Cesarean Birth: Surgical Techniques
Authors
INTRODUCTION
A cesarean section is the delivery of a fetus through an abdominal and uterine incision; technically, it is a laparotomy followed by a hysterotomy.1 This definition considers only the location of the fetus and not whether the fetus is delivered alive or dead. Over recent decades, cesarean delivery has become more commonly used, and this increase has generated a number of controversial issues, including the optimum rate, what constitutes a suitable indication and what is the best technique.
Legends and myths about the abdominal delivery of an infant appear in many cultures. One of the earliest Greek myths includes the birth of Aesculapius, who according to legend, was cut from his mother's abdomen by Apollo, Bacchus, and Jupiter.2 Legend holds that Julius Caesar was also delivered abdominally, but his mother's survival well into adult life makes the story highly unlikely. It is the birth of Caesar that some authors have attributed to the origin of the term cesarean delivery.1, 3 Another possible source for the term is the Latin verb caedare, meaning to cut, or the term for the children who were born by postmortem cesarean sections, who were called caesones. The Roman law Lex Regis, which dates from 600BC, required that infants be delivered abdominally after maternal death to facilitate separate burial; this has also been proposed as the origin of the term. The specific law in question was called the Lex Cesare.4, 5
HISTORY
Historic records that elude to the performance of cesarean section date back as far as the fifth century BC and seem to imply that the outcomes for both mother and child were favorable.1 The earliest authenticated report of a child who survived cesarean birth is a document describing the birth of Gorgias in Sicily in approximately 508BC.4 There are no other accurate descriptions of the performance of a cesarean section or the immediate outcome of the mother or the neonate until 1610.1
Gabert and Bey assessed the evolution of cesarean section by dividing its development into three eras: before 1500, between 1500 and 1877, and from 1878 until the present.1 Before 1500, references to cesarean section are often clouded in mystery and misinformation, although some religious texts lead us to believe that cesarean sections were performed with the survival of both the mother and the infant.
After 1500, the available literature describing delivery by cesarean section and the success of the operation is more plentiful. In 1500, Nufer is reported to have performed the first successful modern cesarean section, with both the mother and infant surviving. The authenticity of this report is doubtful, because it was not documented until 82 years after the operation was performed. In his book Treatise on Caesarean Section published in 1581, Roussett advised that the cesarean operation be performed on a living woman; as such, he was the first physician to do so.4 In 1610, Trautmann performed a well-documented cesarean section in Wittenberg. Unfortunately, the patient died from infectious complications on postoperative day 25. In 1692, a patient who had died 14 years after delivering a child by cesarean section underwent autopsy. The accuracy of the claimed cesarean section was validated by finding a well-healed scar on her uterus.
During this time period, the cesarean operation remained crude at best. The abdominal incision was made lateral to the rectus muscles, and the uterus was incised at whichever portion was accessible through the laparotomy incision. The uterine musculature was not reapproximated, and the patient had to be physically restrained during the procedure because anesthesia was not available.1
Closure of the abdominal incision slowly evolved from choosing to leave the wound open and apply only bandages to allow healing by secondary intention through closing only the skin to full closure of the abdominal wall. Early surgeons often sutured the uterine incision to the anterior abdominal wall to encourage adhesion formation to reinforce the uterus and allow it to tolerate future gestations.1 The first report of uterine closure was not until 1769. Uterine closure was associated with decreased perioperative blood loss. Drainage of the surgical site was also introduced.
By the modern era of cesarean section (1878 to present), several modifications were being made in the cesarean operation. The Porro operation was instituted and became popular in the US and England as it became evident that this procedure was associated with decreased maternal mortality. The operation consisted of a laparotomy and hysterotomy followed by supracervical hysterectomy and bilateral salpingo-oophorectomy. The rationale behind this radical cesarean section was that with removal of the uterus and adnexa, the rates of uterine infection, sepsis, and hemorrhage would decrease.1, 4 Sterility and premature menopause were unfortunate side effects of the Porro procedure.
The first step toward the cesarean operation as it is currently performed was described by Sanger.4, He proposed a procedure that was much less radical and designed to conserve fertility. His operation did not involve hysterectomy and salpingo-oophorectomy, but instead consisted of removing the peritoneum from a portion of the anterior uterine wall and performing a 2 cm-wide wedge resection of the anterior uterine wall. The wedge was cut so that a thick edge of myometrium was adjacent to the peritoneum and a thin edge was adjacent to the endometrial cavity. These modifications allowed the serosal edges to be incorporated into the closure with interrupted silk sutures.1, 4 The technique was further improved by Garrigues, who did not resect the myometrium but instead simply closed the uterine incision. Other modifications included not dissecting the uterine serosa from the uterus and the introduction of silver wire to approximate the myometrium in addition to the interrupted silk sutures on the serosal surface.1
As operative techniques improved cesarean section became safer and could be used at an earlier stage in difficult labors. Further modifications emerged including emptying the bladder and rectum preoperatively, with catheters and enemas, respectively, to decrease the volume of these organs in the operative field, thereby reducing the risk of injury during the surgical procedure. Preoperative antimicrobial preparation was introduced by Lister in 1876 and included shaving the operative area and applying antiseptic solutions to the operative field. Vaginal douching was also introduced and routinely performed before performing cesarean deliveries.1
The technique of laparotomy and site of hysterotomy incision were vigorously debated and modified. Abdominal incisions were made to the right or left of the rectus muscles or in the midline along the linea nigra. The uterine incision was made vertically in the midline, obliquely, transversely through the contractile myometrium, laterally 7.2–10 cm from the fundus, or on the posterior aspect of the uterus.1
Johnson first described a lower segment uterine incision in 1786.1 In 1908, Selheim suggested that a uterine incision made in the lower uterine segment rather than the contractile segment of the myometrium would decrease blood loss at surgery and decrease blood loss in the event of uterine dehiscence.1, 4
The development of the modern cesarean operation has not been a recent accomplishment but instead represents a series of innovations over many centuries. Many aspects of the operation as it is commonly performed today are not based on randomized trial or techniques that have been proven to be superior by rigorous study, but instead are the culmination of many years of trial and error.
EPIDEMIOLOGY
Cesarean section rate is defined as the number of cesarean deliveries over the total number of livebirths, and is usually expressed as a percentage. Increasing cesarean section rates are a cause of concern in both developed and developing countries.6 In 1985 WHO stated: ‘there is no justification for any region to have cesarean section rates higher than 10–15%.6 However, over two decades later, the optimal rate of deliveries by cesarean section remains controversial, and the debate regarding desirable levels of cesarean section continues. Betran et al. (2007) set out to estimate the proportion of births by cesarean section at national, regional and global levels. Data were available for 126 countries, representing nearly 89% of global live births in 2002. The global rate of cesarean section was estimated in that study to be 15% (Table 1). Rates were higher in developed countries, and in Latin America and the Caribbean, but lower in other developing countries.6
Table 1 Cesarean section rates by region and subregion, and coverage of the estimates. Adapted from Betran et al., 20076
Region/ subregion* |
Births by CS (%) |
Range, minimum to maximum (%) |
Coverage of estimates† (%) |
Africa |
3.5 |
0.4–15.4 |
83 |
Eastern Africa |
2.3 |
0.6–7.4 |
93 |
Central Africa |
1.8 |
0.4–6.0 |
26 |
Northern Africa |
7.6 |
3.5–11.4 |
84 |
Southern Africa |
14.5 |
6.9–15.4 |
93 |
Western Africa |
1.9 |
0.6–6.0 |
95 |
Asia |
15.9 |
1.0–40.5 |
89 |
Eastern Asia |
40.5 |
27.4–40.5 |
90 |
South-central Asia |
5.8 |
1.0–10.8 |
93 |
South-eastern Asia |
6.8 |
1.0–17.4 |
83 |
Western Asia |
11.7 |
1.5–23.3 |
75 |
Europe |
19.0 |
6.2–36.0 |
99 |
Eastern Europe |
15.2 |
6.2–24.7 |
100 |
Northern Europe |
20.1 |
14.9–23.3 |
100 |
Southern Europe |
24.0 |
8.0–36.0 |
97 |
Western Europe |
20.2 |
13.5–24.3 |
100 |
Latin America and the Caribbean |
29.2 |
1.7–39.1 |
92 |
Caribbean |
18.1 |
1.7–31.3 |
78 |
Central America |
31.0 |
7.9–39.1 |
98 |
South America |
29.3 |
12.9–36.7 |
90 |
Northern America |
24.3 |
22.5–24.4 |
100 |
Australia/ New Zealand |
21.6 |
20.4–21.9 |
100 |
World total |
15.0 |
0.4–40.5 |
89 |
More developed countries |
21.1 |
6.2–36.0 |
90 |
Less developed countries |
14.3 |
0.4–40.5 |
89 |
Least developed countries |
2.0 |
0.4–6.0 |
74 |
*Countries categorized according to the UN classification. Countries with a population of less than 140,000 in 2000 are not included.
†Refers to population of live births for which nationally representative data were available.
The proportion of births by cesarean section has been proposed as an alternative indicator for measuring access, availability or appropriateness of medical care, as well as for monitoring changes in maternal mortality. In addition rising cesarean section rates will also reflect changes in the demographic risk profile of pregnant women (age, body mass index (BMI) and other medical disorders).
Organisation for Economic Co-operation and Development (OECD) released a comprehensive source of comparable statistics on health and health systems across OECD countries on 29 June 2010.7 According to its website, ‘it is an essential tool for health researchers and policy advisors in governments, the private sector and the academic community, to carry out comparative analyses and draw lessons from international comparisons of diverse health care systems.’
The following figure from the official OECD website shows the cesarean rates of OECD countries from 2006 to 2008 (Fig. 1).
