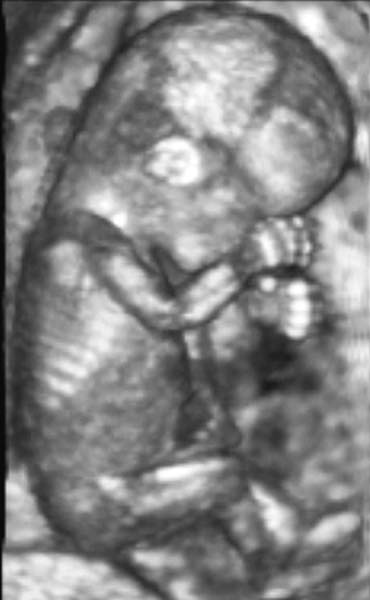
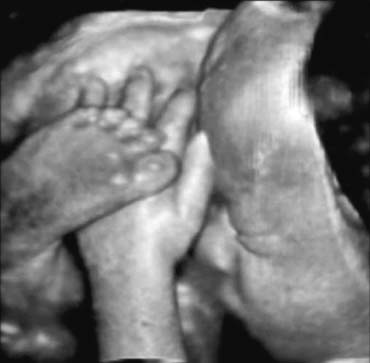
Diagnostic Obstetric Ultrasound
Authors
INTRODUCTION
The role of ultrasonography in obstetric practice has continuously evolved since its introduction more than 40 years ago. Indeed, it is difficult to imagine a modern obstetric practice without access to the information provided by real-time ultrasound. Using this imaging technique, clinicians can detect pregnancy as early as 3 weeks after conception, confirm or revise gestational age with reasonable accuracy, diagnose multiple gestation in early pregnancy, confidently diagnose fetal death at any gestational age, assess fetal well-being, evaluate amniotic fluid volume, and diagnose a broad variety of fetal malformations.
Advances in sonographic imaging in the past decade are attributable to both improved image resolution and to better clinical interpretation of ultrasound findings. It is reassuring that in human pregnancy no adverse bioeffects appear to be caused by diagnostic ultrasound.1 In this chapter, we discuss the basic content of an obstetric ultrasound examination, the use of ultrasound as a screening tool in obstetric patients, and some clinical applications of ultrasound. We also discuss three-dimensional (3D) ultrasound, an exciting new method of imaging, which may lead to further advances in diagnostic obstetric imaging.
OBTAINING FETAL IMAGES
Human pregnancy lasts an average of 280 days from the first day of the last menstrual period in patients with regular 28-day menstrual cycles. Both clinical and ultrasonographic gestational age are expressed using this standard.
The use of endovaginal ultrasound has greatly enhanced our ability to detect pregnancy early. The earliest ultrasonic evidence of pregnancy is the finding of a fluid-filled gestational sac with an echogenic border. A gestational sac should be visible at 4 weeks and 4 days, a yolk sac at 5 weeks, and a fetal pole with cardiac activity at 6 weeks.2 Using a vaginal probe, the gestational sac and fetal pole can be found earlier than with the abdominal approach. The endovaginal transducer eliminates the need for a full bladder before examination and significantly improves resolution in obese patients.
Crown–Rump Length
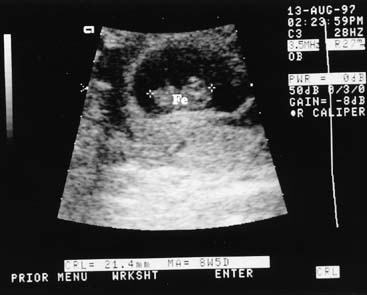
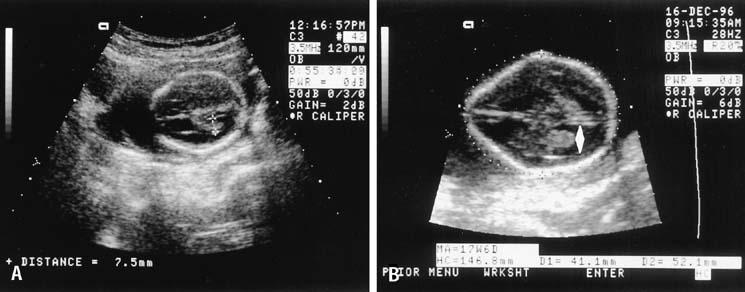
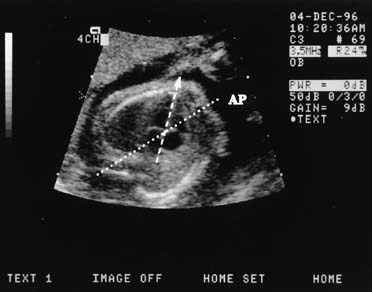
The crown-rump length (CRL) of the fetus is an accurate predictor of gestational age. A practical approximation is that fetal CRL in centimeters plus 6.5 equals gestational age in weeks. A 9-week fetus, for example, would have a CRL of 2.5 cm (Fig. 1). CRL has been promoted as the most accurate method of dating a pregnancy, but experience has shown that the average of the biparietal diameter (BPD), femur length, and abdominal circumference obtained before 20 weeks' gestation is comparable in accuracy.3
Biparietal Diameter
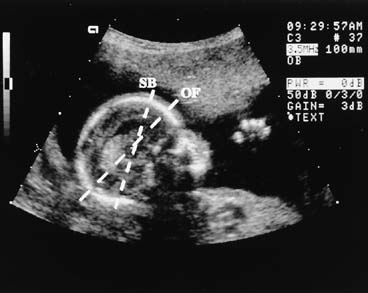
After the first trimester, CRL determination is not practical because of fetal posturing and size. From 14 weeks onward, the BPD is one of several useful estimators of fetal gestational age. The BPD is the largest transverse measurement of the fetal skull and usually is measured from an occipitofrontal scan plane at the level of the fetal thalami and the septum pellucidum cavum (Fig. 2). The fetal head should have an oval shape with clear midline margination. The measurement is determined, by convention, from the outer edge of the proximal fetal parietal bone to the inner edge of the distal parietal bone (Fig. 3). The gestational age is estimated by comparing the observed BPD to tables relating BPD to gestational age.
Because of increasing biologic variation with advancing gestational age, the precision of estimated gestational age from BPD is greatest early in the second trimester. Between 14 and 20 weeks, the BPD correlates with gestational age within 1 week in 95% of cases. Late in the third trimester, the measured BPD may predict gestational age within only 3–4 weeks.4
In approximately 5% of cases, BPD cannot be obtained because of fetal position; in a fetus in occipitoanterior or occipitoposterior presentation, the proper plane of the fetal skull cannot be visualized. Deferring the study for a short time may allow the fetus to assume a more favorable position. If an appropriate BPD cannot be obtained, an alternative fetal dimension is chosen.
The occipitofrontal diameter is measured in the same plane as the BPD and is a measurement of the longitudinal axis, by convention taken from outer skull tables on each side. This measurement can be averaged with the outer-to-outer skull measurement in the transverse plane to provide a basis for estimating head circumference. The cephalic index is the ratio of the transverse to longitudinal diameters measured in a similar fashion; a normal cephalic index is 0.78 ± 0.05. An abnormal cephalic index in not necessarily indicative of fetal pathology, but BPD measurements may be inaccurate estimators of gestational age if the fetal head is either more oval (dolichocephalic) or more round (brachycephalic) than average.5 A low cephalic index, for instance, often is seen in breech presentations and may introduce error into the use of BPD to estimate gestational age.
Measurement of Fetal Long Bones
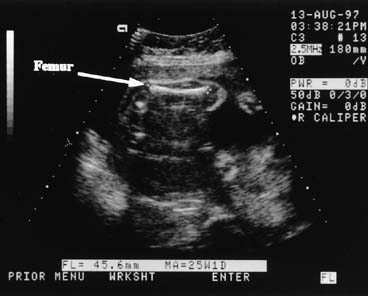
Measurement of the fetal limb bones also may be used to determine gestational age. The femur length usually is chosen because of its relative positional stability. The measurement is obtained by aligning the transducer with the lower end of the fetal spine and rotating toward the ventral aspect of the fetus.6 At first, only part of the femur may be seen; slight rotation and angulation of the transducer allows the entire length to be imaged. When properly visualized, the femur should have clear distal margins. Often, the bone casts a shadow to help identify this landmark (Fig. 4). The ultrasound images typically do not include epiphyses in early gestation; therefore, 8–15% of the palpable bone is not measured. Because the reference tables were derived in this same fashion, accuracy is preserved.
