Prevention and Treatment of Osteoporosis
Authors
INTRODUCTION
Osteoporosis is a worldwide health problem, affecting an estimated 75 million people in Europe, Japan, and the United States,1 mostly women. The incidence of osteoporosis increases greatly with age, and this disease has become a major public health problem among the elderly. Classically, osteoporosis has been defined as a fracture syndrome or a radiographic abnormality; however, advances in bone density measurement now allow diagnosis of low bone mass (osteopenia) and osteoporosis before subjective radiographic changes or the occurrence of fracture. Thus, our awareness of osteoporosis and its prevention and treatment have been acutely enhanced.
Osteoporosis is characterized by low bone mass and microarchitectural deterioration of the skeleton leading to enhanced skeletal fragility and a subsequent increase in fracture risk,2 which can occur even in the absence of trauma. The skeleton consists of two bone types: cortical, or compact, bone provides 80% of total skeletal mass; cancellous, or spongy, bone, the bone of the vertebral bodies, constitutes about 20% of total bone mass. Bone tissue is subject to continuous turnover just as are most tissues in the body. This process, termed bone remodeling, involves osteoclast-mediated bone resorption and osteoblast-mediated bone formation. High rates of bone turnover are associated with low bone mass in women, regardless of menopausal status.3 Bone remodeling occurs on skeletal surfaces. In contrast to bone mass, which is predominantly cortical bone, skeletal surfaces are predominantly (about 80%) cancellous bone. This high surface-to-mass (volume) ratio of cancellous bone makes skeletal sites that are rich in cancellous bone more susceptible to diseases resulting from remodeling disorders. Thus, vertebral bodies and the ends of long tubular bones, such as the proximal femur, are especially vulnerable to fracture. Spinal compression fractures occur in 25% of white women older than 60 years and are associated with acute and chronic pain, loss of height, and postural deformities. On average untreated postmenopausal white woman can expect to shrink 2.5 inches as a result of these fractures.
Because bone mass is greater in the femoral neck than in the spine, osteoporotic fractures of the femur occur later in life, usually between 70 and 75 years of age. The incidence of hip fractures increases from 0.3 per 1000 to 20 per 1000 from age 45 to 85 years, with 20% of all white women sustaining such a fracture by 90 years of age. Of these, 15% die within 3 months from complications of the fracture, including pulmonary edema, myocardial infarction, and pulmonary embolism. Another 25% of these women suffer significant morbidity and in some cases permanent disability. Hip fractures alone occur in about 250,000 women per year in the United States and are associated with a mortality of 40,000 annually and a cost of $8.7 billion.4
Risk factors for osteoporosis include low bone mass at the onset of menopause, white or Asian race, slender build, early onset of menopause, history of estrogen-deficient menstrual disorders, a positive family history for osteoporosis, glucocorticoid therapy, and nulliparity. Avoidable factors contributing to osteoporosis include sedentary lifestyle, inadequate dietary calcium and vitamin D intake, cigarette smoking, excessive caffeine or alcohol intake, and estrogen deficiency. Factors suggested to be protective against osteoporosis include obesity, multiparity, and possibly the use of oral contraceptives for more than 1 year. African American women have both a higher bone mass and a lower incidence and prevalence of osteoporotic fractures than do white women in the United States.
Bone remodeling is regulated by local, humoral, metabolic, nutritional, and mechanical factors; it is not surprising that many disease states can result in osteoporosis. Osteoporosis caused by other disease states, such as excess cortisol in Cushing's disease or from corticosteroid therapy, is termed secondary osteoporosis. The more common form of osteoporosis, postmenopausal osteoporosis, has been clearly demonstrated to result directly from estrogen deficiency but has been termed a primary osteoporosis. The other major primary osteoporosis, senile or involutional osteoporosis, results from age-related bone loss that is universal in humans, the pathogenesis of which is uncertain.
PRIMARY OSTEOPOROSIS
More than 80% of osteoporosis cases occur among postmenopausal and aging populations. Fracture risk at any age is determined primarily by bone density, which is dependent on the peak bone mass achieved at maturity and the subsequent rate of bone loss. Bone density peaks at about 25 years of age in the hip and at about 35 years of age in the spine.5 After 40 years of age, resorption begins to exceed formation by about 0.5% per year. At menopause, there is an acceleration of bone loss, with up to 5% of cancellous bone and 1–1.5% of total bone mass lost per year after menopause. This accelerated loss continues for 10–15 years, after which the rate of bone loss diminishes but continues as an aging-related loss.6 The accelerated loss experienced at menopause is termed type I osteoporosis and is typified by loss of cancellous bone resulting in vertebral fractures, whereas type II osteoporosis is best exemplified by hip fractures that occur in both older men and women (Table 1). Thus, in simple terms, type I osteoporosis is estrogen dependent, and type II osteoporosis is age related. Clearly, the presence of type I disease predisposes patients to type II.
Table 1. Types of osteoporosis
Primary osteoporosis
Postmenopausal (type I) Senile (type II) |
Secondary osteoporosis Corticosteroid induced |
Medications Corticosteroids |
SECONDARY OSTEOPOROSIS
The most common and most serious secondary form of osteoporosis is corticosteroid induced. Usually, this form is associated with therapeutic use of glucocorticoids, but it can be the presenting sign of endogenous Cushing's syndrome.7 Women experience an increased incidence of corticosteroid-induced osteoporosis because of their higher incidence of autoimmune diseases, which may require corticosteroid therapy, coupled with their lower peak bone masses.
A variety of gastrointestinal diseases are associated with osteoporosis and osteomalacia. Subtotal gastrectomy is associated with the development of osteomalacia and osteoporosis in 5–10% of cases many years after surgery.8 The mechanism may be calcium malabsorption, similar to that which occurs with achlorhydria and old age. Increasingly, obese patients are undergoing bariatric surgeries, of which the Roux-en-Y gastric bypass procedure seems to result in the greatest effects upon bone mass.9 Malabsorption associated with sprue and other intestinal diseases is underrecognized as a potential risk factor for osteoporosis. Malnutrition associated with alcoholism and anorexia nervosa likewise may increase the incidence of osteoporosis. Alcohol has additional direct adverse effects on the skeleton and is associated with a pseudo-Cushing's syndrome that includes increased endogenous steroid production. The bone loss of anorexia nervosa is aggravated by the accompanying estrogen deficiency.
Endocrine disorders commonly associated with secondary osteoporosis include hyperthyroidism and iatrogenic hyperthyroidism from overreplacement. Periodic reassessment of the replacement dose is appropriate, especially because a person's daily requirement for thyroid hormone can decrease with age. The incidence of primary hyperparathyroidism may be as high as 1 in 1000. In its mildest form, there may be no symptoms, in which case it is discovered only through routine determinations of serum calcium. In more severe forms, symptoms associated with hypercalcemia or hypercalciuria may lead to its diagnosis. Hypogonadal states are associated with osteoporosis in both genders, and hypogonadism commonly is underdiagnosed in aging men. Clinical conditions resulting in hypogonadism may include anorexia nervosa, hyperprolactinemia, acromegaly, and hypothalamic amenorrhea. Iatrogenic estrogen deficiency, as occurs with gonadotropin releasing hormone agonist or antagonist therapy, also results in accelerated bone loss, which may not be fully reversible when therapy is discontinued.
DIAGNOSIS
Because osteoporosis usually is asymptomatic until the first fracture has occurred, the importance of early diagnosis cannot be overemphasized. Noninvasive studies that allow detection of low bone density in patients without symptoms also allow initiation of therapy and prevention of fractures. Standard radiographs do not provide an early assessment of fracture risk because 25–30% of bone must be lost before radiographic changes become apparent. Low bone mass and increased fracture risk can be assessed only by direct measurement of bone mineral density (BMD) using dual energy X-ray absorptiometry (DXA). Each standard deviation (about 10%) decrement in BMD results in about twice the risk of fracture.10 To assist in patient management, the World Health Organization has defined degrees of deficit in BMD with a value between 1 and 2.5 standard deviations below peak adult bone mass as low bone mass or osteopenia and any value more than 2.5 standard deviations below peak adult bone mass as osteoporosis.10 The association between decreasing BMD and increasing fracture risk is analogous to the association between increasing serum cholesterol and increasing risk of acute myocardial infarction. The sensitivity of the BMD and fracture association, however, is greater than the cholesterol and acute myocardial infarction relation.11 Although BMD measured at any site assesses global fracture risk, site-specific measurements provide more reliable site-specific information. For example, each standard deviation decrement in BMD at the proximal femur is associated with a relative risk of hip fracture of 3, whereas the relative risk of hip fracture for every standard deviation decrease in forearm BMD is only 2. Nonetheless, the forearm measurement is clearly better than no measurement at all, and substantially so.
Table 2 lists the several options for direct, noninvasive measurement of BMD at different skeletal sites. All these methods measure BMD with 95% or greater accuracy, and all have been associated with increasing fracture risk as BMD decreases. Dual-energy X-ray absorptiometry employs photons from two energy sources and provides good precision for all sites of osteoporotic fractures. Quantitative computed tomography (QCT) for bone density measurements can be performed on most commercial CT systems; however, radiation exposure is higher than with DXA. Volumetric CT allows better visualization of the femoral neck as well as improved spinal measurements, but likewise is associated with increased radiation exposure. Single-energy X-ray absorptiometry measures bone density in the calcaneus, which correlates less well with vertebral bone density. Radiographic absorptiometry uses standard radiographic equipment and a calibrated marker to image the phalanges. Radiographs are interpreted using computer technology to determine the BMD. Ultrasonography of the calcaneus and patella has been demonstrated to correlate with vertebral bone density. Most important, each method and site of measurement of bone mass or density has been demonstrated to assess fracture risk, whether or not there is a good statistical correlation with other sites with respect to measured BMD. Magnetic resonance microscopy is under investigation to elucidate trabecular microarchitecture quantitatively, as is high resolution peripheral QCT. Because these are expensive, they probably will not be useful for primary osteoporosis screening; however, they may prove useful for identifying high-risk patients after initial bone densitometry and for research into the pathophysiology of the osteopenic disease process.
Table 2. Bone mineral density measurement
Method | Measurement site |
Dual-energy X-ray absorptiometry | Forearm (proximal and distal radius) |
Lumbar spine (anteroposterior and lateral projections) | |
Proximal femur | |
Total body | |
Quantitative computed tomography | Lumbar spine (cancellous bone only or combined cancellous and cortical bone) |
Single-energy X-ray absorptiometry | Calcaneus |
Radiographic absorptiometry | Phalanges (hand) |
Ultrasonography | Calcaneus |
Patella |
Results of DXA studies are expressed as BMD either in absolute terms (g/cm2), standard deviation units from peak adult BMD (T score), or standard deviation units from BMD adjusted for age and sex (Z score). Both these reporting methods provide an assessment of fracture risk, but the T score allows comparison between the individual patient and study populations reported in the literature. The Z score has most utility in determining which patient might require additional investigation; that is, this measure indicates which patient has a value for BMD that can be accounted for solely on the basis of age, sex, and menopausal status. Any patient in whom the Z score is lower than −2 should be evaluated for secondary causes of low BMD; the yield from such studies is significantly lower in patients with better Z scores. Despite the utility of BMD, this assessment provides only a static measurement of bone mass and gives no information on the rate of bone loss. Also, no matter how reported, BMD provides a continuous measure of fracture risk, and decisions about intervention should be made using all available information about the patient, including BMD, past medical history, lifestyle, and family history. For example, two patients who appear identical in all respects, including BMD, might be treated differently if one had a strong family history of osteoporosis and the other did not.