Repeat cesarean deliveries account for a large percentage (37%) of the cesarean sections in the US.8 While patient and health care provider education may reduce the number of repeat cesarean sections there are other factors influencing cesarean section rates: for example the rate of cesarean delivery was also increased in women older than 35 years, in hospitals with more than 500 beds, in for-profit hospitals, and in patients with private insurance.9 The cesarean section rate in the US, when compared with that in other developed countries, is the among the highest in the world.9, 10
An analysis of cesarean deliveries at the University of Vermont by Pollard and Capeless in 1995 revealed that the primary cesarean section rate was 11.4%.11 Dystocia (arrest of dilatation or descent) accounted for over 35% of cesarean sections and together with abnormal presentation were the major indications for abdominal delivery. A significant problem in analyses of indications for cesarean section is that diagnoses such as dystocia, cephalopelvic disproportion, and failure to progress are inherently vague and do not reflect the true reason why the labor is not progressing as anticipated. An accurate understanding and recording of the reason that labor has not progressed, including the fetal size and position, the strength and frequency of the uterine contractions, and the adequacy of the maternal pelvis would help. It is noteworthy that primary cesarean sections for dystocia predominate in first labors and are a relatively uncommon event in subsequent labors12 and also that the majority of repeat cesarean operations result from women who have had a primary cesarean delivery for dystocia.12 Therefore critical evaluation of patients with evidence of dystocia in their first labor, with identification and alleviation of correctable problems, could significantly impact on overall cesarean section rates.
Fetal malpresentation currently accounts for approximately 3–4% of cesarean sections in the US.12 Because of the International Term Breech Trial, cesarean section is routinely offered to nonvertex infants if external cephalic version is contraindicated or unsuccessful.13, 14, 15, 16
Similar findings were published in the UK National Sentinel Cesarean Section Audit commissioned by the UK Department of Health.17 Data on 99% of births that took place in England, Wales and Northern Ireland over a 3-month period in 2000 were analyzed and the main reasons for cesarean section identified by this audit are illustrated in Table 2.
Table 2 Reasons for cesarean section (CS) in 2000 in the UK17
Of all CS in the UK |
% |
Repeat cesarean |
29 |
Presumed fetal distress |
22 |
Failure to progress |
20 |
Breech birth |
16 |
Maternal request |
1.5 |
Influence of maternal age on CS |
|
Maternal age <20 years |
13 |
Maternal age >40 years |
33 |
The studies above conclude that repeat cesarean delivery and failure of labor progression are the most common causes behind the high rates of this operation in both the UK and North America. Fetal distress was also a significant contributor in the UK (22%), possibly because of the methods adopted to diagnose fetal compromise during labor. On the other hand, in the US, fetal intolerance of labor contributes minimally to the overall cesarean rate. However, and generally speaking, the cesarean section rate has increased with the widespread use of electronic fetal monitoring.18, 19, 20, 21, 22 The use of centralized fetal monitoring increases the cesarean rate even more.22 Because of the well-documented low specificity of a nonreassuring fetal heart rate pattern, further assessment by other diagnostic means should be undertaken in all but the most pressing cases. Recently fetal electrocardiogram (ECG) recording using a scalp electrode (with analysis of the ST segment, called STAN) has been used in combination with cardiotocograph (CTG) recording for intrapartum fetal monitoring in Europe, because initial trials suggested that it might reduce the need for fetal blood sampling and reduce the number of babies delivered with a metabolic acidosis,23 but it is not without problems24 and its value is still being assessed.25 A number of other programs have been implemented at various institutions in an attempt to reduce the cesarean section rate.
In the US a labor-adjusted cesarean rate has been proposed as a more accurate indicator of the appropriateness of the rate of cesarean section than raw numbers and rates.26 This labor-adjusted rate excludes patients who are determined not to be candidates for vaginal delivery by a reasonable physician standard. For example, excluded patients would include women with a history of classical cesarean section, proven pelvic inadequacy, invasive cervical malignancy, suspected ruptured uterus before labor, maternal disease that may be life-threatening because of the physiologic changes involved in labor, macrosomia, macrocephaly, monoamniotic twins, and nonreassuring fetal heart rate on antenatal surveillance. In one population in which the labor-adjusted rate was studied, the adjusted cesarean rate was almost one-third of the raw cesarean rate.
CLASSIFICATION AND INDICATIONS
Traditionally, cesarean section has been classified as emergency or elective. However, with advanced practice in obstetrics, and more complicated deliveries encountered, this definition has become too simplistic and more detailed categories are needed. Therefore, distinguishing between prelabor cesarean section (which may be elective or emergency) and intrapartum cesarean delivery (which is, by default, emergency) is preferable27 (Table 3). Classification of the urgency of cesarean delivery has also been investigated by Lucas and colleagues28 and this has developed into the most consistent method recommended by NCEPOD29 and approved by the Royal College of Obstetricians and Gynaecologists (RCOG) and the Royal College of Anaesthetists (RCA) in the UK.
Table 3 Classification of urgency of cesarean section27, 28
Classification | Indication |
Grade 1: Emergency cesarean section | Immediate threat to the life of the woman or the fetus, i.e. placental abruption: antepartum; or uterine rupture: intrapartum |
Grade 2: Urgent cesarean section | No immediate risk to the life of the woman or baby but delivery should be achieved as soon as possible, i.e. three previous cesarean sections, membranes are ruptured with meconium-stained liquor: antepartum; or nonreassuring CTG and FBS is not possible or contraindicated: intrapartum |
Grade 3: Nonscheduled | Delivery is needed but can fit in with delivery suite workload and allow for fasting/steroid administration and some degree of planning, i.e. preterm IUGR/PET |
Grade 4: Scheduled | Also referred to as elective. No urgency whatsoever, and procedure planned to suit woman, staff, delivery suite, etc. and carried out >39 weeks’ gestation during the working day (i.e. not out of hours) |
CTG, cardiotocograph; FBS, fetal blood sample; IUGR, intrauterine growth retardation; PET, pre-eclamptic toxemia
Therefore, prelabor cesarean section could be any of the four categories explained, whilst intrapartum cesarean will only involve grades 1 and 2.
Indications for cesarean delivery vary depending on the clinical situation, resources available for patient care, and individual physician management techniques. There are no definitive algorithms available to the practicing obstetrician to direct when an abdominal delivery will benefit the mother and/or the fetus in every clinical situation. The decision to perform an abdominal delivery remains a joint judgment between the physician and patient after carefully weighing the pros and cons of a cesarean delivery versus continued labor and/or operative or spontaneous vaginal delivery. Indications for cesarean delivery can be divided into indications that are of benefit to the mother, the fetus, or both as illustrated in Table 4.30
Table 4 Indications for cesarean section30
Indications | Examples |
Maternal | Absolute: · More than 2 previous cesarean sections · Obstructive lesions in the lower genital tract including malignancies, and leiomyomas of the lower uterine segment that interfere with engagement of the fetal head Relative: · Previous uterine surgery: myomectomy or hysterotomy · Situations where the increasing intrathoracic pressure generated by Valsalva maneuvers could lead to maternal complications. These include, dilated aortic valve root and recent retinal detachment. · Women with a prior vaginal or perineal reparative surgery, such as colporrhaphy; ileal pouch - anal anastomosis following colectomy for inflammatory bowel disease |
Fetal | · Abnormal lies or nonvertex presentations · Multiple pregnancies: the first twin in a nonvertex presentation, or higher order multiples (triplets or greater) · Some congenital anomalies · Fetal compromise · Maternal infection: primary genital herpes, HIV |
Maternal–fetal | · Placenta previa · Obstructed labor |
Indications for cesarean delivery for maternal benefit include any situation in which it is inadvisable to continue to strive for a vaginal delivery out of concern for maternal outcome. In these situations, the woman undergoes a major abdominal operation for indications that are likely to decrease her risk for morbidity and/or mortality. In contrast, when a cesarean section is performed for fetal indications, the mother is undergoing major abdominal surgery when there is no immediate benefit to her but there is potential benefit to the neonate. In these situations, fetal health would be compromised if further efforts toward vaginal delivery were pursued. When counseling the patient before cesarean section regarding the risks and benefits of abdominal delivery, the possibility of morbidity and mortality must be discussed.
Before performing an elective repeat cesarean delivery, several considerations must be addressed. In 1995, the ACOG Committee on Quality Assessment published a criteria set outlining these considerations. The committee suggested that the type of previous uterine incision should be documented from the previous operative notes, the risks and benefits of attempting a vaginal birth after a cesarean section (VBAC) should be thoroughly discussed with the patient and documented in the chart, the risks and benefits of repeat cesarean delivery should be thoroughly discussed with the patient and documented in the chart, and fetal maturity should be considered.31
Fetal lung maturity is an important factor to be considered before attempting any prelabor elective CS. Morrison et al. (1995) conducted a study on 33,289 deliveries occurring at or after 37 weeks of gestation and over 9 years. The aim was to establish whether the timing of delivery between 37 and 42 weeks’ gestation influences neonatal respiratory outcome and thus provide information which can be used to aid planning of elective delivery at term. They found a significant reduction in neonatal respiratory morbidity would be obtained if elective cesarean section was performed in the week 39+0 to 39+6 of pregnancy.32 A more recent study from the USA has mirrored these findings33 and prelabor elective procedures should be deferred until after 39 completed weeks.
TECHNIQUE
As noted in the historical review at the beginning of this chapter, the cesarean operation has undergone a number of technical changes as the procedure has evolved. Many different practitioners extol the benefits of various techniques of skin incision, uterine incision, uterine closure, and many other technical aspects of the operation. However, there are relatively few randomized trials to support many of the commonly used practices at cesarean section.
