Hirsutism
Authors
INTRODUCTION
Hirsutism is the presence of terminal (coarse) hairs in females in a male-like pattern; it affects between 5% and 15% of women surveyed.1,2,3,4 This chapter outlines the normal metabolism of androgens; the physiology and pathophysiology of hair growth; and the differential diagnosis, workup, and treatment of the hirsute patient.
NORMAL ANDROGEN METABOLISM
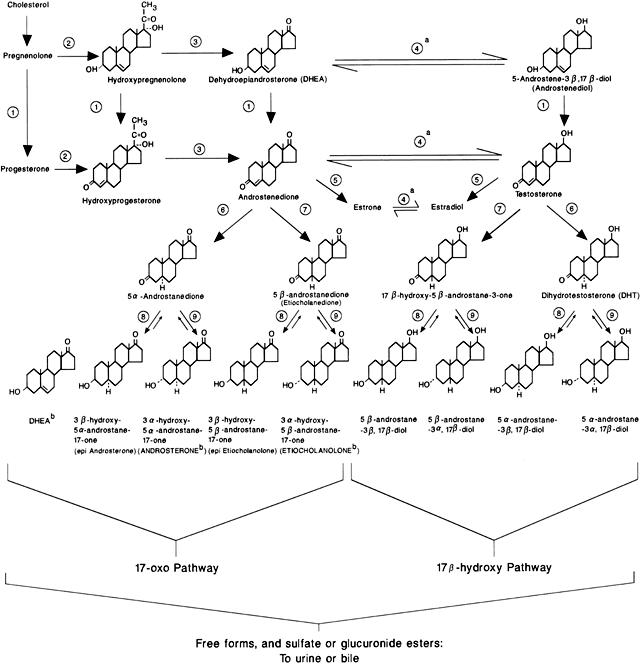
Androgens are C19 steroids that are derived from cholesterol and secreted by the adrenal cortex and ovaries. Androgens may also be derived (not secreted) from the conversion of other steroids by the liver and some peripheral tissues (e.g. adipose tissue, muscle, skin). Principal circulating androgens include testosterone (T) and its 5α-reduced metabolite dihydrotestosterone (DHT), androstenedione (A4), and dehydroepiandrosterone (DHA) and its metabolite dehydroepiandrosterone sulfate (DHEAS) (Fig. 1). Androgens act through a specific cytoplasmic/nuclear intracellular receptor.
In adult women, T originates approximately 25% from the ovary, 25% from the adrenal, and 50% from the peripheral conversion of A4.5 T and A4 are also metabolized to DHT, a potent androgen, through the action of 5α-reductase in the liver and skin. A4 is produced equally from the ovary and the adrenal cortex, whereas approximately 95% of DHA and 99% of DHEAS are secreted by the zona reticularis of the adrenal cortex. DHA, DHEAS, and A4 exhibit a circadian rhythm similar to that of cortisol, with peak serum concentrations in early morning and the nadir in late evening.6,7,8 DHA and A4 are much weaker androgens than are T and DHT, and under normal circumstances they have a very limited androgenic effect. Although DHEAS is the most abundant androgen in circulation, in a concentration approximately 8000 to 10,000 times that of T, it has almost no androgenic activity.
T and DHT circulate tightly bound to the hepatic α-globulin sex hormone-binding globulin (SHBG) and, in a lesser extent, to albumin. A4 is only weakly bound, and DHA and DHEAS circulate mostly unbound.9,10 Because only free (unbound) steroid is able to act on the androgen receptor, the action of T and DHT is greatly influenced by the circulating SHBG level. Assuming that the level of total hormone remains unchanged, a decrease in the circulating concentration of SHBG results in higher free fractions of T and DHT and an increase in their effect.11 In addition, the SHBG level influences the clearance of T and DHT from circulation,12 because only free androgen can be metabolized by the liver and peripheral tissues. Paradoxically, the circulating SHBG level is decreased by androgens and increased by estrogens.13,14,15,16 If the circulating level of androgens increases, the SHBG concentration drops resulting in a higher T and DHT clearance. The final result may be that, although the total T level remains relatively normal, T production and its free (active or unbound) fraction are increased.11 Increased insulin levels, as seen in patients with the polycystic ovary syndrome (PCOS) and the hyperandrogenic-insulin resistant-acanthosis nigricans (HAIRAN) syndrome, are a more important factor in suppressing SHBG levels than are androgens (see later discussion).
Androgen production and clearance are affected by various physiologic states. In obesity, androgen production and clearance are accelerated.17 The increased clearance is caused, in part, by an obesity-related decrease in SHBG, which leads to higher levels of rapidly metabolizable free T and DHT. In addition, because steroids are fat soluble, the increased amount of adipose tissue in obese women tends to “trap” androgens, increasing their extravascular pool. In obesity a greater percentage of the androgens are metabolized to estrogens through the action of aromatase, which is present in the stroma of adipose tissue. Because androgen levels in obese women are generally normal or only slightly decreased, their production rate must be increased to compensate for their higher clearance.
NORMAL HAIR PHYSIOLOGY
Hair follicles cover the entire body with the exception of the soles of the feet, the palms of the hands, and the lips. Most hair follicles are present at birth, with very few new ones formed thereafter.18 Hair follicles are lost in a generalized fashion beginning in the middle to late forties, leading to the thinning of hair that is noted thereafter. Hair follicles are usually present in conjunction with a sebaceous gland, forming the pilosebaceous unit. Hair is composed of keratin proteins, which form the hair shaft. The hair shaft grows within the outer hair root sheath, which forms part of the epidermis. Structurally, there are three types of hair. Lanugo is the soft, unmedullated hair covering the surface of the fetus, which is shed sometime in late gestation or in the early postpartum period. Vellus hairs are short (2 to 5 mm), soft, fine, unmedullated, and usually nonpigmented, and cover the apparently “hairless” areas of the body. Terminal hairs are long, coarse, and medullated; they make up the eyebrows, the eyelashes, and the scalp, pubic, and axillary hair.
Although there are racial and ethnic differences in follicle numbers, there is little difference in follicle number between sexes within each race or ethnic group.19,20 The visible differences between men and women do not relate to the number of hair follicles, but rather to the type of hair arising from these follicles (i.e. vellus versus terminal hairs). Whites have a higher density of hair follicles than Blacks, who in turn have more follicles than persons of Asian descent. Although in women many of the hair follicles produce vellus hairs, in men these same follicles produce terminal hairs, leading to the appearance of visible facial, chest, and abdominal hair.
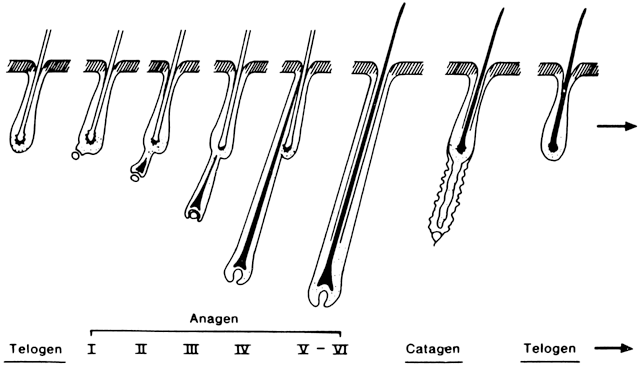
The hair growth cycle can be divided into three phases.18 The hair grows actively during the anagen phase, which is associated with downward progression of the dermal papilla (Fig. 2). During the catagen phase, the hair follicle involutes. This is followed by the telogen phase, in which the hair is eventually shed. Although in many animal species the growth cycles of all hair follicles are in synchrony, in humans this is not the case, which accounts for the impression that hair continuously grows. Occasionally a greater number of hair follicles shift into synchrony, as in pregnancy, in menopause, or on initiation of various hormonal therapies such as oral contraceptive medications. A temporary increase in the number of hairs in anagen is noted, but eventually these hairs reach telogen and begin to shed in excessive numbers. Normally approximately 15% of scalp hairs are in telogen; and if more than 25% of hairs are in telogen, the condition is known as telogen effluvium. Telogen effluvium is most frequently observed in the postpartum period or after discontinuation of oral contraceptives. There is not an actual reduction in the number of hair follicles, and patients should be assured that this condition is only temporary, although full normalization of hair growth may take up to 3 years (i.e. approximately the duration of the life cycle of scalp hairs).
There are a number of hormones that control hair growth. Thyroid and growth hormones produce a generalized increase in hair growth. Hyperthyroidism yields a fine, friable hair that is easily lost, whereas hypothyroidism produces a coarse, brittle hair that also is easily lost. Of the reproductive hormones, progesterone has a minimal effect. Estrogens basically oppose the effect of androgens, most importantly by increasing circulating SHBG, leading to a decrease in free T and DHT. Direct effects of estrogen on the hair follicle have been suggested, but the exact extent of this effect and its mechanism remain unclear.
Androgens are the most important determinant of the type and distribution of hairs throughout the body. Circulating androgens, particularly T and A4, are converted in the hair follicle to DHT through the action of 5α-reductase. DHT (and, to a limited extent, T, A4, and DHA) then acts on the dermal papilla of androgen-sensitive hair follicles and sebaceous glands to increase the growth rate and thickness of terminal hairs and sebum production, respectively. Androgens also transform vellusproducing follicles to terminal hair-producing follicle, an irreversible process.
The effect of androgens on the hair follicle depends on whether the hair follicle is sensitive to these hormones.18,20,21 Some skin areas (e.g. eyelashes, eyebrows, lateral and occipital aspects of the scalp) appear to be relatively resistant and independent of the effects of androgens and are designated as nonsexual skin areas. Other areas, including the lower pubic triangle and the axilla, are very sensitive to the effects of even low levels of androgens because of their high 5α-reductase activity and are designated as ambosexual skin areas. The hair follicles in these areas develop terminal hairs quite early in puberty, coinciding with the minimal increases in adrenal androgens. Other parts of the skin contain lower levels of 5α-reductase and respond only to higher concentrations of androgens. These include the chin, cheek, sideburn, and neck areas; the upper and lower back; the chest; the upper and lower abdomen (i.e. male scutcheon); the upper thighs and buttocks; and the upper arms. These areas are designated as sexual skin areas. Terminal hair growth in these areas is characteristically masculine and, if present in women, is considered hirsutism.
HIRSUTISM
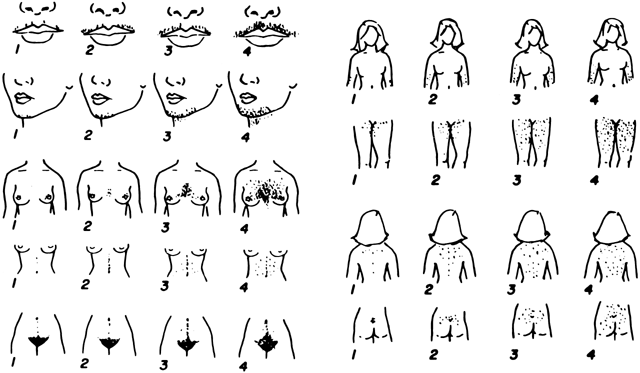
Hirsutism is the presence of terminal (coarse) hairs in females in a male-like pattern (Fig. 3). Excessive growth of coarse hairs of the lower forearms and lower legs alone does not constitute hirsutism, although women with hirsutism may note an increase in the pigmentation and growth rate of hairs on these body areas. Although hirsutism usually is obvious, it is important to standardize the examination for future reference by use of a scoring system. Such a system is depicted in Figure 3, and a few other schemes have also been proposed. It is also important to remember that not all hyperandrogenic women demonstrate hirsutism, such as Asian women.22
OTHER SIGNS OF ANDROGEN EXCESS
In addition to hirsutism, hyperandrogenism in women may have other manifestations, including acne, androgenic alopecia, ovulatory dysfunction, and, if very severe, virilization and/or masculinization.
Acne
The pilosebaceous unit, in addition to the hair follicle, also contains a sebaceous gland which produces an oily protective secretion, sebum. The excessive production of sebum in response to androgen action leads to oily skin, clogged hair follicles, folliculitis, and subsequent acne. Increased serum androgen levels have been noted in patients with acne,23,24,25,26,27 particularly in those with concurrent hirsutism, although not all investigators agree.28,29,30,31 Although all androgens may lead to acne, the sole presence of acne without hirsutism is often associated with excessive levels of the adrenal androgens, DHA and/or DHEAS.32,33,34,35 These steroids are relatively weak in their stimulation of terminal hair growth, but they are relatively effective in increasing sebum production. Nevertheless, there are conflicting reports as to the incidence of adrenocortical abnormalities in women with acne.36,37
Some investigators have observed that, although serum androgen levels may be relatively normal, the metabolism of such androgens to DHT via 5α-reductase is increased in the affected skin.38,39,40 It does appear clear, however, that acne frequently improves after treatment with oral contraceptives, glucocorticoid suppression, spironolactone (SPA), or cyproterone acetate (CPA).41,42,43,44,45,46,47 Although many teenagers suffer from acne, skin problems that persist past 20 years of age should raise the suspicion of androgen excess. Empiric treatment with SPA, low-dose glucocorticoid, or oral contraceptives may be justified in acneic patients without other evidence of overt androgen excess.
