Antibiotics in Maternal-Fetal Medicine
Authors
INTRODUCTION
In the middle of May 1847, Semmelweis1 introduced washing the hands of medical students with “chlorine liquida” after their handling of cadaveric material and before their examinations of laboring women. The results were dramatic and highlighted the importance of infection in obstetrics and the crucial role of hygiene and antibiotics in obstetrics. In the 17 months before June 1, 1847, there were 5486 births and 576 (10%) maternal deaths at First Clinic in Vienna, Austria. In the subsequent 19 months after the initiation of chlorine washing, there were 5397 births and 101 (1.9%) maternal deaths. These observations heralded the realization that infection, along with hemorrhage and hypertension, is one of the three major causes of maternal death. Today, maternal mortality is measured in number of maternal deaths per 100,000 births rather than number per 100 births. The introduction of hygiene by Semmelweis and the subsequent development of antibiotics in mid-twentieth century led to this success story. Despite the successes, the pregnant woman and her fetus remain challenged by microbes and the antibiotics that treat them.
More than one third of pregnant women receive antibiotics for urinary tract infections, group B streptococcus (GBS) prophylaxis, intra-amniotic infections, cesarean prophylaxis, or endometritis (Table 1) or for infections that occur equally in nonpregnant and pregnant women (e.g., upper respiratory tract infections, pneumonia, human immunodeficiency virus [HIV], and other sexually transmitted diseases [STDs]). This chapter reviews the microbes found in obstetric infections, the pathophysiology of obstetric infection, the antibiotics used and their pharmacology during pregnancy, adverse fetal effects of antibiotics, cost considerations in antibiotic choices, and antibiotic management of specific and common obstetric infections. Management issues are STDs in pregnancy, treatment of urinary tract infection, adjunctive antibiotic therapy in preterm labor and premature rupture of membranes, HIV therapy, prophylactic antibiotics, treatment of peripartum infection (e.g., intra-amniotic infection, endometritis, mastitis), and vaginitis.
TABLE 1. Frequency of Infection in Obstetric Patients
Asymptomatic bacteriuria | 4–8% |
Cystitis | 1–3% |
Pyelonephritis | 3–7% |
Intra-amniotic infection | 2–10% |
Group B streptococcus genital colonization | 10–30% |
Endometritis | |
| Vaginal delivery | 1–5% |
| Cesarean delivery | 5–30% |
Wound infection | |
| Episiotomy | 1–5% |
| Cesarean section | 2–10% |
Mastitis | |
| Nonbreastfeeding | 1–4% |
Breastfeeding | 5–15% |
Endocervicitis | |
| Chlamydial | 2–25% |
| Gonococcal | 0.5–10% |
Trichomoniasis | 1–10% |
Bacterial vaginosis | 15–30% |
Yeast vaginitis | 5–20% |
HIV infection | 0.5–30/1000 |
MICROBES IN OBSTETRIC INFECTION
Microbes that cause obstetric infection can be considered of exogenous or endogenous origin. Exogenous organisms are usually single pathogens acquired through environmental exposure and are not a normal part of the genital tract flora. Prominent examples are STD organisms (Neisseria gonorrhoeae, Chlamydia trachomatis, Trichomonas vaginalis, Treponema pallidum, herpes simplex virus type 2, HIV); organisms or entities associated with obstetric or surgical epidemics (toxic shock syndrome, group A streptococci, Staphylococcus aureus [mastitis], methicillin-resistant S. aureus, vancomycin-resistant Enterococcus); organisms or entities associated with poor hygiene or sanitation on a community level (Salmonella, Vibrio cholerae, tuberculosis, staphylococcal toxin–related food poisonings); and organisms associated with community epidemics (“common cold,” Neisseria meningitis, influenza virus types A and B). The acquired immunodeficiency syndrome (AIDS) epidemic has expanded the role of antibiotics to treat infections caused by opportunistic organisms.
The most common obstetric infections (urinary tract infections, intra-amniotic infection, postpartum endometritis, and postoperative wound infections) are associated with endogenous flora of the gastrointestinal tract, vagina, or skin. The most important source is the vaginal flora; it is the source of the common, polymicrobial upper genital tract infections, postpartum endometritis, and intra-amniotic infection. The anatomy, microenvironment, and function of the vagina are uniquely suited for colonization and growth of bacteria. The vagina is a warm, moist environment that is enriched regularly with nutrient-containing body fluids (genital tract fluids, semen). Colonization from the gastrointestinal tract and exogenous bacterial communities (i.e., sexual activity) occur regularly.2 Without adequate host defense, the sterile upper genital tract, products of conception, and abdomen are vulnerable to infections.
The immunology of the female genital tract is poorly studied and poorly understood, especially during pregnancy.3 A coalition of systems involved with cellular and humoral immunity provides immunoglobulins and an array of general and committed neutrophils, lymphocytes, and macrophages as an excellent first defense against upper genital tract invasion. The location, interaction, and control of vaginal immunology are being explored.
A second major mechanism of defense is the normal microflora, which competitively inhibits and reduces other more pathogenic microflora, such as the Enterobacteriaceae or anaerobes. The organisms that have received the most interest and study are the lactobacilli that produce hydrogen peroxide (H2O2). These organisms play a major role in maintaining a normal vaginal microenvironment.4 Women with greater than 10 colonies/g secretion of H2O2-producing lactobacilli have a lower vaginal pH, lower incidence, and a lower colony count of the pathogens. The lactobacilli act through a reduced ability to buffer an acid environment, a greater tolerance of acidic environments, and direct killing effects of H2O2 and other compounds.4
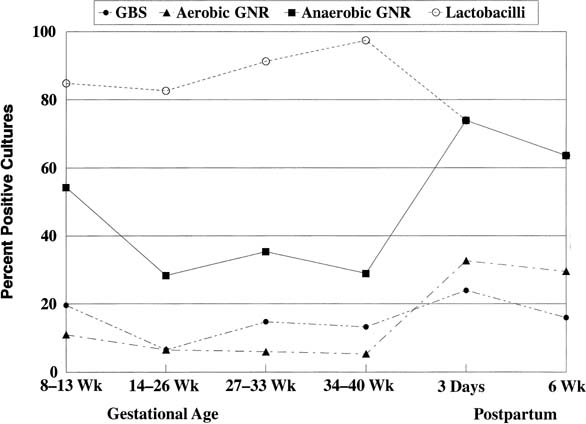
Although it is relatively easy (but expensive) to determine the incidence of various bacteria in the vagina, quantitative measurement, especially with obligate anaerobes, is difficult and has been conducted only by a few research laboratories on primarily nonpregnant women. The incidence of Enterobacteriaceae, anaerobes, and lactobacilli during pregnancy is shown in Figure 1.5 For most pregnancies, high levels of lactobacilli maintain a normal vaginal microenvironment: a vaginal pH less than 4.0, a lowered ratio of anaerobes to aerobes, a succinate-to-lactate ratio less than 4, and a lowered incidence of Gardnerella vaginalis, anaerobes, and Mycoplasma hominis. During delivery, the vaginal flora is greatly disturbed. A flood of alkaline (pH 7.40) bacterial culture media, amniotic fluid, and blood enters the vagina. Often the resident flora is challenged by prophylactic antibiotics, multiple interventions (e.g., vaginal examinations), and foreign bodies (e.g., electronic fetal monitoring), which enhance and distribute pathogens. As a result, the vaginal pH and the incidence and concentration of pathogens increase dramatically.
PATHOPHYSIOLOGY OF OBSTETRIC INFECTIONS
The microflora of the normal vagina and various clinical infections are remarkably different. Table 2 describes the microflora found in the vagina of normal pregnant women6 and the specimens of common obstetric infections: urinary tract infection,7 intra-amniotic infections,8 and postpartum endometritis.9 Of urinary tract infections, 90% involve a single organism. In contrast, 80% to 90% of intra-amniotic infections and postpartum endometritis involve more than one isolate, usually two to four aerobic or anaerobic isolates or both. Wound infections usually involve one or two aerobic isolates; however, more serious infections, such as necrotizing fasciitis, involve many more isolates, including anaerobes.
TABLE 2. Urogenital Tract Microflora (Percentage of Isolates)
| Normal Flora6 | UTI7 | Chorioamnionitis8 | Endometritis9 | |
Group A streptococcus | <1 | <1 | <1 | 2 |
Group B streptococcus | 21 | 3 | 15 | 10 |
Enterococcus | 4 | 2 | 5 | 25 |
Staphylococcus aureus | 3 | 2 | 2 | 6 |
Lactobacillus | 73 | <1 | 4 | |
Escherichia coli | 1 | 76 | 8 | 15 |
Klebsiella | <1 | 5 | 2 | 12 |
Proteus | <1 | 2 | <1 | 8 |
Gardnerella vaginalis | 56 | 16 | 35 | 33 |
Ureaplasma urealyticum | 75 | 2% | 47 | |
Mycoplasma hominis | 33* | 30 | 28 | |
Chlamydia trachomatis | 9 | <1 | 2 | |
Neisseria gonorrhoeae | 1 | <1 | <1 | |
Trichomonas vaginalis | 13 | 2 | ||
Anaerobic streptococci | 7 | 2 | 9 | 4 |
Prevotella | 10 | <1 | 30 | 25 |
Bacteroides | 10 | <1 | 4 | 4 |
Fusobacterium | <1 | <1 | 5 | 4 |
Clostridium | 4 | <1 | <1 | |
Candida | 8 | <1 | ||
Bacterial vaginosis | 16 | 25 | 28 |
UTI, = urinary tract infection.
*Most studies do not culture for Mycoplasma; these percentages are based on 44 patients.12
A wide variety of factors cause the differences in microflora and the frequency and intensity of clinical infection. An understanding of the pathophysiology of obstetric infection is crucial in the prevention and treatment of obstetric infection. The following simple equation provides an organized approach to this understanding:
Infection = [inoculum size × virulence of organism × site or media characteristics]/host resistance
The size of the inoculum can be divided into two major components: obstetric management and surgical technique. Much obstetric infection is associated with organisms found in the vagina. Abnormal vaginal microecology (bacterial vaginosis) is associated with 100-fold to 1000-fold higher concentrations of organisms per gram of vaginal secretion, but obstetric interventions inoculate the upper genital tract. Multiple authors have shown many clinical risk factors for intra-amniotic infection, endometritis, and postcesarean wound infection, including duration of labor, duration from time of rupture of membranes, number of vaginal examinations, duration of fetal scalp electrode placement, and duration of internal pressure catheter placement.9,10,12,13
Attempts at changing the vaginal flora at the time of cesarean section have had mixed success. Neither preoperative chlorhexidine14,15 nor povidine-iodine vaginal washes16 reduce the risk of endometritis after cesarean section. There may be some benefit to preoperative intravaginal metronidazole in addition to intravenous prophylactic antibiotics.17
Surgical techniques have a great influence on the development of wound infection and postcesarean endometritis. Poor skin preparation and failure to isolate the abdominal wound as sometimes occur with emergency cesarean section have be shown to increase wound infection after cesarean section. Abdominal entry using a Joel-Cohen technique followed by nonclosure of the pelvic and parietal peritoneum reduces the incidence of wound infection.18 Wound contamination by the delivering hand, which is thoroughly inoculated with vaginal secretions, is a major challenge to the success of prophylactic antibiotics.19 Removal of contaminated gloves and reisolation of the abdominal wound with sterile drapes after delivery of the placenta may reduce contamination considerably. Copious irrigation or aspiration with warm, sterile saline reduces further the size of the inoculum. Manual removal of the placenta has been shown to increase the likelihood of postcesarean infections.20,21 The placenta should be allowed to deliver spontaneously.
Differences in the virulence of different bacteria are well recognized. No obstetrician or surgeon would prefer Clostridium perfringens to Staphylococcus epidermidis. In general, high-virulence organisms include group A and group B streptococci, Enterobacteriaceae, Listeria, many anaerobes (Prevotella, Bacteroides, Fusobacterium, and Clostridium), and STD organisms. Patients with abnormal vaginal microecology (bacterial vaginosis) are more likely to have endometritis10,11 or intra-amniotic infection.12,13 Clinical predictors of abnormal vaginal microecology include antepartum hospitalization, prior use of antibiotics, current vaginal symptoms, history of STD during pregnancy, and history of multiple partners. The latter two factors may explain partially the consistent association of the teenage years or low parity with obstetric infections. Hospitalization and prophylactic antibiotics9 change significantly the virulence of microflora found in clinical infections. The basis of the virulence is the development of β-lactamases producing strains of bacteria.
A variety of genital tract organisms produce β-lactamases, which hydrolyze the β-lactam ring of cephalosporins and penicillins; this is the basis for their resistance to these antibiotics. Selective pressure by antibiotics creates the more virulent strains. Eschenbach22 reviewed the role of β-lactamase-producing bacteria in pelvic infection. The endometrium of women with pelvic infection was associated with bacteria that produced intermediate (15% of isolates) or high (24% of isolates) levels of β-lactamases. Blood isolates from similar pelvic infections revealed 11% of isolates to be intermediate and 32% of isolates to be high β-lactamase-producing bacteria.
The site and media characteristics are often variables over which the surgeon has control. These variables refer to the tissue and fluid conditions at the incision. The anaerobic conditions created by incision and suture at cesarean section permit the growth of an anaerobic inoculum and create the need for anaerobic coverage with subsequent antibiotic treatment. By removing excess blood and amniotic fluid, the obstetrician improves the site characteristics and reduces the need for and duration of antibiotic therapy. The most important adjunct to improving the site and media characteristics is the use of prophylactic antibiotics. The inclusion of antibiotics in the serum and blood that collect in the surgical site during and shortly after surgery makes the site more resistant to bacterial colonization and growth. Prophylactic antibiotics for the prevention of post–cesarean section endometritis is the standard of care.