Preoperative evaluation
In the case of a planned procedure, the preoperative assessment should include a full history and physical examination, past medical and surgical history, current medications, drug allergies, consent, and indication for cesarean section. In the uncomplicated patient checking a full blood count and having serum in the laboratory usually suffice should blood transfusion become necessary. In more complex instances preoperative consultation with an anesthetist, or other relevant specialist should be considered on an individual basis. The obstetrician should usually highlight women who are at high risk of anesthetic complications during the antenatal period. The risks should be documented in the medical notes and communicated with the anesthetist nearer the time. The form of anesthetic to be used will be discussed and decided on by the anesthetist who is also responsible for discussing all anesthetic risks and complications.27
Conversely in cases of an emergency cesarean section, once the decision to operate has been made by the obstetrician, discussion with the patient, consent and preoperative preparation should be carried out as best as circumstances allow.27
In very high-risk cases, such as placenta previa or suspected accreta, other preoperative measures should also be considered:27 these include the presence of a senior obstetrician and anesthetist at the time of the operation; it may be necessary to involve interventional radiologists; and a cell saver may be made available. Most importantly, the woman should have been fully counseled and consented for the different treatment options including the possibility of hysterectomy in extreme circumstances. Protocols should be present on every delivery suite for the event of massive obstetric hemorrhage
Consent
The person performing the procedure is responsible for checking that written informed consent has been given. He/she should explain the reasons why this operation is needed. Associated risks and complications should be clearly communicated with the patient and care must be taken when explaining the frequently occurring complications and those that are less likely but serious. The importance of good communication is essential and has been highlighted in the report Safer Childbirth.34
Abdominal preparation
There is evidence that any abdominal shave performed should be performed in the operating room just before applying the antibacterial preparations and not the night before. Shaving the patient the night before surgery actually increases the bacterial count on the maternal abdomen.35 Shaving should be performed only to remove the hair that will physically interfere with the operation itself.
Patient preparation
Placing the patient in the left lateral tilt position using either a hip wedge or an operative table with lateral tilt capability will help minimise uterine compression of the inferior vena cava. Before the abdominal preparation and draping of the patient, a Foley catheter should be placed to allow the bladder to drain during the operation keeping the operative field clear and allowing urinary output to be evaluated intraoperatively.
Surgical principles
Any kind of surgery should be carried out with adequate but not excessive access. Gentle handling and respect of tissues, together with meticulous attention to hemostasis are essential and important factors in all aspects of surgery. Anatomical knowledge should be thorough in order to avoid unplanned damage, especially when pathology is encountered.27
Skin incision
The skin incision should be made in one sweeping motion using the belly of the scalpel. It should be of an adequate size to give sufficient access and is usually about 12 cm long.27 A number of skin incisions have been used in abdominal deliveries (Fig. 2): two lower abdominal (Pfannenstiel and Joel-Cohen) and one midline vertical. Historically, a vertical midline skin incision was implemented, however, this scar is cosmetically less acceptable and is associated with higher incidence of postoperative wound discomfort, dehiscence, infection, and hernia formation. It may still be necessary if access is required to the upper uterus or to other abdominal organs. At present, the most frequently used type of skin incision is the Pfannenstiel incision. In general, the skin incision should be determined by the physician based on the clinical situation and the skill of the surgeon. Transverse incisions fall along the lines of expression of the anterior abdominal wall and therefore should create less pronounced scarring and risk of dehiscence. Transverse incisions have also been associated with less postoperative pain. Midline vertical incisions are generally more hemostatic and require less dissection; therefore, less time from incision to birth than transverse incisions.36
TRANSVERSE INCISIONS
The Pfannenstiel incision is made transversely in the maternal abdomen approximately 2–3 cm above the symphysis pubis and is curvilinear, with the lateral apices of the incision smiling up toward the anterior superior iliac spines (Fig. 3). This incision is performed sharply to the level of the anterior rectus fascia which is then sharply incised with the scalpel in a transverse manner in the midline to expose the belly of the rectus muscle on either side of the midline. At this time, the incision in the anterior rectus fascia may be extended laterally using either the scalpel or Mayo scissors. Extending this incision laterally through the subcutaneous tissue risks damage to the superficial epigastric and superficial circumflex iliac veins, respectively; attention to hemostasis is therefore important to minimize the risk of hematoma formation.
Care should be taken to avoid cutting the transverse oblique muscle when incising the fascia. After the fascia is incised, the anterior rectus fascia can then be dissected from the underlying rectus muscles in both the cephalic and (if needed) caudal directions by a combination of blunt and sharp dissection. During this dissection, care must be taken to identify and ligate or electrocoagulate the perforating vessels between the rectus muscles and the anterior fascia; this can be performed at entry, or in the event of an emergency cesarean delivery, at the time of closure. Once the sheath has been mobilized the peritoneum should be exposed staying in the midline (avoid hooking fingers under the rectus muscle which can damage the inferior epigastric vascular bundle). Then the entry point through the peritoneum should be made high in the operative field to avoid injury to the maternal bladder using sharp or blunt dissection: elevating the peritoneal membrane between two hemostats and palpating the opposing pieces of membrane to exclude entrapped bowel then incising with a scalpel; or pushing a finger through the peritoneum. Once the peritoneal cavity is entered and a check has been made to exclude or divide adhesions, the peritoneal incision is extended either bluntly or using scissors, to maximize surgical exposure, with care being taken to avoid inadvertent damage.
The Joel-Cohen incision is performed in a transverse manner above the location of a Pfannenstiel incision and is linear, not curvilinear. Once the fascia is incised the rest of the dissection is performed bluntly. An advantage of this type of incision is speed; however, there are no maternal or fetal advantages other than this.37, 38
In the moderately obese patient, a variation of the Pfannenstiel incision is performed a few centimeters higher than the true Pfannenstiel to avoid placing the incision in the fold created by the abdominal pannus and thereby decreasing the rate of wound complications.
VERTICAL INCISIONS
Historically, the midline vertical skin incision was the preferred incision for cesarean section because of the speed and ease of entry into the peritoneal cavity with minimal dissection required. Vertical incisions remain useful in situations where access high on the uterus is needed. The incision is performed vertically from just below the umbilicus and extended to just above the symphysis pubis and can easily be extended around the umbilicus if exposure of the upper abdomen is required. When making a midline vertical incision, it is important to remember that the linea nigra may not represent the true midline. The incision is carried sharply down to the level of the rectus sheath, which is then carefully incised with the scalpel in a vertical direction. This incision may be completed with the scalpel or by using the Mayo scissors. The fascial edge closest to the midline is then grasped with a pair of small clamps, and sharp and blunt dissections are used to separate the rectus muscles and allow entry through the peritoneum vertically as described previously.
In patients undergoing repeat cesarean delivery, the abdominal scar may be revised at the time of repeat operation. In the case of an emergency cesarean section, scar revision can be performed at the time of abdominal closure. It is also important to remember that the choice of skin incision should be that which the primary surgeon believes will be most beneficial for the present operation and should not be dictated by the location of a previous scar. Hypertrophic scars are best excised as this gives a better cosmetic result and is associated with improved wound healing; however, if the old scar is keloid then its margins should be left, as this generates less tissue reaction in the subsequent scar.27
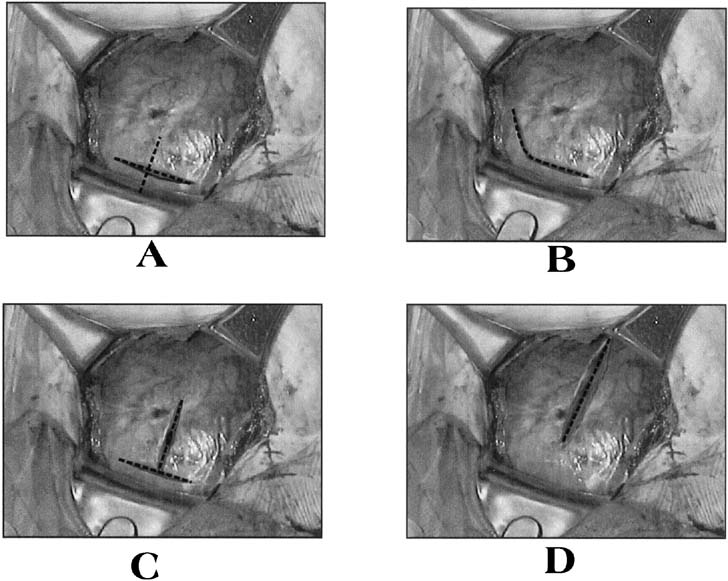
Exposure and access to the uterus
There are three standard uterine incisions that can be performed for delivery of the fetus: low transverse, low vertical, and classical (Fig. 3). The specific type of uterine incision should be determined by the primary surgeon at the time of the operation based on gestational age and lie of the fetus and any uterine anomalies. One of the important factors to be assessed before incising the uterus is the width of the lower segment (the distance between the broad ligaments). This should be assessed in relation to the size of the baby to decide whether a transverse or longitudinal incision is most appropriate. In either case, the peritoneum needs to be reflected inferiorly before the uterine incision is made.27
Historically, the creation of a bladder flap was advocated before making any uterine incisions. More recently, randomized controlled trials have noted that the omission of the bladder flap provides short-term advantages such as reduction of operating time and incision–delivery interval, reduced blood loss and need for analgesics. Practically speaking adequate access to the lower segment may require some dissection but this should be kept to what is needed and not be excessive. The peritoneum is grasped with a pair of forceps, elevated, and then incised transversely with scissors. Next, the inferior portion of the peritoneum is elevated from the lower uterine segment. A Doynes retractor should then be inserted to keep the bladder clear of the surgical field. Before making the uterine incision, the surgeon should also identify the round ligaments to assess the degree of dextra-rotation of the uterus and to evaluate for the presence of any myomas or other malformations that might affect the choice and/or placement of the incision.
