Pulmonary Disease in Pregnancy
Authors
INTRODUCTION
The respiratory system undergoes a number of anatomic and physiologic changes during the course of a normal pregnancy. Some of these changes may predispose the patient to developing several acute pulmonary disorders, such as aspiration, thromboembolic disease, pulmonary edema, and amniotic fluid embolism. Pregnancy can also affect the course of some chronic pulmonary diseases, most notably, asthma and sarcoidosis. Conversely, these pulmonary conditions, if poorly controlled, can adversely affect pregnancy. This chapter describes these anatomic and physiologic alterations, and reviews the clinical presentation and treatment of acute and chronic pulmonary diseases that occur during pregnancy.
CARDIORESPIRATORY PHYSIOLOGY AND PREGNANCY
A number of anatomic and physiologic changes that occur during pregnancy affect the cardiorespiratory system. The upper airway becomes hyperemic, leading to nasal obstruction, increased mucous secretion, and occasionally epistaxis.1 These changes may be more pronounced in patients with preexisting atopic disease, chronic sinusitis, or preeclampsia, and they are more evident during the third trimester.
Blood volume gradually increases from the 6th week to the end of pregnancy, when it is approximately 40% above normal.2,3 The increase is largely due to increased plasma volume, as red blood cell mass remains the same. The result is hemodilution, anemia, and decreased serum protein levels.4 Extracellular fluid also increases, contributing to peripheral edema.5,6
Cardiovascular changes include an increased heart rate, increased stroke volume, and decreased peripheral resistance with an attendant increase in cardiac output.7,8,9,10,11,12 The increase in stroke volume begins around the 10th week and peaks at 20 to 24 weeks, where it is maintained until term.3,13,14 Heart rate begins to increase at 5 to 12 weeks, reaching a maximum value of 10% to 30% above prepregnancy values at 32 weeks.9,10,11,12 In the supine position, the gravid uterus compresses the inferior vena cava, causing decreased venous return and consequently a lower stroke volume and cardiac output. This does not occur in the left lateral decubitus position.13,14 Because of lower systemic vascular resistance, systemic blood pressure may be lower, most notably in midpregnancy. The decrease in diastolic pressure is greater than the decrease in systolic pressure, and hence there is a widened pulse pressure.13,14 Blood pressure generally rises during pregnancy but usually remains below prepregnancy levels. Central venous pressure, pulmonary artery pressure, and pulmonary capillary wedge pressure generally do not change during pregnancy.12
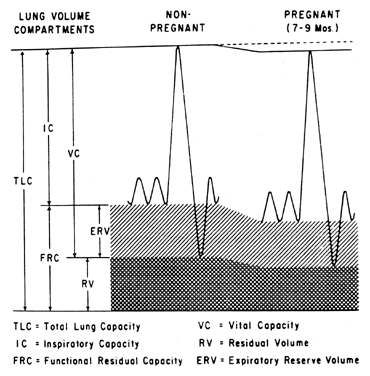
The anatomic changes of pregnancy also affect pulmonary function (Fig. 1). As the uterus enlarges it can cause up to a 4 cm elevation of the diaphragm, although it does not appear to affect diaphragmatic function.15,16 The anteroposterior and transverse diameters of the thoracic cage also increase by 2 cm, in part offsetting the volume loss due to diaphragm elevation.17 Because the diaphragm is displaced upward, there is a decrease in residual volume and expiratory reserve volume, resulting in a decreased functional residual capacity. Conversely, inspiratory capacity and tidal volume increase, so that vital capacity and total lung capacity remain the same.15,17,18,19,20,21
Usually, spirometric measurements of the forced expiratory volume in one second (FEV1), forced vital capacity, and flow rates do not change.4,18,19,20,22 Likewise, there are no changes in lung compliance in a normal pregnancy.23 There may be a slight increase in the carbon monoxide diffusing capacity during early pregnancy, although it usually decreases to normal or slightly below normal as pregnancy progresses.24 The mechanism of these changes is unclear.
A 20% to 50% increase in minute ventilation occurs toward the end of the first trimester and remains constant throughout pregnancy. The increase is thought to be caused by increased progesterone levels,15,18,25,26 and is related to an increase in tidal volume rather than an increase in respiratory rate.7 The increase in alveolar ventilation causes chronic respiratory alkalosis, with PCO2 levels close to 30. Renal bicarbonate excretion partially compensates, keeping serum bicarbonate levels at 18 to 21 mEq/L.27,28 During labor and delivery, minute ventilation and respiratory alkalosis usually worsen. The arterial oxygen tension is usually greater than 100 during pregnancy, although it may decrease closer to term.28,29
FETAL PHYSIOLOGY
Fetal oxygen delivery is dependent on maternal blood oxygen content and uterine blood flow. Uterine blood flow decreases with maternal hypotension, low maternal cardiac output, uterine artery vasoconstriction secondary to increased levels of exogenous or endogenous catecholamines, and maternal alkalosis.12,30 Maternal alkalosis also affects oxygen unloading at the tissue level because of a shift in the oxygen-hemoglobin dissociation relation with greater binding affinity.
Maternal and fetal circulation use a countercurrent exchange mechanism to exchange oxygen and metabolic waste products.31 Umbilical vein blood usually has a PO2 of 26 to 32 mmHg, an oxygen saturation of 80% to 90%, a PCO2 of 38 to 42 mmHg, and a pH of 7.30 to 7.35.32,33,34 Despite this low PO2, the oxygen content of fetal blood is high because of the high concentration of fetal hemoglobin and its affinity for oxygen.4,31 Adequate oxygen delivery is ensured because fetal hemoglobin concentration is increased, and fetal blood flow is high because of an increased cardiac output.4,12 Supplemental oxygen administration to the mother may cause only a small increase in fetal PO2, but it may result in a large increase in fetal oxygen saturation and tissue delivery because of the characteristics of the oxygen-hemoglobin dissociation curve.31,35,36 Thus, supplemental oxygen has been shown to be helpful in the treatment of fetal hypoxemia.35,36,37,38
DYSPNEA IN THE PREGNANT PATIENT
Dyspnea is a common complaint during pregnancy, affecting 60% to 70% of women with no previous history of cardiac or pulmonary disease.18,21,39,40 Although the etiology is unknown, it is thought to be related to physiologic hyperventilation.40
Physiologic dyspnea usually occurs early in pregnancy and improves closer to term. Patients are able to perform daily activities and are usually not dyspneic with rest. Pathologic dyspnea, however, usually worsens with time.41 The pregnant patient may occasionally complain of paroxysmal nocturnal dyspnea, orthopnea, and chest discomfort during late pregnancy. Although this may suggest cardiac disease, it is often due to upward displacement of the diaphragm.13,42,43
A chest radiograph with shielding of the fetus should be performed in any patient being evaluated for pathologic dyspnea. Radiographic findings that are expected in the pregnant patient include straightening of the left upper cardiac border by a prominent pulmonary trunk due to lumbar lordosis3; elevation of the diaphragm, with upward and lateral displacement of the heart; and short, wide lung fields.17 In the lateral view, there may be an increase in the anteroposterior diameter. The increased blood volume results in prominent vascular markings.3
ACUTE PULMONARY DISEASE IN PREGNANCY
Pneumonia
Pneumonia, although infrequent, is the most common nonobstetric infection to cause complications as well as maternal and fetal mortality in the peripartum period.44 The microorganisms that cause pneumonia in the pregnant patient are not unique; however, pregnant women may be more susceptible to organisms that are controlled by cell-mediated immune processes, such as viruses, fungi, and mycobacteria.45 For example, pregnant patients had a higher mortality rate due to influenza A during the 1918 and 1957 epidemics, although this has not been observed in subsequent outbreaks.46 Recent studies have also suggested that the incidence of bacterial pneumonia in pregnancy may be increasing, but this may be due to the increasing number of pregnant women with underlying medical disorders.47
BACTERIAL PNEUMONIA.
Fever, chills, and cough productive of purulent sputum are the most common complaints in bacterial pneumonia. The two most common bacterial pathogens include Streptococcus pneumoniae (Pneumococcus) and Haemophilus influenzae. Mycoplasma pneumoniae, Legionella pneumophila, and Chlamydia pneumoniae also occur, but their incidence in pregnancy is unknown. When bacterial pneumonia complicates viral pneumonia, Staphylococcus aureus or gram-negative organisms should be considered as potential pathogens.
Most community acquired pneumonias can be treated in the outpatient setting with an orally active antibiotic, preferably one that is penicillin-related or a cephalosporin. Erythromycin should be added if Mycoplasma or Legionella are a concern. These antibiotics are classified as category B by the Food and Drug Administration (FDA). If the patient appears severely ill, however, hospital admission should be considered and antibiotics should be administered intravenously. Intravenous aminoglycosides, used for gram-negative bacteria, can cause toxicity to fetal auditory function. Vancomycin, which should be used only for resistant gram-positive infections, may cause renal and auditory nerve damage to the fetus. Antibiotics to be avoided in pregnancy include tetracycline, which can affect fetal skeletal and dental development; chloramphenicol, which can cause newborn “gray syndrome”; and trimethoprim with sulfamethoxazole, which can interfere with folic acid metabolism and has caused cleft palates in rats.47
VIRAL PNEUMONIA.
Clinical symptoms due to influenza A infection include high fever, coryza, headache, malaise, muscle aches, and cough, which usually subside in approximately 3 to 5 days. If chest symptoms persist for longer, a complicating pneumonia should be suspected. Pneumonia can occur from a viral inflammation of lung parenchyma or from a secondary bacterial infection. Amantadine (FDA category C) may be used to treat severe viral pneumonia and can also be used as prophylaxis in high-risk patients. Rimantadine, a newer antiviral agent, has not been studied in pregnant women; however, it has been reported to produce fetal abnormalities when given to animals in doses higher than the recommended dose for humans.48 It is also currently listed as FDA category C. Ribavirin is effective against both influenza A and B but is listed as FDA category X. Although influenza vaccination is not recommended for all pregnant women, it may be used in high-risk patients and should be given after the first trimester.46,49
Varicella, an infection caused by varicella-zoster virus, is usually a benign, self-limited disease in children, but it can affect up to 2% of the adult population with serious consequences.47,50 Compared to nonpregnant persons, pregnant women with varicella pneumonia have a higher rate of increased virulence, greater complications, and increased maternal mortality during third trimester51,52,53,54: the mortality rate is 35% to 40% among pregnant women compared to 11% to 17% among men and nonpregnant women.51,52 Patients with varicella initially present with a rash, fever, and malaise. Pneumonia usually develops 2 to 5 days later, with symptoms of cough, dyspnea, pleuritic chest pain, and occasionally hemoptysis.50,54 Oral mucosal lesions may also be present.54 Chest radiographs may show diffuse miliary or nodular infiltrates, which usually resolve in approximately 2 weeks.50 The disease can vary in severity: some patients may present with few symptoms and an abnormal chest x-ray, whereas others may present with respiratory failure and adult respiratory distress syndrome (ARDS).51,54
Varicella can cause congenital abnormalities as a result of first-trimester infection. Infection late in pregnancy can cause preterm labor and perinatal infection with a high infant mortality rate.52,55 Because of the high rate of maternal mortality, patients with varicella pneumonia should be treated aggressively with early hospitalization and acylovir, both of which have been shown to improve the disease course.51,56 Although acyclovir is listed as FDA category C, it has been given safely in pregnant patients in the second and third trimester.52 The Centers for Disease Control (CDC) recommend that varicella immune globulin be given to all patients without immunity in order to prevent the complications of adult varicella infection.56,57 There is no evidence that administration of immune globulin will prevent congenital abnormalities or perinatal infection of the newborn.