Androgenic Alopecia
Scalp hair loss as a consequence of androgen excess can take two forms. In severe cases, where massive androgen excess and virilization/masculinization is present, patients can demonstrate the typical pattern of balding found in men (i.e. premature male-pattern balding). More common, however, is the so-called androgenic (or androgenetic if an inherited cause is suspected) alopecia (AA) or female-pattern balding. In AA a diffuse thinning of hair throughout the sagittal scalp is noted.48 Approximately 40% of women with AA have some form of hyperandrogenemia.49 In one study of 109 women with AA, 38.5% had evidence of hyperandrogenemia.50 However, in this population 25 women were also hirsute (with and without oligo-ovulation). If only the 84 nonhirsute women with AA were considered, then approximately 20% of these patients were found to be hyperandrogenemic and only 9% had PCOS. Therefore, only one fifth of women with AA and without hirsutism actually are hyperandrogenic.
Oligo-ovulation or Anovulation
Androgens can be converted to estrogens in peripheral and hepatic tissue, and the excessive levels of both androgens and estrogens may alter the hypothalamic-pituitary production of gonadotropins. Furthermore, excessive androgen levels directly inhibit follicular development,51,52 which may result in the accumulation of multiple small cysts within the ovarian cortex (the so-called “polycystic” ovary).
Virilization and Masculinization
If androgen levels become extremely elevated for a substantial period, the patient's body habitus may become masculinized, with a reduction in breast size, an increase in muscle mass and upper body fat deposition, and features of virilization. In turn, virilization includes the appearance of sagittal and frontal balding, clitoromegaly, and severe hirsutism. Masculinized and/or virilized patients are almost always amenorrheic. In general, virilization or masculinization should raise the suspicion of an androgen-producing tumor or classic (but not nonclassic) adrenal hyperplasia (see later discussion). Occasionally girls with severe hyperthecosis or the HAIRAN syndrome (see later discussion) exhibit a moderate degree of virilization.
DIFFERENTIAL DIAGNOSIS OF HIRSUTISM
The differential diagnoses of hirsutism can be divided into those causes that are independent of excessive androgen action (i.e. nonandrogenic causes), those that are related to excessive production or ingestion of androgens (androgen causes), and idiopathic hirsutism (IH), which is presumed to occur in response to the excessive peripheral utilization of androgens.
Nonandrogenic Causes
ACROMEGALY.
Between 0% and 50% of acromegalic women have been reported to present with hirsutism.53 Accompanying the hirsutism is an enlargement of the hands and feet and a coarsening of the facial features. This cause of hirsutism is extremely rare, and the diagnosis is based on a determination of excessive growth hormone secretion. The mechanisms underlying the association between acromegaly and hirsutism, if any exist, remain to be confirmed.
CHRONIC SKIN IRRITATION.
Teleologically, hairs protect the skin, and chronic dermatologic irritation or injury, particularly of the face, can stimulate hair growth.18 Excessive waxing or plucking,54 abuse of depilating agents, and chronic use of retinoic acid for the treatment of age-associated wrinkles55 have all been reported to increase the length and diameter of facial hairs and possibly convert to vellus to terminal hairs.
DRUGS.
Certain medications, such as phenytoin, cyclosporine, minoxidil, calcium channel blockers, diazoxide, and erythropoietin, can cause a generalized growth of body and facial hair.
Androgenic Causes
ANDROGENIC DRUGS.
On occasion the use or abuse of androgenic drugs such as danazol, methyltestosterone, stanazolol, and oxandrolone lead to the development of acne and hirsutism. This problem is most frequently seen in women being treated with long-term danazol for hereditary angioneurotic angioedema, young women engaged in body building, and menopausal women who are receiving androgens as part of the hormonal replacement.
MALE PSEUDOHERMAPHRODITISM.
The peripubertal onset of hirsutism, breast development, and clitoromegaly, accompanied by primary amenorrhea in association with an absent uterus, suggests the presence of either incomplete androgen insensitivity (i.e. partial testicular feminization) or a Y-bearing dysgenetic gonad.57,58 If undiagnosed, progressive masculinization may result. On examination, these persons demonstrate a blind vaginal pouch and varying degrees of clitoromegaly and labial fusion. These patients may also demonstrate bilateral or unilateral vulvar swelling secondary to the descent of one or both gonads. In some patients with XY gonadal dysgenesis, a uterine remnant is present, invariably accompanied by some degree of genital asymmetry. Overall, this cause of hirsutism is extremely rare.
NONCLASSIC ADRENAL HYPERPLASIA.
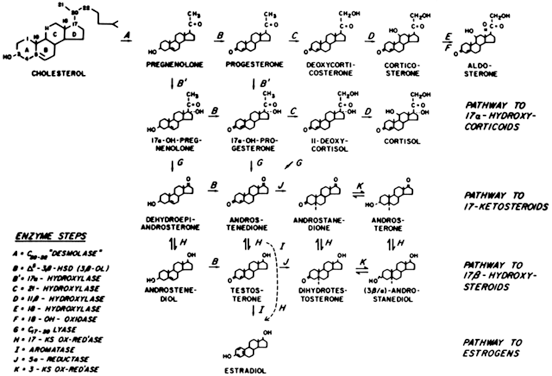
Adrenal enzyme deficiencies that lead to the appearance of hyperandrogenic symptoms at some time after birth have been called late-onset, postpubertal, attenuated, mild, or acquired, although the term nonclassic adrenal hyperplasia (NCAH) is preferred. By definition, NCAH is an autosomal recessive disorder that causes symptomatic hyperandrogenemia (e.g. hirsutism, acne, oligomenorrhea) peripubertally or postpubertally, in the presence of normal female external genitalia. If a girl presents with evidence of an adrenal enzyme deficiency and demonstrates virilization of the genitalia, the disorder should be classified as congenital or classic adrenal hyperplasia (CAH), because the adrenal hyperandrogenemia was present in utero during genital development.
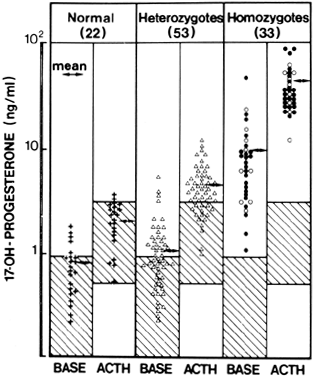
A defect in 21-hydroxylase (21-OH) accounts for more than 95% of all cases of NCAH (and CAH), although defects of 3β-hydroxysteroid dehydrogenase (3β-HSD) and 11β-hydroxylase (11-OH) have also been reported to cause the disorder59 (Fig. 4). These patients have a genetic defect of one of these enzymes that decreases its catalyzing efficiency. Consequently, the production of appropriate amounts of the enzymatic product (e.g. 11-deoxycortisol in the case of 21-OH deficiency) requires greater concentrations of precursor owing to the changes in the enzymatic kinetic constant.60 The excess amounts of precursor (e.g. 17 α-hydroxyprogesterone [17-HP] in the case of 21-OH deficiency) are then also converted to greater amounts of androgens. Overall, and in contrast to CAH, patients with NCAH do not generally demonstrate a decrease in cortisol production nor an elevation in circulating adrenocorticotropic hormone (ACTH) levels.61
21-Hydroxylase-Deficient NCAH.
A deficiency in 21-OH activity constitutes the most common cause of NCAH, affecting between 1% and 10% of hyperandrogenic women, depending on ethnicity.59 Overall, only 1% to 2% of hirsute individuals of Anglo-Saxon descent are affected, and 5% to 6% of hirsute patients of Latin descent (e.g. Spanish, Italian, French) have the disorder. The prevalence of NCAH appears to be particularly high among Ashkenazi Jews and patients from the Middle East.
In contrast to CAH, NCAH patients do not demonstrate virilization of the external genitalia, although occasional patients may have mild clitoromegaly. Hyperandrogenic symptoms most commonly appear peripubertally or postpubertally, although some children may present with premature adrenarche. Women with NCAH generally demonstrate relatively mild symptoms of hyperandrogenism, if any at all.62 Biochemically it is difficult to distinguish women with NCAH from other hyperandrogenic patients, and circulating T and DHEAS levels are not different from those observed in patients with PCOS.62,63,64 In fact, the level of DHEAS, an exclusive adrenal androgen, is often normal.
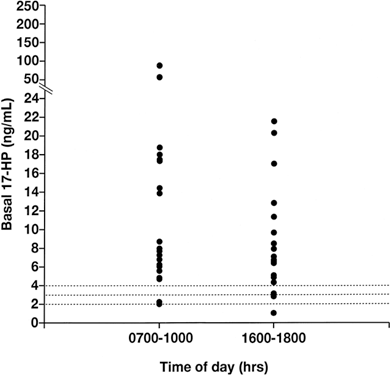
The measurement of a morning 17-HP level in the follicular phase of the menstrual cycle can be used to screen for 21-OH deficiency in NCAH.63,65 The majority of women with untreated NCAH demonstrate basal 17-HP levels higher than 2 ng/ml (Fig. 5). If used to screen for 21-OH deficient NCAH, the basal 17-HP should be obtained in the follicular phase of the menstrual cycle, because this progestogen increases in the luteal phase.65 Furthermore, dexamethasone should not be administrated before sampling, because it will suppress the circulating 17-HP level.
Hyperandrogenic women who demonstrate a basal follicular phase 17-HP level higher than 2 ng/ml merit an ACTH stimulation test to rule out NCAH (see later discussion). If the 17-HP level 30 or 60 minutes after intravenous administration of one vial of ACTH-(1-24) (Cortrosyn) is greater than 10 ng/ml, the diagnosis of 21-OH-deficient NCAH is established, although most patients have ACTH-stimulated 17-HP values in excess of 15 ng/ml (Fig. 6).62,63,64,65,66,67
The genetics of 21-OH-deficient NCAH have been extensively characterized. The 21-OH gene (i.e. CYP21) is located on the short arm of chromosome 6, in the midst of the human leukocyte antigen region. Molecular genetic studies have been able to establish the majority of the mutations resulting in 21-OH-deficient NCAH.68 In close proximity to CYP21 lies a pseudogene (CYP21P) that is not active but is very similar in DNA sequence. Its proximity probably accounts for the high frequency of inherited abnormalities of the 21-OH gene, due to gene conversion or deletion. Patients with NCAH may carry mutations in each of the homologous CYP21 alleles, which encodes for a mild defect (but usually they are different). Alternatively, approximately one half to two thirds of patients with NCAH are “compound heterozygotes,” carrying a mild mutation of one CYP21 and a more severe mutation (which would result in CAH if present in homozygous form) on the homologous allele.
11 β -Hydroxylase-Deficient NCAH.
Patients with 11-OH deficient NCAH, like those with the 21-OH variant, have been reported to be clinically indistinguishable from other hyperandrogenic women.69 The blood pressure in patients with 11-OH-deficient NCAH ranges from normal to moderately elevated. The basal hormonal profile is also not distinctive. However, the prevalence of 11-OH-deficient NCAH and its diagnostic criteria remain unclear. Using a level of 11-deoxycortisol greater than three times the upper limit of controls as a criterion, we69 and others70 have suggested that the prevalence of presumed 11-OH-deficient NCAH among hirsute patients is 1% or less. Nonetheless, the diagnosis of 11-OH-deficient NCAH has yet to be confirmed by molecular analysis of the responsible gene, CYP11a.71
3 β -Hydroxysteroid Dehydrogenase-Deficient NCAH.
The diagnosis of 3β-HSD-deficient NCAH has been assumed to be based on abnormally high levels of 17-hydroxypregnenolone (17-HPREG) or DHA, or both, after ACTH stimulation.72 However, molecular defects of the 3β-HSD-II gene (the enzyme form predominant in the adrenal cortex) have yet to be demonstrated in adult women with suspected 3β-HSD-deficient NCAH.73,74,75,76,77,78 The few cases of molecularly confirmed 3β-HSD deficiency that have been diagnosed in children73,74,75 suggest that the level of 17-HPREG diagnostic of NCAH is more than 50 times the upper normal value.75
Furthermore, even with the much less strict diagnostic criterion of an ACTH-stimulated 17-HPREG level three times greater than control values (similar to the difference in 17-HP levels found in 21-OH-deficient NCAH), we were unable to diagnose any affected patients among 86 consecutive hyperandrogenic women79 or among 30 women with increased DHEAS levels.80 Therefore, it appears that 3β-HSD deficient NCAH, if it exists, is, like its 11-OH-deficient counterpart, extremely rare.