Lower uterine segment incision
The standard low-segment transverse incision accounts for 90% of all uterine incisions.17 This incision should be made 2–3 cm below the upper edge of the uterovesical fold of peritoneum. This is especially important when the cesarean is performed at or near full dilatation, as the tendency is to go in too low, due to the stretched and ballooned out lower segment. A low entry in this situation risks extension of the uterine angles into the broad ligament, or even more dangerously it can risk entry into the vagina (inadvertent laparoelytrotomy) – both complications carry attendant risks to the ureters. The incision is then made sharply with the scalpel in the midline and performed down to the level of the fetal membranes, with care being made not to incise the membranes, and extended laterally using either blunt dissection with the fingers or scissors (Fig. 4). It is best to try to leave the membranes intact at this stage in order to avoid the risk of cutting the baby and to maintain the liquor until the uterine incision is completed (particular attention to avoid cutting the baby is necessary where the membranes have already ruptured, in cases of oligohydramnios, breech presentations, advanced labor or after repeat cesarean, where the lower segment can be very thin).27 There was thought to be no difference between the two methods of extending the uterine incision in amount of blood lost or in the rate of extension of the incision into the lateral uterine vessels when they were compared and correlated by the stage of labor.39 However, a recent investigation revealed a greater risk of subsequent blood transfusion in women whose incision was extended sharply compared to those extended bluntly.40 When blunt dissection is used, an upward curve of the incision may be created by the surgeons placing their thumbs on the patient's anterior superior iliac spines and index fingers in the uterine incision. By keeping the hand in this position, the incision is pulled open in an arc.
Intentional extension of the low-transverse incision is necessary in 1–2% of cases.40 Typically, the extension of the low transverse incision is performed by creating a low vertical incision in the midline, T-extension of the uterine incision, or creating a vertical incision at the lateral aspect of the uterine incision, a J-extension. These extensions are commonly performed for malpresentations, poorly developed lower uterine segment, or deep transverse arrest.40 When performed, extensions of the low-transverse incision are associated with increased incidence of maternal blood loss, broad ligament hematoma, and uterine artery laceration compared with low-segment transverse incisions that do not require extension.
The low-vertical uterine incision is made parallel to the longitudinal axis of the uterus in the midline, with care being taken to remain below the contractile portion of the uterus and within the thin lower uterine segment. Other than the direction of the incision, technical aspects are carried out as described for the low-transverse uterine incision. Studies have shown that there is no increased risk of uterine rupture in patients with this type of incision compared with the low-segment transverse incision as long as the incision remains primarily in the thin lower uterine segment.41
Placenta previa
Placenta previa can be associated with heavy blood loss, and it is important that a senior doctor is present at delivery. Assessment of the placental site and cord insertion by ultrasound scan prior to the operation is helpful in planning the surgical approach on reaching the placenta: blunt dissection should follow preferably gently pushing the placenta aside to access the membranes, but it may have to be entered digitally and the cord insertion should be avoided and the cord clamped quickly after delivery of the baby.27
Upper uterine segment incision
A classical uterine incision is made by incising the uterus parallel to the longitudinal axis of the uterus through the contractile portion of the myometrium. Indications for classical uterine incision include situations in which the lower uterine segment is not adequately developed to accommodate a low-transverse or a low-vertical incision; cases of abnormal fetal lie such as back-down transverse lie, in which the low-transverse or low-vertical incision will not allow the operator adequate access to the fetus for manipulation and delivery, or when myomas or uterine abnormalities distort the uterus in such a way as to make a low transverse incision inadvisable.
Delivery of the fetus – cephalic presentation
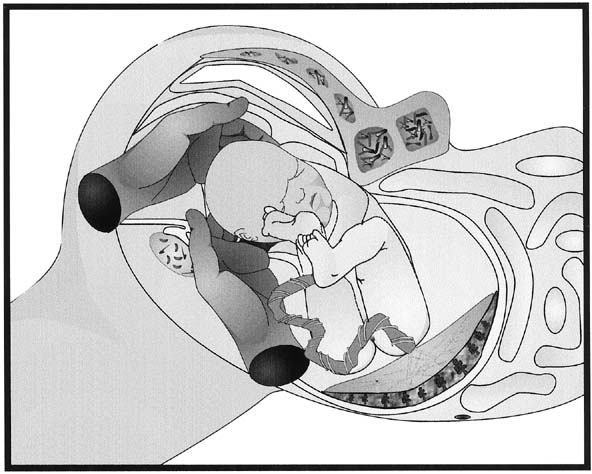
After the uterine incision has been made, the fetal membranes, if still intact, are ruptured carefully. If the fetus is in a noncephalic presentation, leaving the membranes intact until the fetal feet or head can be moved into the uterine incision will increase the ease of delivery. When the fetus is in a cephalic presentation, delivery is performed by the surgeons placing their dominant hand into the lower uterine cavity and elevating the fetal head into the uterine incision (Fig. 5). The Doyen retractor should then be removed. If the fetus is not in an occipito-transverse position, rotating the head into this position will allow the fetal neck to flex laterally around the upper portion of the incised myometrium and deliver into the wound with the aid of fundal pressure. Each shoulder should be gently delivered in turn followed by the trunk of the baby. Facilitating the delivery by the assistant using fundal pressure is important as the operator should apply minimal traction to the baby’s head; neck and brachial plexus injuries are not confined to over-zealous traction at vaginal deliveries.27 If the head is high and delivery is difficult, the Wrigley’s forceps or Kiwi ventouse cup can be applied to gently guide out the baby’s head. When the fetal head is impacted in the maternal pelvis, such as in deep transverse arrest or cesarean at full dilatation, there are a number of options to assist with delivery of the fetal head. The surgeon can place a hand in the lower uterine segment in the standard fashion to cup the head and wait until the uterus relaxes before trying to disengage it. Trying to disimpact the head whilst the uterus is contracting is unlikely to work, will risk extension of the uterine angles, will promote continued uterine tone, and may cause fetal trauma. If waiting does not help, the anesthetists can relax the uterus using a tocolytic such as terbutaline or glyceryl trinitrate GTN. If this does not work, an assistant can place a sterile, gloved hand into the vagina from the introitus and disengage the fetal head from below but again this should not be performed while the uterus is contracting (Fig. 6).
| Fig. 5. Extraction of the fetal head. The surgeon's dominant hand is placed into the uterine incision so that the back of the hand is against the inside of the lower uterine segment and the fingers cup the fetal head. Firm, gentle traction is used to elevate the fetal head toward the incision. The fetal head may then be rotated to an occiput anterior position and delivered through the uterine incision with the assistance of fundal pressure. Courtesy of R Preston McGehee MD. |
After the infant is delivered, it should be quickly dried and then after a short time the cord should be doubly clamped and cut. Depending on the condition of the infant it can either be handed straight to its mother for skin–skin contact, or, if needed it can be handed to the relevant personnel who have been assigned to care for the newborn. The baby should not be lifted up before the cord is clamped, and a time delay to clamping the cord of about a minute, to allow fetal transfusion, should be facilitated where possible.
Delivery of the fetus – breech presentation and transverse lies
In breech cesarean, if the legs are extended the operator’s right hand should be cupped around the bottom and the breech delivered by lateral flexion while the assistant exerts fundal pressure. Alternatively, a foot (recognized by the heel) can be held and the legs delivered first. In either situation the fetal back should be kept anterior, and completion of the delivery is again achieved by fundal pressure with minimal traction: as in the vaginal breech delivery, the shoulders should be delivered with gentle rotation, and a modified Maurcieu Smellie Veit technique can then be used to facilitate delivery of the head.27 If the baby is transverse, a foot should be identified and the baby delivered as breech. In this circumstance, leaving the membranes intact for as long as possible will facilitate the internal rotation of the baby.
Delivery of the placenta
Attention is now turned to the delivery of the placenta. Spontaneous delivery of the placenta, when assisted with uterine massage, 5 IU of intravenous oxytocin and gentle traction on the umbilical cord, is associated with a lower rate of postpartum endomyometritis and maternal blood loss compared with manual extraction.42, 43, 44 Infrequently, the placenta does not separate despite the uterus being well contracted and manual removal is required. Manual removal carries higher risks of hemorrhage and infection, and therefore the operator should guard against impatience and certainly not perform manual removal while the uterus is not contracting, as this will increase blood loss considerably. Any bleeding sinuses on the uterus can be compressed using Green–Armitage clamps while awaiting placental separation. In cases of morbidly adherent placenta (placenta accreta), there are several management options: first, if the placenta has not been breached during uterine entry and delivery of the baby and no placental separation has occurred, the placenta may either be left in situ and the patient managed conservatively, or, second, a hysterectomy may be preferred (depending on the preoperative discussion with and consent by the patient).
Once the placenta has been delivered, the uterine cavity should be checked to ensure it is empty and the uterus may be either exteriorized or left in situ to be repaired. Blood loss is not significantly different with either method.45 Exteriorization of the uterus does allow for better visualization of the adnexal structures and increases the ease with which tubal ligation can be performed but produces more discomfort in women having regional blockade.45
A broad spectrum antibiotic such as co-amoxiclav 1.2 g or, if penicillin allergic, clindamycin 600 mg intravenously should be given to all women at the time of cesarean section after delivery of the baby and placenta. If there is extensive hemorrhage from the placental bed after the placenta has been removed, a number of techniques can be used to help control the bleeding, these include local infiltration with uterotonics, under running the bleeding areas with sutures, local pressure with a Rusch balloon, or embolization by intervention radiology. If the bleeding is due to atony then a B-Lynch compression suture may help. Hysterectomy remains an option in case of failure of these latter measures or with catastrophic bleeding.
Uterine closure
Closing the uterus after cesarean section is best performed with a double layer technique. NICE supports this practice46 as studies have found a four to six-fold increase in the risk of uterine rupture in women who had a single layer closure (Fig. 7) in their previous pregnancy.47, 48, 49, 50 The recent CAESAR study in the UK which compared single and double closure did not look at long-term outcomes.51
Whether single or double layer closure is used, suture material should be of a short-term absorbable type52 (such as polyglycolic acid or polyglactin) as the uterus involutes postnatally and sutures loosen, to avoid loops of thread being present in the pelvis for any longer than necessary. Both uterine angles should be identified, sutured and tied securely.
