This chapter should be cited as follows:
Townsend R, Khalil A, Glob Libr Women's Med
ISSN: 1756-2228; DOI 10.3843/GLOWM.412553
The Continuous Textbook of Women’s Medicine Series – Obstetrics Module
Volume 4
Fetal development and maternal adaptation
Volume Editor: Professor Asma Khalil, The Royal College of Obstetricians and Gynaecologists, London, UK; Fetal Medicine Unit, Department of Obstetrics and Gynaecology, St George’s University Hospitals NHS Foundation Trust, London, UK

Chapter
Multiple Pregnancy
First published: February 2021
Study Assessment Option
By answering four multiple-choice questions (randomly selected) after studying this chapter, readers can qualify for Continuing Professional Development points plus a Study Completion Certificate from GLOWM.
See end of chapter for details.
INTRODUCTION
Multiple pregnancies, an object of fascination from before the stories of Cain and Abel, and Romulus and Remus, still present a special challenge for the clinician today. There is wide international variation in twinning rates, largely driven by differences in the incidence of dizygotic (DZ) twin and multiple pregnancies. While the incidence of monozygotic (MZ) twin pregnancies is relatively constant at 3/1000, DZ twinning is increased by a number of factors including maternal age, parity, ethnicity, maternal family history of twins, and smoking. The highest rates of twinning are seen in Sub-Saharan Africa and the lowest frequency is reported in South East Asia and Latin America (Figure 1).1
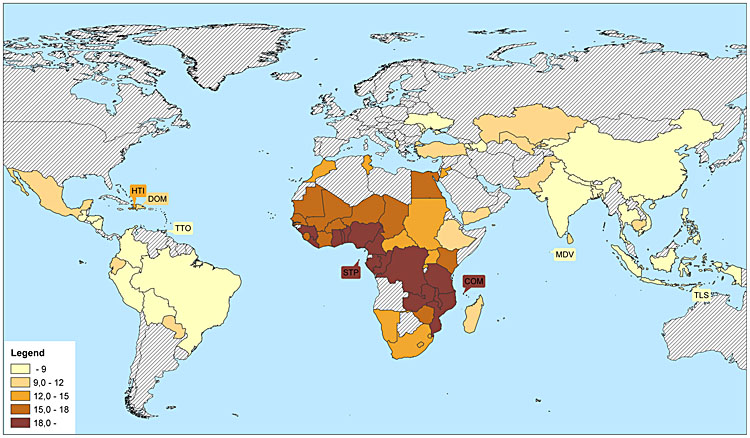
1
Twins per 1000 births in 76 low- and middle-income countries. Reproduced from Smits and Monden, 2011 under the Creative Commons Attribution License.1
The incidence of multiple births in the UK was 16 per 1000 total births in 2015 compared to 10 per 1000 in 1980.2,3. In the US the number of twins almost doubled between 1980 and 2009, increasing from 18.9 to 33.2 per 1000 births.4 The most significant factors behind this observed increase in frequency of multiple pregnancies are increasing maternal age and increasing use of artificial reproductive techniques. Although these have not yet impacted the global incidence of multiple pregnancy, as maternal health improves and artificial reproduction becomes more accessible the incidence of multiple pregnancy is likely to continue increasing globally.3,5
Multiple pregnancy is not as simple as two for the price of one – despite the fact that they constitute fewer than 2% of births, in the UK they contribute to 7% of stillbirths, 18% of neonatal deaths and have 6 times greater risk of cerebral palsy compared to singleton pregnancies.6 In this context, it is clear that all obstetricians need to appreciate the unique development of multiple pregnancies and the importance of specialized antenatal care in their management.
MATERNAL ADAPTATION AND MORBIDITY IN MULTIPLE PREGNANCY
The maternal physiological changes of pregnancy are unsurprisingly greater with multiple pregnancy as the mother adapts to accommodate the greater metabolic demands and physical size of multiple fetuses and placentas. Higher levels of placentally derived hormones can contribute to a higher frequency of hormonally mediated maternal morbidity. For example, human placental lactogen (hPL) is associated with gestational diabetes and is increased in multiple pregnancy, which correlates with the observed clinical increase in gestational diabetes in multiple pregnancy. Equally, increased human chorionic gonadotropin (hCG) is observed in multiple pregnancies and has been linked to an increased frequency of hyperemesis gravidarum.
Cardiovascular adaptation occurs as described in singleton pregnancy and is exaggerated in multiple pregnancy. Plasma volume expansion occurs to a greater degree, leading to reduced oncotic pressure with a higher frequency of both peripheral and pulmonary edema, and also a greater degree of dilutional anemia. By the time the pregnancy is full term, the weight of the gravid uterus in a twin pregnancy may exceed 8 kg and occupy 10 liters of space. The physical effects of this weight on the pelvis, bladder and back exacerbate the common complaints in pregnancy of pelvic girdle pain and urinary frequency.
PRE-ECLAMPSIA
Pre-eclampsia remains one of the most common complications of pregnancy and a significant cause of maternal morbidity and mortality globally. Pre-eclampsia and the other hypertensive disorders of pregnancy are more common in multiple pregnancies, with increasing incidence in higher order multiples. Early and atypical presentations of pre-eclampsia are more common in multiple pregnancy, with renal, hematological and hepatic disturbance more likely to prompt preterm delivery than severe hypertension or proteinuria in higher order multiples.7 It is possible that the exaggerated cardiovascular adaptation, including profound peripheral vasodilation, in multiple pregnancy protects against severe hypertension to some extent. Alternatively, it is possible to speculate that pre-eclampsia in multiple pregnancy is more likely to be immunologically mediated (with two or more foreign genetic profiles rather than one) and thus more likely to be associated with the autoimmune-like microangiopathy of HELLP (hemolysis, elevated liver enzymes and low platelets) syndrome.
There is currently considerable interest in screening for women at high risk of pre-eclampsia using a variety of prediction models. Uterine artery Doppler waveforms measured at 20–22 weeks have been found to be predictive of pre-eclampsia in low-risk singleton pregnancies and are incorporated in most available screening tests. Multiple studies have confirmed that despite the increased placental mass, the uterine artery pulsatility index (UAPI) is lower in twin than in singleton pregnancies.8,9 This supports the thesis that placental dysfunction in singleton pregnancy may be a distinct clinicopathological phenomenon to that observed in multiple pregnancy.
Screening for pre-eclampsia is most beneficial where a proven intervention exists, but the evidence of effective prevention of pre-eclampsia using low-dose aspirin following first trimester screening is limited to singleton pregnancies.10 Nonetheless, UK national guidelines recommend low dose aspirin (LDA) for mothers carrying twins with one additional risk factor for hypertension, which includes maternal age and primiparity.11 This results in the majority of mothers with twins being offered aspirin despite the lack of multiple pregnancy-specific information on its efficacy.
As in singleton pregnancies, the cure for pre-eclampsia is delivery and this should not be delayed where serious concerns about the maternal condition exist. (See the pre-eclampsia chapter for more information on the causes and management of pre-eclampsia)
PRETERM BIRTH IN MULTIPLE GESTATIONS
Multiple pregnancies are at a greater risk of preterm birth than singletons – in fact, 57% of twin pregnancies are delivered before 37 weeks and 11% before 32 weeks through a combination of increased rates of spontaneous preterm labor and a higher risk of antenatal maternal and fetal complications mandating scheduled preterm delivery. For triplet pregnancies, over 75% will be spontaneously delivered before 35 weeks and the majority of deliveries will be indicated by labor or maternal or fetal compromise. Scheduled delivery is the exception, not the rule, but where triplet pregnancies continue beyond 35 weeks, continuing the pregnancy further is associated with increasing risks of perinatal mortality and morbidity.3
The majority of the neonatal morbidity and mortality observed in multiples is attributable to prematurity,12 and effective prevention of spontaneous preterm delivery in multiples is a key target for reducing the perinatal burden associated with multiple pregnancy.
Cervical length screening is used in high-risk singleton pregnancies to identify women who might benefit from interventions to reduce preterm birth. Screening with cervical length at 18–22 weeks in multiple pregnancies is recommended by the Society of Obstetricians and Gynaecologists of Canada (SOGC)13 and the International Society of Ultrasound in Obstetrics and Gynecology (ISUOG)14 where the cut off used is <25 mm. Unfortunately, although cervical length is associated with preterm delivery in twins, the sensitivity is lower than in singleton pregnancies 2010,15,16 suggesting that the mechanism underlying preterm labor in multiple pregnancies may differ from singletons.
Interventions that have been shown to be of benefit in singleton pregnancies have not yet been proven to reduce preterm birth in multiple pregnancy. Cochrane systematic reviews have found that bed rest or progesterone or cervical cerclage are not associated with prolonged pregnancy in multiple gestations.18 Limited trials are available relating to the use of cervical support pessaries in multiple pregnancies, but meta-analysis suggests that these are of limited benefit even in women with a short cervix. There is suggestive meta-analysis evidence that progesterone and cerclage at least might have a role in women with a very short cervix,17,19 but trials in this area are lacking. Since cervical length is less sensitive for preterm birth in multiple pregnancy, interventions that are effective at reducing the risk of preterm delivery in pregnancies where the cervical length is normal are also needed.
