This chapter should be cited as follows:
Tsibizova V, Tosto V, et al., Glob Libr Women's Med
ISSN: 1756-2228; DOI 10.3843/GLOWM.419023
The Continuous Textbook of Women’s Medicine Series – Obstetrics Module
Volume 19
Pregnancy shortening: etiology, prediction and prevention
Volume Editors:
Professor Arri Coomarasamy, University of Birmingham, UK
Professor Gian Carlo Di Renzo, PREIS International School, Florence, Italy
Professor Eduardo Fonseca, Federal University of Paraiba, Brazil

Chapter
Miscarriage: Risk Factors and Prevention
First published: March 2024
Study Assessment Option
By answering four multiple-choice questions (randomly selected) after studying this chapter, readers can qualify for Continuing Professional Development points plus a Study Completion Certificate from GLOWM.
See end of chapter for details.
INTRODUCTION
It is estimated that miscarriage is the most common form of pregnancy loss, with about 80% of early pregnancy loss occurring in the first trimester of gestation. The risk is multivariate and while there are already known risk factors (e.g., maternal age, previous obstetrical history, and maternal comorbidities), it is difficult to establish absolute predictors of future pregnancy loss. Scientific investigation is active in studying possible/probable risk factors that may be involved: identifying the potentially and modifiable pre-pregnant risk factors associated with increased miscarriage risk is crucial to define preventive strategies and optimize obstetrical care and outcomes.
DEFINITION
A miscarriage is the spontaneous expulsion of the embryo/fetus from the uterus before 22 weeks.
The WHO defines a miscarriage as the expulsion or removal of an embryo/fetus weighing less than 500 g, corresponding to approximately 22 weeks of gestation.
This term is defined differently according to local laws and it is sometimes the subject of debate among experts. In the UK, the viability limit is defined by law as up to 24 weeks of pregnancy. The United States defines miscarriage as a clinical pregnancy loss of less than 20 weeks, while Europe defines miscarriage as a loss before 22 weeks of gestation. The viability limit is legally defined in most countries and, especially as neonatal intensive care for preterm infants becomes more effective in high-income countries, often deviates from medical viability limits.1
Common symptoms and clinical presentation of miscarriage include cramping and pain in the lower abdomen, a discharge or blood/fluid from vagina, a discharge of tissue from vagina, disappearing of pregnancy signs, such as breast tenderness and feeling sick.
EPIDEMIOLOGY
Miscarriage is generally defined as the loss of a pregnancy before viability. An estimated 23 million miscarriages occur every year worldwide, translating to 44 pregnancy losses each minute. The pooled risk of miscarriage is 15% of all recognized pregnancies. The population prevalence of women who have had one miscarriage is 10% (10.3–11.4%), two miscarriages are 1–9% (1.8–2.1%), and three or more miscarriages is 0–7% (0.5–0.8%).1
RISK FACTORS
A growing body of scientific evidence supports that miscarriage risk is often increased by multiple potentially identifiable and modifiable risk factors, and a considerable proportion of miscarriages may be preventable.
DEMOGRAPHIC RISK FACTORS
The population's demographics will influence the risk of miscarriage, with the woman's age strongly influencing the risk.
At any age, the number of previous miscarriages profoundly affects the risk of miscarriage. The risk of miscarriage is lowest in women aged 20–29 at 12%, rising sharply to 65% in women aged 45 and older. The risk of miscarriage is most lacking in women without a history of miscarriage (11%). Then, it increases by about 10% with each additional miscarriage, reaching 42% in women with three or more previous miscarriages.
The association between a woman's age and miscarriage is explained by an age-related increase in the frequency of embryonic trisomies and chorion mosaicism, especially trisomies of chromosomes 13, 14, 15, 16, 18, 20, 21, and 22. The trisomy 16 is the most common aneuploidy in miscarriage, and it accounts for approximately 6% of miscarriages.
