Genetic History Taking and Counseling
Authors
INTRODUCTION
The relative burden of major birth defects (2–3% of all deliveries) has increased as deaths due to infection and prematurity decrease. Concurrent with this trend are the genetic advances of the past few decades. Maternal and child health delivery systems must adapt to these trends, and integral to adaptation is a systematic approach for eliciting information about risks of genetic disorders. Public and professional awareness of the importance of genetics in virtually every aspect of medicine has increased expectations that this new knowledge will be introduced rapidly into the healthcare of individuals.
This chapter discusses taking a genetic history. Those disorders amenable to genetic screening are enumerated, along with the principles underlying initiation of screening programs. Finally, the chapter addresses psychological ramifications of genetic counseling. This work also reflects previous contributions by the authors.1, 2
THE ROUTINE GENETIC HISTORY
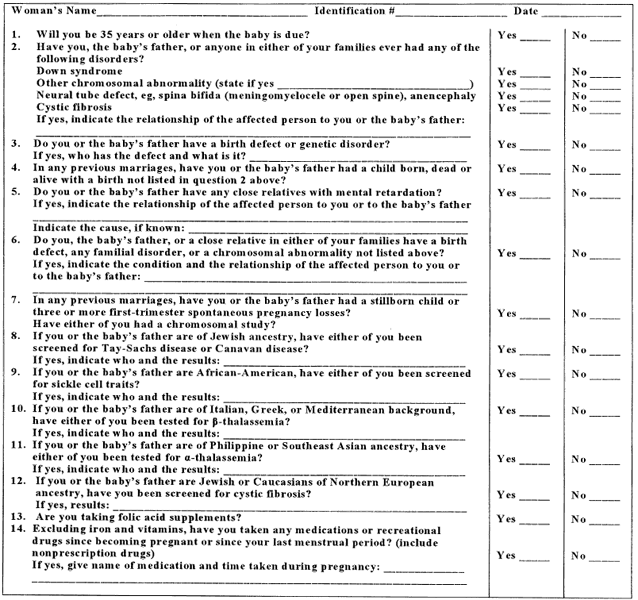
Some obstetricians consider it useful to obtain genetic information through use of questionnaires or checklists, often constructed to require action only to positive responses. Figure 1 reproduces a form that was recommended by the American College of Obstetricians and Gynecologists.2 A similar form was used by the authors in a survey.3 It showed that 21.4% of couples in a prenatal clinic gave at least one positive response, with 7.8% of the original sample couples requiring formal genetic counseling. Advanced maternal age was the most common indication.3 For all pregnancies, it is standard practice to determine whether a couple or anyone on either side of their families has a disorder that might prove heritable. The same holds true for gamete donors.4, 5, 6
|
One should inquire about the health status of first-degree relatives (siblings, parents, offspring), second-degree relatives (uncles, aunts, nephews, nieces, and grandparents), and third-degree relatives (first cousins). Record abnormal reproductive outcomes such as repetitive spontaneous abortions, stillbirths, and anomalous liveborn infants.4, 5, 6
The subsequent counseling may be sufficiently complex to warrant referral to a geneticist, or it may prove sufficiently facile for the informed clinician to handle. If a birth defect is detected in a second- or third-degree relative, the likelihood of that anomaly occurring in a pregnancy rarely proves significantly increased. Identification of a second- or third-degree relative with an autosomal-recessive trait ordinarily places the couple at little increased risk for an affected offspring, the exception being if the couple is consanguineous. Nonetheless, one should inquire about the status of relatives as distant as first cousins (of the fetus) because identification of certain disorders in such relatives may be the only clue that the couple may be at increased risk for autosomal-dominant disorders characterized by decreased penetrance or for X-linked recessive disorders.
In addition to identifying relatives with genetic disorders, one should record drug exposure to the woman and her partner, and identify not only those agents currently received but also noxious agents to which exposure occurred before pregnancy. The latter could be mutagenic.
