Electrosurgery: Principles, Biologic Effects and Results in Female Reproductive Surgery
Authors
INTRODUCTION
Despite a century of therapeutic use, the biologic effects of electricity as used in surgery are poorly understood by most clinicians, and the basics of electrophysics learned in school have been gratefully forgotten. Although we use the “Bovie” almost daily, our ignorance of the basic principles on which it operates is so deep that, for most of us, pressing the button to activate the electrosurgical unit (ESU) constitutes an act of faith rather than the knowledgeable application of scientific principles. It is surprising that extensive training courses, preceptorships, and documentation of cases are required before we receive credentialing for laser privileges, while the surgical application of electrical energy goes essentially unregulated, even though electricity is both more difficult to understand and to control than is laser light. It is the purpose of this chapter to present the basics of electrophysics in an intuitively understandable manner, without intimidating mathematical formulae, to review the English literature dealing with tissue effects, results and complications of electrosurgery, to present basic rules for the safe application of electrical energy in surgery, and to review the basic types of electrosurgical equipment that are available to the clinician.
The authors acknowledge the invaluable assistance of Taha Kadado and Tim Dickens in the preparation of the technical aspects of this manuscript, and Mohammed Kandeel, MD in the preparation and review of this manuscript.
ELECTROPHYSICS
Perhaps the easiest way to understand electrical concepts is by analogy. The properties of electrical current are similar to the flow of water in a stream. If one envisions the stream falling onto a waterwheel, it is easy to understand that the stream's ability to rotate the wheel (power) is a function of the amount of water falling on the wheel (current) and the height from which it falls (pressure or voltage) (Fig. 1). Similarly, the electrical power produced by a generator is the product of the electrical current (amperage) and the electrical pressure (voltage). Therefore, power (watts) = current (amps) × pressure (volts).
If water flows through a hose, the diameter of the hose limits the flow of the water, and is analogous to electrical resistance. If flow is held constant, increases in resistance can only be overcome by increasing pressure proportionately. This is governed by the only other equation that we will use: current (amps) = pressure (voltage)/resistance (ohms). The distance that the water will squirt from the end of the hose is a function of the pressure pushing the flow of the water. If the pressure is increased, the stream of water squirts farther, and with more force. Similarly, when electrical pressure is increased, the current becomes more difficult to control, and arcing from the electrode may be produced (analogous to water squirting forcefully from the end of the hose).
Types of Currents
Electrosurgical generators are capable of producing a variety of current waveforms. Depending on the clinical results desired, different waveforms can be used to produce differing tissue effects. An understanding of the ways in which the electrosurgical generator can modify current is necessary to better understand the options available to the surgeon.
Any current can be classified as either direct current or alternating (varying) current. Direct current is constant, never changing in direction (polarity) or magnitude. Direct current is the type produced by batteries. It is not used in electrosurgery because of its tendency to produce depolarization of neural and muscular tissue.
Alternating current is so named because its direction (polarity) changes (alternates). This type of current is similar to that which comes from electrical wall outlets. The rapidity with which the direction of current flow changes per unit of time is referred to as frequency, and is measured in Hertz (Hz). One complete cycle per second is one Hz. If a current alters polarity one million times per second, it is a one megahertz (MHz) current. Electrosurgical generators typically operate at frequencies between 400,000 Hz and 2.5 MHz, although some generators produce currents with frequencies as high as 3.5 MHz. Because these frequencies fall in the range of radio waves, electrosurgical generators are sometimes called radio frequency generators, and do, in fact, produce radio waves as a byproduct. Either excessively high or low frequencies can cause undesirable effects. Depolarization of susceptible tissues ceases at frequencies above 10,000 Hz. Excessively high frequencies tend to encourage current leakage.
Waveforms can be altered by the multiplication of two or more signals, a process called modulation. Damped and blended currents are examples of this process. There are three basic types of current waveforms used in electrosurgery: cutting current, coagulation current, and blended current.
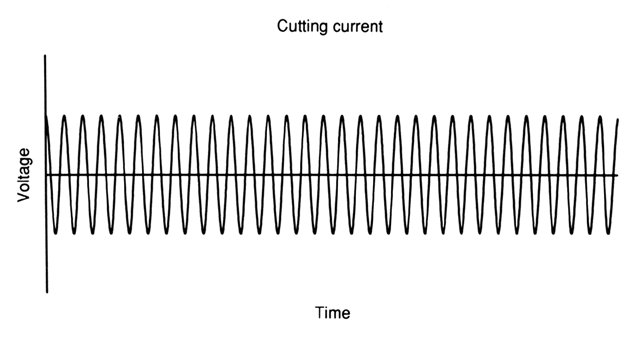
CUTTING CURRENT.
Cutting current uses a pure, nonmodulated sinusoidal waveform (Fig. 2). This waveform achieves a higher average power when compared with any other alternating waveform of equal peak voltage, allowing the voltage to be limited when compared with coagulation current. The high average power creates a higher current density than is allowed by other waveforms, facilitating a smooth cutting action without extensive thermal damage.
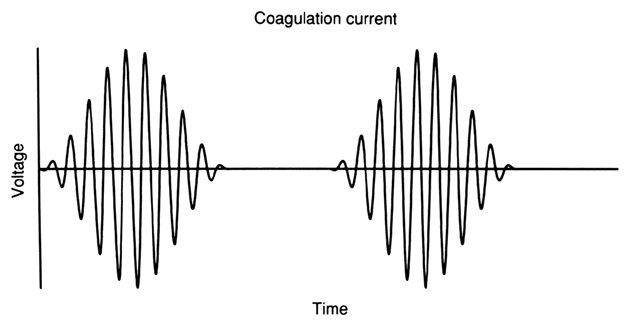
COAGULATION CURRENT.