THE HYPERANDROGENIC-INSULIN RESISTANT-ACANTHOSIS NIGRICANS (HAIRAN) SYNDROME.
Between 1% to 5% of hyperandrogenic women have this disorder. These patients demonstrate extreme insulin resistance with secondary hyperinsulinemia and hyperandrogenism. Although exact diagnostic guidelines have yet to elucidated, it appears that the disorder can be diagnosed by the presence of extremely high circulating levels of insulin, usually more than 80 μU/ml in the fasting state and/or more than 300 μU/ml after a 2- or 3-hour oral glucose tolerance test.81,82,83,84 In the early stages of the disorder, particularly in children or adolescents, glucose levels are relatively normal. Nonetheless, over time many of these patients experience progressive islet cell failure with the development of type 2 diabetes.
The high levels of insulin are generally caused by a genetic defect in post-insulin receptor action.84 Rarely, some of these women demonstrate abnormal insulin receptors85,86,87 or circulating anti-insulin receptor antibodies.88,89 The elevated circulating insulin concentrations act synergistically with luteinizing hormone (LH) to augment ovarian androgen (but not adrenal) production. This syndrome should not be confused with PCOS (discussed later), a disorder that is also associated with insulin resistance, although of a much lesser degree than that of the HAIRAN syndrome.
Patients with HAIRAN syndrome often demonstrate ovarian hyperthecosis, a pathologic finding characterized by islands of hyperplastic luteinized theca cells located throughout the stroma and the presence of relatively few and small atretic follicles.90 In fact, the finding of hyperthecosis usually suggests the presence of the HAIRAN syndrome,91 although a few patients with PCOS may also demonstrate this pathologic finding. In HAIRAN patients with hyperthecosis, circulating levels of LH and follicle-stimulating hormone (FSH) may be normal to low (4 to 8 mIU/ml) due to the extremely high circulating levels of T.
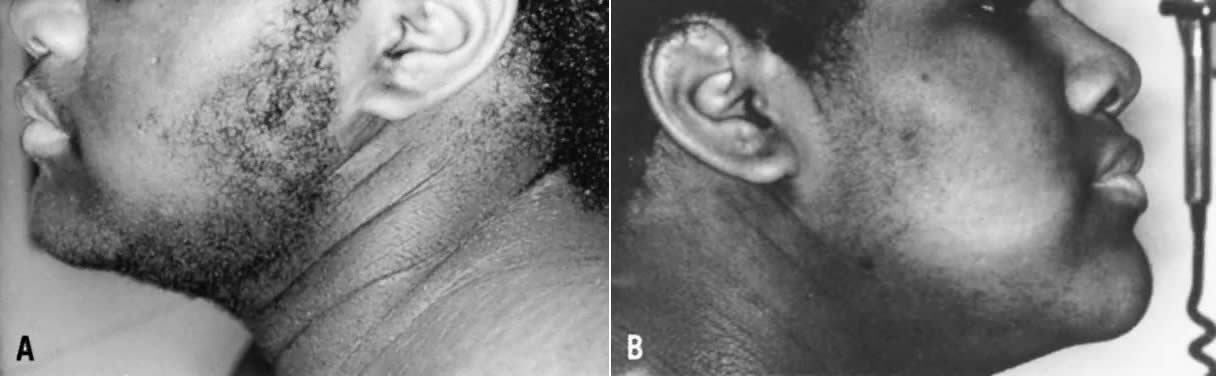
Consequent to the presence of ovarian hyperthecosis, many patients with HAIRAN are severely hyperandrogenic, and even present with a moderate degree of virilization. Patients with HAIRAN syndrome also exhibit acanthosis nigricans, a velvety, hyperpigmented change of the crease areas of the skin (Fig. 7). Some of these patients may also demonstrate forms of lipodystrophy. Many also have, or will have, glucose intolerance or type 2 diabetes mellitus (DM), hypertension, and dyslipidemia, particularly suppressed high-density lipoproteins and hypertrigliceridemia. Overall, both morbidity and mortality in these patients are quite significant, and these women require intensive counseling, follow-up, and treatment of both hyperandrogenic and metabolic abnormalities.
When undergoing treatment for hyperandrogenism, most patients with HAIRAN syndrome respond to standard therapy with antiandrogens and oral contraceptives, although some require more aggressive treatment.92 Ketoconazole93 or longacting gonadotropin-releasing hormone (GnRH) analogues (GnRH-a)92,94 have been used in nonresponsive cases, with good results (Fig. 7). Long-acting GnRH-a inhibits LH-dependent androgen production by reducing circulating gonadotropins to castrate levels. At least 3 to 6 months of gonadotropin suppression is required to achieve full androgen suppression in HAIRAN syndrome, and treatment can be continued for years when combined with add-back estrogen/progestin replacement. If other treatments fail, a bilateral ovarian wedge resection or even oophorectomy may be considered.
CUSHING'S DISEASE.
Hirsutism is present in 60% to 70% of women with Cushing's syndrome. It is generally diffuse, mild, and accompanied by varying degrees of vellus hypertrichosis. Hirsutism, with or without menstrual irregularity, was among the presenting symptoms in 15 of 45 such women, and was the only presenting complaint in 11.95 Overall menstrual irregularities are seen in 80% to 100% of women with Cushing's syndrome, and acne is present in 40% to 50%.96,97,98
Excessive ACTH-dependent adrenocortical function, owing to either an ectopic ACTH-producing tumor or a pituitary tumor (Cushing's disease), can lead to excessive adrenal androgen secretion and hyperandrogenemia. Imura and colleagues did not note virilization in women with ectopic ACTH-producing neoplasms.99 However, all of their patients were Asian, a population that is notably resistant to the development of hirsutism. In contrast, elevated circulating T levels have been documented in women with pituitary Cushing's syndrome.100 An adrenal origin was ascribed to the increased androgen level, because dexamethasone caused a parallel decrease in cortisol and T, ACTH stimulation induced an increase in T that was more profound in women with Cushing's disease compared with normal subjects, and bilateral adrenalectomy dramatically decreased circulating T, correcting the oligomenorrhea and hirsutism.
An increase in the adrenal secretion of androgens may not be the only factor accounting for the hyperandrogenic symptoms in Cushing's disease. Patients with adrenal adenoma generally demonstrate low levels of adrenal androgens, particularly DHEAS.101 Nevertheless, these women demonstrate frequencies of menstrual irregularity, hirsutism, and acne similar to those of patients with generalized adrenal hyperplasia (reflecting increased ACTH stimulation).97 Therefore, it is possible that an increase in ovarian androgen secretion may, in part, account for the hyperandrogenism of these patients. The role played by the increased insulin resistance and gonadotropin abnormalities often noted in patients with Cushing's syndrome in promoting ovarian hyperandrogenemia is unknown. Finally, a direct effect of long-term hypercortisolemia on hair growth, particularly on vellus hairs, cannot be ruled out.
Despite the high frequency of hirsutism reported among women with Cushing's syndrome, hirsute women are rarely diagnosed as cushingoid. As illustration, among approximately 1000 hyperandrogenic women with whose care the author has been involved, only 1 patient has been diagnosed as having Cushing's disease.
ANDROGENIC TUMORS.
Androgen-producing tumors, either ovarian or adrenal, are relatively rare. They should be suspected when the onset of androgenic symptoms is rapid and sudden, and when virilization and masculinization ensue. Steroid-secreting neoplasms are occasionally associated with other systemic symptoms, including weight loss, anorexia, abdominal bloating, and back pain. Suppression and stimulation tests (including corticosteroid, oral contraceptive, human chorionic gonadotropin, and ACTH administration) can be misleading and are not encouraged for the screening or diagnosis of these neoplasms.102 Furthermore, because computed tomographic (CT) scanning for the diagnosis of an adrenal neoplasm, or transvaginal sonography for an ovarian tumor, are relatively sensitive and noninvasive, there is little need today for adrenal or ovarian vein catheterization in the diagnosis of androgen-secreting neoplasms.
Ovarian androgen-secreting neoplasms occur in approximately 1 of 300 to 1 of 1,000 hyperandrogenic patients.103,104,105,106 They are usually palpable on pelvic examination or associated with a unilateral ovarian enlargement seen on ultrasound or CT. Functional ovarian neoplasms generally are not malignant, and they include Sertoli-Leydig cell tumors and lipoid cell tumors. Ovarian tumors should be suspected when hirsutism or virilization is of sudden onset and rapidly progressive. The diagnosis can be further suspected when transvaginal sonography demonstrates an asymmetric ovarian enlargement.
Androgen-producing tumors of the adrenal are less common than ovarian neoplasms and include adenomas and carcinomas. Adrenal carcinomas usually are associated with the development of cushingoid features and can be diagnosed as a large (greater than 6 cm) irregular adrenal mass on CT scanning. The prognosis for patients with adrenocortical carcinoma is poor. The frequency with which these patients present with oligoovulation and hirsutism depends to a significant degree on tumor histopathology. More differentiated tumors tend to produce higher level of androgens, whereas less differentiated tumors produce only minimal amounts of androgens.107 Overall, adrenocortical neoplasias demonstrate less efficient steroidogenesis than normal adrenocortical tissue. However, because these tumors are frequently very large, their overall steroid secretion can be significant.
Because adrenal adenomas secreting solely androgens are exceedingly rare, most patients with an androgen-secreting adrenal neoplasm present with cushingoid features. Furthermore, physicians should keep in mind that signs and symptoms of virilization are encountered with equal frequency in patients with androgen-secreting adrenal adenomas and those with adrenal carcinomas.108
POLYCYSTIC OVARY SYNDROME.
PCOS affects approximately 4% of reproductive-aged women in the United States.4 The disorder is one of the most common causes of oligo-ovulatory infertility109 and hirsutism,110 and it is associated with increased risks of cardiovascular disease (CVD), type 2 DM, endometrial carcinoma, and psychosocial dysfunction.
Patients with PCOS comprise between 60% and 80% of all hyperandrogenic women seen. Although there is no widespread agreement on the definition of PCOS, useful criteria arose from a conference sponsored by the National Institute of Child Health and Human Development in April of 1990.111 Most conference participants indicated that the major criteria for PCOS “should include (in order of importance): (i) hyperandrogenism and/or hyperandrogenemia, (ii) oligo-ovulation, [and] (iii) exclusion of other known disorders, such as Cushing's syndrome, hyperprolactinemia, or congenital [nonclassic] adrenal hyperplasia.” Hence, the diagnosis of PCOS is established by the presence of hyperandrogenic oligo-ovulation, after exclusion of other causes of androgen excess or oligo-ovulation, such as NCAH, hyperprolactinemia, and thyroid dysfunction. The disorder may be evident as early as the age of 12 years.112
Pathologically, the ovarian cortex contains multiple intermediate and atretic follicles measuring 2 to 5 mm in diameter, which give the ovary its “polycystic” appearance. However, not all patients with PCOS have “polycystic” ovaries.113,114 In fact, the sole appearance of “polycystic” ovaries, either on laparoscopy or more commonly at sonography, is not diagnostic for PCOS.113,114,115,116 Excessive levels of circulating androgens from any source will lead to the disruption of follicular development and the accumulation of many small, atretic follicles in the ovarian cortex, producing a “polycystic” picture51,52 (Fig. 8). Phenotypically, 40% to 60% of PCOS patients are obese, predominantly demonstrating abdominal/visceral fat deposition. Although most, if not all, patients with PCOS have ovulatory dysfunction, up to 30% claim to have regular menses.110 In addition, PCOS may manifest as primary amenorrhea.117,118 Although a majority of patients with PCOS demonstrate hirsutism, this clinical finding is by no means uniform.22,119,120,121,122 In one study, 19% of 32 consecutive oligo-ovulatory patients without hirsutism demonstrated increased levels of at least one androgen, suggesting that one fifth of these nonhirsute oligo-ovulatory patients have PCOS.122 Hyperandrogenemia was primarily observed among patients with menstrual cycles greater than 45 days in length, of which 38% had elevated androgens. Hirsutism is also relatively infrequent among Asian women with PCOS.123 Patients with PCOS usually do not present with virilization.