For two-layer closure the first layer includes the deep myometrial edge with minimal decidua. A continuous locking technique is hemostatic and equally distributes the tension, making the suture less likely to cut through (especially useful with friable or thin lower segments). However, continuous suturing of the uterus being more hemostatic may reduce blood supply.53 The second layer completes the myometrial approximation and hemostasis. Locking this layer is not necessary but extra hemostatic sutures may be required if bleeding persists. The second layer effectively buries the first layer but this cosmetic effect is not its purpose which is to maintain the scar integrity and prevent future deficiency. A first layer that is ‘clumsy’ thick and including large chunks of deciduas tends to promote the concept that the second layer is designed to ‘hide it ’ which is often achieved by picking up adventitia from below and above the uterine incision. Such tissue approximation is illogical, can cause bleeding, and can hitch the bladder up towards the incision, making future surgery more treacherous27 (Fig. 8).
|
Decidual exclusion is very important in order to avoid endometrial inversion at the scar site, as this is believed to be the cause of incomplete scar healing. Alfred Wainorek (1967) conducted a study to examine the relationship between type of scar deformity and the suturing method used. He performed hysterographic examination on 270 patients who had previously undergone cesarean delivery, and evaluated the scar on the basis of depth, shape and size. He showed that the severity of scar defect was in direct proportion to the number of operations performed. He found dye penetration deep into the wall in two cases, where in both, inclusion of the decidua in the suture material was clearly recorded in the operative notes. He concluded that the best X-ray pictures with no visible scar defect were obtained when interrupted suture was applied through the myometrium, and when the decidual exclusion approach was adopted.54
Classical uterine incisions are much thicker and they are normally repaired in three layers. The principle to remember is that the dead space needs to be obliterated to achieve hemostasis and reduce the chance of hematoma formation. Sutures should be interrupted and absorbable. The final suture layer is best achieved with a monofilament inert fine continuous locking suture to the serosa to minimize adhesion formation.
After the uterus is closed attention should be turned to ensuring that the operative field is hemostatic, with special attention given to the uterine angles, uterine incision and the raw area of peritoneal reflection below the incision adjacent to the bladder. The paracolic gutters should be checked and cleaned; the tubes and ovaries should be identified to ensure normal anatomy. Hemostasis of the peritoneal edges, the rectus muscles and the underside of the sheath should then be checked and secured by either suture ligation or electrocoagulation of bleeding points.
Closing and drains
There is no advantage to closure of the visceral or parietal peritoneum, this has also been examined in the CAESAR trial and no significant difference was found between the two arms (closure vs. nonclosure of the peritoneum) in terms of maternal infectious morbidity.51 When repaired with suture, the peritoneum undergoes more inflammation and scarring in animal models.55 Operating time and postoperative analgesia requirements are reduced in patients who do not undergo closure of the visceral and parietal peritoneum. There is also a decrease in adhesions found at repeat operation when the visceral and parietal peritoneum is not closed.
The use of drains has also been evaluated by the CAESAR trial;51 there was a higher risk of maternal infectious morbidity associated with the liberal versus restricted use of subrectus sheath drain (20.8% versus 15.6%). If a drain is needed in the peritoneal cavity, a soft large-bore nonsuctioned drain such as a Robinson drain is suitable. If the sheath requires drainage, then a Redivac drain may be used but in such cases the parietal peritoneum should be closed to avoid direct communication of the suction drain with the abdomen contents.27
Fascial closure in a Pfannenstiel incision is performed with a continuous synthetic absorbable stitch. In patients who have undergone more than one laparotomy through the same scar, or in patients who are at increased risk of fascial separation or dehiscence such as diabetic patients or patients who are on corticosteroids, the use of a synthetic delayed absorbable suture such as polydioxanone56 or a permanent monofilament suture such as prolene may be preferable because of its ability to maintain suture strength for a longer period of time.57 For the closure of a vertical fascial incision, a continuous unlocked running delayed absorbable or permanent suture should be used. Whenever sutures are placed within the fascia, they need to be secure but not overly tight as over-tightening causes postoperative pain: reverse locking one or two sutures at even distances across the wound can help to distribute tension. It is also important to remember that a 10-mm zone of collagenolysis occurs surrounding the incision; therefore, sutures should be placed more than 1 cm from the fascial edge to achieve maximal wound strength and to avoid hernia formation.58
The subcutaneous tissue may be closed with an absorbable suture in women with more than 2 cm of subcutaneous fat or if a previous scar has been excised in order to minimize the risk of wound hematoma and infection.27 In slimmer patients, closing this layer has not been associated with decreased rates of superficial wound disruption in several studies.59 The point of this layer is to close the dead space and support the skin layer, so Scarpa’s fascia should be deliberately included in it.27 The skin should then be closed with a subcuticular stitch. Subcuticular stitches have been associated with less immediate postoperative pain and are more cosmetically appealing at 6 weeks when compared to the stapling device.59
Postoperative care
There is little literature to support any specific postoperative regimen in postcesarean patients; however, common sense and extrapolation of data from other postlaparotomy patients allow for the development of a rational plan of care. Most cesarean sections are relatively uncomplicated, and in these patients, care should be given according to the needs dictated by the method of anesthesia and any obstetric or medical complications present, while maximizing normality, skin–skin contact of mother and baby and other midwifery issues such as initiating breastfeeding.
In the first hour after an uncomplicated cesarean section, the patient should be monitored closely in a recovery area where urine output, pulse, blood pressure, respirations, and any evidence of bleeding can be closely observed; if the patient remains stable and without complication, she may then be transferred to the postpartum ward. Once any nausea has abated, the patient should be encouraged to take fluids orally and she can eat when she feels hungry. Early institution of feeding in the postsurgical patient with minimal intraoperative bowel manipulation does not increase the incidence of postoperative ileus.60, 61
All women should be given compression stockings and kept well hydrated after cesarean section to minimize the risk of thromboembolism. Heparin thromboprophylaxis should be given if there are risk factors and in accordance with local guidelines. Early ambulation should also be encouraged. Getting the patient out of bed as soon as regional anesthesia has worn off or as soon as she has recovered from general anesthesia will decrease the incidence of pulmonary complications such as atelectasis and pneumonia, and the incidence of thrombotic complications. Encouragement of deep breathing and coughing will also help prevent collapse of alveoli in the lung and subsequent infection.
In the uncomplicated patient with adequate urine output, the catheter should be removed 12 hours postoperatively unless this would be in the evening in which case it should wait to be removed until the following morning. Encouraging mobilization will also facilitate the removal of bladder catheters, therefore decreasing the incidence of catheter-associated urinary tract infections.
Routine laboratory studies are probably unnecessary in most postcesarean patients who have no unexpected symptoms. However, a single hemoglobin determination on postoperative day 2 is probably reasonable to screen for significant anemia. Most postpartum patients with asymptomatic anemia respond well to oral iron therapy.
The wound should be cared for in the standard manner, with occlusive dressings removed on the first postoperative day and the wound examined daily during the hospitalization for evidence of infection, seroma, or hematoma. The patient may be discharged when she is able to care for herself and her newborn. Many patients are ready to leave the hospital by postoperative day 2 or 3. Discharge instructions should include patient education concerning expectations on activity level, lochia, breastfeeding or milk suppression, contraception, and newborn care, and the plans for suture removal.
SPECIAL SITUATIONS
Vaginal birth after cesarean section
The vaginal birth after cesarean section (VBAC) rate is defined as the number of vaginal births to women with a previous cesarean section per 100 deliveries to women who had a previous cesarean delivery. New evidence is emerging to state that VBAC may not be as safe as its originally thought.62 In addition, fear of medico-legal litigations have led to a decline in the number of clinicians offering and women accepting planned VBAC in the UK and North America.63 There are no randomized controlled trials comparing planned VBAC with planned elective repeat cesarean delivery (ERCD) and this may be an unrealistic aspiration. The agency for health quality and research in the US ranks the current available evidence related to VBAC as level two or three, and acknowledges considerable heterogeneity in the reported outcomes and poor comparability between the treatment groups.
In the USA in the late eighties and nineties there was a drive to increase VBAC and the rate rose from 6.6% in 1985 to 28.3% in 1996,64 but enthusiasm waned as complications arose and by 2006 it had fallen to just under 9%64 (Fig. 9)
Fig. 9. Rates of VBAC in the US, 1981–200664 |
In the UK, we now have a better idea of the overall VBAC rate. The National Sentinel Cesarean Section Audit Report, October 2001, found that of all mothers who fell pregnant following previous cesarean section, 33% achieved a vaginal delivery. Although this can seem low when set against the 70–85% VBAC rates found in most research studies, these studies explore the rates of success in women who embark on attempting VBAC and in routine care many women are not offered or decline the option of VBAC.17
Several authors have attempted to predict which patients are more likely to undergo successful VBAC by various historic parameters and the physical examination at the time of admission for delivery. Jakobi and colleagues65 found that: previous cesarean section performed for a nonrepetitive indication such as breech presentation; a history of a previous successful VBAC; a fetal station of one centimeters or less above the ischial spines; unruptured membranes at admission; more than 2 years since the cesarean delivery and dilation of 4 cm or more at admission were all positively correlated with increased likelihood of successful VBAC. A history of previous cesarean section for arrest of labor, diabetes mellitus, hypertension in pregnancy, induction of labor, oxytocin use in labor, and meconium-stained liquor were significantly associated with an increased risk of unsuccessful VBAC. Using these criteria retrospectively, the authors would have correctly predicted the success of a trial of labor in more than 94% of candidates but would have correctly predicted failure of trial of labor in only 33.3% of candidates.