FETAL DEVELOPMENT IN MULTIPLE PREGNANCY
In multiple pregnancy a number of features relating to the embryonic origins and development are important to the clinical outcome of the pregnancy. These primarily relate to the initiation of the twinning process – how and when separate embryos become established determines zygosity, chorionicity and amnionicity. These features in turn determine prognosis, management and clinical outcome.
Zygosity
A multiple pregnancy may arise from either superfecundation (fertilization of more than one ovum in the same cycle) or cleavage of a single fertilized ovum into multiple embryos. The former will be dizygous (genetically distinct) and represents two-thirds of all twin pregnancies, while the latter will be monozygous (genetically identical) and make up one-third of all twin pregnancies. Higher order multiple pregnancies may include both mono- and dizygous fetuses.
All dizygous (DZ) twins will develop independently and will be dichorionic (two placentas) and diamniotic (two membrane sacs), but monozygous (MZ) twins may share some structures. The degree of sharing depends on the timing of the embryo cleavage. Very early division, prior to development of the blastocyst (<3 days), occurs in around 25% of MZ pregnancies and will lead to separate placentas and membranes (dichorionic diamniotic (DCDA) in twins or trichorionic triamniotic (TCTA) in triplets). Division after the development of a single inner cell mass (day 4–8) will lead to a shared chorion, but separate membrane sacs (monochorionic diamniotic). It is important to recognize that although all monochorionic pregnancies should be monozygous, not all dichorionic pregnancies are dizygous (Figure 2).
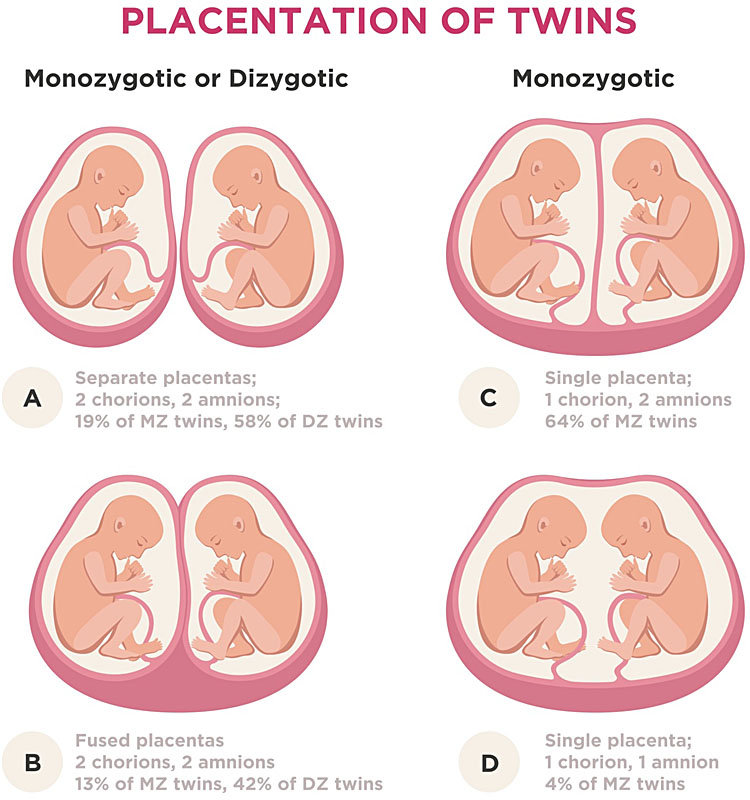
2
Zygosity and chorionicity in multiple pregnancy. DZ, dizygotic; MZ, monozygotic. From ID 125347863 © Andrii Bezvershenko | Dreamstime.com.
Later division of the embryonic plate is rare, but division between day 8 and 13 will lead to a shared placenta and membrane sac (monochorionic monoamniotic) which is observed in only 1% of all twin pregnancies. Division after 14 days leads to conjoined twins, with an estimated incidence of 1.5/100,000 births.
Zygosity has an important role in the clinical outcome of multiple pregnancy, but can only be conclusively demonstrated antenatally when discordant fetal sex is observed on ultrasound or invasive genetic testing is undertaken. Monozygous twins are at higher risk of congenital anomalies, but most of the increased perinatal risks of MZ pregnancies are related to the fact that most monozygous pregnancies are monochorionic: chorionicity is the most important determinant of outcome in multiple pregnancy.
Chorionicity
When the blastocyst divides, the developing embryos may share a single chorion or develop their own independently to become dichorionic (DC). As the chorion develops into the placenta, and the fetal circulation is established, a common chorion allows the development of anastamoses between the fetoplacental circulations of each developing fetus. The number, size and direction of these anastamoses determine the development of all fetuses and mediate the complications associated with monochorionic (MC) pregnancy.
Identifying chorionicity determines the frequency and type of surveillance that should be planned for the remainder of the pregnancy, allows counseling of the parents about the expected outcome of the pregnancy, determines which screening tests should be used, and guides the timing and mode of delivery. The risk of intrauterine fetal death (IUFD) is 11.6% in MC pregnancies compared to 5% in DC pregnancies.20 Moreover, following single intrauterine demise, the risk of IUFD and neurological damage is far greater to the surviving twin in MC than DC twin pregnancy.21 In addition, spontaneous as well as medically indicated preterm delivery and congenital abnormalities are all more common in MC pregnancies.22
The determination of chorionicity is straightforward when entirely distinct placental masses are observed and the pregnancy is clearly dichorionic (or trichorionic in the case of triplet pregnancies). Careful assessment is required in order to distinguish between DC and MC pregnancies where the placental masses appear to be fused.
Where there are two separate placentas, the membranes surrounding the twins will include four layers – the chorion and amnion of each. Where there is only one placenta but two membrane sacs each twin has their own amnion but are both enveloped in a single chorionic membrane. The thickness of the inter-twin membrane is thus affected by chorionicity and in monochorionic twins the membrane will have a thinner appearance on ultrasound. The thickness of the four layer intertwin membranes with chorionic villi invading the intermembrane potential space leads to the classical appearances of the ‘lambda’ sign (where the thicker membranes meet the ‘chorionic peak’ of the placentas) that identifies DC pregnancies in comparison to the ‘T sign’ where the thinner monochorionic diamniotic membranes meet the single placenta (Figure 3).
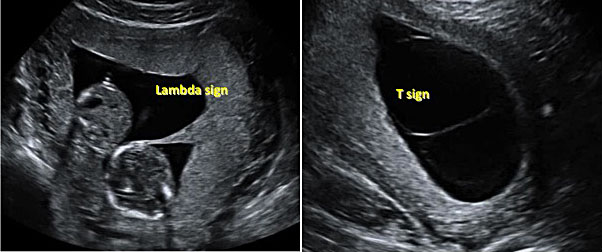
3
The ‘lambda’ and the ‘T’ signs to distinguish monochorionic from dichorionic pregnancies on first trimester ultrasound.
As the pregnancy progresses the chorionic peak becomes less pronounced and in the second trimester this feature is not as reliable for determination of chorionicity. Many of the ultrasound features used to determine chorionicity vary according to gestation; in general, the diagnosis is more accurate the earlier in pregnancy that the assessment is performed (Table 1). Ultrasound accuracy at less than 14 weeks has been reported at 99%, but the sensitivity for monochorionicity falls to only 77% after 14 weeks’ gestation.23 Ideally both chorionicity and amnionicity should be determined and documented before 14 weeks’ gestation.
1
Ultrasound indicators of chorionicity.
Gestation visible | Dichorionic diamniotic (DCDA) | Monochorionic diamniotic (MCDA) | Monochorionic monoamniotic (MCMA) |
<10 weeks | Two yolk sacs | Two yolk sacs | Single yolk sac |
<10 weeks | Two amniotic sacs | Two amniotic sacs | Single amniotic sac |
<10 weeks | Two gestational sacs | Two gestational sacs | Single gestational sac |
Up to 16–20 weeks | Chorionic peak ‘lambda sign’ | ‘T sign’ | No inter-twin membrane visible |
From 14–16 weeks | Discordant fetal sex | Concordant fetal sex is expected | Concordant fetal sex is expected |
Throughout pregnancy | Distinct placental masses | Single placental mass | Single placental mass |
PRACTICAL TIPS FOR CONFIRMING CHORIONICITY AND AMNIONICITY IN MULTIPLE PREGNANCY
Pitfalls in the assessment of chorionicity include:
- The possibility that monochorionic placentas may be bilobar and thus appear as two distinct masses – use color flow Doppler to look for placental anastamoses;
- The rare occurrence of dizygotic monochorionic pregnancies in which fetal sex discordance may mislead the clinician into diagnosing dichorionic pregnancy;
- The disappearance of the chorionic peak (the ‘lambda sign’) at later gestations.
Where chorionicity cannot be determined, it is safer to manage the pregnancy as monochorionic.
Differentiation of MCDA and MCMA twin pregnancies can be supported in early pregnancy by the identification of one or two yolk sacs, but in later pregnancy it relies on identification of the presence or absence of the inter-twin membrane. This is best confirmed by transvaginal scan. Another useful finding is the demonstration of cord entanglement, which is almost universal and pathognomic of monoamniotic twin pregnancies. Using pulsed wave Doppler, two distinct arterial waveform patterns with different heart rates are seen within the same sampling gate. This is known as the ‘galloping horse sign’.