Although some measurement techniques offer advantages in precision or accuracy, BMD should be measured by the technique that is most readily available at the lowest cost, at least for initial decision making in individual patients. This is because the indications for the study are to identify women who will benefit most from pharmacologic intervention to reduce fracture risk (T score) and to identify women in whom additional diagnostic studies may be indicated (Z score). If the Z score is lower than −2 and the likelihood of secondary causes of bone loss is increased, it would seem prudent to re-measure BMD at more than one site because short-term follow-up BMD may be needed. Patients with low Z scores should be screened for other conditions that lead to osteoporosis. A careful history and, when indicated, appropriate laboratory studies should be obtained to rule out hypercortisolism, alcohol abuse, and metastatic cancer. Patients should be screened for primary hyperparathyroidism, including serum parathyroid hormone, calcium, phosphorus, and alkaline phosphatase. Renal function tests should be obtained because secondary hyperparathyroidism is associated with chronic renal failure. Hyperthyroidism should be excluded by thyroid function tests. Blood count and smear, sedimentation rate, and protein electrophoresis are appropriate tests to screen for multiple myeloma, leukemia, and lymphoma, which can present with osteoporosis.
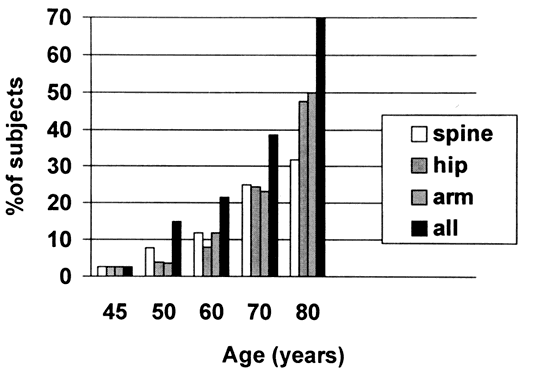
False-negative results occur with each method of measuring BMD; that is, some subjects have a normal T score at one site and a low T score at another. This is shown graphically in Figure 1, which is redrawn from the data of Melton and colleagues.12 In this figure, BMD measurement at three sites identifies more subjects with a low T score than any single site measurement. In the study reported, there was no apparent superiority of one site over any other. Alternatively, the clinician can order BMD measurement at other skeletal sites, or simply repeat the measurement using the original method after an interval of 2 years. The risk of fracture during that 2-year interval is small, and if the follow-up measurement is abnormal, intervention can be initiated at that time.
Indications for bone mineral density measurement
As shown in Figure 1, in the early postmenopausal years, BMD is normal in more than 80% of women; yet by 80 years of age, more than 50% of women have osteoporosis. Therefore, hormone replacement therapy (HRT) should be offered without reliance on BMD measurement. Women who accept a recommendation to begin HRT after counseling of the benefits and risks probably do not need a BMD measurement, although it has been suggested that women are more likely to agree to HRT if it can be demonstrated that the BMD is low.13, 14, 15 The act of measuring BMD may serve to reinforce the serious nature of osteoporosis; those women tested and found to have normal BMD measurements are more likely to agree to HRT than their nontested counterparts.13, 15 BMD measurement should be an integral part of the menopausal evaluation in women who are unwilling to begin HRT after full counseling on its benefits and risks and in women with an absolute or relative contraindication to HRT because there are alternative therapies for bone loss.
Biochemical markers of bone remodeling
Bone is lost when resorption exceeds formation; these processes can be assessed by a number of biochemical studies (Table 3). Because of individual variability, the relation between any of these markers and BMD is poor, although statistically significant, and these biochemical tests are inadequate to diagnose low BMD. The markers, however, are useful in determining the efficacious dose of HRT both at initiation of therapy and at follow-up. A favorable biochemical response (i.e., values on treatment are within the premenopausal reference interval) after 3 months of HRT indicates adequate skeletal effect at a specific dose. Conversely, if the biochemical response is suboptimal, an increased dose is indicated. Changes in the biochemical markers can be identified 4–12 weeks after initiating therapy.16 This approach can be particularly helpful when initiating HRT in older women who are more removed from menopause and more concerned about short-term side effects because a lower dose may yield optimal skeletal protection. When the HRT dose is effective for bone mass preservation, each of the markers should be within the premenopausal reference interval after 3 months of therapy. Measurement of biochemical markers is also useful in evaluation of patients already using HRT. A low BMD measurement in a woman with biochemical markers in the premenopausal range is more likely to reflect that the BMD was low before initiation of HRT and less likely to indicate that her current dosage is ineffective. In these patients, increased doses of estrogen or additional therapy with an antiresorptive drug (discussed later) are unlikely to provide further benefit.
Table 3. Biochemical markers of bone remodeling
Markers | Reference/Interval |
Bone resorption | |
Lysylpyridinoline (LP) | 24–52 nmol Pyd/mmol Cr |
Deoxylysylpyridinoline (Dpd) | 2.5–6.2 nmol Dpd/mmol Cr |
Urinary C-telopeptide of collagen cross-links (PICP) | 13–96 nmol/mmol |
Urinary N-telopeptide of collagen cross-links (uNTX) | 5–79 nmol/mmol creatinine based on 95% CI |
| Serum C-telopeptide of type 1 collagen (sCTX) | 40–465 pg/mL |
| Serum N-telopeptide of type 1 collagen (sNTX) | |
Bone formation | |
Bone-specific alkaline phosphatase (BSAP) | 11.6–30.6 BAP, U/L |
Carboxy-terminal propeptide of type 1 collagen (P1CP) | 45–190 μg/L |
N-terminal propeptide of type 1 collagen (P1NP) | 21–85 ng/mL |
| Osteocalcin | 7.0–27.9 ng/mL |
Pyd, pyridinoline; Cr, creatinine; CI, confidence interval; BAP, bone-specific alkaline phosphatase.
Markers of resorption respond earlier than the serum-based markers of formation (4 weeks versus 12 weeks), and this makes them the preferred investigation in some clinical circumstances. Decreased N-telopeptide of collagen cross-links (NTX) was seen after only 2 weeks of HRT. Eighty-eight percent of women who experience at least a 30% decrease in NTX from baseline to 6 months had the same or better BMD measurements compared to 57% of those women who had less than a 30% decrease.17 The serum-based markers of bone formation are not affected by diurnal variation, unlike the resorption markers, therefore, unless follow-up is required after just 4 weeks of HRT rather than after 3 months, formation markers may be preferable. Two studies16, 18 have clearly demonstrated the clinical utility of these markers in patients who are candidates for estrogen replacement therapy (ERT). In cases in which markers of bone turnover were lowest, there was minimal further loss of bone when only calcium supplementation was given, and only minimal gain in BMD on ERT. At the other end of the spectrum, women with the highest rates of remodeling assessed by these markers lost substantial bone when only calcium was provided and gained significant bone on ERT.
PREVENTION AND TREATMENT
Osteoporosis is more effectively prevented than treated. The primary goal of prevention is to achieve as high a peak bone mass as genetically possible and to reduce the rate of bone loss (in aging). Peak bone mass is to a great extent genetically determined. Optimizing peak bone mass can be accomplished only with proper nutrition, sufficient weight-bearing activity, and minimization of risk factors (e.g., smoking, excessive alcohol use, immobilization). The average calcium intake in the United States is below the recommended daily requirement, and physician-directed education during childhood and adolescence may encourage improved calcium intake with resultant increased bone density and protection against osteoporosis in later life. Adults require at least 1200 mg/day of elemental calcium. Although adequate calcium intake and exercise are critical for building bone, they are inadequate for prevention of bone loss in estrogen-deficient states.
Controlled clinical trials have now demonstrated that the combination of supplemental calcium and vitamin D can reduce the risk of fracture.19, 20 Providing adequate daily calcium and vitamin D is a safe and inexpensive way to help reduce fracture risk. The National Osteoporosis Foundation (NOF) recommends an intake of 800–1000 international units (IU) per day for adults over age 50. Elderly people are frequently deficient in vitamin D and may require greater amounts of supplementation. While the safe upper limit for vitamin D intake for the general adult population was set at 2000 IU per day,21 recent evidence indicates that intakes as high as 10,000 units per day are safe and that some elderly patients will need at least 2000 IU daily to maintain optimal vitamin D levels.22
BMD and clinical risk factors predict hip and other osteoporotic fractures, and the combination of the two provide higher specificity and sensitivity than either alone.23 The clinical risk factors include body mass index (BMI, as a continuous variable), a prior history of fracture, a parental history of hip fracture, use of oral glucocorticoids, rheumatoid arthritis and other secondary causes of osteoporosis, current smoking, and alcohol intake (3 or more units daily). The WHO has developed a fracture risk assessment tool (FRAXTM) based on the use of these clinical risk factors with or without bone mineral density tests which predicts the 10-year risk of fracture in men and women.24 The model suggests that osteoporosis treatment was cost-effective when the 10-year probability of hip fracture reached 3%. This tool is currently available online at http://www.shef.ac.uk/FRAX/ for patients and clinicians. The NOF applied the FRAX model to previously untreated individuals in formulating guidelines for cost-effective treatment. They concluded that it is cost-effective to treat individuals with a prior fracture and those with DXA femoral neck (or total hip) T scores of <−2.5. Previous analyses have established that a spine T score <−2.5 also warrants treatment. The US-adapted WHO algorithm is most helpful in identifying patients with low bone mass (T score −1.0 to −2.5) most likely to benefit from treatment. Unfortunately, spinal bone mass has not been validated and is not included in the algorithm, so the algorithm does not address the patient with low bone mass at the spine and risk factors but normal bone mass at the hip.25 Clinical judgment remains important regardless of which algorithm is used to determine treatment.
PHARMACOLOGIC THERAPY
Agents currently approved by the FDA in the United States for prevention or treatment of osteoporosis are classified as antiresorptive agents or anabolic agents. Antiresorptive agents inhibit osteoclastic function and reduce the frequency of the remodeling units within bone. To some extent, they may also inhibit osteoblast function and have the potential for also inhibiting formation of new bone, and it is the ratio of the effect on osteoclast to osteoblast that determines whether the net balance will be an increase or decrease in BMD. Antiresorptive agents have been shown to increase BMD by laying down new bone on the surface of existing bone, but do not create new trabeculae within gaps left by prior resorption. Thus, their ability to repair bone left weakened by prior resorption has limitations. Anabolic agents, on the other hand, appear to "fill in the gaps" in bone which has sustained structural weakness and are associated with some of the most dramatic increases in BMD of the osteoporotic agents.
Table 4 shows the agents which are currently approved by the FDA in the US for prevention or treatment of osteoporosis. Additionally, there are several new agents which are in phase III trials and which may be available within the next several years.
Table 4. Osteoporosis agents approved by the FDA
| Indication | Fracture reduction | ||||
| Prevention | Treatment | Lumbar spine | Hip | Other | |
| Antiresorptive agents | |||||
| Alendronate | + | + | + | + | + |
| Risedronate | + | + | + | + | + |
| Ibandronate | + | + | + | − | − |
| Zoledronate | + | + | + | + | + |
| Raloxifene | + | + | + | − | + |
| Estrogen | + | − | + | + | + |
| Calcitonin | − | + | + | − | − |
| Anabolic agents | |||||
| Teriparatide | − | + | + | + | + |
Hormonal therapy
ESTROGEN
Bone metabolism changes dramatically when estrogen production declines in women. There is evidence for an increase in activation frequency of new bone remodeling units and an increase in remodeling imbalance. The remodeling imbalance results from increased osteoclastic activity, possibly coupled with impairment of osteoblastic activity. Many different factors can affect osteoclast activity, but it appears that the receptor activator of nuclear factor kappa B (RANK) ligand (RANKL) is required to mediate or permit their effects on bone resorption. Osteoprotegrin (OPG) appears to mediate bone formation and to antagonize the actions of RANKL. Emerging research indicates that estrogen replacement therapy (ERT) may inhibit RANKL and stimulate the expression of OPG.26, 27 Estrogen deficiency leads to increased osteoblast apoptosis while increasing osteoclast number, activity and lifespan, resulting in a negative balance in bone remodeling. Because unopposed ERT results in an increased incidence of endometrial hyperplasia and carcinoma in women with intact uteri, estrogen almost always is administered in conjunction with progestogen. The term hormone replacement therapy (HRT) is used when discussing combined estrogen and progestogen therapy. Progestogen does not counteract the beneficial influence of estrogen on bone metabolism. Whether progestogen offers any independent benefit on BMD is controversial and is discussed later.