The site or media characteristic modifies the choice and timing of prophylactic antibiotic. When the inoculum is small and infection is not established, broad-spectrum coverage is not needed and does not improve effectiveness. What is more important is that the antibiotics reach blood and serum levels in the wound before the natural process of inflammation and surgical compression reduce the blood flow in the area (e.g., intravenous injection after cord clamping).
When host resistance is high, the risk of infection is low, and the success of antibiotic treatment is improved—an inversely proportional relationship. Variables associated with reduced host resistance include medication (chemotherapy, steroids), substance abuse (alcohol, illicit drugs, tobacco), chronic disease (diabetes, autoimmune disease, HIV, renal/hepatic failure), and nutrition (obesity, starvation). Acute starvation, such as what is seen during a long, induced labor, may seriously affect the mother’s ability to overcome the challenges posed by size of the inoculum or virulence of the organisms. Often the obstetrician can do little to alter the influence of these variables; however, their presence may encourage the use of prophylactic antibiotics in an otherwise low-risk patient. A meta-analysis of controlled trials with random assignment of nonlaboring patients with intact membranes to treatment or no-treatment groups showed lower infection rates when prophylactic antibiotics were used than when they were not.23
SPECIFIC ANTIBIOTICS IN OBSTETRICS
There are three strategies to defeat the β-lactamases present in polymicrobial genital tract infections: (1) modification of the β-lactam molecule to reduce the enzymatic attachment and hydrolysis of the β-lactam ring (third-generation cephalosporins or penicillins); (2) use of antibiotics that do not have a β-lactam ring (aminoglycosides, macrolides); or (3) combination of a β-lactam antibiotic with a competitive β-lactamase inhibitor (clavulanate potassium) or a noncompetitive inhibitor (sulbactam, tazobactam). Table 3 summarizes the mechanism of action for various antibiotics used in obstetrics. Modification of the β-lactam antibiotics was the most efficient and profitable strategy for the development of new antibiotics by pharmaceutical companies in the 1970s and 1980s. In the 1990s, the pharmaceutical companies focused on development of effective β-lactamase inhibitors (clavulanate potassium, sulbactam, tazobactam), which has allowed combination with inexpensive, well-known penicillins and cephalosporins. In the new millennium, the rapidity of bacterial resistance to current antibiotics has led to increasing focus on novel antibiotics.
TABLE 3. Mechanism of Action of Antimicrobials
Inhibits synthesis of essential metabolites
Isoniazid
Para-aminosalicylic acid
Sulfonamides
Trimethoprim
Inhibits cell wall synthesis
Bacitracin
Carbapenems
Cephalosporins
Cephamycins
Cycloserine
Monobactams
Penicillins
Vancomycin
Damages cell membranes
Amphotericin B
Colistin
Nystatin
Polymyxin B
Inhibits protein synthesis through 30S subunit of ribosomes
Aminoglycosides
Tetracyclines
Inhibits protein synthesis through 50S subunit of ribosomes
Chloramphenicol
Macrolides (erythromycin, azithromycin, clindamycin)
Damages DNA
Actinomycin
Fluoroquinolones
Rifampin
Table 4 presents the susceptibilities of common genital tract organisms to representative antibiotics that are used in obstetrics. In general, broad-spectrum antibiotics are required for most obstetric infections. Adequate coverage is obtained by (1) a second-generation or third-generation cephalosporin (cefotetan, cefotaxime, ceftriaxone); (2) β-lactam plus inhibitor combination (ampicillin plus sulbactam; ticarcillin plus clavulanate potassium; piperacillin plus tazobactam); or (3) a combination of an aminoglycoside plus clindamycin. The role of Enterococcus in infection is controversial; the addition of ampicillin to the gentamicin/clindamycin combination may be reasonable.24
Click To View Table
PHARMACOLOGY AND SAFETY OF ANTIBIOTICS
Because the efficacy of many antibiotic regimens is similar, the choice depends on the pharmacokinetics, safety, and cost considerations. The safety and pharmacokinetics of antibiotics in pregnancy are poorly studied and poorly understood. Pregnant women are one of the least-studied populations. Most human subject review boards resist the use of pregnant women out of concern for the unknown fetal risks. Granting agencies, especially pharmaceutical agencies, do not fund research on pregnant women because of the liability risk. The clinician often relies on recommendations based on studies in laboratory animals; neonatal usage; and studies performed at the time of abortion, cordocentesis, delivery, or cesarean section. None of these latter situations accurately reflects the physiologic state of a nonlaboring pregnant woman, however.
Table 5 lists the major pregnancy-associated physiologic changes and their potential influence on antimicrobial pharmacokinetic parameters.25 The net influences of these physiologic changes are that maternal antibiotic concentrations tend to be 10% to 50% lower than in the nonpregnant state, oral absorption is less certain, and the late-gestation fetus is exposed to higher levels of antimicrobials.
TABLE 5. Pregnancy-Associated Physiologic Changes and Their Potential Influence on Antimicrobial Pharmacokinetics
Physiologic Changes | Kinetic Influences | Possible Clinical Effects |
Expanded intravascular/extravascular volume (about 50%) | Increased volume or distribution | Need for larger loading dose |
Reduced plasma protein concentration | Underestimation of free drug levels | |
Increased (about 50%) renal blood flow and glomerular filtration rate | Increased drug clearance | Subtherapeutic drug concentrations |
Need to increase dose or decrease dosing interval | ||
Increased progesterone-activated hepatic metabolism | Increased rate of biotransformation | Need to increase dose or decrease dosing interval |
Decreased gastric motility | Reduced rate of absorption from small bowel | Unpredictable absorption of orally administered drugs |
Increased rate of absorption from large bowel | ||
Decreased gastric acid production | Absorption of weak basic compounds | Unpredictable absorption of orally administered drugs |
Thinning of fetomaternal barrier with advancing gestation | Increased transplacental diffusion | Increased fetal concentration Decreased maternal concentration Need to increase dose or decrease dosing interval |
Antimicrobial agents cross the maternal-fetal interface primarily by simple diffusion, although some active and facilitated transfer can occur. The rate of diffusion across the placental barrier is directly proportional to the maternal-fetal concentration gradient and the surface area of the placenta but inversely proportional to the thickness of the placental barrier. In general, drugs with a molecular weight of less than 500 kd, high lipid solubility, low degree of ionization, and low protein binding (high free drug level) cross to the fetus more readily.
Most antibiotics follow a similar pattern of transplacental passage after a single intravenous infusion into the mother. Peak umbilical blood levels occur within 30 to 60 minutes after the time they peak in maternal serum.24 Fetal-to-maternal peak serum level ratios range from 0.3 to 0.9 for ampicillin, penicillin, cefotaxime, cefuroxime, ticarcillin, clindamycin, and metronidazole and are less than or equal to 1 for erythromycin and dicloxacillin.25 Maternal protein binding reduces fetal drug levels. Ampicillin is only 20% protein bound, whereas dicloxacillin is 96% bound. Protein binding can have a profound effect on choice of antibiotic in pregnancy.
One of the crucial questions an obstetrician must ask is who is being treated for the infection—the mother, the fetus, or both? If the mother has a cellulitis (mastitis), the use of dicloxacillin will treat her, but it will have little effect on the fetus or neonate. If the mother has syphilis, a major consideration is effective fetal therapy; erythromycin is inadequate because of poor transplacental passage, and penicillin is standard treatment (after desensitization in a penicillin-allergic patient). If the mother has an intra-amniotic infection, in which the mother and fetus need therapy, fetal blood levels of antibiotic active against GBS and Escherichia coli are key to reducing neonatal sepsis and pneumonia. In this case, ampicillin plus sulbactam, with rapid transplacental passage, is preferred to cefotetan, which passes poorly as a result of extensive protein binding.
Similar to the nonpregnant woman, the pregnant woman is at risk for adverse reactions owing to antimicrobial therapy. The overall incidence or severity of drug reactions is similar in pregnant and nonpregnant women: 5% to 10% of women require cessation of antibiotic therapy because of drug effects.25 The most common reactions are skin rash (β-lactam antibiotics), renal impairment (aminoglycosides), and elevated liver function tests (modified β-lactam antibiotics, macrolides, antifungals, antivirals). Any antibiotic, even a prophylactic antibiotic, can cause pseudomembranous ulcerative colitis.erythromycin estolate (hepatotoxicity), tetracycline (hepatotoxicity), and isoniazid (hepatotoxicity and neurotoxicity). Isoniazid is a crucial component of antituberculosis therapy, however, and should not be withheld for usual toxicity concerns. Pyridoxine (50 mg daily) and folic acid (1 mg daily) should be supplied as well.
FETAL EFFECT OF ANTIBIOTICS
The fetal risks of antibiotic use are governed by the mechanism of action of the antibiotic (see Table 3); the pharmacokinetics of the antibiotic, including the intensity, frequency, and duration of therapy; and concurrent high-risk maternal/fetal conditions. Maternal-to-fetal transfer of antibiotics has been poorly studied because of the reluctance of pharmaceutical companies to solicit the Food and Drug Administration (FDA) for approval for use during pregnancy and the hesitation of institutional review boards for the protection of human subjects to approve phase 1 pharmacokinetics studies in pregnant women. The aggressive climate for litigation and the likelihood of adverse pregnancy outcome independent of any medication make the cost prohibitive for pharmaceutical companies, especially when physicians freely use medications for the appropriate indication with an unapproved use (e.g., during pregnancy). Few drugs of any type are approved by the FDA for use during pregnancy.
Animal studies are limited in their ability to predict fetal risk.27 The concordance rate (fetal animal risk equals fetal human risk) for known human teratogens is as follows: mouse, 85%; rat, 80%; rabbit, 60%; hamster, 45%; monkey, 30%; two or more species, 80%. The likelihood that at least one other species is susceptible to the teratogen is 97%. The concordance rate for human nonteratogens is as follows: mouse, 35%; rat, 50%; rabbit, 70%; hamster, 35%; monkey, 80%; two or more species, 50%; and all species, 28%.
Antibiotics place the fetus at risk for the following five adverse complications: spontaneous abortion, carcinogenic effects, fetal growth restriction, teratogenic effects, and physiologic changes. Antibiotics seem to pose the greatest risk in terms of their teratogenic and physiologic effects. The risk of congenital defects occurs only if the antibiotic is given during the embryonic stage of pregnancy: days 17 to 56 of gestation. An example of a physiologic defect is the selective binding of sulfonamides to the serum proteins of the fetus or newborn. Subsequently, albumin is displaced, and neonatal jaundice may occur.
The pharmaceutical companies apply to the FDA for approval indications and use. Their application contains extensive animal and human study; however, the number of human studies in pregnancy is limited. The pharmaceutical companies usually do not seek nor does the FDA grant approval for use of these drugs during pregnancy. The fetal risk classification in the Physicians’ Desk Reference reflects the data and studies submitted to the FDA at the time of the approval. Rarely, if ever, does the FDA research the issue and upgrade the designation beyond that submitted by the pharmaceutical company. A pharmaceutical company that wishes to limit liability for use of the drug during pregnancy is content with a risk factor C classification. Other authors, notably Briggs and associates,28 go beyond the data supplied by the pharmaceutical companies (if available) and also designate a risk factor classification (Table 6).
TABLE 6. Fetal Risk Summary
Category A | Controlled studies in women fail to show a risk to the fetus in the first trimester (and there is no evidence of a risk in the later trimesters), and the possibility of fetal harm appears remote |
Category B | Either animal-reproduction studies have not shown a fetal risk but there are no controlled studies in pregnant women, or animal-reproduction studies have shown adverse effects (other than a decrease in fertility) that were not confirmed in controlled studies in women in the first trimester (and there is no evidence of risk in the later trimesters) |
Category C | Either studies in animals have revealed adverse effects on the fetus (teratogenic, embryocidal, or other) and there are no controlled studies in women, or studies in women and animals are not available |
Category D | There is positive evidence of human fetal risk, but the benefits from use in pregnant women may be acceptable despite the risk |
Category X | Studies in animals or humans have shown fetal abnormalities or there is evidence of fetal risk based on human experience, and the use of the drug clearly outweighs any possible benefit |
Adapted from Briggs GG, Freeman RK, Summer JY: Drugs in Pregnancy and Lactation. 6th ed. Philadelphia, Lippincott Williams & Wilkins, 2002.
Most antibiotics have low fetal risks in pregnancy and lactation, especially the large group of β-lactam antibiotics. Tables 7, 8, 9, 10, 11, and 12 list the fetal risk categories for various antimicrobials. These tables are meant to provide a preliminary estimate and do not replace a review of literature resources concerning the risk (maternal or fetal) of a drug.
