Rectovaginal Fistulas
Authors
INTRODUCTION
This chapter is dedicated to our friend and teacher, Dr Thomas Elkins.
A rectovaginal fistula is an epithelialized communication between the rectum and the vagina (when distal to the dentate line, it is an anovaginal fistula). Rectovaginal fistulas may cause distressing symptoms, and their severity depends on the size and site of the fistulous tract. They are within the scope of both the gynecologist and the colorectal surgeon.
INCIDENCE
In parts of the world with limited obstetrical resources, rectovaginal fistulas are common. The true incidence of rectovaginal fistulas is not readily available because the majority of series are small and much of the literature reflects a particular author’s clinical experience.
ETIOLOGY
Rectovaginal fistulas may be acquired or congenital. Acquired causes may be infective, inflammatory, neoplastic, or radiation-induced, or they may be posttraumatic; secondary to penetrating trauma, blunt perineal trauma, forceful coitus; and the result of postoperative and postobstetric injuries. Congenital anorectal abnormalities occur in approximately 1 in 5000 births.1 In female infants with high anorectal agenesis, the rectum may open into the vagina via a fistula to the posterior fornix. This chapter focuses on the diagnosis and treatment of acquired defects.
Obstetric
Lacerations of the birth canal and perineum are common with vaginal birth. Fetal weight greater than 3500 g, second stage longer than 120 minutes and instrumental deliveries, are associated with perineal lacerations but not other types of lacerations. Routine episiotomy had been ascribed to protect the pelvic floor, reduce perineal trauma, and be easier to repair than the ragged lacerations incurred without episiotomy. Indeed, two decades ago 80% of nulliparous women and 20% of multiparous women had routine episiotomy.2 However, several studies strongly linked an excess of third- and fourth-degree lacerations to routine episiotomy.3, 4, 5 Coats and colleagues found that anal sphincter and rectal injury was significantly more common after midline than after mediolateral episiotomy (11.6 vs. 2%).6 A recent article noted that operative vaginal delivery, particularly in combination with midline episiotomy, was associated with a significant increase in the risk of anal sphincter trauma in both primigravid and multigravid women. This synergistic relationship of operative vaginal delivery and episiotomy is evidenced by a more than threefold excess of risk of anal sphincter injury than when a patient has both procedures rather than an operative vaginal delivery alone.7 Controversy has been generated by another article which found that episiotomy had no impact on perineal lacerations and had a protective effect for other lacerations. It did, however, support that instrumental deliveries are by far the most significant risk factor for third- and fourth-degree perineal lacerations.8 Although the majority of these injuries are successfully repaired at the time of delivery, factors that may lead to a fistula include failure to recognize and repair a laceration of the rectovaginal septum or subsequent infection, abscess, or hematoma formation. Homsi and coworkers, in a review of the literature, found that episiotomy with subsequent third- and fourth-degree lacerations increased the risk for rectovaginal fistula by 4.6-fold.9 In another series, it has been reported that the incidence of rectovaginal fistula in patients with fourth-degree tears is as high as 1.5%.10 Importantly, women who have experienced a prior anal sphincter laceration are at three times increased risk for subsequent sphincter laceration when compared to women who have not experienced a sphincter laceration.11
Obstetric fistulas are of two varieties. Usually, midzone fistulas are secondary to pressure necrosis of the rectovaginal septum resulting from prolonged or obstructed labor. Repair may be attempted through the perineum or vagina, but surgical access may be poor and the repair is often unsatisfactory. Transanal repair and advancement flaps have become popular. Low fistulas are more commonly seen today because prolonged labor is rarely permitted in modern obstetrics. The fistulas are usually caused by a combination of infection and inadequate repair of a third- or fourth-degree tear. There may also be a history of a forceps delivery, and the fistula may not be recognized early in the postpartum course because of difficulty with postdelivery examination or later because of problems with constipation. There is also usually a significant amount of edema or a hematoma, making this assessment difficult postpartum. What about postpartum endoanal ultrasound? Even when anal sphincter tears are recognized and repaired immediately after delivery, almost half of the women remain symptomatic.12
Iatrogenic
Fistulas may result from direct or unrecognized rectal injury, cautery injury, or anastomotic leak or infection at the time of a surgical procedure. Procedures at higher risk for this complication include low anterior resection of the rectum, fulguration of an anterior rectal tumor, and operations to correct defects of posterior vaginal wall support.
Obviously, the location of a postoperative fistula depends on the type of operation performed. Midzone or low fistulas may occur after vaginal hysterectomy, posterior colporrhaphy, perineal body reconstruction, or local excision of rectal tumors. Often, these fistulas can close spontaneously without repair if they are small, and larger fistulas can close without repair but may require more aggressive management with wound drainage and the creation of a proximal colostomy. High iatrogenic defects can be repaired by coloanal sleeve resection. Advancement flap techniques may be satisfactory for low fistulas, provided access is adequate and there is no sphincter injury.