HRT has a positive effect on BMD at all skeletal sites. In the Womens' Health Initiative (WHI), total hip BMD increased a mean of 1.7% during the first year of estrogen-plus-progestin treatment and improved by 3.7% by year 3 compared with a loss of 0.44% at year 1 and a 0.14% improvement at year 3 in the placebo group (p <0.001), although adherent women had a 7.6% increase in the lumbar spine and 4.5% increase in the total hip by the third year of therapy. By year 6, the percentage increase in lumbar spine BMD was 7.5% in women in the estrogen-plus-progestin group compared with 2.6% in the placebo group.
In addition to preservation of bone mass, estrogen use is associated with a reduced prevalence of osteoporotic fractures.28 In one study, women taking HRT had less than half the fractures of the placebo group, with the most apparent decline seen after 5 years of therapy or longer.29 The preservation of bone mass and reduction in fracture incidence are less clearcut for the proximal femur than for the forearm or spine; some studies suggest that higher doses may be needed to obtain similar responses at the hip.30 Women initiating therapy within 2 years of the onset of menopause have optimal reduction in fracture risk,31 but the Rancho Bernardo Study demonstrated nearly equivalent results in women currently taking HRT who initiated therapy after 60 years of age.32 This also supports the concept that maximal increases in BMD occur during the first few years of ERT, then show a trend toward stabilization or slow decline.33 The WHI demonstrated that even women without significant reduction in bone mass may benefit from the effect of HRT on fracture rate. Women taking HRT had a reduction in all fractures (hazard ratio [HR] 0.76; 95% confidence interval [CI] 0.69–0.83). HRT reduced the risk of hip fracture by 33% (HR 0.67; 95% nCI 0.47–0.96; 95% aCI 0.41–1.10). Women taking HRT who reported a baseline calcium intake of more than 1200 mg/day experienced a 60% reduction in the risk of hip fracture with no reduction seen in women with lower calcium intake (p for interaction = 0.02).28
There is considerable uncertainty about the optimal duration of HRT from a skeletal perspective. The WHI demonstrated a difference in fracture rate between HRT and placebo groups which began to develop soon after randomization.28 Other studies have suggested that it may take as long as 7–10 years of treatment to reduce risk. In the Rancho Bernardo cohort, past users had low BMD measurements, even when estrogen was taken for the same duration as current users, although the more recent the estrogen use in past users, the higher the BMD.32 Almost every study has suggested that estrogen therapy taken only at the time of menopause preserves bone during use but may not preserve bone density late in life34, 35 or protect against osteoporotic fractures.36, 37, 35, 38 The concept that continued HRT remains effective at advanced ages is supported by Cauley and associates,31 who reported that the risk for wrist fractures was reduced by 61% among current users but by only 19% among previous users of estrogen. Estrogen use between the ages of 65 and 74 years has been documented to protect against fractures.36 The average age of women in the WHI was 63 years with 44% of the women over 65 years of age at enrollment suggesting fracture protection at least a decade after menopause.28 Increased bone density has been reported in estrogen users up to the age of 80 years.39 The response appears to be greatest among women who are farthest from menopause and who have the lowest pretreatment bone mass.40, 41, 42 Lufkin and colleagues43 reported that estrogen therapy improved BMD by about 5% during the first year of therapy and reduced the rate of new fractures even in women with existing osteoporotic fractures. It is estimated that initiation of estrogen in women as old as 70 years could reduce the risk of fracture by about one third.44 As seen with estrogen use for prevention of osteoporosis, estrogen use in women with established osteoporosis has a greater effect on the BMD of the lumbar spine than on that of the hip.
Data concerning the skeletal response to stopping HRT also are controversial, with one study indicating that bone loss accelerates45 and another suggesting that bone loss is not accelerated.30 These two studies differed in important aspects, which probably minimizes the differences in their conclusions. One study initiated HRT at the time of bilateral oophorectomy with a more abrupt decrease in estrogen levels; the other included only women further from menopause. It is most likely that cessation of HRT restores the rate of bone loss to that occurring at the time therapy was initiated. Because it is well established that this rate of loss declines naturally with increasing time after menopause, the earlier HRT is started, the more rapid the anticipated loss when therapy is stopped.
A major factor in ensuring efficacy of any medication is the side effects, perceived or real, associated with the medication. The use of estrogen in HRT regimens is a particularly good example. Market research data indicate that up to one third of women do not fill their prescriptions for estrogen and that less than 40% of women who begin therapy continue it after 1 year.46 The reasons generally fall into two categories: the occurrence of vaginal bleeding and the fear of cancer.
Vaginal bleeding commonly is cited as a problem when therapy is used in asymptomatic women, especially in women more removed from menopause. This can often be avoided by providing continuous combined therapy in which the progestin is administered daily with the estrogen. Although some spotting and irregular bleeding is not uncommon during the first 6 months of therapy, about 90% of women are amenorrheic by 1 year after commencement of therapy. Studies support the safety of this regimen when used for 3–5 years.40, 47 Longer-term data are not yet available, and unexplained bleeding requires further evaluation.
The second most commonly cited concern is fear of cancer, particularly uterine and breast cancer. Because data suggest that sequential progestin therapy returns the level of risk of endometrial malignancy at least to that expected in the untreated postmenopausal population,48 providing accurate information regarding the protective effect of progestin usually reassures the patient. A more difficult issue is the concern about breast cancer. Both observational and prospective placebo-controlled trials have demonstrated an increase in the rate of breast cancer with HRT49, 50 with no increase observed in estrogen only treated women.50, 51 The problem remains that the data are far from clear. While combined hormone therapy may increase the risk of breast cancer, the magnitude of that increase is less than that of other established risk factors, and no studies have shown an increase in mortality from breast cancer in estrogen-treated women. Risk of stroke increased with both HRT and ERT, while deep vein thrombosis (DVT) and breast cancer were increased with HRT. Because the results of the WHI suggested more harm than benefit from the use of HRT, it is currently reserved for symptomatic women and multiple organizations have issued statements limiting its use. The decision to use HRT or ERT remains a risk/benefit analysis determined on an individual basis, but estrogen remains one of the most cost effective ways to reduce osteoporotic fracture. A forthright discussion of the issues usually allows most women to balance the benefits with the risks of therapy. Other nuisance side effects, such as water retention, bloating, and nausea, usually respond to use of another estrogen and progestin preparation.
A dose dependent effect on bone has been noted with generally higher doses required to increase BMD at the hip compared to the spine.52 Doses of oral conjugated equine estrogens (CEE) as low as 0.3 mg/day resulted in significant increases in BMD at both spine and hip. Transdermal estradiol doses of 0.025–0.1 mg/day all resulted in increases in BMD during a 2 year trial, with the largest effect noted at the highest dose.53 The minimal dose of estrogen required to preserve bone mass has been researched intensively. While older studies suggested that the minimal effective dose for maintenance of axial and peripheral bone mass was equivalent to 0.625 mg CEE, newer studies such as that above have suggested that lower doses of estrogen may be effective in maintaining and, in some cases, increasing BMD. A transdermal estrogen patch delivering very low doses of estrogen (i.e., 0.014 mg/day) increased lumbar spine BMD in women 60–80 years of age with T scores >−2.0 and maintained hip BMD over a 2 year study.54 Median plasma estradiol level in the estradiol group increased from 4.8 pg/mL at baseline to only 8.6 pg/mL at 2 years (p <0.001 versus baseline), which is still within the menopausal range. The rate of development of endometrial hyperplasia was not significantly greater at the end of 2 years with the unopposed estrogen patch. While these lower doses using various types and routes of administration of estrogen have been demonstrated to increase or maintain BMD, there are no data regarding their efficacy in reducing the risk of fracture. While they probably will be effective in this regard, reduction in fracture risk has only been demonstrated prospectively in the WHI in which CEE 0.625 mg/day was administered either alone or with a progestin.28 Observational population studies are in agreement, but have relied on traditional estrogen doses of CEE 0.625 mg/day or 17ß-estradiol 1–2 mg/day.55
Not all types of estrogen or estrogen preparations are equipotent in preserving bone mass. We have been able to find only one reference suggesting that estrone has bone-sparing potential,56 and estriol has no apparent role in skeletal protection in postmenopausal women.57 The most effective approach is to provide sufficient estrogen to ensure that the rates of bone remodeling assessed biochemically remain within the premenopausal range. Although intellectually less than ideal, this can be accomplished in most women by a single measurement of a biochemical marker after 3 months of therapy, with dose adjustment if needed. Again, these remarks refer only to skeletal protection and are untested in other areas that benefit from ERT or HRT.
Table 4. Estrogen only preparations available in the US and Canada
| Oral estrogens | Available dosages (mg) |
Conjugated estrogens (formerly known as conjugated equine estrogens) | 0.3, 0.45*, 0.625, 0.9, 1.25 |
| Synthetic conjugated estrogens, A | 0.3, 0.45, 0.625, 0.9, 1.25 |
| Synthetic conjugated estrogens, B | 0.3, 0.45, 0.625, 0.9, 1.25 |
| Esterified estrogens | 0.3, 0.625, 0.9, 1.25, 2.5** |
| 17β-estradiol | 0.5, 1.0, 2.0 |
| Estradiol acetate* | 0.45, 0.9, 1.8 |
| Estropipate (formerly piperazine estrone sulfate) | 0.625 (0.75 estropipate, calculated as sodium estrone sulfate 0.625), 1.25 (1.5), 2.5 (3.0) |
| Transdermal estrogens | Delivery rate (mg/day) |
| 17β-estradiol matrix patch | 0.025, 0.0375, 0.05, 0.075, 0.1 |
| 17β-estradiol reservoir patch | 0.05, 0.1 |
| 17β-estradiol transdermal gel | 0.003, 0.009, 0.0125, 0.027, 0.035 |
| 17β-estradiol topical emulsion | 0.05 |
| 17β-estradiol transdermal spray | 0.021 |
| Vaginal ring | 0.05, 0.1 |
* Available in the United States but not Canada
** Available in Canada but not in the United States
The role of repeat BMD measurements in women on HRT is uncertain. Some investigators believe that the potential for nonresponders at the standard dose justifies routine BMD measurement before initiation of HRT and within 2 years after its commencement. Also, studies indicate that compliance is improved with positive feedback in terms of the BMD response, and this may justify repeat measurements in selected patients but clearly not in most women. In women in whom BMD measurement has been an integral part of the initial decision not to begin HRT and in those with established osteopenia or osteoporosis, follow-up measurement after an interval of 2–3 years is appropriate.
PROGESTINS
It is controversial whether the addition of progestin modifies the skeletal response to estrogen, in part because the different types of progestin have different qualities and because their effects on bone and calcium metabolism must be studied separately. Only a few studies have been published on the individual effects of progestin on the postmenopausal calcium metabolism. Medroxyprogesterone acetate (MPA) given cyclically (10 mg/day or 20 mg/day for 12 or 15 days each month) or continuously (2.5 mg/day) with CEE did not increase spinal BMD over that seen with CEE alone;40, 58 even at high doses, MPA failed to reduce trabecular bone loss.59 MPA may be more effective in women with established osteoporosis; use of a combination of estrogen and MPA (5 mg MPA and 0.625 mg of conjugated estrogens daily) was associated with a 65% increase in spinal BMD measurements over those observed in osteoporotic women treated with unopposed estrogen.60 Norethindrone acetate, however, is associated with a greater dose-dependent increase in spinal BMD measurements than that seen with ethinyl estradiol alone, even in the absence of established osteoporosis.61 Norethisterone likewise increased bone mineral content when administered alone for 2 years,62 as did gestronol hexanoate.63 Because it lacks the cardioprotective features associated with estrogen compounds, progestin rarely is indicated alone for the prevention or treatment of osteoporosis. However, because of the increased incidence of endometrial carcinoma in women taking unopposed estrogen, progestin should be routinely used in women with intact uteri.
