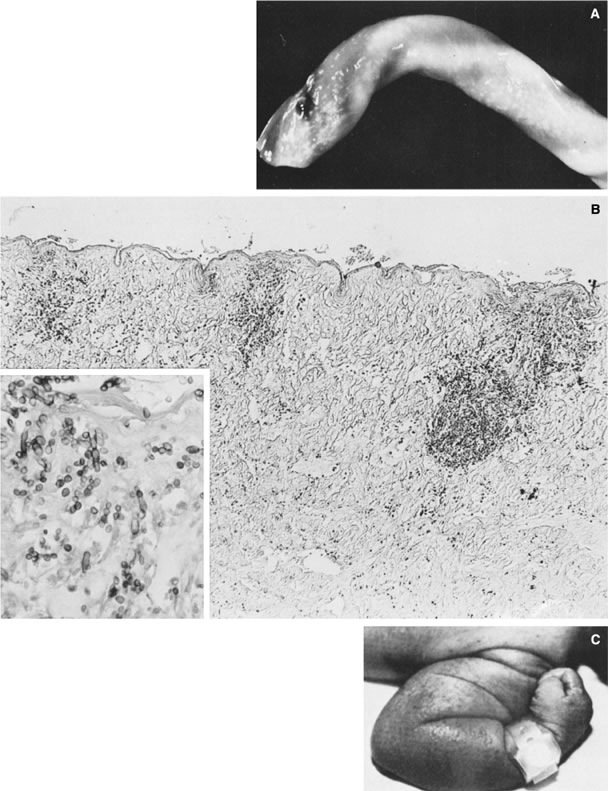
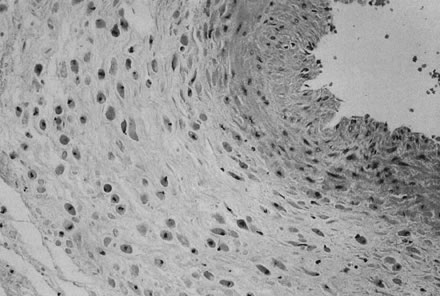
in the delivered placenta. 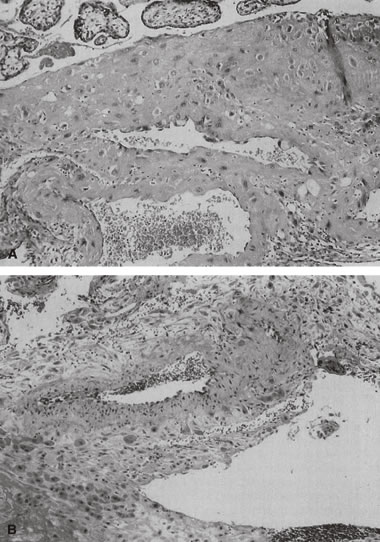 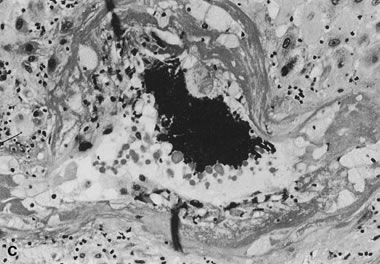 Fig. 13. A. Normally adapted maternal spiral arteries with trophoblasts within the
vessel lumen and invading the wall, seen at the end of the second trimester (hematoxylin
and eosin, ×10). B. A vessel with failed adaptation, with retained smooth muscle, seen near
term (hematoxylin and eosin, ×10). C. Acute atheroma with intimal and medial proliferation, fibrinoid necrosis, foamy
macrophages, and increased numbers of lymphocytes (“chronic
vasculitis”) from an early third-trimester placenta from a
woman with pregnancy-induced hypertension (hematoxylin and eosin, ×20). Fig. 13. A. Normally adapted maternal spiral arteries with trophoblasts within the
vessel lumen and invading the wall, seen at the end of the second trimester (hematoxylin
and eosin, ×10). B. A vessel with failed adaptation, with retained smooth muscle, seen near
term (hematoxylin and eosin, ×10). C. Acute atheroma with intimal and medial proliferation, fibrinoid necrosis, foamy
macrophages, and increased numbers of lymphocytes (“chronic
vasculitis”) from an early third-trimester placenta from a
woman with pregnancy-induced hypertension (hematoxylin and eosin, ×20).
|
When the uteroplacental vasculature is abnormal, there is a greater chance
for “uteroplacental vascular accidents” such as placental
infarcts124 or abruption. When uteroplacental arteries are occluded, intervillous
flow ceases, the intervillous space collapses, and villi become compressed
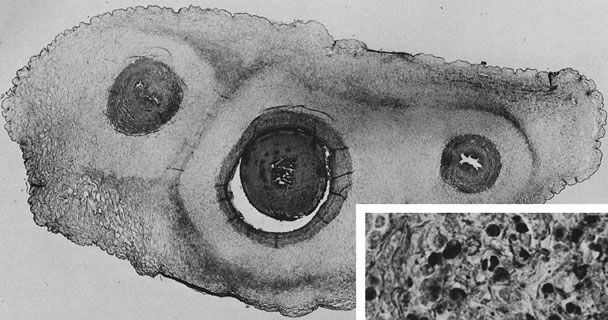
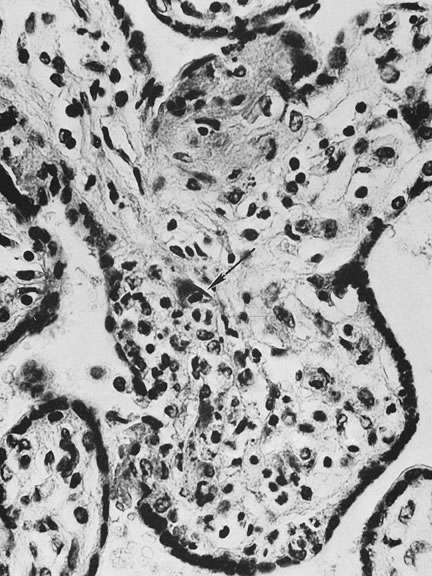
and undergo ischemic necrosis (an infarct, Fig. 14). Infarcts located in the center of the placenta rather than the perimeter (where
there are fewer invasive trophoblasts)85 are considered more significant to the fetus, because the center of the
placenta, with optimal trophoblast vascular conversion, should be the
most healthy part of the placenta. As many as 10% of completely uncomplicated
term births will have one placental infarct, but 90% of these
are single, less than 1cm3 in volume, and located at the placental margin. 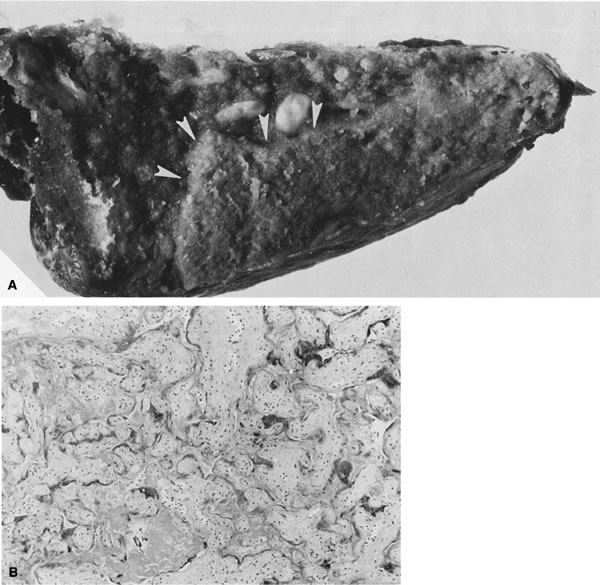 Fig. 14. A. Infarcts are based at the maternal surface. The villous granular character ( arrowheads) is retained. B. True infarct of the placental villi, occurring some time ago. Collapse
of the maternal intervillous space is seen, and ghost-like villi with
minimal basophilia remain in the syncytiotrophoblast nuclei (hematoxylin