Parental ages should also be recorded. Indeed, the most common indication for prenatal diagnosis is advanced maternal age, which warrants discussion regardless of a patient's difficulties in achieving pregnancy and regardless of a physician's personal convictions regarding prenatal diagnosis and pregnancy termination. Offspring of fathers in their fifth decade and beyond are at increased risk of new dominant mutations.
Ethnic origin should be recorded. This is relevant to the genetic screening issues discussed later in this chapter. Ashkenazi Jews are at increased risk for offspring with Tay-Sachs disease and, hence, should be screened to determine heterozygote status (frequency is 1 in 27). The same holds true for Canavan disease. In the United States, Jewish people often are uncertain whether they are of Ashkenazi or Sephardic descent; thus, we recommend screening to all Jewish couples, as well as couples in which only one partner is Jewish. It also is advisable to offer routine heterozygote screening for β-thalassemia in Mediterranean people, sickle cell anemia in blacks, and α-thalassemia in Southeast Asians, Chinese, and Filipinos.7 Table 1 summarizes recommendations for screening.
Table 1. Heterozygote screening
Ethnic Group | Disorder | Screening Test | Definitive Test |
Ashkenazi Jews | Tay-Sachs disease | Decreased serum hexosamidase-A, possibly molecular analysis | CVS or amniocentesis for enzymatic assays or molecular analysis to detect affected fetus for hexosamidase-A |
Canavan disease | DNA analysis to detect most common alleles | CVS or amniocentesis for molecular analysis to determine genotype of fetus | |
Familial dysautonomia | DNA analysis to detect most common alleles | CVS or amniocentesis for molecular analysis to determine genotype of fetus | |
Blacks | Sickle cell anemia | Presence of sickle cell hemoglobin, confirmatory hemoglobin electrophoresis | CVS or amniocentesis for genotype determination (direct molecular analysis) |
Mediterranean people | β-Thalassemia | MCV <80%, followed by hemoglobin electrophoresis | CVS or amniocentesis for genotype determination (direct molecular analysis or linkage analysis) |
Southeast Asians and Chinese (Vietnamese, Laotian, Cambodian, Filipinos) | α-Thalassemia | MCV <80%, followed by hemoglobin electrophoresis | CVS or amniocentesis for genotype determination (direct molecular analysis or linkage analysis) |
All ethnic groups | Cystic fibrosis | DNA to detect specified panel of CFTR mutations (those present in | CVS or amniocentesis for fetal genotype determination. Definitive diagnosis on all fetuses is not possible, sensitivity varying by ethnic group |
CVS, chorionic villus sampling; MCV, mean corpuscular volume.
Some centers offer not only Tay-Sachs, Canavan disorders, cystic fibrosis, and familial dysautonomia to Jewish couples but also screening for Gaucher disease, Niemann-Pick disease, and other disorders (e.g., Bloom syndrome). Relatively efficient screening is possible in Ashkenazi Jews as a result of that ethnic group having only a limited number of mutations. Overall, one in seven Ashkenazi Jews is heterozygous for one of the disorders listed above.
Additional information about disorders recommended for screening is provided elsewhere by the authors.1, 8 Refer also to the American College of Obstetricians and Gynecologists (AGOC) guidelines.7, 9, 10, 11, 12, 13, 14
PRINCIPLES AND PREREQUISITES OF GENETIC SCREENING
Genetic screening implies monitoring a population to identify clinically normal individuals who have genotypes that are associated with a detectable disease or that may lead to that disease in their offspring. Several aspects of genetic screening deserve emphasis, in particular the contrast between screening programs and case detection programs.
A key principle is that genetic screening should be voluntary unless specifically mandated by law.15 (Legal requirements usually dictate neonatal screening for phenylketonuria [PKU], hypothyroidism, and in some states other disorders.) Of course, voluntary screening does not mean that a physician must remain neutral or even fail to express his or her opinion. A given test should not be performed without a patient's knowledge, however, because a patient may not wish to be faced with the dilemma of deciding among options raised by the results of the screening process.
