This chapter should be cited as follows:
Muzny CA, Stockdale CK, et al., Glob Libr Women's Med
ISSN: 1756-2228; DOI 10.3843/GLOWM.419923
The Continuous Textbook of Women’s Medicine Series – Gynecology Module
Volume 12
Infections in gynecology
Volume Editors:
Professor Francesco De Seta, Department of Medical, Surgical and Health Sciences, Institute for Maternal and Child Health, University of Trieste, IRCCS Burlo Garofolo, Trieste, Italy
Dr Pedro Vieira Baptista, Lower Genital Tract Unit, Centro Hospitalar de São João and Department of Gynecology-Obstetrics and Pediatrics, Faculdade de Medicina da Universidade do Porto, Portugal

Chapter
Trichomoniasis
First published: August 2023
Study Assessment Option
By answering four multiple-choice questions (randomly selected) after studying this chapter, readers can qualify for Continuing Professional Development points plus a Study Completion Certificate from GLOWM.
See end of chapter for details.
INTRODUCTION
Trichomonas vaginalis is estimated to be the most common, curable non-viral sexually transmitted infection (STI) among women worldwide.1 It is associated with adverse birth outcomes,2 increased risk of acquisition and transmission of human immunodeficiency virus (HIV) and other STIs,3,4,5 pelvic inflammatory disease (PID),6,7 infertility,8,9 and cervical cancer.10 African Americans are significantly more likely to be T. vaginalis-infected, signifying a predominant health disparity.11 Beyond screening recommendations for HIV-infected women at entry to care and annually thereafter,12 there are no screening, surveillance, or control programs for T. vaginalis. Thus, it is frequently considered a neglected STI.13 This chapter reviews the etiology, pathophysiology, epidemiology, clinical manifestations, diagnosis, and treatment (including partner management) of T. vaginalis infection, focusing on cisgender women.
ETIOLOGY AND PATHOPHYSIOLOGY
T. vaginalis is a parasitic pathogen primarily infecting squamous epithelial cells of the human genital tract, causing damage to these cells. It infects the female lower genital tract (vagina, urethra, and endocervix) and the male urethra and prostate. Humans are its only known natural host.14,15 While transmission by fomites has been occasionally reported,16,17,18,19 transmission mainly occurs during sex.20 There have been conflicting data on the association between personal hygiene and T. vaginalis infection,21,22 but in general, it is not thought to be a major factor. The incubation period of T. vaginalis is 4–28 days.23 It does not have a cyst form and does not survive well in the environment, but has been identified outside the human body in warm and wet locations (i.e. moist towels) for >3 hours.16 It has its own microbiota, harboring two Mycoplasma species and a double-stranded RNA virus, T. vaginalis virus (TVV), which can contribute to its pathogenesis.24,25 Of the four known TVV viruses, TVV1 and TVV2 have been linked to severity of genital symptoms26 and TVV2 and TVV3 to surface expression of an immunogenic protein, P270, which is associated with cytotoxicity, cytoadherence, and host immune evasion.27 The role of TVV4 is not elucidated. However, in a study of 355 US clinical T. vaginalis isolates, 40% of which were positive for TVV, there was no association between TVV positivity and genital symptoms, repeat infection, or metronidazole (MTZ) resistance, suggesting that TVV may be a commensal to T. vaginalis.28
Infection is more common in women perhaps due to the anatomy of the female genital tract.29,30,31,32,33 Other possibilities could be due to spontaneous resolution in men (which may occur in some cases)34,35 or less effective testing among asymptomatic men.11,31,36,37 More T. vaginalis research has been done in symptomatic women, thus, its natural history in women has not been fully elucidated. However, several studies have found that, even after several months without treatment, most women still test positive.38,39 A potential long natural history coupled with no screening recommendations (outside of HIV-infected women) may explain the finding that T. vaginalis is more prevalent in older women.11 In contrast to women, persistence of T. vaginalis in men can be shorter (i.e. <1 month in some cases).34 The greater likelihood of persistence in women is linked to greater availability of iron, an essential nutrient.15,33,40,41,42,43 Menstrual blood creates a rich growth medium which promotes attachment and growth of the parasite in the vaginal canal.29,30
PREVALENCE AND EPIDEMIOLOGY
While not reportable, global estimates indicate that there are 156 million new cases of T. vaginalis infection among women and men annually.1 The global prevalence of T. vaginalis among women (5.3%) is higher than that of chlamydia (3.8%), gonorrhea (0.9%), and syphilis (0.5%) combined.1 In the US, the prevalence of T. vaginalis by urine nucleic acid amplification (NAAT) testing in a recent population-based study was 1.8% in women and 0.5% in men.11 African Americans had a 4-fold higher prevalence than other groups, constituting a dramatic health disparity.11 Unlike many STIs, T. vaginalis prevalence can be higher among older persons >45 years of age with rates ranging from 0.2% to 21.4%.44 Population-based studies have also found that T. vaginalis rates are highest among those ≥25 years.45 The prevalence of urethral infection in men who have sex with men (MSM), while low to non-existent in the US,46 has been found to be 7% in South African MSM.47 Although detection of T. vaginalis DNA occasionally occurs in extra-genital (oral, rectal) locations,48,49 screening or diagnostic testing of these regions is not recommended.
RISK FACTORS
While present in all races,11 T. vaginalis infection is more common in African American women engaging in high-risk sexual behaviors. This includes multiple sex partners,50,51 inconsistent condom use, illicit drug use during sex,50 sex with partners using illicit drugs,31,50,52,53 and transactional sex.54,55,56,57 Other risk factors include early coitarche,58 older age,11,31,52,59 history of incarceration,60,61 having less than a high school education,11 and living below the national poverty level.12,51
Women with bacterial vaginosis (BV) are at higher risk for T. vaginalis infection.12,20,62 While vaginal dysbiosis has been associated with increased pathogenicity of T. vaginalis,63 it is not clear if the presence of BV interferes with T. vaginalis treatment. In two randomized controlled trials, BV was found to increase metrodinazole (MTZ) treatment failure in HIV-infected women64 but not in HIV-uninfected women.65 This difference may be due to impaired immunity in HIV-infected women,64 altered pharmacokinetics and pharmacodynamics of MTZ,66 or inadequate power in the studies.65 Additionally, women with HIV are at higher risk for T. vaginalis.54,67 Several studies have also shown that women who have sex with women and men (WSWM) are at higher risk for T. vaginalis than women who have sex with women (WSW) and women who have sex with men (WSM).31,68
COMPLICATIONS
Complications associated with T. vaginalis infection among women include adverse birth outcomes, risk of HIV and other STIs, PID, infertility, cervical infection, and cervical cancer.
Adverse Birth Outcomes
In a meta-analysis of 19 studies, significant associations were found between T. vaginalis and preterm delivery (PTD), premature rupture of membranes (PROM), and low birth weight.2 However, the physiological mechanisms linking T. vaginalis and adverse birth outcomes are not well understood. One hypothesis is that PTD and PROM in T. vaginalis-infected pregnant women are related to maternal innate immune inflammatory responses to the parasite, which involve elevated cervical interleukin-8 (IL-8) and vaginal defensin levels.8,69 These cytokines are markers of neutrophil activation, which has been associated with PTD and other adverse birth events. For example, cervical IL-8 is thought to trigger cervical ripening and dilatation.70,71 Additionally, one study has shown an association between maternal T. vaginalis infection and intellectual disability in children born to infected mothers.72
Risk of HIV and other STIs
A meta-analysis of 19 studies found that persons infected with T. vaginalis were 1.5 times more likely to acquire HIV than non-infected individuals (95% CI 1.3–1.7; p <0.001).73 In another meta-analysis of 32 studies reporting k = 97 effect size, estimates of HIV acquisition risk due to non-viral STI infections among high-risk heterosexuals with chlamydia, gonorrhea, syphilis, Mycoplasma genitalium, and/or T. vaginalis, HIV acquisition risk was statistically significant for T. vaginalis-infected women (RR 1.54; 95% CI 1.31–1.82; k = 17).74 The greater susceptibility to HIV among T. vaginalis-infected individuals is plausible for several reasons: (1) T. vaginalis damages host epithelial cell membranes which act as a structural barrier to HIV, (2) the host immune response to T. vaginalis stimulates an increased number of HIV target cells in the genital tract mucosa, and (3) T. vaginalis alters the normal vaginal microbiota, rendering it more permissive to the development of BV, which, in turn, increases HIV acquisition risk.73
There is less direct evidence suggesting that HIV-infected individuals with T. vaginalis are more likely to transmit HIV. A review paper found that only 7 of 14 studies demonstrated a higher likelihood of HIV shedding in the genital tract of T. vaginalis-coinfected individuals compared to HIV-infected individuals without coinfection.75 In other studies, vaginal shedding of HIV-1 RNA was decreased after T. vaginalis treatment in women from Kenya76 and New Orleans, LA.77 However, in the Kenyan cohort, the prevalence of vaginal HIV-1 DNA remained unchanged despite T. vaginalis treatment.