Coagulation current is characterized by extensive wave modulation, which produces intermittent bursts of damped sine waves of high peak voltages (Fig. 3). These peak voltages result in high tissue temperatures, and hence significant thermal destruction, making this type of current particularly suited for the coagulation of bleeding vessels.
BLENDED CURRENTS.
Blended currents allow the surgeon to cleanly divide tissue while maintaining a variable degree of hemostasis, depending on the amount of coagulating current used. Blended currents are created by modulating a second, lower frequency, higher amplitude sine wave with the sine wave from the cutting generator, producing a higher peak-to-peak voltage. The new waveform is then delivered in intermittent bursts at a rate determined by the settings of the electrosurgical generator (i.e., Blend 1) (Fig. 4). This burst effect, although delivered at higher peak-to-peak voltage, contains a lower average power than a pure sinusoidal waveform because of the duty cycle.
A second form of time varying current is produced by the process of rectification, in which an alternating current is modified so that it becomes unidirectional. This may be done by either blocking the portion of the wave with a negative polarity (half wave or partial rectification) or by reversing the negative portion of the wave (full wave rectification). The current is time varying because its flow is not constant, even though its polarity does not change. Rectified currents are commonly used in dental and dermatologic surgery.
The Electrical Circuit
An electrical circuit is a closed current pathway along which electricity flows. Any current flowing in a direction or a path other than the intended one may lead to undesired outcomes, such as alternate site burns or shocks.
The type of circuit used in electrosurgery is termed a dipole circuit. A dipole circuit consists of two electrodes in contact with a dielectric or substance with limited electrical conductivity. In electrosurgery, human tissue is the dielectric. Diathermic heating occurs from electrical losses (transformation of the electrical energy into heat), which occur in the dielectric (tissue) located between the two electrodes. Two types of dipole circuits are possible: bipolar and unipolar.
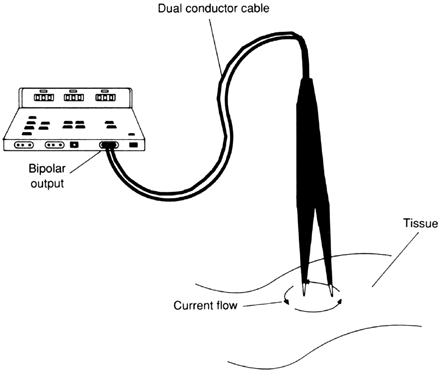
BIPOLAR.
A bipolar circuit uses two electrodes of equal size to create a dipole circuit. The bipolar circuit is the classical form of diathermic heating, in that the loss created in the dielectric is evenly dispersed between the electrodes. In electrosurgery, a high current density is applied through bipolar forceps, coagulating the small amount of tissue contained between the tips of the forceps with minimal effect on surrounding tissue. Both electrodes of the dipole are located at the site of coagulation (Fig. 5). Because the path of least resistance is the shortest distance between the electrodes, the probability of the current traveling via an alternate pathway is reduced.
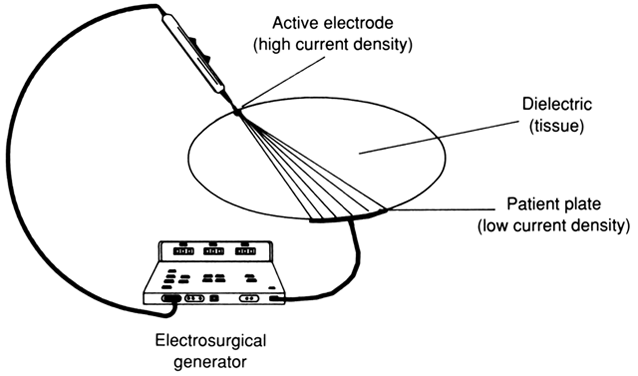
UNIPOLAR.
In unipolar electrosurgery, a small active electrode relative to the patient plate is used at the site of surgery. The high current density produced at the active electrode creates a pronounced diathermic effect, causing tissue destruction at the operative site. The patient plate's large size lowers the current density at its placement site, preventing unwanted burns. The term unipolar or monopolar is used because only one electrode of the dipole is located at the site of surgery (Fig. 6). Because the two electrodes (active and ground electrodes) and the patient's tissue form a circuit, the current's likely path is from the active electrode to the ground electrode and back to the electrosurgical generator, completing the circuit.
TISSUE EFFECTS
It is difficult to precisely predict the effects of electrical energy on tissue in the clinical setting. Many variables have the potential to affect results in electrosurgery. Because many of these parameters are not detailed by investigators, it is difficult to interpret differences in results reported in electrosurgical studies.
To understand electrosurgery, it must be clear that the effects obtained are the result of heat. This heat may be derived from an external source and transmitted to tissue by conductance (cautery), or, as in the case of both laser and electrosurgery, be produced within the tissue by an external source of energy. Due to the rapid changes in the direction (polarity) of current flow with the use of high frequency alternating current, there is no net transfer of electrons, and likewise, no movement of ions across cell membranes (depolarization). Part of the heat generated is from the tissue's impedance (resistance to current flow), but the majority of heat stems from the rapid vibration of molecules within the tissue under the effect of the changing electromagnetic field. That there is no essential difference in the effect of heat produced by any method was demonstrated by Lounsberry and associates,1 who were unable to detect any difference in tissue effect between cutting and coagulating currents of the same amperage, applied over the same area and for the same duration. Indeed, lesions produced by a soldering iron (cautery) were similar to electrical lesions. These findings were confirmed by Zervas and Kuwayama2 who found that lesions produced in rat brain and liver by induction heating, radio frequency electrocoagulation, and direct application of heat were similar for similar degrees of temperature elevation. The only application in which there may be a difference in both mechanism and effect is cutting with electrical current.