Inflammatory bowel disease
Crohn's disease is characterized by transmural bowel inflammation and a high tendency to form fistulas with adjacent structures. Anorectal abscesses, anorectal fistulas and rectovaginal fistulas may complicate Crohn's disease in 1.7–23% of cases.13, 14, 15 The more distal the location of the intestinal disease, the higher the risk of associated anorectal fistulas. Less than one quarter of patients with Crohn’s disease involving the small bowel have anal disease, whereas more than half of patients with colorectal involvement have anorectal fistulas.16 The fistulas that occur may be multiple or single. Not uncommonly, a single vaginal ostium may lead to several tracts opening in the anus.
The management of fistulas associated with Crohn’s disease is controversial and remains a challenge for clinicians. Medical treatments provide only low rates of long term symptomatic control and have an unacceptably high rate of recurrence even when initially successful.17 For enterovaginal fistulas complicating Crohn’s, the ileal or colonic segments associated with a high fistula need to be resected. Fistulas with rectal disease may benefit from a temporary defunctioning loop ileostomy or a proctocolectomy. Closure of the rectovaginal fistula may be warranted in selected patients. Prior to surgery the patient needs to optimized with medical therapy and ideally to have no active disease, if the patient is agreeable to a permanent solution. Reconstruction and repair are probably justified only for low rectovaginal or anovaginal fistulas without gross evidence of rectal involvement or in those patients without active disease. Radcliffe and associates reported that 67% of their 90 patients with rectovaginal fistula thought the symptoms of the intestinal disease overshadowed those of the fistula.13 Closure of the rectovaginal fistula may be warranted in selected patients, who have symptoms attributable to the fistula. Prior to surgery the patient should be optimized from a medical standpoint and ideally have no active disease. A rectal advancement flap technique is advocated for low repairs.13, 17
Rectovaginal fistulas are more commonly associated with Crohn’s disease than ulcerative colitis. Approximately 3% of patients with ulcerative colitis develop rectovaginal fistulas.18 Ileoanal pouch construction has been reported to be successful in managing rectovaginal fistulas complicated by ucerative colitis.18
Infection
A rectovaginal fistula can result from an anterior cryptoglandular abscess that extends into the vagina. Most are caused by nonspecific infection from Escherichia coli and Bacteroides species in an anal gland.19 Lymphogranuloma venereum, tuberculosis, and Bartholin’s abscess may also fistulize into the vagina. Acute acquired rectovaginal fistula in infants was first described in Zimbabwe in 1990. It is suggested that this is an early manifestation of human immunodeficiency virus (HIV) infection in young girls.20 Patients with an acquired spontaneous rectovaginal fistula should be screened for HIV. In HIV positive individuals, conservative treatment seems to be the best option because the success of surgical intervention is disappointing.21 If surgery is considered, it should be delayed until the viral load is minimal. The role of human papillomavirus in poor healing and dehiscence of episiotomies and subsequent development of rectovaginal fistula remains controversial.22 Pelvic abscesses in the pouch of Douglas from such processes as diverticulitis, ileal Crohn’s disease, appendicitis, or tubo-ovarian abscess may drain into the upper vagina and may mimic rectovaginal fistulas. Treatment of infectious causes often involves fistulotomy with drainage. Fistulas that are suprasphincteric typically require advancement flaps and low transphincteric fistulas can sometimes be laid open. Most anal gland infections are best managed by exploration of the fistula in the perineum.23
Radiation
It has been reported that up to 6% of women receiving pelvic irradiation will develop rectovaginal fistulas.24 Rectovaginal fistulas are most commonly associated with radiation therapy for endometrial, cervical, and vaginal cancers and are dose-dependent.25 Tissue hypoxia and progressive endarteritis obliterans are the purported etiology of radiation injury. Most postradiation fistulas occur within 2 years of therapy, but late injury has also been described.24 These late fistulas may be associated with a rectal stricture.24 It is important to distinguish recurrent carcinoma from radiation injury as the cause of the rectovaginal fistula. This is accomplished by biopsying the margins of the fistula at the time of an examination under anesthesia. Fistulas caused by radiation injury usually open into the mid or high vagina; the point of connection with the lower intestinal tract must be determined prior to proceeding with surgical correction. Fistulas that present during radiation therapy itself are probably the result of destruction of a carcinoma that had deeply invaded into the rectovaginal septum. Diversion of the fecal stream will give symptomatic relief for many patients, however it is rarely associated with closure of a radiation fistula.26 Most authors recommend raising a stoma before any form of surgical treatment is contemplated because local repairs do not heal unless defunctioned.27 Whatever the surgical approach, most experts recommend interposition of vascular nonirradiated tissue to improve chances for healing. Other authors have also studied the utility of hyperbaric oxygen therapy either in spontaneously healing these lesions or as an adjunct to surgery.28
CLINICAL PRESENTATION
Rectovaginal fistulas may be asymptomatic. Complaints range from the occasional passage of vaginal flatus to the discharge of feces through the vagina. Small fistulas can present as chronic vaginitis with a malodorous discharge. Associated abnormalities may be present such as anal sphincter disruption following obstetric trauma or proctitis in Crohn’s disease or in radiation injury, and these conditions may obscure the symptoms of the fistula. Loss of perineal support from obstetric lacerations may predispose the patient to anterior vaginal compartment prolapse. These patients may present for evaluation of a vaginal bulge or urinary incontinence.
