Pulmonary Physiology and Airway Management
Authors
INTRODUCTION
Respiration is the exchange of oxygen and carbon dioxide across permeable membranes; ventilation is the movement of gases into and out of the lungs; breathing is the mechanical process of attempting to ventilate. We breathe to ventilate and ventilate to respire. Pregnancy entails clinically insignificant alterations of the airways and lung parenchyma, thereby minimally affecting respiratory gas exchange. However, pregnancy encompasses significant alterations of chest wall and diaphragm function as well as hormonal factors that diminish the efficiency of pulmonary mechanics and increase myocardial demands. The physiology and assessment of normal gas exchange and pulmonary mechanics must be understood before pregnancy-induced alterations can be evaluated.
PULMONARY FUNCTION
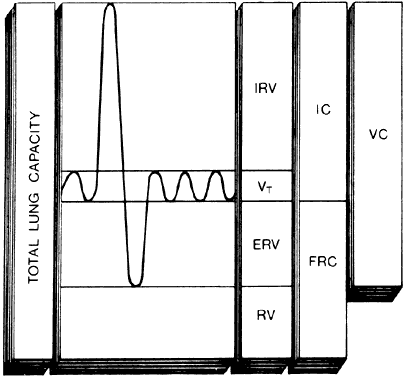
The quantification of pulmonary gas volumes and dynamics is the traditional reference point for documentation and assessment of pulmonary function. Total lung capacity (TLC) is defined as the volume of gas in the lungs at the end of a maximal inspiratory effort. The TLC is divided into four volumes (Fig. 1): inspiratory reserve volume (IRV), tidal volume (VT), expiratory reserve volume (ERV), and residual volume (RV). RV is that volume remaining in the lungs after a maximal expiratory effort.
Combinations of two or more lung volumes are referred to as capacities. Functional residual capacity (FRC) is composed of RV and ERV, representing the amount of gas remaining in the lungs at the end of expiration. Pregnancy entails diminishment of FRC.1 Decreases in FRC result in smaller alveoli being well-perfused (low V/Q), resulting in diminished alveolar oxygen tensions and thereby diminished arterial oxygen tensions. Additionally, the smaller the FRC, the faster alveolar gas tensions equilibrate with the inspired gas tensions, accounting for the relatively rapid induction and emergence of general anesthesia during pregnancy.
Inspiratory capacity (IC) = VT + IRV, which reflects the patient's ability to take a deep breath. Normally, IC is 75% of the vital capacity.
Vital capacity (VC) = IRV + VT + ERV, which represents the patient's total reserves available for breathing and normally is 55–80 mL/kg. A VC of less than 15 mL/kg represents a severe limitation in ventilatory reserves and demands close observation. Most important, the VC is a readily obtainable measurement requiring only a simple spirometer and patient cooperation. The forced vital capacity (FVC) maneuver can be visualized as the forced expiratory spirogram. The FEV1 is the volume of air forcibly exhaled in the first second of an FVC. Normally, at least 80% of the FVC should be exhaled in the first second. FEV1% = measured FEV1/measured FVC.
Abnormal spirometric studies generally are categorized as restrictive or obstructive abnormalities. A restrictive component implies a condition in which VC is diminished contrasted to a restrictive disease, which is diagnosed by measurement of a reduced TLC. An obstructive component implies airway obstruction, characterized by a reduction in expiratory flow rates. This must be distinguished from obstructive disease, which is diagnosed by measurement of normal or increased TLC plus decreased flow rates. Spirometric studies during pregnancy are relatively comparable to nongravid studies in patients free of lung disease.
RESPIRATORY GAS EXCHANGE
External respiration occurs between alveolar gas and pulmonary capillary blood; internal respiration occurs between systemic capillary blood and tissue. Arterial blood gas measurements reflect the balance between internal and external respiration. Ventilation is composed of gas that respires (alveolar ventilation) and gas that does not respire (dead-space ventilation). The balance between alveolar ventilation and the CO2 production is reflected in the arterial PCO2 value. The arterial PO2 reflects the adequacy with which the pulmonary blood flow is oxygenated by the lungs.
Pregnancy does not affect blood gas values in the first two trimesters, but both arterial PCO2 and PO2 are diminished in the last trimester.2, 3 Changes in pulmonary mechanics resulting in diminished end-expiratory lung volumes as well as hormonally induced increases in the ventilatory drive result in arterial PCO2 values slightly less than the normal nongravid values. Increases in total oxygen consumption as well as increased perfusion of gravity-dependent lung areas result in arterial PO2 values slightly less than the normal nongravid values. Although these variances must be accounted for when interpreting arterial blood gas measurements in the last trimester of pregnancy, significant abnormalities seldom are attributable to normal processes and should be assumed to reflect acute cardiopulmonary pathology.
THE PHYSIOLOGY OF PULMONARY MECHANICS
The lungs function as a to-and-fro valveless pump powered by muscles that are anatomically and functionally separate. This mechanically inefficient system is far more susceptible to “failure” than more efficiently designed systems such as the heart.
Autonomic control of respiration
The normal drive to breathe is an unconscious process originating in the midbrain respiratory center comprised of groups of cells with stimulatory and inhibitory effects. The sum of the activities of these cells results in a rhythmic initiation of inspiratory muscle activity that is modulated by central chemoreceptors sensitive to changes in the hydrogen ion concentration of the cerebrospinal fluid, which is acutely affected by fluctuations of the arterial PCO2. In addition to these central chemoreceptors, there are peripheral chemoreceptors located in the aortic arch and at the bifurcation of the internal and external carotid arteries that further modify discharge from the respiratory center. The peripheral chemoreceptors are sensitive to oxygen availability and increase ventilation (and cardiac output) in response to a fall in oxygen tension or a decrease in hemoglobin content or saturation. Marked increases in arterial carbon dioxide tensions or increases in hydrogen ion concentration also stimulate the peripheral chemoreceptors.
Muscles of ventilation
The “muscles of ventilation” often are considered as three distinct groups: the diaphragm, intercostal muscles, and accessory muscles.