and eosin, ×10). Fig. 14. A. Infarcts are based at the maternal surface. The villous granular character ( arrowheads) is retained. B. True infarct of the placental villi, occurring some time ago. Collapse
of the maternal intervillous space is seen, and ghost-like villi with
minimal basophilia remain in the syncytiotrophoblast nuclei (hematoxylin
and eosin, ×10).
|
Abruption is a “hemorrhagic infarct.” In abruption, the placenta
is forcibly separated from the uterine wall by retroplacental hemorrhage
from abnormal uteroplacental vessels.125 Placental compression by a retroplacental hematoma increases fetal blood
volume and may be associated with villous stromal hemorrhage (Fig. 15). 126 Villous stromal hemorrhage may indicate placental trauma and appears as
a bruise. Villous stromal hemorrhage may also be a precursor to fetomaternal
transfusion. In our experience, in cases of fetomaternal blood
group compatibility (in which preformed maternal antibodies to fetal
blood do not exist), an acute abruption that is clinically stabilized
may be followed by fetal decompensation as a result of chronic fetomaternal
transfusion and severe fetal anemia, which may lead to fetal death. Separation
from the uterine lining precludes effective blood flow
to the involved placental area, acutely reducing fetoplacental oxygen
availability.125 Endothelial damage due to hypoxia is complicated by increased intravascular
volume (due to placental compression) to villous stromal hemorrhage.126 This process may also explain the commonly extensive fetal visceral and
germinal matrix hemorrhages in abruption.125 Basal intervillous thrombi are primarily maternal blood;127 these lesions may be very mild forms of an abruption-type pathology. Intervillous
thrombi off the basal plate may be foci of coagulation initiated
by fetomaternal hemorrhage.128 Marginal separation of the placenta may be related to prematurity.129 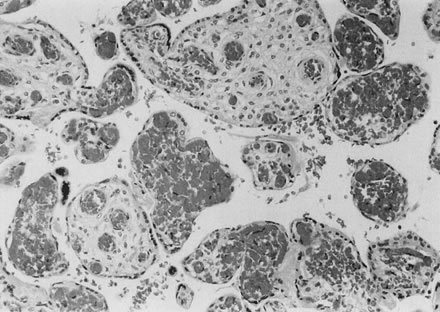 Fig. 15. Intravillous hemorrhage due to unequal blood pressure within the maternal
intervillous space and the fetal capillaries after premature separation
of the placenta (clinical abruption). The result is rupture of the
fetal capillaries with bleeding into the loose villous stroma, a placental “bruise” (hematoxylin and eosin, ×20). Fig. 15. Intravillous hemorrhage due to unequal blood pressure within the maternal
intervillous space and the fetal capillaries after premature separation
of the placenta (clinical abruption). The result is rupture of the
fetal capillaries with bleeding into the loose villous stroma, a placental “bruise” (hematoxylin and eosin, ×20).