Biochemically, patients with PCOS frequently demonstrate increased circulating levels of free T and a reduction in SHBG, accompanied by variable increases in total T.11,124,125 Approximately 30% demonstrate an abnormally high DHEAS level, although rarely higher than 6000 ng/ml.126,127 The prolactin concentration is usually normal, although as many as 17% to 23% of such patients demonstrate mild intermittent elevations (usually less than 80 ng/ml, depending on the assay) without any other apparent cause.128 An increased LH/FSH ratio, exceeding a 2:1, is observed in approximately 60% of these patients.129,130,131
Most patients with PCOS also demonstrate a variety of metabolic abnormalities. Most significant among these is the presence of insulin resistance, accompanied by compensatory hyperinsulinemia, which affects approximately 30% to 80% of these patients.123,132 The “resistance” to the action of insulin in PCOS refers to the impaired action of this hormone on glucose transport and antilipolysis in adipocytes, in the presence of normal insulin binding.133,134,135 The insulin resistance of PCOS appears to be associated with postreceptor signaling aberrations, although these remain to be confirmed. It should be noted that the insulin resistance of PCOS differs from that observed in type 2 DM and simple obesity. However, although the unique insulin resistance of PCOS occurs even in normal-weight individuals, it is further aggravated by the concomitant presence of obesity.
This resistance to the cellular actions of insulin leads to a compensatory hyperinsulinemia in PCOS, with the excess insulin causing an exaggerated effect in other, less traditionally responsive, tissues. These effects include androgen secretion by the ovarian theca, excess growth of the basal cells of the skin resulting in varying degrees of acanthosis nigricans, increased vascular and endothelial reactivity, and abnormal hepatic and peripheral lipid metabolism.
The increased insulin levels found in patients with PCOS appear to directly enhance LH-stimulated androgen secretion from the ovary.136,137 The persistence of hyperinsulinemia in women who have been both surgically and medically castrated refutes hyperandrogenism as the cause of hyperinsulinemia.138,139 More obviously, normal men have free androgen levels some 30-fold higher than those in women, yet they do not exhibit a greater prevalence of insulin resistance or hyperinsulinemia. Increased levels of insulin also serve to decrease circulating SHBG levels, thus resulting in higher concentrations of free androgens.140
The polycystic ovary syndrome has a strong familial component, appearing to demonstrate a multifactorial form of inheritance with a single dominant gene effect.141,142 For example, if a patient has PCOS, her sister and her daughters have an approximately 50% risk of also having the disorder.143 Therefore, patients should be counseled appropriately. Multiple attempts have been made to detect a molecular genetic abnormality. Although some promising associations have been reported,144,145,146 no single defect has yet been found to account for the disorder in even a subgroup of patients. Finally, patients with PCOS should be counseled regarding their increased risks of developing gestational diabetes, type 2 diabetes, hypertension, endometrial cancer, and, possibly, CVD.
Idiopathic Hirsutism
Although IH is often referred to as “familial hirsutism,” this represents a gross misnomer. It is now well established that other forms of hyperandrogenism (e.g. PCOS) also demonstrate a strong familial predisposition. Therefore, it is misleading to regard IH as being solely familial, because this often translates to “untreatable” in the mind of the patient and the clinician. The diagnosis of IH is reached when an obviously hirsute patient demonstrates normal ovulatory function and, frequently, normal circulating androgen levels. Approximately 15% to 25% of hirsute women are diagnosed as having IH, depending on the extent of endocrine evaluation.110,147 It is critical, however, to confirm the presence of normal ovulatory function (e.g. by using a basal body temperature chart and luteal progesterone measurements) in those hirsute patients who claim to have regular menstrual cycles. Up to 40% of these women actually are oligo-ovulatory.110
Some women with IH demonstrate excessive 5α-reductase activity of the hair follicle, which results in hirsutism despite “normal” circulating androgen levels.148 The measurement of serum 3α-androstanediol glucuronide, a metabolite of DHT, may reflect peripheral 5α-reductase activity and help to confirm “hyperfunction” of this enzyme.148,149,150 Nevertheless, the diagnosis of IH is usually one of exclusion, made when repeated measurements of free and total circulating androgen levels are found to be normal in a patient with hirsutism and regular ovulatory function.
EVALUATION OF THE HIRSUTE PATIENT
History
The pace of pubertal development and its relation to the onset of hirsutism, acne, and/or obesity should be established. Drug or medication use and exposure to or use of skin irritants should be elicited. A detailed menstrual history should be obtained, with an emphasis on determining whether evidence of ovulatory function (e.g. premenstrual molimina) is present. The onset and progression of hirsutism, acne, balding and hair loss, change in extremity or head size, and changes in face contour should be noted.
A detailed family history of endocrine, reproductive, and metabolic disorders should be obtained. A family history of similar hyperandrogenic signs and symptoms is a powerful clue to the inherited basis of the disorder, although a familial association also may be noted in patients with PCOS, HAIRAN syndrome, NCAH, or IH. The cause of hirsutism can often be suspected from the history alone. For example, rapid progression of androgenization, particularly when it appears some time after puberty or in the menopause, is most suggestive of an androgen-secreting neoplasm and not a functional cause of androgen excess.
Physical Examination
Overall, it is most important during the physical examination to determine whether hirsutism or other hyperandrogenic features are truly present and whether there are signs or symptoms of related disorders. The type and pattern of excessive hair growth and/or acne should be noted and scored (Fig. 3). The excessive growth of vellus hairs, producing a “fuzzy” appearance, is termed vellus hypertrichosis and should not be considered hirsutism. Although a number of medical problems or medications can lead to vellus hypertrichosis, it is more commonly an ethnic variant noted particularly in individuals of Scandinavian or Mediterranean extraction. Furthermore, excessive hair growth affecting only the lower legs or forearms does not constitute hirsutism, because these areas are normally covered by a mixture of terminal and vellus hairs.
The presence of alopecia, acne, acanthosis nigricans, lipodystrophy, clitoromegaly, virilization and/or masculinization, pelvic and/or abdominal mass, abdominal and generalized obesity, cushingoid features, coarseness or bluntness of facial features, galactorrhea, thyroid enlargement, or signs of systemic illness should be sought and excluded.
If the degree of androgenization is severe, resulting in clitoromegaly, male-pattern balding, masculinization of the body (e.g. decrease in breast size, increase in muscle mass, loss of hip-to-waist discordance), an androgenic tumor or syndrome of severe insulin resistance (i.e. HAIRAN syndrome) should be suspected. The presence of cushingoid features (centripetal obesity, muscle wasting of the extremities, moon facies, generalized facial rubor, or purple-red abdominal striae) in association with hirsutism is particularly worrisome, because it suggests the possibility of either an adrenocortical carcinoma or ACTH excess caused by an ectopic ACTH-producing tumor or a pituitary tumor.
Laboratory Assessment
The laboratory evaluation should not be excessive or unfocused. For diagnostic purposes, at a minimum the levels of TSH, prolactin, and 17-HP should be measured, to exclude thyroid dysfunction, hyperprolactinemia, and 21-OH-deficient NCAH, respectively. As noted previously, screening for 21-OH-deficient NCAH can be done by measuring the circulating 17-HP in the follicular phase of the menstrual cycle (if the patient is ovulatory), preferably in the morning.65 If the 17-HP level is higher than 2 ng/ml (200 ng/dl) and it is certain that the sample was obtained in the preovulatory phase, the patient should undergo acute adrenal stimulation testing. For this test, 250 μg (l vial) of ACTH-(1-24) (Cortrosyn) is injected intravenously and the 17-HP level is checked 30 to 60 minutes later. If the 17-HP level after ACTH-(1-24) administration is higher than 10 ng/ml, the diagnosis of 21-OH-deficient NCAH is highly probable; and if it is higher than 15 ng/ml, the diagnosis is established. It is unnecessary to rule out 3β-HSD- and 11-OH-deficient NCAH, because these conditions are extremely rare.
Levels of total and free T, and DHEAS may also be measured, although the added diagnostic value of these tests is probably minimal. The serum T levels are often increased in patients with adrenal or ovarian androgen-secreting tumors, usually to more than 200 ng/dl.151,152 However, the use of this specific cutoff greatly depends on the particular T assay used by each laboratory and does not allow differentiation between an ovarian and an adrenal neoplasm. Furthermore, 20% to 50% of patients with androgen-producing ovarian tumors have T levels lower than this value,151,152 and the majority of women with T levels higher than 200 ng/dl have either HAIRAN syndrome, hyperthecosis, or PCOS, and not a functional tumor.153 In fact, a study of 478 consecutive hyperandrogenic patients noted that the level of total T had a very low positive predictive value for an ovarian androgen-secreting neoplasm.106 Most, but not all, ovarian tumors can be detected by transvaginal ultrasonography.
Likewise, the predictive value of DHEAS as a marker for an androgen-secreting neoplasm appears to be low. Yeun and colleagues, based on a single case report, suggested that a DHEAS measurement greater than 6600 ng/ml was consistent with the presence of an adrenal tumor.154 Nonetheless, these researchers emphasized the need for serial measurements of serum T and DHEAS, in order for these levels to be useful in the screening of virilizing adrenal adenomas. Multiple reports, however, indicate that androgenized females with adrenal carcinoma or adenoma can have normal or only slightly elevated DHEAS values.152,155,156,157 Surrey and colleagues noted, in a series of patients with virilizing tumors, that the circulating DHEAS level was greater than 7000 ng/ml in one patient with an ovarian lipoid cell tumor and not in their only patient with an adrenal adenoma.158 Overall, the clinical evaluation remains the most sensitive marker of an androgen-secreting tumor, and the measurement of total T or DHEAS is of limited value and can actually be misleading. The diagnosis of an adrenal neoplasm is usually established by a CT scan of the adrenal, with images obtained at 0.5-cm intervals.
Patients with features suggestive of acromegaly, Cushing's syndrome, thyroid dysfunction, or hyperprolactinemia should be evaluated further, as indicated. If male pseudohermaphroditism or XY gonadal dysgenesis is suspected because of the presence of a blind vaginal pouch and/or abnormality of the external genitalia, a karyotype should be obtained.
Finally, if the diagnosis of PCOS is confirmed, an assessment of the patient's metabolic status should be made. At a minimum this should include measurement of the fasting glucose level. Although specific guidelines do not exist, it may be appropriate to also obtain a lipid profile and a glycosylated hemoglobin (i.e. hemoglobin A1C) determination every 2 to 3 years in women with PCOS who are older than 35 years of age, particularly if they have a strong family history of type 2 DM, CVD, or obesity.
TREATMENT OF HIRSUTISM
The treatment of hirsutism should be undertaken as soon as the diagnosis is established. Although in young children or teenagers the degree of hair growth may not be alarming, one must remember that these disorders are usually progressive and the formation of terminal hairs is not reversible. Furthermore, the presence of hirsutism can be cosmetically disfiguring and can have a significant negative impact on a young woman's social and emotional development.
It is extremely important for optimum patient compliance to present realistic therapeutic expectations. The primary goal of treatment in hirsutism is to stop the development of any new terminal hairs. Generally, hormonal therapy also decreases the growth rate, diameter, and pigmentation of terminal hairs that are already present. However, it does not generally reverse the transformation of vellus to terminal hairs. Finally, it is important to emphasize that therapy for hirsutism may take up to 6 months before a clinical response is noted. Therapy for hirsutism consists of mechanical, hormonal, and surgical means. Any terminal hairs remaining after 6 to 12 months of adequate suppression with medical therapy must be destroyed mechanically.
Hormonal Treatment
Current hormonal therapy consists of drugs that primarily suppress androgen secretion (from the ovary, the adrenal, or both) and drugs that primarily inhibit the action of androgens at the periphery (i.e., the skin). Often these drugs are combined to simultaneously suppress production of androgens while peripherally blocking their effect.
SUPPRESSION OF ANDROGEN PRODUCTION.
Oral Contraceptive Pills.
The mainstay of androgen excess treatment is oral contraceptive pills (OCPs), which, by suppressing the secretion of LH, lead to a decrease in ovarian androgen production.159 In addition, the estrogen fraction in the OCPs increases circulating SHBG, with a subsequent decrease in free T levels. Full hormonal suppression is usually seen within 3 months.
OCPs also decrease adrenal androgen production, although minimally, by a mechanism that is not yet clear but which may be related to a decrease in circulating ACTH. The progestin in OCPs can also compete for 5α-reductase and the androgen receptor.159 Because the progestin in the OCPs may in turn decrease SHBG and increase free T, it is most important to select a pill with the least androgenic progestin (e.g. norethindrone acetate, ethynodiol diacetate desogesterol). OCPs should be administered in the usual cyclic fashion, and the circulating androgen levels should be checked periodically to assess the adequacy of hormonal suppression and stimulation of SHBG levels.