As is the case with BPD, the reliability of femur length in prediction of gestational age is best in early pregnancy. In the third trimester, however, femur length shows less variation than the BPD. Femur length may be difficult to obtain in a breech presentation, and measurement of the fetal humerus provides a good alternative. The humerus usually can be identified arising from the lateral upper fetal chest.
Abdominal Circumference
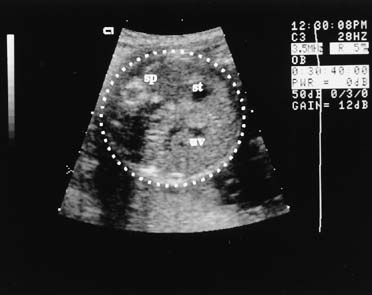
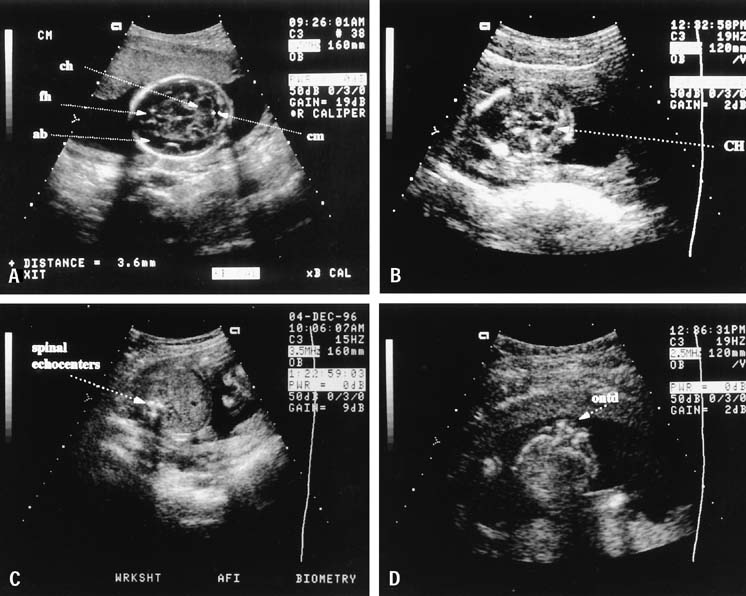
The fetal abdominal circumference, or mean abdominal diameter, also is useful in predicting gestational age.7 This measurement is obtained in a transverse plane, perpendicular to the fetal spine, at the level of the stomach and umbilical vein (Fig. 5). The fetal image in this traverse plane is nearly circular; the abdominal circumference is the average of the anteroposterior and transverse diameters, measured from the outer diameter to outer diameter. The circumference can be obtained by multiplying the mean abdominal diameter by π (approximately 3.14), or by directly measuring the circumference using a perimeter system on the ultrasound image, which is provided on most current ultrasound systems.
ASSESSMENT OF GESTATIONAL AGE
The relation between ultrasonographically derived fetal dimensions and gestational age is purely empiric. The older a normal fetus is, the larger its dimensions are. The reproducible ultrasonographic measurement of any fetal dimension in a normal reference population of fetuses of known gestational age allows the construction of a regression relation between that dimension and age. Subsequently, the determination of the gestational age of a pregnancy for which the conception date is unknown can be made by comparing that dimension with the reference data (Table 1).
Table 1. Fetal growth parameters*
| Gestational age (wk) | Biparietal diameter (mm) | Femur length (mm) | Humerus length (mm) | Head circumference (mm) | Average head diameter (mm) | Abdominal circumference (mm) | Average abdominal diameter (mm) |
| 14 | 28 | 15 | 15 | 101 | 032 | 084 | 027 |
| 15 | 32 | 18 | 18 | 114 | 036 | 093 | 030 |
| 16 | 36 | 20 | 21 | 128 | 041 | 106 | 034 |
| 17 | 39 | 23 | 23 | 141 | 045 | 117 | 037 |
| 18 | 42 | 26 | 26 | 154 | 049 | 128 | 041 |
| 19 | 45 | 29 | 28 | 167 | 053 | 139 | 044 |
| 20 | 48 | 32 | 31 | 179 | 057 | 150 | 048 |
| 21 | 51 | 35 | 33 | 192 | 061 | 161 | 051 |
| 22 | 54 | 37 | 35 | 204 | 065 | 172 | 055 |
| 23 | 58 | 40 | 37 | 215 | 068 | 183 | 058 |
| 24 | 61 | 42 | 39 | 227 | 072 | 194 | 062 |
| 25 | 64 | 45 | 41 | 238 | 076 | 205 | 065 |
| 26 | 67 | 48 | 44 | 249 | 079 | 216 | 069 |
| 27 | 70 | 50 | 46 | 259 | 082 | 227 | 072 |
| 28 | 72 | 53 | 48 | 269 | 086 | 238 | 076 |
| 29 | 75 | 55 | 49 | 279 | 089 | 249 | 079 |
| 30 | 78 | 57 | 51 | 288 | 092 | 260 | 083 |
| 31 | 80 | 60 | 53 | 297 | 095 | 271 | 086 |
| 32 | 82 | 62 | 55 | 306 | 097 | 282 | 090 |
| 33 | 85 | 64 | 57 | 314 | 100 | 293 | 093 |
| 34 | 87 | 67 | 59 | 322 | 102 | 304 | 097 |
| 35 | 88 | 69 | 60 | 329 | 105 | 315 | 100 |
| 36 | 90 | 71 | 62 | 336 | 107 | 326 | 104 |
| 37 | 92 | 73 | 64 | 342 | 109 | 337 | 107 |
| 38 | 93 | 76 | 66 | 348 | 111 | 348 | 111 |
| 39 | 94 | 78 | 67 | 354 | 113 | 359 | 114 |
| 40 | 95 | 80 | 69 | 359 | 114 | 370 | 118 |
*95% confidence intervals: biparietal diameter—12–20 weeks ± 5–7 days, 20–30 weaks ± 10 days, 30–40 weeks ± 14–21 days; femur/humerus length—12–20 weeks ± 6–7 days, 20–30 weeks ± 10 days, 30–40 weeks ± 12 days; head circumference—16–28 weeks ± 1.5 cm; 28–40 weeks ± 2.5 cm; abdominal circumference—16–40 weeks ± 13%.
(Seeds JW, Cefalo RC: Practical Obstetrical Ultrasound, p 57. Rockville, MD, Aspen, 1986)
Averaging the gestational ages derived from two or more measurements has been shown to be more accurate than using any single parameter.8 Determinations made early in pregnancy are more accurate than those made later. Because of the greater accuracy of the early study, ultrasound examinations subsequent to an early study should not be used to revise the estimated date of confinement (EDC), but rather should be used as a measure of the quality of fetal growth between the two studies. Similarly, it is not appropriate to revise an EDC on the basis of an ultrasound examination if the patient's menstrual dates are within the range of error of the ultrasound method.
If significant discrepancy is seen between two ultrasonographically measured fetal dimensions (more than a 2-week difference), then the ultrasonographer must consider the possibility of an error in measurement technique. If a critical reevaluation reveals no error, then asymmetry in fetal growth may be present. Growth asymmetry may occur as a result of physiologic alteration in fetal head shape (brachycephaly or dolichocephaly) or in association with intrauterine growth restriction (IUGR), macrosomia, or a fetal anomaly.
CONTENT OF AN ULTRASOUND EXAMINATION
A basic ultrasound examination should contain a fetal survey, an evaluation of fetal biometry, and an anatomic screening examination. The survey includes a confirmation of fetal number, viability, position, assessment of amniotic fluid volume, and location of the placenta. In assessing fetal biometry, the applicable standard fetal measurements already discussed, including CRL, BPD, abdominal circumference, and femur length, should be taken. The estimation of fetal weight is a clinically useful parameter computed from the fetal biometric measurements. Several equations have been produced based on fetal biometry that estimate fetal weight. One or more of these usually is incorporated into the software of most contemporary ultrasound machines. An estimation is provided automatically after the biometry is recorded. These weight estimations differ little in accuracy, and most provide an estimated fetal weight that has a standard deviation of ±8% to 10%. Therefore, 95% (2 standard deviations [SD]) of the estimates are within 20% of the actual birth weight.
An ultrasound examination also should include specific documentation of the fetal anatomic images obtained. These images are usually stored in the form of Polaroid pictures, videotape, thermal prints, or digital image files.