ANDROGEN
Androgen deficiency at menopause also may affect bone metabolism. Both androgen and estrogen receptors have been found in bone cells from both sexes, and positive correlations between bone mass and androgen levels have been observed in premenopausal and postmenopausal women. There are some data that, unlike estrogen therapy, androgens may stimulate bone formation.64 Although androgenic hormones generally are not used as single-agent therapy in women because of the potential for hirsutism as well as adverse cardiovascular effects, they may potentiate the effects of estrogen on BMD, particularly because HRT actually may reduce endogenous androgen production by reducing the high levels of luteinizing hormone and follicle-stimulating hormone that can stimulate androgen production by the climacteric ovary. Anabolic androgenic hormones, such as nandrolone decanoate, have been demonstrated to increase bone mass in postmenopausal women alone65, 66 and in combination with estrogen.67 Combination therapy of CEE with methyltestosterone demonstrated not only decreases in bone resorption markers but also increases in serum markers of bone formation, whereas CEE resulted only in reduction of the bone formation markers.68 These findings are in agreement with the greater increase in BMD of the spine and hip observed in women taking a CEE and methyltestosterone combination therapy than that seen in women taking CEE alone.69 Thus, in patients with established osteoporosis, combined androgen and estrogen therapy may be indicated until other therapies that stimulate bone formation become available.
Tibolone, a synthetic C-19 steroid with weak estrogenic, progestational, and androgenic properties, has been studied as an alternative to standard HRT. At doses of 1.25 mg/day and 2.5 mg/day, tibolone appears to be effective in increasing BMD and improving biochemical markers in postmenopausal women for prevention70, 71 and treatment72 of osteoporosis. One of the benefits of tibolone therapy is improvement of climacteric symptoms, seemingly without adverse effects on the endometrium or breasts.73 A large multicenter trial evaluating tibolone was able to demonstrate 45% reduction in new vertebral and 26% reduction in nonverbral fractures after 34 months. The tibolone group also had a decreased risk of invasive breast cancer (relative hazard 0.32; 95% CI 0.13–0.80) and colon cancer (relative hazard 0.31; 95% CI 0.10–0.96). However, the tibolone group had an increased risk of stroke (relative hazard 2.19; 95% CI 1.14–4.23; p = 0.02), for which the study was stopped. There were no significant differences in the risk of either coronary heart disease or venous thromboembolism between the two groups.29 Tibolone remains available in Europe but will probably not get US approval based on the above data.
Anabolic steroids, such as stanozolol, are effective in preserving bone mass in postmenopausal women,74 but the optimal dose has not been defined, and it is unclear whether this optimal dose is sufficiently free of unwanted androgenic side effects. Early studies of ipriflavone demonstrated maintenance of bone density at the spine as well as reduction in back pain in women with established osteoporosis, but bone mineral content of the hip was not significantly different in patients taking ipriflavone.75 One study has suggested a synergistic effect of ipriflavone in increasing forearm bone density when administered with a subtherapeutic dose (0.3 mg) of CEE for 1 year.76
Selective estrogen receptor modulators (estrogen agonists/antagonists)
Although HRT is the regimen of choice for many women because of its low cost and its concomitant utility in treating vasomotor symptoms and urogenital atrophy, other preventive and therapeutic options are available for women who cannot or will not take estrogen. Tamoxifen is a viable alternative to HRT as adjuvant therapy for breast cancer. Tamoxifen acts as an estrogen agonist on bone; increased lumbar BMD was demonstrated after 2 years of tamoxifen therapy,77, 78, 79 but the magnitude of increase was less than that typically observed with estrogen. This suggests that the degree of protection is not equivalent to that of estrogen, although long-term studies have yet to be completed. Because tamoxifen is associated with increased incidence of endometrial hyperplasia and carcinoma, there is little enthusiasm for its use for skeletal protection in circumstances other than breast cancer. Often, its use is limited to 5 years, which presents a major problem for younger women, in whom premature menopause resulted from the chemotherapy for the breast cancer. There is mounting evidence that HRT may be safe in selected women after successful treatment of breast cancer,80 but many breast cancer survivors, and their treating physicians, are reluctant to consider hormonal therapy.
Several antiestrogenic compounds are available or under development. These compounds are termed selective estrogen receptor modulators (SERMs) or estrogen agonists/antagonists because of their differential effects at various estrogen receptors. Raloxifene is approved for the prevention and treatment of osteoporosis and is administered orally as a 60 mg/day dose. Raloxifene is effective in preventing the development of carcinogen-induced rat mammary tumors. Although it is not as potent an antitumor agent as tamoxifen,81 it is effective in minimizing the risk of breast cancer in a subset of women and is FDA approved with this indication. In a short-term study, similar effects on markers of bone turnover as well as serum lipids were seen in women treated with raloxifene and estrogen, whereas no proliferative effect on endometrial histology was noted in the raloxifene-treated women.82 Raloxifene does not seem to increase BMD as much as some other antiresorptive therapies such as estrogen or bisphosphonates. In the MORE trials of raloxifene for osteoporosis, raloxifene resulted in a 2.6% increase in BMD of the lumbar spine and a 2.1% increase in hip BMD after 4 years of treatment. Raloxifene reduced vertebral fractures by 49% at 4 years.37 There was no reduction in nonvertebral fractures. Vertebral fracture reduction seems rapid and one study demonstrated a significant reduction in new fractures as early as 3 months after initiation.36 Raloxifene has also been shown to reduce invasive breast cancer in both low risk women83 and women at increased risk for breast cancer.84 It was equivalent to tamoxifen in reducing the incidence of invasive breast cancer, although, unlike tamoxifen, raloxifene did not reduce the incidence of carcinoma in situ.
Side effects from raloxifene include increased vasomotor symptoms, which seem less troublesome to those women not initially symptomatic, muscle cramps, DVT, and development of uterine polyps.85 Raloxifene does not increase the incidence of myocardial infarction, stroke, uterine cancer, endometrial hyperplasia, ovarian cancer or postmenopausal bleeding. The incidence of DVT is approximately equivalent to that observed in women on estrogen therapy. Raloxifene has a generally favorable impact on lipids similar to that seen with estrogen, but unlike estrogen does not seem to raise either triglycerides or high density lipoprotein (HDL) levels.
Several other selective estrogen receptors are currently in clinical trials and may be more active on bone remodeling than the currently available SERMs.
Bisphosphonates
The bisphosphonates share a common P-C-P structure and are analogs of pyrophosphate; they differ in their side chains, imparting differences in potency and skeletal effectiveness and toxicity. Bisphosphonates bind avidly to hydroxyapatite, inhibit osteoclastic bone resorption, and are then buried within the skeleton, where they may remain for 10–12 years or longer. Bisphosphonates also have the potential to inhibit mineralization of bone in a dose-dependent manner. The ratio of the dose that inhibits resorption to the dose that inhibits mineralization determines the clinical usefulness of one bisphosphonate over another. Nearly all bisphosphonates that have been evaluated have been shown to prevent bone loss effectively. The first-generation bisphosphonate etidronate (Didronel) has been studied in several controlled trials and causes a small increment in bone mass, compatible with a reduction in activation frequency, with some reduction in fracture occurrence during the initial 2 years of study.86, 87 Long-term studies with etidronate have been open-label, however, and although they demonstrated its continued effectiveness in increasing BMD and reducing vertebral fracture rate,88 verification of these data from placebo-controlled trials is lacking. Concerns about a mineralization defect with longer-term etidronate therapy did not materialize when the drug was administered intermittently (2 weeks on therapy, 12 weeks off),89 which was done in the controlled trials. Initial studies indicate that combined HRT and etidronate therapy may result in greater increases in BMD than are seen with either therapy alone, with no evidence of osteomalacia.90
The second- and third-generation bisphosphonates do not impair bone mineralization at doses that maximally inhibit bone resorption and are not associated with osteomalacia, effects that have been attributed to etidronate. Alendronate sodium (Fosamax), a potent aminobisphosphate, is approved for the prevention of postmenopausal osteoporosis and the treatment of postmenopausal osteoporosis, glucocorticoid induced osteoporosis, osteoporosis in men, and Paget's disease. In women with postmenopausal osteoporosis, alendronate reduced markers of bone remodeling and increased BMD at the spine, hip, and total body, progressively during 3 years of therapy at doses of 5 and 10 mg daily.91, 92, 93 In addition, the increased bone mass was associated with a 48% reduction in the proportion of women with new vertebral fractures and with a reduction in the number of new vertebral fractures, the progression of vertebral deformity, and height loss.94 Only 8% of women with at least one vertebral fracture at baseline treated with alendronate experienced a subsequent vertebral fracture, compared with 15% of the placebo-treated group. In women with two or more fractures at baseline, the difference was even greater, with 90% fewer subsequent vertebral fractures in alendronate-treated women. Alendronate also reduces nonvertebral fractures,95 with about a 50% reduction in the incidence of hip fractures.94, 95
Alendronate has been used for as long as 10 years in one clinical trial with continued increases in BMD at both spine and hip. Patients completing the 5 year Fracture Intervention Trial (FIT) were either continued on alendronate or switched to placebo for an additional 5 years. Compared with continuing alendronate, switching to placebo resulted in declines in BMD at the total hip (−2.4%; 95% CI −2.9% to −1.8%) and spine (−3.7%; 95% CI −4.5% to −3.0%), but mean levels remained at or above pretreatment levels 10 years earlier with nothing to suggest a phase of accelerated loss. Similarly, those discontinuing alendronate had increased serum markers of bone turnover compared with those continuing alendronate.96 While no difference in nonvertebral fractures could be observed in the small number of patients completing the trial, there was a significant reduction in clinically recognized vertebral fractures (5.3% for placebo and 2.4% for alendronate; RR 0.45; 95% CI 0.24−0.85) when compared to women who were switched to placebo. Alendronate is also approved for prevention of osteoporosis. At a dose of 5 mg daily, alendronate resulted in significant increases in BMD of the spine, hip, and total body in women without established osteoporosis.97 Daily and weekly alendronate have been shown to be equally effective.98 Alendronate is now available both with and without vitamin D (2800 or 5600 IU/week).
Several other bisphosphonates, including risedronate, ibandronate, and zoledronic acid, are also approved for treatment of osteoporosis. Risedronate is available as a daily (5 mg/day), weekly (35 mg/week), or monthly agent (150 mg/month or two consecutive doses of 75 mg/month) and is approved for both prevention and treatment of postmenopausal and glucocorticoid induced osteoporosis. Bone mineral density increased significantly in patients treated with risedronate compared with placebo at the lumbar spine (5.4% vs 1.1%), femoral neck (1.6% vs −1.2%), femoral trochanter (3.3% vs −0.7%), and midshaft of the radius (0.2% vs −1.4%) during a 3 year trial. Bone formed during risedronate treatment was histologically normal. Risedronate resulted in a significant decrease with a 41% reduction the incidence of new vertebral fractures over 3 years (11.3% vs 16.3%), with protection noted as early as the first year of treatment (fracture rate 2.4% vs 6.4%).99 The incidence of nonvertebral fractures over 3 years was reduced by 39%, but was not reduced at the hip in the pivotal trial. In the Hip Intervention Program (HIP) trial, risedronate was compared to placebo in women who were older and/or had worse osteoporosis than in the prior trial. The incidence of hip fracture among women treated with risedronate was 2.8%, as compared with 3.9% among those assigned to placebo (relative risk 0.7; 95% CI 0.6–0.9), with the greatest effects noted in women with the lowest T scores. Women treated only based on age and risk for fracture did not have a reduction in their fracture risk in this trial, confirming that antiresorptives are most effective in preventing fracture when the BMD is low.100
Ibandronate is available as a daily (2.5 mg/day) or monthly (150 mg/month) oral agent and is approved for prevention and treatment of osteoporosis. There is also an injection (150 mg) which is approved for use every 3 months for the treatment of osteoporosis. In a 3 year clinical trial, daily treatment with oral ibandronate 2.5 mg resulted in a significant increase in BMD of the lumbar spine (6.5%) and hip, with normalization of bone turnover and less height loss than in the placebo group. The incidence of vertebral fractures was reduced in patients receiving ibandronate by 62%, however, no reduction in nonvertebral fractures was observed. In a higher-risk subgroup (femoral neck BMD T score <−3.0), the risk of nonvertebral fractures was reduced by 69%.101 Monthly treatment with oral ibandronate 150 mg resulted in greater increases in lumbar spine BMD (6.4%) after 2 years of treatment compared to daily ibandronate 2.5 mg (4.8%); a similar finding was seen at the hip, where monthly ibandronate resulted in greater increases in BMD than did daily dosing102 Ibandronate has also been shown to decrease bone turnover and maintain or increase BMD in the spine and proximal femur in women without osteoporosis.103
Clinical use of bisphosphonates has been associated with esophagitis and upper gastrointestinal symptoms such as abdominal pain and flatulence. No significant differences in the frequencies of such disorders were generally noted between the various bisphosphonates and placebo groups during their trials, however, there are many clinical reports of such symptoms in postmarketing studies. Comparative trials between the various bisphosphonates have been published which have demonstrated variable results depending on the methodology used in the study and the patient population. In practice, there probably is little difference between the various agents. Regimens with less frequent administration generally result in significantly fewer gastrointestinal side effects, and weekly regimens have resulted in much better patient tolerance. It remains controversial whether there is further improvement by using monthly regimens. Patients intolerant to one medication may sometimes have better tolerance to another drug within the class, in spite of the fact that the side effects seem to be common to the class. Regardless of which drug is prescribed, careful attention to dosing instructions may reduce the risk of esophageal irritation. Women using bisphosphonates should be instructed to take the medication with at least 100 mL of water and not to lie down during the next 30 minutes. If patients complain of irritation, a drug holiday may allow the drug to be reinstituted, however, care must be taken because rare reports of ulcerations and strictures have occurred.
