This chapter should be cited as follows:
Oyelese Y, Perez M, et al., Glob Libr Women's Med
ISSN: 1756-2228; DOI 10.3843/GLOWM.419203
The Continuous Textbook of Women’s Medicine Series – Obstetrics Module
Volume 18
Ultrasound in obstetrics
Volume Editors:
Professor Caterina M (Katia) Bilardo, Amsterdam UMC, Amsterdam and University of Groningen, Groningen, The Netherlands
Dr Valentina Tsibizova, PREIS International School, Florence, Italy

Chapter
Ultrasound Evaluation of the Placenta, Umbilical Cord and Membranes: Normal and Abnormal Findings
First published: August 2025
Study Assessment Option
By answering four multiple-choice questions (randomly selected) after studying this chapter, readers can qualify for Continuing Professional Development points plus a Study Completion Certificate from GLOWM.
See end of chapter for details.
INTRODUCTION
The placenta and umbilical cord play a pivotal role in fetal wellbeing, serving as the lifeline that supports the developing fetus.1 Ultrasound is the primary – and often the only – tool for assessing these critical structures during pregnancy.2 Unfortunately, the focus of most obstetric imaging is almost exclusively the fetus, leaving the placenta and cord relatively underexamined.1,3,4,5 This oversight can have significant consequences, as undiagnosed abnormalities of the placenta or umbilical cord can lead to severe complications for both the mother and the fetus.1,2,3,4 Indeed, issues related to the placenta and cord are among the leading causes of stillbirth, with one large study attributing 23% of stillbirths to cord abnormalities.6 In addition, these conditions increase the risk for long-term sequelae in the offspring, including cerebral palsy.7,8 Many of these abnormalities are detectable by ultrasound, and adverse outcomes resulting from them are preventable by instituting appropriate management plans.
Those who perform obstetric imaging often encounter placental or umbilical cord abnormalities with which they may be unfamiliar, making it challenging to interpret the findings, assess the associated risks and determine the appropriate course of action to mitigate perinatal or maternal morbidity and mortality.2,9,10,11,12 This chapter is designed to equip sonographers and physicians with the knowledge to assess accurately the placenta, umbilical cord and membranes during prenatal ultrasound. It explores the normal and abnormal appearances of these structures, highlights key abnormalities that can be detected and discusses their clinical implications, and describes the management pathways when abnormal findings occur, providing a comprehensive guide to optimizing prenatal care and improving outcomes.
THE PLACENTA
Development
The placenta is a unique disc-shaped temporary organ that is attached to the uterine wall and forms the interface between the mother and the fetus. The placenta’s primary functions include facilitating the exchange of oxygen, nutrients and waste products between the mother and fetus. It also is responsible for producing hormones essential for maintaining pregnancy and acts as a protective barrier against harmful substances and organisms.
The maternal surface of the placenta or basal plate is derived from the decidua basalis and is in direct contact with the maternal endometrium, anchoring the placenta to the uterine wall. Nitabuch’s fibrinoid layer, also known as the uteroplacental fibrinoid, is an uninterrupted layer that separates trophoblastic cells from decidual cells, marking the maternofetal border.
The fetal surface, or chorionic plate, is a segment of the continuous chorionic tissue surrounding the fetus. Unlike the chorion laeve (fetal membranes), the chorionic plate has a thicker connective tissue layer containing fetal vessels that branch from the umbilical cord and spread across the fetal surface.
Between the chorionic plate (fetal surface) and the basal plate (maternal surface) lies the villous parenchyma, which consists of fetally-derived chorionic villi. These villi are immersed in the intervillous space, where maternal blood circulates. Gas exchange, as well as the transfer of nutrients and waste products between maternal and fetal circulations, occurs at the level of the chorionic villi.
The umbilical cord serves as the connecting stalk, acting as a conduit for fetal vessels between the placenta and the fetus.
Normal ultrasound appearance of the placenta
Placental characteristics by trimester: thickness, echogenicity and location
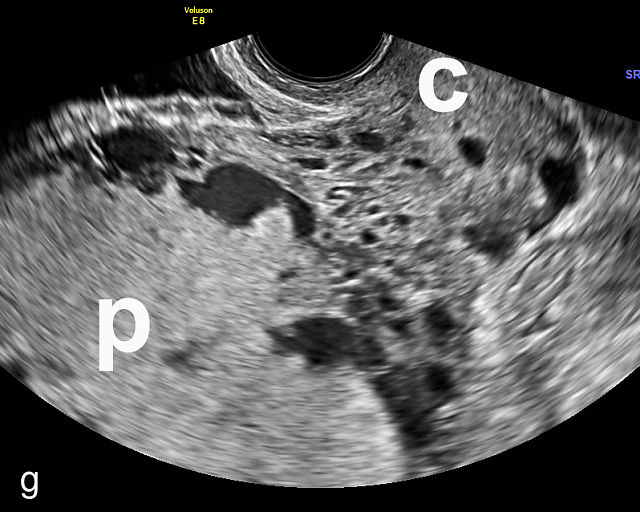
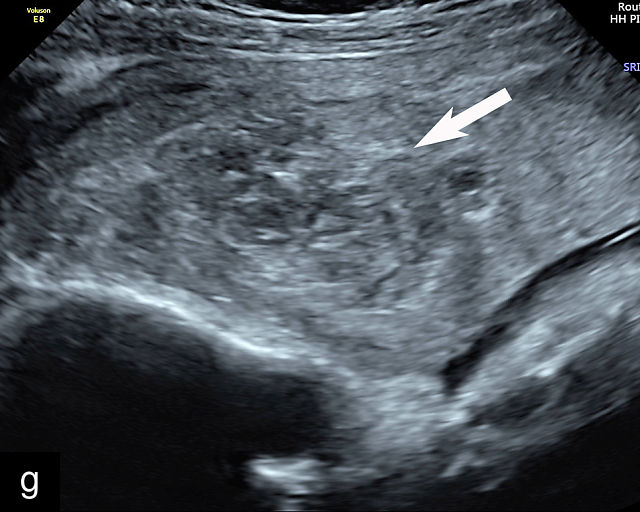
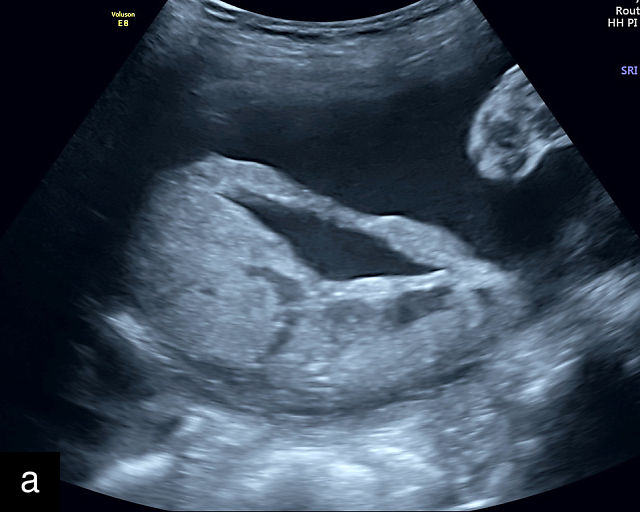
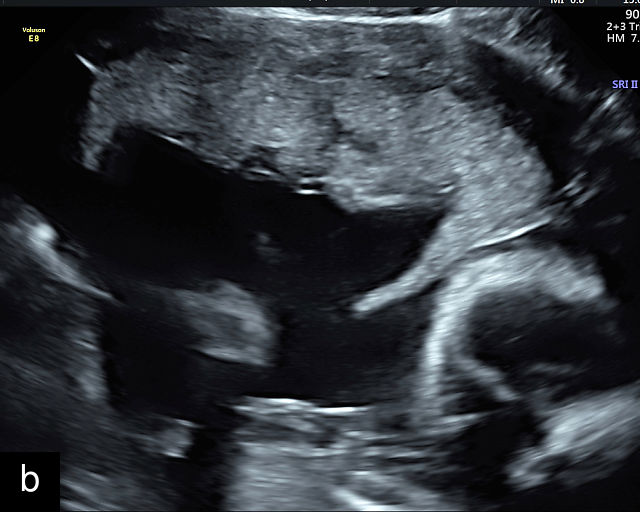
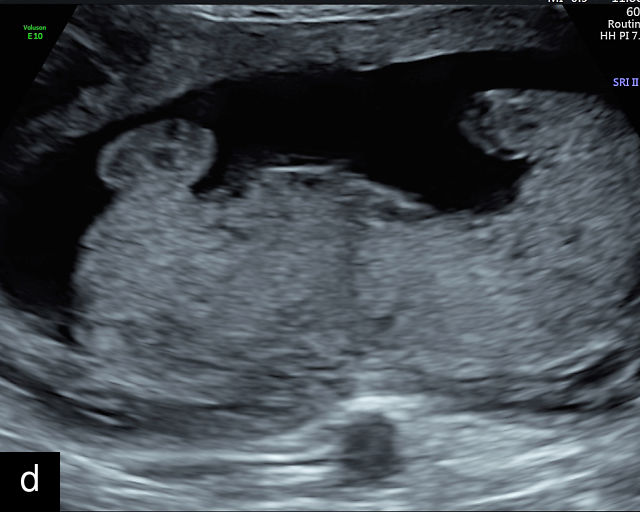
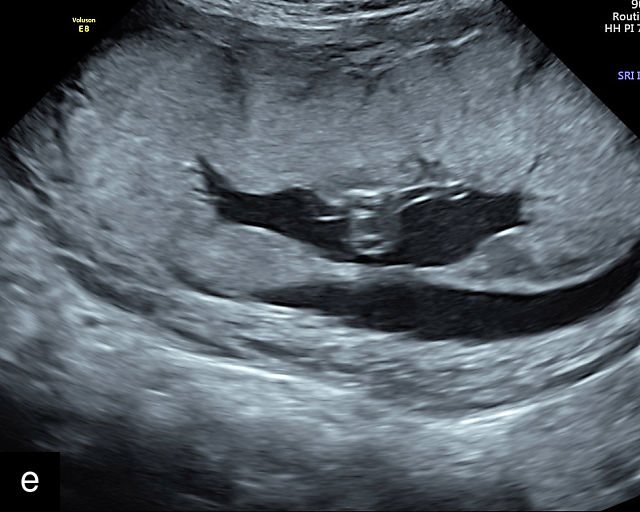
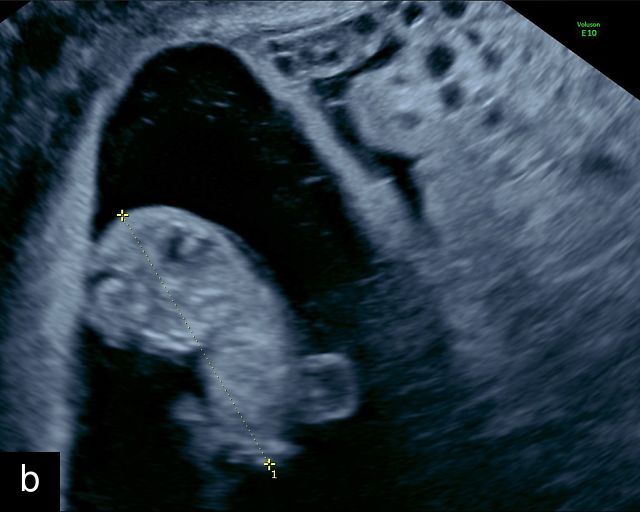
In the early first trimester, trophoblastic tissue appears as an echogenic ring encircling the gestational sac (Figure 1a). By the late first trimester, the placenta becomes recognizable as a distinct structure on ultrasound (Figure 1b). Initially, it presents as a homogeneous echogenic mass (Figure 1b–d) but undergoes progressive differentiation, becoming more heterogeneous as pregnancy advances from the second to third trimester (Figure 1e–g). By the third trimester, cotyledons become discernible, and in the late third trimester, calcifications frequently appear basally and around the cotyledons (Figure 1g).

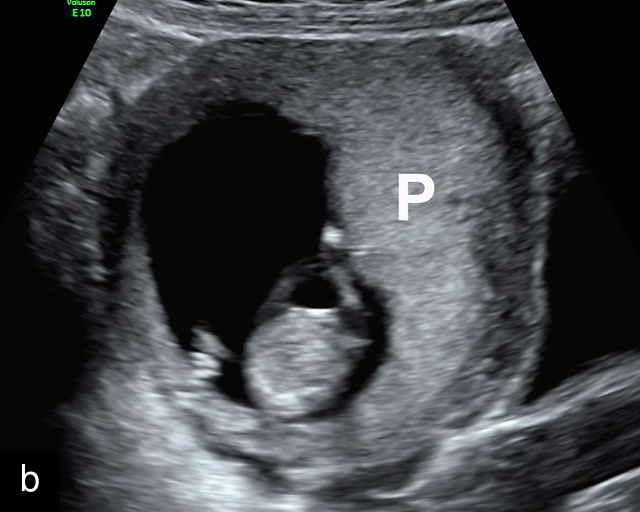
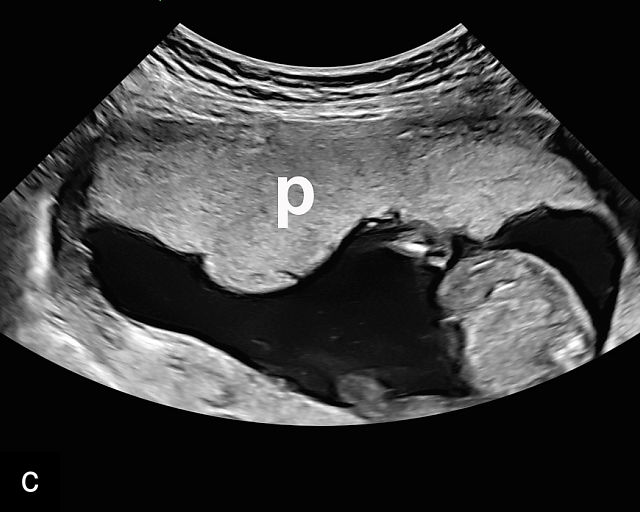
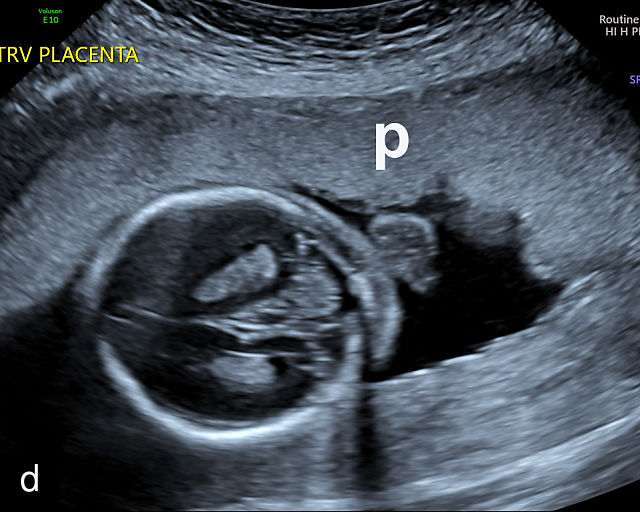

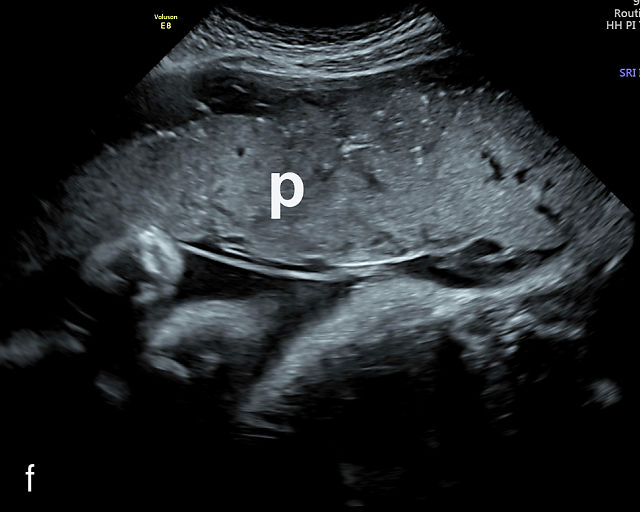

1
Ultrasound images of development of the placenta (P/p). (a) Trophoblastic tissue appearing as an echogenic ring surrounding the gestational sac at 6 weeks' gestation. (b) Placenta at 12 weeks has become a discrete, uniformly echogenic mass. (c) Placenta at 17 weeks. (d) Placenta at 20 weeks. (e) Placenta at 27 weeks. (f) Placenta at 33 weeks. Increasingly, differentiation and heterogeneous appearance is seen, with demarcation of the cotyledons. Basal calcifications are beginning to appear. (g) Placenta at 40 weeks, showing a distinctly heterogeneous appearance, with clear demarcation of the cotyledons and presence of calcifications.
The placenta can be located anteriorly, posteriorly, fundally or laterally or in a low-lying position, including placenta previa (covering or near the internal cervical os). A low-lying placenta or placenta previa is common before 20 weeks, with over 90% resolving before term. Most placentas covering the internal os in early pregnancy will migrate away by 20 weeks; therefore, the terms 'placenta previa' or 'low-lying placenta' should not be used before 18 weeks.
Placental thickness increases with gestational age, averaging approximately 1 mm per week. Subchorionic hematomas, commonly seen in the first and second trimesters, are typically of little or no clinical significance and most often resolve as pregnancy progresses. Additionally, hypoechoic areas known as placental lakes are frequently observed and generally hold no clinical significance.13
Systematic approach to sonographic placental examination
Ultrasound identification of the placenta was first described by Gottesfeld and associates in 1966.14 Examination of the placenta is now a part of routine obstetric ultrasound. However, this assessment is typically limited to determining the placenta’s location, particularly in relation to the internal os, and, in some cases, identifying the site of umbilical cord insertion.2 More detailed evaluations may include the number of placental lobes. Overall, examination of the placenta has been limited by the absence of a standardized approach.2
Recently, Hernandez-Andrade and colleagues proposed a systematic method for sonographic evaluation of the placenta, highlighting the importance of assessing more than just placental location and cord insertion.² Their approach includes performing a sagittal sweep of the entire placenta, moving from side to side and top to bottom, to ensure a thorough evaluation.
Their method involves evaluating four key placental regions:
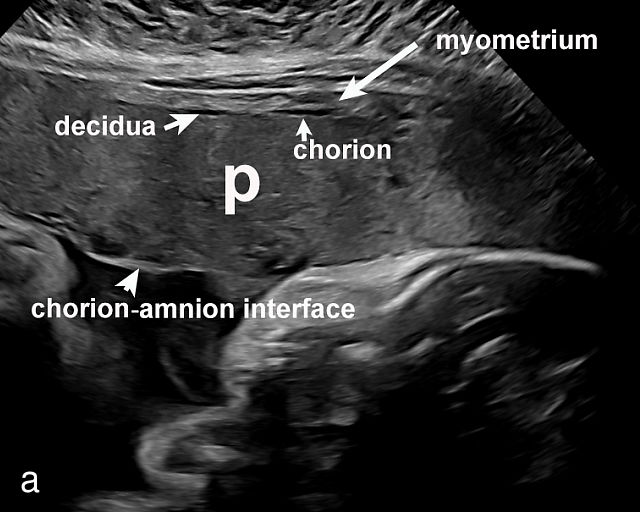
- The uterine wall (myometrium) and the decidua/chorion interface – The outermost layer, the uterine wall or myometrium, appears as a gray area with varying thickness (Figure 2a) depending on placental location. The myometrium is generally thicker when the placenta is located in the upper uterus or fundus. Just beneath the myometrium lies the decidua, a hypoechoic (black) layer where the spiral arteries develop.
- The amnion–chorion (or chorion–amnion) interface – The amnion, the innermost fetal membrane, is more echogenic and appears as a gray line, while the underlying chorion is slightly less echogenic. This interface is important for identifying abnormalities in the fetal surface of the placenta.
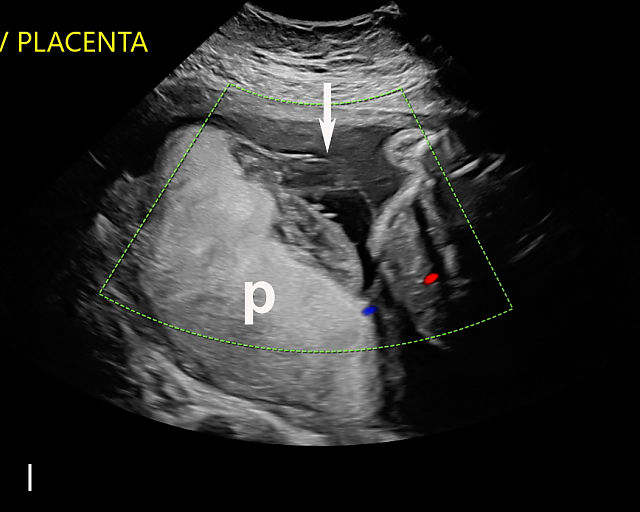
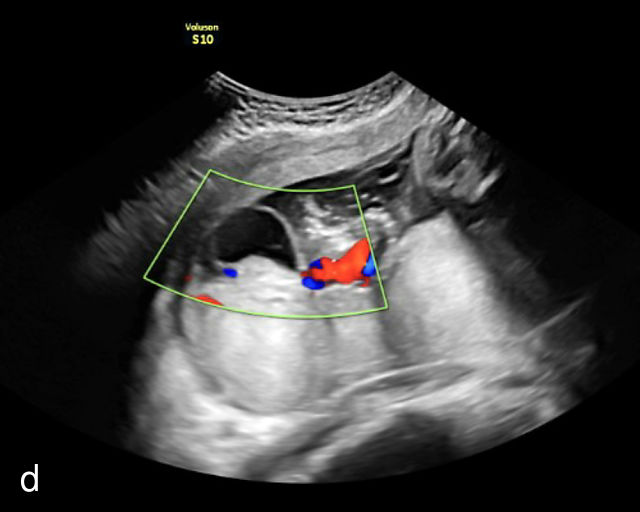
- The placental body – The main mass of the placenta, where most abnormalities are detected. The texture, thickness and uniformity of the placental body should be carefully examined for any irregularities.
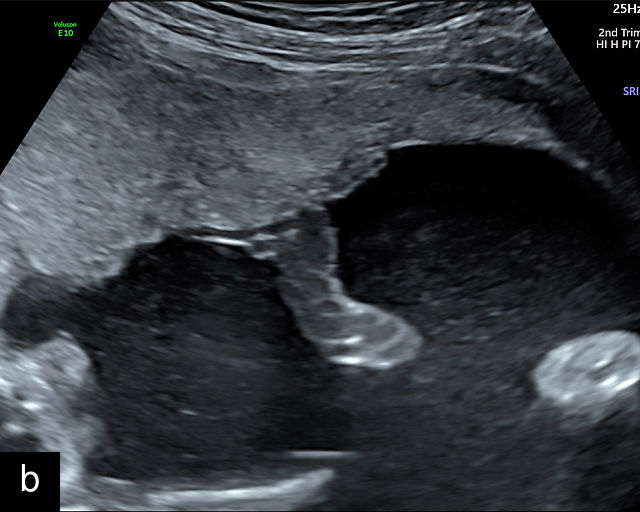
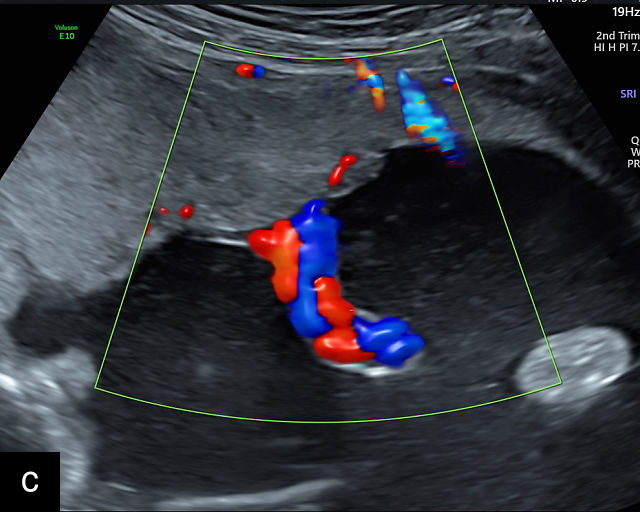
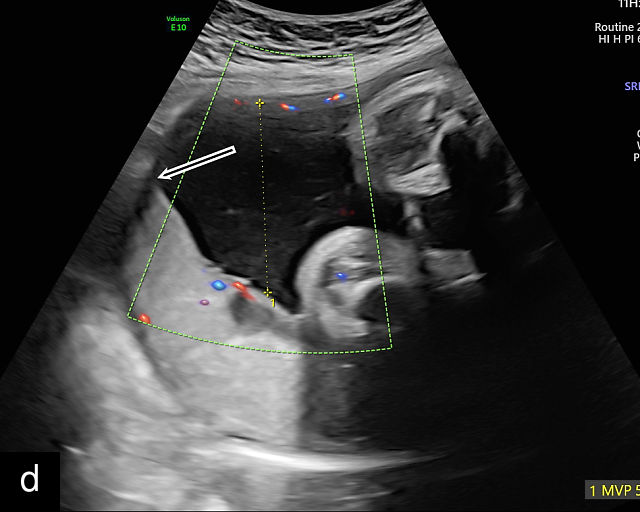
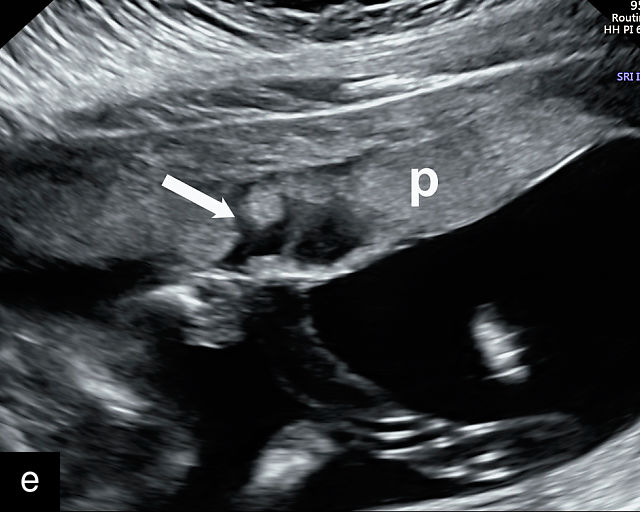
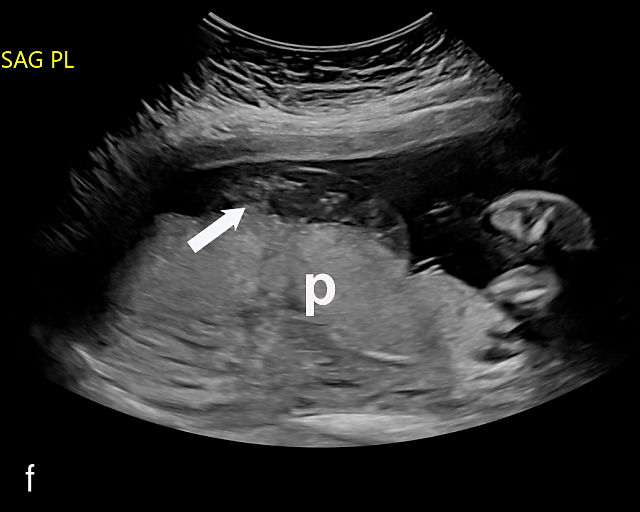
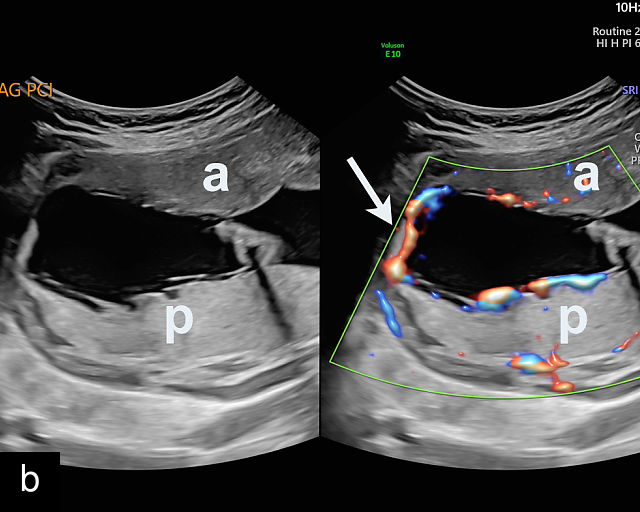
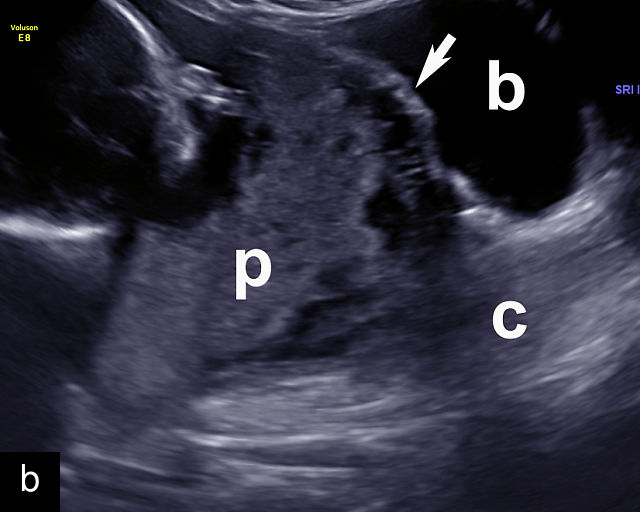
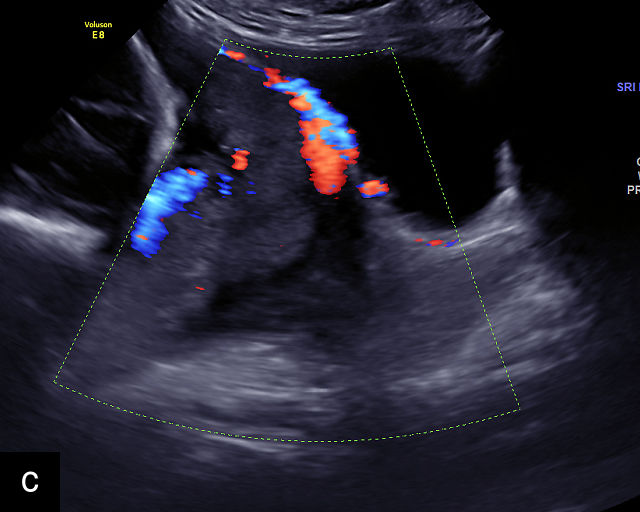
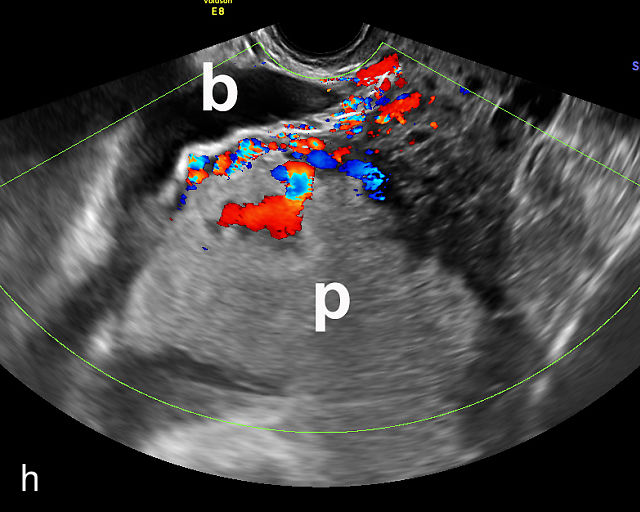
- The umbilical cord insertion (Figure 2b,c) and placental margin (Figure 2d) – Special attention should be paid to the site at which the umbilical cord or vessels insert into the placenta, as well as the marginal regions of the placenta, to identify potential abnormalities.

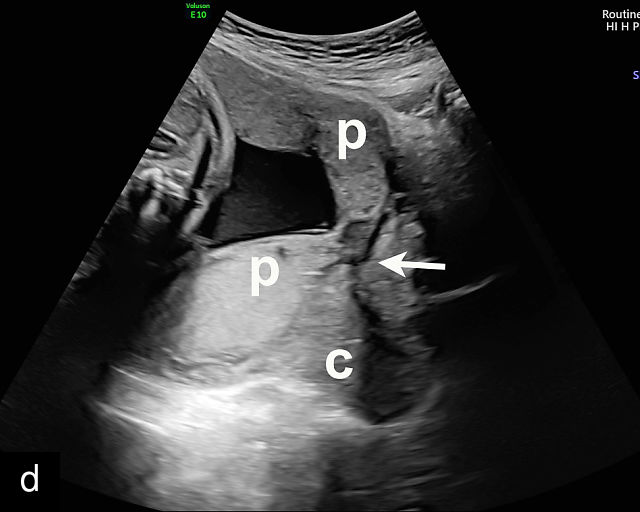
2
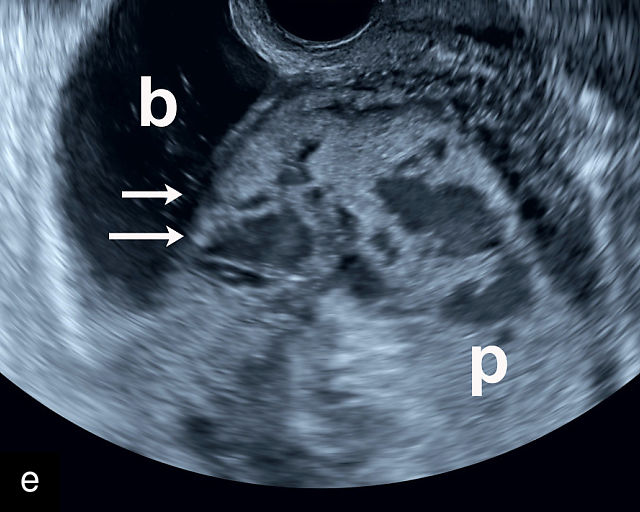
(a) Transabdominal grayscale ultrasound image of the placenta showing the four key placental regions: the myometrium (gray area), decidua (black line), chorion (light gray thin line), placental body (p), and the chorion–amnion interface. (b) Grayscale ultrasound image of the umbilical cord insertion into the placenta. (c) Same view as in (b) but with color Doppler applied. (d) Normal placental edge (arrow), which is a common location for placental lakes. (e) Rolled placental edges (arrows) in circumvallate placenta (p).
Hernandez-Andrade and colleagues assert that most placental abnormalities can be accurately identified using gray-scale ultrasound alone, but color Doppler can be a valuable tool in distinguishing between certain lesions.2,15 By following this structured approach, these authors propose that the majority of placental and cord abnormalities can be effectively recognized, leading to improved diagnostic accuracy and better prenatal care.2
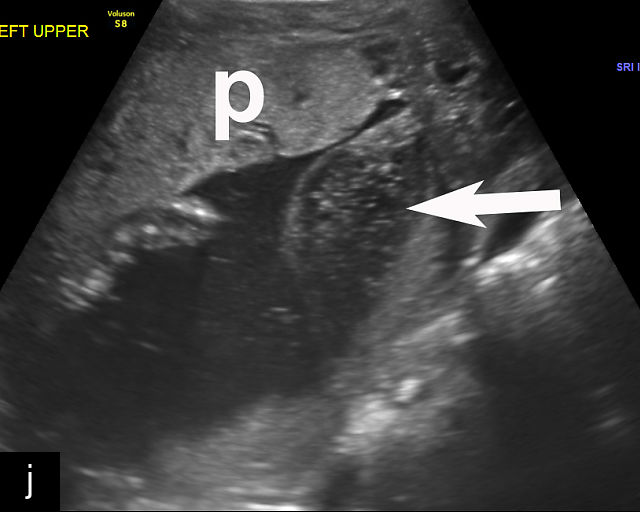
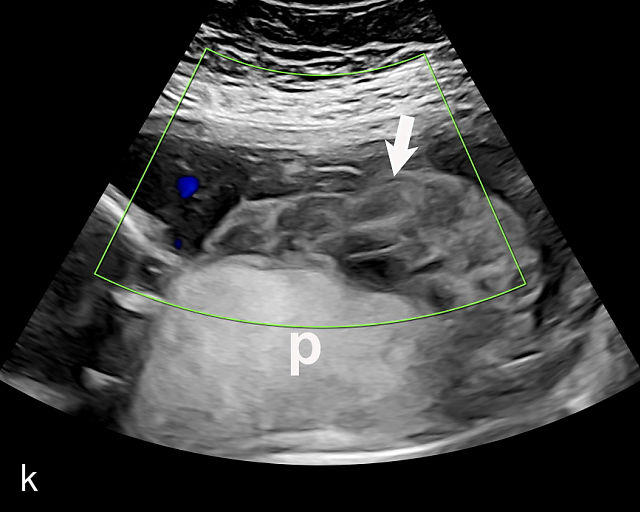
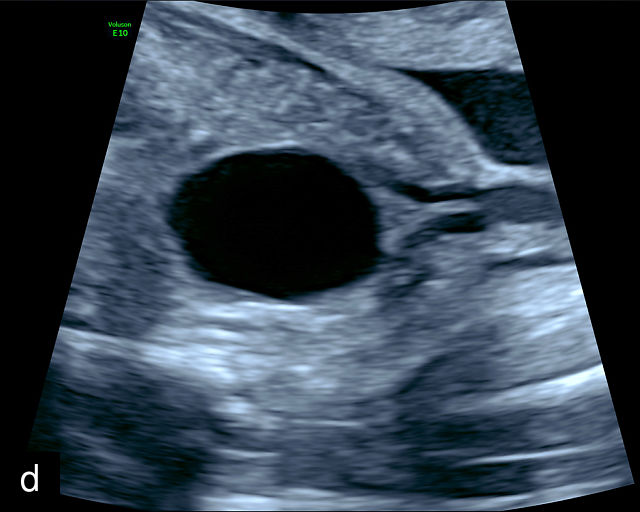
Placental lakes
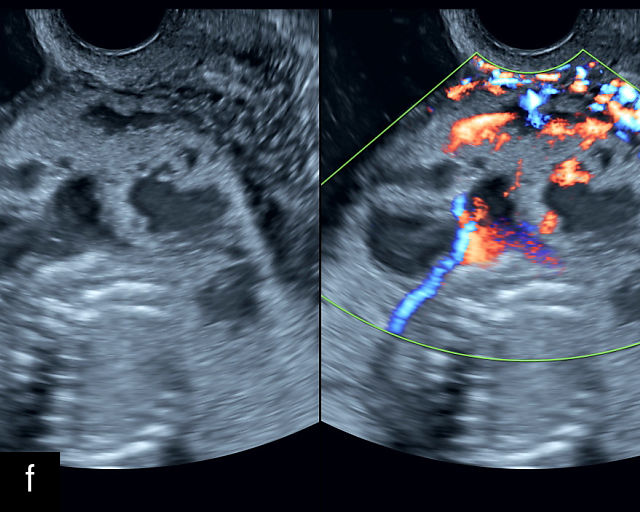
Placental lakes are hypoechoic regions within the placenta and are the most common finding on ultrasound of the placenta (Figure 3; Video 1).2,9,13,16 They are surrounded by placental tissue of normal echogenicity.16 Some reports specify that hypoechoic areas should be at least 2 cm in size to qualify as lakes.13,17 On grayscale ultrasound, swirling of blood can be observed in lakes (Video 1). Low-velocity flow can be demonstrated using sensitive Doppler techniques. Lakes tend to be located in the center of placental cotyledons or under the chorionic plate (Figure 3).16 Other common locations are at the placental edge and between lobes in a bilobed placenta where they may be very large. Lakes tend to fluctuate in size over time. Several studies have found that placental lakes are not associated with an increase in adverse pregnancy outcome, regardless of size and number.13,18,19 However, one study found an increased risk of small-for-gestational age fetuses with larger placental lakes.20 Lakes must be differentiated from lacunae, which contain high-velocity blood flow and are associated with placenta accreta spectrum.16