FETAL GROWTH AND ANTENATAL MONITORING IN MULTIPLE PREGNANCY
Multiples do not simply grow as if they were entirely independent single fetuses, even when they have individual placentation. Although multiples and singletons seem to have similar growth patterns in the second trimester,24 by the third trimester growth velocity in multiples is consistently found to be less than in singletons, with the differences most pronounced and noted earlier in MC pregnancies and higher order multiple pregnancies. Estimated fetal weight (EFW) calculations are observed to be less accurate in twin pregnancies than in singletons, which calls into question the validity of using singleton norms for management of multiple pregnancies.25
The key question for clinicians to consider is does the difference in observed growth represent physiological adaptation or restriction? If growth in multiple pregnancy is limited by the ability of the mother to supply the metabolic demands of multiple growing fetuses, does that suggest the need for multiple-specific growth standards because they are physiologically distinct from singleton fetuses or represent a pathophysiological explanation for the observed increase in incidence of growth restriction and adverse perinatal outcomes in twins? If the former, then there is a need for multiple-specific fetal growth assessment, but if the latter then there is an argument to continue using singleton growth charts in order to avoid failing to diagnose growth restriction in multiple pregnancies.
Since it can be observed that multiple pregnancies are genuinely more at risk of stillbirth and perinatal loss than singletons, the finding that fetuses in a multiple pregnancy are smaller than singletons plausibly reflects an increased prevalence of true growth restriction in multiple pregnancies. There is, however, evidence to suggest that the use of twin-specific charts leads to fewer babies being diagnosed with FGR without failing to identify those small babies that go on to suffer IUFD or neonatal death.26,27,28,29 It seems that there is an element of physiological adaptation to a shared intrauterine environment, and growth parameters alone are insufficient to determine those babies most at risk of perinatal complications.
Caution should be used in assuming that twins are physiologically normal when smaller than the equivalently aged singletons until prospective trials of antenatal management using twin specific antenatal growth assessment are available. As in singleton pregnancies, the absolute size of the fetus is of less importance than the trend in growth and change in the Doppler parameters and these should be taken into consideration together with the clinical presentation and maternal history in making treatment decisions.
Placental vasculature in monochorionic pregnancies
In addition to placental development, total placental mass and relative placental share, the clinical outcome of MC pregnancies is determined as much, if not more, by the number and type of vascular anastomoses between the twins. Placental anastomoses can be artery to artery (AA), artery to vein (AV) or vein to vein (VV). AV anastomoses typically have unidirectional flow from one twin to another. VV anastomoses permit bidirectional flow and are observed with similar frequency (18–25%) in uncomplicated, twin to twin transfusion syndrome (TTTS) and selective fetal growth restriction (sFGR) MC pregnancies. AA anastomoses permit bidirectional flow at higher pressures, are present in >80% of uncomplicated MCDA pregnancies and can allow compensatory flow from the normally grown twin to compensate for placental insufficiency in sFGR and may be protective against TTTS by compensating for volume imbalances caused by unidirectional flow through AV anastomoses. AA anastamoses with larger diameters (>2 mm) can, however, also allow acute feto–fetal transfusion events.30 With many large AA anastomoses the larger fetus is vulnerable to sudden changes in the smaller twin’s blood pressure. Even without death of the smaller twin, transient periods of bradycardia and hypotension can lead to significant volume shifting and neurological damage to either or both twins.
Frequency of growth assessment
Most international bodies recommend scanning dichorionic (DC) twins every 4 weeks and monochorionic (MC) pregnancies every 2 weeks, on the basis that MC pregnancies are at greater risk of all adverse perinatal outcomes than DC twins (Figures 4 and 5).3,13,14,31 Longer scan intervals are likely to be associated with a more severe presentation at diagnosis of complications and consequently with poorer outcomes.32,33 Although additional tests are likely to identify problems earlier, there are significant resource implications for increased screening, particularly in DC pregnancies which represent the majority of twin clinic attendees. Clinicians must also weigh carefully the risk of missing a struggling fetus against the fact that additional screening, particularly with imperfect diagnostic tools, will inevitably lead to additional iatrogenic deliveries of babies suspected to be compromised. In multiple pregnancies these iatrogenic deliveries affect not only the mother but also any healthy but potentially immature siblings.
The majority of small babies are well and suffer no adverse fetal or neonatal outcomes and can safely be managed expectantly until maturity. Using appropriate tools to measure fetal growth and additional prognostic markers to identify the highest risk pregnancies can reduce the risk of iatrogenic harm. ISUOG recommends the assessment of the middle cerebral artery (MCA), umbilical artery doppler and fetal growth parameters at each antenatal ultrasound in the monitoring of multiple pregnancy (Figure 5).
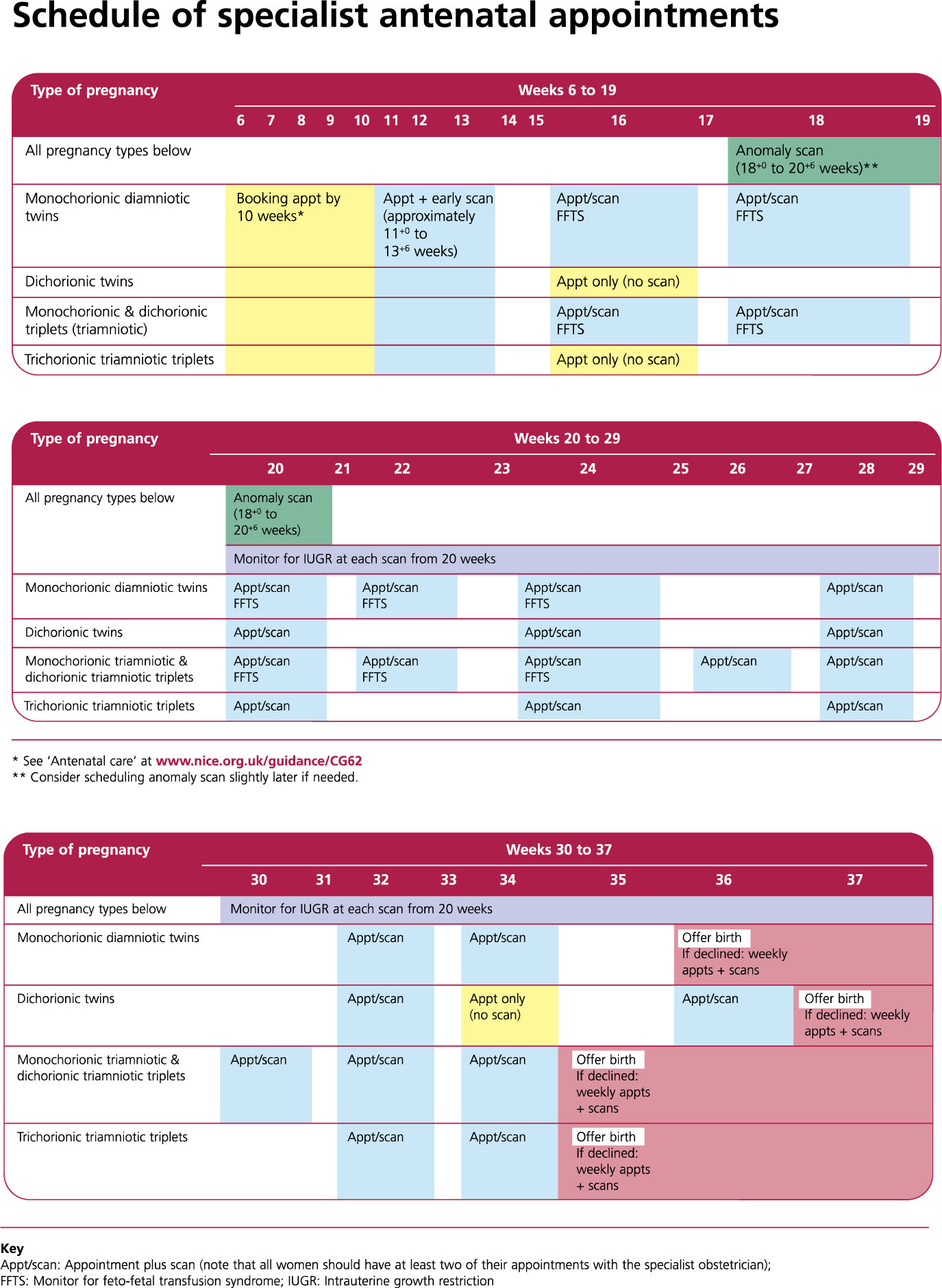
4
NICE antenatal monitoring of multiple pregnancy (from NICE CG129, Sept 2011).3
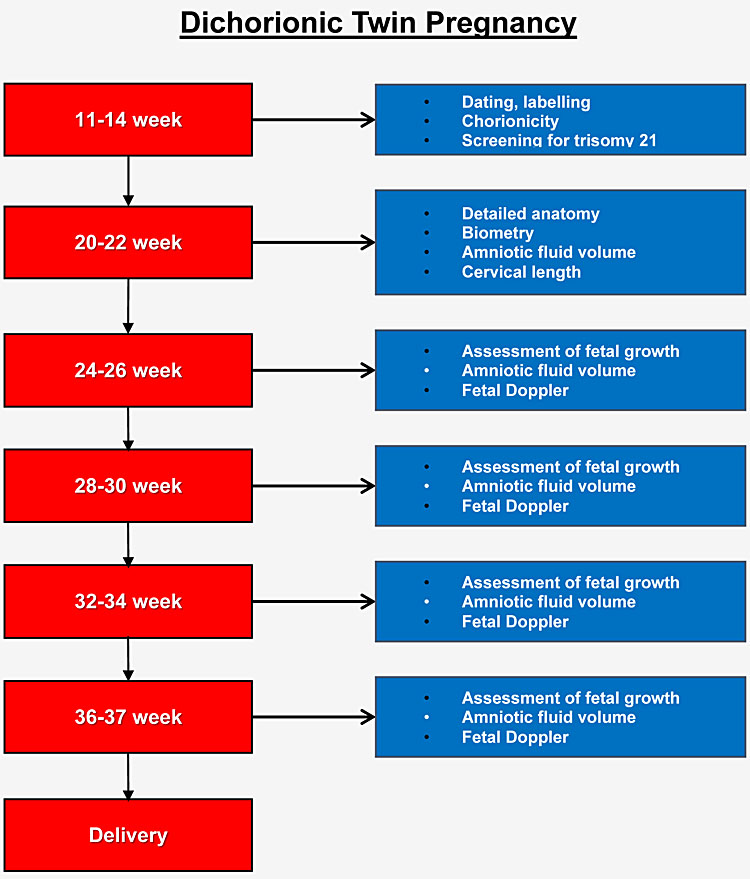
5a
ISUOG practice guideline: recommended schedule for antenatal ultrasound assessment of dichorionic twins.14

5b
ISUOG practice guideline: recommended schedule for antenatal ultrasound assessment of monochorionic twins.14
Twin labeling
Labeling of the twins begins at the first scan and should be consistent at every subsequent scan. The strategy must be applied by all professionals undertaking studies of the same pregnancy.14 It is good practice to describe each twin as fully as possible (e.g. twin 1 is female, on maternal left and has anterior placenta, while twin 2 is male, on maternal right and has a posterior placenta) to minimize the chance of confusion. It may be helpful to represent the twins relative positions diagrammatically in the maternal notes.