Although numerous electrosurgical tissue effects have been described and named, we can find evidence to justify defining only two effects by histologic changes (desiccation and coagulation) and two by observed surgical effects (fulguration and vaporization, commonly called cutting). We will briefly discuss each of these effects.
Desiccation
In 1924, Clark and colleagues gave the first histologic description of desiccation and coagulation.3 His description is classic and is accepted today without modification. Desiccation is produced by low current and relatively higher voltage applied over a broad area, producing a low current density. In desiccation, the cells are shrunken and shriveled with elongated nuclei. Cellular detail is preserved. This effect is produced by the loss of water from the cells, without extensive coagulation of proteins.
Coagulation
Coagulation occurs at higher current densities than are used in desiccation, resulting in higher tissue temperatures. The tissue fluids boil away and the proteins become denatured, forming a white coagulum similar to that produced when an egg white is boiled. There is loss of cellular definition as all tissue structures fuse into a formless, homogenous mass with a hyalinized appearance. This is the classic appearance of coagulation necrosis.
Fulguration
Fulguration results from the action of electrical arcs striking the tissue at widely divergent locations, producing a high localized instantaneous current density, but a low average current density. The characteristics of fulguration are the superficial nature of the tissue destruction, and the presence of large amounts of carbonization. The latter effect is due to the high temperature of the tissue at the point of the arc strike. The former occurs because, after the arc strikes, current is dispersed widely, causing rapid diminution of current density and little generation of heat in the deeper structures. The thin layer of carbon and the desiccated tissue beneath it form an insulating barrier, decreasing the probability of subsequent arc strikes in the same location. Fulguration requires low amperage and high voltages to overcome the resistance of the large distances between electrode and tissue.
Vaporization
The cutting of tissue by electrical current is due to the vaporization of cells. This is a special and interesting case, since the actual mechanisms of cutting remain controversial. As with the other tissue effects, the cutting action of electrical current is a product of current density. A dampened (coagulating) current can be made to divide tissue (albeit at the expense of great lateral thermal damage) by increasing the power or decreasing the electrode size, and an undamped, sinusoidal current (cutting) will produce coagulation if the current density is low, and the electrode contacts tissue. Every gynecologist who has performed tubal sterilization with the Wolf bipolar unit has made use of cutting current to produce coagulation of tissue.
Cutting tissue requires that a spark be present between the electrode and the tissue. An arc may be present in coagulating currents, and is necessary in fulguration. In the formation of an arc, little happens until a sufficient voltage is reached to allow the electrons to traverse the air gap between electrode and tissue. When this voltage is reached, electrons jump across the gap, causing ionization of the air molecules along the path of the spark. This ion path presents a low resistance pathway to the tissue, so long as the plasma (gas composed of ionized molecules) cloud is maintained. In a cutting current, the rapidly repetitive peak voltages occur before the plasma cloud can dissipate, so that each spark tends to follow the same pathway, maintaining ionization of the air along the spark path and striking the tissue at the same spot, generating a locally high current density. A dampened current reaches higher peak voltages than does an undamped one, but the peaks are separated by a longer period of time so that the plasma cloud dissipates between each peak. The low resistance path to tissue is lost, and the charring and dehydration of the tissue caused by the previous spark cause a locally higher resistance, decreasing the likelihood of a subsequent spark striking the same spot. The net result is a lower current density, and coagulation. McLean4 demonstrated the essential difference between the arcs used in cutting and the other effects by photographing the sparks caused by damped and undamped currents and found that the former caused a broad, brushlike spark (low current density), while the latter caused a tight, pencil-like arc (high current density).
It is thought that the cutting effect on tissue stems from the extremely high localized current density causing boiling of the intracellular water, and exploding the cells.5 Lateral damage is minimized by heat being carried off by the steam.
Pearce5 postulated a second mechanism of cellular disruption. When an intense electromagnetic field impinges on an absorbing tissue, the rate of vapor formation cannot keep up with the rate of energy input. To maintain thermodynamic equilibrium, an acoustic wave is generated, disrupting the cell. This may account for the description by Eisenmann and co-workers6 of an individual mast cell divided without disturbance of the intracellular structures.
The degree of tissue damage caused by electrical energy is determined by numerous factors in addition to waveforms and current density. The electrical resistance of the tissue is important as is the inherent sensitivity of the tissue to damage by heat. Maness and associates 7 found that epithelium is more sensitive to damage than is connective tissue or muscle in hamster tongue. Luciano and colleagues8 found that, in rabbits, the ovary was less susceptible to damage than the uterus.
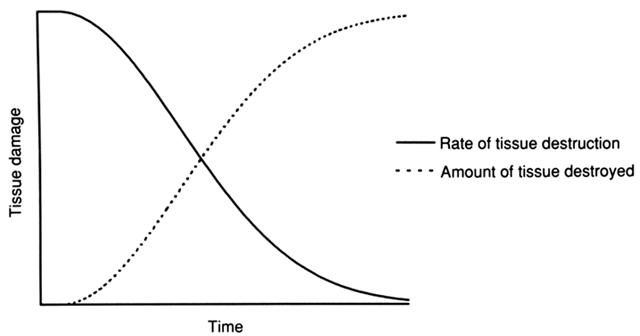
The degree of tissue damage is also affected by the duration of the energy application, with increasing levels of damage being seen with longer applications. The rate of tissue destruction, however, decreases with increasing duration of application; after the resistance of the destroyed tissue becomes greater than the ability of the current to penetrate it, no further damage occurs (see Fig. 7). Increasing levels of power also tend to increase the degree of tissue damage, with increasing amperage causing more damage than increased voltage.4 Because current density increases inversely as the square of the radius of the electrode, tissue damage is likely to increase with increasing electrode size.7
|
The case of bipolar coagulation deserves special consideration because of its unique features, and widespread use in laparoscopic surgery. Greenwood9 first described bipolar electrocoagulation in 1940. Ramsay and colleagues 10 found that bipolar coagulation required less power than unipolar, and would operate regardless of the medium in which it was used, permitting coagulation in a fluid environment, a great advantage when attempting to coagulate in a wet field. The main advantage of bipolar coagulation to the gynecologist is its limited spread of electrical effect. Due to the fact that the tissue to be coagulated is nearly, but not entirely, isolated from the rest of the body between the blades of the forceps, the current flow is essentially limited to this area. It must be noted that some current leakage does occur11 with the use of bipolar forceps, and there is the attendant potential for unintended tissue damage. Because bipolar coagulators require less voltage,5 there is less likelihood that current will follow unexpected pathways, such as sparking to adjacent structures.