3
(a–f) Placental lakes. Multiple hypoechoic spaces present in placenta (p), surrounded by placental tissue of normal echogenicity. Arrows in (e) and (f) indicate lakes.
1
(a–c) Placental lakes. Multiple hypoechoic spaces present in placenta, surrounded by placental tissue of normal echogenicity. The spaces demonstrate low-velocity swirling, visible on grayscale ultrasound.
Accessory placental lobes
While most placentas consist of a single disc with several cotyledons, some placentas may have two or more separate lobes. These variations can include a bilobate placenta, where two lobes of roughly equal size are present, or a main placental body accompanied by one or more smaller accessory or succenturiate lobes (Figure 4).21,22,23,24 The cord insertions in these pregnancies are often velamentous or marginal, and blood vessels always traverse the membranes between the placental lobes. When these connecting vessels pass over the cervix, the condition is classified as Type- 2 or Type- 3 vasa previa.25,26,27,28,29,30
4
Accessory placental lobes. (a) Color Doppler transabdominal ultrasound image depicting a bilobed placenta with an anterior (a) and posterior (p) lobe, into which the cord inserts. (b) Grayscale ultrasound with color flow Doppler showing a bilobed placenta with anterior (a) and posterior (p) lobes. Color flow Doppler shows unprotected fetal vessels traversing the membranes between the lobes (arrow). (c) Grayscale ultrasound showing a bilobed placenta with anterior (a) and posterior (p) lobes.
The sonographic features of a bilobed or succenturiate-lobed placenta include the presence of two distinct placental masses, typically one anterior and the other posterior, which may vary in size or be of equal size.21,22,23,24,26,31,32,33 These masses are connected by blood vessels, which can be visualized using color flow Doppler (Figure 4).28
It is crucial to confirm that the masses are truly separate and not connected as part of a single lateral placenta, which may be mistaken for a bilobate placenta. Additionally, careful scanning should verify that both masses share similar echogenicity to distinguish true placental tissue from other structures such as subchorionic hematomas, myometrial contractions or uterine fibroids, which can mimic accessory lobes. Prominent placental lakes may also be observed between the lobes.23,26
The placental cord insertion in these cases is often velamentous and may be located between the placental lobes (Figure 4b; Video 2). In rare cases, furcate insertion, in which the cord vessels branch before reaching the placental tissue, between the lobes has been reported.24
2
Accessory placental lobes. (a) Grayscale (left) and color flow Doppler (right) ultrasound of bilobed placenta with cord insertion between the placental lobes. The cord insertion is marginal into the anterior lobe and unprotected vessels run through the membranes for a short distance into the posterior placental lobe. (b) Grayscale ultrasound showing a bilobed placenta.
Pregnancies conceived via assisted reproductive techniques, particularly in-vitro fertilization, are at increased risk of developing placentas with accessory lobes.22 Bilobate placentas have also been linked to an elevated risk of pre-eclampsia, though risks for small-for-gestational-age infants and preterm birth were not significantly increased.22
In most cases, pregnancies with accessory lobes have favorable outcomes in the absence of vasa previa. However, because accessory lobes are strongly associated with vasa previa, transvaginal ultrasound with color Doppler imaging is recommended whenever they are identified.3,4,25,28,34,35,36
At delivery, it is critical to examine the placenta thoroughly to ensure that it is complete and that no lobes have been retained. Retained accessory lobes can lead to postpartum hemorrhage, a potentially life-threatening complication.22
Placental grading
Placental grading was introduced by Grannum et al. as a method for assessing fetal lung maturity.37 They classified the sonographic appearance of the placenta into four grades. Grade 0 is characterized by a smooth chorionic plate with a uniform, homogeneous texture. Grade I shows mild undulations of the chorionic plate with scattered echogenic areas within the placental texture. Grade II is marked by linear hyperechoic calcifications within the basal plate (Figure 1f). Grade III features extensive calcifications distributed throughout the contours of the cotyledons (Figure 1g).
However, Quinlan and colleagues found no strong correlation between placental grading and fetal lung maturity.38 Their study reported that only 5% of pregnancies with confirmed fetal pulmonary maturity on amniotic fluid analysis had a Grade-III placenta. Additionally, placental Grade III on ultrasound incorrectly predicted fetal pulmonary maturity in 42% of cases. Another study found that, even beyond 40 weeks of gestation, only 20% of pregnancies exhibited a Grade-III placenta.39 Maternal smoking has been associated with Grade-III placentas.40
Subsequently, Vintzileos and colleagues proposed incorporating placental grading into a modified biophysical profile to assess fetal wellbeing.41,42 However, placental grading is no longer considered a primary tool for fetal assessment, partly due to poor inter- and intraobserver reliability in ultrasound interpretation.43,44,45 In addition, placental grading alone is a poor predictor of adverse perinatal outcomes.45,46 Despite this, some studies have suggested that the presence of a Grade-III placenta before term may be associated with an increased risk of adverse perinatal outcome including stillbirth.47,48 Therefore, when a Grade-III placenta is identified on ultrasound before 37 weeks, closer fetal monitoring may be warranted.
Abnormal placental findings
Placenta previa and low-lying placenta
In most pregnancies, the placenta implants in the upper part of the uterus. However, when the placenta implants in the lower uterine segment and overlies the cervix, it results in a condition called placenta previa, meaning the placenta is positioned ahead of the fetus in the birth canal.49 This positioning poses a serious risk during labor, as cervical dilation can cause premature placental separation, leading to severe, life-threatening bleeding for the pregnant patient.50,51 To prevent this hemorrhage, patients with placenta previa must be delivered by cesarean section before the onset of labor.51 Placenta previa complicates approximately 0.4% (1 in 250) of pregnancies at delivery.52 The strongest risk factor for placenta previa is a history of prior cesarean delivery.53,54,55 Maternal smoking, advanced maternal age and assisted reproductive technologies are also strong risk factors for placenta previa.56,57,58,59
Historically, placenta previa was most often identified in the early third trimester when patients presented with painless vaginal bleeding.49,60,61 However, with the widespread use of obstetric ultrasound in the second trimester, nearly all cases of placenta previa are now diagnosed incidentally in asymptomatic patients during routine scans. Notably, approximately 90% of placenta previa cases diagnosed in the second trimester will resolve by term due to placental ‘migration’ as the uterus grows.51,61,62,63,64,65 Thus, when placenta previa is diagnosed in the second half of pregnancy, the patient should have an examination close to delivery to determine whether the placenta previa is persistent or whether it has resolved. Generally, when a placenta previa is found at 20 weeks, a repeat ultrasound should be performed at about 32 weeks. In cases that persist at 32 weeks, a repeat scan should be performed around 36 weeks of gestation.66
In the past, placenta previa was classified as complete, partial, marginal or low-lying.50,61 However, with advancements in ultrasound technology allowing precise assessment of the relationship between the placenta and the internal cervical os, the classification has been simplified.67 Placenta previa now refers to cases in which the placenta overlies the internal os to any degree, while low-lying placenta describes cases in which the lower placental edge lies within 2 cm of the internal os (Figure 5).51,66,67
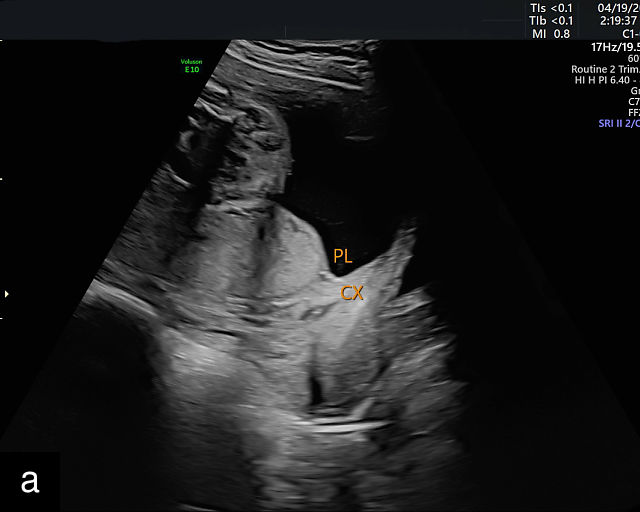
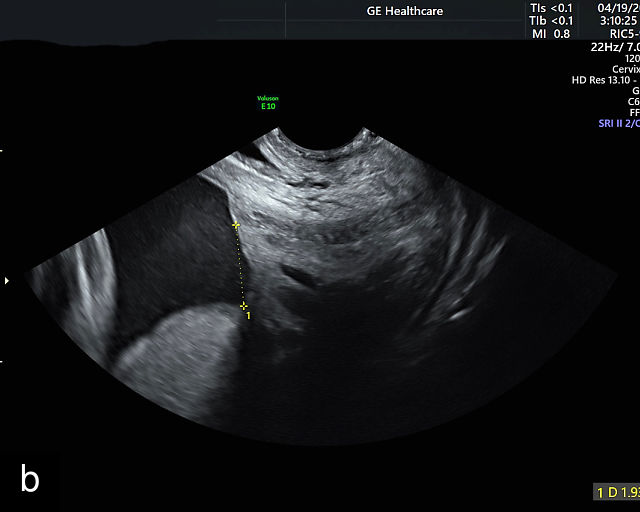

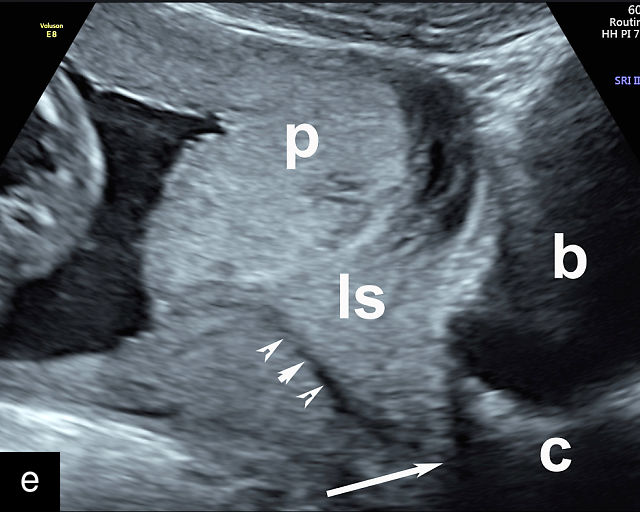
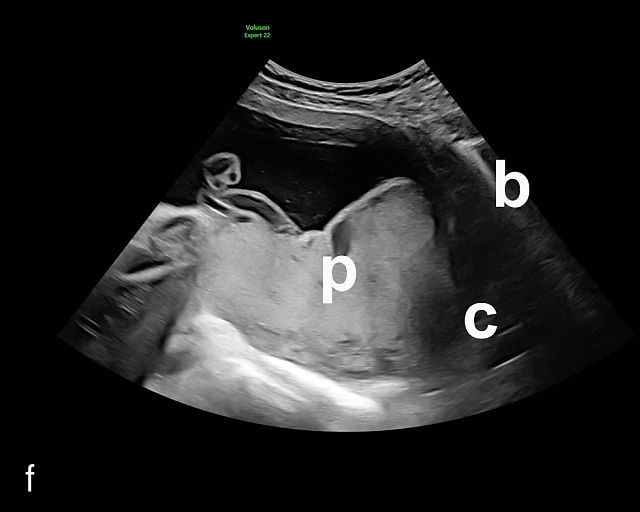
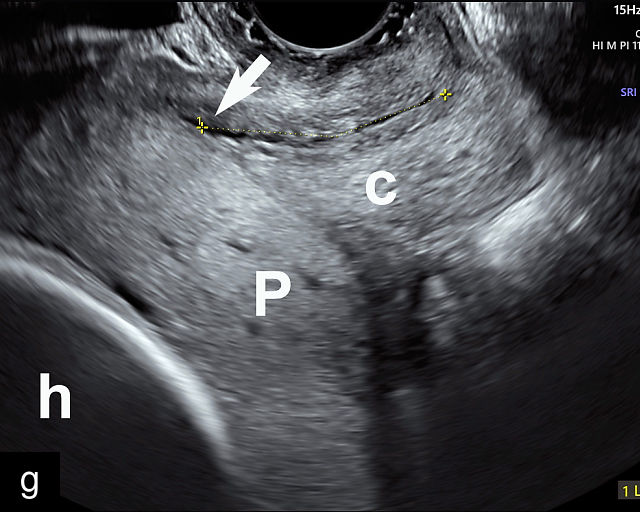
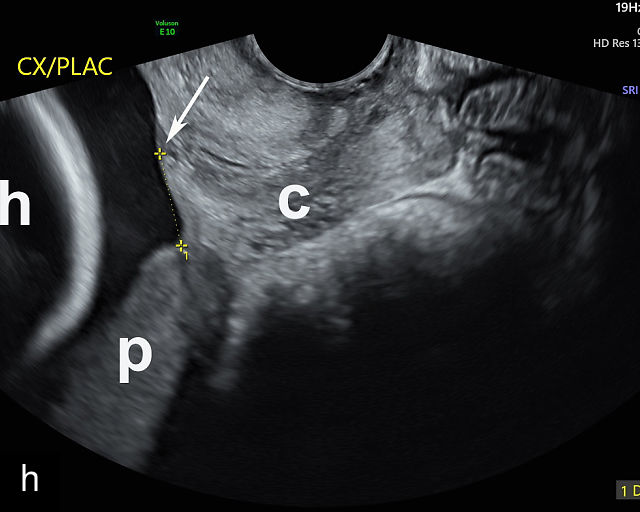
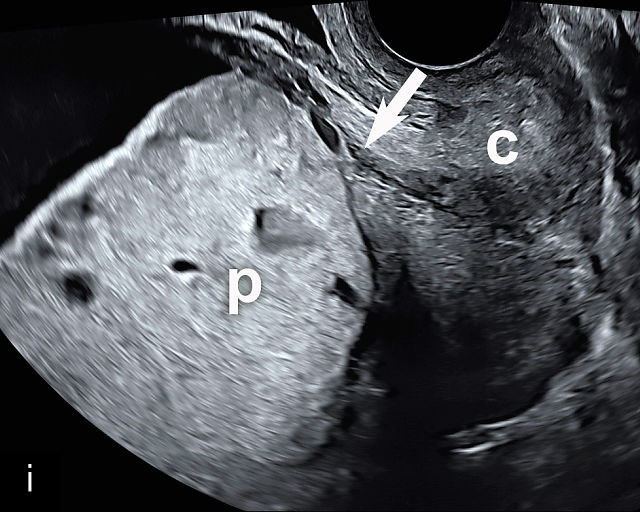
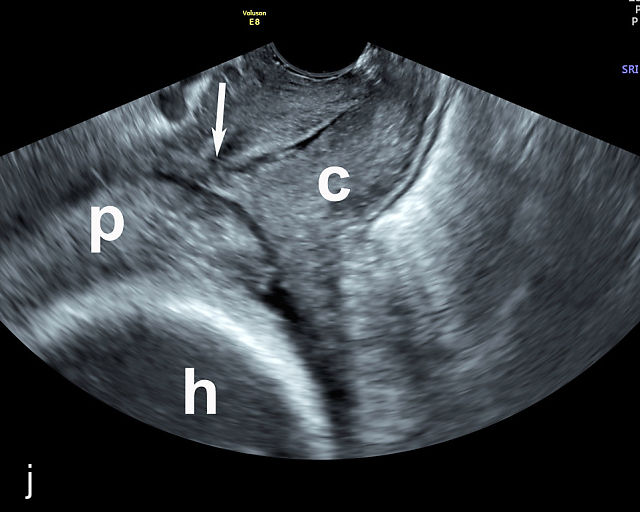


5
Placenta previa and low-lying placenta. (a) Transabdominal grayscale ultrasound image suspicious for placenta previa. The placenta (PL) appears to overlie the internal cervical os (CX). Note the time stamp: 2:19:37. (b) Transvaginal ultrasound of the same patient taken approximately 50 minutes later. Note the time stamp: 3:10:25. The internal os and the lower placental edge are both clearly seen, and the placenta does not overlie the internal os. Because the lower placental edge is 1.93 cm from the internal os, it will likely resolve by the third trimester. (c) Transabdominal grayscale ultrasound of placenta previa. The placenta (p) covers the cervix, but the cervix, especially the internal os, cannot be visualized due to shadowing. (d) Transabdominal grayscale ultrasound of placenta previa. The placenta (p) covers the cervix (c) but shadowing obscures adequate visualization. The internal os is indicated by the arrow. (e) False-positive image of placenta previa on transabdominal grayscale ultrasound. The bladder (b) is full, pushing the anterior and posterior walls of the lower uterine segment (ls) together making it appear that the placenta (p) overlies the internal os of the cervix. In reality, the line depicted by the arrowheads is where the anterior and posterior walls of the lower segment are in proximity to each other. The cervix is much lower and is obscured by shadowing (c). (f) Transabdominal grayscale ultrasound image of placenta previa. The placenta (p) covers the cervix (c), but the cervix, especially the internal os, cannot be visualized due to shadowing. b, bladder. (g) Transvaginal grayscale ultrasound image of placenta previa. The placenta (p) completely covers the internal os (arrow) of the cervix (c). The internal os can be seen clearly. h, fetal head. (h) Transvaginal grayscale ultrasound image of posterior low-lying placenta (p). The lower placental edge is clearly seen and is 1.56 cm from the internal os (arrow) of the cervix (c). The placental edge and the internal os are clearly seen. h, fetal head. (i) Transvaginal grayscale ultrasound image of a posterior placenta previa (p). The internal cervical os is clearly seen (arrow). c, cervix. (j) Transvaginal grayscale ultrasound image of an anterior placenta previa (p). The internal cervical os is clearly seen (arrow). c, cervix; h, fetal head. (k) Transvaginal grayscale ultrasound image of a posterior placenta that was thought to be low-lying on transabdominal sonography but could not be adequately assessed. This examination clearly shows the lower edge of the placenta (p) to be 2.18 cm from the internal os (arrow) of the cervix, firmly establishing that the placenta is not low-lying and allowing the patient to undergo labor safely and deliver vaginally. c, cervix. (l) Transvaginal grayscale ultrasound image of placenta previa. The placenta (p) completely covers the internal os (arrow) of the cervix (c). The internal os can be seen clearly.
This distinction is critical in determining the mode of delivery.49,51 All patients with placenta previa persisting into late pregnancy require cesarean delivery to avoid complications such as severe bleeding.50,60,61 Studies suggest that patients with a lower placental edge located more than 1 cm from the internal os may safely attempt a vaginal delivery without a significant increase in bleeding risk.68,69,70,71
In patients with placenta previa and one or more prior cesarean deliveries, careful evaluation is necessary to rule out placenta accreta spectrum disorders, in which the placenta abnormally adheres to or implants deeply into the uterine wall.50,60
Most cases of placenta previa will be suspected prenatally by transabdominal ultrasound.49 However, this approach has several limitations and may be inaccurate.72,73,74 because the relationship between the placenta and the internal cervical os may be difficult to assess by transabdominal ultrasound.72,73,74 The bladder may be full, pushing the anterior and posterior walls of the lower uterine segment together, falsely creating the impression of a placenta previa (Figure 5e).61 There may be considerable shadowing, including by the fetal presenting part, which may limit the accuracy of transabdominal ultrasound (Figure 5d,f).75 Posterior placentas may be more difficult to assess.
Transvaginal ultrasound overcomes these limitations (Figure 5g–l).72,73,74,76 The probe is inserted into the vagina and therefore is closer to the region of interest.51,61 In addition, transvaginal transducers have higher frequencies and superior resolution compared to transabdominal transducers. Transvaginal ultrasound is safe and is not associated with increased bleeding.72,73,74,75,76 As such, transvaginal ultrasound should be the imaging modality of choice whenever there is suspicion of placenta previa.49,67,77
Placenta accreta spectrum
Placenta accreta spectrum (PAS) refers to abnormal implantation of the placenta into the uterine wall, where the normal plane of separation, known as Nitabuch’s layer, is absent.50,60 As a result, the placenta cannot detach from the uterine wall after delivery, leading to severe and potentially life-threatening hemorrhage.50,60,61 Fortunately, the condition can be diagnosed by ultrasound prenatally, allowing appropriate management to avoid severe maternal morbidity and mortality (Figure 6a–h; Video 3).



6
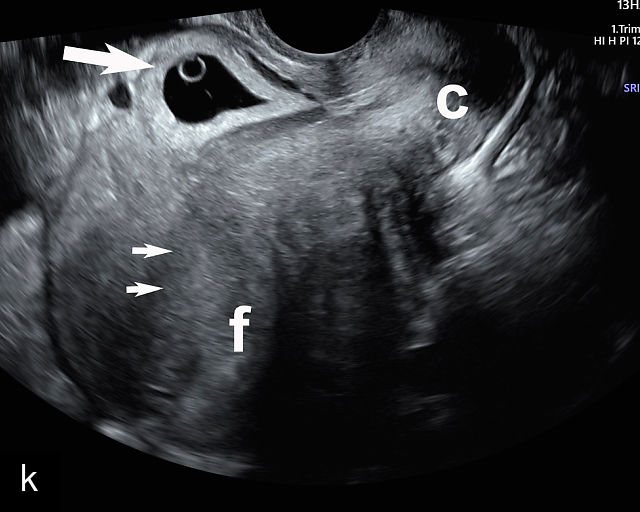
Placenta accreta spectrum. (a,b) Sagittal transabdominal grayscale ultrasound of the lower uterus and cervix demonstrating placenta previa accreta, with the placenta (p) containing prominent irregular hypoechoic lacunae. There is absence of the myometrium at the bladder (b) interface (arrows) c, cervix. (c) Sagittal transabdominal color Doppler ultrasound of the lower uterus and cervix demonstrating placenta previa accreta, with increased vascularity of the lower uterus and the myometrial bladder interface. (d) Transvaginal ultrasound of placenta previa accreta showing multiple lacunae involving the cervix with no clear demarcation between the placenta and the cervix. (e) Transvaginal ultrasound of placenta previa accreta showing multiple large irregular lacunae involving the cervix with no clear demarcation between the placenta (p) and the cervix. The placenta has a ‘moth-eaten’ appearance. There is loss of myometrium (arrows). (f) Transvaginal ultrasound of placenta previa accreta without and with color flow Doppler showing multiple large irregular lacunae. The placenta has a ‘moth-eaten’ appearance. There is hypervascularity of the myometrial interface. (g) Transvaginal ultrasound of placenta previa accreta showing multiple lacunae involving the cervix with no clear demarcation between the placenta and the cervix. (h) Transvaginal ultrasound of placenta previa accreta with color flow Doppler showing hypervascularity of the myometrial interface and irregularity of the bladder wall. b, bladder; p, placenta. (i) Transabdominal grayscale ultrasound of cesarean scar pregnancy. The uterine fundus (f) is empty (arrowhead). The gestational sac (arrow) is located in the lower uterus, anterior to the uterine canal, and lies above the cervix (c). b, bladder. (j) Transvaginal grayscale ultrasound image of cesarean scar pregnancy showing the empty fundus (f) and the normal cervix (c). The gestational sac lies below the fundus, above the cervix and anterior to the uterine cavity. (k) Transvaginal grayscale ultrasound image of cesarean scar pregnancy showing the empty cavity (arrowheads) of the uterine fundus (f), and the normal cervix (c). The gestational sac lies below the fundus, above the cervix and anterior to the uterine cavity (arrow).
3
Placenta accreta. Transvaginal ultrasound shows placenta previa with multiple irregular lacunae in the placenta and irregularity of the uterine wall and myometrium–bladder interface.
Historically, PAS was classified into three distinct categories: placenta accreta, where the placenta is superficially attached to the myometrium; placenta increta, where it invades deeper into the myometrium; and placenta percreta, where it penetrates through the serosal surface and may extend into adjacent structures such as the bladder, bowel or parametrium.60,61 These classifications are now grouped under the umbrella term 'placenta accreta spectrum'.
Nearly all cases of PAS occur in the presence of placenta previa and a history of one or more prior cesarean deliveries, with the risk increasing as the number of previous cesareans rises.78,79,80 All women with both these risk factors should have screening for PAS.81,82,83,84 There is strong evidence that most cases of PAS originate as first-trimester cesarean scar pregnancies, which can be identified by ultrasound early in pregnancy (Figure 6i–k).83,85,86,87,88,89 In these cases, the embryo implants within or on the scar from a prior cesarean section. On first-trimester ultrasound, this appears as a gestational sac implanted low and anterior in the uterine cavity, with an empty endometrial cavity in the fundus (Figure 6i–k).90
Early recognition of cesarean scar pregnancy and PAS is crucial, as prenatal diagnosis and appropriate delivery planning significantly impact outcomes.50,60,84 When PAS is not diagnosed before birth, it frequently results in severe maternal morbidity, including massive hemorrhage, multi-organ failure, and the need for extensive surgical intervention.61,91 It is also associated with a substantial maternal mortality rate, particularly in cases in which delivery occurs in an inadequately prepared setting without a multidisciplinary surgical team experienced in PAS management.92
The standard approach to management involves scheduled cesarean delivery before labor, followed by hysterectomy with the placenta left in situ to prevent catastrophic hemorrhage.60,61 However, an increasing number of reports describe successful conservative management strategies that allow for uterine preservation without hysterectomy.93,94
Several characteristic sonographic findings are associated with PAS.90,95,96,97 These include placental lacunae, which appear as hypoechoic, irregular spaces within the placenta with high-velocity turbulent flow on color Doppler.16,50 Other features include obliteration of the retroplacental clear space, thinning of the myometrium in the lower uterine segment, irregularity and hypervascularity of the myometrial–bladder interface, and placental bulging into the bladder (Figure 6a–h).90,96,98,99
A more detailed discussion of placenta accreta spectrum is available in another chapter in this volume (Anomalies of Placentation: Placenta Accreta Spectrum Disorders | Article | GLOWM) and will not be expanded upon further here.
Abnormal placental thickness
Thick placenta
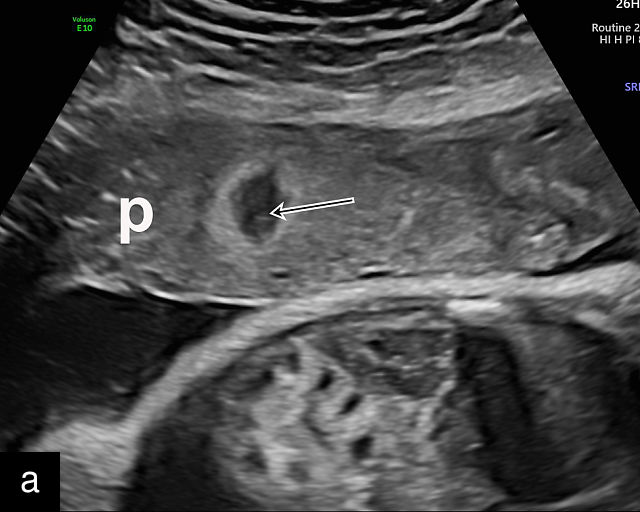
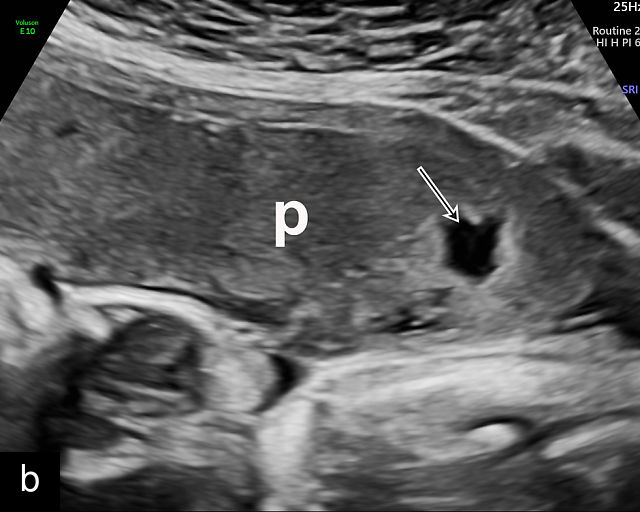
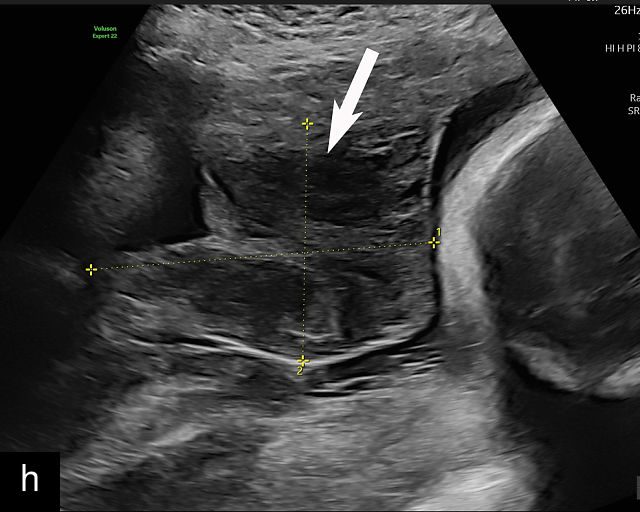
The normal placental thickness is approximately 1 mm for each week of gestational age after 20 weeks, and it generally does not exceed 40 mm.100 However, varying definitions of placental thickness have led to inconsistent estimates of the prevalence of a thick placenta, ranging from 0.6% to 7.6%.101 The term ‘placentomegaly’ is commonly used to describe a thickened placenta, which should be measured sagitally and under the umbilical cord insertion, if this is central (Figure 7a). Thickened placentas may result from a variety of fetal, maternal or placental conditions.101,102


7
Thick placenta (p). (a) Placentomegaly in a pregnancy affected by parvovirus with severe fetal anemia. (b) Thickened placenta in a patient with syphilis. (c,d) Thickened placenta in two patients with cytomegalovirus infection. (d) Note the placental calcifications (white spots).
Fetal causes of placentomegaly include hydrops fetalis, which may be immune or non-immune, hemoglobinopathy, particularly fetal thalassemia, and fetal infection, such as cytomegalovirus (CMV), toxoplasmosis, rubella, syphilis, Zika virus and malaria (Figure 7).2,9,103 Structural abnormalities, such as sacrococcygeal teratoma and genetic conditions like Beckwith–Wiedemann syndrome, have also been associated with thickened placenta.104 Maternal conditions, including pre-eclampsia, diabetes and anemia, may also contribute. In monochorionic twin pregnancies, placentomegaly may occur in the context of twin–twin transfusion syndrome (TTTS) or twin anemia–polycythemia sequence (TAPS). PAS disorders are another potential cause of placental thickening. Heterogeneous thickening, specifically, may result from placental mesenchymal dysplasia or placental hemorrhage.105,106 Laterally positioned placentas are more likely to be measured as thickened, which may reflect either challenges in measurement or compensatory hypertrophy due to impaired blood flow. Anterior placentas tend to be thinner than posterior or fundal ones.107
When a thickened placenta is detected on ultrasound, a detailed sonographic examination of both the fetus and placenta is essential, including an assessment of amniotic fluid volume.101 Tests for fetal infection may be indicated, with specific investigations tailored to the clinical context, and antibody screening should be reviewed to rule out alloimmunization. Thickened placentas are associated with an increased risk of adverse outcome, including fetal anomalies, fetal growth restriction (FGR), perinatal acidemia, fetal death and preterm delivery.104 Management should focus on addressing the underlying condition when an etiology is identified. In cases in which no specific cause is found, serial growth scans and fetal surveillance in the late third trimester are recommended to monitor for potential complications.101
Thin placenta
Polyhydramnios may lead to placental compression and a sonographically thin placenta. This may occur in the recipient twin in TTTS. The placental compression may result in uteroplacental insufficiency. A thin placenta covering the entire uterine surface may be found in placenta membranacea, also known as placenta diffusa.108 This condition is rare, estimated to occur in 1 : 20 000 pregnancies.109 It may result in bleeding in pregnancy, FGR and retained placenta. If the placenta appears unusually thin on ultrasound, a detailed anatomical survey should be performed, and consideration given to serial ultrasounds to evaluate fetal growth and wellbeing.
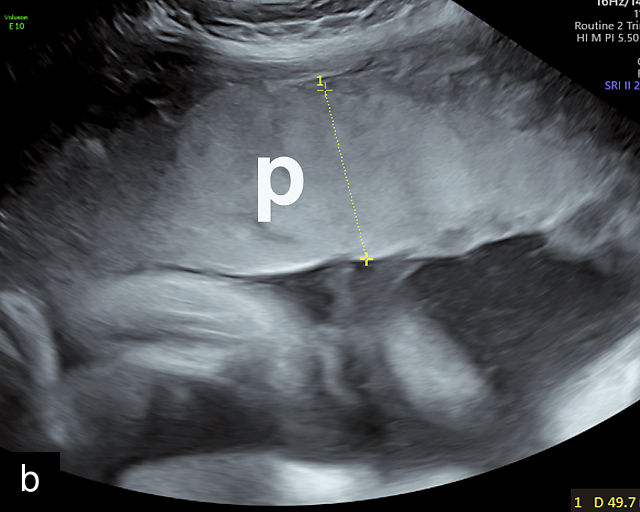
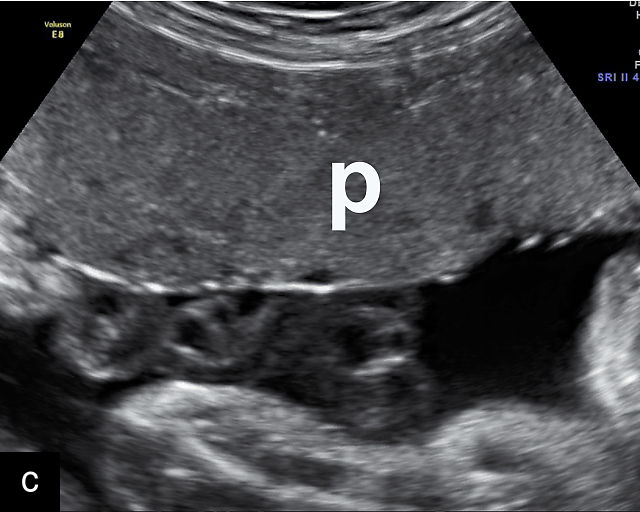
Placental infarcts
Placental infarcts are hypoechoic structures within the placental body with hyperechoic rims.2,9,16 (Figure 8; Video 4). The central hypoechoic region will not demonstrate any flow using sensitive Doppler techniques (Figure 8d).9,15 In addition, they do not change size or shape over time. It is these three findings (absence of flow, the hyperechoic rim and stability in size over time) that differentiate them from placental lakes.2,9 Small infarcts are found in 20% of normal pregnancies and are generally of no consequence.2,9,15 However, multiple infarcts may be found in pregnancies with pre-eclampsia and FGR, and may be associated with increased risk for abnormal umbilical artery Doppler velocimetry and adverse outcomes.2,9

8
(a–d) Placentas (p) showing infarcts (arrows). Note the hypoechoic structures with hyperechoic rims. There is no flow demonstrable with color Doppler (d).
4
(a–c) Placental infarcts. Videoclips show multiple hypoechoic placental structures with hyperechoic rims, consistent with infarcts.
Placental abruption and hemorrhagic lesions of the placenta
Placental abruption, which complicates 0.5–1% of births, refers to the premature separation of a normally implanted placenta before the delivery of the baby.110,111,112 As the placenta is the primary source of oxygen and nutrients for the fetus, this separation poses significant risks, including fetal death or asphyxia.111,112 Placental abruption is also a major cause of preterm birth.113,114,115 Risk factors for abruption include hypertensive disorders of pregnancy, smoking, cocaine use, trauma and preterm prelabor rupture of membranes.110,116
Classically, the diagnosis of placental abruption is based on clinical symptoms such as vaginal bleeding, abdominal pain, uterine tenderness, contractions and signs of fetal distress.110,111,112 When more than 50% of the placenta separates, stillbirth is often the outcome.112,114 Maternal complications can also be severe and include disseminated intravascular coagulation (DIC), renal failure and the need for intensive care unit (ICU) admission.110,112,115
While the diagnosis of placental abruption is generally considered clinical, ultrasound plays an important adjunctive role.111,117 Historically, the criteria for diagnosing abruption relied on symptoms; however, in some cases, ultrasound findings may precede clinical manifestations.118,119 The definitive sonographic finding of placental abruption is retroplacental hematoma.117,120 However, various hemorrhagic placental lesions may be observed in both symptomatic and asymptomatic patients, suggesting these findings exist along a continuum that culminates in the clinical presentation of abruption.110,111,112,117,120
Ultrasound findings associated with placental abruption include (Figure 9a–l):117,120,121,122,123,124 retroplacental hematoma (Figure 9a,b; Video 5c,f); subchorionic hematoma (Figure 9c–f,h,j; Video 5d,e); heterogeneous thickened placenta and intraparenchymal hematoma (Figure 9g; Video 5a); intra-amniotic hematoma (Figure 9i); and preplacental hematoma located beneath the chorionic plate (Figure 9k,l).125




9
Placental abruption and hemorrhagic lesions of the placenta (p). (a,b) Grayscale transabdominal ultrasound showing a retroplacental hematoma (arrow). (c–f) Transabdominal grayscale ultrasound image showing an anterior subchorionic hematoma (arrow). In (c) and (f), the posterior placenta is marked by 'p'. (g) Transabdominal ultrasound showing a thickened heterogeneous placenta in a patient with clinical abruption. The arrow points to the intraparenchymal hematoma. (h) Transabdominal grayscale ultrasound image showing an anterior subchorionic hematoma (arrow). Note the heterogeneous appearance with ‘layering’. (i) Large intra-amniotic hematoma (arrow). Note the ‘layering’. (j) Posterior subchorionic hematoma (arrow). The anterior placenta is marked by ‘p’. (k,l) Posterior placenta (p) with preplacental hematoma (arrow) of the chorionic plate (also known as Breus’ mole).
5
Placental abruption and hemorrhagic lesions of the placenta. (a,b) Posterior placenta containing an intraparenchymal hematoma. Note the ‘layering’ caused by organization of the clot. (c) Anterior placenta containing a retroplacental hematoma. Note the ‘layering’ caused by organization of the clot. (d,e) Anterior subchorionic hematoma. (f) Anterior placenta containing a retroplacental hematoma. Note the ‘layering’ caused by organization of the clot.
Importantly, the appearance of the hematomas may evolve over time as the clot organizes, varying from hypoechoic, when it has recently occurred to heterogeneous with layering and echogenic when it changes structure (or becomes organized) over time. Understanding this spectrum of ultrasound findings is crucial for identifying placental abruption, particularly in its earlier stages, and optimizing management to improve maternal and fetal outcomes.117,120 When these sonographic findings are encountered in the presence of a clinical diagnosis of abruption, the specificity for abruption is over 90%.123,124 Studies on ultrasound diagnosis of abruption have shown considerable variability in their sensitivity, from about 30% to 80%, with most suggesting that ultrasound has poor sensitivity for diagnosing abruption.110,126 However, these studies are limited because of the wide variety of what is considered sonographic findings diagnostic of abruption.123 Some consider only a retroplacental hematoma to be a sonographic finding consistent with abruption.117,120,127 However, when Yeo and colleagues assessed multiple ultrasound findings, listed above, in symptomatic patients, they found a sensitivity of 80%.123 Importantly, the retroplacental complex which is hypoechoic, or the presence of a localized subplacental contraction, should not be mistaken for abruption, nor should uterine fibroids.
Placental calcifications
Scattered placental calcifications can be associated with fetal infections such as toxoplasmosis, rubella, herpes, SARS-CoV-2, Zika virus and cytomegalovirus (Figure 10).128,129,130,131,132 When identified, a detailed fetal sonographic evaluation is warranted, and serologic testing for fetal infection should be considered.

10
Scattered placental calcifications in a case of congenital cytomegalovirus infection. The placenta is also thickened.
Placental cysts
Placental cysts are relatively uncommon and are most often located on the fetal surface of the placenta or along the chorionic plate.133,134,135,136,137 When identified, it is important to assess them carefully to rule out hemorrhagic lesions, as hematomas resulting from ruptured chorionic vessels can lead to fetal anemia.138,139 This distinction can typically be made sonographically: true cysts appear anechoic, whereas hematomas tend to contain echogenic fluid. Placental cysts are generally associated with favorable pregnancy outcomes; however, some case reports have noted an association with FGR.137,140 Thus, serial ultrasound evaluations are recommended to monitor for any changes in the cysts and to assess fetal growth (Figure 11; Video 6).