|
In addition to these large-scale placental lesions, chronically abnormal
uteroplacental vascular perfusion may impair the growth and development
of the placenta (Fig 16). Alternatively, it may lead to diffuse villous lesions that cannot be
identified grossly. Scarred, shrunken, fibrotic, and hypovascular villi, with
reduced placental capillary number and/or caliber, may be caused
by capillary destruction by abnormal turbulent uteroplacental flow (Fig. 17).130 Villous capillary damage may lead to fetomaternal hemorrhage. Fetomaternal
hemorrhages in the midtrimester (when the barrier between maternal
and fetal bloodstreams is still comparatively thick and “sturdy”) are
more frequent in hypertensive pregnancies.131 Chronic placental perfusion injury and/or nutritional deprivation may
impair terminal villous arborization. The small placenta with uteroplacental
vascular lesions may often show histologic increased intervillous
volume and decreased villous parenchymal volume, smaller terminal villi, and
apparently sparser villous numbers. The placental mass, in these
circumstance, may simply fail to develop. The net effect of poor
placental growth is a reduced total villous capillary bed. This anatomy
would be analogous to an emphysematous lung. Just as significant to
the fetus is the effect of reduced placental capillary bed on total peripheral
resistance. The smaller placental vascular tree would have increased
placental resistance and cause increased cardiac work. Five hundred
milliliters per minute of fetal cardiac output is directed to the
placenta; the cardiac work effects may be dramatic. An indirect reflection
of umbilical-placental resistance is the umbilical systolic/diastolic
ratio. This ratio approaches infinity and end-diastolic flow in
the umbilical artery may be negative when the placental capillary bed
is reduced by more than 50%.132 A reduction in the total fetoplacental capillary bed is paralleled by
a reduction in fetoplacental volume. Reduced fetal blood volume can result
in a reduced fetal glomerular filtration rate and oligohydramnios.133 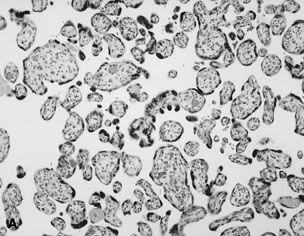 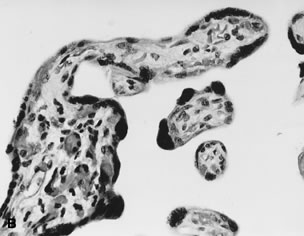 Fig. 16. A. Placenta at 26 weeks' gestation with “accelerated maturation” due
to preeclampsia (hematoxylin and eosin, ×10). B. Unlike normal syncytial knots, exaggerated syncytial knots project above
the surface of the villus (hematoxylin and eosin, ×40). Fig. 16. A. Placenta at 26 weeks' gestation with “accelerated maturation” due
to preeclampsia (hematoxylin and eosin, ×10). B. Unlike normal syncytial knots, exaggerated syncytial knots project above
the surface of the villus (hematoxylin and eosin, ×40).
|
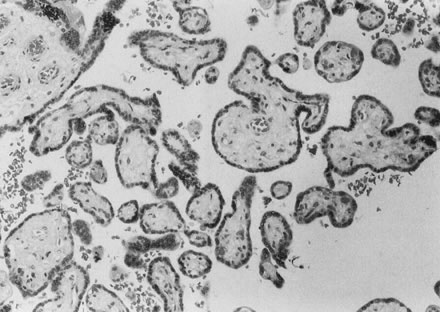 Fig. 17. Hypovascular villi with decreased number and caliber of fetal villous capillaries
and stromal fibrosis (hematoxylin and eosin, ×10). Fig. 17. Hypovascular villi with decreased number and caliber of fetal villous capillaries
and stromal fibrosis (hematoxylin and eosin, ×10).
|
Coagulation Coagulation is a principal defense mechanism of organisms against invading
microorganisms. It plays a central role in both the damage and repair
stages of tissue or cell injury or death. Pathologic initiation of
coagulation has been used as a marker of clinically significant allograft
rejection.134,135 Coagulopathy has been implicated in the pathogenesis of certain types
of obstetric compromise, including antiphospholipid antibody-related fetal
death136 and preeclampsia.137 Maternal hypercoagulable states may accompany maternal autoimmune diseases (e.g., systemic
lupus erythematosus) or may occur in clinically healthy
patients. The increased risk of pregnancy failure in the context
of maternal hypercoagulability is independent of maternal clinical disease
status.138–142 The characteristic pathology of pregnancy loss in these conditions (uteroplacental
thrombosis and placental infarction)143 is not pathognomonic of any specific laboratory abnormality, although
it may be due as much to the inadequacy or incompleteness of our laboratory
screening tests for coagulopathy as to the nonspecificity of the
pathologic process. For a decade, all patients followed in a university-based
rheumatology clinic were prospectively studied. Chronic inflammatory
lesions were a significant component of the pathology in mothers
with systemic lupus erythematosus, antiphospholipid antibodies, or
both. However, the maternal clinical disease and serologic profiles correlated
poorly with clinical outcome, with the type of placental pathology (coagulation-related, chronic inflammation, or uteroplacental vascular), and
with the site of placental injury. It is not surprising, therefore, that
serologic delineation of the syndromes of obstetric compromise
is controversial.144,145 Generally accepted laboratory assays include lupus anticoagulant and anticardiolipin
and antiphospholipid antibodies.143,144,145,146 Deficiencies of protein C, S,147,148 and antithrombin III may cause hypercoagulability during pregnancy, fetal
wastage, and similar placental findings. The lack of specificity in
the clinical and laboratory assessment of pregnancy outcome and the
apparent pathophysiologic mechanism of placental damage probably means
that we have not completely delineated all clinical and laboratory markers
that are directly causative of obstetric compromise. The uterine vasculature is particularly susceptible to thrombosis (because
its endothelium is normally eroded and the basement membranes and
decidual stromal collagen are normally exposed to circulating maternal
platelets) for up to at least 24 weeks. Complete conversion includes
re-endothelialization of the maternal artery. Failure to accomplish maternal
endothelial regrowth over the converted uteroplacental vascular
wall of fibrinoid material and embedded trophoblasts is part of the vascular
pathology of preeclampsia.149 The mechanisms of action of aspirin may include alterations in both maternal
systemic and placental prostacyclin/thromboxane production.150 Not all trials have identified beneficial effects of anticoagulants on
pregnancy outcome; this variance may be explained by differences in the
therapeutic effects of a uniform dosage within a study population151 or by the reduced efficacy of therapy instituted late in the pathophysiologic
process.152,153 Therapy begun after there is a clinically detectable disorder may not
be able to normalize uteroplacental perfusion in an abnormally developed
vascular anatomy. If the early development of the fetoplacental unit
is compromised, it may be impossible for later therapy to yield normal
placental (and fetal) growth and development. The reports of potentially
deleterious effects, such as an increased risk of fetal death after