The problem of unintended tissue damage has often been attributed to current's purported tendency to concentrate around, and follow, certain structures such as blood vessels, causing unphysiologic heating and subsequent damage. Our review of the literature found little evidence to support this concept. It must be realized that current will tend to distribute through tissue in such a manner as to minimize the potential difference between the electrodes. In simpler terms, current will follow the path of least resistance. If an alternate parallel pathway is available, which taken together with the first path will lower its resistance, a portion of the current will flow along this alternate path. Because heat is a product of the current and the resistance across which it is flowing, it is unlikely that, assuming an adequate dispersive electrode is used, the current density distant from the treatment electrode would be high enough to cause sufficient temperature elevation to damage the tissue in a location other than at the treatment site. The idea that current sufficient to cause tissue damage could travel preferentially along blood vessels or nerves seems to have little support in the literature. Lounsberry and co-workers 1 found no evidence for electrocoagulation effects following vessels or lymphatics, and in fact found that the cooling of the circulation appeared to have a protective effect.
Even though the reason for the use of radiofrequency current (as opposed to low frequency current) is to avoid stimulation of excitable tissues, particularly nervous tissue and muscle, we have all noted localized contractions when attempting to cut muscle or to coagulate bleeding points on it. This paradox is explained by the observation that, as the duration of the stimulus is decreased (i.e., increasing the frequency of the current), stimulation can still be obtained by increasing the strength (current density) of the stimulus. Thus, high frequency current will cause localized muscle contraction where the current density is high, but generalized muscle contraction will be avoided as the current disperses away from the treatment electrode and the current density drops.
HEALING OF ELECTROSURGICAL WOUNDS
Results achieved with electrosurgery, after healing has taken place, depend on the goal of the surgeon and his or her ability to tailor the current to suit the task at hand. Electrosurgery can be used to achieve either the mass destruction of large volumes of tissue as in endometrial ablation, fine incision as required by the reconstructive microsurgeon, or tasks requiring both cutting and coagulation, such as incision of the skin and subcutaneous tissue with hemostasis. Because of failure to control for variations in technique and equipment, it is difficult to draw firm conclusions from studies comparing healing in tissue divided with electricity and other methods. Reported results are often contradictory. While acknowledging the difficulties inherent in any attempt to interpret the literature in this area, we will review available data dealing with results achieved in animal studies, and then proceed to examine the clinical studies in both reproductive and general gynecologic surgery.
The manner in which tissue heals following electrosurgery is a function of the lateral thermal damage and attendant tissue necrosis caused by the application of electrical energy. Higher current densities and decreased duration of exposure cause less damage, and lower current densities and longer exposure cause more. McLean4 showed that all electrosurgical lesions demonstrated areas of desiccation, coagulation, and carbonization regardless of the power or waveform used. Regardless of the technique used, some degree of lateral thermal damage will always exist, although its extent can be limited to minute areas. The tissue necrosis resulting from heat damage, if excessive, can result in poor healing and increased likelihood of infection.
Numerous animal studies comparing electrosurgical incision to incision by other modalities are available. Although results are often contradictory, it would appear from review of these studies that electrosurgical incisions do not heal as well as those made by scalpel,12 apparently due to the volume of tissue destroyed.13,14 Electrosurgical incision also appears to be more susceptible to infection.14 While some investigators found that after healing was complete electrosurgical incisions were weaker than scalpel incisions,13 others15 found no difference in strength when the incision was made with cutting current and a small electrode.
Studies have shown that electrosurgery may be more,16 equally,17 or less18 adhesiogenic when compared to laser surgery in various animal models.
CLINICAL RESULTS IN INFERTILITY SURGERY
In surgery, the purpose of which is to restore or preserve reproductive function, hemostasis and minimal tissue trauma are of paramount importance. Techniques that minimize postoperative bleeding and tissue necrosis should produce fewer adhesions to compromise reproductive function, and thus offer improved results.
Of the various mechanical factors contributing to female infertility, the two most common are pelvic adhesive disease and endometriosis. Several studies have reported results achieved by the use of either CO2 laser or electrosurgery in the treatment of these conditions, but only a few have directly compared the two modalities.
Tulandi and Vilos19 performed salpingostomy in 67 patients with distal tubal occlusion using either CO2 laser at 5000 to 10,000 W/cm2 or unipolar electrosurgery, and showed that the intrauterine pregnancy rates in both groups were similar. The same group of investigators,20 performing salpingoovariolysis for pelvic adhesions with either CO2 laser or electrosurgery showed similar intrauterine pregnancy rates. In an earlier study, Tulandi and colleagues 21 performed salpingostomy with either CO2 laser 5000 to 10,000 W/cm2 or bipolar electrosurgery, and showed similar intrauterine pregnancy rates after 1 year of follow-up.