11
Placental cysts. (a–d) Hypoechoic cystic structures on the fetal surface of the placenta. (e) Hypoechoic cyst within the placental parenchyma.
6
Placental surface cysts.
Placental chorioangiomas
Chorioangiomas are benign tumors of the placenta, arising from chorionic tissue.141 These tumors consist of capillaries and cellular stroma in varying proportions.141,142 While clinical diagnosis of chorioangiomas is relatively uncommon, histopathological analysis reveals their presence in approximately 1% of all placentas.142 Most chorioangiomas identified on prenatal ultrasound are incidental findings during routine examination and are typically those that are larger in size.9 Consequently, the prevalence of chorioangiomas detected sonographically is significantly lower than the prevalence observed in pathological examinations of placentas. More rarely, chorioangiomas may be detected during ultrasound evaluation for polyhydramnios or FGR.143 Large chorioangiomas (greater than 4 cm in diameter) are thought to occur in approximately 1 : 3500–1 : 9000 pregnancies (0.01–0.03%).141 These larger chorioangiomas may be associated with significant complications, including polyhydramnios, fetal hydrops, fetal anemia, FGR and even fetal death.9,141,143,144,145 A systematic review reported a fetal death rate of 8.2% in cases of prenatally diagnosed chorioangioma in which no intervention was performed.145 Additionally, preterm birth before 37 weeks occurred in 34.1% of these pregnancies (95% CI, 21.1–48.3%). The study also found that 24.0% of infants (95% CI, 13.5–36.5%) were born small-for-gestational age.145 The most important predictor of outcomes was the size of the tumor.145
The typical sonographic appearance of a chorioangioma on grayscale ultrasound is a well-circumscribed mass arising from the fetal surface of the placenta, protruding into the amniotic cavity, and distinctly separate from the placenta (Figure 12; Video 7). These tumors are most commonly located near the umbilical cord insertion site on the placenta. Sonographically, they may appear hypoechoic or hyperechoic and are often heterogeneous, with possible calcifications, hemorrhage or infarction visible within the mass.9 The appearance of these masses may change over time.141 Color flow Doppler imaging typically reveals vascularity within the tumor, showing low-resistance vessels and arteriovenous shunts (Figure 12c–e; Video 7).9 These shunts are believed to contribute to fetal complications such as high-output cardiac failure, anemia and hydrops.141 Three-dimensional ultrasound may be helpful in assessment of the mass.146 Importantly, the differential diagnosis includes placental hemorrhage, and color flow Doppler is essential to assist in making the diagnosis.


12
Grayscale (a,b), color Doppler (c,e) and power Doppler (d) images of chorioangiomas, showing heterogeneous masses protruding from the placental surface.
7
(a–c) Color flow and power Doppler imaging of placental chorioangiomas.
When a chorioangioma is identified on prenatal ultrasound, close fetal surveillance is essential. Initial monitoring includes weekly ultrasound examinations to assess amniotic fluid volume and fetal cardiac function, as polyhydramnios and hydrops can develop rapidly. Polyhydramnios is the most common complication of chorioangioma, complicating between 14% and 28% of cases, and when severe, may lead to maternal discomfort, respiratory embarrassment and preterm labor.141,143,147,148 Fetal growth should be evaluated every 4 weeks. Maternal mirror syndrome, with severe pre-eclampsia has also been described.149,150
Management of complications related to chorioangioma may include interventions such as amnioreduction to manage severe polyhydramnios.149,151 More recently, a variety of fetal surgical procedures have been developed to treat these tumors, with varying degrees of success. Additional interventions include intrauterine fetal transfusion to correct anemia and various fetoscopic techniques to occlude tumor vessels, such as embolization with injection of vascular plugs, and radiofrequency or laser ablation of the feeding vessels.143,144,152,153,154,155,156,157
Rare placental tumors
Rarer placental tumors include teratomas and choriocarcinomas.158 Metastatic malignancies have rarely been found in the placenta, though they should be considered in the differential diagnosis when placental masses are found on ultrasound.
Circumvallate placenta
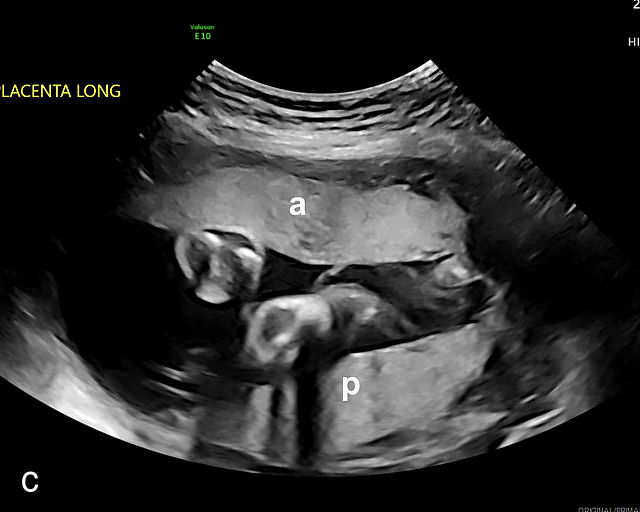
A circumvallate placenta is one in which the chorionic plate (the fetal surface of the placenta) is smaller than the basal plate, resulting in placental tissue extending beyond the margins of the membrane insertion.9 This creates rolled edges with a cuplike appearance. Rather than inserting at the edges of the placenta, the membranes insert into the fetal surface. In a review of 16 042 placental pathology reports collected over 13 years, Stuijt et al. found that circumvallate placentas were present in 2.2% of cases.159 Circumvallate placentas have been associated with an increased risk of adverse pregnancy outcomes, including FGR, placental abruption, preterm birth and fetal demise.160,161
Sonographically, a circumvallate placenta appears as a placenta with raised, rolled edges, which can be identified on grayscale ultrasound (Figure 13). Additionally, a placental shelf or band may be observed at the placental margin. The differential diagnosis includes amniotic bands, uterine synechiae and uterine septa. However, achieving an accurate prenatal diagnosis can be challenging. An early study by Harris et al. found a poor correlation between the sonographic diagnosis of circumvallate placenta and confirmation on placental examination after delivery.162 In that study, placental sonograms reviewed by expert sonologists showed that 17 of 49 normal placentas were incorrectly diagnosed as probably or definitely circumvallate by one or more observers. Conversely, in a study of 10 patients in which prenatal ultrasound had revealed raised rolled placental edges thought to represent circumvallate placenta, the diagnosis was confirmed on gross placental examination in eight cases.163 This high false-positive rate poses a significant challenge in prenatal ultrasound diagnosis and impacts the interpretation of reported outcomes. The difficulty in achieving an accurate diagnosis likely contributes to the wide range of reported prevalence of circumvallate placenta.

13
(a–e) Grayscale images of circumvallate placentas showing the rolled edges of the placentas.
Recent evidence provides additional insight into the clinical significance of this condition. A retrospective cohort study by Herrera et al. examined 179 patients with a prenatal diagnosis of circumvallate placenta at an average of 19.8 weeks.164 The study found no correlation between a circumvallate placenta and adverse pregnancy outcome. These authors suggested that no additional surveillance was necessary when a circumvallate placenta is suspected on prenatal ultrasound. This difference in reported outcomes may reflect the false-positive diagnoses of circumvallate placenta in sonographic studies.
When findings consistent with a circumvallate placenta are observed on prenatal ultrasound, efforts should focus on achieving an accurate diagnosis. The use of three-dimensional ultrasound may improve diagnostic accuracy.9,165,166 If a circumvallate placenta is confirmed or strongly suspected, serial growth scans and consideration of antepartum fetal surveillance in the late third trimester are prudent to monitor for potential complications.
Placental mesenchymal dysplasia
Placental mesenchymal dysplasia (PMD) was first described by Moscoso et al. in 1991.167 It is an uncommon placental vascular abnormality characterized by mesenchymal stem villous hyperplasia, edema and cystic dilation.9,105,168 PMD complicates approximately 1 in 500 pregnancies (0.2%) and is often associated with placental mosaicism.9 There is a 3.6 : 1 female: male preponderance. Maternal serum alpha-fetoprotein (AFP) levels may also be markedly elevated in these pregnancies.169 Despite its clinical significance, PMD is underreported, and many sonographers, obstetricians and pathologists are unfamiliar with the condition, often confusing it with a hydatidiform mole.106,153,170,171,172
PMD is strongly associated with adverse pregnancy outcomes, including pre-eclampsia, FGR and fetal demise.106,173 It has also been found in association with Beckwith–Wiedemann syndrome and various congenital anomalies, including skeletal dysplasias and CHARGE syndrome.106,174,175,176 A systematic review by Nayeri et al. reported that only 9% of pregnancies with PMD resulted in a normal outcome for the baby106.
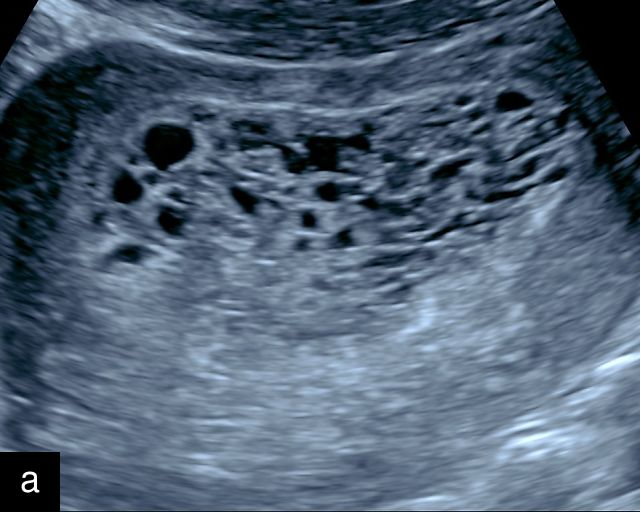
On ultrasound, PMD appears as a thickened placenta with multiple small cysts of varying size, creating a 'snowstorm' appearance that closely resembles a hydatidiform molar pregnancy (Figure 14; Video 8).9,105,106,169,172 Differentiating PMD from a molar pregnancy can be challenging, particularly in twin pregnancies in which a normal twin coexists with a cotwin that has an enlarged, cystic placenta.169 In complete molar pregnancy, the uterus is entirely filled with cystic tissue, showing a snowstorm appearance without identifiable fetal structures. Partial molar pregnancies, on the other hand, typically feature an enlarged cystic placenta coexisting with an abnormal fetus.105



14
(a–e) Grayscale ultrasound of placental mesenchymal dysplasia showing thickened placentas with multiple round cysts of varying size.
8
(a–c) Ultrasound imaging of placental mesenchymal dysplasia.
Several features can help distinguish PMD from molar pregnancy. In molar pregnancy, human chorionic gonadotropin (hCG) levels are typically dramatically elevated, whereas in PMD, hCG levels are usually normal or only mildly elevated.106,177 Elevated maternal AFP levels are more characteristic of PMD. Doppler imaging in molar pregnancies shows hypervascularity with low-resistance flow, a consequence of trophoblastic proliferation, while vascularity in PMD is generally normal or only mildly increased, with normal resistance. Low-risk results from non-invasive prenatal testing (NIPT) using cell-free DNA and the presence of a normal-appearing fetus further support a diagnosis of PMD and make triploidy or partial molar pregnancy unlikely.
Management of these two conditions differs significantly. Molar pregnancies require uterine evacuation and follow-up with serial hCG measurements to monitor for persistent trophoblastic disease. In contrast, PMD may result in a viable pregnancy but carries greatly increased risk of FGR and fetal demise, necessitating close monitoring and intervention when indicated.106
Molar pregnancy
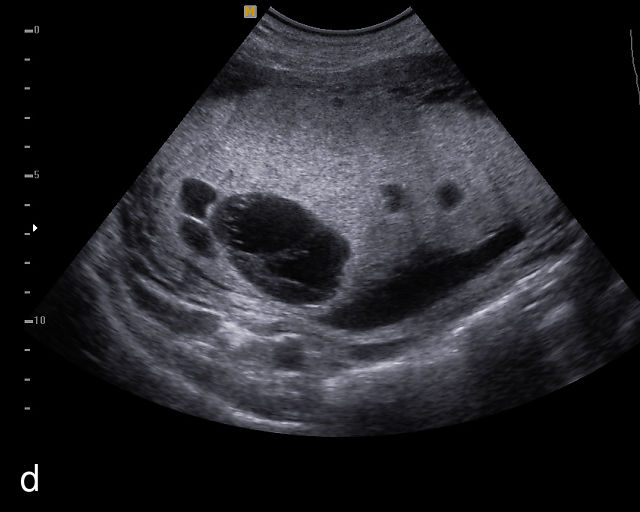
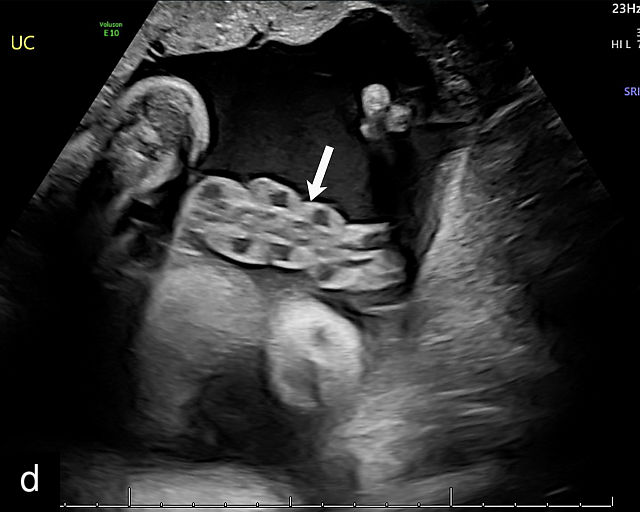
Molar pregnancy, or hydatidiform mole, is a form of gestational trophoblastic disease characterized by abnormal proliferation of trophoblastic tissue and hydropic degeneration of chorionic villi.178,179,180 It is broadly classified into complete and partial types. A complete mole arises from fertilization of an enucleate ovum by one or two sperm, resulting in a diploid karyotype, typically 46,XX, composed entirely of paternal genetic material and lacking an embryo or fetus. A partial mole, by contrast, results from fertilization of a normal ovum by two sperm, leading to a triploid karyotype, most commonly 69,XXY or 69,XXX, and often includes an abnormal fetus.179 Clinically, patients may present with vaginal bleeding, excessive uterine size for gestational age, hyperemesis gravidarum and significantly elevated beta-hCG levels.178 Ultrasound is the diagnostic modality of choice: a complete mole demonstrates a heterogeneous intrauterine mass with numerous anechoic cystic spaces, often described as a ‘snowstorm’ or ‘cluster of grapes’, with no identifiable embryo or amniotic sac (Figure 15).178,181 Color Doppler typically reveals a hypervascular pattern with high-velocity, low-resistance flow due to abnormal trophoblastic vasculature. In partial moles, sonography may reveal a thickened, cystic placenta alongside a growth-restricted fetus, often with structural anomalies such as hydrocephalus, cardiac defects or syndactyly. Doppler evaluation may show signs of placental insufficiency, including absent or reversed end-diastolic flow in the umbilical artery.182 In addition, in molar pregnancies, there may be bilateral enlargement of both ovaries with multiple theca lutein cysts.183 Accurate and timely diagnosis is essential to prevent complications such as persistent gestational trophoblastic neoplasia and to guide appropriate clinical management.181,184
15
Molar pregnancy. (a) Transvaginal grayscale ultrasound at 8 weeks’ gestation demonstrating features of a complete molar pregnancy. The uterus is enlarged and filled with a heterogeneous echogenic mass containing multiple small anechoic cystic spaces, producing a characteristic 'snowstorm' or 'cluster-of-grapes' appearance. No identifiable embryo or normal gestational sac is seen. The myometrium appears normal in echotexture, and there is no evidence of adnexal mass or free fluid. (b) Transabdominal grayscale ultrasound at 9 weeks’ gestation demonstrating a dichorionic twin pregnancy with a complete mole co-existing with a normal appearing embryo in a separate sac. One sac is filled with a heterogeneous echogenic mass containing multiple small anechoic cystic spaces, producing a characteristic 'snowstorm' or 'cluster-of-grapes' appearance.
UMBILICAL CORD
Normal anatomy, development and ultrasound appearance
The umbilical cord serves as the lifeline connecting the fetus to the placenta. It arises from the midportion of the fetal abdomen and typically inserts into the center of the placenta (Figure 16). The average length of the umbilical cord is approximately 55 cm (22 inches).185 It contains three vessels: two arteries, which originate from the fetal left and right internal iliac arteries, and one vein, collectively referred to as a three-vessel cord.


16
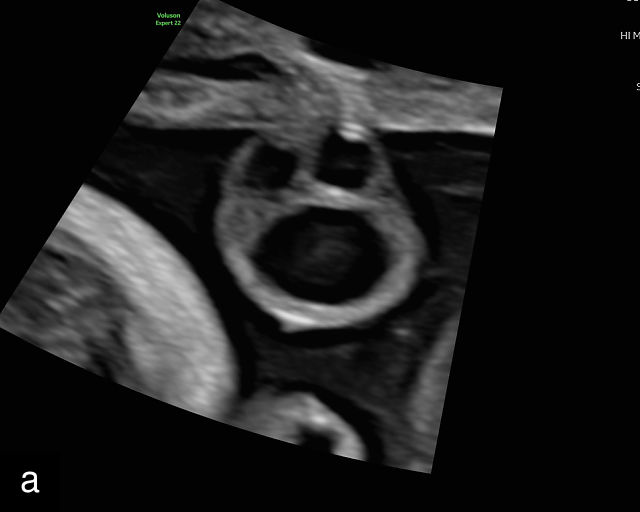
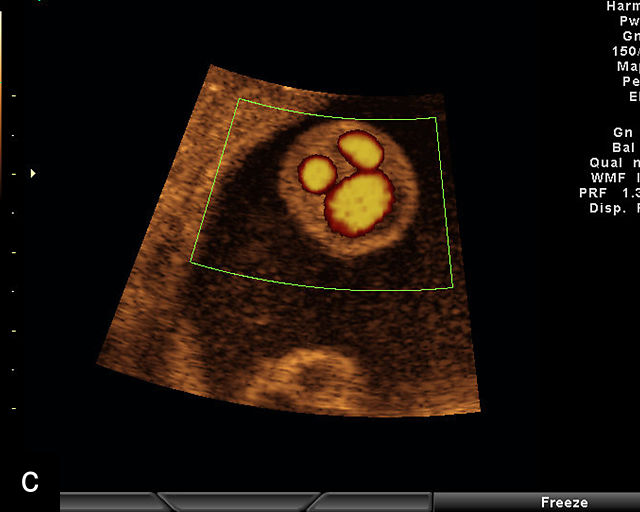
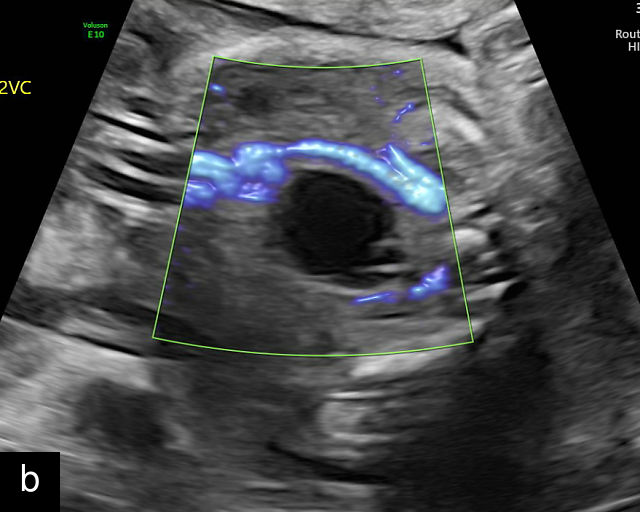
Normal appearance of the umbilical cord with two umbilical arteries and a single vein. (a–c) Cross-section of the umbilical cord on grayscale (a,b) and power Doppler (c) ultrasound. In the normal cord, three vessels are visible: a larger umbilical vein and two smaller umbilical arteries. This characteristic appearance is often referred to as the 'Mickey Mouse' sign, the larger circle (umbilical vein) representing Mickey’s face while the smaller circles (arteries) form his ears. (d–g) Grayscale (d) and color Doppler (e–g) images of transverse section of the lower fetal abdomen showing the two umbilical arteries diverging around the fetal bladder. (h) Longitudinal ultrasound view with color Doppler of a normal umbilical cord showing three vessels present in each coil: two arteries with flow in one direction and a single vein with flow in the opposite direction.
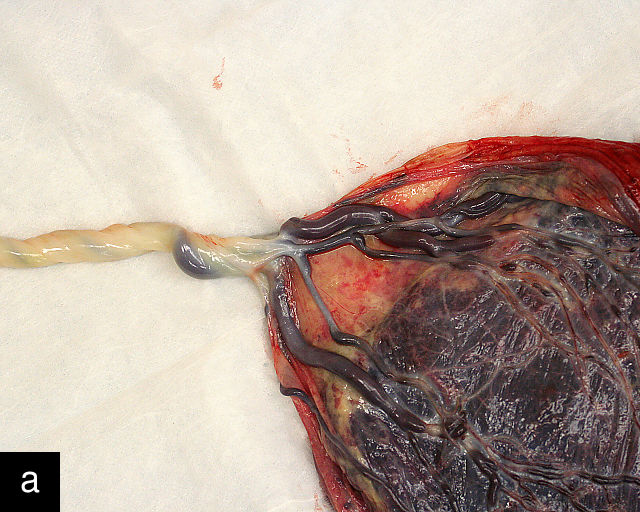
The umbilical vein carries oxygenated blood from the placenta to the fetus, while the two umbilical arteries return deoxygenated blood and waste products from the fetus to the placenta for exchange with the maternal circulation. These vessels are surrounded and protected by Wharton’s jelly, a specialized connective tissue derived from the extraembryonic mesoblast, which cushions the vessels and prevents compression. As the umbilical cord approaches its placental insertion, the two arteries form Hyrtl’s anastomosis, a connection that helps equalize blood flow between the arteries.
At a minimum, the mid-trimester ultrasound should include identification and documentation of the umbilical cord's fetal and placental insertions, as well as the number of cord vessels.66,186,187 In a transverse grayscale section of the cord, the two umbilical arteries can be visualized alongside the larger, thinner-walled umbilical vein, creating a characteristic ‘Mickey Mouse’ appearance (Figure 16a–c). Additionally, in a transverse section of the lower fetal abdomen, the umbilical arteries are seen encircling the fetal bladder. This can be seen on grayscale ultrasound and confirmed with color flow Doppler (Figure 16d–g).
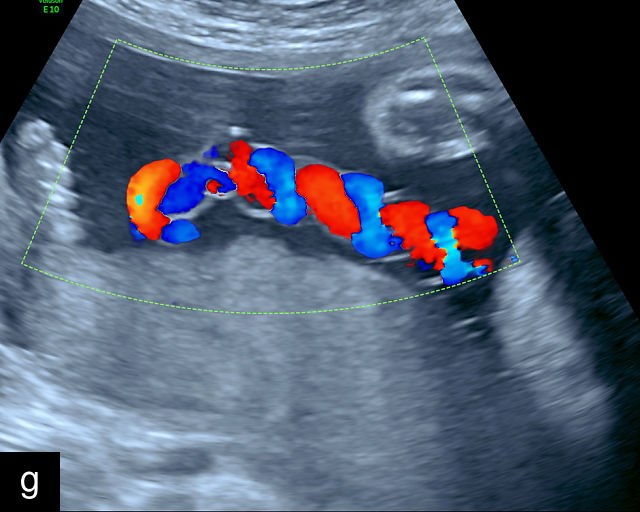
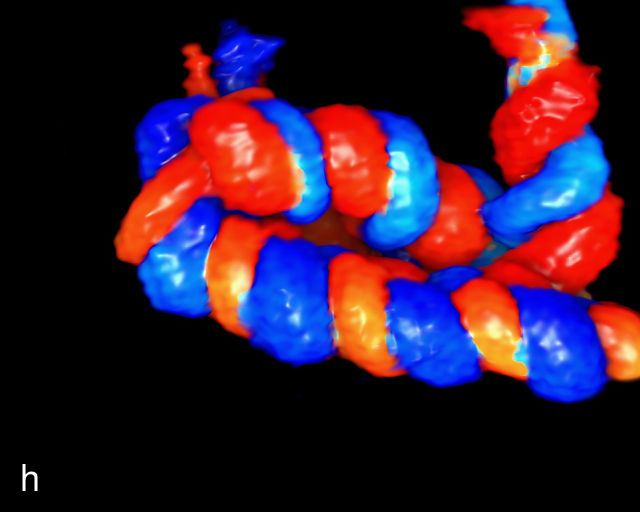
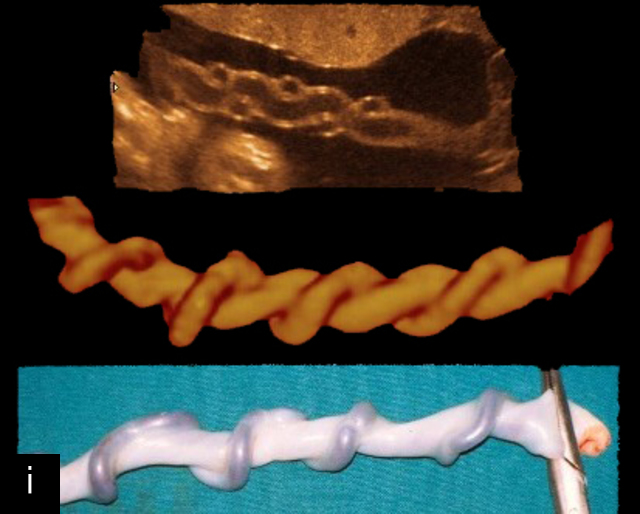
A normal umbilical cord is helical, with an average of 2.1 coils per 10 cm of cord length.188,189 The majority of umbilical cords exhibit a leftward (counterclockwise) twist, with a left-to-right twist ratio of approximately 7 : 1, making the rightward (clockwise) twist significantly less common.185,190,191 This twisting structure is believed to provide additional protection against compression and kinking during pregnancy.192
Thus, several mechanisms work together to protect the fetal vessels from occlusion. These include the cushioning effect of Wharton’s jelly, the helical twisting of the cord, normal length of the cord (which prevents excessive tension, twisting, knots or compression), Hyrtl’s anastomosis (which equalizes blood flow between the arteries) and the surrounding amniotic fluid, which provides additional protection and buoyancy. Aberrations in any of these protective factors may compromise the cord and increase the risk of fetal death or other adverse outcomes.192,193
After birth, the umbilical arteries undergo obliteration and eventually form the medial umbilical ligaments within the abdominal wall.
Abnormal umbilical cord findings
Single umbilical artery
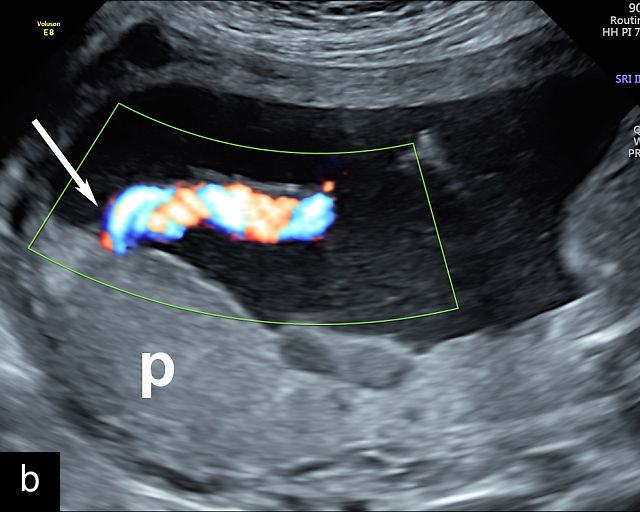
Assessment of the number of vessels in the umbilical cord is a component of routine obstetric ultrasound.66 As mentioned above, the umbilical cord typically contains two arteries and one vein, however, a single umbilical artery (SUA) is observed in approximately 1% of pregnancies.4,194,195,196 Several hypotheses have been proposed to explain the etiology of SUA: primary agenesis of one umbilical artery, obliteration of a previously formed artery or fusion of two separate arteries. When there is a SUA, there is a compensatory increased diameter of the single artery.197
SUA is one of the most common umbilical cord abnormalities detected on prenatal ultrasound.191,198 When a SUA is present, only a single vessel is seen along one side of the bladder (Figure 17a–d), compared with the two vessels seen with a normal cord (Figure 16e–g). In a cross-section of the cord with SUA, only two vessels, one artery and one vein, are visible (Figure 17e), and the Mickey Mouse sign (Figure 16a–c) is no longer present. In a longitudinal view of the cord, a SUA is identified by the presence of only two vessels within the helix, with the single artery being almost equal in size to the vein (Figure 17f–i). Fusion of the umbilical arteries has also been noted, such that a fetus may have a single umbilical artery and two separate umbilical arteries at different points along the cord.199,200

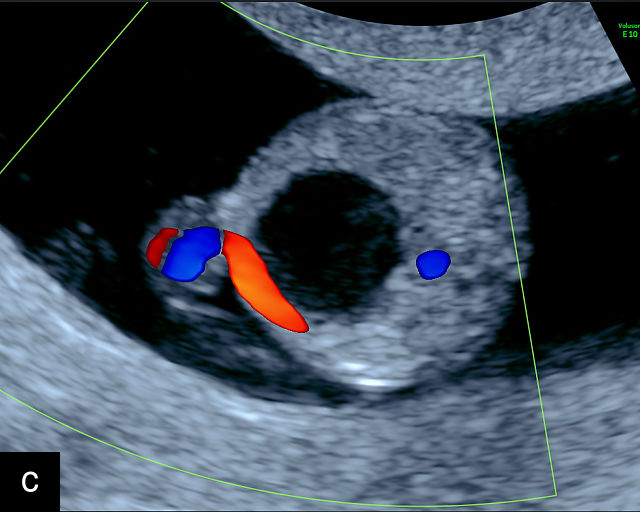


17
Single umbilical artery (SUA). (a–d) Transverse section of lower fetal abdomen at the level of the fetal bladder. Only a single vessel is seen coursing around one side of the bladder, compared with two vessels on each side of the bladder in the normal three-vessel cord. (e) Cross section through the umbilical cord showing only two circles, representing two vessels: a single umbilical artery and a vein. Compare with the three-vessel Mickey Mouse sign in Figure 16a–c. (f–h) Longitudinal ultrasound views with color flow Doppler of an umbilical cord with a SUA, showing two vessels with flow in opposite directions. This contrasts with a normal umbilical cord, in which three vessels are present in each coil (Figure 16h). In Figure 17h, the cord is also hypercoiled. (i) Grayscale and 3D power Doppler ultrasound images of a SUA and the umbilical cord after delivery.
When SUA is identified on obstetric ultrasound, a detailed fetal anatomical survey is recommended.201 The prognosis of SUA depends on whether it is an isolated finding or associated with other fetal anomalies. SUA has been linked to a range of genetic and structural abnormalities, particularly when additional anomalies are present.201 The most commonly associated abnormalities involve the cardiovascular and renal systems.202 SUA is also associated with trisomies 21, 18 and 13 when multiple anomalies coexist. As such, screening results should be carefully reviewed. If multiple anomalies are detected, invasive testing such as amniocentesis should be offered. Conversely, if SUA is an isolated finding and aneuploidy screening produces a low-risk result, no further evaluation for aneuploidy is recommended.201 A detailed cardiac assessment, or fetal echocardiography, should be performed, although studies suggest that a normal detailed cardiac examination typically obviates the need for additional echocardiographic evaluation.203,204,205
SUA has also been associated with FGR, and there is conflicting evidence regarding an increased risk of stillbirth.201,206,207 Therefore, the Society for Maternal-Fetal Medicine recommends a third-trimester growth scan and consideration of antepartum fetal surveillance beginning at approximately 36 weeks.201 However, such a monitoring protocol is not universally recommended, especially if the fetal growth is normal.
Abnormal cord coiling
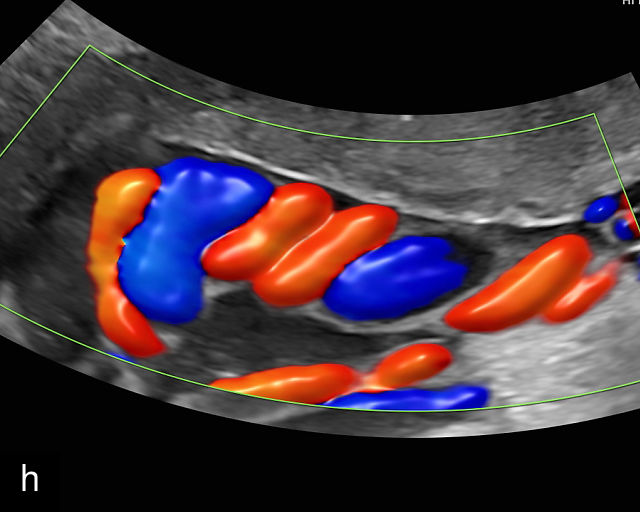
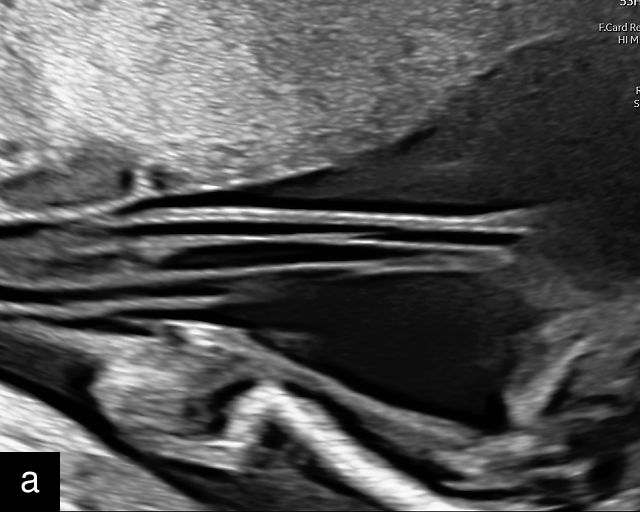
The umbilical cord coiling index (UCI) is defined as the number of complete vascular coils per cm of cord, with a normal UCI averaging around 0.2.185,191 A hypocoiled cord is typically defined as having an UCI of less than 0.2, or below the 10th percentile, with approximately one coil per 10 cm of cord (Figure 18a–c). In contrast, a hypercoiled cord has an UCI greater than 0.3, or above the 90th percentile (Figure 18d; Video 9). It is estimated that hypocoiling of the cord occurs in between 4% and 15% of pregnancies.185,191

18
Umbilical cord hypocoiling and hypercoiling. (a) Grayscale ultrasound image of hypocoiled cord. (b) Color flow Doppler image of hypocoiled cord. (c) Grayscale and color flow Doppler image of hypocoiled cord. (d) Grayscale image of hypercoiled cord.
9
Imaging of umbilical cord hypercoiling using grayscale (a–c) and color Doppler (b) ultrasound.
Several factors influence cord coiling, including cord length and fetal movement.191,208 Coiling tends to increase with gestational age and serves as a protective mechanism, shielding the fetal vessels from compression. Excessive coiling may obstruct blood flow, while hypocoiling may increase the risk of vessel compression and adverse perinatal outcome.209 Hypocoiling has also been associated with conditions characterized by reduced fetal movement.
While sonographic assessment of cord coiling is feasible, the sensitivity and specificity of this finding in isolation are limited. As a result, there are no current recommendations for routine evaluation of the UCI during obstetric ultrasound. This limitation is further compounded by the fact that most UCI reference values have been derived from postnatal examination of the cord rather than by prenatal imaging. Most adverse outcomes associated with abnormal umbilical cord coiling have been reported based on pathological examination of the placenta and cord following stillbirths or other adverse events, rather than on the prenatal ultrasound detection of abnormal cord coiling.6
Nonetheless, efforts have been made to assess coiling prenatally.208 Predanic et al. reported a correlation between second-trimester ultrasound measurements of cord coiling and findings at birth but noted that prenatal ultrasound was less accurate in identifying hypocoiled or hypercoiled patterns.188,210,211 In contrast, Ma’ayeh and colleagues, in a prospective study of 72 patients, found that second-trimester sonographic assessment of the UCI did not reliably predict coiling at birth or correlate with perinatal outcomes.212 Therefore, routine assessment of umbilical cord coiling is not recommended. However, if a cord appears markedly hypocoiled or hypercoiled during a standard ultrasound examination, closer surveillance with serial ultrasounds should be considered.
Abnormal cord length
A long umbilical cord is defined as one measuring above the 90th percentile for gestational age, while a short cord falls below the 5th percentile.191,213,214 Long cords (>80 cm at term) are associated with an increased risk of complications, including nuchal cords, cord entanglement, true knots and cord prolapse, as well as fetal demise.215,216 Excessive fetal movements are thought to contribute to the development of longer cords. However, it remains unclear whether these complications are primarily a result of the cord’s length or the excessive fetal movements themselves. The risk of these complications rises with increasing cord length, and there is some evidence suggesting that longer cords may be linked to FGR.217 Remarkably, cords as long as 300 cm have been documented, although measuring cord length remains challenging and is not a routine component of sonographic assessment.
In contrast, excessively short or absent umbilical cords are frequently associated with fetal anomalies, most notably limb–body wall complex (body–stalk anomaly) and ectopia cordis. Short cords, defined as less than 35–40 cm in length, are also linked to conditions such as fetal hypokinesia/akinesia sequence (Pena–Shokeir syndrome), arthrogryposis and constrictive dermatopathy, conditions that are typically characterized by inadequate or absent fetal movements.191,214
Even in the absence of fetal anomalies, short cords can increase the risk of adverse perinatal outcome, including placental abruption, preterm prelabor rupture of membranes (PPROM), intrapartum fetal heart rate abnormality, inadequate fetal descent in labor, operative delivery, uterine inversion and postpartum hemorrhage.213
It is important to note that these definitions of cord length are primarily based on examinations of the placenta and umbilical cord after delivery, rather than on prenatal ultrasound assessment.213 Furthermore, these measurements are typically obtained at term, which may differ significantly from measurements taken earlier in gestation. While some have attempted to assess umbilical cord length prenatally, this is an extremely challenging task that is difficult to standardize or reproduce. As such, routine measurement of cord length via prenatal ultrasound is not recommended and is often a qualitative rather than quantitative exercise. However, if the umbilical cord appears subjectively abnormally short (with only a small segment visible) or excessively long (with numerous free loops observed), a detailed fetal anatomy examination should be performed, with particular attention to fetal movements. In these cases, serial growth ultrasounds are also recommended to monitor fetal development.
Abnormal cord thickness
Lean umbilical cord
Thin or lean umbilical cords are often associated with FGR.185,218 In many such cases, there is marked deficiency of Wharton’s jelly, rendering the cord more susceptible to compression and increasing the risk of fetal acidemia. Oligohydramnios, frequently observed in growth-restricted pregnancies, can exacerbate this susceptibility by eliminating the cushioning effect of amniotic fluid, signficantly elevating the risk of fetal acidemia and stillbirth.
Silver and colleagues reported higher rates of smaller umbilical cord diameters in post-term pregnancies complicated by oligohydramnios.219 These pregnancies also exhibited a greater incidence of intrapartum fetal heart rate decelerations. Similarly, Raio and colleagues evaluated the cross-sectional area of the umbilical cord after 20 weeks' gestation in 860 patients.218 They identified a four-fold increase in small-for-gestational-age infants at birth among cases with lean umbilical cord. These pregnancies were also more likely to present with oligohydramnios at delivery and low 5-min Apgar score (< 7).
The umbilical artery cross-sectional diameter may be increased when there is a single umbilical artery.220,221
Thick umbilical cord
Thick umbilical cords with an abundance of Wharton’s jelly are often observed in pregnancies complicated by diabetes, fetal macrosomia and overgrowth syndromes such as Beckwith–Wiedemann syndrome.222 Predanic and colleagues, in a small retrospective study, found that umbilical cord thickness was greater in fetuses with aneuploidy than in euploid fetuses.223
Although umbilical cord nomograms exist, umbilical cord thickness is not routinely measured during obstetric ultrasound examination.224,225 Observations such as a ‘thick’ or ‘lean’ cord are therefore typically subjective. However, when the umbilical cord appears unusually thick or thin, a detailed ultrasound evaluation of the fetus and placenta is recommended. In such cases, serial ultrasound examinations may also be advisable.
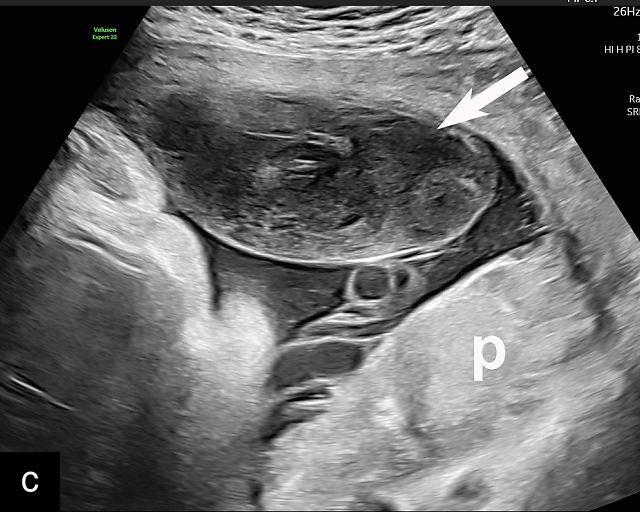
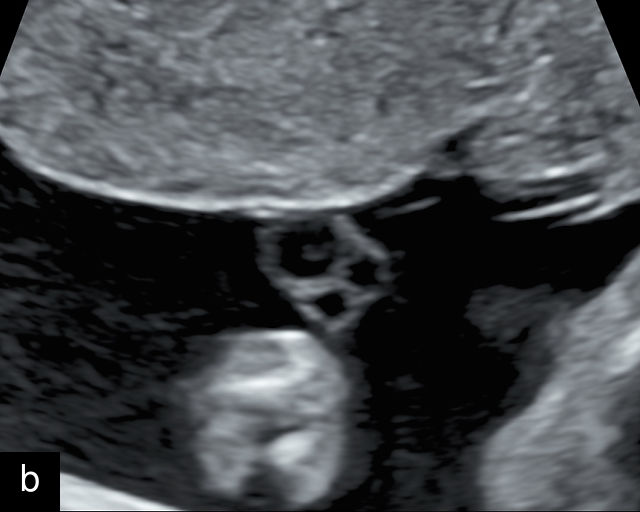
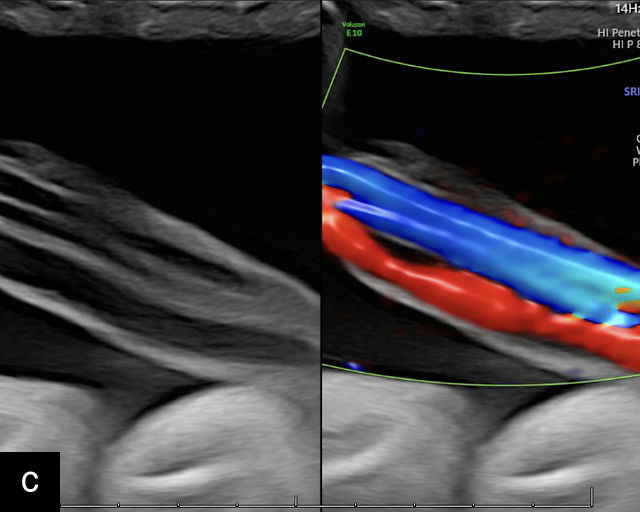
Marginal cord insertion
In some cases, the umbilical cord inserts close to the edge of the placenta, a condition known as marginal cord insertion (Figure 19; Video 10). The definition of marginal cord insertion varies, with some investigators using a distance of 2 cm from the placental edge, while others use 1 cm or 1.5 cm as the cutoff.226 In true marginal cord insertion, the fetal vessels remain protected by Wharton’s jelly throughout their entire course, with no exposed vessels.