ANATOMY
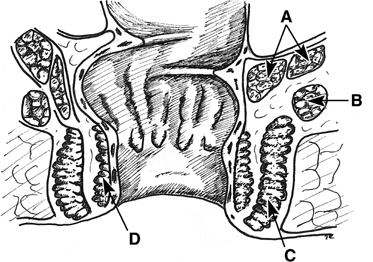
The rectum may be divided into thirds. The upper third is protected by peritoneum anteriorly. A fistula may arise here, or there may be an associated fistula from the contents of the pouch of Douglas, including small bowel and sigmoid colon. The lower two thirds of the rectum is extraperitoneal. The middle third is intimately related to the upper vagina, in particular the posterior fornix. This area is often involved by uterine cancer or radiation injury. The vagina is separated from the middle third of the rectum by the muscular wall of the rectum and the endopelvic fascia. The anal sphincters and the deep transverse perineal muscles distance the lower third of the rectum from the lower half of the vagina. The lower third of the rectum accounts for the majority of obstetric injury. At the level of the anorectal ring, the internal sphincter, the external sphincter, the transverse perineal muscles, the bulbocavernosus, and the perineal body distance the vagina from the anal canal. In the intersphincteric plane, one can find the anal glands, which open into the anal canal at the base of the anal crypts (Fig. 1).
THE CONTINENCE MECHANISM
The internal anal sphincter is responsible for the majority of the resting tone of the anal canal. This smooth muscle group has the major responsibility for continence of liquid stool and flatus. The external anal sphincter and the puborectalis, both striated muscle groups, are mainly responsible for continence of solid stool. The external sphincter responds to the sudden filling of the rectum secondary to the peristalsis of the bowel. The puborectalis is a muscular sling that forms the posterior rectal angle.
CLASSIFICATION
Based on anatomic considerations, fistulas to the vagina have been classified into six types. (1) Enterovaginal fistulas involve the ileum or sigmoid or a surgically created anastomosis as they lie in the pouch of Douglas and communicate to the posterior fornix. The usual etiology includes Crohn’s disease, diverticular disease, cancer, or surgery. (2) High rectovaginal fistulas communicate from the posterior fornix to the middle third of the rectum. They usually occur secondary to radiotherapy for cervical or endometrial cancer or after operations on the rectum or the uterus. (3) Midzone fistulas involve the lower third of the rectum and the midportion of the vagina and may be obstetric or related to rectal lesions or inflammatory bowel disease. (4) Low rectovaginal fistulas lie at the anorectal ring and may result from obstetric injury, foreign bodies. or local trauma; they may follow operations for pelvic organ prolapse. There are also (5) suprasphincteric and (6) transsphincteric anovaginal fistulas associated with anal gland infections, perirectal abscess, Bartholin’s abscess, Crohn’s disease, or previous anal surgery (Fig. 2).
ASSESSMENT
The anatomic site of the fistula needs to be carefully defined. The ostia in the anorectum and in the vagina must be described. The direction, length, width, and associated blind tracks must be appreciated. The size of the fistula affects the choice for repair. Examination under anesthesia and biopsies may be necessary to rule out a primary or recurrent malignancy or whether the injury is secondary to radionecrosis. Proctoscopy, sigmoidoscopy, and speculum examination with careful palpation of both openings with a lacrimal duct probe may be useful. If Crohn’s is a possible etiology, colonoscopy should be considered.
Radiologic studies including vaginogram, fistulogram, barium enema, or small bowel series may be necessary to demonstrate some fistulas, e.g. postepisiotomy repair rectovaginal fistulas and those related to systemic illnesses may present as tiny pinholes in the vaginal epithelium and can be difficult to visualize. Another means of identifying the tract was described by Carey.29 Air is instilled into the rectum, the vaginal vault is filled with a soapy solution, and one observes for air bubbles from the rectum identifying the fistula tract. Alternately, an enema of saline solution colored with methylene blue may be administered with a tampon in the vagina, and if after 15 minutes, the retained enema does not stain the tampon, one should reconsider the diagnosis of rectovaginal fistula. Some caution is needed if the tampon fails to stain: Shieh and Gennaro reported that the methylene blue test was diagnostic in only 9% in their series.30
When considering a surgical repair, the anal continence mechanism must be assessed to evaluate for associated sphincter deficiency or scarring of the perineal body. Endoanal ultrasonography is validated to accurately identify both external and internal anal sphincter defects.31, 32 Anal manometry can be helpful in quantitating an abnormality of the sphincter and in defining sensation of the rectum.33, 34
Bowel preparation should be done for all patients with rectovaginal fistula. By lowering the fecal load and decreasing bacterial seeding, the risk of postoperative infection and dehiscence of repair is reduced. Perioperative antibiotics should be given in a fashion similar to that commonly used for colon resection.