USE OF ULTRASOUND AS A SCREENING TEST
Generally recognized indications for obstetric ultrasound imaging are found in Table 2. Although the routine use of ultrasound in early pregnancy is not considered the standard of care by any public organization in the United States,9 it is promoted by many practitioners. Some investigators have noted a significant reduction in the frequency of labor induction for postdate fetuses when routine ultrasound is performed. Routine ultrasound early in pregnancy can result in a significant revision of the estimated date of delivery in up to 10% of patients with an accurate normal last menstrual period. Routine ultrasound use may decrease the incidence of antepartum fetal assessment and induction of labor for postdate fetuses.
Table 2. Indications for ultrasound in pregnancy
| Unsure last menstrual period |
| Vaginal bleeding during pregnancy |
| Uterine size not equal to that expected for dates |
| Use of ovulation-inducing drugs in early pregnancy |
| Obstetric complications in a prior pregnancy: prior cesarean section, preterm delivery |
| Screen for fetal anomaly in selected high-risk pregnancies: elevated maternal serum α-fetoprotein level, prior fetal malformation, certain drug exposure in early pregnancy, maternal diabetes |
| Antepartum fetal assessment in high-risk pregnancy: postdate fetus |
| Following fetal growth in certain high-risk situations: twins, suspected intrauterine growth restriction |
Belfrage and colleagues10 compared the routine use of ultrasound in early pregnancy with the use of ultrasound for selected indications. Routine ultrasound use resulted in correction of the estimated date of confinement by more than 2 weeks in 7% of cases. There also was a significant reduction in the incidence of labor induction for postdate fetuses in the routine ultrasound group. Neonatal outcome was similar in both groups.
The use of ultrasound as a routine screening tool in obstetric patients applies to low-risk patients who have none of the indications shown in Table 2. The multicenter Routine Antenatal Diagnostic Imaging with UltraSound (RADIUS) study intended to determine the clinical benefits of routine ultrasound in a low-risk population.11 Patients were recruited from 109 obstetricians and family practitioners from private, academic, and health maintenance organization practices. Of the initial patient population of approximately 53,800 patients, in the final analysis, only 15,530 patients (29%) of the initial registered group were randomized to receive either selective or routine ultrasound. The study concluded that there was no benefit from routine ultrasound in this low-risk population. The RADIUS study has been criticized in that the patients selected for final inclusion in this study bear little similarity to the average obstetric population. Only approximately one third of fetal anomalies were detected antenatally. If only serious malformations are considered, the detection rate was significantly improved at 78%. Clearly, the detection of anomalies in tertiary centers was significantly more accurate than those in primary care ultrasound clinics.
The Helsinki ultrasound trial included 9310 women: half were randomly allocated to ultrasound screening and half to routine obstetric care.12 In the ultrasound screening group, perinatal mortality was significantly lower by a factor of approximately 50%, mainly because of improved early detection of major malformations in the group randomized to ultrasound, which led to an increased rate of pregnancy termination. It also appeared in this trial that twin pregnancies detected earlier had a lower perinatal mortality rate.
Clearly, the use of ultrasound as a screening test is a controversial matter. The most likely benefits of ultrasound screening in low-risk patients are obstetric and include confirmation of dates, early detection of multiple gestation, location of the placenta, and baseline growth data. In virtually every complication that may occur later in pregnancy, clinical decisions may be facilitated if the patient had an ultrasound earlier in pregnancy.
To be used as a screening test, ultrasound should be widely available and have a relatively low cost and high sensitivity and specificity rates. One of the stated intents of the RADIUS study was to determine whether routine ultrasound screening would improve perinatal outcome. Ultrasound, however, is a diagnostic modality, not a therapeutic one. Ultrasound alone cannot reduce perinatal mortality, but the information obtained from an ultrasound can be used to guide the clinician to choose the appropriate therapy. In the final analysis, whether ultrasound is used as a screening test may depend on its cost. Direct cost savings from screening ultrasound examinations may be closely related to the accuracy of these examinations in detecting fetal anomalies. One study concluded that cost-savings from screening ultrasounds in low-risk patients could only be realized if examinations were performed in tertiary centers.13
In the RADIUS study, approximately two-thirds of the subjects were thought to have an obstetric indication for ultrasound; however, it is only in the small proportion of truly low-risk patients that a screening ultrasound should be considered. Should every obstetric patient have a routine ultrasound examination? Only if it is adequately performed, properly recorded, and the patient is counseled regarding appropriate goals and limitations of ultrasound.
Routine screening is best performed between 18 and 20 weeks' gestation. At this time, the fetus usually is large enough that the fetal anatomy can be surveyed well, and yet the gestation is early enough that the accuracy of biometric measurements is preserved.
LEVEL 1 VERSUS LEVEL 2 ULTRASOUND
The introduction of maternal serum alpha feto-protein (AFP) screening gave rise to a two-tiered system of ultrasound. The basic, screening, or level 1 ultrasound was performed to assess fetal biometry and rule out multiple gestation. A level 2, referral, high-detail, or targeted scan, performed by a more experienced sonologist, could then assess for fetal anomalies. The distinction between a level 1 scan and level 2 scan has blurred in modern practice, and there is no official credentialing for referral ultrasound.
We believe that all ultrasound examinations should have at least a minimum content to include: documentation of fetal number, position, placentation, fluid volume, assessment of biometry, and cardiac activity. The report should also include what anatomic features were noted and whether they appeared normal; images should be recorded.14 In the patient at higher risk for fetal anomalies, referral to a tertiary center for a high-detail scan may be appropriate. Although anomalies may be missed at all imaging centers, it is clear from the RADIUS study that tertiary centers have increased accuracy in detection, which may allow for optimizing perinatal care.
CLINICAL APPLICATION OF OBSTETRIC ULTRASOUND
Assessment of First-Trimester Complications
First-trimester ultrasonography can be of significant value in predicting the outcome in patients with bleeding in early pregnancy. Except in the unusual circumstance of a combined pregnancy (incidence 1 in 12,000 to 30,000), the finding of a pregnancy within the uterus excludes an ectopic pregnancy. This distinction, however, is not always clear. The normal gestational sac has a well-defined, echogenic border. In ectopic gestation, decidua and blood may distend the uterine cavity, and the ultrasound image can mimic a gestational sac, resulting in the so-called pseudogestational sac. These entities often can be distinguished by ultrasound; in a pseudogestational sac, the echogenic rim usually is absent, ill defined, or not centrally positioned in the uterus. In questionable cases, serial growth of the sac can be assessed. In a normal pregnancy, the gestational sac should grow at least 0.6 mm daily.15
Using a quantitative assay of human chorionic gonadotropin (hCG) with ultrasound improves diagnostic accuracy. An intrauterine gestational sac should be visualized by transvaginal ultrasound with β-hCG values between 1000 and 2000 mIU/mL.16 If the level is below this value, in a clinically stable patient, serial hCG values can be followed. In a normal early pregnancy, the β-hCG level should increase at least two-fold in 72 hours.
In practice, a clinical problem often faced is differentiation of a threatened abortion from an ectopic pregnancy. In both these conditions, a subnormal increase in β-hCG usually is seen. Ultrasound findings in a pregnancy destined to abort include a poorly-defined gestational sac, a large yolk sac (6 mm or greater in size), a low site of sac location in the uterus, or an empty gestational sac at 8 weeks' gestational age (the blighted ovum).
The only absolute assurance that a pregnancy is intrauterine is the finding of a fetal pole within the uterine cavity. The endovaginal ultrasound can be useful in this clinical setting because the fetal pole can be seen at 6 weeks. In a normal pregnancy, the fetal pole should be visible if the gestational sac is 25 mm or larger in diameter.
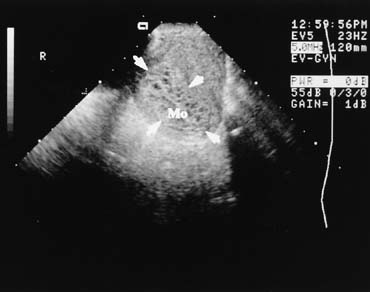
The presence of a fetal pole with demonstrable cardiac activity is reassuring and greatly decreases the likelihood of spontaneous abortion. In an ultrasonically normal gestation without bleeding at 8–9 weeks, there is a 3% chance of subsequent pregnancy loss.17 If bleeding is present, this chance increases to approximately 13%.18 Also included in the differential diagnosis of vaginal bleeding in pregnancy is the hydatid mole, which has a characteristic ultrasound appearance (Fig. 6).