Although oral bisphosphonates remain the primary treatment of osteoporosis, they may be contraindicated because of clinical concerns such as upper gastrointestinal disease or inability to remain upright for the required length of time after administration. For this reason, there has been much progress toward development of alternative routes of administration for the bisphosphonates. At this time, two intravenous (IV) bisphosphonates are approved for the treatment of osteoporosis, ibandronate and zoledronate.
Intravenous ibandronate 3 mg was administered every 3 months in a trial comparing its efficacy to 2.5 mg daily oral ibandronate in a 2 year trial. The IV regimen resulted in a greater increase in the lumbar spine BMD compared with the daily regimen (6.3% vs 4.8%; p <0.001). Greater increases were also obtained with IV ibandronate versus daily in proximal femur BMD. Serum bone turnover markers were reduced to a similar extent. The tolerability profile of the IV regimen was similar to that observed with daily oral therapy.104 Because the study was designed to compare efficacy to the FDA approved oral therapy, it was not powered to evaluate the efficacy of the IV drug in preventing osteoporotic fractures and its efficacy of fracture reduction must be extrapolated from the data obtained in the trials of the oral agent. Thus, we assume efficacy in reducing vertebral fracture but no reduction in nonvertebral fractures.
Zoledronic acid has been utilized for malignancy related bone disease, but was recently approved for the treatment of osteoporosis. In a 3 year trial, IV zoledronic acid reduced the risk of morphometric vertebral fracture by 70%, as compared with placebo (3.3% in the zoledronic-acid group vs. 10.9% in the placebo group; relative risk 0.30; 95% CI 0.24–0.38) and reduced the risk of hip fracture by 41% (1.4% in the zoledronic-acid group vs. 2.5% in the placebo group; hazard ratio 0.59; 95% CI 0.42–0.83). Nonvertebral fractures, clinical fractures, and clinical vertebral fractures were reduced by 25%, 33%, and 77%, respectively (p <0.001 for all comparisons). Zoledronic acid was also associated with a significant improvement in BMD with approximately 6% increases at both hip and spine. Bone turnover markers were all significantly reduced. Adverse events, including change in renal function, were similar in the two study groups. However, serious atrial fibrillation occurred more frequently in the zoledronic acid group (in 50 vs. 20 patients, p <0.001).105
Treatment with intravenous bisphosphonates has been associated with renal toxicity manifested as deterioration of renal function and in rare cases acute renal failure. For this reason, patients treated with IV bisphosphonates should have serum creatinine measured prior to each infusion. Most IV bisphosphonates require rather long infusion times, with zolendronate requiring infusion times of 15 minutes. Ibandronate injection has not been associated with renal toxicity in patients with estimated glomerular filtration rate >30 ml/min and with no known diabetes mellitus or hypertension and may be injected over an interval of 15–30 s. Both IV bisphosphonates may result in an acute phase reaction with flu-like symptoms occurring within 24 hours of the infusion and lasting 2–4 days. While uncommon in general, this complication is seen most often after the first course of therapy.
Osteonecrosis of the jaw (ONJ) associated with the use of bisphosphonate therapy has been reported. ONJ is defined as an area of exposed bone in the maxillofacial region that does not heal within 8 weeks after identification. The condition is uncommon in the general population but risk factors include poor dental hygiene, gingivitis, periodontal disease, temperomandibular joint disease, bone tumors or metastases, and exposure to radiation therapy. The risk of ONJ associated with oral bisphosphonate therapy for osteoporosis seems to be low, estimated between 1 in 10,000 and <1 in 100,000 patient-treatment years. However, because ONJ is probably underdiagnosed and under reported, the true incidence may be higher. The risk of ONJ in patients with cancer treated with high doses of intravenous bisphosphonates is much higher, in the range of 1–10 per 100 patients. These patients have usually been treated with high doses of intravenous bisphosphonates, so it is unlikely that the intravenous agents used for treatment of osteoporosis will result in similar rates of ONJ. Because this is an evolving concern, patients should be informed about the condition and the fact that it appears to be rarely an issue for patients treated for osteoporosis. Patients who are starting or continuing to take bisphosphonates should be encouraged to practice good oral hygiene and have regular dental visits during which they can receive proper dental care.106
Although bone biopsies have been performed in small number of patients on long-term alendronate and did not show any abnormalities in bone remodeling, it has been suggested that long-term use may predispose to excessive inhibition of bone remodeling. Case reports have suggested that there may be markedly suppressed bone formation, with reduced or absent osteoblastic surface107 and it remains controversial whether this could result in atypical fractures.108 For this reason, it is particularly important to ensure that these agents are not started inappropriately but reserved for women at increased risk for fracture. Since there are few comparative studies that have demonstrated real differences between the various agents, individual patient tolerance, cost, and availability remain the principal means of selecting one bisphosphonate over another.
Calcitonin
Calcitonin, a polypeptide hormone produced by the parafollicular C cells of the normal human thyroid, inhibits bone resorption in pharmacologic doses, although the precise physiologic role of this hormone in humans is unknown. Synthetic calcitonin from several species (salmon, eel, human) has been extensively studied as a potential therapy for osteoporosis and has been found to be both effective and safe. The salmon hormone appears to be the most potent and is the most widely used. Most of the studies have been conducted in patients with established osteoporosis, but there is little reason to believe that it is not equally effective in preventing early postmenopausal bone loss.109 Until recently, this drug was available only in a parenteral (subcutaneous injection) formulation (Calcimar, Miacalcin), limiting its appeal for prophylaxis against bone loss. The recent availability of an intranasal salmon calcitonin spray (Miacalcin nasal spray, 200 U/day, Fortical) has resulted in the reappraisal of this drug for prevention of early postmenopausal bone loss, and it is a viable alternative to HRT in women who cannot or will not take HRT. Because the only current indication for calcitonin in postmenopausal women is the inhibition of bone loss, use of this drug should be limited to women in whom BMD measurement has demonstrated increased risk of fracture. Overall, the gains in bone mass seen with calcitonin therapy do not appear to be as great as those seen with the bisphosphonates, and the data on antifracture efficacy are less comprehensive.110, 111, 112 As noted with other antiresorptive drugs, the women with the lowest baseline bone mass demonstrate the greatest response to calcitonin therapy.111 One study113 has reported some synergy with respect to increases in BMD when eel calcitonin and HRT were given in combination. Combining calcitonin with the anabolic steroid nandrolone decanoate does not result in further increases in BMC, however; rather, it yields results inferior those seen with either drug used alone.114
There is abundant anecdotal evidence from many physicians, and supporting documentation from some controlled clinical trials,115, 116 that calcitonin has analgesic properties when administered during the acute phase after osteoporotic vertebral fracture. Calcitonin stimulates endorphin synthesis and release,117 which is the putative mechanism for its analgesic properties. It remains unclear why this analgesic effect is seen only with acute fracture pain and not in the chronic back pain associated with vertebral fractures. The analgesic effect has not been documented after femoral neck fractures. There are almost three decades of clinical experience with calcitonin (mainly for treatment of Paget's disease), and the long-term safety of this drug is well established. It is easier to take as a nasal spray than alendronate and has an important role for many patients. An abstract reported on the use of an inhaled form of calcitonin,118 which should expand the potential for its use.
Anabolic therapies
TERIPARATIDE
Recombinant human parathyroid hormone (PTH 1-34; Teriparatide) is the only anabolic agent currently approved for the treatment of osteoporosis. Analogous to the differential effect seen with intermittent or long-acting gonadotropin releasing hormone (GnRH) on reproductive hormones, administration of PTH as a single daily injection results in increases in BMD and reductions in fracture, whereas secretion of PTH from parathyroid adenomas results in bone loss. PTH treatment initially stimulates new bone formation and subsequently stimulates bone resorption and bone formation, with the balance still positive for bone formation even in the latter phase. The growth of new bone with teriparatide permits restoration of bone micro-architecture, including improved trabecular connectivity and enhanced cortical thickness. There may also be induction of new bone on the outer periosteal surface of bone, possibly affecting bone size and geometry, resulting in increased bone strength. For patients with the most severe osteoporosis, these characteristics may result in a greater reduction in fracture risk compared to that of the antiresorptives, though there are no comparative studies of fracture efficacy at this time. Trials of teriparatide in osteoporotic women utilized teriparatide, 20 or 40 μg, or placebo by daily subcutaneous injection. After a median treatment period of 19 months, teriparatide increased spine BMD by 9.7% (20 μg dose) and 13.7% (40 μg dose). Hip BMD increased, with a small decline in BMD seen in the radius, which was significant only at the higher dose. For this reason, the 20 μg dose was chosen for release. At this dose, vertebral fractures were reduced by 65%, with an absolute fracture risk of 5% compared to 14% in placebo treated women. Nonvertebral fractures were reduced by 40%; though the number of hip fractures was too small to evaluate statistically, there were fewer numerically.119
The study was stopped early because of an observed increase in osteogenic sarcomas in animal studies; however, this is not thought to be relative to humans. In patients with endogenous hyperparathyroidism or parathyroid cancer, there is no evidence of an increased risk for osteogenic sarcoma. There has been one reported case of osteogenic sarcoma in a patient treated with teriparatide but the incidence of less than 1:250,000 is lower than the background incidence in the community. Side effects of teriparatide include redness and irritation at injection sites, dizziness, leg cramps, nausea, arthralgias, weakness, and hypercalcemia, although only leg cramps and hypercalcemia were statistically different from placebo. Although transient increases in serum calcium were common when measured within 6 hours of the PTH injection, sustained increases were seen in only 3% of patients. Leg cramps were noted by 3% of teriparatide treated patients compared with 1% of placebo treated women.
Teriparatide induced BMD changes were not dependent upon patient age, baseline BMD, or prior fracture history, but were related to baseline biochemical bone turnover markers. Optimal duration of treatment is still under investigation. Treatment with teriparatide is approved for 2 years because the bone turnover markers suggested no further increase in bone formation after about 18 months. A series of observational studies suggests that an antiresorptive must be given after cessation of PTH because BMD begins to fall soon after discontinuation, whereas antiresorptive therapy can maintain PTH-induced BMD gains or even provide further incremental increases. Repeat administration of PTH was administered to a small group of women treated with concomitant PTH and alendronate 1 year after discontinuation of PTH. The rechallenge resulted in similar changes in bone turnover markers and BMD seen during the first course of therapy.
One of the most powerful agents available for the treatment of osteoporosis today, the use of teriparatide is limited secondary to its cost and also to the need for daily injection. Good candidates for teriparatide are those individuals who have had vertebral compression fractures, other osteoporotic related fractures with a low BMD, or those who have very low BMD, even in the absence of fractures. It is also appropriate for those individuals who have had a suboptimal response to other treatment regimens, including fracture on therapy, continued bone loss, or persistent osteoporosis.
