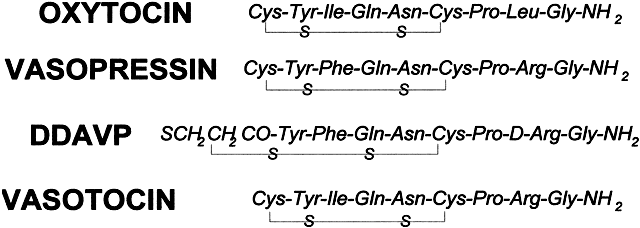Neurosecretory Peptides
Authors
INTRODUCTION
The concept that neurons would be capable of secretory function was first proposed by Scharrer in 1928 and was based on observations made in fish.1 These observations were later extended to mammals in experiments that demonstrated that extracts from certain neurons contained bioassayable vasopressin activity. Because the initial identification of vasopressin, a growing number of neuropeptides, some of which directly influence reproductive function, have been identified.
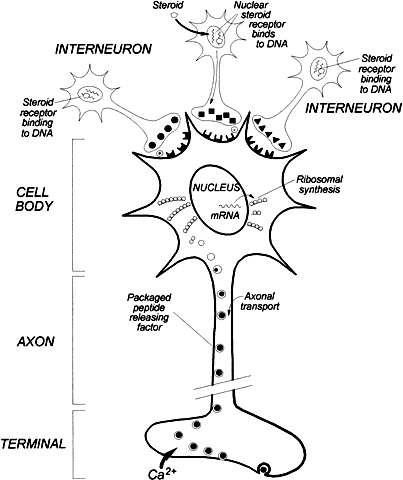
Unlike the classic neurotransmitters that are secreted after enzymatic conversions at the nerve terminal, peptide synthesis requires transcription of mRNA from DNA, subsequent translation on ribosomes, and posttranslational processing. This synthesis occurs in the neuronal cell body rather than at the nerve terminals. The initial product is usually a larger molecular weight precursor molecule that is then transported down the axon and further processed by enzymatic cleavage into the active peptide before secretion (Fig. 1). From a phylogenetic perspective, the widespread distribution of brain peptides probably reflects evolutionary conservation of vital regulatory functions imposed by these peptides. For example, a vasopressin-like peptide occurs in the freshwater snail, in which it functions as a neurotransmitter. The importance of neuropeptides is further supported by the presence in higher animals of multiple subtypes of many of the neuropeptides and their receptors, with each having different physiologic roles.2 Although it is likely that these multiple subtypes of neuropeptides and their receptors arose through genome duplication events, their retention and diversification into separate physiologic roles convey their importance and suggest that additional subtypes remain to be discovered.2
On the basis of evidence from many biologic systems, information transfer from the neurosecretory neuron can modulate biologic activity by four principal means: autocrine, paracrine, neuroendocrine, and neurotransmission. The most direct form of neurohumoral signaling is the autocrine mode, in which the release of a substance from the cell acts to autoregulate activity within that same cell. For example, gonadotropin-releasing hormone (GnRH) inhibits its own gene transcription in hypothalamic neurons.3 The paracrine mode of communication occurs when secretion of a substance controls activity on adjacent cells. This mode of regulation functions in the pituitary. When gonadotrophs (i.e. luteinizing hormone [LH]- and follicle stimulating hormone [FSH]-secreting cells) are exposed to GnRH, a paracrine product is secreted that allows adjacent lactotrophs to respond with prolactin release.4 A third form of informational transfer, the neuroendocrine mode, is characterized by a release of a neurohumoral product such as oxytocin into the general circulation to regulate distant tissues that have oxytocin receptors, such as the myoepithelial cells of the breast, causing these cells to eject milk.
Neurotransmission can be conceptualized as a special form of paracrine secretion in which there is cell-to-cell communication through typical synaptic contacts. The classic neurotransmitters include acetylcholine, norepinephrine, epinephrine, dopamine, histamine, serotonin, and γ-aminobutyric acid (GABA). In general, neurons that contain these transmitters are activated by an action potential above a threshold leading to membrane depolarization presynaptically. Ion fluxes then mobilize release of a small amount of the transmitter substance, which diffuses across the synaptic gap and causes excitatory or inhibitory postsynaptic potentials. The transmitter is then removed by enzymatic degradation or a reuptake mechanism. It is unclear whether all brain peptides are capable of synaptic transmission; however, it is estimated that the classic neurotransmitters only account for 40% of the synaptic contacts in the central nervous system.5 Peptide-secreting hypothalamic neurons make synaptic contact with each other (Fig. 1), and it is likely that neuropeptides play an important role in the modulation of synaptic transmission.
ISOLATION AND CHARACTERIZATION OF NEUROPEPTIDES
The development of a bioassay system in which the biologic effect of an unknown substance can be accurately and reproducibly measured is the first step in the characterization of new peptides. With conventional biochemistry techniques, tissue extracts are subjected to chemical manipulations such as differential solvent extraction and chromatographic procedures to purify the unknown factor. After each purification step, the biologic activity of the fractions is determined, and the fractions of interest are further purified to the final active material. Using this approach, Schally and Guillemin's laboratories were able to isolate gonadotropinreleasing factor, a process requiring more than 500,000 hypothalami.6,7 Through similar meticulous extraction procedures, Guillemin's group also isolated somatostatin.8
Recombinant DNA techniques have been rapidly applied to this area and have been combined with conventional biochemical approaches. One series of isolation strategies is illustrated in Figure 2. After amino acid sequencing, antibodies against the new peptide can be raised, and immunocytochemical staining of tissue sections can provide an overall view of the localization and distribution of the new peptide. These techniques are responsible for the rapid progress in the discovery of new peptides. With immunocytochemistry, many peptides that were originally isolated in the gastrointestinal tract, such as vasoactive intestinal polypeptide (VIP), have also been localized to the central nervous system. However, neuropeptides such as oxytocin and β-endorphin have been identified in nonneural tissue such as the ovary, testes, and adrenal gland (Table 1).9
TABLE 1. Classification of Neuroactive Peptides
Secretin-glucagon family | Tachykinin peptides |
Glucagon | Substance P |
Gastric-inhibitory peptide | Bombesin |
Vasoactive intestinal polypeptide | Neurotensin |
Secretin | Neurokinin A |
Histidine-isoleucine peptide | Neurokinin B |
Hypothalamic-releasing peptides | Gastrin-type peptides |
Thyrotropin-releasing hormone | Gastrin |
Gonadotropin-releasing hormone | Cholecystokinin |
Somatostatin | Pancreatic family peptides |
Corticotropin-releasing hormone | Neuropeptide Y |
Growth hormone-releasing factor | Peptide YY |
Neurohypophyseal peptides | Pancreatic peptide |
Vasopressin | Others |
Oxytocin | Angiotensin II |
Oxytocin-vasotocin–like peptide | Bradykinin |
Opiate peptides | Atrial-natriuretic factor |
ACTH | Galanin |
β-Endorphin | |
α-Melanocyte–stimulating hormone | |
Met-enkephalin | |
Leu-enkephalin | |
Dynorphin | |
Nociceptin/orphanin FQ |
Adapted from Cooper JR, Bloom FE, Roth RH: The biochemical basis of neuropharmacology. In Cooper JR, Bloom FE, Roth RH (eds): Neuroactive Peptides, p 295. New York, Oxford University Press, 1982.
TRANSGENIC TECHNOLOGY AND ITS APPLICATION TO THE STUDY OF NEUROSECRETORY PEPTIDES
An important advance in our understanding of the physiology of neurosecretory peptides occurred with the development of transgenic technology, or the ability to precisely alter the genetic makeup of an experimental animal. These techniques result in the generation of animals, usually mice, with genetic alterations that include the introduction of human genes, an increase in the expression of specific single genes, and the complete lack of expression of specific genes, or gene knockouts. A detailed analysis of these animals can yield important information about the physiology of a neuropeptide that cannot be obtained in any other way.
The knockout mouse,10 in which the animal is completely normal except for the precise deletion of a single gene, is the most widely used of these transgenic techniques. With the exception of naturally occurring knockouts, such as Kallmann's syndrome, that are found for a variety of genes in a number of species, experimentally generated knockouts are only produced in mice. These mice are generated by a complex series of experiments that begins with the cloning of the gene of interest (Fig. 3). This information is used to generate a mutant gene that is inactive or irrelevant. The mutant gene is exchanged for the normal gene through a process known as homologous recombination, a normal cellular process allowing for the exchange of genetic information between chromosomes. This exchange is carried out in highly specialized cells known as embryonic stem cells, which have the unique property that they can be incorporated into mouse embryos. The mutant cells are then incorporated into these first-generation mice essentially at random. To generate the full knockout mouse, a large number of these mice must be screened to find one in which the mutant cells were correctly incorporated into germ cells. When this mouse reproduces, its offspring will carry the mutant gene. Further breeding can be performed to generate mice in which both copies of the gene are mutant, creating the full knockout mouse. This technology has been applied to a large number of neurosecretory peptides, as described in the following sections.
It would initially appear that the development of a knockout mouse for a neuropeptide would provide specific information about the physiology of the peptide: what does this neuropeptide really do? Fortunately, this is often the case, but in many instances, the knockout mouse yields surprising and unanticipated information. For example, a knockout mouse may be completely normal, indicating that the peptide of interest does not subserve proposed functions, at least not in the mouse, or more likely that there are other peptides or systems that compensate for its loss. Another common result is that the knockout mouse is nonviable. When this occurs in the embryonic period, such findings support a critical role for the peptide in development but may yield little other useful physiologic information. Although the knockout mouse often provides extremely useful information about the physiologic roles of a neurosecretory peptide, there are also significant limitations to its use.
The ubiquitous distribution of neurosecretory peptides has raised many questions regarding their evolution, mechanism of action, function, and ultimate physiologic roles. In this chapter, neuropeptides are discussed with special emphasis on hypothalamic peptides and peptide derivatives of the opioidergic system and on their recognized or putative roles in reproduction.