Concomitant infection with T. vaginalis has been associated with a higher incidence of genital HSV-2 infection78 as well as genital HSV-2 shedding.79 It has also been associated with the presence of other STIs including chlamydia, gonorrhea, and human papillomavirus (HPV).4,80
Pelvic Inflammatory Disease
T. vaginalis is not traditionally considered an STI associated with pelvic inflammatory disease (PID). However, in an older study of 119 South African women, those infected with T. vaginalis had a significantly higher risk of PID than those without (p = 0.03).6 When these women were stratified according to their HIV status, the risk of PID in HIV-infected women with T. vaginalis increased significantly (p = 0.002); no association was found in women without HIV.6 More recently, among 647 women in the PID Evaluation and Clinical Health (PEACH) study, T. vaginalis was isolated from the vagina in 12.8% of women and the odds of having endometritis at baseline was twice as high among T. vaginalis-infected women with compared to women without T. vaginalis infection (adjusted OR (AOR): 1.9, 95% CI 1.0–3.3). Infertility and recurrent PID were also more common among T. vaginalis-infected women.7
Infertility
A meta-analysis of eight studies found that T. vaginalis was associated with a 1.7 times greater risk of infertility in women (95% CI 1.25–2.31).81 Similarly, a meta-analysis of five studies found that T. vaginalis was associated with a 1.91 times greater risk of infertility in men (95% CI 1.02–3.58).81 This is thought to be due to inflammatory damage of female reproductive organs and changes in the vaginal environment resulting in decrease or loss of reproductive function in women.81 In men, T. vaginalis itself or the induced inflammatory response can impair sperm cells, causing a decrease in cell viability or death, resulting in a decrease or loss of reproductive function.81
Cervical Infection and Cervical Cancer
While thought to be a vaginal infection, there is some evidence to suggest that T. vaginalis causes cervical involvement. A small study of 64 symptomatic female STI clinic attendees presenting with cervico-vaginal discharge found that 6.4% of cervical infections were attributable to T. vaginalis.82 Regarding the risk of cervical cancer, a study of Tanzanian women found that those infected with T. vaginalis were 6.5 times more likely to have high-risk HPV, suggesting an indirect link between T. vaginalis and cervical neoplasia.4 In addition, a meta-analysis of 17 studies found that T. vaginalis-infected women had a higher risk of cervical neoplasia (OR 2.06, 95% CI: 1.77–2.39), with HPV co-infection playing an important role.10
SIGNS AND SYMPTOMS
The “classic” symptoms of T. vaginalis infection include vaginal odor and a yellow-green, frothy, malodorous vaginal discharge.12,29,52,83,84 However, a large number of infected women have minimal or no symptoms (83–89%).85 Half of infected asymptomatic women, however, may become symptomatic within 6 months.14 Infected women can also develop cyclic symptoms that are worse during menstruation.29 Symptomatic women may note additional symptoms including genital pruritis, dysuria, and dyspareunia.
On exam, signs may include vaginal erythema, malodorous, frothy, vaginal discharge, colpitis macularis or “strawberry cervix” (present in <5% of women;86,87 rises to nearly 50% with colposcopy)87, and elevated vaginal pH >4.5.20 However, infection may also be present in the setting of a normal vaginal pH.20
DIAGNOSIS
T. vaginalis is traditionally diagnosed at the point-of-care (POC) by wet mount microscopy of vaginal secretions for motile trichomonads (sensitivity 44–68%; specificity 100%)88 (Figure 1). This test must be performed within 10–20 minutes or the trichomonads will lose their viability, increasing the likelihood of a false negative test. The OSOM® rapid test (Sekisui Diagnostics, California) is another POC test (results ≤10 minutes) that uses antibodies to detect T. vaginalis protein antigens in vaginal secretions (sensitivity 82–95%; specificity 97–100%, compared to wet mount and culture). It is a qualitative test that should be used in symptomatic women or contacts to T. vaginalis.88 When present, T. vaginalis antigens bind antibodies resulting in the formation of a blue line on the test strip. This test does not require microscopy however is more expensive than wet mount.
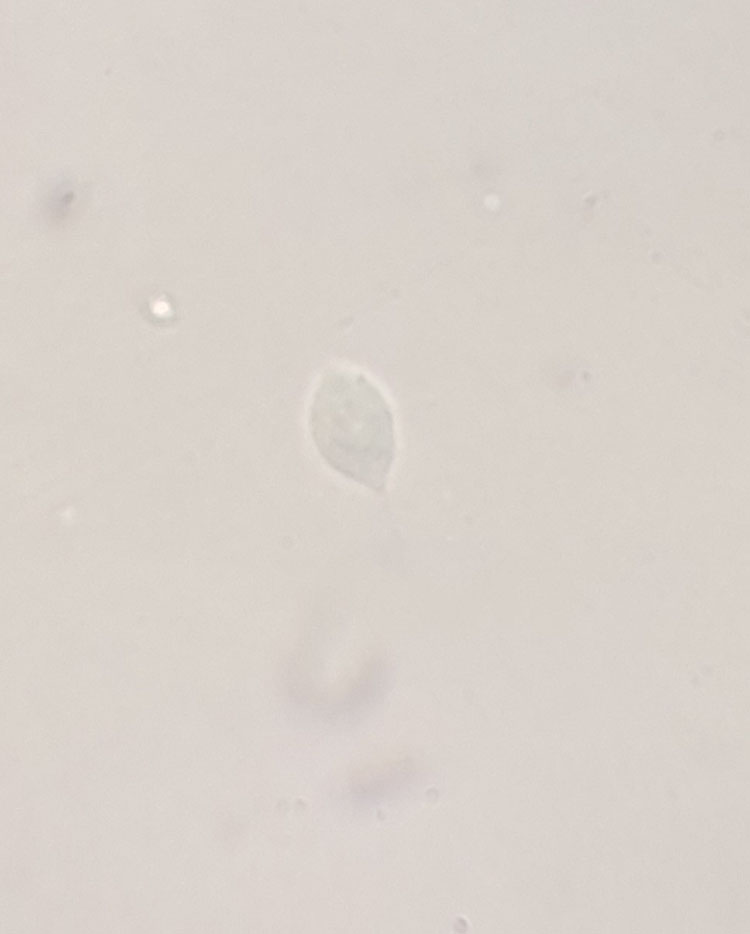
1
Wet mount showing a motile trichomonad. (Photo courtesy of Keonte Graves, MS.)
An additional POC sero-diagnostic test is currently in development (MedMira Rapid Vertical Flow (RVF®) Technology).89 This novel test detects α-actinin blood antibodies, a highly immunogenic T. vaginalis protein not found among other microorganisms that has no identity with the human homolog, in serum or whole blood specimens.89 The MedMira Rapid Vertical Flow test cartridge has a membrane including a vertical procedural/reagent control line and a spot that becomes readily visible within 5 minutes if there is a positive reaction with α-actinin antibody present in serum or whole blood specimens. One area of future research associated with this test is the need to address the temporal nature and duration of serum anti-T. vaginalis antibody response after infection and cure, including the possibility of long-lasting serum antibody, which may not be indicative of active infection.