SODIUM FLUORIDE
Sodium fluoride has been studied extensively for more than 35 years for the treatment of osteoporosis and is available for this purpose in many countries. As a nonpatentable compound, the larger pharmaceutical companies have been appropriately reluctant to invest the time and money needed to complete pharmacologic studies with sodium fluoride, which has hampered the development of this drug for clinical use. Pak and associates120, 121 completed a series of controlled clinical trials with a slow-release preparation of sodium fluoride (Neosten). Unlike ERT, the bisphosphonates, and calcitonin, sodium fluoride directly stimulates bone formation rather than inhibiting bone resorption. Accordingly, gains in bone mass are generally greater than those seen with antiresorptive drugs, and the increment appears to be linear and continuous over many years. This is in contrast to the antiresorptive drugs, with which most of the gains in bone mass are seen in the first year of therapy. A particular concern with sodium fluoride is that it accumulates in the skeleton and, when allowed to accumulate to excess, results in a paradoxical decrease in bone strength. This important side effect is dose and duration dependent. The seeming advantage of slow-release preparation is the delayed intestinal absorption, low peak serum levels, and lesser skeletal accumulation than were reported with earlier studies of plain sodium fluoride. Nonetheless, the drug must be administered intermittently, with a 2-month drug-free interval at the end of each year of therapy. As with all therapies that result in bone gain, it is imperative that the patient ingest enough “bone substrate” (calcium) to form properly mineralized bone. This is particularly important for fluoride therapy122 because osteomalacia has been reported in some patients who do not ingest enough calcium. On the horizon are newer preparations of fluoride, particularly a slow-release preparation of monosodium fluorophosphate, which appears to have a safety and efficacy profile similar to that of the slow-release preparation of sodium fluoride.123
Developing therapies
DENOSUMAB
The process of bone remodeling is mediated through RANKL as discussed in an earlier section of this chapter. RANKL binds to its receptor, RANK, on the surface of precursor and mature osteoclasts and stimulates these cells to mature and resorb bone. It is physiologically inhibited by osteoprotegerin (OPG), a soluble receptor that competes with RANK for binding to RANKL, thus neutralizing the effects of RANK. Many basic science studies have demonstrated that inhibition of RANKL leads to improved bone architecture and increased bone density and strength. Both estrogen and the SERM raloxifene stimulate OPG synthesis and there is evidence that estrogen may reduce RANK activation. A human monoclonal antibody to RANKL, Denosumab, is undergoing phase III trials with excellent results with respect to BMD, and also with respect to fracture reduction. FDA approval is likely in the near future. Preclinical studies in cynomolgus monkeys demonstrated similar effects to OPG on bone density and turnover, with differing mechanism of action for Denosumab from that of bisphosphonates. Denosumab binds with high affinity and specificity to RANKL, inhibiting osteoclast-mediated bone resorption as well as osteoclast maturation and survival, whereas bisphosphonates bind to the mineralized surface of bone and reduce osteoclast activity. In patients treated with Denosumab 60 mg every 6 months for 2 years, the lumbar spine BMD increased by 6.5% compared to –0.6% for placebo. BMD increases with Denosumab were rapid, with significant increases compared with placebo observed as early as month 1. Total hip BMD increased 3.4% compared to –1.1% with placebo.124 Markers of bone resorption were rapidly reduced by Denosumab treatment. Clinical fractures occurred in 4% of placebo group and in 1% of the Denosumab group. The overall incidence of adverse events over 24 months was similar between Denosumab and placebo groups; however, Denosumab resulted in a greater number of serious adverse events, primarily infections resulting in hospitalization. The types of infections were common to the subject population and were not thought to be related to the study drug; however, further studies will be required to ensure that there are not adverse effects on the immune function of patients receiving Denosumab.
Discontinuation of Denosumab was investigated in a group of patients receiving Denosumab 210 mg every 6 months for 24 months during a preliminary trial. Discontinuing therapy resulted in a decrease in BMD within the first year of discontinuation comparable to the gains in BMD with 24 months of Denosumab treatment. Bone loss then plateaued at values near baseline by the end of the fourth year. Biochemical markers of bone turnover increased after discontinuation of Denosumab to values above baseline and greater than those of the placebo group but returned to baseline within 24 months, suggesting no long-term residual effect.125 At this time, Denosumab appears promising as an alternative to existing osteoporosis therapies and may result in fewer side effects than current agents.
EMERGING THERAPIES
The need to develop drugs that stimulate bone formation and thereby elevate bone mass (anabolics), as opposed to preventing bone loss (antiresorptives), has opened new research areas for therapeutic intervention in the treatment of osteoporosis. There is increasing understanding of the underlying mechanisms which regulate osteoblast proliferation and differentiation. One of the areas intensively researched is the Wnt pathway. Alterations in this pathway have been associated with bone disorders characterized by either low or high bone mass. However, because the Wnt pathway is a common signaling mechanism involved in many organ systems, targeting elements within the pathway may be a challenge because of unwanted and potentially serious adverse effects. Nevertheless, specific pharmacological targets to influence bone formation have been identified in this pathway. Sclerostin antagonizes Wnt signaling and inhibits bone formation. Because sclerostin expression is highly restricted to osteocytes, it is an ideal target for anabolic drug therapy and trials are underway to develop antagonists to sclerostin activity.126 It is only a matter of time until new, highly effective anabolics will be available for clinical use. Strontium ranelate is an anabolic agent already approved for use in several countries but appropriate clinical trials have not been completed in the United States.
Selection of therapy and combination therapy
Choosing between the antiresorptive drugs (bisphosphonate, calcitonin, estrogen, raloxifene) remains an individual physician and patient preference. From a cost standpoint, ERT is the most appropriate, particularly given the many nonskeletal benefits of therapy. However, many patients and physicians are concerned because of risk of adverse events. Because estrogen is often used only for a few years following menopause, its utility for older patients at the highest risks of fracture may be limited in spite of data that have demonstrated continued effectiveness in preventing fracture in older patients. Raloxifene has both skeletal and extraskeletal benefits and is probably under prescribed; however, its use is often directed to those women at highest risk for the development of breast cancer because of its perceived lesser effectiveness at increasing BMD and reducing fracture. Another reason for the lesser use of raloxifene is the development or exacerbation of hot flashes in some women. Use of tamoxifen is restricted to patients with breast cancer for whom it is prescribed independent of any skeletal benefit. The bisphosphonates and calcitonin are generally more expensive than ERT/HRT and have no known extraskeletal benefit. Alendronate is now available as a generic formulation with substantially lower cost. The bisphosphonates are the most commonly employed agents for treatment of osteoporosis and have sometimes been inappropriately used in patients at low risk for fracture. Concern regarding oversuppression of bone turnover remains an issue, but seems relatively unfounded considering the millions of prescriptions written without observation of increased fracture rates among long-term users. Calcitonin might well be the treatment of choice from a side-effect point of view as there are few, if any, recognized contraindications to calcitonin therapy, and the drug is extremely well tolerated by most patients using the nasal spray preparation. Since its effectiveness seems less than the other antiresorptives, it is usually chosen when the patient cannot tolerate any other medication.
Teriparatide the only anabolic agent currently available in the USA but others including Denosumab, slow-release sodium fluoride strontium, and sclerostin may become available in the next several years. There is no clear indication of when anabolic agents should be selected over an antiresorptive drug as first line therapy. In theory, antiresorptive drugs should be the treatment of choice for patients with high turnover, and formation – stimulation drugs should be used for those with low turnover. There is evidence that the response to ERT39, 40, 41 and calcitonin111 is greatest in those with high turnover, but in controlled clinical trials the antifracture effectiveness of all FDA approved therapies is independent of pre-treatment bone turnover.
As more therapeutic options become available, discussions concerning combination therapies always follow. There is limited formal documentation of synergism when more than one antiresorptive therapy is used,127, 128 although no study has demonstrated a blunting of effect. Lumbar spine BMD by about 14% over 3 years in women receiving PTH plus HT, with evidence of the most rapid rise in BMD within the first 6 months of administration. Total hip BMD increased by about 4%. Although too small to make statistical comparisons, the combined HT/PTH group had no new vertebral fractures, compared to 22% of women on HT alone. Prior treatment with HT does not seem to reduce response to PTH as women starting PTH after HT had an increase of BMD of the spine by 11% and the hip 3%. Concomitant use of raloxifene with teriparatide likewise did not impair response. The bone formation marker P1NP rose similarly in women treated with teriparatide alone and in women treated with combined raloxifene/teriparatide; however, the increment in bone resorption marker was lower in the combined group. The increase in spine BMD was similar in both groups, but the increase in hip BMD was greater with combined therapy. Pretreatment with raloxifene also does not seem to impair the response to PTH.119
Prior or concomitant use of bisphosphonates has also been studied. In patients who are established on alendronate, PTH has been shown to stimulate bone formation as assessed by biochemical markers, with no increase in markers of bone resorption in a short trial. Data from a study in women pre-treated with alendronate for a 6-month period also suggested that spine BMD gain was lower in those given PTH with continued alendronate, compared with those given PTH alone. In another trial assessing addition of teriparatide given continuously or cyclically to women on alendronate for at least 1 year, PTH stimulated both bone formation and resorption markers, though change for bone formation was significantly higher than for resorption. Spine BMD increased similarly for both regimens compared to alendronate alone. Institution of teriparatide after cessation of long-term raloxifene or alendronate is associated with a blunted response of BMD and bone formation markers with prior alendronate but not prior raloxifene therapy, however, study was observational and small.
The question remains as to how best to combine therapies to cope with resumption of bone resorption upon discontinuation of teriparatide. Current strategies suggest that addition of an antiresorptive such as raloxifene or a bisphosphonate after discontinuation of teriparatide is important to maintain the increased BMD achieved while on the anabolic agent. A number of pharmacoeconomic models can be developed to assist in selecting the most cost-efficient strategy for prevention and treatment of osteoporosis, but until more complete data are available, these are heavily biased by unproven assumptions. Physicians should adopt the same approach to osteoporosis therapy as they do in other circumstances in which multiple options are available: become familiar with one or two alternatives and limit the use of other (possibly equally effective) therapies to patients not responding appropriately to the initial choice.