In addition, studies reported that metabolic issues may be relevant contributors in influencing obstetric outcomes. In this regard, high BMI ≥25 kg/m2 increased the risk of miscarriage, and being overweight or obese was associated with a higher risk of miscarriage. It also is significantly higher in African ethnicity than in Caucasian ethnicity, showing a probable influence of genetic ethnicity.1,2
EMBRYONIC CHROMOSOMAL ERRORS
Chromosomal abnormalities are found in 60% of miscarried material but less than 1% of live births when prenatal diagnosis is not used. Most frequent are autosomal trisomy, monosomy X, and triploidy. Also, developmental anomalies of embryos are found in miscarriages with normal chromosomes.3
ENDOMETRIAL DEFECTS
Endometrium transforming into decidua is essential for implantation to accommodate the invading trophoblast. Inappropriate decidualization can result from changes in immune cells, primarily because of uterine natural killer cells, Th1 cytokines high release and as a result activation of pro-inflammatory reaction, especially the thrombosis mechanism in the trophoblast which result in miscarriage. Multiple risk factors of recurrent miscarriage, including metabolic (e.g., obesity) and endocrine (e.g., hypothyroidism) disorders, have been shown to affect the decidual process in the endometrium adversely.4
MATERNAL CONDITIONS
Antiphospholipid syndrome, thyroid autoantibodies, and subclinical hypothyroidism are associated with miscarriage and recurrent pregnancy loss (RPL).1 Mild endocrine diseases are not associated with RPL, but thyroid dysfunction and pregestational diabetes mellitus may increase risk. Endometriosis is associated with increased RPL as a complex gynecological condition. High BMI and polycystic ovary syndrome (PCOS), common conditions in childbearing age, increase RPL risk and are associated with anovulation and infertility.
UTERINE ANOMALIES
Bicornuate uterus, unicornuate, duplication of reproductive organs, and uterine septae have been associated with spontaneous and recurrent miscarriage. All of these anomalies are congenital, but uterine septae is usually linked with repeated uterine curettage after cervical dilatation and could be a cause of injury to the cervix and endometrial cavity or change in the uterine microbiome, increasing the risk of miscarriage/preterm birth due to damage to the cervix or chronic endometritis. It is also a cause of abnormal placentation, which increases the risk of placental abruption and placenta previa.5
Other disease of the uterus, such as adenomyosis, can destroy the junctional zone, leading to inappropriate spiral artery remodeling or defective placentation and, consequently, results in miscarriage or placenta-related complications.6 Moreover, adenomyosis shows a pro-inflammatory microenvironment. It has been demonstrated that oxidative stress and inflammatory cytokines such as IL-1, IL-8, and TNF-α are increased in the affected tissues, which may impact the pregnancy outcome.7
INFECTION
Bacterial (i.e., bacterial vaginosis, brucellosis, Chlamydia trachomatis, and syphilis), viral (i.e., herpes simplex virus type 1 [HSV-1] and HSV-2, human cytomegalovirus, human papillomavirus, parvovirus, adeno-associated viruses, parvovirus B19, bocavirus, HIV, polyomavirus, dengue virus, hepatitis B, hepatitis C, rubella, and coronaviruses [SARS, MERS, and H1N1]), and protozoa (i.e., malaria and toxoplasmosis) infections have all been linked to miscarriage.1
MICROBIOME
The vaginal microbiome has a strong association with the chance of miscarriage and other obstetric conditions, such as preterm labor and preterm birth. The vaginal microbiome is a dynamic ecosystem of microbes that are essential for preserving vaginal health and avoiding infections, and thus a consequent cascade of negative events that may variably contribute to increase the risk of pregnancy loss. During pregnancy, the composition of the vaginal microbiota has a substantial impact on the health of both the mother and the fetus, and the microbiome of pregnant women is characterized by greater stability and lower diversity than in non-pregnant women. Recognized factors that can influence the vaginal microbiome include hormonal changes, food, sexual practices, medical treatments, and urogenital infections.
Miscarriage is more commonly associated with a deficiency of lactobacillus microbiota. In a healthy pregnancy, the vaginal bacterial composition remains stable, with a predominance of Lactobacillus species. Grewal has shown that vaginal microbiota with low mount of Lactobacillus spp. combined with a local inflammatory response, predispose to euploid miscarriage. Also, the cytokine response, which is mentioned above, leads to specific pro-inflammatory reactions. This study has shown that in some cases, antibiotic or pre- or probiotic treatments can reduce the risk of miscarriage. Further studies are needed to validate these findings.8
PATERNAL AGE
The study of Oldereid et al. found associations between advanced paternal age and psychiatric disorders like autism spectrum and schizophrenia, but also with stillbirth and several congenital disabilities.9 The father's age and the mutation rate in the offspring are strongly related, possibly due to the larger number of germline divisions in older males.10 Increasing paternal age is associated with a higher frequency of point mutations, sperm DNA strand breaks, genetic imprinting errors, and chromosomal anomalies, all factors related to miscarriage.11 Thus, from a biological point of view, it seems justified to consider paternal age as an independent risk factor for miscarriage.