Women of childbearing age are increasingly infected with human immunodeficiency virus (HIV). Pneumocystis carinii pneumonia is the most common opportunistic infection. In 1989, the United States Public Health Service, in order to prevent harm to the fetus, recommended that pregnant patients not receive prophylactic medication to prevent Pneumocystis carinii pneumonia.58 The American College of Obstetricians and Gynecologists (ACOG), however, believes that potentially beneficial therapies should not be withheld during pregnancy unless there is a known adverse maternal or fetal side effect that outweighs the potential maternal benefit. Thus ACOG suggests that prophylactic treatment be initiated as it would for a nongravid HIV-infected patient.59,60,61 There is a dearth of data on the effect of these medications on pregnancy and on their teratogenicity. Trimethoprim-sulfamethoxazole is the most effective drug but has many side effects, including rash, fever, nausea, vomiting, and leukopenia. It also crosses the placenta, thus increasing the risk of exposure and toxicity to the fetus. Aerosolized pentamidine has minimal systemic absorption and fewer side effects, but it is not as effective. Other medications currently in use in the nonpregnant population include dapsone, pyrimethamine, and clindamycin.60,61 Patients with documented Pneumocystis carinii pneumonia have been treated with trimethoprim-sulfamethoxazole in combination with corticosteroids with a good outcome.62 The safety of other treatments for documented infection in pregnancy is not known.
FUNGAL PNEUMONIA.
Coccidioidomycosis is the most extensively studied fungal infection in pregnancy. Although coccidioidomycosis is rare and does not affect the outcome of pregnancy unless it becomes disseminated,63 several studies suggest that the risk of dissemination is higher in pregnant women than in the general population, especially in the third trimester.63,64 Others have suggested that the high reported rate of dissemination does not correspond to general clinical experience in endemic areas and may be exaggerated as a result of confounding socioeconomic and racial factors.65 Mortality can be avoided by early treatment of disseminated disease with antifungal agents, such as amphotericin B.47,63,64,65 Ketoconazole has been used in one case successfully but needs further study.64 There has been one case report suggesting a possible link between fluconazole and congenital abnormalities, and therefore it is not recommended for use at this time.66 Other fungal infections, including cryptococcosis, blastomycosis, and sporotrichosis, have also been reported in pregnant patients, although the evidence does not suggest that pregnancy is a risk factor for these infections.65
Tuberculosis
The incidence of mycobacterial disease has increased in recent years, possibly because of the increased rate of HIV infection, increased numbers of immigrants from endemic countries, and a decline in public health services. It is especially prevalent in certain high-risk groups (Table 1).67,68,69
TABLE 1. Persons at Risk for Tuberculosis
Household contact of patients with known TB infection
Immigrants from endemic countries
Patients infected with HIV
Patients with underlying medical conditions (hematologic and reticuloendothelial malignancies, treatment with immunosuppressive agents, diabetes mellitus, end-stage renal disease, silicosis, jejunoileal bypass, gastrectomy)
Malnourished individuals
Intravenous drug users
The homeless
Patients in correctional facilities and nursing homes
People living in medically underserved, low-income areas
Tuberculosis (TB) is caused by inhalation of aerosol droplets carrying the organism. Tubercle bacilli multiply in the lung and migrate via the lymphatic and cardiovascular system to other organs including the spleen, liver, bones, meninges, joints, genitalia, endometrium, and placenta. As the patient develops immunity to the organism, the affected areas heal, scar, and become calcified. Despite this repair process, some viable organisms may remain.
The American Thoracic Society (ATS) and the CDC have developed the following criteria for determining when a tuberculin skin test is considered positive68:
- A 5-mm or greater induration is considered positive in patients infected with HIV, those who have close contact with a person with TB, or those with a chest radiograph demonstrating old, healed TB.
- A 10-mm or greater induration is considered positive in patients who are immigrants from endemic areas, homeless, live in a nursing home or correctional facility, inject drugs, are part of a high-risk minority population, or have another medical disease that increases their risk of contracting TB.
- A 15-mm or greater induration is considered positive in all other patients. Patients who have had a prior BCG vaccination should have their skin test interpreted in the same manner.67,68
Approximately 10% of adults with active TB may have a false-negative skin test because of age, malnutrition, immunosuppression due to disease or drugs, viral infections, or overwhelming TB infection. Conversely, false-positive tests can occur in patients infected with nontuberculous mycobacteria.67
Once it is determined that a patient has a positive skin test, a chest radiograph with shielding of the fetus should be performed. The most common symptoms of pulmonary TB are cough, weight loss, fever, fatigue, and hemoptysis. Diagnosis is usually obtained by culture from expectorated sputum, although occasionally a more invasive means, such as bronchoscopy, is needed to obtain samples.
Treatment recommendations have changed recently because of the increased prevalence of drug-resistant TB in the United States.70 Initial treatment of the nonpregnant patient involves four medications: isoniazid, rifampin, and pyrazinamide plus either streptomycin sulfate or ethambutol. This regimen should be continued until drug susceptibility test results are available. Since streptomycin sulfate can interfere with fetal auditory development and can cause congenital deafness, its use is not recommended in the pregnant patient. Although there are no data on the teratogenicity of pyrazinamide, it should be avoided if possible in the pregnant patient. The pregnant patient, who is not at high risk for drug-resistant TB, should be treated with isoniazid, rifampin, and ethambutol for a total of 9 months. If the risk of drug resistance is high, pyrazinamide may be added with the understanding there are unknown risks. Pyridoxine should be given to all patients receiving isoniazid to avoid nerve damage to the mother.
The current recommendations of the ATS and CDC for prophylactic treatment of a pregnant patient with a positive skin test and no evidence of active disease are stated as follows69:
Although no harmful effects of isoniazid to the fetus have been observed, preventive therapy generally should be delayed until after delivery. There does not appear to be any substantial increase in tuberculosis risk for women as a result of pregnancy. However, for pregnant women likely to have been recently infected or with high risk medical conditions, especially HIV infection, isoniazid preventive therapy should begin when infection is documented.
Congenital TB of the newborn occurs as a result of placental transmission to the fetus via the umbilical vein or by aspiration of infected amniotic fluid.71 A tuberculin skin test is usually negative in the newborn, but it may turn positive after 1 to 3 months.67 Congenital TB has a high mortality rate, especially if there is a delay in establishing the diagnosis and starting antibiotic therapy.72
Aspiration Pneumonia
Aspiration pneumonia is a significant cause of maternal morbidity and mortality. Factors that predispose the pregnant patient to aspiration during labor and delivery include increased intragastric pressure caused by abdominal compression, decreased gastroesophageal sphincter tone due to progesterone effects, delayed gastric emptying, repeated abdominal palpations during examinations, and alteration of consciousness secondary to analgesics and anesthesia.47,73 Most cases of aspiration occur if general anesthesia or endotracheal intubation is needed.73
Aspiration of gastric acid is usually followed by chemical pneumonitis and pulmonary edema due to increased vascular permeability. The clinical presentation includes tachypnea, cyanosis, hypoxemia, hypotension, tachycardia, and bronchospasm. The chest radiograph shows either isolated or diffuse infiltrates.73 The volume aspirated is important in the timing of the onset of clinical symptoms. When smaller volumes are aspirated the symptoms usually do not become apparent until 6 to 8 hours after the event. Large volumes can cause immediate asphyxiation. The pH of the aspirate is also important. Minimal injury has been found when the pH is greater than 2.5.74
The treatment of aspiration pneumonia is supportive, and consists of oxygen, bronchodilators, and if needed, ventilatory support. Unless there are complications due to secondary bacterial pneumonia, improvement is usually seen after a few days75; however, the condition may also progress to respiratory failure and ARDS. The most common bacterial pathogens, including S. aureus, gram-negative organisms, and anaerobic organisms, originate from the oropharynx. Antibiotics should be started early if bacterial infection is suspected.
Pleural Disease
Small, benign pleural effusions may occur in the immediate postpartum period in patients with no history of underlying cardiac or pulmonary disease.76,77 These small effusions are believed to result from the combination of increased blood volume, decreased colloid oncotic pressure, and the increased intrathoracic pressures generated by repeated Valsalva maneuvers during labor and delivery.77,78 The occurrence of postpartum effusions has been challenged in a study that failed to find evidence of pleural effusions with ultrasound evaluation.79 Despite the controversy, a small, asymptomatic pleural effusion should be considered benign. In contrast, a moderate to large pleural effusion is suspicious of an underlying cardiopulmonary disorder.78
Deep Vein Thrombosis and Pulmonary Embolism
Pregnant patients are at increased risk for thromboembolic disease for several reasons: (1) decreased venous tone and blood flow in the lower extremities, leading to venous stasis80,81; (2) compression of the inferior vena cava and left iliac vein by the uterus, leading to venous outflow obstruction and stasis81,82; and (3) an increase in several clotting factors and a decrease in fibrinolytic activity, leading to a hypercoagulable state.83
In the nonpregnant patient the most specific diagnostic test to determine the presence of deep vein thrombosis (DVT) is contrast venography. A normal venogram excludes the possibility of DVT.84 During pregnancy, in order to minimize radiation exposure to the fetus, only limited venography can be performed. This enables visualization of the calf veins, popliteal veins, and most of the superficial femoral veins, but not the iliac veins. The limited visualization decreases the sensitivity of the test.
Impedance plethysmography is a good test for diagnosis of a proximal DVT located above the popliteal vein in the nonpregnant patient. In the pregnant patient, however, especially during the third trimester, this test may be difficult to interpret because of compression of the iliac vein by the uterus, which causes a false-positive test.85 Therefore, if a positive test result is obtained, the patient should be placed in the left lateral recumbent position for 20 to 30 minutes and the test should be repeated. It is not a good test for the diagnosis of DVT below the knee.
Duplex ultrasonography is also good for identification of a proximal DVT, but it does not visualize the iliac vein well.86,87 The sensitivity of the test is very reliant on the knowledge and experience of the technician performing it and the radiologist's interpretation. It can be used in place of impedance plethysmography. Radioactive fibrinogen uptake scanning is contraindicated in the pregnant patient.