SURGICAL TECHNIQUE
Rectovaginal fistulas can be repaired through a multitude of approaches including transanal, vaginal, perineal, abdominal, and transsacral. A transabdominal approach is usually reserved for high rectovaginal fistulas. The most important aspect of rectovaginal fistula repair is closing the high-pressure or rectal side of the defect. Fine, delayed absorbable suture should be used in the closure of the rectal defect because of prolonged tensile strength and the greater integrity of knots.
Low/midzone fistulas
Simple fistulotomy may be used in superficial anoperineal fistulas. Here, the tract extends from an opening in the anal canal to the perineum, and usually, the sphincter is not involved. The fistulous tract is laid open with a simple incision. There are reports of women with Crohn’s disease and anovaginal fistulas successfully treated by fistulotomy.23 More commonly, a transsphincteric approach with layered closure is used for low fistulas because of proximity to the anal sphincter.35 An episioproctotomy is made and allows for excision of the entire fistulous tract followed by a repair similar to that of a fresh fourth-degree laceration at delivery. This technique requires meticulous reapproximation of both the internal and the external anal sphincters. There is a risk of fistula recurrence as well as anal incontinence if the healing is compromised. This approach allows for reconstruction of the perineal body as well. Episioproctotomy has been favorably described for patients with quiescent Crohn’s disease without prior fecal diversion.36
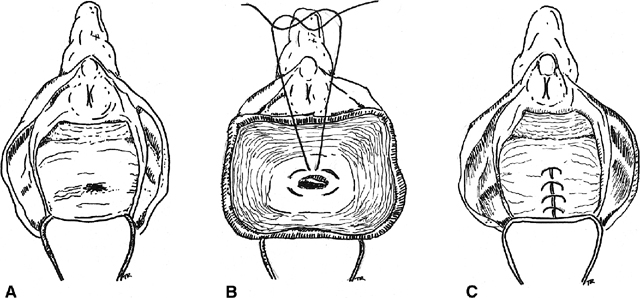
Inversion of the fistula into the rectal lumen via a transvaginal approach is also suitable for small low rectovaginal fistulas37 (Fig. 3). The vaginal mucosa is incised around the fistula ostium, and the fistula is closed with purse-string sutures tied in such a fashion as to invert the soft tissue into the anorectum. The vaginal mucosa is then reapproximated. For high rectovaginal fistulas, the anterior and posterior vaginal walls, once mobilized, can be utilized in the inversion of the fistula into the rectum (a modification of the Latzko procedure for vesicovaginal fistulas).37
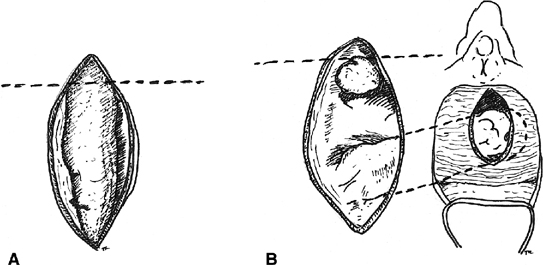
Boronow reported excellent results in patients with radiation-induced rectovaginal fistulas, utilizing a layered closure with interposition of a modified Martius graft of bulbocavernosis–labial fat pad tissue38 (Fig. 4). A vertical incision is utilized on the labia majora. The fatty tissue is mobilized sharply with careful attention to preserving the blood supply of the graft either inferiorly or superiorly. The thumb-sized graft is tunneled subcutaneously beneath the vaginal mucosa and the labia minora to overlay the repaired fistula site. The donor site may require a drain. Boronow emphasized a delay of surgery for 6 months after radiation exposure, a biopsy of the margins of the tract, a diverting colostomy in advance of the repair, interposition of vascularized tissue, and a tension-free closure. Elkins and colleagues have also utilized the Martius technique to repair a number of rectovaginal fistulas with satisfactory results.39
|
Hoexter and coworkers, proposed a transanal repair.40 They excise the fistula tract, approximate the rectal musculature, and advance the rectal mucosa caudad to protect the repair. The vagina mucosa is left open to heal by secondary intention. The authors found no recurrence in 35 patients with benign rectovaginal fistulas followed for 1–6 years. Repair from the rectal side has the advantage of correction of the defect from the high-pressure side, perhaps decreasing the rate of failure of repair.