In genetic screening, one does not expect to detect all affected cases in a given population. This contrasts with case detection programs, an example of which is cervical cytologic screening. Yearly Papanicolaou smears are recommended to guard against laboratory errors and the inability of physicians to correctly sample all endocervices. If such pitfalls did not exist, a normal smear would categorically exclude squamous cervical cancer for about 5 years, the interval required for a normal cervix to progress through dysplasia to carcinoma. By contrast, in genetic screening, one accepts failure to detect all affected infants on fetuses.
Establishing technical feasibility for screening a given disorder alone does not justify screening. Indeed, many genetic disorders are amenable to screening. For example, all chromosomal abnormalities could be detected during the neonatal period, and almost all disorders of amino acid metabolism are amenable to screening. Conversely, screening actually is recommended only for those disorders that fulfill prerequisites essential for initiating screening programs. Four prerequisites must be fulfilled as is discussed in Table 2.
Table 2. Prerequisites of genetic screening
Capacity to alter clinical management of affected individuals
Ability to identify matings between two heterozygotes
Cost-effectiveness
Reliable methods of assessment of genetic status (reproducible assays)
Capacity to handle ancillary problems
Variants not requiring action
Potential stigmatization of heterozygotes
Capacity to alter clinical management
Although screening to achieve research objectives (e.g., determining the incidence of a disorder) sometimes is appropriate, widespread testing is ordinarily performed only if an abnormal finding could alter clinical management. Thus, neonates are screened for those metabolic disorders amenable to treatment (e.g., PKU and hypothyroidism) but not for those untreatable disorders (e.g., Lesch-Nyhan syndrome). Neonatal screening for sickle cell anemia was recommended only after it became clear that prophylactic antibiotics might prevent life-threatening infections. Relatively common disorders that are not treatable and therefore for which neonates should not be subjected to screening include chromosomal abnormalities, Tay-Sachs disease, and Duchenne muscular dystrophy.
If neonatal screening is undesirable for a given disorder, it still may be reasonable to screen adults to determine whether they are at increased risk of having offspring with the same disorder. Identification of individuals heterozygous for an autosomal-recessive disorder could alter reproductive choices and, thus, clinical management. The object is to identify matings between two individuals heterozygous for the same mutant allele. Screening adults to determine heterozygote status ordinarily is applicable only if prenatal diagnosis is possible. Ideally, screening should be performed before pregnancy; however, if the patient is already pregnant, screening should be completed as early in gestation as possible to allow maximum family planning options.
Cost-effectiveness
Ability to identify a heterozygote or even to detect an affected neonate with a treatable disorder does not necessarily dictate that screening be undertaken. For only a few disorders does the cost of screening justify the monetary and emotional savings of detecting the rare affected case. Indeed, technology now exists to screen for scores of mendelian disorders. Yet only hypothyroidism, PKU, and perhaps sickle cell anemia unequivocally fulfill the criteria of cost-effectiveness in neonatal screening. Screening for other treatable disorders is arguable, although some states mandate screening for galactosemia, maple syrup urine disease, adenosine deaminase deficiency, and adrenal 21-hydroxylase deficiency. Chromosomal analysis and α-fetoprotein assays are performed routinely whenever amniotic fluid is obtained at amniocentesis, but testing for other detectable disorders (e.g., rare metabolic traits) is not pursued. Indeed, in the future, we can anticipate that virtually all common mendelian disorders will be detectable in utero; however, neonatal screening, heterozygote detection, or prenatal testing will be applicable for only a few disorders in the general population.
Reliable method of assessment
The requisite assay must have a high predictive value. Although a general axiom of laboratory medicine, this statement is especially applicable for genetic diseases. Because genetic disorders are individually rare, even low false-negative rates could result in a greater likelihood that a given abnormal value represents a false-positive rather than a true-positive value.