Symptoms of a DVT include calf pain and swelling. Impedance plethysmography or duplex ultrasonography should be performed first in all patients suspected of having a DVT. If the initial test is negative, serial testing should be done over a 7- to 14-day period to exclude extension of a clot. If the initial test is positive and the patient is in the first or second trimester, she should undergo treatment. If she is in the third trimester, external compression should be ruled out first with a limited venogram.
Clinical symptoms of pulmonary embolism (PE) include the sudden onset of dyspnea, tachypnea, tachycardia, and pleuritic chest pain. In massive PE, arrhythmias, syncope, and cardiovascular collapse may develop. The best diagnostic test to diagnose PE is the pulmonary angiogram, although it has the disadvantage of being an invasive test.88,89 Ventilation and perfusion lung scanning is a reliable noninvasive test used to determine the presence of PE, but the results may be difficult to interpret in some cases. While a normal scan excludes PE90 and a high-probability scan usually indicates PE, approximately 40% to 70% of all patients have scans interpreted as low to intermediate probability.88,91,92 The prevalence of PE in this group ranges from 21% to 40%.92 To diagnose a PE in these patients, one can either proceed to perform pulmonary angiography or evaluate the patient first for the presence of a DVT.88,91 The presence of a DVT increases the likelihood that the patient has a PE.
Treatment of DVT and PE involves the use of anticoagulants. Since warfarin is contraindicated in pregnancy, heparin is the drug of choice. Heparin can be given by intravenous infusion or subcutaneously, and the dose should be adjusted by maintaining the patient's partial thromboplastin time at a level 1.5 to 2.5 times the control. Treatment should be given for the duration of pregnancy and continued for approximately 4 to 6 weeks after delivery. If thrombosis occurs late in the pregnancy, treatment may be required for up to 3 months after delivery. Patients who have a history of a previous DVT or PE can be managed prophylactically by either empiric treatment with subcutaneous heparin at a dose of 5000 units every 12 hours or by weekly surveillance with impedance plethysmography or duplex ultrasonography.93
Patients receiving adjusted-dose subcutaneous heparin have an increased risk of bleeding during delivery because of the prolonged half-life of heparin when administered subcutaneously. It may be best to induce labor in these patients after they have discontinued subcutaneous heparin for 24 hours. Intravenous heparin is substituted and can be stopped 4 to 6 hours before delivery.
Low-molecular-weight heparin is an alternative to standard heparin treatment and has been used safely during pregnancy. It has the advantages of once-daily dosing, no need for monitoring partial thromboplastin time, lack of local side effects at the injection site, and less thrombocytopenia.94,95,96 There have also been some studies suggesting that the use of low-molecular-weight heparin decreases the patient's risk of osteoporosis compared to standard heparin, but this needs to be investigated further.96,97 Pregnancy is a relative contraindication to the use of thrombolytic therapies such as streptokinase, urokinase, and tissue plasminogen activator, and these should be used only in patients suffering from massive PE and cardiovascular instability.98,99,100 The use of vena cava filters is indicated for those patients who cannot be anticoagulated or for those who have recurrent PE while on adequate anticoagulant therapy.
Amniotic Fluid Embolism
Amniotic fluid contains fetal debris, including desquamated squamous cells, meconium, lanugo hair, and mucin. As long as the fetal membranes remain intact, fluid cannot enter the maternal circulation. In addition, there must also be disruption of the uterine veins and a pressure gradient favoring fluid entry.101 Two possible sites of entry are the uterine veins at the site of placental separation and small tears in the lower uterus and endocervix.
Clinical signs of amniotic fluid embolism include sudden onset of severe dyspnea, hypoxemia, cyanosis, cardiovascular collapse, seizures, and coma occurring either during or shortly after delivery.102,103,104 Progression to ARDS and disseminated intravascular coagulation is not uncommon. Risk factors include premature rupture of membranes, meconium staining of amniotic fluid, advanced age, multiparity, and the use of uterine stimulants during labor and delivery.102,104,105
Mechanisms proposed for cardiopulmonary collapse caused by amniotic fluid embolism include (1) mechanical obstruction of the pulmonary vasculature; (2) alveolar capillary leak (ARDS) secondary to extensive microembolic insult; (3) pulmonary edema due to left ventricular failure; and (4) anaphylaxis due to sudden exposure to fetal antigen.105
The diagnosis can be made by cytologic examination of blood removed from the distal lumen of a wedged pulmonary artery catheter, which will show a large amount of fetal squamous cells, mucin, and lanugo.105 Treatment is largely supportive, and the mortality rate ranges from 80% to 90%.102
Tocolytic-Induced Pulmonary Edema
β-Adrenergic agents such as ritodrine and terbutaline are commonly used to inhibit preterm labor and, in this setting, can cause acute respiratory failure due to pulmonary edema. Symptoms include chest pain, dyspnea, tachypnea, tachycardia, and cough. The physical examination demonstrates crackles on auscultation, and chest radiograph suggests pulmonary edema. It can happen during administration of the medication or even 24 to 48 hours after discontinuation.106
The etiology is unclear, but several mechanisms have been suggested, including the concomitant use of large amounts of intravenous fluids106,107; sodium and water retention due to the administration of glucocorticoids, which are used to ensure fetal lung maturity; β-adrenergic stimulation of antidiuretic hormone release108; and drug-related tachycardia and increased cardiac output in a cardiovascular system already stressed by the physiology of pregnancy.106,109
Treatment consists of discontinuation of the β-adrenergic agent, diureses, and supplemental oxygen. The response is usually rapid. If there is no improvement within 24 hours, an alternative cause for pulmonary edema should be considered.106,107 Risk factors for pulmonary edema include use of the β-adrenergic agent for more than 24 hours, administration of large volumes of intravenous fluids, multiple gestations, sepsis, and preeclampsia.110
Adult Respiratory Distress Syndrome
ARDS is a form of acute lung injury characterized by increased vascular permeability and pulmonary edema. Diagnostic criteria for ARDS include (1) radiologic evidence of pulmonary edema; (2) elimination of cardiac causes for pulmonary edema (i.e., a pulmonary capillary wedge pressure of less than 12 mmHg); (3) severe hypoxemia requiring greater than 50% inspired oxygen; and (4) reduced lung compliance.111 The overall mortality rate for ARDS in the general population ranges from 30% to 70%, the highest mortality rate being among patients suffering from multisystem organ failure.111
In the pregnant patient, there are several factors that could place the patient at risk for ARDS. Martin and associates112 developed the mnemonic GESTOSIS: Gestational complications; Embolic events; Sepsis; Transfusion (rapid and massive); Other (diabetic ketoacidosis, sickle cell crisis); S ubstance abuse; Irritants (aspiration, burns); and Severe pregnancy-induced hypertension.
Symptoms of ARDS include dyspnea, tachypnea, cyanosis, tachycardia, and changes in mental status that occur 12 to 72 hours after the inciting event.110,113 Treatment consists of elimination of the cause and supportive care consisting of hemodynamic and nutritional support and mechanical ventilation. The most important treatment is delivery of the baby if the patient can tolerate it and the baby is at a safe gestational age.
Guidelines for mechanical ventilation of the pregnant patient are the same as those for the nonpregnant patient with a few exceptions. Because of the mucosal edema of pregnancy, patients are at increased risk for trauma during intubation. Therefore, a smaller endotracheal tube may be needed, and nasotracheal intubation should be avoided.114 Because of a decrease in functional residual capacity and an increase in oxygen consumption, pregnant patients may have a lower oxygen reserve. Since even a short period of apnea can cause significant oxygen desaturation, patients should be given 100% oxygen before intubation.114,115 Hyperventilation should be avoided, however, because respiratory alkalosis can result in decreased uterine blood flow.
Once the patient has been safely intubated, the ventilator settings should be adjusted to maintain a PCO2 of 30 to 32 mmHg, which is normal in pregnancy. The goal of oxygen therapy is to maintain a PO2 of 65 mmHg or higher using the lowest possible level of inspired oxygen so as to minimize oxygen toxicity. Positive end-expiratory pressure is often helpful in maintaining adequate oxygenation in ARDS.
Hemodynamic monitoring is essential, and pulmonary artery catheters may be helpful for managing fluids and following cardiovascular parameters. The lowest possible pulmonary capillary wedge pressure should be maintained in order to minimize further capillary leak in the lungs. However, left ventricular filling pressure should be sufficient to maintain adequate urine output and to avoid maternal hypotension, which may shunt blood from the placenta.116 Hypotensive patients who do not respond to fluid resuscitation or a change of body position to the left lateral decubitus position should be given vasopressor agents to help maintain adequate blood pressure. Ephedrine is the only agent that maintains maternal blood pressure as well as uterine blood flow.117 If the maternal blood pressure remains low, then other vasopressor agents (e.g., dobutamine, epinephrine, dopamine) should be added, despite their adverse effects on uterine blood flow.32,117
CHRONIC PULMONARY DISEASE IN PREGNANCY
Asthma
EPIDEMIOLOGY.
Asthma affects approximately 4% to 6% of adults of all ages and is one of the most common medical conditions complicating pregnancy.118 Asthma may develop de novo during the childbearing years, or it may exist as a persistent abnormality since childhood. In many patients, adult-onset asthma actually represents a recurrence of disease that was originally present during childhood but went into remission during adolescence.119 For unknown reasons, asthma severity appears to be greater in women versus men between the ages of 20 and 50 years.120
Experience suggests that pregnancy has a variable effect on the course of asthma. Different studies have suggested that asthma may improve, worsen, or remain the same during pregnancy.121,122,123,124 This reported variability is likely related to differences in asthma severity in the populations studied, as well as to differences in how asthma severity was assessed. As a rule, however, women with severe asthma before pregnancy are at greater risk of having their asthma worsen during pregnancy.121,122,123,124 Moreover, the effect of pregnancy on asthma symptoms is likely to be consistent during successive pregnancies in an individual patient.122
Although the effects of pregnancy on asthma appear biologically significant rather than random, unpredictable changes in disease control, the mechanisms responsible for these changes are unknown. It has been suggested that airway responsiveness improves during pregnancy, although the changes reported were of small magnitude and the observations were made in very few subjects.125 Although unproven, it is suspected that hormonal changes associated with pregnancy may be related to improvement in symptoms in some patients. Pregnancy-associated deterioration in asthma, however, may be linked to an increased incidence of viral respiratory infections, increased gastroesophageal reflux, and changes in glucocorticoid-receptor responsiveness. It also seems likely that the changes in ventilatory mechanics that occur during pregnancy (i.e., breathing at low lung volumes, where some airways are closed or near closure) may result in increased symptoms when superimposed on the underlying airway obstruction associated with asthma.
Some studies suggest that asthma may have an adverse effect on pregnancy in terms of an increased rate of preterm births, decreased birth weight, and increased rates of neonatal and maternal mortality.126,127 Although other factors may be involved, poor asthma control seems the most likely explanation for these epidemiologic findings.128 Patients with impaired pulmonary function or those requiring hospitalizations for asthma during pregnancy tend to have infants with lower birth weight than asthmatics whose disease is better controlled.129,130 Although the mechanisms for these findings are unclear, maternal hypoxia and alkalosis due to hypocapnia and the hyperventilation of pregnancy may play a role.