LIFESTYLE RISK FACTORS
In a systematic review and meta-analysis Coomarasamy et al. provided evidence of a protective association between diet and miscarriage. An overall dietary exposure that is high in quality with healthy nutrient sources and low in pro-inflammatory factors or unhealthy food groups, such as highly refined, processed meat or sugar substitutes may be associated with a reduction in miscarriage risk. This supports the positive effect of healthy dietary choices on fertility status and maternal–fetal–neonatal outcomes. Therefore, women who wish to reduce their risk of pregnancy loss should be encouraged to make healthy food choices.12
Smoking is an important modifiable risk factor for miscarriage. Miscarriage risk increases with the amount smoked (1% increase in relative risk per cigarette smoked daily). Alcohol use is also an important modifiable risk factor, as high alcohol consumption during the first trimester is associated with increased miscarriage risk. A high caffeine intake might be related to miscarriage. Furthermore, any association between caffeine and miscarriage is likely to be confounded as a healthy pregnancy is associated with nausea and vomiting (due to pregnancy hormones), which might reduce caffeine consumption. Night shift work is also associated with an increased risk of miscarriage. This risk appeared to follow a dose-response relationship.12
ENVIRONMENTAL RISK FACTORS
Air pollution is associated with stillbirth, preterm delivery, and low birth weight. A large study assessed the effect of exposure to air pollution on miscarriage rates in Beijing, China, showing a strong relationship with miscarriage (OR 1.51; 95% CI 1.33–1.69).13 Similarly, a case-control study on women attending an emergency department in Utah, USA, found that 10 parts per billion rise in nitrogen oxide concentrations was associated with an increased risk of miscarriage (OR 1.16; 95% CI 1.01–1.33).14 Pesticides have also been linked to recurrent miscarriage, especially sprayed pesticides in rural South Africa in the first 3 months of pregnancy, which were associated with an increased risk of miscarriage (OR 2.8; 95% CI 1.1–7.2).15 This epidemiological study correlates with a clinical study showing higher concentrations of serum organochlorine pesticides in women with recurrent miscarriage than controls (i.e., who have live term births).
CLINICAL RISK FACTORS
Gestational age is also an essential factor in determining the risk of miscarriage. Moreover, as the pregnancy progresses, the risk of miscarriage decreases. If the pregnancy reaches 8 weeks, the risk of miscarriage is significantly reduced, and the chance of a live birth approaches 97–98%. Another important risk factor is the number of previous miscarriages.
Threatened miscarriage, defined as vaginal bleeding in early pregnancy (in the first 12 weeks), is among the most common reasons for adverse outcomes and complications such as antepartum hemorrhage due to placenta previa (OR 1.62; 95% CI 1.19–2.22) or antepartum bleeding of unknown origin (OR 2.47; 95% CI 1.52–4.02), preterm prelabor rupture of membranes (OR 1.78; 95% CI 1.28–2.48), preterm delivery (OR 2.05; 95% CI 1.76–2.40) (Table 1), and fetal growth restriction (OR 1.54; 95% CI 1.18–2.00), higher perinatal mortality rates (OR 2.15; 95% CI 1.41–3.27) and low-birth weight neonates (OR 1.83; 95% CI 1.48–2.28).1,16
1
Miscarriage and preterm birth: similar risk factors.