TABLE 7. Fetal Risk Categories for Common Antibacterial Antibiotics
Antibiotic | Risk Category |
Amikacin | C |
Azithromycin | B |
Carbapenems | C |
Cephalosporins | B |
Chloramphenicol | C |
Clindamycin | B |
Erythromycin | B |
Gentamicin | C |
Metronidazole | B |
Monobactams | C |
Nitrofurantoin | B |
Quinolones | C |
Penicillins | B |
Spectinomycin | B |
Spiramycin | B |
Sulfonamides | B |
Tobramycin | C |
Trimethoprim | C |
Vancomycin | C |
TABLE 8. Fetal Risk Categories for Antiparasitics
Antiparasitic | Risk Category |
Chloroquine | C |
Mebendazole | C |
Mefloquine | C |
Metronidazole | B |
Piperazine | B |
Primaquine | C |
Pyrimethamine | C |
TABLE 9. Fetal Risk Categories for Antivirals (Non-HIV)
Antivirals | Risk Category |
Acyclovir | B |
Amantadine | C |
Ganciclovir | C |
Famciclovir | B |
Valacyclovir | B |
TABLE 10. Fetal Risk Categories for Antituberculosis Drugs
Drug | Risk Category |
Cycloserine | C |
Ethambutol | B |
Isoniazid | C |
Para-aminosalicylic acid | C |
Pyrazinamide | C |
Rifampin | C |
Streptomycin | D |
TABLE 11. Fetal Risk Categories for Antifungals
Antifungal | Risk Category |
Amphotericin B | B |
Butoconazole | C |
Clotrimazole | B |
Fluconazole | C |
Griseofulvin | C |
Ketoconazole | C |
Miconazole | C |
Nystatin | B |
TABLE 12. Fetal Risk Categories for Antiretroviral Antibiotics
Antiretroviral Antibiotics | Risk Category |
Nucleoside reverse-transcriptase inhibitors | |
| Abacavir | C |
| Didanosine | B |
| Lamivudine | C |
| Stavudine | C |
| Zalcitabine | C |
| Zidovudine | C |
Non-nucleoside reverse-transcriptase inhibitors | |
| Delavirdine | C |
| Efavirenz | C |
| Nevirapine | C |
Protease inhibitors | |
| Amprenavir | C |
| Indinavir | C |
| Lopinavir | C |
| Nelfinavir | B |
| Ritonavir | B |
| Saquinavir | B |
Relatively few antibiotics should be limited or are contraindicated (Table 13). Podophyllin should not be used because of the risk of neurotoxicity. Some clinical situations arise in which the use of other drugs may be justifiable. For example, quinine may be required to treat effectively chloroquine-resistant Plasmodium falciparum malaria.
TABLE 13. Antibiotics of Limited Use or Contraindicated in Pregnancy
Antibiotic | Risk Category |
Podophyllin | X |
Quinine | D |
Streptomycin | D |
Tetracyclines | D |
ANTIBIOTICS IN THE BREASTFEEDING MOTHER
Most medications taken by the mother appear in breast milk, but the calculated dose consumed by the breastfeeding infant ranges from 0.001% to 5% of the standard therapeutic doses tolerated by infants without toxicity.chloramphenicol, phenylbutazone, atropine, and the ergot alkaloids.
When the literature does not reveal the concentration of an antibiotic in breast milk, an approximation can be derived.29,30 First, the apparent volume in which the antibiotic is distributed is calculated as Vd = [dose/Cp0], where Vd is the apparent volume of distribution (L/kg), dose is the quantity (mg/kg) administered to the mother, and Cp0 is the antibiotic concentration in the plasma at time 0. Cp0 is a derived value reflecting the concentration in maternal plasma, if instantaneous distribution of the antibiotic had taken place before metabolism and elimination from the body; it is the plasma concentration predicted from extrapolation of the elimination phase to time 0. The Cp0 is available for most drugs given to nonpregnant patients. In the first 2 to 3 weeks postpartum, the literature values for Cp0 may be unreliable because of differences in the physiology of postpartum women—greater blood volume, decreased serum albumin, and increased renal clearance.
When the volume of distribution is known, the concentration of the antibiotic in breast milk can be calculated as CB = dose/Vd, where CB is the concentration (mg/L) of drug in breast milk, Vd is the volume of distribution (L/kg), and dose is the amount delivered to the maternal systemic circulation (mg/kg). In general, the maternal serum concentration of unbound drug determines the amount excreted in breast milk. Drug characteristics that predict a higher breast milk concentration are low molecular weight, high lipid solubility, low protein binding, small volume of distribution, and long half-life. Antibiotics prescribed in megadoses and antibiotics prescribed for chronic conditions have higher concentrations in breast milk.
The risk of a drug to the infant is related to the dose consumed, the oral bioavailability, and the elimination. Solubility, gastrointestinal pH, gastric emptying time, interaction with food constituents, and gastrointestinal membrane permeability determine absorption of drugs, and each of these characteristics is affected by the infant’s age. In the first several weeks of life, low gastric acid secretion enhances the absorption of drugs unstable in acid, such as penicillin, and the neonate’s gastrointestinal tract is more permeable to these macromolecules. Gastric emptying time is prolonged for 6 to 8 months, however, and may decrease antibiotic absorption.
There are several differences between infants and adults that affect the elimination of drugs. Extracellular fluid volume decreases from 50% of the total fluid volume to 25% after 1 year of age. Water-soluble drugs are distributed freely into this compartment, reducing and delaying the elimination of free drug. The total body fat content varies with age: 3% in the preterm neonate, 12% at full term, and 30% at 1 year. Highly lipid-soluble drugs are more likely to affect the neonatal brain because of its fewer fat-storage sites. Neonates have decreased protein binding because of lower albumin concentrations and affinity, which results in more free drug, greater drug metabolism, and more drug displacement of unconjugated bilirubin. Neonates also have deficient mechanisms for drug metabolism and elimination because of hepatic enzymatic deficiencies and renal immaturity; the half-lives of many drugs in neonates are 2 to 10 times longer than those in adults.
Given the complexities of antibiotic transfer to breast milk, neonatal tolerance, and the lack of data regarding specific antibiotics, the following guidelines are helpful:
- Evaluate the therapeutic benefit of antibiotics. Antibiotics for viral upper respiratory infection provide fewer benefits than the same antibiotics used for pneumonia. Are antibiotics really necessary?
- Choose intravenous antibiotics most widely tested and with the lowest milk-to-plasma ratio.
- Choose antibiotics with the lowest oral bioavailability.
- Select the least toxic drug with the shortest half-life.
- Avoid long-acting forms. Usually, these antibiotics are detoxified by the liver or bound to protein.
- Schedule doses so that the least amount gets into the milk. The rate of maternal absorption and the peak maternal serum concentration are helpful in scheduling dosage. Usually, it is best for the mother to take the antibiotic immediately after a breastfeeding session.
- Monitor the infant during the course of therapy. Many antibiotics for maternal use also are used for infants. This implies the availability of knowledge about therapeutic doses and the signs and symptoms of toxicity. Toxic effects include sedation, anorexia, diarrhea, rash, and hyperkinesis. In most cases, the dose received by the infant is less than the therapeutic dose. The dose can be calculated as follows:
Dose/24 hours = CB × weight × volume of milk
COST EVALUATION OF ANTIBIOTICS
Cost-effective antibiotic therapy has been highlighted as health care systems seek to reduce costs. Table 14 provides representative daily costs of common antibiotic regimens: the cost of acquisition of the antibiotic plus diluent. Although this comparison is an important first step, these figures reflect only a small portion of the total cost and are inadequate for cost-containment decisions. Cost evaluation must take into account other material, personnel, and efficacy issues, including the indications for use, nursing personnel time and effort, adjunctive laboratory test (drug levels), the speed and proportion of patients cured with initial therapeutic antibiotics, the drug and maternal costs of the regimen, the cost associated with adverse reactions, and the ease with which patients who fail the initial antibiotic regimen are cured.
TABLE 14. Daily Cost of Commonly Ordered Antibiotic Regimens in Obstetrics and Gynecology
Antibiotics | Daily Cost |
Prophylaxis | |
Ampicillin 2 g IV | $5.00 |
Cefazolin 2 g IV | $6.00 |
Cefotetan 2 g IV | $36.00 |
Penicillin G 5 million U IV | $10.00 |
Ampicillin 2 g plus sulbactam 1 g IV | $46.00 |
Intra-amniotic Infection | |
Cefuroxime 1.5 g IV q 8 h | $26.00 |
Ampicillin 2 g plus sulbactam 1 g IV q 6 h | $46.00 |
Piperacillin/tazobactam 3.375 g IV q 6 h | $52.00 |
Ampicillin 2 g IV q 6 h plus gentamicin 120 mg q 8 h | $18.00 |
Vancomycin 1 g IV q 12 h plus gentamicin 120 mg q 8 h | $25.00 |
Endometritis | |
Cefotetan 2 g IV q 12 h | $40.00 |
Piperacillin/tazobactam 3.375 g IV q 6 h | $52.00 |
Cefotaxime 2 g IV q 8 h | $50.00 |
Ampicillin 2 g IV q 6 h plus gentamicin 120 mg q 8 h plus clindamycin 900 mg q 8 h | $45.00 |
Several general principles help explain the concept of true cost. The largest cost item is the number of hospitalized days. If the antibiotic regimen saves only 1 day of hospitalization, the differences in daily cost (see Table 14), differences in nursing personnel effort with differences in multidrug regimen (“triple antibiotics”) versus single agent, and differences in dosing frequency (cefoxitin versus cefotetan) are relatively minor. Wound infection after cesarean section often prolongs hospitalization considerably, however, and if an antibiotic is associated with a greater number of cases of wound infection because it fails to treat Enterococcus, this antibiotic may be a poor choice.9,24
A second principle is the clarity of indication and diagnosis. If the antibiotic regimen is started for weak diagnostic criteria, uninfected patients are treated unnecessarily and the hospitalization days, material, and personnel costs are high. Delay in diagnosis of pelvic infection can lead to a more serious infection (i.e., pelvic abscess), however, and increased costs. A history and physical examination by a knowledgeable health care provider and a focused laboratory workup is the best way to ensure cost-effective diagnosis of infection.
If all other issues are equivalent, the cost of a drug may play a role. Single-agent therapy with a broad-spectrum cephalosporin or penicillin is effective in most obstetric infections. Agents with a reduced dosing frequency (e.g., cefotetan every 12 hours) reduce the cost of personnel time and effort. A multiagent regimen, which usually includes an aminoglycoside, is more expensive because of its differing dose frequency and the need to monitor drug levels.
SPECIFIC USES OF ANTIBIOTICS IN OBSTETRICS
Asymptomatic Bacteriuria
A combination of host defense inefficiency, anatomy, behavior, and microbial virulence factors identifies a cohort of women who have episodes of bacteriuria throughout their lifetimes.31,32,33,34,35,36 Cross-sectional prevalence studies identify 1% to 8% of women with asymptomatic bacteriuria.21,22 In longitudinal studies, 30% to 50% of nonpregnant women with bacteriuria have symptomatic lower urinary tract infections during 3 to 5 years of follow-up.32,33,34 Most episodes cluster over a 3- to 4-month period, followed by an asymptomatic interval of variable length. A series of 9- to 19-year follow-up studies37 on 60 asymptomatic bacteriuric school girls (6 to 10 years old) were compared with studies on 38 nonbacteriuric control school girls matched for age, race, and school. Episodes of bacteriuria in the 5-year study period for infected girls and controls were 22% and 3%; episodes during pregnancy were 64% and 27%. The children of bacteriuric women were more likely to have urinary tract infections than were the children of controls.
Of 30% of women who are bacteriuric during pregnancy, 20% are bacteriuric on long-term follow-up cultures when they are not pregnant.35,36 Radiologic examination at follow-up of women who were bacteriuric during pregnancy revealed abnormalities in 316 (41%) of 777 women (range, 5% to 75%). Chronic pyelonephritis was the most common radiologic diagnosis (47% of abnormalities). The incidence of bacteriuria during first pregnancies was significantly greater in women with (47%) than without (27%) renal scarring from childhood urinary infections. Similar controls who had not had childhood urinary infections had an incidence of 2%.
The cohort of women with chronic, episodic bacteriuria is identified by routine screening of urine cultures at the first prenatal visit. The prevalence of asymptomatic bacteriuria (≥2 cultures having≥105↑ colony-forming units per milliliter) is higher among women with prior renal/urinary tract disease, diabetes, sickle cell trait/disease, poor hygiene, high parity, increased age, and lower socioeconomic status.33,39 The overall prevalence varies between 1.9% and 11.8%, with the lowest prevalence in primiparous patients of upper socioeconomic class and the highest among indigent multiparas. Although most women with asymptomatic bacteriuria are identified shortly after entering prenatal care, approximately 1% to 2% acquire bacteriuria later in pregnancy.
Uncomplicated, asymptomatic bacteriuria is a significant health risk for pregnant women but not for nonpregnant women. Asymptomatic bacteriuria has been associated with pyelonephritis, preterm birth, growth retardation, hypertension, and fetal neuropathology. The most consistent association is a greater likelihood of pyelonephritis. Sweet40 reviewed the relationship between asymptomatic bacteriuria and acute pyelonephritis. In 1699 patients with untreated asymptomatic bacteriuria (18 studies), pyelonephritis developed in 471 (27.8%; range, 16% to 65%). Antibiotics in placebo-controlled trials reduced by 80% the frequency of pyelonephritis in women with asymptomatic bacteriuria. The incidence of pyelonephritis in the treated groups ranged from 0% to 5.3%. On the basis of these observations, treatment of asymptomatic bacteriuria in pregnancy is warranted to reduce the incidence of pyelonephritis.
The association between preterm birth and asymptomatic bacteriuria first was identified by Kass41 at Boston City Hospital between 1955 and 1960. As is true of many early studies, prematurity was defined as birth weight less than or equal to 2500 g, a definition that would include 30% to 50% of growth-retarded term infants. Kass’ initial study reported that 32 (17.8%) of 179 bacteriuric patients delivered low-birth-weight (LBW) infants, whereas 88 (8.8%) of 1000 nonbacteriuric patients delivered LBW infants. Since that report, many studies of small numbers and heterogeneous populations have supported and rejected this observation. In 1990, Sweet and Gibbs39 reviewed 19 studies that related bacteriuria to LBW infants. In these studies, 3619 bacteriuric pregnant women delivered 400 (11%; range, 4.4% to 23%) LBW infants. In these same studies, 31,277 nonbacteriuric women delivered 2725 (8.7%; range, 3% to 13.5%) LBW infants. Some cohort studies designed to adjust for socioeconomic demographic variables failed to show a difference in LBW between women with and without asymptomatic bacteriuria. Perhaps asymptomatic bacteriuria is not associated with LBW per se, but it is a marker for low socioeconomic status, which in turn predicts LBW.