19
Grayscale (a) and color Doppler (b) ultrasound images of marginal cord insertion, showing the cord inserting close to placental edge (arrows).
10
Grayscale and color Doppler imaging of marginal placental cord insertion.
Complicating the matter, studies often group velamentous cord insertions, in which vessels are unprotected for some distance, together with marginal cord insertions, in which the vessels are protected.227 Differentiating between these two conditions on obstetric ultrasound can also be challenging, further complicating the assessment of the clinical significance of true marginal cord insertion. Therefore, when a marginal cord insertion is suspected, it is crucial to differentiate it from a velamentous cord insertion, in which unprotected fetal vessels are seen traversing the membranes for a variable distance before inserting into the placental edge.
Some studies suggest that marginal cord insertions may be associated with an increased risk of adverse perinatal outcome, particularly FGR.228,229 Conversely, other studies have found no significant increase in risk.230,231,232,233,234 To compound the problem, some of the studies that show increased adverse outcomes were based on examinations of placentas after birth rather than by prenatal ultrasound.235 Importantly, the majority of isolated marginal cord insertions are associated with favorable perinatal outcome, allowing for patient reassurance.
In some centers, the identification of a marginal cord insertion prompts a third-trimester ultrasound to assess fetal growth and rule out FGR. This approach helps ensure appropriate monitoring and management while addressing potential concerns.
Velamentous cord insertion
When the umbilical cord inserts into the membranes rather than the main placental mass, it is known as a velamentous cord insertion (VCI).191 In this condition, unprotected fetal vessels traverse the membranes for a variable distance before inserting into the placental edge. Velamentous cord insertions are relatively common, occurring in approximately 1% of pregnancies and about 10% of twin pregnancies.191 A large population-based study from Norway reported a 1.5% prevalence of VCI in singleton pregnancies compared to 6.0% in twins.227 Risk factors for VCI include twin gestation, pregnancy conceived via assisted reproductive technology and advanced maternal age.34,236,237,238,239
VCI is associated with an increased risk of fetal structural abnormalities.240 When a VCI is identified, a detailed fetal anatomical survey is recommended. VCI is also linked to adverse perinatal outcomes, including intrauterine fetal demise, FGR, preterm birth and complications during delivery, such as cord avulsion leading to retained placenta and an increased risk of postpartum hemorrhage.241,242,243,244 VCI has also been associated with increased risks for cerebral palsy in the offspring.7 While rare, spontaneous rupture of VCIs has been reported, potentially resulting in stillbirth or other adverse outcomes. In the absence of vasa previa, however, labor and vaginal delivery are typically recommended. Serial ultrasound examinations to assess fetal growth are advised when VCI is detected during prenatal ultrasound. Importantly, VCI in monochorionic twins is associated with an increased risk for TTTS, selective FGR and birth-weight discordance.245,246,247
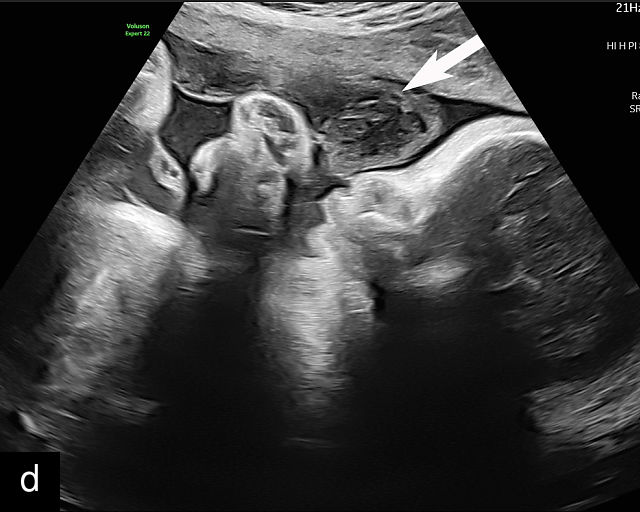
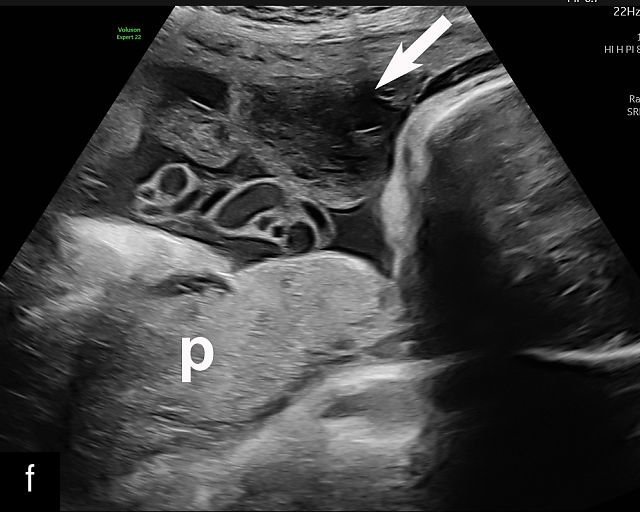
Ultrasound is the primary diagnostic method for identifying VCI. Normally, the cord inserts into the center or edge of the placenta and is fully protected by Wharton’s jelly. In contrast, a velamentous cord insertion is visualized on ultrasound as the cord inserting into the membranes, with unprotected fetal vessels running to the placental margin (Figure 20; Video 11). Color flow Doppler and three-dimensional ultrasound can enhance diagnostic accuracy. Velamentous cord insertion may also occur into the dividing membrane in a multifetal pregnancy (Figure 20e). When unprotected fetal vessels from a VCI cross over the cervix, the condition is termed vasa previa, posing a significant risk of fetal exsanguination if these vessels rupture. Because VCI is a strong risk factor for vasa previa, a color flow Doppler sweep over the cervix is recommended when VCI is identified. If concerns about vasa previa persist, transvaginal ultrasound with color Doppler may be performed for confirmation.30


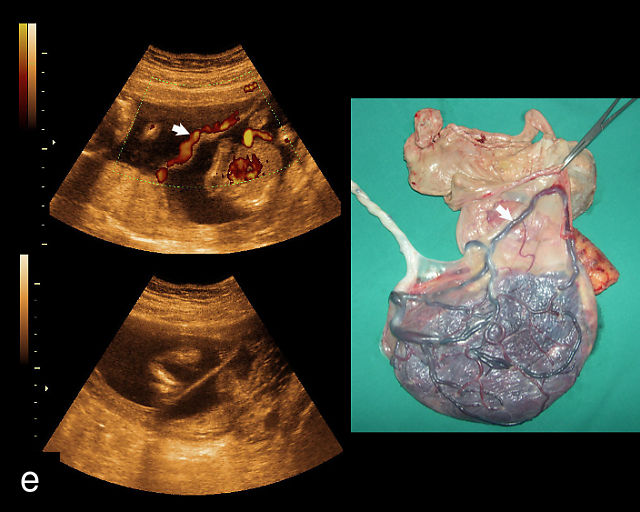
20
(a–c) Grayscale (a) and color Doppler (b,c) ultrasound images of velamentous cord insertion. The cord inserts into the membranes (arrow), from where unprotected fetal vessels (arrowheads) traverse the membranes to insert into the edge of the anterior (a,b) or posterior (c) placenta (p). (d) Color Doppler ultrasound image of velamentous cord insertion, showing the cord inserting into the membranes posteriorly (arrow), from where unprotected fetal vessels (arrowheads) traverse the membranes to insert into the edge of the anterior placenta (p). (e) Ultrasound with color Doppler showing velamentous vessels (arrow) running through the dividing membrane in a dichorionic diamniotic twin pregnancy. The right image shows the placenta after delivery.
11
Color Doppler ultrasound imaging of velamentous cord insertion. The cord inserts into the membranes from where unprotected fetal vessels traverse the membranes to insert into the edge of the posterior (a) or anterior (b) placenta.
Nomiyama and colleagues conducted a study in which color Doppler screening of placental cord insertion was routinely performed on 587 patients during second-trimester anatomy scans.248 They successfully identified the placental cord insertion in 586 cases (99.8%) and correctly diagnosed five cases of VCI, with one false-positive result.248 The study reported a sensitivity of 100%, specificity of 99.8%, positive predictive value of 83% and negative predictive value of 100%. However, the study population in Japan had a significantly lower average body mass index (BMI) compared to populations in the Western world. Increased BMI, fetal position, uterine fibroids and posterior placenta may hinder visualization of the cord, making these results less generalizable to populations with higher BMI prevalence.
Sepulveda and colleagues evaluated 832 patients during the second and third trimesters using color Doppler and successfully visualized placental cord insertion in 825 cases (99%), diagnosing seven cases of VCI.249 These authors, in a separate study, also screened 533 pregnancies during the first trimester and correctly identified five cases of VCI.250
In the USA, consensus guidelines recommend assessing placental cord insertion during the second-trimester anatomical ultrasound examination when feasible. This approach is being increasingly recommended in other countries.251 At our center, this assessment has been routinely performed as part of the second-trimester anatomy scan for several years, and we strongly advocate for its inclusion as a standard component of the examination.
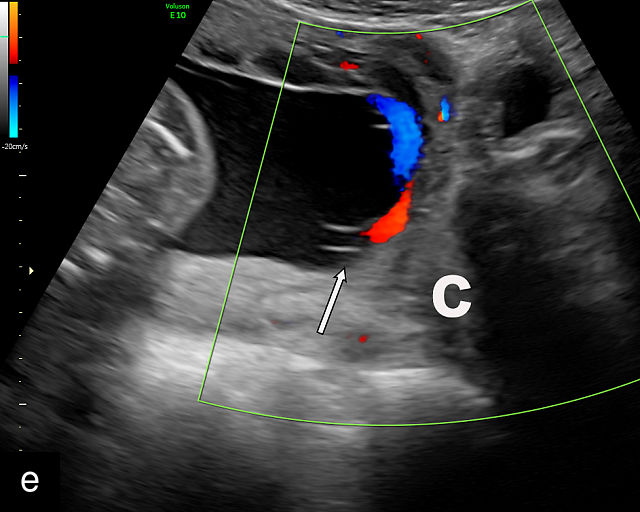
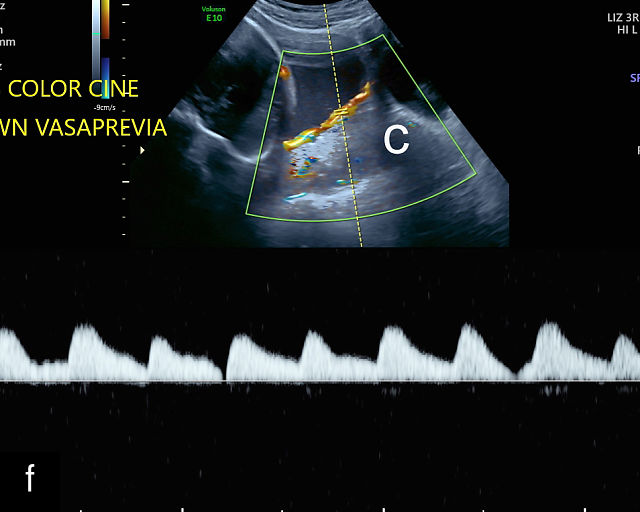
Vasa previa
Vasa previa refers to unprotected fetal vessels running through the membranes over the cervix.30,50,252,253 These vessels often rupture when the membranes rupture during labor or in late pregnancy, resulting in fetal hemorrhage and often exsanguination.252 As a result, this condition is associated with high perinatal mortality.35,252 A large study found a 56% perinatal mortality when vasa previa was not diagnosed prenatally.35 Prenatal diagnosis with ultrasound and cesarean delivery before labor or rupture of the membranes prevents this high perinatal mortality.254,255,256 Risk factors for vasa previa include second-trimester placenta previa or low-lying placenta, velamentous cord insertion, pregnancy resulting from in-vitro fertilization, multifetal gestation and placenta with accessory lobe.30,36,50,257,258,259,260,261
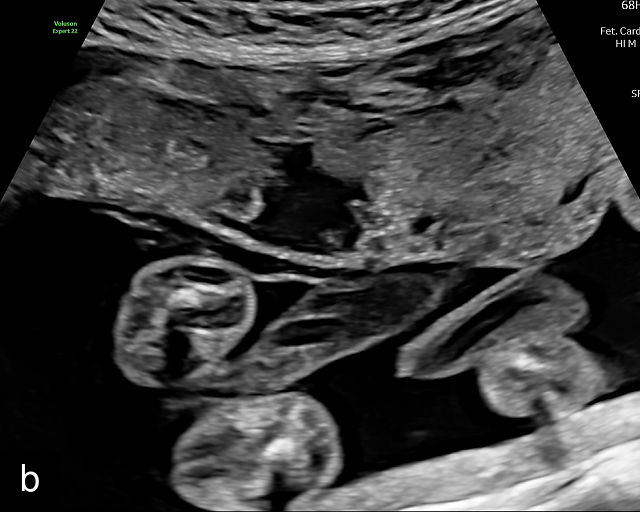
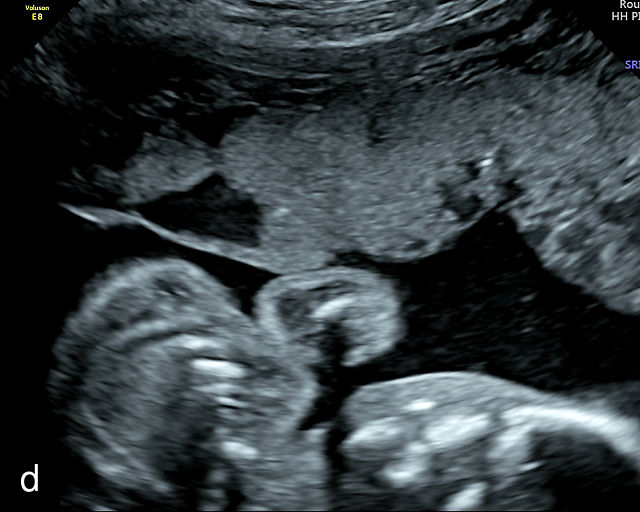
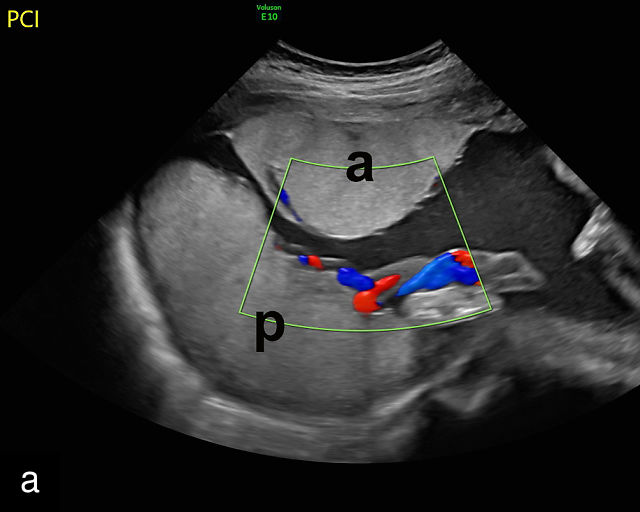
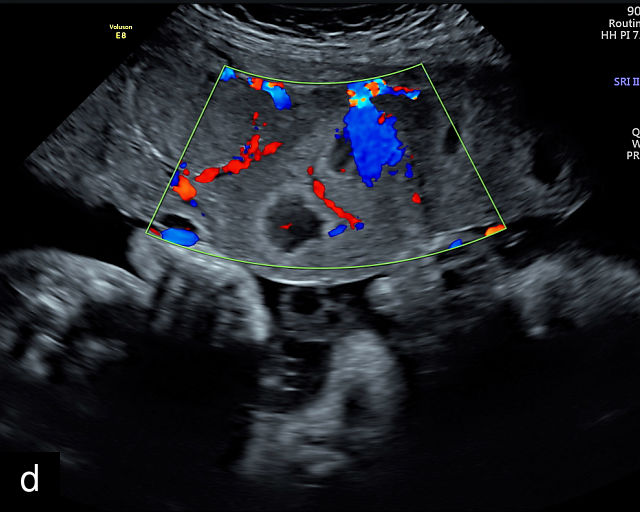
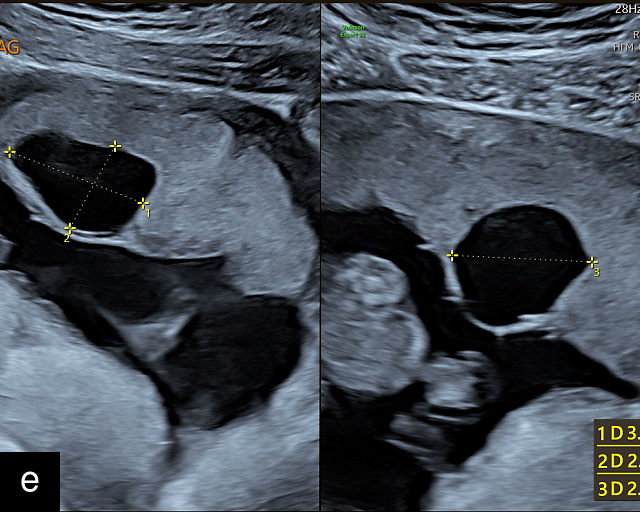
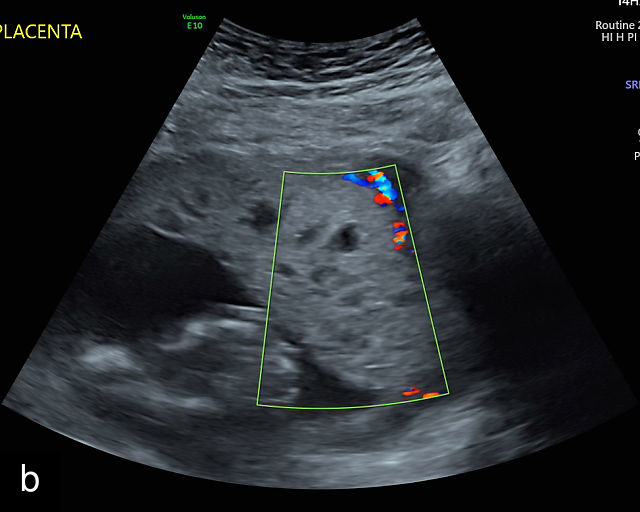
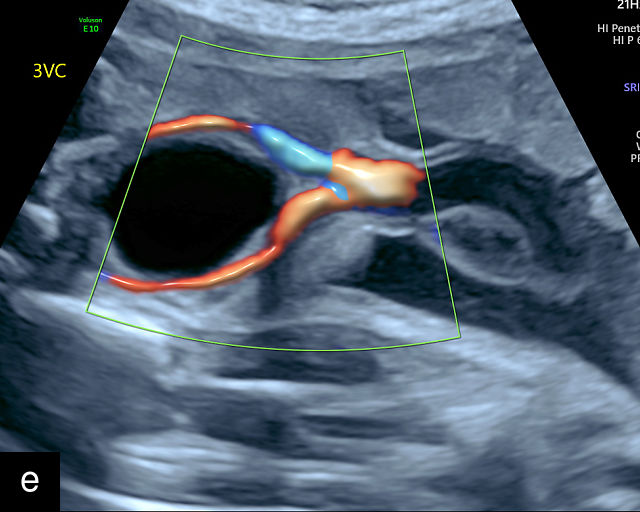
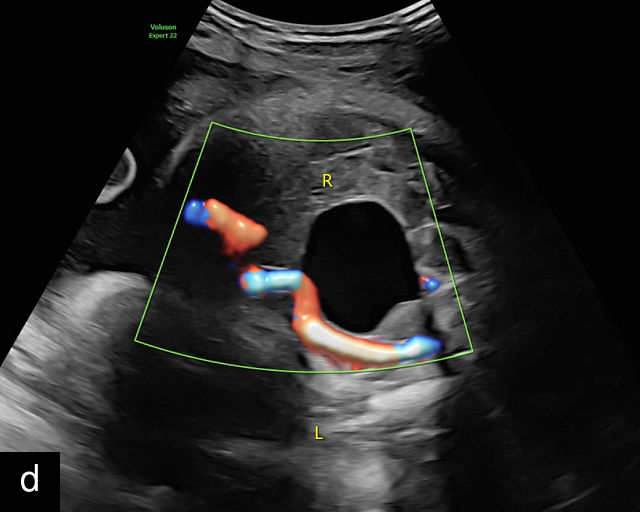
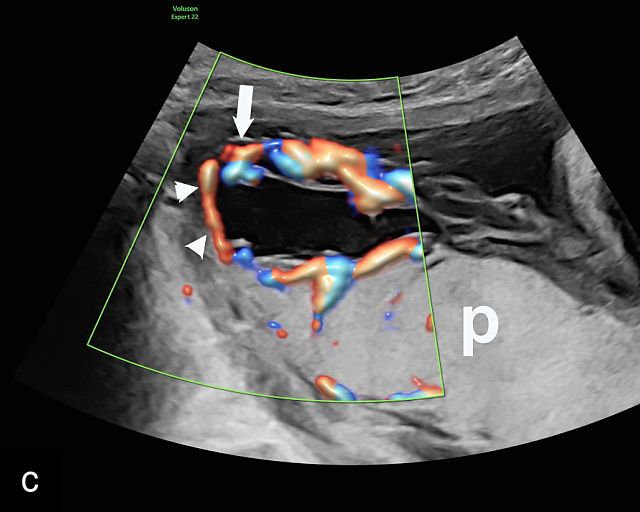
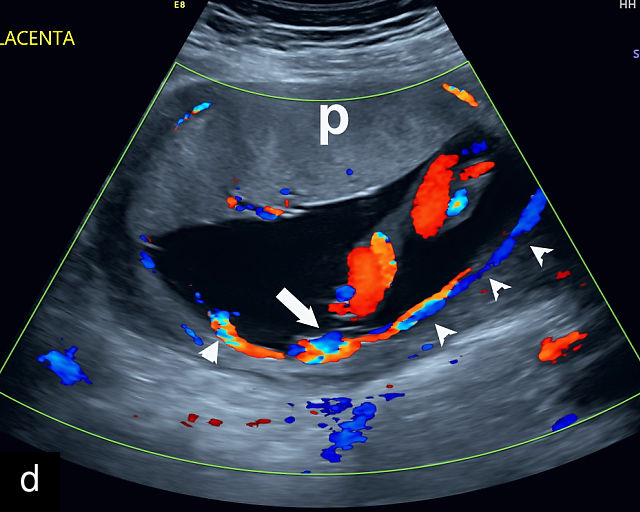
There are three types of vasa previa.30,262 In Type 1, the cord inserts into the membranes rather than the placenta. Unprotected vessels then traverse the membranes over the cervix to insert into the placenta (Figure 21a; Video 12). In Type 2, unprotected vessels running through the membranes over the cervix connect the main placental lobe with an accessory lobe (Figure 21b).263 In Type 3, there is generally a normal placental cord insertion, and unprotected vessels exit one placental edge, run through the membranes over the cervix and then boomerang to insert into the placental edge at another site (Figure 21c).25,28,29,264 Regardless of the type, all these expose the fetus to the same risks.
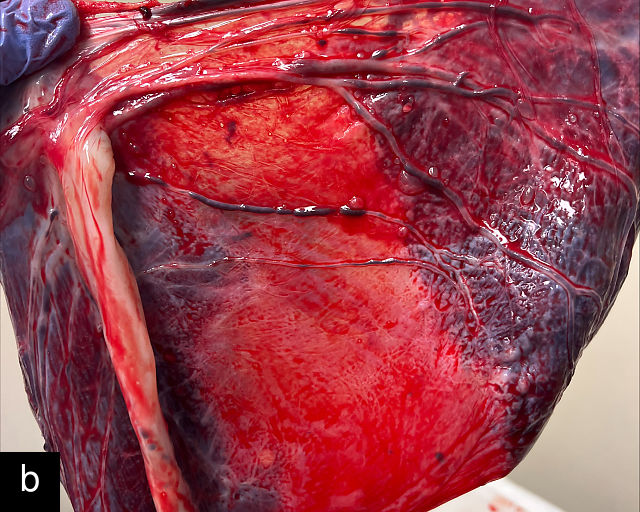
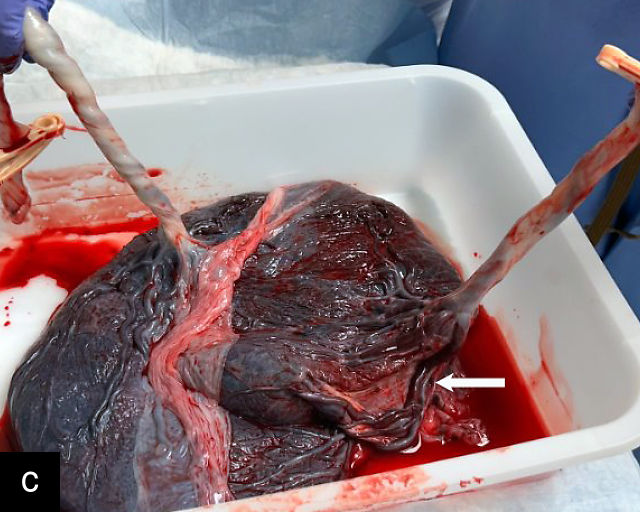

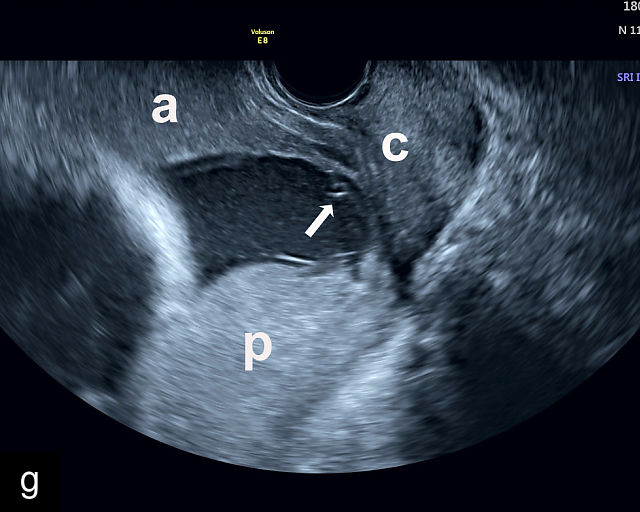
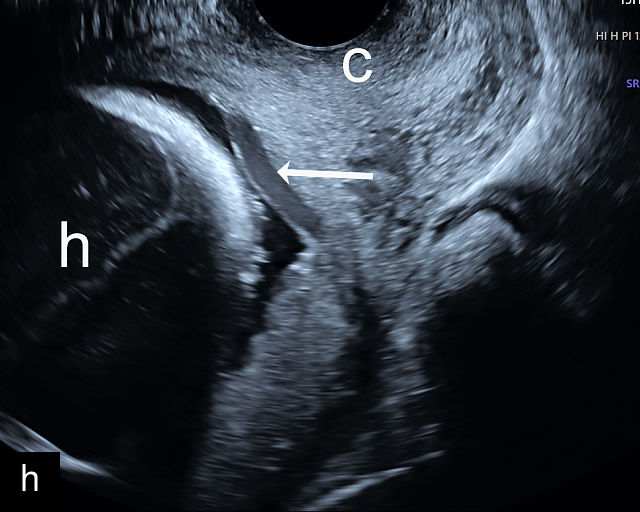

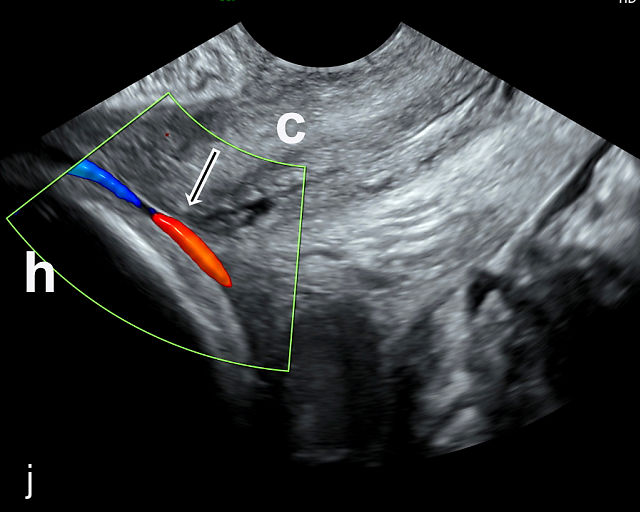
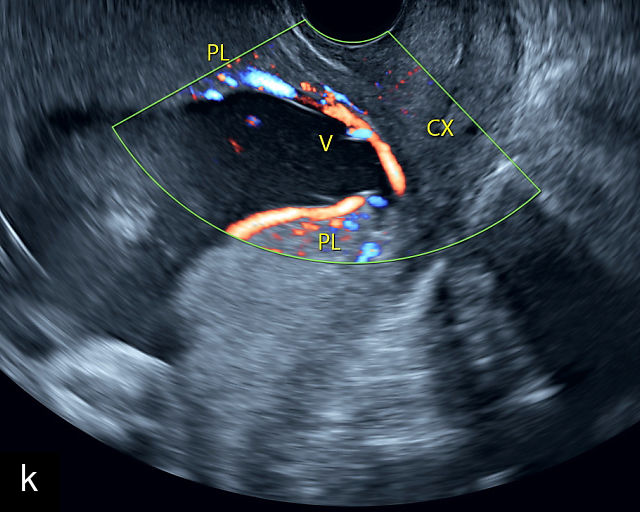
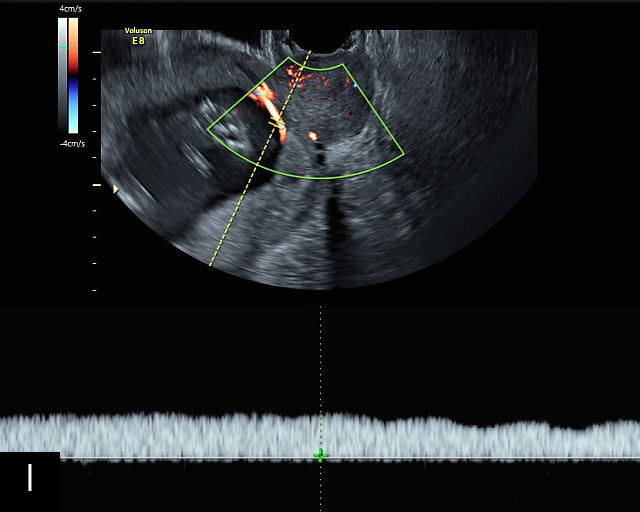
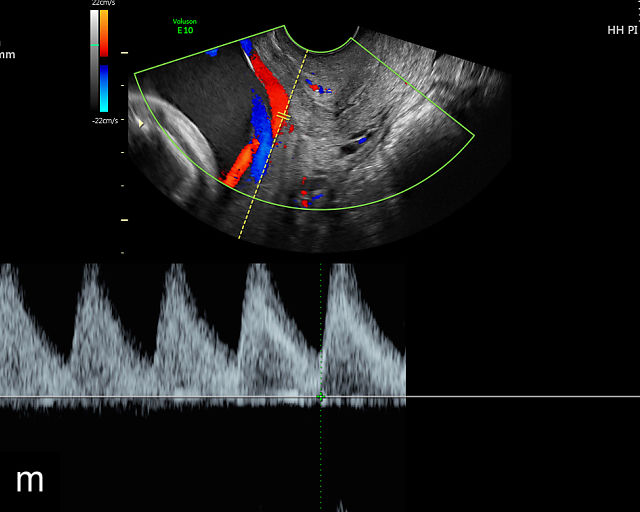
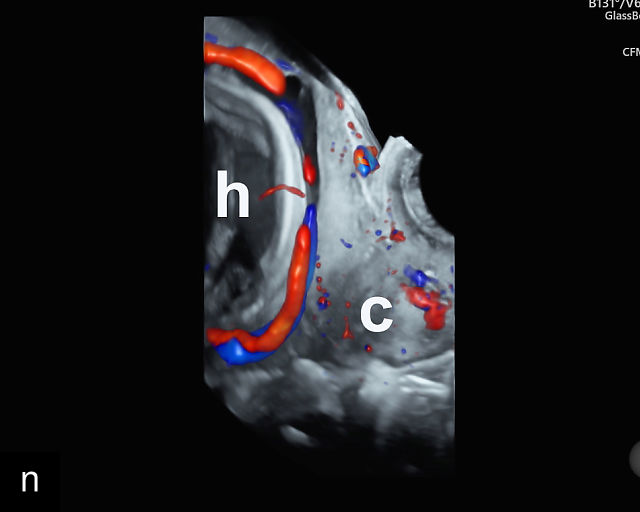
21
(a) Type-1 vasa previa. Photograph after delivery showing the umbilical cord inserting into the membranes through which unprotected fetal vessels run to insert into the placental edge. (b) Type-2 vasa previa. Photograph after delivery showing bilobed placenta with unprotected fetal vessels running though the membranes between the lobes. (c) Type-3 vasa previa in a twin pregnancy. Photograph after delivery showing unprotected vessels running through the membranes from one edge of the placenta to another (arrow). (d) Transabdominal grayscale ultrasound of the lower uterine segment showing a linear hypoechoic structure (fetal vessel) running over the cervix (c) indicating vasa previa (Type 2). b, bladder; h, fetal head. (e) Transabdominal color Doppler ultrasound of the lower uterine segment showing fetal vessels (arrow) running over the cervix (c) indicating vasa previa (Type 2). (f) Transabdominal ultrasound with color flow and pulsed-wave Doppler of the lower uterine segment showing a fetal vessel running over the cervix (c) indicating vasa previa (Type 2). Pulsed-wave Doppler demonstrates an umbilical arterial waveform. (g) Transvaginal grayscale ultrasound with showing a Type-2 vasa previa. There are two placental lobes, an anterior (a) and a posterior (p) lobe. There is a linear and circular hypoechoic structure (arrow) running over the cervix (c) between the lobes. (h) Transvaginal grayscale ultrasound image of vasa previa. A hypoechoic linear structure (arrow) is seen running through the membranes over the cervix (c). h, fetal head. (i) Transvaginal grayscale ultrasound of vasa previa. Hypoechoic circular and linear structures are seen close to the internal os. (j) Transvaginal color Doppler ultrasound image of vasa previa. A fetal vessel is seen running through the membranes over the internal os (arrow) of the cervix (c). h, fetal head. (k) Transvaginal ultrasound with color Doppler showing a Type-2 vasa previa. There are two placental lobes, an anterior and posterior lobe (pl). Fetal vessels run over the cervix between the lobes. (l) Transvaginal color flow ultrasound with pulsed-wave Doppler image of vasa previa. Color Doppler shows flow through the vessel and pulsed-wave Doppler shows a fetal umbilical venous waveform. (m) Transvaginal color ultrasound with pulsed-wave Doppler image of vasa previa. Color flow Doppler shows flow through the vessel and pulsed-wave Doppler shows a fetal umbilical arterial waveform. (n) Transvaginal three-dimensional ultrasound with color Doppler image of vasa previa. h, fetal head; c, cervix.
12
Transvaginal ultrasound imaging of Type-1 vasa previa. Color Doppler (a,c,d) and grayscale (b) imaging shows fetal vessels running over the cervix.
In all pregnancies, the placental cord insertion should be identified at the time of the second-trimester anatomy scan. In addition, a color flow Doppler sweep of the region overlying the cervix should be performed. The ultrasound appearance of vasa previa is of hypoechoic linear or circular structures overlying or in close proximity to the internal os (Figure 21).30,187,252,263 When these are observed, color flow Doppler should be employed to confirm flow through these structures.30,187 Transvaginal ultrasound with color and pulsed-wave Doppler is essential for diagnosing vasa previa; the presence of a fetal arterial or venous waveform on pulsed-wave Doppler confirms the diagnosis (Figure 21). A common misconception is that vasa previa occurs only when a fetal artery overlies the cervix; however, a fetal vein overlying the cervix also constitutes vasa previa (Figure 21l).265 The precise distance of fetal vessels from the internal os required to define vasa previa has been debated.266 While some have used a distance of 2 cm, others argue that unprotected fetal vessels within 5 cm of the internal os are at risk of rupture, particularly given that the cervix dilates to 10 cm during labor. Some experts advocate for a 5-cm threshold to define vasa previa.266,267 An international Delphi consensus of vasa previa experts recommended that the definition should not be limited to a 2 cm cutoff, acknowledging the variability in clinical practice.268
Transvaginal ultrasound with color flow Doppler is highly accurate in the diagnosis of vasa previa, with a systematic review and meta-analysis finding sensitivity and specificity of greater than 99%.269
False positives may arise due to several factors, including funic presentation (where free loops of the cord overlie the cervix), movement artifacts on color Doppler, placenta previa or maternal vessels.30,263 Free loops of the umbilical cord typically move away from the cervix with changes in maternal position and do not persist on serial ultrasound examinations.187
When vasa previa is suspected during the second trimester, transvaginal sonography with color Doppler should be repeated at around 32 weeks to confirm the diagnosis. This follow-up is essential, as 15–40% of vasa previa cases identified in the second trimester resolve spontaneously before delivery.270,271 It is also critical to ensure that the fetal head is not engaged, as this can compress and obscure fetal vessels, potentially leading to a false-negative diagnosis.272 In such cases, manual elevation of the fetal head may be necessary, but care must be taken to avoid compressing the vessels during this maneuver, as it can also result in a missed diagnosis.273
Three-dimensional ultrasound may assist in confirming the diagnosis and mapping the course of fetal vessels (Figure 21n).274,275 Routine screening for vasa previa in all pregnancies is recommended by assessing the placental cord insertion and performing a color Doppler sweep of the lower uterine segment.30,255,268
For asymptomatic patients with a prenatal diagnosis of vasa previa, delivery by scheduled cesarean at 36–37 weeks is recommended.254,276,277 Earlier delivery may be warranted in the presence of labor, bleeding or rupture of membranes.
Furcate cord
In a furcate cord, the fetal vessels separate from the umbilical cord before reaching the placental surface.24,278 A velamentous cord is often mistakenly referred to as a furcate cord due to the incorrect assumption that the term ‘furcate’ means to ‘fork out’, implying that unprotected vessels running through the membranes are furcate. However, a true furcate cord ends short of the placenta and does not insert peripherally into the membranes. Instead, the fetal vessels separate from the umbilical cord, float freely in the amniotic fluid and insert unprotected most often into the central fetal surface of the placental body.
This contrasts with a velamentous cord insertion, in which the unprotected vessels run through the membranes before inserting peripherally into the edge of the placenta. A furcate cord insertion is extremely rare and is associated with significant risks, including compression or rupture of the exposed vessels, particularly during labor. It is often diagnosed only after delivery and carries a substantial risk of fetal demise or fetal distress during labor.279 Traction on the cord after delivery often leads to avulsion of the cord.
Nuchal cord
A nuchal cord, when the umbilical cord encircles the fetal neck, is a common finding, occurring in approximately one-third of all births.280,281 Nuchal cords can often be detected as early as the first trimester, commonly noted during first-trimester nuchal translucency measurements. While most nuchal cords consist of a single loop, cases with two, three or more loops have been described.282 Remarkably, one published report documented a nuchal cord with eight loops in a pregnancy (despite the cord length being normal at 55 cm) complicated by FGR, breech presentation and non-reassuring fetal heart rate patterns.283 That pregnancy required a cesarean delivery, resulting in a live birth. Nuchal cords frequently unravel with advancing gestation, reducing their potential impact.
One of the most common concerns expressed by pregnant patients during ultrasound examinations is whether the cord is around the baby’s neck. Historically, most nuchal cords were only detected incidentally at birth in uncomplicated pregnancies. However, with the widespread use of routine obstetric ultrasound, they are now frequently identified prenatally.280 This often causes significant anxiety for parents.185,282,284 In stillbirth cases, one or more nuchal cord loops may sometimes be identified. While the nuchal cord is often an incidental finding, in some cases – especially when there are multiple or tightly wrapped loops – it may be the direct cause of stillbirth. The risk of stillbirth increases with the number of loops and the tightness of the cord.282,285
Although most nuchal cords diagnosed prenatally are associated with favorable outcome,281 certain factors can increase the risk of adverse outcome. An adequate amount of Wharton’s jelly plays a critical role in protecting the fetal vessels from compression or shearing.282 In cases of FGR, in which Wharton’s jelly may be significantly reduced, the risk of complications with a nuchal cord increases. Other risk factors include hypocoiling of the cord, co-existing cord knots, multiple or tight loops of nuchal cord and a single umbilical artery.285
Nuchal cords have also been associated with intrapartum fetal heart rate abnormality, increased rates of cesarean and operative vaginal delivery, and neonatal acidemia.281,282 A long cord length is the primary risk factor for the development of a nuchal cord.286 However, one study found that the nuchal cord was more likely to be tight when the cord length was short or normal.286
The ultrasound appearance of a nuchal cord typically shows loops of the cord encircling the fetal neck (Figure 22). However, loops of cord in the vicinity of the neck are often misdiagnosed as nuchal cords. The divot sign, described by Ranzini et al., is considered diagnostic confirmation.287 This sign appears as an indentation in the subcutaneous tissue of the fetal neck caused by the cord (Figure 22a–c). Color flow and power Doppler significantly enhance visualization of nuchal cords, improving diagnostic accuracy (Video 13).288 Three-dimensional ultrasound may further aid in diagnosis, particularly in complex cases. The sensitivity of third-trimester ultrasound for detecting nuchal cords varies widely, ranging from 30% to 90%.
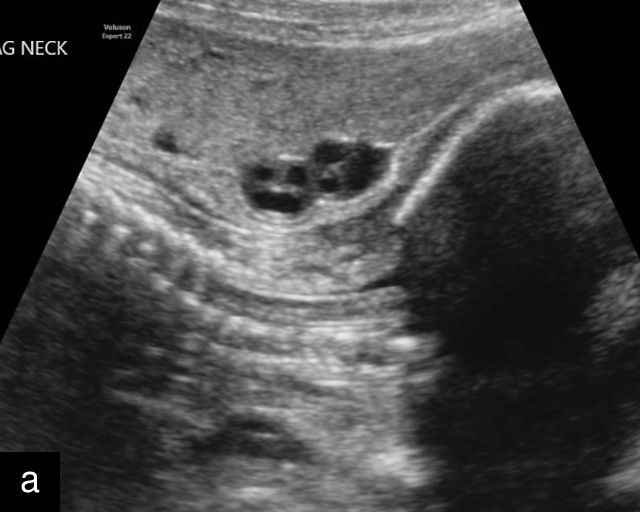
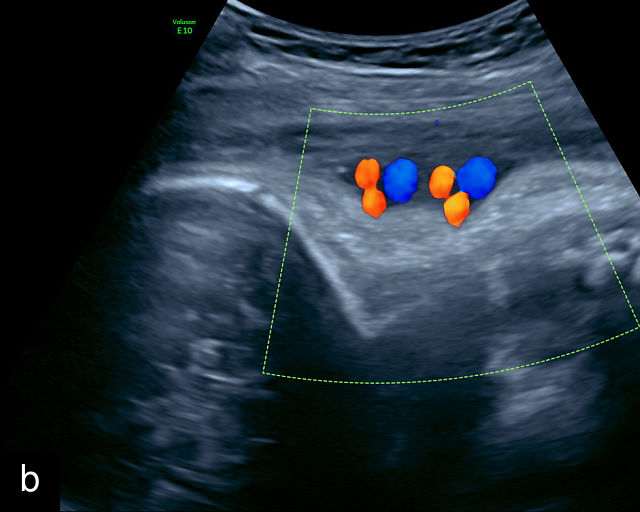