The diaphragm is composed of both a membranous and muscular portion. The muscle fibers originate and insert on the inferior surface of the lower ribs. The two muscular hemidiaphragms are connected by a membranous midline portion. Motor innervation originates from the spinal cord at the third through fifth cervical roots, which combine to form the phrenic nerve. The diaphragm is the major muscle of ventilation and it performs all the work associated with normal breathing; the other inspiratory muscles provide great reserve.
The intercostal muscles form two layers of fibers connecting the ribs and are innervated by spinal nerves from T-1 through T-11. The external intercostal muscles elevate the anterior chest wall and facilitate inspiration, whereas the internal intercostals act in an opposite direction and are believed to play a role in forceful expiration.
The accessory muscles of ventilation serve to elevate and stabilize the chest wall in its greatest diameters and thereby improve the efficiency of diaphragmatic excursion. The major accessory muscles are the scalene, sternocleidomastoid, trapezius, and pectoralis, the innervations of which are derived from the upper cervical roots. Accessory muscles normally are inactive in the absence of exercise and stress.
Inspiration is always an active muscular event, whereas expiration normally is passive; elastic recoil of the lung and chest wall provides the expiratory energy. Abdominal muscles (external oblique, rectus abdominis, internal oblique, and transverse abdominis) play a major part in active expiration. Innervation of the abdominal muscles is through spinal nerves T6 through L1.
The lung–thorax relationship
Elasticity describes forces working to return matter to its “resting” status. Lung elasticity results from interstitial elastic fibers and alveolar surface tension, producing forces that act to collapse the alveoli. LaPlace's law of elastic spheres dictates that elastic forces increase as the radius decreases, inferring that smaller alveoli should collapse by emptying into larger alveoli. This phenomenon is prevented by a pulmonary surfactant complex that reduces surface tension forces. In general, every alveolus has a critical volume at which the distending forces are equal to the elastic forces; below this critical volume, the elastic forces are overwhelming and total collapse ensues; above this critical volume, the surfactant complex acts to balance the opposing distending and elastic forces. The term compliance is used to describe forces opposing distortion from the resting state. Because lung elastic forces must be overcome to achieve inspiration, low lung compliance is associated most often with increased elastic forces.
As depicted in Figure 2, the mechanics of ventilation depend in large part on the interrelationship of elastic forces in lung and chest wall structures. After inspiration, the elastic forces return the chest cage and lungs to a state in which all elastic forces are balanced. At the end of a normal expiration, the chest wall elastic forces are equal and opposite to the lung elastic forces, resulting in a stable intrathoracic volume—the FRC.
Pregnancy does not appear to affect surfactant production or function but does entail diminution of the chest wall elastic forces resulting in the normal lung elastic forces, exerting a relatively greater influence on FRC.4 The decrease in FRC that accompanies pregnancy can only be partly accounted for by diminution of chest wall elasticity. Another significant factor is increased pulmonary blood volume resulting from progesterone- and prostaglandin-induced vasodilation. Regardless of the various mechanisms, the decrease in FRC begins by the 15th week of gestation and cannot be attributed solely to the enlarging gravid uterus.1, 5
THE WORK OF BREATHING
Spontaneous breathing improves cardiac output by augmenting venous return to the heart and improves matching of ventilation to perfusion by enhancing distribution of ventilation to areas of greatest pulmonary blood flow. The work of breathing is the muscular energy required during spontaneous breathing to overcome airway resistance and lung/chest wall elastic forces.
Detrimental work of breathing
Increases in the work of breathing accomplished within the limits of cardiopulmonary reserves (as with moderate exercise or stress) are anticipated to be beneficial to the maintenance of respiratory homeostasis. However, when increased work of breathing places demands on the cardiopulmonary system that exceed their functional reserves, the result often is cardiopulmonary deterioration.6 The concept of detrimental work of breathing describes a clinical picture of progressively increasing work of breathing that eventually destabilizes cardiopulmonary homeostasis.7 Such detrimental work of breathing is manifest by progressive increases in respiratory rate, heart rate, and systolic blood pressure and onset of diaphoresis and mental confusion, delirium, or even obtundation. Patients able to communicate invariably report dyspnea.
Extreme degrees of detrimental work of breathing are clinically recognized as acute respiratory distress (progressive tachypnea, tachycardia, dyspnea, hypertension, intercostal retraction, use of accessory muscles of ventilation, diaphoresis, and mental status changes). The patient often is described as appearing “fatigued” or “tiring out.” Lesser degrees of detrimental work of breathing undoubtedly exist but are clinically difficult to recognize or quantitate.
Assessment of ventilatory reserves
Development of detrimental work of breathing should be anticipated in any clinical circumstance in which ventilatory demand is increased concomitantly with reduced ventilatory reserve. Because increases in airway resistance usually are not associated with pregnancy, it is appropriate to consider increased work of breathing in pregnancy mainly in relation to the elastic forces (i.e., compliance factors).
Normally, 5% of total oxygen consumption goes to the work of breathing. The relationship of minute volume (MV) to oxygen consumption for the work of breathing can be graphically depicted (Fig. 3A); a similar relationship can be derived relating oxygen consumed by the work of breathing to tidal ventilation represented as a percentage of VC (see Fig. 3B). The greater the portion of the VC used for tidal ventilation, the greater the work required to further increase tidal ventilation. Significant diminishment in VC signals a decrease in ventilatory reserves. This concept of ventilatory reserve is applicable in the obstetric patient because VC usually is not significantly altered during pregnancy.1, 5, 8
PULMONARY CHANGES DURING PREGNANCY
Antepartum pulmonary changes begin in the fourth week of gestation. The earliest changes of clinical significance are related to mucosal capillary engorgement, resulting in swelling and congestion of the nose and throat.9 Manipulations of the upper airway, such as intubation and suctioning, can more readily lead to upper airway obstruction than in the nongravid patient.