Trichomonas culture (InPouch® system [BioMed Diagnostics, White City, OR]) has previously been the gold standard for diagnosis (sensitivity 44–81%; specificity 100%).88,90 Specimens from women (vaginal swabs) or men (urethral swabs, urine sediment, and/or semen; multiple specimens recommended to increase yield) should be used to inoculate the culture medium <1 hour after collection.88 However, this test is categorized by the Clinical Laboratory Improvement Amendments (CLIA) as moderately complex, as it requires incubation at 37°C and reading over multiple days.91
The availability of highly sensitive and specific T. vaginalis molecular diagnostic assays has grown rapidly. These assays can be further divided into molecular amplified assays (i.e. AmpliVue and Solana assays),92,93 instrument-based assays (i.e. Hologic Aptima T. vaginalis nucleic acid amplification test [NAAT], Becton Dickinson [BD] ProbeTec Qx T. vaginalis NAAT, BD Max CT/GC/TV2 NAAT, Cepheid GeneXpert T. vaginalis NAAT, Roche Cobas MG/TV NAAT, and the Abbott Alinity m STI assay (including T. vaginalis NAAT testing),94,95,96,97,98 and instrument-free assays (i.e. Visby GC/CT/TV NAAT testing device).99 These assays, with their respective sensitivities and specificities, specimen types in women, complexity, and time to results, are detailed in Table 1. Several of these assays can provide testing results within 1 hour or less (i.e. AmpliVue [45–50 minutes]; Solana [<40 minutes]; Cepheid GeneXpert [40–63 minutes], and Visby [25 minutes]).
1
T. vaginalis diagnostic tests in women. Reproduced, with permission, from the International Society for the Study of Vulvovaginal Disease.100Test | Sample | Sensitivity/specificity for T. vaginalis | Complexity/time |
Wet mount microscopy88 | Vaginal specimens | Sensitivity: 44–68%; Specificity: 100% | CLIA waived; POC test (results in ≤10 minutes) |
OSOM®88 | Vaginal specimens | Sensitivity: 83–92%; Specificity: 99–100% | CLIA waived; POC test (results in ≤10 minutes) |
BD Affirm VPIII88 | Vaginal specimens | Sensitivity: 91–100%; Specificity: 93–96% | Moderate complexity Results <1 hour |
Vaginal specimens | Sensitivity: 44–81%; Specificity: 100% | Moderate complexity. Requires incubation at 37°C; should be read for 5 days over a 7 day period91 | |
AmpliVue92 | Vaginal specimens from symptomatic and asymptomatic women | Sensitivity 90.7%; | Results in 45–50 minutes |
Solana93 | Vaginal specimens from symptomatic and asymptomatic women; urine specimens | Sensitivity/Specificity 98.6–100%/98.5–98.9% for vaginal specimens and 92.9–98%/97.9–98.4% for urine specimens | Results in <40 minutes |
Hologic Aptima T. vaginalis NAAT94 | Vaginal, endocervical, ThinPrep Pap, and urine specimens from symptomatic and asymptomatic women | Sensitivity: 95.2–100%; Specificity: 98.9–99.6% | High complexity. Requires Panther, Viper, or Tigris system. Results in <8 hours |
BD ProbeTec Qx T. vaginalis NAAT95 | Vaginal, endocervical, and urine specimens from symptomatic and asymptomatic women | Sensitivity: 98%-100%; Specificity: 98%-100% | High complexity. Requires Viper system. Results in <8 hours |
BD Max CT/GC/TV2 NAAT98 | Vaginal, endocervical, and urine specimens from symptomatic and asymptomatic women | Sensitivity: 86.6%-100%; Specificity: 99.2%-99.8% | High complexity. |
Cepheid GeneXpert96 | Self-collected vaginal, clinician-collected endocervical, and urine specimens from symptomatic and asymptomatic women | Sensitivity: 99.5%-100%; Specificity: 99.4–99.9% | Moderate complexity. Results in 40–63 minutes |
Roche Cobas MG/TV NAAT97 | Vaginal and endocervical specimens from symptomatic and asymptomatic women | Sensitivity: 96.4–100%; Specificity: 96.5–98.8% | High complexity. For use on Cobas 6800/8800 systems |
Abbott Alinity m STI assay* | Vaginal, endocervical, ThinPrep Pap, and urine specimens from symptomatic and asymptomatic women | Sensitivity, Specificity not yet published; refer to Abbott Molecular website* | Results in <115 minutes |
Visby GC/CT/TV NAAT Testing Device99 | Self-collected vaginal specimens | Sensitivity: 99.2%; Specificity 96.9% | CLIA waived; POC test (results in 25 minutes) |
CLIA, Clinical Laboratory Improvement Amendments; POC, point-of-care; BD, Becton Dickinson; STI, sexually transmitted infection; NAAT, nucleic acid amplification test; MG, Mycoplasma genitalium; GC, Neisseria gonorrhoeae; CT, Chlamydia trachomatis; TV, Trichomonas vaginalis
*FDA-approved 5/4/22; https://www.molecular.abbott/int/en/products/infectious-disease/alinity-m-sti-assay.
TREATMENT AND FOLLOW-UP
5-nitroimidazoles (MTZ, tinidazole [TDZ], and secnidazole [SEC]) are the primary class of drugs used for T. vaginalis treatment. For decades, the CDC and World Health Organization have recommended single dose 2-g oral MTZ as the preferred treatment for T. vaginalis, with oral MTZ 400–500 mg twice daily for 7 days or single-dose 2-g oral TDZ as alternative therapies. The recommended treatment was changed to the 7-day oral MTZ dose for HIV-infected women more than a decade ago in response to a multi-center randomized controlled trials demonstrating superiority of the 7-day oral MTZ dose over singe-dose.64 A subsequent meta-analysis101 and multi-center trials65 more recently found similar results in HIV-uninfected women. In vivo pharmacokinetic and pharmacodynamic effects of MTZ may be playing a role in single-dose oral MTZ treatment failure, necessitating a longer treatment regimen in women.66 Two hypotheses for this finding are (1) competition for oral MTZ by BV-associated bacteria in the vaginal microbiota of T. vaginalis-infected women and (2) inadequate accumulation of the active metabolites of MTZ when only a single, oral dose is given.66 Thus, the 7-day oral MTZ regimen is currently the recommended treatment regimen for all women with single dose 2-g oral TDZ remaining as an alternative.12,102 Given the lack of a comparable randomized controlled trials in men, single dose 2-g oral MTZ remains the recommended therapy for men with single dose 2-g oral TDZ as an alternate.12 While it is never recommended to drink alcohol with any medication, it is now understood that moderate alcohol consumption should not be a barrier to oral metronidazole use during the treatment of T. vaginalis.