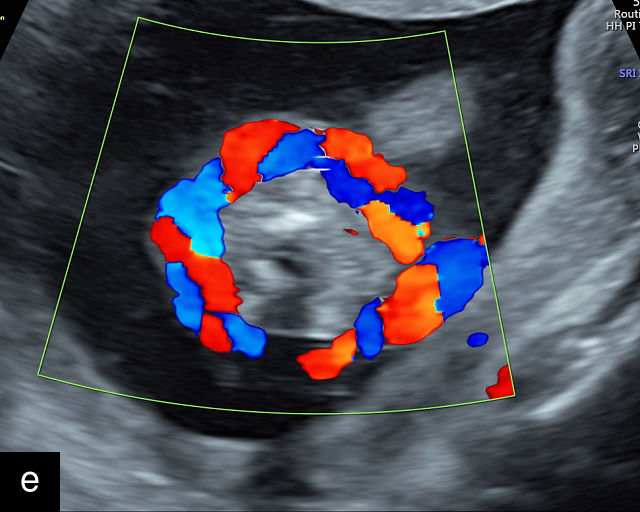
22
(a,b) Sagittal grayscale (a) and color Doppler (b) ultrasound images of fetal neck showing two loops of umbilical cord indenting the fetal neck – the ‘divot sign’. (c) Sagittal image of fetal neck with color flow Doppler showing four loops of cord encircling fetal neck. (d,e) Transverse grayscale (d) and color Doppler (e) ultrasound images showing loop of cord entirely encircling the fetal neck at 20 weeks of gestation.
13
(a) Transverse color flow Doppler showing at least two loops of umbilical cord encircling the fetal neck. (b) Sagittal scan sweep with color flow Doppler showing a nuchal cord. (c) Transverse sweep of fetal neck with color flow Doppler showing nuchal cord with three loops of cord encircling fetal neck.
Sherer and colleagues have provided practical guidance on managing findings of a nuchal cord during obstetric ultrasound.284 They suggest it is unethical not to inform the patient of a detected nuchal cord in the late third trimester.282,284 They recommend that when a nuchal cord is identified in the mid to late third trimester, both the patient and the referring physician should be informed. Patients should be advised to monitor fetal movements closely and report any perceived decrease in movements immediately. Fetal surveillance is recommended in cases with additional risk factors, such as FGR, hypocoiling, co-existing cord knots, multiple or tight loops or a SUA.282
Sepulveda and colleagues conducted routine examinations for nuchal cords after 24 weeks using color flow Doppler.289 They identified 10 singleton fetuses with triple nuchal cords. In cases diagnosed after 36 weeks, all pregnancies underwent cesarean delivery, confirming the presence of triple nuchal cords. For cases diagnosed before 36 weeks, at least one loop unraveled in 83% of pregnancies as gestation progressed. All pregnancies in the study had favorable outcomes, demonstrating that even a triple nuchal cord is often associated with excellent results.
When a nuchal cord is observed early in pregnancy, reporting it is of limited clinical value since interventions are unavailable and nuchal cords are exceedingly common and typically benign. At greater than 39 weeks of gestation, induction of labor and vaginal delivery with close intrapartum monitoring is a reasonable and effective approach.
Umbilical cord knots
Knots of the umbilical cord are found most often incidentally at delivery (Figure 23a,b) The incidence of cord knots depends on whether umbilical cords at birth or on prenatal ultrasound are analyzed. It is thought that most cord knots occur at the end of the first trimester when the ratio between the fetal size and that of the amniotic cavity is relatively small, allowing fetal movement. It is also thought that the initial cord knot is loose and tightens with advancing gestation and fetal movement. Factors that predispose to cord knots include long umbilical cord and excessive fetal movement. Because the fetal vessels in the umbilical cord in most pregnancies are protected by a good amount of Wharton’s jelly, in most cases, cord knots do not result in fetal death. However, cord knots may predispose to stillbirth (with a four-fold increased odds) and to intrapartum fetal heart rate abnormality and perinatal asphyxia.285,290,291 Most often, antenatal detection of umbilical cord knots occurs by chance during an obstetric ultrasound examination.
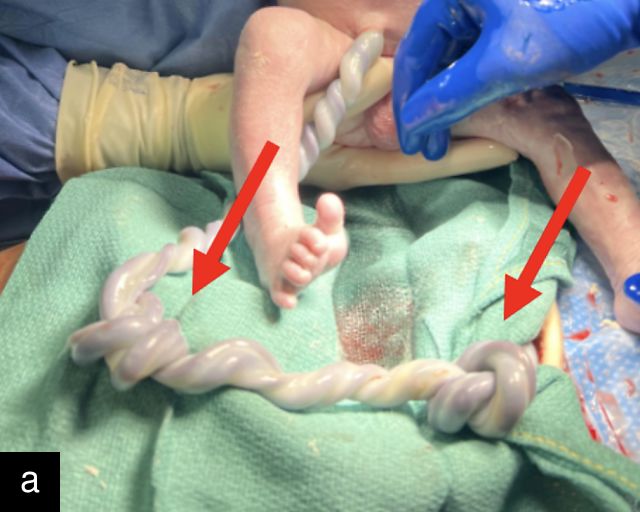

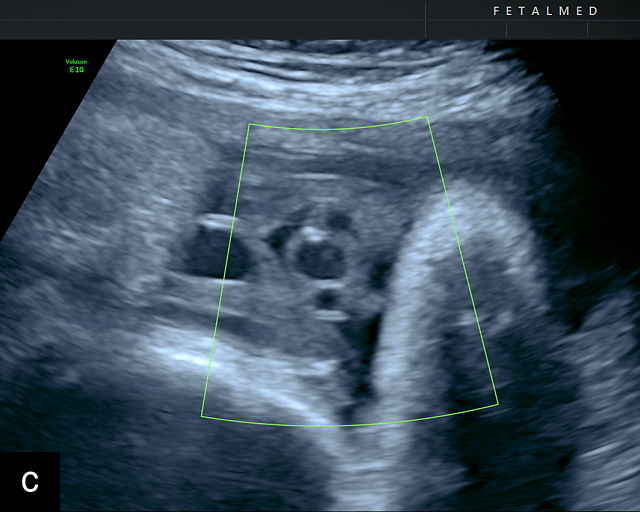

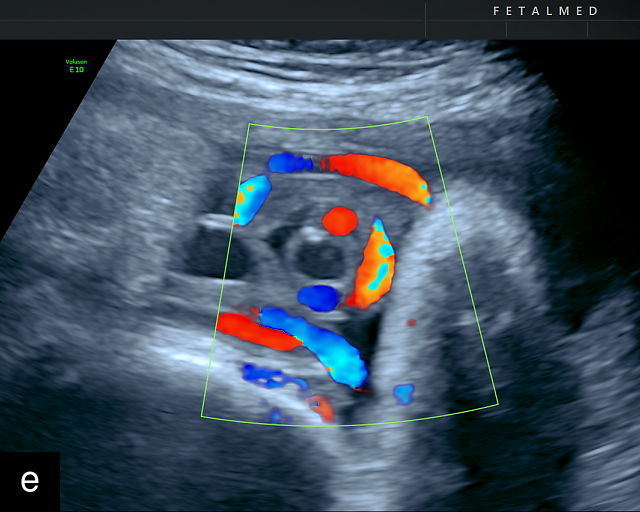
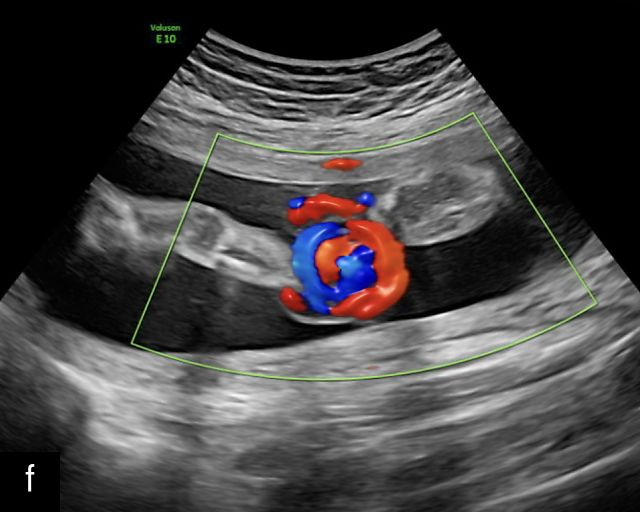
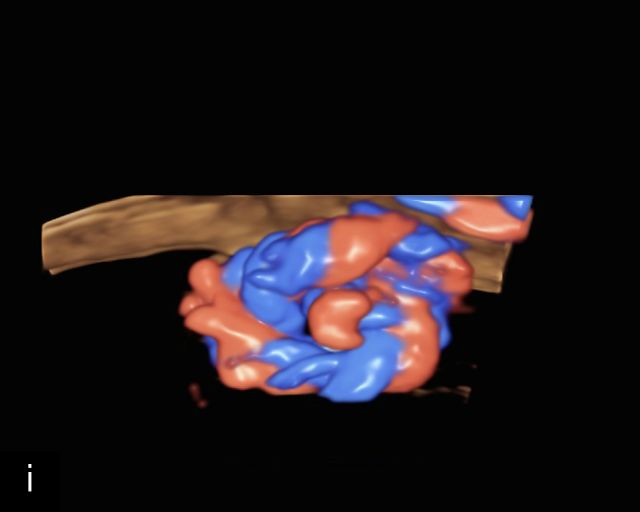
23
(a,b) Postnatal photographs showing double (a) and single (b) umbilical cord knots (arrows). (c–i) Grayscale (c), two-dimensional color Doppler (d–f) and three-dimensional color Doppler (g–i) images showing umbilical cord knots.
The sonographic finding of a cord knot has been described as resembling a four-leaf clover. Others have described a ‘cyclone’ sign.292 While grayscale ultrasound may lead to a suspicion of a cord knot (Figure 23c), color flow Doppler may enhance the diagnosis (Figure 23d–f). The use of three-dimensional and four-dimensional ultrasound with color flow Doppler may reveal convincing evidence of a cord knot (Figure 23g–i)).293 The reported sensitivity of ultrasound for the diagnosis of a true cord knot varies, but the diagnosis can be challenging with both false positives and false negatives. Redundancy of Wharton’s jelly may lead to a ‘false knot’ which is important to distinguish from a true knot.
Because of the association of a cord knot with adverse perinatal outcome, instituting antepartum fetal surveillance with biophysical profiles, serial growth ultrasounds and fetal heart rate monitoring with non-stress tests/cardiotocography is recommended.294 While it is tempting to perform cesarean delivery when a cord knot is found, labor with continuous fetal heart rate monitoring and early intervention for fetal heart rate abnormalities is a reasonable course of action. Rarely, two or more true knots of the cord may be found.
Ugurlucan and colleagues reported on the evaluation of the entire umbilical cord length between 18 and 23 weeks of gestation, tracing from the fetal insertion site to the placental insertion using grayscale ultrasound, with color Doppler applied as needed.295 They successfully performed this assessment in all but one patient. The mean time required to examine the umbilical cord was 41.5 ± 46.7 seconds, with color Doppler utilized in 22.8% of scans.295 Their study identified 104 cases (18.9%) of nuchal cord, six cases (1.1%) of SUA, two cases (0.4%) of umbilical cord knot and eight cases (1.5%) of VCI.
While these results are impressive, they have not been replicated by other studies. Additionally, some findings, such as nuchal cord and umbilical cord knot, have wide-ranging clinical significance. Detection at such an early gestational age is likely to cause considerable parental anxiety, given that the majority of these findings are associated with favorable outcome. Moreover, there are limited interventions available for these conditions and any intervention carries a high risk of iatrogenic complications, including preterm delivery.
Ultrasound also has a high false-positive rate for both nuchal cords and cord knots, further complicating the clinical utility of routine imaging of the entire cord during the mid-trimester. For these reasons, we do not currently recommend routine imaging of the entire umbilical cord. When cord knots are suspected during the mid-trimester, we suggest follow-up imaging in the third trimester, around 34 weeks, at which time practical management strategies, such as fetal kick count monitoring and electronic fetal heart rate monitoring, can be more effectively implemented.
Entangled cords in monoamniotic twins
Monoamniotic twins share a single amniotic cavity and are at significant risk for fetal death.296,297,298,299 They occur in approximately 1 in 10 000 pregnancies.300 Entanglement of the umbilical cords of the twins is the most commonly reported complication related to monoamniotic twins (Figure 24). Studies have shown that at least 90% of monoamniotic twin pregnancies have cord entanglement, which can be detected as early as the first trimester.301,302,303,304,305,306 In fact, the presence of cord entanglement is diagnostic for monoamniotic twins.304,307 This finding can be demonstrated using both grayscale ultrasound and color flow Doppler (Video 14). Cord entanglement may also occur in pseudo-monoamniotic twins when iatrogenic or spontaneous rupture of the dividing membrane occurs.308,309,310,311


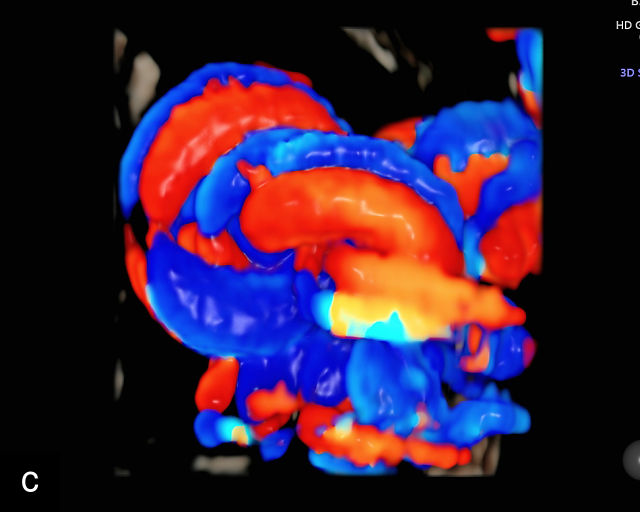
24
Photograph after delivery (a) and prenatal three-dimensional color Doppler images (b,c) of entangled cords in a monoamniotic twin pregnancy.
14
Grayscale (a) and color Doppler (b) ultrasound imaging of entangled cords in a monoamniotic twin pregnancy.
Distinguishing true cord entanglement from mere clumping or proximity of the cords can be challenging. However, when entanglement is present, the two umbilical cords will often exhibit different fetal heart rates, which can be confirmed with Doppler imaging. Color flow Doppler and three-dimensional ultrasound may further assist in diagnosis by providing clearer visualization of cord relationships.312,313,314,315 Early observations suggested that Doppler-detected notching of the umbilical artery waveform might indicate cord constriction or fetal compromise in monoamniotic twins with cord entanglement.316,317 However, subsequent studies have not consistently supported this association.318
Although cord entanglement in monoamniotic twins is a significant concern, most fetal deaths in these pregnancies are due to congenital malformations rather than entanglement itself.319 Given the high-risk nature of these pregnancies, most monoamniotic twins are admitted for close inpatient monitoring between 24 and 28 weeks of gestation and are typically delivered by cesarean section between 32 and 34 weeks to reduce the risk of fetal demise.320
Cord cysts
Cysts are relatively uncommon findings in the umbilical cord, with a reported incidence of 0.4% to 3.4% of pregnancies.185,191 They may present as single or multiple structures and are broadly categorized into true cysts and pseudocysts. True cysts are lined by epithelium and may arise from embryologic remnants, such as the urachus, allantois or yolk sac. Amniotic inclusion cysts, caused by amniotic epithelium encased in the cord, can also occur. Rarely, omphalomesenteric cysts have been reported. In contrast, pseudocysts lack an epithelial lining and result from focal degeneration of Wharton’s jelly. Differentiating between true cysts and pseudocysts is often challenging during prenatal imaging. The most common location for umbilical cord cysts is near the fetal abdominal insertion. Their size can vary greatly and pseudocysts are more commonly observed than true cysts.
Sonographically, umbilical cord cysts appear as hypoechoic, rounded structures with well-defined contours, either embedded within or attached to the cord (Figure 25; Video 15). These structures move with the cord, and color Doppler examination is essential to exclude vascular anomalies such as aneurysms or varices. The differential diagnosis for umbilical cord cysts includes cord tumors, omphalocele and gastroschisis.


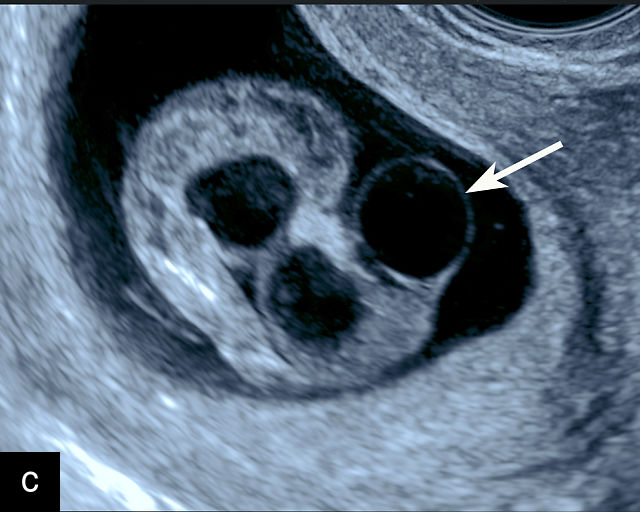
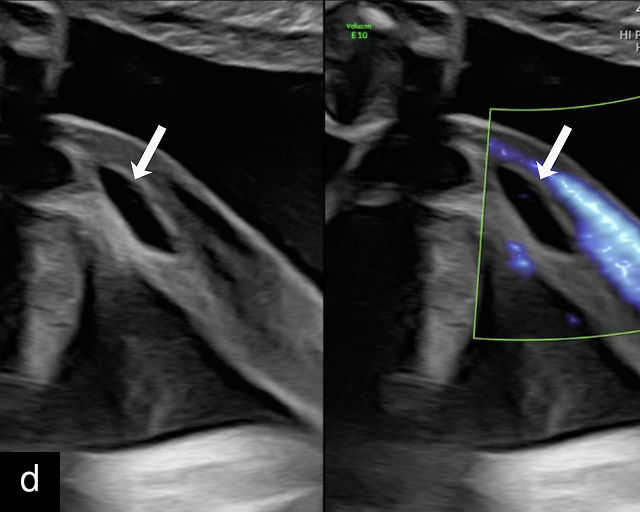
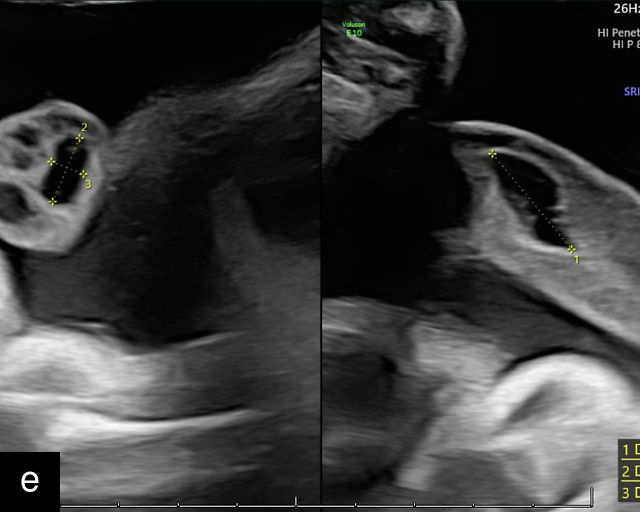

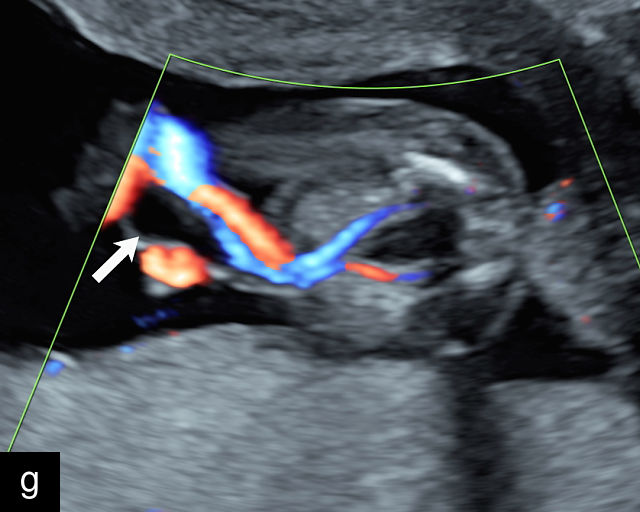
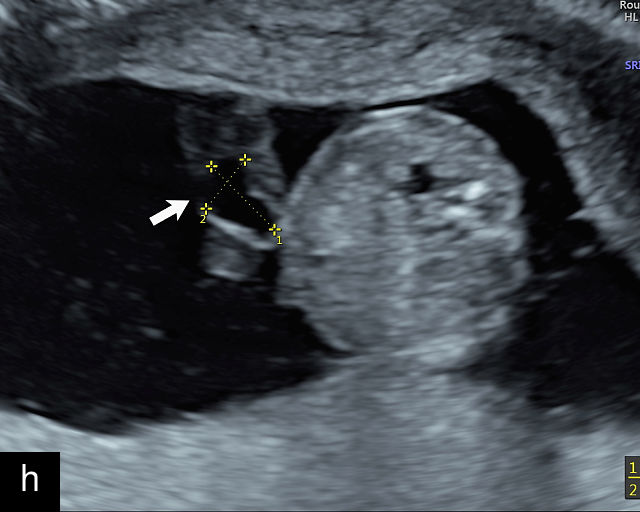
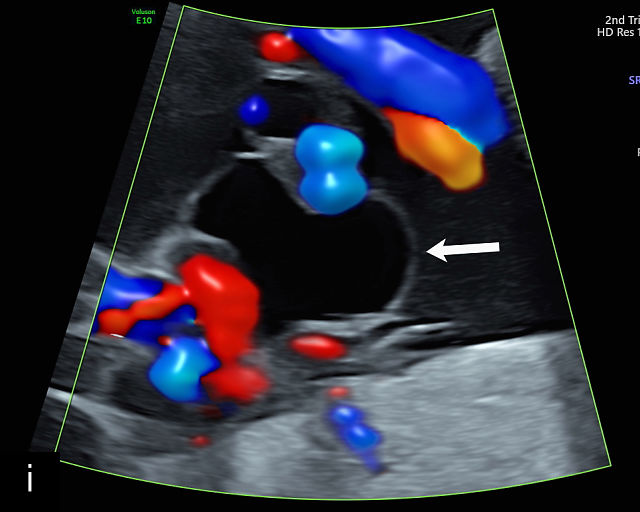
25
Umbilical cord cysts. (a) Grayscale and color flow Doppler images showing an umbilical cord cyst close to the fetal cord insertion at 20 weeks of gestation. (b) Measurement of the cord cyst. (c) Grayscale cross section of umbilical cord showing a cord cyst (arrow). (d) Grayscale and power Doppler image of umbilical cord showing a cord cyst (arrow). (e,f) Grayscale images of umbilical cord cyst. (g) Grayscale and color flow Doppler image of umbilical cord showing a cord cyst. (h) Grayscale images of umbilical cord cyst (arrow). (i) Color Doppler image of umbilical cord cyst (arrow).
15
(a–d) Grayscale and color Doppler imaging of umbilical cord cysts.
The detection of umbilical cord cysts can understandably cause anxiety for patients, sonographers and physicians, as their clinical significance is often uncertain. Cysts may be identified as early as the first trimester. Early studies suggested a potential association with chromosomal abnormalities, but more recent evidence indicates that isolated first-trimester umbilical cord cysts most often resolve and are frequently associated with favorable outcomes. However, caution is warranted when interpreting the literature, as reports may be biased toward cases with associated anomalies. Older studies, particularly those from the 1980s and 1990s, often reported an association between umbilical cord cysts and chromosomal abnormalities such as trisomy 18. Advances in ultrasound technology and high-sensitivity screening for trisomies 13, 18 and 21 have improved diagnostic accuracy and reduced false associations. Thus, isolated umbilical cord cysts with low-risk screening and no other anomalies on ultrasound are highly unlikely to be associated with chromosomal anomalies.
A patent urachus is one specific cause of cord cysts and numerous cases of prenatal diagnosis of this condition have been documented. An umbilical cord cyst may be associated with early-onset megacystis, and in this circumstance may represent a pop-off mechanism to reduce bladder tension.321 When an umbilical cord cyst is detected, a detailed fetal anatomical survey should be performed to assess for associated anomalies. The prognosis largely depends on whether additional abnormalities are present. In the presence of other fetal anomalies, invasive testing is recommended. Isolated cord cysts, with no other anomalies and low-risk screening results, may be reassuring.
Serial growth scans are advised for pregnancies with umbilical cord cysts. These cysts may enlarge or rupture, potentially increasing the risk of adverse outcomes, including stillbirth. Additionally, umbilical cord cysts have been associated with FGR, underscoring the need for close monitoring throughout pregnancy.
Supernumerary umbilical cord blood vessels
There have been rare reports of umbilical cords containing more than three vessels.322,323,324 However, in the majority of reports, this finding was noted only after delivery, rather than prenatally. Prenatal diagnosis of supernumerary umbilical vessels is exceedingly rare. Many of these are thought to be due to persistence of the right umbilical vein. When more than three vessels are observed in the cord, a detailed ultrasound examination of the fetus should be performed, since supernumerary fetal vessels may be associated with fetal structural abnormalities and genetic syndromes.
Umbilical cord tumors
Tumors of the umbilical cord are exceedingly rare.185 The most common is the umbilical cord angiomyxoma. Teratomas and hemangiomas of the cord have also been reported.192,193,325,326 Angiomyxoma is a benign soft-tissue solid mesenchymal tumor which contains numerous small blood vessels and a variable amount of myxoid material.327,328,329 These tumors are often referred to as cord hemangiomas, but this terminology can be misleading. Hemangiomas are predominantly vascular in nature, with large, prominent vessels that typically originate from the umbilical vessels. In contrast, angiomyxomas are characterized by a combination of abnormal small vessels and significant myxoid degeneration of the Wharton’s jelly, distinguishing them from true hemangiomas.327 These tumors may be associated with elevated maternal serum alpha-fetoprotein.329,330
Prenatal ultrasound diagnosis of angiomyxomas is uncommon, with most cases identified incidentally during placental examination after birth.330 On prenatal ultrasound, angiomyxomas typically appear as echogenic masses within the cord, adjacent to the umbilical blood vessels (Figure 26).327,329,330 Sensitive Doppler techniques may reveal low velocity flow within the tumor, and co-existing edema of the cord is often present. These tumors may occur anywhere along the cord and may even involve the entire cord length.330 In many cases, pseudocysts of the umbilical cord are also observed.


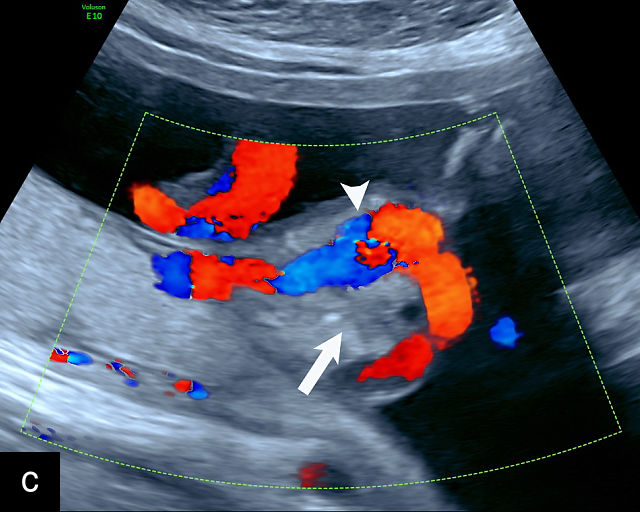
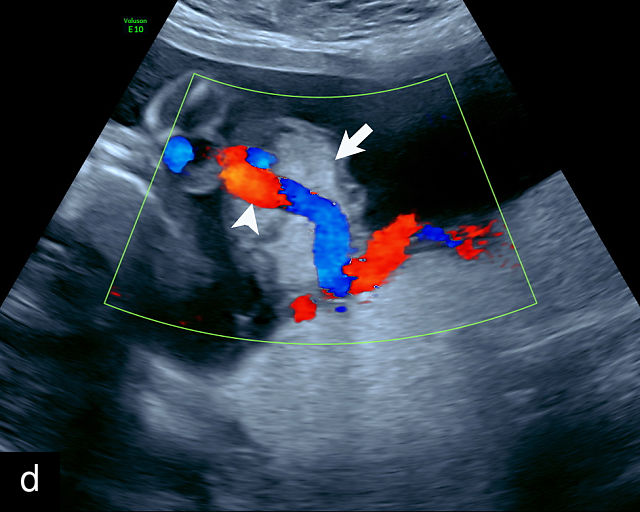
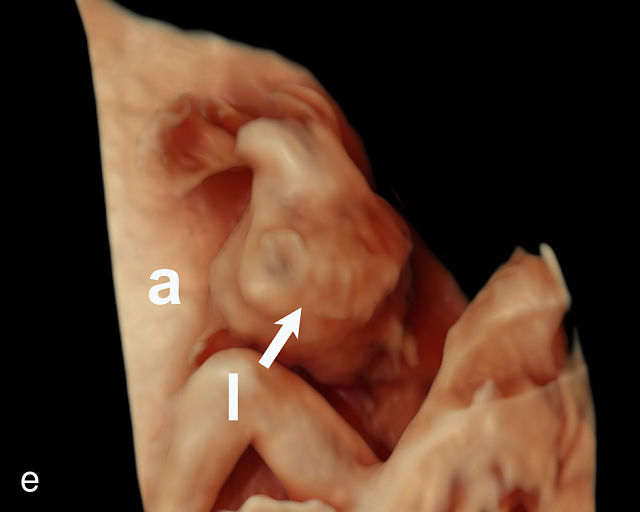
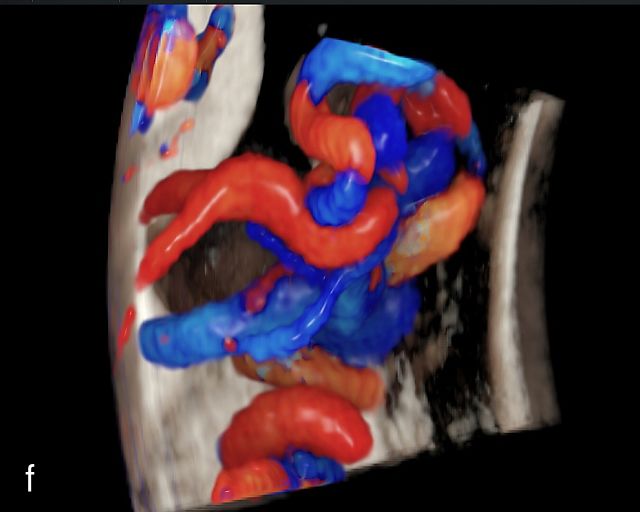
26
(a–d) Grayscale (a,b) and color Doppler (c,d) ultrasound images of umbilical cord angiomyxoma. There is an echogenic thickened area (arrow) surrounding the umbilical cord vessels (arrowhead). (e) Three-dimensional ultrasound image of umbilical cord angiomyxoma (arrow) at the fetal abdominal cord insertion. This could be mistaken for an omphalocele. a, fetal abdomen; l, fetal leg. (f) Three-dimensional color Doppler ultrasound image of umbilical cord angiomyxoma at the fetal cord insertion.
The differential diagnosis includes acardiac twin, hemangioma, teratoma, hematoma and even omphalocele, particularly when the mass is located close to the umbilical cord insertion into the fetal abdomen (Figure 26e).325,327 Angiomyxomas are associated with significant potential complications, including compression of fetal vessels, rupture of the tumor, FGR, cardiac overload, hydrops and, in severe cases, fetal death.327,331 There are also reports of umbilical artery occlusion, resulting in a SUA.
Given the potential for adverse perinatal outcome, it is essential for obstetric imaging specialists to recognize this abnormality.327 When cord thickening or a solid mass is identified within the cord, a high index of suspicion for angiomyxoma is warranted. Serial growth ultrasounds should be performed to monitor fetal wellbeing. If there is evidence of fetal distress, or as the pregnancy nears term, delivery should be considered, typically at around 37 weeks of gestation to mitigate risks.
Cord aneurysms
Umbilical cord aneurysms are very uncommon and rarely reported. They may be associated with chromosomal abnormalities, most notably trisomy 18. The advent of more detailed ultrasound means that trisomy 18 will be diagnosed prenatally in almost all cases. These fetuses have multiple abnormalities, and most cases will be detected by screening. When isolated aneurysms of the cord are identified on prenatal ultrasound, there is an increased risk for stillbirth. Thus, these patients should be monitored closely and delivered late preterm.
Umbilical vein varix
Umbilical vein varix (UVV) refers to a focal dilation of the intra-abdominal portion of the umbilical vein.332,333,334 Various definitions have been proposed, including dilation greater than 9 mm, more than 1.5 times the diameter of the intrahepatic umbilical vein or the non-dilated portion of the umbilical vein, or exceeding 2 standard deviations above the mean measurement for gestational age.335 The umbilical vein naturally increases in diameter with gestational age, from approximately 2 mm in the early second trimester to about 8 mm at term.336 The reported incidence of UVV ranges from 0.4 to 1 per 1000 pregnancies.337,338
On grayscale ultrasound, UVV appears as a focal dilation of the fetal intra-abdominal umbilical vein shortly after its entrance into the abdominal cavity (Figure 27).338,339 It presents as an anechoic cystic mass between the fetal abdominal wall and the lower margin of the fetal liver. Color Doppler imaging is essential for distinguishing UVV from other avascular intra-abdominal cystic structures. Imaging in multiple planes is recommended to confirm the diagnosis. Three-dimensional ultrasound may help with accurate diagnosis (Figure 27e,f).340
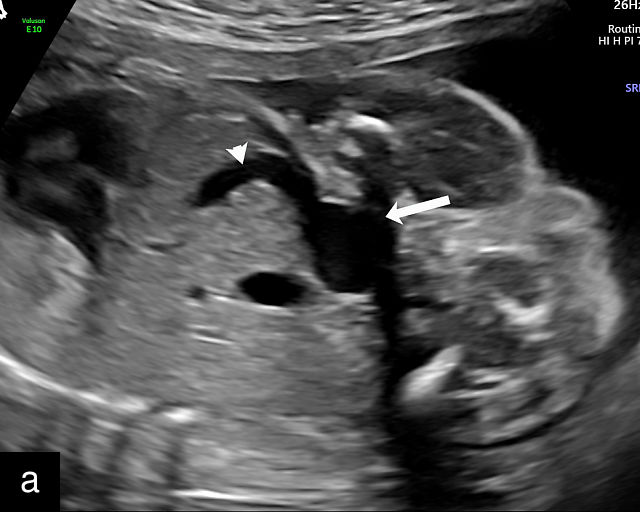
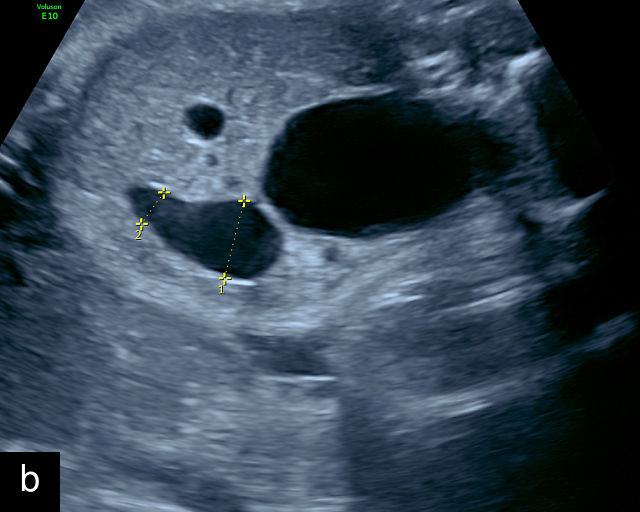