Goligher has described a transperineal approach, through which the anus and rectum are separated from the vagina and the fistula tract is divided.41 The mobilization of the vaginal and rectal walls permits a tension-free closure. In addition, he recommends slight rotation of the rectum and vagina in opposite directions to protect the suture lines. This approach to repair allows the surgeon to preserve an intact internal and external anal sphincter. Wiskind and Thompson devised another transperineal approach akin to Goligher’s.42 Using a transverse incision across the perineal body above the sphincter complex, dissection is carried out between the anterior rectal wall and the posterior vaginal wall. The external anal sphincter is preserved, vaginal and rectal defects are closed separately, the fistula tract is transected, and attention is directed toward reapproximation of the endopelvic fascia in the midline and interposing the approximated puborectalis muscle between the vaginal and the rectal defects. These defects can be closed transversely or longitudinally. By closing the defects longitudinally, vaginal and anal length is maintained. In the series by Wiskind and Thompson, 21 patients with low fistulas, including seven with Crohn’s disease, had no recurrence during a follow-up ranging from 3 months to 8 years (mean, 18 months). Sher and associates also emphasize the importance of approximating the levator ani muscles in the midline between the rectal and the vaginal suture lines.43 Laparoscopic upper rectovaginal mobilization facilitating the transvaginal repair of recurrent rectovaginal fistulas has also been described by Pelosi and Pelosi.44
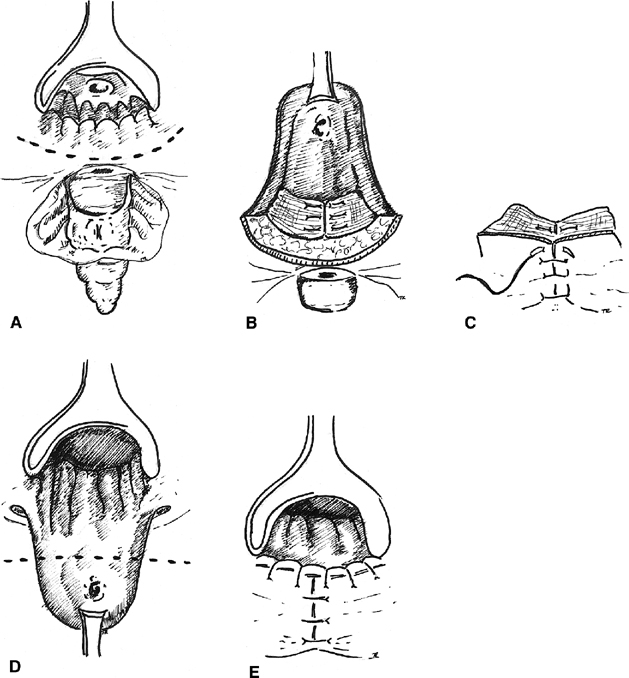
Many feel the best method of repairing low and midzone fistulas is the sliding or advancement flap. Based on the work by Noble,45 there have been many modifications. Stowe and Goldberg describe developing a broad-based flap of rectal mucosa, submucosa, and circular muscle and advancing it caudad46 (Fig. 5). Before the flap is sutured over the fistula site, the tract is excised from the distal end of the flap and the circular muscle is reapproximated with absorbable suture. The flap is then advanced over the repair and secured with interrupted absorbable sutures. The vaginal side is left open to heal by secondary intention and provide drainage of the surgical site. Several authors report a high rate of success with this method even when used for the treatment of rectovaginal fistula secondary to Crohn’s disease.43, 47 Some have described advancing the entire circumference (sleeve) of rectum for lesions involving more than one third of the circumference of the anus.47, 48 The advancement flap can be mucosa only or full thickness. Some feel the full thickness advancement has a higher rate of success without increasing the complications. 49
High fistula repair
Most often, a transabdominal approach is used for a colovaginal or high rectovaginal fistula because of coexisting pelvic disease and the inaccessibility of the fistula through the vagina. However, Lawson has successfully used a perineal approach to treat fistulas near the cervix.50 The vagina is divided up to the lateral fornix, and the pouch of Douglas may be opened behind the cervix to improve exposure. The rectum is drawn downward for a layered repair. Lawson reported success with this technique in 42 of 53 cases. The Latzko technique of using the anterior and posterior walls of the vagina in the closure of a vesicovaginal fistula can also be adapted for high rectovaginal fistulas.37
The etiology of the fistula must be appreciated when anticipating a transabdominal repair for a high fistula. If the fistula is secondary to trauma or pelvic drainage of an inflammatory process (e.g., diverticulitis), the defect can be successfully closed by opening the rectovaginal septum and closing the rectal and vaginal defects with interposition of an omental J-flap. Bowel resection or the interposition of nondiseased tissue is usually required in cases involving radiation injury or advanced malignancy.