Before pregnancy | During pregnancy | Mechanisms |
Genetics and heritability | Stress and hormones | Premature myometrial activation |
Ethnicity | Cervical insufficiency | |
Maternal age | Uterine overdistension | Premature cervical ripening |
Reproductive system diseases | Ischemia | |
Maternal medical disorders | Inflammation/infection | Premature rupture of membranes |
Environmental factors and epigenetics | Male fetus | |
Previous abortion/preterm delivery | ||
Previous cesarean section |
RECURRENT MISCARRIAGE SYNDROME
Recurrent pregnancy loss is defined as two or more consecutive miscarriages before the 20th week of gestation17 and affects 1–5% of women of fertile age. Several conditions have been associated, such as chromosome anomalies in parents or embryos, prothrombotic states, structural anomalies of the uterus, endocrine dysfunction, infections, and immunological factors. It is estimated that 35–60% of cases are still idiopathic, suggesting that genetic, epigenetic, and environmental factors contribute to the recurrent pregnancy loss phenotype. Moreover, in couples with recurrent pregnancy loss (RPL), identification of mutations in the SYCP3 gene, which encodes a fundamental component of the synaptonemal complex involved in meiotic segregation, has demonstrated a correlation between meiosis, aneuploidy, and recurrent miscarriages.18
Recurrent miscarriage can also be linked to thrombophilia. Mutations in the Leiden factor V gene (F5), coagulation factor II gene (F2) and annexin A5 gene (ANXA5 encoding an anticoagulant protein active in placental villi) have been associated with an increased risk of recurrent pregnancy loss. Factor V Leiden mutation has been related to the RPL and Activated Protein C Resistance and Prothrombin G20210A mutation. MTHFR mutation, deficiency of Protein C, S and Antithrombin III have not conclusively been related to RPL.
Finally, mutations in NLRP7 and KHDC3 L have been associated with hydatidiform mole, a trophoblast disease. Hydatidiform mole is due to a fertilization defect and is characterized by trophoblast proliferation that prevents normal embryo development. Mutations in the two genes have been reported in 1% of cases of hydatidiform mole.19
MISCARRIAGE AND LONG-TERM HEALTH RISKS
Recurrent miscarriage is associated with long-term health problems beyond pregnancy. The recurrent miscarriage is associated with an increased risk of cardiovascular disease and venous thromboembolism. These findings are important because they add to the concept of a recurrent miscarriage syndrome and could mean that a history of repeated miscarriage is an opportunity to reduce risks for cardiovascular and thromboembolic disease. The psychological consequences of miscarriage involve both trauma and bereavement, and these consequences might have little or no outward physical manifestation so that they can go unrecognized by healthcare professionals, family, and friends. This scenario occurs particularly in a society that views miscarriage as unimportant or shameful, thus leading to the concealment of a pregnancy loss and its consequences. Also, anxiety, depression, and suicide are strongly associated with miscarriage. A multicenter, prospective cohort study of 537 women following a miscarriage found that 9 months after a pregnancy loss, 18% met the criteria for post-traumatic stress, 17% for moderate or severe anxiety, and 6% for moderate or severe depression.20 Women at risk of psychological distress after miscarriage should have an optimal treatment strategy before planning their subsequent pregnancy.
PREVENTION OF MISCARRIAGE IN WOMEN AT HIGH RISK OF MISCARRIAGE
Several interventions have targeted asymptomatic women with no vaginal bleeding or pelvic pain in early pregnancy but have other risk factors for miscarriage, such as a history of recurrent pregnancy losses. The key interventions may include lifestyle changes, progestogens, anticoagulants, levothyroxine, metformin, human chorionic gonadotropin, immunomodulatory agents, micronutrient supplementation, depending on identified risk factors and should be tailored to the patient.21,22,23
When a pregnancy is planned, a pre-conceptional visit represents a key moment in which each woman can be evaluated for risk factors for adverse pregnancy outcomes, especially in the presence of a previous obstetrical history and/or already known maternal comorbidities, in order to plan the conception in a moment of clinical and therapeutic stability.