NEUROHYPOPHYSIAL HORMONES
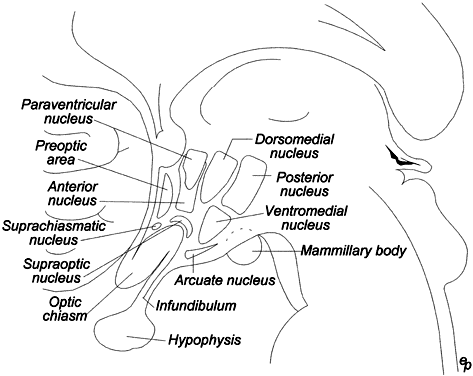
There is a common structural organization of hypothalamic peptide neurons. Most of the neuronal cell bodies are concentrated in the arcuate, paraventricular, and periventricular nuclei (Fig. 1). With more refined immunocytochemical techniques, it is evident that more than one neuropeptide can be synthesized by a single group of neurons. For example, neurons that secrete corticotropin-releasing hormone (CRH) may also contain vasopressin or oxytocin.
The neurohypophysial hormones oxytocin and vasopressin are synthesized in the paraventricular and supraoptic nuclei of the hypothalamus. Each peptide is 9 amino acids long and contains an internal disulfide bridge at the 1 through 6 positions (Fig. 4). Their similarity to neurohypophysial hormones such as vasotocin in other species suggests that they evolved from a common precursor by a single point mutation. Both peptides are synthesized as part of a larger prohormone; oxytocin is associated with an estrogen-stimulated neurophysin, and vasopressin is associated with a nicotine-stimulated neurophysin.11 Immunocytochemical studies of individual neurons have shown that they contain oxytocin, vasopressin, and their neurophysins.12
Clinical studies have shown that the vasopressin secretion is tightly controlled and governed primarily by plasma osmolarity and circulating blood volume. For example, serum osmolarity is kept constant within strict tolerances (+ 1.8%).13 Vasopressin secretion can also be stimulated by administration of angiotensin II.
Clearly established roles for oxytocin that is released into the circulation are milk ejection from the breast and stimulation of uterine contractions during the second stage of parturition. Controversy still exists concerning the role of oxytocin in labor in humans, because no appreciably higher levels are detected during early labor14 and because an oxytocin knockout mice deliver normally.15 Perhaps a more important factor is that the activity of circulating oxytocin is amplified by the increase in uterine-myometrial oxytocin receptors.16
Oxytocin and vasopressin have also been shown to affect the adrenocorticotropic hormone (ACTH)-adrenal axis. Vasopressin is capable of stimulating ACTH release and is considered to have weak corticotropin-releasing activity. After the isolation of ovine CRH, it was shown under experimental conditions that vasopressin acted synergistically with CRH to release fivefold more ACTH than CRH alone in humans.17 Alternatively, oxytocin has been reported to have effects opposite that of vasopressin, causing suppression of ACTH secretion.18 These potential physiologic interactions are reinforced by the findings of Bergland and Page that the neurohypophysis is vascularly interconnected with the anterior pituitary.19
Many other diverse actions of these nonapeptides have been hypothesized. Preliminary evidence in rats suggests that these two peptides are differentially released in response to different types of stress.20 Moreover, an increase in oxytocin activity has been associated with a decrease in GnRH secretion. Both nonapeptides have been postulated to have effects on cognitive functions. Vasopressin or its synthetic analogs has been shown to enhance memory in normal and cognitive-impaired adults.21 Oxytocin appears to have the opposite effect, impairing memory functions,22 but in addition oxytocin can induce maternal behavior in rats.23
A knockout mouse for oxytocin has been developed. Oxytocin-deficient mice cannot lactate and display mild behavioral changes. Surprisingly, these mice deliver normally and do not display problems in mating or maternal behavior. This finding supports a stronger role for prostaglandins in uterine contractions of parturition, and mice deficient in cyclooxygenase-1 (COX-1), the enzyme that synthesizes prostaglandins, have delayed labor and subsequent neonatal death. Mice deficient in oxytocin and COX-1 do deliver normally, an effect attributed to the luteotrophic action of oxytocin. A "natural" knockout model for vasopressin has been known for many years: the Brattleboro rat.24 Although these rats display the expected diabetes insipidus of vasopressin deficiency, they also have several other central nervous system (CNS) defects, affecting behavior and cognition.25
HYPOTHALAMIC-RELEASING FACTORS
Thyrotropin-Releasing Hormone
The first hypothalamic-releasing factor isolated was the tripeptide thyrotropin-releasing hormone (TRH), which was discovered independently by Schally and Guillemin's group.7,26 Like other neuropeptides, TRH is derived from a much larger 30-kd precursor (Fig. 5) and specifically stimulates release of thyroid-stimulating hormone (TSH). TRH also has prolactin-releasing properties, because lactotropes have receptors for TRH. TRH may play other roles besides its physiologic role in TSH release. It has been localized by immunocytochemical techniques to the cerebral cortex, spinal cord, paraventricular nucleus, pineal gland, neurohypophysis, and ventral horn motor cells.27 A diagrammatic view of the circumventricular hypothalamic nuclei is shown in Figure 6. Neuropharmacologic studies implicate TRH as a neuromodulator with general stimulant activity. Experimentally observed properties of TRH are summarized in Table 2.28
TABLE 2. Central Nervous System---Mediated Actions of Thyrotropin-Releasing Hormone
Hypothermia
Reversal of spinal shock
Increased spontaneous motor activity
Alteration in sleep patterns
Anorexia
Inhibition of avoidance behavior
Reversal of depression
Neuromodulator of classic transmitter action
Releases norepinephrine and dopamine from synaptosomal preparations
Enhances disappearance of norepinephrine from nerve terminals
Potentiates excitatory actions of acetylcholine on cerebral cortical neurons
Vale W, Rivier C, Brown M: Regulatory peptides of the hypothalamus. Ann Rev Physiol 39:473, 1977.
|
Transgenic mice deficient in TRH have been generated.29 These mice develop normally and are fertile. The affected mice are hypothyroid with elevated TSH levels (i.e. tertiary hypothyroidism). The mice also display hyperglycemia and abnormal insulin secretion.29
Because of its ability to reverse spinal shock in animals, TRH has been used experimentally in patients with spinal cord trauma as an adjunctive treatment with promising results. Because of a short half-life, additional clinical applications will require development of long-acting analogs.
Gonadotropin-Releasing Hormone
GnRH is a decapeptide that was also isolated and characterized by Schally and Guillemin's laboratories in 1971 from porcine and ovine hypothalami.6,7 The structure of this peptide is identical in all mammals, including humans (Fig. 5). GnRH has been localized primarily to the hypothalamus in the interstitial nucleus (medial preoptic area) and has its highest concentrations in the arcuate nucleus adjacent to the median eminence30 (Fig. 6). Small amounts of GnRH are also found in the circumventricular organs of the brain. There are an estimated 1,500 GnRH neurons in the hypothalamus.
GnRH, like many other neuropeptides, is synthesized as part of a much larger precursor peptide containing 92 amino acids.31 GnRH is cleaved from the precursor, leaving a 56-amino acid peptide known as GnRH-associated peptide (GAP).32 GAP has been shown to have potent activity to inhibit the release of prolactin. The placenta, which also secretes GnRH, forms a different precursor protein through the use of a different promoter sequence.33
Because of the importance of GnRH in the control of reproduction, intensive efforts have been made to determine its physiologic role. When administered to humans, GnRH stimulates a rapid and large release of LH and a smaller release of FSH. In the absence of GnRH, as in patients with Kallmann's syndrome, there is virtually no secretion of LH and FSH, emphasizing the importance of GnRH in the reproductive axis. As with other knockouts, the unexpected finding of anosmia in these patients led to the discovery that GnRH neurons arise in the olfactory placode and migrate to the hypothalamus.34
The half-life of GnRH in the peripheral circulation is 2 to 4 minutes.35 Because GnRH is rapidly degraded by peptidases, analogs have been synthesized that have much longer half-lives (Fig. 5). By trial and error testing of thousands of GnRH analogs, it was discovered that substitution of amino acids at the 2 or 3 position results in analogs with antagonistic properties, whereas modifications at the 6 or 10 position result in analogs with agonist action.
Two unique functional properties of GnRH have permitted its widespread clinical application. When GnRH is administered as a continuous infusion, pituitary LH response is initially brisk but is followed by a decrease in LH secretion and decrease in GnRH receptors (i.e. downregulation). Agonistic analogs of GnRH promote this downregulation phenomenon and can be used to induce "medical castration" by shutting down the pituitary-gonadal axis. Several GnRH analogs are available clinically and are widely used for a number of diseases, including endometriosis and uterine leiomyomas. If GnRH is administered in a pulsatile fashion at 60- to 90-minute frequencies, LH receptors and LH response increase. Continuation of this mode of administration for prolonged periods results in stimulation of a normal menstrual cycle36 or normal testosterone production and spermatogenesis in patients with a deficiency or disorder of GnRH secretion. A summary of the clinical applications for GnRH and its analogs is provided in Table 3.
TABLE 3. Clinical Application of Gonadotropin-Releasing Hormone and Its Analogs
Activation of pituitary-gonadal axis
Dynamic testing of pituitary
Ovulation induction
Induction of spermatogenesis
Cryptorchidism
Downregulation of pituitary-gonadal axis
Contraception
Gonadal steroid-dependent tumors
Prostate cancer
Breast cancer
Endometrial cancer
Endometriosis
Polycystic ovarian disease
Uterine leiomyoma
Premenstrual syndrome
Precocious puberty
Controlled ovarian hyperstimulation
Besides its role in the control of reproduction, GnRH also has intriguing behavioral effects. When administered to ovariectomized, estrogen-primed female rats intracerebrally, GnRH induces lordotic posturing and mating behavior.37 In contrast, administration of GnRH antibodies has suppressed sexual behavior.38 This effect is a unique property of this peptide. From a phylogenetic perspective, these behavioral effects appear similar to a peptide that has been isolated from yeast, the α-mating factor. The α-mating factor has sequence homology with GnRH and can stimulate LH release from gonadotrophs.39
The role of GnRH as a true neurotransmitter is not established. GnRH has been shown to elicit a slow excitatory synaptic potential in frog sympathetic ganglia.40 Whether GnRH plays a modulatory role on other neurotransmitter systems remains to be determined.
Somatostatin
During the screening of multiple fractions from hypothalamic extracts, fractions with the ability to inhibit secretion of growth hormone (GH) from anterior pituitary cells were isolated. One factor subsequently isolated and characterized by Guillemin's group was a 14-amino acid peptide named somatostatin (Fig. 5).8 This peptide is derived from a 210-amino acid precursor.