When confounding variables are controlled, a strong relationship between asymptomatic bacteriuria and LBW remains. In 1989, Romero and associates42 reported on the relationship between asymptomatic bacteriuria and LBW. A meta-analysis was performed to increase the statistical power for primary and secondary outcome variables and to improve estimations of the effect of sample size on treatment trials. Previous cohort, case-controlled, and randomized antibiotic trials, many of which also were reviewed by Sweet,39,40 were analyzed for comparable and appropriate study design. Seventeen cohort studies met their criteria for “good” studies. The typical relative risk for a nonbacteriuric woman to deliver a LBW infant compared with a bacteriuric woman was 0.65 (95% confidence interval [CI] = 0.52 to 0.72). One case-controlled study compared the prevalence of bacteriuria in women delivering at less than 36 weeks (33 of 404 [8.1%]) with the prevalence of bacteriuria in women delivering at or beyond 37 weeks (15 of 404 [3.7%]) (p = .0036) after matching for maternal race, age, parity, smoking habits, physical dimensions, and sex of the newborn. Eight randomized clinical trials of antibiotic therapy showed a significant reduction in the frequency of LBW after antibiotic therapy (typical relative risk = 0.56; 95% CI = 0.43 to 0.73). These analyses support the hypothesis that untreated asymptomatic bacteriuria is associated directly with a higher incidence of LBW. It is unclear whether the benefit from antibiotics results from a reduction in asymptomatic or symptomatic pyelonephritis or from beneficial changes in abnormal genital tract flora, which are associated with LBW.
A variety of antimicrobial agents and treatment regimens have been used to treat asymptomatic bacteriuria during pregnancy.43 Most community-acquired pathogens associated with asymptomatic bacteriuria during pregnancy are sensitive to sulfa drugs (sulfisoxazole, 1 g four times daily for 7 days), nitrofurantoin (100 mg four times daily for 7 days), or cephalosporins (cephalexin, 500 mg four times daily for 7 days). Ampicillin (500 mg four times daily for 7 days) is a time-honored, safe, effective, and inexpensive therapy; however, there are a growing number of resistant E. coli strains.
Patient education should accompany any prescription for antibiotics to treat urinary tract infection. The essentials of behavior intervention include (1) avoiding the female superior position during sexual activity; (2) avoiding anal intercourse before vaginal intercourse; (3) voiding within 15 minutes after sexual activity; (4) avoiding bubble baths and oils; (5) avoiding vaginal douching or deodorant sprays; and (6) always wiping the urethra, perineum, and anus from front to back. These interventions reduce the frequency of recurrent urinary tract infections in high-risk women.44
Fihn and Stamm45 reviewed 62 treatment trials for uncomplicated urinary tract infections to assess whether methodologic problems compromised the validity of the study. These trials fulfilled an average of 56% of 12 standards necessary for accurate interpretation and comparability. The standards least often met were sufficient power to detect a meaningful difference (21%), double-blind assignment of treatment regimens (37%), and clear definitions of cure and failure (35%). Those deficiencies were especially true when comparing single-dose versus multidose therapy. None of 14 randomized controlled trials had sufficient power to prevent a type II error. When roughly comparable studies were pooled, single-dose amoxicillin (3 g) was significantly less effective than conventional multidose therapy (69% versus 84%). Until a larger study is performed, single-dose therapy should not be used in the treatment of urinary tract infections in pregnancy.
Antibiotics sterilize the urine in asymptomatic bacteriuria in 80% to 90% of women. The cure rate depends on compliance, length of regimen, preexisting risk factors, asymptomatic renal infection, and sensitivity of the organism. A test of cure by culture within 2 weeks after the end of the antibiotic regimen discriminates between relapse and reinfection. Relapse (a positive test-of-cure culture) has been associated with complicated asymptomatic bacteriuria. These women may have urinary tract abnormalities, asymptomatic renal infections, or silent urolithiasis. Unusual organisms or antibiotic sensitivity patterns alert the clinician to a reservoir of partially protected bacteria (e.g., renal abnormality, urolithiasis, or noncompliance). A urine pH greater than 6.0 (Proteus) and persistent hematuria are clues indicating a stone-related infection. During pregnancy, a renal ultrasound helps identify a renal stone as a cause of relapse. A postpartum intravenous pyelogram is warranted in any case of relapse. Relapse should be treated with another 10-day course of antibiotics chosen by the sensitivity pattern from the test-of-cure culture. The therapeutic regimen should be followed by suppressive therapy.
Suppressive antibiotic therapy is effective in reducing recurrent cystitis in nonpregnant women and recurrent pyelonephritis in pregnant women. The prophylactic efficacy depends on nightly bactericidal activity against sensitive reinfecting bacteria entering the bladder urine. Vaginal colonization with uropathogenic Enterobacteriaceae continues unabated, depending on the regimen chosen. The rectal reservoir for potential uropathogen is rarely sterilized by either therapeutic or prolonged suppressive regimens. One danger of suppressive therapy is the emergence of antibiotic-resistant strains. High-dose cephalexin (500 mg four times daily), but not low-dose cephalexin (250 mg four times daily), induces resistant E. coli strains.36 Nitrofurantoin macrocrystals (100 mg every night) neither reduce the prevalence of Enterobacteriaceae in rectal or periurethral flora nor induce antibiotic resistance. Trimethoprim, 40 mg, plus sulfamethoxazole, 200 mg, given every night reduces the incidence of Enterobacteriaceae in rectal and periurethral flora, but it generally is not associated with antibiotic resistance.36 Lincoln and coworkers46 reported resistant urinary infections resulting from sulfonamide suppression therapy.
In motivated patients, a combination of patient education and urine testing biweekly for leukocyte esterase and nitrite is just as effective as prophylactic antibiotic suppression in reducing the frequency of recurrent pyelonephritis after an initial episode during pregnancy. The frequency of recurrent pyelonephritis in the antibiotic suppression group was 7% versus 8% in the close surveillance group.47 The latter surveillance regimen may be enhanced further by antibiotic prophylaxis (nitrofurantoin macrocrystals, 100 mg, or cephalexin monohydrate, 500 mg) after each episode of sexual intercourse or masturbation.36,48
Acute Cystitis
Acute cystitis occurs in 0.3% to 2% of pregnancies.39 The reported frequency is only minimally greater than the frequency of cystitis in sexually active nonpregnant women. The diagnosis is more difficult to make during pregnancy. Most pregnant women have urgency, frequency, or suprapubic discomfort. Suprapubic discomfort in pregnancy often results from pressure from the presenting fetal part or early labor. Suprapubic discomfort from cystitis is unique, however, and most women with a history of acute cystitis can discriminate accurately between cystitis and pregnancy-related discomfort. The most reliable findings are dysuria and hematuria. Acute dysuria also may result from labial or perivaginal irritation secondary to vaginitis, vulvitis, herpes simplex, condylomata acuminata, or genital ulcers. Because of the separate pregnancy risks associated with these factors, an inspection of the vulva and vagina is warranted in patients with acute cystitis during pregnancy.
Patients with preterm labor and impending second-trimester loss often present with signs and symptoms similar to those of acute cystitis. As the lower uterine segment expands and the presenting fetal part descends, hesitancy, urgency, frequency, and suprapubic discomfort occur. A bloody vaginal discharge may contaminate and confuse urine testing and may lead to misdiagnosis of urinary tract infection. Pelvic examination is warranted in patients presenting with signs and symptoms of lower urinary tract infection to rule out preterm labor.
Treatment of acute cystitis is similar to that of asymptomatic bacteriuria:43 nitrofurantoin, 50 mg four times daily for 7 days; cephalosporin, 500 mg four times daily for 7 days; or sulfonamide, 1 g four times daily for 7 days. Because these patients are symptomatic, therapy is initiated as soon as a midstream, clean-catch urine culture is obtained. Studies in nonpregnant women suggest that 3 days of antibiotics may be as efficacious and cost less than a 7-day regimen.49 The comparative efficacy of 3- versus 7-day regimens has not been studied in pregnancy. A test-of-cure culture is obtained within 2 weeks after therapy is complete. Of women, 10% to 20% have a positive test-of-cure culture, representing a relapse. These women should be retreated with another antibiotic, as determined by bacterial sensitivities. After retreatment, these patients should be placed on suppressive antibiotic therapy. Without suppressive therapy, an additional 20% to 30% of women acquire another urinary tract infection (i.e., a reinfection) during the remainder of pregnancy and the puerperium. Because of the risk of recurrence, patients with cystitis should be followed intensively with a urine screen biweekly for nitrite and leukocyte esterase.
The delivery process constitutes a significant risk period for symptomatic urinary tract infections. Trauma to the urethra, periurethra, and labia creates swelling and pain that inhibits frequent and complete voiding. Multiple vaginal examinations and the pumping action of the fetal head in the second stage inoculate the urine with periurethral flora. Urinary retention is exacerbated by epidural anesthesia and perineal trauma. Interventions such as simple in-and-out catheterization to relieve urinary retention pose a 10% to 15% risk of bacteriuria.50 As a result, 10% to 25% of all pyelonephritis cases associated with pregnancy occur in the first 14 days postpartum.
Acute Pyelonephritis
Acute pyelonephritis is the most common serious medical complication of pregnancy.39,51 The incidence of pyelonephritis is 1% to 5%. Often these patients present for prenatal care in the second half of pregnancy with signs and symptoms of pyelonephritis. Only 40% to 67% of pyelonephritis cases occur in patients with a known history of asymptomatic bacteriuria. Three fourths of women with pyelonephritis present in the antepartum period, 5% to 10% present in labor, and 10% to 25% present postpartum. Antepartum pyelonephritis occurs mainly after the first trimester: 10% to 20% during the first trimester, 45% to 70% during the second trimester, and 8% to 45% during the third trimester. The predominance of pyelonephritis in late pregnancy and the puerperium relates to the partial obstruction caused by the growing uterus and to trauma or interventions at birth.
The diagnosis of acute pyelonephritis is based on clinical presentation: fever (≥38°C), costovertebral angle tenderness, and either bacteriuria or pyuria. Among patients meeting these criteria (n = 656),52 12% had fevers greater than 40°C; costovertebral angle tenderness was on the right side in 54%, on the left side in 16%, and bilateral in 27%. Chills and back pain were a presenting complaint in 82% of patients, whereas only 40% had dysuria, frequency, urgency, or hematuria; 24% had nausea and vomiting.
Overt septic shock or adult respiratory distress syndrome occurs in 1% to 2% of pregnant women with acute pyelonephritis. Clinical clues to the development of these life-threatening complications are leukocytopenia (<6000 cells/mm3), hypothermia (≤35°C), elevated respiratory rate, and widened pulse pressure. In the late stages, hypothermia, mental confusion, and symptomatic hyperstimulation of the sympathetic nervous system (cold, clammy extremities) herald a scenario that often leads to maternal or fetal death. In all cases, the mother and fetus should be treated in facilities having the expertise and equipment to handle critically ill mothers and infants.
All pregnant women with pyelonephritis should be hospitalized because of the additional fetal and maternal risks of acute pyelonephritis in pregnancy. Intravenous antibiotics (2 g of cefazolin every 6 hours or 2 g of ampicillin plus 1 g of sulbactam every 6 hours) should be initiated as soon as possible after urine and blood cultures are obtained. Because many patients are dehydrated as a result of nausea and vomiting, careful rehydration is started. The degree of endothelial damage in the lungs may not be apparent, so careful attention to fluid intake and output and vital signs, especially respiratory rate, is imperative. Respiratory symptoms (e.g., an increased respiratory rate), peripheral cyanosis, and mental confusion prompt an immediate x-ray study and measurement of arterial blood gases. Colloid oncotic pressure and serum albumin measurements are important in the fluid management of these critically ill patients.
Endotoxins stimulate cytokine and prostaglandin production by decidual macrophages and fetal membranes. The ensuing preterm contractions raise concern for preterm birth. Three major problems confront the physician at this point. First, although pyelonephritis is often a clear diagnosis, the presence of lower abdominal pain and contraction raises the possibility of intra-amniotic infection, a diagnosis that precludes tocolytic therapy. The presence of white blood cells and bacteria on an unspun Gram stain of amniotic fluid is sufficiently sensitive in the diagnosis of intra-amniotic infection to preclude the use of tocolysis. Second, premature contractions may not indicate labor. Often uterine irritability ceases after hydration and administration of antibiotics. If contractions are of sufficient frequency and strength to change the cervix on serial pelvic examinations (≥2 cm in dilation, ≤1 cm in length, and ≥50% effacement), the diagnosis of preterm labor is made. Third, preterm labor must be treated with an appropriate tocolytic agent if no other contraindication to tocolysis is present (e.g., intra-amniotic infection, fetal lung maturity, fetal abnormalities, or rupture of membranes). Ritodrine hydrochloride, the only FDA-approved tocolytic, exacerbates the cardiovascular effect of endotoxemia. The risk of pulmonary edema, cardiac toxicity, and adult respiratory distress syndrome is increased. Magnesium sulfate (4 g intravenous slow bolus, followed by 2 to 4 g/h) is the tocolytic of choice. Serum magnesium levels (≤10 mEq/L) and physical signs of toxicity (loss of deep tendon reflexes) are especially important to follow, however, because half of patients with acute pyelonephritis have renal dysfunction.