FIRST TRIMESTER MORBIDITY AND SCREENING IN MULTIPLE PREGNANCY
Miscarriage and ‘vanishing twins’
Multiple pregnancies have a higher rate of first trimester loss than singleton pregnancies. The whole pregnancy may miscarry, but the phenomenon of a ‘vanishing twin’ is also observed in nearly 1 in 5 twin pregnancies, where one embryo ceases to develop early in the first trimester.34 MC pregnancies are at higher risk of vanishing twins than DC pregnancies, and the incidence is even higher in higher order multiple pregnancies, occurring in 53% of triplets and 65% of quadruplets.35 The prognosis for the remaining fetus is good and these pregnancies should be managed as singletons where one fetus remains or as twins when one triplet has been lost.
Twin reversed arterial perfusion
Twin reversed arterial perfusion (TRAP) is a rare complication of MC pregnancy (<1% all recognized MC pregnancies) usually detected in the first trimester. In this pregnancy, an abnormal ‘acardiac’ twin with no heart or only rudimentary cardiac tissue is perfused by a normal ‘pump’ twin via superficial arterial anastamoses in the common placenta. The appearances on scan may mimic a ‘vanishing twin’ but continued growth in the acardiac mass indicates the occurrence of the TRAP sequence. This can lead to a hyperdynamic circulation and progressive high output cardiac failure in the pump twin, leading to demise of the healthy twin in up to 30% of these pregnancies by 18 weeks’ gestation.36
If TRAP is suspected, the patient will benefit from referral to a fetal medicine center with facilities to conduct invasive fetal interventions. In many cases the acardiac fetus spontaneously resolves, but where this does not occur the management options include cord coagulation, cord ligation and photocoagulation of the anastomoses, as well as intra-fetal methods such as radiofrequency ablation (RFA) and intrafetal laser therapy. The survival rate for the pump twin using these minimally invasive techniques is approximately 80%, and earlier intervention (<16 weeks' gestation) is likely to be better than later.37
Aneuploidy screening in twins
Dizygotic (DZ) pregnancies have two fetal chromosomal arrangements and two fetuses at risk of aneuploidy. The risk of aneuploidy affecting one or both fetuses is therefore higher than the risk of aneuploidy affecting a singleton pregnancy, with the estimated age related risk of a 33-year old carrying a DZ pregnancy being equivalent to that of a 35-year-old woman with a singleton pregnancy.38 National guidelines recommend assigning a risk ‘per fetus’ in DZ pregnancies, while MZ pregnancies are assumed by virtue of their common embryological origin to have a risk equivalent to the age-adjusted risk for an equivalent singleton pregnancy.3,14,31 In fact, the observed incidence of Down’s syndrome has been found to be lower, most significantly so in MZ pregnancies, where the observed-to-expected ratio is only 33.6%.39 The reasons for this finding, consistent over a number of studies,40 could include higher rates of pregnancy loss obscuring the true incidence of Down’s syndrome. This could also explain the finding of lower risk in MC pregnancies, where the fragility of the pregnancy is increased and pregnancy loss rates are higher than in DC pregnancies.
The combined screening test [nuchal translucency (NT), maternal serum beta-human chorionic gonadotropin (βhCG) and pregnancy-associated plasma protein-A (PAPP-A)] is the first line screening test for aneuploidy in singleton pregnancies. In singletons, the combined test has a reported detection rate (DR) for trisomy 21 of up to 90% for a 5% false positive rate (FPR).41 In twin pregnancies, the DR is reported to be 72–100% for a FPR of 5%.42 While rare, monozygotic pregnancies discordant for aneuploidy43 and dichorionic pregnancies with the appearance of monochorionic placentation have been reported,44 so high risk results of the combined test or non-invasive prenatal testing of cfFDNA require amniocentesis of both amniotic sacs and careful identification of each twin and sample to confirm a possible diagnosis of aneuploidy and identify the affected fetus.
Non-invasive prenatal testing in twins
Cell free fetal DNA (cffDNA) testing has recently been introduced into practice for aneuploidy screening in singleton pregnancies; DRs have been excellent (>99% for trisomy 21 with a FPR of <0.1%).45 Since twins have been thought to be associated both with an increased risk of aneuploidy and greater risks of pregnancy loss after invasive diagnostic testing, the advantages of non-invasive prenatal testing (NIPT) could potentially be even more significant in twin pregnancies than in singletons.
Theoretically in monozygotic pregnancies the fraction of fetal DNA should be greater and testing therefore more effective than in singletons, while in dizygotic pregnancies the presence of not one but two additional haplotypes can complicate the detection of aneuploidy. Although intuitively it would seem that each fetus would contribute equally to the free fraction of fetal DNA, it has been shown that, in fact, there can be wide variation in contribution to the free fetal fraction by each fetus. This limits screening performance – in the event where one fetus contributes <4% FF then a false negative result may be recorded.46 In addition, where a positive result is obtained, it is not possible to determine which fetus is affected. No studies large enough to accurately report the test performance in twins have yet been published, but the findings of a recent meta-analysis suggest that where a result is obtained the detection rates are similar to those in singleton pregnancies which are reportedly as high as 99.7% (95% CI 99.1–99.9%) and 0.04% (95% CI 0.02–0.08%) for trisomy 21.47
Structural anomalies
As with singleton pregnancies, detailed scans are recommended at 18–22 weeks’ gestation to identify structural anomalies in twin pregnancies.3,14 This is particularly important because there is an increased risk of congenital anomalies observed in twin pregnancies.48 Around 1 in 25 dichorionic diamniotic (DCDA) and 1 in 15 monochorionic diamniotic (MCDA) twin pairs will be affected by major congenital anomaly, usually affecting only one twin.49 The most commonly observed anomalies are renal and cardiac.50 Cardiac anomalies in particular are more common in MC pregnancies and therefore detailed examination of the heart is recommended in MC pregnancies.51 The embryological reason for the observed differences is unclear – it has been hypothesized that the twinning cleavage process in some manner disturbs laterality in at least one of the twins and predisposes to congenital anomaly, particularly in the heart.52
Since most anomalies only affect one twin, the problem of managing a pregnancy with only one affected fetus frequently arises. The management of discordant anomalies is dependent on chorionicity and the prognosis of the anomaly. Detailed fetal assessment, including MRI evaluation, may provide additional information on the nature of the anomaly and aid in counseling parents regarding the treatment options. Even with a high risk of intrauterine demise of the affected twin, expectant management is usually preferred in DC pregnancies. In MC pregnancies, the connected placental circulations put the healthy fetuses at risk of death and serious neurological morbidity in addition to the risk of preterm delivery in the event of the death of their sibling.21 Selective reduction where intra-uterine death of the affected twin is anticipated can protect the surviving fetuses.53 The remaining options are termination of the entire pregnancy or preterm scheduled delivery. The choice of management will depend on the nature of the anomaly, the facilities available for intrauterine interventions and postnatal care and the values and wishes of the family.
GROWTH RESTRICTION IN MULTIPLE PREGNANCY
Although not all small babies are truly growth restricted, the fetus that struggles to reach its full growth potential is at substantial risk of fetal and neonatal complications, even more so if not identified antenatally. In FGR in multiples, as in singleton pregnancies, simply being small for gestational age is not a guarantee of adverse perinatal outcomes. Like singletons, a large number of babies born small (<10th centile) suffer no adverse outcome, and a significant proportion of apparently appropriately grown twins demonstrate antenatal and neonatal sequelae of limited intrauterine development. The presence of interdependent fetal circulations in monochorionic pregnancies further complicates the underlying pathology of FGR and alters the presentation of pathology antenatally.