Clinical studies of this peptide demonstrated that somatostatin inhibited GH, TSH, prolactin, glucagon, and insulin secretion in humans.41 Subsequently, somatostatin was found to be widely distributed in the gastrointestinal tract, the delta cells of the pancreas, and the thyroid-parafollicular cells. In the central nervous system, somatostatin has been localized to the hypothalamus, limbic system, septum, hippocampus, cortex, and medulla.42 The secretion of hypothalamic somatostatin is episodic and probably plays a role in the regulation of episodic GH secretion.43 Because somatostatin is localized adjacent to other hormone-secretory cells, control of secretion can be affected by paracrine action. In the case of the thyroid, parafollicular cells that contain calcitonin and somatostatin regulation may occur through autocrine action.
In general, somatostatin injected intraventricularly in animals acts to decrease spontaneous motor activity and displays a sedative effect. The half-life of somatostatin in the peripheral circulation is only 2 to 4 minutes.40
Mice overexpressing somatostatin have been generated.44 These mice displayed motor hyperactivity, especially in females, supporting a role for somatostatin as a neurotrophic factor. Mice deficient in somatostatin receptor type 2 do not respond to the negative feedback of GH45 and have high gastric acid secretion.46
Corticotropin-Releasing Hormone
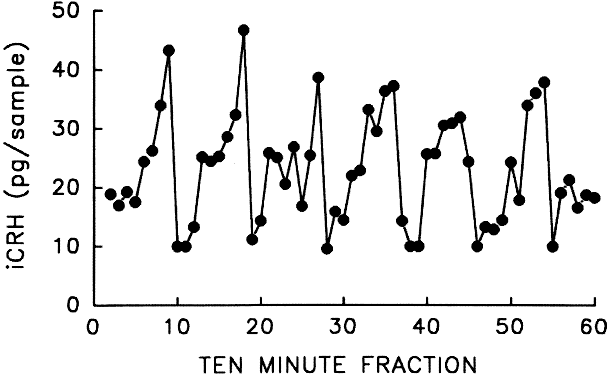
CRH was the first hypothalamic-releasing hormone to be recognized. However, it was not until 1981 that Vale and his colleagues first reported the isolation and characterization of ovine CRH as a 41-amino acid peptide.47 Through genetic engineering techniques, the structure of human CRH was determined to be a 41-amino acid peptide that differed from the ovine hormone in 7 positions and was derived from a 196-amino acid precursor.48 Using immunocytochemical techniques, CRH was found to be highly concentrated in the hypothalamus (i.e. paraventricular nucleus and parvocellular neurons) and pituitary stalk. Immunoreactive CRH was found in the posterior pituitary, thalamus, cerebral cortex, cerebellum, pons, medulla oblongata, and spinal cord.49 CRH neurons are also capable of synthesizing and storing oxytocin and vasopressin. There is evidence that CRH, like GnRH, is released in regular episodic pulses from a pulse generator intrinsic to the hypothalamus (Fig. 7).50 The physiologic significance of the pulsatile nature of CRH remains unknown.
In initial studies, intracerebroventricular injection of CRH in rats activated the sympathetic nervous system, which increased plasma levels of epinephrine, norepinephrine, glucose, glucagon, and insulin.51 These responses suggest that CRH is a principal mediator of ACTH/cortisol secretion during stress in addition to its ability to activate the sympathetic nervous system. CRH may also play a role in depression, because elevated concentrations of CRH-like peptides have been reported in the cerebrospinal fluid of depressed patients.52 CRH also can suppress secretion of GnRH-LH release in vitro53 and in vivo.54,55 This effect can be reversed by a CRH antagonist53 or naloxone.56 This effect remains controversial, because other studies have found no effect of CRH on LH pulsatility.57
Outside the central nervous system, CRH has been localized to pancreas, stomach, duodenum, liver, lung, placenta, and adrenal gland. The molecular weight of immunoreactive CRH from these tissues suggests that it is similar to hypothalamic CRH.49 The physiologic role of CRH in these tissues is unknown.
In clinical studies, the half-life of human CRH is 5 minutes when injected into the circulation.58 When injected into humans, the ACTH-releasing effects of CRH are synergistically potentiated by vasopressin.17 These responses suggest that ACTH release may be modulated by peptide-peptide interaction.
Transgenic technology has been used to generate mice that overexpress CRH and mice deficient in CRH and its receptor. Mice overexpressing CRH show elevated ACTH and glucocorticoid and display the signs of Cushing's syndrome, including female infertility.59 The CRH knockout mice are viable, reproduce, and have normal longevity despite severe glucocorticoid deficiency.60 Vasopressin and oxytocin expression were not increased. Knockout mice have a decreased glucocorticoid response to stress, with males much more severely affected. The most interesting finding in CRH-deficient mice was the effect of CRH deficiency on the offspring; lack of lung maturation led to neonatal death within 12 hours of life. The mice could be rescued by the administration of glucocorticoid to the maternal drinking water.61 This is reminiscent of the beneficial effect of glucocorticoid on lung maturation to women in preterm labor. Mice deficient in CRH receptor type 1 also show glucocorticoid deficiency but display increased exploratory activity and decreased anxiety, suggesting a role for CRH in stress and the anxiety response.
CRH administration is limited to research and diagnostic applications. CRH may be useful in the differentiation between pituitary and nonpituitary Cushing's syndrome.62 The therapeutic potential for CRH lies in its involvement in memory, obesity, and Alzheimer's disease.63 Additional diagnostic and therapeutic applications await development of long-acting analogs.
A new member of the CRH family, urocortin, was cloned from rat brain.64 It was later described in humans, sharing 95% identity with the rat molecule.65 This 40-amino acid peptide shares 45% identity with CRH and stimulates ACTH release in vitro and in vivo.66 Urocortin has greater ability to bind and activate type 2 CRH receptors than CRH, suggesting that it may be the endogenous ligand for the type 2 CRH receptor.64 When administered intravenously to rats, urocortin produces a significant reduction in blood pressure,67 similar to the activity of urotensin, a closely related, vasoactive hormone in fish.
Growth Hormone-Releasing Hormone
The most recent hypothalamic-releasing factor to be characterized, sequenced, and synthesized is growth hormone-releasing hormone (GHRH). Because many brain peptides have been localized outside the central nervous system, it was not surprising that GHRH was isolated from two patients with pancreatic tumors that induced acromegaly. From these tumors, two peptides (40 and 44 amino acids long) were isolated that possessed GH-releasing properties.68,69 Both have been shown to be equally potent and specific for GH release that is inhibited by somatostatin.70,71 Subsequent reports indicate that both peptides are derived from a 108-amino acid precursor and are localized predominantly to the arcuate nuclei.
Studies in patients with acromegaly have demonstrated no significant different responses to GHRH,72 whereas in patients with hyperprolactinemia and galactorrhea responses to GHRH are blunted.73 The discovery of this peptide has obvious clinical applications. Studies that use GHRH for the treatment of GH-deficient children are already in progress.74 As with other hypothalamic peptides, long-acting analogs of GHRH may be an alternative to the use of synthetic human GH in the treatment for this condition. There are now several nonpeptide analogues of GHRH, such as MK-677, in clinical development for the stimulation of the GH axis.75
Neuropeptide Y, Galanin, Substance P, and Angiotensin II
A number of other neuropeptides that are localized to the circumventricular nuclei have been shown to modulate release of prolactin, GH, and GnRH. To review the interaction of these peptides is beyond the scope of this chapter; however, four of the better-known neuropeptides are discussed.
Neuropeptide Y (NPY) is the most abundant neuropeptide in the brain. It is one of several gastrointestinal peptides that are also localized in the CNS. NPY shares many similarities with the pancreatic family of peptides, showing a high degree of homology to peptide YY and pancreatic peptide (Table 1). There is considerable evidence that NPY plays a significant role in the regulation of appetite, body weight, and reproduction. In this capacity, NPY may be an important factor in the complex interplay between nutrition and reproduction.
NPY is a 36-amino acid peptide and is the only member of the pancreatic peptide family with localization in the CNS. NPY has a CNS distribution that is similar to somatostatin and is localized to the many hypothalamic nuclei, including the arcuate nucleus, median eminence, amygdala, septum, cortex, and the hippocampus.76,77 NPY receptors are also widely distributed in the CNS, especially the hypothalamus and median eminence.77 Pharmacologic and molecular biologic studies have confirmed the presence of at least four subtypes of NPY receptor.78
Considerable evidence supports a major role for NPY in the control of reproductive function. Initial studies demonstrated that NPY stimulated LH release from intact rats but inhibited LH release from ovariectomized animals. The stimulatory effect of NPY on LH release was later shown to be mediated by an increase in the hypothalamic release of GnRH, in estrogen-treated animals or tissues. NPY is secreted in an episodic manner that corresponds to GnRH pulsatility. There is evidence that NPY is involved in the pulsatile secretion of GnRH and in the burst of GnRH seen at the time of the LH surge.79 There is also evidence that NPY influences LH secretion at the pituitary level. NPY has a pronounced inhibitory effect on sexual behavior.80
NPY is recognized as the most potent appetite-stimulating substance in the brain. Injection of NPY into the brains of satiated rats consistently stimulates ongoing feeding behavior and the development of obesity. Evidence that NPY is an endogenous appetite stimulator came from the neutralization of NPY by administration of NPY antibody, which eliminated the normal feeding pattern of rats. It is likely that NPY interacts with other neurosecretory peptides, such as galanin and β-endorphin, to regulate feeding behavior.79
It was expected that a mouse deficient in NPY would display alterations in feeding behavior and body weight, but this is not the case. NPY-deficient mice maintain a normal body weight and have normal reproductive function.81 In addition, the response of these animals to the newly discovered obesity hormone, leptin, is normal. Surprisingly, NPY-deficient mice have an increased propensity for seizure activity.81 It is likely that other neuropeptides, such as galanin, play a compensatory role in feeding and reproductive function in these animals.82
Galanin is an important neurosecretory peptide. Originally isolated in 1983 in extracts of porcine intestine, galanin is composed of 29 amino acids in all species except humans, in whom it is 30 amino acids long.83 The first 14 amino acids, which are associated with biologic activity, are fully conserved across 13 species in which galanin has been characterized.83 The name galanin was derived from the first and last amino acids of the sequence: glycine and alanine. Galanin is recognized as a neurosecretory peptide, with wide distribution in the central nervous system. Most important is its secretion from the hypothalamus, which also has a high concentration of galanin receptors. There is evidence that hypothalamic galanin has physiologic roles involving feeding, cognition, gastrointestinal function, and regulation of hypothalamic and pituitary hormone secretion.