Capacity to handle problems
Fulfilling all the above prerequisites still does not mean that screening for a particular disorder should be initiated. A final requirement is the ability to handle the difficulties that inevitably arise in screening programs. Sometimes the inability to handle these problems obviates introduction of a screening program that would otherwise be desirable. It should be anticipated that unexpected problems will arise. After screening programs for PKU were initiated, it became clear that elevated neonatal phenylalanine often was not the result of PKU but rather the result of other conditions that required no dietary treatment. Ability to separate false-positive from true PKU proved crucial.
When normal populations are screened to detect heterozygotes (sickle cell disease, Tay-Sachs disease, α-thalassemia, β-thalassemia), one must ensure that individuals identified as heterozygotes do not become stigmatized or do not develop erroneous impressions that their health is threatened.
PRINCIPLES OF GENETIC COUNSELING
Genetic counseling is the process in which an individual or family is given information about a real or possible genetic problem. In educating and counseling about genetics, the counselor must provide information in an understandable way about the nature of genetic risks and our ability to predict such risks. This depends on how a disease or condition is inherited, its severity, and other important factors, such as the interactions of environment and genetic background that must be present before a genetic susceptibility is expressed.
Genetic counseling is a dynamic communicative process that begins with trying to establish a diagnosis. This involves taking a medical and family history, performing clinical examinations, and obtaining relevant laboratory tests. The prediction of genetic risks based on laboratory tests depends on the sensitivity and specificity of the tests themselves and the quality of the laboratory performing the tests. The counselor must then provide information regarding risks of recurrence, genetic and medical implications of the disorder, prevention, family planning, and medically managing the condition. Because genetic counseling is directly concerned with human behavior, it must be based on an understanding of the psychological meanings of health and illness, procreation, and parenthood. Genetic information can bring bad news, and the counselor must be a resource for individuals and families dealing with sadness, loss, anger, guilt, or anxiety. The counselor has a responsibility to help individuals and families adjust psychologically and socially to their genetic condition.
PROVIDERS OF GENETIC COUNSELING
Genetic counseling cannot and should not be provided exclusively by a single specialist but is an interdisciplinary activity. There also is a growing recognition of the need to ensure that those providing genetic counseling are competent. Increasingly, genetic services are being provided by primary care physicians who are not necessarily trained in human genetics. In the clearest situations, the primary care physician may also be the most appropriate person to provide the counseling because he or she knows the family, their personal attitudes, and their socioeconomic background better than a consultant. However, in more complex situations, the primary care physician may lack the specific knowledge, time commitment, availability of necessary diagnostic tests, or skills required for genetic counseling.
Although there are no state or federal licensing bodies for genetic counselors, certification is provided by the American Board of Medical Genetics. To be eligible for certification, an individual must meet the criteria in the area of desired certification and pass a written examination. The areas in which certification is offered include clinical geneticists (e.g., MD, DDS, DMD, DO), clinical laboratory geneticists (e.g., MD, PhD, DDS), and medical geneticists (e.g., PhD). The National Board of Genetic Counselors certifies individuals who hold postbaccalaureate degrees, most often a Masters of Science.
All who hold themselves out to the public as engaging in genetic counseling services must possess sufficient knowledge, training, and skill to provide these services in a reasonable manner. Practitioners must respect the limits of their individual competence and avoid acting beyond the scope of their ability. The use of a genetics team approach has been endorsed as consistent with the notions of competence and appropriately shared responsibilities.
Confirming the diagnosis
Good genetic advice requires certainty of diagnosis; even the best counseling cannot compensate for an inaccurate diagnosis. In addition to taking a detailed family history, the proband (i.e., the family member through whom the family was identified) should be examined carefully, as should other family members at risk. If the proband is no longer living, the appropriate medical records should be sought and reviewed. The possibility of nonpaternity must also be considered.
Laboratory studies needed to establish the diagnosis often include chromosome analysis; DNA studies; or biochemical tests of blood, urine, or cultured cells. Nongenetic factors can mimic genetic factors in the production of disease (so-called phenocopies); a good history and various clinical and laboratory studies may help resolve questionable cases.