The results of studies of the surgical management of endometriosis indicate that when the disease is appropriately staged, there appears to be no significant difference in outcome whether the CO2 laser or electrosurgery is used.22,23
There are several potential problems when interpreting results of studies of this nature. Study design, randomization, and staging of disease vary between studies. Moreover, in the case of pelvic inflammatory disease, the degree of damage and subsequent function of the endosalpinx is never known.
CLINICAL RESULTS IN GENERAL GYNECOLOGIC SURGERY
Several studies have examined the role of electrosurgery in abdominal wall incision in humans. In a large, prospective, unrandomized, uncontrolled study of 23,649 surgical wounds, Cruse and Foord24 found that use of the electrosurgical knife doubled the infection rate across all categories of operations. In a prospective, randomized study of 88 cholecystectomy incisions, Pearlman and associates25 determined that electrosurgical incisions were faster to perform, and more hemostatic than incisions made with either a scalpel or a laser, but that there was no difference in postoperative pain and wound healing. The number of infections and wound hematomas was similar among the three groups. The power of this study was such that it was calculated to have an 80% chance of detecting a 25% difference in each parameter. In a prospective study26 of 49 consecutive radical mastectomies randomized between scalpel and electrosurgery, it was found that there was significantly less blood loss with electrosurgery, but no difference in the postoperative hemovac drainage, and the infection rate was the same. Johnson and Serpell27 randomized 240 patients undergoing abdominal incisions between scalpel and electrosurgery, and found no difference in incision time or infection rate. Finally, Mann and Klippel28 studied 125 children with bilateral inguinal hernias. One side was opened electrosurgically, and the other with a scalpel, so that each patient functioned as his own control. There were no infections, and 4 to 6 weeks postoperatively healing appeared to be equal.
Despite animal data to the contrary, we found little evidence in human clinical studies to support the hypothesis that electrosurgical incision is in any way inferior to the scalpel, and, in fact, may offer the advantages of decreased operating time and decreased blood loss.
COMPLICATIONS
Complications of electrosurgery can be categorized as: the potential for the explosion of combustible gases, either anesthetics or bowel gas, interference with pacemakers and monitors, neuromuscular stimulation including ventricular fibrillation, accidental burns, and the potential for the transmission of infection.
One of the advantages attributed to electrosurgery was the purported ability to sterilize the field in which it was used. Recent concern sparked by the acquired immunodeficiency syndrome epidemic has prompted reevaluation of this concept. Transfer of both bacteria29 and viruses30 by electrosurgical electrodes has been demonstrated, proving that the electrode is not sterilized by the electrical discharge. Because gynecologists are, we hope, unlikely to use electrodes on more than one patient without intervening sterilization, the transmission of disease between patients is unlikely. With the advent of in office electrosurgical excision procedures involving tissues presumably infected by oncogenic subtypes of the human papillomavirus, the gynecologist performing these procedures must be vigilant regarding sterilization technique if disposable electrodes are not used. Steam or gas sterilization is preferred over soaking. Of greater concern to the gynecologist is the potential for transmission of disease from patient to physician or ancillary personnel. Colver and Peutherer30 demonstrated that discharge of an electrosurgical current over a droplet of fluid caused spattering over a distance of at least 5 cm. Because electrosurgery causes an expansion of tissue fluids resulting in the explosion of cells, an aerosol of blood and fluid droplets is created that can potentially transmit infectious agents. It is important, regardless of the surgical procedure being performed, that all personnel observe universal precautions. Smoke generated by electrosurgical procedures is mutagenic, giving additional impetus to the recommendation that surgical masks be worn.
Burns to the patient's skin can occur in a variety of ways. The most common mechanism is the alternate site burn, which results from a high current density either at a poorly applied ground electrode, at the site of monitoring devices such as ECG electrodes or temperature probes, or at the sight of accidental contact with a grounded metal object. These burns must be distinguished from decubitus ulcers and chemical burns. All electrosurgical burns are visible at the time of occurrence. Late appearing burns are due to some other factor. Most modern electrosurgical generators are isolated from earth ground, and have fault monitors that will disable the machine and sound an alarm if the ground electrode circuit is not intact. While these features minimize the incidence of alternate pathway burns, several measures can be taken to make them even less likely to occur. Ground electrodes should not be placed under warming blankets, as there will be an additive heating of tissue. The best site for placement of the dispersive electrode is one with low tissue impedance between it and the active electrode. In pelvic surgery, the top of the thigh is the preferred location to minimize the distance between the electrodes. Monitoring devices should not be placed between the active and ground electrodes. There is a marked increase in current through ECG leads when they are positioned in this manner. Most importantly, if an ESU requires unusually high settings to function, a faulty ground should be suspected, and the ground electrode and its entire circuit checked.
Other mechanisms of skin burns involve the ignition of paper drapes or antiseptic solutions, particularly alcohol, used in skin preparation. If alcohol is used to prepare the skin prior to surgery, time should be allowed to ensure that it has fully evaporated prior to draping the patient.
Historically, explosive anesthetic gases posed the greatest explosion risk in the operating room. Fortunately, these agents are rarely used today. If they are used, the surgeon should be so informed, and use of the ESU avoided. Of greater concern is bowel gas, which frequently contains a mixture of methane and hydrogen which, when mixed with oxygen, even in low concentrations, are highly explosive. This is a real hazard when operating around the large bowel, or when performing anorectal surgery.
Nitrous oxide supports combustion, as well as pure oxygen. Many gynecologists use nitrous oxide as a laparoscopic distention medium to avoid the peritoneal irritation caused by carbon dioxide. If electrosurgery is to be used during a laparoscopic operation, the use of nitrous oxide to distend the abdomen should be avoided.