The galanin gene was cloned in 1987 from rat pituitary and hypothalamus.84 Analysis of the galanin peptide suggests that it is unrelated to any other known families of neuropeptides, making it unique among neurosecretory peptides. Like all other neurosecretory peptides, the galanin receptor (GALR) is a member of the G protein-coupled receptors with seven transmembrane domains.85 Galanin receptors are found predominantly in the brain in three regions: cerebral cortex, amygdala, and hypothalamus. The first GALR was cloned from a human melanoma in 1994.86 There are three known GAL receptors, designated GALR1, GALR2, and GALR3, which differ considerably in primary structure and distribution.85 Based on the effects of different galanin agonists and antagonists, it is likely that there are other galanin receptors.83
Galanin has a well-recognized role in reproductive function. One of the first indications of this action was the demonstration that galanin expression is markedly stimulated by estrogen.87 There is evidence that galanin is co-released with GnRH and that it modulates LH secretion from the pituitary. In this regard, galanin secretion is modulated by estrogen and testosterone levels and varies with the estrus cycle, peaking in proestrus with the LH surge.88,89 Galanin stimulates the release of GnRH from the hypothalamus in a dose-dependent manner.90 Based on these findings, it has been proposed that galanin expression may modulate the pulsatility of GnRH release.88 The pituitary gland is another important site of galanin secretion, with expression stimulated by estrogen.84 Galanin was also shown to stimulate prolactin release and proliferation of prolactin-secreting cells (i.e. lactotrophs).91
Galanin, in conjunction with NPY and other hypothalamic neuropeptides, has a well-characterized role in the regulation of feeding and body weight.92,93 Hypothalamic galanin secretion is modulated by circulating hormones, such as insulin and leptin, and nutrient ingestion. There is evidence that galanin secretion from one particular site within the hypothalamus, the anterior paraventricular nucleus, mediates these effects.94 Galanin responds to these metabolic signals by regulating energy balance through its effects on behavior, hormone secretion, and metabolism.95 Galanin has orexigenic action, or stimulation of eating behavior. In this regard, galanin may act through β-endorphin, which also stimulates appetite.96 Galanin is specifically linked to ingestion of fats, in contrast to NPY, which stimulates the ingestion of fats and carbohydrates.95 Galanin not only stimulates feeding behavior, but also has effects on metabolism, acting to reduce energy expenditure. These effects tend to increase body weight, especially of adipose tissue.93
A knockout mouse for the galanin gene has been developed and characterized.97 The mice develop normally and can reproduce, indicating that galanin does not play critical roles in these physiologic processes. The mice have a normal body weight compared with wild-type littermates. The mice do demonstrate a significant reduction in pituitary prolactin content, supporting an important role of galanin in the development or maintenance of the lactotroph cell. Mammary development was also delayed. Affected mothers cannot lactate, so that the pups die shortly after birth if not transferred to a foster mouse.91 The stimulatory response of the lactotroph cell to estrogen is also abolished. The mice also demonstrate several neurologic abnormalities, including important changes in the responses of sensory neurons to injury and pain.97
NEUROSECRETORY PEPTIDES AND THE LINK BETWEEN NUTRITION AND REPRODUCTION.
Two neurosecretory peptides, NPY and galanin, play major physiologic roles in nutrition and reproduction, suggesting that these peptides may be involved in the reproductive changes seen in disorders such as anorexia nervosa and the chronic anovulation of obesity. The most important part of this puzzle was the discovery of the obesity hormone leptin.98 This important discovery provided the missing piece of the puzzle: how does the hypothalamus know how much fat tissue is present in the organism? Produced by fat tissue, circulating levels of leptin correlate directly with the amount of adipose tissue present.99 Receptors for leptin are located in the hypothalamus, where leptin acts to regulate energy expenditure and body weight. In this model (the "lipostat" hypothesis of body weight regulation), leptin serves as the feedback signal to the hypothalamus that allows the animal to maintain body weight in a narrow range, despite daily fluctuations in food consumption and energy expenditure.100
It is likely that the primary mechanism by which leptin modulates appetite and energy balance is through its effects on NPY, galanin, and possibly other neurosecretory peptides, such as melanin-concentrating hormone (MCH), opiates, and CRH.100,101 Leptin receptor is coexpressed in NPY neurons,102 where leptin acts to decrease NPY mRNA and neural signaling.103,104 Leptin also inhibits the expression of galanin in the hypothalamus.101 Although not yet proved, it is likely that these effects of leptin on NPY, galanin, and other neurosecretory peptides modulate the reproductive axis.90 The requirement for both NPY and galanin is demonstrated in studies showing markedly decreased GnRH pulsatility when the activity of both neuropeptides is inhibited but only partial effect when only one is blocked.105 This study helps to explain why mice deficient in either neuropeptide have normal weight. Taken together, the interaction of leptin, NPY, galanin, and other neuropeptides is beginning to shed light on the long-recognized link between nutritional status and reproductive function.
Substance P was one of the first nonhypothalamic-releasing hormones shown to affect pituitary function. It is related to the tachykinin family of neuropeptides and has been localized to the brain stem, amygdala, cortex, medulla, and hippocampus.106 Receptors for substance P-like peptides are found in brain, pituitary lactotrophs (i.e. prolactin-releasing cells), and gonadotrophs.107 In animal studies, administration of substance P to rats stimulates prolactin secretion and inhibits GH release.108 Mice deficient in substance P or its receptor are viable and fertile but do show altered responses to pain.109
Although angiotensin II is well known for its regulation of salt-water balance, this octapeptide is also found in the paraventricular and supraoptic nuclei. It is often colocalized with another osmoregulating hormone, vasopressin. With regard to its modulatory role on pituitary function, angiotensin II has been shown to stimulate LH release and inhibit prolactin release.110 Mice deficient in angiotensin II are fertile and show the predicted alterations in salt balance and blood pressure.111 Male mice lacking angiotensin-converting enzyme show altered sperm mobility and decreased fertility.112
OPIOID PEPTIDES
For many years, the existence of endogenous opiate compounds similar to morphine was postulated. However, it was not until 1975 that two endogenous opiate-like substances, methionine enkephalin and leucine enkephalin, were discovered by Hughes and coworkers.113 Subsequently, a much larger peptide with potent opiate activity was isolated by Li and was named β-endorphin.114 A comparison of the structure of this peptide indicated that β-endorphin was identical with the carboxyl-terminal sequence of β-lipotropin (β-LPH), a previously isolated protein that did not appear to have a specific function.115
These observations led to a series of elegant experiments by Eipper and Main.116 Using the pulse-chase technique for differential labeling of peptides, they were able to establish that β-endorphin and ACTH were major intermediate peptide products of a larger precursor molecule with a molecular weight of 31 kd. This precursor was subsequently named proopiomelanocortin (POMC). The sequence for POMC was determined by genetic engineering techniques.117 Further examination of the POMC mRNA revealed that differential processing of this precursor occurred in different tissues. For example, in the rat anterior pituitary lobe the POMC molecule is processed predominantly to ACTH and β-LPH. In the pituitary intermediate lobe, the hypothalamus, and placenta, ACTH and β-LPH are further cleaved to form α-melanocyte-stimulating hormone (α-MSH) and β-endorphin.118 A schematic representation of the POMC molecule and its known processed products is shown in Figure 7.
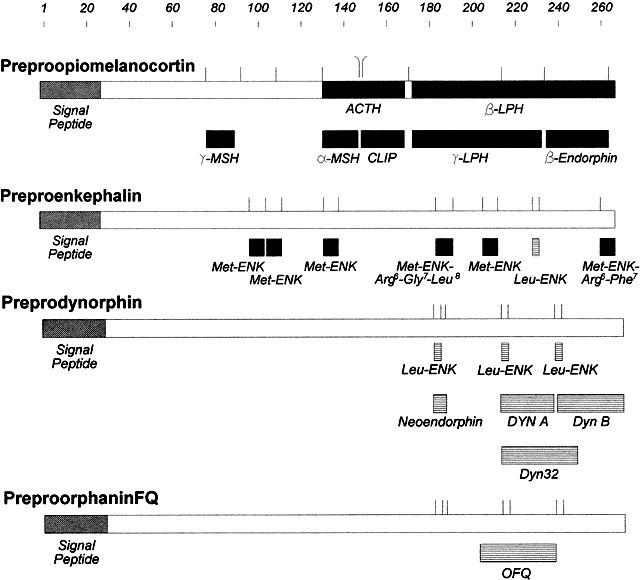
Since the discovery of the POMC family of peptides, three other classes of opioid peptides have been characterized: enkephalins, dynorphins, and the newest opioid peptide, nociceptin or orphanin FQ. The endogenous opiate peptides are all derived from posttranslational processing of larger precursor peptides (i.e. POMC, proenkephalin A, proenkephalin B, and pronociceptin), each of which are encoded by separate genes (Fig. 8). Preproenkephalin A is processed to yield four met-enkephalins, one leu-enkephalin, and two other modifications of met-enkephalin. Preprodynorphin, or preproenkephalin B, gives rise to dynorphins A and B, and neoendorphin. In a similar manner, nociceptin and perhaps other uncharacterized peptides are derived from pronociceptin.119
Opioid Receptors
The primary action of the opioidergic peptides is through binding to specific opioid receptors. In the past, pharmacologic studies suggested the presence of many different opiate receptor subtypes. Molecular biologic techniques have determined four major classes of opioid receptors, each arising from a separate gene.120 The three major classes of opiate receptor are classified as μ, δ, and κ. A fourth opiate receptor, known as ORL-1, was cloned in 1994.121 It is likely that subtypes exist within these four major groups.
Originally localized to the vas deferens (hence the δ designation), the first opioid receptor to be cloned and definitively established was the δ receptor, also known as DOR and OP1.122 The δ receptor displays preferential binding of enkephalins. The highest δ receptor density is found in the olfactory bulb, neocortex, caudate putamen, and nucleus accumbens.123 It is likely that there are two subtypes of δ receptors. Proposed functions of the δ receptor include analgesia, olfaction, motor function, cognition, and mood.
The second opioid receptor to be established was the κ receptor, named for its binding to a synthetic opiate ketocyclazocine.124 Also known as KOR and OP2, this receptor is localized to the cerebral cortex and other sites within the CNS. Pharmacologic data support the existence of three subtypes of the κ receptor. Proposed functions include pain, appetite, diuresis, and neuroendocrine regulation.