First Trimester Down Syndrome Screening
In 2007, the American College of Ob Gyn endorsed offering aneuploidy screening to all gravidas.19 This screening can be between 11 and 13 weeks 6 days (fetal crown–rump length 42–79 mm). The test involves measurement of the fetal nuchal translucency and evaluating levels of two chemicals in maternal blood, β-hCG and pregnancy-associated plasma protein A (PAPP-A). This test can detect approximately 85% of fetuses with Down syndrome, with a 5% false positive rate.20 The risk of Down syndrome is increased with increasing values of nuchal skin thickness, as is seen in the second trimester. In fetuses with increased nuchal translucency and normal chromosomes, there is an increased incidence of fetal cardiac anomalies. 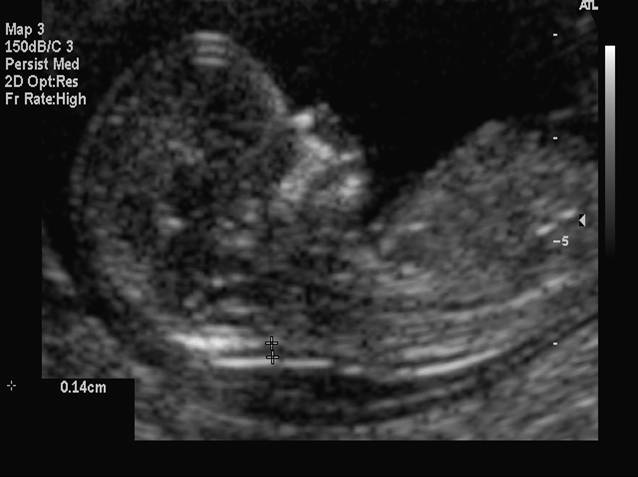
Fig 7. Measurement of first trimester fetal nuchal translucency.
Absence of the fetal nasal bone in the first trimester (at 11 or more weeks' gestation) has been observed to occur more frequently in fetuses with Down syndrome. Although this is not as an important sonographic marker as increased nuchal translucency, fetuses with Down syndrome are more likely to have an absent nasal bone then chromosomally normal fetuses.21, 22 Fig 8. Fetal nasal bone (arrow) seen at first trimester ultrasound screening test. 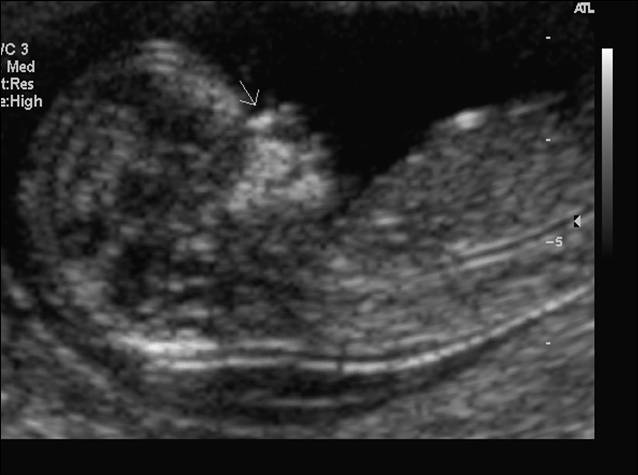
Evaluation of Abnormal Biochemical Testing
For two decades, AFP testing has been offered to obstetric patients in the United States. Many practitioners are now using multiple markers, which may include AFP, hCG, estriol, and inhibin. These biochemical markers are used as a screening test for fetal anatomic abnormalities and Down syndrome. Fetal abnormalities associated with elevated AFP levels include fetal death, spina bifida, anencephaly, abdominal wall defect such as omphalocele and gastroschisis, and renal abnormalities. Low AFP levels have been associated with Down syndrome and other chromosomal abnormalities. Because AFP levels and the other biochemical markers vary with gestational age, the interpretation of these tests demands accurate clinical dating. The first step in evaluation of a patient with an abnormal serum AFP level is ultrasound.
Because maternal serum AFP levels normally increase with advancing gestational age, an apparent elevation might result from an error in dates. Twins also cause an elevation of AFP levels. In a basic ultrasound examination to evaluate elevated maternal serum AFP levels, as many as one-third of patients are shown to have incorrect dates, multiple gestation, or fetal death. If the dates are correct and the elevated maternal serum AFP level is confirmed, referral for a high-detail ultrasound examination is appropriate. This should be performed by a sonologist with experience in diagnosing fetal anomalies by ultrasound. Detection of these anomalies is discussed later in the sections on clinical applications.
In patients who have elevated AFP levels and normal high-detail ultrasound examinations, determination must be made about whether amniocentesis is appropriate. The information from the ultrasound can then be used when counseling the patient. In the past, amniocentesis was recommended for determination of amniotic fluid AFP and karyotype. High-detail ultrasound now can detect up to 95% of neural tube defects, so the risk of missing spina bifida is low. It is appropriate to inform the patient regarding the risk of an invasive procedure such as amniocentesis as well as about the very low risk of a normal high-detail ultrasound missing spina bifida.23
In patients who have low AFP levels or abnormal biochemical screen results, ultrasound can play an important role in interpretation and diagnosis.24 An inaccurate gestational age may explain up to half of cases of apparently low maternal serum AFP values. Usually, if the ultrasound gestational age is within 2 weeks of that determined by the last menstrual period, the gestational age used for interpretation should not be changed. Ultrasound is less accurate for determination of chromosomal abnormalities, such as Down syndrome, than it is for determination of neural tube defects. Benacerraf and colleagues25 reported the finding of occipital nuchal skin thickening in approximately half of fetuses with trisomy 21; these data subsequently were confirmed by other investigators.26 Other abnormalities seen with increased incidence in fetuses with Down syndrome include short femur, short humerus, dilation of the fetal renal pelvis, ventriculomegaly, and fetal cardiac abnormalities. Combining multiple minor dysmorphic features into an aneuploidy scoring system may prove useful both in the detection of abnormal fetuses and in the reduction of risk.27
Evaluation of Fetal Growth Abnormalities
Fetal growth can be evaluated by comparing individual dimensions to normative data, by comparing various fetal dimensions to assess symmetry, or by integrating selected measurements to produce an estimation of fetal weight.28 Several investigators have derived equations useful for estimating weight, using fetal dimensions, such as BPD, mean abdominal diameter, abdominal circumference, and femur length. The differences between these tables and published methods for fetal weight determination is small when compared with the overall accuracy of fetal weight estimation. Generally, the ultrasonic predicted weight is within 10% of the actual weight in two-thirds of cases and within 20% in 95% of cases.
In the preterm fetus in which clinical estimation of fetal gestational age is often inaccurate, estimating fetal weight using ultrasound even with known error can be the only objective data indicating survival probability available. In the term fetus, however, because of the greater actual error, ultrasound-estimated fetal weight may be no more accurate than a clinical weight estimation.29
Fetal growth restriction is commonly observed in fetuses with chromosome abnormalities. These growth abnormalities can often been detected at less than 20 weeks gestation. 30
Assessment of Third-Trimester Bleeding
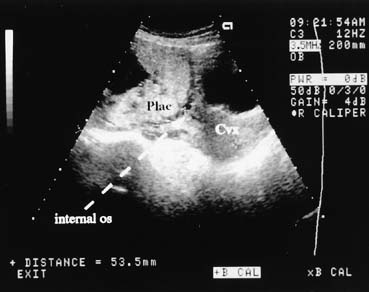
Ultrasound examination of the placenta is useful in differentiating causes of third-trimester bleeding.31 The placenta can be identified easily with the real-time scanner. Placenta previa can be diagnosed with a high degree of accuracy (Fig. 7). In cases of marginal previa, the examination should be performed with both an empty and a full bladder for greatest accuracy. The internal os may be located by identifying the bladder angle in a moderately full bladder.
|
Placenta previa is found in approximately 5% of second-trimester scans performed for other indications. In marginal previa or central previa (central insertion of the placenta over the cervix) seen on a second-trimester scan, a follow-up scan in the third trimester is indicated; many of these cases will have spontaneously resolved on subsequent examination because of asymmetry in uterine growth, not placental migration.32, 33
The persistence of placenta previa depends on the gestational age at which it is detected. For example if previa is detected at 15–19 weeks, it persists in 12% of patients. If it is detected at 24–27 weeks, it may persist in up to 50%.34
Other causes of third-trimester bleeding include abruptio placentae and vasa previa. Although these conditions have been diagnosed with ultrasound, they cannot be excluded confidently in most cases. A retroplacental clot may be seen in some cases of abruptio, but ultrasound cannot exclude abruption, and the wise course in most cases is to treat the patient as clinical circumstances dictate.