placental abruption, are of greater concern.154,155 One study (in which abruption was self-reported) did not confirm an increased
risk.156 A limitation of most clinical studies of anticoagulant therapy in pregnancy
is the lack of placental histopathologic examination. In one study, data
suggested that clinical outcome was improved after aspirin therapy, although
no improvement in placental histopathology was noted.157 If the results of this study are confirmed, therapeutic interventions
that alter the maternal course but do not correct the intrauterine pathophysiology
may require extremely careful fetal monitoring to avoid fetal
compromise. If therapy also improves fetal hemodynamics and hence
fetal ability to compensate for uteroplacental pathology,158 then less concern about subclinical fetal compromise is warranted. In our analyses of pregnancies before and after anticoagulant therapy, we
devised a diagnostic schema that provides a semiquantitative score
of the severity of the lesion and identifies a target tissue for pathologic
coagulation. The potential targets for pathologic coagulation include
the (maternal) uteroplacental vasculature, the basal plate (including
Nitabuch's fibrin), the intervillous space, the villous (syncytiotrophoblast) surface, and the fetoplacental vasculature. In our
experience, single thrombotic lesions in the uteroplacental arteries that
occlude less than 50% of the lumen and are not accompanied by villous
evidence of abnormal uteroplacental perfusion are not uncommon at
term. In fact, these lesions may represent part of the uteroplacental
vascular preparation for parturition, which allows for rapid cessation
of uteroplacental flow at placental delivery and protects the mother
against exsanguination.87 Multifocal thromboses completely occluding the uteroplacental arterial
lumina are not consistent with uncomplicated term delivery. It has been
our experience that maternal anticoagulant therapy is most effective
when the maternal vasculature is the target of pathologic coagulopathy, and
that it is specifically not effective when coagulation is initiated
on the villous trophoblast surface or within the fetoplacental vasculature. The laying down of a fibrinoid layer at the maternal/placental interface
is a standard process of normal pregnancy. This layer of Nitabuch's
fibrin is progressively laid down throughout gestation, leading to, at
term, a smooth plane of cleavage for the delivering placenta. Even
this “normal” amount of coagulation in the basal plate
has been proposed to occur as a result of maternal/placental immunologic
interactions. The authors also identified increased basal plate coagulation
in cases of pregnancy compromise believed to be of immunologic
origin.159 This increase in basal coagulation results in a thick band of basal plate
fibrin/fibrinoid, with up to several layers of entrapped and variably
well-preserved basal villi, often with focal villitis. We have often
seen a subjective increase in basal cytotrophoblasts in such cases, a
finding that may be reflected in the observations by Redline and Patterson123 of increased basal cytotrophoblast in preeclampsia. We speculate that
although an intrinsic trophoblast defect cannot be ruled out, trophoblast
invasion is impaired by the markedly abnormal basal matrix. It is
also possible that deposition of Nitabuch's fibrin occurs in response
to basal cytotrophoblast. Much further work is required to dissect
the causal pathways of defective placentation and pregnancy compromise. A
highly unusual placental lesion, maternal floor infarction (Fig. 18), shares the histologic features of uteroplacental and decidual “sparing” and
intervillous fibrin/fibrinoid but is much more massive, generally
involving 80% to 95% of the villous parenchyma. We have
seen potential intermediate lesions in the central basal plate: irregularly
increased basal fibrin/fibrinoid and 5 to 15 layers of entrapped
necrotic villi in cases of recurrent pregnancy loss. Perhaps maternal
floor infarction, a rare lesion recognized to recur in subsequent pregnancies, is
an extreme example of a coagulopathy initiated by pathologic
maternal/placental interactions in the basal plate. Milder forms
of this lesion (in which 5 to 20-plus villi are entrapped in encroaching
parabasal fibrin/fibrinoid) are not uncommonly found in serial pregnancies
from women with recurrent pregnancy loss. Anecdotally, this lesion
may not be modified by maternal aspirin/heparin therapy. 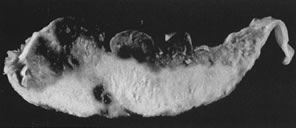 Fig. 18. Maternal floor infarct associated with intrauterine demise at 26 weeks. Fig. 18. Maternal floor infarct associated with intrauterine demise at 26 weeks.
|
Coagulation on the placental trophoblast surface distant from the basal
plate may be initiated by a variety of stimuli, including circulating
endotoxin and trophoblast apoptosis,160 and it may play a critical role in syncytial repair. The common finding
of placental trophoblast surface coagulation in association with chronic
villitis and/or intervillositis supports a hypothesis that inflammatory
processes, including local immune complex formation, may damage
syncytiotrophoblast. Massive perivillous fibrin/fibrinoid (Fig. 19) with or without chronic placental inflammation is, in our experience, the
most common pathology in patients who fail to respond to anticoagulant
therapy for antiphospholipid antibody-related pregnancy loss. Some
women have never had uteroplacental thromboses in previous (untreated) pregnancies. Other
patients have had prior uteroplacental vascular
thromboses; in those patients, anticoagulant therapy may have remedied
the maternal coagulopathy but not prevented the evolution of coagulopathy
at other sites (e.g., the placental surface). 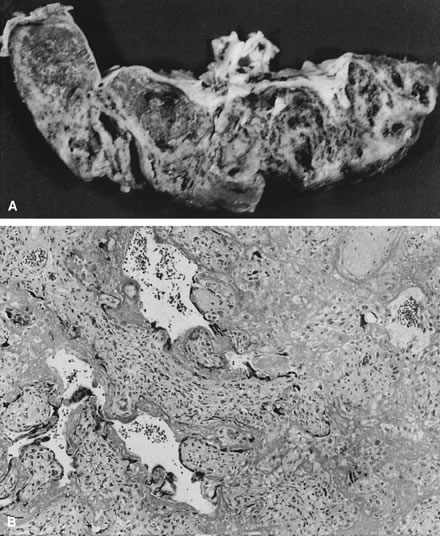 Fig. 19. A. Cross-section of a 27-week-gestation placenta. Delivery was due to intrauterine
growth retardation and fetal distress. Massive perivillous fibrinoid
is seen as perpendicular bands of white tissue separating nests
of normal-appearing villous tissue. B. Microscopic appearance is quite distinct from an infarct. The villi are
not infarcted, but they are avascular and there is loss of the syncytial
trophoblast layer, which is replaced by firm eosinophilic material
within which is proliferation of “X cells” (hematoxylin
and eosin, ×10). Fig. 19. A. Cross-section of a 27-week-gestation placenta. Delivery was due to intrauterine
growth retardation and fetal distress. Massive perivillous fibrinoid
is seen as perpendicular bands of white tissue separating nests
of normal-appearing villous tissue. B. Microscopic appearance is quite distinct from an infarct. The villi are
not infarcted, but they are avascular and there is loss of the syncytial
trophoblast layer, which is replaced by firm eosinophilic material
within which is proliferation of “X cells” (hematoxylin
and eosin, ×10).