The third major class of opioid receptor is designated μ for its affinity for morphine.125 Other designations for the μ receptor are OP3 and MOR. Pharmacologic data are largely derived from studies on the guinea pig ileum, which has a high density of these receptors. The widely used opioid antagonist naloxone has a high affinity for the μ receptor, whereas the orally active naltrexone is less selective. The μ receptor is localized throughout the CNS, spinal cord, and peripheral nervous system, especially the gut. Binding studies support the existence of two subtypes of the μ receptor.123 Believed to mediate the actions of morphine and its derivatives, the μ receptor generally exerts a depressant effect on many functions, including respiration, cardiac function, gastrointestinal function, locomotor activity, and memory.
In the search for opioid receptor subtypes, a fourth opioid receptor, initially designated an "orphan opiate receptor" or ORL-1, was identified.121 Despite a high degree of homology with the other opioid receptors, ORL-1 showed poor binding to known opioid compounds. Later, the endogenous ligand for ORL-1 was found and given the names nociceptin126 and orphanin FQ.127
Distribution
The POMC-derived peptides have a ubiquitous distribution and have been identified in the placenta, gastrointestinal tract, and brain (Table 4). Within the brain, the highest concentrations of β-endorphin have been localized to the basomedial and basolateral hypothalamus128 from which neuronal processes project to the anterior hypo thalamus. Other areas where β-endorphin is distributed include the locus ceruleus (pons), periventricular nucleus, amygdala, and periaqueductal gray matter.
TABLE 4. Distribution of Proopiomelanocortin-Derived Peptides
Brain
Globus pallidus
Nucleus accumbens
Mesencephalon
Amygdala
Medial hypothalamus
Locus ceruleus
Pituitary gland
Male reproductive tract
Vas deferens
Epididymis
Seminal vesicles
Female reproductive tract
Ovary (corpus luteum)
Placenta
Gastrointestinal tract
Stomach
Pancreas
Intestine
Lung
Adapted from Krieger DT: The multiple faces of proopiomelanocortin, a prototype precursor molecule. Clin Res 31:342, 1983.
The enkephalin family of peptides is also widely distributed in the brain, with the concentrations of met-enkephalin exceeding leu-enkephalin by a 5:1 ratio. Areas that have high concentrations include medial hypothalamus, amygdala, mesencephalon globus pallidus, and the substantia gelatinosa of the spinal cord.129
The third major opioid peptide group, dynorphin, has been localized to the supraoptic and paraventricular nucleus of the hypothalamus and the posterior pituitary.130 Dynorphins also are colocalized with vasopressin, oxytocin, and CRH and are found within pituitary gonadotrophs.131,132 The presence of all three groups of peptides in the brain reflects the complexity of the opioid system.
Nociceptin and its receptor are widely distributed in the central nervous system, where it has a primarily inhibitory effect. Nociceptin is highly expressed in the hippocampus, stria terminalis, amygdaloid nucleus, and selective thalamic nuclei.133 The nociceptin receptor, known as ORL-1, is expressed in the diencephalon, mesencephalon, pons/medulla, and spinal cord.134
Measurement in Biologic Fluids
Because POMC-derived peptides share similar amino acid sequences, antisera that are generated to individual peptides such as β-endorphin and β-LPH often cross-react with other POMC peptides. Measurement of these peptides requires confirmation by rigorous chromatographic characterization.135 Peptidases are present in biologic fluids such as plasma, and further cleavage of the peptide to other opiate fragments can potentially take place. Current approaches to measurement of β-endorphin and β-LPH use extraction techniques such as C-18 minicolumns before chromatographic separation of peptides and radioimmunoassay.
In general, measurements of peripheral levels of β-endorphin and β-LPH parallel that of ACTH in pathologic states such as Cushing's syndrome or Nelson's syndrome. Under CRH stimulation, simultaneous elevations of β-endorphin, β-LPH, and ACTH are also observed.136 Interpretation of changes in peripheral β-endorphin levels under basal conditions is difficult. Because opiate peptides do not cross the blood-brain barrier, changes in peripheral β-endorphin levels may not reflect central opioidergic activity. Wehrenberg and coworkers demonstrated that large changes in the concentration of β-endorphin in the portal blood are not accompanied by concomitant changes in peripheral β-endorphin levels.137
Behavioral Effects
Beyond the analgesic effects of opiates, the behavioral effects in humans have not been well characterized. After one dose of morphine, human volunteers report a positive euphoric effect, mild nausea, giddiness, and a subjective sense of mental clouding. On the basis of animal studies, β-endorphin is 50 to 100 times more potent than morphine in achieving this effect. In contrast, injection of nociceptin into mice increases their sensitivity to pain (hence the name nociceptin).138
Animal studies suggest that the opioidergic system may influence memory, blood pressure, temperature, feeding, and sexual activity. These findings are not surprising, because it appears that the opioidergic system can modulate other major neuron systems.128
Opioid Modulation of Anterior Pituitary Function
Two approaches have been used to study the effects of opiates. In initial studies, morphine or opiate analogs were administered to animals or humans, and corresponding changes in hormonal levels were measured. These experiments were primarily pharmacologic and probably did not reflect physiologic changes that might be mediated by opiates. Later studies took a more physiologic approach using naloxone, a nonspecific opioid receptor antagonist, to assess the action of endogenous opiates. The effects of opiates can be determined with greater confidence using both approaches.
In early in vivo studies, β-endorphin or morphine was shown to stimulate GH, prolactin, and corticosteroid release when administered intracerebrally or intravenously. These effects were abolished by naloxone.129 Under conditions in which morphine is given chronically, a tolerance to these effects is observed. From these findings, it is unclear whether opiates play a role in modulation of basal secretion of these hormones. The observed effects of opiates on anterior pituitary function in humans are summarized in Table 5.
TABLE 5. Proposed Actions of Opioid Peptides on Pituitary Function
Acute stimulation of growth hormone release
Acute stimulation of prolactin release
Decrease in corticotropin release
Decrease in thyroid-stimulating hormone secretion
Inhibition of gonadotropin-releasing hormone release
Inhibition of suckling-induced oxytocin release
Morley SE: The endocrinology of the opiates and opioid peptides. Metabolism 30:195, 1981.
Opioid Modulation of Neurotransmitter Function
The long-term effects of opiates have been characterized at the neuronal level. In general, the biologic activity of opiate peptides is inhibitory. Opiates have been shown to suppress the firing of adrenergic neurons presynaptically in morphineaddicted animals. Because of the decrease in adrenergic input at the synapse, a compensatory increase of adrenergic receptors occurs postsynaptically. If opiate inhibition is interrupted, normal firing of adrenergic neurons resumes in the face of increased sensitivity postsynaptically, resulting in overactivation of the sympathetic nervous system.139 Observed effects include tremors, restlessness, irritability, sweating, and other symptoms typical of heroin withdrawal.
Opiate peptides also exert similar effects on the dopaminergic system. In isolated dopaminergic neurons, opiates inhibit depolarization.140 In vivo studies by Gudelsky and Porter show a reduction in dopamine concentration in rat pituitary stalk plasma when morphine or β-endorphin is administered.141 These observations suggest that the increases in prolactin levels after exposure to βendorphin or morphine reflect opioid inhibition of dopamine activity, resulting in increased prolactin release.
Opiates in the Control of Reproduction
As in other anterior pituitary hormones, the predominant effect of opiate peptides on gonadotropin secretion is inhibitory. When morphine, human β-endorphin, or enkephalin analogs are administered to women, acute suppression of LH levels is observed.142,143 If naloxone is used to block the action of endogenous opiates during the late follicular and mid-luteal phases of the menstrual cycle, increases in LH, the opposite effect,144 occur. What then governs the activity of these endogenous opiate peptides?
Evidence suggests that the gonadal steroid environment may influence endogenous opiate activity. Measurement of β-endorphin concentrations in the pituitary portal blood in monkeys during the menstrual cycle show an increase in release of hypothalamic β-endorphin during late follicular and mid-luteal phases of the menstrual cycles.137 In ovariectomized monkeys, increased hypothalamic β-endorphin release was observed only in monkeys with estrogen and/or progesterone replacement.145 These experiments suggest that opioid activity may be influenced by the gonadalsteroidal environment.
In an analogous situation in humans, after naloxone administration during the menstrual cycle, there appears to be a progressive increase in LH release from the late follicular to the mid-luteal phase of the cycle, suggesting indirectly that endogenous opiate activity was suppressing gonadotropin levels.144 In low-estrogen environments such as in menopausal women, naloxone infusions have no effects on LH levels. However, replacement with progesterone146 or with estrogen and progesterone result in reestablishing a gonadotropin rise in response to naloxone.147 Collectively, these observations suggest that endogenous opiate inhibition is mediated in part by the gonadal-steroidal environment.
At what point in development does functional coupling between the opiate and GnRH system occur? If we examine the hypothalamic-pituitarygonadal axis of the fetus, it is evident that the human fetal pituitary gland can secrete gonadotropins during early life in sufficient amounts to stimulate production of gonadal steroids. If fetal hypothalami are exposed to naloxone, increased GnRH release occurs, indicating that the suppressive effect of endogenous opiates is already established by 20 weeks' gestation.148
On the basis of these observations, it is clear that the opioidergic system is a major regulator of GnRH secretion and may be involved in several clinical hypogonadotropic states. One example in which endogenous opiates have been implicated is the postpartum period. The puerperium is characterized by an extended interval of low gonadotropin levels. Administration of naloxone during the second postpartum week reverses this situation and is accompanied by significant elevations of LH and FSH, suggesting that endogenous opiate activity is in part responsible for gonadotropin suppression.149 Two other clinical conditions in which hypogonadotropinism occur are anorexia nervosa and hypothalamic amenorrhea. In both conditions, blockage of endogenous opiate activity with naloxone results in elevations of gonadotropin.150,151
Transgenic Mice and the Opioidergic System
Many of the opioid peptides and their receptors have been investigated by gene knockout experiments, providing new information on the physiologic role of opiates. For example, mice deficient in β-endorphin are viable and reproduce.152 They do not show abnormalities in the adrenal axis. These mice do respond to morphine but show an abnormal analgesic response to stress. However, mice deficient in the μ opiate receptor do not respond to morphine with an analgesic response,153 and mice lacking the κ receptor154 or the δ receptor155 respond to morphine normally. This evidence supports a role for the μ receptor in mediating the actions of this drug.153 Affected mice develop normally and reproduce. Altered pain responses under basal conditions in these mice support a role of opioidergic peptides in the endogenous response to pain. Other findings in the μ receptor knockout mice were changes in hematopoiesis, motor function, and sexual behavior.156 Alterations in the function of the κ and δ receptor in mice deficient in the μ receptor support a physiologic interaction among the opioid receptors, as suspected from pharmacologic data.155 Enkephalin-deficient mice are healthy, are fertile, and show normal maternal behavior. The mice are, however, anxious, aggressive, and show abnormal responses to pain.157 Mice lacking the nociceptin receptor (ORL-1) have been developed. Surprisingly, these mice appear to have a normal response to pain, but have primarily hearing and memory deficits.158
Although the genetically altered mice have provided important insights into our understanding of opioidergic peptides, these results certainly underscore the vast amount of opiate physiology that remains to be defined. It is apparent that we are just beginning to unravel the complex interactions of the opioidergic system. From these early studies, it appears that opiates play an inhibitory role in the control of gonadotropin secretion. This activity appears to be functionally coupled by gonadal steroids and is active during early fetal life. With our enhanced ability to isolate and characterize new peptides, the answers should be forthcoming.