REFERENCES
Consensus Development Conference: Who are candidates for prevention and treatment for osteoporosis? Osteoporos Int. 1997;7(1):1-6. |
|
Consensus Development Conference: Prophylaxis and treatment of osteoporosis. Osteoporos Int 1991;1:118 |
|
Ravn P, Fledelius C, Rosenquist C, et al: High bone turnover is associated with low bone mass in both pre- and postmenopausal women. Bone 1996;19:291 |
|
Burge R, Dawson-Hughes B, Solomon DH, et al. Incidence and economic burden of osteoporosis-related fractures in the United States, 2005-2025. 2007; J Bone Miner Res 22:465. |
|
Laitinen K, Valimaki M, Keto P: Bone mineral density measured by dual-energy x-ray absorptiometry in healthy Finnish women. Calcif Tissue Int 1991;48:224 |
|
Riggs Bl, Wahner HW, Dunn WL, et al: Differential changes in bone mineral density of the appendicular and axial skeleton with aging: Relationship to spinal osteoporosis. J Clin Invest 1981;67:328 |
|
Kleerekoper M, Rao SD, Frame B, et al: Occult Cushing's syndrome presenting with osteoporosis. Henry Ford Med 1980;J28:132 |
|
Rao SD, Kleerekoper M, Rogers M, et al: Is gastrectomy a risk factor for osteoporosis? In: Christiansen C, Arnaud CD, Nordin BEC, et al (eds): Osteoporosis, p 775. Aalborg, Denmark, Aalborg Stifsbogtrykkeri, 1984 |
|
von Mach MA, Stoeckli R, Bilz S et al: Changes in bone mineral content after surgical treatment of morbid obesity. Metabolism. 2004 Jul;53(7):918-21. |
|
World Health Organization 1994: Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Geneva, WHO, Technical Report Series |
|
Marshall D, Johnell O, Wedel H: Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. Br Med J 1996;312:1254 |
|
Melton LJ III, Atkinson EJ, O'Fallon WM, et al: Long-term fracture prediction by bone mineral assessed at different skeletal sites. J Bone Miner Res 1993;8:1227 |
|
Silverman SL, Greenwald M, Klein RA, Drinkwater BL: Effect of bone density information on decisions about hormone replacement therapy: A randomized trial. Obstet Gynecol 1997;89:321 |
|
Ryan PJ, Harrison R, Blake GM, Fogelman I: Compliance with hormone replacement therapy (HRT) after screening for post menopausal osteoporosis. Br J Obstet Gynaecol 1992;99:325 |
|
Torgerson DJ, Donaldson C, Russell IT, Reid DM: Hormone replacement therapy: Compliance and cost after screening for osteoporosis. Eur J Obstet Gynecol Reprod Biol 1995;59:57 |
|
Prestwood KM, Pilbeam CC, Burleson JA, et al: The short term effects of conjugated estrogen on bone turnover in older women. J Clin Endocrinol Metab 1994;79L:366 |
|
Chesnut CH III, Bell NH, Clark GS, et al: Hormone replacement therapy in postmenopausal women: Urinary N-telopeptide of type I collagen monitors therapeutic effect and predicts response of bone mineral density. Am J Med 1997;102:29 |
|
Fuleihan GEH, Brown EM, Curtis K, et al: Effect of sequential and daily continuous hormone replacement therapy on indexes of mineral metabolism. Arch Intern Med 1992;152:1904 |
|
Meunier PJ, Chapuy MC, Arlot ME, et al: Effects of a calcium and vitamin D3 supplement on non-vertebral fracture and femoral bone density and parathyroid function in elderly women: A prospective placebo-controlled study. J Bone Miner Res 1991;6:S135 |
|
Heikinheimo RJ, Inkovaara JA, Harju EJ, et al: Annual injection of vitamin D and fractures of aged bones. Calcif Tissue Int 1992;51:105 |
|
Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. Dietary reference intakes for calcium, phosphorus, magnesium, vitamin D, and fluoride. Washington, D.C.: National Academy Press; 1997 |
|
Heaney RP: Vitamin D in Health and Disease.of calcium absorption) and an autocrine mechanism Clin J Am Soc Nephrol. 2008 Jun 4;. |
|
Kanis JA, Oden A, Johnell O et al: The use of clinical risk factors enhances the performance of BMD in theprediction of hip and osteoporotic fractures in men and women. Osteoporos Int. 2007 Aug;18(8):1033-46. Epub 2007 Feb 24. |
|
Kanis JA, Johnell O, Oden A et al: FRAX and the assessment of fracture probability in men and women from the UK. Osteoporos Int. 2008 Apr;19(4):385-97. Epub 2008 Feb 22. |
|
Dawson-Hughes B, Tosteson AN, Melton LJ 3rd et al: Implications of absolute fracture risk assessment for osteoporosis practiceguidelines in the USA. Osteoporos Int. 2008 Apr;19(4):449-58. Epub 2008 Feb 22. |
|
Eghbali-Fatourechi G, Khosla S, Sanyal A et al: Role of RANK ligand in mediating increased bone resorption in earlypostmenopausal women. J Clin Invest. 2003 Apr;111(8):1221-30. |
|
Hofbauer LC, Khosla S, Dunstan CR et al: Estrogen stimulates gene expression and protein production of osteoprotegerin in human osteoblastic cells. Endocrinology. 1999 Sep;140(9):4367-70. |
|
Cauley JA, Robbins J, Chen Z et al: Effects of estrogen plus progestin on risk of fracture and bone mineral density:the Women's Health Initiative randomized trial. JAMA. 2003 Oct 1;290(13):1729-38. |
|
Cummings SR, Ettinger B, Delmas PD et al: The effects of tibolone in older postmenopausal women. N Engl J Med. 2008 Aug 14;359(7):697-708. |
|
Lindsay R, Hart DM, Clark DM: The minimum effective dose of estrogen for prevention of postmenopausal bone loss. Obstet Gynecol 1984;63:759 |
|
Cauley JA, Seeley DG, Ensrud K, et al: Estrogen replacement therapy and fractures in older women. Study of Osteoporotic Fractures Research Group. Ann Intern Med 1995;122:9 |
|
Schneider DL, Barrett-Connor EL, Morton DJ: Timing of postmenopausal estrogen for optimal bone mineral density: The Rancho Bernardo study. JAMA 1997;277:543 |
|
Heaney RP: Estrogen-calcium interactions in the postmenopause: A quantitative description. Bone Miner 1990;11:67 |
|
Felson DT, Zhang Y, Hannan MT, et al: The effect of postmenopausal estrogen therapy on bone density in elderly women. N Engl J Med 1993;329:1141 |
|
Nguyen TV, Jones G, Sambrook PH, et al: Effects of estrogen exposure and reproductive factors on bone mineral density and osteoporotic fractures. J Clin Endocrinol Metab 1995;80:2709 |
|
Qu Y, Wong M, Thiebaud D et al: The effect of raloxifene therapy on the risk of new clinical vertebral fracturesat three and six months: a secondary analysis of the MORE trial. Curr Med Res Opin. 2005 Dec;21(12):1955-9. |
|
Delmas PD, Ensrud KE, Adachi JD et al: Efficacy of raloxifene on vertebral fracture risk reduction in postmenopausal J Clin Endocrinol Metab. 2002 Aug;87(8):3609-17. |
|
Ettinger B, Genant HK, Cann CE: Long-term estrogen replacement therapy prevents bone loss and fractures. Ann Intern Med 1985;102:319 |
|
Quigley MER, Martin PL, Burnier AM, Brooks P: Estrogen therapy arrests bone loss in elderly women. Am J Obstet Gynecol 1987;156:1516 |
|
The Writing Group for the PEPI Trial. Effects of hormone therapy on bone mineral density. JAMA 1996;276:1389 |
|
Marx CW, Dailey GE, Cheney C, et al: Do estrogens improve bone mineral density in osteoporotic women over age 65? J Bone Miner Res 1992;7:1275 |
|
Lindsay R, Tohme JF: Estrogen treatment of patients with established osteoporosis. Obstet Gynecol 1990;76:290 |
|
Lufkin EG, Wahner HW, O'Fallon WM, et al: Treatment of postmenopausal osteoporosis with transdermal estrogen. Ann Intern Med 1992;117:1 |
|
Ettinger B, Grady D: The waning effect of postmenopausal estrogen therapy on osteoporosis. N Engl J Med 1993;329:1192 |
|
Christiansen C, Christensen MS, Tranbol I: Bone mass in postmenopausal women after withdrawal of oestrogen/gestagen replacement therapy. Lancet 1981;1:459 |
|
Ravnnikar VA: Compliance with hormone replacement therapy: Are women receiving the full impact of hormone replacement therapy preventative health benefits? Womens Health Issues 1992;2:75 |
|
Anderson GL, Chlebowski RT, Rossouw JE et al: Prior hormone therapy and breast cancer risk in the Women's Health Initiativerandomized trial of estrogen plus progestin. Maturitas. 2006 Sep 20;55(2):103-15. Epub 2006 Jul 11. |
|
Gambrell RD Jr: The prevention of endometrial cancer in postmenopausal women with progestogens. Maturitas 1978;1:107 |
|
Chlebowski RT, Hendrix SL, Langer RD et al: Influence of estrogen plus progestin on breast cancer and mammography in healthy postmenopausal women: the Women's Health Initiative Randomized Trial. JAMA. 2003 Jun 25;289(24):3243-53. |
|
Schairer C, Lubin J, Troisi R et al: Menopausal estrogen and estrogen-progestin replacement therapy and breast cancer risk. JAMA. 2000 Jan 26;283(4):485-91. |
|
Anderson GL, Limacher M, Assaf AR et al: Effects of conjugated equine estrogen in postmenopausal women with hysterectomy:the Women's Health Initiative randomized controlled trial. JAMA. 2004 Apr 14;291(14):1701-12. |
|
Lindsay R, Gallagher JC, Kleerekoper M et al: Effect of lower doses of conjugated equine estrogens with and without medroxyprogesterone acetate on bone in early postmenopausal women. JAMA. 2002 May 22-29;287(20):2668-76. |
|
McKeever C, McIlwain H, Greenwald M et al: An estradiol matrix transdermal system for the prevention of postmenopausal boneloss. Clin Ther. 2000 Jul;22(7):845-57. |
|
Ettinger B, Ensrud KE, Wallace R et al: Effects of ultralow-dose transdermal estradiol on bone mineral density: arandomized clinical trial. Obstet Gynecol. 2004 Sep;104(3):443-51. |
|
Mosekilde L, Beck-Nielsen H, Sorensen OH et al: Hormonal replacement therapy reduces forearm fracture incidence in recent Maturitas. 2000 Oct 31;36(3):181-93. |
|
Lagrelius A: Treatment with oral estrone sulphate in the female climacteric. III. Effects on bone density and on certain biochemical parameters. Acta Obstet Gynecol Scand 1981;60:481 |
|
Lindsay R, Hart DM, Maclean A, et al: Bone loss during oestriol therapy in postmenopausal women. Maturitas 1979;1:279 |
|
Adachi JD, Sargeant EJ, Sagle MA, et al: A double-blind randomised controlled trial of the effects of medroxyprogesterone acetate on bone density of women taking oestrogen replacement therapy. Br J Obstet Gynaecol 1997;104:64 |
|
Gallagher JC, Kable WT: Effect of progestin therapy on cortical and trabecular bone: Comparison with estrogen. Am J Med 1991;90:171 |
|
Grey A, Cundy T, Evans M, Reid I: Medroxyprogesterone acetate enhances the spinal bone mineral density response to oestrogen in late post-menopausal women. Clin Endocrinol 1996;44:293 |
|
Speroff L, Rowan J, Symons J, et al, for the CHART Study Group: The comparative effect on bone density, endometrium, and lipids of continuous hormones as replacement therapy (CHART Study). JAMA 1996; 276:1397 |
|
Abdalla HI, Hart DM, Lindsay R, et al: Prevention of bone mineral loss in postmenopausal women by norethisterone. Obstet Gynecol 1985;66:789 |
|
Lindsay R, Hart DM, Purdie D, et al: Comparative effects of estrogen and a progestogen on bone loss in postmenopausal women. Clin Sci 1978;54:193 |
|
Isaia G, Mussetta M, Pecchio F, et al: Effect of testosterone on bone in hypogonadal males. Maturitas 1992;15:47 |
|
Riggs BL, Jowsey J, Goldsmith RS, et al: Short- and long-term effects of estrogen and a synthetic anabolic hormone in postmenopausal osteoporosis. J Clin Invest 1972;51:1659 |
|
Passeri M, Pedrazzoni M, Pioli G, et al: Effects of nandrolone decanoate on bone mass in established osteoporosis. Maturitas 1993;17:211 |
|
Savvas M, Studd JWW, Norman S, et al: Increase in bone mass after one year of percutaneous oestradiol and testosterone implants in post-menopausal women who have previously received long-term oral oestrogens. Br J Obstet Gynaecol 1992;99:757 |