Chest wall changes are apparent by the fifth month of gestation.1 The anteroposterior diameter is increased up to 2 cm with “flaring” of the lower ribs. The substernal angle is increased from a normal 70 degrees to 105 degrees, and a predominantly thoracic (intercostal) breathing pattern is apparent at term.10, 11 The diaphragm becomes elevated earlier and to a greater extent than can be attributed solely to the enlarging gravid uterus. At term, the elevation approaches 4 cm while diaphragmatic movement essentially is unimpeded.1, 8 Approximately, a 45% loss of chest wall elasticity and a normal lung compliance result in a 30% decrease in total pulmonary compliance at term. Total pulmonary resistance decreases 50%, mostly as a result of progesterone-induced changes in bronchial smooth muscle.1 FEV1 remains relatively normal throughout pregnancy.12
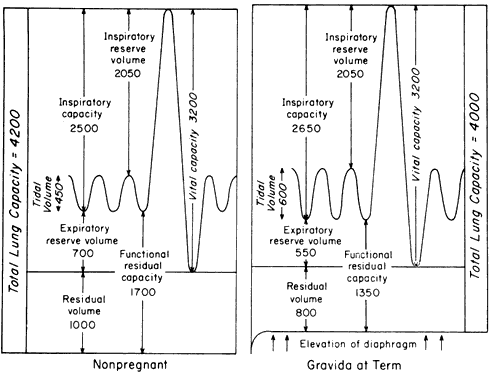
Figure 4 compares lung volumes and capacities between the nonpregnant woman and the term gravida. There is less than a 5% decrease in TLC, which does not meet criteria for the diagnosis of restrictive disease; however, FRC is diminished approximately 20%, which does meet criteria for restrictive pulmonary disease.8 The fact that the RV-to-TLC relationship remains essentially intact is the most probable explanation for the observation that the decreased FRC is associated with little, if any, morbidity.1, 5, 8, 9, 11 VC (ventilatory reserve) essentially is unchanged throughout pregnancy. Apparently, the increased anteroposterior diameter compensates for elevation of the diaphragm. A significant decrease in VC during pregnancy indicates diminished ventilatory reserve just as in the nongravid patient.
At term, the MV has increased 30–50%. This increase is caused by more than just fetal oxygen consumption and carbon dioxide production demands since the MV takes up to 6 weeks postpartum to return to normal limits.11, 12 One explanation is that increased progesterone levels are known to cause a decreased medullary chemoreceptor carbon dioxide response.2 The increasing MV is secondary to increases in VT to a much greater extent than increases in the respiratory rate.12 Because there is no increase in dead-space ventilation, the gradually increasing MV results in the arterial PCO2 gradually decreasing to approximately 32 mmHg.2 Maternal alkalosis does not occur because the renal compensatory mechanism increases serum bicarbonate.3
Arterial PO2 is diminished (70–90 mmHg) in the last trimester.2, 3 Mechanisms accounting for this relative hypoxemia include increased maternal metabolism, decreased FRC (low  /
/ ), and small airway closure. Because small airway closure has been suggested as the major mechanism for hypoxemia during labor, it deserves special consideration. Small airways in dependent portions of the lung are known to collapse during forced exhalation, a mechanism intended to protect the small, dependent alveoli from collapsing. This small airway closure phenomenon is studied by the closing volume (CV) pulmonary function test. CV measurements in the supine pregnant patient show that one third of patients develop airway closure during normal tidal ventilation5, 12; most show the phenomenon during forced exhalation.13 All these factors underscore the point that the parturient is very susceptible to having significant hypoxemia develop with even minor degrees of stress or hypoventilation. Because all these mechanisms produce low
), and small airway closure. Because small airway closure has been suggested as the major mechanism for hypoxemia during labor, it deserves special consideration. Small airways in dependent portions of the lung are known to collapse during forced exhalation, a mechanism intended to protect the small, dependent alveoli from collapsing. This small airway closure phenomenon is studied by the closing volume (CV) pulmonary function test. CV measurements in the supine pregnant patient show that one third of patients develop airway closure during normal tidal ventilation5, 12; most show the phenomenon during forced exhalation.13 All these factors underscore the point that the parturient is very susceptible to having significant hypoxemia develop with even minor degrees of stress or hypoventilation. Because all these mechanisms produce low  /
/ alveoli, as contrasted to zero
alveoli, as contrasted to zero  /
/ units, arterial hypoxemia should be responsive to oxygen administration in the range of 30–50%.
units, arterial hypoxemia should be responsive to oxygen administration in the range of 30–50%.
MV may be increased 300% when pain becomes severe during labor. The result is transient maternal hypocarbia (PCO2 less than 25) and alkalemia (pH greater than 7.55). The increased work of breathing and resulting oxygen deficit can lead to lactic acidemia. Effective analgesia alone can diminish this hyperventilation.14, 15
AIRWAY MANAGEMENT
Airway mismanagement accounts for one third of cases of fatal aspiration and 50% of anesthetic-related maternal mortality. The most recent studies in England, the United States, and other parts of the globe show that the incidence of failed intubation has remained at one in 250.16, 17, 18 Editorials, articles, and reports refer to the problem as intractable, unsolvable, or irreducible.19, 20, 21 There are several reasons for this:
- There is no accurate method to predict difficult intubation.
- The current technology or techniques are not suited for handling the situation of a failed rigid laryngoscopy.
- Failed intubation occurs rarely, so few practitioners can gain experience.
- General anesthesia is the method of last resort when regional anesthesia fails.
- Anesthesiologists ignore the data and belie their experiences.
- Maternal physiology is not conducive to surviving prolonged periods of apnea.
For the past 30 years, rapid sequence induction has reigned as the unchallenged standard of care to secure the airway because of the urgency of obstetric emergencies. There is a tendency to underestimate that which is quantifiably small, leaving awake intubation as an underutilized technique. Surveys show a wholesale undermining of safety. Even in the presence of a known difficult intubation, 60% of anesthesiologists prefer to secure the airway with the patient asleep.22
Prediction
The current state of predicting difficult intubation is similar to the situation in the 1970s when x-ray pelvimetry was considered to be the gold standard for predicting successful vaginal delivery.23 Every few years, articles are published stating that if one uses a specific set of airway measurements derived from physical examination, x-rays, computed tomography, or magnetic resonance imaging, one can predict difficult intubation within a reasonable degree of certainty.24, 25 Later, these studies are refuted and replaced by a different technique or method that purports greater accuracy and reproducibility.26, 27 None of these tests have been validated by large prospective studies, and thus the reports are the scatologic equivalent of worthless. Patients intubated fiberoptically because of abnormal external anatomy prove to be easy when intubated at a later date. Patients who appear normal often are found to be difficult. Only 6.6% of patients with Mallampati IV classification often were found to be difficult.28 In the Confidential Enquiry into Maternal Mortality (CEMD) series from 1967 to 1984, 83% (44/53) of the deaths due to failed intubation occurred in patients with normal-appearing airways.