Estrogens and Progestins.
The use of oral estrogen (with a progestin administered in a cyclic fashion when the uterus is present) has been advocated for older hyperandrogenic women and those not tolerating OCPs. The doses of estrogen needed are higher than those required for regular postmenopausal hormonal therapy. Oral, but not transdermal, estrogen therapy increases circulating SHBG, decreasing free T. Estrogens also decrease circulating LH levels, with a reduction in gonadal androgen secretion, and potentially antagonize the effect of androgens at the hair follicle.
High doses of progestins (e.g. 20 to 30 mg of medroxyprogesterone acetate daily) have been used for the treatment of hirsutism.160 Progestins increase the clearance of T by the liver and may lead to a decrease in circulating LH. However, this therapy also leads to a decrease in the circulating SHBG concentration, which may reduce its antiandrogenic effectiveness. Furthermore, side effects may become unacceptable and include breakthrough bleeding, liver dysfunction, possibly vascular changes, weight gain, depression, and hot flushes.
Long-Acting Gonadotropin-Releasing Hormone Analogues.
A long-acting GnRH-a (e.g. Lupron—depot 3.75 mg/mo) is rarely required to suppress the hypothalamic-pituitary-ovarian axis in patients with PCOS. However, this drug is particularly useful for patients with severe insulin resistance and hyperinsulinism92,94,161,162 and for those with severe androgen excess and hyperthecosis.
Ketoconazole.
Ketoconazole, an imidazole antimycotic agent, is a known inhibitor of cytochrome P450 steroidogenic enzymes, specifically P450ssc, P45017α, and P450arom. Therapy with 800 to 1200 mg of ketoconazole daily decreases hirsutism163,164; treatment with lower doses (400 mg/day) is less effective.165 However, side effects are common and include headaches, nausea, scalp hair loss, and liver function abnormalities. Treatment with ketoconazole may be considered for patients not responding to conventional therapy,93,166 although its use remains experimental.
Insulin-Sensitizing Agents.
Because up to 70% of patients with PCOS demonstrate overt insulin resistance and hyperinsulinism with dyslipidemia, the administration of insulin-sensitizing agents, principally metformin and troglitazone, has been proposed for the treatment of this disorder. These agents have various potential advantages over traditional therapies: they correct both the metabolic and the endocrinologic aberrations of the disorder; they permit the resumption of normal endogenous ovulatory function, with little or no risk of ovarian hyperstimulation and multiple gestation; and they may possibly decrease the long-term risk of type 2 DM and CVD.
It is important to note that only small studies or preliminary data concerning the use of these agents in PCOS have been published.167,168,169,170,171 Conclusive data regarding outcome, patient selection, and risks and complications, while in the process of being collected, are not yet available. Data regarding their effect on hair growth in the hirsute patient remain anecdotal at best.
Glucocorticoids.
Dexamethasone (0.25 to 0.5 mg every evening to every other evening) or prednisone (5 to 10 mg/day) can be used as an adjuvant for the treatment of patients with significant adrenal hyperandrogenemia and those with NCAH. Corticosteroid administration reduces ACTH stimulation to the adrenal cortex, leading to a decrease in the production of morning cortisol and various androgens in hyperandrogenic women.172,173,174 However, glucocorticoids alone appear to have limited effect on ovulatory dysfunction175 or hair growth175 in PCOS or NCAH,178 and they may be associated with weight gain and osteoporosis. Nonetheless, glucocorticoids appear to be an effective adjuvant for the treatment of androgen-associated acne.179
BLOCKADE OF ANDROGEN ACTION.
Drugs in this category include antiandrogens and 5α-reductase inhibitors.
Spironolactone.
SPA is an aldosterone antagonist and a mild diuretic. It also is an effective therapy for hirsutism,180,181,182,183,184,185 competing for the androgen receptor, 5α-reductase, and the binding of T to SHBG.186 Furthermore, SPA blocks the action of various enzymes involved in androgen biosynthesis.186,187,188 Initially, a dose as high as 200 mg a day should be used. If the dose is slowly increased from 25 mg to 200 mg per day, by 25 mg increments every 3 days or so, patients will develop a minimum of side effects. The side effects most commonly associated with SPA use include dyspepsia, nausea, polyuria, fatigue, headaches, and irregular menses. The incidence of metrorrhagia may be decreased by administering the SPA in conjunction with an OCP. Hypertensive patients already receiving a potassium-saving diuretic may rarely develop hyperkalemia when SPA is taken simultaneously.
Flutamide.
Flutamide is an androgen receptor blocker that appears to be as effective as SPA for the treatment of hirsutism, in doses of 250 to 500 mg daily.189,190,191 Side effects include liver enzyme abnormalities, the appearance of greenish urine, and excessive dryness of skin or scalp hair. It has some teratogenic potential, as does SPA.
Cyproterone Acetate.
CPA is a strong progestin and antiandrogen and an effective treatment for hirsutism.192 It produces a decrease in circulating testosterone and androstenedione levels through a reduction in LH. Furthermore, CPA antagonizes the effect of androgens at the androgen receptor level. Side effects may include adrenal insufficiency and loss of libido. This drug currently is not available in the United States, and it is unlikely that it will be approved by the U.S. Food and Drug Administration (FDA), because SPA appears to be equally effective.
Finasteride.
Finasteride is a 5α-reductase inhibitor that is FDA-approved for the treatment of benign prostatic hyperplasia. Finasteride may be useful for the treatment of hirsutism in women (5 mg/day),193 despite a paradoxic increase in circulating testosterone levels. Teratogenicity (i.e. feminization of a male infant) is a major concern.
MECHANICAL MEANS OF HAIR REMOVAL.
Mechanical means, by themselves, do little to improve the long-term course of hirsutism and may actually worsen the problem. However, while awaiting the full effect of hormonal therapy, it is best to encourage the patient to maintain optimum cosmesis by either shaving or bleaching. Although shaving can lead to a blunt hair end, which may feel like “stubble,” it does not result in a worsening of the hirsutism,194,195 and it causes little trauma to the skin. It is surprising how well shaving and the proper application of makeup results in a very satisfactory appearance. Recording the interval between shavings before each office visit provides a relatively simple way to monitor the efficacy of treatment.
Plucking or waxing should be discouraged because it may stimulate the growth of surrounding follicles, at least in animal models,54 and may lead to folliculitis with the subsequent development of ingrown hairs. Excessive use of depilating agents, particularly on the face, can result in chronic irritation and worsening of the skin condition.
Electrolysis (i.e. electroepilation) provides long-term destruction of hair follicles, albeit slowly.196,197 Efficacy ranges from 15% to 50% permanent hair loss198 with repeated treatments, which may take months to years. Scarring can occur after electrolysis, especially if it is inexpertly performed.199 However, hirsute patients often turn for assistance first to electrologists.200 Therefore, it behooves all physicians caring for these women to establish and maintain communication with the electrology profession, referring patients to those electrologists who are properly trained. It should also be noted that certification and training requirements for electrology vary widely from state to state.
Directed damage to hair follicles based on the theory of selective photothermolysis by lasers has been reported.201,202 Various lasers have been evaluated for their effectiveness in treating body hair. High-energy ruby light was shown to be effective as a tool for removal of unwanted hair growth in 1979 and was introduced for this clinical use in 1992, with the free-running long-pulse ruby laser introduced for routine use after 1995. The mode of action has been proposed to be selective hair thermolysis, which implies that light is selectively absorbed in certain structures of the hair follicle. Grey and fair-colored hair should not be expected to disappear. Laser-treated hair follicles appear to go into telogen, which may last for up 2 years after treatment.201,202
Peer-reviewed reports of the true long-term benefit of laser ablation in hirsutism are lacking, notwithstanding the fact that many dermatologists today are offering laser treatment in their practices as an “effective” means of removing body hair. In a 1998 study,201 a single treatment with the normal-mode ruby laser (694 nm, 270 microseconds, 6-mm beam diameter) of six skin areas on the thighs or back was compared with shaved and wax-epilated control sites in 13 volunteers. Terminal hairs were counted before and after laser exposure. After a single treatment session, 4 of the 13 subjects demonstrated permanent hair loss when evaluated 2 years later, and 6 others had partial hair loss; in 3 subjects, hair regrowth was complete or almost complete.
Overall, laser treatment may be effective in permanently removing hair in some patients, although most women are likely to require multiple treatments. Nonetheless, it is still unclear whether hirsute women experience the same degree of therapeutic effectiveness as reported in nonhirsute patients, and further studies including these patients are required. Side effects of laser epilation include pigmentary changes of treated skin and burning and scarring of the treated area. Most current laser technologies have minimized these side effects with the use of more selective wavelengths and cooling devices attached to the probe.
SURGICAL TREATMENT OF ANDROGEN EXCESS.
In the past, ovarian wedge resection was used with satisfactory improvement in menstrual cycles,203,204 although with little impact on hirsutism. Patients today rarely require this treatment, with the exception of those with suspected ovarian tumor and some patients with hyperthecotic or HAIRAN syndrome. Rarely are individuals so affected that a bilateral oophorectomy need be considered, although it may be applicable in some severely affected young girls who have little hope of improvement otherwise. However, these patients are very much the exception, particularly since the advent of long-acting GnRH-a. More recently, laparoscopic wedge resection205 or fulguration of multiple areas of the ovarian cortex206,207,208 has been proposed, although long term follow-up information is lacking. In general, these destructive procedures should be used as the last line of therapy, because they almost invariably lead to ovarian adhesions. Furthermore, although they may improve ovulatory function, they have little impact on hirsutism.
CONCLUSIONS
Women with hirsutism require long-term follow-up, counseling, and emotional support, because these disorders are usually lifelong and progressive unless adequately treated. Most of these patients have PCOS, IH, NCAH, HAIRAN syndrome, or, rarely, androgen-secreting ovarian tumor (Table 1). The primary goal of hormonal therapy is to stop the progression of hirsutism. Hormonal therapy may also decrease the growth rate, diameter, and pigmentation of terminal hairs that are already present. A clinical response may not be apparent before 6 months of therapy. Because hormonal treatment does not reverse the transformation of vellus to terminal hairs, electrolysis or possibly laser therapy, may serve as a useful adjuvant. Encouraging the patient to shave or bleach, rather than pluck or wax, is also helpful.