If a woman is still infected with T. vaginalis after multi-dose oral MTZ and has been re-exposed to an untreated sexual partner, she should be re-dosed with the same treatment regimen. If she has not been re-exposed, she should be re-treated with either 2-g of oral MTZ or TDZ daily for 7 days.12 If a male is still infected with T. vaginalis after treatment with single dose 2-g oral MTZ and has been re-exposed to an untreated sexual partner, he should be re-dosed with single dose 2-g oral MTZ. If he has not been re-exposed, he should be given a course of oral MTZ 500 mg twice daily for 7 days.12
Most recently, a randomized, double-blind, placebo-controlled, delayed-treatment study evaluating the efficacy and safety of a single 2-g dose of oral SEC, a second generation 5-nitroimidazole with a longer half-life (17–19 hours), in 147 women with trichomoniasis was conducted.103 At the test-of-cure (TOC) visit 6−12 days after randomization, the microbiologic cure rate was 92.2% (95% CI: 82.7–97.4) in the SEC group and 1.5% (95% CI: 0.0–8.0) in the placebo group (p <0.001).103 For women who received placebo at baseline, the opposite treatment was given at TOC to ensure all participants were treated per standard of care. SEC was well tolerated in this study. The most frequent adverse events were vulvovaginal candidiasis and nausea (2.7% each). SEC has since been FDA approved for T. vaginalis treatment in adolescent and adult women and men ≥12 years. It is also FDA approved for BV treatment in women12 and is the only single-dose oral regimen available for both vaginal infections.104
Re-testing for T. vaginalis is recommended, preferably by NAAT, for all sexually active women between 3 weeks and 3 months after the end of treatment regardless of whether or not their sexual partner(s) were treated.12 The optimal time for repeat T. vaginalis NAAT testing after completion of multi-dose oral MTZ was ≥3 weeks in a recent study;105 repeat NAAT testing before this time carries the risk of detecting remnant T. vaginalis nucleic acid that can still exist. If re-testing by 3 months is not possible, women should be re-tested whenever they next seek medical care.12
MANAGEMENT OF T. VAGINALIS IN SPECIAL SITUATIONS
Infants
T. vaginalis has been noted to be transmitted perinatally in case reports,106 although it is rare. In female newborns, T. vaginalis acquisition during birth may cause vaginal discharge during the first week of life.107 Respiratory infection in newborns is also possible.108
Pregnant and Lactating Women
Several meta-analyses have found MTZ to be safe in all stages of pregnancy109,110 and its use in pregnancy is supported by current national guidelines.12 TDZ use should be avoided in pregnancy based on preclinical data suggesting it poses a moderate risk.12 Limited data are available on the use of SEC in pregnancy; however, there is no evidence of adverse developmental outcomes in animal studies.104
In lactating women administered MTZ, withholding breastfeeding during treatment and for 12–24 hours after the last dose will reduce the exposure of the infant to MTZ. For women treated with TDZ, interruption of breastfeeding is recommended during treatment and for 3 days after the last dose.111
5-nitroimidazole Hypersensitivity
The most common reactions associated with 5-nitroimidazoles (primarily MTZ) are immediate, type I IgE-mediated hypersensitivity reactions, occurring with 1–2 hours of drug exposure. This includes urticaria and hives with potential life-threatening manifestations such as angioedema, bronchospasm, and anaphylaxis.112 Type II and IV hypersensitivity reactions have been less commonly described.113,114,115,116 The prevalence of MTZ hypersensitivity was found to be ~0.15% in a study of 2,375,424 Kaiser Permanente health plan members.117
Although uncommon, treatment for T. vaginalis-infected patients with a true history of 5-nitroimidazole hypersensitivity is challenging.118 If a prior IgE-mediated hypersensitivity reaction has been confirmed based on history and/or drug provocation testing,118 desensitization to the 5-nitroimidazole is the first line of treatment.12 Oral113,119 and intravenous120 protocols for desensitization have been published, primarily involving MTZ. Intensive monitoring is required during the desensitization process due to the need for frequent drug administration and monitoring for reactions; thus, it should be performed in the inpatient setting.118 After completion of a desensitization protocol, patients are able to safely take oral MTZ for 4–5 half-lives of the drug (half-life = 7–8 hours), approximately 2 days.117 If the drug is not continued at regular intervals after completion of the desensitization protocol, it will need to be restarted from the beginning to avoid breakthrough reactions.117
For patients in which desensitization is not an option, use of other 5-nitroimidazoles such as TDZ or SEC is not recommended because of the risk of cross-reactivity115 and alternative treatment options outside of the 5-nitroimidazoles should be used.12,118 Use of these alternative treatments is anecdotal, limited to vaginal formulations (the majority of which have to be compounded), and may not reach all sites infected with T. vaginalis (i.e. Bartholin’s and Skene’s glands).12,118 One option is a prolonged course of vaginal boric acid 600 mg twice daily for 60 days, either alone121,122 or in combination with vaginal clotrimazole.123 Another option is vaginal paromomycin 6.25% cream daily for 8–14 days.124,125,126 However, topical use of this medication can result in painful vulvar ulcers that are self-limited and resolve once treatment is discontinued. Lubricating jelly applied to the vulva before use has been successful in preventing the development of these ulcers in some women.126
Persistent Infection
For those experiencing persistent infection not due to sexual re-exposure, a trichomonas culture kit from the Centers for Disease Control and Prevention (CDC) can be requested to perform drug resistance testing (404–718–4141; https://www.cdc.gov/laboratory/specimen-submission/detail.html?CDCTestCode=CDC-10239). CDC has experience with MTZ and TDZ susceptibility testing for 5-nitroimidazole-resistant T. vaginalis as well as management of infected patients. Based on resistance testing results, an alternative treatment regimen may be recommended.
Resistance rates in T. vaginalis for MTZ and TDZ have ranged from 4.3 to 10%,127 although these data are not contemporary; resistance rates of SEC among clinical T. vaginalis isolates are unknown. In vitro resistance may not always correlate with clinical treatment failure,128 especially in pregnant women,57 but use of alternative treatment regimens following drug resistance testing results in cure of resistant infections in >80% of cases, suggesting that there is a benefit to this testing.129
Alternative treatment regimens for infections demonstrating in vitro resistance may include 2-g oral MTZ or TDZ daily for 7 days.12 If a patient fails the 7-day regimen of high-dose oral MTZ or TDZ, additional treatment options have had successful results in women. One is high-dose oral TDZ 2-g daily plus vaginal TDZ 500 mg twice daily for 14 days.130 If this fails, high-dose oral TDZ (1-g three times daily) plus vaginal paromomycin (4 g of 6.25% cream nightly) for 14 days can be considered.131 Recently, a case report of successful treatment of a T. vaginalis-infected woman with 5-nitroimidazole resistance was also published using an extended regimen of 2-g oral SEC daily in combination with intra-vaginal boric acid 600 mg twice daily for 14 days.132
HIV-Infected Women
In a randomized controlled trial of HIV-infected women co-infected with T. vaginalis, 7-day oral MTZ was found to be superior to single-dose 2-g oral MTZ;64 this superiority only occurred in the presence of BV.133 Studies have found that protease inhibitors used for the treatment of HIV may interfere with the efficacy of single-dose 2-g oral MTZ among HIV-infected women.134,135
T. vaginalis screening (and treatment for positive cases) at entry to care and annually is recommended for HIV-infected women.12 It has been estimated that if the recommendation for T. vaginalis screening and treatment among HIV-infected women were followed, the lifetime cost of new HIV infections prevented would be ~$159,264,000 and could potentially prevent new HIV cases due to female-to-male transmission.136
Partner Management
Male sexual partners of persons infected with T. vaginalis also likely to also have infection. One recent study in Melbourne, Australia found that, among men with T. vaginalis-infected partners, the rate of infection was 18.6% compared to 0.5% among those who did not have a T. vaginalis-infected partner (p <0.001).137 Sexual partners of index cases of T. vaginalis infection should be treated. Commonly, patients are told to tell their partners to seek testing and treatment. Providers can also consider treating partners of positive patients presumptively. One method is expedited partner therapy (EPT), the practice of treating sexual partner(s) of patients diagnosed with an STI by providing a prescriptions or the medication to the patient to take to his/her partner(s) without the health care provider first examining the partner.
One randomized controlled trial demonstrated that partner treatment with single-dose 2-g oral TDZ resulted in a >4 fold reduction in repeat infections among T. vaginalis-infected index women.138 However, two other studies using single-dose 2-g oral MTZ for male partners of T. vaginalis-infected women found either no effect of EPT139 or a borderline effect.140 While it is possible that the two studies that used MTZ were either underpowered or did not use a correct control arm, it is also possible that oral TDZ is a better treatment for men. No data exist on the use of oral SEC in the setting of EPT.
FUTURE DIRECTIONS
Given that most studies have examined outcomes of symptomatic T. vaginalis infection in women, additional studies are needed to examine the importance of asymptomatic infection. This is necessary given the proliferation of T. vaginalis molecular diagnostic tests, including POC tests.99 Additional study of the role of T. vaginalis in contributing to PID is also needed, especially among HIV-uninfected women. Regarding treatment, contemporary data on rates of T. vaginalis resistance among 5-nitroimidazoles, including SEC, are needed. In addition, the role of oral SEC in the treatment of persistent T. vaginalis infection and in the setting of EPT should also be further investigated.
PRACTICE RECOMMENDATIONS*
- Screening at entry to care and then annually is recommended for women living with HIV.
- T. vaginalis infection may occur in the setting of a normal vaginal pH or an elevated pH.
- Wet mount microscopy should be performed when trichomoniasis is suspected, but a negative result does not exclude the diagnosis.
- Trichomonas InPouch® culture is a moderately complex test requiring incubation and reading over several days. Culture can be used to perform drug susceptibility testing.
- Molecular tests are currently the gold standard for the diagnosis of trichomoniasis; multiple assays are available.
- Screening or diagnostic testing for non-genital (i.e., oral, rectal) trichomoniasis is not recommended.
- Oral metronidazole 400–500 mg twice daily for 7 days is currently the recommended treatment for trichomoniasis in all women; single-dose 2-g oral metronidazole is no longer recommended in women for treatment for trichomoniasis.
- Single-dose 2-g oral metronidazole is the recommended treatment in men.