DIAGNOSIS.
The diagnosis of asthma is made on the basis of a clinical history of typical symptoms and confirmed by the demonstration of reversible airway obstruction on spirometry. The typical symptoms of asthma include wheezing, chest tightness, cough, and sometimes dyspnea. Patients who have symptoms of a persistent cough or episodes of recurrent “bronchitis” should be suspected of having asthma. If asthma symptoms develop for the first time during pregnancy, it is important to rule out laryngeal dysfunction or upper airway obstruction, pulmonary edema, gastroesophageal reflux with aspiration, and viral respiratory infection. The diagnosis of asthma is facilitated during the childbearing years by a relative paucity of other conditions that mimic asthma. As a rule, chronic obstructive pulmonary disease secondary to smoking does not cause symptoms until the sixth decade of life, unless the patient has associated α1-antitrypsin deficiency.
MANAGEMENT.
It is important to note that asthma is heterogeneous with respect to severity, natural history, and response to therapy. Thus, a single management approach is unlikely to work for all patients. In addition, there may be considerable variation in symptom severity in an individual patient over time. Accordingly, therapy should be tailored to the individual patient, monitored regularly, and adjusted to meet the current level of severity.
Although pharmacologic therapy is a vital component of good management, patient education and avoidance of asthma triggers are also important. The goal of patient education is to improve adherence to treatment recommendations by improving knowledge of the disease and its management. The patient should understand that symptoms fluctuate and that occasional exacerbations are expected. With proper treatment, however, these events can be minimized, and in most cases, a relatively normal lifestyle and normal pregnancy can be anticipated. The patient's individual treatment plan, including the purpose of different drugs and their side effects as well as instruction in the proper use of inhaled medications, should be provided. Another aim of education is to engage the patient in self-management practices, particularly in terms of identifying and avoiding triggers, and recognizing and treating exacerbations in their earliest stages.
The importance of airway inflammation in the pathogenesis of asthma has been demonstrated by pathologic studies revealing inflammatory cell infiltration of airway tissues and air spaces, primarily with eosinophils, but also with activated helper (CD4+) T-lymphocytes and mast cells as well.131,132,133 Thus, asthma, a disorder that manifests clinically by acute episodes superimposed on chronic airway obstruction, displays histologic features of both acute (eosinophilic) and chronic (lymphocytic) inflammation. Because inflammatory processes play such an important role in asthma pathogenesis, recommended chronic therapy for asthma now places greater emphasis on agents that interfere with inflammatory pathways than on agents designed to relax airway smooth muscle.
Drugs currently available to treat asthma may be classified as “reliever” or “controller” agents, depending on their principal pharmacodynamic effect. Short-acting bronchodilators such as inhaled β-agonists are considered reliever agents, because they are to be taken for acute symptom relief on an as-needed basis. Corticosteroids, cromolyn sodium, nedocromil sodium, sustained-release theophylline, and long-acting β-agonists are considered controller agents, because they are used to achieve and maintain control of symptoms and are used daily on a long-term basis. In general, medications that are commonly used for chronic management of asthma may be taken safely by the pregnant patient.
Inhaled β2-adrenergic agonists are the drugs of choice for acute symptoms.134 Activation of the β2-adrenergic receptor leads to relaxation of airway smooth muscle and bronchodilatation. There is also evidence that β-agonists inhibit mast cell and eosinophil secretory function in vitro, although the clinical importance of these effects is unknown. Short-acting β-agonists provide rapid bronchodilatation and have a 3- to 6-hour duration of activity. Tachycardia, palpitations, and tremor may be seen with excessive dosing, and hypokalemia may occur at even higher doses. Since short-acting inhaled β-agonists are used to relieve acute symptoms, patients with well-controlled asthma should require only minimal use of these agents. There is no evidence that their use is associated with adverse outcomes for the mother or fetus.135 Long-acting inhaled β-agonists have at least a 12-hour duration of activity and are not recommended for short-term relief of acute symptoms. Rather, they are considered controller agents and should be given twice daily on a long-term basis. Because these long-acting agents are relatively new, data are scarce regarding their use in pregnancy. Since selective β2-adrenergic agonists are effective and safe, the use of nonselective sympathomimetics, particularly inhaled epinephrine, is discouraged during pregnancy. Subcutaneous epinephrine, however, may be considered in the treatment of severe, acute asthma when there is difficulty delivering β-agonists by the inhaled route.
Theophylline was used for many years as first-line treatment for asthma. More recently, however, slow-release theophylline has been used primarily as added therapy in patients whose symptoms fail to abate with inhaled corticosteroids. Theophylline has relatively weak bronchodilator activity in therapeutic doses, and recent evidence suggests that it may also have anti-inflammatory properties.136 The dose-related adverse effects of theophylline are well known, as are the multiple drug interactions that lead to altered theophylline metabolism. Nervousness, anxiety, tachycardia, and nausea are the earliest manifestations of toxicity. Recent studies have shown that the use of slow-release theophylline during pregnancy was not associated with an increased risk of premature contractions, placenta previa, placental abruption, prolonged third phase of delivery, or postpartum hemorrhage.137 In the same study, an increased incidence of preeclampsia and neonatal jaundice was observed in asthmatics taking theophylline, compared to asthmatics not taking theophylline and to nonasthmatic pregnant controls. The latter findings are confounded, however, on two counts: (1) it was indicated that patients who were taking theophylline had severer asthma compared to the other groups; and (2) previous studies have demonstrated a relation between asthma severity and the occurrence of preeclampsia.128 Because there is a reduction in protein binding to theophylline during pregnancy, it is recommended that plasma theophylline concentrations be kept between 8 and 12 mg/L for optimum management. At these levels, there are usually no adverse effects on the mother or fetus. Patients who are taking theophylline may also breastfeed, since less than 1% of the theophylline diffuses from plasma to breast milk.138
Glucocorticoids are the most effective agents available for moderate to severe asthma.139 Glucocorticoids are available for systemic or inhalational use. Side effects such as adrenal suppression, osteoporosis, weight gain, hypertension, diabetes, dermal thinning, cataracts, myopathy, and psychotic reactions are dose related and are usually seen with systemic steroids. Local side effects, including oral thrush and dysphonia, may occur at lower doses of inhaled glucocorticoids. Although chronic use of oral or systemic glucocorticoids has been associated with decreased birth weight,140 similar effects have not been reported with inhaled glucocorticoids. Inhaled glucocorticoids available in the United States include beclomethasone, triamcinolone, and flunisolide. Of these, beclomethasone is recommended during pregnancy because of its safety record in large numbers of pregnant patients.
Cromolyn sodium and nedocromil sodium are classified as anti-inflammatory agents and are thought to inhibit chemical mediator release from mast cells. Both agents are remarkably safe for the patient, although there is no reported experience with nedocromil during pregnancy. In adults, these agents are most often prescribed for patients with mild disease.
Optimal management requires a stepwise approach to drug therapy based on disease severity.141 Such an approach recognizes that severity may vary over time, and it considers treatment an ongoing process whereby increments and decrements in therapy are made in accordance with changes in severity. Although errors in management are often related to undertreatment with drugs, overtreatment can also be a problem, especially in patients with moderate to severe asthma, who tend to remain on a static treatment regimen even after they have achieved symptom control and clinical stability. Principles of management in the pregnant asthmatic patient have been thoroughly reviewed in the Report of the Working Group on Asthma and Pregnancy, written by an expert panel of specialists in obstetrics and gynecology, pulmonology, allergy, and pharmacology.142
Intermittent Asthma.
Most asthmatics experience only mild, intermittent symptoms that are easily reversed by inhaled bronchodilators, which usually provide satisfactory results with no major side effects. If symptoms occur under predictable circumstances (e.g., with exercise), preventive treatment with an inhaled β-agonist or cromolyn sodium is recommended. As a rule, sole therapy with inhaled β-agonists on an as-needed basis is recommended for patients with normal to near-normal lung function (FEV1 greater than 80% predicted) and little peak flow variability (less than 20%).
Mild Persistent Asthma.
The addition of a controller agent is recommended when mild symptoms are no longer intermittent.141 There is no consensus, however, on the frequency with which symptoms must occur before asthma is no longer considered intermittent. Although some guidelines recommend adding a controller agent if symptoms occur more than once per week or if nocturnal symptoms occur more than twice per month, in our experience, few physicians prescribe inhaled glucocorticoids until β-agonists are needed more than once daily to control symptoms. Whether current treatment guidelines are too conservative in their recommendations concerning early inhaled glucocorticoid use, or whether many physicians are withholding appropriate treatment, is unclear. The intent of early glucocorticoid use is not only to alleviate current symptoms, but also to prevent long-term complications of asthma. Although unproven, it is thought that untreated airway inflammation can lead to airway remodeling and chronic, persistent airway obstruction. Autopsy studies in patients with longstanding asthma who died of unrelated causes have disclosed narrowing of small airways with peribronchial fibrosis.143 The finding that elderly patients with longstanding asthma have persistent abnormalities in lung function and tend to have less reversibility144 also favors the notion that longstanding inflammation might have adverse effects.
The most compelling argument for the use of inhaled glucocorticoids in patients with mild persistent asthma derives from a controlled comparison of the effects of inhaled glucocorticoids versus regularly scheduled inhaled β-agonists in patients with newly detected, mild persistent asthma in a 2-year trial.145,146 Compared to β-agonists, inhaled glucocorticoids provided better peak expiratory flow rates, fewer symptoms, and less use of rescue β-agonists.145 When inhaled glucocorticoids were discontinued or reduced in dosage, patients tended to deteriorate.146 After 2 years, patients in the β-agonist treatment arm were switched to inhaled glucocorticoids, and their asthma control improved significantly. Of particular note was the finding that after 2 years of β-agonist use, those patients who switched to inhaled glucocorticoids had less improvement in airway responsiveness and peak flow than those given inhaled glucocorticoids from the beginning as first-line therapy.146 This suggests that earlier intervention with inhaled glucocorticoids can result in better lung function as well as better symptom control over the long term.
As a rule, dosages less than 1000 μg/day are considered very effective and safe for most patients using currently available formulations of beclomethasone, triamcinolone, and flunisolide. Toxicity of inhaled glucocorticoids relates to pituitary-adrenal suppression and osteoporosis, although osteoporosis is largely a concern in postmenopausal women. Studies of inhaled glucocorticoid effects on pituitary-adrenal function have yielded conflicting results because of differences in methodology and drug-delivery techniques.
We recommend inhaled glucocorticoid therapy for any patient who requires rescue β-agonists on a daily basis to control symptoms, or for patients with moderate impairment of lung function (FEV1 less than 75% predicted). Nonglucocorticoid anti-inflammatory drugs (e.g., cromolyn) and even long-acting β-agonists offer alternative approaches to controlling symptoms in patients with mild persistent asthma. Cromolyn is thought to benefit patients with mild persistent asthma whose symptoms are increasing in frequency but occur less than once per day. In patients with symptoms that are severer or more frequent, however, better results are achieved with the use of inhaled glucocorticoids.