With increasing gestational age, the placenta increases in echogenicity because of increased fibrosis and calcium content. This feature of placental maturation has led to a grading of placentas from immature (grade 0) to mature (grade 3).35 In a grade 3 placenta, individual cotyledons are outlined by dense fibrotic calcified septa. The correlation of a grade 3 placenta with fetal pulmonary maturity is high. A grade 3 placenta can be observed in the third trimester in a variety of high-risk fetal conditions (e.g., IUGR, chronic hypertension) but also can occur in normal pregnancy. Conversely, a grade 3 placenta may not be seen in many normal patients in labor at term.
Clinical Applications
Ultrasound estimation of fetal weight can be invaluable in many clinical situations, such as obstetric management of the patient presenting with labor and no previous prenatal care.
Identification of fetal macrosomia also is of particular interest to the clinician because this condition is associated with an increased risk of a variety of obstetric complications. The accuracy of ultrasound in predicting macrosomia depends on the clinical situation in which the ultrasound is used.36 The sensitivity of ultrasound in predicting macrosomia is approximately 60%.37 As the prevalence of macrosomia increases, such as in the case of a mother with diabetes, the accuracy of ultrasound in predicting macrosomia increases. The percent error in sonographic estimation of fetal weight, however, is similar in both diabetic and nondiabetic pregnancies.38
Some clinicians have advocated prophylactic cesarean section when fetal macrosomia is found, to prevent brachial plexus injury. Any management decisions must take all other clinical features into account, including the estimated fetal weight presentation, progress in labor, clinical pelvimetry, and obstetric history.
Diagnosis of Intrauterine Growth Restriction
If the gestational age in a pregnancy is confidently established, ultrasound can provide an accurate diagnosis of IUGR. IUGR can result from uteroplacental insufficiency, drug exposure, intrauterine infection, or genetic factors.39 Symmetric IUGR, in which all fetal parameters are small for a given gestational age, occurs with extrinsic conditions that are active early in pregnancy (e.g., tobacco or alcohol abuse, congenital rubella). Symmetric IUGR also can result from intrinsic conditions that limit fetal growth potential (e.g., chromosomal abnormalities). Symmetrically reduced growth also is seen in the constitutionally small, normal fetus.
Fetal growth may be normal until the late second or early third trimester, at which time the limit of uteroplacental circulation in sustaining fetal growth is reached. This results in asymmetric fetal growth, in which the fetus adapts to the relative decrease in utero placental blood flow by redistributing cardiac output to favor the brain at the expense of the muscles and abdominal viscera.
For these reasons, the use of BPD alone can detect only half of IUGR cases. When IUGR is suspected, ultrasound-estimated fetal weight can detect up to 90% of affected infants.40, 41 An estimated fetal weight below the 10th percentile for gestational age is predictive of IUGR. When IUGR is suspected, the ultrasonographer should look critically at amniotic fluid volume. Oligohydramnios, resulting from decreased fetal urine production as blood is shunted away from the fetal kidneys, commonly is associated with severe IUGR. Manning and associates noted that when a 1-cm pocket of amniotic fluid could not be found, IUGR was present in 90% of cases.42 The amniotic fluid index also has been described as a useful tool in the evaluation of fluid volume.43
When the gestational age is not precisely known, assessment of fetal growth over a 2-week interval is useful in distinguishing IUGR from incorrectly dated pregnancies. In the third trimester, fetal weight should increase approximately 300 grams in 2 weeks. Another gestational age-independent assessment is the abdominal circumference-to-fetal length or mean abdominal diameter-to-femur length ratio.44 Several investigators 45, 46 have reported that the transverse fetal cerebellar diameter is unaffected by IUGR; this additional parameter may help to discriminate IUGR from incorrectly dated pregnancies.
Evaluation of Multiple Gestations
In evaluating multiple gestations, the ultrasonographer should note the number and presentation of the fetuses and the position and number of placentas. A membrane almost always can be seen between each fetus in early gestation. Monoamniotic twins, which comprise only 3% of all twin gestations, have no intervening membrane and are at much greater risk of perinatal mortality than other twins. The thickness of the membrane between twins may provide a clue about whether both chorion and amnion are present, because a thicker membrane is present in dichorionic–diamniotic twins, and the placenta appears to funnel down into the proximal intervening membrane (Figure 10).47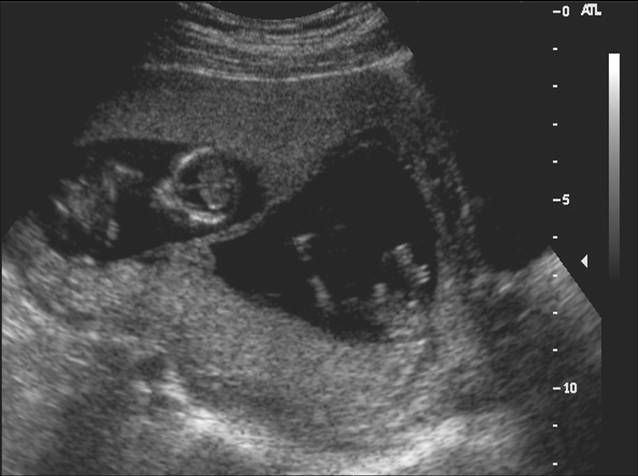
Fig 10. Dichorionic twin gestation, with thick intervening membrane and twin peak sign.
As is the case with ultrasound diagnosis of IUGR, discordance is better predicted by estimated fetal weight calculation than by BPD alone.48 Discordance is determined by dividing the observed fetal weight difference by the weight of the larger twin. Mild discordance (15–25% difference in fetal weights) occurs in approximately 20% of twin pregnancies, and severe discordance (more than 25% difference) occurs in approximately 5% of twins.49 Discordant twins are at increased risk for perinatal mortality; the smaller twin is at greatest risk antenatally. A reasonable approach to assess growth in twin gestation is to perform serial estimated fetal weight measurements on both twins every 4 weeks beginning at 20 weeks' gestation. If discordance is present, or if intertwin weight differences are increasing, weight determination is recommended more often.
Detection of Fetal Malformations
The ability of ultrasound to detect and characterize a broad array of fetal malformations is well known. Ultrasound is best suited to the detection of obstructive malformations or major distortions of surface anatomy. Each ultrasound examination performed should comprise an anatomic survey of the fetus, including cranial and intracranial structures, spinal anatomy, a four-chamber cardiac view, the abdomen with stomach and umbilicus, the bladder, and at least one long bone. Any suspicion of abnormality should result in referral for a more detailed ultrasound examination.
A basic office ultrasound examination is adequate for most patients. A targeted scan, performed by an ultrasonographer with experience in the evaluation of fetal malformations, is performed when there is a suspected fetal abnormality on a basic ultrasound examination or to evaluate fetal anatomy when the patient is in a high-risk category based on history, physical findings such as polyhydramnios, or laboratory testing (e.g., AFP).
Determination of fetal karyotype should be considered in any case in which a malformation is ultrasonographically detected. Aneuploidy is discovered in approximately 10% of cases of ultrasonographic dysmorphology. The finding of a lethal karyotype could alter obstetric management because fetal distress during labor often occurs in this setting, and a cesarean section delivery might otherwise result. Additionally, the finding of one structural malformation should prompt a more detailed search for other anomalies.
CENTRAL NERVOUS SYSTEM ANOMALIES
The incidence of fetal open neural tube defects in the United States is 1 to 2 per 1000 births. Anencephaly, in which the cranial vault is absent, accounts for approximately half of these. By locating the fetal spine and moving the ultrasound transducer cephalad, the absence of a cranial vault can be confirmed. In anencephaly, the fetal orbits should be visible. The cranium should be visualized by 14 weeks' gestation.
Spina bifida is more difficult to detect; the fetal spine must be meticulously examined in three planes. Successful ultrasonographic detection of spina bifida requires examination of both spinal and cranial anatomy. Experience strongly suggests that intracranial abnormalities are present in most all cases of spina bifida.23, 50 Five cranial signs associated with spina bifida are frontoparietal notching (the lemon sign) (Fig. 8), mild hydrocephalus, a small-for-date BPD, abnormal cerebellar hemispheres, and obliteration of the cisterna magna. The finding of a normal fetal cerebellum and cisterna magna virtually excludes spina bifida. This view is obtained by finding the axial plane of the BPD and rotating the transducer to view the posterior fossa (Fig. 9).