Flamm and Geiger66 examined similar data to develop a scoring system in an attempt to predict the success of trial of labor. These authors found that maternal age younger than 40 years, BMI less than 30, white ethnicity, indication other than failure to progress, cervical effacement of more than 75% on admission, and cervical dilation of more than 4 cm at admission were all significantly correlated with increased success of trial of labor. When these factors were weighted and placed in a scoring system in an attempt to predict the success of attempted VBAC, the authors found that as the number of these factors increased, the likelihood of successful trial of labor increased. Patients with only one or two of these characteristics had a 49–59% success rate, whereas patients with four or more of these characteristics had a greater than 90% success rate. The ability to more accurately predict the likely success of trial of labor is clinically useful, because there is increased maternal morbidity is associated with a failed trial of labor.67 These women have an increased risk of operative injury, infectious morbidity and uterine rupture.
Perimortem cesarean section
One of the first indications for cesarean section was for the delivery of the fetus in the case of maternal death. Currently, the performance of a rapid cesarean delivery in the event of sudden maternal cardiac arrest is performed to assist in maternal resuscitation. If performed quickly, it can be lifesaving for the fetus68 but this is not the primary aim. In the event of maternal cardiac arrest a cesarean section should be initiated within 4 min of cardiac arrest, with the goal of delivering the fetus within 5 min of onset of cardiac arrest, where possible, but while resuscitation continues it is still worth emptying the uterus to assist in maternal resuscitation even if this timeframe has been overstepped.
COMPLICATIONS
Maternal mortality
As anesthesia and operative techniques have improved, cesarean section has become an increasingly safe and common procedure; however, the obstetrician must always bear in mind that the abdominal delivery of an infant is still a major operative procedure and can be associated with significant maternal mortality and morbidity. Maternal mortality after cesarean section has been estimated to be between 5.81 and 6.1 per 100,000 procedures.69, 70 Most of these deaths result from the complications that led to the cesarean section, but a few are as a direct consequence of the procedure. In general, the complications associated with cesarean section are similar to those observed after any laparotomy, with the exception of an increased incidence of endomyometritis. Complications may be divided into those encountered intraoperatively and those encountered postoperatively.
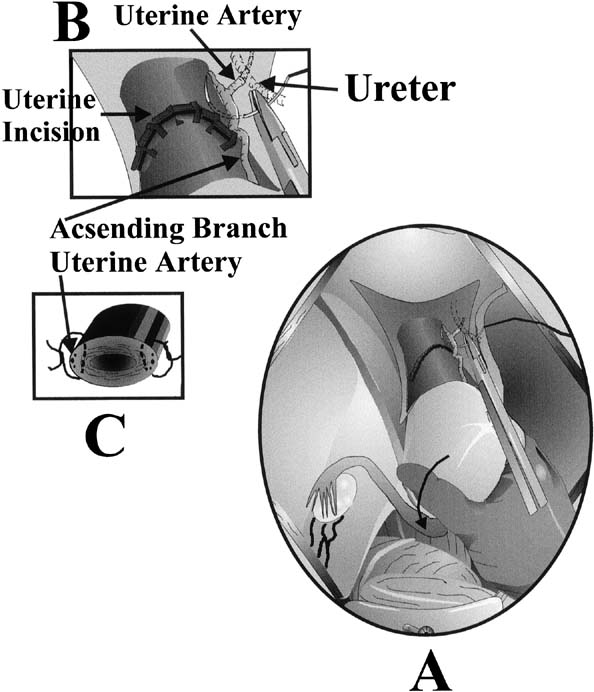
Potential intraoperative complications include uterine hemorrhage from the surgery itself but also from inadvertent uterine angle extensions, atony or placenta pathology. Uterine atony continues to be the major cause of hysterectomy at the time of cesarean section71, 72 and is best managed with uterotonics and massage the uterus while repairing the uterine incision. Additional uterotonics that can be given include ergometrine (not with hypertension), Hemabate (not with asthma), or misoprostol. Fluid resuscitation and blood transfusion should be instituted as clinically indicated. If the atony persists a B-Lynch compression suture72 can be inserted (Fig. 10) and other options for hemorrhage control include bilateral uterine artery ligation, with sutures placed to obliterate both the ascending uterine artery at the level of the lower uterine segment and its anastomosis with the ovarian artery at the uterine cornua (Fig. 11). This will control bleeding from an atonic uterus in 75% of cases.71 Localization of the uterine artery can be facilitated by careful palpation of its course along the lateral edge of the uterus. The surgeon should then pull the engorged uterine veins laterally into the broad ligament and away from the operative field to avoid laceration of these veins during suture placement. Isolating the uterine artery with a Babcock clamp is often helpful in these cases. Care should be taken to avoid incorporating the ureter in the ligature.73 In cases that do not respond to bilateral uterine artery ligation, bilateral hypogastric artery ligation is no longer recommended because it is successful in less than 50% of patients and training opportunities are not readily available, thus an experienced operator is unlikely. Interventional radiology is more commonly used now and requires access via the internal iliac artery. In patients who do not respond to these management strategies, hysterectomy is appropriate and life saving.74
In cases of hemorrhage not caused by uterine atony, careful exploration of the uterus for possible retained placental fragments and exploration of the operative field for unrecognized lacerations should be performed. Placental fragments may be removed manually or with a swab. Areas of placental adherence should be examined for evidence of placenta accreta. Genital tract lacerations should be identified, isolated, and closed in a hemostatic manner.
Urinary tract injuries
Injury to the urinary tract is a relatively rare complication of cesarean delivery. The incidences of bladder and ureteral injury are 0.3% and 0.1%, respectively.73, 75 Bladder injuries are more common with a history of previous cesarean section. The most common site for bladder injury during cesarean delivery is at the dome of the bladder. Laceration of the bladder should be evaluated by first ensuring that the trigone and ureters are not involved. This may be accomplished by direct visualization of the ureters through the cystotomy. If the trigone is not involved and the ureters are functioning, the cystotomy can be closed in two layers using an absorbable suture. Whenever there is a possibility of inadvertent cystotomy at the time of cesarean section, this can be evaluated by distending the bladder with sterile saline through the Foley catheter and observing the operative field for the appearance of the fluid.
Ureteral injury is less common than injury to the bladder. If there is concern during the operative procedure that the ureter has been compromised, the situation should be thoroughly evaluated and consultation with a specialist urologist is needed.
Gastrointestinal tract injury
Injury to the bowel at the time of cesarean section is exceedingly rare. An incidence of less than 0.1% has been reported.75 This low incidence is caused by the displacement of the bowel out of the operative field by the enlarged, gravid uterus. The risk of bowel injury is increased in patients with previous abdominal surgery or intra-abdominal adhesions. Injury is usually obvious because of the appearance of bowel contents in the surgical field. These injuries should be quickly identified and isolated to minimize contamination of the peritoneal cavity. Injury to the small bowel can be primarily repaired with a two-layer closure using a delayed absorbable suture. The closure should be performed at 90 degrees to the bowel lumen to decrease its constriction. Larger lacerations of the small bowel or multiple lacerations may require resection of a length of bowel, and a specialist surgeon should be called for this and for any injuries to the large bowel which may require a defunctioning colostomy as well as primary closure.
Wound infections
Wound infections occur at a rate of approximately 7% after cesarean section when prophylactic antibiotics are not given;76 this incidence is reduced to 2% with the use of prophylactic antibiotics.77 Wound infections that occur after cesarean section include endomyometritis, pelvic abscess, incisional abscess, and wound cellulitis. The antibiotic of choice for each infection depends on the location of the infection and the suspected pathogen. Antibiotic therapy should be instituted empirically and adjusted as needed based on culture results. For pelvic abscesses treatment includes drainage of pus and broad-spectrum antibiotics including anaerobic coverage are required; for superficial wound infections, simply opening the incision and draining the infectious source usually alleviates the problem in patients who do not have signs of systemic infection. Superficial wound cellulitis can usually be treated using penicillinase-resistant penicillin.
Endomyometritis
Endomyometritis complicates up to 80% of cesarean sections performed after the membranes have been ruptured for more than 6 hours in patients who are not administered antibiotic prophylaxis78 and 30% in patients with intact membranes. The incidence has been shown to be high in patient populations of lower socioeconomic status, in patients who have had six or more vaginal examinations during labor and in patients with longer duration of rupture of membranes.79 The rate of uterine infection can be reduced to 5% or less with the use of prophylactic antibiotics given at the time of cord clamp.80 A single dose of a broad-spectrum antibiotic is relatively inexpensive and effectively decreases the infection rate.
Incomplete scar healing
A deficient cesarean section scar has become one of the recognized complications associated with this type of operation. The exact cause and mechanism of incomplete healing of the scar and whether this leads to functional uterine deficiency is not well understood. As more women undergo transvaginal ultrasonography, the morphology of section scars has come under increasing scrutiny. Currently it is not known if the appearances of a cesarean section scar using ultrasound translate into any relationship to the functional integrity of the uterus, risk of ectopic pregnancy, pathological placentation, uterine rupture or performance in labor. There is an urgent need to explore this relationship so that we can understand how to interpret images of the uterus following cesarean section and the implications of various types of scar on patient management. Studying the natural history of cesarean section scar in the pregnant state and following it to delivery could achieve this, and analyzing whether certain scar features on ultrasound scan can lead to a successful vaginal delivery or a repeat cesarean section.
There is growing evidence to suggest that complete healing of the previous cesarean scar and myometrial thickness at the lower uterine segment are important factors in achieving uneventful pregnancy outcome, whether by ERCD or VBAC.81 Over the past 10 years there have been multiple attempts to study these factors by ultrasonography and different reports have been published in the literature regarding the prevalence and the clinical significance of incompletely healed cesarean scars.82 VBAC is considered to be a safe alternative to ERCD when the risk of uterine rupture in minimal, and as mentioned above several factors must be examined before considering this option.65 However, to better assess the risk of uterine rupture, some authors have proposed sonographic measurement of the cesarean scar and the thickness of the lower uterine segment, assuming that certain scar features and cut-off values are correlated with the uterine scar defect.83 These ultrasound measurements may increase the safety of labor after cesarean section because they provide additional information on the risk of uterine rupture. Recent evidence also states that transvaginal ultrasound scan is a reliable and reproducible method for measuring the myometrial thickness near term,83 and concludes that this value may serve as a predictor of uterine scar defect in women contemplating VBAC. However, an ideal cut-off value cannot be recommended yet, underlining the need for further well-designed prospective and longitudinal studies during pregnancy.