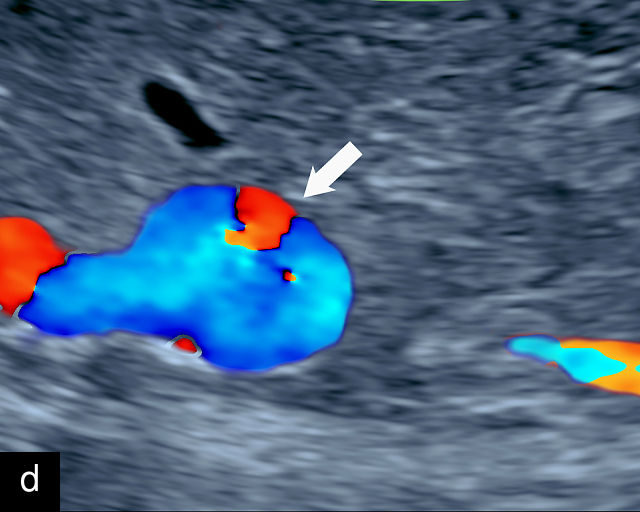
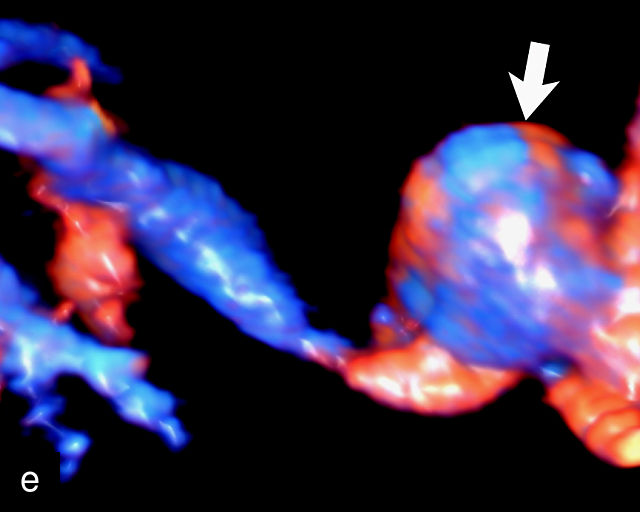

27
(a) Grayscale ultrasound image of umbilical vein varix. The fetal intra-abdominal umbilical vein (arrowhead) is noted to have a focal dilation (arrow). (b–f) Grayscale (b), two-dimensional color Doppler (c,d) and three-dimensional color Doppler (e,f) ultrasound images of umbilical vein varix, showing dilation of the umbilical vein (arrow) shortly after entering the fetal abdomen.
Early reports suggested that UVV might be associated with an increased risk of fetal demise, with some studies reporting stillbirth rates as high as 43%.332,339,341 The proposed pathophysiological mechanisms include umbilical vein thrombosis and fetal cardiac failure.332,342 Additionally, FGR has been observed in some cases.334 Up to 20% of fetuses with UVV have been found to have chromosomal or structural abnormalities.338
When UVV is identified on prenatal ultrasound, a detailed fetal anatomical survey is warranted to assess for associated anomalies.338,343,344 Serial ultrasound examinations are typically recommended to monitor fetal growth and evaluate the varix using grayscale and color Doppler for signs of turbulence or thrombosis. Novoa et al. suggested serial growth ultrasounds, fetal echocardiography and weekly biophysical profile (BPP) assessments starting at 32 weeks.343 While the optimal timing of delivery remains controversial, early-term delivery has been proposed in certain cases.
Recent systematic reviews and retrospective cohort studies suggest that adverse outcomes predominantly occur in non-isolated UVV cases (i.e. those with additional anomalies), whereas isolated UVV is associated with favorable outcomes.334,335,338,344 A 2017 systematic review and meta-analysis by Di Pasquo et al. reported that 19.2% of fetuses with UVV had additional anomalies, similar to the 20% reported in Novoa et al.’s study.338,343 Importantly, no chromosomal abnormalities or stillbirths were identified in cases of isolated UVV and the pooled incidence of small-for-gestational-age infants was 3.7%. In contrast, among non-isolated UVV cases, the fetal death rate was 7.3%.338
These findings suggest that isolated UVV is associated with an excellent prognosis, while the presence of additional anomalies significantly increases the risk of adverse outcomes.333,334,338,344 Patients with isolated UVV can generally be reassured, though serial ultrasounds and third-trimester fetal surveillance may still be considered as a precaution.
Cord strictures, torsion and absence of Wharton's jelly
The umbilical cord may rarely undergo torsion, have local areas of stricture, or may have absence of Wharton’s jelly. Spontaneous rupture of Warton’s jelly and the umbilical cord have been described. All these conditions are associated with increased risk for stillbirth. Most cases are identified incidentally on examination of the placenta and cord after stillbirth. Only extremely rarely are they found on prenatal ultrasound.
Funic presentation
Funic presentation refers to the presence of free loops of the umbilical cord overlying the internal cervical os (Figure 28; Video 16). This is not an uncommon finding on prenatal ultrasound. In most cases, the cord loops are mobile and do not remain in that position permanently, typically shifting away from the cervix with maternal position changes or as the pregnancy progresses.
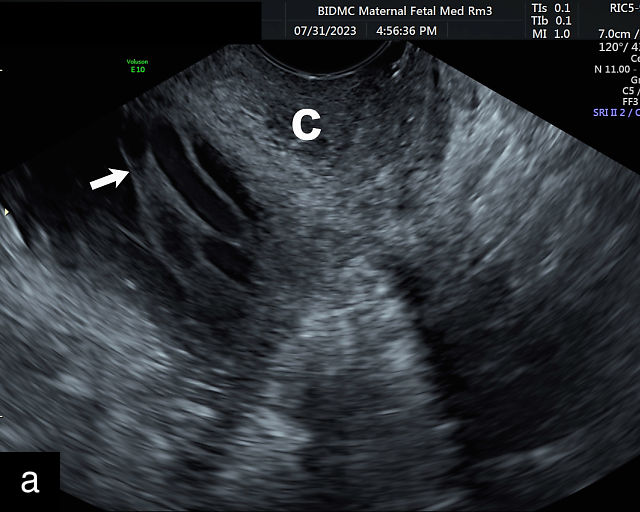
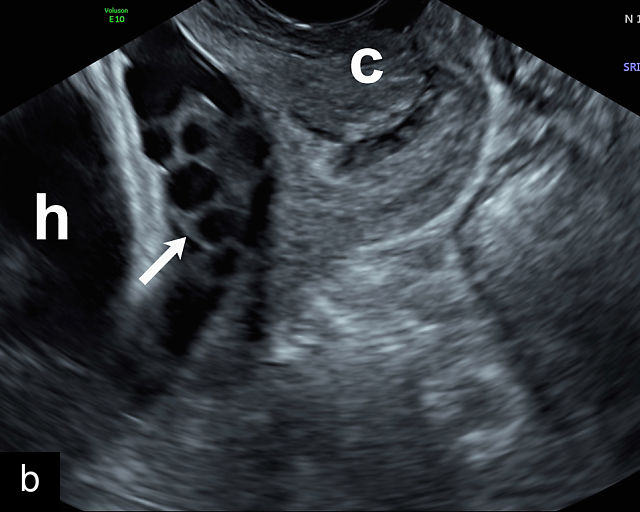

28
(a–c) Grayscale (a,b) and color Doppler (c) transvaginal ultrasound images of funic presentation, showing loops of cord (arrow) overlying the cervix. In these cases, it is important to rule out vasa previa. c, cervix; h, fetal head.
16
Transvaginal ultrasound imaging with color flow Doppler of funic presentation, showing loops of cord overlying the cervix. In these cases it is important to rule out vasa previa.
In the earlier days of ultrasound, funic presentation raised concern due to the perceived increased risk of umbilical cord prolapse during labor. However, the majority of funic presentations identified on ultrasound resolve spontaneously on follow-up imaging. That said, when funic presentation is associated with a marginal cord insertion near the edge of a low-lying placenta, it is more likely to persist. In such cases, labor may still be allowed, but careful intrapartum monitoring is warranted due to the elevated risk of cord prolapse.
Whenever funic presentation is observed, it is essential to rule out vasa previa. In contrast to funic presentation, vasa previa involves fetal vessels that are fixed within the membranes and do not change position with maternal movement or on serial examinations.
THE MEMBRANES
Abnormalities of the membranes
Chorioamniotic separation
In early pregnancy, the chorion and amnion are not fused (Figure 29a; Video 17a) but fusion typically occurs by 16 weeks. Delayed fusion has been associated with chromosomal abnormalities.345 Chorioamniotic separation may be spontaneous or iatrogenic, following such procedures as amniocentesis or fetoscopy (Figure 29b; Video 17b,c).346 Chorioamniotic separation has been associated with increased risks for stillbirth, premature rupture of the membranes and preterm delivery.347,348,349 Some stillbirths result from cord strangulation by the detached membrane.349,350 On sonographic examination, chorioamniotic separation typically presents as membranes within the amniotic cavity, shown as a fine linear or crumpled appearing hyperechoic structure that is mobile and separated from the chorion and uterine wall by a hypoechoic space.351,352