TABLE 1. Distribution of the Various Causes of Hirsutism
Etiology | Proportion of total patients |
Polycystic ovary syndrome | 60%–80% |
Idiopathic hirsutism | 15%–30% |
HAIRAN syndrome | 2%–4% |
Nonclassic adrenal hyperplasia (21-hydroxylase deficient) | 1%–10% |
Ovarian tumors | 1/300–1/1,000 |
Others | Very rare |
REFERENCES
Ferriman D, Gallwey JD: Clinical assessment of body hair growth in women. J Clin Endocrinol Metab 21: 1440, 1961 |
|
McKnight E: The prevalence of “hirsutism” in young women. Lancet 1: 410, 1964 |
|
Hartz AJ, Barboriak PN, Wong A et al: The association of obesity with infertility and related menstrual abnormalities in women. Int J Obes 3: 57, 1979 |
|
Knochenhauer ES, Key TJ, Kahsar-Miller M et al: Prevalence of the polycystic ovarian syndrome in unselected Black and White women of the Southeastern United States: A prospective study. J Clin Endocrinol Metab 83: 3078, 1998 |
|
James VHT, Goodall A: Androgen production in women. In Jeffcoate SL (ed): Androgens and Anti-Androgen Therapy, p 23. New York: John Wiley & Sons, 1982 |
|
Rosenfeld RS, Rosenberg BJ, Fukushima DK et al: 24-Hour secretory pattern of dehydroisoandrosterone and dehydroisoandrosterone sulfate. J Clin Endocrinol Metab 40: 850, 1975 |
|
Huq MS, Pfaff M, Jespersen D et al: Concurrence of aldosterone, androgen and cortisol secretion in adrenal venous effluents. J Clin Endocrinol Metab 42: 230, 1976 |
|
Suzuki T: Circadian rhythm of adrenocortical secretory activity. In Suzuki T (ed): Physiology of Adrenocortical Secretion, p 72. New York: Karger, 1983 |
|
Kato T, Horton R: Studies of testosterone binding globulin. J Clin Endocrinol Metab 28: 1160, 1968 |
|
Dunn JF, Nisula BC, Rodbard D: Transport of steroid hormones: Binding of 21 endogenous steroids to both testosterone-binding globulin and corticosteroid-binding globulin in human plasma. J Clin Endocrinol Metab 53: 58, 1981 |
|
Rosenfield RL: Plasma testosterone binding globulin and indexes of the concentration of unbound plasma androgens in normal and hirsute subjects. J Clin Endocrinol Metab 32: 717, 1971 |
|
Vermeulen A, Verdonck L, Van der Straeten M et al: Capacity of the testosterone-binding globulin in human plasma and influence of specific binding of testosterone on its metabolic clearance rate. J Clin Endocrinol Metab 29: 1470, 1969 |
|
Plymate R, Leonard JM, Paulsen CA et al: Sex hormone-binding globulin changes with androgen replacement. J Clin Endocrinol Metab 57: 645, 1983 |
|
Tochimoto S, Olivo J, Southren AL et al: Studies of plasma β-globulin: Sex difference and effect of ethinyl estradiol and testosterone. Proc Soc Exp Biol Med 134: 700, 1970 |
|
Musa BU, Doe RP, Seal US: Serum protein alterations produced in women by synthetic estrogens. J Clin Endocrinol Metab 27: 1463, 1967 |
|
Chetkowski RJ, Meldrum DR, Steingold KA et al: Biologic effects of transdermal estradiol. N Engl J Med 314: 1615, 1986 |
|
Azziz R: Reproductive endocrinologic alteration in female asymptomatic obesity. Fertil Steril 52: 703, 1989 |
|
Uno H: Biology of hair growth. Semin Reprod Endocrinol 4: 131, 1986 |
|
Bernstein RM, Rassaman WR: The aesthetics of follicular transplantation. Dermatol Surg 23: 785, 1997 |
|
Bernstein RM, Rassaman WR: Follicular transplantation: Patient evaluation and surgical planning. Dermatol Surg 23: 771, 1997 |
|
Randall VA: Androgens and human hair growth. Clin Endocrinol 40: 439, 1994 |
|
McKenna TJ, Loughlin T, Daly L et al: Variable clinical and hormonal manifestations of hyperandrogenemia. Metabolism 33: 714, 1984 |
|
Darley CR, Moore JW, Besser GM et al: Androgen status in women with late-onset or persistent acne vulgaris. Clin Exp Dermatol 9: 28, 1984 |
|
Steinberger E, Rodriguez-Rigau LJ, Smith KD et al: The menstrual cycle and plasma testosterone levels in women with acne. J Am Acad Dermatol 4: 54, 1981 |
|
Lucky AW, McGuire J, Rosenfield RL et al: Plasma androgens in women with acne vulgaris. J Invest Dermatol 81: 70, 1983 |
|
Lawrence DM, Katz M, Robinson TWE et al: Reduced sex hormone binding globulin and derived free testosterone levels in women with severe acne. Clin Endocrinol (Oxf) 15: 87, 1981 |
|
Held BL, Nader S, Rodriguez-Rigau LJ et al: Acne and hyperandrogenism. J Am Acad Dermatol 10: 112, 1984 |
|
van der Meeren HLM, Thijssen JH: Circulating androgens in male acne. Br J Dermatol 110: 609, 1984 |
|
Sultan C, Oliel V, Audran F et al: Free and total plasma testosterone in men and women with acne. Acta Derm Venereol 66: 301, 1986 |
|
Sheehan-Dare RA, Hughes BR, Cunliffe WJ: Clinical markers of androgenicity in acne vulgaris. Br J Dermatol 119: 723, 1988 |
|
Levell MJ, Cawood ML, Burke B et al: Acne is not associated with abnormal plasma androgens. Br J Dermatol 120: 649, 1989 |
|
Ginsberg GS, Birnbaum MD, Rose LI: Androgen abnormalities in acne vulgaris. Acta Derm Venereol 61: 431, 1981 |
|
Marynick SP, Chakmakjian ZH, McCaffree DL et al: Androgen excess in cystic acne. N Engl J Med 308: 981, 1983 |
|
Vexiau P, Husson C, Chivot M et al: Androgen excess in women with acne alone compared with women with acne and/or hirsutism. J Invest Dermatol 94: 279, 1990 |
|
Pekkarinen A, Sonck CE: Adrenocortical reserves in acne vulgaris. Acta Derm Venereol 42: 200, 1962 |
|
Chrousos GP, Peck GL, Gross EG et al: Adrenal function in women with idiopathic acne. J Invest Dermatol 78: 468, 1982 |
|
Lucky AW, Rosenfield RL, McGuire J et al: Adrenal androgen hyperresponsiveness to adrenocorticotropin in women with acne and/or hirsutism: Adrenal enzyme defects and exaggerated adrenarche. J Clin Endocrinol Metab 62: 840, 1986 |
|
Sansone G, Reisner RM: Differential rates of conversion of testosterone to dihydrotestosterone in acne and in normal human skin: A possible pathogenic factor in acne. J Invest Dermatol 56: 366, 1971 |
|
Lookingbill DP, Horton R, Demers LM et al: Tissue production of androgens in women with acne. J Am Acad Dermatol 12: 481, 1985 |
|
Hay JB, Hodgins MB: Metabolism of androgens by human skin in acne. Br J Dermatol 91: 123, 1974 |
|
Palatsi R, Reinila M, Kivinen S: Pituitary function and DHEA-S in male acne and DHEA-S, prolactin and cortisol before and after oral contraceptive treatment in female acne. Acta Derm Venereol 66: 225, 1986 |
|
Hammerstein J, Moltz L, Schwartz U: Antiandrogens in the treatment of acne and hirsutism. J Steroid Biochem 19: 592, 1983 |
|
Darley CR, Moore JW, Besser GM et al: Low dose prednisolone or oestrogen in the treatment of women with late onset or persistent acne vulgaris. Br J Dermatol 108: 345, 1983 |
|
Goodfellow A, Alaghband-Zadeh J, Carter G et al: Oral spironolactone improves acne vulgaris and reduces sebum excretion. Br J Dermatol 11: 209, 1984 |
|
Muhlemann MF, Carter GD, Cream JJ et al: Oral spironolactone: An effective treatment for acne vulgaris in women. Br J Dermatol 115: 227, 1986 |
|
Nader S, Rodriguez-Rigau LJ, Smith KD et al: Acne and hyperandrogenism: Impact of lowering androgen levels with glucocorticoid treatment. J Am Acad Dermatol 11: 256, 1984 |
|
Ettinger B, Goldfield B, Burrill E et al: Plasma testosterone stimulation-suppression dynamics in hirsute women. Am J Med 54: 195, 1973 |
|
Ludwig E: Classification of the types of androgenetic alopecia (common baldness) occurring in the female sex. Br J Dermatol 97: 247, 1977 |
|
Strickler JH, Garrett AB: Androgen excess and hair loss in women. In Azziz R, Nestler JE, Dewailly D (eds): Androgen Excess Disorders in Women, p 141. Philadelphia: Lippincott-Raven, 1977 |
|
Futterweit W, Dunaif A, Yeh H-C et al: The prevalence of hyperandrogenism in 109 consecutive female patients with diffuse alopecia. J Am Acad Dermatol 19: 831, 1988 |
|
Spinder T, Spijkstra JJ, van den Twell JG et al: The effects of long term testosterone administration on pulsatile luteinizing hormone secretion and on ovarian histology in eugonadal female to male transsexual subjects. J Clin Endocrinol Metab 69: 151, 1989 |
|
Amirikia H, Savoy-Moore RT, Sundareson AS et al: The effects of long-term androgen treatment on the ovary. Fertil Steril 45: 202, 1986 |
|
Deroubaix-Allard D, Prevost G, Dewailly D: Acromegaly and androgen excess. In Azziz R, Nestler JE, Dewailly D (eds): Androgen Excess Disorders in Women, p 585. Philadelphia: Lippincott-Raven, 1977 |
|
Silver AF, Chase HB, Arsenault CT: Early anagen initiated by plucking compared with early spontaneous anagen. In Montagna W, Dobson RL (eds): Advances in Biology of Skin. Vol 9: Hair Growth, p 265. New York: Pergamon Press, 1967 |
|
Caputo R, Monti S, et al: The treatment of visible signs of senescence: The Italian experience. Br J Dermatol 112 (Suppl 35): 97, 1990 |
|
Tosti A, Misciali C, Piraccini BM et al: Drug-induced hair loss and hair growth. Drug Safety 10: 310, 1994 |
|
Philip J, Trolle D: Familial male hermaphroditism with delayed and partial masculinization. Am J Obstet Gynecol 93: 1076, 1965 |
|
Rosen GF, Kaplan B, Lobo RA: Menstrual function and hirsutism in patients with gonadal dysgenesis. Obstet Gynecol 71: 677, 1988 |
|
Azziz R, Dewailly D, Owerbach D: Non-classic adrenal hyperplasia: Current concepts. J Clin Endocrinol Metab 78: 810, 1994 |
|
Azziz R, Slayden SM: Mechanisms of steroid excess in 21-hydroxylase deficient non-classic adrenal hyperplasia. J Soc Gynecol Investig 3: 297, 1996 |
|
Feuillan P, Pang S, Schurmeyer T et al: The hypothalamic-pituitary-adrenal axis in partial (late-onset) 21-hydroxylase deficiency. J Clin Endocrinol Metab 67: 154, 1988 |
|
Dewailly D, Vantyghem-Haudiquet M-C, Sainsard C et al: Clinical and biological phenotypes in late-onset 21-hydroxylase deficiency. J Clin Endocrinol Metab 63: 418, 1986 |
|
Azziz R, Zacur HA: 21-Hydroxylase deficiency in female androgenism: Screening and diagnosis. J Clin Endocrinol Metab 69: 577, 1989 |
|
Kuttenn F, Couillin P, Girard F: Late-onset adrenal hyperplasia in hirsutism. N Engl J Med 313: 224, 1985 |
|
Azziz R, Hincapie LC, Knochenhauer ES et al: Screening for 21-hydroxylase deficient non-classic adrenal hyperplasia: A prospective study. Fertil Steril 72: 996, 1999 |
|
Fiet J, Gueux B, Gourmelen M et al: Comparison of basal and adrenocorticotropin-stimulated plasma 21-deoxycortisol and 17-hydroxyprogesterone values as biological markers of late-onset adrenal hyperplasia. J Clin Endocrinol Metab 66: 659, 1988 |
|
New MI, Lorenzen F, Lerner DJ et al: Genotyping steroid 21-hydroxylase deficiency: Hormonal reference data. J Clin Endocrinol Metab 57: 320, 1983 |
|
Azziz R: 21-Hydroxylase deficient non-classic adrenal hyperplasia. The Endocrinologist 5: 297, 1995 |
|
Azziz R, Boots LR, Parker CR Jr et al: 11β-hydroxylase deficiency in hyperandrogenism. Fertil Steril 55: 733, 1991 |
|
Carmina E, Malizia G, Pagano M et al: Prevalence of late-onset 11β-hydroxylase deficiency in hirsute patients. J Endocrinol Invest 11: 595, 1988 |
|
Joeher K, Geley S, Strasser-Wozak EMC et al: CYP11B1 mutations causing nonclassic congenital adrenal hyperplasia due to 11β-hydroxylase deficiency. Hum Mol Genet 6: 1829, 1997 |
|
Pang S, Lerner AJ, Stoner E et al: Late-onset adrenal steroid 3β-hydroxysteroid dehydrogenase deficiency: I. A cause of hirsutism in pubertal and postpubertal women. J Clin Endocrinol Metab 60: 428, 1985 |