Bricker and colleagues described a complex repair for radiation-induced rectovaginal fistula.51 The procedure involves an onlay patch of well-vascularized intestine to the damaged tissue and a descending colostomy. The advantages of Bricker’s technique are that it requires only anterior mobilization of the rectum, avoids denervation injury to the anorectum and hemorrhage, and that the patch or flap retains its native blood supply.
Timing of repair
The majority of fistulas secondary to trauma or obstetric causes may be repaired early. Repair should be deferred until the resolution of active infection, inflammation, induration, or local cellulitis. The authors agree with Hibbard, who feels that the decision of when to operate should depend on the condition of the tissue and not on an arbitrary period of time.52
Adjuvant materials
The goal in the treatment of rectovaginal and anal fistulas is to eliminate the fistula without a change in continence. No single technique exists that is appropriate for the treatment of all fistulas. The classic approach of excision of the fistula tract and layered closure has had a high rate of recurrence. Cutting sectons and fistulotomy have been used successfully to eliminate the fistula tract but cutting through the sphincter many times leads to some degree of anal sphincter dysfunction and varying degrees of incontinence. Other options that have been reported include cryotherapy,53 fibrin sealant,54 and rectal advancement flaps used individually or in combination. A biologic fistula plug fashioned from small intestine submucosa has been used to occlude the fistula tract and early reports have been promising.55, 56, 57, 58, 59 While the lithotomy position is often preferred by gynecologists, the prone jackknife position is preferred by colorectal surgeons. The prone position affords distinct advantage when using the anal plug. The prone position allows identification of the internal fistula tract opening, which is then irrigated with hydrogen peroxide and curetted to de-epithelialize the tract. An anal epithelial flap is raised around the primary opening. An anal plug is pulled through the fistula tract and secured to the submucosa with fine suture. The rectal flap is then closed. The excess plug material that is exposed in the vagina is excised and the plug is tacked to the periphery of the secondary opening so that the tract is left open. Multiple fistula tracts can be managed in the same way. The success of the plugs is at least equal to layered closure and has the advantage of being a less invasive and quicker procedure. The anal plugs are also effective in patients with Crohn's disease.60
Diverting colostomy
Diverting colostomy is typically used in the management of radiation-induced fistulas, very large rectovaginal fistulas, and some fistulas secondary to inflammatory bowel disease. A repair of the fistula can then be accomplished after all evidence of cellulitis and inflammation has resolved (usually 8–12 weeks). Colostomy takedown may be scheduled for 3–4 months after the repair.
POSTOPERATIVE CARE
Some surgeons prescribe a clear liquid diet for 2–3 days postoperatively, whereas others advocate clear liquids for an entire week. A low-residue diet and the use of stool softeners are recommended by some so that a patient has infrequent but loose stools. Others advocate high-fiber diets or the addition of bulk enhancers like psyllium (Metamucil). Sitz baths are often very comforting to the patient and also serve to keep the wound clean. Patients should not have intercourse for 6–8 weeks.
CONCLUSION
Rectovaginal fistulas can be a vexing problem for women. The successful management of this problem depends on the etiology, size, and location of the fistula, as well as assessing the competence of the continence mechanism.
REFERENCES
Stern HS, Dreznik Z: Rectovaginal fistula. Adv Surg 21:245, 1987 |
|
Kozok LJ: Surgical and non-surgical procedures associated with hospital delivery in the United States: 1980–1987. Birth 16:209, 1989 |
|
Thorp JM, Bowes W: Episiotomy: Can its routine use be defended? Am J Obstet Gynecol 160:1027, 1989 |
|