- While it is never recommended to drink alcohol with any medication, it is now understood that moderate alcohol consumption should not be a barrier to metronidazole use.
- Single-dose 2-g oral tinidazole or secnidazole can be considered as alternative treatments in both women and men.
- If a male is still infected with T. vaginalis after treatment with single-dose 2-g oral metronidazole and has been re-exposed to an untreated sexual partner, he should be re-dosed with another single-dose of 2-g oral metronidazole.
- If a male is still infected with T. vaginalis and he has not been re-exposed, he should be given a course of oral metronidazole 500 mg twice daily for 7 days.
- Re-testing for T. vaginalis is recommended, preferably by a molecular test, for all sexually active women between 3 weeks and 3 months after the end of treatment regardless of whether or not their sexual partner(s) were treated.
- If re-testing by 3 months is not possible, women should be re-tested whenever they next seek medical care <12 months after treatment.
- Metronidazole is safe for use in pregnant women in all stages of pregnancy.
- Tinidazole use should be avoided in pregnant women.
- Limited data are available on the use of secnidazole in pregnant women, but there is no evidence of adverse developmental outcomes in animal studies.
- In lactating women who are administered metronidazole, withholding breastfeeding during treatment and for 12–24 hours after the last dose is recommended.
- In lactating women who are administered tinidazole, interruption of breastfeeding is recommended during treatment and for 3 days after the last dose.
- In cases of 5-nitroimidazole hypersensitivity, desensitization is the first-line option of treatment.
- For patients in which metronidazole desensitization is not an option, use of other 5-nitroimidazoles such as tinidazole or secnidazole are not recommended because of the risk of cross-reactivity within the same drug class.
- For patients who are experiencing persistent infection not due to sexual re-exposure, culture and drug resistance testing are recommended.
- Sexual partners of patients with T. vaginalis infection should be tested and treated presumptively.
- Providers can consider treating partner(s) of positive patients presumptively, without the need of observing or testing them.
- A single-dose 2-g oral tinidazole as expedited partner therapy may be superior to single dose metronidazole for male partners of infected women.
*Adapted from the International Society for the Vulvovaginal Disease.100
CONFLICTS OF INTEREST
Christina A. Muzny, MD, MSPH has received research grant support from NIH/NIAID, Lupin Pharmaceuticals, Abbott Molecular, and Gilead Sciences, Inc.; is a consultant for Abbott, BioNTech, Scynexis, and Cepheid, and has received honoraria from Visby, Elsevier, Abbott Molecular, Cepheid, Roche Diagnostics, and Lupin Pharmaceuticals. Patricia J. Kissinger, PhD has received grants from NIH/NIAID and NIH/NICHD.
Feedback
Publishers’ note: We are constantly trying to update and enhance chapters in this Series. So if you have any constructive comments about this chapter please provide them to us by selecting the "Your Feedback" link in the left-hand column.
REFERENCES
Rowley J, Vander Hoorn S, Korenromp E, et al. Chlamydia, gonorrhoea, trichomoniasis and syphilis: global prevalence and incidence estimates, 2016. Bull World Health Organ 2019;97(8):548–62P. | |
Van Gerwen OT, Craig-Kuhn MC, Jones AT, et al. Trichomoniasis and adverse birth outcomes: a systematic review and meta-analysis. BJOG 2021;128(12):1907–15. | |
Sorvillo F, Smith L, Kerndt P, et al. Trichomonas vaginalis, HIV, and African-Americans. Emerg Infect Dis 2001;7(6):927–32. | |
Lazenby GB, Taylor PT, Badman BS, et al. An association between Trichomonas vaginalis and high-risk human papillomavirus in rural Tanzanian women undergoing cervical cancer screening. Clin Ther 2014;36(1):38–45. | |
Allsworth JE, Ratner JA, Peipert JF. Trichomoniasis and other sexually transmitted infections: results from the 2001–2004 National Health and Nutrition Examination Surveys. Sex Transm Dis 2009;36(12):738–44. | |
Moodley P, Wilkinson D, Connolly C, et al. Trichomonas vaginalis is associated with pelvic inflammatory disease in women infected with human immunodeficiency virus. Clin Infect Dis 2002;34(4):519–22. | |
Wiringa AE, Ness RB, Darville T, et al. Trichomonas vaginalis, endometritis and sequelae among women with clinically suspected pelvic inflammatory disease. Sex Transm Infect 2020;96(6):436–8. | |
Mielczarek E, Blaszkowska J. Trichomonas vaginalis: pathogenicity and potential role in human reproductive failure. Infection 2016;44(4):447–58. | |
Van Gerwen OT, Camino AF, Sharma J, et al. Epidemiology, Natural History, Diagnosis, and Treatment of Trichomonas vaginalis in Men. Clin Infect Dis 2021;73(6):1119–24. | |
Yang S, Zhao W, Wang H, et al. Trichomonas vaginalis infection-associated risk of cervical cancer: A meta-analysis. European journal of obstetrics, gynecology, and reproductive biology 2018;228:166–73. | |
Patel EU, Gaydos CA, Packman ZR, et al. Prevalence and Correlates of Trichomonas vaginalis Infection Among Men and Women in the United States. Clin Infect Dis 2018;67(2):211–7. | |
Workowski KA, Bachmann LH, Chan PA, et al. Sexually Transmitted Infections Treatment Guidelines, 2021. MMWR Recomm Rep 2021;70(4):1–187. | |
Muzny CA. Why Does Trichomonas vaginalis Continue to be a "Neglected" Sexually Transmitted Infection? Clin Infect Dis 2018;67(2):218–20. | |
Petrin D, Delgaty K, Bhatt R, et al. Clinical and microbiological aspects of Trichomonas vaginalis. Clin Microbiol Rev 1998;11(2):300–17. | |
Lizarraga A, Munoz D, Strobl-Mazzulla PH, et al. Toward incorporating epigenetics into regulation of gene expression in the parasite Trichomonas vaginalis. Mol Microbiol 2021;115(5):959–67. | |
Burch TA, Rees CW, Reardon LV. Epidemiological studies on human trichomoniasis. Am J Trop Med Hyg 1959;8(3):312–8. | |
Muzny CA, Rivers CA, Mena LA, et al. Genotypic characterization of Trichomonas vaginalis isolates among women who have sex with women in sexual partnerships. Sex Transm Dis 2012;39(7):556–8. | |
Crucitti T, Jespers V, Mulenga C, et al. Non-sexual transmission of Trichomonas vaginalis in adolescent girls attending school in Ndola, Zambia. PLoS One 2011;6(1):e16310. | |
Charles SX. Epidemiology of trichomonas vaginalis (TV) in rural adolescent and juvenile children. J Trop Pediatr 1991;37(2):90. | |
Kissinger PJ, Gaydos CA, Sena AC, et al. Diagnosis and Management of Trichomonas vaginalis: Summary of Evidence Reviewed for the 2021 Centers for Disease Control and Prevention Sexually Transmitted Infections Treatment Guidelines. Clin Infect Dis 2022;74(Supplement_2):S152–61. | |
Zhu X, Liu L, Yixi L, et al. The prevalence and risk factors of Trichomonas vaginalis in Wuhan and the Tibetan area, China: a two-center study. Parasitol Res 2023;122(1):265–73. | |
Dogan N, Gitmez F. The relationship between hygiene-social variables and Trichomonas vaginalis. International Journal of Infectious Diseases 2019;79:80–8. | |
Schwebke JR, Burgess D. Trichomoniasis. Clin Microbiol Rev 2004;17(4):794–803. | |