Sometimes, however, despite the most intensive efforts, a precise diagnosis cannot be established. For some families, the answer “we do not know” leads to understandable frustration and dissatisfaction with the counseling experience. Conversely, some families receive a measure of satisfaction and relief from the knowledge that all reasonable steps have been taken to answer their questions.
Estimating and interpreting risks
It is important for the counselor to establish precisely what information the individual, couple, or family wants to know about their genetic situation. People often have less interest in the label of a disorder and its mechanism of action than how tests may predict a disorder; what affect the disorder has on physical and mental functioning; and how intrusive, difficult, or effective an existing treatment or alternatives might be. Research has shown that individuals generally remember the levels of risks they were told at counseling, not always in numeric form, but whether it was high or low. However, how different people perceive their genetic risks varies widely from overly cautious to reckless. The counselor should strive to help the family understand the consequences of the genetic problem. Factual information must be conveyed concerning its significance and natural history. Such explanations should be developed in small, discreet steps, with frequent pauses so that the counselees can ask questions. This may require several counseling sessions.
Perception of risk is highly dependent on the individuals’ subjective experiences and expectations and is related to the manner in which they receive the information and their experiential, emotional, religious, and situational concerns. For example, a cleft lip may be perceived as a major tragedy by some parents. Conversely, some couples may readily accept a child with Down's syndrome.
The manner in which risk figures are presented has been an important influence in how they are interpreted. For example, telling a 35-year-old woman that her risk of having a child with Down's syndrome is 1 in 385 might be interpreted differently than saying that she has a fourfold higher risk compared with a 20-year-old woman. Recent data indicate that parents better understand magnitude of risk when comparisons are presented as rates (e.g., 2.6 versus 8.9 per 1000 women) compared with the traditional use of proportions to express risk (e.g., 1 in 384 versus 1 in 112).16 Regardless of the actual risk and how it is presented, counselees often perceive risks as being “all or nothing” (i.e., it will either happen or not happen). Although most counselors claim to use a nondirective approach (discussed below), few deny that an element of counselor bias is always present. Risk figures might usefully be presented in several alternative ways; however, beyond near-comprehension of numeric risks, genetic counseling must assist individuals in determining their own acceptable risk. Therefore, the counselor must always bear in mind that for the individual, couple, or family for which the feared event actually occurs, the risk is now 100%; the gamble was lost.
All of this, of course, implies that the more accurate information the couple has, the more likely they are to make a final decision that is consistent with their own values. As a policy matter, this conclusion underlies the doctrine of informed consent in genetic counseling and its purpose: the promotion of self-determination and rational decision making in situations that critically affect one's own life.
Communication
At the heart of genetic counseling lies the necessity to educate patients about the genetic facts and issues relevant to their circumstances. One would assume that the higher an individual's educational level, the better he or she should be able to understand complicated and unfamiliar biologic and medical concepts. For common genetic counseling situations (e.g., advanced maternal age), prepared literature packets can be provided before the counseling session so that the patient can become familiar with the information to be discussed during the counseling session. Such printed materials not only initiate the education process but also serve as a resource to which the patient may wish to refer at a later time. They also have the advantage of emphasizing that a patient's problem is not unique and standardizes the informational content so that the counseling process is consistent.
Nondirective approach to counseling
In 1942, a clinical psychologist, Carl Rogers, coined the term nondirectiveness to describe his psychotherapeutic approach of not advising, interpreting, or guiding his clients. In 1969, the World Health Organization Expert Committee on Genetic Counseling endorsed the nondirective approach to genetic counseling, an attitudinal strategy that has become universally accepted by virtually all professionals providing genetic counseling services. The counselor makes clear from the onset that the process is educational and no decisions will be made for the counselees. The counselor tries to remain impartial and objective in providing information that will allow the counselees to make their own rational decisions commensurate with their private concerns and desires. A variation on this theme is that some counselors will not comment on the decisions families suggest, whereas others will support any decisions families make. This is in contrast to the usual directive approach physicians take in dealing with patients having medical problems. For example, a physician who detects a breast mass in a patient usually gives a strong recommendation as to what steps should or should not be taken next.