Hydrocephalus is most sensitively detected by measurement of the width of the atrium of the lateral ventricle. A value of 9 mm or less is normal. In early hydrocephalus, the choroid plexus may be displaced anteriorly and does not fill the atrium of the ventricle. Early diagnosis is thus made on morphologic as well as measurement criteria. If the lateral ventricle is enlarged, this should prompt a careful evaluation of the fetal brain and spine.
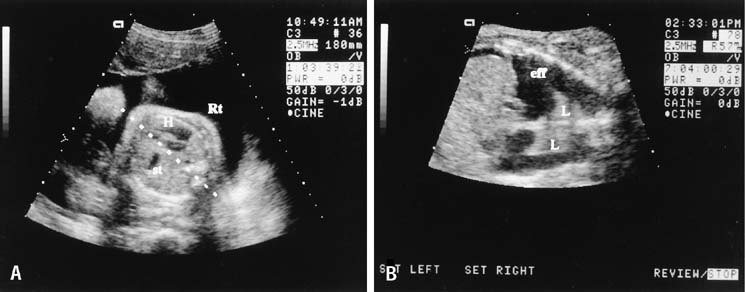
CARDIOVASCULAR DISORDERS
The four-chamber view of the fetal heart is obtained from a near transverse midchest scan plane at the level of the heart (Fig. 10). The normal fetal heart is angled at 45° (±10 degrees [1 SD]) to the left of the anteroposterior midline.51 The anteroposterior midline passes through the left atrium and the right ventricle. Asymmetry in chamber size, defects in the septum, or displacement of the fetal heart should lead to referral for a more detailed ultrasonographic evaluation (Fig. 11). In one study, screening ultrasound had an overall detection rate of 75% for major congenital heart disease.52 A normal four-chamber view excludes many cardiac anomalies,53 but accuracy of anomaly detection can be improved by the addition of outflow tract views.
GASTROINTESTINAL DISORDERS
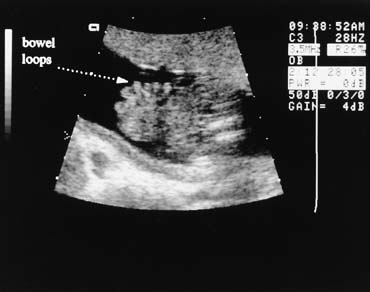
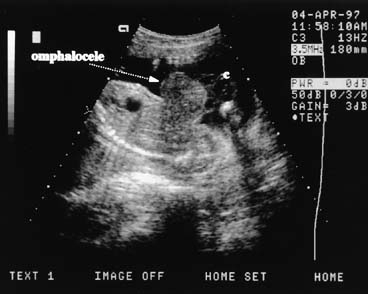
The fetal stomach can be seen as a fluid-filled structure on the left side of the abdomen caudal to the four-chamber view (see Fig. 5). The normal umbilical cord insertion can be seen in a transverse section below the fetal stomach (Fig. 12). In this area, ventral wall defects of the fetus are seen, including gastroschisis and omphalocele. Gastroschisis is an open defect in the abdominal wall with evisceration of the abdominal organs that usually occurs to the right of a normal cord insertion. There is no membrane covering the eviscerated fetal bowel loops, which can be seen in the amniotic fluid at the site of the defect (Fig. 13). Omphalocele, a failure of the embryonic gut to return into the abdomen by 12 weeks' gestation, occurs at the cord insertion (Fig. 14) and usually is covered by a membrane consisting of peritoneum and amnion. Omphalocele is associated with fetal chromosome abnormalities in 33–50% of cases and should prompt a careful search for other fetal anomalies. IUGR commonly is associated with both gastroschisis and omphalocele.
|
|
The double-bubble sign, classically associated with duodenal obstruction, is not specific for atresia but also may be seen with obstruction from malrotation or an annular pancreas.
RENAL DISORDERS
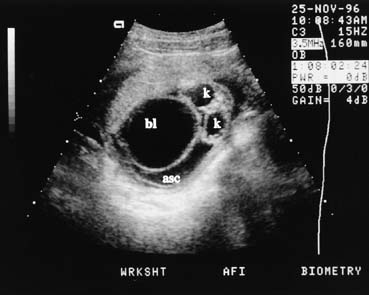
The fetal kidneys can be seen on a transverse scan plane just below the level of the stomach, in the dorsal paraspinal areas (Fig. 15). The fetal bladder is seen as a round fluid-filled structure in the anterior midline of the fetal pelvis. The amniotic fluid volume provides a clue to fetal renal function. In the fetus with absent kidneys, renal obstruction, or nonfunctioning kidneys (renal dysplasia) after 20 weeks' gestation, the amniotic fluid volume is markedly decreased. Before 20 weeks' gestation, however, the amniotic fluid volume may be normal despite renal malformations.
|
Obstructive uropathy may be caused by posterior urethral valves, urethral atresia, or persistent cloaca syndrome. Ultrasound findings include a large bladder and enlarged ureters, which are seen as cystic convoluted structures in the lower abdomen (Fig. 16). Ureteropelvic junction obstruction is suspected when an enlarged fetal renal pelvis, without an enlarged bladder, is seen.
Assessment of Changes in Amniotic Fluid Volume
Amniotic fluid is a physiologic extension of the fetus. Abnormalities of amniotic fluid volume are nonspecific findings that warrant further investigation. Polyhydramnios is defined as an amniotic fluid volume in excess of 2000 mL. The clinical diagnosis is made by the findings of a large-for-date uterine size, increased fluid, and a normal-size fetus. The excess fluid is seen as large anechoic areas on ultrasound. A single pocket of fluid that is 8 cm or larger suggests polyhydramnios; this occurs in approximately 1% of pregnancies.54 An excess of fluid may be found in maternal diabetes, erythroblastosis, or fetal malformation.55 In severe erythroblastosis, fetal hydrops may be seen at the time of ultrasound study.
In one-third of pregnancies complicated by polyhydramnios, no cause is determined.56 Fetal malformations can complicate up to 40% of cases when twins, diabetes, and blood group incompatibility are excluded.
Oligohydramnios can be associated with fetal renal abnormalities, IUGR, postdate fetuses, or membrane rupture. The clinical diagnosis is made when the fundal height is small and ultrasound examination shows little or no amniotic fluid. When amniotic fluid is decreased, the fetal small parts are crowded and the fetus is in contact with the uterine wall in most scanning planes.
The amniotic fluid index, a sum of the largest vertical pocket of amniotic fluid in each of four quadrants of the uterus, is a useful semiquantitative measurement of fluid volume.57
Severe oligohydramnios is present when the largest vertical pocket of amniotic fluid measures less than 1 cm. Major fetal anomalies were observed in 13% of patients with severe oligohydramnios. In the structurally normal fetus with intact membranes, severe oligohydramnios may be an indication for delivery.58
Assessment of Fetal Well-Being
The biophysical profile is a test used in conjunction with antepartum fetal heart rate testing to evaluate fetal well-being in high-risk pregnancies.59 The test quantifies fetal breathing and body movements, fetal tone, and amniotic fluid volume. It also incorporates the nonstress test as a measure of fetal reactivity. Each of these five parameters are assigned a score of 2 if normal and 0 if abnormal (Table 3). The fetus is observed until all parameters are seen or until 30 minutes has elapsed.
Table 3. Biophysical profile scoring
| Variable | Score 2 | Score 0 |
| Fetal breathing movements | At least 30 s of sustained fetal breathing movements in 30 min of observation | Less than 30 s of fetal breathing movements min 30 min |
| Fetal movements | Three or more gross body movements min 30 min | Two or fewer gross body movements min 30 min |
| Fetal tone | At least one limb motion from flexion to extension and rapid return to flexion | Partial or full limb extension, with no flexion; absence of fetal movement |
| Fetal reactivity | Two or more fetal heart accelerations of 15 beats/min for 15 s in 40-min observation period | Fewer than two fetal heart rate accelerations in 40 min |
| Amniotic fluid volume | A pocket of fluid that measures at least 2 cm in two perpendicular planes or AFI of 5 | Largest pocket of fluid measures less than 2 cm in two perpendicular planes or AFI less than 5 |
(Manning FA, Baskett TF, Morrison I et al: Fetal biophysical profile scoring: A prospective study in 1184 high-risk patients. Am J Obstet Gynecol 140:289, 1981)
This observation period helps to distinguish the normal fetus with a physiologic periodic steep cycle from the chronically asphyxiated fetus with central nervous system depression. Fetal breathing movements occur approximately 30% of the time and, when seen, demonstrate that a complex fetal central nervous system function is intact.59 Fetal breathing movements are seen in the normal fetus after 24 weeks' gestation.60
A normal biophysical profile (score of 8 to 10) is associated with a corrected perinatal mortality rate of 1 to 2 per 1000.61 A test score of 6 indicates the need to repeat testing within 24 hours. Scores of 4 or less are highly predictive of intrauterine fetal jeopardy and, in a term fetus, are an indication for delivery. Low scores are associated with fetal distress in labor as well as low Apgar scores. Vintzileos and colleagues62 observed a good correlation between a low biophysical profile and fetal acidosis.