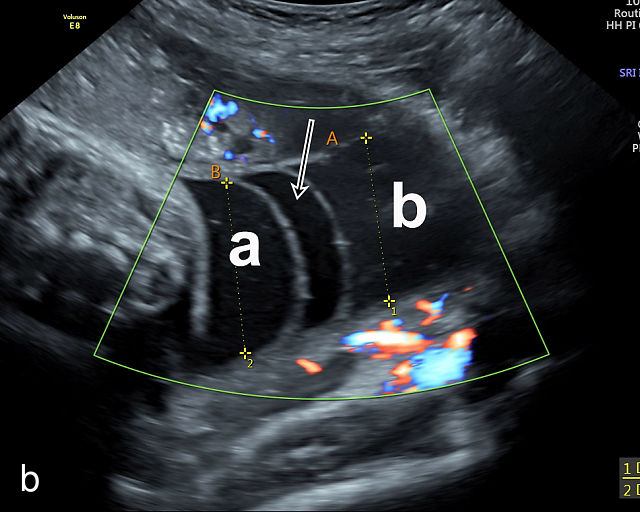
29
(a) Unfused membranes (arrows) at 13 weeks. This is a normal finding at this gestational age. (b) Spontaneous chorioamniotic separation of the membranes between dichorionic twins. The separation is shown by the area (arrow) between the sacs of Twin a (a) and Twin b (b).
17
(a) Unfused membranes at 13 weeks. (b) Chorioamniotic separation in a monochorionic twin pregnancy following fetoscopic laser photocoagulation. (c) Spontaneous separation of the dividing membranes in a dichorionic diamniotic twin pregnancy.
Amniotic band syndrome
Amniotic band syndrome is estimated to occur in approximately 1 in 1200 live births.353 Although multiple etiologies have been proposed, the prevailing theory is that the condition results from an early disruption of the amnion, allowing the fetus to enter the chorionic space. This exposure can lead to the formation of fibrous amniotic strands that entangle fetal structures. The syndrome encompasses a range of anomalies with varying diagnostic criteria, and its etiology is considered multifactorial. This variation contributes to discrepancies in reported incidence.
Remnants of the amnion that are free-floating can become entangled with fetal structures, leading to amputations, strangulation and deformities. Amniotic band syndrome may also occur following chorioamniotic separation, which can happen spontaneously or as a result of fetal interventions such as amniocentesis and fetoscopy.354 The condition is characterized by craniofacial, body-wall and limb anomalies, including irregular amputation of fetal digits and extremities, facial cleft, abdominal wall defect, ectopia cordis, spina bifida, acrania or anencephaly, cephalocele, kyphoscoliosis, bladder and cloacal exstrophy, unusually short limbs or extremities and abnormal limb posturing.353,355,356,357,358,359,360,361,362,363 In some cases, amniotic bands may lead to stricture, occlusion or even transection of the umbilical cord, which can result in fetal death or FGR.
Sonographic findings associated with amniotic band syndrome include otherwise unexplained irregular and eccentrically placed amputations and clefts (Figure 30; Video 18). The condition has been diagnosed as early as the first trimester, typically between 10 and 12 weeks of gestation. In addition to amputations and clefts, isolated swollen limbs may be observed, often with a stricture proximal to the swelling.360 The fetal head and trunk may appear grossly misshapen. In some instances, an amniotic band may be visualized as a linear echogenic structure that moves with the fetus, though often these bands are not sonographically detectable. Whenever facial clefts, abdominal wall defects, neural tube defects, amputations or anencephaly are identified, a detailed ultrasound examination should be performed to assess for amniotic band syndrome.355 Three-dimensional ultrasound may assist in accurate diagnosis of amniotic band syndrome.
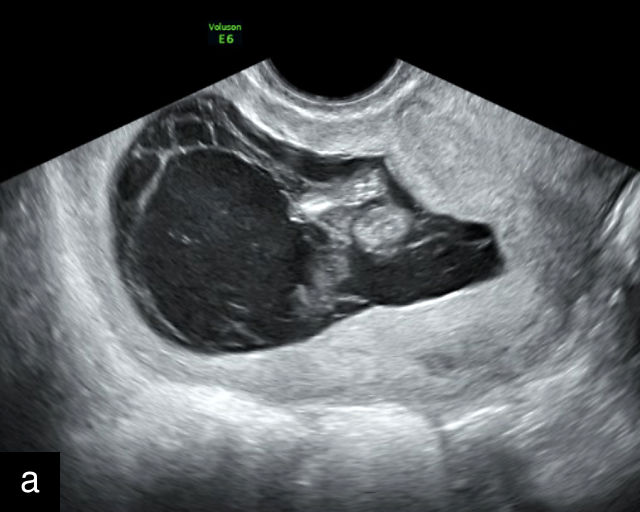
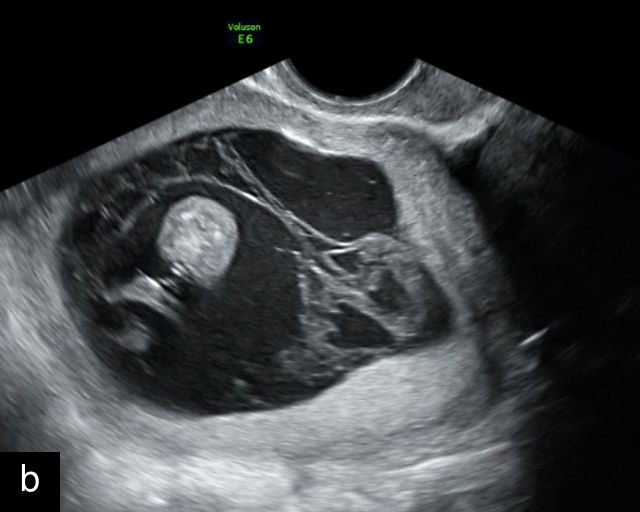
30
(a,b) Grayscale ultrasound images, showing multiple amniotic bands surrounding a 12-week fetus.
18
Grayscale (a) and color Doppler (b) ultrasound imaging, showing multiple amniotic bands surrounding the fetus. The fetus can be seen to be grossly abnormal with several defects, including an abdominal wall defect and absence of a normal cranium.
In selected cases, intrauterine fetoscopic transection of amniotic bands has been successfully performed to release entrapment and prevent further fetal compromise.363,364,365,366,367,368
CONCLUSIONS
Abnormalities of the placenta and umbilical cord are frequently detected on prenatal ultrasound. While some of these abnormalities may have significant consequences for both the fetus and the mother – ranging from serious morbidity to, in the most severe cases, fetal death – others may be benign and associated with favorable outcome. It is essential for those involved in prenatal diagnosis to have a thorough understanding of these entities to provide accurate assessments and counseling.
Advancements in technology, including color flow Doppler, more sensitive Doppler techniques and three-dimensional ultrasound, have significantly improved our ability to detect and accurately diagnose placental and cord abnormalities. Additionally, the development of fetal and intrauterine interventions offers promising new avenues for improving outcomes in selected cases.
However, while some cord abnormalities detected on prenatal ultrasound may be common and benign, they can still cause unnecessary alarm and lead to unwarranted interventions. It is crucial to balance the accurate identification of abnormalities with appropriate management strategies to avoid iatrogenic complications.
The evaluation of the placenta and umbilical cord should extend beyond basic assessments, such as merely evaluating placental location and the number of cord vessels.2,12,369 To make a meaningful impact on perinatal mortality and morbidity and to ensure the safest possible outcomes for mothers and babies, those performing ultrasound examinations must be well-versed in the spectrum of placental and cord abnormalities and the imaging modalities required for their accurate diagnosis.
PRACTICE RECOMMENDATIONS
- Perform detailed ultrasound examinations of the placenta and umbilical cord to identify potential abnormalities.
- Use a systematic approach to evaluate the placenta, including the uterine wall, decidua/chorion interface, placental body and umbilical cord insertion.
- In cases of placenta previa, perform transvaginal ultrasound to confirm the diagnosis and accurately assess the relationship between the placenta and the internal cervical os.
- Be aware of the risk factors for placenta accreta spectrum (PAS) disorders, such as placenta previa and prior cesarean deliveries, and screen high-risk patients appropriately.
- When a thickened placenta is detected, conduct a thorough evaluation to identify potential fetal, maternal or placental causes.
- Differentiate placental lakes from lacunae using Doppler ultrasound to assess blood flow characteristics.
- In cases of accessory placental lobes, examine the placenta at delivery to ensure complete removal and prevent postpartum hemorrhage.
- Recognize that placental grading is not a reliable tool for assessing fetal lung maturity or predicting adverse perinatal outcome.
- When a single umbilical artery (SUA) is identified, perform a detailed fetal anatomical survey to check for associated anomalies.
- Understand that most cases of abnormal umbilical cord coiling do not require intervention but may warrant closer surveillance.
- In cases of velamentous cord insertion (VCI), use color flow Doppler to identify unprotected fetal vessels and assess for vasa previa.
- Perform a color flow Doppler sweep over the cervix to rule out vasa previa, especially in the presence of risk factors like VCI or low-lying placenta.
- When a chorioangioma is detected, conduct weekly ultrasound examinations to monitor amniotic fluid volume and fetal cardiac function.
- Be aware that placental abruption can have variable ultrasound findings, and clinical correlation is essential for diagnosis.
- When umbilical cord cysts are identified, perform a detailed fetal anatomical survey to assess for associated anomalies.
- In cases of monoamniotic twins, recognize the high risk of cord entanglement and consider close inpatient monitoring and scheduled delivery.
- Exercise caution when diagnosing circumvallate placenta due to the high rate of false positives on ultrasound.
- Differentiate placental mesenchymal dysplasia (PMD) from molar pregnancy using Doppler imaging, hCG and AFP levels, and NIPT results.
- When a cord knot is suspected, consider follow-up imaging in the third trimester for appropriate management planning.
- Inform patients of nuchal cords detected in the late third trimester, and advise them to monitor fetal movements closely.
CONFLICTS OF INTEREST
The author(s) of this chapter declare that they have no interests that conflict with the contents of the chapter.
Feedback
Publishers’ note: We are constantly trying to update and enhance chapters in this Series. So if you have any constructive comments about this chapter please provide them to us by selecting the "Your Feedback" link in the left-hand column.
ACKNOWLEDGMENT
The authors thank Dr Martin Chavez for kindly providing images of placental choriangioma (Figure 12 and Video 7).
REFERENCES
Oyelese Y. Placenta, umbilical cord and amniotic fluid: the not-less-important accessories. Clin Obstet Gynecol. Mar 2012;55(1):307–23. doi: 10.1097/GRF.0b013e318248818e. | |
Hernandez-Andrade E, Gerulewicz D, Soto-Torres E, et al. We Should Perform a Systematic Evaluation of the Placenta. J Ultrasound Med. Jan 9 2025;doi: 10.1002/jum.16645. | |
Jauniaux E, Lees C, Bhide A, Daly-Jones E, Srinivasan D, Oyelese Y. The placenta and umbilical cord in prenatal care: Unseen, overlooked and misunderstood. BJOG. Jan 2025;132(1):12–14. doi: 10.1111/1471-0528.17936. | |
Jauniaux E, Ebbing C, Oyelese Y, Maymon R, Prefumo F, Bhide A. European association of perinatal medicine (EAPM) position statement: Screening, diagnosis and management of congenital anomalies of the umbilical cord. Eur J Obstet Gynecol Reprod Biol. Jul 2024;298:61–65. doi: 10.1016/j.ejogrb.2024.04.044. | |
Abramowicz JS, Sheiner E. Ultrasound of the placenta: a systematic approach. Part I: Imaging. Placenta. Mar 2008;29(3):225–40. doi: 10.1016/j.placenta.2007.12.006. | |
Hammad IA, Blue NR, Allshouse AA, et al. Umbilical Cord Abnormalities and Stillbirth. Obstet Gynecol. Mar 2020;135(3):644–652. doi: 10.1097/AOG.0000000000003676. | |
Ebbing C, Rasmussen S, Kessler J, Moster D. Association of placental and umbilical cord characteristics with cerebral palsy: national cohort study. Ultrasound Obstet Gynecol. Feb 2023;61(2):224–230. doi: 10.1002/uog.26047. | |
Nasiell J, Papadogiannakis N, Lof E, Elofsson F, Hallberg B. Hypoxic ischemic encephalopathy in newborns linked to placental and umbilical cord abnormalities. J Matern Fetal Neonatal Med. Mar 2016;29(5):721–6. doi: 10.3109/14767058.2015.1015984. | |
Hernandez-Andrade E, Gerulewicz-Vannini D, Soto-Torres EE, Papanna R. Rare pathologic Placenta Ultrasound Findings. Clin Obstet Gynecol. Dec 2 2024;doi: 10.1097/GRF.0000000000000912. | |
Harris RD, Barth RA. Sonography of the gravid uterus and placenta: current concepts. AJR Am J Roentgenol. Mar 1993;160(3):455–65. doi: 10.2214/ajr.160.3.8430536. | |
Harris RD, Cho C, Wells WA. Sonography of the placenta with emphasis on pathological correlation. Semin Ultrasound CT MR. Feb 1996;17(1):66–89. doi: 10.1016/s0887-2171(96)90045-1. | |
Sepulveda W, Wong AE, Gomez L, Alcalde JL. Improving sonographic evaluation of the umbilical cord at the second-trimester anatomy scan. J Ultrasound Med. Jun 2009;28(6):831–5. doi: 10.7863/jum.2009.28.6.831. | |
Keller NA, Kouba I, Stefanov DG, et al. Presence and Size of Placental Lakes on 20-Week Fetal Anatomy Ultrasound and Obstetrical Outcomes. J Obstet Gynaecol Can. Jun 2024;46(6):102458. doi: 10.1016/j.jogc.2024.102458. | |
Gottesfeld KR, Thompson HE, Holmes JH, Taylor ES. Ultrasonic placentography–a new method for placental localization. Am J Obstet Gynecol. Oct 15 1966;96(4):538–47. doi: 10.1016/s0002-9378(16)34691-9. | |
Hernandez-Andrade E, Huntley ES, Bartal MF, Soto-Torres EE, Tirosh D, Jaiman S, Johnson A. Doppler evaluation of normal and abnormal placenta. Ultrasound Obstet Gynecol. Jul 2022;60(1):28–41. doi: 10.1002/uog.24816. | |
Jauniaux E, Zosmer N, D'Antonio F, Hussein AM. Placental lakes vs lacunae: spot the differences. Ultrasound Obstet Gynecol. Feb 2024;63(2):173–180. doi: 10.1002/uog.27453. | |
Jauniaux E, Ramsay B, Campbell S. Ultrasonographic investigation of placental morphologic characteristics and size during the second trimester of pregnancy. Am J Obstet Gynecol. Jan 1994;170(1 Pt 1):130–7. doi: 10.1016/s0002-9378(94)70397-3. | |
Reis NS, Brizot ML, Schultz R, Nomura RM, Zugaib M. Placental lakes on sonographic examination: correlation with obstetric outcome and pathologic findings. J Clin Ultrasound. Feb 2005;33(2):67–71. doi: 10.1002/jcu.20086. | |
Thompson MO, Vines SK, Aquilina J, Wathen NC, Harrington K. Are placental lakes of any clinical significance? Placenta. Sep-Oct 2002;23(8–9):685–90. doi: 10.1053/plac.2002.0837. | |
Hwang HS, Sohn IS, Kwon HS. The clinical significance of large placental lakes. Eur J Obstet Gynecol Reprod Biol. Jun 2012;162(2):139–43. doi: 10.1016/j.ejogrb.2012.02.015. | |
Angtuaco TL, Boyd CM, Marks SR, Quirk JG, Galwas B. Sonographic diagnosis of the bilobate placenta. J Ultrasound Med. Nov 1986;5(11):672–4. doi: 10.7863/jum.1986.5.11.672. | |
Giouleka S, Siargkas A, Tsakiridis I, Mamopoulos A, Athanasiadis A, Dagklis T. Prenatal diagnosis of bilobate placenta: incidence, risk factors and impact on pregnancy outcomes. J Perinat Med. Nov 27 2023;51(9):1132–1138. doi: 10.1515/jpm-2023-0122. | |
Has R, Yuksel A, Gunay S, Aydinhan E, Ozgun T. Antenatal sonographic detection of an unusual placental lake under the cord insertion. Arch Gynecol Obstet. Jan 2005;271(1):59–61. doi: 10.1007/s00404-004-0649-3. | |
Smith M, McCullum B. Furcate umbilical cord insertion with a bilobed placenta identified on prenatal ultrasonography. Australas J Ultrasound Med. May 2022;25(2):98–102. doi: 10.1002/ajum.12293. | |
Kamijo K, Miyamoto T, Ando H, et al. Clinical characteristics of a novel 'Type- 3' vasa previa: case series at a single center. J Matern Fetal Neonatal Med. Dec 2022;35(25):7730–7736. doi: 10.1080/14767058.2021.1960975. | |
Larcher L, Jauniaux E, Lenzi J, et al. Ultrasound diagnosis of placental and umbilical cord anomalies in singleton pregnancies resulting from in-vitro fertilization. Placenta. Jan 2023;131:58–64. doi: 10.1016/j.placenta.2022.11.010. | |
Matsuzaki S, Ueda Y, Matsuzaki S, et al. The Characteristics and Obstetric Outcomes of Type II Vasa Previa: Systematic Review and Meta-Analysis. Biomedicines. Dec 15 2022;10(12) doi: 10.3390/biomedicines10123263. | |
Tachibana D, Misugi T, Pooh RK, et al. Placental Types and Effective Perinatal Management of Vasa Previa: Lessons from 55 Cases in a Single Institution. Diagnostics (Basel). Jul 29 2021;11(8) doi: 10.3390/diagnostics11081369. | |
Suekane T, Tachibana D, Pooh RK, Misugi T, Koyama M. Type-3 vasa previa: normal umbilical cord insertion cannot exclude vasa previa in cases with abnormal placental location. Ultrasound Obstet Gynecol. Apr 2020;55(4):556–557. doi: 10.1002/uog.20347. | |
Oyelese Y, Javinani A, Shamshirsaz AA. Vasa Previa. Obstet Gynecol. Sep 1 2023;142(3):503–518. doi: 10.1097/AOG.0000000000005287. | |
Spirt BA, Kagan EH, Gordon LP, Massad LS. Antepartum diagnosis of a succenturiate lobe: sonographic and pathologic correlation. J Clin Ultrasound. Mar 1981;9(3):139–40. doi: 10.1002/jcu.1870090309. | |
Suzuki S, Igarashi M. Clinical significance of pregnancies with succenturiate lobes of placenta. Arch Gynecol Obstet. Apr 2008;277(4):299–301. doi: 10.1007/s00404-007-0482-6. | |
Cavaliere AF, Rosati P, Ciliberti P, Buongiorno S, Guariglia L, Scambia G, Tintoni M. Succenturiate lobe of placenta with vessel anomaly: a case report of prenatal diagnosis and literature review. Clin Imaging. Sep-Oct 2014;38(5):747–50. doi: 10.1016/j.clinimag.2014.01.018. | |
Fukuda E, Hamuro A, Kitada K, et al. The Impact of Assisted Reproductive Technology on Umbilical Cord Insertion: Increased Risk of Velamentous Cord Insertion in Singleton Pregnancies Conceived through ICSI. Medicina (Kaunas). Sep 25 2023;59(10) doi: 10.3390/medicina59101715. | |
Oyelese Y, Catanzarite V, Prefumo F, et al. Vasa previa: the impact of prenatal diagnosis on outcomes. Obstet Gynecol. May 2004;103(5 Pt 1):937–42. doi: 10.1097/01.AOG.0000123245.48645.98. | |
Pavalagantharajah S, Villani LA, D'Souza R. Vasa previa and associated risk factors: a systematic review and meta-analysis. Am J Obstet Gynecol MFM. Aug 2020;2(3):100117. doi: 10.1016/j.ajogmf.2020.100117. | |
Grannum PA, Berkowitz RL, Hobbins JC. The ultrasonic changes in the maturing placenta and their relation to fetal pulmonic maturity. Am J Obstet Gynecol. Apr 15 1979;133(8):915–22. doi: 10.1016/0002-9378(79)90312-0. | |
Quinlan RW, Cruz AC, Buhi WC, Martin M. Changes in placental ultrasonic appearance. I. Incidence of Grade III changes in the placenta in correlation to fetal pulmonary maturity. Am J Obstet Gynecol. Oct 15 1982;144(4):468–70. | |
Hill LM, Breckle R, Ragozzino MW, Wolfgram KR, O'Brien PC. Grade 3 placentation: incidence and neonatal outcome. Obstet Gynecol. Jun 1983;61(6):728–32. | |
Brown HL, Miller JM, Jr., Khawli O, Gabert HA. Premature placental calcification in maternal cigarette smokers. Obstet Gynecol. Jun 1988;71(6 Pt 1):914–7. | |
Vintzileos AM, Campbell WA, Feinstein SJ, Lodeiro JG, Weinbaum PJ, Nochimson DJ. The fetal biophysical profile in pregnancies with grade III placentas. Am J Perinatol. Apr 1987;4(2):90–3. doi: 10.1055/s-2007-999746. | |
Vintzileos AM, Campbell WA, Ingardia CJ, Nochimson DJ. The fetal biophysical profile and its predictive value. Obstet Gynecol. Sep 1983;62(3):271–8. doi: 10.1097/00006250-198309000-00001. | |
Moran M, Ryan J, Higgins M, Brennan PC, McAuliffe FM. Poor agreement between operators on grading of the placenta. J Obstet Gynaecol. 2011;31(1):24–8. doi: 10.3109/01443615.2010.522266. | |
Sau A, Seed P, Langford K. Intraobserver and interobserver variation in the sonographic grading of placental maturity. Ultrasound Obstet Gynecol. Apr 2004;23(4):374–7. doi: 10.1002/uog.1004. | |
Walker MG, Hindmarsh PC, Geary M, Kingdom JCP. Sonographic maturation of the placenta at 30 to 34 weeks is not associated with second trimester markers of placental insufficiency in low-risk pregnancies. J Obstet Gynaecol Can. Dec 2010;32(12):1134–1139. doi: 10.1016/S1701-2163(16)34736-3. | |
Mastrolia SA, Weintraub AY, Sciaky-Tamir Y, Tirosh D, Loverro G, Hershkovitz R. Placental calcifications: a clue for the identification of high-risk fetuses in the low-risk pregnant population? J Matern Fetal Neonatal Med. Mar 2016;29(6):921–7. doi: 10.3109/14767058.2015.1023709. | |
McKenna D, Tharmaratnam S, Mahsud S, Dornan J. Ultrasonic evidence of placental calcification at 36 weeks' gestation: maternal and fetal outcomes. Acta Obstet Gynecol Scand. Jan 2005;84(1):7–10. doi: 10.1111/j.0001-6349.2005.00563.x. | |
Chen KH, Seow KM, Chen LR. The role of preterm placental calcification on assessing risks of stillbirth. Placenta. Sep 2015;36(9):1039–44. doi: 10.1016/j.placenta.2015.06.015. | |
Oyelese Y. Placenta previa: the evolving role of ultrasound. Ultrasound Obstet Gynecol. Aug 2009;34(2):123–6. doi: 10.1002/uog.7312. | |
Silver RM. Abnormal Placentation: Placenta Previa, Vasa Previa, and Placenta Accreta. Obstet Gynecol. Sep 2015;126(3):654–668. doi: 10.1097/AOG.0000000000001005. | |
Oyelese Y, Shainker SA. Placenta Previa. Clin Obstet Gynecol. Dec 10 2024;doi: 10.1097/GRF.0000000000000911. | |
Iyasu S, Saftlas AK, Rowley DL, Koonin LM, Lawson HW, Atrash HK. The epidemiology of placenta previa in the United States, 1979 through 1987. Am J Obstet Gynecol. May 1993;168(5):1424–9. doi: 10.1016/s0002-9378(11)90776-5. | |
Getahun D, Oyelese Y, Salihu HM, Ananth CV. Previous cesarean delivery and risks of placenta previa and placental abruption. Obstet Gynecol. Apr 2006;107(4):771–8. doi: 10.1097/01.AOG.0000206182.63788.80. | |
Faiz AS, Ananth CV. Etiology and risk factors for placenta previa: an overview and meta-analysis of observational studies. J Matern Fetal Neonatal Med. Mar 2003;13(3):175–90. doi: 10.1080/jmf.13.3.175.190. | |
Ananth CV, Smulian JC, Vintzileos AM. The association of placenta previa with history of cesarean delivery and abortion: a metaanalysis. Am J Obstet Gynecol. Nov 1997;177(5):1071–8. doi: 10.1016/s0002-9378(97)70017-6. | |
Shobeiri F, Jenabi E. Smoking and placenta previa: a meta-analysis. J Matern Fetal Neonatal Med. Dec 2017;30(24):2985–2990. doi: 10.1080/14767058.2016.1271405. | |
Zhang J, Fried DB. Relationship of maternal smoking during pregnancy to placenta previa. Am J Prev Med. Sep-Oct 1992;8(5):278–82. | |
Sheiner E, Shoham-Vardi I, Hallak M, Hershkowitz R, Katz M, Mazor M. Placenta previa: obstetric risk factors and pregnancy outcome. J Matern Fetal Med. Dec 2001;10(6):414–9. doi: 10.1080/714052784. | |
Rosenberg T, Pariente G, Sergienko R, Wiznitzer A, Sheiner E. Critical analysis of risk factors and outcome of placenta previa. Arch Gynecol Obstet. Jul 2011;284(1):47–51. doi: 10.1007/s00404-010-1598-7. | |
Jauniaux E, Alfirevic Z, Bhide AG, et al. Placenta Praevia and Placenta Accreta: Diagnosis and Management: Green-top Guideline No. 27a. BJOG. Jan 2019;126(1):e1-e48. doi: 10.1111/1471-0528.15306. | |
Oyelese Y, Smulian JC. Placenta previa, placenta accreta, and vasa previa. Obstet Gynecol. Apr 2006;107(4):927–41. doi: 10.1097/01.AOG.0000207559.15715.98. | |
Jansen C, Kleinrouweler CE, Kastelein AW, Ruiter L, van Leeuwen E, Mol BW, Pajkrt E. Follow-up ultrasound in second-trimester low-positioned anterior and posterior placentae: prospective cohort study. Ultrasound Obstet Gynecol. Nov 2020;56(5):725–731. doi: 10.1002/uog.21903. | |
Yadav S, Shah A. Low placenta at the mid trimester anomaly scan-A cause for concern? J Clin Ultrasound. Jan 2025;53(1):155–162. doi: 10.1002/jcu.23841. | |
Jansen C, Kleinrouweler CE, van Leeuwen L, et al. Final outcome of a second trimester low-positioned placenta: A systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. Sep 2019;240:197–204. doi: 10.1016/j.ejogrb.2019.06.020. | |
Heller HT, Mullen KM, Gordon RW, Reiss RE, Benson CB. Outcomes of pregnancies with a low-lying placenta diagnosed on second-trimester sonography. J Ultrasound Med. Apr 2014;33(4):691–6. doi: 10.7863/ultra.33.4.691. | |
Reddy UM, Abuhamad AZ, Levine D, Saade GR, Fetal Imaging Workshop Invited P. Fetal imaging: Executive summary of a Joint Eunice Kennedy Shriver National Institute of Child Health and Human Development, Society for Maternal-Fetal Medicine, American Institute of Ultrasound in Medicine, American College of Obstetricians and Gynecologists, American College of Radiology, Society for Pediatric Radiology, and Society of Radiologists in Ultrasound Fetal Imaging Workshop. Am J Obstet Gynecol. May 2014;210(5):387–97. doi: 10.1016/j.ajog.2014.02.028. | |
Oppenheimer LW, Farine D. A new classification of placenta previa: measuring progress in obstetrics. Am J Obstet Gynecol. Sep 2009;201(3):227–9. doi: 10.1016/j.ajog.2009.06.010. | |
Ornaghi S, Vaglio Tessitore I, Vergani P. Pregnancy and Delivery Outcomes in Women With Persistent Versus Resolved Low-Lying Placenta in the Late Third Trimester. J Ultrasound Med. Jan 2022;41(1):123–133. doi: 10.1002/jum.15687. | |
Bronsteen R, Valice R, Lee W, Blackwell S, Balasubramaniam M, Comstock C. Effect of a low-lying placenta on delivery outcome. Ultrasound Obstet Gynecol. Feb 2009;33(2):204–8. doi: 10.1002/uog.6304. | |
Vergani P, Ornaghi S, Pozzi I, Beretta P, Russo FM, Follesa I, Ghidini A. Placenta previa: distance to internal os and mode of delivery. Am J Obstet Gynecol. Sep 2009;201(3):266 e1–5. doi: 10.1016/j.ajog.2009.06.009. | |
Al Wadi K, Schneider C, Burym C, Reid G, Hunt J, Menticoglou S. Evaluating the safety of labour in women with a placental edge 11 to 20 mm from the internal cervical Os. J Obstet Gynaecol Can. Aug 2014;36(8):674–677. doi: 10.1016/S1701-2163(15)30508-9. | |
Farine D, Fox HE, Jakobson S, Timor-Tritsch IE. Is it really a placenta previa? Eur J Obstet Gynecol Reprod Biol. May 1989;31(2):103–8. doi: 10.1016/0028-2243(89)90170-6. | |
Farine D, Fox HE, Jakobson S, Timor-Tritsch IE. Vaginal ultrasound for diagnosis of placenta previa. Am J Obstet Gynecol. Sep 1988;159(3):566–9. doi: 10.1016/s0002-9378(88)80009-7. | |
Farine D, Peisner DB, Timor-Tritsch IE. Placenta previa–is the traditional diagnostic approach satisfactory? J Clin Ultrasound. May 1990;18(4):328–30. doi: 10.1002/jcu.1870180418. | |
Leerentveld RA, Gilberts EC, Arnold MJ, Wladimiroff JW. Accuracy and safety of transvaginal sonographic placental localization. Obstet Gynecol. Nov 1990;76(5 Pt 1):759–62. doi: 10.1097/00006250-199011000-00006. | |
Timor-Tritsch IE, Yunis RA. Confirming the safety of transvaginal sonography in patients suspected of placenta previa. Obstet Gynecol. May 1993;81(5 ( Pt 1)):742–4. | |
Oyelese Y. Placenta previa and vasa previa: time to leave the Dark Ages. Ultrasound Obstet Gynecol. Aug 2001;18(2):96–9. doi: 10.1046/j.1469-0705.2001.00511.x. | |
Clark EA, Silver RM. Long-term maternal morbidity associated with repeat cesarean delivery. Am J Obstet Gynecol. Dec 2011;205(6 Suppl):S2–10. doi: 10.1016/j.ajog.2011.09.028. | |
Clark SL, Koonings PP, Phelan JP. Placenta previa/accreta and prior cesarean section. Obstet Gynecol. Jul 1985;66(1):89–92. | |
To WW, Leung WC. Placenta previa and previous cesarean section. Int J Gynaecol Obstet. Oct 1995;51(1):25–31. doi: 10.1016/0020-7292(95)80004-v. | |
Coutinho CM, Giorgione V, Noel L, et al. Effectiveness of contingent screening for placenta accreta spectrum disorders based on persistent low-lying placenta and previous uterine surgery. Ultrasound Obstet Gynecol. Jan 2021;57(1):91–96. doi: 10.1002/uog.23100. | |
Bhide A. Routine screening for placenta accreta spectrum. Best Pract Res Clin Obstet Gynaecol. Aug 2023;90:102392. doi: 10.1016/j.bpobgyn.2023.102392. | |
Society for Maternal-Fetal Medicine . Electronic address pso, Miller R, Timor-Tritsch IE, Gyamfi-Bannerman C. Society for Maternal-Fetal Medicine (SMFM) Consult Series #49: Cesarean scar pregnancy. Am J Obstet Gynecol. May 2020;222(5):B2-B14. doi: 10.1016/j.ajog.2020.01.030. | |
Society of Gynecologic O, American College of O, Gynecologists, et al. Placenta Accreta Spectrum. Am J Obstet Gynecol. Dec 2018;219(6):B2-B16. doi: 10.1016/j.ajog.2018.09.042. | |
Timor-Tritsch I, Buca D, Di Mascio D, et al. Outcome of cesarean scar pregnancy according to gestational age at diagnosis: A systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. Mar 2021;258:53–59. doi: 10.1016/j.ejogrb.2020.11.036. | |
Timor-Tritsch IE, Monteagudo A, Cali G, D'Antonio F, Agten AK. Cesarean Scar Pregnancy: Patient Counseling and Management. Obstet Gynecol Clin North Am. Dec 2019;46(4):813–828. doi: 10.1016/j.ogc.2019.07.010. | |
Timor-Tritsch IE, Monteagudo A, Cali G, D'Antonio F, Kaelin Agten A. Cesarean Scar Pregnancy: Diagnosis and Pathogenesis. Obstet Gynecol Clin North Am. Dec 2019;46(4):797–811. doi: 10.1016/j.ogc.2019.07.009. | |
Timor-Tritsch IE, Monteagudo A, Cali G, et al. Cesarean scar pregnancy and early placenta accreta share common histology. Ultrasound Obstet Gynecol. Apr 2014;43(4):383–95. doi: 10.1002/uog.13282. | |
Timor-Tritsch IE, Monteagudo A, Cali G, et al. Cesarean scar pregnancy is a precursor of morbidly adherent placenta. Ultrasound Obstet Gynecol. Sep 2014;44(3):346–53. doi: 10.1002/uog.13426. | |
Shainker SA, Coleman B, Timor-Tritsch IE, et al. Special Report of the Society for Maternal-Fetal Medicine Placenta Accreta Spectrum Ultrasound Marker Task Force: Consensus on definition of markers and approach to the ultrasound examination in pregnancies at risk for placenta accreta spectrum. Am J Obstet Gynecol. Jan 2021;224(1):B2-B14. doi: 10.1016/j.ajog.2020.09.001. | |
Publications Committee SfM-FM, Belfort MA. Placenta accreta. Am J Obstet Gynecol. Nov 2010;203(5):430–9. doi: 10.1016/j.ajog.2010.09.013. | |
Shamshirsaz AA, Fox KA, Erfani H, et al. Multidisciplinary team learning in the management of the morbidly adherent placenta: outcome improvements over time. Am J Obstet Gynecol. Jun 2017;216(6):612 e1–612 e5. doi: 10.1016/j.ajog.2017.02.016. | |
Palacios-Jaraquemada JM, Fiorillo A, Hamer J, Martinez M, Bruno C. Placenta accreta spectrum: a hysterectomy can be prevented in almost 80% of cases using a resective-reconstructive technique. J Matern Fetal Neonatal Med. Jan 2022;35(2):275–282. doi: 10.1080/14767058.2020.1716715. | |
Hessami K, Kamepalli S, Lombaard HA, Shamshirsaz AA, Belfort MA, Munoz JL. Conservative management of placenta accreta spectrum is associated with improved surgical outcomes compared to cesarean hysterectomy: a systematic review and meta-analysis. Am J Obstet Gynecol. Jan 28 2025;doi: 10.1016/j.ajog.2025.01.030. | |
Jauniaux E, D'Antonio F, Bhide A, et al. Modified Delphi study of ultrasound signs associated with placenta accreta spectrum. Ultrasound Obstet Gynecol. Apr 2023;61(4):518–525. doi: 10.1002/uog.26155. | |
Comstock CH, Bronsteen RA. The antenatal diagnosis of placenta accreta. BJOG. Jan 2014;121(2):171–81; discussion 181–2. doi: 10.1111/1471-0528.12557. | |
Comstock CH, Love JJ, Jr., Bronsteen RA, Lee W, Vettraino IM, Huang RR, Lorenz RP. Sonographic detection of placenta accreta in the second and third trimesters of pregnancy. Am J Obstet Gynecol. Apr 2004;190(4):1135–40. doi: 10.1016/j.ajog.2003.11.024. | |
Finberg HJ, Williams JW. Placenta accreta: prospective sonographic diagnosis in patients with placenta previa and prior cesarean section. J Ultrasound Med. Jul 1992;11(7):333–43. doi: 10.7863/jum.1992.11.7.333. | |
Comstock CH. Antenatal diagnosis of placenta accreta: a review. Ultrasound Obstet Gynecol. Jul 2005;26(1):89–96. doi: 10.1002/uog.1926. | |
Azagidi AS, Ibitoye BO, Makinde ON, Idowu BM, Aderibigbe AS. Fetal Gestational Age Determination using Ultrasound Placental Thickness. J Med Ultrasound. Jan-Mar 2020;28(1):17–23. doi: 10.4103/JMU.JMU_127_18. | |
Strebeck R, Jensen B, Magann EF. Thick Placenta in Pregnancy: A Review. Obstet Gynecol Surv. Sep 2022;77(9):547–557. doi: 10.1097/OGX.0000000000001051. | |
Degani S. Sonographic findings in fetal viral infections: a systematic review. Obstet Gynecol Surv. May 2006;61(5):329–36. doi: 10.1097/01.ogx.0000216518.85796.88. | |
Khullar P, Hon JD, Sethi S, Kim J, Iqbal M, Chavez MR. Placental Infections. Clin Obstet Gynecol. Mar 1 2025;68(1):119–129. doi: 10.1097/GRF.0000000000000919. | |
Rawal S, Ray S, Sharma N. Correlation Between Ultrasonographic Placental Thickness and Adverse Fetal and Neonatal Outcomes. Cureus. Mar 2024;16(3):e56410. doi: 10.7759/cureus.56410. | |
Woo GW, Rocha FG, Gaspar-Oishi M, Bartholomew ML, Thompson KS. Placental mesenchymal dysplasia. Am J Obstet Gynecol. Dec 2011;205(6):e3–5. doi: 10.1016/j.ajog.2011.08.019. | |
Nayeri UA, West AB, Grossetta Nardini HK, Copel JA, Sfakianaki AK. Systematic review of sonographic findings of placental mesenchymal dysplasia and subsequent pregnancy outcome. Ultrasound Obstet Gynecol. Apr 2013;41(4):366–74. doi: 10.1002/uog.12359. | |
Lee AJ, Bethune M, Hiscock RJ. Placental thickness in the second trimester: a pilot study to determine the normal range. J Ultrasound Med. Feb 2012;31(2):213–8. doi: 10.7863/jum.2012.31.2.213. | |
Tang L, Xu L, Hu Y, Zhang L, Wei Q, Wu L. Placenta membranacea: an anormaly of the placenta: Three case reports. Medicine (Baltimore). Jun 2019;98(26):e16166. doi: 10.1097/MD.0000000000016166. | |
Pereira N, Yao R, Guilfoil DS, Richard SD, Plante LA. Placenta membranacea with placenta accreta: radiologic diagnosis and clinical implications. Prenat Diagn. Dec 2013;33(13):1293–6. doi: 10.1002/pd.4224. | |
Oyelese Y, Ananth CV. Placental abruption. Obstet Gynecol. Oct 2006;108(4):1005–16. doi: 10.1097/01.AOG.0000239439.04364.9a. | |
Schneider E, Kinzler WL. Placental Abruption: Pathophysiology, Diagnosis, and Management. Clin Obstet Gynecol. Nov 18 2024;doi: 10.1097/GRF.0000000000000903. | |
Brandt JS, Ananth CV. Placental abruption at near-term and term gestations: pathophysiology, epidemiology, diagnosis, and management. Am J Obstet Gynecol. May 2023;228(5S):S1313-S1329. doi: 10.1016/j.ajog.2022.06.059. | |
Ananth CV, Berkowitz GS, Savitz DA, Lapinski RH. Placental abruption and adverse perinatal outcomes. JAMA. Nov 3 1999;282(17):1646–51. doi: 10.1001/jama.282.17.1646. | |
Ananth CV, Wilcox AJ. Placental abruption and perinatal mortality in the United States. Am J Epidemiol. Feb 15 2001;153(4):332–7. doi: 10.1093/aje/153.4.332. | |
Downes KL, Grantz KL, Shenassa ED. Maternal, Labor, Delivery, and Perinatal Outcomes Associated with Placental Abruption: A Systematic Review. Am J Perinatol. Aug 2017;34(10):935–957. doi: 10.1055/s-0037-1599149. | |
Tikkanen M. Placental abruption: epidemiology, risk factors and consequences. Acta Obstet Gynecol Scand. Feb 2011;90(2):140–9. doi: 10.1111/j.1600-0412.2010.01030.x. | |
Nyberg DA, Cyr DR, Mack LA, Wilson DA, Shuman WP. Sonographic spectrum of placental abruption. AJR Am J Roentgenol. Jan 1987;148(1):161–4. doi: 10.2214/ajr.148.1.161. | |
Walker M, Whittle W, Keating S, Kingdom J. Sonographic diagnosis of chronic abruption. J Obstet Gynaecol Can. Nov 2010;32(11):1056–1058. doi: 10.1016/S1701-2163(16)34713-2. | |
Sherer DM, Kheyman M, Benayoun J, Dalloul M. Incidental sonographic finding of a concealed placental abruption leading to delivery at 37 weeks' gestation. J Clin Ultrasound. Jul 2021;49(6):630–631. doi: 10.1002/jcu.22971. | |
Nyberg DA, Mack LA, Benedetti TJ, Cyr DR, Schuman WP. Placental abruption and placental hemorrhage: correlation of sonographic findings with fetal outcome. Radiology. Aug 1987;164(2):357–61. doi: 10.1148/radiology.164.2.3299486. | |
Mor L, Erteschik N, Gandelsman E, et al. Obstetric and neonatal outcomes in pregnancies complicated by placental abruption with vs. without supporting sonographic findings- A retrospective cohort study. Placenta. Oct 2024;156:14–19. doi: 10.1016/j.placenta.2024.08.017. | |
Fadl SA, Linnau KF, Dighe MK. Placental abruption and hemorrhage-review of imaging appearance. Emerg Radiol. Feb 2019;26(1):87–97. doi: 10.1007/s10140-018-1638-3. | |
Yeo L AC, Vintzileos AM. Placental Abruption. In: P. vD, ed. The Global Library of Women’s Medicine. International Federation of Gynecology and Obstetrics; 2008. | |
Shinde GR, Vaswani BP, Patange RP, Laddad MM, Bhosale RB. Diagnostic Performance of Ultrasonography for Detection of Abruption and Its Clinical Correlation and Maternal and Foetal Outcome. J Clin Diagn Res. Aug 2016;10(8):QC04–7. doi: 10.7860/JCDR/2016/19247.8288. | |
Alanjari A, Wright E, Keating S, Ryan G, Kingdom J. Prenatal diagnosis, clinical outcomes, and associated pathology in pregnancies complicated by massive subchorionic thrombohematoma (Breus' mole). Prenat Diagn. Oct 2013;33(10):973–8. doi: 10.1002/pd.4176. | |
Glantz C, Purnell L. Clinical utility of sonography in the diagnosis and treatment of placental abruption. J Ultrasound Med. Aug 2002;21(8):837–40. doi: 10.7863/jum.2002.21.8.837. | |
Yiu-Chiu V, Chiu L. Sonographic features of placental complications in pregnancy. AJR Am J Roentgenol. May 1982;138(5):879–85. doi: 10.2214/ajr.138.5.879. | |
Iordache M, Dumitru A, Turcan N, Cirstoiu MM. Sonographic Signs of SARS-CoV-2 Placentitis. Association with Pregnancy Outcome. Maedica (Bucur). Jun 2024;19(2):317–321. doi: 10.26574/maedica.2024.19.2.317. | |
Kim Y, Kim YM, Kim DR, et al. The Multifaceted Clinical Characteristics of Congenital Cytomegalovirus Infection: From Pregnancy to Long-Term Outcomes. J Korean Med Sci. Aug 14 2023;38(32):e249. doi: 10.3346/jkms.2023.38.e249. | |
Kouba I, Bracero L, Millington K, Blitz MJ. Placental calcifications after coronavirus disease 2019 in first trimester of pregnancy: ultrasound and pathology findings. Med Ultrason. Jun 26 2023;25(2):233–235. doi: 10.11152/mu-3753. | |
Mlakar J, Korva M, Tul N, et al. Zika Virus Associated with Microcephaly. N Engl J Med. Mar 10 2016;374(10):951–8. doi: 10.1056/NEJMoa1600651. | |
Bailao LA, Osborne NG, Rizzi MC, Bonilla-Musoles F, Duarte G, Bailao TC. Ultrasound markers of fetal infection part 1: viral infections. Ultrasound Q. Dec 2005;21(4):295–308. doi: 10.1097/01.ruq.0000187025.61943.ff. | |
De Leon-Luis J, Oneson RH, Santolaya-Forgas J. Placental surface cyst with contents less echogenic than amniotic fluid on a second-trimester ultrasonographic evaluation. Ultrasound Obstet Gynecol. Jun 2004;23(6):627–8. doi: 10.1002/uog.1057. | |
Shipley CF, 3rd, Nelson GH. Prenatal diagnosis of a placental cyst: comparison of postnatal biochemical analyses of cyst fluid, amniotic fluid, cord serum, and maternal serum. Am J Obstet Gynecol. Jan 1993;168(1 Pt 1):211–3. doi: 10.1016/s0002-9378(12)90916-3. | |
Larciprete G, Arduini D, Cirese E. Choroid plate cyst: not such a rare finding. Ultrasound Obstet Gynecol. Jul 2005;26(1):99–100. doi: 10.1002/uog.1942. | |
Ferrara N, Menditto C, Di Marino MP, Ciccarelli A, Gerosolima G, Menditto V. Subchorionic placental cyst: histopathological and clinical aspects in two cases. Pathologica. Oct 1996;88(5):439–43. | |
Brown DL, DiSalvo DN, Frates MC, Davidson KM, Genest DR. Placental surface cysts detected on sonography: histologic and clinical correlation. J Ultrasound Med. Jun 2002;21(6):641–6; quiz 647–8. doi: 10.7863/jum.2002.21.6.641. | |
Hong SC, Yoo SW, Kim T, Yeom BW, Kim YT, Lee KW, Kim SH. Prenatal diagnosis of a large subchorionic placental cyst with intracystic hematomas. A case report. Fetal Diagn Ther. 2007;22(4):259–63. doi: 10.1159/000100786. | |
Soyama H, Miyamoto M, Sasa H, Yoshida M, Takano M, Furuya K. Multiple placental surface cysts with intracystic hemorrhaging: A case report and review of the literature. J Obstet Gynaecol Res. Aug 2017;43(8):1346–1349. doi: 10.1111/jog.13365. | |
Boulis TS, Rochelson BL, Williamson AK. Massive Subchorionic Placental Cyst and Poor Fetal Growth: A Case Report. J Reprod Med. Sep-Oct 2015;60(9–10):458–60. | |
Fan M, Skupski DW. Placental chorioangioma: literature review. J Perinat Med. May 2014;42(3):273–9. doi: 10.1515/jpm-2013-0170. | |
Wallenburg HC. Chorioangioma of the placenta. Thirteen new cases and a review of the literature from 1939 to 1970 with special reference to the clinical complications. Obstet Gynecol Surv. Jun 1971;26(6):411–25. doi: 10.1097/00006254-197106000-00001. | |
Zanardini C, Papageorghiou A, Bhide A, Thilaganathan B. Giant placental chorioangioma: natural history and pregnancy outcome. Ultrasound Obstet Gynecol. Mar 2010;35(3):332–6. doi: 10.1002/uog.7451. | |
Saeed B, Tulbah A, Bintalib M, et al. Chorioangioma: a single tertiary care center retrospective study. J Perinat Med. Jun 27 2023;51(5):664–674. doi: 10.1515/jpm-2021-0085. | |
Buca D, Iacovella C, Khalil A, et al. Perinatal outcome of pregnancies complicated by placental chorioangioma: systematic review and meta-analysis. Ultrasound Obstet Gynecol. Apr 2020;55(4):441–449. doi: 10.1002/uog.20304. | |
Arora S, Khera N, Kaul A. Three-dimensional color Doppler acquisition of a highly vascular giant placental chorioangioma. Am J Obstet Gynecol. Feb 2022;226(2):259–260. doi: 10.1016/j.ajog.2021.08.021. | |
Ropacka-Lesiak M, Gruca-Stryjak K, Breborowicz G. Nontrophoblastic placental tumors. Neuro Endocrinol Lett. 2012;33(4):375–9. | |
Ma H, Liu Z, Ruan J. Placental chorioangioma and pregnancy outcome: a ten-year retrospective study in a tertiary referral centre. BMC Pregnancy Childbirth. May 25 2023;23(1):381. doi: 10.1186/s12884-023-05719-x. | |
Garcia-Diaz L, Carreto P, Costa-Pereira S, Antinolo G. Prenatal management and perinatal outcome in giant placental chorioangioma complicated with hydrops fetalis, fetal anemia and maternal mirror syndrome. BMC Pregnancy Childbirth. Jul 28 2012;12:72. doi: 10.1186/1471-2393-12-72. | |
Wu PT, Huang KL, Tsai CC, Cheng HH, Lai YJ, Hsu TY. A singleton pregnancy with placental chorioangioma and hydrops fetalis complicated with mirror syndrome and ritodrine-induced side effects: a case report. BMC Pregnancy Childbirth. Mar 20 2024;24(1):213. doi: 10.1186/s12884-024-06391-5. | |
As AK, Hagen P, Webb JB, Wijesinghe D. Therapeutic amniodrainage in chorioangioma. J Obstet Gynaecol. Mar 1997;17(2):169–70. doi: 10.1080/01443619750113799. | |
Sepulveda W, Wong AE, Herrera L, Dezerega V, Devoto JC. Endoscopic laser coagulation of feeding vessels in large placental chorioangiomas: report of three cases and review of invasive treatment options. Prenat Diagn. Mar 2009;29(3):201–6. doi: 10.1002/pd.2197. | |
Quarello E, Bernard JP, Leroy B, Ville Y. Prenatal laser treatment of a placental chorioangioma. Ultrasound Obstet Gynecol. Mar 2005;25(3):299–301. doi: 10.1002/uog.1848. | |
Hosseinzadeh P, Shamshirsaz AA, Javadian P, et al. Prenatal Therapy of Large Placental Chorioangiomas: Case Report and Review of the Literature. AJP Rep. Oct 2015;5(2):e196–202. doi: 10.1055/s-0035-1558829. | |
Agarwal N, Papanna R, Bergh EP, et al. Management of large placental chorioangioma: two-port laser approach for fetal intervention. Ultrasound Obstet Gynecol. Dec 2023;62(6):882–890. doi: 10.1002/uog.26307. | |
Jones K, Tierney K, Grubbs BH, Pruetz JD, Detterich J, Chmait RH. Fetoscopic laser photocoagulation of feeding vessels to a large placental chorioangioma following fetal deterioration after amnioreduction. Fetal Diagn Ther. 2012;31(3):191–5. doi: 10.1159/000331944. | |
Emery SP, Orons PD, Bonadio JF. Successful Management of Giant Placental Chorangioma by Microcoil Embolization. AJP Rep. Oct 2018;8(4):e230-e233. doi: 10.1055/s-0038-1669944. | |
Ahmed N, Kale V, Thakkar H, Hanchate V, Dhargalkar P. Sonographic diagnosis of placental teratoma. J Clin Ultrasound. Feb 2004;32(2):98–101. doi: 10.1002/jcu.10231. | |
Stuijt DG, Bos M, Nikkels PGJ, Wolterbeek R, van der Meeren LE. Significant association between circumvallate placenta, placental abruption and acute chorioamnionitis in preterm birth: A 23-year retrospective cohort study. Placenta. Feb 2024;146:25–29. doi: 10.1016/j.placenta.2023.12.018. | |
Taniguchi H, Aoki S, Sakamaki K, Kurasawa K, Okuda M, Takahashi T, Hirahara F. Circumvallate placenta: associated clinical manifestations and complications-a retrospective study. Obstet Gynecol Int. 2014;2014:986230. doi: 10.1155/2014/986230. | |
Celik OY, Obut M, Keles A, Calik MG, Dagdeviren G, Yucel A, Sahin D. Outcomes of pregnancies diagnosed with circumvallate placenta, and use of uterine artery pulsatility index and maternal serum alpha-fetoprotein for prediction of adverse outcomes. Eur J Obstet Gynecol Reprod Biol. Jul 2021;262:57–61. doi: 10.1016/j.ejogrb.2021.05.008. | |
Harris RD, Wells WA, Black WC, Chertoff JD, Poplack SP, Sargent SK, Crow HC. Accuracy of prenatal sonography for detecting circumvallate placenta. AJR Am J Roentgenol. Jun 1997;168(6):1603–8. doi: 10.2214/ajr.168.6.9168736. | |
McCarthy J, Thurmond AS, Jones MK, Sistrom C, Scanlan RM, Jacobson SL, Lowensohn R. Circumvallate placenta: sonographic diagnosis. J Ultrasound Med. Jan 1995;14(1):21–6. doi: 10.7863/jum.1995.14.1.21. | |
Herrera CL, Chu TM, Stanteen SM, et al. Prenatal Ultrasound Findings of Circumvallate Placenta and Pregnancy Outcomes. Am J Perinatol. May 2024;41(S 01):e2069-e2072. doi: 10.1055/s-0043-1770337. | |
de Castro Rezende G, Araujo Junior E. Prenatal diagnosis of placenta and umbilical cord pathologies by three-dimensional ultrasound: pictorial essay. Med Ultrason. Dec 2015;17(4):545–9. doi: 10.11152/mu.2013.2066.174.pdg. | |
Arlicot C, Herve P, Simon E, Perrotin F. Three-dimensional surface rendering of the chorionic placental plate: the 'tire' sign for the diagnosis of a circumvallate placenta. J Ultrasound Med. Feb 2012;31(2):340–1. doi: 10.7863/jum.2012.31.2.340. | |
Moscoso G, Jauniaux E, Hustin J. Placental vascular anomaly with diffuse mesenchymal stem villous hyperplasia. A new clinico-pathological entity? Pathol Res Pract. Mar 1991;187(2–3):324–8. doi: 10.1016/s0344-0338(11)80791-0. | |
Guenot C, Kingdom J, De Rham M, et al. Placental mesenchymal dysplasia: An underdiagnosed placental pathology with various clinical outcomes. Eur J Obstet Gynecol Reprod Biol. Mar 2019;234:155–164. doi: 10.1016/j.ejogrb.2019.01.014. | |
Lolli NB, McWhirter AM, Lesser KB, Rush DS, Aboul-Nasr AG, Coppola LM. Zebras in a snowstorm: ultrasound guidance for differentiating placental mesenchymal dysplasia from hydatidiform mole. Am J Obstet Gynecol. Jul 25 2024;doi: 10.1016/j.ajog.2024.07.025. | |
Tang P, Jin X, Li J, Zhang L, Li Y, Xu S. Misdiagnosis of placental mesenchymal dysplasia as pregnancy with hydatidiform mole: A case report and literature review. Medicine (Baltimore). Apr 14 2023;102(15):e33438. doi: 10.1097/MD.0000000000033438. | |