REFERENCES
Scharrer E, Scharrer B: Secretory cells within the hypothalamus. In The Hypothalamus: Association for Research on Nervous and Mental Disease. New York: Hafner, 1940 |
|
Darlison M, Richter D: Multiple genes for neuropeptides and their receptors: co-evolution and physiology. Trends Neurosci 22: 81, 1999 |
|
Cho S, Han J, Sun W et al: Evidence for autocrine inhibition of gonadotropin-releasing hormone (GnRH) gene transcription by GnRH in hypothalamic GT1–1 neuronal cells. Mol Brain Res 50: 51, 1997 |
|
Denef C, Andries M: Evidence for paracrine interaction between gonadotrophs and lactrotrophs in pituitary cell aggregates. Endocrinology 112: 813, 1983 |
|
Krieger DT: Brain peptides: What, where, and why. Science 222: 975, 1983 |
|
Matsuo H, Baba Y, Nair RMG et al: Structure of the porcine LH and FHS releasing factor: I. The proposed amino acid sequence. Biochem Biophys Res Commun 43: 1334, 1971 |
|
Burgus R, Butcher M, Ling N et al: Molecular structure of the hypothalamic factor (LRF) of ovine origin monitoring the secretion of pituitary gonadotropic hormone of luteinization (LH). Comptes Rendus Hebdomadaires Seances Acad Sci 273: 1611, 1971 |
|
Brazeau P, Vale W, Burgus R et al: Hypothalamic polypeptide that inhibits the secretion of immunoreactive pituitary growth hormone. Science 179: 77, 1973 |
|
Cooper JR, Bloom FE, Roth RH: The biochemical basis of neuropharmacology. In: Cooper JR, Bloom FE, Roth RH (eds): Neuroactive Peptides. New York: Oxford University Press, 1982 |
|
Majzoub JA, Muglia LJ: Knockout mice. N Engl J Med 334: 904, 1996 |
|
Robinson AG: Isolation, assay and secretion of individual human neurophysins. J Clin Invest 55: 360, 1975 |
|
Dierickx K, Van de Sande F: Immunocytochemical demonstration of separate vasopressin-neurophysin and oxytocin-neurophysin neurons in the human hypothalamus. Cell Tissue Res 196: 203, 1979 |
|
Robertson GL: The regulation of vasopressin function in health and disease. Recent Prog Horm Res 33: 333, 1977 |
|
Ritta-Fuchs, Fuchs F: Endocrinology of human parturition: A review. Br J Obstet Gynecol 91:948, 1984 |
|
Nishimori K, Young LJ, Guo Q et al: Oxytocin is required for nursing but is not essential for parturition or reproductive behavior. Proc Natl Acad Sci USA 93: 11699, 1996 |
|
Fuchs AR, Fuchs F, Husslein P et al: Oxytocin receptor and human parturition: A dual role for oxytocin in the initiation of labor. Science 215: 1396, 1982 |
|
Liu JH, Muse K, Contreras P et al: Augmentation of ACTH-releasing activity of synthetic corticotropin releasing factor by vasopressin in women. J Clin Endocrinol Metab 57: 1087, 1983 |
|
Legros JJ, Chiodera P, Demey-Ponsart E: Inhibitory influence of exogenous oxytocin on adrenocorticotropin secretion in normal human subjects. J Clin Endocrinol Metab 55: 1035, 1982 |
|
Bergland RM, Page RB: Can the pituitary secrete directly to the brain? (affirmative anatomical evidence). Endocrinology 102: 1326, 1978 |
|
Gibbs DM: Dissociation of oxytocin, vasopressin, and corticotropin secretion during different types of stress. Life Sci 35: 487, 1984 |
|
Weingartner H, Gold P, Ballenger JL et al: Effects of vasopressin on human memory functions. Science 211: 601, 1981 |
|
Ferrier BM, Kennett DJ, Deviin ML: Influence of oxytocin on human memory processes. Life Sci 27: 2311, 1980 |
|
Pedersen CA, Prange AJ Jr: Induction of maternal behavior in virgin rats after intracerebroventricular administration of oxytocin. Proc Natl Acad Sci USA 76: 6661, 1979 |
|
Valtin H, Schroeder HA: Familial hypothalamic diabetes insipidus in rats (Brattleboro strain) [1964 classic article]. J Am Soc Nephrol 8: 1333, 1997 |
|
Bohus B, de Wied D: The vasopressin deficient Brattleboro rats: a natural knockout model used in the search for CNS effects of vasopressin. Prog Brain Res 119: 555, 1998 |
|
Boler J, Enzmann F, Folkers K et al: The identity a chemical and hormonal properties of the thyrotropin releasing hormone and pyro-glutamyl-histadyl proline amide. Biochem Biophys Res Commun 37: 705, 1970 |
|
Jackson IMD: Extrahypothalamic and phylogenetic distribution of hypothalamic peptides. In: Greae RS (ed): The Hypothalamus. New York: Raven Press, 1978 |
|
Vale W, Rivier C, Brown M: Regulatory peptides of the hypothalamus. Annu Rev Physiol 39: 473, 1977 |
|
Yamada M, Saga Y, Shibusawa N et al: Tertiary hypothyroidism and hyperglycemia in mice with targeted disruption of the thyrotropin-releasing hormone gene. Proc Natl Acad Sci USA 94: 10862, 1997 |
|
Silverman AS, Antunes JL, Ferin M et al: The distribution of luteinizing hormone-releasing hormone in the hypothalamus of the rhesus monkey: Light microscope studies using immunoperoxidase technique. Endocrinology 101: 134, 1977 |
|
Seeburg PH, Adelman JP: Characterization of cDNA for precursor of human luteinizing hormone releasing hormone. Nature 311: 666, 1984 |
|
Adelman JP, Mason AJ, Hayflick JS, Seeburg PH: Isolation of the gene and hypothalamic cDNA for the common precursor of gonadotropin-releasing hormone and prolactin release-inhibiting factor in human and rat. Proc Natl Acad Sci USA 83: 179, 1986 |
|
Radovick S, Wondisford FE, Nakayama Y et al: Isolation and characterization of the human gonadotropin-releasing hormone gene in the hypothalamus and placenta. Mol Endocrinol 4: 476, 1990 |
|
Schwanzel-Fukuda M, Jorgenson KL, Bergen HT, Weesner GD, Ffaff DW: Biology of normal luteinizing hormone-releasing hormone neurons during and after their migration from olfactory placode. Endocr Rev 13: 623, 1992 |
|
Jeffcoate SL, Greenwood RH, Holland DT: Blood and urine clearance of luteinizing hormone releasing hormone in man measured by radioimmunoassay. J Endocrinol 60: 305, 1974 |
|
Belchetz P, Plant TM, Nakai Y et al: Hypophysial responses to continuous intermittent delivery of hypothalamic gonadotropin-releasing hormone. Science 202: 631, 1980 |
|
Sakuma Y, Pfaff D: LHRH in the mesencephalic central grey can potentiate lordosis reflex of female rats. Nature 283: 566, 1980 |
|
Sakuma Y, Pfaff DW: Modulation of the lordosis reflex of female rats by LHRH, its antiserum and analogs in the mesencephalic gray. Nature 283: 566, 1980 |
|
Loumaye E, Thorner J, Catt KJ: Yeast mating pheromone activates mammalian gonadotrophs: Evolutionary conservation of a reproductive hormone? Science 218: 1323, 1982 |
|
Jan YN, Jan LY, Kuffler SW: A peptide as possible transmitter in sympathetic ganglia of the frog. Proc Natl Acad Sci USA 76: 1501, 1979 |
|
Yen SSC, Lasley BL, Wang CF et al: The operating characteristic of the hypothalamic pituitary system during the menstrual cycle and observations of biological action of somatostatin. Recent Prog Horm Res 31: 321, 1975 |
|
Tavianini M, Hayest T, Magazin M et al: Isolation, characterization of DNA sequence of the rat somatostatin gene. J Biol Chem 259: 11798, 1984 |
|
Plotsky PM, Vale W: Patterns of growth hormone releasing factor and somatostatin secretion into the hypophyseal portal circulation of the rat. Science 230: 461, 1985 |
|
Schwartz JP, Taniwaki T, Messing A, Brenner M: Somatostatin as a trophic factor: Analysis of transgenic mice overexpressing somatostatin in astrocytes. Ann N Y Acad Sci 780: 29, 1996 |
|
Zheng H, Bailey A, Jiang M et al: Somatostatin receptor subtype 2 knockout mice are refractory to growth hormone-negative feedback on arcuate neurons. Mol Endocrinol 11: 1709, 1997 |
|
Martinez V, Curi AP, Torkian B et al: High basal gastric acid secretion in somatostatin receptor subtype 2 knockout mice. Gastroenterology 114: 1125, 1998 |
|
Vale W, Spiess J, Rivier C et al: Characterization of a 41 residue ovine hypothalamic peptide that stimulates secretion of corticotropin and β-endorphin. Science 213: 1394, 1981 |
|
Shibahara S, Morimoto Y, Furutani Y et al: Isolation and sequence analysis of the human corticotropin-releasing factor precursor gene. EMBO J 2: 775, 1983 |
|
Suda T, Tomori N, Tozawa F et al: Distribution and characterization of immunoreactive corticotropin-releasing factor in human tissues. J Clin Endocrinol Metab 55: 861, 1984 |
|
Mershon JL, Sehlhorst CS, Rebar RW, Liu JH: Evidence of a corticotropin-releasing hormone pulse generator in the macaque hypothalamus. Endocrinology 130: 2991, 1992 |
|
Brown MR, Fisher LH, Spiess J et al: Corticotropin-releasing factor: Actions on the sympathetic nervous system and metabolism. Endocrinology 111: 928, 1982 |
|
Nemeroff CB, Widerlov E, Bissette G et al: Elevated concentrations of CSF corticotropin-releasing factor-like immnoreactivity in depressed patients. Science 226: 1342, 1984 |
|
Gambacciani M, Yen SSC, Rasmussen DD: GnRH release from the mediobasal hypothalamus: In vitro inhibition by corticotropin-releasing factor. Neuroendocrinology 43: 533, 1986 |