Uterine scar rupture
In spite of the recent advances in modern obstetric practice, rupture of the pregnant uterus is still one of the most life-threatening complications of pregnancy, and it is associated with high rates of maternal morbidity and fetal morbidity and mortality.84 Previous cesarean section has been shown to be the most important risk factor,85 but no difference in the outcome of labor with regard to uterine rupture, between women with and those without previous cesarean section has also been reported.84 The most commonly quoted scar rupture for lower segment cesarean section (LSCS) is 0.5%, or one in 200.86
The incidence of uterine rupture may be increased in patients with a previous single-layer closure.47, 48, 49, 50, 87 However, in patients with a previous classic uterine incision, the risk of uterine rupture may be as high as 9%, with one-third of these occurring before the onset of clinical labor.88 In one study on preterm cesarean sections the risk of uterine rupture was similar for both types of uterine incision.89 In the event of uterine rupture, fetal mortality and morbidity are highly significant. When a classic uterine incision ruptures, the fetal mortality is in excess of 50%, compared with 12% in the event of rupture of a prior low transverse incision.90
Future fertility
Women who have had a cesarean section are less likely than those who have had a vaginal delivery to have further children, but it is hard to separate out the reasons for this in terms of voluntary family planning, consequences of the indications for the cesarean and due to the actual operation. There appears to be a lesser desire for further children as well as a decreased ability to conceive.91 There is a concept developing that abnormal scar healing may affect endometrial receptivity as well as uterine contractility due to abnormal signaling, and this is believed to compromise the uterine favorability for successful implantation.92 However, no hard evidence has been established to support this. Ben-Nagi92 recently hypothesized the possibility of a link between altered uterine immunobiology, LSCS and embryo implantation. They studied the effect of LSCS on the endometrium in premenopausal women with history of LSCS; they took endometrial samples from both cesarean scar site and posterior uterine wall, and compared the results with similar samples obtained from women who had spontaneous vaginal delivery (SVD). The most significant difference found was fewer leukocytes and less vascularization at the scar site, than in the endometrium of the unscarred uterus. In addition, they found a delay in endometrial maturation at the scar site, and this delay might have been a result of disruption in steroid receptor expression, leading to abnormal response to estrogen and progesterone. These studies raised considerable interest and emphasized that endometrium at LSCS scar site may not cycle in synchrony with the rest of endometrium in the uterine cavity; however, further studies are required to explore this hypothesis.
CONCLUSION
Over the past several decades the incidence of cesarean delivery has increased dramatically. Although the operation continues to become safer, the incidence of maternal mortality and morbidity is still significant. Continued efforts on the part of the obstetrician must be made to ensure that cesarean deliveries are not performed for inappropriate indications and that each woman is counseled carefully according to her individual characteristics. Every effort should be made to allow the patient to deliver vaginally when fetal and maternal statuses are reassuring. When a cesarean section is indicated and embarked upon the preparation should be as carefully planned as time allows, and the surgical technique should adhere to good surgical principles. Continuing research is required to evaluate specific techniques of the cesarean operation further, particularly relating to uterine closure and scar integrity in the longer term.
REFERENCES
Gabert HA, Bey M: History and development of cesarean operation. Obstet Gynecol Clin North Am 15:591, 1988 |
|
Speert H: A Pictorial History of Gynecology and Obstetrics. Philadelphia, Davis, 1973 |
|
Katz VL, Cefalo RC: History and evolution of cesarean delivery. In: Phelan JP, Clark SL, (eds): Cesarean Delivery. New York, Elsevier, 1988 |
|
Boley JP: The history of caesarean section. CMAJ 145:319, 1991 |
|
Horley JMG: Cesarean section. Clin Obstet Gynecol 7:529, 1980 |
|
Betrán AP, Merialdi M, Lauer JA et al: Rates of cesarean section: analysis of global, regional and national estimates. Paediatr Perinat Epidemiol. 2007 Mar; 21(2): 98-113 |
|
OECD Health Data 2010 - Version: June 2010 |
|
Clark SC, Taffel SM: State variation in rates of cesarean and VBAC delivery: 1989 and 1993. Stat Bull Metrop Insur Co 77:28-36, 1996 |
|
Notzon FC, Cnattingius S, Bergsjo P et al: Cesarean section delivery in the 1980s: International comparison by indication. Am J Obstet Gynecol 170:495-504, 1994 |
|
Norton FC: International differences in the use of obstetric interventions. JAMA 283:3286, 1990 |
|
Pollard JK, Capeless EL: Cesarean deliveries at a university hospital: Analysis of rates and indications. Am J Perinatol 14:245, 1997 |
|
Ziadeh SM, Sunna FI: Decreased cesarean birth rates and improved perinatal outcome: A seven-year study. Birth 22:144, 1995 |
|
US Department of Health and Human Services: Healthy Children 2000. DHHS Pub. No. HRSA-M-CH 91–2. Washington, DC: US Government Printing Office, 1991 |
|
Hogle KL, Kilburn L, Hewson S et al: Impact of the international term breech trial on clinical practice and concerns: A survey of centre collaborators. J Obstet Gynaecol Can 25:14-16, 2003 |
|
American College of Obstetrics and Gynecology. Technical Bulletin No. 218. December 1995 |
|
Renou P, Chang A, Anderson I et al: Controlled trial of fetal intensive care. Am J Obstet Gynecol 126:470, 1976 |
|
Parliamentary Office of Science and Technology. Cesarean sections. Postnote 2002;(184): 1–4 [www.parliament.uk/ post/pn184.pdf]. |
|
Kelso AM, Parsons RJ, Lawrence GF et al: An assessment of continuous fetal heart rate monitoring in labor. Am J Obstet Gynecol 131:526, 1978 |
|
Haverkamp AD, Orleans M, Langendoerfer S et al: A controlled trial of the differential effects of intrapartum fetal monitoring. Am J Obstet Gynecol 134:399, 1979 |
|
MacDonald D, Grant A, Sheridan-Pereira M et al: The Dublin randomized controlled trial of intrapartum fetal heart rate monitoring. Am J Obstet Gynecol 152:524, 1985 |
|
Leveno KJ, Cunninhgam FG, Nelson S et al: A prospective comparison of selective and universal electronic fetal monitoring in 34,995 pregnancies. N Engl J Med 315:615, 1986 |
|
Weiss PM, Balducci J, Reed J et al: Does centralized monitoring affect perinatal outcome. J Matern Fetal Med 6:317, 1997 |
|
Ragupathy K, Ismail F, Nicoll AE. The use of STAN monitoring in the labour ward. J Obstet Gynaecol 2010 30:5, 465-469 |
|
Ojala K, Makikallio K, Haapsamo M, Ijas H, Tekay A. Interobserver agreement in the assessment of intrapartum automated fetal electrocardiography in singleton pregnancies. Acta Obstetr Gynecol Scand 2008. 87:536–540 |
|
Neilson JP. Fetal electrocardiogram (ECG) for fetal monitoring during labour. Cochrane Database Syst Rev 2006, Issue 3. Art. No.: CD000116. DOI: 10.1002/14651858.CD000116.pub2 |
|
American College of Obstetrics and Gynecology. Criteria Set Number 13. December 1995 |
|
Story L, Paterson-Brown S: Cesarean deliveries: indications, techniques and complications. Chapter 10: Best Practice in Labour and Delivery, ed. R. Warren and S. Arulkumaran. Published by Cambridge University Press 2009. |
|
Lucas DN, Yentis SM, Kinsella SM, et al. Urgency of cesarean section: a new classification. J R Soc Med 2000; 93: 346–50 |
|
Report of the National Confidential Enquiry into Perioperative Deaths 1992/1993. London: NCEPOD, 1995 |
|
Joy S, Contag SA. eMedicine Specialties > Obstetrics and Gynaecology > Labor and Delivery: Cesarean delivery 2010 |
|
American College of Obstetrics and Gynecology. Committee Opinion No. 98. September 1991 |
|
Morrison JJ, Rennie JM, Milton PJ. Neonatal respiratory morbidity and mode of delivery at term: influence of timing of elective cesarean section. Br J Obstet Gynaecol. 1995 Feb; 102(2): 101-6 |
|
Tita AT, Landon MB, Spong CY et al. Timing of elective repeat cesarean delivery at term and neonatal outcomes. N Engl J Med 2009 Jan 8;360(2):111-20. |
|
RCOG, RCM, RCA, RCPCH. Safer Childbirth. Minimum Standards for the Organisation and Delivery of Care in Labour. London: RCOG Press, 2007. Available online at: http://www.rcog.org.uk/resources/ public/pdf/safer_childbirth_report_web.pdf (accessed 30 January 2008). |
|