Maternal hyperthermia (≥38.3°C) should be treated aggressively with antipyretics, such as acetaminophen. Maternal hyperthermia, hence fetal hyperthermia (an additional 0.5°C), increases the metabolic demand of the fetus. Glucocorticoids should not be used to enhance fetal lung maturity because they may exacerbate maternal infection. Of these pyelonephritis patients, 80% to 90% become afebrile within 48 hours, and an additional 5% to 15% become afebrile by 72 hours; 5% to 10% are classified as initial treatment failures. In patients with significant deterioration of their condition after the first 18 hours of therapy or in patients with temperatures greater than 38°C at 48 hours of therapy, 1.5 mg/kg of gentamicin every 8 hours should be added. The dosing frequency is extended for serum creatinine greater than 1 mg/dL (dosing frequency = 8 × the serum creatinine). Antibiotic therapy should be continued until the patient is afebrile (<37°C) for more than 24 hours. The patient should finish a 14-day course of antibiotics with a bactericidal oral medication (500 mg of an oral cephalosporin four times daily). A test-of-cure urine culture should be performed 2 weeks after therapy. Without suppressive antibiotic therapy, reinfection is common in these patients: 20% have asymptomatic bacteriuria, and 23% have recurrent pyelonephritis. Frequent surveillance (nitrate/leukocyte esterase testing biweekly) or suppressive antibiotic therapy (100 mg of nitrofurantoin every night) is warranted.47 The risk of recurrent pyelonephritis is less than 10% (with either regimen).47,51
The differential diagnosis in patients with persistent fever and costovertebral tenderness at 72 hours of therapy includes a resistant organism, urolithiasis, renal abscess, complete ureteral obstruction, or another source of infection (e.g., appendicitis or intra-amniotic infection). A radiologic evaluation of the urinary tract is warranted after re-examination of the patient and review of culture and sensitivity reports.53 Many radiologists are unduly concerned regarding the fetal dangers of intravenous pyelograms during pregnancy and advocate renal ultrasound. A renal ultrasound is useful for evaluating renal abscess, but not for evaluating function or ureteral abnormalities, the more common issues associated with antibiotic failure. A “one-shot” intravenous pyelogram (no plain film and one 20-minute film) is appropriate.
Intra-Amniotic Infection
Intra-amniotic infection is a clinical infection of the amniotic fluid membranes, placenta, fetus, or uterus that occurs during labor or immediately after birth (<6 hours). Intra-amniotic infection occurs when an intrapartum temperature is greater than 37.8°C and two or more of the following conditions are present: maternal tachycardia (>100 beats/min), fetal tachycardia (>160 beats/min), uterine tenderness, foul odor of the amniotic fluid, and maternal leukocytosis (>15,000 cells/mm3).13,54 This definition was associated with a higher incidence (81% versus 31%; p < .002) and more virulent organisms (69% versus 8%; p < .001) cultured from the amniotic fluid of laboring women.13,54 The incidence of intra-amniotic infection varies between 0.5% and 10% of laboring women and is associated with significant maternal and neonatal morbidity. Maternal age younger than 20, nulliparas, longer duration of rupture of membranes, longer duration of fetal scalp sample, greater number of vaginal examinations, and preterm gestation are risk factors for intra-amniotic infection.13,55
Potential maternal complications include sepsis, adult respiratory distress syndrome, prolonged labor, cesarean wound infection, and persistent fever postpartum.54 As long as intra-amniotic infection is recognized promptly, broad-spectrum antibiotic therapy reduces additional maternal infectious morbidity to a minimum. In a randomized trial of intrapartum versus postpartum antibiotic therapy, intrapartum treatment was associated with a lower maximum temperature postpartum and fewer postpartum hospitalization days (4.0 ± 1.0 versus 5.0 ± 1.9; p < .05).56 The duration of labor was not altered by antibiotic therapy; however, the sample size in this study was small (n = 45). Several cohort studies showed a deterioration in uterine contractibility with intra-amniotic infection.54 It is possible that early antibiotic therapy may improve myometrial performance and reduce the need for cesarean section secondary to dystocia.
In a population of primarily term infants, the neonatal morbidity and mortality rates of untreated intra-amniotic infection were as follows: sepsis, 10% to 15%; pneumonia, 1% to 4%; and perinatal death, 0.5% to 3%.54,57 Intra-amniotic infection in preterm neonates probably results in higher morbidity rates; however, the observed risk is reduced by the routine use of intrapartum antibiotics in preterm gestations complicated by intra-amniotic infection. GBS and E. coli contribute a disproportionate number of cases of neonatal morbidity, and the goal of intrapartum therapy is to provide fetal blood levels of antibiotics to which GBS and E. coli are sensitive.
Reducing fetal neonatal morbidity and mortality is the major focus of therapy in intra-amniotic infection, and three issues dominate fetal management in these cases: (1) the need to provide effective antimicrobial therapy for the fetus; (2) the need to provide direct intravenous antibiotics to the neonate; and (3) the use of fetal monitoring, antipyretics, and surgical technique at cesarean section. In the past, obstetric services delayed antibiotic therapy in intra-amniotic infection until after birth because the therapy would result in negative newborn cultures and delay the diagnosis of neonatal sepsis. The results of two retrospective studies58,59 and one prospective, randomized study56 have shown unequivocally a benefit of administering intrapartum antibiotic therapy as soon as an infection is diagnosed. Overall, intrapartum antibiotics were associated with 8 (2%) of 389 cases of neonatal sepsis versus 21 (9.3%) of 225 (p < .01) cases among women treated postpartum. The incidence of neonatal pneumonia (3.2%) reported in these studies was too small to determine the effectiveness of intrapartum antibiotics on pneumonia.
The two essential qualities of an antibiotic regimen are the ability to cover the organisms associated with early neonatal sepsis and intra-amniotic infection and the ability to cross the placenta in quantities sufficient to begin fetal/neonatal therapy.60 GBS and Enterobacteriaceae are the most common organisms associated with early neonatal sepsis and are commonly found in intra-amniotic infection (see Table 2). Ampicillin, 2 g intravenously every 6 hours, plus gentamicin, 1.5 mg/kg intravenously every 8 hours, or ampicillin, 2 g, plus sulbactam, 1 g intravenously every 6 hours, provide effective therapy. Ampicillin is always given first because it crosses the placenta rapidly (<30 minutes) in high concentrations (ratio of maternal blood to cord blood, 0.71). Gentamicin is used in higher doses (1.5 mg/kg every 8 hours) in pregnant women than in nonpregnant women because of the high renal clearance associated with pregnancy.
Anaerobes play a major role in the pathogenesis of preterm birth, the amniotic fluid flora of intra-amniotic infection (see Table 2), and complications associated with postcesarean endometritis. The addition of anaerobic coverage has reduced failure rates in postcesarean endometritis, and because of this finding, we add clindamycin, 900 mg intravenously every 8 hours, after cord clamping to the primary antibiotics (ampicillin plus gentamicin or ampicillin plus sulbactam) if the patient is undergoing a cesarean section. Maberry and Gilstrap60 compared the effect of ampicillin plus gentamicin (n = 69) with ampicillin, gentamicin, and clindamycin (n = 64) in a randomized, comparative trial. One infant in each group (1.5%) had positive blood cultures, and there were no differences in maternal outcomes. The sample size was insufficient to show a difference in postcesarean complications (abscess or septic pelvic thrombophlebitis), which may have been affected by the addition of clindamycin.
Modified cephalosporins and penicillins enter the market frequently. Many have an antimicrobial spectrum including anaerobic coverage, which would indicate they are effective therapy for intra-amniotic infection. Newer agents should not be used, however, unless they have limited fetal or neonatal effects and have been shown to cross the placenta. There are several alternatives based on safety and the study of their transplacental pharmacokinetics (Table 15).
TABLE 15. Antimicrobial Regimens for Treatment of Intra-amniotic Infection
First Choice
- Piperacillin plus tazobactam
- Cefuroxime
- Ampicillin plus gentamicin*
Second Choice
- Cefoxitin
- Ampicillin plus sulbactam*
Penicillin Allergy
- Cefuroxime
- Cefazolin plus gentamicin*
- Vancomycin plus gentamicin*
*Add clindamycin if cesarean section is performed.
Many clinicians recognize the potential neonatal risks of intra-amniotic infection and believe that the longer the fetus stays in the infected environment, the greater the likelihood of neonatal infection or stillbirth. This urgency may be reflected in a greater risk of cesarean section; however, current data suggest that this urgency is not warranted. First, intrapartum antibiotics provide bactericidal concentrations of antibiotics to the fetus, membranes, and amniotic fluid within 0.5 to 1 hour after infusion. Second, the average time between diagnosis of intra-amniotic infection and delivery is 3 to 5 hours.47 It is not likely that 3 to 5 hours will change the neonatal outcome if the fetus is receiving adequate antibiotics transplacentally. Third, the duration of infection does not correlate with adverse neonatal outcomes, such as pneumonia and early neonatal sepsis.54
The use of continuous electronic fetal monito ring is appropriate for observing the development of fetal compromise in cases of intra-amniotic infection. The combination of villous edema, hyperthermic stress, and fetal infection can lead to fetal acidosis. Although no particular pattern of periodic changes signifies fetal infection, a nonreassuring tracing (e.g., one with absent variability and late decelerations) predicts fetal acidosis and poor short-term outcomes. Fetal tachycardia is a predictor of fetal sepsis or pneumonia but may be due only to fetal hyperthermia. The use of an antipyretic (e.g., 625 mg of acetaminophen rectal suppository every 4 hours) is therapeutic and may be diagnostic.61 The lowering of maternal temperature reduces the metabolic stress of fetal hyperthermia and decreases the fetal heart rate. If the tachycardia is not due to maternal fever, the acetaminophen will not reduce the fetal heart rate. In these cases of persistent fetal tachycardia, health care providers must prepare for a hemodynamically unstable neonate. Preparation includes the personnel, skill, and drugs required for neonatal resuscitation in the delivery room.
In the past, extraperitoneal cesarean section was recommended in patients with intra-amniotic infection to reduce the surgical and infectious complications.62 More recently, the use of extraperitoneal cesarean section was not found to reduce major complications when compared with traditional transperitoneal cesarean section.13 Newton63 found no difference in blood loss, duration of surgery, febrile index, or postpartum hospital stay among patients with antibiotic-treated intra-amniotic infection who underwent cesarean section compared with patients without intra-amniotic infection who underwent cesarean section. The need to continue antibiotic therapy of intra-amniotic infection after cesarean section has been questioned.64 The sample size was not sufficient to eliminate a beta error based on persistent endometritis. Antibiotics should be continued until the patient’s temperature is less than 37.8°C for 48 hours.
Prophylactic Antibiotics in Maternal-Fetal Medicine
Prophylactic antibiotics generally are used in one of four situations: prevention of subacute bacterial endocarditis, prevention of GBS sepsis in the neonate, prevention of endometritis after a cesarean section, and prevention of recurrent pyelonephritis. Prophylactic antibiotics for recurrent pyelonephritis was discussed in the section on urinary tract infection.
PREVENTION OF BACTERIAL ENDOCARDITIS.
Bacterial endocarditis is a life-threatening complication of pregnancy. Infection of the heart valves occurs in the presence of bacteremia and pre-existing injury to the valves (e.g., rheumatic heart disease, congenitally abnormal valves, artificial valves). Intravenous drug abusers seem to be at higher risk because of the frequency of bacteremia and, perhaps, their compromised immune systems. Women with mitral valve prolapse constitute a large segment (20% to 30%) of the population of patients with bacterial endocarditis, but the prevalence of mitral valve prolapse is common (3% to 6% of young women), and the risk of bacterial endocarditis is small when prophylaxis is not used. Nevertheless, these women should have bacterial endocarditis prophylaxis for most obstetric procedures.
The incidence of bacteremia during labor and delivery is understudied and can only be estimated. The major hindrance to study is timing of the sample in relationship to labor, delivery of the infant, or delivery of the placenta. Sugrve and coworkers65 found that bacteremia occurred in 3.5% of normal deliveries and argued that prophylaxis is not routinely indicated. This opinion is in the minority, however; most authors believe that the seriousness of bacterial endocarditis outweighs the low incidence of its occurrence and that the risks of prophylaxis are minimal. In circumstances in which the risk of bacteremia is higher (e.g., prolonged rupture of membranes, operative delivery, manual removal of the placenta), antibiotic prophylaxis is clearly indicated. The recommended regimen for bacterial endocarditis prophylaxis is ampicillin, 2 g intravenously, plus gentamicin, 1.5 mg/kg intravenously, 1 hour before any surgical procedure or every 8 hours after onset of the active phase of labor. One dose is given 8 hours after the procedure. For patients allergic to penicillin, vancomycin, 1 g intravenously every 8 hours, replaces the ampicillin.
PREVENTION OF EARLY-ONSET GROUP B STREPTOCOCCAL NEONATAL SEPSIS.
Early-onset neonatal sepsis is a rare but dramatic complication in the first 3 to 7 days of life. Early-onset neonatal sepsis occurs in 3.5 per 1000 live births (GBS, 1.4 per 1000 live births; non-GBS, 0.6 per 1000 live births).66 The overall case-fatality rate is 16% and varies by gestational age (<34 weeks, 30%; 34 to 36 weeks, 10%; = 36 weeks, 2%)67 and organism (GBS, 6.7%; non-GBS, 22.3%).66 Intrapartum antibiotics reduce the incidence of early-onset GBS sepsis by 80% to 90%. Because 90% of births occur at term and 84% of early-onset GBS sepsis occurs in the term infant,67 most mothers will have had a relatively normal term pregnancy. There is tremendous opportunity for confusion, anger, and blame on the part of patients and their family. Consequently, malpractice litigation is common in these cases. Given these facts, there is tremendous pressure to identify and treat prophylactically women who carry GBS in their genital tract.