In the extremely preterm fetus, a low score may not necessarily indicate the need for immediate delivery. The clinician must weigh the risk of fetal distress or demise with the risks of prematurity. In this clinical situation, stabilizing any adverse maternal condition (such as elevated blood glucose in a diabetic patient or high blood pressure in a hypertensive patient) and repeating the biophysical test a short time later may be indicated.
Assessment of the Risk of Preterm Labor
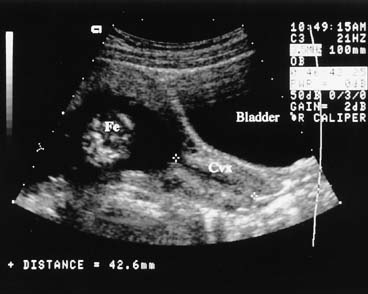
There has been considerable interest in ultrasonographic evaluation of the cervix as a marker for assessing the risk of preterm birth (Fig. 17).63 Iams and colleagues64 published a large multicenter study confirming that patients with a short cervix, as determined by endovaginal ultrasonography, are at increased risk for preterm birth. It is clear that the length of the cervix decreases with advancing gestational age and that there is no cervical length at which the risk of preterm birth is zero. A cervical length of less than 25 mm indicates an increased risk of preterm birth, both in high-risk and in low-risk populations. A single endovaginal cervical length measurement at 24 weeks can be used to assess risk status.65 Cervical length may also provide a more objective and clinically effective means of assessing the cervix in patients with cervical cerclage.
|
Guiding Invasive Procedures
Ultrasound can be used to localize a favorable pocket of amniotic fluid before performing amniocentesis.66 The needle can be inserted under direct visualization using the real-time scanner. A needle guide is available to aid the obstetrician with needle placement, but this can limit the operator's ability to move the needle in three dimensions. It is preferable to insert the needle in a coplanar fashion (i.e., in the same longitudinal plane as the ultrasound transducer), so that the entire length of the needle track can be seen. Transplanar placement (i.e., perpendicular to the ultrasound plane) limits ultrasound view of the needle.
In chorionic villus sampling, the catheter is inserted into the placental bed under ultrasound guidance. Similarly, simultaneous real-time ultrasound guidance is used in percutaneous umbilical blood sampling or cordocentesis.67
Three-Dimensional Ultrasound
3D ultrasound offers a significant new method of imaging which may revolutionize obstetric ultrasound. Initially, 3D images were obtained by computer processing of multiple two-dimensional (2D) images. Presently, real-time 3D images can be obtained (Fig. 18).68 The 3D ultrasound image consists of a volume of data that can subsequently be manipulated to display multiple longitudinal, transverse, and coronal images. The sonologist can obtain images which may not be readily available by conventional 2D sonography, and the images may not be as operator dependent as with 2D sonography. A saggital view of the fetal brain to look at the corpus callosum, for example, not readily obtained on 2D study, can be obtained readily during volume manipulation of a 3D data set. It is clear that if image quality is poor in 2D, then it may also be suboptimal in 3D.
There is an increasing body of research which indicates that 3D images may improve the accuracy of anomaly detection of the fetal face, ears, and distal extremities when compared to 2D images (Fig. 19).69, 70
SUMMARY
Obstetric ultrasound is an essential tool for the practicing clinician that can be used to improve obstetric care. The ultrasound examination should include information about fetal number, presentation, biometry, placentation, fluid volume, and anatomy, and the images should be documented. Routine or screening ultrasound may be useful to confirm dates, detect multiples earlier in pregnancy, assess placenta site, and obtain baseline biometry data. For patients at increased risk, or those with an abnormal screening examination, referral to a tertiary center may be indicated.
The information from diagnostic obstetric imaging can be used to assess fetal well-being in at-risk pregnancy and to facilitate invasive procedures such as amniocentesis. 3D ultrasound provides an exciting new and potentially revolutionary method of fetal imaging.
REFERENCES
Fowlkes JB, Holland CK: Mechanical bioeffects from diagnostic ultrasound: AIUM consensus statements. J Ultrasound Med 2000; 19:68 |
|
Timor-Tritsch IE, Rottem S, Thaler I: Review of transvaginal ultrasonography: A description with clinical application. Ultrasound Q 1988; 6:1 |
|
Kopta MM, May RR, Crane JP: A comparison of the reliability of the estimated date of confinement predicted by crown-rump length and biparietal diameter. Am J Obstet Gynecol 1983; 145:562 |
|
Kopta MM, Tomich PG, Crane JP: Ultrasonic methods of predicting the estimated date of confinement. Obstet Gynecol 1981; 57:657 |
|
Hadlock FP, Deter RL, Carpenter RJ et al: Estimating fetal age: The effect of head shape on BPD. AJR 1981; 137:83 |
|
O'Brien GD, Queenan JT: Growth of the ultrasound fetal femur length during normal pregnancy. Am J Obstet Gynecol 1981; 141:833 |
|
Tamura RK, Sabbagha RE: Percentile ranks of sonar fetal abdominal circumference measurements. Am J Obstet Gynecol 1980; 138:475 |
|
Hadlock FP, Harrist RB, Shah YP et al: Estimating fetal age using multiple parameters: A prospective evaluation in a racially mixed population. Am J Obstet Gynecol 1987; 156:955 |
|
National Institute of Health Consensus Development Conference: Consensus Statement: Diagnostic Ultrasound in Pregnancy. Washington, DC, US Government Printing Office, 1984 |
|
Belfrage P, Fernstrom I, Hallenberg G: Routine or selective ultrasound examinations in early pregnancy. Obstet Gynecol 1987; 69:747 |
|
Ewigman BG, Crane JP, Frigoletto FD et al: Effect of prenatal ultrasound screening on perinatal outcome. N Engl J Med 1996; 329:821 |
|
Saari-Kemppainen A, Karjalainen O, Ylostalo P et al: Ultrasound screening and perinatal mortality: Controlled trial of systematic one-stage screening in pregnancy. Lancet 1990: 336:387 |
|
Vintzileos AM, Ananth CV, Smulain JC et al: Routine second-trimester ultrasonography in the United States: A cost benefit analysis. Am J Obstet Gynecol 2000; 182:655 |
|
AIUM practice guideline for the performance of an antepartum obstetric ultrasound examination. J Ultrasound Med 2003; 22:1116. |
|
Nyberg DA, Mack LA, Laing FC et al: Distinguishing normal from abnormal gestational sac growth in early pregnancy. J Ultrasound Med 1987; 6:23 |
|
Dogra V, Paspulati RM, Bhatt S. First trimester bleeding evaluation. Ultrasound Q 2005; 21:69. |
|
Simpson JL, Mills JL, Holmes LB et al: Low fetal loss rates after ultrasound-proved viability in early pregnancy. JAMA 1987; 258:2555 |
|
Mantoni M: Ultrasound signs in threatened abortion and their prognostic significance. Obstet Gynecol 1985; 65:471 |
|
ACOG Committee on Practice Bulletins. Practice Bulletin No. 77: Screening for Fetal Chromosomal Abnormalities. Obstet Gynecol 2007; 109:217 |
|
Malone FD, Canick JA, Ball RH et al. First-trimester or second-trimester screening, or both, for Down's syndrome. N Engl J Med 2005 353:2001 |
|
Cicero S, Curcio P, Papageorghiou A et al: Absence of nasal bone in fetuses with trisomy 21 at 11-14 weeks of gestation: an observational study. Lancet 2001; 358:1665 |
|