Fichorova R, Fraga J, Rappelli P, et al. Trichomonas vaginalis infection in symbiosis with Trichomonasvirus and Mycoplasma. Res Microbiol 2017;168(9–10):882–91. | |
Mercer F, Johnson PJ. Trichomonas vaginalis: Pathogenesis, Symbiont Interactions, and Host Cell Immune Responses. Trends Parasitol 2018;34(8):683–93. | |
Fraga J, Rojas L, Sariego I, et al. Species typing of Cuban Trichomonas vaginalis virus by RT-PCR, and association of TVV-2 with high parasite adhesion levels and high pathogenicity in patients. Arch Virol 2012;157(9):1789–95. | |
Bessarab IN, Nakajima R, Liu HW, et al. Identification and characterization of a type III Trichomonas vaginalis virus in the protozoan pathogen Trichomonas vaginalis. Arch Virol 2011;156(2):285–94. | |
Graves KJ, Ghosh AP, Schmidt N, et al. Trichomonas vaginalis Virus Among Women With Trichomoniasis and Associations With Demographics, Clinical Outcomes and Metronidazole Resistance. Clin Infect Dis 2019;69(12):2170–6. | |
Bouchemal K, Bories C, Loiseau PM. Strategies for Prevention and Treatment of Trichomonas vaginalis Infections. Clin Microbiol Rev 2017;30(3):811–25. | |
Harp DF, Chowdhury I. Trichomoniasis: evaluation to execution. European Journal of Obstetrics, Gynecology and Reproductive Biology 2011;157(1):3–9. | |
Bassey GB, Clarke AIL, Elhelu OK, et al. Trichomoniasis, a new look at a common but neglected STI in African descendance population in the United States and the Black Diaspora. A review of its incidence, research prioritization, and the resulting health disparities. J Natl Med Assoc 2022;114(1):78–89. | |
Ryan CM, de Miguel N, Johnson PJ. Trichomonas vaginalis: current understanding of host-parasite interactions. Essays Biochem 2011;51:161–75. | |
Secor WE, Meites E, Starr MC, et al. Neglected parasitic infections in the United States: trichomoniasis. Am J Trop Med Hyg 2014;90(5):800–4. | |
Krieger JN, Verdon M, Siegel N, et al. Natural history of urogenital trichomoniasis in men. J Urol 1993;149(6):1455–8. | |
Schwebke JR, Rompalo A, Taylor S, et al. Re-evaluating the treatment of nongonococcal urethritis: emphasizing emerging pathogens–a randomized clinical trial. Clin Infect Dis 2011;52(2):163–70. | |
Van der Pol B. Trichomonas vaginalis infection: the most prevalent nonviral sexually transmitted infection receives the least public health attention. Clin Infect Dis 2007;44(1):23–5. | |
Poole DN, McClelland RS. Global epidemiology of Trichomonas vaginalis. Sex Transm Infect 2013;89(6):418–22. | |
Van Der Pol B, Williams JA, Orr DP, et al. Prevalence, incidence, natural history, and response to treatment of Trichomonas vaginalis infection among adolescent women. J Infect Dis 2005;192(12):2039–44. | |
Juliana NCA, Omar AM, Pleijster J, et al. The Natural Course of Chlamydia trachomatis, Neisseria gonorrhoeae, Trichomonas vaginalis, and Mycoplasma genitalium in Pregnant and Post-Delivery Women in Pemba Island, Tanzania. Microorganisms 2021;9(6). | |
Figueroa-Angulo EE, Rendon-Gandarilla FJ, Puente-Rivera J, et al. The effects of environmental factors on the virulence of Trichomonas vaginalis. Microbes Infect 2012;14(15):1411–27. | |
Beltran NC, Horvathova L, Jedelsky PL, et al. Iron-induced changes in the proteome of Trichomonas vaginalis hydrogenosomes. PLoS One 2013;8(5):e65148. | |
Lehker MW, Alderete JF. Iron regulates growth of Trichomonas vaginalis and the expression of immunogenic trichomonad proteins. Mol Microbiol 1992;6(1):123–32. | |
Lehker MW, Arroyo R, Alderete JF. The regulation by iron of the synthesis of adhesins and cytoadherence levels in the protozoan Trichomonas vaginalis. J Exp Med 1991;174(2):311–8. | |
Lindrose AR, Htet KZ, O'Connell S, et al. Burden of trichomoniasis among older adults in the United States: a systematic review. Sex Health 2022;19(3):151–6. | |
Lewis FMT, Spicknall IH, Flagg EW, et al. Incidence and Prevalence of Trichomonas vaginalis Infection Among Persons Aged 15 to 59 Years: United States, 2018. Sex Transm Dis 2021;48(4):232–7. | |
Kelley CF, Rosenberg ES, O'Hara BM, et al. Prevalence of urethral Trichomonas vaginalis in black and white men who have sex with men. Sex Transm Dis 2012;39(9):739. | |
Le Roux M, Ngwenya IK, Nemarude AL, et al. Sexually transmitted infections and sexual behaviour among men having sex with men from Tshwane, South Africa. Int J STD AIDS 2023;34(3):183–90. | |
Carter-Wicker K, Utuama O, Omole F. Can trichomoniasis cause pharyngitis? A case report. SAGE Open Med Case Rep 2016;4:2050313X16682132. | |
Francis SC, Kent CK, Klausner JD, et al. Prevalence of rectal Trichomonas vaginalis and Mycoplasma genitalium in male patients at the San Francisco STD clinic, 2005–2006. Sex Transm Dis 2008;35(9):797–800. | |
Miller M, Liao Y, Gomez AM, et al. Factors associated with the prevalence and incidence of Trichomonas vaginalis infection among African American women in New York city who use drugs. J Infect Dis 2008;197(4):503–9. | |
Rogers SM, Turner CF, Hobbs M, et al. Epidemiology of undiagnosed trichomoniasis in a probability sample of urban young adults. PLoS One 2014;9(3):e90548. | |
Sutton M, Sternberg M, Koumans EH, et al. The prevalence of Trichomonas vaginalis infection among reproductive-age women in the United States, 2001–2004. Clin Infect Dis 2007;45(10):1319–26. | |
Shafir SC, Sorvillo FJ, Smith L. Current issues and considerations regarding trichomoniasis and human immunodeficiency virus in African-Americans. Clin Microbiol Rev 2009;22(1):37–45, Table of Contents. | |
Cu-Uvin S, Ko H, Jamieson DJ, et al. Prevalence, incidence, and persistence or recurrence of trichomoniasis among human immunodeficiency virus (HIV)-positive women and among HIV-negative women at high risk for HIV infection. Clin Infect Dis 2002;34(10):1406–11. | |
Kissinger P. Trichomonas vaginalis: a review of epidemiologic, clinical and treatment issues. BMC Infect Dis 2015;15:307. | |
Najafi A, Chaechi Nosrati MR, Ghasemi E, et al. Is there association between Trichomonas vaginalis infection and prostate cancer risk?: A systematic review and meta-analysis. Microb Pathog 2019;137:103752. | |
Lazenby GB, Thompson L, Powell AM, et al. Unexpected High Rates of Persistent Trichomonas vaginalis Infection in a Retrospective Cohort of Treated Pregnant Women. Sex Transm Dis 2019;46(1):2–8. | |
Ijasan O, Okunade KS, Oluwole AA. The prevalence and risk factors for Trichomonas vaginalis infection amongst human immunodeficiency virus-infected pregnant women attending the antenatal clinics of a university teaching hospital in Lagos, South-Western, Nigeria. Niger Postgrad Med J 2018;25(1):21–6. | |
Muzny CA, Blackburn RJ, Sinsky RJ, et al. Added benefit of nucleic acid amplification testing for the diagnosis of Trichomonas vaginalis among men and women attending a sexually transmitted diseases clinic. Clin Infect Dis 2014;59(6):834–41. | |
Sutcliffe S, Newman SB, Hardick A, et al. Prevalence and correlates of Trichomonas vaginalis infection among female US federal prison inmates. Sex Transm Dis 2010;37(9):585–90. | |
Freeman AH, Katz KA, Pandori MW, et al. Prevalence and correlates of Trichomonas vaginalis among incarcerated persons assessed using a highly sensitive molecular assay. Sex Transm Dis 2010;37(3):165–8. | |
Balkus JE, Richardson BA, Rabe LK, et al. Bacterial Vaginosis and the Risk of Trichomonas vaginalis Acquisition Among HIV-1-Negative Women. Sex Transm Dis 2014;41(2):123–8. | |