|
Rivier C, Rivier J, Vale W: Stress-induced inhibition of reproductive function: Role of endogenous corticotropin releasing factor. Science 231: 607, 1986 |
|
Williams CL, Nishihara M, Thalabard JC, Grosser PM, Hotchkiss J, Knobil E: Corticotropin-releasing factor and gonadotropin-releasing hormone pulse generator activity in the rhesus monkey: Electrophysiological studies. Neuroendocrinology 52: 133, 1990 |
|
Barbarino A, DeMarinis L, Tofani A et al: Corticotropin releasing hormone inhibition of gonadotropin release and the effects of opioid blockade. J Clin Endocrinol Metab 68: 523, 1989 |
|
Thomas MA, Rebar RW, LaBarbera AR et al: Dose-response effects of exogenous pulsatile human corticotropin-releasing hormone on adrenocorticotropin, cortisol, and gonadotropin concentrations in agonadal women. J Clin Endocrinol Metab 72: 1249, 1991 |
|
Schurmeyer TH, Augerinos PC, Gold PW et al: Human corticotropin-releasing factor in man: Pharmacokinetic properties and dose-response of plasma adrenocorticotropin and cortisol secretion. J Clin Endocrinol Metab 59: 1103, 1984 |
|
Stenzel-Poore MP, Heinrichs SC, Rivest S et al: Overproduction of corticotropin-releasing factor in transgenic mice: A genetic model of anxiogenic behavior. J Neurosci 14: 2579, 1994 |
|
Muglia L, Jacobson L, Majzoub JA: Production of corticotropin-releasing hormone-deficient mice by targeted mutation in embryonic stem cells. Ann N Y Acad Sci 780: 49, 1996 |
|
Muglia L, Jacobson L, Dikkes P, Majzoub JA: Corticotropin-releasing hormone deficiency reveals major fetal but not adult glucocorticoid need. Nature 373: 427, 1995 |
|
Chrousos GP, Schulte HM, Oldfield EH et al: The corticotropin-releasing factor stimulation test. N Engl J Med 310: 622, 1984 |
|
Chalmers DT, Lovenberg TW, Grigoriadis DE et al: Corticotrophin-releasing factor receptors: From molecular biology to drug design. Trends Pharmacol Sci 17: 166, 1996 |
|
Vaughan J, Donaldson C, Bittencourt J et al: Urocortin, a mammalian neuropeptide related to fish urotensin I and to corticotropin-releasing factor. Nature 378: 287, 1995 |
|
Danaldson CJ, Sutton SW, Perrin MH et al: Cloning and characterization of human urocortin. Endocrinology 137: 3896, 1996 |
|
Asaba K, Makino S, Hashimoto K: Effect of urocortin on ACTH secretion from rat anterior pituitary in vitro and in vivo: Comparison with corticotropin-releasing hormone. Brain Res 806: 95, 1998 |
|
Schilling L, Kanzler C, Schmiedek P, Ehrenreich H: Characterization of the relaxant action of urocortin, a new peptide related to corticotropin-releasing factor in the rat isolated basilar artery. Br J Pharmacol 125: 1164, 1998 |
|
Rivier I, Spiess J, Thorner MO et al: Characterization of a growth hormone-releasing factor from a human pancreatic islet tumor. Nature 300: 276, 1982 |
|
Guillemin R, Brazeau P, Bohlen P et al: Growth hormone-releasing factor from a human pancreatic tumor that caused acromegaly. Science 218: 585, 1982 |
|
Vance ML, Borges JL, Kaiser DL et al: Human pancreatic tumor growth hormone releasing factor: Dose-response relationships in normal man. J Clin Endocrinol Metab 58: 838, 1984 |
|
Gelato MC, Pescovitz O, Cassorla F et al: Effects of growth hormone releasing factor in man. J Clin Endocrinol Metab 57: 674, 1983 |
|
Gelato MC, Merriam GR, Vance ML et al: Effects of growth hormone-releasing factor on growth hormone secretion in acromegaly. J Clin Endocrinol Metab 60: 251, 1985 |
|
Weidlin N, Culler F, Villanueva A et al: Diminished growth hormone release in response to human pancreatic growth hormone releasing factor (hpGRF-44) in hyperprolactinemic women. Presented before the Seventh International Congress of Endocrinology, 1984 |
|
Thorner MO, Reschke J, Chitwood J et al: Acceleration of growth in two children treated with human growth hormone releasing factor. N Engl J Med 312: 4, 1985 |
|
Chapman IM, Pescovitz OH, Murphy G et al: Oral administration of growth hormone (GH) releasing peptidemimetic MK-677 stimulates the GH/insulin-like growth factor-I axis in selected GH-deficient adults. J Clin Endocrinol Metab 82: 3455, 1997 |
|
Allen YS, Adrian TG, Allen JM et al: Neuropeptide 5 distribution in the brain. Science 221: 877, 1983 |
|
Kalra SP, Crowley WR: Neuropeptide Y: A novel neuroendocrine peptide in the control of pituitary hormone secretion and its relation to luteinizing hormone. Front Neuroendocrinol 13: 1, 1992 |
|
Balasubramaniam A: Neuropeptide Y family of hormones: Receptor subtypes and antagonists. Peptides 18: 445, 1997 |
|
Kalra SP, Kalra PS: Nutritional infertility: The role of the interconnected hypothalamic neuropeptide Y-galanin-opioid network. Front Neuroendocrinol 17: 371, 1996 |
|
Clark JT, Kalra PS, Kalra SP: Neuropeptide Y stimulate feeding but inhibits sexual behavior in rats. Endocrinology 117: 2435, 1985 |
|
Erickson JC, Clegg KE, Palmiter RD: Sensitivity to leptin and susceptibility to seizures of mice lacking neuropeptide Y. Nature 381: 415, 1996 |
|
Stephens TW: Fat regulation: Life without neuropeptide Y. Nature 381: 377, 1996 |
|
Langel U, Bartfai T: Chemistry and molecular biology of galanin receptor ligands. Ann N Y Acad Sci 863: 86, 1998 |
|
Vrontakis M, Peden L, Duckworth M, Friesen H: Isolation and characterization of a complementary DNA (galanin) clone from estrogen-induced pituitary tumor messenger RNA. J Biol Chem 262: 16755, 1987 |
|
Branchek T, Smith K, Walker M: Molecular biology and pharmacology of galanin receptors. Ann N Y Acad Sci 863: 94, 1998 |
|
Habert-Ortoli E, Amiranoff B, Loquet I et al: Molecular cloning of a functional human galanin receptor. Proc Natl Acad of Sci USA 91: 9780, 1994 |
|
Rokaeus A, Jiang K, Spyrou G, Waschek J: Transcriptional control of the galanin gene: Tissue-specific expression and induction by NGF, protein kinase C, and estrogen. Ann N Y Acad Sci 863: 1, 1998 |
|
Hohmann J, Clifton D, Steiner R: Galanin: analysis of its coexpression in gonadotropin-releasing hormone and growth hormone-releasing hormone neurons. Ann NY Acad Sci 863: 221, 1998 |
|
Leibowitz S, Akabayashi A, Alexander J, Wang J: Gonadal steroids and hypothalamic galanin and neuropeptide Y: Role in eating behavior and body weight control in female rats. Endocrinology 139: 1771, 1998 |
|
Kalra S, Horvath T: Neuroendocrine interactions between galanin, opioids, and neuropeptide Y in the control of reproduction and appetite. Ann NY Acad Sci 863: 236, 1998 |
|
Wynick D, Small C, Bacon A et al: Galanin regulates prolactin release and lactotroph proliferation. Proc Natl Acad Sci USA 95: 12671, 1998 |
|
Akabayashi A, Koenig JI, Watanabe Y et al: Galanin-containing neurons in the paraventricular nucleus: a neurochemical marker for fat ingestion and body weight gain. Proc Natl Acad Sci USA 91: 10375, 1994 |
|
Kalra SP, Dube MG, Pu S et al: Interacting appetite-regulating pathways in the hypothalamic regulation of body weight. Endocr Rev 20: 68, 1999 |
|
Leibowitz S: Differential functions of hypothalamic galanin cell grows in the regulation of eating and body weight. Ann NY Acad Sci 863: 206, 1998 |
|
Wang J, Akabayashi A, Yu H et al: Hypothalamic galanin: control by signals of fat metabolism. Brain Res 804: 7, 1998 |
|
Dube M, Horvath T, Leranth C et al: Naloxone reduces the feeding evoked by intracerebroventricular galanin injection. Physiol Behav 56: 811, 1994 |
|
Wynick D, Small C, Bloom S, Pachnis V: Targeted disruption of the murine galanin gene. Ann NY Acad Sci 863: 22, 1998 |
|
Zhang Y, Proenca R, Maffei M et al: Positional cloning of the mouse obese gene and its human homologue. Nature 372: 425, 1994 |
|
Considine RV, Sinha MK, Heiman ML et al: Serum immunoreactive-leptin concentrations in normal-weight and obese humans [see comments]. N Engl J Med 334: 292, 1996 |
|
Inui A: Feeding and body-weight regulation by hypothalamic neuropeptides—mediation of the actions of leptin. Trends Neurosci 22: 62, 1999 |
|
Sahu A: Leptin decreases food intake induced by melanin-concentrating hormone (MCH), galanin (GAL) and neuropeptide Y (NPY) in the rat. Endocrinology 139: 4739, 1998 |
|
Mercer JG, Hoggard N, Williams LM et al: Coexpression of leptin receptor and preproneuropeptide Y mRNA in arcuate nucleus of mouse hypothalamus. J Neuroendocrinol 8: 733, 1996 |
|
Schwartz MW, Seeley RJ, Campfield LA et al: Identification of targets of leptin action in rat hypothalamus. J Clin Invest 98: 1101, 1996 |
|
Wang Q, Bing C, Al-Barazanji K et al: Interactions between leptin and hypothalamic neuropeptide Y neurons in the control of food intake and energy homeostasis in the rat. Diabetes 46: 335, 1997 |
|