|
Raisz LG, Wiita B, Artis A, et al: Comparison of the effects of estrogen alone and estrogen plus androgen on biochemical markers of bone formation and resorption in postmenopausal women. J Clin Endocrinol Metab 1996;81:37 |
|
Watts NB, Notelovitz M, Timmons MC, et al: Comparison of oral estrogens and estrogen plus androgen on bone mineral density, menopausal symptoms, and lipid-lipoprotein profiles in surgical menopause. Obstet Gynecol 1995;85:668 |
|
Rymer J, Chapman MG, Fogelman I: Effect of tibolone on postmenopausal bone loss. Osteoporos Int 1994;4:314 |
|
Bjarnason NH, Bjarnason K, Haarbo J, et al: Tibolone: Prevention of bone loss in late postmenopausal women. J Clin Endocrinol Metab 1996;81:2419 |
|
Geusens P, Dequeker J, Gielen J, Schot LPC: Non-linear increase in vertebral density induced by a synthetic steroid (ORG OD 14) in women with established osteoporosis. Maturitas 1991;13:155 |
|
Lyritis GP, Karpathios S, Basdekis K, et al: Prevention of post-oophorectomy bone loss with tibolone. Maturitas 1995;22:247 |
|
Chesnut CH, Ivey Jl, Gruber HE, et al: Stanozolol in postmenopausal osteoporosis: Therapeutic efficacy and possible mechanisms of action. Metabolism 1983;32:571 |
|
Agnusdei D, Zacchei F, Bigazzi S, et al: Metabolic and clinical effects of ipriflavone in established post-menopausal osteoporosis. Drugs Exp Clin Res 1989;15:97 |
|
Agnusdei D, Gennari C, Bufalino L: Prevention of early postmenopausal bone loss using low doses of conjugated estrogens and the non-hormonal, bone-active drug ipriflavone. Osteoporos Int 1995;5:462 |
|
Ward RL, Morgan G, Dalley D, Kelly PJ: Tamoxifen reduces bone turnover and prevents lumbar spine and proximal bone loss in early postmenopausal women. Bone Miner 1993;22:87 |
|
Love RR, Mazzess RB, Barden HS, et al: Effects of tamoxifen on bone mineral density in postmenopausal women with breast cancer. N Engl J Med 1992;326:852 |
|
Cosman F, Nieves J, Sherwood D, et al: Comparative effects of tissue selective estrogens and estrogen on bone mineral turnover. J Bone Miner Res 1996;11:835 |
|
Wile AG, Opfell RW, Margileth DA: Hormone replacement therapy in previously treated breast cancer patients. Am J Surg 1993;165:372 |
|
Gottardis MM, Jordan VC: Antitumor actions of keoxifene and tamoxifen in the N-nitrosomethylurea-induced rat mammary carcinoma model. Cancer Res 1987;46:4020 |
|
Draper MW, Flowers DE, Huster WJ, et al: A controlled trial of raloxifene (LY139481) HCI: Impact on bone turnover and serum lipid profile in healthy postmenopausal women. J Bone Miner Res 1996;11:835 |
|
Cauley JA, Norton L, Lippman ME et al: Continued breast cancer risk reduction in postmenopausal women treated with raloxifene: 4-year results from the MORE trial. Breast Cancer Res Treat. 2001 Jan;65(2):125-34. |
|
Vogel VG, Costantino JP, Wickerham DL et al: Effects of tamoxifen vs raloxifene on the risk of developing invasive breastcancer and other disease outcomes: the NSABP Study of Tamoxifen and Raloxifene JAMA. 2006 Jun 21;295(23):2727-41. Epub 2006 Jun 5. |
|
Martino S, Disch D, Dowsett SA et al: Safety assessment of raloxifene over eight years in a clinical trial setting. Curr Med Res Opin. 2005 Sep;21(9):1441-52. |
|
Watts NB, Harris ST, Genant HK, et al: Intermittent cyclical etidronate treatment of postmenopausal osteoporosis. N Engl J Med 1990;323:73 |
|
Storm T, Thamsborg G, Steiniche T, et al: Effects of intermittent cyclical etidronate therapy on bone mass and fracture rate in women with postmenopausal osteoporosis. N Engl J Med 1990;322:1265 |
|
Storm T, Kollerup G, Thamsborg G, et al: Five years of clinical experience with intermittent cyclical etidronate for postmenopausal osteoporosis. J Rheumatol 1996;23:1560 |
|
Harris ST, Watts NB, Jackson RD, et al: Four-year study of intermittent cyclic etidronate treatment of osteoporosis: Three years of blinded therapy followed by one year of open therapy. Am J Med 1993;95:557 |
|
Wimalawansa SJ. Combined therapy with estrogen and etidronate has an additive effect on bone mineral density in the hip and vertebrae: Four-year randomized study. Am J Med 1995;99:36 |
|
Bone HG, Downs RW Jr, Tucci JR, et al: Dose-response relationships for alendronate treatment in osteoporotic elderly women. J Clin Endocrinol Metab 1997;82:265 |
|
Tucci, JR, Tonino RP, Emkey RD, et al, for the US Alendronate Phase III Osteoporosis Treatment Study Group. Effect of three years of oral alendronate treatment in postmenopausal women with osteoporosis. Am J Med 1996;101:488 |
|
Liberman UA, Weiss SR, Broll J, et al: Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. N Engl J Med 1995;333:1437 |
|
Black DM, Cummings SR, Karpf DB, et al, for the Fracture Intervention Trial Research Group: Randomized trial of effect of alendronate on the risk of fracture in women with existing vertebral fractures. Lancet 1996;348:1535 |
|
Karpf DB, Shapiro DR, Seeman E, et al, for the Alendronate Osteoporosis Treatment Study Groups: Prevention of nonvertebral fractures by alendronate: A meta-analysis. JAMA 1997;277:1159 |
|
Black DM, Schwartz AV, Ensrud KE et al: Effects of continuing or stopping alendronate after 5 years of treatment: the JAMA. 2006 Dec 27;296(24):2927-38. |
|
Hosking DJ, McClung MR, Ravn P, et al, for the EPIC Study Group: Alendronate in the prevention of osteoporosis: EPIC study two-year results. J Bone Miner Res 1996;11(S):153 |
|
Rizzoli R, Greenspan SL, Bone G 3rd et al: Two-year results of once-weekly administration of alendronate 70 mg for thetreatment of postmenopausal osteoporosis. J Bone Miner Res. 2002 Nov;17(11):1988-96. |
|
Harris ST, Watts NB, Genant HK et al: Effects of risedronate treatment on vertebral and nonvertebral fractures in womenwith postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group. JAMA. 1999 Oct 13;282(14):1344-52. |
|
McClung MR, Geusens P, Miller PD et al: Effect of risedronate on the risk of hip fracture in elderly women. Hip Intervention Program Study Group. N Engl J Med. 2001 Feb 1;344(5):333-40. |
|
Chesnut III CH, Skag A, Christiansen C et al: Effects of oral ibandronate administered daily or intermittently on fracture riskin postmenopausal osteoporosis. J Bone Miner Res. 2004 Aug;19(8):1241-9. Epub 2004 Mar 29. |
|
Reginster JY, Adami S, Lakatos P et al: Efficacy and tolerability of once-monthly oral ibandronate in postmenopausal osteoporosis: 2 year results from the MOBILE study. Ann Rheum Dis. 2006 May;65(5):654-61. Epub 2005 Dec 8. |
|
McClung MR, Wasnich RD, Recker R et al: Oral daily ibandronate prevents bone loss in early postmenopausal women without osteoporosis. J Bone Miner Res. 2004 Jan;19(1):11-8. |
|
Eisman JA, Civitelli R, Adami S et al: Efficacy and tolerability of intravenous ibandronate injections in postmenopausal osteoporosis. J Rheumatol. 2008 Mar;35(3):488-97. Epub 2008 Feb 1. |
|
Black DM, Delmas PD, Eastell R et al: Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. 2007 May 3;356(18):1809-22. |
|
Khosla S, Burr D, Cauley J et al: Bisphosphonate-associated osteonecrosis of the jaw: report of a task force of theAmerican Society for Bone and Mineral Research. J Bone Miner Res. 2007 Oct;22(10):1479-91. |
|
Odvina CV, Zerwekh JE, Rao DS et al: Severely suppressed bone turnover: a potential complication of alendronate therapy. J Clin Endocrinol Metab. 2005 Mar;90(3):1294-301. Epub 2004 Dec 14. |
|
Lenart BA, Lorich DG, Lane JM: Atypical fractures of the femoral diaphysis in postmenopausal women taking alendronate. N Engl J Med. 2008 Mar 20;358(12):1304-6. |
|
Cranney A, Moher D, Shea B, et al: Meta-analysis of calcitonin in the treatment of postmenopausal osteoporosis. Arthritis Rheum 1995;38:S360 |
|
Overgaard K: Effect of intranasal calcitonin therapy on bone mass and bone turnover in early postmenopausal women: A dose-response study. Calcif Tissue Int 1994;55:82 |
|
Ellerington MC, Hillard TC, Whitcroft SI, et al: Intranasal salmon calcitonin for the prevention and treatment of postmenopausal osteoporosis. Calcif Tissue Int 1996;59:6 |
|
Rico H, Revilla M, Hernandez ER, et al: Total and regional bone mineral content and fracture rate in postmenopausal osteoporosis treated with salmon calcitonin: A prospective study. Calcif Tissue Int 1995;56:181 |
|
Meschia M, Brincat M, Barbacini P, et al: Effect of hormone replacement therapy and calcitonin on bone mass in postmenopausal women. Eur J Obstet Gynecol Reprod Biol 1992;47:53 |
|
Flicker L, Hopper JL, Larkins RG, et al: Nandrolone decanoate and intranasal calcitonin as therapy in established osteoporosis. Osteoporos Int 1997;7:29 |
|
Pontiroli AE, Pajetta E, Scaglia L, et al: Analgesic effect of intranasal and intramuscular salmon calcitonin in post-menopausal osteoporosis: A double-blind, double-placebo study. Aging 1994;6:459 |
|
Lyritis GP, Tsakalakos N, Magiasis B, et al: Analgesic effect of salmon calcitonin in osteoporotic vertebral fractures: A double-blind placebo-controlled clinical study. Calcif Tissue Int 1991;49:3699 |
|
Laurian L, Oberman Z, Graf E, et al: Calcitonin induced increase in ACTH, beta-endorphin and cortisol secretion. Horm Metab Res 1986;18:268 |
|
Deftos LJ, Nolan JJ, Seely BL, et al: Intrapulmonary drug delivery of bone-acting peptides: Bioactivity of inhaled calcitonin approximates injected calcitonin. J Bone Miner Res 1996;11(suppl 1):95 |
|
Cosman F: Anabolic therapy for osteoporosis: parathyroid hormone. Curr Osteoporos Rep. 2005 Dec;3(4):143-9. |
|
Pak CYC, Sakhaee K, Adams-Huet B, et al: Treatment of postmenopausal osteoporosis with slow-release sodium fluoride: Final update of a randomized controlled trial. Ann Intern Med 1995;123:401 |
|
Pak CYC, Sakhaee K, Bell N, et al: Comparison of non-randomized trials with slow-release sodium fluoride with a randomized placebo-controlled trial in postmenopausal osteoporosis. J Bone Miner Res 1996;11:160 |
|
Lundy MW, Stauffer M, Wergedal JE, et al: Histomorphometric analysis of iliac crest bone biopsies in placebo-treated versus fluoride-treated subjects. Osteoporos Int 1995;5:115 |
|
Sebert JL, Richard P, Mennecier I, et al: Monofluorophosphate increases lumbar bone density in osteopenic patients: A double-masked randomized study. Osteoporos Int 1995;5:108 |
|
Bone HG, Bolognese MA, Yuen CK et al: Effects of denosumab on bone mineral density and bone turnover in postmenopausalwomen. J Clin Endocrinol Metab. 2008 Jun;93(6):2149-57. Epub 2008 Apr 1. |
|
Miller PD, Bolognese MA, Lewiecki EM et al: Effect of denosumab on bone density and turnover in postmenopausal women with low bone mass after long-term continued, discontinued, and restarting of therapy: a randomized blinded phase 2 clinical trial. Bone. 2008 Aug;43(2):222-9. Epub 2008 Apr 26. |
|
Grey A, Reid IR: Emerging and potential therapies for osteoporosis. Expert Opin Investig Drugs. 2005 Mar;14(3):265-78. |
|
Greenspan SL. Emkey RD. Bone HG. , et al:Significant differential effects of alendronate, estrogen, or combination therapy on the rate of bone loss after discontinuation of treatment of postmenopausal osteoporosis. A randomized, double-blind, placebo-controlled trial.[summary for patients in Ann Intern Med. 2002 Dec 3;137(11):I31 |
|
Pinkerton JV, Dalkin AC: Combination therapy for treatment of osteoporosis: A review. Am J Obstet Gynecol. 2007 Dec;197(6):559-65. |