Completely nondirective counseling probably is unrealistic. The natural tendency for counselors to interject their own biases by either verbal or nonverbal messages is always present. Indeed, to provide counseling implies that it is necessary. Despite conscious efforts to provide nondirective counseling, counselors may unknowingly give directive signals. For example, a simple gesture (i.e., nonverbal body language), such as shifting in one's chair or raising eyebrows, may be interpreted as approval or disapproval of a decision. Repetition of certain points or presenting them in a louder voice may influence the way an individual weighs the information. Silence at certain crucial moments in a discussion may give the loudest message of all. These biases may reflect the attitudes of the genetic counselor about the nature and meaning of health and disease, the seriousness of the genetic condition in question, perception of quality of life, and the appropriateness of decisions related to genetic counseling and testing, acceptance or rejection of advice, and other important issues.
Perhaps the most surreptitious directive signals occur in the managed care environment. For example, the availability of a test may be viewed as an endorsement whereas its unavailability as a condemnation. The reality is that inclusion or exclusion of certain genetic services by a managed care organization may be more based on cost-efficiency determinations than on risk–benefit concerns and patient autonomy. Similar issues arise when one weighs decisions that follow genetic counseling with respect to reducing the societal burdens of genetic disease when they compete with the particular harms to an individual. This turns into societal stigmatization. For example, as prenatal diagnosis for advanced maternal age becomes more popular, there may now be less tolerance for an older woman having a child with Down's syndrome. This reduced tolerance may not only be directed toward the mother but also toward the affected child.
PSYCHOLOGICAL CONSIDERATIONS IN COUNSELING COUPLES WITH A PREVIOUS ABNORMAL PREGNANCY OUTCOME
Counseling couples who already have been delivered of an abnormal child requires additional appreciation for the psychological adaptations that such couples have undergone. Great sensitivity and considerable investment of time are necessary. Such couples are thus best managed by a geneticist who can efficiently set aside the necessary time. The role of most obstetricians/gynecologists is to recognize the problem and reinforce salient genetic data. Again, even if the diagnosis seems obvious, confirmation is obligatory. The clinician should not merely accept a patient's word nor even accept a diagnosis made by a physician who is not highly knowledgeable about the condition. If possible, the anomalous individual should be examined. If the affected relative is still alive, cells may need to be frozen in the event that diagnosis based on DNA studies is or will be feasible. Examining first-degree relatives also may be helpful if an autosomal-dominant disorder such as neurofibromatosis needs to be excluded. Alternatively, one may need to rely on medical records or laboratory tests.
Psychological defenses are always operative in the couple receiving genetic counseling. Such defenses can impede the entire counseling process. Anxiety is low in couples requesting genetic counseling because of parental age or because of an abnormality in a distant relative. As long as anxiety remains low, emotions are easily controlled and comprehension of information is not impaired. However, couples who have experienced a stillborn infant, an anomalous offspring, or multiple repetitive abortions are understandably more anxious. Their ability to retain information, thus, may be impaired.
Stages of psychological response
The psychological impact of a genetic diagnosis varies with its severity, treatability, and the unique responses of different individuals and families. Indeed, psychological defenses underlie all genetic counseling. If not appreciated, these defenses can impede communication and interfere with the entire counseling process. For example, anxiety generally is low when the risks are perceived as remote for an adverse event actually happening (counseling couples for advanced maternal age) or for an abnormality in a distant relative. As long as the anxiety level remains low, comprehension of information usually is not impaired. However, when the genetic condition directly affects the individual or a close family member, anxiety is understandably high and the ability to retain information is often hindered.
The impact of diagnosing a genetic problem may be potentially devastating to an individual or family (e.g., the delivery of an infant with multiple birth defects and predicted severe mental retardation or the discovery of a mutated gene that indicates a very high likelihood of cancer developing in an individual and other family members who inherit that gene). The physician or counselor needs to recognize that individuals receiving such bad news can be expected to manifest clearly identifiable sequential coping responses in dealing with such situations. Several distinct stages can be recognized in this process.