Although most modern cardiac pacemakers are resistant to interference by extraneous electromagnetic signals, several incidences of asystole and cardiac arrest have been reported when electrosurgery is used in patients with pacemakers.31 These problems occur predominantly in patients with older demand pacers. In these units, the electrosurgical signal may block the pacer's inhibition amplifier allowing an R-on-T phenomenon to occur, leading to ventricular fibrillation. Aside from the special case of the cardiac pacemaker patient, with the use of radio frequency currents, cardiac arrhythmia due to discharge from an ESU should be an almost nonexistent event.
Until the recent upsurge in interest in operative laparoscopy, reports of burns of the bowel during laparoscopic sterilization had rendered the use of unipolar techniques in laparoscopy almost untenable. In 1973, the Complications Committee of the Association of Gynecologic Laparoscopists32 reported burns to skin or bowel occurring at a rate of 2.3 per 1000 patients undergoing sterilization by unipolar electrosurgery. In the same year, Thompson and Wheeless33 reported 10 burn injuries of the intestines occurring in a cohort of 3600 patients undergoing unipolar laparoscopic sterilization. Four of these injuries were noted at the time of surgery and treated with observation alone due to the small, superficial nature of the injury. This group experienced uneventful recovery. A fifth patient with a small burn had the site oversewn, although she could have been treated by observation. In five additional cases, the injury was unrecognized and resulted in delayed perforation. Most of the burns occurred on the terminal ileum. As we shall see presently, it is important that burns occurred with both one- and two-puncture techniques. In 1975, Loffer and Pent34 reviewed the 71 electrical complications of laparoscopy that had been reported at that time. Twenty-five cases involved burn injuries to the abdominal wall, and 44 cases, burns of the bowel. Of these 44 cases, the ileum was involved in 39 and the colon in five. Schwimmer35 reported two superficial intestinal burns occurring in 410 sterilization procedures performed with a two-puncture technique, using unipolar current. In 1979, Maudsley and Qizilbash36 reported an additional four small bowel injuries among 7466 consecutive procedures, all performed with a two-puncture technique. The mechanism of these injuries is controversial. Mechanisms involving current arcing to the bowel at distant sites, arcing from tube to bowel, and creation of a capacitor have been proposed. Each of these mechanisms appears equally improbable. It requires about 30,000 volts to cause dielectric breakdown of a 2.5 cm air space.37 Because the bowel, the active electrode and the dispersive electrode are connected by much lower resistance circuits than air, it is difficult to rationalize sparking between biologic structures although surgeons have reported seeing sparks jumping from the tube to the bowel.
Theoretically and in practice, a capacitor can be inadvertently constructed using unipolar current and a single puncture technique. This would allow several thousands of volts of electrical energy to accumulate in the laparoscope barrel—more than enough to allow arc formation between laparoscope and nearby bowel. Although this mechanism may account for some of the reported injuries, it is unlikely to account for the majority because most cases occurred with a double puncture technique. It is possible that faulty insulation on the unipolar electrode, passed either through an insulated or a metal sheath may account for some of the injuries. Perhaps the most likely mechanism was proposed by Engle and Harris37 who studied the electrodynamics of tubal coagulation. It was found that, initially, with the electrode in good contact with the intact tube, as tissue heating began, the resistance dropped, and no sparking occurred. As coagulation proceeded and the tissue fluids boiled away, the resistance increased. When resistance became so high that tissue contact was poor, sparking occurred from the electrode to the nearest moist tissue. This effect was related to the peak voltage. It is recommended that tubal coagulation be carried out with the lowest effective power and a cutting current to limit peak voltage. The bipolar electrosurgical forceps have been adapted to gynecologic use as a highly successful remedy to the problem of inadvertent bowel burns.
One additional potential cause of inadvertent electrical injury deserves discussion. When unipolar current is applied to a structure on a stalk, the current tends to concentrate at the base of the stalk causing coagulation of the blood supply to the structure. While this may be applied to advantage in treatment of condylomas and papillomas, there is a potential for disastrous consequences if unipolar coagulation is used to control bleeding during circumcision.
GENERATORS
Electrosurgical generators are devices that produce currents of various waveforms suitable for surgical applications. The earliest electrosurgical generators used a spark gap system to produce a highly damped, high frequency current. Shortly thereafter, valve (vacuum tube) generators, which had the capability of producing smooth, sine wave currents, suitable for cutting of tissue, were introduced. The tube generator has since been replaced by the modern, solid state generator. The spark gap and the solid state generators are the two types in modern usage. Solid state generators produce the coagulation waveform by using transistors and solid state components to produce high voltage bursts of current. Spark gap generators produce damped, high voltage bursts of current by discharge of air-spaced plates. Spark gap systems are incapable of producing undamped, cutting current.
Even though solid state generators are capable of producing peak voltages in excess of 10,000 volts, unlike spark gap units, they usually cannot maintain the same level of power output at high impedance loads, as when working in a fluid environment.
Cutting and coagulating currents are produced from two independent generators contained within the ESU. Contrary to what is popularly understood, blended currents from solid state electrosurgical systems are produced by the cutting current generator, and consist of a simple, damped waveform, much less drastic in modulation than the coagulation current. Earlier models, such as combination spark-gap/valve generators, combined the outputs of the cutting generator and the coagulation generator to produce a blended waveform with a much higher root mean square power. In either case, the desired effect is to enable the standard electrode to cleanly divide tissue, while causing sufficient thermal effect to produce hemostasis.
SAFETY ISSUES
Frequency
Several considerations place limits upon the frequencies at which electrosurgical generators can operate.
Frequencies in excess of 10,000 Hz are necessary to produce the diathermic effect without neuromuscular stimulation. Frequencies below this threshold cause undesirable depolarization of susceptible tissues (heart, muscle, and nervous tissue), leading to tonic and clonic muscle contractions with rhabdomyolysis and cardiac dysrythmias.