PROGESTERONE
Progesterone is the oldest known hormone. Since the discovery of progesterone in walnut trees over 500,000 years ago, its history is extensive. It is among the oldest bio-regulators, possibly pre-dating contemporary plants and animals.24,25 It is essential for the conception and maintenance of a pregnancy. Progesterone deficiency could be a cause of miscarriage and RPL. Micronized vaginal progesterone treatment can therefore be considered for asymptomatic women with recurrent miscarriages and is likely to be more effective in women with many previous miscarriages.26
The NICE committee guideline on management, "Ectopic pregnancy and miscarriage: diagnosis and initial management (NG126)" recommends using vaginal micronized progesterone to treat women with the dual risk factors of a history of one or more previous miscarriages and early pregnancy bleeding. This should be a course of treatment with vaginal micronized progesterone 400 mg twice daily, started at the time of presentation of vaginal bleeding up to 16 weeks.22 Giving micronized progesterone to women with recurrent miscarriages early in their pregnancy may lower the rates of miscarriage and is likely to be a cost-effective intervention (ESHRE guidelines).27,28
On the other hand, the overall available evidence suggests that progestogens such as dydrogesterone and medroxyprogesterone make little or no difference in the live birth rate for women with threatened or recurrent miscarriage. Moreover, there are serious safety concerns regarding the use of dydrogesterone in support of the luteal phase and the first trimester of pregnancy, with a significant increase in the incidence of congenital anomalies in the offspring.29,30
ANTICOAGULANT THERAPY
Thrombophilia, whether acquired (e.g., antiphospholipid antibodies) or inherited (e.g., factor V Leiden), is associated with vascular thrombosis and adverse pregnancy outcomes, such as recurrent miscarriage. Anticoagulant therapy with low-dose aspirin, heparin, or both reduced the miscarriage rate (relative risk [RR] 0.48; 95% CI 0.32–0.71; low certainty evidence) and increased live birth rate (RR 1.27; 95% CI 1.09–1.49; low certainty evidence),31 compared with aspirin alone, in women with antiphospholipid syndrome and a history of recurrent miscarriage. The professional body guidelines recommend low-dose aspirin and heparin in women with antiphospholipid syndrome and recurrent miscarriage.21 There is no evidence to support the use of aspirin and heparin in women with inherited thrombophilia or in women who do not have thrombophilia.
LEVOTHYROXINE
Treating evident thyroid disorders in the periconceptional period and pregnancy is an established practice for reducing adverse pregnancy outcomes, including miscarriage. There is no clear understanding of managing women with subclinical hypothyroidism or thyroid autoimmunity. Therefore, there is some evidence that subclinical hypothyroidism and autoantibodies without thyroid dysfunction are linked to miscarriage.32 It is suggested to start levothyroxine treatment in the periconceptional time or early pregnancy for subclinical hypothyroidism in women who are trying to conceive, and treatment could be considered for women with subclinical hypothyroidism in whom thyroid-stimulating hormone concentrations are above 4.0 mIU/L. Nevertheless, women with normal thyroid function but with thyroid antibodies do not require levothyroxine treatment.
METFORMIN
Polycystic ovary syndrome is a common endocrine disorder that affects up to 15% of women of reproductive age. Increased insulin resistance, hyperandrogenism, and obesity are closely linked to polycystic ovary syndrome, and all substantially affect reproductive outcomes, including miscarriage. As insulin resistance and resulting hyperinsulinemia are key metabolic features in women with polycystic ovary syndrome, their improvement, through metformin treatment, could improve pregnancy outcomes.33
IMMUNOTHERAPY
A fetus is semi-allogenic and contains antigens of maternal and paternal origins. The physiological mechanisms that allow a woman to accept the paternal antigens are poorly understood; however, a dysfunction in immune tolerance has been hypothesized to cause miscarriage. Various immunological markers, including elevated concentrations of natural killer cells, dysregulated cytokines, and the presence of antiphospholipid antibodies or other autoantibodies, have been linked to miscarriages. Three systematic reviews evaluated immunological interventions, which included oral prednisolone, intravenous immunoglobulins, lymphocyte immunotherapy, and trophoblast membrane immunization. The studies included in the reviews are small and were of low or moderate quality. None of the interventions studied across the three reviews were associated with a reduction in miscarriages or an increase in live births. Therefore, there is insufficient evidence to recommend immunotherapy to prevent recurrent miscarriage.34,35