Xu B, Pu S, Kalra PS et al: An interactive physiological role of neuropeptide Y and galanin in pulsatile pituitary luteinizing hormone secretion. Endocrinology 137: 5297, 1996 |
|
Brownstein MJ, Mroz EA, Kizer JS et al: Regional distribution of substance P in the brain of the rat. Brain Res 116: 299, 1976 |
|
Morel G, Chayvialle JA, Kerdelhue B et al: Ultrastructural evidence for endogenous substance P-like immunoreactivity in the rat pituitary gland. Neuroendocrinology 35: 86, 1982 |
|
Vijayan E, McCann SM: In vivo and in vitro effects of substance P and neurotensin on gonadotropin and prolactin release. Endocrinology 105: 64, 1979 |
|
Woolf CJ, Mannion RJ, Neumann S: Null mutations lacking substance: elucidating pain mechanisms by genetic pharmacology. Neuron 20: 1063, 1998 |
|
Dornelles RC, Franci CR: Alpha- but not beta-adrenergic receptors mediate the effect of angiotensin II in the medial preoptic area on gonadotropin and prolactin secretion. Eur J Endocrinol 138: 583, 1998 |
|
Umemura S, Kihara M, Sumida Y et al: Endocrinological abnormalities in angiotensinogen-gene knockout mice: Studies of hormonal responses to dietary salt loading. J Hypertens 16: 285, 1998 |
|
Hagaman JR, Moyer JS, Bachman ES et al: Angiotensin-converting enzyme and male fertility. Proc Natl Acad Sci USA 95: 2552, 1998 |
|
Hughes JT, Smith W, Kosterlitz HW et al: Identification of two related pentapeptides from the brain with potent opiate agonist activity. Nature 258: 577, 1975 |
|
Li CH, Chung D: Isolation and structure of an untriakonta peptide with opiate activity from camel pituitary glands. Proc Natl Acad Sci USA 73: 1145, 1975 |
|
Li CH: Lipotropin, a new active peptide from pituitary glands. Nature 201: 924, 1964 |
|
Eipper BA, Main RE: Structure and biosynthesis of pro-adrenocorticotropin/endorphin and related peptides. Endocr Rev 1: 1, 1980 |
|
Nakanishi SO, Inoue A, Kita T et al: Nucleotide sequence of cloned DNA for bovine corticotropin-83-lipotropin precursor. Nature 257: 238, 1979 |
|
Krieger DT: The multiple faces of pro-opiomelanocortin, a prototype precursor molecule. Clin Res 31: 342, 1983 |
|
Mollereau C, Simons MJ, Soularue P et al: Structure, tissue distribution, and chromosomal localization of the prepronociceptin gene. Proc Natl Acad Sci USA 93: 8666, 1996 |
|
Connor M, Christie MD: Opioid receptor signalling mechanisms. Clin Exp Pharmacol Physiol 26: 493, 1999 |
|
Mollereau C, Parmentier M, Mailleux P et al: ORL1, a novel member of the opioid receptor family: Cloning, functional expression and localization. FEBS Lett 341: 33, 1994 |
|
Kieffer BL, Befort K, Gaveriaux-Ruff K, Hirth CG: The delta-opioid receptor: Isolation of a cDNA by expression cloning and pharmacological characterization. Proc Natl Acad Sci USA 89: 12048, 1992 |
|
Dhawan BN, Cesselin F, Raghubir R et al: International Union of Pharmacology. XII. Classification of opioid receptors. Pharmacol Rev 48: 567, 1996 |
|
Akil H, Watson SJ: Cloning of kappa opioid receptors: Functional significance and future directions. Prog Brain Res 100: 81, 1994 |
|
Wang JB, Imia Y, Eppler CM et al: Mu opiate receptor: cDNA cloning and expression. Proc Natl Acad Sci USA 90: 10230, 1993 |
|
Meunier JC, Mollereau C, Toll L et al: Isolation and structure of the endogenous agonist of opioid receptor-like ORL1 receptor [see comments]. Nature 377: 532, 1995 |
|
Reinscheid RK, Nothacker HP, Bourson A et al: Orphanin FQ: A neuropeptide that activates an opioidlike G protein-coupled receptor. Science 270: 792, 1995 |
|
Bloom FG: Endorphins. In: Bloom FG, Roth RH (eds): The Biochemical Basis of Neuropharmacology. New York: Oxford University Press, 1982 |
|
Morley SE: The endocrinology of the opiates and opioid peptides. Metabolism 30: 195, 1981 |
|
Weber E, Roth KA, Barchas JD: Immunohistochemical distribution of a-neo-endorphin/dynorphin neuronal systems in rat brain: Evidence for colocalization. Proc Natl Acad Sci USA 79: 3062, 1982 |
|
Watson SJ, Akil H, Frichli W et al: Dynorphin and vasopressin: Common localization in magnocellular neurons. Nature 216: 88, 1982 |
|
Khatchaturian H, Sherman TG, Lloyd RV et al: Prodynorphin is endogenous in the anterior pituitary and is colocalized with LH and FSH in gonadotrophs. Endocrinology 199: 409, 1986 |
|
Boom A, Mollereau C, Meunier JC et al: Distribution of the nociceptin and nocistatin precursor transcript in the mouse central nervous system. Neuroscience 91: 991, 1999 |
|
Anton B, Fein J, To T et al: Immunohistochemical localization of ORL-1 in the central nervous system of the rat. J Comp Neurol 368: 229, 1996 |
|
Wilkes MM, Stewart RD, Bruni JF et al: A specific homologous radioimmunoassay for human-endorphin: Direct measurement in biological fluids. J Clin Endocrinol Metab 50: 309, 1980 |
|
McLoughlin L, Tomlin S, Grossman A et al: CRF-41 stimulates the release of beta lipotrophin and beta endorphin in normal human subjects. Neuroendocrinology 38: 282, 1984 |
|
Wehrenberg WB, Wardiaw SL, Frantz AG et al: β-Endorphin in hypophyseal portal blood: Variations throughout the menstrual cycle. Endocrinology 111: 879, 1982 |
|
Yamamoto T, Nozaki-Taguchi N, Kimura S: Effects of intrathecally administered nociceptin, an opioid receptor-like 1 (ORL1) receptor agonist, on the thermal hyperalgesia induced by carrageenan injection into the rat paw. Brain Res 754: 329, 1997 |
|
Lloren SC, Martres MP, Baudry M et al: Hypersensitivity to noradrenaline in cortex after chronic morphine: Relevance to tolerance and dependence. Nature 274: 603, 1978 |
|
Myers PR, Livengood DR, Shain W: Effect of morphine on a depolarizing dopamine response. Nature 257: 238, 1975 |
|
Gudelsky GA, Porter JC: Morphine and opioid peptide induced inhibition of the release of dopamine from tuberoinfundibular neurons. Life Sci 25: 1697, 1979 |
|
Reid RL, Hoff JD, Yen SSC et al: Effects of exogenous β-endorphin on pituitary hormone secretion and its disappearance rate in normal human subjects. J Clin Endocrinol Metab 52: 1179, 1981 |
|
Stubbs WA, Jones A, Edwards CRW et al: Hormonal and metabolic responses to an enkephalin analogue in normal man. Lancet 2: 1225, 1978 |
|
Quigley ME, Yen SSC: The role of endogenous opiates on LH secretion during the menstrual cycle. J Clin Endocrinol Metab 51: 179, 1980 |
|
Wardlaw SL, Wehrenberg WB, Ferin M et al: Effect of sex steroids on β-endorphin in hypophyseal portal blood. J Clin Endocrinol Metab 55: 877, 1982 |
|
Casper RF, Alapin-Rubillovitz S: Progestins increase endogenous opioid peptide activity in postmenopausal women. J Clin Endocrinol Metab 60: 34, 1985 |
|
Shoupe D, Montz FJ, Lobo RA: The effects of estrogen and progestin on endogenous opioid activity in oophorectomized women. J Clin Endocrinol Metab 60: 178, 1985 |
|
Rasmussen DD, Liu IH, Wolf PL et al: Endogenous opioid regulation of gonadotropin-releasing hormone release from the human fetal hypothalamus in vitro. J Clin Endocrinol Metab 57: 881, 1983 |
|
Ishizuka B, Quigley ME, Yen SSC: Postpartum hypogonadotrophinism: Evidence for increased opioid inhibition. Clin Endocrinol 20: 573, 1984 |
|
Baranowska B, Rozbicka G, Jeske W et al: The role of endogenous opiates in the mechanism of inhibited luteinizing hormone (LH) secretion in women with anorexia nervosa: The effect of naloxone on LH, FSH, prolactin and β-endorphin secretion. J Clin Endocrinol Metab 59: 412, 1984 |
|
Quigley ME, Sheehan KL, Casper RF et al: Evidence for an increased dopaminergic and opioid activity in patients with hypothalamic hypogonadotropic amenorrhea. J Clin Endocrinol Metab 50: 949, 1980 |
|
Rubinstein M, Mogil JS, Japon M et al: Absence of opioid stress-induced analgesia in mice lacking beta-endorphin by site-directed mutagenesis. Proc Natl Acad Sci USA 93: 3995, 1996 |
|
Sora I, Takahashi N, Funada M et al: Opiate receptor knockout mice define mu receptor roles in endogenous nociceptive responses and morphine-induced analgesia. Proc Natl Acad Sci USA 94: 1544, 1997 |
|
Slowe S, Simonin F, Kieffer B, Kitchen I: Quantitative autoradiography of mu-, delta- and kappa 1 opioid receptors in kappa-opioid receptor knockout mice. Brain Res 818: 335, 1999 |
|
Kieffer BL: Opioids: First lessons from knockout mice. Trends Pharmacol Sci 20: 19, 1999 |
|
Tian M, Broxmeyer HE, Fan Y et al: Altered hematopoiesis, behavior, and sexual function in mu opioid receptor-deficient mice. J Exp Med 185: 1517, 1997 |
|
Konig M, Zimmer AM, Steiner H et al: Pain responses, anxiety and aggression in mice deficient in pre-proenkephalin. Nature 383: 535, 1996 |
|
Nishi M, Houtani T, Noda Y et al: Unrestrained nociceptive response and disregulation of hearing ability in mice lacking the nociceptin/orphanin FQ receptor. EMBO J 16: 1858, 1997 |