Technology and technique
The current practice of general anesthesia requires intubation for management of the obstetric airway. This can be accomplished with a rigid laryngoscope in 99.6% (249/250) of the cases. Although no single company or person has a monopoly on the correct technology or equipment to solve the problem of failed intubation, a panoply of apparatus is promoted. Thus, the anesthesiologist is inundated by a host of pulse oximeters, end-tidal carbon dioxide detectors, Combitubes, cuffed oropharyngeal airways, laryngeal mask airways, light-wands, jet ventilators, and retrograde wires. These devices are popular because they require minimal training and skill. Conversely, the reviews by Pierce and Cheney29, 30 of the American Society of Anesthesiologists (ASA) Closed-Claim Database conclude that increased monitoring will not prevent all cases of airway disasters. Whereas cases of death due to unrecognized esophageal intubation have virtually disappeared, deaths due to airway mismanagement still occur. Despite the myriad of proposed solutions, the best epistemologists of our specialty are stymied by the problem of preventing maternal death after failed intubation. With the invention of the laryngeal mask airway (LMA), they recommend that our efforts should focus on ventilation instead of intubation.31 It is difficult using only positive pressure and a mask to ventilate the parturient with an abdomen distended by a 9-month gravid uterus. Whether the patient weighed 140 or 300 lb (65 or 140 kg) when the LMA was successful, spontaneous ventilation returned for the duration of the cesarean delivery.32, 33 When positive pressure ventilation is planned, the LMA is best suited as a helpful guide for sedated endotracheal intubation before the administration of muscle relaxants.34 There is little time to react to the problem of failed intubation because of the speed by which anesthesia induces apnea, with subsequent loss of the airway, and oxygen desaturation. To be of any value, it is imperative that the LMA and other airway aids are physically present in the operating room before induction.
It has long ago been recognized that more important than the technology are the approach and technique.35 Rather than hire a tailor with a different style to make new clothes from old cloth for the emperor, we are tempted to buy new fabric to effect a sartorial result.36 With minimal sedation, liberal topicalization, and maintenance of spontaneous ventilation, the airway can be secured before rendering the patient unconscious. This was first suggested by Walts in 196537 and is now a recommended option in the ASA Task Force for management of the difficult airway.38 Older solutions to airway management were forgotten and abandoned because the technology was not appropriate for the task. Species are most successful when their genetics promote a very active atavism, enabling certain traits to disappear when the environment becomes hostile and allowing their re-emergence in later generations when conditions become more favorable. The same rules apply to engineering, when newer technology ensures the success of older solutions.
The fiberoptic laryngoscope is ideally suited for the task of securing the airway with the patient awake, although a recent editorial did not even mention its existence.19 Fiberoptic skills are not connatal; they must be developed. Similar to following ASA pathways, guidelines, algorithms, or drills, training and practice are the keys to successful outcome. Keeping the fiberoptic laryngoscope locked in a cabinet for use only in emergency situations leads to failure and frustration. Nor should its use be limited to the sacerdotal services of attending physicians training novices in academic centers. Other than isolated case reports, there is no large series of using fiberoptics before induction of anesthesia to reduce maternal mortality.39, 40, 41, 42
Rare occurrences
Failed intubation accounts for 20% of the obstetric anesthesia malpractice cases in which severity of outcome determines both physician reviews and jury awards.43, 44 There are few articles from major institutions on this subject because errors of judgment concerning the airway are clothed in secrecy for fear of being misconstrued as cases of medical malfeasance.45 In a recently published American study, fiberoptics were used preinduction of general anesthesia in only 1.3% (7/538) of the parturients and the incidence of failed intubation remained at one in 269.17 Of the two cases of failed intubation, one resulted in a maternal death. When compared with the British data from Hawthorne's study, in which no fiberoptics were used, the ratio of the two standardized incidence ratios is 0.97.46
Why not take a passive approach and use the fiberoptic only after failed rigid laryngoscopy? This question is of eschatologic magnitude because one in every 87 (40/3333 = 1/87) cases of failed intubation results in a maternal death.21 Following the old adage of closing the barn door after the horse escapes leads to maternal death since 1.2% (40/3333) of failed intubations are in parturients who are impossible to mask ventilate. This is 13 times the incidence in the nonobstetric patient.21
When prospective studies rather than mortality statistics are reviewed, impossible mask ventilation occurs in 9% of failed intubations whether the patient is pregnant (2/23) or not (1/11). Analysis shows that the combination of failed intubation and impossible mask ventilation occurs in nonobstetric patients with risk factors that include body-mass index higher than 26 kg/m2 and presence of sleep apnea.47 Pharyngeal edema frequently occurs during pregnancy with gain in body weight.48 The linkage of difficult mask ventilation with failed intubation is roughly the same in the two populations: 36% (4/11) in the nonobstetric patients compared with 30% (7/23) in the obstetric patients.
In contrast to the CEMD series, 26% of the patients presenting for general anesthesia in an obstetric population from South Africa had difficult-appearing airways.28 They were described as having a short neck and swollen face or tongue and as being obese. Unfortunately, the endomorphic parturient who weighs more than 300 lb is no longer a rare event in the United States, in which 17% of the population is overweight and medical complications from obesity account for more than 300,000 deaths annually.49, 50 In the United States, Endler and coworkers51 in Michigan found that 80% of general anesthesia deaths were caused by obesity. In a different American population of parturients weighing more than 300 lb (Hood, North Carolina), difficult intubation was encountered in 35% (6/17) of the cases.52 Only one patient was intubated awake with rigid laryngoscope. After induction, two patients required multiple attempts at securing the airway.