Hinderfeld AS, Simoes-Barbosa A. Vaginal dysbiotic bacteria act as pathobionts of the protozoal pathogen Trichomonas vaginalis. Microb Pathog 2020;138:103820. | |
Kissinger P, Mena L, Levison J, et al. A randomized treatment trial: single versus 7-day dose of metronidazole for the treatment of Trichomonas vaginalis among HIV-infected women. J Acquir Immune Defic Syndr 2010;55(5):565–71. | |
Kissinger P, Muzny CA, Mena LA, et al. Single-dose versus 7-day-dose metronidazole for the treatment of trichomoniasis in women: an open-label, randomised controlled trial. Lancet Infect Dis 2018;18(11):1251–9. | |
Legendre D, Muzny CA, Kissinger P. Pharmacokinetic and Pharmacodynamic Effects of Metronidazole May Account for the Superior Efficacy of Multidose Therapy Among Women With Trichomoniasis. Sex Transm Dis 2019;46(11):751–2. | |
Balkus JE, Richardson BA, Mochache V, et al. A prospective cohort study comparing the effect of single-dose 2 g metronidazole on Trichomonas vaginalis infection in HIV-seropositive versus HIV-seronegative women. Sex Transm Dis 2013;40(6):499–505. | |
Muzny CA, Sunesara IR, Martin DH, et al. Sexually transmitted infections and risk behaviors among African American women who have sex with women: does sex with men make a difference? Sex Transm Dis 2011;38(12):1118–25. | |
Simhan HN, Anderson BL, Krohn MA, et al. Host immune consequences of asymptomatic Trichomonas vaginalis infection in pregnancy. Am J Obstet Gynecol 2007;196(1):59 e1–5. | |
Fichorova RN. Impact of T. vaginalis infection on innate immune responses and reproductive outcome. J Reprod Immunol 2009;83(1–2):185–9. | |
Tanaka Y, Narahara H, Takai N, et al. Interleukin-1beta and interleukin-8 in cervicovaginal fluid during pregnancy. Am J Obstet Gynecol 1998;179(3 Pt 1):644–9. | |
Mann JR, McDermott S, Barnes TL, et al. Trichomoniasis in pregnancy and mental retardation in children. Ann Epidemiol 2009;19(12):891–9. | |
Masha SC, Cools P, Sanders EJ, et al. Trichomonas vaginalis and HIV infection acquisition: a systematic review and meta-analysis. Sex Transm Infect 2019;95(1):36–42. | |
Barker EK, Malekinejad M, Merai R, et al. Risk of Human Immunodeficiency Virus Acquisition Among High-Risk Heterosexuals With Nonviral Sexually Transmitted Infections: A Systematic Review and Meta-Analysis. Sex Transm Dis 2022;49(6):383–97. | |
Kissinger P, Adamski A. Trichomoniasis and HIV interactions: a review. Sex Transm Infect 2013;89(6):426–33. | |
Wang CC, McClelland RS, Reilly M, et al. The effect of treatment of vaginal infections on shedding of human immunodeficiency virus type 1. J Infect Dis 2001;183(7):1017–22. | |
Kissinger P, Amedee A, Clark RA, et al. Trichomonas vaginalis treatment reduces vaginal HIV-1 shedding. Sex Transm Dis 2009;36(1):11–6. | |
Gottlieb SL, Douglas JM Jr, Foster M, et al. Incidence of herpes simplex virus type 2 infection in 5 sexually transmitted disease (STD) clinics and the effect of HIV/STD risk-reduction counseling. J Infect Dis 2004;190(6):1059–67. | |
Boselli F, Chiossi G, Bortolamasi M, et al. Prevalence and determinants of genital shedding of herpes simplex virus among women attending Italian colposcopy clinics. European journal of obstetrics, gynecology, and reproductive biology 2005;118(1):86–90. | |
Ginocchio CC, Chapin K, Smith JS, et al. Prevalence of Trichomonas vaginalis and coinfection with Chlamydia trachomatis and Neisseria gonorrhoeae in the United States as determined by the Aptima Trichomonas vaginalis nucleic acid amplification assay. J Clin Microbiol 2012;50(8):2601–8. | |
Zhang Z, Li Y, Lu H, et al. A systematic review of the correlation between Trichomonas vaginalis infection and infertility. Acta Trop 2022;236:106693. | |
Bansal S, Bhargava A, Verma P, et al. Etiology of cervicitis: Are there new agents in play? Indian J Sex Transm Dis AIDS 2022;43(2):174–8. | |
Sena AC, Miller WC, Hobbs MM, et al. Trichomonas vaginalis infection in male sexual partners: implications for diagnosis, treatment, and prevention. Clin Infect Dis 2007;44(1):13–22. | |
Swygard H, Sena AC, Hobbs MM, et al. Trichomoniasis: clinical manifestations, diagnosis and management. Sex Transm Infect 2004;80(2):91–5. | |
Landers DV, Wiesenfeld HC, Heine RP, et al. Predictive value of the clinical diagnosis of lower genital tract infection in women. Am J Obstet Gynecol 2004;190(4):1004–10. | |
Edwards T, Burke P, Smalley H, et al. Trichomonas vaginalis: Clinical relevance, pathogenicity and diagnosis. Crit Rev Microbiol 2016;42(3):406–17. | |
Wolner-Hanssen P, Krieger JN, Stevens CE, et al. Clinical manifestations of vaginal trichomoniasis. JAMA 1989;261(4):571–6. | |
Hobbs MM, Sena AC. Modern diagnosis of Trichomonas vaginalis infection. Sex Transm Infect 2013;89(6):434–8. | |
Alderete JF, Chan H. Point-of-Care Diagnostic for Trichomonas vaginalis, the Most Prevalent, Non-Viral Sexually Transmitted Infection. Pathogens 2023;12(1). | |
Ohlemeyer CL, Hornberger LL, Lynch DA, et al. Diagnosis of Trichomonas vaginalis in adolescent females: InPouch TV culture versus wet-mount microscopy. The Journal of adolescent health: official publication of the Society for Adolescent Medicine 1998;22(3):205–8. | |
Rivers CA, Muzny CA, Schwebke JR. Diagnostic rates differ on the basis of the number of read days with the use of the InPouch culture system for Trichomonas vaginalis screening. J Clin Microbiol 2013;51(11):3875–6. | |
Gaydos CA, Hobbs M, Marrazzo J, et al. Rapid Diagnosis of Trichomonas vaginalis by Testing Vaginal Swabs in an Isothermal Helicase-Dependent AmpliVue Assay. Sex Transm Dis 2016;43(6):369–73. | |
Gaydos CA, Schwebke J, Dombrowski J, et al. Clinical performance of the Solana(R) Point-of-Care Trichomonas Assay from clinician-collected vaginal swabs and urine specimens from symptomatic and asymptomatic women. Expert Rev Mol Diagn 2017;17(3):303–6. | |
Schwebke JR, Hobbs MM, Taylor SN, et al. Molecular testing for Trichomonas vaginalis in women: results from a prospective U.S. clinical trial. J Clin Microbiol 2011;49(12):4106–11. | |
Van Der Pol B, Williams JA, Taylor SN, et al. Detection of Trichomonas vaginalis DNA by use of self-obtained vaginal swabs with the BD ProbeTec Qx assay on the BD Viper system. J Clin Microbiol 2014;52(3):885–9. | |
Schwebke JR, Gaydos CA, Davis T, et al. Clinical Evaluation of the Cepheid Xpert TV Assay for Detection of Trichomonas vaginalis with Prospectively Collected Specimens from Men and Women. J Clin Microbiol 2018;56(2). | |
Van Der Pol B. A profile of the cobas(R) TV/MG test for the detection of Trichomonas vaginalis and Mycoplasma genitalium. Expert Rev Mol Diagn 2020;20(4):381–6. | |
Van Der Pol B, Torres-Chavolla E, Kodsi S, et al. Clinical Performance of the BD CTGCTV2 Assay for the BD MAX System for Detection of Chlamydia trachomatis, Neisseria gonorrhoeae, and Trichomonas vaginalis Infections. Sex Transm Dis 2021;48(2):134–40. | |
Morris SR, Bristow CC, Wierzbicki MR, et al. Performance of a single-use, rapid, point-of-care PCR device for the detection of Neisseria gonorrhoeae, Chlamydia trachomatis, and Trichomonas vaginalis: a cross-sectional study. Lancet Infect Dis 2021;21(5):668–76. | |