Moderate Persistent Asthma.
Moderate persistent asthma is characterized by symptoms that require inhaled β-agonists several times a day for relief. Moderate persistent asthma is best treated with inhaled glucocorticoids to control inflammation and with inhaled β-agonists taken as needed to relieve breakthrough symptoms. The benefits of inhaled glucocorticoids are usually appreciable within the first 2 weeks of therapy.145 Although quite effective for controlling asthma, glucocorticoids do not provide a cure, as discontinuation of medication after long-term treatment can result in deterioration.147
The threshold dose for efficacy varies considerably within the asthmatic population. Thus, the choice of an initial dose is largely empiric. We recommend starting at a higher dose initially (600 to 1000 μg/day) and then reducing the dose after control is achieved. Comparisons of different inhaled glucocorticoid preparations in terms of relative therapeutic or toxic potencies have not been carried out. It is apparent, however, that different products are not equivalent on a puff-per-puff basis. Rather, they are considered equivalent in terms of efficacy on a microgram-per-microgram basis, keeping in mind that they may not be equivalent in terms of toxicity. Most patients achieve adequate control of asthma at doses less than 1000 μg/day.
In a small number of patients, inhaled glucocorticoid doses less than 1000 μg/day may not be sufficient to control symptoms. In such cases, therapy can be increased by increasing the dose of inhaled glucocorticoids or by adding another medication, particularly a long-acting β-agonist or sustained-release theophylline. Although either approach is likely to improve asthma control, recent studies suggest that the addition of a long-acting β-agonist provides greater benefits than an increase in inhaled glucocorticoids.148 Thus, for many patients with borderline severe persistent asthma, control can be achieved without increasing inhaled glucocorticoid doses to levels where systemic effects are encountered.
Severe Persistent Asthma.
Severe persistent asthma is characterized by persistent symptoms despite treatment with inhaled glucocorticoids (less than 1000 μg/day) and additional therapy with either long-acting β-agonists or theophylline. Since severe persistent asthma is often caused by poor adherence to drug regimens or exposure to environmental factors or drugs that trigger asthma episodes, such as aspirin or β-blockers, a thorough investigation of these factors is indicated before augmenting drug therapy.
In severe persistent asthma, there are limited therapeutic options, including increasing the dose of inhaled glucocorticoid, adding sustained-release theophylline in patients already taking inhaled glucocorticoids and long-acting β-agonists, or adding a long-acting β-agonist in patients already taking inhaled glucocorticoids and theophylline. Patients who are not controlled despite treatment with inhaled glucocorticoids up to 2000 μg/day and one or more long-acting bronchodilators are considered candidates for oral steroid therapy.
Oral glucocorticoids should be given in the lowest dose possible on alternate mornings, although daily or even twice-daily doses may be required in some patients. One strategy is to give oral glucocorticoids in a moderate to high doses (0.5 mg/kg/day) for a period of 8 to 21 days, followed by a tapering course to the lowest dose that maintains control. This strategy is often sufficient to modify the underlying airway inflammation and permit better control at lower steroid doses. Although a threshold dose is required to maintain stability in some patients, in others this strategy may achieve sufficient results to permit complete discontinuation of oral glucocorticoids.
Whereas potent and higher dose formulations of inhaled glucocorticoids are on the horizon, the administration of increasing doses of inhaled glucocorticoids may become more appropriate than the use of oral steroids in severe persistent asthma. For the same therapeutic effect, inhaled glucocorticoids have been shown to produce fewer steroid-related side effects compared to oral glucocorticoids.149 A comparison of oral prednisolone (40 mg/day) with budesonide (3.2 mg/day) has also demonstrated greater systemic toxicity with prednisolone despite greater efficacy with budesonide.150 High doses of fluticasone (1000 μg twice daily) have also been shown to improve asthma control and quality of life measures while at the same time permitting a reduction of oral prednisone in severe asthmatics.151 Thus, improved formulations of inhaled glucocorticoids may in the future obviate the need for chronic oral prednisone.
TREATMENT OF ACUTE EXACERBATIONS.
The principle goal of treatment is rapid relief of airflow obstruction. In patients with severe obstruction, correction of hypoxemia and hypercapnia is also of importance. Because symptoms and physical findings do not accurately predict the severity of airflow obstruction, spirometry or peak flow measurements are required for proper assessment of disease severity as well as response to treatment. Reversal of airflow obstruction is most effectively achieved by early administration of inhaled β2-adrenergic bronchodilators. Systemic corticosteroids are usually required for successful resolution of severe exacerbations. Because of a delayed onset of action, they should be given early in the course of treatment to patients who fail to respond rapidly to β-agonists. These general principles apply to the emergency care of all asthmatic patients. In caring for the pregnant asthmatic, additional attention should be paid to preventing fetal hypoxemia. Thus, administration of supplemental oxygen to keep maternal oxygen saturation greater than 95% is recommended. Intensive fetal monitoring by continuous electronic monitoring or by intermittent auscultation is also recommended. Patients with a history of prior life-threatening episodes of asthma are at high risk and should be managed aggressively in the hospital setting.
ASTHMA MANAGEMENT DURING LABOR AND DELIVERY.
As a rule, asthma exacerbations are uncommon during labor and delivery.122 It is recommended that stable patients be given their usual medications during labor and delivery. To verify stability, a peak flow measurement should be obtained when the patient is admitted. If the patient has required chronic oral glucocorticoids or numerous short courses of oral steroids to control asthma during pregnancy, stress doses of parenteral steroids should be given until 24 hours postpartum to prevent exacerbations during labor. In all cases, fetal monitoring is advised. In the stable asthmatic, monitoring can be performed by intermittent auscultation if an initial period of electronic monitoring fails to disclose fetal distress. In patients with uncontrolled asthma during labor, however, continuous electronic monitoring is advised.
Oxytocin is not associated with bronchoconstriction and is therefore the drug of choice for labor induction. Analogs of prostaglandin F2α (PGF2α) and PGE2, however, are known to cause bronchospasm in asthmatics and should be avoided.152,153 Similarly, oxytocin is preferred for treating postpartum hemorrhage. If oxytocin is not successful in arresting hemorrhage, a PGE2 suppository may be the safest of additional alternatives, since both methylergonovine and PGF2α have been associated with bronchospasm.153,154,155 Topical use of PGE2 for cervical ripening has not been reported to cause adverse respiratory effects.
For relief of pain and anxiety during labor, the narcotic analgesic fentanyl is preferred over morphine because of the histamine-releasing properties of the latter. Regional analgesia by the lumbar epidural approach is also recommended for asthmatics because it reduces minute ventilation, respiratory alkalosis, and increased risk of fetal hypoxia. If general anesthesia is required for surgical delivery, halogenated anesthetics in low doses are recommended because of their bronchodilator properties.156,157 Ketamine is the preferred anesthetic agent for use during induction, since it, too, has bronchodilator effects.158 In patients with hypertension and preeclampsia, however, ketamine should be avoided. If muscle relaxation is required, pancuronium is the preferred agent because of the histamine-releasing properties of succinylcholine.159
Cystic Fibrosis
Cystic fibrosis is an autosomal-recessive disorder resulting from a mutation of a single gene on chromosome 7.160 Among whites in the United States, it occurs in approximately 1 in 3000 live births.161 It is characterized by exocrine gland dysfunction, with the lungs and pancreas being primarily affected. The pulmonary disease consists of chronic, recurrent infections, bronchiectasis, and airway obstruction. With improved treatment, patients are surviving well into their childbearing years. Those with mild disease and good nutritional status tend to tolerate pregnancy well. Those with more advanced disease and evidence of cor pulmonale, hypoxemia, severe obstructive lung disease, and malnutrition should be advised against pregnancy. Many patients lie between these two extremes, and the advisability of pregnancy should be based on disease stability. As a rule, patients with a significant decline in pulmonary function over a short time period and those with frequent infectious exacerbations are best advised against pregnancy.161
Patients should be observed closely during pregnancy and should be treated early with antibiotics and bronchodilators when they exhibit signs of an exacerbation. Termination of pregnancy should be considered if worsening respiratory function, right heart failure, refractory hypoxemia, or respiratory acidosis occurs.161
Sarcoid
Sarcoid is a disease of unknown etiology characterized by granulomatous inflammation of body tissues, most commonly the lung, liver, lymph nodes, eyes, and skin. Clinical manifestations include dyspnea, nonproductive cough, chest pain, fever, joint/muscle aches, visual changes, fatigue, cardiac arrhythmias, neurologic symptoms, and erythema nodosum. Patients are often asymptomatic, the disease being discovered incidentally on a chest radiograph performed for other reasons. The diagnosis is made by a biopsy of an involved organ (usually a transbronchial biopsy) that shows noncaseating granulomas. The patient may also have laboratory abnormalities including hypercalcemia, elevated liver function, and an increased angiotensin-converting enzyme (ACE) level. An increased ACE level is not diagnostic of sarcoid, but monitoring levels is helpful in following disease activity.
The stage of sarcoid is determined by the abnormalities found on chest radiograph.162Stage I demonstrates hilar adenopathy alone. Stage II involves both hilar adenopathy and parenchymal infiltrates. Stage III shows parenchymal infiltrates and pulmonary fibrosis. Pulmonary function tests may be normal, restricted, or obstructed if there is endobronchial involvement. There may also be a reduction in the diffusion capacity.
Sarcoid is treated with corticosteroids, and the indications are the same for pregnant and nonpregnant patients. Patients who show progression of disease as evidenced by a decreased diffusion capacity, elevation of liver function, or poorly controlled skin involvement should be placed on a moderate dose of steroids. They should remain on medication for several months, with the dose gradually decreased in conjunction with careful monitoring to ensure against disease reactivation. After initial treatment, some patients are able to remain off steroids indefinitely. Patients with central nervous system, cardiac, or ophthalmologic involvement may need to stay on low-dose corticosteroids on a long-term basis.
Sarcoid has not been found to increase the incidence of fetal or obstetric complications.163 The literature suggests that pregnancy causes an improvement in sarcoid, which is possibly due to an increased concentration of circulating corticosteroids.164,165 It remains unclear from these studies, however, whether this improvement was truly secondary to pregnancy or whether patients improved solely as a natural progression of the disease. Some patients with more active disease demonstrated improvement during pregnancy but relapsed 3 to 6 months postpartum.164 Factors believed to be indicative of a poor prognosis in pregnancy include parenchymal infiltrates on chest radiograph, advanced stage, advanced maternal age, low inflammatory activity, disease control requiring medications other than corticosteroids, and the presence of extrapulmonary disease.165
Lymphangioleiomyomatosis
Lymphangioleiomyomatosis is an idiopathic disease characterized by the proliferation of smooth muscle in the pulmonary vasculature, airways, and lymphatics. It occurs exclusively in women during their childbearing years.166 Clinical manifestations include dyspnea with exertion, cough, chest pain, recurrent pneumothorax, hemoptysis, and a chylous pleural effusion. Chest radiography demonstrates increased lung volumes and diffuse reticular nodular infiltrates. Computerized tomography of the chest shows characteristic thin-walled cystic air spaces.167 Pulmonary function testing reveals a severe decrease in diffusion capacity and an obstructive defect.