The relative scale of the problem must be brought into perspective. Greater concern should be made of managing the maternal airway and less worry about neonatal respiratory depression. The 300-lb parturient at 28 weeks' gestation presenting for emergency cesarean delivery due to a prolapsed cord is better served by a sedated fiberoptic intubation than a failed rapid sequence induction. It is easier to intubate an 800-g neonate than to struggle with the airway of the mother. The neonate will be intubated and ventilated anyway because of fetal lung immaturity. An order-of-magnitude mentality can easily mislead. Although the neonatal airway is much smaller than the adult, intubation equipment is appropriately scaled to the task. Larger laryngoscope blades are used in adults. However, this does not guarantee success because of the challenge of aligning less plastic airway anatomy with the usual field of the intubator.
Gaining experience
There is increasing concern that as the number of cesarean deliveries to patients receiving general anesthesia decreases, there will be insufficient cases for training.53 Because of the acuteness of obstetric care, there must be a designated fiberoptic assigned to labor and delivery. Unlike the situation in other institutions, the fiberoptic cannot be shared with respiratory care or the general operating room.54 In emergency situations or when the abdomen is already open, it is appropriate for an attending physician to perform the fiberoptic intubation. When facing a nonurgent situation, such as a retained placenta or termination, it is appropriate for a tyro to try his or her skills with fiberoptics.
How can we ensure that attending physicians covering obstetric patients are qualified in the use of fiberoptics? Experience performing three awake fiberoptic intubations per month is considered the minimum to maintain proficiency.55 It is doubtful whether most of the attending staff in the United States who are responsible for covering labor and delivery suites currently possess these skills.
Age of the physician seems less of a handicap in using the fiberoptic laryngoscope than lack of hand–eye coordination. When using a straight or curved laryngoscope blade, presbyoptics are at a disadvantage because it is impossible to focus on an anterior larynx through their lower set of bifocal lenses. By enhancing the visual acuity of the optically challenged physician, the fiberoptic soon becomes an indispensable part of their armamentarium.
The video game generation quickly grasps the use of the fiberoptic because the controls on the handle are similar to the controls of a joystick. Simulators are being developed to take advantage of the generation's youthful proclivity in navigating through virtual environments from Mario World to the Virtual Larynx.56
Promoting regional anesthesia
The use of general anesthesia for cesarean deliveries has fallen steadily from 50% to 5% during the past 25 years. However, regional anesthesia may fail in situations in which it is needed the most, or the opposite may happen: too high a level is reached in a parturient with a difficult airway.
Denying the data
Some contest the veracity of Medic-Alert periapts emblazoned with “difficult intubation.” Others hold firm to their inveterate beliefs and inveigh a litany of complaints against sedated fiberoptic intubation. They protest that the hypermetric use of the fiberoptic exposes patients to the very dangers we are trying to avoid: (1) delays in delivery, (2) lower neonatal Apgar and umbilical cord gases, (3) maternal aspiration and asphyxiation, and (4) psychological trauma.
The data show the following:
- Time from transtracheal injection to intubation is less than 2 minutes.
- When stratified by gestational age and indication for cesarean delivery, there is no difference between rigid and fiberoptic groups.
- No cases of maternal aspiration have occurred in the fiberoptic groups. Parturients with a full stomach, sedated but breathing spontaneously, who can cough do not aspirate.57