The first stage is shock and disbelief. Denial is a psychological mechanism in which a person attempts to maintain the integrity of his or her personality simply by denying the stressful situation. This may take the form of the individual insisting that a mistake has been made. Sometimes he or she appears not to comprehend the situation at all. The counselor must be alert to this coping response, because frequently such individuals appear to be composed on the surface and “very mature in handling the situation”. If prolonged, such a response can be self-defeating and interfere with an understanding and acceptance of a genetic problem. It is also important for the counselor to recognize that during this period, the counselee usually is not able to properly assimilate important information that is being given and that must be acted on.
In the second stage, anger, the individual begins to comprehend the reality of the situation and attempts to deal with it at an intellectual level. Anxiety may be manifest by nervousness, irritability, hyperactivity, fatigue, insomnia, loss of appetite, or other physical complaints such as headaches and indigestion. Sometimes, this may precipitate a panic attack. Although the stressful event has been recognized at the intellectual level, the individual has not achieved a psychological equilibrium at the emotional level. The individual may become frustrated and angry. Fault may be assigned to others (e.g., spouse, affected child, physician, genetic counselor) without a rational basis. Alternatively, the anger may be self-directed and manifest by guilt.
The third stage of the coping reaction is depression. The symptoms of depression are feelings of worthlessness, uselessness, despair, and helplessness. Physical symptoms include loss of appetite, weight loss, fatigue, constipation, headaches, crying spells, and poor sleep. Often, individuals have early morning wakening, a time when depressive symptoms usually are heightened. The counselor must be particularly aware of severe depression that may be associated with a high risk of suicide. In such cases, psychiatric care is mandatory.
The fourth stage, bargaining or rationalization, occurs when an individual begins to abandon old modes of behavior and thought and reaches a new psychological homeostasis. The person actively tries adaptive changes. This is a critical time in the genetic counseling process because the individual is most receptive to new ideas. During this stage, the role of the counselor should chiefly be supportive and to encourage the counselee to evaluate self-generated alternative courses of action with regard to decision making and outcome.
CONFIDENTIALITY
Genetic information may not only have an impact on healthcare decisions for individuals and families but also result on a cornucopia of social and legal problems, including stigmatization, discrimination in the workplace, and loss or inability to obtain employment and health insurance. For these reasons, those entrusted with obtaining genetic information have a great responsibility to maintain it in strict confidence.
One such problem is whether a physician or counselor has an obligation to inform potentially affected relatives of a genetic finding. An analogy can be made to the legal obligation of informing sexual contacts when a venereal disease such as syphilis is diagnosed. Does the same hold true for genetic conditions? Suppose a mother learns through having an affected son that she is a carrier for Duchenne-Becker muscular dystrophy. Should the physician or counselor inform the mother's sisters, who also are at an increased risk of having affected sons? The mother should naturally be urged to notify her sisters herself, but if she refuses, what should be done? Unfortunately, there is not entirely a satisfactory answer to this dilemma. Perhaps the most analogous situation is a case adjudicated in California involving a psychiatrist who failed to warn a woman that his patient, a disturbed graduate student from India who wanted to marry her, had threatened to kill her. When the woman subsequently was indeed murdered, her parents sued the psychiatrist for failure to warn her (or them) of the danger. The court gave the following opinion: “Where a doctor … in the exercise of his professional skill and knowledge, determines, or should determine, that a warning is essential to avert danger arising from the medical or psychological condition of his patient, he incurs a legal obligation to give that warning.” The basis for this decision was the court's view that we live in an “interdependent” and “risk-infested ” society and that society cannot tolerate additional risks that physicians could eliminate by a simple act of communication. It is questionable whether this decision could be used to find a duty on the part of a physician or genetic counselor to warn other family members that their offspring might be in danger because of a genetic condition a family member might be carrying. However, in view of the public policy enunciated by the courts, a strong argument can be made that such a disclosure would be legally permissible even though not required.