Hormonal influences are believed to be important in the pathogenesis of the disease. Receptors sites for estrogen and progesterone have been found in the lungs of these patients, but not in the normal population.168,169,170 Since lymphangioleiomyomatosis may progress during pregnancy, patients should be advised against childbearing.169,171 There is no known treatment at this point, although there have been some reports suggesting that treatment with progesterone or oophorectomy may halt disease progression.166,169,170
Eosinophilic Granuloma
Eosinophilic granuloma is a disease of unknown etiology that occurs mainly in current or former smokers between the ages of 20 to 40. It is more common in men (64%) than in women (36%).172 It is usually confined to the lungs but may cause diabetes insipidus and cystic bone lesions. Symptoms include cough, dyspnea, chest pain, weight loss, fever, and pneumothorax. The classic radiologic findings include ill-defined or stellate nodules, reticulonodular infiltrates, upper zone cysts, preservation of lung volumes, and costophrenic angle sparing. Pulmonary function tests may be normal or may demonstrate either restrictive or obstructive disease. There is usually a reduced diffusion capacity. These patients may demonstrate hypoxemia with exercise and develop pulmonary hypertension and cor pulmonale.
The diagnosis is made by a lung biopsy that reveals stellate lesions located near bronchioles and blood vessels.173 The classic cell found in these lesions is the histiocyte or Langerhans' cell. Although eosinophilic granuloma is progressive, there have been reports of improvement with steroid treatment. A few patients who became pregnant demonstrated no adverse outcome.172
Kyphoscoliosis
Kyphoscoliosis is a bony abnormality of the spine characterized by either excessive posterior or lateral curvature. It can cause respiratory failure and death due to decreased lung volumes, atelectasis, hypoxemia from ventilation-perfusion abnormalities, hypercapnia, increased work of breathing, and respiratory muscle fatigue.172 The risk of progression of the curvature during pregnancy is low unless the patient has an unstable scoliosis at the time of pregnancy. Premature birth rates are higher in these patients compared to the normal population.174
CONCLUSION
Physiologic dyspnea and hyperventilation are common symptoms of pregnancy, but they are usually benign and not reflective of serious underlying pathology. Several serious pulmonary complications of pregnancy may occur, however, and because they can adversely affect outcomes for both mother and fetus, physicians should be aware of their occurrence and familiar with prevention and treatment strategies. Thromboembolic disease, amniotic fluid embolism, aspiration, pneumonia, and pulmonary edema are the most commonly encountered serious complications. Chronic pulmonary diseases, most notably asthma and sarcoid, can adversely affect pregnancy if disease is poorly controlled; conversely, pregnancy can often affect the clinical course of patients with these chronic conditions. Although pregnant patients with pulmonary disorders are often treated with medications commonly used in the nongravid patient, the effects of many of these medications on pregnancy and fetal development are unknown. More studies of the effects of new therapies are needed in pregnant women to ensure patient access to the most effective treatments, and to provide knowledge of the risks of such treatments.
REFERENCES
Fishburne JI: Physiology and disease of the respiratory system in pregnancy. J Reprod Med 22: 177, 1979 |
|
Ueland K: Maternal cardiovascular dynamics: VII. Intrapartum blood volume changes. Am J Obstet Gynecol 126: 671, 1976 |
|
Zeldis SM: Dyspnea during pregnancy: Distinguishing cardiac from pulmonary causes. Clin Chest Med 13: 567, 1992 |
|
Elkus R, Popovich J: Respiratory physiology in pregnancy. Clin Chest Med 13: 555, 1992 |
|
Hughes CW, Popovich J Jr: Pinpointing the cause of respiratory crises in pregnancy. J Crit Illness 5: 559, 1990 |
|
Ratigan TR: Anatomic and physiologic changes of pregnancy: Anesthetic considerations. J Am Assoc Nurse Anesth 51: 38, 1983 |
|
James CF, Banner T, Levelle P, Caton D: Noninvasive determination of cardiac output throughout pregnancy (abstr). Anesthesiology 63: A434, 1985 |
|
Mashini IS, Albazzaz SJ, Fadel HE et al: Serial noninvasive evaluation of cardiovascular hemodynamics during pregnancy. Am J Obstet Gynecol 156: 1208, 1987 |
|
Robson SC, Hunter S, Boys RJ, Dunlop W: Serial study of factors influencing changes in cardiac output during human pregnancy. Am J Physiol 256:(H)1060, 1989 |
|
Mabie WC, DiSessa TG, Crocker LG et al: A longitudinal study of cardiac output in normal human pregnancy. Am J Obstet Gynecol 170: 849, 1994 |
|
Katz R, Karliner JS, Resnick R: Effects of a natural volume overload state (pregnancy) on left ventricular performance in normal human subjects. Circulation 58: 434, 1978 |
|
Lapinsky SE, Kruczynski K, Slutsky AS: Critical care in the pregnant patient. Am J Respir Crit Care Med 152: 427, 1995 |
|
Elkayam U, Gleicher N: Hemodynamics and cardiac function during normal pregnancy and the puerperium. In Elkayam U, Gleicher N (eds): Cardiac Problems in Pregnancy, pp 5 – 24. New York, Alan R Liss, 1990 |
|
Elkayam U: Pregnancy and cardiovascular disease. In Braunwald E (ed): Heart Disease, pp 1790 – 1791. Philadelphia, WB Saunders, 1992 |
|
Contreras G, Guttierrez M, Beroiza T et al: Ventilatory drive and respiratory muscle function in pregnancy. Am Rev Respir Dis 144: 837, 1991 |
|
McGinty AP: The comparative effects of pregnancy and phrenic nerve interruption on the diaphragm and their relation to pulmonary tuberculosis. Am J Obstet Gynecol 35: 237, 1938 |
|
Bonica JJ: Maternal respiratory changes during pregnancy and parturition. In Marx (ed): Parturition and Perinatology, pp 2 – 19. Philadelphia, FA Davis, 1973 |
|
Cugell DW, Frank NR, Gaensler EA, Badger TL: Pulmonary function in pregnancy: I. Serial observations in normal women. Am Rev Tuberc 67: 568, 1953 |
|
Alaily AB, Carrol KB: Pulmonary ventilation in pregnancy. Br J Obstet Gynaecol 85: 518, 1978 |
|
Baldwin GR, Moorthi DS, Whelton JA, MacDonnell KF: New lung function and pregnancy. Am J Obstet Gynecol 127: 235, 1977 |
|
Prowse CM, Gaensler EA: Respiratory and acid-base changes during pregnancy. Anesthesiology 26: 381, 1965 |
|
Milne JA, Mills RJ, Howie AD, Pack AI: Large airways function during normal pregnancy. Br J Obstet Gynaecol 84: 448, 1977 |
|
Gee JBL, Packer BS, Millen JE, Robin ED: Pulmonary mechanics during pregnancy. J Clin Invest 46: 945, 1967 |
|
Milne JA, Mills RJ, Coutts JR et al: The effect of human pregnancy on the pulmonary transfer factor for carbon monoxide as measured by the single breath method. Clin Sci Mol Med 53: 271, 1977 |
|
Rees GB, Pipkin FB, Symonds EM, Patrick JM: A longitudinal study of respiratory changes in normal human pregnancy with cross-sectional data on subjects with pregnancy-induced hypertension. Am J Obstet Gynecol 162: 826, 1990 |
|
Skatrud JB, Bempsey JA, Kaisen DG: Ventilatory response to medroxyprogesterone acetate in normal subjects: Time course and mechanism. J Appl Physiol 44: 939, 1978 |
|
Lucius H, Gahlenbeck H, Kleine HO et al: Respiratory functions, buffer system, and electrolyte concentrations of blood during human pregnancy. Respir Physiol 9: 311, 1970 |
|
Andersen GJ, James GB, Mathers NP et al: The maternal oxygen tension and acid-base status during pregnancy. J Obstet Br Commonw 76: 16, 1969 |
|
Templeton A, Kelman GR: Maternal blood-gases (PAO2 -PaO2 ), physiological shunt and VD/VT in normal pregnancy. Br J Anaesth 48: 1001, 1976 |
|
Buss DD, Bisgard GE, Rawling CA, Rankin JHG: Uteroplacental blood flow during alkalosis in the sheep. Am J Physiol 228: 1497, 1975 |
|
Meschia G: Supply of oxygen to the fetus. J Reprod Med 23: 160, 1979 |
|
Clark RB, Brunner JA: Dopamine for the treatment of spinal hypotension during cesarean section. Anesthesiology 53: 514, 1980 |
|
Crawford JS: Maternal and cord blood at delivery: Vertex vaginal delivery of mature infant: Parameters of respiratory exchange. Biol Neonatol 8: 131, 1965 |
|
Wulf H: The oxygen and carbon dioxide tension gradients in the human placenta at term. Am J Obstet Gynecol 88: 38, 1964 |
|
Khazin AF, Hon EH, Hehre FW: Effects of maternal hyperoxia on the fetus I: Oxygen tension. Am J Obstet Gynecol 109: 628, 1971 |
|
Willcourt RJ, King JC, Queenan JT: Maternal oxygen administration and the fetal transcutaneous PO2. Am J Obstet Gynecol 146: 714, 1983 |
|
Althabe O, Schwarcz RL, Pose SV et al: Effects of fetal heart rate and fetal PO2 of oxygen administration to the mother. Am J Obstet Gynecol 98: 858, 1967 |
|
Arduini D, Rizzo G, Mancuso S, Romanini C: Shortterm effects of maternal oxygen administration on blood flow velocity waveforms in healthy and growth retarded fetuses. Am J Obstet Gynecol 159: 1077, 1988 |
|
Tenholder MF, South-Paul J: Dyspnea in pregnancy. Chest 96: 381, 1989 |
|
Milne J, Howie A, Pack A: Dyspnea during normal pregnancy. Br J Obstet Gynaecol 85: 260, 1978 |
|