Of pregnant women, 10% to 25% are carriers of GBS in their genital tract. The incidence of positive culture is increased with the use of modified Todd-Hewitt or Lim growth media and simultaneous anorectal sampling.68 When serially cultured over a 1-year period, about half of women have persistent colonization, and about 25% are episodically and transiently colonized. Risk factors for a higher prevalence of rectovaginal GBS colonization include black race, age older than 30, not living with family or partner, less than 9 years of school, current tobacco use, and increasing years of sexual experience.69 The risk factors for invasive GBS disease in the neonate are gestational age less than 37 weeks, intrapartum temperature greater than or equal to 38°C, rupture of membranes 18 hours before delivery, GBS bacteriuria during pregnancy, multiple gestation, and a previous infant with invasive GBS disease.68,70 Of early-onset GBS and non-GBS sepsis, 49% and 79% are associated with one or more risk factors.66
Since the early 1990s, the American Trial Lawyers Association, the American Academy of Pediatrics, the American College of Obstetrics and Gynecology (ACOG), and the Centers of Disease Control and Prevention (CDC) have proposed different indications for the use of intrapartum antibiotics based on antepartum culture results, presence of risk factors for early-onset GBS sepsis, or both.68,70 In mid-2002, a population-based study that was sponsored by the CDC on 629,912 live births in 1998 and 1999 defined much better the advantages of a screening culture–based approach compared with a risk identification–based approach.72 The adjusted relative risk of early-onset GBS neonatal sepsis was 0.48 (95% CI, 0.37 to 0.63) in favor of the universal screening–based approach. Given the powerful results of this study, the following management is recommended: A rectovaginal screening culture is performed on all pregnant women at 34 to 36 weeks’ gestation using appropriate culture media; prophylactic antibiotics are recommended for any patient with a positive culture or the presence of a risk factor for early-onset GBS sepsis (see previous paragraph). If GBS status is unknown, prophylactic antibiotics are recommended regardless of risk factor status.70
Identification and therapy of GBS carrier status benefit the mother and the neonate. Many different studies at different institutions have shown an association between rectovaginal GBS and infectious morbidity in the mother. In a study involving centers in Houston, Pittsburgh, and Seattle, 7806 women were cultured at admission to labor and delivery. Of women, 22% had positive cultures; 5.2% were heavily colonized. Heavy GBS colonization was associated with intra-amniotic infection (adjusted odds ratio [OR], 2.0; 95% CI, 1.1 to 3.7) and postpartum endometritis (adjusted OR, 1.8; 95% CI, 1.3 to 2.6).73 The universal GBS screening and intrapartum antibiotics (CDC-endorsed method) reduced the incidence of clinical chorioamnionitis when compared with the risk factor method (ACOG-endorsed)—5.2% of 4453 deliveries(CDC) versus 7.7% of 7917 deliveries (ACOG). Similarly, universal screening was associated with lower rates of postpartum endometritis—2.8% (CDC) versus 4.6% (ACOG).74
There are three potential strategies to treat the GBS carrier state: antepartum, intrapartum, or neonatal therapy. Because GBS is a part of normal bowel microflora, oral antibiotic therapy has not been successful in treating antepartum GBS carrier states. The selective pressure may increase the likelihood of resistant aerobic gram-negative rods. Neonatal therapy with 50,000 U of aqueous penicillin within 1 hour of birth has been shown to reduce early-onset GBS sepsis (incidence in treated patients, 1.1 per 1000 live births).75 The results need to be interpreted with caution, however, because 10% to 20% of septic neonates present in the first 4 hours of birth, and this prophylactic therapy may delay definitive therapy in these neonates. Septic preterm neonates are more difficult to diagnosis; it is not clear that early neonatal therapy is efficacious. The standard of therapy is intrapartum therapy occurring more than 4 hours before delivery.
The recommended prophylactic treatment is penicillin G (an initial dose of 5 million U followed by 2.5 million U intravenously every 4 hours until delivery). The treatment reduces neonatal colonization by 80% to 90%, and the efficacy of antibiotics against early-onset GBS sepsis is 85% (95 CI, 42% to 98%) after adjustment for intrapartum fever.71 Ampicillin (2 g intravenously every 6 hours) is equally efficacious, but there a risk of sepsis from ampicillin-resistant gram-negative aerobic rods (E. coli).76,77,78 Penicillin-allergic mothers should receive clindamycin (900 mg intravenously every 8 hours until delivery) or erythromycin (500 mg intravenously every 6 hours until delivery). There is an increasing resistance (15% to 20%) of GBS to macrolide antibiotics (clindamycin or erythromycin). Consideration should be given to cefazolin, 2 g intravenously every 6 hours, in cases in which the reported allergy to penicillin does not include respiratory distress or immediate urticaria. About 25% of mothers with neonates who had early-onset GBS sepsis received intrapartum antibiotics (antibiotic failures).
About 25% of laboring women receive intrapartum antibiotics regardless of the screening method used. The management of an asymptomatic term infant whose asymptomatic mother received intrapartum antibiotics is not clear. Of these neonates, 70% receive a workup or antibiotic treatment.60 The expense and risk of managing more than 0.5 million neonates per year in the United States in this fashion is a potential liability.
The other hidden risk is that of selection-resistant bacteria caused by antibiotic usage for GBS prophylaxis. McDuffie and coworkers78 reported a series of four cases of adverse perinatal outcome caused by resistant Enterobacteriaceae after ampicillin or amoxicillin usage for preterm premature rupture of membranes or GBS carriage. In very-low-birth-weight neonates, E. coli is the most common pathogen associated with early-onset sepsis; 85% were resistant to ampicillin, and it was associated with a higher death rate (41%).76 These are early observations, and more research is needed, but it is prudent to use intrapartum penicillin rather than ampicillin for GBS prophylaxis.79
PREVENTION OF POSTCESAREAN ENDOMETRITIS.
Multiple studies and reviews have established that prophylactic antibiotics reduce the incidence of postcesarean endometritis. The reported incidence of postoperative infection in patients after labor or prolonged rupture of membranes is 30% to 85%; the incidence in patients who undergo elective cesarean section without prophylactic antibiotics is less than 10%.80 Prophylactic antibiotics reduce the incidence of postcesarean endometritis by 40% to 60%. Table 16 summarizes the results of a review by Swartz and Grolle in 198181 concerning comparative trials of prophylactic antibiotics versus no prophylactic antibiotics for cesarean section. Prophylactic antibiotics reduced the risk of endometritis, wound infection, and urinary tract infection.
TABLE 16. Effect of Prophylactic Antibiotics on Puerperal Infection After Cesarean Section
Infection | Control | Prophylactic Antibiotics |
Endometritis | 406/1188 (34%) | 178/1509 (12%)* |
Wound infection | 87/888 (10%) | 32/1115 (3%)* |
Urinary tract infection | 107/709 (14%) | 69/1025 (6%)* |
*p < 0.001 versus controls.
Adapted from Swartz WH, Grolle K: The use of prophylactic antibiotics in cesarean section. J Reprod Med 26:595, 1981.
Since the 1980s, the focus has been on patient selection, timing of the first dose, single versus multiple doses, narrow-spectrum versus broad-spectrum antibiotics, and the development of resistant bacteria after prophylaxis. Harger and English,82 in a randomized comparison of cefoxitin with placebo prophylaxis in 386 women, identified maternal age less than 21 years, lower socioeconomic status, gestational age less than 38 weeks, use of an intrauterine pressure catheter plus fetal scalp electrode, duration of internal monitoring greater than 9 hours, and obesity as predictors of postcesarean endometritis. Cefoxitin reduced the incidence of endometritis in the presence of one or more risk factors but not in the absence of risk factors (2 of 61 versus 5 of 61; p = not significant). A meta-analysis of the efficacy of prophylactic antibiotics for nonlaboring patients undergoing cesarean delivery with intact membranes showed significant reductions in postoperative fever, endometritis, and wound infection.23
In an animal model83 and in general surgery,84 the use of prophylactic antibiotics concomitantly with skin incision is associated with a lower rate of endometritis than if the antibiotics are delayed until after the procedure. Delay in cefazolin prophylaxis at cesarean section until after the cord clamping was not associated with a higher rate of endometritis, however.85 The use of antibiotics before cord clamping was associated with more neonatal intervention and cost.85 As a result of the latter retrospective study,85 it is recommended that prophylactic antibiotics be given after cord clamping.
The available literature suggests that a single dose of prophylactic antibiotics is as effective as multiple doses. At least four studies with randomized assignment of patients showed equal efficacy between single-dose and multidose schemes.86 At least three studies with random assignment of subjects to first-generation agents versus extended-spectrum cephalosporins (cefazolin versus ampicillin versus cefotaxime, cefazolin versus cefoxitin, cefazolin versus moxalactam) showed no differences in the incidence of endometritis (2.5% to 7.7%). In 1990, Faro and colleagues87 reported on a large study (N = 1568) with randomized assignment of subjects to 1 of 10 prophylactic regimens. The rate of endometritis in each group (n = 142 to n = 217) was as follows:
1 g cefazolin for three doses, 22.5%
1 g cefazolin, 20.3%
2 g cefazolin, 10.6%
1 g cefoxitin, 15.5%
2 g cefoxitin, 16.7%
1 g cefotetan, 6.1%
1 g ceftizoxime, 17.9%
1 g cefonicid, 15.1%
2 g ampicillin, 12.8%
4 g piperacillin, 8.4%
Faro’s study87 showed the following: (1) A single dose of 2 g of ampicillin, 2 g of cefazolin, 1 g of cefotetan, or 4 g of piperacillin was superior to three doses of 1 g of cefazolin. (2) Extended-spectrum antibiotics (cefoxitin, cefotetan, ceftizoxime, cefonicid, piperacillin) do not offer a clear advantage over first-generation cephalosporins (cefazolin) or penicillin (ampicillin). (3) Cephalosporin prophylaxis was associated with an increase in Enterococcus faecalis colonization of the vagina.
Prophylactic antibiotics fail to prevent approximately 20% to 30% of cases of postcesarean endometritis. In a classic study, Gonik and associates88 showed what many experts have thought to be true: Postcesarean endometritis is the postpartum clinical manifestation of subclinical intra-amniotic infection. Bacteria were shown within the decidua and myometrium of asymptomatic women at the time of cesarean section and in women in whom endometritis subsequently developed. These observations supported the clinical observation that clinical characteristics can predict the development of postcesarean endometritis or failure of prophylactic antibiotics.
In the large study by Faro and coworkers,87 in which all 1568 women received prophylactic antibiotics, rupture of membranes lasting more than 4 hours and duration of internal monitoring predicted failure of prophylactic antibiotics. In a retrospective review of 766 women who received either ampicillin or cefazolin prophylaxis for cesarean sections, Chang and Newton89 identified the following predictors of antibiotic prophylactic failure: multiparity (OR, 1.77; 95% CI, 1.26 to 2.48), six or more vaginal examinations (OR, 3.39; 95% CI, 2.17 to 5.28), gestational age less than 37 weeks (OR, 1.55; 95% CI, 1.04 to 2.33), and cefazolin prophylaxis (OR, 1.69; 95% CI, 1.21 to 2.36). The latter study suggests that in certain clinical situations, prophylactic antibiotics with a broader coverage and longer duration may reduce postoperative morbidity. The number of vaginal examinations during labor is a distinct, objective clinical measure of risk and might be used to select patients for a more intensive prophylactic antibiotic regimen.
A major concern of prophylactic antibiotics has been the changes in genital tract flora. Four published studies have examined these changes. In 1981, Gibbs and colleagues90 compared the endometrial flora of 100 patients who were randomly assigned to receive 2 g of cefamandole intravenously every 4 hours for three doses or placebo prophylaxis. Cefamandole was associated with significant increases in aerobic gram-negative rods and isolation of enterococci from the endometrium postpartum when compared with preantibiotic amniotic fluid cultures. In 1984, Stiver and associates91 compared the aerobic and anaerobic cervical microflora before and 4 days after cesarean sections in women randomly assigned to receive one dose of 1 g of cefazolin, 2 g of cefoxitin, or normal saline for prophylaxis. The three-dose cephalosporin regimens resulted in statistically significant increased isolation of Enterococcus from the cervix but no increase in nosocomial infection. In 1990, Faro and coworkers87 reported changes in vaginal isolates after treatment with prophylactic antibiotics for cesarean section. A total of 1568 patients were randomized to one of seven antibiotic regimens. Cephalosporin prophylaxis was associated with increases in the isolation of vaginal E. faecalis. In 1998, Newton and Wallace9 reported the differences in endometrial microflora in patients with postcesarean endometritis whether they had received ampicillin, cefazolin, or no prophylaxis. Patients who had received ampicillin prophylaxis were more likely to have Klebsiella pneumoniae, E. coli, or any gram-negative rod. Ampicillin prophylaxis was associated with a decrease in Prevotella bivia or any anaerobe. Women who received cefazolin were more likely to have Enterococcus and less likely to have Proteus. There were no differences between cures; however, cefazolin prophylaxis followed by treatment with an extended-spectrum cephalosporin was associated with an increase in wound infection and dehiscence.9
The use of prophylactic antibiotics is recommended for most cesarean sections, especially for any patient who has a cesarean section with rupture of membranes. The efficacy of prophylactic antibiotics in women without rupture of membranes and labor, who are undergoing elective cesarean section, is not clear. Most authors do not recommend their use. When prophylactic antibiotics are used, a single dose of 2 g of ampicillin intravenously (for penicillin-allergic patients, 2 g of cefazolin or 900 mg of clindamycin intravenously) immediately after cord clamping is the most cost-effective choice. In high-risk patients having more than five vaginal examinations in the active phase, an extended-spectrum prophylactic antibiotic should be given for 24 hours: 2 g of cefotetan intravenously every 12 hours for two doses or 2 g of ampicillin plus 1 g of sulbactam intravenously every 6 hours for four doses.