|
Damage to the fetoplacental vasculature commonly results in the initiation
of coagulation. One may attempt, with variable success, to distinguish
fetoplacental vascular lesions primarily related to coagulation from
lesions in which coagulation is secondarily invoked. Thrombotic lesions
occurring in the absence of other placental pathology most often
involve the chorionic or large fetal stem vessels but can occur at any
level of the villous tree. In preterm infants, acute inflammatory thrombi
in the amniotic-surface side of the chorionic vessel may be the
most common cause of placental vascular thrombosis. In term infants, the
most common etiology may vary by patient population. In some populations
with a high prevalence of thrombophilic mutations, fetal thrombophilia
may be an important contribution to this serious placental lesion. When
these lesions are identified in a compromised fetus, detailed
maternal and paternal family histories may be useful to clarify inheritance
and better estimate recurrence risk in subsequent pregnancies.161 Bland (noninflammatory) eosinophilic mural thrombi are often calcified
and grossly visible on careful inspection of the chorionic surface. Such
lesions may result from mechanical trauma to the chorionic plate vessels (e.g., fetal
kicking of the chorionic plate). In our experience, these
lesions are commonly seen in areas of vasa previa and velamentous
vessels, if the vessels running in the membranes and/or near the site
of rupture are adequately sampled. In cases of acute ascending infection, chorionic
plate inflammation and chorionic vasculitis may be accompanied
by chorionic mural thrombi, generally on the superficial side
of the vessel (closest to the inflammatory stimuli in the amniotic
fluid, and in the area of greatest density of neutrophil infiltration). Similarly, bland
thrombi can occur in the fetal stem vessels (Fig. 20), although in these cases it is difficult to posit a role of mechanical
trauma. These lesions often accompany other evidence of processes that
damage endothelia, such as chronic villitis and so-called hemorrhagic
endovasculitis. If thrombi in the fetal stem vessels are associated
with inflammatory infiltrates, they are most commonly chronic in nature
and are associated with chronic villitis in other sites. Many times, vascular
lesions may be seen in cases of stillbirth. When vascular lesions
are caused by stillbirth (stillbirth being a “global” process), the
vascular lesions are more generalized and may begin in
the capillary bed. We also believe that “multiple lumens,” recanalization
of thrombi, are a marker of antemortem thrombus. 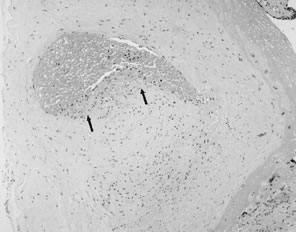 Fig. 20. Band of eosinophilic thrombus adherent to endothelial wall. “Cushion
defect” most commonly seen within chorionic plate and stem arteries, possibly
at branch points. Endothelium beneath thrombus is absent (hematoxylin
and eosin, ×10). Fig. 20. Band of eosinophilic thrombus adherent to endothelial wall. “Cushion
defect” most commonly seen within chorionic plate and stem arteries, possibly
at branch points. Endothelium beneath thrombus is absent (hematoxylin
and eosin, ×10).
|
Thrombosis of large fetal stem arteries in the placenta results in downstream
vascular obliteration and avascular villi. Thrombosis of large
fetal stem veins could also result in placental capillary injury but might
involve local capillary rupture due to increased venous pressure
from local venous obstruction. Venous thrombi are considered to carry
the greater risk for fetal cerebral thromboembolism and cerebral injury.162 Redline and Pappin163 have shown that when avascular villi account for more than 2.5% of the
placental parenchyma, there are increased rates of intrauterine growth
restriction, abnormal antepartum fetal monitoring test results, oligohydramnios, and
maternal coagulopathy. Chronic villitis, membrane hemosiderin, meconium
in all three membrane layers, and chorangiosis are
also more common. The neonates also had increased immediate morbidity, including
thrombotic events. Both mother and neonate must be carefully
evaluated when extensive avascular villi are identified. Abnormal Fetoplacental Perfusion Associated With Intraplacental Vascular
Pathology If the developmental program of the conceptus is normal (normal karyotype) and
there is adequate maternal nutrient provision to the conceptus, then
an appropriate vasculature to transport those nutrients from the
placenta to the fetus proper is still required for successful completion
of pregnancy. Placental vasculogenesis and angiogenesis have been
reviewed.164 While early villous growth primarily reflects trophoblast differentiation
and proliferation, later villous development is marked by extensive
terminal villous capillary growth.165 Placental Vascular Obliterative Lesions Placental vessels can develop normally but then be removed from function
by thrombi, which can occur as a primary process or develop as a result
of other placental pathophysiology (e.g., chronic villitis). They
can also be modified by extrinsic forces (related to maternal perfusion) and
remodeled by aberrant flow patterns within the placenta itself. Lesions
that either preclude the subsequent normal development of the
placental capillary bed or remove parts of it from function would be
expected to affect umbilical artery Doppler velocimetry. Indeed, altered
placental morphology is observed in patients with abnormal umbilical
artery velocity waveforms.166,167,168,169,170,171,172 A reduced number of small muscular arteries in the fetal stem villi165–169 and more extensive ischemic pathology171 have been reported in patients with abnormal umbilical artery Doppler
velocimetry compared with normal controls. These two lesions may be causally
related; reduced uteroplacental perfusion can result in uteroplacental