Gilbert R, Auchincloss J: Dyspnea of pregnancy: Clinical and physiological observation. Am J Med Sci 252: 270, 1966 |
|
Leontic E: Dyspnea in pregnancy. J Perinat Med 3: 154, 1975 |
|
Elkayam U, Gleicher N: Changes in cardiac findings during normal pregnancy. In Elkayam U, Gleicher N (eds): Cardiac Problems in Pregnancy, pp 31 – 38. New York, Alan R Liss, 1990 |
|
Kaunitz AM, Hughes JM, Grimes DA et al: Causes of maternal mortality in the United States. Obstet Gynecol 65: 605, 1985 |
|
Lederman MM: Cell-mediated immunity and pregnancy. Chest 86 (suppl): 6S, 1984 |
|
McKinney WP, Volkert P, Kaufman J: Fatal swine influenza pneumonia during late pregnancy. Arch Intern Med 150: 213, 1990 |
|
Rodrigues J, Niederman MS: Pneumonia complicating pregnancy. Clin Chest Med 13: 679, 1992 |
|
New drug bulletin: Rimantadine (Flumadine). RN 57:43, 1994 |
|
American Thoracic Society: Prevention of influenza and pneumonia. Am Rev Respir Dis 142:487, 1990 |
|
Cox SM, Cunningham FG, Luby J: Management of varicella pneumonia complicating pregnancy. Am J Perinatol 7: 300, 1990 |
|
Haake DA, Zakowski PC, Haake DL, Bryson YJ: Early treatment with acyclovir for varicella pneumonia in otherwise healthy adults: Retrospective controlled study and review. Rev Infect Dis 12: 788, 1990 |
|
Smego RA, Asperilla MO: Use of acyclovir for varicella pneumonia during pregnancy. Obstet Gynecol 78: 1112, 1991 |
|
Esmonde TF, Herdman G, Anderson G: Chickenpox pneumonia: An association with pregnancy. Thorax 44: 812, 1989 |
|
Harris RE, Rhoades ER: Varicella pneumonia complicating pregnancy: Report of a case and review of literature. Obstet Gynecol 25: 734, 1965 |
|
Steyn DW, Odendaal HJ: Fetal resuscitation in a patient with varicella pneumonia and preterm labor. Int J Gynecol Obstet 30: 171, 1989 |
|
Eder SE, Apuzzio JJ, Weiss G: Varicella pneumonia during pregnancy: Treatment of two cases with acyclovir. Am J Perinatol 5: 16, 1988 |
|
Centers for Disease Control, Department of Health and Human Services: Varicella-Zoster immune globulin for the prevention of chicken pox: Recommendations of the immunization practices advisory committee. Ann Intern Med 100:859, 1984 |
|
Masur H, Allegra C, Armstrong D et al: Guidelines for prophylaxis against Pneumocystis carinii pneumonia for persons infected with human immunodeficiency virus. MMWR 38 (suppl 5): 1, 1989 |
|
American College of Obstetricians and Gynecologists: Human immunodeficiency virus infections. ACOG Technical Bulletin No. 169, June 1992. Int J Gynecol Obstet 41:307, 1993 |
|
Sperling RS, Stratton P: Treatment options for human immunodeficiency virus-infected pregnant women. Obstetric-Gynecologic Working Group of the AIDS Clinical Trials Group of the National Institute of Allergy and Infectious Disease. Obstet Gynecol 79: 443, 1992 |
|
Connelly RT, Lourwood DL: Pneumocystis carinii pneumonia prophylaxis during pregnancy. Pharmacotherapy 14: 424, 1994 |
|
Albino JA, Shapiro JM: Respiratory failure in pregnancy due to Pneumocystis carinii: Report of a successful outcome. Obstet Gynecol 83: 823, 1994 |
|
Harris RE: Coccidioidomycosis complicating pregnancy. Obstet Gynecol 28: 401, 1966 |
|
Wack EE, Ampel NM, Galgiani JN, Bronnimann DA: Coccidioidomycosis during pregnancy: An analysis of ten cases among 47,120 pregnancies. Chest 94: 376, 1988 |
|
Cantanzaro A: Pulmonary mycosis in pregnant women. Chest 86 (suppl): 14S, 1984 |
|
Lee BE, Feinberg M, Abraham JJ, Murthy AR: Congenital malformations in an infant born to a woman treated with fluconazole. Pediatr Infect Dis J 11: 1062, 1992 |
|
Vallejo JG, Starke JR: Tuberculosis and pregnancy. Clin Chest Med 13: 693, 1992 |
|
American Thoracic Society: Diagnostic standards and classification of tuberculosis. Am Rev Respir Dis 142:725, 1990 |
|
American Thoracic Society: Treatment of tuberculosis and tuberculosis infection in adults and children. Am J Respir Crit Care Med 149:1359, 1994 |
|
Centers for Disease Control: Initial therapy for tuberculosis in the era of multidrug resistance: Recommendations of the advisory council for the elimination of tuberculosis. MMWR 42(RR-7):1, 1993 |
|
Smith MHD, Teele DW: Tuberculosis. In Remington JS, Klein JO (eds): Infectious Diseases of the Fetus and Newborn, 3rd ed, pp 1078 – 1079. Philadelphia, WB Saunders, 1990 |
|
Hageman J, Shulman S, Schreiber M et al: Congenital tuberculosis: Critical reappraisal of clinical findings and diagnostic procedures. Pediatrics 66: 980, 1980 |
|
Baggish MS, Hooper S: Aspiration as a cause of maternal death. Obstet Gynecol 43: 327, 1974 |
|
Hollingsworth HM, Pratter MR, Irwin RS: Acute respiratory failure in pregnancy. J Intensive Care Med 4: 11, 1989 |
|
Bynum LJ, Pierce AK: Pulmonary aspiration of stomach contents. Am Rev Respir Dis 114: 1129, 1976 |
|
Hessen I: Roentgen examination of pleural fluid. Acta Radiologica 86 (suppl): 62, 1951 |
|
Hughson WG, Friedman PJ, Feigin DS et al: Postpartum pleural effusion: A common radiologic finding. Ann Intern Med 97: 856, 1982 |
|
Heffner JE, Sahn SA: Pleural disease in pregnancy. Clin Chest Med 13: 667, 1992 |
|
Udeshi UL, McHugo JM, Selwyn Crawford J: Postpartum pleural effusion. Br J Obstet Gynecol 95: 894, 1988 |
|
Goodrich SM, Wood JE: Peripheral venous distensibility and velocity of venous blood flow during pregnancy or during oral contraceptive therapy. Am J Obstet Gynecol 90: 740, 1964 |
|
Ikard RW, Ueland K, Folse R: Lower limb venous dynamics in pregnant women. Surg Gynecol Obstet 132: 483, 1971 |
|
Kerr MG, Scott DB, Samuel E: Studies of the inferior vena cava in late pregnancy. Br Med J 1: 532, 1964 |
|
Stirling Y, Woolf L, North WRS et al: Haemostasis in normal pregnancy. Thromb Haemost 52: 176, 1984 |
|
Hull R, Hirsh J, Sackett DL et al: Clinical validity of a negative venogram in patients with clinically suspected venous thrombosis. Circulation 64: 622, 1981 |
|
Ginsberg J, Turner C, Brill-Edwards P et al: Pseudothrombosis in pregnancy. Can Med Assoc J 139: 409, 1988 |
|
Lensing AW, Prandoni P, Brandjes D et al: Detection of deep vein thrombosis by real-time B-mode ultrasonography. N Engl J Med 320: 342, 1989 |
|
White RH, McGahan JP, Daschback NM, Hartling RP: Diagnosis of deep vein thrombosis using duplex ultrasound. Ann Intern Med 111: 297, 1989 |
|
Hull RD, Raskob GE, Hirsh J: The diagnosis of clinically suspected pulmonary embolism. Chest 89 (suppl): 417s, 1986 |
|
Goodman PC: Pulmonary angiography. Clin Chest Med 5: 465, 1984 |
|
Hull RD, Raskob GE, Coates G, Panju AA: Clinical validity of a normal perfusion lung scan in patients with suspected pulmonary embolism. Chest 97: 23, 1990 |
|
Hull RD, Hirsh J, Carter CJ et al: Diagnostic value of ventilation-perfusion lung scanning in patients with suspected pulmonary embolism. Chest 88: 819, 1985 |
|
The PIOPED Investigators: Value of the ventilation/perfusion scan in acute pulmonary embolism: Results of the prospective investigation of pulmonary embolism diagnosis (PIOPED). JAMA 263:2753, 1990 |
|
Demers C, Ginsberg JS: Deep vein thrombosis and pulmonary embolism in pregnancy. Clin Chest Med 13: 645, 1992 |
|
Gouault-Heilmann M, Huet Y, Adnot S et al: Low molecular weight heparin fractions as an alternative therapy in heparin-induced thrombocytopenia. Haemostasis 17: 134, 1987 |
|
Messmore HL, Fareed J, Corey J et al: In vitro assessment of low molecular weight heparin in patients with thrombocytopenia induced by standard heparin (abstr). Blood 64: 238a, 1984 |
|
Gillis S, Shushan A, Eldor A: Use of low molecular weight heparin for prophylaxis and treatment of thromboembolism in pregnancy. Int J Gynecol Obstet 39: 297, 1992 |
|
Melissari E, Parker CJ, Wilson NV et al: Use of low molecular weight heparin in pregnancy. Thromb Haemost 68: 652, 1992 |
|
Delclos GL, Davila F: Thrombolytic therapy for pulmonary embolism in pregnancy: A case report. Am J Obstet Gynecol 155: 375, 1986 |
|
Hall RJ, Young C, Sutton GC, Cambell S: Treatment of acute pulmonary embolism with streptokinase during labor and delivery. Br Med J 4: 647, 1972 |
|
Rutherford SE, Phelan JP: Deep vein thrombosis and pulmonary embolism in pregnancy. Obstet Gynecol Clin North Am 18: 345, 1991 |
|
Cunningham FG, MacDonald PC, Grant NF: Amniotic fluid embolism. In Williams Obstetrics, 18th ed, p 719. Norwalk, CT, Appleton-Century-Crofts, 1989 |
|
Morgan M: Amniotic fluid embolism. Anaesthesia 34: 20, 1979 |
|
Clark SL: Amniotic fluid embolism. Crit Care Clin 7: 877, 1991 |
|
Peterson EP, Taylor HB: Amniotic fluid embolism: An analysis of 40 cases. Obstet Gynecol 35: 787, 1970 |
|
Masson RG: Amniotic fluid embolism. Clin Chest Med 13: 657, 1992 |
|
Pisani RJ, Rosenow EC III: Pulmonary edema associated with tocolytic therapy. Ann Intern Med 110: 714, 1989 |
|
Hankins GDV: Complications of beta-sympathomimetic tocolytic agents. In Clark SL, Cotton DB, Hankins GDV, Phelan JP (eds): Critical Care Obstetrics, pp 223 – 250. Boston, Blackwell Scientific Publications, 1991 |
|
Schrier RW, Lieberman R, Ufferman RC: Mechanism of antidiuretic effect of beta-adrenergic stimulation. J Clin Invest 51: 97, 1972 |
|
Finley J, Katz M, Rojas-Perez M et al: Cardiovascular consequences of beta-agonist tocolysis: An echocardiographic study. Obstet Gynecol 64: 787, 1984 |