Postpartum Endomyometritis
Infection is the most common complication of cesarean section or vaginal delivery (see Table 1). Some type of infection occurs in 15% to 20% of postcesarean patients; endometritis is the most common (15%). Approximately 10% to 20% of endometritis cases involve severe complications, including septicemia, pelvic cellulitis, septic pelvic thrombophlebitis, abscess formation, and pneumonia. The most important single factor in predicting a puerperal infection is the presence of a uterine wound (OR, 12.84).10 The relative risk of endometritis is increased with internal monitoring, multiple vaginal examinations, rupture of membranes, and labor. Bacterial vaginosis organisms in the vagina and amniotic fluid predict a higher incidence of endometritis.10,11,92
Puerperal infection results from a multiorganism invasion of the endometrium by vaginal flora (see Table 2). Endometrial cultures show 70% of the isolates to be aerobic. Aerobic gram-negative rods (E. coli), GBS, and group D enterococci predominate. Of the isolates, 80% have anaerobic organisms as well (Prevotella, Bacteroides, and anaerobic Streptococcus). Chlamydia and Mycoplasma are present in 5% and 15% of cultures. C. trachomatis is associated with 25% of late-occurring endometritis cases after a vaginal delivery.93 This observation has not been verified by the examination of post–cesarean section patients.94 The acute morbidity of cesarean section may mask the low-grade symptoms of the infection; infertility may result.
The diagnosis of a postoperative puerperal infection can be difficult. Normal postoperative pain and postpartum contractions obscure the discomfort from mild infection. The clinical markers are fever, uterine tenderness, foul-smelling lochia, uterine subinvolution, and a persistent paralytic ileus. Standard febrile morbidity is the occurrence of two temperature elevations greater than 38°C at least 6 hours apart, taken by standard technique at least four times daily and occurring between the 2nd and 10th postpartum days. Despite the definition, fever in the first 24 hours should not be ignored, and the source should be sought by complete physical examination and appropriate testing. Possible caus
REFERENCES
Semmelweis IP: The Etiology, the Concepts and the Prophylaxis of Childhood Fever. 1861. Reprinted by the Classics of Obstetrics and Gynecology Birmingham, AL, Library Gryphon Editions, 1990 |
|
Newton ER, Piper JM, Shain ST, Peairs W: Predictors of vaginal microflora. Am J Obstet Gynecol 184:845, 2001 |
|
Wilkin SS: Immunology of the vagina. Clin Obstet Gynecol 36:122, 1983 |
|
Redondo-Lopez V, Cook RL, Sobel JD: Emerging role of lactobacilli in the control and maintenance of the vaginal bacterial microflora. Rev Infect Dis 12:856, 1990 |
|
Goplerud CP, Ohm MJ, Galask RP: Aerobic and anaerobic flora of the cervix during pregnancy and the puerperium. Am J Obstet Gynecol 126:858, 1976 |
|
Carey JC, Yaffe SJ, Catz C: for the VIP Study Group: The vaginal infections and prematurity study: An overview. Clin Obstet Gynecol 36:821, 1993 |
|
Campbell-Brown M, McFadyen IR, Seal DV, Stephenson ML: Is screening for bacteriuria in pregnancy worth while? BMJ 294:1579, 1987 |
|
Sperling RS, Newton ER, Gibbs RS: Intraamniotic infection in low-birth-weight infants. Infect Dis 157:113, 1988 |
|
Newton ER, Wallace PA: Effects of prophylactic antibiotics on endometrial flora in women with post cesarean endometritis. Obstet Gynecol 92:262, 1998 |
|
Newton ER, Prihoda TJ, Gibbs RS: A clinical and microbiologic analysis of risk factors for puerperal endometritis. Obstet Gynecol 75:402, 1990 |
|
Watts DH, Krohn MA, Hillier SL, Eschenbach DA: Bacterial vaginosis as a risk factor for post-cesarean endometritis. Obstet Gynecol 75:52, 1990 |
|
Clark P, Kurtzer T, Duff P: Role of bacterial vaginosis in peripartum infections. Infect Dis Obstet Gynecol 2:179, 1994 |
|
Newton ER: Chorioamnionitis and intraamniotic infection. Clin Obstet Gynecol 36:795, 1993 |
|
Rouse DJ, Hauth JC, Andrews WW, et al: Chlorhexidine vaginal irrigation for the prevention of peripartal infection: A placebo-controlled randomized clinical trial. Am J Obstet Gynecol 176:617, 1997 |
|
Sweeten KM, Eriksen NL, Blanco JD: Chlorhexidine versus sterile water vaginal wash during labor to prevent peripartum infection. Am J Obstet Gynecol 176:426, 1997 |
|
Reid VC, Hartmann KE, McMahon M, et al: Vaginal preparation with povidone iodine and postcesarean infectious morbidity: A randomized controlled trial. Obstet Gynecol 97:147, 2001 |
|
Pitt C, Sanchez-Ramos L, Kaunitz AM: Adjunctive intravaginal metronidazole for the prevention of postcesarean endometritis: a randomized controlled trial. Obstet Gynecol 98:745, 2001 |
|
Franchi M, Ghezzi F, Balestreri D, et al: A randomized clinical trial of two surgical techniques for cesarean section. Am J Perinatol 15:589, 1998 |
|
Sherman D, Lurie S, Betzer M, et al: Uterine flora at cesarean and its relationship to postpartum endometritis. Obstet Gynecol 94:787, 1999 |
|
Lasley DS, Eblen A, Yancey M, et al: The effect of placental removal method on the incidence of postcesarean infections. Am J Obstet Gynecol 176:1250, 1997 |
|
Atkinson MW, Owen J, Wren A, et al: The effect of manual removal of the placenta on post-cesarean endometritis. Obstet Gynecol 87:99, 1996 |
|
Eschenbach DA: A review of the role of β-lactamase-producing bacteria in obstetric-gynecologic infections. Am J Obstet Gynecol 156:495, 1987 |
|
Chelmow D, Ruehli MS, Huang E: Prophylactic use of antibiotics for nonlaboring patients undergoing cesarean delivery with intact membranes: A meta-analysis. Am J Obstet Gynecol 184:656, 2001 |
|
Walmer D, Walmer ER, Gibbs RS: Enterococci in post-cesarean endometritis. Obstet Gynecol 71:159, 1988 |
|
Chow AW, Jewesson PJ: Pharmacokinetics and safety of antimicrobial agents during pregnancy. Rev Infect Dis 7:287, 1985 |
|
Arsura EL, Fazio RA, Wickremesinghe PC: Pseudomembranous colitis following prophylactic antibiotic use in primary cesarean section. Am J Obstet Gynecol 151:87, 1985 |
|
Brown NA, Fabro S: The value of animal teratogenicity testing for predicting human risk. Clin Obstet Gynecol 26:467, 1983 |
|
Briggs GG, Freeman RK, Sumner JY: Drugs in Pregnancy and Lactation. 4th ed. Baltimore, Williams & Wilkins, 1994 |
|
Rivera-Calimlim L: The significance of drugs in breast milk. Clin Perinatol 14:51, 1987 |
|
American Academy of Pediatrics, Committee on Drugs: The transfer of drugs and other chemicals into human breast milk. Pediatrics 93:137, 1994 |
|
McDowall DR, Buchanan JD, Fairley KF, Gilbert GL: Anaerobic and other fastidious microorganisms in asymptomatic bacteriuria in pregnant women. J Infect Dis 144:114, 1981 |
|
Stamm WE, Hooten TM: Management of urinary tract infections in adults. N Engl J Med 329:1328, 1993 |
|
Andriole VT, Patterson TF: Epidemiology, natural history and management of urinary tract infections in pregnancy. Med Clin North Am 74:359, 1991 |
|
Gillenwater JY, Harrison RB, Kunin CM: Natural history of bacteriuria in schoolgirls. N Engl J Med 301:396, 1979 |
|
Stamm WE, Hooton TM, Johnson JR, et al: Urinary tract infections: From pathogenesis to treatment. J Infect Dis 159:400, 1989 |
|
Stamey T: Recurrent urinary tract infections in female patients: An overview of management and treatment. Rev Infect Dis 9:195, 1987 |
|
Zinner SH, Kass EH: Long-term (10 to 14 years) follow-up of bacteriuria of pregnancy. N Engl J Med 235:820, 1971 |
|
Little PJ, McPherson DR, Wardener HE: The appearance of the intravenous pyelogram during and after acute pyelonephritis. Lancet 1:186, 1965 |
|
Sweet RL, Gibbs RS: Urinary tract infections. Infectious Diseases of the Female Genital Tract. pp 274, 277 Baltimore, Williams & Wilkins, 1990 |
|
Sweet RL: Bacteriuria and pyelonephritis during pregnancy. Semin Perinatol 1:25, 1977 |
|
Kass EH: Asymptomatic infections of the urinary tract. Trans Assoc Am Phys 69:56, 1956 |
|
Romero R, Oyarzun E, Mazor M, et al: Meta-analysis of the relationship between asymptomatic bacteriuria and preterm delivery/low birthweight. Obstet Gynecol 73:576, 1989 |
|
McNeeley SG: Treatment of urinary tract infections during pregnancy. Clin Obstet Gynecol 31:480, 1988 |
|
Lunsden L, Hyner GC: Effects of an educational intervention on the rate of recurrent urinary tract infections in selected female outpatients. Womens Health 310:79, 1985 |
|
Fihn ST, Stamm WE: Interpretation and comparison of treatment studies for uncomplicated urinary tract infections in women. Rev Infect Dis 7:468, 1985 |
|
Lincoln K, Lidin-Janson G, Winberg J: Resistant urinary infections resulting from changes in resistance pattern of fecal flora induced by sulfonamide and hospital environment. BMJ 3:305, 1970 |
|
Lenke RR, van Dorsten JP, Schifin BS: Pyelonephritis in pregnancy: A prospective randomized trial to prevent recurrent disease evaluating suppressive therapy with nitrofurantoin and close surveillance. Am J Obstet Gynecol 146:953, 1983 |
|
Wong ES, McKevitt M, Running K, et al: Management of recurrent urinary tract infections with patient-administered single-dose therapy. Ann Intern Med 102:302, 1985 |
|
Hooton TM, Winter C, Tiv F, Stamm WE: Randomized comparative trial and cost analysis of 3-day antimicrobial regimens for the treatment of acute cystitis in women. JAMA 273:329, 1995 |
|
Turck M, Goffe B, Petersdorf RG: The urethral catheter and urinary tract infection. J Urol 88:834, 1962 |
|
Newton ER: The urinary tract in pregnancy. pp 388, 408 Walters MD, Karram MM (eds): Clinical Urogynecology. St Louis, Mosby-Year Book, 1993 |
|
Gilstrap LC III, Cunningham FG, Whalley PJ: Acute pyelonephritis in pregnancy, an anterospective study. Obstet Gynecol 57:409, 1981 |
|
Benson M, Purna JP, Resnick MI: The role of imaging studies in urinary tract infection. Urol Clin North Am 13:605, 1986 |
|
Newton ER: Intraamniotic Infection. In Rose BD (ed): UpToDate. Wellesley, MA, www.uptodate.com2002 |
|
Newton ER, Prihoda TJ, Gibbs RS: Logistic regression analysis of risk factors for intraamniotic infection. Obstet Gynecol 73:571, 1989 |
|
Gibbs RS, Dinsmoor MJ, Newton ER, Ramamurthy RS: A randomized trial of intrapartum versus immediate postpartum treatment of women with intra-amniotic infection. Obstet Gynecol 72:823, 1988 |
|
Morales WJ, Washington SR III, Lazar AJ: The effect of chorioamnionitis on perinatal outcome in preterm gestation. J Perinatol 7:105, 1987 |
|
Sperling RS, Ramamurthy FS, Gibbs RS: A comparison of intrapartum versus immediate postpartum treatment of intraamniotic infection. Obstet Gynecol 70:862, 1987 |
|
Gilstrap LC III, Leveno KJ, Cos SM, et al: Intrapartum treatment of acute chorioamnionitis: Impact on neonatal sepsis. Am J Obstet Gynecol 159:579, 1988 |
|
Maberry MC, Gilstrap LC III: Intrapartum antibiotic therapy for suspected intraamniotic infection: Impact on the fetus and neonate. Clin Obstet Gynecol 34:345, 1991 |
|
Kirshon B, Moise KJ Jr, Wasserstrum N: Effect of acetaminophen on fetal acid-base balance in chorioamnionitis. J Reprod Med 34:955, 1989 |
|
Perkins RP: Role of extraperitoneal cesarean section. Clin Obstet Gynecol 23:583, 1980 |
|
Newton ER: Treated intraamniotic infection and operative morbidity. Am J Obstet Gynecol 66:389, 1992 |
|
Turnquest MA, How HY, Cook CR, et al: Chorioamnionitis: Is continuation of antibiotic therapy necessary after cesarean section? Am J Obstet Gynecol 179:1261, 1998 |
|
Sugrve D, Blake S, Troy P, MacDonald P: Antibiotic prophylaxis against infective endocarditis after a normal delivery: Is it necessary? Br Heart J 44:499, 1980 |
|
Schuchat A, Deaver-Robinson K, Plikaytis BD, et al: Multistate case-control study of maternal risk factors for neonatal group B streptococcal disease. Pediatr Infect Dis J 13:623, 1994 |
|
Schrag SJ, Zywicki S, Farley MM, et al: Group B streptococcal disease in the era of intrapartum antibiotic prophylaxis. N Engl J Med 342:15, 2000 |