As with most pregnancy complications, the risk of FGR is increased in twin pregnancies, and more so in monochorionic (MC) twin pregnancies.1 Where one fetus is identified as small and the other is not, this is termed selective fetal growth restriction (sFGR). sFGR occurs in both DC and MC pregnancies but the shared placental circulation in MC pregnancies significantly affects presentation, progression and outcome.
Causes of fetal growth restriction in twins
Although FGR in multiple pregnancies is most often placentally mediated, other conditions that are associated with FGR in singleton pregnancies can also cause FGR in multiple pregnancies and should not be overlooked in the initial assessment of women referred with suspected FGR.
Intrauterine infections including parvovirus and cytomegalovirus (CMV) can selectively affect a single twin54,55 and the risk of both twins being affected does not seem to be related to the chorionicity.56 Equally, the first presentation of discordant anomaly may be the detection of growth restriction. For these reasons the clinical assessment of women presenting with fetal growth restriction in multiple pregnancy should include testing for infection and screening for chromosomal and structural anomalies.
Selective fetal growth restriction
Unlike singletons, multiples, especially monochorionic twins, come with a built in control. The difference between twins can help to discriminate between well small babies and pregnancies affected by growth restriction. Although twins are rarely identical in size, significant growth discrepancies are associated with poor perinatal outcomes in a continuous fashion and may be more important in relation to perinatal outcome than the absolute size of individual babies.57 An increase in perinatal adverse outcomes can be observed when the fetal growth discrepancy is only 18%, even after the exclusion of TTTS pregnancies.58 Most national bodies recommend a discrepancy of 20–25% as a trigger for referral to fetal medicine experts or additional monitoring.14,59 Consensus diagnostic criteria for selective fetal growth restriction in twin pregnancies has recently been established (Figure 6).60

6
Consensus diagnostic criteria for selective fetal growth restriction in twins.
While sFGR in DC pregnancies is similar to placental insufficiency in singleton pregnancies, sFGR in MC pregnancies relates not to placental dysfunction but primarily to discordance in placental share. The greater the discrepancy in placental share in MC pregnancies, the greater the growth discordance that should be observed. This is consistent with the observation that abnormal cord insertion (marginal or velamentous) is associated with sFGR, since laterally inserted cords are naturally associated with less equitable division of placental territory.61
Diagnosis and monitoring of multiple pregnancies affected by selective fetal growth restriction
In DC pregnancy the timing of delivery will be determined by evidence of deteriorating placental dysfunction and fetal compensation as assessed by the umbilical artery, middle cerebral artery (MCA) and ductus venosus (DV) Dopplers in accordance with the monitoring used in singleton pregnancies.
In an MC pregnancy the UA flow in the smaller twin is significantly affected by the hemodynamics of the larger twin and the compensatory effect of the co-twin’s support via bidirectional AA anastamoses may delay or even fully prevent deterioration in the smaller twin. Ultrasound assessment of MC pregnancies affected by sFGR should include assessment of placental structure and vasculature in order to inform assessment of the severity and prognosis of the condition. Cord insertion should be identified, placental anastamoses can be mapped using color Doppler and the degree of the compensatory effect of the larger twins circulation on the smaller twin can be documented in assessing the umbilical artery end diastolic blood flow.
Although the UA Doppler findings do not predict progressive deterioration as they do in singleton and DC pregnancies, the UA end diastolic flow (EDF) observed at diagnosis is still associated with the type of anastomoses connecting the fetal circulations and thus with the probability of compensatory supply from the larger twin, acute feto-fetal transfusion events and sudden intra-uterine demise.
The presence or absence of UA EDF in the affected twin at the time of diagnosis forms the basis of the classification system devised by Gratacos et al. (outlined in Table 2).62 Positive EDF is classified as type I and absent or reversed EDF (AREDF) as type II. The ‘intermittent AREDF’ unique to MC pregnancies is classified as type III (Figure 7). In this group, the presence of large diameter AA anastomoses permits a cyclical compensatory flow from the normal twin that can be observed in cyclical normalization in the EDF in the small twin, promoting longer survival of the small twin but also facilitating acute transfusion events that can lead to unexpected fetal death or neurological damage.
2
The classification, placental anatomy and pregnancy outcomes of selective fetal growth restriction in monochorionic twin pregnancy.
Umbilical artery Doppler findings in the smaller twin62 | Corresponding placental vascular findings | Perinatal mortality65 | |
Type I | Normal end-diastolic flow (EDF) | Similar to uncomplicated cases | 4.1% (95% CI 1.2–8.7) |
Type II | Absent or reversed EDF (AREDF) | Few large AA anastomoses | 16.1% (95%CI 4.6–32.7) |
Type III | Intermittently absent or reversed EDF (iAREDF) | Many large diameter AA anastamoses | 11.5% (95%CI 7.7–16.0) |
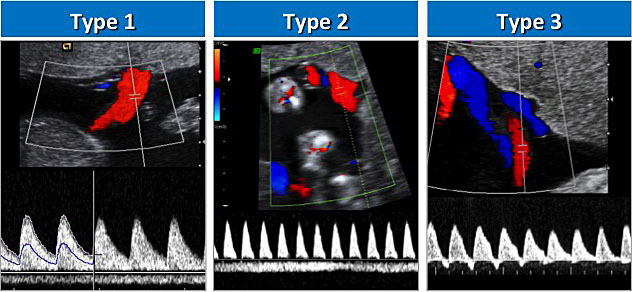
7
Umbilical artery Doppler in selective fetal growth restriction.
Management of selective fetal growth restriction
While managing sFGR in DC pregnancies, clinicians must factor in the additional consideration that delivery may cause iatrogenic harm to the normally grown fetus. In extremely preterm gestations parents may choose to continue the pregnancy in the expectation of losing the smaller twin but allowing the larger twin to continue to a later gestation. Since the fetal circulations are independent, single intrauterine fetal demise is associated with a lower risk of complications in the larger babies compared to single intrauterine demise in monochorionic placentation. In MC pregnancies demise of one twin is associated with a 15% risk of death and 25% risk of neurodevelopmental impairment in the co-twin.21
Type I sFGR has an excellent prognosis and elective delivery at 34–36 weeks is usually safely possible. Although most type I cases do remain stable, recent reports suggest that progression may occur in 11–26% of cases,63,64,65 underlining the importance of regular ultrasound surveillance even in this group.
In type II sFGR between 60 and 90% of cases can be expected to deteriorate and intact survival occurs in only 37%.63,64,65 Most studies describing outcomes in sFGR are small and retrospective, but meta-analysis suggests that perinatal mortality and neurological morbidity are highest in type II sFGR.65 While perinatal mortality in type III sFGR is comparable or less than in type II, it is more difficult to predict acute events, and the incidence of unexpected fetal demise with attendant neurological injury or death is likely to be higher.64,65 For type II or Type III sFGR, optimal management should be individualized to the gestation at diagnosis, severity of ultrasound findings, parental wishes and locally available treatments. Options include expectant management with delivery if and when fetal deterioration is detected, selective reduction of the smaller twin in order to pre-empt an intrauterine demise that also damages the larger twin or fetoscopic laser separation of the placental circulations. Laser reduces the risk of sequelae for the larger twin after death of the smaller co-twin but also removes from the smaller baby the beneficial effects of the placental circulation and may in fact precipitate fetal demise.
COMPLICATIONS SPECIFIC TO MONOCHORIONIC PREGNANCIES
MC twins are vulnerable to complications of interdependent placental circulations in a way that DC pregnancies are not. These complications include twin to twin transfusion syndrome (TTTS), selective fetal growth restriction (sFGR), twin anemia-polycythemia sequence (TAPS) and twin reversed arterial perfusion sequence (TRAP). As we have already described in sFGR, the presence and type of placental anastamoses between the fetal circulations are key to the pathophysiology and prognosis of these conditions. Although these complications carry a heavy burden of fetal morbidity and mortality, ultrasound screening can facilitate interventions and delivery to improve perinatal outcomes.
Twin to twin transfusion syndrome
Twin to twin transfusion syndrome (TTTS) is the most common serious complication of monochorionc pregnancies, occurring in 10–15% of MCDA pregnancies. TTTS accounts for more than one-third of all perinatal deaths in MC pregnancies.20 TTTS is a volume discrepancy between monochorionic twins created by unidirectional flow through AV anastamoses and not relieved by bidirectional flow through AA anastamoses. The placental anastamoses are unbalanced, preferentially allowing flow from one fetus (the ‘donor’) to the other (the ‘recipient’). In higher order pregnancies with monochorionic placentation it is possible to observe multiple recipients or donors. The donor twin is underperfused while the recipient suffers the effects of volume overload. The donor develops oligohydramnios and is often growth restricted, while the recipient twin develops polyhydramnios and may show evidence of hydrops.