ischemia and chronic uteroplacental vasoconstriction.172 However, at least one report170 identified severe chronic villitis associated with abnormal umbilical
artery Doppler, suggesting that the pathophysiology of abnormal umbilical
artery Doppler may be more complex. We recently studied 52 consecutive, nonanomalous
singletons delivered between January 1989 and June 1995. These
infants were in less than the 10th percentile for birthweight (fetal
growth retardation) and were admitted to the neonatal intensive
care unit with umbilical artery Doppler velocimetry obtained within 3 days
of delivery.173 We expected that intraplacental vaso-occlusive processes would show a
continuum from normal to a reversal of end-diastolic flow. Instead, we
identified two major subsets of fetal growth retardation by characterizing
them according to the level of intraplacental vaso-occlusive lesions. Lesions
considered to reflect intraplacental vascular pathology
included the following: - Mural or occlusive fibrin thrombi in chorionic and fetal stem vessels
- Avascular terminal villi, diagnosed when nutrient villi lacked capillaries
and had a dense paucicellular eosinophilic stroma (Fig. 21)
- Hemorrhagic endovasculitis
- Reduction or obliteration of chorionic or fetal stem vessels by mural hyperplasia, identified
by a decreased or absent lumen area and increased
wall thickness for the level of the fetal vessel (chorionic, large
fetal stem artery, small fetal stem artery)
- Mural disorganization, diagnosed when the normal concentric organization
of the arterial wall was interrupted over at least 50% of its circumference
- Abnormally thin-walled arteries, diagnosed when all vessels in a large
or small fetal stem were histologically similar, implying that the obligatory
artery in the fetal stem had veinlike characteristics
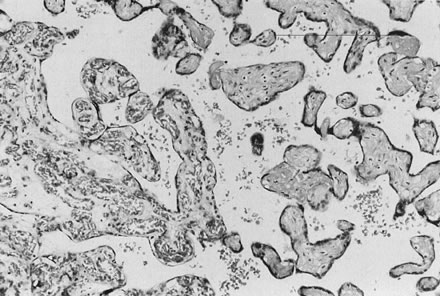 Fig. 21. Avascular villi on the right compared with the normally vascularized villi
on the left. Absence of fetal capillaries is the result of obstruction
of fetal blood flow to the region, usually caused by hemorrhagic
endovasculosis (hematoxylin and eosin, ×10). Fig. 21. Avascular villi on the right compared with the normally vascularized villi
on the left. Absence of fetal capillaries is the result of obstruction
of fetal blood flow to the region, usually caused by hemorrhagic
endovasculosis (hematoxylin and eosin, ×10).
|
Baseline levels of placental lesions were defined as the mean number of
lesions in fetal growth retardation placentas with normal umbilical artery
Doppler waveforms, and excess levels were defined as numbers of
vascular lesions that were greater than baseline. The first growth retardation
subset defined consisted of those with excess intraplacental
lesions. In these cases, there were more uteroplacental vascular lesions, more
excess lesion scores, and lighter placentas, and these features
were related to worsening umbilical artery Doppler status, consistent
with our hypothesis of a continuum of pathologic anatomy from normal
umbilical artery Doppler to reversal of end-diastolic flow. These findings
support the observations of Arabin and associates169 and Laurini and coworkers,170 who related histologic evidence of uteroplacental ischemia to abnormal
umbilical artery Doppler velocimetry. Perfusion of the placenta at abnormally
low oxygen tension is associated with increased basal perfusion
pressure, consistent with placental vasoconstriction.174 Chronic vasoconstriction (and increased intraluminal pressure) could lead
to vascular obliteration by way of progressive mural hyperplasia. However, increased
intraluminal pressure could predispose to endothelial
damage and luminal obliteration by way of hemorrhagic endovasculitis
lesions. We observed histologic changes that could support both of these
pathophysiologic mechanisms. We also observed that placentas with
intraplacental vaso-occlusive lesions were lighter, implying a diffuse
reduction in placental growth as a whole. Others have observed that
there is a global reduction or underdevelopment of villous arborization, rather
than a selective destruction of one vessel type.175,176,177 Macara and colleagues178 recently showed that terminal villi in cases of fetal growth retardation
with absent end-diastolic flow have increased syncytial nuclei, reduced
cytotrophoblast nuclei, and thickened basal lamina, with increased
stromal collagen and extracellular matrix materials. Their findings
suggest that prematurely aged syncytiotrophoblast (with increased nuclei
arranged in knots) is less rapidly replenished by cytotrophoblast (reduced
cytotrophoblast cell number and proliferation index), which led
them to conclude that there was no evidence of villous hypoxia per se but rather a capillary congestion that supports the hypothesis of a primary
vasomotor pathology in absent end-diastolic flow. The timely observations
of Burton and Jauniaux179 led them to conclude that umbilical vascular resistance may be heavily
influenced by the number of parallel circulatory units offered by the
placental lobules. In a study of the placental villous response to hypoxia
caused by high altitude, maternal iron-deficiency anemia, and preeclampsia, they
observed a trend for increased capillary volume, apparently
due to capillary dilatation with accompanying thinning of the vasculosyncytial
membrane barrier.180 The second group of growth retardation was defined as those cases with
baseline intraplacental vaso-occlusive lesions. Among these, an increased
chronic inflammation score was related to worsening umbilical artery
Doppler status, despite no recognizable increase in intraplacental
vaso-occlusive lesions. Chronic inflammation-associated abnormal umbilical
artery Doppler waveforms may represent the effects of abnormal vascular