MICRONUTRIENTS
Vitamins are essential nutrients required for normal metabolism and reproduction. Vitamin D plays an important role in supporting women's reproductive health through its metabolism of 1.25(OH) D (calcitriol, (hormone D)). Its receptors (VDR) are found in the endometrium, placenta, deciduous cells, ovarian granule cells, uterine tube epithelium, pituitary gland, and hypothalamus. The placenta was described as one of the first extrarenal tissues capable of synthesizing calcitriol with CYP27B1 activity in decidual tissue and trophoblast cells. It expresses enzymes and proteins necessary for metabolism and cholecalciferol regulation, such as VDR, VDBP, CYP2R1, CYP27B1, and CYP24A1. It is important to emphasize that vitamin D is an endometrial expression regulator of the HOXA-10 gene, a transcription regulator. It plays an important role in the functional differentiation of the endometrium, which correlates with the quality of implantation processes at the early stages of pregnancy. The prescription of extra vitamin D supplementation should be based on the blood concentration of 25-hydroxy vitamin D and individual habit of daily sun uptake.36
SURGICAL INTERVENTIONS FOR UTERINE ANOMALIES
Surgical interventions on uterine malformations depends on the type of anatomical anomalies and for some circumstances the surgical approach is controversial among experts. For example, hysteroscopic septum resection does not improve outcomes in women with septate uterus, according to the TRUST trial (Dutch Trial Register NTR1676).37
CONCLUSION
The adequate evaluation of women with miscarriage is complex due to the possible multifactorial etiology (Figure 1).38 The pre-conceptional visit is the key moment to screen and stratify the patient/couple risk. Minimum investigations, analyses, and treatments should be offered to couples with repeated miscarriages. Services for couples who have had recurrent miscarriages should provide physical support and psychological support needs. Other further specialist investigations for effective treatments may be needed to improve conceptional chances and better obstetrical outcomes. Thus, a multidisciplinary management may be required. Furthermore, an important priority is the exploration of optimal management approaches for women with mental health distress after miscarriages.
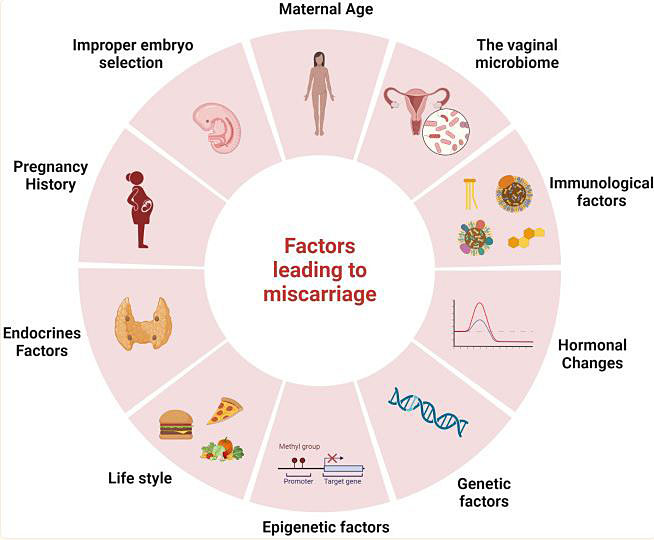
1
Factors that may lead to miscarriage: the possible multifactorial origin. Reproduced with permission from Rikken et al., 2021.38
PRACTICE RECOMMENDATIONS
Early prenatal care (ideally pre-conceptional):
- Schedule an appointment with healthcare worker for early visit. This can help identify any health issues or known risks that may predispose to miscarriage.
- Screen and actively treat any vaginal infection that might alter the vaginal ecosystem.
- Avoid smoking, alcohol, and illicit drugs that can all raise the chance of miscarriage.
- Note the importance of pre-gestational weight and gestational weight gain and BMI.
- Maintain a healthy diet.
- Undertake physical activity can assist in maintaining general health and lower the chance of difficulties during pregnancy.
- Manage stress conditions/interferences, investigate potential social risk factors: since stress during pregnancy increases the chance of miscarriage, it is critical to discover techniques to control stress.
- Avoid exposure to several chemicals, pollutants, and radiation which might raise your risk of miscarriage.
- Note positive previous obstetric and/or gynecological history and other medical conditions (comorbidities – cardiovascular disease, hematological disorders, immunological conditions, nephrological illness, oncological history . . .): plan an individualized course of investigations with a multidisciplinary team and in a referral center.
- Implement tailored recommendations and treatments (pharmacological, surgical, immunomodulatory . . .) depending on identified risk factors and specific clinical conditions.
CONFLICTS OF INTEREST
The author(s) of this chapter declare that they have no interests that conflict with the contents of the chapter.
Feedback
Publishers’ note: We are constantly trying to update and enhance chapters in this Series. So if you have any constructive comments about this chapter please provide them to us by selecting the "Your Feedback" link in the left-hand column.