It has been our opinion that a policy of strict nondisclosure without patient consent should be adhered to in the genetic counseling setting. Such a policy fosters patient self-determination and confidence in the integrity of the counseling process. Conversely, we recognize the serious implications of many genetic disorders and understand the reasoning in which one might want to breach confidentiality in certain instances. In any event, given the uncertainty in the law and the fact that this situation can be anticipated easily, we believe that individuals providing genetic counseling should have a clear written policy defining the circumstances, if any, under which they will disclose information or test results learned in the counseling program. Such a program should be spelled out before the counseling and genetic diagnosis process begins, so that the individual, couple, or family can opt out immediately if the policy is not acceptable. Finally, many individuals who attend genetic counseling perceive themselves as having a duty to disseminate information they obtain to other relatives. Given that individuals often experience difficulty recalling information they receive during consultations, there is a potential for the miscommunication of genetic information within families. Thus, a written summary of the salient genetic issues often is beneficial.17
REFERENCES
Simpson JL, Elias S: Genetics in Obstetrics and Gynecology. 3rd ed. Philadelphia, WB Saunders, 2003 |
|
American College of Obstetricians and Gynecologists (ACOG): Technical Bulletin No. 108, Antenatal Diagnosis of Genetic Disorders. Washington, DC, ACOG, 1987 |
|
Simpson JL, Elias S, Gatlin M et al: Genetic counseling and genetic services in obstetrics and gynecology: Implications for educational goals and clinical practice. Am J Obstet Gynecol 140: 70, 1981 |
|
American College of Obstetricians and Gynecologists (ACOG): Committee Opinion No. 192, Genetic Screening of Gamete Donors. Washington, DC, ACOG, 1999 |
|
American College of Obstetricians and Gynecologists (ACOG): Guidelines for Perinatal Care/American Academy of Pediatrics. 6th ed. Washington, DC, ACOG, 2007 |
|
American Society for Reproductive Medicine (ASRM): Minimal genetic screening for gamete donors. Fertil Steril 70: 12S, 1998 |
|
ACOG Practice Bulletin No. 78: hemoglobinopathies in pregnancy. Obstet Gynecol 109: 229–37, 2007 |
|
Simpson JL, Holzgreve W: Genetic Counseling and Genetic Screening. In Gabbe SA, Niebyl JF, Simpson JL. (Eds): Obstetrics: Normal and Problem Pregnancies. 5th ed. Pennsylvania, Churchill-Livingstone, pp. 138–151, 2007. |
|
ACOG committee opinion. Number 318, October 2005. Screening for Tay-Sachs disease. Obstet Gynecol 106: 893–4, 2005 |
|
ACOG committee opinion No. 298, August 2004. Prenatal and preconceptional carrier screening for genetic diseases in individuals of Eastern European Jewish descent. Obstet Gynecol 104: 425–8, 2004 |
|
ACOG Committee Opinion No. 393, December 2007. Newborn screening. Obstet Gynecol 110: 1497–500, 2007 |
|
ACOG Committee Opinion No. 325, December 2005. Update on carrier screening for cystic fibrosis. Obstet Gynecol 106: 1465–8, 2005 |
|
ACOG committee opinion No. 212. Screening for canavan disease. November 1998. Committee on Genetics. American College of Obstetricians and Gynecologists. Int J Gynaecol Obstet 65: 91–2, 1999 |
|
ACOG Committee Opinion: Genetics In Obstetrics and Gynecology. Washington DC, American College of Obstetricians and Gynecologists, 2002. |
|
National Academy of Sciences: Genetic Screening: Programs, Principles and Research. Washington, DC, National Academy of Sciences, 1975 |
|
Grimes DA, Snively GR: Patient's understanding of medical risks: Implications for genetic counseling. Obstet Gynecol 93: 910, 1999 |
|
Hallowell N, Murton F: The value of written summaries of genetic consultations. Patient Educ Couns 35: 27, 1998 |