Mehedintu C, Frincu F, Ionescu OM, et al. A Challenging Diagnosis: Placental Mesenchymal Dysplasia-Literature Review and Case Report. Diagnostics (Basel). Jan 24 2022;12(2) doi: 10.3390/diagnostics12020293. | |
Ang DC, Rodriguez Urrego PA, Prasad V. Placental mesenchymal dysplasia: a potential misdiagnosed entity. Arch Gynecol Obstet. Jun 2009;279(6):937–9. doi: 10.1007/s00404-008-0812-3. | |
Pham T, Steele J, Stayboldt C, Chan L, Benirschke K. Placental mesenchymal dysplasia is associated with high rates of intrauterine growth restriction and fetal demise: A report of 11 new cases and a review of the literature. Am J Clin Pathol. Jul 2006;126(1):67–78. doi: 10.1309/RV45-HRD5-3YQ2-YFTP. | |
Soejima H, Hara S, Ohba T, Higashimoto K. Placental Mesenchymal Dysplasia and Beckwith-Wiedemann Syndrome. Cancers (Basel). Nov 12 2022;14(22) doi: 10.3390/cancers14225563. | |
de Vasconcelos Gaspar A, Branco M, Galhano E, Ramos F. Ultrasound and molecular prenatal diagnosis of Beckwith-Wiedemann syndrome: Two case reports. Radiol Case Rep. Dec 2022;17(12):4914–4919. doi: 10.1016/j.radcr.2022.09.066. | |
Jatavan P, Tongsong T, Traisrisilp K. Fetal Beckwith-Wiedemann syndrome associated with abnormal quad test, placental mesenchymal dysplasia and HELLP syndrome. BMJ Case Rep. Jun 24 2021;14(6) doi: 10.1136/bcr-2021-243415. | |
Colpaert RM, Ramseyer AM, Luu T, Quick CM, Frye LT, Magann EF. Diagnosis and Management of Placental Mesenchymal Disease. A Review of the Literature. Obstet Gynecol Surv. Oct 2019;74(10):611–622. doi: 10.1097/OGX.0000000000000716. | |
Berkowitz RS, Goldstein DP. Clinical practice. Molar pregnancy. N Engl J Med. Apr 16 2009;360(16):1639–45. doi: 10.1056/NEJMcp0900696. | |
Seckl MJ, Sebire NJ, Berkowitz RS. Gestational trophoblastic disease. Lancet. Aug 28 2010;376(9742):717–29. doi: 10.1016/S0140-6736(10)60280-2. | |
Lurain JR. Gestational trophoblastic disease I: epidemiology, pathology, clinical presentation and diagnosis of gestational trophoblastic disease, and management of hydatidiform mole. Am J Obstet Gynecol. Dec 2010;203(6):531–9. doi: 10.1016/j.ajog.2010.06.073. | |
Kirk E, Papageorghiou AT, Condous G, Bottomley C, Bourne T. The accuracy of first trimester ultrasound in the diagnosis of hydatidiform mole. Ultrasound Obstet Gynecol. Jan 2007;29(1):70–75. doi: 10.1002/uog.3875. | |
Jauniaux E, Nicolaides KH. Early ultrasound diagnosis and follow-up of molar pregnancies. Ultrasound Obstet Gynecol. Jan 1997;9(1):17–21. doi: 10.1046/j.1469-0705.1997.09010017.x. | |
Frates MC, Feinberg BB. Early prenatal sonographic diagnosis of twin triploid gestation presenting with fetal hydrops and theca-lutein ovarian cysts. J Clin Ultrasound. Mar 2000;28(3):137–41. doi: 10.1002/(sici)1097-0096(200003/04)28:3<137::aid-jcu6>3.0.co;2-4. | |
Eagles N, Sebire NJ, Short D, Savage PM, Seckl MJ, Fisher RA. Risk of recurrent molar pregnancies following complete and partial hydatidiform moles. Hum Reprod. Sep 2015;30(9):2055–63. doi: 10.1093/humrep/dev169. | |
Pinette MG, Tropepe M. Umbilical Cord Abnormalities. Clin Obstet Gynecol. Dec 19 2024;doi: 10.1097/GRF.0000000000000915. | |
Wax JR, Pinette MG. Imaging the Placental Cord Insertion: Just Do It. J Ultrasound Med. Apr 2020;39(4):811–815. doi: 10.1002/jum.15143. | |
Ranzini AC, Oyelese Y. How to screen for vasa previa. Ultrasound Obstet Gynecol. May 2021;57(5):720–725. doi: 10.1002/uog.23520. | |
Predanic M, Perni SC, Chasen ST, Baergen RN, Chervenak FA. Assessment of umbilical cord coiling during the routine fetal sonographic anatomic survey in the second trimester. J Ultrasound Med. Feb 2005;24(2):185–91; quiz 192–3. doi: 10.7863/jum.2005.24.2.185. | |
Qin Y, Lau TK, Rogers MS. Second-trimester ultrasonographic assessment of the umbilical coiling index. Ultrasound Obstet Gynecol. Nov 2002;20(5):458–63. doi: 10.1046/j.1469-0705.2002.00846.x. | |
Nakamura M, Oba T, Salafia CM, et al. Ultrasonographic study of umbilical cord twist direction during second trimester. J Matern Fetal Neonatal Med. Nov 2022;35(22):4233–4239. doi: 10.1080/14767058.2020.1849098. | |
Sherer DM, Al-Haddad S, Cheng R, Dalloul M. Current Perspectives of Prenatal Sonography of Umbilical Cord Morphology. Int J Womens Health. 2021;13:939–971. doi: 10.2147/IJWH.S278747. | |
Tonni G, Lituania M, Grisolia G, et al. Placental and Umbilical Cord Anomalies Detected by Ultrasound as Clinical Risk Factors of Adverse Perinatal Outcomes. Case Series Review of Selected Conditions. Part 3: Vascular Anomalies of the Umbilical Cord and Fetoplacental Vascular Malperfusion. J Clin Ultrasound. Mar 4 2025;doi: 10.1002/jcu.23904. | |
Tonni G, Lituania M, Grisolia G, et al. Placental and Umbilical Cord Anomalies Detected by Ultrasound as Clinical Risk Factors of Adverse Perinatal Outcome: Case Series Review of Selected Conditions. Part 2: Umbilical Cord Abnormalities. J Clin Ultrasound. Mar 4 2025;doi: 10.1002/jcu.23909. | |
Sepulveda WH. Antenatal sonographic detection of single umbilical artery. J Perinat Med. 1991;19(5):391–5. doi: 10.1515/jpme.1991.19.5.391. | |
Persutte WH, Hobbins J. Single umbilical artery: a clinical enigma in modern prenatal diagnosis. Ultrasound Obstet Gynecol. Sep 1995;6(3):216–29. doi: 10.1046/j.1469-0705.1995.06030216.x. | |
Siargkas A, Giouleka S, Tsakiridis I, Mamopoulos A, Kalogiannidis I, Athanasiadis A, Dagklis T. Prenatal Diagnosis of Isolated Single Umbilical Artery: Incidence, Risk Factors and Impact on Pregnancy Outcomes. Medicina (Kaunas). Jun 3 2023;59(6) doi: 10.3390/medicina59061080. | |
Sepulveda W. Umbilical artery diameter or umbilical vein/artery ratio: prenatal detection of single umbilical artery? J Ultrasound Med. Apr 1995;14(4):262, 288–9. doi: 10.7863/jum.1995.14.4.262. | |
Voskamp BJ, Fleurke-Rozema H, Oude-Rengerink K, Snijders RJ, Bilardo CM, Mol BW, Pajkrt E. Relationship of isolated single umbilical artery to fetal growth, aneuploidy and perinatal mortality: systematic review and meta-analysis. Ultrasound Obstet Gynecol. Dec 2013;42(6):622–8. doi: 10.1002/uog.12541. | |
Kaymak D, Dobral A, Oruc SBB, Daglar HK. The significance of assessing the umbilical cord on the placental side: A case report on fused umbilical arteries. J Clin Ultrasound. Nov-Dec 2024;52(9):1485–1489. doi: 10.1002/jcu.23805. | |
Rosenak D, Meizner I. Prenatal sonographic detection of single and double umbilical artery in the same fetus. J Ultrasound Med. Dec 1994;13(12):995–6. doi: 10.7863/jum.1994.13.12.995. | |
Society for Maternal-Fetal Medicine . Electronic address pso, Prabhu M, Kuller JA, Biggio JR. Society for Maternal-Fetal Medicine Consult Series #57: Evaluation and management of isolated soft ultrasound markers for aneuploidy in the second trimester: (Replaces Consults #10, Single umbilical artery, October 2010; #16, Isolated echogenic bowel diagnosed on second-trimester ultrasound, August 2011; #17, Evaluation and management of isolated renal pelviectasis on second-trimester ultrasound, December 2011; #25, Isolated fetal choroid plexus cysts, April 2013; #27, Isolated echogenic intracardiac focus, August 2013). Am J Obstet Gynecol. Oct 2021;225(4):B2-B15. doi: 10.1016/j.ajog.2021.06.079. | |
Yu J, Wu Q, Kong F, Ning Y. Diagnosis of single umbilical artery and risk of foetal congenital malformations by prenatal ultrasound: a retrospective study. BMC Pregnancy Childbirth. Mar 12 2024;24(1):193. doi: 10.1186/s12884-024-06375-5. | |
Tai C, Conner T, Pevyandi S, Moon-Grady AJ. Prevalence of Congenital Heart Disease in an Isolated Single Umbilical Artery Is Low at a Tertiary Referral Center. J Ultrasound Med. Aug 2021;40(8):1729–1730. doi: 10.1002/jum.15542. | |
Tonismae T, Kline JH, Choe JJ, Schubert F, Tuuli M, Shanks A. Anatomical Survey Versus Fetal Echocardiograms for Diagnosis of Cardiac Defects with a Single Umbilical Artery Cases: A Retrospective Cohort Study and Diagnostic Meta-analysis. J Ultrasound Med. May 2021;40(5):1031–1036. doi: 10.1002/jum.15483. | |
Gurram P, Figueroa R, Sipusic E, Kuhnly N, Clark S, Janicki MB. Isolated Single Umbilical Artery and Fetal Echocardiography: A 25-Year Experience at a Tertiary Care City Hospital. J Ultrasound Med. Feb 2018;37(2):463–468. doi: 10.1002/jum.14353. | |
Ebbing C, Kessler J, Moster D, Rasmussen S. Isolated single umbilical artery and the risk of adverse perinatal outcome and third stage of labor complications: A population-based study. Acta Obstet Gynecol Scand. Mar 2020;99(3):374–380. doi: 10.1111/aogs.13747. | |
Gutvirtz G, Walfisch A, Beharier O, Sheiner E. Isolated single umbilical artery is an independent risk factor for perinatal mortality and adverse outcomes in term neonates. Arch Gynecol Obstet. Nov 2016;294(5):931–935. doi: 10.1007/s00404-016-4088-8. | |
de Laat MW, Franx A, van Alderen ED, Nikkels PG, Visser GH. The umbilical coiling index, a review of the literature. J Matern Fetal Neonatal Med. Feb 2005;17(2):93–100. doi: 10.1080/14767050400028899. | |
Mittal A, Nanda S, Sen J. Antenatal umbilical coiling index as a predictor of perinatal outcome. Arch Gynecol Obstet. Apr 2015;291(4):763–8. doi: 10.1007/s00404-014-3456-5. | |
Predanic M, Perni SC, Chasen ST, Baergen RN, Chervenak FA. Ultrasound evaluation of abnormal umbilical cord coiling in second trimester of gestation in association with adverse pregnancy outcome. Am J Obstet Gynecol. Aug 2005;193(2):387–94. doi: 10.1016/j.ajog.2004.12.092. | |
Predanic M, Perni SC, Chervenak FA. Antenatal umbilical coiling index and Doppler flow characteristics. Ultrasound Obstet Gynecol. Oct 2006;28(5):699–703. doi: 10.1002/uog.2745. | |
Ma'ayeh M, McClennen E, Chamchad D, Geary M, Brest N, Gerson A. Hypercoiling of the umbilical cord in uncomplicated singleton pregnancies. J Perinat Med. Aug 28 2018;46(6):593–598. doi: 10.1515/jpm-2017-0034. | |
Rayburn WF, Beynen A, Brinkman DL. Umbilical cord length and intrapartum complications. Obstet Gynecol. Apr 1981;57(4):450–2. | |
Ente G, Penzer PH. The umbilical cord: normal parameters. J R Soc Health. Aug 1991;111(4):138–40. doi: 10.1177/146642409111100406. | |
Taweevisit M, Thorner PS. Massive fetal thrombotic vasculopathy associated with excessively long umbilical cord and fetal demise: case report and literature review. Pediatr Dev Pathol. Mar-Apr 2010;13(2):112–5. doi: 10.2350/09-07-0680-CR.1. | |
Hanna M, Martini N, Deeb Y, Mahmoud W, Issa SY. A misdiagnosed case of a 150-cm umbilical cord coiled twice around the fetal neck with a true cord knot: A rare Syrian case report. SAGE Open Med Case Rep. 2023;11:2050313X231164858. doi: 10.1177/2050313X231164858. | |
Schmid A, Jacquemyn Y, Loor JD. Intrauterine growth restriction associated with excessively long umbilical cord. Clin Pract. Aug 2 2013;3(2):e23. doi: 10.4081/cp.2013.e23. | |
Raio L, Ghezzi F, Di Naro E, Franchi M, Maymon E, Mueller MD, Bruhwiler H. Prenatal diagnosis of a lean umbilical cord: a simple marker for the fetus at risk of being small for gestational age at birth. Ultrasound Obstet Gynecol. Mar 1999;13(3):176–80. doi: 10.1046/j.1469-0705.1999.13030176.x. | |
Silver RK, Dooley SL, Tamura RK, Depp R. Umbilical cord size and amniotic fluid volume in prolonged pregnancy. Am J Obstet Gynecol. Sep 1987;157(3):716–20. doi: 10.1016/s0002-9378(87)80036-4. | |
Li TG, Guan CL, Wang J, Peng MJ. Comparative study of umbilical cord cross-sectional area in foetuses with isolated single umbilical artery and normal umbilical artery. J Obstet Gynaecol. Jul 2022;42(5):935–940. doi: 10.1080/01443615.2021.1962818. | |
Contro E, Larcher L, Lenzi J, Valeriani M, Farina A, Jauniaux E. Changes in Artery Diameters and Fetal Growth in Cases of Isolated Single Umbilical Artery. Diagnostics (Basel). Feb 3 2023;13(3) doi: 10.3390/diagnostics13030571. | |
Cromi A, Ghezzi F, Di Naro E, Siesto G, Bergamini V, Raio L. Large cross-sectional area of the umbilical cord as a predictor of fetal macrosomia. Ultrasound Obstet Gynecol. Nov 2007;30(6):861–6. doi: 10.1002/uog.5183. | |
Predanic M, Perni SC, Chasen S, Chervenak FA. Fetal aneuploidy and umbilical cord thickness measured between 14 and 23 weeks' gestational age. J Ultrasound Med. Sep 2004;23(9):1177–83; quiz 1185. doi: 10.7863/jum.2004.23.9.1177. | |
di Pasquo E, Dall'Asta A, Volpe N, Corno E, Di Ilio C, Bettinelli ML, Ghi T. Ultrasound evaluation of the size of the umbilical cord vessels and Wharton's jelly and correlation with intrapartum CTG findings. Eur J Obstet Gynecol Reprod Biol. Feb 2025;305:42–47. doi: 10.1016/j.ejogrb.2024.11.048. | |
Nunes CM, Biancolin SE, Miyadahira MY, Peres SV, de Carvalho MHB, Francisco RPV, Brizot ML. Sonographic evaluation of umbilical cord thickness in monochorionic diamniotic twin pregnancies. Prenat Diagn. May 2022;42(5):636–642. doi: 10.1002/pd.6141. | |
Wax IR, Cartin A, Craig WY, Pinette MG, Wax JR. Second-Trimester Ultrasound-Measured Umbilical Cord Insertion-to-Placental Edge Distance: Determining an Outcome-Based Threshold for Identifying Marginal Cord Insertions. J Ultrasound Med. Feb 2020;39(2):351–358. doi: 10.1002/jum.15113. | |
Ebbing C, Kiserud T, Johnsen SL, Albrechtsen S, Rasmussen S. Prevalence, risk factors and outcomes of velamentous and marginal cord insertions: a population-based study of 634,741 pregnancies. PLoS One. 2013;8(7):e70380. doi: 10.1371/journal.pone.0070380. | |
Allaf MB, Andrikopoulou M, Crnosija N, Muscat J, Chavez MR, Vintzileos AM. Second trimester marginal cord insertion is associated with adverse perinatal outcomes. J Matern Fetal Neonatal Med. Sep 2019;32(18):2979–2984. doi: 10.1080/14767058.2018.1453798. | |
Siargkas A, Tsakiridis I, Pachi C, Mamopoulos A, Athanasiadis A, Dagklis T. Impact of marginal cord insertion on perinatal outcomes: a systematic review and meta-analysis. Am J Obstet Gynecol MFM. Apr 2023;5(4):100876. doi: 10.1016/j.ajogmf.2023.100876. | |
Asoglu MR, Crimmins S, Kopelman JN, Turan OM, Goetzinger KR. Marginal placental cord insertion: the need for follow up? J Matern Fetal Neonatal Med. May 2022;35(9):1629–1635. doi: 10.1080/14767058.2020.1763297. | |
Lutz AB, Young-Lin N, Leon-Martinez D, Bianco IC, Seckel E, Mrazek-Pugh B, Bianco K. Measurement of Marginal Placental Cord Insertion by Prenatal Ultrasound Was Found Not to Be Predictive of Adverse Perinatal Outcomes. J Ultrasound Med. Oct 2021;40(10):2079–2086. doi: 10.1002/jum.15586. | |
O'Quinn C, Cooper S, Tang S, Wood S. Antenatal Diagnosis of Marginal and Velamentous Placental Cord Insertion and Pregnancy Outcomes. Obstet Gynecol. Apr 2020;135(4):953–959. doi: 10.1097/AOG.0000000000003753. | |
Liu CC, Pretorius DH, Scioscia AL, Hull AD. Sonographic prenatal diagnosis of marginal placental cord insertion: clinical importance. J Ultrasound Med. Jun 2002;21(6):627–32. doi: 10.7863/jum.2002.21.6.627. | |
Bigelow CA, Robles BN, Pan S, et al. Placental cord insertion distance from the placental margin and its association with adverse perinatal outcomes. Eur J Obstet Gynecol Reprod Biol. Dec 2020;255:51–55. doi: 10.1016/j.ejogrb.2020.10.006. | |
Nkwabong E, Njikam F, Kalla G. Outcome of pregnancies with marginal umbilical cord insertion. J Matern Fetal Neonatal Med. Apr 2021;34(7):1133–1137. doi: 10.1080/14767058.2019.1628206. | |
Siargkas A, Tsakiridis I, Gatsis A, De Paco Matallana C, Gil MM, Chaveeva P, Dagklis T. Risk Factors of Velamentous Cord Insertion in Singleton Pregnancies-A Systematic Review and Meta-Analysis. J Clin Med. Sep 19 2024;13(18) doi: 10.3390/jcm13185551. | |
Somiya A, Tsuda H, Tsugeno E, et al. Prevalence, Risk Factors, and Perinatal Outcomes of Velamentous Umbilical Cord Insertion in Twin Pregnancies: A Single-Center Retrospective Study. J Clin Med. Feb 28 2024;13(5) doi: 10.3390/jcm13051396. | |
Matsuzaki S, Ueda Y, Matsuzaki S, et al. Assisted Reproductive Technique and Abnormal Cord Insertion: A Systematic Review and Meta-Analysis. Biomedicines. Jul 17 2022;10(7) doi: 10.3390/biomedicines10071722. | |
Furuya S, Kubonoya K, Yamaguchi T. Incidence and risk factors for velamentous umbilical cord insertion in singleton pregnancies after assisted reproductive technology. J Obstet Gynaecol Res. May 2021;47(5):1772–1779. doi: 10.1111/jog.14727. | |
Yerlikaya G, Pils S, Springer S, Chalubinski K, Ott J. Velamentous cord insertion as a risk factor for obstetric outcome: a retrospective case-control study. Arch Gynecol Obstet. May 2016;293(5):975–81. doi: 10.1007/s00404-015-3912-x. | |
Siargkas A, Tsakiridis I, Pachi C, Mamopoulos A, Athanasiadis A, Dagklis T. Impact of velamentous cord insertion on perinatal outcomes: a systematic review and meta-analysis. Am J Obstet Gynecol MFM. Feb 2023;5(2):100812. doi: 10.1016/j.ajogmf.2022.100812. | |
Ebbing C, Kiserud T, Johnsen SL, Albrechtsen S, Rasmussen S. Third stage of labor risks in velamentous and marginal cord insertion: a population-based study. Acta Obstet Gynecol Scand. Aug 2015;94(8):878–83. doi: 10.1111/aogs.12666. | |
Zahedi-Spung LD, Stout MJ, Carter EB, Dicke JM, Tuuli MG, Raghuraman N. Obstetric Outcomes in Singleton Pregnancies with Abnormal Placental Cord Insertions. Am J Perinatol. Jan 2023;40(1):89–94. doi: 10.1055/s-0041-1729163. | |
de Los Reyes S, Henderson J, Eke AC. A systematic review and meta-analysis of velamentous cord insertion among singleton pregnancies and the risk of preterm delivery. Int J Gynaecol Obstet. Jul 2018;142(1):9–14. doi: 10.1002/ijgo.12489. | |
Couck I, Mourad Tawfic N, Deprest J, De Catte L, Devlieger R, Lewi L. Does site of cord insertion increase risk of adverse outcome, twin-to-twin transfusion syndrome and discordant growth in monochorionic twin pregnancy? Ultrasound Obstet Gynecol. Sep 2018;52(3):385–389. doi: 10.1002/uog.18926. | |
Kalafat E, Thilaganathan B, Papageorghiou A, Bhide A, Khalil A. Significance of placental cord insertion site in twin pregnancy. Ultrasound Obstet Gynecol. Sep 2018;52(3):378–384. doi: 10.1002/uog.18914. | |
Costa-Castro T, Zhao DP, Lipa M, et al. Velamentous cord insertion in dichorionic and monochorionic twin pregnancies – Does it make a difference? Placenta. Jun 2016;42:87–92. doi: 10.1016/j.placenta.2016.04.007. | |
Nomiyama M, Toyota Y, Kawano H. Antenatal diagnosis of velamentous umbilical cord insertion and vasa previa with color Doppler imaging. Ultrasound Obstet Gynecol. Dec 1998;12(6):426–9. doi: 10.1046/j.1469-0705.1998.12060426.x. | |
Sepulveda W, Rojas I, Robert JA, Schnapp C, Alcalde JL. Prenatal detection of velamentous insertion of the umbilical cord: a prospective color Doppler ultrasound study. Ultrasound Obstet Gynecol. Jun 2003;21(6):564–9. doi: 10.1002/uog.132. | |
Sepulveda W. Velamentous insertion of the umbilical cord: a first-trimester sonographic screening study. J Ultrasound Med. Aug 2006;25(8):963–8; quiz 970. doi: 10.7863/jum.2006.25.8.963. | |
Ward S, Sun Z, Maresse S. Current practice of placental cord insertion documentation in Australia – A sonographer survey. Australas J Ultrasound Med. Aug 2023;26(3):157–168. doi: 10.1002/ajum.12360. | |
Oyelese KO, Turner M, Lees C, Campbell S. Vasa previa: an avoidable obstetric tragedy. Obstet Gynecol Surv. Feb 1999;54(2):138–45. doi: 10.1097/00006254-199902000-00024. | |
Jain V, Gagnon R. Guideline No. 439: Diagnosis and Management of Vasa Previa. J Obstet Gynaecol Can. Jul 2023;45(7):506–518. doi: 10.1016/j.jogc.2023.05.009. | |
Oyelese Y. Vasa previa: time to make a difference. Am J Obstet Gynecol. Dec 2019;221(6):539–541. doi: 10.1016/j.ajog.2019.08.034. | |
Oyelese Y, Lees CC, Jauniaux E. The case for screening for vasa previa: time to implement a life-saving strategy. Ultrasound Obstet Gynecol. Jan 2023;61(1):7–11. doi: 10.1002/uog.26085. | |
Society of Maternal-Fetal Publications C, Sinkey RG, Odibo AO, Dashe JS. #37: Diagnosis and management of vasa previa. Am J Obstet Gynecol. Nov 2015;213(5):615–9. doi: 10.1016/j.ajog.2015.08.031. | |
Francois K, Mayer S, Harris C, Perlow JH. Association of vasa previa at delivery with a history of second-trimester placenta previa. J Reprod Med. Oct 2003;48(10):771–4. | |
Jauniaux E, Melcer Y, Maymon R. Prenatal diagnosis and management of vasa previa in twin pregnancies: a case series and systematic review. Am J Obstet Gynecol. Jun 2017;216(6):568–575. doi: 10.1016/j.ajog.2017.01.029. | |
Jauniaux E, Savvidou MD. Vasa praevia: more than 100 years in preventing unnecessary fetal deaths. BJOG. Jul 2016;123(8):1287. doi: 10.1111/1471-0528.13869. | |
Jauniaux E, Ben-Ami I, Maymon R. Do assisted-reproduction twin pregnancies require additional antenatal care? Reprod Biomed Online. Feb 2013;26(2):107–19. doi: 10.1016/j.rbmo.2012.11.008. | |
Silver RM. Vasa praevia: improved diagnosis through recognition of risk factors. BJOG. Jul 2016;123(8):1288. doi: 10.1111/1471-0528.13870. | |
Catanzarite V, Cousins L, Daneshmand S, et al. Prenatally Diagnosed Vasa Previa: A Single-Institution Series of 96 Cases. Obstet Gynecol. Nov 2016;128(5):1153–1161. doi: 10.1097/AOG.0000000000001680. | |
Catanzarite V, Maida C, Thomas W, Mendoza A, Stanco L, Piacquadio KM. Prenatal sonographic diagnosis of vasa previa: ultrasound findings and obstetric outcome in ten cases. Ultrasound Obstet Gynecol. Aug 2001;18(2):109–15. doi: 10.1046/j.1469-0705.2001.00448.x. | |
Oyelese Y. Evolution from placenta previa to Type-3 vasa previa. Ultrasound Obstet Gynecol. Jan 2024;63(1):128–130. doi: 10.1002/uog.27505. | |
Srinivasan D, Daly-Jones E, Oyelese Y, Papanikolaou K, Lees CC. Challenging common misconceptions in vasa previa screening and diagnosis. Ultrasound Obstet Gynecol. Oct 2024;64(4):435–442. doi: 10.1002/uog.27567. | |
Oyelese Y. A 2-cm Distance Should Not Be Used to Define Vasa Previa. J Ultrasound Med. Apr 2024;43(4):811–814. doi: 10.1002/jum.16420. | |
Schenone CV, Aghajani F, Javinani A, et al. Vasa Previa: Prenatal Diagnosis and the Rationale Behind Using a 5 cm Distance from Internal Os. J Clin Med. Feb 5 2025;14(3) doi: 10.3390/jcm14031009. | |
Oyelese Y, Javinani A, Gudanowski B, et al. Vasa previa in singleton pregnancies: diagnosis and clinical management based on an international expert consensus. Am J Obstet Gynecol. Dec 2024;231(6):638 e1–638 e24. doi: 10.1016/j.ajog.2024.03.013. | |
Ruiter L, Kok N, Limpens J, Derks JB, de Graaf IM, Mol BW, Pajkrt E. Systematic review of accuracy of ultrasound in the diagnosis of vasa previa. Ultrasound Obstet Gynecol. May 2015;45(5):516–22. doi: 10.1002/uog.14752. | |
Erfani H, Haeri S, Shainker SA, et al. Vasa previa: a multicenter retrospective cohort study. Am J Obstet Gynecol. Dec 2019;221(6):644 e1–644 e5. doi: 10.1016/j.ajog.2019.06.006. | |
Klahr R, Fox NS, Zafman K, Hill MB, Connolly CT, Rebarber A. Frequency of spontaneous resolution of vasa previa with advancing gestational age. Am J Obstet Gynecol. Dec 2019;221(6):646 e1–646 e7. doi: 10.1016/j.ajog.2019.06.040. | |
Kagan KO, Hoopmann M, Sonek J. Vasa previa: easy to miss. Ultrasound Obstet Gynecol. Feb 2018;51(2):283–284. doi: 10.1002/uog.17532. | |
Oyelese Y, Reforma L, Sewell McGough R, O'Brien B. Manual elevation of fetal head as potential cause of missed vasa previa. Ultrasound Obstet Gynecol. Sep 2022;60(3):429–431. doi: 10.1002/uog.24982. | |
Lee W, Kirk JS, Comstock CH, Romero R. Vasa previa: prenatal detection by three-dimensional ultrasonography. Ultrasound Obstet Gynecol. Sep 2000;16(4):384–7. doi: 10.1046/j.1469-0705.2000.00188.x. | |
Oyelese Y, Chavez MR, Yeo L, Giannina G, Kontopoulos EV, Smulian JC, Scorza WE. Three-dimensional sonographic diagnosis of vasa previa. Ultrasound Obstet Gynecol. Aug 2004;24(2):211–5. doi: 10.1002/uog.1097. | |
Mitchell SJ, Ngo G, Maurel KA, et al. Timing of birth and adverse pregnancy outcomes in cases of prenatally diagnosed vasa previa: a systematic review and meta-analysis. Am J Obstet Gynecol. Aug 2022;227(2):173–181 e24. doi: 10.1016/j.ajog.2022.03.006. | |
McMahon C, Laiu S, Oyelese Y, Rolnik DL. Vasa previa guidelines and their supporting evidence. J Perinat Med. Jan 7 2025;doi: 10.1515/jpm-2024-0473. | |
Kosian P, Henrich W, Entezami M, Weichert A. Furcate insertion of the umbilical cord: pathological and clinical characteristics in 132 cases. J Perinat Med. Oct 25 2020;48(8):819–824. doi: 10.1515/jpm-2019-0459. | |
Xu H, Lu JP, Xu QL. Intrauterine fetal death due to rupture of umbilical vessels: a rare case of furcate cord insertion. BMC Pregnancy Childbirth. Jul 5 2024;24(1):464. doi: 10.1186/s12884-024-06660-3. | |
Peesay M. Nuchal cord and its implications. Matern Health Neonatol Perinatol. 2017;3:28. doi: 10.1186/s40748-017-0068-7. | |
Narang Y, Vaid NB, Jain S, Suneja A, Guleria K, Faridi MM, Gupta B. Is nuchal cord justified as a cause of obstetrician anxiety? Arch Gynecol Obstet. Apr 2014;289(4):795–801. doi: 10.1007/s00404-013-3072-9. | |
Sherer DM, Ward K, Bennett M, Dalloul M. Current Perspectives of Prenatal Sonographic Diagnosis and Clinical Management Challenges of Nuchal Cord(s). Int J Womens Health. 2020;12:613–631. doi: 10.2147/IJWH.S211124. | |
Herkelman C, Nicoara D, Thiel L, Florido J. Caesarean delivery of a breech singleton with eight loops of nuchal cord. BMJ Case Rep. Feb 2 2022;15(2) doi: 10.1136/bcr-2021-246023. | |
Sherer DM, Manning FA. Prenatal ultrasonographic diagnosis of nuchal cord(s): disregard, inform, monitor or intervene? Ultrasound Obstet Gynecol. Jul 1999;14(1):1–8. doi: 10.1046/j.1469-0705.1999.14010001.x. | |
Hayes DJL, Warland J, Parast MM, et al. Umbilical cord characteristics and their association with adverse pregnancy outcomes: A systematic review and meta-analysis. PLoS One. 2020;15(9):e0239630. doi: 10.1371/journal.pone.0239630. | |
Rogers MS, Ip YW, Qin Y, Rogers SM, Sahota D. Relationship between umbilical cord morphology and nuchal cord entanglement. Acta Obstet Gynecol Scand. Jan 2003;82(1):32–7. doi: 10.1034/j.1600-0412.2003.820106.x. | |
Ranzini AC, Walters CA, Vintzileos AM. Ultrasound diagnosis of nuchal cord: the gray-scale divot sign. Obstet Gynecol. May 1999;93(5 Pt 2):854. doi: 10.1016/s0029-7844(98)00373-1. | |
Aksoy U. Prenatal color Doppler sonographic evaluation of nuchal encirclement by the umbilical cord. J Clin Ultrasound. Nov-Dec 2003;31(9):473–7. doi: 10.1002/jcu.10211. | |
Sepulveda W. Antenatal course and perinatal outcome after ultrasound detection of triple nuchal cord: a case series. J Matern Fetal Neonatal Med. Oct 2021;34(19):3246–3251. doi: 10.1080/14767058.2019.1659773. | |
Hashiramoto S, Takita H, Arakaki T, Yamashita Y, Kaneko M, Matsuoka R, Sekizawa A. Antenatal Detection of a True Umbilical Cord Knot: Facilitating Optimal Delivery Management. J Clin Ultrasound. Oct 29 2024;doi: 10.1002/jcu.23873. | |
Airas U, Heinonen S. Clinical significance of true umbilical knots: a population-based analysis. Am J Perinatol. Apr 2002;19(3):127–32. doi: 10.1055/s-2002-25311. | |
Zacharias NM, Rhinehart-Ventura J. Cyclone sign: prenatal ultrasound diagnosis of a true umbilical cord knot. Am J Obstet Gynecol. Apr 2023;228(4):471. doi: 10.1016/j.ajog.2022.10.036. | |
Sherer DM, Dalloul M, Zigalo A, Bitton C, Dabiri L, Abulafia O. Power Doppler and 3-dimensional sonographic diagnosis of multiple separate true knots of the umbilical cord. J Ultrasound Med. Sep 2005;24(9):1321–3. doi: 10.7863/jum.2005.24.9.1321. | |
Sherer DM, Amoabeng O, Dryer AM, Dalloul M. Current Perspectives of Prenatal Sonographic Diagnosis and Clinical Management Challenges of True Knot of the Umbilical Cord. Int J Womens Health. 2020;12:221–233. doi: 10.2147/IJWH.S192260. | |
Ugurlucan FG, Yuksel A. Is complete umbilical cord scanning possible at the second-trimester ultrasound scan? J Clin Ultrasound. May 2015;43(4):249–253. doi: 10.1002/jcu.22242. | |
Post AL. Managing Monoamniotic Twin Pregnancies. Clin Obstet Gynecol. Dec 1 2023;66(4):841–853. doi: 10.1097/GRF.0000000000000817. | |
D'Antonio F, Odibo A, Berghella V, et al. Perinatal mortality, timing of delivery and prenatal management of monoamniotic twin pregnancy: systematic review and meta-analysis. Ultrasound Obstet Gynecol. Feb 2019;53(2):166–174. doi: 10.1002/uog.20100. | |
Van Mieghem T, Abbasi N, Shinar S, Keunen J, Seaward G, Windrim R, Ryan G. Monochorionic monoamniotic twin pregnancies. Am J Obstet Gynecol MFM. Mar 2022;4(2S):100520. doi: 10.1016/j.ajogmf.2021.100520. | |
Ishii K. Prenatal diagnosis and management of monoamniotic twins. Curr Opin Obstet Gynecol. Apr 2015;27(2):159–64. doi: 10.1097/GCO.0000000000000160. | |
Vasciaveo L, Zanzarelli E, De Lucia GS, De Feo V, Mastricci AL, Nappi L. Monochorionic monoamniotic twin pregnancy: Inpatient or outpatient management? Our experience. J Clin Ultrasound. Jun 2023;51(5):833–836. doi: 10.1002/jcu.23434. | |
Sherer DM, Sokolovski M, Haratz-Rubinstein N. Diagnosis of umbilical cord entanglement of monoamniotic twins by first-trimester color Doppler imaging. J Ultrasound Med. Nov 2002;21(11):1307–9. doi: 10.7863/jum.2002.21.11.1307. | |
Liberty G, Reyzer SM, Shenhav S, Anteby EY, Cohen SM, Yagel S. Ultrasound diagnosis of first trimester umbilical cord entanglement in monochorionic monoamniotic twins – case report and review of the literature. BMC Pregnancy Childbirth. Nov 28 2024;24(1):801. doi: 10.1186/s12884-024-06962-6. | |
Murata M, Ishii K, Kamitomo M, et al. Perinatal outcome and clinical features of monochorionic monoamniotic twin gestation. J Obstet Gynaecol Res. May 2013;39(5):922–5. doi: 10.1111/jog.12014. | |
Dias T, Mahsud-Dornan S, Bhide A, Papageorghiou AT, Thilaganathan B. Cord entanglement and perinatal outcome in monoamniotic twin pregnancies. Ultrasound Obstet Gynecol. Feb 2010;35(2):201–4. doi: 10.1002/uog.7501. | |
Vayssiere C, Plumere C, Gasser B, Neumann M, Favre R, Nisand I. Diagnosing umbilical cord entanglement in monoamniotic twins: becoming easier and probably essential. Ultrasound Obstet Gynecol. Oct 2004;24(5):587–9. doi: 10.1002/uog.1745. | |
Overton TG, Denbow ML, Duncan KR, Fisk NM. First-trimester cord entanglement in monoamniotic twins. Ultrasound Obstet Gynecol. Feb 1999;13(2):140–2. doi: 10.1046/j.1469-0705.1999.13020140.x. | |
Zollner U, Rehn M, Heuer S, Morr AK, Dietl J. Umbilical cord entanglement in monoamniotic twins. Ultrasound Obstet Gynecol. Jul 2012;40(1):121–2. doi: 10.1002/uog.11129. | |
Ito A, Nakata M, Oji A, et al. Diagnosis of umbilical cord entanglement in a monochorionic diamniotic twin pregnancy with spontaneous septostomy of the dividing membranes using dual-gate Doppler imaging. J Med Ultrason (2001). Jan 2018;45(1):189–192. doi: 10.1007/s10396-017-0793-6. | |
Fleming T, Miller T. Spontaneous septostomy in monochorionic diamniotic twins resulting in cord entanglement and fetal demise. Australas J Ultrasound Med. Aug 2012;15(3):103–106. doi: 10.1002/j.2205-0140.2012.tb00014.x. | |
Nasrallah FK, Faden YA. Antepartum rupture of the intertwin-dividing membrane in monochorionic diamniotic twins: a case report and review of the literature. Prenat Diagn. Sep 2005;25(9):856–60. doi: 10.1002/pd.1157. | |
Sherer DM, Bitton C, Stimphil R, Dalloul M, Khoury-Collado F, Abulafia O. Cord entanglement of monochorionic diamniotic twins following spontaneous antepartum septostomy sonographically simulating a true knot of the umbilical cord. Ultrasound Obstet Gynecol. Nov 2005;26(6):676–8. doi: 10.1002/uog.2612. | |
Hanaoka U, Tenkumo C, Ito M, Mori N, Tanaka H, Hata T. Three-dimensional surface-rendered imaging of cord entanglement in monoamniotic twins. Arch Gynecol Obstet. Oct 2012;286(4):1091–2. doi: 10.1007/s00404-012-2406-3. | |
Kuwata T, Matsubara S, Suzuki M. 3D color Doppler of monoamniotic twin cord entanglement. Arch Gynecol Obstet. May 2010;281(5):973–4. doi: 10.1007/s00404-009-1304-9. | |
Henrich W, Tutschek B. Cord entanglement in monoamniotic twins: 2D and 3D colour Doppler studies. Ultraschall Med. Dec 2008;29 Suppl 5:271–2. doi: 10.1055/s-2007-963641. | |
Faber R, Stepan H. Umbilical cord entanglement in monoamniotic twins. Ultrasound Obstet Gynecol. Oct 2004;24(5):592–3. doi: 10.1002/uog.1735. | |
Hugon-Rodin J, Guilbert JB, Baron X, Camus E. Notching of the umbilical artery waveform associated with cord entanglement in a monoamniotic twin pregnancy. J Matern Fetal Neonatal Med. Oct 2013;26(15):1559–61. doi: 10.3109/14767058.2013.794204. | |
Abuhamad AZ, Mari G, Copel JA, Cantwell CJ, Evans AT. Umbilical artery flow velocity waveforms in monoamniotic twins with cord entanglement. Obstet Gynecol. Oct 1995;86(4 Pt 2):674–7. doi: 10.1016/0029-7844(95)00210-i. | |
Aurioles-Garibay A, Hernandez-Andrade E, Romero R, et al. Presence of an umbilical artery notch in monochorionic/monoamniotic twins. Fetal Diagn Ther. 2014;36(4):305–11. doi: 10.1159/000361020. | |
Rossi AC, Prefumo F. Impact of cord entanglement on perinatal outcome of monoamniotic twins: a systematic review of the literature. Ultrasound Obstet Gynecol. Feb 2013;41(2):131–5. doi: 10.1002/uog.12345. | |
Ezra Y, Shveiky D, Ophir E, Nadjari M, Eisenberg VH, Samueloff A, Rojansky N. Intensive management and early delivery reduce antenatal mortality in monoamniotic twin pregnancies. Acta Obstet Gynecol Scand. May 2005;84(5):432–5. doi: 10.1111/j.0001-6349.2005.00683.x. | |
Brinkman LAM, Duin LK, Adama van Scheltema PN, et al. Pop-off mechanisms in fetal megacystis: extravasation, umbilical cord cyst, ureterocele and megaureter. Ultrasound Obstet Gynecol. Apr 2025;65(4):487–494. doi: 10.1002/uog.29200. | |
Allen MK, Minto O. Four-Vessel Umbilical Cord: Supernumerary Right Umbilical Vein With No Associated Congenital Anomalies. Cureus. Aug 2024;16(8):e67283. doi: 10.7759/cureus.67283. | |
Karatza A, Tsamandas A, Varvarigou A, Davlouros P, Pavlou V, Mantagos S. Supernumerary umbilical vein in a hydropic neonate with hypertrophic cardiomyopathy. Fetal Pediatr Pathol. 2011;30(3):173–6. doi: 10.3109/15513815.2010.547557. | |
Lei T, Xie HN, Feng JL. Prenatal diagnosis of four-vessel umbilical cord with supernumerary vein varix: A case report and literature review. J Obstet Gynaecol Res. Jul 2017;43(7):1200–1204. doi: 10.1111/jog.13324. | |
Adams KV, Bernieh A, Morris RW, Saad AG. Umbilical Cord Teratomas Associated With Congenital Malformations. Arch Pathol Lab Med. Feb 2020;144(2):156–159. doi: 10.5858/arpa.2019-0161-RA. | |
Travessa AM, Santo S, Luis R, Carvalho Afonso M, Carvalho R, Vitorino E, Sousa AB. A fetus with an immature umbilical cord teratoma associated with exomphalos: case report and review of the literature. Rom J Morphol Embryol. Jul-Sep 2020;61(3):953–957. doi: 10.47162/RJME.61.3.37. | |
Grossi AP, Astori AF, Nakatani ET, Jure R, Salazar D, Tonni G, Sepulveda W. Prenatal Diagnosis of Umbilical Cord Angiomyxoma: Case Studies and Literature Review of 45 Cases. J Ultrasound Med. Sep 2024;43(9):1769–1784. doi: 10.1002/jum.16506. | |
Gramellini D, Pedrazzoli G, Sacchini C, Montalto M, Piantelli G. Color Doppler ultrasound in prenatal diagnosis of umbilical cord angiomyxoma. Case report. Clin Exp Obstet Gynecol. 1993;20(4):241–4. | |
Cheng HP, Hsu CY, Chen CP, Su TH. Angiomyxoma of the umbilical cord. Taiwan J Obstet Gynecol. Dec 2006;45(4):360–2. doi: 10.1016/S1028-4559(09)60262-X. | |
Jauniaux E, Moscoso G, Chitty L, Gibb D, Driver M, Campbell S. An angiomyxoma involving the whole length of the umbilical cord. Prenatal diagnosis by ultrasonography. J Ultrasound Med. Jul 1990;9(7):419–22. doi: 10.7863/jum.1990.9.7.419. | |
Lisovaja I, Franckevica I, Vedmedovska N. Large Angiomyxoma of the Umbilical Cord-Uncomplicated Rupture of Tumor Membranes at 32 Weeks of Gestation. Diagnostics (Basel). May 28 2022;12(6) doi: 10.3390/diagnostics12061339. | |
Sepulveda W, Mackenna A, Sanchez J, Corral E, Carstens E. Fetal prognosis in varix of the intrafetal umbilical vein. J Ultrasound Med. Mar 1998;17(3):171–5. doi: 10.7863/jum.1998.17.3.171. | |
Ghahremani T, Britt AB, Ray E, Jones A, Du R, Magann EF. Perinatal Outcomes and Management of Umbilical Vein Varix: A Comprehensive Review of 392 Cases. J Clin Med. Jan 11 2025;14(2) doi: 10.3390/jcm14020441. | |
Koorn I, Heinrich H, Nelissen A, et al. Isolated fetal umbilical vein varix and the association with intrauterine fetal death and fetal growth restriction: A systematic review, meta-analysis, and nested retrospective cohort study. Prenat Diagn. May 2024;44(5):595–613. doi: 10.1002/pd.6538. | |
Ozek MA, Calis P, Bayram M, Karcaaltincaba D. Fetal intraabdominal umbilical vein varix: antenatal diagnosis and management. J Matern Fetal Neonatal Med. Jan 2018;31(2):245–250. doi: 10.1080/14767058.2016.1278208. | |
Mahony BS, McGahan JP, Nyberg DA, Reisner DP. Varix of the fetal intra-abdominal umbilical vein: comparison with normal. J Ultrasound Med. Feb 1992;11(2):73–6. doi: 10.7863/jum.1992.11.2.73. | |
Beraud E, Rozel C, Milon J, Darnault P. Umbilical vein varix: Importance of ante- and post-natal monitoring by ultrasound. Diagn Interv Imaging. Jan 2015;96(1):21–6. doi: 10.1016/j.diii.2014.01.009. | |
di Pasquo E, Kuleva M, O'Gorman N, Ville Y, Salomon LJ. Fetal intra-abdominal umbilical vein varix: retrospective cohort study and systematic review and meta-analysis. Ultrasound Obstet Gynecol. May 2018;51(5):580–585. doi: 10.1002/uog.18895. | |
Valsky DV, Rosenak D, Hochner-Celnikier D, Porat S, Yagel S. Adverse outcome of isolated fetal intra-abdominal umbilical vein varix despite close monitoring. Prenat Diagn. Jun 2004;24(6):451–4. doi: 10.1002/pd.897. | |
Lu M, Kakani N, Romagnoli C, Yue L, Xiong W, An S, Song B. Two- and three-dimensional sonographic diagnosis of fetal intra-abdominal umbilical vein varix: a case report. J Clin Ultrasound. Nov-Dec 2012;40(9):586–9. doi: 10.1002/jcu.21880. | |
Fung TY, Leung TN, Leung TY, Lau TK. Fetal intra-abdominal umbilical vein varix: what is the clinical significance? Ultrasound Obstet Gynecol. Feb 2005;25(2):149–54. doi: 10.1002/uog.1815. | |
Suryadevara M, Gupta R, Mishra GV, Nagendra V, Bhansali PJ. Antenatal Hydrops Fetalis with Umbilical Vein Varix and Thrombosis – Ultrasound Imaging: A Rare Case. J Med Ultrasound. Oct-Dec 2024;32(4):337–340. doi: 10.4103/jmu.jmu_11_23. | |
Novoa V, Shazly S, Ibirogba ER, et al. Perinatal outcomes of fetal intra-abdominal umbilical vein varix: a multicenter cohort study. J Matern Fetal Neonatal Med. Oct 2021;34(20):3393–3396. doi: 10.1080/14767058.2019.1685969. | |
Byers BD, Goharkhay N, Mateus J, Ward KK, Munn MB, Wen TS. Pregnancy outcome after ultrasound diagnosis of fetal intra-abdominal umbilical vein varix. Ultrasound Obstet Gynecol. Mar 2009;33(3):282–6. doi: 10.1002/uog.6233. | |
Bromley B, Shipp TD, Benacerraf BR. Amnion-chorion separation after 17 weeks' gestation. Obstet Gynecol. Dec 1999;94(6):1024–6. doi: 10.1016/s0029-7844(99)00457-3. | |
Nassr AA, Hessami K, Shazly SA, et al. Perinatal outcomes of iatrogenic chorioamniotic separation following fetoscopic surgery: systematic review and meta-analysis. Ultrasound Obstet Gynecol. Sep 2021;58(3):347–353. doi: 10.1002/uog.23588. | |
Bibbo C, Little SE, Bsat J, Botka KA, Benson CB, Robinson JN. Chorioamniotic Separation Found on Obstetric Ultrasound and Perinatal Outcome. AJP Rep. Jul 2016;6(3):e337–43. doi: 10.1055/s-0036-1593407. | |
Zhu KH, Young BC, Shamshirsaz AA, et al. Outcomes of prenatally diagnosed spontaneous chorioamniotic membrane separation in singleton pregnancies: A systematic review of case series and case reports. Prenat Diagn. Oct 2020;40(11):1366–1374. doi: 10.1002/pd.5767. | |
Graf JL, Bealer JF, Gibbs DL, Adzick NS, Harrison MR. Chorioamniotic membrane separation: a potentially lethal finding. Fetal Diagn Ther. Mar-Apr 1997;12(2):81–4. doi: 10.1159/000264436. | |
Lewi L, Hanssens M, Spitz B, Deprest J. Complete chorioamniotic membrane separation. Case report and review of the literature. Fetal Diagn Ther. Jan-Feb 2004;19(1):78–82. doi: 10.1159/000074266. | |
Kaufman AJ, Fleischer AC, Thieme GA, Shah DM, James AE, Jr. Separated chorioamnion and elevated chorion: sonographic features and clinical significance. J Ultrasound Med. Mar 1985;4(3):119–25. doi: 10.7863/jum.1985.4.3.119. | |
Burrows PE, Lyons EA, Phillips HJ, Oates I. Intrauterine membranes: sonographic findings and clinical significance. J Clin Ultrasound. Jan 1982;10(1):1–8. doi: 10.1002/jcu.1870100102. | |
Burton DJ, Filly RA. Sonographic diagnosis of the amniotic band syndrome. AJR Am J Roentgenol. Mar 1991;156(3):555–8. doi: 10.2214/ajr.156.3.1899757. | |
Rujiwetpongstorn J, Tongsong T. Amniotic band syndrome following septostomy in management of twin-twin transfusion syndrome: a case report. J Perinatol. May 2008;28(5):377–9. doi: 10.1038/sj.jp.7211927. | |
Chen CP. Prenatal diagnosis of atypical facial clefting should alert amniotic band syndrome and prompt a search for associated amniotic bands and other structural anomalies. Genet Couns. 2007;18(2):255–7. | |
Chen CP, Chang TY, Lin YH, Wang W. Prenatal sonographic diagnosis of acrania associated with amniotic bands. J Clin Ultrasound. Jun 2004;32(5):256–60. doi: 10.1002/jcu.20021. | |
Dabkowska S, Kucinska-Chahwan A, Beneturska A, et al. Prenatal diagnosis and clinical significance of cephalocele-A single institution experience and literature review. Prenat Diagn. Apr 2020;40(5):612–617. doi: 10.1002/pd.5654. | |
Finberg HJ, Glass M. Craniofacial damage from amniotic band syndrome subsequent to pathologic chorioamniotic separation at 10 weeks' gestation. J Ultrasound Med. Sep 1996;15(9):665–8. doi: 10.7863/jum.1996.15.9.665. | |
Muraskas JK, McDonnell JF, Chudik RJ, Salyer KE, Glynn L. Amniotic band syndrome with significant orofacial clefts and disruptions and distortions of craniofacial structures. J Pediatr Surg. Apr 2003;38(4):635–8. doi: 10.1053/jpsu.2003.50141. | |
Paladini D, Foglia S, Sglavo G, Martinelli P. Congenital constriction band of the upper arm: the role of three-dimensional ultrasound in diagnosis, counseling and multidisciplinary consultation. Ultrasound Obstet Gynecol. May 2004;23(5):520–2. doi: 10.1002/uog.1042. | |
Sriwastwa A, Kline-Fath BM, Bierbrauer KS, Venkatesan C, Nagaraj UD. Fetal craniofacial anomalies in amniotic band sequence. Pediatr Radiol. Oct 2024;54(11):1933–1934. doi: 10.1007/s00247-024-05993-7. | |
Society for Maternal-Fetal M, Monteagudo A. Exencephaly-anencephaly Sequence. Am J Obstet Gynecol. Dec 2020;223(6):B5-B8. doi: 10.1016/j.ajog.2020.08.176. | |
Richter J, Wergeland H, DeKoninck P, De Catte L, Deprest JA. Fetoscopic release of an amniotic band with risk of amputation: case report and review of the literature. Fetal Diagn Ther. 2012;31(2):134–7. doi: 10.1159/000335026. | |
Ferrer-Marquez F, Peiro JL, Tonni G, Ruano R. Fetoscopic Release of Amniotic Bands Based on the Evidence-A Systematic Review. Prenat Diagn. Sep 2024;44(10):1231–1241. doi: 10.1002/pd.6636. | |
Gueneuc A, Chalouhi GE, Borali D, Mediouni I, Stirnemann J, Ville Y. Fetoscopic Release of Amniotic Bands Causing Limb Constriction: Case Series and Review of the Literature. Fetal Diagn Ther. 2019;46(4):246–256. doi: 10.1159/000495505. | |
Waerlop F, Dehaene I, Roelens K. Successful management of fetal hypoxia due to amniotic banding at 26 weeks of pregnancy: A case report of a rare survival. Eur J Obstet Gynecol Reprod Biol. Jun 2024;297:264–266. doi: 10.1016/j.ejogrb.2024.04.010. | |
Li J, Li GX, Dong L, et al. [Fetoscopy for intrauterine diagnosis and treatment of amniotic band syndrome: a clinical analysis of 7 cases and literature review]. Zhonghua Fu Chan Ke Za Zhi. Jul 25 2024;59(7):530–539. doi: 10.3760/cma.j.cn112141-20240228-00125. | |
Javadian P, Shamshirsaz AA, Haeri S, et al. Perinatal outcome after fetoscopic release of amniotic bands: a single-center experience and review of the literature. Ultrasound Obstet Gynecol. Oct 2013;42(4):449–55. doi: 10.1002/uog.12510. | |
Sepulveda W. Time for a more detailed prenatal examination of the umbilical cord? Ultrasound Obstet Gynecol. Mar 1999;13(3):157–60. doi: 10.1046/j.1469-0705.1999.13030157.x. |
Online Study Assessment Option
All readers who are qualified doctors or allied medical professionals can automatically receive 2 Continuing Professional Development points plus a Study Completion Certificate from GLOWM for successfully answering four multiple-choice questions (randomly selected) based on the study of this chapter. Medical students can receive the Study Completion Certificate only.
(To find out more about the Continuing Professional Development awards program CLICK HERE)