Wilcox LS, Strobine DM, Baraffi G, et al: Episiotomy and its role in the incidence of perineal lacerations in a maternity center and a tertiary hospital obstetric service. Am J Obstet Gynecol 160:1067, 1983 |
|
Shiono P, Klebanoff M, Christopher J: Midline episiotomies: More harm than good? Obstet Gynecol 75:765, 1990 |
|
Coats PM, Chan KK, Wilkins M, et al: A comparison between midline and mediolateral episiotomies. Br J Obstet Gynecol 87:408, 1980 |
|
Kudish B, Blackwell S, Mcneeley SG, et al: Operative vaginal delivery and midline episiotomy: a bad combination for the perineum. Am J Obstet Gynecol. 2006;195(3):749-54 |
|
Mikolajczyk RT, Zhang J, Troendle J, et al: Risk factors for birth canal lacerations in primiparous women. Am J Perinatol. 2008;25(5):259-64 |
|
Homsi R, Daikoku NH, Littlejohn J, et al: Episiotomy: Risks of dehiscence and rectovaginal fistula. Obstet Gynecol Surv 49:803, 1994 |
|
Legino LJ, Woods MP, Rayburn WF, et al: Third and fourth degree perineal tears. 50 years’ experience at a university hospital J Reprod Med 33:423, 1988 |
|
Lowder JL, Burrows LJ, Krohn MA, et al: Risk factors for primary and subsequent anal sphincter lacerations: a comparison of cohorts by parity and prior mode of delivery. Am J Obstet Gynecol 2007;196(4):344.e1-5 |
|
Sultan AH, Kamm MA, Hudson CN, et al: Third degree obstetric anal sphincter tears: Risk factors and outcome of primary repair. BMJ 308:887, 1994 |
|
Radcliffe AF, Ritchie DM, Hawley PR, et al: Anovaginal and rectovaginal fistulas in Crohn’s disease. Dis Colon Rectum 31:94, 1988 |
|
Van Patter WN, Bargen JA, Dockery MB, et al: Regional enteritis. Gastroenterology 26:347, 1954 |
|
Ritchie JK, Lennard-Jones JE: Crohn’s disease of the distal large bowel. Scand J Gastroenterol 11:433, 1976 |
|
Rankin GB, Watts HD, Clifford S, et al: National cooperative Crohn’s disease study: Extraintestinal manifestations and perianal complications. Gastroenterology 4:914, 1979 |
|
Andreani SM, Dang HH, Grondona P, et al: Rectovaginal fistula in Crohn's disease. Dis Colon Rectum. 2007;50(12):2215-22. Epub 2007 Sep 11 |
|
Froines EJ, Palmer DL: Surgical therapy for rectovaginal fistulas in ulcerative colitis. Dis Colon Rectum 34:925, 1991 |
|
Eykyn S, Grace RH: The relevance of microbiology in the management of anorectal sepsis. Ann R Coll Surg Engl 68:237, 1986 |
|
Borgstein ES, Broadhead RL: Acquired rectovaginal fistula. Arch Dis Child 70:165, 1994 |
|
Uba AF, Chirdan LB, Ardill W, et al: Acquired rectal fistula in human immunodeficiency virus-positive children: a causal or casual relationship? Pediatr Surg Int 2004;20(11-12):898-901 |
|
Snyder RR, Hammond TL, Hankins GD: Human papillomavirus associated with poor healing of episiotomy repairs. Obstet Gynecol 76:664, 1990 |
|
Francois Y, Descos L, Vignal J: Conservative treatment of low rectovaginal fistula in Crohn’s disease. Int J Colorectal Dis 5:12, 1990 |
|
Kimose HH, Fischer L, Spjeldnaes N, et al: Late radiation injury of the colon and rectum: Surgical management and outcome. Dis Colon Rectum 32:684, 1989 |
|
Sandeman TF: Radiation injury of the anorectal region. Aust N Z J Surg 50:169, 1980 |
|
Piekarski JH, Jereczek-Fossa BA, Nejc D, et al: Does fecal diversion offer any chance for spontaneous closure of the Int J Gynecol Cancer 2008;18(1):66-70. Epub 2007 Apr 26 |
|
Anseline PF, Lavery IC, Fasio VW, et al: Radiation injury to the rectum. Evaluation of surgical treatment Ann Surg 194:716, 1981 |
|
Williams JA, Clarke D, Dennis WA, et al: The treatment of pelvic soft tissue radiation necrosis with hyperbaric oxygen. Am J Obstet Gynecol 167:412, 1992 |
|
Carey JC: A new method of diagnosing rectovaginal fistula: A case report. J Reprod Med 33:789, 1988 |
|
Shieh CJ, Gennaro AR: Rectovaginal fistula: A review of 11 years experience. Int Surg 69:69, 1984 |
|
Sultan AH, Kamm MA, et al: Anal endosonography: Precision of identifying sphincter defects confirmed histologically. Br J Surg 81:466, 1994 |
|
Frudinger A, Ballon M, Taylor SA, et al: The natural history of clinically unrecognized anal sphincter tears over 10 yearsafter first vaginal delivery. Obstet Gynecol. 2008;111(5):1058-64 |
|
Titi MA, Jenkins JT, Urie A, et al: Correlation between anal manometry and endosonography in females with faecalincontinence. Colorectal Dis 2008;10(2):131-7 |