- Because general anesthesia results in 17 times the mortality of regional anesthesia, it is no surprise that there is great fear of its use, even when appropriate.20 During the past quarter of a century (1975–2000), more patients have had to endure cesarean deliveries with poor anesthesia because of an inadequate regional block than the few patients who reported a brief choking sensation after fiberoptic intubation.
Surviving prolonged periods of apnea
The physiologic changes of pregnancy include increased oxygen consumption and cardiac output but a decrease in alveolar volume. During apnea, these factors shift the maternal hemoglobin de saturation curve to the left. The curve is shifted to the right by increased maternal blood volume. Overall, there is a faster rate of hemoglobin de saturation in the gravid patient compared with the nongravid patient58 (Fig. 5). The nonpareil benefit of preoxygenation only adds 4–6 minutes before hemoglobin desaturation to 65%. Benumof59 showed that return to 50% twitch height after succinylcholine can extend beyond 8 minutes. A rapid sequence induction without preoxygenation should be proscribed. The whole scenario of failed intubation and impossible ventilation can be avoided by securing the airway before induction. Maternal, and thereby fetal, oxygenation then can be maintained up to the time of delivery. In preparation for fiberoptic intubation, a nasal cannula should be placed on the mother before sedation and topicalization. Even in the presence of high maternal PaO2, the fetal pvO2 cannot exceed maternal mixed venous PO2.60
|
Newer studies
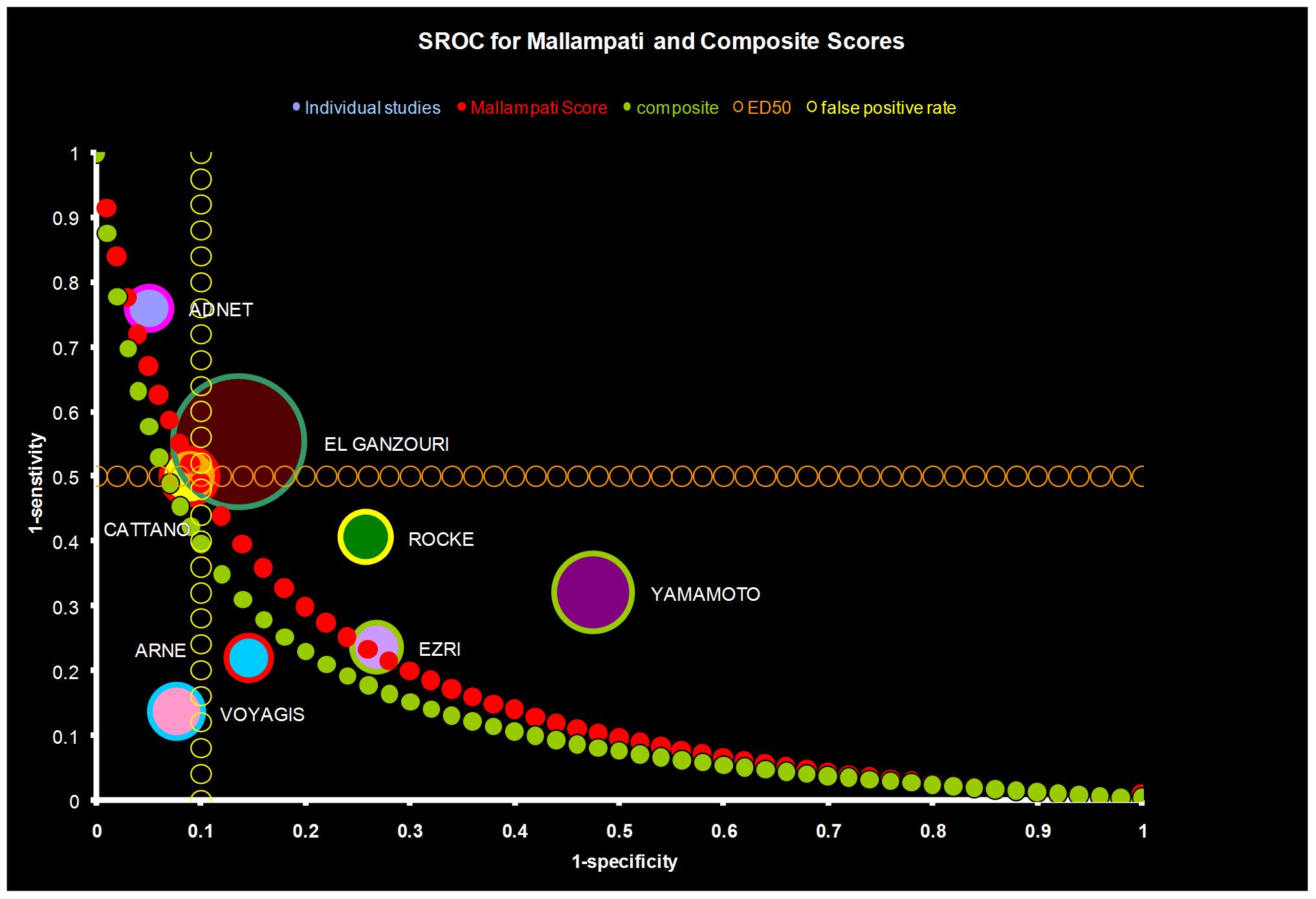
A review of the CEMD data shows a dramatic reduction in maternal deaths due to aspiration from 34% (1967–1984) to 9% (1985–2002) (Fig. 6). This is due primarily to the widely accepted practice of preoperatively administering nonparticulate antiacids, H2 receptor blockers, and proton pump inhibitors. Surprisingly, the availability of end-tidal carbon dioxide detectors has only reduced mortality due to esophageal intubation from 14% to 10%. The concept of prophylactically securing the airway with a laryngeal mask was proved effective in over 1000 parturients where only seven patients required endotracheal intubation.61 Recent meta-analysis of predictive airway scores reveals that only 1% of parturient airways would need to be secured prophylactically prior to induction of anesthesia to prevent 50% of the failed intubations (Fig. 7).62, 63 The recommendation that all intubations be performed electively with a fiberoptic prior to induction of anesthesia is perhaps the only way to prevent airway disasters.64 This may be practical in the future given the lower costs of the equipment, increasing user skill, and the ability to quickly sedate the parturient and topicalize the airway.
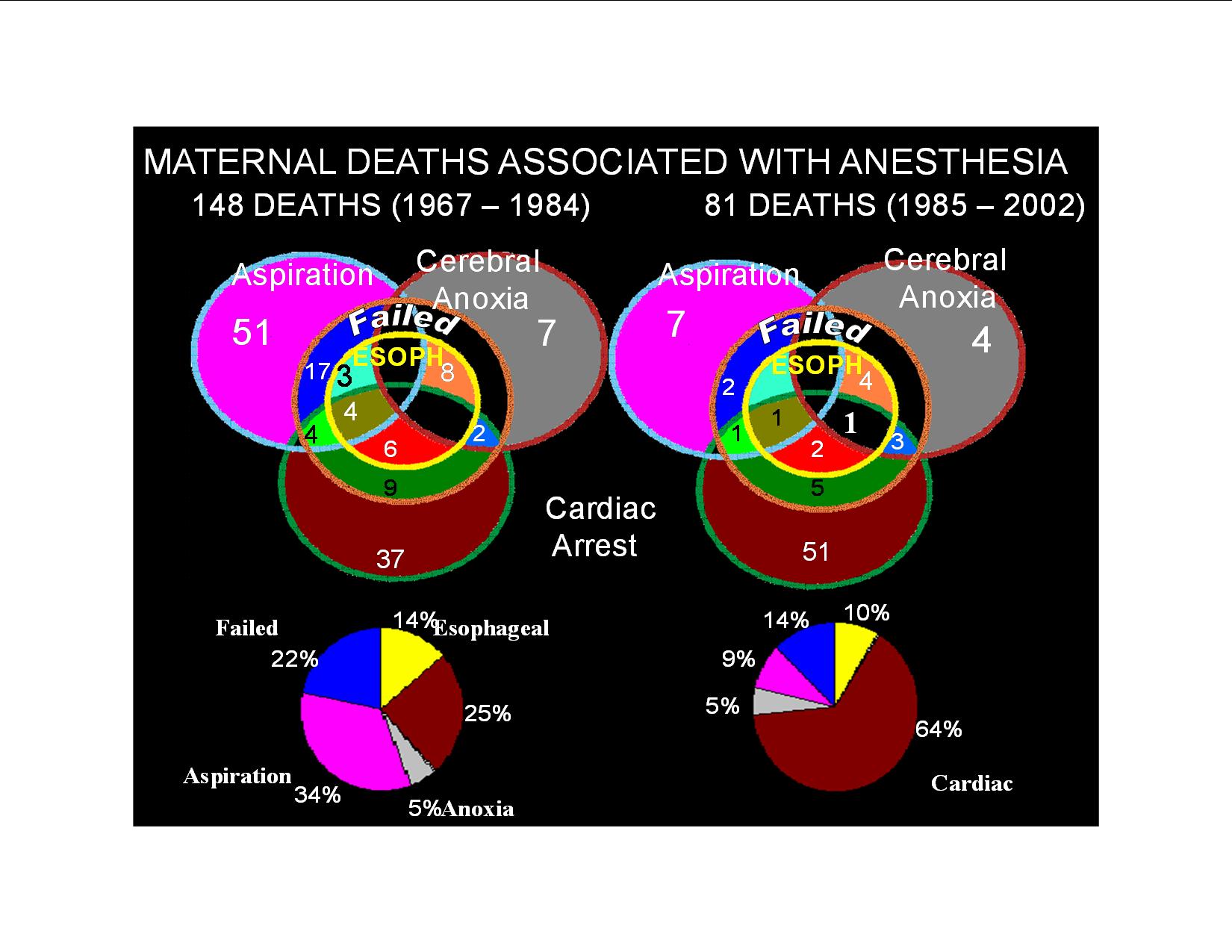 Fig. 6. Maternal deaths associated with anesthesia 1967–2002. CEMD
Fig. 6. Maternal deaths associated with anesthesia 1967–2002. CEMD
REFERENCES
Gee JBL, Packe BS, Miller JE et al: Pulmonary mechanics during pregnancy. J Clin Invest 46: 945, 1967 |
|
Prowse CM, Gaensler EA: Respiratory and acid-base changes during pregnancy. Anesthesiology 26: 381, 1965 |
|
Shapiro BA, Harrison RA, Cane RD et al: Clinical Application of Blood Gases. 4th ed. Chicago, Year Book Medical Publishers, 1989 |
|
Marx GF, Murthy PK, Orkin LR: Static compliance before and after vaginal delivery. Br J Anaesth 42: 1100, 1972 |
|
Russell IF, Chambers WA: Closing volume in normal pregnancy. Br J Anaesth 53: 1043, 1981 |
|
Roussos C, Macklem PT: Inspiratory muscle fatigue. In Fishman AP, Macklem PT, Mead J (eds): Handbook of Physiology. The Respiratory System, pp 511–527. Vol 3. Baltimore, Waverly Press, 1986 |
|
Shapiro BA: Editorial: When is an increase in the work of breathing clinically significant? Crit Care Med 18: 681, 1990 |
|
Baldwin GR, Moorthi DS, Whalton JA et al: New lung functions and pregnancy. Am J Obstet Gynecol 127: 235, 1977 |