Similarly, the use of excessively high frequencies can lead to surgical complications. The primary concern in the design of electrosurgical equipment is the containment of the current pathway within its intended circuit. Travel of the current via alternate, undesired pathways can produce electrosurgical burns. Such an alternate pathway may occur via a pathway of less capacitive resistance to ground. Capacitance, technically defined as leakage or loss of current through an insulator, occurs with much greater efficiency as the frequency of the current increases, allowing shunting of current to occur (short circuit). Capacitive coupling can occur if other electrical circuits, such as monitoring equipment, are in contact with the patient, causing alternate site burns.
It is for these reasons that almost all contemporary electrosurgery generators use a frequency of between 400,000 Hz and 2.2 MHz, with the most commonly used generators producing either 600,000 Hz or 750,000 Hz.
Grounding
In electrosurgery, the term grounding refers to the relationship between the dipole circuit (active electrode—patient—return electrode) and earth ground. Three relationships between the dipole circuit and earth ground are possible: direct grounding, isolated grounding circuitry, and capacitive grounding circuitry.
DIRECT GROUNDING.
The return portion of the circuit is connected directly to earth ground. Direct grounding is considered unsafe and is not used in modern ESUs because it invites numerous alternate pathway possibilities with the potential for unintended burns.
ISOLATED GROUNDING CIRCUITRY.
The output current is floating free of ground. This means that the patient ground is referenced to the ESU rather than earth ground. The machine is, in turn, referenced to earth ground via special circuitry that avoids alternate pathways. If the patient should inadvertently be placed in contact with another grounded object, this circuit will detect the loss of current and disable the ESU. It should be noted that if an ESU using isolated grounding circuitry is activated while the active electrode is not in contact with the patient, it is possible for alternate site burns to occur if the patient is in contact with a direct path to ground (i.e., a metal operating table), and the ESU is not equipped with a sentry circuit.
CAPACITIVE GROUNDING CIRCUITRY.
The return circuit is referenced to earth ground via a capacitive circuit, which reduces some of the hazards of electrosurgery by acting as an electrical filter, permitting only currents of specific frequencies to flow through the return plate attached to the patient.
Each grounding scheme has inherent advantages and disadvantages. In response to the need of many surgical disciplines for a very effective spray coagulation current, many manufacturers of ESUs have increased the performance of their solid state generators to deliver 10,000 or more volts. Since high voltages have the ability to seek and find earth ground effectively, it is difficult to contain these higher voltages with an isolated circuit. Because direct grounding would only compound this problem, when high voltages are used, capacitive grounding becomes the method of choice. Most generators producing lesser voltages use isolated circuitry because this grounding scheme reduces alternate pathway possibilities and is generally considered to be the safest.
Monitoring of the Return Electrode
Electrosurgical generators built prior to the late 1960s did not incorporate any type of monitoring device to ensure proper connection of the return electrode, either to the patient or the ESU. Reports of burn injuries caused by a breach of the return electrode pathway led manufacturers of electrosurgical equipment to develop a monitor circuit, which incorporated a dual conductor patient cable, rather than a simple single conductor cable. A low voltage current is introduced into the circuit and travels through one conductor of the patient cable, through the patient plate, and back to the unit via the second conductor of the cable. If this low voltage current is interrupted for any reason, an alarm sounds and the unit's output is disabled.
This should be considered the minimum requirement from a safety standpoint for any electrosurgical generator. This monitoring system does have one limitation: it cannot detect proper adherence of the return electrode to the patient. It only assures that the patient cable is connected to the generator.
The return electrode monitor (REM) system was introduced by ValleyLab in the early 1980s, and offers one further level of safety in preventing alternate site burn injuries by ensuring that the return electrode is properly attached to the patient. With REM monitoring, a special return electrode with two separate, isolated areas is used. A low voltage current is introduced into one of the conductive areas of the pad. If the pad is evenly applied to the patient's skin, the current is transmitted to the second conductive area, and back to the ESU, completing the circuit.
The REM circuit is calibrated to detect a range of impedance, which would be expected between one side of the return electrode pad and the other with the patient's skin as the dielectric. Impedances outside this precalibrated range will engage the REM alarm and disable the unit's output. This circuit is not impossible to defeat, as surfaces such as the conductive floor of the operating room may provide an impedance within the monitor's range. It must, therefore, be stressed that there is still no substitute for carefully and properly applying the return electrode to the patient.
The two mechanisms by which patient injury is most likely to occur are the use of excessively high peak voltages and improper patient grounding. To avoid injury, the surgeon should personally ensure that the patient is positioned in such a manner that there is no contact with metal or other conductive objects that may potentially be grounded, and that the return electrode is properly applied and connected to the ESU. The lowest effective settings on the ESU should be used to limit peak voltages and, for the same reason, a cutting rather than a coagulating waveform should be employed where possible. If excessively high power settings are needed to achieve the desired tissue effect, a faulty ground should be suspected, and the use of the ESU stopped until safe function has been assured.