function rather than fixed anatomic lesions. Cytokines and prostanoids (known
to affect placental vasomotor tone)181 may be generated in the process of chronic inflammation and cause abnormal
placental resistance in the absence of anatomic lesions. This distinction
could be clinically relevant because malfunction of an anatomically
normal placenta may be amenable to in utero therapy (e.g., transplacental administration of anti-inflammatory agents). It may be too optimistic to expect the range of umbilical artery Doppler
from normal to reversal of end-diastolic flow to show a pathologic continuum, but
it is reasonable to expect absent and reversed end-diastolic
flow to have a close resemblance. Although our findings may show
the effects of small sample size, reversed end-diastolic flow cases were
striking for the relative paucity of vaso-occlusive lesions and for
a significant increase in structures best described as veinlike fetal
stem arteries. One explanation may be that some cases of reversed end-diastolic
flow represent unique pathophysiologic processes. Alternatively, a
pattern of degenerative and/or dysplastic aortic architecture
that effectively reverses the normal differentiation pattern of aortic
wall development has been reported in cases of twin-reversed arterial
perfusion.182 This maldevelopment has been suggested to reflect the effects of abnormal
levels and gradients of intraluminal pressure. Whether the embryonal
vessels forming within the primitive chorionic membrane of the placenta
will become arteries or veins is believed to be “determined
by the direction of flow and its pressure.”183 Thus, our observations suggest that in fetal growth retardation with reversed
end-diastolic flow, the placental arterial wall structure may
be modified (and lesions obscured) by the reversed blood flow. The placental
vasculature is a dynamic structure, the development and expansion
of which continue throughout most of gestation. The potential for mechanical
factors (e.g., intraluminal pressure and flow direction) to
remodel and in effect deform the evolving placental vasculature cannot
be underestimated. Karimu and Burton184,185 have confirmed the mechanical effect of intraplacental perfusion pressure
on villous capillary growth. They noted that more proliferating endothelial
nuclei were found at 100 mm Hg than at 40 mm Hg. This suggests
that mechanical factors may play a role in villous angiogenesis and
the formation of terminal villi. They have also shown similar effects
of maternal perfusion on the placental capillary bed. The study showed
that placental capillaries are elastic and deformable, and also that
if a sufficiently high pressure is generated in the intervillous space, they
may be compressed. They suggested that this may also provide an
explanation for the observed increase in umbilical vascular resistance
shown by Doppler studies in pregnant women scanned in the supine position. Vascular Proliferative Lesions Abnormally proliferative placental vascular development is commonly related
to maternal diseases involving overgrowth of the placenta as a whole (Fig. 22). Cardinal among these conditions is maternal diabetes mellitus. In maternal
diabetes mellitus, abnormal growth factor expression and effect
may be associated with overgrowth of the placental vessels, as well as
with a wide range of direct fetal effects, including macrosomia, polycythemia, and
late fetal death.186 Vascular proliferation can be seen in other conditions, including fetal
growth retardation187 and twin gestations. In the latter case, two fetuses are competing for
a single volume of uterine blood flow. In maternal diabetes mellitus, fetal
growth retardation, and multiple gestation, capillary proliferation
may be an endothelial response to decreased oxygen availability. Capillary
proliferation may increase placental microvascular resistance
and the diffusion distance of nutrient across the placental membrane, and
it may increase fetoplacental intravascular volume and fetal cardiac
work. We have repeatedly observed in our clinical practice that umbilical
systolic/diastolic ratios generally remain within normal limits
even when there is a massive increase in villous capillary number, as
long as other placental lesions that obliterate the placental vasculature
are not present. Chorangioma, a focal placental vascular tumor
or malformation, occurs singly or multifocally and may function as an
arteriovenous malformation or vascular shunt (Fig. 23). This lesion may present with stillbirth, hydrops fetalis due to congestive
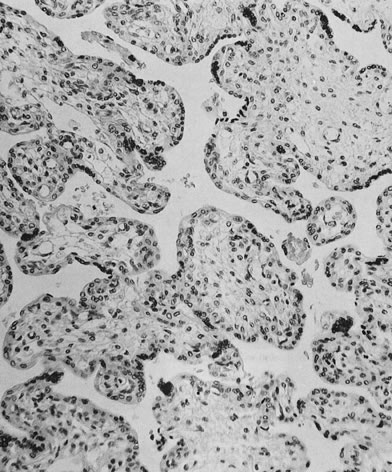
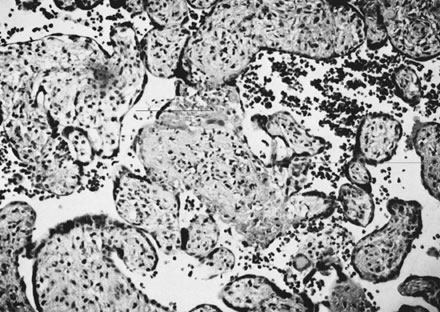
heart failure, or unexplained fetal tachycardia. 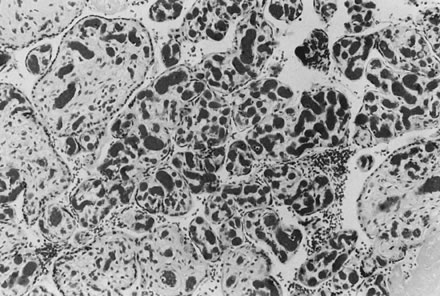 Fig. 22. Marked proliferation of capillaries in fetal terminal villi, chorangiosis, is
thought to be due to prolonged abnormalities in uteroplacental
blood flow (hematoxylin and eosin, ×10). Fig. 22. Marked proliferation of capillaries in fetal terminal villi, chorangiosis, is
thought to be due to prolonged abnormalities in uteroplacental
blood flow (hematoxylin and eosin, ×10).
|
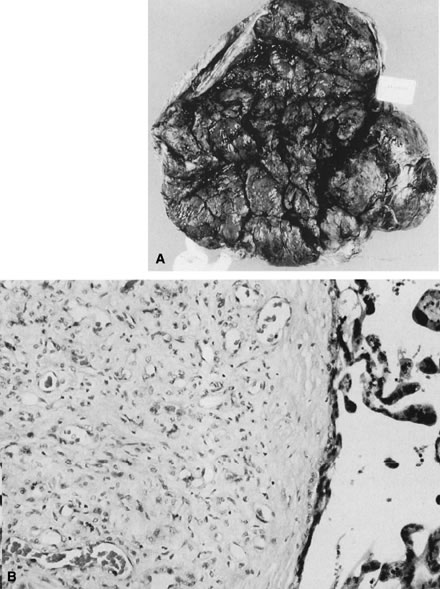 Fig. 23. A. Chorangioma, seen at the right of the photograph, is fed by a large chorionic
plate vessel, in this case a vein. This blood supply may become
thrombosed, or torsion may occur, resulting in infarction. B. Small, closely packed vessels frequently make it difficult to identify
the vascular nature of this lesion (hematoxylin and eosin, ×10). Fig. 23. A. Chorangioma, seen at the right of the photograph, is fed by a large chorionic
plate vessel, in this case a vein. This blood supply may become
thrombosed, or torsion may occur, resulting in infarction. B. Small, closely packed vessels frequently make it difficult to identify
the vascular nature of this lesion (hematoxylin and eosin, ×10).