|
Prevention of early-onset group B streptococcal disease in newborns. ACOG Committee Opinion, 173, 1996 |
|
Regan JA, Klebanoff MA, Nugent RP: The epidemiology of group B streptococcal colonization in pregnancy. Obstet Gynecol 77:604, 1991 |
|
Rouse DJ, Goldenberg RL, Cliver SP, et al: Strategies for the prevention of early-onset neonatal group B streptococcal sepsis: A decision analysis. Obstet Gynecol 83:483, 1994 |
|
Schuchat A, Zywicki SS, Dinsmoor MJ, et al: Risk factors and opportunities for prevention of early-onset neonatal sepsis: A multicenter case-control study. Pediatrics 105:21, 2000 |
|
Schrag SJ, Zell ER, Lynfield R, et al: A population-based comparison of strategies to prevent early-onset group B streptococcal disease in neonates. N Engl J Med 347:233, 2002 |
|
Krohn MA, Hillier SL, Baker CJ: Maternal peripartum complications associated with vaginal group B streptococci colonization. J Infect Dis 179:1410, 1999 |
|
Locksmith GJ, Clark P, Duff P: Maternal and neonatal infection rates with three different protocols for prevention of group B streptococcal disease. Am J Obstet Gynecol 180:416, 1999 |
|
Patel DM, Rhodes PG, LeBlanc MH, et al: Role of postnatal penicillin prophylaxis in prevention of neonatal group B streptococcus infection. Acta Paediatr 88:874, 1999 |
|
Stoll BJ, Hansen N, Fanaroff AA, et al: Changes in pathogens causing early-onset sepsis in very-low-birth-weight infants. N Engl J Med 347:240, 2002 |
|
Hickman ME, Rench MA, Ferrieri P, et al: Changing epidemiology of group B streptococcal colonization. Pediatrics 104:203, 1999 |
|
McDuffie RS, McGregor JA, Gibbs RS: Adverse perinatal outcome and resistant Enterobacteriaceae after antibiotic usage for premature rupture of the membranes and group B streptococcus carriage. Obstet Gynecol 82:487, 1993 |
|
Amstey MS, Gibbs RS: Is penicillin G a better choice than ampicillin for prophylaxis of neonatal group B streptococcal infections? Obstet Gynecol 84:1058, 1994 |
|
Crombleholme WR: The use of prophylactic antibiotics in obstetrics and gynecology. Clin Obstet Gynecol 313:466, 1988 |
|
Swartz WH, Grolle K: The use of prophylactic antibiotics in cesarean section. J Reprod Med 26:595, 1981 |
|
Harger JH, English DH: Selection of patients for antibiotic prophylaxis in cesarean sections. Am J Obstet Gynecol 141:752, 1981 |
|
Burke J: The effective period of preventive antibiotic action in experimental incisions and dermal lesions. Surgery 50:161, 1961 |
|
Polk HC, Simpson CJ, Simmons BP, Alexander JW: Guidelines for prevention of surgical wound infection. Arch Surg 118:1213, 1983 |
|
Cunningham FG, Leveno KJ, DePalma RT, et al: Perioperative antimicrobials for cesarean delivery: Before or after cord clamping. Obstet Gynecol 62:151, 1983 |
|
Duff P: Prophylactic antibiotics for cesarean delivery: A simple cost-effective strategy for prevention of postoperative morbidity. Am J Obstet Gynecol 157:794, 1987 |
|
Faro S, Martens MG, Hammill HA, et al: Antibiotic prophylaxis: Is there a difference? Am J Obstet Gynecol 162:900, 1990 |
|
Gonik B, Shannon RL, Shawar R, et al: Why patients fail antibiotic prophylaxis at cesarean delivery: Histologic evidence for inpatient infection. Obstet Gynecol 179:72, 1992 |
|
Chang PL, Newton ER: Predictors of antibiotic prophylactic failure in post-cesarean endometritis. Obstet Gynecol 80:117, 1992 |
|
Gibbs RS, St. Clair P, Castillo M, Castaneda Y: Bacteriologic effects of antibiotic prophylaxis in high risk cesarean section. Obstet Gynecol 57:277, 1981 |
|
Stiver HG, Forward KR, Tyrello DL, et al: Comparative cervical microflora shifts after cefoxitin or cefazolin prophylaxis against infection following cesarean section. Am J Obstet Gynecol 149:718, 1984 |
|
Newton ER, Piper J, Peairs W: Bacterial vaginosis and intraamniotic infection. Am J Obstet Gynecol 176:672, 1997 |
|
Wager GP, Martin DH, Koutsky L, et al: Puerperal infectious morbidity: Relationship to route of delivery and to antepartum Chlamydia trachomatis infection. Am J Obstet Gynecol 138:1028, 1980 |
|
Blanco JD, Diaz KC, Lipscomb KA, et al: Chlamydia trachomatis isolation in patients with endometritis after cesarean section. Am J Obstet Gynecol 152:278, 1985 |
|
Filker R, Monif GRG: The significance of temperature during the first 24 hours postpartum. Obstet Gynecol 53:358, 1979 |
|
Hartmann KE, Barrett KE, Reid VC, et al: Clinical usefulness of white blood cell count after cesarean delivery. Obstet Gynecol 96:295, 2000 |
|
Spandorfer SD, Graham E, Forouzan I: Postcesarean endometritis: Clinical risk factors predictive of positive blood cultures. J Reprod Med 41:797, 1996 |
|
Yonekura ML: Treatment of post-cesarean endometritis. Clin Obstet Gynecol 31:488, 1988 |
|
Mitra AG, Whitten MK, Laurent SL, et al: A randomized, prospective study comparing once-daily gentamicin versus thrice-daily gentamicin in the treatment of puerperal infection. Am J Obstet Gynecol 177:786, 1997 |
|
Soper DE, Kemmer CT, Conover WB: Abbreviated antibiotic therapy for the treatment of post-partum endometritis. Obstet Gynecol 69:127, 1987 |
|
Stovall TG, Ambrose SE, Ling FW, Anderson GD: Short course antibiotic therapy for the treatment of chorioamnionitis and post-partum endometritis. Am J Obstet Gynecol 159:404, 1988 |
|
Dinsmoor MJ, Newton ER, Gibbs RS: A randomized, double-blinded placebo controlled-blind or oral antibiotics therapy following intravenous antibiotic therapy for post-partum endometritis. Obstet Gynecol 77:60, 1991 |
|
Thomsen AC: Infectious mastitis and the occurrence of antibody-coated bacteria in milk. Am J Obstet Gynecol 144:350, 1982 |
|
Thomsen AC, Espersen T, Maigaard S: Course treatment of milk stasis, non-infectious inflammation of the breast, and infectious mastitis in nursing women. Am J Obstet Gynecol 149:492, 1984 |
|
Thomsen AC, Hansen KPB, Moller BR: Leukocyte counts and microbiological cultivation in the diagnosis of puerperal mastitis. Am J Obstet Gynecol 146:938, 1983 |
|
Thomsen AC, Mogensen SC, Jepsen FL: Experimental mastitis in mice induced by coagulase-negative staphylococcus isolated from cases of mastitis in nursing women. Acta Obstet Gynecol Scand 64:163, 1985 |
|
Newton M, Newton NR: Breast abscess: A result of lactation failure. Surg Gynecol Obstet 91:651, 1950 |
|
Benson EA, Goodman MA: Incision with primary suture in the treatment of acute puerperal breast abscess. Br J Surg 57:55, 1970 |
|
Gibbs RS, Romero R, Hillier SL, et al: A review of premature birth and subclinical infection. Am J Obstet Gynecol 166:1515, 1992 |
|
Minkoff H: Prematurity: Infection as an etiologic factor. Obstet Gynecol 62:137, 1983 |
|
Kass EH, McCormack WM, Lin JS, et al: Genital mycoplasmas as a cause of excess premature delivery. Trans Assoc Am Phys 94:260, 1981 |
|
Eschenbach DA, Nugent RP, Rao AV, et al: A randomized placebo-controlled trial of erythromycin for the treatment of Ureaplasma urealyticum to prevent premature delivery. Am J Obstet Gynecol 164:734, 1991 |
|
Klebanoff MA, Regan JA, Rao VR, et al: for the Vaginal Infections and Prematurity Study Group: Outcome of the Vaginal Infections and Prematurity Study: Results of a clinical trial of erythromycin among pregnant women colonized with group B streptococci. Am J Obstet Gynecol 172:1540, 1995 |
|
Thomsen AC, Morup L, Brogaard Hansen K: Antibiotic elimination of group B streptococci in urine of preterm labor. Lancet 1:591, 1987 |
|
McGregor JA, French JI, Parker R, et al: Prevention of premature birth by screening and treatment for common genital tract infections: Results of a prospective controlled evaluation. Am J Obstet Gynecol 173:157, 1995 |
|
Carey JC, Klebanoff MA, Hauth JC, et al: Metronidazole to prevent preterm delivery in pregnant women with asymptomatic bacterial vaginosis. N Engl J Med 342:534, 2000 |
|
Klebanoff MA, Carey JC, Hauth JC, et al: Failure of metronidazole to prevent preterm delivery among pregnant women with asymptomatic trichomonas vaginalis infection. N Engl J Med 345:487, 2001 |
|
Newton ER, Dinsmoor MJ, Gibbs RS: A randomized, blinded, placebo-controlled trial of antibiotics in idiopathic preterm labor. Obstet Gynecol 74:562, 1989 |
|
Newton ER, Shields L, Ridgway LE, et al: Combination antibiotics and indomethacin in idiopathic preterm labor: A randomized double-blind clinical trial. Am J Obstet Gynecol 165:1753, 1991 |
|
Romero R, Sibai B, Caritis S, et al: Antibiotic treatment of preterm labor with intact membranes: A multicenter, randomized, double-blinded, placebo-controlled trial. Am J Obstet Gynecol 169:764, 1993 |
|
Gordon M, Samuels P, Shubert P, et al: A randomized, prospective study of adjunctive ceftizoxime in preterm labor. Am J Obstet Gynecol 172:1546, 1995 |
|
Kenyon SL, Taylor DJ, Tarnow-Mordi W: Broad-spectrum antibiotics for spontaneous preterm labour: The ORACLE II randomized trial. Lancet 357:989, 2001 |
|
King J, Flenady V: Antibiotics for preterm labour with intact membranes. Cochrane Database of Systematic Reviews No. 2, CD000246 2000 |
|
Ehrenberg HM, Mercer BM: Antibiotics and the management of preterm premature rupture of the fetal membranes. Clin Perinatol 28:807, 2001 |
|
McGregor JA, French JI: Use of antibiotics for preterm premature rupture of membranes: Rationale and results. Obstet Gynecol Clin North Am 19:327, 1992 |
|
Kenyon S, Boulvain M, Neilson J: Antibiotics for preterm premature rupture of membranes. Cochrane Database of Systematic Reviews 4:CD001058 2001 |
|
Kenyon SL, Taylor DJ, Tarnow-Mordi W: Broad-spectrum antibiotics for preterm, prelabour rupture of fetal membranes: The ORACLE I randomized trail. Lancet 357:979, 2001 |
|
Centers for Disease Control: 1993 Sexually transmitted diseases treatment guidelines. MMWR Morb Mortal Wkly Rep 42:RR-14, 1993 |
|
Mead PB, Hager WD (eds): Infection Protocols for Obstetrics and Gynecology. Montvale, NJ, Medical Economics, 1992 |
|
Wendel GD, Stark BJ, Jamison RG, et al: Penicillin allergy and desensitization in serious maternal/fetal infections. N Engl J Med 312:1229, 1985 |
|
Turrentine MA, Troyer L, Gonik B: Randomized prospective study comparing erythromycin, amoxicillin, and clindamycin for the treatment of Chlamydia trachomatis in pregnancy. Infect Dis Obstet Gynecol 2:205, 1995 |
|
Haddad J, Langer B, Astruc D, et al: Oral acyclovir and recurrent genital herpes during late pregnancy. Obstet Gynecol 82:102, 1993 |
|
Hader SL, Smith DK, Moore JS, et al: HIV infection in women in the United States: Status at the millennium. JAMA 285:1186, 2001 |
|
Lindsay MK: Human immunodeficiency virus and the obstetrician. Clin Obstet Gynecol 36:821, 1993 |
|
Lindsay MK, Johnson N, Peterson HB, et al: Human immunodeficiency virus among inner-city adolescent parturients undergoing routine voluntary screening: July 1987 to March 1991. Am J Obstet Gynecol 167:1096, 1992 |
|
Lindsay MK, Peterson HB, Boing J, et al: Crack cocaine: A risk factor for human immunodeficiency virus infection type I among inner-city parturients. Obstet Gynecol 80:981, 1992 |
|
Watts DH: Drug therapy: Management of human immunodeficiency virus infection in pregnancy. N Engl J Med 346:1879, 2002 |
|
Pediatric AIDS Clinical Trials Group Protocol 076 Study Group: Reduction of maternal-infant transmission of human immunodeficiency virus type 1 with zidovudine treatment. N Engl J Med 331:1173, 1994 |
|
Yeni PG, Hammer SM, Carpenter CCJ, et al: Antiretroviral treatment for adult HIV infection in 2002. JAMA 288:222, 2002 |
|
Jordan R, Gold L, Cummins C, et al: Systematic review and meta-analysis of evidence for increasing numbers of drugs in antiretroviral combination therapy. BMJ 324:757, 2002 |
|
Tuomala RE, Shapiro DE, Mofenson LM, et al: Antiretroviral therapy during pregnancy and the risk of an adverse outcome. N Engl J Med 346:1863, 2002 |
|
Biswas MJ: Bacterial vaginosis. Clin Obstet Gynecol 36:166, 1993 |