REFERENCES
Lounsberry W, Goldschmidt V, Linke CA et al: The early histologic changes following electrocoagulation. J Urol 86 (3): 321, 1961 |
|
Zervas NT, Kuwayama A: Pathological characteristics of experimental thermal lesions. Comparison of induction heating and radiofrequency electrocoagulation. J Neurosurg 37: 418, 1972 |
|
Clark WL, Morgan JD, Asnis EJ: Electrothermic methods in treatment of neoplasms and other lesions, with clinical and histological observations. Radiology 2: 233, 1924 |
|
McLean AJ: Characteristics of adequate electrosurgical current. Am J Surg 18 (3): 417, 1932 |
|
Pearce JA: Electrosurgery. New York, John Wiley and Sons, 1986 |
|
Eisenmann D, Malone WF, Kusek J: Electron microscopic evaluation of electrosurgery. Oral Surg 29 (5): 660, 1970 |
|
Maness WL, Rober FW, Clark RE et al: Tissue damage from electrosurgical power output variations in hamster tongues. J Prosthet Dent 42 (4): 456, 1979 |
|
Luciano AA, Randolph J, Whitman P et al: A comparison of thermal injury, healing patterns, and postoperative adhesion formation following CO2 laser and electromicrosurgery. Fertil Steril 48 (6): 1025, 1987 |
|
Greenwood J: Two point coagulation. A new principle and instrument for applying coagulation current in neurosurgery. Am J Surg 50: 267, 1940 |
|
Ramsay JWA, Shepherd NA, Butler M et al: A comparison of bipolar and monopolar diathermy probes in experimental animals. Urol Res 13: 99, 1985 |
|
Vallfors B, Erlandson B, Hanson H, Weick B: Current leakage in bipolar electrocoagulation. Neurosurgery 13 (2): 111, 1983 |
|
Ellis JD: The rate of healing of electrosurgical wounds as expressed by tensil strength. JAMA 96: 16, 1931 |
|
Ritter EF, Demas CP, Thompson DA, Devereux DF: Effects of method of hemostasis on wound infection rate. Am Surg 56 (10): 648, 1990 |
|
Goltesman J, Perla D, Zeigler JM: The effects of electrocautery on normal tissue. Surg Gynecol Obstet 51: 667, 1930 |
|
Sozio RB, Riley EJ, Shklar GS: A histologic and electronic evaluation of electrosurgical currents: Nonfiltered fullwave modulated vs. filtered current. J Prosthet Dent 33 (3): 300, 1975 |
|
Bellina JH, Hemming SR, Varos JI, Ross LF: Carbon dioxide laser and electrosurgical wound study with an animal model: A comparison of tissue damage and healing patterns in peritoneal tissue. Am J Obstet Gynecol 148 (3): 327, 1984 |
|
Filmar S, Jetha N, McComb P, Gomel V: A comparative histologic study on the healing process after tissue transection: I. Carbon dioxide laser and electromicrosurgery. Am J Obstet Gynecol 160 (5): 1062, 1989 |
|
Fayez JA, McComb JS, Harper MA: Comparison of tubal surgery with the CO2 laser and the unipolar microelectrode. Fertil Steril 40 (4): 476, 1983 |
|
Tulandi T, Vilos GA: A comparison between laser surgery and electrosurgery for bilateral hydrosalpinx: A two year follow-up. Fertil Steril 44 (6): 846, 1985 |
|
Seiler JC, Gidwani G, Ballard L: Laparoscopic cauterization of endometriosis for fertility: A controlled study. Fertil Steril 46 (6): 1098, 1986 |
|
Tulandi T, Farag R, McInnes RA et al: Reconstructive surgery of hydrosalpinx with and without the carbon dioxide laser. Fertil Steril 42 (6): 839, 1984 |
|
Adamson GD, Lu JJ, Subak LL: Laparoscopic CO2 laser vaporization of endometriosis compared with traditional treatment. Fertil Steril 58 (5): 704, 1988 |
|
Murphy AA, Schlaff WD, Hassiakos D et al: Laparoscopic cautery in the treatment of endometriosis related infertility. Fertil Steril 55 (2): 246, 1991 |
|
Cruse PJE, Foord R: A five year prospective study of 23,649 surgical wounds. Arch Surg 107: 206, 1973 |
|
Pearlman NW, Steigman GV, Vance V et al: A prospective study of incisional time, blood loss, pain and healing with carbon dioxide laser, scalpel, and electrosurgery. Arch Surg 126: 1018, 1991 |
|
Miller E, Paull DE, Morrissey K et al: Scalpel versus electrocautery in modified radical mastectomy. Am Surg 54 (5): 284, 1988 |
|
Johnson CD, Serpell JW: Wound infection after abdominal incision with scalpel or diathermy. Br J Surg 77: 626, 1990 |
|
Mann W, Klippel CH: Electrosurgical skin incision. J Pediatr Surg 12 (5): 725, 1977 |
|
Bennett RG, Kraffert CA: Bacterial transference during electrodessication and electrocoagulation. Arch Dermatol 126: 751, 1990 |
|
Colver GB, Peutherer JF: Herpes simplex virus dispersal by Hyfrecator electrodes. Br J Dermatol 117: 627, 1987 |
|
Batra YK, Bali IM: Effect of coagulating and cutting current on a demand pacemaker during transurethral resection of the prostate. A case report. Can Anaesth Soc J 25 (1): 65, 1978 |
|
Hulka JF, Soderstrom RM, Corson SL et al: First annual report of the complications committee of the American Association of Gynecologic Laparoscopists. J Reprod Med 10: 301, 1973 |
|
Thompson BH, Wheeless CR: Gastrointestinal complications of laparoscopy sterilization. Obstet Gynecol 41 (5): 669, 1973 |
|
Loffer FD, Pent D: Indications, contraindications and complications of laparoscopy. Obstet Gynecol Surv 30 (7): 407, 1975 |
|
Schwimmer WB: Electrosurgical burn injuries during laparoscopy sterilization. Obstet Gynecol 44 (4): 526, 1974 |
|
Maudsley RF, Qizilbash AH: Thermal injury to the bowel as a complication of laparoscopic sterilization. Can J Surg 22 (3): 232, 1979 |
|
Engle T, Harris FW: The electrodynamics of laparoscopic sterilization. J Reprod Med 15 (1): 33, 1975 |