REFERENCES
Quenby S, Gallos ID, Dhillon-Smith RK, et al. Miscarriage matters: the epidemiological, physical, psychological, and economic costs of early pregnancy loss. The Lancet 2021;397(10285):1658–67. | |
Caserta D, Ralli E, Matteucci E, et al. The influence of socio-demographic factors on miscarriage incidence among Italian and immigrant women: a critical analysis from Italy. Journal of Immigrant and Minority Health 2015;17:843–51. | |
Bettio D, Venci A, Setti PL. Chromosomal abnormalities in miscarriages after different assisted reproduction procedures. Placenta 2008;29:126–8. | |
Muter J, Lynch VJ, McCoy RC, et al. Human embryo implantation. Development 2023;150(10):dev201507. | |
Carbonnel M, Pirtea P, de Ziegler D, et al. Uterine factors in recurrent pregnancy losses. Fertility and Sterility 2021;115(3):538–45. | |
Boucher A, Brichant G, Gridelet V, et al. Implantation failure in endometriosis patients: etiopathogenesis. Journal of Clinical Medicine 2022;11(18):5366. | |
Wang Q, Sun Y, Fan R, et al. Role of inflammatory factors in the etiology and treatment of recurrent implantation failure. Reproductive Biology 2022;22(4):100698. | |
Grewal K, Lee YS, Smith A, et al. Chromosomally normal miscarriage is associated with vaginal dysbiosis and local inflammation. BMC Medicine 2022;20(1):38. | |
Oldereid NB, Wennerholm U-B, Pinborg A, et al. The effect of paternal factors on perinatal and paediatric outcomes: a systematic review and meta-analysis. Human Reproduction Update 2018;24(3):320–89. | |
Kong A, Frigge ML, Masson G, et al. Rate of de novo mutations and the importance of father's age to disease risk. Nature 2012;488(7412):471–5. | |
du Fosse NA, Van der Hoorn M-LP, van Lith JM, et al. Advanced paternal age is associated with an increased risk of spontaneous miscarriage: a systematic review and meta-analysis. Human Reproduction Update 2020;26(5):650–69. | |
Chung Y, Melo P, Pickering O, et al. The association between dietary patterns and risk of miscarriage: a systematic review and meta-analysis. Fertility and Sterility 2023. | |
Zhang L, Liu W, Hou K, et al. Air pollution-induced missed abortion risk for pregnancies. Nature Sustainability 2019;2(11):1011–7. | |
Butler LJ. The impact of meteorological factors and air pollution on adverse birth outcomes. Boston University, 2021. | |
Arbuckle TE, Sever LE. Pesticide exposures and fetal death: a review of the epidemiologic literature. Critical Reviews in Toxicology 1998;28(3):229–70. | |
Saraswat L, Bhattacharya S, Maheshwari A, et al. Maternal and perinatal outcome in women with threatened miscarriage in the first trimester: a systematic review. BJOG: An International Journal of Obstetrics & Gynaecology 2010;117(3):245–57. | |
Shapira E, Ratzon R, Shoham-Vardi I, et al. Primary vs. secondary recurrent pregnancy loss–epidemiological characteristics, etiology, and next pregnancy outcome. Journal of Perinatal Medicine 2012;40(4):389–96. | |
Bolor H, Mori T, Nishiyama S, et al. Mutations of the SYCP3 gene in women with recurrent pregnancy loss. The American Journal of Human Genetics 2009;84(1):14–20. | |
Guerri G, Maniscalchi T, Barati S, et al. Non-syndromic monogenic female infertility. Acta Bio Medica: Atenei Parmensis 2019;90(Suppl 10):68. | |
Farren J, Jalmbrant M, Falconieri N, et al. Post-traumatic stress, anxiety and depression following miscarriage and ectopic pregnancy: a multicenter, prospective, cohort study. American Journal of Obstetrics and Gynecology 2020;222(4):367.e1–22. | |
Coomarasamy A, Dhillon-Smith RK, Papadopoulou A, et al. Recurrent miscarriage: evidence to accelerate action. The Lancet 2021;397(10285):1675–82. | |