Supulveda W, Wong AE, Dezerega V: First trimester ultrasonographic screening for trisomy 21 using fetal nuchal translucency and nasal bone. Obstet Gynecol 2007; 109:1040 |
|
Watson WJ, Chescheir NC, Katz VL et al: The role of ultrasound in evaluation of patients with an elevated maternal serum alpha-fetoprotein: A review. Obstet Gynecol 1991; 78:123 |
|
Seeds JW, Watson WJ: Ultrasound and maternal serum alphafetoprotein screening: A complementary relationship. Ultrasound Q 1990; 8:145 |
|
Benacerraf BR, Barss VA, Laboda LA: A sonographic sign for the detection in the second trimester of the fetus with Down's syndrome. Am J Obstet Gynecol 1985; 151:1078 |
|
Watson WJ, Miller RC, Menard MK et al: Ultrasonographic measurement of fetal nuchal skin to screen for chromosomal abnormalities. Am J Obstet Gynecol 1994; 170:583 |
|
Souter V, Nyberg DA: Use of genetic sonography for adjusting the risk for fetal Down syndrome. Semin Perinatol 2003; 27:130 |
|
Snijders RJ, Nicolaides KH. Fetal biometry at 14-40 weeks' gestation. Ultrasound Obstet Gynecol 1994; 4:34 |
|
Watson WJ, Soisson AP, Harlass FH: Estimated weight of the term fetus: Accuracy of ultrasound vs. clinical exam. J Reprod Med 1988; 33:369 |
|
Watson WJ, Miller RC, Wax JR et al: Sonographic detection of trisomy 13 in the first and second trimesters of pregnancy. J Ultrasound Med 2007; 26:1209 |
|
Abramowicz JS, Sheiner E: Ultrasound of the placenta: a systematic approach. Part 1: imaging. Placenta 2008; 29:225 |
|
Laing FC: Placenta previa: Avoiding false negative diagnoses. J Clin Ultrasound 1981; 9:109 |
|
Wexler P, Gottesfeld KR: Early diagnosis of placenta previa. Obstet Gynecol 1979; 54:231 |
|
Dashe JS, McIntire DD, Ramus RM et al: Persistence of placenta previa according to gestational age at ultrasound detection. Obstet Gynecol 2002; 99:692 |
|
Grannum PAT, Berkowitz RL, Hobbins JC: The ultrasonic changes in the maturing placenta and their relation to fetal pulmonic maturity. Am J Obstet Gynecol 1979; 133:915 |
|
Miller JM Jr, Brown HL, Khawli OF et al: Ultrasonographic identification of the macrosomic fetus. Am J Obstet Gynecol 1988; 159:1110 |
|
Rouse DJ, Owen J: Prophylactic cesarean delivery for fetal macrosomia diagnosed by means of ultrasonography–A Faustian bargain. Am J Obstet Gynecol 1999; 181:332 |
|
Sacks DA, Chen W: Estimating fetal weight in the management of macrosomia. Obstet Gynecol Survey 2000; 55:229 |
|
Seeds JW: Impaired fetal growth: Definition and clinical diagnosis. Obstet Gynecol 1984; 64:303 |
|
Seeds JW: Impaired fetal growth: Ultrasonic evaluation and clinical management. Obstet Gynecol 1984; 64:577 |
|
Ott WJ: Sonographic diagnosis of fetal growth restriction. Clin Obstet Gynecol 2006; 49:295 |
|
Manning FA, Hill LM, Platt LD: Qualitative amniotic fluid detection by ultrasound: Antepartum detection of intrauterine growth determination. Am J Obstet Gynecol 1981; 139:254 |
|
Rutherford SE, Phelan JP, Smith CV et al: The four-quadrant assessment of amniotic fluid volume: An adjunct to antepartum fetal heart rate testing. Obstet Gynecol 1987; 70:353 |
|
Egley CC, Seeds JW, Cefalo RC: Femur length versus biparietal diameter for estimating gestational age in the third trimester. Am J Perinatol 1986; 3:77 |
|
Reece EA, Goldstein I, Pilu G et al: Fetal cerebellar growth unaffected by intrauterine growth retardation: A new parameter for prenatal diagnosis. Am J Obstet Gynecol 1987; 157:632 |
|
Chavez, MR, Ananth CV, Smulian JC et al: Fetal transcerebellar diameter measurement with particular emphasis in the third trimester: a reliable predictor of gestational age. Am J Obstet Gynecol 2004; 191: 979 |
|
Wood SL, St Onge R, Connors G, Elliott PD: Evaluation of the twin peak or lambda sign in determining choroinicity in multiple pregnancy. Obstet Gynecol 1996: 88:6 |
|
Brown CE, Guzick DS, Leveno KJ et al: Prediction of discordant twins using ultrasound measurements of biparietal diameter and abdominal perimeter. Obstet Gynecol 1987; 70:677 |
|
Blickstein I, Lancet M: The growth discordant twin. Obstet Gynecol Surv 1988; 43:509 |
|
Campbell J, Gilbert WM, Nicholaides KH: Ultrasound screening for spina bifida: Cranial and cerebellar signs in a high risk population. Obstet Gynecol 1987; 70:247 |
|
Comstock CH: Normal fetal heart axis and position. Obstet Gynecol 1987; 70:255 |
|
Carvalho JS, Mavrides E, Shinebourne EA et al: Improving the effectiveness of routine prenatal screening for major congenital heart defects. Heart 2002; 88:387 |
|
Copel JA, Pilu G, Green J et al: Fetal echocardiographic screening for congenital heart disease: The importance of the four chamber view. Am J Obstet Gynecol 1987; 157:648 |
|
Hill LM, Breckle R, Thomas ML et al: Polyhydramnios: Ultrasonically detected prevalence and neonatal outcome. Obstet Gynecol 1987; 69:21 |
|
Hauge M, Bugge M, Nielsen J: Early prenatal diagnosis of omphalocele constitutes indication for amniocentesis. Lancet 1983; 2:507 |
|
Cardwell MS: Polyhydramnios: A review. Obstet Gynecol Surv 1987; 42:612 |
|
Rutherford SE, Smith CV, Phelan JP et al: Four quadrant assessment of amniotic fluid volume: Interobserver and intraobserver variation. J Reprod Med 1987; 32:587 |
|
Bastide AB, Manning FM, Harman C et al: Ultrasound evaluation of amniotic fluid: Outcome of pregnancies with severe oligohydramnios. Am J Obstet Gynecol 1986; 154:895 |
|
Manning FA, Baskett TF, Morrison I et al: Fetal biophysical profile scoring: A prospective study in 1184 high-risk patients. Am J Obstet Gynecol 1981; 140:289 |
|
Natale R, Nasello-Paterson C, Connors G: Patterns of fetal breathing activity in the human fetus at 24-28 weeks' gestation. Am J Obstet Gynecol 1988; 158:294 |
|
Manning FA, Morrison L, Lange IR et al: Fetal assessment based on fetal biophysical scoring: Experience in 12,620 referred high risk pregnancies. 1. Perinatal mortality by frequency and etiology Am J Obstet Gynecol 1985; 151:343 |
|
Vintzileos AM, Gaffney SE, Salinger LM et al: The relationship between fetal biophysical profile and cord pH in patients undergoing cesarean section before the onset of labor. Obstet Gynecol 1987; 70:196 |
|
Berghella V, Roman A, Daskalakis C et al: Gestational age at cervical length measurement and incidence of preterm birth. Obstet Gynecol 2007; 110:311 |
|
Iams JD, Goldenberg RL, Meis PJ et al: The length of the cervix and the risk of spontaneous premature delivery. N Engl J Med 1996; 334:567 |
|
Watson WJ, Stevens D, Welter S et al: Observations on the sonographic measurement of cervical length and the risk of premature birth. J Maternal Fetal Med 1999; 8:17 |
|
Seeds JW Diagnostic mid trimester amniocentesis: how safe? Am J Obstet Gynecol 2004: 191: 607 |
|
Ralston SJ, Craigo SD. Ultrasound-guided procedures for prenatal diagnosis and therapy. Obstet Gynecol Clin North Am 2004; 31:101 |
|
Baba K, Okai T, Kozuma S et al: Fetal abnormalities: evaluation with real-time processible three-dimensional US – prelimary report. Radiology 1999; 211:441 |
|
Pretorious DH, Borok N, Coffler MS et al: Three dimensional ultrasound in obstetrics and gynecology. Rad Clin North Am 2001; 39:499-521 |
|
Chmail R, Pretorious D, Jones M et al: Prenatal evaluation of facial clefts with two-dimensional and adjunctive three-dimensional ultrasonography: A prospective study. Am J Obstet Gynecol 2002; 187:946 |