Vieira-Baptista P, Stockdale CK, Sobel J (eds). International Society for the Study of Vulvovaginal Disease recommendations for the diagnosis and treatment of vaginitis. Lisbon: Admedic, 2023. https://www.issvd.org/guidelines | |
Howe K, Kissinger PJ. Single-Dose Compared With Multidose Metronidazole for the Treatment of Trichomoniasis in Women: A Meta-Analysis. Sex Transm Dis 2017;44(1):29–34. | |
Committee on Practice Bulletins. Vaginitis in Nonpregnant Patients: ACOG Practice Bulletin, Number 215. Obstet Gynecol 2020;135(1):e1–17. | |
Muzny CA, Schwebke JR, Nyirjesy P, et al. Efficacy and Safety of Single Oral Dosing of Secnidazole for Trichomoniasis in Women: Results of a Phase 3, Randomized, Double-Blind, Placebo-Controlled, Delayed-Treatment Study. Clin Infect Dis 2021;73(6):e1282–9. | |
Muzny CA, Van Gerwen OT, Legendre D. Secnidazole: a treatment for trichomoniasis in adolescents and adults. Expert Rev Anti Infect Ther 2022:1–10. | |
Craig-Kuhn MC, Granade C, Muzny CA, et al. Optimal Timing for Trichomonas vaginalis Test of Cure Using Nucleic Acid Amplification Testing. Sex Transm Dis 2019;46(5):312–6. | |
Schwandt A, Williams C, Beigi RH. Perinatal transmission of Trichomonas vaginalis: a case report. J Reprod Med 2008;53(1):59–61. | |
Trintis J, Epie N, Boss R, et al. Neonatal Trichomonas vaginalis infection: a case report and review of literature. Int J STD AIDS 2010;21(8):606–7. | |
Carter JE, Whithaus KC. Neonatal respiratory tract involvement by Trichomonas vaginalis: a case report and review of the literature. Am J Trop Med Hyg 2008;78(1):17–9. | |
Burtin P, Taddio A, Ariburnu O, et al. Safety of metronidazole in pregnancy: a meta-analysis. Am J Obstet Gynecol 1995;172(2 Pt 1):525–9. | |
Caro-Paton T, Carvajal A, Martin de Diego I, et al. Is metronidazole teratogenic? A meta-analysis. Br J Clin Pharmacol 1997;44(2):179–82. | |
Evaldson GR, Lindgren S, Nord CE, et al. Tinidazole milk excretion and pharmacokinetics in lactating women. Br J Clin Pharmacol 1985;19(4):503–7. | |
Garcia-Rubio I, Martinez-Cocera C, Santos Magadan S, et al. Hypersensitivity reactions to metronidazole. Allergol Immunopathol (Madr) 2006;34(2):70–2. | |
Gendelman SR, Pien LC, Gutta RC, et al. Modified oral metronidazole desensitization protocol. Allergy Rhinol (Providence) 2014;5(2):66–9. | |
Madsen JT, Thormann J, Kerre S, et al. Allergic contact dermatitis to topical metronidazole – 3 cases. Contact Dermatitis 2007;56(6):364–6. | |
Mishra D, Mobashir M, Zaheer MS. Fixed drug eruption and cross-reactivity between tinidazole and metronidazole. Int J Dermatol 1990;29(10):740. | |
Kanwar AJ, Sharma R, Rajagopalan M, et al. Fixed drug eruption due to tinidazole with cross-reactivity with metronidazole. Dermatologica 1990;180(4):277. | |
Macy E, Romano A, Khan D. Practical Management of Antibiotic Hypersensitivity in 2017. J Allergy Clin Immunol Pract 2017;5(3):577–86. | |
Van Gerwen OT, Camino AF, Bourla LN, et al. Management of Trichomoniasis in the Setting of 5-Nitroimidazole Hypersensitivity. Sex Transm Dis 2021;48(8):e111-e5. | |
Kurohara ML, Kwong FK, Lebherz TB, et al. Metronidazole hypersensitivity and oral desensitization. J Allergy Clin Immunol 1991;88(2):279–80. | |
Pearlman MD, Yashar C, Ernst S, et al. An incremental dosing protocol for women with severe vaginal trichomoniasis and adverse reaction to metronidazole. Am J Obstet Gynecol 1996;174(3):934–6. | |
Muzny C, Barnes A, Mena L. Symptomatic Trichomonas vaginalis infection in the setting of severe nitroimidazole allergy: successful treatment with boric acid. Sex Health 2012;9(4):389–91. | |
Backus KV, Muzny CA, Beauchamps LS. Trichomonas vaginalis Treated With Boric Acid in a Metronidazole Allergic Female. Sex Transm Dis 2017;44(2):120. | |
Aggarwal A, Shier RM. Recalcitrant Trichomonas vaginalis infections successfully treated with vaginal acidification. J Obstet Gynaecol Can 2008;30(1):55–8. | |
Nyirjesy P, Sobel JD, Weitz MV, et al. Difficult-to-treat trichomoniasis: results with paromomycin cream. Clin Infect Dis 1998;26(4):986–8. | |
Helms DJ, Mosure DJ, Secor WE, et al. Management of trichomonas vaginalis in women with suspected metronidazole hypersensitivity. Am J Obstet Gynecol 2008;198(4):370 e1–7. | |
Keating MA, Nyirjesy P. Trichomonas vaginalis Infection in a Tertiary Care Vaginitis Center. Sex Transm Dis 2015;42(9):482–5. | |
Kirkcaldy RD, Augostini P, Asbel LE, et al. Trichomonas vaginalis antimicrobial drug resistance in 6 US cities, STD Surveillance Network, 2009–2010. Emerg Infect Dis 2012;18(6):939–43. | |
Schwebke JR, Barrientes FJ. Prevalence of Trichomonas vaginalis isolates with resistance to metronidazole and tinidazole. Antimicrob Agents Chemother 2006;50(12):4209–10. | |
Bosserman EA, Helms DJ, Mosure DJ, et al. Utility of antimicrobial susceptibility testing in Trichomonas vaginalis-infected women with clinical treatment failure. Sex Transm Dis 2011;38(10):983–7. | |
Sobel JD, Nyirjesy P, Brown W. Tinidazole therapy for metronidazole-resistant vaginal trichomoniasis. Clin Infect Dis 2001;33(8):1341–6. | |
Nyirjesy P, Gilbert J, Mulcahy LJ. Resistant trichomoniasis: successful treatment with combination therapy. Sex Transm Dis 2011;38(10):962–3. | |
McNeil CJ, Williamson JC, Muzny CA. Successful Treatment of Persistent 5-Nitroimidazole-Resistant Trichomoniasis with an Extended Course of Oral Secnidazole Plus Intravaginal Boric Acid. Sex Transm Dis 2022; e-published online 11/26/22. | |
Gatski M, Martin DH, Levison J, et al. The influence of bacterial vaginosis on the response to Trichomonas vaginalis treatment among HIV-infected women. Sex Transm Infect 2011;87(3):205–8. | |
Adamski A, Clark RA, Mena L, et al. The influence of ART on the treatment of Trichomonas vaginalis among HIV-infected women. Clin Infect Dis 2014;59(6):883–7. | |
Kissinger P, Adamski A, Clark RA, et al. Does Antiretroviral Therapy Interfere With the Treatment of Trichomonas vaginalis Among HIV+ Women? Sex Transm Dis 2013;40(6):506–7. | |
Lazenby GB, Unal ER, Andrews AL, et al. Cost-effectiveness analysis of annual Trichomonas vaginalis screening and treatment in HIV-positive women to prevent HIV transmission. Sex Transm Dis 2014;41(6):353–8. | |
Abraham E, Fairley CK, Aung ET, et al. Trichomoniasis among men presenting to a sexual health clinic in Melbourne, Australia. Sex Health 2022;19(1):70–3. | |
Lyng J, Christensen J. A double-blind study of the value of treatment with a single dose tinidazole of partners to females with trichomoniasis. Acta Obstet Gynecol Scand 1981;60(2):199–201. | |
Kissinger P, Schmidt N, Mohammed H, et al. Patient-delivered partner treatment for Trichomonas vaginalis infection: a randomized controlled trial. Sex Transm Dis 2006;33(7):445–50. | |
Schwebke JR, Desmond RA. A randomized controlled trial of partner notification methods for prevention of trichomoniasis in women. Sex Transm Dis 2010;37(6):392–6. |
Online Study Assessment Option
All readers who are qualified doctors or allied medical professionals can automatically receive 2 Continuing Professional Development points plus a Study Completion Certificate from GLOWM for successfully answering four multiple-choice questions (randomly selected) based on the study of this chapter. Medical students can receive the Study Completion Certificate only.
(To find out more about the Continuing Professional Development awards program CLICK HERE)