Alexander JW, Fisher JE, Boyajiam M et al: The influence of hair removal methods on wound infections. Arch Surg 118:347, 1983 |
|
Greenmall MJ, Evans M, Pollack AV: Mid-line or transverse laparotomy? A random controlled clinical trial period Br J Surg 67:188, 1980 |
|
Franchi M, Ghezzi F, Raio L et al: Joel-Cohen or Pfannenstiel incision at cesarean delivery: does it make a difference? Acta Obstet Gynecol Scand 81:1040-1046, 2002 |
|
Hohlagschwandtner M, Ruecklinger E, Husslein P et al: Is the formation of a bladder flap at cesarean necessary? A randomized trial Obstet Gynecol 98:1089-1092, 2001 |
|
Rodriquez AI, Porter KB, O'Brien WF: Blunt versus sharp expansion of the uterine incision in low-segment transverse cesarean section. Am J Obstet Gynecol 152:971, 1985 |
|
Boyle JG, Gabbe SG: T and J Vertical extensions in low transverse cesarean births. Obstet Gynecol 87:238, 1996 |
|
Martin JN Jr, Perry KG Jr, Roberts WE et al: The case for trial of labour in the patient with a prior low-segment vertical cesarean incision. Am J Obstet Gynecol 177:144, 1997 |
|
Atkinson M, Owen J, Wren A et al: The effect of manual removal of the placenta on post-cesarean endometritis. Obstet Gynecol 87:99, 1996 |
|
McCurdy CM Jr, Magann EF, McCurdy CJ et al: The effect of placental management at cesarean delivery on operative blood loss. Am J Obstet Gynecol 167:1363, 1992 |
|
Khan GQ, John IS, Wani S et al: Controlled cord traction versus minimal intervention techniques in delivery of the placenta: A randomized controlled trial. Am J Obstet Gynecol 177:770, 1997 |
|
Wilkinson C, Enkin MW Uterine exteriorization versus intraperitneal repair at cesarean section Cochrane Database Syst Rev. 2000;(2):CD000085. |
|
National Institute of Clinical Excellence. Clinical Guideline 13 Cesarean Section. London: RCOG Press, 2004. Available online at: http://www.nice.org.uk/ nicemedia/pdf/CG013fullguideline.pdf (accessed 30 September 2010). |
|
Hauth JC, Owen J, Davis RO: Transverse uterine incision closure: One versus two layers. Am J Obstet Gynecol 167:1108, 1992 |
|
Tucker JM, Hauth JC, Hodgkins P et al: Trial of labour after a one or two-layer closure of a low transverse uterine incision. Am J Obstet Gynecol 168:545, 1993 |
|
Chapman SJ, Owen J, Hauth JC: One-versus two layer closure of a low transverse cesarean: The next pregnancy. Obstet Gynecol 89:16, 1997 |
|
Bujold E, Bujold C et al: The impact of a single-layer or double-layer closure on uterine rupture. Am J Obstet Gynecol 186:1326-1330, 2002 |
|
The CAESAR study collaborative group. Cesarean section surgical techniques: a randomised factorial trial (CAESAR). BJOG 2010; 117:1366–1376 |
|
Wound healing: Techniques and materials. In Visscher HC, (ed): Precis V: An Update in Obstetrics and Gynecology. Washington, American College of Obstetrics and Gynecology, 1994 |
|
Potter M, Johnston DC, Buffalo NY, et al. Uterine closure in cesarean section. Am J Obstet Gynecol 1954; 76 (4): 760-7 |
|
Wainorek A. Hysterography after c/s for evaluation of suturing technique. Obstet Gynecol 1967; 29 (2): 192-9 |
|
Wilkinson CS, Enkin MW: Peritoneal non-closure at cesarean section. Cochrane Database Syst Rev 2:CD000163, 2000 |
|
Ray JA, Doddi N, Regula D et al: Polydioxanone (PDS), a novel monofilament synthetic absorbable suture. Surg Gynecol Obstet 153:497, 1981 |
|
Wound healing: Techniques and materials. In Visscher HC, (ed): Precis V: An Update in Obstetric Cesarean section and Gynecology. Washington, American College of Obstetrics and Gynecology, 1994 |
|
Magann EF, Chauhan SP, Rodts-Palenik S et al: Subcutaneous stitch closure versus subcutaneous drain to prevent wound disruption after cesarean delivery: A randomized clinical trial. Am J Obstet Gynecol 186:1119-1123, 2002 |
|
Frishman GN, Schwartz T, Hogan JW: Closure of Pfannenstiel skin incisions. Staples vs. subcuticular suture J Reprod Med 42:627-630, 1997 |
|
Mangesi L, Hofmeyr GJ: Early compared with delayed oral fluids and food after cesarean section. Cochrane Database Syst Rev 3:CD003516, 2003 |
|
Miller DA, Diaz FG, Paul RH: Vaginal birth after cesarean: A 10-year experience. Obstet Gynecol 84:255, 1994 |
|
Landon MB, Hauth JC, Leveno KJ et al. Maternal and perinatal outcomes associated with a trial of labor after prior cesarean delivery. N Engl J Med 2004; 351:2581–9 |
|
Menacker F.Trends in cesarean rates for first births and repeat cesarean rates for low-risk women: United States, 1990–2003. Natl Vital Stat Rep 2005; 54:1–8 |
|
Kimberly D. Gregory, M.D., M.P.H.; Moshe Fridman, Ph.D.;Lisa Korst, M.D., Ph.D. Trends and Patterns of Vaginal Birth After Cesarean Availability in the United States. 2010 Abstract from: consensus.nih.gov/2010/vbac.htm |
|
Jakobi P, Weissman A, Peretz KBA et al: Evaluation of prognostic factors for vaginal delivery after cesarean section. J Reprod Med 38:729, 1993 |
|
Flamm BL, Geiger AM: Vaginal birth after cesarean delivery: An admission scoring system. Obstet Gynecol 90:907, 1997 |
|
McMahon MJ, Luther ER, Bowes WA Jr et al: Comparison of a trial of labour with an elective second cesarean section. N Engl J Med 335:689, 1996 |
|
Cummin RO, (ed): Advanced Cardiac Life Support. Dallas, TX, American Heart Association, 1994 |
|
Clark SL, Yeh SY, Phelan JP et al: Emergency hysterectomy for the control of obstetric hemorrhage. Obstet Gynecol 64:376, 1984 |
|
Chestnut DH, Eden RD, Gall SA et al: Peripartum hysterectomy: A review of cesarean and postpartum hysterectomy. Obstet Gynecol 65:365, 1985 |
|
Adrabbo F, Salah J: Stepwise uterine devascularization: A novel technique for management of uncontrollable postpartum hemorrhage with preservation of the uterus. Am J Obstet Gynecol 171:694, 1984 |
|
B-Lynch C, Coker A, Lawal AH et al: The B-Lynch surgical technique for the control of massive postpartum haemorrhage: an alternative to hysterectomy? Five cases reported Br J Obstet Gynaecol 104:372-375, 1997 |
|
Eisenkip SM, Richman R, Platt LD et al: Urinary tract injury during cesarean section. Obstet Gynecol 60:591, 1982 |
|
Nielsen TF, Hokegard KH: Cesarean section and intraoperative surgical complications. Acta Obstet Gynaecol Scand 63:104, 1984 |
|
Smith CV, Gallup DG: Management of urinary and gastrointestinal tract injuries. In: Phelan JP, Clark SL, (eds): Cesarean Delivery. New York, Elsevier, 1988 |
|
Faro S: Soft tissue infections. In: Gilstrap LC, Faro S, (eds): Infections in Pregnancy. New York, Wiley-Liss, 1990 |
|
Cunningham FG, Hauth JC, Strong JD et al: Infectious morbidity following cesarean section: Comparison of two treatment regimens. Obstet Gynecol 52:656, 1978 |
|
Magann EF, Dodson MK, Ray MA et al: Preoperative skin preparation and intraoperative pelvic irrigation: Impact on post-cesarean endometritis and wound infection. Obstet Gynecol 81:922, 1993 |
|
Chang PL, Newton ER: Predictors of antibiotic prophylactic failure in post-cesarean endometritis. Obstet Gynecol 80:117, 1992 |
|
Soper DE, Brockwell WJ, Dalton HP: The importance of wound infection in antibiotic failures in the therapy of postpartum endometritis. Surg Gynecol Obstet 174:265, 1992 |
|
Ozdemir I, Yucel N, Yucel O. Rupture of the pregnant uterus: a 9-year review. Arch Gynecol Obstet 2005; 272: 229–231 |
|
Chen H-Y, Chen S-J, H F-J. Observation of LSCS scars by transvaginal ultrasonography. Ultrasound Med Biol 1990; 16(5): 443-7 |
|
Jastrow N, Antonelli E, Robyr R et al. Inter- and intra-observer variability in sonographic measurement of the lower uterine segment after a previous Cesarean section. Ultrasound Obstet Gynecol. 2006 Apr; 27(4):420-4 |
|
Eden RD, Parker RT, Gall SA. Rupture of the pregnant uterus: a 53-year review. Obstet Gynecol 1986; 68: 671–674 |
|
Chazotte C, Cohen WR. Catastrophic complications of previous cesarean section 1990. Am J Obstet Gynecol 163:738–742 |
|
Enkin M, Keirse MJNC and Chalmers I (1994). Effective Care in Pregnancy and Childbirth, Oxford Medical Publications |
|
Rosen MG, Dickinson JC, Westhoff CL: Vaginal birth after cesarean: A meta-analysis of morbidity and mortality. Obstet Gynecol 77:465, 1991 |
|
Scott J: Avoiding labour problems during vaginal birth after cesarean delivery. Clin Obstet Gynecol 40:533, 1997 |
|
Halperin ME, Morre DC, Hannah WJ: Classical vs. low-segment transverse incision for preterm cesarean section: Maternal complications and outcome of subsequent pregnancies Br J Obstet Gynaecol 95:990, 1988 |
|
Flamm BL, Goings JR, Fuelberth NJ et al: Oxytocin during labour after previous cesarean section: Results of a multicenter study. Obstet Gynecol 70:709, 1987 |
|
Jolly J, Walker J, Bhabra K. Subsequent obstetric performance related to primary mode of delivery. Br J Obstet Gynaecol 1999; 106: 227–32 |
|
Ben-Nagi J, Walker A, Yazbek J et al. Effect of Cesarean Delivery on the endometrium. International Journal of Gynaecology and Obstetrics 2009; 106: 30-4. |