|
Sanchez R, Rheaume E, Laflamme N et al: Detection and functional characterization of the novel missense mutation Y254D in type II 3β-hydroxysteroid dehydrogenase (3βHSD) gene of a female patient with nonsalt-losing 3βHSD deficiency. J Clin Endocrinol Metab 78: 561, 1994 |
|
Chang YT, Zhang L, Alkaddour HS et al: Absence of molecular defect in the type II 3β-hydroxysteroid dehydrogenase (3β-HSD) gene in premature pubarche children and hirsute female patients with moderately decreased adrenal 3β-HSD activity. Pediatr Res 37: 820, 1995 |
|
Pang S: The molecular and clinical spectrum of 3βhydroxysteroid dehydrogenase deficiency disorder. Trends Endocrinol Metab 9: 82, 1998 |
|
Zerah M, Rheaume E, Mani P et al: No evidence of mutations in the genes for type I and type II 3β-hydroxysteroid dehydrogenase (3βHSD) in nonclassical 3βHSD deficiency. J Clin Endocrinol Metab 79: 1811, 1994 |
|
Sakkal-Alkaddour H, Zhang L, Yang X et al: Studies of 3β-hydroxysteroid dehydrogenase genes in infants and children manifesting premature pubarche and increased adrenocorticotropin-stimulated Δ5-steroid levels. J Clin Endocrinol Metab 81: 3961, 1996 |
|
Tajima T, Nishi Y, Takase A et al: No genetic mutation in type II 3β-hydroxysteroid dehydrogenase gene in patients with biochemical evidence of enzyme deficiency. Horm Res 47: 49, 1997 |
|
Azziz R, Bradley EL Jr, Potter HD et al: 3β-Hydroxysteroid dehydrogenase deficiency in hyperandrogenism. Am J Obstet Gynecol 168: 889, 1993 |
|
Moran C, Potter HD, Reyna R et al: Prevalence of 3β-hydroxysteroid dehydrogenase deficient non-classic adrenal hyperplasia in hyperandrogenic women with adrenal androgen excess. Am J Obstet Gynecol 181: 596, 1999 |
|
Barbieri RL, Ryan KJ: Hyperandrogenism, insulin resistance, and acanthosis nigricans syndrome: A common endocrinopathy with distinct pathophysiologic features. Am J Obstet Gynecol 147: 90, 1983 |
|
Barth JH, Ng LL, Wojnarowska F et al: Acanthosis nigricans, insulin resistance and cutaneous virilism. Br J Dermatol 118: 613, 1988 |
|
Richards GE, Cavallo A, Meyer WJ III et al: Obesity, acanthosis nigricans, insulin resistance, and hyperandrogenemia: pediatric perspective and natural history. J Pediatr 7: 893, 1985 |
|
Vidal-Puig A, Moller DE: Classification, prevalence, clinical manifestations, and diagnosis. In Azziz R, Nestler JE, Dewailly D (eds): Androgen Excess Disorders in Women, p 227. Philadelphia: Lippincott-Raven, 1977 |
|
Moller DE, Flier JS: Detection of an alteration in the insulin-receptor gene in a patient with insulin resistance, acanthosis nigricans, and the polycystic ovary syndrome (type A insulin resistance). N Engl J Med 319: 1526, 1988 |
|
Kadowaki T, Bevins CL, Cama A et al: Two mutant alleles of the insulin receptor gene in a patient with extreme insulin resistance. Science 240: 787, 1988 |
|
Moller DE, Cohen O, Yamaguchi Y et al: Prevalence of mutations in the insulin receptor gene in subjects with features of the type A syndrome of insulin resistance. Diabetes 43: 247, 1994 |
|
Taylor SI, Dons RF, Hernandez E et al: Insulin resistance associated with androgen excess in women with autoantibodies to the insulin receptor. Ann Intern Med 97: 851, 1982 |
|
Kellett HA, Collier A, Raylor R et al: Hyperandrogenism, insulin resistance, acanthosis nigricans, and systemic lupus erythematosus associated with insulin receptor antibodies. Metabolism 7: 656, 1988 |
|
Scully RE: Correspondence: Reply to Dr. Fienberg. Am J Obstet Gynecol 119: 864, 1974 |
|
Nagamani M, Lingold JC, Gomez LG et al: Clinical and hormonal studies in hyperthecosis of the ovaries. Fertil Steril 36: 326, 1981 |
|
Azziz R: The hyperandrogenic-insulin resistance-acanthosis nigricans (HAIRAN) syndrome: therapeutic response. Fertil Steril 61: 570, 1994 |
|
Pepper GM, Poretsky L, Gabrilove JL et al: Ketoconazole reverses hyperandrogenism in a patient with insulin resistance and acanthosis nigricans. J Clin Endocrinol Metab 65: 1047, 1987 |
|
Corenblum B, Baylis BW: Medical therapy for the syndrome of familial virilization, insulin resistance, and acanthosis nigricans. Fertil Steril 53: 421, 1990 |
|
Ross EJ, Marshall-Jones P, Friedman M: Cushing's syndrome: diagnostic criteria. Q J Med 35: 149, 1966 |
|
Gold EM: The Cushing syndromes: Changing views of diagnosis and treatment. Ann Intern Med 90: 829, 1979 |
|
Ross EJ, Linch DC: Cushing's syndrome-killing disease: Discriminatory value of signs and symptoms aiding early diagnosis. Lancet 646, 1982 |
|
Urbanic RC, George JM: Cushing's disease: 18 years' experience. Medicine (Baltimore) 60: 14, 1981 |
|
Imura H, Matsukura S, Yamamoto H et al: Studies on ectopic ACTH-producing tumors: II. Clinical and biochemical features of 30 cases. Cancer 35: 1430, 1975 |
|
Smals AGH, Kloppenborg PWC, Benraad TJ: Plasma testosterone profiles in Cushing's syndrome. J Clin Endocrinol Metab 45: 240, 1977 |
|
Yamaji T, Ishibashi M, Sekihara H et al: Serum dehydroepiandrosterone sulfate in Cushing's syndrome. J Clin Endocrinol Metab 59: 1164, 1984 |
|
Padilla SL: Androgen-producing tumors in children and adolescents. Adolesc Pediatr Gynecol 2: 135, 1989 |
|
Barbieri RL: Hyperandrogenic disorders. Clin Obstet Gynecol 33: 640, 1990 |
|
O'Driscoll JB, Mamtora H, Higginson J et al: A prospective study of the prevalence of clear-cut endocrine disorders and polycystic ovaries in 350 patients presenting with hirsutism or androgenic alopecia. Clin Endocrinol 41: 231, 1994 |
|
Moran C, Tapia M del C, Hernandez E et al: Etiologic review of hirsutism in 250 patients. Arch Med Res 25: 311, 1994 |
|
Waggoner W, Azziz R: Total testosterone and DHEAS levels as predictors of androgen-secreting neoplasms: A populational study. Gynecol Endocrinol 13: 1, 1999 |
|
Hogan TF, Gilchrist KW, Westring DW et al: A clinical and pathological study of adrenocortical carcinoma. Cancer 45: 2880, 1980 |
|
Gabrilove JL, Seman AT, Sabet R et al: Virilizing adrenal adenoma with studies on the steroid content of the adrenal venous effluent and a review of the literature. Endocr Rev 2: 462, 1981 |
|
Hull MGR: Epidemiology of infertility and polycystic ovarian disease: Endocrinological and demographic studies. Gynecol Endocrinol 1: 235, 1987 |
|
Azziz R, Waggoner WT, Ochoa T et al: Idiopathic hirsutism: An uncommon cause of hirsutism in Alabama. Fertil Steril 70: 274, 1998 |
|
Zawadzki JK, Dunaif A: Diagnostic criteria for polycystic ovary syndrome: Towards a rational approach. In Dunaif A, Givens JR, Haseltine F et al (eds): Polycystic Ovary Syndrome, p 377. Boston: Blackwell Scientific, 1992 |
|
Rao JK, Chihal HJ, Johnson CM: Primary polycystic ovary syndrome in a premenarchal girl. J Reprod Med 30: 361, 1985 |
|
el Tabbakh GH, Lotfy I, Azab I et al: Correlation of the ultrasonic appearance of the ovaries in polycystic ovarian disease and the clinical, hormonal, and laparoscopic findings. Am J Obstet Gynecol 154: 892, 1986 |
|
Orsini LF, Venturoli S, Lorusso R et al: Ultrasonic findings in polycystic ovarian disease. Fertil Steril 43: 709, 1985 |
|
Swanson M, Sauerbrei EE, Cooperberg PL: Medical implications of ultrasonically detected polycystic ovaries. J Clin Ultrasound 9: 219, 1981 |
|
Polson DW, Wadsworth J, Adams J et al: Polycystic ovaries: A common finding in normal women. Lancet 870, 1988 |
|
Stanhope R, Adams J, Brook CGD: Evolution of polycystic ovaries in a girl with delayed menarche. J Reprod Med 33: 482, 1988 |
|
Canales ES, Zarate A, Castelazo-Ayala L: Primary amenorrhea associated with polycystic ovaries. Obstet Gynecol 37: 205, 1971 |
|
Goldzieher JW, Green JA: The polycystic ovary: I. Clinical and histologic features. J Clin Endocrinol Metab 22: 325, 1962 |
|
Hosseinian AH, Kim MH, Rosenfield RL: Obesity and oligomenorrhea are associated with hyperandrogenism independent of hirsutism. J Clin Endocrinol Metab 42: 765, 1976 |
|
Lobo RA, Goebelsmann U, Horton R: Evidence for the importance of peripheral tissue events in the development of hirsutism in polycystic ovary syndrome. J Clin Endocrinol Metab 57: 393, 1983 |
|
Allen SE, Potter HD, Azziz R: Prevalence of hyperandrogenemia among nonhirsute oligo-ovulatory women. Fertil Steril 67: 569, 1997 |
|
Carmina E, Koyama T, Chang L et al: Does ethnicity influence the prevalence of adrenal hyperandrogenism and insulin resistance in polycystic ovary syndrome. Am J Obstet Gynecol 167: 1807, 1992 |
|
Mathur RS, Moody LO, Landgrebe S et al: Plasma androgens and sex hormone-binding globulin in the evaluation of hirsute females. Fertil Steril 35: 29, 1981 |
|
Vermeulen A, Stoica T, Verdonck L: The apparent free testosterone concentration, an index of androgenicity. J Clin Endocr 33: 759, 1971 |
|
Hoffman DI, Klove K, Lobo RA. The prevalence and significance of elevated dehydroepiandrosterone sulfate levels in anovulatory women. Fertil Steril 42:76, 1924 |
|
Moran C, Knochenhauer ES, Boots LR et al: Adrenal androgen excess in hyperandrogenism: Relationship of age and body mass. Fertil Steril 71: 671, 1999 |
|
Zacur HA, Foster GV: Hyperprolactinemia and polycystic ovarian syndrome. Semin Reprod Med 10: 236, 1992 |
|
Taymor ML, Barnard R: Luteinizing hormone excretion in the polycystic ovary syndrome. Fertil Steril 13: 501, 1962 |
|
Yen SSC, Vela P, Rankin J: Inappropriate secretion of follicle-stimulating hormone and luteinizing hormone in polycystic ovarian disease. J Clin Endocrinol Metab 30: 435, 1970 |
|
Kazer RR, Kessel B, Yen SSC: Circulating luteinizing hormone pulse frequency in women with polycystic ovary syndrome. J Clin Endocrinol Metab 65: 233, 1987 |
|
Dunaif A: Insulin resistance and the polycystic ovary syndrome: Mechanism and implications for pathogenesis. Endocr Rev 18: 774, 1997 |
|
Ciaraldi TP, Abrams L, Nikoulina S et al: Glucose transport in cultured human skeletal muscle cells: Regulation by insulin and glucose in nondiabetic and non-insulin-dependent diabetes mellitus subjects. J Clin Invest 96: 2820, 1995 |
|
Dunaif A, Xia J, Book C-B et al: Excessive insulin receptor phosphorylation in cultured fibroblasts and in skeletal muscle: A potential mechanism for insulin resistance in the polycystic ovary syndrome. J Clin Invest 96: 801, 1995 |
|
Marsden PJ, Murdoch A, Taylor R: Severe impairment of insulin action in adipocytes from amenorrheic subjects with polycystic ovary syndrome. Metab Clin Exp 43: 1536, 1994 |
|
Barbieri RL, Makris A, Randall RW et al: Insulin stimulates androgen accumulation in incubations of ovarian stroma obtained from women with hyperandrogenism. J Clin Endocrinol Metab 62: 904, 1986 |
|
Nestler JE, Jakubowicz DJ, de Vargas AF et al: Insulin stimulates testosterone biosynthesis by human thecal cells from women with polycystic ovary syndrome by activating its own receptor and using inositolglycan mediators as the signal transduction system. J Clin Endocrinol Metab 83: 2001, 1998 |
|
Dunaif A, Green G, Futterweit W et al: Suppression of hyperandrogenism does not improve peripheral or hepatic insulin resistance in the polycystic ovary syndrome. J Clin Endocrinol Metab 70: 699, 1990 |
|
Geffner ME, Kaplan SA, Bersch N et al: Persistence of insulin resistance in polycystic ovarian disease after inhibition of ovarian steroid secretion. Fertil Steril 45: 327, 1986 |