|
Bordeianou L, Lee KY, Rockwood T et al: Anal resting pressures at manometry correlate with the Fecal Incontinence Severity Index and with presence of sphincter defects on ultrasound. Dis Colon Rectum. 2008 Apr 25;2008 Apr 25. [Epub ahead of print] |
|
Nichols DH, Randall CL: Rectovaginal fistula. In Nichols DH, Randall CL (eds): Vaginal Surgery, 4th edn. Baltimore, Williams & Wilkins, 1996: 290 |
|
Bandy LC, Addison WA, Parker RT: Surgical management of rectovaginal fistulas in Crohn’s disease. Am J Obstet Gynecol 147:359, 1983 |
|
Wiskind AK, Thompson JD: Fecal incontinence and rectovaginal fistulas. In Rock JA, Thompson JD (eds): TeLinde’s Operative Gynecology, 8th edn. Philadelphia, JB Lippincott, 1997: 618 |
|
Boronow RC: Repair of the radiation-induced fistula utilizing the Martius technique. World J Surg 10:237, 1986 |
|
Elkins TE, DeLancey JO, McGuire EJ: The use of modified Martius graft as an adjunctive technique in vesicovaginal and rectovaginal fistula repair. Obstet Gynecol 75:727, 1990 |
|
Hoexter B, Labow SB, Moseson MS: Transanal rectovaginal fistula repair. Dis Colon Rectum 28:572, 1985 |
|
Goligher JC: Rectovaginal fistula. In Goligher JC (ed): Surgery of the Anus, Rectum, and Colon, 4th edn. London, Spottiswoode Ballantyne, 1980: 191 |
|
Wiskind AK, Thompson JD: Transverse transperineal repair of rectovaginal fistulas in the lower vagina. Am J Obstet Gynecol 167:694, 1992 |
|
Sher ME, Bauer JJ, Gelernt I: Surgical repair of rectovaginal fistulas in patients with Crohn’s disease: Transvaginal approach. Dis Colon Rectum 34:641, 1991 |
|
Pelosi MA 3rd, Pelosi MA: Transvaginal repair of recurrent rectovaginal fistula with laparoscopic-assisted rectovaginal mobilization. J Laparoendosc Adv Surg Tech 7:379, 1997 |
|
Noble GH: A new operation for complete laceration of the perineum designed for the purpose of eliminating danger of infection from the rectum. Trans Am Gynecol Soc 27:357, 1902 |
|
Stowe JM, Goldberg SM: The endorectal advancement flap procedure: How I do it. Int J Colorect Dis 5:232, 1990 |
|
Simmang CL, Lacey SW, Huber PJ: Rectal sleeve advancement: Repair of rectovaginal fistula associated with anorectal stricture in Crohn’s disease. Dis Colon Rectum 41:787, 1998 |
|
Berman IR: Sleeve advancement anorectoplasty for complicated anorectal/vaginal fistula. Dis Colon Rectum 34:1032, 1991 |
|
Dubsky PC, Stift A, Friedl J et al: Endorectal advancement flaps in the treatment of high anal fistula of cryptoglandular origin: full-thickness vs. mucosal-rectum flaps. Dis Colon Rectum 2008; 51(6): 852-7 |
|
Lawson J: Rectovaginal fistulae following difficult labour. Proc R Soc Med 65:283, 1972 |
|
Bricker EM, Kraybill WG, Lopez MJ: Functional results after postirradiation rectal reconstruction. World J Surg 10:249, 1986 |
|
Hibbard LT: Surgical management of rectovaginal fistulas and complete perineal tears. Am J Obstet Gynecol 130:139, 1978 |
|
O'Connor JJ: The role of cryosurgery in management of anorectal disease: A study of cryosurgical techniques. Dis Colon Rectum. 1975; 18(4): 298-300 |
|
Johnson EK, Gaw JU, Armstrong DN: Efficacy of anal fistula plug vs. fibrin glue in closure of anorectal fistulas. Dis Colon Rectum 2006; 49(3): 371-6 |
|
Champagne BJ, O'Connor LM, Ferguson M, et al: Efficacy of anal fistula plug in closure of cryptoglandular fistulas: long-term Dis Colon Rectum 2006; 49(12): 1817-21 |
|
O'Connor L, Champagne BJ, Ferguson MA, et al: Efficacy of anal fistula plug in closure of Crohn's anorectal fistulas. Dis Colon Rectum 2006; 49(10): 1569-73 |
|
Ellis CN: Bioprosthetic plugs for complex anal fistulas: an early experience. J Surg Educ 2007; 64(1): 36-40 |
|
van Koperen PJ, D'Hoore A, Wolthuis AM, et al: Anal fistula plug for closure of difficult anorectal fistula: a prospectivestudy. Dis Colon Rectum 2007; 50(12): 2168-72 |
|
Ky AJ, Sylla P, Steinhagen R, et al: Collagen fistula plug for the treatment of anal fistulas. Dis Colon Rectum 2008; 51(6): 838-43. Epub 2008 Mar 11 |
|
Schwandner O, Stadler F, Dietl O, et al: Initial experience on efficacy in closure of cryptoglandular and Crohn's transsphincteric fistulas by the use of the anal fistula plug. Int J Colorectal Dis 2008; 23(3): 319-24. Epub 2007 Nov 22. |