|
Pattern Recognition in Placental Histopathology: Clinical Applications
in Prematurity A comprehensive prospective data collection on all pregnancies delivered
at a university health center formed the basis of a data set of extreme
prematurity (deliveries at less than 32 weeks' gestation). As extensively
reported elsewhere,14,73,79 the study population excluded clinical factors that could be considered
predisposing to preterm birth (e.g., multiple gestation, fetal congenital
anomaly, maternal diabetes mellitus, and isolated chronic hypertension), conditions
resulting in elective preterm birth (e.g., placenta
previa or isolated fetal growth restriction with fetal distress), and
cases in which there was a discrepancy between the obstetric and neonatal
assessments of gestational age. For 432 pregnancies, preterm birth
could be attributed to one of three principal indications for delivery: premature
membrane rupture, premature labor with intact labor, or
preeclampsia. Placental histopathologic features were scored semiquantitatively
in five primary pathophysiology categories: acute inflammation, chronic
inflammation, uteroplacental vascular pathology and related
villous lesions, intraplacental vascular pathology, and coagulation-related
pathology. To this data set we applied factor analysis (a statistical
technique that extracts complex patterns of correlations within
a data set and generates sets of equations that identify interrelations
among lesions) and weighted the contribution of the particular lesion
to the overall pattern. Essentially, a factor represents a group
of variables that are correlated with each other but are relatively uncorrelated
with other variables. Our goal in using factor analysis was
to reduce the complexity of analyses required in univariate studies of
large numbers of specific lesions. We accomplished this by analyzing
instead a smaller number of factors and weighting the different lesions
within them. The results of factor analysis of placental histopathologic lesions in
preterm preeclampsia have been published.72 Factor analysis generated 15 lesion categories in preterm labor and 15 in
premature rupture of membranes that combined different lesions with
different strengths of associations (coefficients) among lesions (Tables 2 and 3). Our goal was to identify patterns that combined several lesions into
a single numeric score that would better relate to outcome than any one
lesion in isolation. To test this, we examined the relation between
the factors and gestational age at delivery. Histopathologic factors
generated regression equations highly predictive of gestational age at
delivery in both premature membrane rupture (R2 = 0.18) and preterm labor (R2 = 0.28). At least 66% of cases had high scores for more than one factor. We
needed to determine whether the placental factors showed additive
or synergistic effects on gestational age. For each case, the scores
for the categories related to gestational age were summed and plotted
against gestational age (Fig. 24). These data suggest that preterm labor or premature rupture of membranes
is infrequently due to a single problem; it more often is the result
of the application of different “straws” that culminate
in “breaking the camel's back” (premature membrane rupture
or preterm labor).
Table 2. Premature Membrane Rupture Factor Analysis
Click here to view Table
2.
Table 3. Preterm Labor Factor Analysis
Click here to view Table
3.
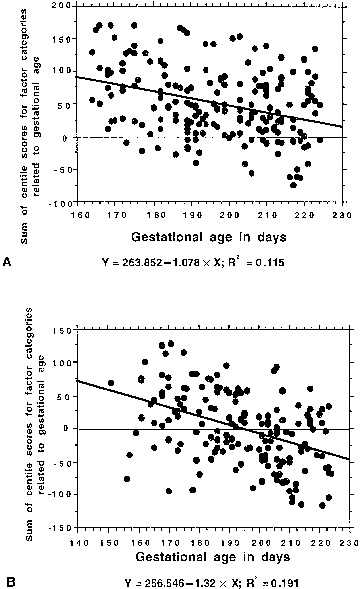 Fig. 24. Values greater than 0 indicate that factors related to longer gestational
length predominate. Values less than 0 indicate that factors related
to shorter gestational length predominate. A. Relationship of summation of factor centiles to gestational age in premature
membrane rupture. B. Relationship of summation of factor centiles to gestational age in preterm
labor. Fig. 24. Values greater than 0 indicate that factors related to longer gestational
length predominate. Values less than 0 indicate that factors related
to shorter gestational length predominate. A. Relationship of summation of factor centiles to gestational age in premature
membrane rupture. B. Relationship of summation of factor centiles to gestational age in preterm
labor.
|
The implications of this thesis for current investigations of methods to
reduce prematurity (e.g., by antibiotic therapy) are significant. Our
analysis indicates that a decrease in amnion inflammation from grade 4 to
grade 3 is associated with 1 more day of gestation, whereas a similar
unit increase in severity of uteroplacental vascular lesions is
associated with 4 fewer days of gestation. The underlying pathophysiology
of prematurity in any population and the interplay of the various
factors that predispose to prematurity must be carefully taken into consideration
in the interpretation of analyses. The factor categories for premature membrane rupture and preterm labor
appear to uniquely characterize these two processes. However, if this
is true, the factors and their associations with clinical features should
also be unique in the two different outcomes. For both premature membrane
rupture and preterm labor, a factor category was generated that
primarily represented the effects of histologic markers of acute ascending
infection (factor 1; see Tables 2 and 3), with very similar coefficients. However, when the premature membrane
rupture factor of acute inflammation was used to “predict” gestational
age in the preterm labor group, it was only 26% as strong
a predictor of gestational age as the similar equation generated in
the preterm labor data set itself. Although the factors appear to be quite
similar, they are in fact quite different and have different associations
with such basic clinical concerns as gestational length. We have applied factor analysis to a series of cases involving infants
delivered to women with antiphospholipid antibodies, systemic lupus erythematosus, or
both.57 We have also identified patterns of lesions that may contribute to an
understanding of the mechanisms of placental damage in these conditions
and the differences in patient responses to therapeutic interventions. In summary, we use a diagnostic approach to placental pathology that is
directed toward identifying the underlying pathophysiologic processes (acute
inflammation, chronic inflammation, uteroplacental vascular pathology
and related villous lesions, intraplacental vascular pathology, and
coagulation-related pathology) and the target or targets of tissue
injury. The processes and targets revealed can then be related to the
clinically manifest pathophysiology of mother and fetus/neonate. This
provides, in our opinion, the best mechanism for clinically relevant
clinicopathologic correlation. This can also be used in the generation
and testing of new hypotheses regarding relations among placental anatomic
changes, placental functional disturbances, and maternal and fetal/neonatal
outcome. |