TTTS is diagnosed when a significant amniotic fluid volume discrepancy (deepest vertical pocket [DVP] >8 cm in one twin and <2 cm in the other) is observed in MC pregnancies. Staging of TTTS is according to the Quintero classification (Table 3), but the Quintero stage is only partially associated with prognosis.66 Twin pairs may progress very rapidly from stage I to V without passing through the other stages, or remain stable at stage I or II for a prolonged period of time.67 Additional ultrasound markers can be used to guide prognosis and risk of progression, particularly in cases where difficult decisions regarding fetal interventions (fetoscopic laser coagulation or amnioreduction) or delivery are being weighed.
4
Quintero staging of twin to twin transfusion syndrome.
Stage | Description |
I | Discordant amniotic fluid volumes – DVP <2 cm in one and >8 cm in the other (before 20 weeks) or >10 cm after 20 weeks |
II | Bladder of donor twin not visible |
III | Critically abnormal Doppler studies in either twin (umbilical artery Doppler in the donor and/or venous Doppler in the recipient) |
IV | Ascites, pericardial or pleural effusion or overt hydrops |
V | Single or double intrauterine death |
Since the advent of fetoscopic laser coagulation, intervention can ameliorate the previously dismal prognosis in MC pregnancies affected by TTTS. Survival of at least one twin at 6 months is significantly improved after laser at 76% compared to 51% in those treated with amnioreduction alone and around 20% in pregnancies managed expectantly. Laser is also associated with improved neurological outcomes in the surviving fetuses.68 Fetoscopic interventions are limited to high volume, well resourced units. Where laser is not available, the management options are amnioreduction, termination of pregnancy or preterm delivery. Amnioreduction is inferior to fetoscopic laser treatment, but may increase survival 2–3 fold compared to no intervention at all. It is thought to confer benefit by reducing uterine stretch and the risk of preterm labor associated with polyhydramnios as well as relieving pressure that may be occluding venous circulation in the placenta.
Twin anemia polycythemia sequence
Twin anemia polycythemia sequence (TAPS) is an imbalance in hemoglobin without a volume distribution disparity in MC twins. It is most commonly seen after fetoscopic laser coagulation for TTTS, but may also arise spontaneously where there are only very small AV placental anastamoses with few protective AA anastamoses.69 Because spontaneous TAPS is seen in only around 2–5% of MC pregnancies and optimal management is not actually known,70 routine screening in all MC pregnancies has not previously been recommended, except after fetoscopic laser coagulation for TTTS. The ISUOG guidance has now recommended routine monitoring of the MCA peak systolic velocity (PSV) from 20 weeks in order to detect TAPS and allow decisions to be made about management in a timely fashion.14
TAPS is diagnosed when there is a significant discrepancy between the twins’ MCA PSVs. A PSV >1.5 MoM (indicative of anemia) associated with an MCA PSV in the other twin of <1 MoM (indicative of polycythemia) is required for the diagnosis of TAPS.70 The combination of the degree of MCA PSV discrepancy and abnormal fetal Dopplers or hydrops has been used to generate a staging system that can guide intervention and postnatal expectations (Table 4).70
4
Staging of twin anemia polycythemia sequence (TAPS).
Stage | Antenatal | Postnatal |
I | MCA-PSV donor >1.5 MoM and MCA-PSV recipient <1.0 MoM, without other signs of fetal compromise | Intertwin Hb difference (g/dL) >8.0 |
II | MCA-PSV donor >1.7 MoM and MCA-PSV recipient <0.8 MoM, without other signs of fetal compromise | >11.0 |
III | Stage 1 or 2 changes in MCA-PSV, with cardiac compromise of donor | >14.0 |
IV | Hydrops in donor | >17.0 |
V | IUFD of either fetus in a pregnancy known to be affected by TAPS | >20.0 |
MoM, multiples of the median; IUFD, intrauterine fetal demise; MCA, middle cerebral artery; PSV, peak systolic velocity.
The optimal management of TAPS is yet to be determined. The available management options include in-utero transfusion, which carries a risk of worsening the polycythemia in the recipient twin, fetoscopic laser coagulation or early delivery after a course of steroids.
Monoamniotic pregnancies
In 5% of MC pregnancies the fetuses are also monoamniotic. This is usually identified at the time of the first scan. Monoamnionicity can also be an iatrogenic complication of fetal intervention during the pregnancy, particularly after laser for TTTS. Sharing the amniotic sac presents additional fetal risks and historically very high perinatal mortality was associated with monoamniotic pregnancy, although with intensive antenatal monitoring, steroid use and early identification of fetal anomalies the perinatal mortality after 24 weeks may now be as low as 4.5%.71
Intrauterine demise is usually thought to be due to cord accidents, but cord entanglement is nearly universal in MCMA pregnancies. The most significant difference between monoamniotic and other multiple pregnancies is the near universal presence of large AA anastamoses between the fetal circulations in MCMA pregnancies, which potentiates large volume shifts between the fetuses. These sudden and often unpredictable shifts may also be the cause of many intrauterine complications in normally formed MCMA fetuses. A further significant proportion of intrauterine death in MCMA pregnancies is related to the high incidence of fetal anomalies in these twins.
In spontaneous MCMA pregnancies the large AA anastamoses protect against TTTS, which only occurs in 3% of MCMA pregnancies. Because the sac is shared, TTTS should be suspected when polyhydramnios is observed with a small or empty bladder in one twin and a full bladder in the other. MCMA pregnancies also seem to be at a lower risk of FGR, a finding that has also been attributed to the protective effects of AA anastamoses.
Although large prospective studies have not been possible in monoamniotic pregnancies, a large retrospective review of nearly 200 cases found that the risk of stillbirth is higher than the risk of neonatal mortality and morbidity after 32 weeks.72 Most experts recommend elective cesarean delivery after the administration of steroids at 32–34 weeks, but the role of inpatient admission, ultrasound surveillance and cardiotocography in monitoring these pregnancies remains controversial.
Conjoined twins
The rarest complication of monochorionic pregnancy is conjoined twins, a condition resulting from very late splitting of the blastocyst and occurring in only 1% of monochorionic twin pregnancies.14 The majority of conjoined twins are female. Advances in ultrasound mean that conjoined twins are most commonly identified in the first trimester when many parents will opt for termination of pregnancy in view of the high risk of morbidity and mortality in an ongoing pregnancy. In families choosing to continue pregnancies, around 25% would be expected to survive to discharge and almost all with significant morbidity.73 The prognosis is ultimately determined by the degree and site of the junction between the twins, and therefore detailed ultrasound studies are necessary to fully explore the nature of the connections between the twin pair. The most common site of union is at the thorax with the twins facing each other, and bowels, liver and hearts may be shared. Mapping blood vessels and structures can help plan postnatal surgery – where delivery is planned, it should be by cesarean section in a unit equipped to meet the surgical needs of the babies.
TWIN CLINICS AND ANTENATAL CARE FOR MULTIPLE PREGNANCY
The use of dedicated clinics staffed by obstetricians and midwives with expertise in the management of multiple pregnancy has been associated with reduced preterm birth, fetal growth restriction and perinatal mortality.74 This is in keeping with the finding that continuity of carer improves pregnancy outcomes in general, but there is no doubt that families with higher risk pregnancies particularly benefit from the sustained involvement of experts from early pregnancy. Example schedules for antenatal visits are described in Figures 4 and 5, and the UK-based Twin and Multiple Birth Association (TAMBA) has produced a detailed multiple pregnancy antenatal care pathway that reflects UK national guidelines for management of multiple pregnancy, available online as supporting material for NICE Clinical Guideline 129 “Multiple pregnancy: antenatal care for twin and triplet pregnancies” at https://www.nice.org.uk/guidance/cg129.
Although ultrasound is key to monitoring of multiple pregnancy, it is also important not to lose sight of holistic clinical assessment and management of the mother and her babies. Women should be advised to take high dose folic acid in early pregnancy and should follow local protocols relating to iron supplementation (if anemia is detected) and malaria prophylaxis. Women with risk factors for gestational diabetes should still receive screening by the oral glucose tolerance test (OGTT) in accordance with local guidelines. Where women with a multiple pregnancy do develop gestational diabetes clear communication between the maternal medicine and multiple pregnancy clinicians is important to ensure that the full clinical picture is taken into account when planning interventions and delivery. Blood pressure and proteinuria must be tested at every antenatal encounter and women must be well informed about the signs and symptoms of pre-eclampsia that should prompt self referral to clinical services. In many countries specialist peer support is available for families navigating the challenges of preparing for the birth of multiples, for example TAMBA offers in person, online and phone support to parents of multiples in the UK.
ANTENATAL CARE OF MULTIPLE PREGNANCY IN LOW RESOURCE SETTINGS
The regular use of ultrasound assessment is fundamental to the optimal management of multiple pregnancy, although not feasible in all settings. In particular first trimester ultrasound is necessary to diagnose the presence of multiple pregnancy and permit the assessment of the gestational age, number of fetuses, chorionicity and amnionicity of the pregnancy. Even in later pregnancy, ultrasound monitoring requires skilled operators with experience in handling the detailed assessment of multiple fetuses.
Where multiple pregnancy is suspected on abdominal palpation in later pregnancy, every effort to obtain ultrasound confirmation should be made. Palpation may determine the lie of the presenting fetus but cannot determine the number or chorionicity of the fetuses present. It may be possible to determine chorionicity even in late pregnancy using the site of the placentae or by identifying discordant fetal sex. Where chorionicity cannot be determined, the pregnancy should be managed as if monochorionic.