|
Bonica JJ: Principles and Practice of Obstetric Analgesia. Philadelphia, FA Davis, 1967 |
|
Skaredoff MD, Ostheimer GW: Physiologic changes during pregnancy: Effects of major regional anesthesia. Reg Anaesth 6: 28, 1981 |
|
Knuttgen HG, Emerson K Jr: Physiological response to pregnancy at rest and during exercise. J Appl Physiol 36: 549, 1974 |
|
Cugell DW, Frank NR, Gaensler EA et al: Pulmonary function in pregnancy. I. Serial observations in normal women. Am Rev Tuberc 67: 568, 1953 |
|
Bevan DR, Holdcroft A, Loh L et al: Closing volume and pregnancy. Br Med J 1: 13, 1974 |
|
Marx GF, Greene WM: Maternal lactate, pyruvate, and excess lactate production during labor and delivery. Am J Obstet Gynecol 90: 786, 1964 |
|
Sangoul F, Fox GS, Houle GL: Effect of regional analgesia on maternal oxygen consumption during the first stage of labor. Am J Obstet Gynecol 121: 1080, 1975 |
|
Hawthorne L, Wilson R, Lyons G et al: Failed intubation revisited: 17-yr experience in a teaching maternity unit. Br J Anaesth 76: 680–684, 1996 |
|
Tsen L, Pitner R, Camann W: General anaesthesia for cesarean section in a tertiary care hospital 1990–1995: Indications and implications. Int J Obstet Anaesth 7: 147–152, 1998 |
|
Ramadhani S, Mohamed L, Rocke DA et al: Sternomental distance as the sole predictor of difficult laryngoscopy in obstetric anaesthesia. Br J Anaesth 77: 312–316, 1996 |
|
Chestnut D: Anesthesia and Maternal Mortality: Anesthesiology 86: 273–276, 1997 |
|
Hawkins J, Koonin L, Palmer S et al: Anesthesia-related Deaths during Obstetric Delivery in the United States, 1979–1990. Anesthesiology 86: 277– 284, 1997 |
|
Glassenberg R: General Anesthesia and Maternal Mortality: Semin Perinatol 15: 386–396, 1991 |
|
Rosenblatt WH, Wagner PJ, Ovassapian A et al: Practice patterns in managing the difficult airway by anesthesiologists in the United States. Anesth Analg 87: 153–157, 1998 |
|
Parsons M, Spellacy W: Prospective randomized study of x-ray pelvimetry in the primigravida. Obstet Gynecol 66: 76, 1985 |
|
Bellhouse CP, Dore C: Criteria for estimating likelihood of difficulty of endotracheal intubation with the Macintosh laryngoscope. Anesth Intens Care 16: 329–337, 1988 |
|
Chou HC, Wu TL: Mandibulohyoid distance in difficult laryngoscopy. Br J Anaesth 71: 335–339, 1993 |
|
Samra SK, Schork MA, Guinto FC: A study of radiologic imaging techniques and airway grading to predict a difficult endotracheal intubation. J Clin Anesth 7: 373–379, 1995 |
|
Naguib M, Malabarey T, AlSatli RA et al: Predictive models for difficult laryngoscopy and intubation. A clinical, radiologic and three-dimensional computer imaging study. Can J Anesth 46: 748–759, 1999 |
|
Rocke DA, Murray WB, Rout CC et al: Relative risk analysis of factors associated with difficult intubation in obstetric anesthesia. Anesthesiology 7: 67–73, 1992 |
|
Pierce EC: The 34th Rovenstine Lecture: 40 years behind the mask: Safety revisited. Anesthesiology 84: 965–975, 1996 |
|
Cheney FW: The American Society of Anesthesiologists Closed Claims Project: What have we learned, how has it affected practice, and how will it affect practice in the future? Anesthesiology 91: 552–556, 1999 |
|
Benumof JL: Laryngeal mask airway and the ASA difficult airway algorithm. Anesthesiology 84: 686–699, 1996 |
|
McClune S, Regan M, Moore J: Laryngeal mask airway for caesarean section. Anaesthesia 45: 227–228, 1990 |
|
McFarlane C: Failed intubation in an obese obstetric patient and the laryngeal mask (letter). Int J Obstet Anesth 2: 183–185, 1993 |
|
Shung J, Avidan MS, Ing R et al: Awake intubation of the difficult airway with the intubating laryngeal mask airway. Anesthesia 53: 645–649, 1998 |
|
Benumof JL: Management of the difficult adult airway with special emphasis on awake tracheal intubation. Anesthesiology 75: 1087–1110, 1991 |
|
Orkin FK: Practice Standards: The Midas touch or the emperor's new clothes? Anesthesiology 70: 567–571, 1989 |
|