|
Hollingsworth HM, Irwin RS: Acute respiratory failure in pregnancy. Clin Chest Med 13: 723, 1992 |
|
Rinaldo JE: Adult respiratory distress syndrome. In Rippe JM, Irwin RS, Alpert JS et al (eds): Intensive Care Medicine, p 476. Boston, Little, Brown & Co, 1991 |
|
Martin JN, Martin RW, Hess LW: Gestosis: Acronym for the diverse etiologies of pregnancy associated with adult respiratory distress syndrome (ARDS) (abstr). Presented at the American College of Obstetrics and Gynecologists, Atlanta, Georgia, 1989 |
|
McColgin SW, Glee L, Brian BA: Pulmonary disorders complicating pregnancy. Obstet Gynecol Clin North Am 19: 697, 1992 |
|
Cheek TG, Gutsche BB: Maternal physiologic alterations during pregnancy. In Shnider SM, Levinson G (eds): Anaesthesia for Obstetrics, pp 3 – 13. Baltimore, Williams & Wilkins, 1987 |
|
Archer GW, Marx GF: Arterial oxygen tension during apnoea in parturient women. Br J Anaesth 46: 358, 1974 |
|
Erickson NL, Parisi VM: Adult respiratory distress syndrome and pregnancy. Semin Perinatol 14: 68, 1990 |
|
Cosmi EV, Shnider SM: Obstetric anesthesia and uterine blood flow. In Shnider SM, Levinson G (eds): Anesthesia for Obstetrics, pp 22 – 40. Baltimore, Williams & Wilkins, 1987 |
|
Dodge RR, Burrows B: The prevalence and incidence of asthma and asthma-like symptoms in a general population sample. Am Rev Respir Dis 122: 567, 1980 |
|
Bronnimann S, Burrows B: A prospective study of the natural history of asthma. Chest 90: 480, 1986 |
|
Skobeloff EM, Spivey WH, St. Clair SS, Schoffstall JM: The influence of age and sex on asthma admissions. JAMA 268: 3437, 1992 |
|
Gluck JC, Gluck P: The effects of pregnancy on asthma: A prospective study. Ann Allergy 37: 164, 1997 |
|
Schatz M, Harden K, Forsythe A et al: The course of asthma during pregnancy, post partum, and with successive pregnancies: A prospective analysis. J Allergy Clin Immunol 81: 509, 1988 |
|
Stenius-Aarniala B, Piirila P, Teramo K: Asthma and pregnancy: A prospective study of 198 pregnancies. Thorax 43: 12, 1988 |
|
White RJ, Coutts II, Gibbs CJ, MacIntyre C: A prospective study of asthma during pregnancy and the puerperium. Respir Med 83: 103, 1989 |
|
Juniper EF, Daniel EE, Roberts RS et al: Improvement in airway responsiveness and asthma severity during pregnancy: A prospective study. Am Rev Respir Dis 140: 924, 1989 |
|
Bahna SL, Bjerkedal T: The course and outcome of pregnancy in women with bronchial asthma. Acta Allergol 27: 397, 1972 |
|
Gordon M, Niswander KR, Berendes H, Kantor AG: Fetal morbidity following potentially anoxigenic obstetric conditions: VII. Bronchial asthma. Am J Obstet Gynecol 106: 421, 1970 |
|
Schatz M, Zeiger RS, Hoffman CP et al: Perinatal outcomes in the pregnancies of asthmatic women: A prospective controlled analysis. Am J Respir Crit Care Med 151: 1170, 1995 |
|
Greenberger PA, Patterson R: The outcome of pregnancy complicated by severe asthma. Allergy Proc 9: 539, 1988 |
|
Schatz M, Zeiger RS, Hoffman CP: Intrauterine growth is related to gestational pulmonary function in pregnant asthmatic women: Kaiser-Permanente Asthma and Pregnancy Study Group. Chest 98: 389, 1990 |
|
Zangrilli JG, Fish JE, Peters SP: Inflammation in allergic asthma: Findings from invasive studies. In Middleton E, Reed CE, Ellis EF et al (eds): Update: Allergy Principles and Practice, 4th ed, Update 16, pp 1 – 15. St. Louis, CV Mosby, 1994 |
|
Holgate S: Mediator and cytokine mechanisms in asthma. Thorax 48: 103, 1993 |
|
Robinson DS, Hamid Q, Ying S et al: Predominant TH-2-like bronchoalveolar T-lymphocyte population in atopic asthma. N Engl J Med 326: 298, 1992 |
|
Nelson HS: B-adrenergic bronchodilators. N Engl J Med 333: 499, 1995 |
|
Schatz M, Zeiger RS, Harden KM et al: The safety of inhaled beta-agonist bronchodilators during pregnancy. J Allergy Clin Immunol 82: 686, 1988 |
|
Kidney J, Dominguez M, Taylor PM et al: Immunomodulation by theophylline in asthma. Am J Respir Crit Care Med 151: 1907, 1995 |
|
Stenius-Aarniala B, Riikonen S, Teramo K: Slow-release theophylline in pregnant asthmatics. Chest 107: 642, 1995 |
|
Yurchak AM, Jusko WJ: Theophylline secretion into breast milk. Pediatrics 57: 518, 1976 |
|
Barnes PJ, Pedersen S: Efficacy and safety of inhaled corticosteroids in asthma. Am Rev Respir Dis 148 (suppl): S1, 1993 |
|
Reinisch JM, Simon NG, Karow WG, Gandelman R: Prenatal exposure to prednisone in humans and animals retards intrauterine growth. Science 202: 436, 1978 |
|
Global Initiative for Asthma: Global strategy for asthma management and prevention: NHLBI/WHO Workshop report. Department of Health and Human Services Publication No. 95–3659. Bethesda, MD, DHHS, 1995 |
|
Report of the Working Group on Asthma and Pregnancy: Management of asthma during pregnancy. National Institutes of Health Publication No. 93–3279. Bethesda, MD, NIH, 1993 |
|
Sobonya RE: Quantitative structural alterations in long-standing allergic asthma. Am Rev Respir Dis 130: 289, 1984 |
|
Braman SS, Kaemmerlen JT, Davis SM: Asthma in the elderly: A comparison between patients with recently acquired and long-standing disease. Am Rev Respir Dis 143: 336, 1991 |
|
Haahtela T, Järvinen M, Kava T et al: Comparison of a β2 -agonist, terbutaline, with an inhaled corticosteroid, budesonide, in newly detected asthma. N Engl J Med 325: 388, 1991 |
|
Haahtela T, Järvinen M, Kava T et al: Effects of reducing or discontinuing inhaled budesonide in patients with mild asthma. N Engl J Med 331: 700, 1994 |
|
Chervinsky P, van As A, Bronsky EA et al: Fluticasone propionate aerosol for the treatment of adults with mild to moderate asthma. J Allergy Clin Immunol 94: 676, 1994 |
|
Greening AP, Wind P, Northfield M, Shaw G: Added salmeterol versus higher-dose corticosteroid in asthma patients with symptoms on existing inhaled corticosteroids. Lancet 344: 219, 1994 |
|
Toogood JH, Baskerville J, Jennings B et al: Bioequivalent doses of budesonide and prednisone in moderate and severe asthma. J Allergy Clin Immunol 84: 688, 1989 |
|
Hodsman AB, Toogood JH, Jennings B et al: Differential effects of inhaled budesonide and oral prednisolone on serum osteocalcin. J Clin Endocrinol Metab 72: 530, 1991 |
|
Noonan M, Chervinsky P, Busse WW et al: Fluticasone propionate reduces oral prednisone use while it improves asthma control and quality of life. Am J Respir Crit Care Med 152: 1467, 1995 |
|
Smith AP: The effects of intravenous infusion of graded doses of prostaglandins F2a and E2 on lung resistance in patients undergoing termination of pregnancy. Clin Sci 44: 17, 1973 |
|
Mathé AA, Hedqvist P: Effect of prostaglandins F2a and E2 on airway conductance in heathy subjects and asthmatic patients. Am Rev Respir Dis 111: 313, 1975 |
|
Hill H, Geraghty I, Hughes A et al: Ergometrine and bronchospasm. Anaesthesia 42: 1115, 1987 |
|
Crawford JS: Letter to the Editor: Bronchospasm following ergometrine. Anesthesiology 35: 397, 1980 |
|
Schwartz SH: Treatment of status asthmaticus with halothane. JAMA 251: 2688, 1984 |
|
Parnass SM, Feld JM, Chamberlin WH et al: Status asthmaticus treated with isoflurane and enflurane. Anesth Analg 66: 193, 1987 |
|
Corssen G, Gutierrez J, Reeves JG, Huber FC Jr: Ketamine in the anaesthetic management of asthmatic patients. Anesth Analg 51: 588, 1972 |
|
Ertama PM: Histamine liberation in surgical patients following administration of neuromuscular blocking drugs. Ann Clin Res 14: 27, 1982 |
|
Tsui LC, Buchwald M: Biochemical and molecular genetics of cystic fibrosis. Adv Hum Genet 20: 153, 1991 |
|
Nelson-Piercy C, Waldron M, Mooe-Gillon J: Respiratory disease in pregnancy. Br J Hosp Med 51: 398, 1994 |
|
Hunninghake GW: Staging of pulmonary sarcoidosis. Chest 89 (suppl): 178S, 1986 |
|
Selroos O: Sarcoidosis and pregnancy: A review with results of a retrospective survey. J Intern Med 227: 221, 1990 |
|
Agha FP, Vade A, Amendola MA, Cooper RF: Effects of pregnancy on sarcoidosis. Surg Gynecol Obstet 155: 817, 1982 |
|
Haynes de Regt R: Sarcoidosis and pregnancy. Obstet Gynecol 70: 369, 1987 |
|
Eliasson AH, Phillips YY, Tenholder MF: Treatment of lymphangioleiomyomatosis: A meta-analysis. Chest 96: 1352, 1989 |
|
Aberle DR, Hansell DM, Brown K, Tashkin DP: Lymphangiomyomatosis: CT, chest radiographic, and functional correlations. Radiology 176: 381, 1990 |
|
Brentani MM, Carvalho CR, Saldiva PH et al: Steroid receptors in pulmonary lymphangioleiomyomatosis. Chest 85: 97, 1984 |
|
Graham ML II, Spelsberg TC, Dines DE et al: Pulmonary lymphangioleiomyomatosis: With particular reference to steroid receptor assay studies and pathologic correlations. Mayo Clin Proc 59: 3, 1984 |
|
McCarty KS Jr, Mossler JA, McLelland R, Sieker HO: Pulmonary lymphangioleiomyomatosis responsive to progesterone. N Engl J Med 303: 1461, 1980 |
|
Yockey CC, Riepe RE, Ryan K: Pulmonary lymphangioleiomyomatosis complicated by pregnancy. Kans Med 87: 277, 1986 |
|
King TE: Restrictive lung disease in pregnancy. Clin Chest Med 13: 607, 1992 |
|
Colby TV, Lombard C: Histiocytosis X in the lung. Hum Pathol 14: 847, 1983 |
|
Visscher W, Lonstein JE, Hoffman DA et al: Reproductive outcomes in scoliosis patients. Spine 13: 1096, 1988 |