Women carrying a multiple pregnancy should be encouraged to seek care at a facility equipped to deal with preterm infants and provide comprehensive emergency obstetric care (CEmOC) because of the fetal and maternal risks of twin delivery. (The intrapartum management of multiple pregnancy is discussed in detail later in this textbook.)
CONCLUSION
The management of multiple pregnancy requires the input of skilled sonographers, midwives and obstetricians from the very beginning of pregnancy. Although multiple pregnancies may come with more than ‘double trouble’, with appropriate monitoring and timely intervention a good outcome can be achieved in the majority of cases. All obstetricians should familiarize themselves with the normal development of multiple pregnancies in order to identify and support those pregnancies that develop maternal or fetal complications.
PRACTICE RECOMMENDATIONS
- Early identification of gestation, number of fetuses and chorionicity is key to the management of multiple pregnancy. Fetuses should be clearly labeled and described at each ultrasound.
- Specialized multiple pregnancy clinics providing continuity of care have proven clinical benefit and should be the norm in caring for multiple pregnancies.
- Regular ultrasound assessment is the key to detection and management of fetal complications, but is only of use in management of complicated monochorionic pregnancies when accompanied by a clear understanding of placental vasculature and the physiological implications for both fetuses.
- Do not lose focus on the mother – remember that maternal death is more common in multiple pregnancy and monitor for the development of pre-eclampsia, gestational diabetes and other obstetric complications, remembering that presentation may be atypical.
CONFLICTS OF INTEREST
The author(s) of this chapter declare that they have no interests that conflict with the contents of the chapter.
Feedback
Publishers’ note: We are constantly trying to update and enhance chapters in this Series. So if you have any constructive comments about this chapter please provide them to us by selecting the "Your Feedback" link in the left-hand column.
REFERENCES
Smits J, Monden C. Twinning across the Developing World. Newell M-L (ed.) PLoS One Public Library of Science; 2011;6(9):e25239. | |
Smith LK, Manktelow BN, Draper ES, et al. Trends in the incidence and mortality of multiple births by socioeconomic deprivation and maternal age in England: population-based cohort study. BMJ Open 2014;4(e004514). | |
NICE. CG 129: Multiple pregnancy : antenatal care for twin and triplet pregnancies. National Institute for Health and Care Excellence – Guidance and Guidelines, 2011. | |
Martin J, Hamilton B, Osterm. Three decades of twin births in the United States, 1980–2009. NCHS Data Brief 2012;(80):1–8. | |
Martin JA, Hamilton BE, Osterman MJKS, et al. Births: Final Data for 2015. Natl Vital Stat Reports 2017;66(1). | |
Scher AI, Petterson B, Blair E, et al. The Risk of Mortality or Cerebral Palsy in Twins: A Collaborative Population-Based Study. Pediatr Res Nature Publishing Group; 2002;52(5):671–81. | |
Hardardottir H, Kelly K, Bork MD, et al. Atypical presentation of preeclampsia in high-order multifetal gestations. Obstet Gynecol 1996;87(3):370–4. | |
Yu CKH, Papageorghiou AT, Boli A, et al. Screening for pre-eclampsia and fetal growth restriction in twin pregnancies at 23 weeks of gestation by transvaginal uterine artery Doppler. Ultrasound Obstet Gynecol 2002;20(6):535–40. | |
Rizzo G, Pietrolucci ME, Aiello E, et al. Uterine artery Doppler evaluation in twin pregnancies at 11 + 0 to 13 + 6 weeks of gestation. Ultrasound Obstet Gynecol 2014;44(5):557–61. | |
Rolnik DL, Wright D, Poon LC, et al. Aspirin versus Placebo in Pregnancies at High Risk for Preterm Preeclampsia. N Engl J Med Massachusetts Medical Society; 2017;377(7):613–22. | |
NICE. Hypertension in pregnancy. Guidelines. NICE; 2010;CG 107. | |
Blondel B, Macfarlane A, Gissler M, Breart G, Zeitlin J, PERISTAT Study Group. General obstetrics: Preterm birth and multiple pregnancy in European countries participating in the PERISTAT project. BJOG An Int J Obstet Gynaecol 2006;113(5):528–35. | |
Morin L, Lim K, Bly S, et al. Ultrasound in Twin Pregnancies. J Obstet Gynaecol Canada Elsevier Masson SAS; 2011;33(6):643–56. | |
ISUOG. ISUOG Practice Guidelines: role of ultrasound in twin pregnancy. Ultrasound Obstet Gynecol 2016;47:247–63. | |
Lim K, Butt K, Crane JM. SOGC Clinical Practice Guideline. Ultrasonographic cervical length assessment in predicting preterm birth in singleton pregnancies. J Obstet Gynaecol Can 2011;33(5):486–99. | |
Conde-Agudelo A, Romero R, Hassan SS, et al. Transvaginal sonographic cervical length for the prediction of spontaneous preterm birth in twin pregnancies: a systematic review and metaanalysis. Am J Obstet Gynecol 2010;203(2):128.e1–128.e12. | |
Romero R, Conde-Agudelo A, El-Refaie W, et al. Vaginal progesterone decreases preterm birth and neonatal morbidity and mortality in women with a twin gestation and a short cervix: an updated meta-analysis of individual patient data. Ultrasound Obstet Gynecol Wiley-Blackwell, 2017;49(3):303–14. | |
da Silva Lopes K, Takemoto Y, Ota E, et al. Bed rest with and without hospitalisation in multiple pregnancy for improving perinatal outcomes. Cochrane Database Syst Rev John Wiley & Sons, Ltd, 2017;(3). | |
Li C, Shen J, Hua K. Cerclage for women with twin pregnancies: a systematic review and metaanalysis. Am J Obstet Gynecol Elsevier Inc.; 2019; | |
Hack K, Derks J, Elias S, et al. Increased perinatal mortality and morbidity in monochorionic versus dichorionic twin pregnancies: clinical implications of a large Dutch cohort study. BJOG An Int J Obstet Gynaecol Blackwell Publishing Ltd, 2007;115(1):58–67. | |
Hillman SC, Morris RK, Kilby MD. Co-twin prognosis after single fetal death: a systematic review and meta-analysis. Obstet Gynecol 2011;118(4):928–40. | |
Kenneth J. Moise JM, Pedro S. Argotti M. The importance of determining chorionicity in twin gestations. Contemp Ob Gyn Advanstar Communications Inc, 2013. | |
Stenhouse E, Hardwick C, Maharaj S, et al. Chorionicity determination in twin pregnancies: how accurate are we? Ultrasound Obstet Gynecol 2002;19(4):350–2. | |
Chervenak FA, Skupski DW, Romero R, et al. How accurate is fetal biometry in the assessment of fetal age? Am J Obstet Gynecol 1998;178(4):678–87. | |
Khalil A, D’Antonio F, Dias T, Cooper D, Thilaganathan B, Southwest Thames Obstetric Research Collaborative (STORK). Ultrasound estimation of birth weight in twin pregnancy: comparison of biometry algorithms in the STORK multiple pregnancy cohort. Ultrasound Obstet Gynecol 2014;44(2):210–20. | |
Kalafat E, Sebghati M, Thilaganathan B, Khalil A, Southwest Thames Obstetric Research Collaborative (STORK). Predictive accuracy of the Southwest Thames Obstetric Research Collaborative (STORK) chorionicity-specific twin growth charts for stillbirth: a validation study. Ultrasound Obstet Gynecol 2018. | |
Stirrup OT, Khalil A, D’Antonio F, Thilaganathan B, Southwest Thames Obstetric Research Collaborative (STORK). Fetal growth reference ranges in twin pregnancy: analysis of the Southwest Thames Obstetric Research Collaborative (STORK) multiple pregnancy cohort. Ultrasound Obstet Gynecol 2015;45(3):301–7. | |
Gielen M, Lindsey PJ, Derom C et al. Twin-Specific Intrauterine ‘Growth’ Charts Based on Cross-Sectional Birthweight Data. Twin Res Hum Genet Cambridge University Press; 2008;11(02):224–35. | |
Odibo AO, Cahill AG, Goetzinger KR, et al. Customized growth charts for twin gestations to optimize identification of small-for-gestational age fetuses at risk of intrauterine fetal death. Ultrasound Obs Gynecol 2013;41:637–42. | |
Lewi L, Deprest J, Hecher K, et al. The vascular anastomoses in monochorionic twin pregnancies and their clinical consequences. Am J Obstet Gynecol Elsevier, 2013;208(1):19–30. | |
ACOG. Practice Bulletin 169: Multifetal gestations: Twin, triplet and higher-order multifetal pregnancies. Obstet Gynecol 2016;128(4):e131–46. | |
McDonald R, Hodges R, Knight M, et al. Optimal Interval between Ultrasound Scans for the Detection of Complications in Monochorionic Twins. Fetal Diagn Ther 2017;41(3):197–201. | |