Di Renzo GC, Tosto V, Tsibizova V, et al. Prevention of Preterm Birth with Progesterone. J Clin Med 2021;10(19):4511. doi: 10.3390/jcm10194511. | |
Di Renzo GC. Preface. Best Pract Res Clin Obstet Gynaecol 2020;69:1. doi: 10.1016/j.bpobgyn.2020.10.008. | |
Di Renzo GC, Tosto V, Tsibizova V. Progesterone: History, facts, and artifacts. Best Pract Res Clin Obstet Gynaecol 2020;69:2–12. doi: 10.1016/j.bpobgyn.2020.07.012. | |
Di Renzo GC, Fonseca E. Effect of progestogen for women with threatened miscarriage: a systematic review and meta-analysis. BJOG 2020;127(10):1304–5. Doi: 10.1111/1471-0528.16370. | |
Coomarasamy A, Devall AJ, Brosens JJ, et al. Micronised vaginal progesterone to prevent miscarriage: a critical evaluation of randomised evidence. Am J Obstet Gynecol 2020;223(2):167–76. doi: 10.1016/j.ajog.2019.12.006. | |
van Oppenraaij RH, Jauniaux E, Christiansen OB, et al. ESHRE Special Interest Group for Early Pregnancy (SIGEP). Predicting adverse obstetric outcome after early pregnancy events and complications: a review. Hum Reprod Update 2009;15(4):409–21. doi: 10.1093/humupd/dmp009. | |
Henry A, Santulli P, Bourdon M, et al. Birth defects reporting and the use of oral dydrogesterone in assisted reproductive technology: a global pharmacovigilance study Abst 093–177 ESHRE 39th Intl Congress 2023, Copenhagen. | |
Devall AJ, Papadopoulou A, Podesek M, et al. Progestogens for preventing miscarriage: a network metanalysis. Cochrane Database of Systematic Reviews 2021;4:CD013792. DOI: 10.1002/14651858.CD013792.pub2. | |
Devall AJ, Melo P, Coomarasamy A. Progesterone for the prevention of threatened miscarriage. Obstetrics, Gynaecology & Reproductive Medicine 2022;32(3):44–7. | |
Rasmark Roepke E, Hellgren M, Hjertberg R, et al. Treatment efficacy for idiopathic recurrent pregnancy loss–a systematic review and meta‐analyses. Acta Obstetricia et Gynecologica Scandinavica 2018;97(8):921–41. | |
Dhillon‐Smith RK, Boelaert K, Jeve YB, et al. Subclinical hypothyroidism and antithyroid autoantibodies in women with subfertility or recurrent pregnancy loss: Scientific Impact Paper No. 70 June 2022. BJOG: An International Journal of Obstetrics & Gynaecology 2022;129(12):e75-e88. | |
Orio F, Vuolo L, Palomba S, et al. Metabolic and cardiovascular consequences of polycystic ovary syndrome. Minerva ginecologica 2008;60(1):39–51. | |
Kwak‐Kim J, Yang KM, Gilman‐Sachs A. Recurrent pregnancy loss: a disease of inflammation and coagulation. Journal of Obstetrics and Gynaecology Research 2009;35(4):609–22. | |
Wong LF, Porter TF, Scott JR. Immunotherapy for recurrent miscarriage. Cochrane Database of Systematic Reviews 2014(10). | |
Reddy AM, Iqbal M, Chopra H, et al. Pivotal role of vitamin D in mitochondrial health, cardiac function, and human reproduction. EXCLI Journal 2022;21:967. | |
Rikken J, Kowalik C, Emanuel M, et al. Septum resection versus expectant management in women with a septate uterus: an international multicentre open-label randomised controlled trial. Human Reproduction 2021;36(5):1260–7. | |
Saadaoui M, Singh P, Ortashi O, et al. Role of the vaginal microbiome in miscarriage: exploring the relationship. Front Cell Infect Microbiol 2023;13:1232825. doi: 10.3389/fcimb.2023.1232825. ECollection, 2023. |
Online Study Assessment Option
All readers who are qualified doctors or allied medical professionals can automatically receive 2 Continuing Professional Development points plus a Study Completion Certificate from GLOWM for successfully answering four multiple-choice questions (randomly selected) based on the study of this chapter. Medical students can receive the Study Completion Certificate only.
(To find out more about the Continuing Professional Development awards program CLICK HERE)