Walts LF: Anesthesia of the larynx in the patient with a full stomach. JAMA 192: 705, 1965 |
|
Practice guidelines for management of the difficult airway. A report by the ASA task force on management of the difficult airway. Anesthesiology 78: 597–602, 1993 |
|
Fayek SS, Isaac PA, Shah J: Awake fiberoptic intubation in a 38-week pregnant patient with submandibular abscess. Int J Obstet Anesth 3: 103–105, 1994 |
|
D'Alessio JG, Ramanathan J: Fiberoptic intubation using intraoral glossopharyngeal nerve block in a patient with severe preeclampsia and HELLP syndrome. Int J Obstet Anesth 4: 168– 171, 1995 |
|
Broomhead CJ, Davies W, Higgins D: Awake oral fiberoptic intubation for caesarean section. Int J Obstet Anesth 4: 172–174, 1995 |
|
Boyne IC, O'Connor R, Marsh D: Awake fiberoptic intubation, airway compression and lung collapse in a parturient: Anaesthetic and intensive care management. Int J Obstet Anesth 8: 138–141, 1999 |
|
Chadwick HS: An analysis of obstetric anesthesia cases from the ASA closed claims project database. Int J Obstet Anesth 5: 258–263, 1996 |
|
Caplan RA, Posner KL, Cheney FW: Effect of outcome on physician judgments of appropriateness of care. JAMA 265: 1957–1960, 1991 |
|
Gostin L: A public health approach to reducing error: Medical malpractice as a barrier. JAMA 283: 1742–1743, 2000 |
|
Gardner MJ, Altman DJ: Statistics with Confidence. London, British Medical Journal, 1989 |
|
Langeron O, Massom E, Huraux C et al: Prediction of difficult mask ventilation. Anesthesiology 92: 1229–1236, 2000 |
|
Pilkington S, Carli F, Dakin MJ et al: Increase in Mallampati score during pregnancy. Br J Anaesth 74: 638–642, 1995 |
|
Mokdad AH, Serdula MK, Dietz WH et al: The spread of the obesity epidemic in the United States, 1991–1998. JAMA 282: 1519–1522, 1999 |
|
Allison DB, Fontaine JE, Manson JE et al: Annual deaths attributable to obesity in the United States. JAMA 282: 1530–1538, 1999 |
|
Endler GC, Mariona FG, Sokol RJ et al: Anesthesia related maternal mortality in Michigan, 1972 to 1984. Am J Obstet Gynecol 159: 187–193, 1988 |
|
Hood DD, Dewan DM: Anesthetic and obstetric outcome in morbidly obese parturients. Anesthesiology 79: 1210–1218, 1993 |
|
Johnson RV, Lyons GR, Wilson RC et al: Training in obstetric general anaesthesia: A vanishing art? Anaesthesia 55: 163–183, 2000 |
|
Anderson KJ, Quinian MJ, Popat M et al: Failed intubation in a parturient with spina bifida. Int J Obstet Anesth 9: 64–68, 2000 |
|
Popat M, Srivastava, Russell R: Awake fiberoptic intubation skills in obstetric patients: A survey of anaesthetics in Oxford region. Int J Obstet Anesth 9: 78–82, 2000 |
|
Lema MJ (ed): The Virtual Larynx. ASA Newsletter 63: 16, 1999 |
|
Ovassapian A, Krejcie TC, Yelich SJ et al: Awake fiberoptic intubation in the patient at high risk of aspiration. Br J Anaesth 62: 13–16, 1989 |
|
Farmery AD, Roe PG: A model to describe the rate oxyhaemoglobin desaturation during apnoea. Br J Anaesth 76: 284–291, 1996 |
|
Benumof JL, Dagg R, Benumof R: Critical hemoglobin desaturation will occur before return to an unparalyzed state following 1 mg/kg intravenous succinylcholine. Anesthesiology 87: 979–982, 1997 |
|
Bartels H, Moll W: Passage of inert substances and oxygen in the human placenta. Pflugers Arch 280: 165–177, 1964 |
|
Han TH, Brimacombe J, Lee EJ et al: The laryngeal mask airway is effective (and probably safe) in selected healthyparturients for elective Cesarean section: a prospective study of 1067 cases. Can J Anaesth 48: 1117, 2001 |
|
Shiga T, Wajima Z, Inoue T et al: Predicting difficult intubation in apparently normal patients: a meta-analysis ofbedside screening test performance. Anesthesiology 103: 429–37, 2005 |
|
Lee A, Fan LT, Gin T et al: A systematic review (meta-analysis) of the accuracy of the Mallampati tests topredict the difficult airway. Anesth Analg 102: 1867–78, 2006 |
|
Cormack RS, Lehane JR: Intubation training in the real world: a defence of the Northwick Park drill. Anaesthesia 62: 975–8, 2007 |