Corcoran S, Breathnach F, Burke G, et al. Dichorionic twin ultrasound surveillance: Sonography every 4 weeks significantly underperforms sonography every 2 weeks: Results of the Prospective Multicenter ESPRiT Study. Am J Obstet Gynecol Elsevier Inc.; 2015;213(4):551.e1–551.e5. | |
Márton V, Zádori J, Kozinszky Z, et al. Prevalences and pregnancy outcome of vanishing twin pregnancies achieved by in vitro fertilization versus natural conception. Fertil Steril 2016;106(6):1399–406. | |
Dickey RP, Taylor SN, Lu PY, et al. Spontaneous reduction of multiple pregnancy: incidence and effect on outcome. Am J Obstet Gynecol 2002;186(1):77–83. | |
Lewi L, Valencia C, Gonzalez E, et al. The outcome of twin reversed arterial perfusion sequence diagnosed in the first trimester. Am J Obstet Gynecol 2010;203(3):213.e1–213.e4. | |
Tan TYT, Sepulveda W. Acardiac twin: a systematic review of minimally invasive treatment modalities. Ultrasound Obstet Gynecol 2003;22(4):409–19. | |
Rodis JF, Egan JF, Craffey A, et al. Calculated risk of chromosomal abnormalities in twin gestations. Obstet Gynecol 1990;76(6):1037–41. | |
Sparks TN, Norton ME, Flessel M, et al. Observed Rate of Down Syndrome in Twin Pregnancies. Obstet Gynecol 2016;128(5):1127–33. | |
Boyle B, Morris J, McConkey R, et al. Prevalence and risk of Down syndrome in monozygotic and dizygotic multiple pregnancies in Europe: implications for prenatal screening. BJOG An Int J Obstet Gynaecol 2014;121(7):809–20. | |
Nicolaides KH, Spencer K, Avgidou K, et al. Multicenter study of first-trimester screening for trisomy 21 in 75 821 pregnancies: results and estimation of the potential impact of individual risk-orientated two-stage first-trimester screening. Ultrasound Obstet Gynecol John Wiley & Sons, Ltd.; 2005;25(3):221–6. | |
Gagnon A, Audibert F, Titulaire P. Prenatal screening and diagnosis of aneuploidy in multiple pregnancies. Best Pract Res Clin Obstet Gynaecol 2014;28:285–94. | |
McFadden P, Smithson S, Massaro R, et al. . Monozygotic Twins Discordant for Trisomy 13. Pediatr Dev Pathol 2017;20(4):340–7. | |
Lu J, Cheng YKY, Ting YH, et al. Pitfalls in assessing chorioamnionicity: novel observations and literature review. Am J Obstet Gynecol Elsevier, 2018;0(0). | |
Royal College of Obstetricians and Gynaecologists. Non-invasive Prenatal Testing for Chromosomal Abnormality using Maternal Plasma DNA. Scientific Impact Paper 15. London; 2014. | |
Struble CA, Syngelaki A, Oliphant A, et al. Fetal Fraction Estimate in Twin Pregnancies Using Directed Cell-Free DNA Analysis. Fetal Diagn Ther 2014;35:199–203. | |
Gil M, Accurti V, Santacruz B, et al. Analysis of Cell-Free DNA in Maternal Blood in Screening For Aneuploidies: Updated Meta-Analysis. Ultrasound Obstet Gynecol 2017. | |
Hardin J, Carmichael SL, Selvin S, et al. Increased prevalence of cardiovascular defects among 56,709 California twin pairs. Am J Med Genet A NIH Public Access, 2009;149A(5):877–86. | |
Lewi L, Lewi P, Diemert A, et al. The role of ultrasound examination in the first trimester and at 16 weeks’ gestation to predict fetal complications in monochorionic diamniotic twin pregnancies. Am J Obstet Gynecol 2008;199(5):493.e1–493.e7. | |
D’Antonio F, Familiari A, Thilaganathan B, et al. Sensitivity of first-trimester ultrasound in the detection of congenital anomalies in twin pregnancies: population study and systematic review. Acta Obstet Gynecol Scand 2016;95(12):1359–67. | |
Carvalho J, Allan L, Chaoui R, et al. ISUOG Practice Guidelines (updated): sonographic screening examination of the fetal heart. Ultrasound Obstet Gynecol 2013;41(3):348–59. | |
Burn J, Corney G. Congenital heart defects and twinning. Acta Genet Med Gemellol (Roma) 1984;33(1):61–9. | |
Gaerty K, Greer RM, Kumar S. Systematic review and metaanalysis of perinatal outcomes after radiofrequency ablation and bipolar cord occlusion in monochorionic pregnancies. Am J Obstet Gynecol 2015;213(5):637–43. | |
Bekhit MT, Greenwood PA, Warren R, et al. In utero treatment of severe fetal anaemia due to parvovirus B19 in one fetus in a twin pregnancy–a case report and literature review. Fetal Diagn Ther Karger Publishers; 2009;25(1):153–7. | |
De la Calle M, Baquero F, Rodriguez R, et al. Successful treatment of intrauterine cytomegalovirus infection with an intraventricular cyst in a dichorionic diamniotic twin gestation using cytomegalovirus immunoglobulin. J Matern Neonatal Med 2017;1–4. | |
Yinon Y, Yagel S, Tepperberg-Dikawa M, et al. Prenatal diagnosis and outcome of congenital cytomegalovirus infection in twin pregnancies. BJOG An Int J Obstet Gynaecol 2006;113(3):295–300. | |
Harper LM, Weis MA, Odibo AO, et al. Significance of growth discordance in appropriately grown twins. Am J Obstet Gynecol Elsevier Inc.; 2013;208(5):1–5. | |
Breathnach FM, McAuliffe FM, Geary M, et al. Definition of Intertwin Birth Weight Discordance. Obstet Gynecol 2011;118(1):94–103. | |
NICE. Multiple pregnancy: antenatal care for twin and triplet pregnancies. Clinical Guideline CG129. NICE; 2011. | |
Khalil A, Beune I, Hecher K, et al. Consensus definition and essential reporting parameters of selective fetal growth restriction in twin pregnancy: a Delphi procedure. Ultrasound Obstet Gynecol 2018. | |
Kalafat E, Thilaganathan B, Papageorghiou A, et al. The Significance of Placental Cord Insertion Site in Twin Pregnancy. Ultrasound Obstet Gynecol 2017. | |
Gratacós E, Lewi L, Muñoz B, Acosta-Rojas R, et al. A classification system for selective intrauterine growth restriction in monochorionic pregnancies according to umbilical artery Doppler flow in the smaller twin. Ultrasound Obstet Gynecol 2007;30(1):28–34. | |
Rustico MA, Consonni D, Lanna M, et al. Selective intrauterine growth restriction in monochorionic twins: changing patterns in umbilical artery Doppler flow and outcomes. Ultrasound Obstet Gynecol 2016. | |
Ishii K, Murakoshi T, Takahashi Y, et al. Perinatal Outcome of Monochorionic Twins with Selective Intrauterine Growth Restriction and Different Types of Umbilical Artery Doppler under. Fetal Diagn Ther 2009;26:157–61. | |
Buca D, Pagani G, Rizzo G, et al. Outcome in monochorionic twin pregnancies with selective intrauterine growth restriction according to the umbilical artery Doppler pattern of the smaller twin: a systematic review and meta-analysis. Ultrasound Obstet Gynecol 2017;50(5):559–68. | |
Khalil A, Cooper E, Townsend R, Thilaganathan B. Evolution of Stage 1 Twin-to-Twin Transfusion Syndrome (TTTS): Systematic Review and Meta-Analysis. Twin Res Hum Genet 2016;19(03):207–16. | |
Duryea EL, Happe SK, McIntire DD, et al. The natural history of twin–twin transfusion syndrome stratified by Quintero stage*. J Matern Neonatal Med 2016;29(21):1–5. | |
Senat M-V, Deprest J, Boulvain M, et al. Endoscopic Laser Surgery versus Serial Amnioreduction for Severe Twin-to-Twin Transfusion Syndrome. N Engl J Med 2004;351(2):136–44. | |
Slaghekke F, Oepkes D. Solomon Technique Versus Selective Coagulation for Twin–Twin Transfusion Syndrome. Twin Res Hum Genet 2016;19(03):217–21. | |
Slaghekke F, Kist WJ, Oepkes D, et al. Twin anemia-polycythemia sequence: Diagnostic criteria, classification, perinatal management and outcome. Fetal Diagn Ther 2010;27(4):181–90. | |
Prefumo F, Fichera A, Pagani G, et al. The natural history of monoamniotic twin pregnancies: a case series and systematic review of the literature. Prenat Diagn 2015;35(3):274–80. | |
Van Mieghem T, De Heus R, Lewi L, et al. Prenatal Management of Monoamniotic Twin Pregnancies. Obstet Gynecol 2014;124(3):498–506. | |
Baken L, Rousian M, Kompanje EJO, et al. Diagnostic techniques and criteria for first-trimester conjoined twin documentation: a review of the literature illustrated by three recent cases. Obstet Gynecol Surv 2013;68(11):743–52. | |
Luke B, Brown MB, Misiunas R, et al. Specialized prenatal care and maternal and infant outcomes in twin pregnancy. Am J Obstet Gynecol 2003;189(4):934–8. |
Online Study Assessment Option
All readers who are qualified doctors or allied medical professionals can automatically receive 2 Continuing Professional Development points plus a Study Completion Certificate from GLOWM for successfully answering four multiple-choice questions (randomly selected) based on the study of this chapter. Medical students can receive the Study Completion Certificate only.
(To find out more about the Continuing Professional Development awards program CLICK HERE)