|
Nestler JE, Powers LP, Matt DW et al: A direct effect of hyperinsulinemia on serum sex-hormone-binding globulin levels in obese women with the polycystic ovary syndrome. J Clin Endocrinol Metab 72: 83, 1991 |
|
Legro RS, Driscoll D, Strauss JF 3rd et al: Evidence for a genetic basis for hyperandrogenemia in polycystic ovary syndrome. Proc Natl Acad Sci U S A 95: 14956, 1998 |
|
Kahsar-Miller M, Azziz R: The development of the polycystic ovary syndrome: Family history as a risk factor. Trends Endocrinol Metab 9: 55, 1998 |
|
Kahsar-Miller M, Azziz R: Heritability and the risk of developing androgen excess. J Steroid Biochem Mol Biol 69: 261, 1999 |
|
Urbanek, Waterworth DM, Bennett ST et al: Linkage and association of insulin gene VNTR regulatory polymorphism with polycystic ovary syndrome. Lancet 5: 986, 1997 |
|
Gharani N, Waterworth DM, Batty S et al: Association of the steroid synthesis gene CYP11a with polycystic ovary syndrome and hyperandrogenism. Hum Mol Genet 6: 397, 1997 |
|
Urbanek M, Legro RS, Driscoll DA et al: Thirty-seven candidate genes for polycystic ovary syndrome: Strongest evidence for linkage is with follistatin. Proc Natl Acad Sci USA 96: 8573, 1999 |
|
Carmina E: Prevalence of idiopathic hirsutism. Eur J Endocrinol 139: 421, 1998 |
|
Gompel A, Wright F, Kuttenn F et al: Contribution of plasma androstenedione to 5α-androstanediol glucuronide in women with idiopathic hirsutism. J Clin Endocrinol Metab 62: 441, 1986 |
|
Greep N, Hoopes M, Horton R: Androstanediol glucuronide plasma clearance and production rates in normal and hirsute women. J Clin Endocrinol Metab 62: 22, 1986 |
|
Kirschner MA, Samojlik E, Szmal E: Clinical usefulness of plasma androstanediol glucuronide measurements in women with idiopathic hirsutism. J Clin Endocrinol Metab 65: 597, 1987 |
|
Meldrum DR, Abraham G: Peripheral and ovarian venous concentrations of various steroid hormones in virilizing ovarian tumors. Obstet Gynecol 53: 36, 1979 |
|
Derksen J, Nagesser SK, Meinders AE et al: Identification of virilizing adrenal tumors in hirsute women. N Engl J Med 331: 968, 1994 |
|
Friedman CI, Schmidt GE, Kim MH et al: Serum testosterone concentrations in the evaluation of androgenproducing tumors. Am J Obstet Gynecol 153: 44, 1985 |
|
Yuen BH, Moon YS, Mincey EK et al: Adrenal and sex steroid hormone production by a virilizing adrenal adenoma and its diagnosis with computerized tomography. Am J Obstet Gynecol 145: 164, 1983 |
|
McKenna TJ, O'Connell Y, Cunningham S et al: Steroidogenesis in an estrogen-producing adrenal tumor in a young woman: Comparison with steroid profiles associated with cortisol- and androgen-producing tumors. J Clin Endocrinol Metab 70: 28, 1990 |
|
Kaplowitz PB, Mandell J: Virilizing adrenal tumor in a 3-year-old girl: Diagnosis by computed tomographic scan and ultrasound. Am J Dis Child 137: 406, 1983 |
|
Kamilaris TC, DeBold CR, Manolas KJ et al: Testosterone-secreting adrenal adenoma in a peripubertal girl. JAMA 258: 2558, 1987 |
|
Surrey ES, de Ziegler D, Gambone JC et al: Preoperative localization of androgen-secreting tumors: Clinical, endo-crinologic, and radiologic evaluation of ten patients. Am J Obstet Gynecol 58: 1313, 1988 |
|
Azziz R, Gay F: The treatment of hyperandrogenism with oral contraceptives. Semin Reprod Endocrinol 7: 246, 1989 |
|
de Oliveira RFC, Novaes LP, Lima MB et al: A new treatment for hirsutism. Ann Intern Med 83: 817, 1975 |
|
Pascale MM, Pugeat M, Roberts M et al: Androgen suppressive effect of GnRH agonist in ovarian hyperthecosis and virilizing tumours. Clin Endocrinol 41: 571, 1994 |
|
Steingold KA, Judd HL, Nieberg RK et al: Treatment of severe androgen excess due to ovarian hyperthecosis with a long-acting gonadotropin-releasing hormone agonist. Am J Obstet Gynecol 154: 1241, 1986 |
|
Martikainen H, Heikkinen J, Ruokonen A et al: Hormonal and clinical effects of ketoconazole in hirsute women. J Clin Endocrinol Metab 66: 987, 1988 |
|
Pepper G, Brenner SH, Gabrilove JL: Ketoconazole use in the treatment of ovarian hyperandrogenism. Fertil Steril 54: 438, 1990 |
|
Venturoli S, Fabbri R, Dal Prato L et al: Ketoconazole therapy for women with acne and/or hirsutism. J Clin Endocrinol Metab 71: 335, 1990 |
|
Vidal-Puig AJ, Munos-Torres M, Joder-Gimeno E et al: Ketoconazole therapy: Hormonal and clinical effects in non-tumoral hyperandrogenism. Eur J Endocrinol 130: 333, 1994 |
|
Velazquez EM, Mendoza S, Hamer T et al: Metformin therapy in polycystic ovary syndrome reduced hyperinsulinemia, insulin resistance, hyperandrogenemia, and systolic blood pressure, while facilitating normal menses and pregnancy. Metabolism 43: 647, 1994 |
|
Velasquez E, Acosta A, Mendoza SG: Menstrual cyclicity after metformin therapy in polycystic ovary syndrome. Obstet Gynecol 90: 392, 1997 |
|
Dunaif A, Scott D, Finegood D et al: The insulin-sensitizing agent troglitazone improves metabolic and reproductive abnormalities in the polycystic ovary syndrome. J Clin Endocrinol Metab 81: 3299, 1996 |
|
Ehrmann DA, Schneider DJ, Sobel BE et al: Troglitazone improves defects in insulin action, insulin secretion, ovarian steroidogenesis, and fibrinolysis in women with polycystic ovary syndrome. J Clin Endocrinol Metab 82: 2108, 1997 |
|
Nestler JE, Jakubowicz DJ, Evans WS et al: Effects of metformin on spontaneous and clomiphene-induced ovulation in the polycystic ovary syndrome. N Engl J Med 338: 1876, 1998 |
|
Meikle AW, Odell WD: Effect of short- and long-term dexamethasone on 3α-androstanediol glucuronide in hirsute women. Fertil Steril 46: 227, 1986 |
|
Abraham GE, Maroulis GB, Buster JE et al: Effect of dexamethasone on serum cortisol and androgen levels in hirsute patients. Obstet Gynecol 47: 395, 1976 |
|
Steinberger E, Rodriguez-Rigau LJ, Smith KD: The prognostic value of acute adrenal suppression and stimulation tests in hyperandrogenic women. Fertil Steril 37: 187, 1982 |
|
Azziz R, Black VY, Knochenhauser ES et al: Ovulation after glucocorticoid suppression of adrenal androgens in the polysystic ovary syndrome is not predicted by the basal dehydroepiandrosterone sulfate level. J Clin Endocrinol Metab 84: 946, 1999 |
|
Emans SJ, Grace E, Woods ER et al: Treatment with dexamethasone of androgen excess in adolescent patients. J Pediatr 112: 821, 1988 |
|
Rittmaster RS, Givner ML: Effect of daily and alternate day low dose prednisone on serum cortisol and adrenal androgens in hirsute women. J Clin Endocrinol Metab 67: 400, 1988 |
|
Spritzer P, Billaud L, Thalabard J-C et al: Cyproterone acetate versus hydrocortisone treatment in late-onset adrenal hyperplasia. J Clin Endocrinol Metab 70: 642, 1990 |
|
Slayden SM, Azziz R: The role of androgen excess in acne. In Azziz R, Nestler JE, Dewailly D (eds): Androgen Excess Disorders in Women, p 131. Philadelphia: Lippincott-Raven, 1997 |
|
Ober KP, Hennessy JF: Spironolactone therapy for hirsutism in hyperandrogenic woman. Ann Intern Med 89: 643, 1978 |
|
Boisselle A, Tremblay RR: New therapeutic approach to the hirsute patient. Fertil Steril 32: 276, 1979 |
|
Shapiro G, Evron S: A novel use of spironolactone: Treatment of hirsutism. J Clin Endocrinol Metab 51: 429, 1980 |
|
Cumming DC, Yang JC, Rebar RW et al: Treatment of hirsutism with spironolactone. JAMA 247: 1295, 1982 |
|
Lobo RA, Shoupe D, Serafini P et al: The effects of two doses of spironolactone on serum androgens and anagen hair in hirsute women. Fertil Steril 43: 200, 1985 |
|
Barth JH, Cherry CA, Wojnarowska F et al: Spironolactone is an effective and well tolerated systemic antiandrogen therapy for hirsute women. J Clin Endocrinol Metab 68: 966, 1989 |
|
Taylor A, Pita JC, Santen R: Spironolactone and endocrine dysfunction. Ann Intern Med 85: 630, 1976 |
|
Young RL, Goldzieher JW, Elkind-Hirsch K: The endocrine effects of spironolactone used as an antiandrogen. Fertil Steril 48: 223, 1987 |
|
Serafini P, Lobo RA: The effects of spironolactone on adrenal steroidogenesis in hirsute women. Fertil Steril 44: 595, 1985 |
|
Cusan L, Dupont A, Gomez JL et al: Comparison of flutamide and spirolactone in the treatment of hirsutism: A randomized controlled trial. Fertil Steril 61: 281, 1994 |
|
Erenus M, Gurbuz O, Durmusoglu F et al: Comparison of the efficacy of spironolactone verses flutamide in the treatment of hirsutism. Fertil Steril 61: 613, 1994 |
|
Marugo M, Bernasconi D, Meozzi M et al: The use of flutamide in the management of hirsutism. J Endocrinol Invest 17: 195, 1994 |
|
Belisle S, Love EJ: Clinical efficacy and safety of cyproterone acetate in severe hirsutism: Results of a multicentered Canadian study. Fertil Steril 46: 1015, 1986 |
|
Rittmaster RS: Finasteride. N Engl J Med 330: 120, 1994 |
|
Saitoh M, Uzuka M, Sakamoto M: Rate of hair growth. In Montagna W, Dobson RL (eds): Advances in Biology of Skin. Vol 9: Hair Growth, p 813. New York: Pergamon Press, 1967 |
|
Peereboom-Wynia JDR: Effect of various methods of depilation on density of hair growth in women with idiopathic hirsutism. Arch Derm Forsch 272: 164, 1972 |
|
Peereboom-Wynia JDR, Stolz E, van Joost T et al: A comparative study of the effects of electrical epilation of beard hairs in women with hirsutism by diathermy and by the blend method. Arch Dermatol Res 278: 84, 1985 |
|
Richards RN, Meharg GE: Electrolysis: Observations from 13 years and 140,000 hours of experience. J Am Acad Dermatol 33: 662, 1995 |
|
Wagner RF: Physical methods for the management of hirsutism. Cutis 45: 19, 1990 |
|
Kligman AM, Peters L Histologic changes of human hair follicles after electrolysis: A comparison of 2 methods. Cutis 34: 169, 1984 |
|
Farah L, Lazenby AJ, Boots LR, et al: Prevalence of the polycystic ovary syndrome among women seeking treatment from community electrologists. J Reprod Med 44: 870, 1999 |
|
Dierickx CC, Grossman MC, Farinelli WA et al: Permanent hair removal by normal mode ruby laser. Arch Dermatol 134: 837, 1998 |
|
Lin TYD, Manuskiatti W, Dierickx CC et al: Hair growth cycle affects hair follicle destruction by ruby laser pulses. J Invest Dermatol 111: 107, 1998 |
|
Adashi EY, Rock RA, Guzick D et al: Fertility following bilateral ovarian wedge resection: A critical analysis of 90 consecutive cases of the polycystic ovary syndrome. Fertil Steril 36: 320, 1981 |
|
Lunde O: Polycystic ovarian syndrome: A retrospective study of the therapeutic effect of ovarian wedge resection after unsuccessful treatment with clomiphene citrate. Ann Chir Gynaecol 71: 330, 1982 |
|
Campo S, Garcea N, Caruso A et al: Effect of celioscopic ovarian resection in patients with polycystic ovaries. Gynecol Obstet Invest 15: 213, 1983 |
|
Greenblatt E, Casper RF: Endocrine changes after laparoscopic ovarian cautery in polycystic ovarian syndrome. Am J Obstet Gynecol 156: 279, 1987 |
|
Sumioki H, Utsunomyiya T, Matsuoka K et al: The effect of laparoscopic multiple punch resection of the ovary on hypothalamo-pituitary axis in polycystic ovary syndrome. Fertil Steril 50: 567, 1988 |
|
Sakata M, Tasaka K, Kurachi H et al: Changes of bioactive luteinizing hormone after laparoscopic ovarian cautery in patients with polycystic ovarian syndrome. Fertil Steril 53: 610, 1990 |